Abstract
Metal(loid)s have a dual biological role as micronutrients and stress agents. A few geochemical and natural processes can cause their release in the environment, although most metal-contaminated sites derive from anthropogenic activities. Actinobacteria include high GC bacteria that inhabit a wide range of terrestrial and aquatic ecological niches, where they play essential roles in recycling or transforming organic and inorganic substances. The metal(loid) tolerance and/or resistance of several members of this phylum rely on mechanisms such as biosorption and extracellular sequestration by siderophores and extracellular polymeric substances (EPS), bioaccumulation, biotransformation, and metal efflux processes, which overall contribute to maintaining metal homeostasis. Considering the bioprocessing potential of metal(loid)s by Actinobacteria, the development of bioremediation strategies to reclaim metal-contaminated environments has gained scientific and economic interests. Moreover, the ability of Actinobacteria to produce nanoscale materials with intriguing physical-chemical and biological properties emphasizes the technological value of these biotic approaches. Given these premises, this review summarizes the strategies used by Actinobacteria to cope with metal(loid) toxicity and their undoubted role in bioremediation and bionanotechnology fields.
Keywords: metal resistance mechanisms, actinobacteria, metal stress response, metal-based nanostructures, biogenic nanoscale materials
1. Introduction
Metals [e.g., silver (Ag), aluminum (Al), cadmium (Cd), cobalt (Co), chromium (Cr), cesium (Cs), copper (Cu), mercury (Hg), iron (Fe), magnesium (Mg), manganese (Mn), molybdenum (Mo), nickel (Ni), lead (Pb), strontium (Sr), zinc (Zn), uranium (U)] and metalloids [i.e., arsenic (As), selenium (Se), tellurium (Te)], although naturally occurring throughout the Earth’s crust, are not generally present in soluble forms, but suspended as colloids, or in association with organic matters, minerals, and rocks [1]. The release of metals and metalloids (henceforth indicated as metal(loid)s) into the environment derives from geochemical alterations, the type and age of the rock material, as well as natural events, such as volcanic eruptions, atmospheric deposition, leaching of the Earth, earthquakes, typhoons, and climate changes. Some metal(loid)s (e.g., Cu, Co, Fe, Mn, Se, Zn) are essential trace micronutrients needed to support several biological functions in all the domains of life, acting as cofactors featuring important structural and catalytic properties in enzymes and proteins, electron carriers, and regulators of the cellular osmotic pressure [2]. As opposed to this scenario, the presence of “non-physiological” levels of metal(loid)s into the environment has nowadays gained growing concern worldwide, and the reasons behind this phenomenon must be sought elsewhere. Anthropogenic activities (e.g., alloy production, use of pesticides, leather tanning, battery production, and electroplating industries) inexorably determined the buildup of wastes containing metal(loid)s during the 20th century. Moreover, metal(loid) anthropogenic source is more bioavailable and (bio)reactive as compared to those normally found in nature, being this aspect strictly correlated to the enhanced metal(loid) mobility and accumulation at different levels of the trophic chain [3]. Indeed, the reduction or destruction of these substances is a challenging task, making the excess of metal(loid)-containing compounds in the environment a persisting long-term problem [4]. In the case of metal(loid)s, they can cause severe environmental and human health issues even at low concentrations, exerting mutagenic and carcinogenic effects and impairing, among others, the central and peripheral nervous system or the circulatory one [5]. The toxic effects of metal(loid)s depend on the chemical element itself and the load at which it is encountered, as well as a variety of physical-chemical (e.g., temperature, pH, redox-potential, presence/absence of chelating compounds) and biological parameters (e.g., the metal(loid)-lipid solubility, physiological state of the organism, presence/absence of metal interacting proteins, organism adaptation phenomena), including also the way all these factors interplay with each other [6]. Furthermore, the overexposure to metal(loid)s can both compromise soil and water micro-flora fitness, severely worsening the chances of recovering environments contaminated by new xenobiotics [7,8], and contribute to the emergence, maintenance, and transmission of antimicrobial-resistant pathogens [9,10,11]. Among possible solutions for metal(loid) removal or recovery, chemical precipitation, ultrafiltration, ion-exchange, reverse osmosis, and electro-dialysis are procedures that generate toxic wastes and are economically disadvantageous, mostly considering the large-scale application necessary to face the diffuse contamination of metal(loid)s [12,13]. Conversely, bioremediation strategies represent promising eco-friendly, economically feasible, efficient, and pollutant-selective approaches, involving the use of metal(loid) tolerant and/or resistant microorganisms [14], which have evolved specific metabolic processes and adaptation mechanism(s) associated with their survival under different stress conditions [15]. In particular, the survival and development of bacteria in extreme and harsh environments (e.g., in the presence of toxic metals) is allowed by the evolution of inducible mechanisms associated with specific genetic traits (i.e., referred to as resistance) and/or adaptation ones related to intrinsic biochemical features (i.e., referred to as tolerance) [15]. These metabolic activities can be exploited to lower metal(loid) concentration to acceptable and safe levels in order to restore contaminated ecological niches and metal transformation for their recovery (e.g., metal-based nanostructure bioproduction) [16].
Among bacteria that are frequently recovered from toxic metal(loid) and, in turn, utilized for bioremediation strategies, members of Actinobacteria phylum have acquired extensive research attention. Actinobacteria are distributed among 221 phenotypically and morphologically different genera that are highly ubiquitous on Earth and inhabit diverse and extreme environments (e.g., water, soil, plant surface, arctic, and deserted areas) [17]. This bacterial group includes microorganisms exhibiting highly variable physiological and metabolic properties as a reflection of their wide genomic heterogeneity [17]. Among Actinobacteria, members of Streptomyces genus and the CMNR group (Corynebacterium, Mycobacterium, Nocardia, and Rhodococcus) possess large genomes and multiple linear and circular megaplasmids, which contribute to their extraordinary metabolic capabilities and toxic metal resistance [18,19,20]. One interesting aspect of combining bioremediation with metal recovery strategies involves the utilization of microbes to produce nanostructures (NSs) containing metal(loid)s, which are typically more toxic in bulk. Metal(loid) nanomaterials (MeNMs) play a central role in nanotechnology, the field of science dealing with materials that possess at least one dimension in the nanorange (10−9 m), where they hold physical-chemical characteristics (high surface-to-volume ratio, large surface energy, and spatial confinement) from which a plethora of intriguing properties—e.g., size-dependent, optical, catalytic, electronic, mechanical—arise as compared to the corresponding bulk materials. This aspect has enabled the extensive use of metal(loid) nanomaterials (MeNMs) at a multidisciplinary level in various application fields [21,22].
Given these premises, this review will overview the mechanisms of interaction between some model strains of Actinobacteria phylum and metal(loid)s in terms of (i) resistance strategies adopted to counteract metal(loid) toxicity, (ii) transformation of metal(loid) ion/oxyanions into less toxic and bioavailable forms, (iii) synthesis of biogenic MeNMs with different physical-chemical features (e.g., size, morphology, and properties) and relevant biotechnological applications.
2. Mechanism(s) of Metal Tolerance and Resistance in Actinobacteria
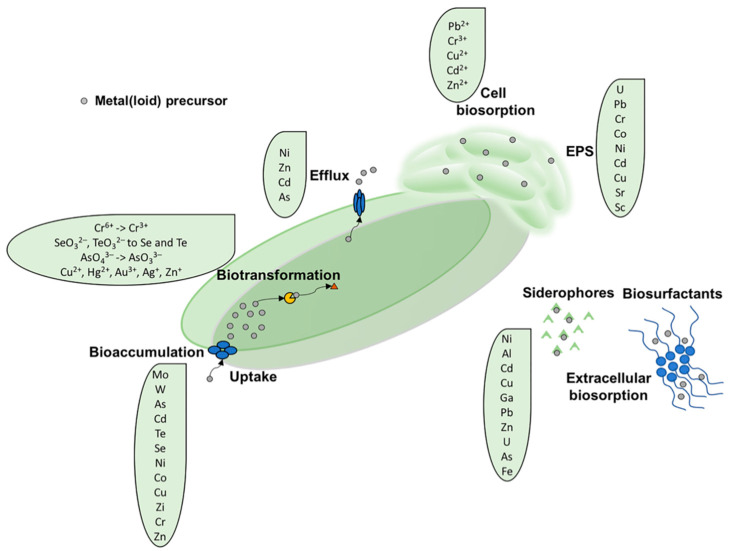
Various members of Actinobacteria phylum hold a positive and long-standing reputation for environmental biotechnology applications, as they (i) can utilize inexpensive and/or toxic compounds as carbon and energy sources, (ii) feature an environmental resilience, persistence, and robustness– which derives from a versatile catabolic and anabolic potential, (iii) usually are not pathogens, and (iv) generally display a very good basal resistance to metals. All these aspects make Actinobacteria excellent candidates for the bioremediation of metal(loid)-polluted environments [23,24,25]. The main actinobacterial tolerance/resistance strategies to metal(loid) toxicity is associated with biosorption, bioaccumulation, and biotransformation processes [26], which are visually summarized in Figure 1 and described in the paragraphs below.
Figure 1.
Schematic representation of biotic mechanisms (i.e., biosorption and extracellular sequestration by siderophores, biosurfactants, and extracellular polymeric substances (EPS), bioaccumulation, biotransformation, and metal efflux processes) of interaction between Actinobacteria and metal(loid)s.
2.1. Biosorption
Biosorption represents a microbial tolerance mechanism that allows the lowering of the local metal(loid) concentration. This process occurs through electrostatic interactions between metal(loid) ions and functional groups present on microbial surfaces such as proteins, carbohydrates, cell wall polymers, extracellular polymeric substance (EPS), and metal chelating factors (e.g., siderophores and metalloproteins) [27]. Biosorption is recognized, from an industrial perspective, as a highly efficient, economically, and ecologically sustainable process since it avoids the formation of hazardous wastes in removing metal(loid)s, radionuclides, and toxic organic xenobiotics from polluted environments. From a physical-chemical perspective, microorganisms are generally considered good biosorbents due to their small size and high surface-to-volume ratio, thus offering a large surface area for their interaction with metal(loid)s to successfully occur [28,29,30,31].
Depending on the metal(loid) speciation found in a specific ecological niche [3] and its physical-chemical factors, diverse members of Actinobacteria phylum have been described to carry out metal(loid) sorption strategies [32]. The affinity, yield, and specificity of this bioprocess rely on the cell wall and membrane surface structural chemistry—i.e., charge and orientation of the metal(loid)-binding groups [33]. Indeed, the actinobacterial cell surface has a complex macromolecular composition (lipids, peptidoglycan, teichoic acid, lipo- and surface-proteins, enzymes and mycolic acids) that overall expose diverse reactive functional groups (e.g., hydroxyl [OH−], carbonyl [CO], carboxyl [COO−], phosphate [PO43−], sulfate [SO42−], and amino [NH2+/NH3+]), which can mediate the sorption and deposition of metal(loid)s on the bacterial surface [34,35,36]. For instance, S. zinciresistens exhibited a bald—no spore-forming—phenotype, which showed a short and thin mycelium upon cell exposure to Zn2+ and Cd2+, likely due to the interaction occurring between OH−, CO, and NH surface moieties and these cations, resulting in dense granules visible on the cell surface by scanning electron microscopy (SEM) [37]. The availability of proton binding sites, as a function of pH and salt level, will determine a diverse net charge of the actinobacterial cell surface, widening the possibility of eliciting different sorption phenomena. This impacts the general pKa, isoelectric point, and the number of active moieties of the biosorbent, as highlighted in work carried out by Plette and colleagues (1995) using Rhodococcus erythropolis A177 [34]. At pH values lower than the isoelectric point of the biosorbent, the biosorbent will feature a positive net charge due to its association to hydronium (H3O+) ions, therefore repulsing metal(loid) cations. The opposite effect (i.e., negative net charge) is expected at pH values higher than the biosorbent isoelectric point, leading to the cation adsorption. In this context, the biosorption capacity of Al3+ by R. opacus strain was enhanced at pH values around 5, as the general isoelectric point of this biosorbent was 3.26, as well as in the case of other metal(loid)s (e.g., Pb2+, Cr3+, Cu2+, Cd2+, and Zn2+) [38,39,40]. Moreover, proton exchange with metal(loid)s will be favored in the case of pH values above the pKa of the binding moieties characterizing the biosorbent [41]. Fourier-transform infrared (FTIR) spectroscopy analyses performed on Streptomyces VITSVK5, VITSVK9, and S. werraensis LD22 strains exposed to Pb2+, Cd2+, and Cr3+ revealed that ion-exchange takes place between metals and hydrogens of OH−, COO−, and NH2+ groups [42,43,44]. The importance of these charged moieties for actinobacterial biosorption was further proved by their chemical modifications, i.e., through methylation of NH2+/NH3+ groups or esterification of both OH− and COO− ones that dramatically impaired the sorption of Cu2+ as compared to untreated biomass [45].
If the pH, the type of metal(loid), and the nature of the biosorbent material represent important factors that must be accounted for the optimization of biosorption processes, in natural settings, the contemporary presence of a variety of metal(loid)s may lead to their competition for the ligand sites present on the surface of actinobacterial cells, compromising sorption bioprocesses. This is the case of Ca2+ that is the main competitor for metal sorption in contaminated soil matrices [46,47,48], yet R. erythropolis A177 showed good sorption capacity towards Cd2+, even in the presence of Ca2+ ions. Upon exposure of Rhodococcus cells to Ca2+, the cell wall drastically changed because of breakings of the cross-links between different chemical surface moieties and alteration in the structure of the peptidoglycan layer, increasing the number of active binding sites for Cd2+ [49]. On the other hand, it has been reported that the cell amount in a specific contaminated site influences the single-cell sorption capacity, likely due to cross-links between functional groups [39,41,50].
2.1.1. Extracellular Sequestration by Siderophores
The scarce aqueous solubility and bioavailability of Fe and its importance for several biological functions led microorganisms to evolve specific mechanisms of Fe-uptake and Fe-trapping, mostly represented by siderophore biosynthesis [51,52]. Siderophores are low-molecular-weight (200–2000 Da) and diffusible molecules featuring a high affinity and selectivity for the insoluble ferric (Fe3+) as compared to ferrous (Fe2+) cations, which guarantees a low level of competition with other bivalent yet essential metal cations(e.g., Zn2+, Cu2+, Ni2+, and Mn2+) [53]. Thus, siderophores’ biosynthesis and secretion usually depend on Fe3+ environmental concentration [52,53,54], as they act as Fe-chelators, creating extracellular iron-siderophore complexes from which Fe3+ is taken up by bacterial cells through redox processes [53,55]. After the first study reporting the production of the siderophores arthrobactin and mycobactin by Arthrobacter terregens and Mycobacterium johnei in the 1950s [53], actinobacterial members have been described to be able to synthesize siderophores, which, depending on their chemical structure, are indicated as phenolates, catecholates, carboxylates, hydroxamates, or mixed types [56,57,58] (Table 1).
Table 1.
Siderophores produced by Actinobacteria as metal(loid) chelating agents.
| Actinobacteria Strains | Siderophores | Metal Tolerance | References |
|---|---|---|---|
| Microbacterium flavescens JG-9 | Desferrioxamine-B | U and Pu | [59] |
| Arthrobacter spp., Microbacterium spp., Curtobacterium spp., Leifsonia spp. | N.D. 1 | Co, Cr, Cu, Ni, Zn | [60] |
| Streptomyces sp. AR16 and AR36 | N.D. 1 | Zn, Cd, Pb | [61] |
| Microbacterium G16 | N.D. 1 | Cu, Cd, Ni, Pb, Zn | [62] |
| Streptomyces acidiscabies E13 | Coelichelin and desferrioxamines | Ni | [63] |
| Streptomyces tendae F4 | Coelichelin and desferrioxamines | Ni, Cd | [64,65] |
| Methylobacterium and Frigobacterium spp. | N.D. 1 | Zn, Cd | [66] |
| Arthrobacter spp. and M. kitamiense YM31 | N.D. 1 | Cu | [67] |
| Microbacterium sp. NCr-8, Arthrobacter sp. NCr-1, Kokuria sp. NCr-3 | N.D. 1 | Ni | [68] |
| Streptomyces mirabilis P16B-1 | Linear and cyclic ferrioxamines | Zn, Al, Ni, Cu | [69] |
| Streptomyces spp. | Hydroxamates | Pb | [70] |
| Streptomyces spp. | Ferrioxamines, phenolates, and catecholates | Cd | [71] |
| Arthrobacter oxydans ATW2 and ATW3, Kocuria rosea ATW4, Rhodococcus erythropolis ATW1 and S43 | N.D. 1 | As | [72] |
| Rothia mucilaginosa ATCC 25,296 | Enterobactin | Zn, Mg | [73] |
1 N.D. indicates not determined, which is referred to the nature of siderophores produced by Actinobacteria.
Among these, hydroxamates (e.g., desferrioxamines G1, B, and E, rhodotorulic acid, coelichelin) appear to be the most common [54,74,75]. Streptomyces spp. were also found to produce catecholate siderophores, such as enterobactin and myxochelin, although they are typical of Gram-negative bacterial strains [54]. The production of structurally diverse siderophores seems to follow either the desferrioxamine or the multiple nonribosomal peptide synthetase (NRPS) biosynthetic pathways, which are upregulated under Fe-deficiency [54]. Although it is not obvious to understand the advantage of Actinobacteria and, specifically, of Streptomyces spp., to biosynthesize diverse siderophores, this phenomenon may depend on the need of these bacterial strains to face competitive interactions with other microorganisms [54]. Recent studies also demonstrated the ability of actinobacterial siderophores to bind several toxic metal(loid)s other than Fe3+, contributing to their bioavailability reduction and, therefore, enhancing the bacterial survival in contaminated environments [52,65,76]. In this regard, hydroxamates produced by Streptomyces spp. can chelate Ni, Al, Cd, Cu, gallium (Ga), Pb, Zn, U, and As, although the binding strength of these metal(loid)s is lower as compared to Fe [63,65,69,71,76]. Once metal(loid)s form a complex with siderophores, they can either be assimilated or sequestered in the extracellular environment, the latter alleviating the cell stress deriving from toxic metal(loid)s [63,69,72]. Particularly, coelichelin, desferrioxamines, and hydroxamates produced by S. acidiscabies E13 and S. tendae F4 showed good binding selectivity and, therefore, toxicity protection towards Ni2+ Leifsonia spp. and Cd2+ [63,64,65]. In this regard, the low amount of available Fe3+ in the culture media, and its competition with other metals, likely stimulated an active biosynthesis of siderophores by these bacteria to obtain enough Fe-content for their survival [64,65]. Catecholates and phenolates were also found responsible for counteracting Cd2+ toxicity in a wide number of Streptomyces spp., suggesting the presence of regulatory systems for siderophores’ synthesis in these microorganisms [71]. Siderophores were described to be involved in the post-efflux chelation of Cd and Zn also in Methylobacterium and Frigobacterium spp. [66,77], and in the binding and chelating of As3+ cations in A. oxydans ATW2 and ATW3, Kokuria rosea ATW4, R. erythropolis ATW1 and S43 under Fe-deficiency [72]. Particularly, Fe- or As-siderophore complexes extracted from A. oxydans ATW3 and R. erythropolis ATW1 showed distinct physical-chemical characteristics, indicating the occurrence, in Actinobacteria, of multiple co-existing chelators with different metal(loid) selectivity [16,72].
2.1.2. Extracellular Sequestration Mediated by Extracellular Polymeric Substance (EPS)
A common feature of bacteria is the production and release of biological exudates, such as polysaccharides and their derivatives, lipids, secondary metabolites, proteins, and nucleic acids, which are generally referred to as EPS [78,79]. These extracellular biomolecules constitute a complex hydrogel matrix that confers fluid and elastic properties to microorganisms, allowing them to adapt to environmental changes, particularly when growing as biofilm [80]. EPS forms a protective layer surrounding bacterial cells, whose physical-chemical structure guarantees to microorganisms an efficient adhesion to surfaces, the accumulation and dissolution of nutrients, and the protection towards desiccation, predation, as well as toxic and exogenous substances, such as metal(loid)s, antimicrobials, and reactive oxygen species (ROS) present in the environment [52,78]. EPS features a great ability to bind and immobilize metal(loid) ions from the extracellular environment, avoiding their entry within bacterial cells and, hence, playing a fundamental role as biosorbents for these substances [81,82,83]. The biosorption capability of EPS is selective, and it depends on (i) its structure and functional groups, (ii) environmental parameters (i.e., pH, temperature, and carbon source availability), and (iii) the initial concentration, size, and bond energy of the metal(loid) ions in the extracellular environment [81,83,84,85]. Overall, the high hydrophilicity, chemical reactivity, and stability of polysaccharides make them good biosorbents, as they display several chemical reactive groups [e.g., COO−, CO, NH3+, OH−, PO43−, and acetamido (AcNH+)] that can dynamically interact and sequester metal(loid) ions [78,86]. EPS metal(loid) biosorption occurs through electrostatic interactions between the charged biomolecules and metal(loid)s, forming stable metal-EPS complexes [27,81,83,87,88]. The biosorption potential of EPS is correlated with the number of functional groups present within this matrix, enhancing the metal(loid) binding to bacterial cell membranes [89,90]. Once EPS immobilizes metal(loid) ions, they can be sequestered and transformed by enzymes and proteins through ion exchange, complexation, and precipitation [27,52].
Several members of the Actinobacteria phylum were demonstrated to produce EPS in response to metal stress, representing the first defense mechanism against the toxicity of these inorganic compounds (Table 2). Within this phylum, EPS biosynthesis is influenced by the bacterial species, the growth phase, the availability of nutrients, and the selective pressure exerted by toxic compounds [84,88].
Table 2.
Extracellular polymeric substances (EPS) produced by Actinobacteria as biosorbents for metal(loid)s.
| Actinobacteria Strains | EPS Features | Metal Tolerance | References |
|---|---|---|---|
| Arthrobacter D9 | N.D. 1 | Cd | [91] |
| Amycolatopsis sp. AB0 | Polysaccharides containing glucose | Cu | [92] |
| Kocuria rizophila DE2008 | Polysaccharides and uronic acid | Cu, Pb | [93,94] |
| Arthrobacter viscosus | N.D. 1 | Cr | [95] |
| Nocardia amarae | N.D. 1 | Na, Ca, Al, Fe | [96] |
| Arthrobacter ps-5 | Polysaccharides containing glucose and galactose | Pb, Cu, Cr | [81] |
| Rhododoccus opacus and Rhodococcus rhodochrous | N.D. 1 | Ni, Cd, Pb, Co, Cr | [86] |
| Kocuria rizophila BPB1 | N.D. 1 | As | [97] |
| Frankia sp. strain EAN1pec | Polysaccharides and proteins | Pb | [98] |
| Kokuria sp. BRI 36 | Polysaccharides and uronic acid | Pb, Cd, Cr | [99] |
| Nocardiopsis sp. 13H | Polysaccharides, proteins, and nucleic acids | Sr and Cs | [87,100] |
| Streptomyces sp. CuOff24 | Arabinose, galactose, mannose, glucose, and uronic acid | Sr | [88] |
| Glutamicibacter halophytocola KLBMP 5180 | Rhamnose, glucuronic acid, glucose, galactose, xylose, and arabinose | Mn, Cu, Fe, Ca, Mg | [83] |
1 N.D. indicates not determined, which is referred to the nature of EPS produced by Actinobacteria.
Amycolatopsis sp. AB0 produced EPS mainly constituted by polysaccharides containing ca. 20 repeating glucose units that could bind Cu2+ [92]. Upon addition of Cu2+ and Pb2+, cells of K. rizophila (former Micrococcus luteus) DE2008 enhanced the secretion of EPS that was characterized by a larger amount of carbohydrate and uronic acid moieties as compared to the EPS produced in the absence of the metal challenge, which determined an improvement in metal immobilization efficiency [94]. The role of EPS in metal biosorption by the DE2008 strain was suggested by the formation of Cu and Pb deposits visible in the extracellular matrix [93,94]. Stable precipitates of Pb2+ were also detected on the cell surface of Frankia sp. strain EAN1pec, in which a specific Pb-binding mechanism was demonstrated to occur. The involvement of EPS, as well as cell wall lipids, in the adsorption of Pb2+, was suggested by proteomic analyses, which highlighted peculiar surface and lipid–protein expression patterns in EAN1pec cells exposed to Pb2+ [98].
The EPS biosorption behavior towards different metal cations and the influence of metal concentration, pH, and ionic strength on biosorption efficiency was assessed using cultures of Arthrobacter ps-5 [81], R. opacus 89 UMCS, and R. rhodochrous 202 DSM [86]. These actinobacterial strains showed a biosorption preference towards Pb cations followed by Cu and Cr in the case of Arthrobacter ps-5 [81] and Co, Cr, Cd, and Ni for Rhodococcus spp. strains [86]. Particularly, the adsorption equilibrium was reached within 30 min of incubation for all the metals tested for both 89 UMCS and 202 DSM strains, being the highest Pb biosorption rate observed with the latter strain (ca. 200 mg/g Pb2+ in 5 min) [86]. The EPS secreted by Arthrobacter ps-5 contained polysaccharides composed of glucose and galactose units, whose amount strongly influenced its metal biosorption ability, as a high number of functional groups acting as binding sites for metal cations were available [81]. Specifically, functional groups containing oxygen [i.e., OH−, COO−, CO, and ether (COC)] were involved in the complexation of metal cations in Arthrobacter ps-5 and Rhodococcus spp. [81,86], due to the oxygen tendency to reduce the electron cloud density of these chemical groups [101]. However, reactive NH3+, methyl (CH3), methylene (CH2), primary (NH) and secondary (NH2) amine groups were also found to be partially responsible for EPS biosorption of Ni2+, Pb2+, Co2+, Cd2+, and Cr4+ in the case of rhodococci [86]. Moreover, the pH must be accounted for the biosorption process, as H3O+ ions present in an acid (pH = 2–3) solution could compete with Pb, Cu, Co, and Cr cations for the same EPS functional groups [102,103], while at pH > 6 these metals can precipitate in the medium forming metal hydroxides [104], impairing their immobilization within the bacterial EPS. Thus, a pH value of 5 was the optimal condition for Pb, Cu, Co, and Cr biosorption in Arthrobacter ps-5 cultures [81] and Rhodoccus spp. [86], while the latter better immobilized Cd2+ cations when the pH was fixed at 6.5, as, at values greater than 8, Cd(OH) complexes dominate [86]. A similar conclusion can be drawn for the greatest Ni2+ EPS-mediated adsorption in the case of rhodococci, which occurred at pH between 3 and 4, as higher values led to the formation of Ni(OH)2 and Ni(OH)3 species, whose interaction with EPS’ functional groups are less favored [86]. A competition for the EPS binding sites can also arise between metal cations and metal salts (e.g., KCl and CaCl2) generally present in the growth medium, as the positive charges of the latter can electrostatically interact with the actinobacterial extracellular matrix [81]. In this regard, a decrease in the EPS biosorption ability was detected when metal salt concentration was increased in cultures of Arthrobacter ps-5, being Ca2+ more effective in competing with Pb, Cd, and Cr [81].
In some Actinobacteria members, EPS-mediated biosorption seemed to be also involved in the detoxification of metal radionuclides, such as strontium (Sr) and cesium (Cs) [87,88,100]. Indeed, Nocardiopsis sp. 13H could adsorb both Sr+ and Cs+ species through the production of EPS, which featured layers that were loosely (outer and prone to dissolution) and tightly (inner) bound to bacterial cells [87,100]. Compositional analysis of EPS revealed that the carbohydrate content largely increased in both layers, consequently to Sr or Cs exposure, followed by proteins, nucleic acids, and unidentified compounds. Moreover, Sr+ and Cs+ were only found in the inner layer of EPS, which featured a large number of OH−, PO43−, NH2 and thiol (SH) groups that probably have a significant role in the biosorption of these metal radionuclides [87,100]. Similarly, EPS produced by Streptomyces sp. CuOff24 showed a good proficiency as a biosorbent for Sr+, being arabinose, galactose, mannose, glucose, and uronic acid the major components identified along with NH, NH2, AcNH+, and SH reactive moieties [88].
2.2. Bioaccumulation
A further strategy that microorganisms can exploit to detoxify the surrounding environment from metal(loid)s is based on bioaccumulation phenomena, which imply the active transport of metals within the cells through specific uptake systems [3,105,106]. Bioaccumulation occurs through slow metabolic-dependent and energy-demanding processes, which generally follow the rapid metal(loid) biosorption by bacteria, resulting in a net decrease of the metal(loid) concentration in the extracellular environment. Thus, bioaccumulation can be performed only by living cells that feature specific genetic and biochemical assets involved in metal transport and can undergo physiological adaptations in response to the presence of metal(loid)s [107]. Moreover, it has been reported that the temperature can affect metal(loid) bioaccumulation as if, on one hand, a temperature increase is expected to enhance the rate of chemical reactions; on the other hand, it can influence the fluidity of biological membranes, compromising the respiratory chain kinetics and permeability of microbial cell, which are crucial for the bacterial survival [108]. Once inside the cells, some metal(loid)s (e.g., As, Cd, Te, Se) can be used as terminal electron acceptors and/or cofactors in metalloproteins and enzymes [109,110,111]. They can also be transformed through either redox processes or alkylation reactions [3], where such changes in speciation can alter metal(loid) mobility and toxicity. The internalization of metal(loid)s in the bacterial cytoplasm can be mediated by (i) ion channels or carriers exploiting a concentration gradient and (ii) active transporters dependent on either ATP consumption or the electrochemical gradient as an energy source. This aspect is particularly important for those bacteria holding enzymatic activities (e.g., nitrogenases and oxidoreductases) that use molybdoproteins as cofactors, making it imperative for these microorganisms to acquire Mo (as the oxyanion MoO42−), which generally occurs through either the high-affinity ATP-binging cassette (ABC) transporter or low-affinity carrier systems [112]. Non-essential and toxic metal(loid)s can be internalized through promiscuous transport systems typically involved in the uptake of the essential ones, as in the case of the molybdate ABC transporter that does not discriminate tungsten (W) in its oxyanion from (i.e., WO42−), which can therefore be bioaccumulated. In regard to the oxyanions, those containing metalloid elements, such as arsenate (AsO43−), arsenite (AsO33−), tellurate (TeO42−), tellurite (TeO32−), selenate (SeO42−), and selenite (SeO32−), are extremely toxic for many living organisms; for instance, TeO32− exert its toxicity at a concentration as low as 1 µg mL−1 towards both prokaryotic and eukaryotic organisms [113]. Moreover, although Se represents an essential micronutrient for living systems [114], the overexposure to Se-compounds can cause severe health issues, while any biological function has not been reported for those containing As and Te [115]. Due to anthropogenic activities, these oxyanions can now be found as pollutants at critical environmental concentrations, whose toxicity relies on their high reactivity, mobility—through soil and water streams—and bioavailability thanks to their association with oxygen [116]. Despite the oxyanions’ toxicity, bacteria can take up either SeO42− or SeO32− using transporters and/or permeases involved in sulfate and sulfite uptake [117,118,119] and using polyol ABC transporters [120]. Further, the phosphate transporter has been proposed to be involved in the internalization of TeO32− and AsO43−, since these oxyanions are strong competitors of PO43− groups [121,122,123,124,125], as well as the acetate permease (ActP) monocarboxylate transporter [126].
Other than oxyanions, bacterial strains have evolved specific systems of bioaccumulation to assure a proper supply of metal cations and detoxification bioprocesses. In this context, Abbas and Edwards (1990), alongside Amoroso and colleagues (1998), isolated diverse Streptomycetes that showed significant resistance levels against Hg2+, Co2+, Cd2+, Cu2+, Zn2+, Cr6+, Ni2+, and Mn2+ [127,128,129]. Some of these cations were taken up by bacterial cells exploiting the transport system of Mg2+, which represents a strong competitor for the uptake of transition metals [129]. Transporters that can be used for Ni2+ internalization are high-affinity permeases that are characterized by low transport capacity [130,131,132]. In the case of M. tuberculosis and Streptomyces spp., it has been reported that they can utilize these permeases to support the activity of Ni-dependent ureases [133] and superoxide dismutases (Sod), respectively. Particularly, Ni plays a crucial role in S. coelicolor, regulating sod gene expression, post-translational modifications, and Sod enzymatic activity [134]. Moreover, the Ni-responsive transcriptional regulator Nur, a homolog of the iron one (Fur), was identified as responsible for Ni uptake in S. coelicolor, binding the promoter region of nikA of the putative Ni transporter gene cluster (nikABCDE) [135]. On the same line, Lu and colleagues (2014) deciphered, in S. coelicolor A3(2), the molecular function of a hypothetical protein that binds four Ni2+ at its surface. The gene coding for this protein is only upregulated by the presence of Ni2+ as compared to other cations, such as Cu2+, Co2+, and Mn2+ [136]. This evidence underlines how the intracellular nickel homeostasis and antioxidative response are finely regulated in S. coelicolor. On the other hand, R. rhodochrous strain J1 used the Nh1F transporter to take up physiological amounts of Co2+ to sustain the nitrile hydratase activity, which exploits non-corrin Co as a cofactor [137]. In line with this, Pogorelova and colleagues (1996) described the importance of Co2+ for the nitrile utilization by R. rhodochrous strain M8 since Co deficiency seemed to determine the inhibition of the nitrile hydratase production either at the transcriptional or translational level [138]. Due to the high amino acid sequence similarity with Ni2+ HoxN transporter of Alcaligenes eutrophus [139], the Nh1F transporter was also proposed to promote the Ni2+ uptake in some actinobacterial members because of the metal-binding promiscuity [137], despite the preferential binding of Co2+. This aspect was further proven by the enhanced Ni-dependent urease activity showed by a recombinant strain of E. coli heterologously expressing the Rhodococcus nhlF gene and the successful cross-complementation of a hoxN deletion with the nhlF gene in A. eutrophus strain [140]. Moreover, Amoroso and coworkers (2000) highlighted that some of the 25 streptomycetes isolated from a uranium mining site displayed a great tolerance against Ni2+ ions (10 mM), among others (e.g., Co2+, Cd2+, Cu2+, Cr3+, and Zi2+) [141], likely due to the presence of a high-affinity Ni-transport system, which was identified in Streptomyces F4 and E8 strains using degenerated primer pairs designed on the basis of the Ni-transporter genes hoxN and nixA from Ralstonia eutropha [132] and H. pylori [142], respectively.
Zinc is another important transition metal that can act as a stabilizing element during protein folding and catalytic cofactor of several enzymes [143]. However, Zn poisoning can inhibit the electron transport chain and either block thiols or compete with other essential metal(loid)s for binding sites present in proteins, as reported in the case of cytochrome c oxidase [144,145]. Thus, bacteria must finely regulate and maintain Zn homeostasis [146]. To this aim, various microorganisms feature specific zinc regulators (Zur), which belong to the Fur family and are responsible for the regulation of genes devoted to Zn transport and homeostasis maintenance. In S. coelicolor, the Zn regulator Zur is located downstream of the znuACB operon transcribing for a putative Zn transporter. In particular, Zn could repress the transcript level of the znuA gene, while the deletion mutant in the zur gene did not show any repression. Moreover, the purified Zur protein could bind the znuA promoter as a homodimer in the presence of Zn, further highlighting the role of this repressor responsive to Zn [147]. Later, Choi and colleagues (2017) demonstrated that not only Zur negatively regulates genes encoding for proteins involved in Zn uptake, but it induces the expression of the zitB gene, which encodes for a Zn exporter in S. coelicolor [148]. At low Zn concentration (e.g., femtomolar), the regulator Zur binds, as a dimer, its box motif present in the upstream region of the zitB promoter, which results in a weak zitB expression. On the other hand, at high Zn concentrations (e.g., micromolar), oligomeric Zur binds its target region with a footprint that goes beyond the zinc box motif, resulting in an enhanced zitB induction [148], thus highlighting how a single metal regulator can maintain the intracellular free level of Zn within a narrow range by controlling the expression of genes coding for both uptake and export systems. Despite the fine regulation system for Zn homeostasis in bacteria, it is worth noting the good adaptability of some Streptomycetes strains to the high metal load, as reported in the case of Streptomyces K11, which could tolerate and bioaccumulate very high concentrations of Zn2+ ions [149].
Consequently, to metal(loid) bioaccumulation, microbial cells can show non-orthodox features such as electron-dense inclusions due to the localization of the sequestered metal(loid) within cellular components or their binding to metallothioneins [150,151]. For instance, electron-dense inclusions were visible through SEM microscopy in Streptomyces sp. MC1 cells as a possible result of the resistance mechanism of this strain to Cr6+ [152]. An additional effect of Cr was the generation of more rounded and shorter filamentous cells with respect to unexposed ones [153]. Further, some Actinobacteria members featured the ability to uptake radionuclides as Cs, whose presence on the Earth’s crust gained a global concern since the Chernobyl accident in 1986. In this regard, several rhodococci can cope with the toxicity derived from both Cs and rubidium (Rb), as reported for R. erythropolis CS98 and Rhodococcus sp. strain CS402 accumulating high levels of Cs, whose process was inhibited by both potassium (K) and Rb [154], probably because of the involvement of the poorly specific K-transport system [155,156].
2.3. Biotic Metal(loid) Reduction Reaction
The tolerance of bacteria to metal(loid) polluted environments is enhanced by reducing these chemical species into less toxic and bioavailable forms, guaranteeing the microbial survival under harsh conditions. Numerous studies reported on Actinobacteria’s capability of coping with metal(loid) cation and oxyanion toxicity through either intra- or extracellular bioconversion strategies (Table 3), being Arthrobacter and Streptomyces spp. considered as model microorganisms [14].
Table 3.
Overview of metal(loid) reduction processes in Actinobacteria.
| Actinobacteria Strains | Metal(loid) Reduction | Mediating Biomolecules | References |
|---|---|---|---|
| Micrococcus lactilyticus | Au3+, Ag+, SeO32−, TeO32−, TeO42−, AsO43−, bismuthate (BiO3−), vanadate (VO34−), molybdate (MoO42−), ruthenium (Ru3+) | N.D. 1 | [157] |
| Streptomyces lividans 1326 | Hg2+ | Hg-constitutive reductase | [158] |
| Streptomyces sp. 3 M | CrO42− | Constitutive reductase | [159] |
| Microbacterium sp. MP30 | CrO42− | N.D. 1 | [160] |
| Streptomyces griseus | CrO42− | N.D. 1 | [161] |
| Thermomonospora sp. | Au3+ | Four proteins and enzymes (80–100 kDa) | [162] |
| Rhodococcus sp. | Au3+ | Intracellular proteins | [163] |
| Streptomyces thermocarboxydus NH50 | CrO42− | Extracellular “nonenzymatic” substance | [164] |
| Arthrobacter crystallopoietes ES 32 | CrO42− | NADH-dependent chromate reductase | [165] |
| Corynebacterium strain SH09 | Ag+ | Aldehyde and ketone groups of biomolecules | [166] |
| Actinobacter sp. | K3Fe(CN)6/ K4Fe(CN)6 mixture | Extracellular hydrolases | [167] |
| Corynebacterium glutamicum | AsO43− | NAD(P)H-dependent intracellular ArsC proteins | [168] |
| Streptomyces spp., Amycolatopsis sp. | CrO42− | N.D. 1 | [152] |
| Actinobacter sp. | Au3+ | Cytochrome oxidase | [169] |
| Actinobacter sp. | FeCl3 or FeCl3/FeSO4 mixture | Ferrisiderophore reductase, phosphoadenosyl sulfate, and sulfite reductases | [170] |
| Streptomyces sp. MS-2 | CrO42− | N.D. 1 | [171] |
| Brevibacterium casei AP6 | Co2+ | Intra- and extracellular proteins | [172] |
| Streptomyces sp. MC1 | CrO42− | NAD(P)H-dependent constitutive chromate reductase | [153,173,174] |
| Arthroobacter rhombi-RE | CrO42− | Intracellular enzymes | [175] |
| Streptomyces hygroscopicus | Ag+ | Secondary metabolites | [176] |
| Corynebacterium glutamicum | Ag+ | Proteins | [177] |
| Arthrobacter ramosus | Hg2+, CrO42− | MerA enzyme, intracellular enzymes | [178] |
| Arthrobacter aurescens MM10 | CrO42− | Intracellular chromate reductase | [179] |
| Flexivirga alba ST13 | CrO42− | Intracellular proteins | [180] |
| Streptomyces sp. | Ag+ | Intracellular enzymes | [181] |
| Nocardia farcinica | Au3+ | extracellular nitrate reductase | [182] |
| Arthrobacter sp. SUK 1201 | CrO42− | N.D. 1 | [183,184] |
| Arthrobacter sp. SUK 1205 | CrO42− | N.D. 1 | [185] |
| Streptomyces sp. RSF17, CRF14, Streptomyces matansis BG5, Streptomyces vinaceus CRF2, Streptomyces pulcher CRF17, Streptomyces griseoincarnatus SCF18 | CrO42− | N.D. 1 | [186] |
| Rhodococcus NCIM 2891 | Ag+ | Intracellular NADH-dependent nitrate reductase; peptides, proteins, and carbohydrates | [187,188] |
| Streptomyces sp. JAR1 | Ag+ | Intracellular NADH-dependent nitrate reductase | [189] |
| Micrococcus luteus NCIM 2379 | Au3+ | Extracellular α-amylase and TUA | [190] |
| Streptomyces sp.VITDDK3 | Au3+ | (2S,5R,6R)-2-hydroxy-3,5,6-trimethyloctan-4-one | [191] |
| Streptomyces sioyaensis Lv81-138 | CrO42− | Intracellular reductases | [192] |
| Rhodococcus pyridinivorans NT2 | Zn2+ | NAD(P)H-dependent reductase and secreted reductase | [193] |
| Streptomyces spp. and Amycolatopsis tucumanensis | CrO42− and lindane | N.D. 1 | [194] |
| Kocuria flava M-7 | Cu2+ | Intracellular proteins | [195] |
| Streptomyces minutiscleroticus M10A6 | SeO32− | Intracellular proteins | [196] |
| Acidithrix ferrooxidans PY-F3 | Fe3+ | N.D. 1 | [197] |
| Streptomyces bikiniensis strain Ess_amA-I | SeO32− | Intracellular proteins and enzymes | [198] |
| Intraspongium chromatireducens Q5-1 | CrO42− | Extracellular constitutive enzyme | [199] |
| Streptomyces rochei MHM13 | Ag+ | Intracellular proteins | [200] |
| Corynebacterium paurometabolum SKPD 1204 | CrO42− | N.D. 1 | [201] |
| Gordonia amarae | Au3+ | Glycolipids | [202] |
| Streptomyces sp. ES2-5 | SeO32− | Mycothiols and thiol-containing molecules | [203] |
| Rhodococcus etherivorans BCP1 | TeO32− | Mycothiols and thiol-containing molecules | [204] |
| Streptomyces sp. NH21 | Au3+, Ag+ | Intra- and extracellular proteins | [205] |
| Streptomyces kasugaensis M338-M1T, Streptomyces celluloflavus NRRL B-2493T | Ag+ | Proteins | [206] |
| Streptomyces parvulus DPUA 1549, Streptomyces owasiensis DPUA 1748 | Ag+ | Proteins | [207] |
| Rhodococcus etherivorans BCP1 | SeO32− | Mycothiols and thiol-containing molecules | [208] |
| Rhodococcus etherivorans BCP1 | TeO32− | Mycothiols and thiol-containing molecules | [209] |
| Streptomyces spp. M7, A5, and MC1, Amycolatopsis tucumanensis | CrO42− | N.D. 1 | [210] |
| Streptomyces sp. Al-Dhabi 89 | Ag+ | Extracellular biomolecules | [211] |
| Streptomyces xinghaiensis OF1 | Ag+ | Organic compounds featuring amino bonds | [212] |
| Streptomyces capillispiralis Ca-1 | Cu2+ | Intracellular proteins | [213] |
| Micrococcus luteus HM 2 and HM 16 | CrO42− | Extracellular proteins | [214] |
| Rhodococcus etherivorans BCP1 | AsO43− | Mycothiols and NAD(P)H-dependent intracellular ArsC proteins | [215] |
| Streptomyces zaomyceticus Oc-5, Streptomyces pseudogriseolus Acv-11 | Cu2+ | Biomolecules containing peptide bonds, -NH, -OH, -CN, and C=C reactive groups | [216] |
| Frankia inefficax strain EuI1c | SeO32− | NADH-dependent reductase and dehydrogenase | [217] |
| Rhodococcus erythropolis ATCC 4277 | Fe3+ | N.D. 1 | [218,219] |
| Streptomyces spongiicola AS-3 (cell-free extracts) | Ag+ | Proteins, alcohols, and terpenoids | [220] |
| Nocardiopsis. dassonvillei-DS013 | Ag+ | Biomolecules containing -OH, -C+O, -CCl, -CBr, and -C≡C reactive groups | [221] |
| Streptomyces sp. M10A65 | SeO32− | Intracellular NAD(P)H-dependent reductase | [222] |
1 N.D. indicates not determined, which is referred to the nature of the biomolecules mediating metal(loid) reduction in Actinobacteria.
Arthrobacter and Streptomyces spp. can reduce, under different microbial growth conditions, Cr6+ (present in the form of chromate, CrO42−) to the less harmful Cr3+ [14]. Similarly, Cr6+ reduction was also described for Actinobacteria belonging to Microbacterium, Flexivirga, Intrasporangium, Corynebacterium, Micrococcus, and Amycolatopsis spp. [152,160,180,199,201,210,214]. In Arthrobacter and Streptomyces spp., Cr6+ reduction can occur either intra- or extracellularly. In the latter, enzymes are produced and secreted by bacterial cells [210], while intracellular mechanisms generally implied the biosorption and uptake of metal cations, their reduction in the cytoplasm, and the accumulation of Cr3+ as intracellular deposits through metal binding with extracellular or intracellular proteins (Table 3) [153,210]. Moreover, Sugiyama and colleagues (2012) proposed the mechanism of Cr6+ bioreduction in Flexivirga alba ST13T, where the metal cations were first reduced either on the cell surface or intracellularly using glucose as an electron donor; the as-produced soluble Cr3+ ions were then extracellularly secreted and precipitate as Cr(OH)3 deposits, which bound the bacterial cell wall [180].
Streptomyces sp. 3 M and A. crystallopoietes ES 32 were the first members belonging to the corresponding species described to bioconvert Cr6+ through either a constitutive enzyme or an NADH-dependent chromate reductase [159,165]. A similar constitutive reduction mechanism was identified in Streptomyces sp. MC1, which featured a high Cr6+-reductase activity [174]. An intracellular enzymatic reduction appeared to be responsible for Cr6+ biotransformation in S. siyoaensis Lv81-138 [192], A. rhombi-RE [175], A. ramosus [178], and A. aurescens MM10 [179]. On the other hand, reducing substances of nonenzymatic nature mediated the bioconversion of Cr6+ into CrOOH in S. thermocarboxydus NH50. These reducing agents were released into the culture supernatants, and their production was induced upon the addition of the metal precursor [164,223].
In actinobacterial members, Cr6+ reduction depends on several factors, including the initial metal concentration, pH and temperature of the system, composition of the culture medium, physiological state and growth phase of the cells, as well as the addition of either cations or anions to the medium. Indeed, the higher the concentration of Cr6+ precursor, the lower is the rate of Cr reduction, likely due to increased toxicity of these cations [152,161,171,173,183,184,185,186,201]. The pH and the temperature of the microbial cultures differentially influenced Cr6+ bioconversion as a function of the actinobacterial species under analysis, revealing maximum reduction rates at optimal bacterial growth conditions [153,161,171,185,186,201]. It is noteworthy mentioning that pH variations from its optimum could change the ionization rate and conformation of the enzyme(s) devoted to Cr6+ bioprocessing, limiting their activity [171]. However, pH alterations from 4 to 9 in Streptomyces sp. MC1 cultures did not affect Cr6+ bioconversion, highlighting the stability of the enzymatic system involved [173]. On the other hand, the addition of glucose, yeast extract, or peptone to the culture media generally led to an increased Cr6+ reduction, as these carbon compounds acted as electron donors for both the uptake and transformation of metal cations and sources of reducing equivalents (i.e., NADH), supporting this bioprocess [152,153,161,164,171,174,180,183,184,185,199,201,210]. In this regard, the addition, at the initial time of incubation, of either organic compounds or metal ions to S. griseus resting cell suspension resulted in fast Cr6+ biotransformation [161] during Streptomyces sp. MC1 cultures showed a low rate of metal bioprocessing at prolonged Cr exposure times [153]. Providing either Streptomyces or Arthrobacter spp. culture media with diverse metal(loid) ions also influenced Cr6+ reduction. The addition of Cu2+ led to an increased rate of intra- and extracellular Cr6+ bioconversion, indicating that Cu could act as a “catalyst” for this bioprocess [164,183,184,185,186]. The presence of Co2+, Zn2+, and several anions in Streptomyces spp. growth media similarly enhanced Cr reduction [186], which was not influenced by Ni2+, Cd2+, and Pb2+ [164,186]. The only ion negatively affecting Cr reduction was Ca2+, likely due to a strong competition between these two metals for the binding site present in the enzymatic system(s) [171]. Conversely, divalent cations, such as Co2+, Hg2+, Ni2+, Cd2+, Zn2+, and Ba2+, greatly inhibited Cr6+ reduction in Arthrobacter spp., being Ca2+ and Cu2+ the only metal species that improved the rate of this bioprocess [175,183,184,185].
Besides Cr bioprocessing, several actinobacterial spp. strains showed a good ability in facing Hg2+, Cu2+, Ag+, Au3+, and SeO32− toxicity mostly through NAD(P)H-dependent reductases and dehydrogenases (Table 3), although a deep characterization of these bioprocesses is still lacking. Particularly, NADH-dependent reductases specific to this actinobacterial genus played a key role in Ag+ and Zn2+ bioconversion performed by Rhodococcus sp. NCIM2891 and R. pyridinivorans NT2, as proven by the high upregulation of these enzymes upon bacterial exposure to the metal precursor [187,193]. The extracellular α-amylase and the cell wall polysaccharide teichuronic acid (TUA) were instead responsible for Au3+ reduction in M. luteus NCIM 2379 [190]. Indeed, the partially purified enzyme was capable of bioconverting Au cations at 55 °C (optimum temperature for α-amylase), possibly through the reaction between Au3+ and thiol (RSH) groups of the amylase, while the reactive D-glucose and N-acetyl-D-mannosaminuronic acid moieties present within TUA were suggested to mediate this bioprocess in the heat-inactivated dried M. luteus biomass [190]. In Actinobacter sp., the observed rapid reduction of Au3+ was ascribed to the abundant secretion of the enzyme cytochrome oxidase, whose activity was enhanced in an anoxic environment at specific temperatures (i.e., 37 °C) [169,224]. The same environmental isolate also showed good proficiency in biotransforming Fe-containing compounds, as either mixture (ferricyanide (K3Fe(CN)6/ferrocyanide (K4Fe(CN)6), ferric chloride (FeCl3)/ferrous sulfate (FeSO4)) or single salts (FeCl3) [167,170]. The exposure of Actinobacter sp. to K3Fe(CN)6/ K4Fe(CN)6 mixture stimulated the induction of two proteins with hydrolyzing activity, likely responsible for Fe-bioconversion [167], while the presence of either FeCl3 or FeCl3/FeSO4 in the culture media triggered a separate cell response [170]. The bioreduction of Fe3+ within FeCl3 to the less toxic Fe2+ was mediated by an extracellular iron reductase with siderophore activity—i.e., ferrisiderophore reductase—whose concentration increased in response to the excess of Fe salt supplied to the growth medium [170]. The importance of these reductases in Fe bioprocessing was confirmed by the addition of Zn salts, as potent inhibitors of Fe reductases, to Actinobacter sp. cultures, which determined the lack of Fe-bioconversion [170]. Similarly, when the same environmental isolate was incubated in the presence of FeCl3/FeSO4 mixture, two additional proteins, identified as phosphoadenosyl sulfate and sulfite reductases, were detected, indicating their implication in handling the stress deriving from SO42− ions, which was confirmed by their competition with CrO42− [170].
In Actinobacteria, the reduction of metal(loid) oxyanions, such as SeO32−, TeO32−, and AsO43−, is mostly mediated by RSH-containing molecules, which are abundantly present within bacterial cells [225], through the so-called Painter-type reaction [226]. According to the latest proposed mechanism for Se-oxyanion bioconversion [227], SeO32− react with RSH-containing compounds producing a relatively stable intermediate featuring Se2+ and superoxide ions (O2−), which undergo several biotransformation steps to prevent oxidative damage. The Se2+-intermediate is then bioconverted by Glutathione (GSH) or thioredoxin (Trx) reductases generating a highly unstable intermediate (i.e., RS-Se−) that spontaneously dismutates in Se0, favoring the regeneration of intracellular RSH-pool [227]. This mechanism was originally proposed for Gram-negative bacterial strains, as they feature high intracellular concentrations of GSHs, which are considered the first stress–response molecules for handling the presence of metal(loid) ions [227]. However, the abundance of MSHs in Actinobacteria and their greater redox stability as compared to GSHs [225] confers to these microorganisms the ability to handle and reduce high amounts of oxyanions [228]. In this regard, the Painter-type reaction appeared to be involved in either the intracellular or membrane-bound reduction of the oxyanions SeO32− and TeO32− to their elemental forms (Se0 and Te0) in both Rhodococcus and Streptomyces spp. [203,204,208,209], as well as the intracellular bioconversion of the oxyanion AsO43− to AsO33− by C. glutamicum [168,229] and R. etherivorans BCP1 [215] through the NADPH-dependent MSH_Mrx1 pathway [230,231]. In both these strains, a second yet important mechanism proposed for AsO43− bioconversion is based on the activity of thioredoxin- and NADPH-dependent ArsC proteins that mediate RSH/disulfide redox reactions [215,229].
2.4. Metal Efflux Systems
Besides the metal(loid) uptake, bacteria have evolved an extraordinary variety of resistance mechanisms that overall rely on multidrug efflux systems (MES) [232,233], which control the accumulation of molecules and their homeostasis between the intra- and extracellular environment. These efflux systems are essentially responsible for the protection of microorganisms against exceeding amounts of organic solvents, metal(loid)s, toxic lipids, quorum sensing molecules, and biocides, to name a few. The substrate specificity of most MES is very broad, thus representing the main factor contributing to the development of the microbial multidrug resistance (MDR) phenotype [234]. Based on the amino acid sequence identity and the energy source (either ATP hydrolysis or chemical gradient), MES can be distinguished into five main families: (i) ABC transporters, (ii) small multidrug resistance (SMR), (iii) resistance-nodulation cell division (RND), (iv) major facilitator superfamily (MFS), and (v) multidrug and toxic compound extrusion (MATE) [235,236,237,238]. The genome analysis approach carried out by Getsin and colleagues (2013) unveiled that S. coelicolor carries a total of 156 systems, which include channel pores, primary channels, secondary carries, translocators, transmembrane electron flow carriers, auxiliary proteins, and other poorly characterized putative transporters, devoted to both intra- and extracellular trafficking of inorganic compounds [239]. This study highlights the genome-based enzymatic systems inferred to have a role in metal import and efflux processes in this actinobacterial strain, therefore being involved in metal homeostasis and cell survival [239]. In this regard, when very high Ni2+ concentrations were present in the growth medium of S. coelicolor A3(2), the cells expressed the Ni-responsive regulator Nur, which is responsible for the downregulation of genes coding for enzymes involved in the Ni influx [135]. Yet, S. coelicolor A3(2) could grow only in media amended with low Ni concentrations [240], likely because of nonspecific Ni-transporters such as those involved in Mg2+ import [129], which mediate the uncontrolled entry of Ni2+ and may compromise the bacterial cell survival. Furthermore, Ni2+ accumulation supported the activity of Ni-dependent enzymes in Streptomyces F4 and E8 strains [141], while excesses of these cations became severely toxic [241]. In another example, R. opacus possesses a finely regulated uptake/efflux system dependent on Zn concentration, which is responsible for the Zn2+ uptake as efficient as the efflux one, thus maintaining physiological levels of these cations in the cell cytoplasm. To prove this point, Zn-loaded cells exposed to an additional amount of either Zn2+ or Cd2+ showed a decrease in the level of intracellular Zn2+, suggesting both an inducible (Zn2+) and a cross-inducible (Cd2+) metal efflux system [242].
Specific metal efflux systems are also involved in arsenic resistance mechanisms. Arsenic detoxification generally relies on the conversion of AsO43− to AsO33−, which is further extruded from the cell using the arsenite efflux proteins ArsB or Acr3 [243]. The genomes of Actinobacteria almost exclusively possess genes encoding Acr3 permease as an arsenite extrusion system. In C. glutamicum, Acr3 mediated arsenite efflux depending on electrochemical energy [244]. On the other hand, in R. etherivorans BCP1, Acr3 seemed to be associated with the ATPase activity of the AsO33− transporting ArsA, resulting in the production of an efficient arsenite extrusion system represented by ATPase ArsA-Acr3 pump, which may contribute to the great resistance shown by this strain towards AsO43− [215].
3. Metal(loid) Nanomaterial Biosynthesis by Actinobacteria
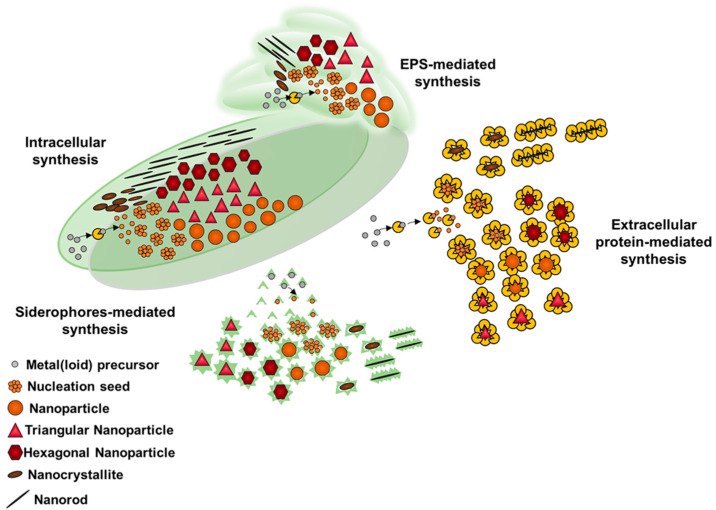
Although diverse biochemical strategies could be exploited by bacteria to regulate metal(loid) concentration in the environmental niches they live in, these microorganisms elicit metabolic traits aimed at detoxifying their habitat, generally involving the reduction of metal(loid) ions to less toxic and bioavailable (e.g., elemental) forms. This process results in a high intra- or extracellular localized concentration of metal(loid) atoms, which, to counteract their thermodynamic instability, tend to aggregate each other, eventually assembling in defined and uniform metal(loid) nanostructures (MeNSs; Figure 2 and Figure 3) [162,204,208,209]. Thus, the proficiency of several bacterial genera, including Actinobacteria, to cope with metal(loid) ions makes them ideal candidates in the microbial nanotechnology field—i.e., the exploitation of microorganisms for the green and eco-friendly production of valuable Me nanomaterials (MeNMs). Indeed, a wide spectrum of Actinobacteria showed a strong attitude in biosynthesizing intra-, extracellular, or membrane-bound MeNMs with different sizes, morphologies, and properties (Table 4). However, parameters and biochemical processes influencing the production of MeNSs and their assembly, in combination with a lack of their extensive physical-chemical characterization and potential applications, are still the black holes of this emerging field, underlining some critical concerns about the possibility of scaling-up the biogenic route behind NMs production stream. In this regard, key aspects to consider in designing innovative MeNM production methods are the choice of reducing agents, metal(loid) precursor-to-reducing agent ratio, temperature, pH, and reaction time. Considering the biogenic synthesis, controlling the reducing agent(s) is a very demanding task, especially when whole cells or EPS are used, as several and complex biomolecules could participate in these bioprocesses [22]. Moreover, whole-cell mediated synthesis also forces to keep constant the temperature and pH of the system, parameters that are optimized for the bacterial strain investigated. Particularly, a temperature between 30 and 37 °C and pH values close to neutrality are the optimum conditions to favor the growth of Actinobacteria [169]. Alternatively, when the metal(loid) reduction occurs through cell-free extracts or EPS, a higher flexibility for these two parameters can lead to an improved optimization of the NM production. For instance, a temperature of 70–100 °C mediated the fast reduction of Au3+ or Ag+ generating MeNSs using cell-free extracts derived from Nocardia farcinica [182] and Gordonia amarae [202], as well as the EPS recovered from Arthrobacter sp. B4 [245].
Figure 2.
Schematic representation of the diverse biogenic nanomaterial morphologies obtained by actinobacterial biotransformation of metal salt precursors through intra- or extracellular (i.e., mediated by siderophores, extracellular proteins, and extracellular polymeric substances (EPS)) processes.
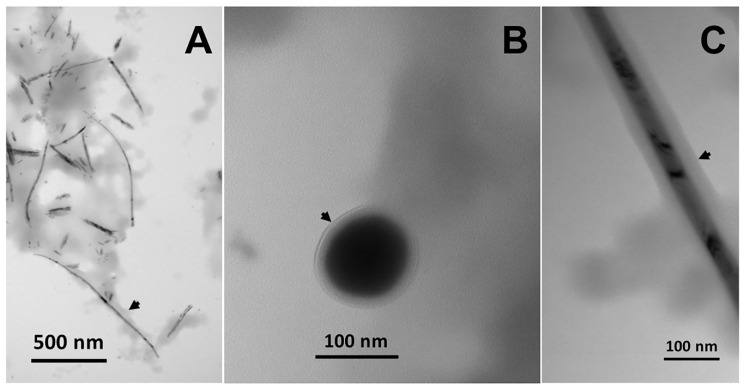
Figure 3.
Biogenic nanomaterials in the form of tellurium nanorods (TeNRs) (A), selenium nanoparticles (SeNPs) (B), and selenium nanorods (SeNRs) (C) produced by the actinomycete Rhodococcus etherivorans BCP1 cells grown in liquid-rich medium amended with either potassium tellurite of sodium selenite as metal salt precursors. Arrowheads point toward the slight electron-dense organic coating featuring biogenically produced nanostructures.
Table 4.
Overview of metal(loid) NMs produced by Actinobacteria, their thermodynamic stabilization, physical-chemical and applicative properties.
| Actinobacteria | Growth Conditions | Shape | Size (nm) | Crystal Structure | Stabilization | Properties | References |
|---|---|---|---|---|---|---|---|
| AuNMs | |||||||
| Thermomonospora sp. | growing cells | AuNPs 1 | ca. 10 | fcc 2 | extracellular secreted proteins | N.D. 3 | [162] |
| Rhodococcus sp. | resting cells | AuNPs | ca. 9 | fcc | N.D. | N.D. | [163] |
| Actinobacter sp. | BSA-exposed biomass | triangular hexagonal AuNPs | 50–500 | fcc | extracellular proteases | N.D. | [169] |
| BSA-exposed biomass (anoxic) | triangular AuNPs | 30–50 | fcc | ||||
| BSA-exposed biomass 25 C | AuNPs | N.D. | fcc | ||||
| Streptomyces viridogens HM10 | resting cells | AuNPs | 18–20 | fcc | N.D. | antibacterial | [246] |
| Arthrobacter sp. 61B; Arthrobacter globiformis 151B | resting cells | AuNPs | 8–40 | fcc | N.D. | N.D. | [247] |
| Streptomyces griseus | resting cells | AuNPs | 50 | fcc | N.D. | N.D. | [248] |
| Nocardia farcinica | cell-free extract | AuNPs | 15–20 | fcc | N.D. | N.D. | [182] |
| Streptomyces hygroscopicus | resting cells (neutral pH) | AuNPs | 2–10 | fcc | intracellular bioactive compounds | antibacterial, electrochemical | [176] |
| Streptomyces hygroscopicus | resting cells (acidic pH) | hexagonal, pentagonal AuNPs | 30–1500 | fcc | intracellular bioactive compounds | antibacterial, electrochemical | [176] |
| Streptomyces sp. ERI-3 | cell-free extract | AuNPs | ca. 9 | fcc | N.D. | N.D. | [249] |
| Micrococcus luteus NCIM 2379 | extracted α-amylase | AuNPs | ca. 6 | N.D. | extracted α-amylase | N.D. | [192] |
| extracted TUA | hexagonal AuNPs | ca. 50 | extracted TUA | ||||
| Streptomyces sp. VITDDK3 | cell-free extract | hexagonal, cubical AuNPs | ca. 90 | fcc | (2S,5R,6R)-2-hydroxy-3,5,6-trimethyloctan-4-one | antifungal | [191] |
| Sacchomonospora sp. | cell-free extract | triangular AuNPs | 40–80 | fcc | polypeptides | N.D. | [250] |
| Streptomyces hygroscopicus | resting cells | AuNPs | 10–20 | N.D. | N.D. | N.D. | [251] |
| Arthrobacter nitroguajacolicus | resting cells | AuNPs | ca.40 | fcc | N.D. | N.D. | [252] |
| Gordonia amarae | cell-free extract | AuNPs | 15–40 | fcc | N.D. | sensor for Cu detection | [202] |
| Gordonia amicalis HS-11 | cell-free extract | AuNPs | 5–25 | fcc | glycolipids | antioxidant | [253] |
| Streptomyces grisoruber | cell-free extract | triangular, hexagonal AuNPs | 5–50 | fcc | extracellular biomolecules | catalytic | [254] |
| Streptomyces sp. NH21 | cell-free extract | AuNPs | 10 | fcc | proteins | antibacterial | [205] |
| Nocardiopsis dassonvillei DS013 | growing cells | AuNPs | 30–80 | fcc | proteins | antibacterial | [221] |
| AgNMs | |||||||
| Corynebacterium strain SH09 | resting cells | AgNPs | 10–15 | fcc | proteins | N.D. | [166] |
| Streptomyces hygroscopicus | cell-free extract | AgNPs | 20–30 | fcc | extracellular biomolecules | antibacterial | [176] |
| Corynebacterium glutamicum | resting cells | irregular AgNPs | 5–50 | fcc | proteins | N.D. | [177] |
| Streptomyces glaucus 71MD | resting cells | AgNPs | 4–25 | fcc | N.D. | N.D. | [255] |
| Streptomyces sp. | cell-free extract | AgNPs | 15–25 | fcc | extracellular proteins | N.D. | [181] |
| Streptomyces rochei | cell-free extract | N.D. | N.D. | N.D. | N.D. | antibacterial | [256] |
| Rhodococcus NCIM 2891 | growing cells cell-free extract |
AgNPs | ca.10 | fcc | N.D. | N.D. | [187] |
| 5–50 | proteins | antibacterial | [257] | ||||
| ca. 15 | mesophilic proteins | fluorescent, antimicrobial, catalytic | [188] | ||||
| Nocardiopsis sp. MBRC-1 | cell-free extract | AgNPs | 30–80 | fcc | proteins | antimicrobial, anticancer | [258] |
| Actinobacteria sp. PSBVIT-13 | cell-free extract | AgNPs | ca. 45 | fcc | proteins | antibacterial | [259] |
| Streptomyces sp. JAR1 | cell-free extract | N.D. | ca. 68 | fcc | extracellular proteins | antibacterial, antifungal | [189] |
| Micrococcus luteus | growing cells | AgNPs | <100 | fcc | N.D. | N.D. | [260] |
| Streptomyces sp. P-311 | crude enzyme extract | AgNPs | 100–200 | fcc | N.D. | antibacterial | [261] |
| Streptomyces sp., Streptoverticillium sp. | growing cells | AgNPs | ca. 8 < 70 | N.D. | N.D. | N.D. | [262] |
| Streptomyces sp. LK3 | cell-free extract | AgNPs | ca. 5 | fcc | N.D. | acaricidal | [263] |
| Streptomyces sp. SS2 | cell-free extract and resting cells | AgNPs | ca. 67 | N.D. | secondary metabolites | antibacterial | [264] |
| Pilimelia columellifera subsp. pallida SF23 and C9 | cell-free extract | AgNPs | 4–36 8–60 | N.D. | extracellular proteins | antifungal | [265] |
| Corynebacterium glutamicum | resting cells | AgNPs | 15 | N.D. | N.D. | antibacterial | [266] |
| Streptomyces rochei MHM13 | cell-free extract | AgNPs | 22–85 | N.D. | proteins | antibacterial, antibiofouling, anticancer | [200] |
| Gordonia amicalis HS-11 | cell-free extract | AgNPs | 5–25 | fcc | glycolipids | antioxidant | [253] |
| purified glycolipid | ca. 20 | ||||||
| Streptomyces sp. M-13 and M-24 | cell-free extract | AgNPs | 10–20 | N.D. | N.D. | antibacterial | [267] |
| Streptomyces parvulus DPUA 1549, Streptomyces owasiensis DPUA 1748 | cell-free extract | AgNPs | 1–40 | fcc | amino acids and peptides | antibacterial | [207] |
| Streptomyces sp. NH21 | cell-free extract | AgNPs | ca. 44 | fcc | proteins | antibacterial | [205] |
| resting cells | ca. 8 | ||||||
| Arthrobacter sp. B4 | EPS | AgNPs | 9–72 | fcc | EPS | antibacterial | [245] |
| Micromonospora KPMS10 | cell-free extract | AgNPs | ca.80 | N.D. | N.D. | antibacterial | [268] |
| Streptomyces sp. Al-Dhabi 89 | cell-free extract | cubic AgNPs | 4–11 | fcc | extracellular metabolites | antibacterial | [211] |
| Nocardiopsis alba | cell-free extract | AgNPs | 20–60 | fcc | N.D. | antibacterial, antiviral | [269] |
| Streptomyces spongiicola AS-3 | cell-free extract | AgNPs | ca.22 | fcc | proteins | antibacterial | [220] |
| Streptomyces sp. 192ANMG and 17ANMG | cell-free extract | AgNPs | ca. 9 ca. 35 | hcp 4 | proteins | antibacterial, antibiofilm | [270] |
| Fe-based NMs | |||||||
| Actinobacter strain EC5 | growing cells | mixture of Fe2O3 and Fe3O4 NPs | 10–40 | fcc | extracellular biomolecules | superpara magnetic | [167] |
| Actinobacter sp. | growing cells | Fe2O3 NPs | 5–7 | fcc | proteins | superpara magnetic | [170] |
| mixture of Fe3S4 and FeS2 NPs | ca. 20 | ||||||
| Rhodococcus erythropolis ATCC 4277 | growing cells, cell-free extract | Fe2O3 NPs | 50–100 | fcc | N.D. | N.D. | [218,219] |
| Cu-based NMs | |||||||
| Streptomyces sp. KUA106 | cell-free extract | CuO NPs | 100–150 | N.D. | reductases | antibacterial, antifungal | [271] |
| growing cells | |||||||
| Kocuria flava M-7 | cell-free extract | CuNPs | 5–30 | N.D. | proteins | N.D. | [195] |
| Streptomyces capillispiralis Ca-1 | cell-free extract | CuNPs | 5–59 | fcc | proteins | antimicrobial, larvicidal | [213] |
| Actinomycete sp. VITBN4 | cell-free extract | CuO NPs | ca. 60 | N.D. | proteins | antimicrobial | [272] |
| Streptomyces zaomyceticus Oc-5; Streptomyces pseudogriseolus Acv-11 | cell-free extract | CuO NPs | ca. 80 | fcc | N.D. | antimicrobial, antioxidant, larvicidal | [216] |
| Zn-based NMs | |||||||
| Streptomyces sp. KUA106 | growing cells, cell-free extract | ZnO NPs | 100–150 | N.D. | reductases | antimicrobial | [271] |
| Streptomyces sp. HBUM171191 | growing cells | ZnSO4 NPs | 10–20 | N.D. | N.D. | N.D. | [273] |
| Streptomyces sp. | growing cells | ZnO NPs | N.D. | N.D. | N.D. | antibacterial | [274] |
| Rhodococcus pyridinivorans NT2 | growing cells, cell-free extract | ZnO NPs | ca. 100 | N.D. | metabolites and proteins | UV-protective, photocatalytic, self-cleaning, antibacterial | [193] |
| SeNMs | |||||||
| Streptomyces bikiniensis strain Ess_amA-I | growing cells | SeNPs | 50–100 | N.D. | proteins and enzymes | N.D. | [198] |
| SeNRs 5 | 600 | anticancer | |||||
| Streptomyces minutiscleroticus M10A62 | resting cells | SeNPs | 100–250 | monoclinic | proteins | antibiofilm, antioxidant, wound healing, anticancer, antiviral | [198] |
| Streptomyces sp. ES2-5 | growing cells | SeNPs | 100–500 | N.D. | N.D. | N.D. | [203] |
| Rhodococcus etherivorans BCP1 | growing cells | SeNPs SeNRs | 50–600 | N.D. | amphiphilic biomolecules | N.D. | [208] |
| Frankia inefficax strain EuI1c | growing cells | SeNPs | N.D. | N.D. | N.D. | N.D. | [217] |
| Streptomyces sp. M10A65 | resting cells | SeNPs | 20–150 | monoclinic | proteins | antibacterial, larvicidal, anthelminthic | [222] |
| TeNMs | |||||||
| Rhodococcus etherivorans BCP1 | growing cells | TeNRs | 100–500 | N.D. | amphiphilic biomolecules | N.D. | [204] |
| resting cells | TeNPs TeNRs | 200–700 | trigonal | electrically conductive | [209] | ||
| Co-based NMs | |||||||
| Brevibacterium casei | cell-free extract | Co3O4 NPs | ca. 6 | crystals | proteins | magnetic | [172] |
| Mn-based NMs | |||||||
| Streptomyces sp. HBUM | growing cells | MnSO4 NPs | 10–20 | N.D. | N.D. | N.D. | [273] |
1 NPs indicates nanoparticles. 2 fcc stands for face-centered cubic. 3 N.D. indicates metal(loid) NM features not determined. 4 hcp stands for hexagonal close-packed. 5 NRs stands for nanorods
Changes in the pH can differentially influence MeNM production, depending on the functional groups present within the bacterial extracts. An acidic pH value (ca. 4) allowed to obtain monodispersed and uniform AuNPs by N. farcinica extract [182], while the EPS extracted from Arthrobacter sp. B4 [245] and G. amarae [202] required a pH either close to neutrality (7–8) or alkaline conditions (10–12) for the reduction of Ag+ or Au3+, respectively, leading to the corresponding MeNM production. Extracting specific biomolecules—i.e., glycolipids [253], α-amylases, and TUA [190]—guarantees better control over both the mechanism responsible for the metal(loid) reduction and the concentration of the reducing agent available. Nevertheless, strict control of temperature and pH is still needed to assure the biomolecules’ functional activity [190]. Another parameter influencing the production of biogenic MeNMs is the metal(loid) precursor concentration, which exerts a positive effect until a certain threshold value—i.e., the maximum amount of metal(loid) ions that can be reduced by the actinobacterial strain—is reached [202]. The effect of the precursor-to-reducing agent ratio on the biogenic MeNS synthesis was explored in the case of AgNPs produced by the EPS extracted from Arthrobacter sp. B4 cultures, for which the addition of a large EPS amount determined a high rate of NP synthesis, likely due to an enhanced number of reactive groups interacting with Ag+ [245]. A similar relationship was observed for resting cells belonging to Rhodococcus and Gordonia genera incubated for different time intervals in the presence of metal(loid) ions, as the adaptation of these microorganisms to these chemical species seemed to mediate a faster ion bioconversion, leading to MeNM generation [202,209]. However, these reduction reactions generally reach a plateau that is related to the redox stability of microbial reduction systems, reflecting the bacterial tolerance towards the metal(loid) challenge [202]. Further, increasing the precursor concentration and/or the time of incubation can induce (i) a morphological transition of MeNMs, determining the generation of cubic or hexagonal NPs and one-dimensional nanorods (NRs), as well as (ii) the growth and elongation of NPs and NRs, consequently to the increased density of elemental atoms that can be reached within the infinitesimal spatial volume represented by the bacterial cytoplasm, the latter being true for those strains capable of generating intracellular MeNMs [22,167,209]. This aspect was studied in detail in the case of TeNRs produced by R. etherivorans BCP1 cells under different conditions [204,209]. Stirring and oxygen flow rates were also parameters to account for the synthesis of Fe3O2 NPs by R. erythropolis ATCC 4277 grown in a tank reactor [218,219]. Indeed, a high, stirring rate resulted in large intracellular NPs due to the prevention in releasing enzymes and proteins generally responsible for the extracellular NP synthesis, while the lack of dissolved oxygen may have forced the production of reductases, enhancing NP generation [219]. Similarly, when the reduction of chloroauric acid (HAuCl4) by Actinobacter sp. was carried out under anoxic conditions, the rate of the reaction itself was enhanced, and the as-produced AuNPs were more uniform in shape and monodisperse in size [169].
The inherent complexity and diversity of a biological system, such as that of a bacterial cell, unveils the importance of choosing the microbial cell factory to use, as well as its cell physiology, to develop biogenic MeNM synthetic processes. Different actinobacterial strains have diverse capabilities of processing metal(loid) ions and producing MeNMs, whose cell localization, size, and shape can greatly change in a given microorganism depending on its growth conditions (Table 4). Such an effect can be achieved by playing with the bacterial cell physiology; for instance, R. etherivorans BCP1 cells exposed twice (i.e., conditioned) to the same metal(loid) precursor (SeO32− or TeO32−) showed a higher rate of oxyanion bioconversion as compared to those exposed once, causing an enlargement or an elongation of SeNPs or TeNRs [204,208], respectively. Nevertheless, the highest reduction rate was detected upon the use of BCP1 resting cells to synthesize long and uniform TeNRs, likely due to the high initial redox potential of these bacterial cells [209]. On the other hand, bacterial strains growing in a minimal growth medium supplied with a single source of carbon could result in a reduced metal(loid) ion removal efficiency, probably due to a lower amount of reducing equivalents available that can support the biotic metal(loid) transformation as compared to bacterial cells growing in complex and rich media. However, the bacterial growth on a minimal medium can give rise to more homogeneous MeNMs, representing an advantage from a biotechnological point of view [275].
Although actinobacterial strains and corresponding cell-free extracts efficiently produce MeNMs, the mechanism(s) governing their assembly in the intra- or extracellular environment is still poorly understood. Some insights were put forward by studying the capability of R. etherivorans BCP1 cells of biosynthesizing Se or TeNMs [204,208,209]. In this regard, the formation of these NSs occurred following the so-called LaMer mechanism, by which, once a critical concentration of Se or Te atoms is reached, these atoms collapse, generating nucleation seeds. The latter then undergo aggregation when a critical concentration value is reached, which determines the formation of amorphous Se or Te NPs. Since these NPs feature high surface energy and are confined in the intracellular environment, they tend to dissolve and deposit along one axis, forming more thermodynamically stable Se or TeNRs. This process is favored by the presence of amphiphilic biomolecules surrounding the forming NRs, which could act as a template for the mono-dimensional growth of these NMs [204,208,209].
A common feature and a first indication of the microbial ability to produce MeNMs is the change in color [depending on the considered metal(loid)] generally observed within bacterial cultures or cell-free extracts, which is due to the arising of surface plasmon resonance (SPR) or SPR-like phenomena. Following the bioconversion of metal(loid)s, the reduced chemical species, as well as the forming NMs, generally develop unique optical properties as a function of the metal(loid) element, size, and morphology of the as-produced NSs [169,202,269,276]. The color variation of the suspensions can also be independent of SPR, as in the case of Fe2O3 NPs, which, depending on their size and crystalline configuration, can acquire red, brown, or black shades [277].
Regardless of the color change, the physical-chemical characterization of MeNMs produced by Actinobacteria revealed their versatility in synthesizing a broad spectrum of NSs with diverse localization, morphologies, size, crystalline configuration, and, therefore, properties, whose overview is presented in Table 4. Among these features, the localization of biogenic MeNSs plays a fundamental role in terms of the easiness of their recovery, being the extracellular production preferred, as it makes it unnecessary to perform expensive and time-consuming procedures, such as the isolation of MeNSs from the cell cytosol [21,22]. However, stable and crystalline MeNRs are generally obtained through intracellular processes [208,209], likely due to the high local concentrations of elemental atoms available for their deposition along one axis. Thus, depending on the desired final product and the actual feasibility of the bioprocesses, different actinobacterial strains can be exploited for NM production, which can be optimized considering the above-mentioned parameters.
Another important physical-chemical feature of biogenic NMs is their high thermodynamic stability under diverse conditions, which results in the absence of aggregation phenomena, avoiding the need for postproduction treatments to ensure the applicability of these NSs [22]. This key aspect is to be traced back to the presence of organic material within the biogenic NM extracts that contain biomolecules either derived from or secreted by actinobacterial cells (Table 4). These biomolecules, mostly represented by proteins and enzymes, but also by polypeptides, glycolipids, secondary metabolites, and amphiphilic compounds (Table 4), can reduce the metal(loid) precursor (Table 3) as well as thermodynamically stabilize the forming NMs, through reactive functional groups and steric hindrance phenomena [22]. These chemical features ensure the development of both electrostatic (between reactive groups) and steric (due to the bulky structure) interactions between the biomolecules themselves and MeNSs, decreasing their high surface energy and conferring them good thermodynamic stability [22]. The nature and strength of these interactions seemed to control size, shape, crystalline configuration, and aggregation of Au, Ag, and ZnO NPs produced by Rhodococcus spp., N. farcinica, Rhodococcus NCIM 2891, and R. pyridinivorans NT2 [163,182,193,257]. Indeed, biomolecules capping AgNPs recovered from Rhodococcus NCIM 2891 conferred to these NMs strong stability in neutral, alkaline, and slightly acidic conditions. At alkaline pH values, COO− groups were fully charged and could mediate strong electrostatic repulsive interactions, which, however, disappeared as the pH became closer to the pKa (ca. 5) of this reactive functional group [257]. Similarly, these biogenic AgNPs were thermodynamically stable up to 60 °C, the temperature at which the surrounding biomolecules may denature, hence determining their inactivation [257]. Since several washing steps were not sufficient to completely remove the organic material from the biogenic MeNM extracts recovered from Rhodococcus NCIM 2891, R. pyridinivorans NT2, and R. etherivorans BCP1 [193,204,208,209], it was suggested that thermodynamic equilibrium is reached between the biomolecules and NSs, thus allowing a strong stabilization of the latter. To further support this hypothesis, Kundu et al. (2014) compared the thermodynamic stability of ZnO NPs produced by R. pyridinivorans NT2 with those commercially available. This indicated that the incubation of these NPs with bacterial extracellular proteins favored their stabilization overtime. Indeed, ZnO NPs synthesized by NT2 cells remained steadily dispersed in water for more than 2 months, while the chemogenic ones revealed lower stability [193].
Properties and Applications of Metal(loid) NMs Produced by Actinobacteria
The quality of biogenic MeNMs allows their use in the (bio)technological field, as they show promising antibacterial, antifungal, antioxidant, antiviral, acaricidal, larvicidal, anti-biofouling, anticancer, electrochemical, fluorescent, conductive, superparamagnetic, (photo)catalytic, and sensory properties, to name a few (Table 4).
Currently, resistance to antibiotics by microorganisms (AMR) is a major problem not easy to solve; hence, the potential of biogenic MeNMs, such as AgNPs, to act as bacteriostatic and/or bactericidal compounds has gained an impressive scientific and technological interest [278]. Overall, AgNPs produced by Actinobacteria displays a higher antibacterial activity against Gram-negative as compared to Gram-positive bacteria, likely due to their thicker peptidoglycan layer [189]. Furthermore, biogenic AgNPs can prevent pathogen biofilm formation [200,270], becoming largely promising for the treatment of hospital-oriented infections. Although several studies reported on the antimicrobial potential of biogenic AgNPs, alongside other MeNMs, three possible mechanisms have been formulated to date to explain their antimicrobial efficiency: (i) NP attachment to the cell membrane negatively affecting its bioenergetic functions (i.e., permeability and respiration), (ii) NP penetration within the cells by interacting with S- and P-containing compounds (i.e., DNA) causing lethal damage and the depletion of ATP intracellular reservoir, or (iii) the release of Ag+, causing DNA to lose its replication ability and cellular proteins to become inactivated [189,279]. AgNPs, as well as SeNPs, have also been proven to be efficient, acting in synergy with antibiotics, enhancing the antimicrobial activity of the latter [189,200,222,265], as multiple resistance mechanisms are needed for the pathogens to simultaneously counteract these bioactive substances. Moreover, antibiotics contain many functional groups that can react with AgNPs, favoring their efficacy [189]. Similarly, Cu-, Zn-, and Se-based NPs produced by Actinobacteria featured high antimicrobial ability in limiting or preventing the proliferation of pathogens as either planktonic cells or biofilms [193,195,213]. In this regard, Cu-based NPs caused the production of hydroxyl radicals, which in turn determine the destruction of plasmid and genomic DNA and the inactivation of essential enzymes through their binding with Cu2+ [213,216]. Additionally, Cu-based NPs showed good larvicidal properties, as their size guarantees them an easy penetration through either the oral canal or the breaching of the larval cuticle membrane, leading to their diffusion within the body. Here, Cu-based NPs can bind, as for bacterial cells, to S- and P-containing biomolecules, causing inhibition of both DNA and protein synthesis [213].
Besides antimicrobials, common features of MeNMs obtained from Actinobacteria are their good anticancer and antioxidant properties (Table 4), yet their mode of action is still poorly understood, as they showed both antioxidant and pro-oxidant behaviors, as a function of the experimental conditions tested [198]. Indeed, although some of these NMs were described to induce apoptosis and cell cycle arrest [196], the most accredited mechanism explaining the anticancer activity of biogenic MeNSs is their ability to promote the generation of ROS in cancer cells, eventually causing cell death. For this situation to happen, a high level of free Cu2+ is needed in the cellular environment, as they can bind with NMs (e.g., SeNPs) along with DNA, forming a ternary complex, which can undergo a redox reaction that determines the generation of Cu+ ions, whose subsequent oxidation produces ROS [198]. Since an elevated level of Cu2+ is quite common within cancer tissues and cells, the mobilization of these ions can cause the pro-oxidation activity of MeNMs [198]. On the other hand, in normal conditions, these NSs exhibit strong antioxidant activity by functioning as free radical scavengers for ROS in living organisms [196,216,253]. The morphology and the size of biogenic MeNMs are the two parameters highly influencing this antioxidant behavior, being monodispersed NPs more suitable for this task [216,253]. All these properties are even emphasized by the low cytotoxicity of biogenic MeNMs towards human cell lines [193,207,216,270], encouraging the potential applications of these nanoformulations in the biomedical field.
From a technological point of view, Au, Ag, ZnO, and Te NMs produced by the Actinobacteria featured good electrochemical [176], (photo)catalytic [193,254,257], sensing [202], superparamagnetic [167,170,171], and conductive [176,208] properties (Table 4). Particularly, AuNPs synthesized by G. amarae extract combined with the amino acid L-cysteine revealed a high proficiency as a sensor for Cu2+ at concentrations as low as 4.0 nM [202]. The addition of L-cysteine to AuNP solutions determines the aggregation of the NPs through -SH groups on the amino acid; however, when Cu2+ are present, L-cysteine undergoes an oxidation reaction becoming cystine, in which any free -SH moiety is not available to interact with AuNPs [202]. Since the aggregation of AuNPs is linked to their SPR phenomena, a change in the absorption profile of NPs themselves occurs, making the interaction between L-cysteine and Cu2+ easily detectable through UV-visible spectroscopy [202]. Moreover, upon addition of other metal ions, AuNP agglomeration was still detected, indicating the specificity of AuNPs recovered from G. amarae cell-free extract for the detection of Cu ions in suspension without the need of additional tags or functionalization with specific ligands [202].
4. Conclusions
Several members of the Actinobacteria phylum are capable of withstanding and converting toxic metals into harmless forms ascribed to metabolism-dependent and -independent cellular activities. Thus, a better comprehension of these biotic processes constitutes an important basis for developing successful bioremediation approaches to reclaim metal(loid)-polluted environments by using either actinobacterial pure cultures or ad hoc consortia, which meet the requested criteria of green chemistry and circular economy. Moreover, a relevant added value of these bioprocesses is represented by the proficiency of Actinobacteria to bioconvert toxic metal(loid) ions, consequently producing nanomaterials of various sizes, shapes, and composition that feature a broad spectrum of applicative physical-chemical (e.g., optical, photoluminescent, photocatalytical, electrical) and biological (anticancer and antimicrobial) properties. Nevertheless, the knowledge regarding (i) biological mechanisms behind metal(loid) nanostructure production, (ii) the influence of cell physiology and metabolism on the final nanoscaled products, (iii) the optimization of nanomaterial recovery from actinobacterial cultures or extracts, (iv) the identity of macromolecules determining the high thermodynamic stabilization of these nanostructures, and (v) their structure-to-properties relationships still needs to be improved to guarantee the translation of biogenic nanostructures from benchtop research to their practical application.
Acknowledgments
We acknowledge the Italian Ministry of Education, University, and Research (MIUR) for the PON project on Research and Innovation 2012–2020 (Attraction and International Mobility—AIM1808223) and L’Oréal-UNESCO “For Women In Science” Award (Italian edition 2020).
Author Contributions
Conceptualization, A.P., E.P., R.J.T., D.Z., M.C.; writing—original draft preparation, A.P., E.P.; writing—review and editing, R.J.T., D.Z., M.C.; supervision, M.C.; funding acquisition, A.P., R.J.T., M.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by 2020–2025 Natural Sciences and Engineering Research Council of Canada (NSERC), Discovery Grant, “Bacterial multiresistance mechanisms: multimetal(loid) sensitivity-tolerance and transformations”.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kabata-Pendias A., Pendias H. Trace Elements in Soils and Plants. CRC Press, Inc.; Boca Raton, FL, USA: 1984. [Google Scholar]
- 2.Maret W. The metals in the biological periodic system of the elements: Concepts and conjectures. Int. J. Mol. Sci. 2016;17:66. doi: 10.3390/ijms17010066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ledin M. Accumulation of metals by microorganisms-processes and importance for soil systems. Earth Sci. Rev. 2000;51:1–31. doi: 10.1016/S0012-8252(00)00008-8. [DOI] [Google Scholar]
- 4.Tchounwou P.B., Yedjou C.G., Patlolla A.K., Sutton D.J. Heavy Metal Toxicity and the Environment. In: Luch A., editor. Molecular, Clinical and Environmental Toxicology. Volume 3. Springer; Basel, Switzerland: 2012. pp. 133–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barbosa F., Jr. Toxicology of metals and metalloids: Promising issues for future studies in environmental health and toxicology. J. Toxicol. Environ. Health A. 2017;80:137–144. doi: 10.1080/15287394.2016.1259475. [DOI] [PubMed] [Google Scholar]
- 6.Hamelink J., Landrum P.F., Bergman H., Benson W.H. Bioavailability: Physical, Chemical and Biological Interactions. CRC Press; Boca Raton, FL, USA: 1994. [Google Scholar]
- 7.Presentato A., Cappelletti M., Sansone A., Ferreri C., Piacenza E., Demeter M.A., Crognale S., Petruccioli M., Milazzo G., Fedi S., et al. Aerobic growth of Rhodococcus aetherivorans BCP1 using selected naphthenic acids as the sole carbon and energy sources. Front. Microbiol. 2018;9:672. doi: 10.3389/fmicb.2018.00672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chakraborty I., Sathe S.M., Khuman C.N., Ghangrekar M.M. Bioelectrochemically powered remediation of xenobiotic compounds and heavy metal toxicity using microbial fuel cell and microbial electrolysis cell. Mater. Sci. Technol. 2020;3:104–115. doi: 10.1016/j.mset.2019.09.011. [DOI] [Google Scholar]
- 9.Gullberg E., Albrecht L.M., Karlsson C., Sandegren L., Andersson D.I. Selection of a multidrug resistance plasmid by sublethal levels of antibiotics and heavy metals. MBio. 2014;5:e01918-14. doi: 10.1128/mBio.01918-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pal C., Bengtsson-Palme J., Kristiansson E., Larsson D.J. Co-occurrence of resistance genes to antibiotics, biocides and metals reveals novel insights into their co-selection potential. BMC Genom. 2015;16:964. doi: 10.1186/s12864-015-2153-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Alduina R., Gambino D., Presentato A., Gentile A., Sucato A., Savoca D., Filippello S., Visconti G., Caracappa G., Vicari D., et al. Is Caretta caretta a carrier of antibiotic resistance in the Mediterranean sea? Antibiotics. 2020;9:116. doi: 10.3390/antibiotics9030116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ali H., Khanb E., Sajad A.M. Phytoremediation of heavy metals-Concepts and applications. Chemosphere. 2013;91:869–881. doi: 10.1016/j.chemosphere.2013.01.075. [DOI] [PubMed] [Google Scholar]
- 13.Abdel-Ghani N., El-Chagbaby G. Biosorption for metal ions removal from aqueous solutions: A review of recent studies. Int. J. Latest Res. Sci. Technol. 2014;3:24–42. [Google Scholar]
- 14.Alvarez A., Saez J.M., Davila Costa J.S., Colin V.L., Fuentes M.S., Cuozzo S.A., Benimeli C.S., Polti M.A., Amoroso M.J. Actinobacteria: Current research and perspectives for bioremediation of pesticides and heavy metals. Chemosphere. 2017;166:41–62. doi: 10.1016/j.chemosphere.2016.09.070. [DOI] [PubMed] [Google Scholar]
- 15.Gadd G.M. Microbial Control of Heavy Metal Pollution. In: Fry J., Gadd G.M., Herbert R.A., Jones C.W., Watson-Craik I.A., editors. Microbial Control of Heavy Metal Pollution. Cambridge University Press; Cambridge, UK: 1992. pp. 59–88. [Google Scholar]
- 16.Cappelletti M., Presentato A., Piacenza E., Firrincieli A., Turner R.J., Zannoni D. Biotechnology of Rhodococcus for the production of valuable compounds. Appl. Microbiol. Biotechnol. 2020;104:8567–8594. doi: 10.1007/s00253-020-10861-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ventura M., Canchaya C., Tauch A., Chandra G., Fitzgerald G.F., Chater K.F., van Sinderen D. Genomics of Actinobacteria: Tracing the evolutionary history of an ancient phylum. Microbiol. Mol. Biol. Rev. 2007;71:495–548. doi: 10.1128/MMBR.00005-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Martínková L., Uhnáková B., Pátek M., Nesvera J., Kren V. Biodegradation potential of the genus Rhodococcus. Environ. Int. 2009;35:162–177. doi: 10.1016/j.envint.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 19.Goodfellow M., Kämpfer P., Busse H.-J., Trujillo M.E., Suzuki K.-I., Ludwig W., Whitman W.B. Bergey’s Manual of Systematic Bacteriology: The Actinobacteria. Volume 5 Springer; New York, NY, USA: 2012. [Google Scholar]
- 20.Cappelletti M., Zampolli J., Di Gennaro P., Zannoni D. Chapter 2: Genomics of Rhodococcus. In: Alvarez H.M., editor. Biology of Rhodococcus. 2nd ed. Springer International Publishing; Basel, Switzerland: 2019. pp. 23–60. [DOI] [Google Scholar]
- 21.Piacenza E., Presentato A., Zonaro E., Lampis S., Vallini G., Turner R.J. Selenium and Tellurium nanomaterials. Phys. Sci. Rev. 2018;3:20170100. doi: 10.1515/psr-2017-0100. [DOI] [Google Scholar]
- 22.Piacenza E., Presentato A., Turner R.J. Stability of biogenic metal(loid) nanomaterials related to the colloidal stabilization theory of chemical nanostructures. Crit. Rev. Biotechnol. 2018;38:1137–1156. doi: 10.1080/07388551.2018.1440525. [DOI] [PubMed] [Google Scholar]
- 23.Park J.H., Kim B.S., Chon C.M. Characterization of iron and manganese minerals and their associated microbiota in different mine sites to reveal the potential interactions of microbiota with mineral formation. Chemosphere. 2018;191:245–252. doi: 10.1016/j.chemosphere.2017.10.050. [DOI] [PubMed] [Google Scholar]
- 24.Solecka J., Zajko J., Postek M., Rajnisz A. Biologically active secondary metabolites from Actinomycetes. Open Life Sci. 2012;7:373–390. doi: 10.2478/s11535-012-0036-1. [DOI] [Google Scholar]
- 25.Liao L., Chen R., Jiang M., Tian X., Liu H., Yu Y., Fan C., Chen B. Bioprospecting potential of halogenases from Arctic marine actinomycetes. BMC Microbiol. 2016;16:34. doi: 10.1186/s12866-016-0662-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shivlata L., Satyanarayana T. Thermophilic and alkaliphilic Actinobacteria: Biology and potential applications. Front. Microbiol. 2015;6:1014. doi: 10.3389/fmicb.2015.01014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gupta P., Diwan B. Bacterial exopolysaccharide mediated heavy metal removal: A review on biosynthesis, mechanism remediation strategies. Biotechnol. Rep. 2017;13:58–71. doi: 10.1016/j.btre.2016.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Volesky B. Biosorption and Biosorbents. In: Volesky B., editor. Biosorption of Heavy Metals. CRC Press; Boca Raton, FL, USA: 1990. pp. 3–6. [Google Scholar]
- 29.Wilde E.W., Benemann J.R. Bioremoval of heavy metals by the use of microalgae. Biotechnol. Adv. 1993;4:781–812. doi: 10.1016/0734-9750(93)90003-6. [DOI] [PubMed] [Google Scholar]
- 30.Bankar A., Kumar A., Zinjarde S. Removal of chromium (VI) ions from aqueous solution by adsorption onto two marine isolates of Yarrowia lipolytica. J. Hazard. Mater. 2009;170:487–494. doi: 10.1016/j.jhazmat.2009.04.070. [DOI] [PubMed] [Google Scholar]
- 31.Bankar A.V., Zinjarde S.S., Kapadnis B.P. Management of Heavy Metal Pollution by Using Yeast Biomass. In: Satyanarayana T., Johri B., Prakash A., editors. Microorganisms in Environmental Management. Springer; Dordrecht, The Netherlands: 2012. pp. 335–363. [DOI] [Google Scholar]
- 32.Gadd G.M., White C. Microbial treatment of metal pollution-a working biotechnology? Trends Biotechnol. 1993;11:353–359. doi: 10.1016/0167-7799(93)90158-6. [DOI] [PubMed] [Google Scholar]
- 33.Goksungur Y., Üren S., Guvenc U. Biosorption of cadmium and lead ions by ethanol treated waste baker’s yeast biomass. Bioresour. Technol. 2005;96:103–109. doi: 10.1016/j.biortech.2003.04.002. [DOI] [PubMed] [Google Scholar]
- 34.Plette A.C.C., van Riemsdijk W.H., Benedetti M.F., van der Wal A. pH dependent charging behavior of isolated cell walls of a gram-positive soil bacterium. J. Colloid Interface Sci. 1995;173:354–363. doi: 10.1006/jcis.1995.1335. [DOI] [Google Scholar]
- 35.Stratton H., Brooks P., Griffiths P., Seviour R. Cell surface hydrophobicity and mycolic acid composition of Rhodococcus strains isolated from activated sludge foam. J. Ind. Microbiol. Biotechnol. 2002;28:264–267. doi: 10.1038/sj.jim.7000241. [DOI] [PubMed] [Google Scholar]
- 36.Botero A.E.C., Torem M.L., de Mesquita L.M.S. Fundamental studies of Rhodococcus opacus as a biocollector of calcite and magnesite. Miner. Eng. 2007;20:1026–1032. doi: 10.1016/j.mineng.2007.03.017. [DOI] [Google Scholar]
- 37.Lin Y., Wang X., Wang B., Mohamad O., Wei G. Bioaccumulation characterization of zinc and cadmium by Streptomyces zinciresistens, a novel actinomycete. Ecotoxicol. Environ. Saf. 2012;77:717. doi: 10.1016/j.ecoenv.2011.09.016. [DOI] [PubMed] [Google Scholar]
- 38.Sheng P.X., Tan L.H., Chen J.P., Ting Y.P. Biosorption performance of two brown marine algae for removal of chromium and cadmium. J. Disper. Sci. Technol. 2004;25:679–686. doi: 10.1081/DIS-200027327. [DOI] [Google Scholar]
- 39.Vasquez T.G.P., Botero A.E.C., de Mesquita L.M.S., Torem M.L. Biosorptive removal of Cd and Zn from liquid streams with a Rhodococcus opacus strain. Miner. Eng. 2007;20:939–944. doi: 10.1016/j.mineng.2007.03.014. [DOI] [Google Scholar]
- 40.Bueno B.Y.M., Torem M.L., Molina F., de Mesquita L.M.S. Biosorption of lead (II), chromium (III) and copper (II) by R. opacus: Equilibrium and kinetic studies. Miner. Eng. 2008;21:65–75. doi: 10.1016/j.mineng.2007.08.013. [DOI] [Google Scholar]
- 41.Cayllahua J.E.B., Torem M.L. Biosorption of aluminum ions onto Rhodococcus opacus from wastewaters. Chem. Eng. J. 2010;161:1–8. doi: 10.1016/j.cej.2010.03.025. [DOI] [Google Scholar]
- 42.Kumar S., Krishnan K. Biosorption of Cd (II) and Pb (II) ions by aqueous solutions of novel alkalophillic Streptomyces VITSVK5 spp. biomass. J. Ocean Univ. China. 2011;10:61–66. doi: 10.1007/s11802-011-1771-z. [DOI] [Google Scholar]
- 43.Kumar S., Krishnan K. Biosorption of Cr (III) and Cr (VI) by Streptomyces VITSVK9 spp. Ann. Microbiol. 2011;61:833–841. doi: 10.1007/s13213-011-0204-y. [DOI] [Google Scholar]
- 44.Latha S., Vinothini G., Dhanasekaran D. Chromium [Cr (VI)] biosorption property of the newly isolated actinobacterial probiont Streptomyces werraensis LD22. 3 Biotech. 2015;5:423432. doi: 10.1007/s13205-014-0237-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kirova G., Velkova Z., Gochev V. Copper (II) removal by heat inactivated Streptomyces fradiae biomass: Surface chemistry characterization of the biosorbent. J. BioSci. Biotechnol. 2012;2012:77–82. [Google Scholar]
- 46.Doyle R.J., Matthews T.H., Streips U.N. Chemical basis for the selectivity of metal ions by the Bacillus subtilis wall. J Bacteriol. 1980;143:471–480. doi: 10.1128/JB.143.1.471-480.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xue H.B., Stumm W., Sigg L. The binding of heavy metals to algal surfaces. Water Res. 1988;22:917–926. doi: 10.1016/0043-1354(88)90029-2. [DOI] [Google Scholar]
- 48.Flemming C.A., Ferris F.G., Beveridge T.J., Bailey G.W. Remobilization of toxic heavy metals absorbed to wall-clay composites. Appl. Environ. Microbiol. 1990;56:3191–3209. doi: 10.1128/AEM.56.10.3191-3203.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Plette A.C.C., Benedetti M.F., Vanriemsdijk W.H. Competitive binding of protons, calcium, cadmium, and zinc to isolated cell walls of a Gram-positive soil bacterium. Environ. Sci. Technol. 1996;30:1902–1910. doi: 10.1021/es950568l. [DOI] [Google Scholar]
- 50.Koch A.L. Growth and form of the bacterial cell wall. Am. Sci. 1990;78:327–341. [Google Scholar]
- 51.Kraemer S.M. Iron oxide dissolution and solubility in the presence of siderophores. Aquat. Sci. 2004;66:3–18. doi: 10.1007/s00027-003-0690-5. [DOI] [Google Scholar]
- 52.Timkova I., Sedlakova-Kadukova J., Pristas P. Biosorption and bioaccumulation abilities of Actinomycetes/Streptomyces isolated from metal contaminated sites. Separations. 2018;5:54. doi: 10.3390/separations5040054. [DOI] [Google Scholar]
- 53.Hider R.C., Kong X. Chemistry and biology of siderophores. Nat. Prod. Rep. 2010;27:637–657. doi: 10.1039/b906679a. [DOI] [PubMed] [Google Scholar]
- 54.Challis G.L., Hopwood D.A. Synergy and contingency as driving forces for the evolution of multiple secondary metabolite production by Streptomyces species. Proc. Natl. Acad. Sci. USA. 2003;100:14555–14561. doi: 10.1073/pnas.1934677100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Schwyn B., Neilands J.B. Universal chemical assay for the detection and determination of siderophores. Anal. Biochem. 1987;160:47–56. doi: 10.1016/0003-2697(87)90612-9. [DOI] [PubMed] [Google Scholar]
- 56.Matzanke B.F. Structures, coordination chemistry and functions of microbial iron chelates. In: Matzanke B.F., editor. Handbook of Microbial Iron Chelates. CRC Press; Boca Raton, FL, USA: 1991. pp. 23–72. [Google Scholar]
- 57.Miethke M., Marahiel M.A. Siderophore-based iron acquisition and pathogen control. Microbiol. Mol. Biol. Rev. 2007;71:413–451. doi: 10.1128/MMBR.00012-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Schalk I.J., Hannauer M., Braud A. New roles for bacterial siderophores in metal transport and tolerance. Environ. Microbiol. 2011;13:2844–2854. doi: 10.1111/j.1462-2920.2011.02556.x. [DOI] [PubMed] [Google Scholar]
- 59.John S.G., Ruggiero C.E., Hersman L.E., Tung C.S., Neu M.P. Siderophore mediated Plutonium accumulation by Microbacterium flavescens (JG-9) Environ. Sci. Technol. 2001;35:2942–2948. doi: 10.1021/es010590g. [DOI] [PubMed] [Google Scholar]
- 60.Barzanti R., Ozino F., Bozzicalupo M., Gabbrielli R., Galardi F., Gonnelli C., Mengoni A. Isolation and characterization of endophytic bacteria from Nickel hyperaccumulator plant Alyssum bertolonii. Microb. Ecol. 2007;53:306–316. doi: 10.1007/s00248-006-9164-3. [DOI] [PubMed] [Google Scholar]
- 61.Kuffner M., Puschenreiter M., Wieshammer G., Gorfer M., Sessitsch A. Rhizosphere bacteria affect growth and metal uptake of heavy metal accumulating willows. Plant Soil. 2008;304:35–44. doi: 10.1007/s11104-007-9517-9. [DOI] [Google Scholar]
- 62.Sheng X.F., Xia J.J., Jiang C.Y., He L.Y., Qian M. Characterization of heavy metal-resistant endophytic bacteria from rape (Brassica napus) roots and their potential in promoting the growth and lead accumulation of rape. Environ. Pollut. 2008;156:1164–1170. doi: 10.1016/j.envpol.2008.04.007. [DOI] [PubMed] [Google Scholar]
- 63.Dimkpa C.O., Svatos A., Merten D., Buchel G., Kothe E. Hydroxamate siderophores produced by Streptomyces acidiscabies E13 bind nickel and promote growth in cowpea (Vigna unguiculate L.) under nickel stress. Can. J. Microbiol. 2008;54:163–172. doi: 10.1139/W07-130. [DOI] [PubMed] [Google Scholar]
- 64.Dimkpa C.O., Svatos A., Dabrowska P., Schmidt A., Boland W., Kothe E. Involvement of siderophores in the reduction of metal-induced inhibition of auxin synthesis in Streptomyces spp. Chemosphere. 2008;74:19–25. doi: 10.1016/j.chemosphere.2008.09.079. [DOI] [PubMed] [Google Scholar]
- 65.Dimkpa C.O., Merten D., Svatos A., Buchel G., Kothe E. Siderophores mediate reduced and increased uptake of cadmium by Streptomyces tendae F4 and sunflower (Helianthus annuus), respectively. J. Appl. Microbiol. 2009;107:1687–1696. doi: 10.1111/j.1365-2672.2009.04355.x. [DOI] [PubMed] [Google Scholar]
- 66.Kuffner M., De Maria S., Puschenreiter M., Fallmann K., Wieshammer G., Gorfer M., Strauss J., Rivelli A.R., Sessitsch A. Culturable bacteria from Zn- and Cd-accumulating Salix caprea with differential effects on plant growth and heavy metal availability. J. Appl. Microbiol. 2010;108:1471–1484. doi: 10.1111/j.1365-2672.2010.04670.x. [DOI] [PubMed] [Google Scholar]
- 67.Sun L.N., Zhang Y.F., He L.Y., Chen Z.J., Qang Q.Y., Qian M., Sheng X.F. Genetic diversity and characterization of heavy metal-resistant-endophytic bacteria from two copper-tolerant species on copper mine wasteland. Biores. Technol. 2010;101:501–509. doi: 10.1016/j.biortech.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 68.Visioli G., D’Egidio S., Vamerali T., Mattarozzi M., Sanangelantoni A.M. Culturable endophytica bacteria enhance Ni translocation in the hyperaccumulator Noccaea caerulescens. Chemosphere. 2014;117:538–544. doi: 10.1016/j.chemosphere.2014.09.014. [DOI] [PubMed] [Google Scholar]
- 69.Schutze E., Ahmed E., Voit A., Klose M., Greyer M., Svatos A., Merten D., Roth M., Holmstrom S.J.M., Kothe E. Siderophores production by Streptomycetes—Stability and alteration of ferrihydroxamates in heavy metal-contaminated soil. Environ. Sci. Pollut. Res. 2015;22:19376–19383. doi: 10.1007/s11356-014-3842-3. [DOI] [PubMed] [Google Scholar]
- 70.El Baz S., Baz M., Barakate M., Hassani L., El Gharmali A., Imziln B. Resistance to and accumulation of heavy metals by Actinobacteria isolated from abandoned mining areas. Sci. World J. 2015;2015:761834. doi: 10.1155/2015/761834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zloch M., Thiem D., Gadzala-Kopciuch R., Hrynkiewicz K. Synthesis of siderophores by plant-associated metallotolerant bacteria under exposure to Cd2+ Chemosphere. 2016;156:312–325. doi: 10.1016/j.chemosphere.2016.04.130. [DOI] [PubMed] [Google Scholar]
- 72.Retamal-Morales G., Menhert M., Schwabe R., Tischer D., Zapata C., Chavez R. Detection of arsenic-binding siderophores in arsenic-tolerating Actinobacteria by a modified CAS assay. Ecotoxicol. Environ. Saf. 2018;157:176–181. doi: 10.1016/j.ecoenv.2018.03.087. [DOI] [PubMed] [Google Scholar]
- 73.Uranga C., Arroyo P.J., Duggan B.M., Gerwick W.H., Edlund A. Commensal oral Rothia mucilaginosa produces enterobactin—A metal chelating siderophore. mSystems. 2020;5:e001621-20. doi: 10.1128/mSystems.00161-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sessitsch A., Kuffner M., Kidd P., Vangronsveld J., Wenzel W.W., Fallmann K., Puschenreiter M. The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol. Biochem. 2013;60:182–194. doi: 10.1016/j.soilbio.2013.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhang L., Zeng Q., Liu X., Chen P., Guo X., Ma L.Z., Dong H., Huang Y. Iron reduction by diverse actinobacteria under oxic and pH-neutral conditions and the formation of secondary minerals. Chem. Geol. 2019;525:390–399. doi: 10.1016/j.chemgeo.2019.07.038. [DOI] [Google Scholar]
- 76.Rajkumar M., Ae N., Freitas H. Endophytic bacteria and their potential to enhance heavy metal phytoextraction. Chemosphere. 2009;77:153–160. doi: 10.1016/j.chemosphere.2009.06.047. [DOI] [PubMed] [Google Scholar]
- 77.Van der Lelie D., Corbisier P., Diels L., Gilis A., Lodewyckx C., Mergeay M., Taghavi S., Spelmans N., Vangronsveld J. The role of bacteria in the phytoremediation of heavy metals. In: Terry N., Banuelos G.S., editors. Phytoremediation of Contaminated Soil and Water. CRC Press; Boca Raton, FL, USA: 2000. pp. 265–281. [Google Scholar]
- 78.Djuric A., Gojgic-Cvijovic G., Jakovljevic D., Kekez B., Stefanovic Kojic J., Mattinen M.L., Haru I.E., Vrvic M.M., Beskoski V. Brachybacterium sp. CH-KOV3 isolated from an oil-polluted environment—A new producer of levan. Int. J. Biol. Macromol. 2017;104:311–321. doi: 10.1016/j.ijbiomac.2017.06.034. [DOI] [PubMed] [Google Scholar]
- 79.Presentato A., Piacenza E., Cappelletti M., Turner R.J. Interaction of Rhodococcus with metals and biotechnological applications. In: Alvarez H.M., editor. Biology of Rhodococcus. Springer Nature; Cham, Switzerland: 2019. pp. 333–357. [Google Scholar]
- 80.Harrison J.J., Turner R.J., Marques L.L.R., Ceri H. Biofilms: A new understanding of these microbial communities is driving a revolution that may transform the science of microbiology. Amer. Sci. 2005;93:508–515. doi: 10.1511/2005.56.508. [DOI] [Google Scholar]
- 81.Shuhong Y., Meiping Z., Hong Y., Han W., Shan X. Biosorption of Cu2+, Pb2+ and Cr6+ by a novel exopolysaccharide from Artrhobacter ps-5. Carbohydr. Polym. 2014;101:50–56. doi: 10.1016/j.carbpol.2013.09.021. [DOI] [PubMed] [Google Scholar]
- 82.Bankar A., Nagaraja G. Recent trends in biosorption of heavy metals by Actinobacteria. In: Singh B.P., Gupta K., Passari A.K., editors. New and Future Developments in Microbial Biotechnology and Bioengineering—Actinobacteria: Diversity and Biotechnological Applications. Elsevier; Amsterdam, The Netherlands: 2018. pp. 257–275. [Google Scholar]
- 83.Xiong Y.W., Ju X.Y., Li X.W., Gong Y., Xu M.J., Zhang C.M., Yuan B., Lv Z.P., Qin S. Fermentation conditions optimization, purification, and antioxidant activity of exopolysaccharides obtained from the plant growth-promoting endophytic actinobacterium Glutamicibacter halophytocola KLBMP 5180. Int. J. Biol. Macromol. 2019;153:1176–1185. doi: 10.1016/j.ijbiomac.2019.10.247. [DOI] [PubMed] [Google Scholar]
- 84.Neal A.L., Dublin S.N., Taylor J., Bates D.J., Burns J.L., Apkarian R., DiChristina T.J. Terminal electron acceptors influence the quantity and chemical composition of capsular exopolymers produced by anaerobically growing Shewanella spp. Biomacromolecules. 2007;8:166–174. doi: 10.1021/bm060826e. [DOI] [PubMed] [Google Scholar]
- 85.Solis M., Solis A., Perez H.I., Manjarrez N., Flores M. Microbial decolouration of azo dyes: A review. Process Biochem. 2012;47:1723–1748. doi: 10.1016/j.procbio.2012.08.014. [DOI] [Google Scholar]
- 86.Dobrowolski R., Szczes A., Czemierska M., Jarosz-Wikolazka A. Studies of cadmium(II), lead(II), nickel(II), cobalt(II) and chromium(IV) sorption on extracellular polymeric substances produced by Rhodococcus opacus and Rhodococcus rhodochrous. Biores. Technol. 2017;225:113–120. doi: 10.1016/j.biortech.2016.11.040. [DOI] [PubMed] [Google Scholar]
- 87.Sivaperumal P., Kamala K., Rajaram R. Adsorption of cesium ion by marine actinobacterium Nocardiopsis sp. 13H and their extracellular polymeric substances (EPS) role in bioremediation. Environ. Sci. Pollut. Res. 2018;25:4254–4267. doi: 10.1007/s11356-017-0818-0. [DOI] [PubMed] [Google Scholar]
- 88.Kamala K., Sivaperumal P., Thilagaraj R., Natarajan E. Bioremediation of Sr2+ ion radionuclide by using marine Streptomyces sp. CuOff24 extracellular polymeric substances. J. Chem. Technol. Biotechnol. 2020;95:893–903. doi: 10.1002/jctb.6018. [DOI] [Google Scholar]
- 89.Liu H., Fang H.H. Characterization of electrostatic binding sites of extracellular polymers by linear programming analysis of titration data. Biotechnol. Bioeng. 2002;30:806–811. doi: 10.1002/bit.10432. [DOI] [PubMed] [Google Scholar]
- 90.Wei X., Fang L., Cai P., Huang Q., Chen H., Liang W., Rong X. Influence of extracellular polymeric substances (EPS) on Cd adsorption by bacteria. Environ. Pollut. 2011;159:1369–1374. doi: 10.1016/j.envpol.2011.01.006. [DOI] [PubMed] [Google Scholar]
- 91.Roane T.M., Josephson K.L., Pepper I.L. Dual-Bioaugmentation Strategy to Enhance Remediation of Cocontaminated Soil. Appl. Environ. Microbiol. 2001;67:3208–3215. doi: 10.1128/AEM.67.7.3208-3215.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Albarracin V.H., Winik B., Kothe E., Amoroso M.J., Abate C.M. Copper bioaccumulation by the actinobacterium Amycolatopsis sp. AB0. J. Basic Microbiol. 2008;48:323–330. doi: 10.1002/jobm.200700360. [DOI] [PubMed] [Google Scholar]
- 93.Maldonado J., Diestra E., Huang L., Domenech A.M., Villagrasa E., Puyen Z.M., Duran R., Esteve I., Solé A. Isolation and identification of a bacterium with high tolerance to lead and copper from a marine microbial mat in Spain. Ann. Microbiol. 2010;60:113–120. doi: 10.1007/s13213-010-0019-2. [DOI] [Google Scholar]
- 94.Puyen Z.M., Villagrasa E., Maldonado J., Diestra E., Esteve I., Sole A. Biosorption of lead and copper by heavy-metal tolerant Micrococcus luteus DE2008. Biores. Technol. 2012;126:233–237. doi: 10.1016/j.biortech.2012.09.036. [DOI] [PubMed] [Google Scholar]
- 95.Quintelas C., da Silva V.B., Silva B., Figueiredo H., Tavares T. Optimization of production of extracellular polymeric substances by Arthrobacter viscosus and their interaction with a 13X zeoilite for the biosorption of Cr(IV) Environ. Technol. 2011;32:1541–1549. doi: 10.1080/09593330.2010.543930. [DOI] [PubMed] [Google Scholar]
- 96.More T.T., Yan S., John R.P., Tyagi R.D., Surampalli R.Y. Biochemical diversity of the bacterial strains and their biopolymer producing capabilities in wastewater sludge. Biores. Technol. 2012;121:304–311. doi: 10.1016/j.biortech.2012.06.103. [DOI] [PubMed] [Google Scholar]
- 97.Vijay V., Vandana K.B., Kumar R.M., Prabagaran S.R. Genetic analysis of arsenic metabolism in Micrococcus luteus BPB1, isolated from the Bengal basin. Ann. Microbiol. 2017;67:79–89. doi: 10.1007/s13213-016-1239-x. [DOI] [Google Scholar]
- 98.Furnholm T., Rehan M., Wishart J., Tisa L.S. Pb2+ tolerance by Frankia sp. strain EAN1pec involves surface-binding. Microbiology. 2017;163:472–487. doi: 10.1099/mic.0.000439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Mulik A., Bhadekar R. Extracellular polymeric substance (EPS) from Kocuria sp. BRI 36: A key component in heavy metal resistance. Int. J. Pharm. Pharm. Sci. 2018;10:50–54. doi: 10.22159/ijpps.2018v10i5.23535. [DOI] [Google Scholar]
- 100.Sivaperumal P., Kamala K., Rajaram R. Biosorption of long half-life radionuclide of Strontium ion (Sr+) by marine actinobacterium Nocardiopsis sp. 13H. Geomicrobiol. J. 2018;35:300–310. doi: 10.1080/01490451.2017.1350891. [DOI] [Google Scholar]
- 101.Pal A., Paul A.K. Microbial extracellular polymeric substances central elements in heavy metal bioremediation. Indian J. Microbiol. 2008;48:49–64. doi: 10.1007/s12088-008-0006-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Han R.P., Li H.K., Li Y.H., Zhang J.H., Xiao H.J., Shi J. Biosorption of copper and lead ions by waste beer yeast. J. Hazard. Mater. 2006;137:1569–1576. doi: 10.1016/j.jhazmat.2006.04.045. [DOI] [PubMed] [Google Scholar]
- 103.Lamelas C., Benedetti M., Wilkinson K.J., Slaveykova V.I. Characterization of H+ and Cd(II) binding properties of the bacterial exopolysaccharides. Chemosphere. 2006;65:1362–1370. doi: 10.1016/j.chemosphere.2006.04.021. [DOI] [PubMed] [Google Scholar]
- 104.Veglio F., Beolchini F. Removal of metals by biosorption: A review. Hydrometallurgy. 1997;44:301–306. doi: 10.1016/S0304-386X(96)00059-X. [DOI] [Google Scholar]
- 105.Avery S.V. Microbial interactions with caesium—Implications for biotechnology. J. Chem. Technol. Biotechnol. 1995;62:3–16. doi: 10.1002/jctb.280620102. [DOI] [Google Scholar]
- 106.Brooks A.N., Turkarslan S., Beer K.D., Lo F.Y., Baliga N.S. Adaptation of cells to new environments. Wiley Interdiscip. Rev. Syst. Biol. Med. 2011;3:544–561. doi: 10.1002/wsbm.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Mowll J.L., Gadd G.M. Cadmium uptake by Aureobasidium pullulans. J. Gen. Microbiol. 1984;130:279–284. doi: 10.1099/00221287-130-2-279. [DOI] [Google Scholar]
- 108.Konings W.N., Albers S., Koning S., Driessen A.J.M. The cell membrane plays a crucial role in survival of bacteria and archaea in extreme environments. Antonie Van Leeuwenhoek. 2002;81:61–72. doi: 10.1023/A:1020573408652. [DOI] [PubMed] [Google Scholar]
- 109.Ehrlich H.L. Microbes and metals. Appl. Microbiol. Biotechnol. 1997;48:687–692. doi: 10.1007/s002530051116. [DOI] [Google Scholar]
- 110.Valls M., de Lorenzo V. Exploiting the genetic and biochemical capacities of bacteria for the remediation of heavy metal pollution. FEMS Microbiol. Rev. 2002;4:327–338. doi: 10.1016/S0168-6445(02)00114-6. [DOI] [PubMed] [Google Scholar]
- 111.Gadd G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology. 2010;156:609–643. doi: 10.1099/mic.0.037143-0. [DOI] [PubMed] [Google Scholar]
- 112.Pau R.N., Klipp W., Leimkühler S. Molybdenum transport, processing and gene regulation. In: Winkelmann G., Carrano C.J., editors. Transition Metals in Microbial Metabolism. Harwood; Amsterdam, The Netherlands: 1997. pp. 217–234. [Google Scholar]
- 113.Taylor D.E. Bacterial tellurite resistance. Trends Microbiol. 1999;7:111–115. doi: 10.1016/S0966-842X(99)01454-7. [DOI] [PubMed] [Google Scholar]
- 114.Natasha S.M., Niazi N.K., Khalid S., Murtaza B., Bibi I., Rashid I.M. A critical review of selenium biogeochemical behavior in soil-plant system with an inference to human health. Environ. Pollut. 2018;234:915–934. doi: 10.1016/j.envpol.2017.12.019. [DOI] [PubMed] [Google Scholar]
- 115.Presentato A., Turner R.J., Vásquez C.C., Yurkov V., Zannoni D. Tellurite-dependent blackening of bacteria emerges from the dark ages. Environ. Chem. 2019;16:266–288. doi: 10.1071/EN18238. [DOI] [Google Scholar]
- 116.Piacenza E., Presentato A., Zonaro E., Lemire J.A., Demeter M., Vallini G., Turner R.J., Lampis S. Antimicrobial activity of biogenically produced spherical Se nanomaterials embedded in organic material against Pseudomonas aeruginosa and Staphylococcus aureus strains on hydroxyapatite-coated surfaces. Microb. Biotechnol. 2017;10:804–818. doi: 10.1111/1751-7915.12700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Springer S.E., Huber R.E. Sulfate and selenate uptake and transport in wild and in two selenate-tolerant strains of Escherichia coli K12. Arch. Biochem. Biophys. 1973;156:595–603. doi: 10.1016/0003-9861(73)90310-X. [DOI] [PubMed] [Google Scholar]
- 118.Brown T.A., Shrift A. Assimilation of selenate and selenite by Salmonella thyphimurium. Can. J. Microbiol. 1980;26:671–675. doi: 10.1139/m80-117. [DOI] [PubMed] [Google Scholar]
- 119.Bryant R.D., Laishley E.J. Evidence for proton motive force dependent transport of selenite by Clostridium paesteurianum. Can. J. Microbiol. 1989;35:481–486. doi: 10.1139/m89-074. [DOI] [PubMed] [Google Scholar]
- 120.Bebien M., Chauvin J.P., Adriano J.M., Grosse S., Vermeglio A. Effect of selenite on growth and protein synthesis in the phototrophic bacterium Rhodobacter sphaeroides. Appl. Environ. Microbiol. 2001;67:4440–4447. doi: 10.1128/AEM.67.10.4440-4447.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Tomas J.M., Kay W.W. Tellurite susceptibility and non-plasmid mediated resistance in Escherichia coli. Antimicrob. Agents Chemother. 1986;30:127–131. doi: 10.1128/AAC.30.1.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Van Veen H.W. Phosphate transporter in prokaryotes: Molecules, mediators and mechanisms. Antoine Van Leeuwenhoek. 1997;72:299–315. doi: 10.1023/A:1000530927928. [DOI] [PubMed] [Google Scholar]
- 123.Harris R.M., Webb D.C., Howitt S.M., Cox G.B. Characterization of PitA and PitB from Escherichia coli. J. Bacteriol. 2001;183:5008–5014. doi: 10.1128/JB.183.17.5008-5014.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Turner M.S., Tan Y.P., Giffard P.M. Inactivation of an iron transporter in Lactococcus lactis results in resistance to tellurite and oxidative stress. Appl. Environ. Microbiol. 2007;73:6144–6149. doi: 10.1128/AEM.00413-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zannoni D., Borsetti F., Harrison J.J., Turner R.J. The bacterial response to the chalcogen metalloids Se and Te. Adv. Microb. Physiol. 2008;53:1–71. doi: 10.1016/S0065-2911(07)53001-8. [DOI] [PubMed] [Google Scholar]
- 126.Borghese R., Zannoni D. Acetate permease (ActP) is responsible for tellurite (TeO32−) uptake and resistance in cells of the facultative phototroph Rhodobacter capsulatus. Appl. Environ. Microbiol. 2010;76:942–944. doi: 10.1128/AEM.02765-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Abbas A., Edwards C. Effects of metals on a range of Streptomyces species. Appl. Environ. Microbiol. 1990;55:2030–2035. doi: 10.1128/AEM.55.8.2030-2035.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Amoroso M.J., Castro G.R., Carlino F.J., Romero N.C., Hill R.T., Oliver G. Screening of heavy metal-tolerant actinomycetes isolated from the Salí River. J. Gen. Appl. Microbiol. 1998;44:129–132. doi: 10.2323/jgam.44.129. [DOI] [PubMed] [Google Scholar]
- 129.Smith R.L., Maguire M.E. Microbial magnesium transport: Unusual transporters searching for identity. Mol. Microbiol. 1998;28:217–226. doi: 10.1046/j.1365-2958.1998.00810.x. [DOI] [PubMed] [Google Scholar]
- 130.Fu C., Javedan S., Moshiri F., Maier R.J. Bacterial genes involved in incorporation of nickel into a hydrogenase enzyme. Proc. Natl. Acad. Sci. USA. 1994;91:5099–5103. doi: 10.1073/pnas.91.11.5099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Eitinger T., Friedrich B. Microbial nickel transport and incorporation into hydrogenases. In: Winkelmann G., Carrano C.J., editors. Transition Metals in Microbial Metabolism. Harwood; Amsterdam, The Netherlands: 1997. pp. 235–256. [Google Scholar]
- 132.Fulkerson J.F., Jr., Garner R.M., Mobley H.L.T. Conserved motifs and residues in the NixA protein of Helicobacter pylori are critical for the high affinity transport of nickel ions. J. Biol. Chem. 1998;273:235–241. doi: 10.1074/jbc.273.1.235. [DOI] [PubMed] [Google Scholar]
- 133.Cole S.T., Broch R., Parkhill J., Garnier T., Churcher C., Harris D., Gordon S.V., Eiglmeir K., Gas S., Barry C.E., III, et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature. 1998;393:537–544. doi: 10.1038/31159. [DOI] [PubMed] [Google Scholar]
- 134.Kim E.J., Chung H.J., Suh B., Hah Y.C., Roe J.H. Transcriptional and post-transcriptional regulation by nickel of sodN gene encoding nickel-containing superoxide dismutase from Streptomyces coelicolor Müller. Mol. Microbiol. 1998;27:187–195. doi: 10.1046/j.1365-2958.1998.00674.x. [DOI] [PubMed] [Google Scholar]
- 135.Ahn B.E., Cha J., Lee E.J., Han A.R., Thompson C.J., Roe J.H. Nur, a nickel-responsive regulator of the Fur family, regulates superoxide dismutases and nickel transport in Streptomyces coelicolor. Mol. Microbiol. 2006;59:1848–1858. doi: 10.1111/j.1365-2958.2006.05065.x. [DOI] [PubMed] [Google Scholar]
- 136.Lu M., Jiang Y.L., Wang S., Jin H., Zhang R.G., Virolle M.J., Chen Y., Zhou C.Z. Streptomyces coelicolor SCO4226 Is a Nickel Binding Protein. PLoS ONE. 2014;9:e109660. doi: 10.1371/journal.pone.0109660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Komeda H., Kobayashi M., Shimizu S. A novel transporter involved in cobalt uptake. Proc. Natl. Acad. Sci. USA. 1997;94:36–41. doi: 10.1073/pnas.94.1.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Pogorelova T.E., Ryabchenko L.E., Sunzow N.I., Yanenko A.S. Cobalt-dependent transcription of nitrile hydratase gene in Rhodococcus rhodochrous M8. FEMS Microbiol. Lett. 1996;144:191–195. doi: 10.1111/j.1574-6968.1996.tb08529.x. [DOI] [Google Scholar]
- 139.Wolfram L., Friedrich B., Eitinger T. The Alcaligenes eutrophus protein HoxN mediates nickel transport in Escherichia coli. J. Bacteriol. 1995;177:1840–1843. doi: 10.1128/JB.177.7.1840-1843.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Degen O., Kobayashi M., Shimizu S., Eitinger T. Selective transport of divalent cations by transition metal permeases: The Alcaligenes eutrophus HoxN and the Rhodococcus rhodochrous NhlF. Arch. Microbiol. 1999;171:139–145. doi: 10.1007/s002030050691. [DOI] [PubMed] [Google Scholar]
- 141.Amoroso M.J., Schubert D., Mitscherlich P., Schumann P., Kothe E. Evidence for high affinity nickel transporter genes in heavy metal resistant Streptomyces spec. J. Basic Microbiol. 2000;40:295–301. doi: 10.1002/1521-4028(200012)40:5/6<295::AID-JOBM295>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 142.Hendricks J.K., Mobley H.L. Helicobacter pylori ABC transporter: Effect of allelic exchange mutagenesis on urease activity. J. Bacteriol. 1997;179:5892–5902. doi: 10.1128/JB.179.18.5892-5902.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Locatelli F.M., Goo K.S., Ulanova D. Effects of trace metal ions on secondary metabolism and the morphological development of Streptomycetes. Metallomics. 2016;8:469–480. doi: 10.1039/C5MT00324E. [DOI] [PubMed] [Google Scholar]
- 144.Aagaard A., Brzezinski P. Zinc ions inhibit oxidation of cytochrome c oxidase by oxygen. FEBS Lett. 2001;494:157–160. doi: 10.1016/S0014-5793(01)02299-2. [DOI] [PubMed] [Google Scholar]
- 145.Mills D.A., Schmidt B., Hiser C., Westley E., Ferguson-Miller S. Membrane potential-controlled inhibition of cytochrome c oxidase by zinc. J. Biol. Chem. 2002;277:14894–14901. doi: 10.1074/jbc.M111922200. [DOI] [PubMed] [Google Scholar]
- 146.Mangold S., Potrykus J., Björn E., Lövgren L., Dopson M. Extreme zinc tolerance in acidophilic microorganisms from the bacterial and archaeal domains. Extremophiles. 2013;17:75–85. doi: 10.1007/s00792-012-0495-3. [DOI] [PubMed] [Google Scholar]
- 147.Shin J.H., Oh A.Y., Kim A.J., Roe J.H. The Zinc-Responsive Regulator Zur Controls a Zinc Uptake System and Some Ribosomal Proteins in Streptomyces coelicolor A3(2) J. Bacteriol. 2007;189:4070–4077. doi: 10.1128/JB.01851-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Choi S.H., Lee K.L., Shin J.H., Cho Y.B., Cha S.S., Roe J.H. Zinc-dependent regulation of zinc import and export genes by Zur. Nat. Commun. 2017;8:15812. doi: 10.1038/ncomms15812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Sedlakova-Kadukova J., Kopcakova A., Gresakova L., Godany A., Pristas P. Bioaccumulation and biosorption of zinc by a novel Streptomyces K11 strain isolated from highly alkaline aluminium brown mud disposal site. Ecotoxicol. Environ. Saf. 2019;167:204–211. doi: 10.1016/j.ecoenv.2018.09.123. [DOI] [PubMed] [Google Scholar]
- 150.Gadd G.M. Microbial influence on metal mobility and application for bioremediation. Geoderma. 2004;122:109–119. doi: 10.1016/j.geoderma.2004.01.002. [DOI] [Google Scholar]
- 151.Malik A. Metal bioremediation through growing cells. Environ. Int. 2004;30:261–278. doi: 10.1016/j.envint.2003.08.001. [DOI] [PubMed] [Google Scholar]
- 152.Polti M.A., Amoroso M.J., Abate C.M. Chromium(VI) resistance and removal by actinomycete strains isolated from sediments. Chemosphere. 2007;67:660–667. doi: 10.1016/j.chemosphere.2006.11.008. [DOI] [PubMed] [Google Scholar]
- 153.Polti M.A., Amoroso M.J., Abaste C.M. Intracellular chromium accumulation by Streptomyces sp. MC1. Water Air Soil Pollut. 2011;214:49–57. doi: 10.1007/s11270-010-0401-5. [DOI] [Google Scholar]
- 154.Tomioka N., Uchiyama H., Yagi O. Cesium accumulation and growth characteristics of Rhodococcus erythropolis CS98 and Rhodococcus sp. strain CS402. Appl. Environ. Microbiol. 1994;60:2227–2231. doi: 10.1128/AEM.60.7.2227-2231.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Avery S.V., Codd G.A., Gadd G.M. Caesium accumulation and interactions with other monovalent cations in the cyanobacterium Synechocystis PCC 6803. J. Gen. Microbiol. 1991;137:405–413. doi: 10.1099/00221287-137-2-405. [DOI] [Google Scholar]
- 156.Avery S.V., Codd G.A., Gadd G.M. Interactions of cyanobacteria and microalgae with caesium. In: Vernet J.P., editor. Impact of Heavy Metals on the Environment. Elsevier; Amsterdam, The Netherlands: 1992. pp. 133–182. [Google Scholar]
- 157.Woolfolk C.A., Whiteley H.R. Reduction of inorganic compounds with molecular hydrogen by Micrococcus lactilyticus. I. Stoichiometry with compounds of arsenic, selenium, tellurium, transition and other elements. J. Bacteriol. 1962;84:647–658. doi: 10.1128/JB.84.4.647-658.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nakahara H., Schottel J.L., Yamada T., Miyakawa Y., Asakawa M., Harville J., Silver S. Mercuric Reductase Enzymes from Streptomyces Species and Group B Streptococcus. J. Gen. Microbiol. 1985;131:1053–1059. doi: 10.1099/00221287-131-5-1053. [DOI] [PubMed] [Google Scholar]
- 159.Das S., Chandra A.L. Chromate reduction in Streptomyces. Experientia. 1990;46:731–733. doi: 10.1007/BF01939949. [DOI] [PubMed] [Google Scholar]
- 160.Pattanapipitpaisal P., Brown N.L., Macaskie L.E. Chromate reduction by Microbacterium liquefaciens immobilised in polyvinyl alcohol. Biotechnol. Lett. 2001;23:61–62. doi: 10.1023/A:1026750810580. [DOI] [Google Scholar]
- 161.Laxman R.S., More S. Reduction of hexavalent chromium by Streptomyces griseus. Miner. Eng. 2002;15:831–837. doi: 10.1016/S0892-6875(02)00128-0. [DOI] [Google Scholar]
- 162.Ahmad A., Senapati S., Islam Khan M., Kumar R., Sastry M. Extracellular Biosynthesis of Monodisperse Gold Nanoparticles by a Novel Extremophilic Actinomycete, Thermomonospora sp. Langmuir. 2003;19:3550–3553. doi: 10.1021/la026772l. [DOI] [Google Scholar]
- 163.Ahmad A., Senapati S., Islam Khan M., Kumar R., Ramani R., Srinivas V., Sastry M. Intracellular synthesis of gold nanoparticles by a novel alkalotolerant actinomycete, Rhodococcus species. Nanotechnology. 2003;14:824–828. doi: 10.1088/0957-4484/14/7/323. [DOI] [Google Scholar]
- 164.Desjardin V., Bayard R., Lejeune P., Gourdon R. Utilisation of supernatants of pure cultures of Streptomyces thermocarboxidus NH50 to reduce Chromium toxicity and mobility in contaminated soils. Water Air Soil Pollut. 2003;3:153–160. doi: 10.1023/A:1023965314747. [DOI] [Google Scholar]
- 165.Camargo F., Bento F., Okeke B., Frankenberger W. Hexavalent chromium reduction by an actinomycete, Arthrobacter crystallopoietes ES 32. Biol. Trace Elem. Res. 2004;97:183–194. doi: 10.1385/BTER:97:2:183. [DOI] [PubMed] [Google Scholar]
- 166.Zhang H., Li Q., Lu Y., Sun D., Lin X., Deng X., He N., Zheng S. Biosorption and bioreduction of diamine silver complex by Corynebacterium. J. Chem. Technol. Biotechnol. 2005;80:285–290. doi: 10.1002/jctb.1191. [DOI] [Google Scholar]
- 167.Bharde A., Wani A., Shouche Y., Joy P.A., Prasad B.L.V., Sastry M. Bacterial Aerobic Synthesis of Nanocrystalline Magnetite. J. Am. Chem. Soc. 2005;127:9326–9327. doi: 10.1021/ja0508469. [DOI] [PubMed] [Google Scholar]
- 168.Mateos L.M., Ordonez E., Letek M., Gil J.A. Corynebacterium glutamicum as a model bacterium for the bioremediation of arsenic. Int. Microbiol. 2006;9:207–215. [PubMed] [Google Scholar]
- 169.Bharde A., Kulkarni A., Rao M., Prabhune A., Sastry M. Bacterial Enzyme Mediated Biosynthesis of Gold Nanoparticles. J. Nanosci. Nanotechnol. 2007;7:4369–4377. doi: 10.1166/jnn.2007.891. [DOI] [PubMed] [Google Scholar]
- 170.Bharde A., Parikh R.Y., Baidakova M., Jouen S., Hannoyer B., Enoki T., Prasad B.L.V., Shouche Y.S., Ogale S., Sastry M. Bacteria-Mediated Precursor-Dependent Biosynthesis of Superparamagnetic Iron Oxide and Iron Sulfide Nanoparticles. Langmuir. 2008;24:5787–5794. doi: 10.1021/la704019p. [DOI] [PubMed] [Google Scholar]
- 171.Mabrouk M.E.M. Statistical optimization of medium components for chromate reduction by halophilic Streptomyces sp. MS-2. Afr. J. Microbiol. Res. 2008;2:103–110. [Google Scholar]
- 172.Kumar U., Shete A., Harle A.S., Kasyutich O., Schwarzacher W., Pundle A., Poddar P. Extracellular Bacterial Synthesis of Protein-Functionalized Ferromagnetic Co3O4 Nanocrystals and Imaging of Self-Organization of Bacterial Cells under Stress after Exposure to Metal Ions. Chem. Mater. 2008;20:1484–1491. doi: 10.1021/cm702727x. [DOI] [Google Scholar]
- 173.Polti M.A., Garcia R.O., Amoroso M.J., Abate C.M. Bioremediation of chromium(VI) contaminated soil by Streptomyces sp. MC1. J. Basic Microbiol. 2009;49:285–292. doi: 10.1002/jobm.200800239. [DOI] [PubMed] [Google Scholar]
- 174.Polti M.A., Amoroso M.J., Abate C.M. Chromate reductase activity in Streptomyces sp. MC1. J. Gen. Appl. Microbiol. 2010;56:11–18. doi: 10.2323/jgam.56.11. [DOI] [PubMed] [Google Scholar]
- 175.Elangovan R., Philip L., Chandraraj K. Hexavalent chromium reduction by free and immobilized cell-free extract of Arthrobacter rhombi-RE. Appl. Biochem. Biotechnol. 2010;160:81. doi: 10.1007/s12010-008-8515-6. [DOI] [PubMed] [Google Scholar]
- 176.Sadhasivam S., Shanmugam P., Yun K.S. Biosynthesis of silver nanoparticles by Streptomyces hygroscopicus and antimicrobial activity against medically important pathogenic microorganisms. Colloids Surf. B Biointerfaces. 2010;81:358–362. doi: 10.1016/j.colsurfb.2010.07.036. [DOI] [PubMed] [Google Scholar]
- 177.Sneha K., Sathishkumar M., Mao J., Kwak I.S., Yun Y.S. Corynebacterium glutamicum—Mediated crystallization of silver ions through sorption and reduction processes. Chem. Eng. J. 2010;162:989–996. doi: 10.1016/j.cej.2010.07.006. [DOI] [Google Scholar]
- 178.Bafana A., Krishnamurthi K., Patil M., Chakrabarti T. Heavy metal resistance in Arthrobacter ramosus strain G2 isolated from mercuric salt-contaminated soil. J. Hazard. Mater. 2010;177:481–486. doi: 10.1016/j.jhazmat.2009.12.058. [DOI] [PubMed] [Google Scholar]
- 179.Patra R.C., Malik S., Beer M., Megharaj M., Naidu R. Molecular characterization of chromium (VI) reducing potential in gram positive bacteria isolated from contaminated sites. Soil Biol. Biochem. 2010;42:1857–1863. doi: 10.1016/j.soilbio.2010.07.005. [DOI] [Google Scholar]
- 180.Sugiyama T., Sugito H., Mamiya K., Suzuki Y., Ando K., Ohnuki T. Hexavalent chromium reduction by an actinobacterium Flexivirga alba ST13 in the family Dermacoccaceae. J. Biosci. Bioeng. 2012;113:367–371. doi: 10.1016/j.jbiosc.2011.11.009. [DOI] [PubMed] [Google Scholar]
- 181.Alani F., Moo-Young M., Anderson W. Biosynthesis of silver nanoparticles by a new strain of Streptomyces sp. compared with Aspergillus fumigatus. World J. Microbiol. Biotechnol. 2012;28:1081–1086. doi: 10.1007/s11274-011-0906-0. [DOI] [PubMed] [Google Scholar]
- 182.Oza G., Pandey S., Gupta A., Kesarkar R., Sharon M. Biosynthetic Reduction of Gold Ions to Gold Nanoparticles by Nocardia farcinica. J. Microbiol. Biotech. Res. 2012;2:511–515. [Google Scholar]
- 183.Dey S., Paul A.K. Optimization of cultural conditions for growth associated chromate reduction by Arthrobacter sp. SUK 1201 isolated from chromite mine overburden. J. Hazard. Mater. 2012;213–214:200–206. doi: 10.1016/j.jhazmat.2012.01.078. [DOI] [PubMed] [Google Scholar]
- 184.Dey S., Paul A.K. In vitro bioreduction of hexavalent Chromium by viable whole cells of Arthrobacter sp. SUK 1201. J. Microbiol. Biotech. Food Sci. 2014;4:19–23. doi: 10.15414/jmbfs.2014.4.1.19-23. [DOI] [Google Scholar]
- 185.Dey S., Paul A.K. Optimization of Chromate Reduction by Whole Cells of Arthrobacter sp. SUK 1205 Isolated from Metalliferous Chromite Mine Environment. Geomaterials. 2012;2:73–81. doi: 10.4236/gm.2012.24012. [DOI] [Google Scholar]
- 186.Javaid M., Sultan S. Plant growth promotion traits and Cr (VI) reduction potentials of Cr (VI) resistant Streptomyces strains. J. Basic Microbiol. 2013;53:420–428. doi: 10.1002/jobm.201200032. [DOI] [PubMed] [Google Scholar]
- 187.Otari S.V., Patil R.M., Nadaf N.H., Gosh S.J., Pawar S.H. Green biosynthesis of silver nanoparticles from an actinobacteria Rhodococcus sp. Mat. Lett. 2012;72:92–94. doi: 10.1016/j.matlet.2011.12.109. [DOI] [Google Scholar]
- 188.Otari S.V., Patil R.M., Gosh S.J., Thorat N.D., Pawar S.H. Intracellular synthesis of silver nanoparticle by actinobacteria and its antimicrobial activity. Spectrochim. Acta A Mol. Biomol. Spectr. 2015;136:1175–1180. doi: 10.1016/j.saa.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 189.Chauhan R., Kumar A., Abraham J. A Biological Approach to the Synthesis of Silver Nanoparticles with Streptomyces sp JAR1 and its Antimicrobial Activity. Sci. Pharm. 2013;81:607–621. doi: 10.3797/scipharm.1302-02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Arunkumar P., Thanalakshmi M., Kumar P., Premkumar K. Micrococcus luteus mediated dual mode synthesis of gold nanoparticles: Involvement of extracellular α-amylase and cell wall teichuronic acid. Colloid Surf. B Biointerfaces. 2013;103:517–522. doi: 10.1016/j.colsurfb.2012.10.051. [DOI] [PubMed] [Google Scholar]
- 191.Gopal J.V., Thenmozhi M., Kannabiran K., Rajakumar G., Velayutham K., Rahuman A.A. Actinobacteria mediated synthesis of gold nanoparticles using Streptomyces sp. VITDDK3 and its antifungal activity. Mat. Lett. 2013;93:360–362. doi: 10.1016/j.matlet.2012.11.125. [DOI] [Google Scholar]
- 192.Gren T., Ostash B., Hrubskyy Y., Tistechok S., Fedorenko V. Influence of transition metals on Streptomyces coelicolor and S. sioyaensis and generation of chromate-reducing mutants. Folia Microbiologica. 2014;59:147–153. doi: 10.1007/s12223-013-0277-z. [DOI] [PubMed] [Google Scholar]
- 193.Kundu D., Hazra C., Chatterjee A., Chaudhari A., Mishra S. Extracellular biosynthesis of zinc oxide nanoparticles using Rhodococcus pyridinivorans NT2: Multifunctional textile finishing, biosafety evaluation and in vitro drug delivery in colon carcinoma. J. Photochem. Photobiol. B. 2014;140:194–204. doi: 10.1016/j.jphotobiol.2014.08.001. [DOI] [PubMed] [Google Scholar]
- 194.Polti M.A., Aparicio J.D., Benimeli C.S., Amoroso M.J. Simultaneous bioremediation of Cr(VI) and lindane in soil by actinobacteria. Int. Biodet. Biodegr. 2014;88:48–55. doi: 10.1016/j.ibiod.2013.12.004. [DOI] [Google Scholar]
- 195.Kaur H., Dolma K., Kaur N., Malhotra A., Kumar N., Dixit P., Sharma D., Mayilraj S., Choudhury A.R. Marine Microbe as Nano-factories for Copper Biomineralization. Biotechnol. Bioproc. Eng. 2015;20:51–57. doi: 10.1007/s12257-014-0432-7. [DOI] [Google Scholar]
- 196.Ramya S., Shanmugasundaram T., Balagurunathan R. Biomedical potential of actinobacterially synthesized selenium nanoparticles with special reference to anti-biofilm, anti-oxidant, wound healing, cytotoxic and anti-viral activities. J. Trace Elem. Med. Biol. 2015;32:30–39. doi: 10.1016/j.jtemb.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 197.Jones R.M., Johnson D.B. Acidithrix ferrooxidans gen. nov., sp. nov.; a filamentous and obligately heterotrophic, acidophilic member of the Actinobacteria that catalyzes dissimilatory oxido-reduction of iron. Res. Microbiol. 2015;166:111–120. doi: 10.1016/j.resmic.2015.01.003. [DOI] [PubMed] [Google Scholar]
- 198.Ahmad M.S., Yasser M.S., Sholkamy E.N., Ali M.A., Mehanni M.M. Anticancer activity of biostabilized selenium nanorods synthesized by Streptomyces bikiniensis strain Ess_amA-1. Int. J. Nanomed. 2015;10:3389–3401. doi: 10.2147/IJN.S82707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Liu H., Huang J., Zhang S., Xu B., Wang G. Chromate interaction with chromate reducing actinobacterium Intrasporangium chromatireducens Q5-1. Geomicrobiol. J. 2015;32:616–623. doi: 10.1080/01490451.2014.971200. [DOI] [Google Scholar]
- 200.Abd-Elnaby H.M., Abo-Elala G.M., Abdel-Raouf U.M., Hamed M.Z. Antibacterial and anticancer activity of extracellular synthesized silver nanoparticles from marine Streptomyces rochei MHM13. Egypt. J. Aquat. Res. 2016;42:301–312. doi: 10.1016/j.ejar.2016.05.004. [DOI] [Google Scholar]
- 201.Dey S., Paul A.K. Assessment of heavy metal tolerance and hexavalent chromium reducing potential of Corynebacterium paurometabolum SKPD 1204 isolated from chromite mine seepage. AIMS Bioeng. 2016;3:337–351. doi: 10.3934/bioeng.2016.3.337. [DOI] [Google Scholar]
- 202.Bennur T., Khan Z., Kshirsagar R., Javdekar V., Zinjarde S. Biogenic gold nanoparticles from the Actinomycete Gordonia amarae: Application in rapid sensing of copper ions. Sens. Actuat. B. 2016;233:684–690. doi: 10.1016/j.snb.2016.04.022. [DOI] [Google Scholar]
- 203.Tan Y., Yao R., Wang R., Wang D., Wang G., Zheng S. Reduction of selenite to Se(0) nanoparticles by filamentous bacterium Streptomyces sp. ES2-5 isolated from a selenium mining soil. Microb. Cell. Fact. 2016;15:157. doi: 10.1186/s12934-016-0554-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Presentato A., Piacenza E., Anikovskiy M., Cappelletti M., Zannoni D., Turner R.J. Rhodococcus aetherivorans BCP1 as cell factory for the production of intracellular tellurium nanorods under aerobic conditions. Microb. Cell. Fact. 2016;15:204. doi: 10.1186/s12934-016-0602-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Składanowski M., Wypij M., Laskowski D., Golinksa P., Dahm H., Rai M. Silver and gold nanoparticles synthesized from Streptomyces sp. isolated from acid forest soil with special reference to its antibacterial activity against pathogens. J. Clust. Sci. 2017;28:59–79. doi: 10.1007/s10876-016-1043-6. [DOI] [Google Scholar]
- 206.Wypij M., Golinska P., Dahm H., Rai M. Actinobacterial-mediated synthesis of silver nanoparticles and their activity against pathogenic bacteria. IET Nanobiotechnol. 2017;11:336–342. doi: 10.1049/iet-nbt.2016.0112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Silva-Vinhote N.M., Duran Caballero N.E., de Amorim Silva T., Veras Quelemes P., de Araujo A.R., de Moraes A.C.M., dos Santos Camara A.L., Longo J.P.F.O., Azevedo R.B., da Silva D.A., et al. Extracellular biogenic synthesis of silver nanoparticles by Actinomycetes from amazonic biome and its antimicrobial efficiency. Afr. J. Biotechnol. 2017;16:2072–2082. doi: 10.5897/AJB2017.16148. [DOI] [Google Scholar]
- 208.Presentato A., Piacenza E., Anikovskiy M., Cappelletti M., Zannoni D., Turner R.J. Biosynthesis of selenium-nanoparticles and -nanorods as a product of selenite bioconversion by the aerobic bacterium Rhodococcus aetherivorans BCP1. New Biotechnol. 2018;41:1–8. doi: 10.1016/j.nbt.2017.11.002. [DOI] [PubMed] [Google Scholar]
- 209.Presentato A., Piacenza E., Darbandi A., Anikovskiy M., Cappelletti M., Zannoni D., Turner R.J. Assembly, growth and conductive properties of tellurium nanorods produced by Rhodococcus aetherivorans BCP1. Sci. Rep. 2018;8:3923. doi: 10.1038/s41598-018-22320-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Aparicio J.D., Saez J.M., Raimondo E.E., Benimeli C.S., Polti M.A. Comparative study of single and mixed cultures of actinobacteria for the bioremediation of co-contaminated matrices. J. Environ. Chem. Eng. 2018;6:2310–2318. doi: 10.1016/j.jece.2018.03.030. [DOI] [Google Scholar]
- 211.Al-Dhabi N.A., Ghilan A.K.M., Arasu M.V., Duraipandiyan V. Green biosynthesis of silver nanoparticles produced from marine Streptomyces sp. Al-Dhabi-89 and their potential applications against wound infection and drug resistant clinical pathogens. J. Photochem. Photobiol. B. 2018;189:176–184. doi: 10.1016/j.jphotobiol.2018.09.012. [DOI] [PubMed] [Google Scholar]
- 212.Wypij M., Czarnecka J., Świecimska M., Dahm H., Rai M., Golinska P. Synthesis, characterization and evaluation of antimicrobial and cytotoxic activities of biogenic silver nanoparticles synthesized from Streptomyces xinghaiensis OF1 strain. World J. Microbiol. Biotechnol. 2018;34:23. doi: 10.1007/s11274-017-2406-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Hassan S.E.D., Salem S.S., Fouda A., Awad M.A., El-Gamal M.S., Abdo A.M. New approach for antimicrobial activity and bio-control of various pathogens by biosynthesized copper nanoparticles using endophytic actinomycetes. J. Radiat. Res. Appl. Sci. 2018;11:262–270. doi: 10.1016/j.jrras.2018.05.003. [DOI] [Google Scholar]
- 214.Katyal P., Kaur G. Reduction of Cr(VI) by Micrococcus luteus isolate from common effluent treatment plants (CETPs) Int. J. Curr. Microbiol. App. Sci. 2018;7:693–710. doi: 10.20546/ijcmas.2018.707.084. [DOI] [Google Scholar]
- 215.Firrincieli A., Presentato A., Favoino G., Marabottini R., Allevato E., Stazi S.R., Scarascia Mugnozza G., Harfouche A., Petruccioli M., Turner R.J., et al. Identification of Resistance Genes and Response to Arsenic in Rhodococcus aetherivorans BCP1. Front. Microbiol. 2019;10:888. doi: 10.3389/fmicb.2019.00888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Hassan S.E.D., Fouda A., Radwan A.A., Salem S.S., Barghoth M.G., Awad M.A., Abdo A.M., El-Gamal M.S. Endophytic actinomycetes Streptomyces spp mediated biosynthesis of copper oxide nanoparticles as a promising tool for biotechnological applications. J. Biol. Inorg. Chem. 2019;24:377–393. doi: 10.1007/s00775-019-01654-5. [DOI] [PubMed] [Google Scholar]
- 217.Rehan M., Alsohim A.S., El-Fadly G., Tisa L.S. Detoxification and reduction of selenite to elemental red selenium by Frankia. Antonie Van Leeuwenhoek. 2019;112:127–139. doi: 10.1007/s10482-018-1196-4. [DOI] [PubMed] [Google Scholar]
- 218.Maas D., Valerio A., Lourenco L.A., de Oliveira D., Hotza D. Biosynthesis of iron oxide nanoparticles from mineral coal tailings in a stirred tank reactor. Hydrometallurgy. 2019;184:199–205. doi: 10.1016/j.hydromet.2019.01.010. [DOI] [Google Scholar]
- 219.Maas D., de Medeiros M.M., Cesa Rovaris B., Bernardin A.M., de Oliveira D., Hotza D. Biomining of iron-containing nanoparticles from coal tailings. Appl. Microbiol. Biotechnol. 2019;103:7231–7240. doi: 10.1007/s00253-019-10001-2. [DOI] [PubMed] [Google Scholar]
- 220.Devagi P., Suresh T.C., Sandhiya R.V., Sairandhry M., Bharathi S., Velmurugan P., Radhakrishnan M., Sathiamoorthi T., Suresh G. Actinobacterial-Mediated Fabrication of Silver Nanoparticles and Their Broad Spectrum Antibacterial Activity Against Clinical Pathogens. J. Nanosci. Nanotechnol. 2020;20:2902–2910. doi: 10.1166/jnn.2020.17440. [DOI] [PubMed] [Google Scholar]
- 221.Dhanaraj S., Thirunavukkarasu S., John H.A., Pandian S., Salmen S.H., Chinnathambi A., Alharbi S.A. Novel marine Nocardiopsis dassonvillei-DS013 mediated silver nanoparticles characterization and its bactericidal potential against clinical isolates. Saudi J. Biol. Sci. 2020;27:991–995. doi: 10.1016/j.sjbs.2020.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Ramya S., Shanmugasundaram T., Balagurunathan R. Actinobacterial enzyme mediated synthesis of selenium nanoparticles for antibacterial, mosquito larvicidal and anthelminthic applications. Part. Sci. Technol. 2020;38:63–72. doi: 10.1080/02726351.2018.1508098. [DOI] [Google Scholar]
- 223.Desjardin V., Bayard R., Huck N., Manceau A., Gourdon R. Effect of microbial activity on the mobility of chromium in soils. Waste Manag. 2002;22:195–200. doi: 10.1016/S0956-053X(01)00069-1. [DOI] [PubMed] [Google Scholar]
- 224.Bansal V., Bharde A., Ramanathan R., Bargava S.K. Inorganic materials using ‘unusual’ microorganisms. Adv. Colloid Interf. Sci. 2012;179–182:150–168. doi: 10.1016/j.cis.2012.06.013. [DOI] [PubMed] [Google Scholar]
- 225.Newton G.L., Ta P., Fahey R.C. A mycothiol synthase mutant of Mycobacterium smegmatis produces novel thiols and has an altered thiol redox status. J. Bacteriol. 2005;187:7309–7316. doi: 10.1128/JB.187.21.7309-7316.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Painter E.P. The chemistry and toxicity of selenium compounds with special reference to the selenium problem. Chem. Rev. 1941;28:179–213. doi: 10.1021/cr60090a001. [DOI] [Google Scholar]
- 227.Kessi J., Hanselmann K.W. Similarities between the abiotic reduction of selenite with glutathione and the dissimilatory reaction mediated by Rhodospirillum rubrum and Escherichia coli. J. Biol. Chem. 2004;279:50662–50669. doi: 10.1074/jbc.M405887200. [DOI] [PubMed] [Google Scholar]
- 228.Cappelletti M., Fedi S., Zampolli J., Di Canito A., D’Ursi P., Orro A., Viti C., Milanesi L., Zannoni D., Di Gennaro P. Phenotype microarray analysis may unravel genetic determinants of the stress response by Rhodococcus aetherivorans BCP1 and Rhodococcus opacus R7. Res. Microbiol. 2016;167:766–773. doi: 10.1016/j.resmic.2016.06.008. [DOI] [PubMed] [Google Scholar]
- 229.Villadangos A.F., Van Belle K., Whani K., Tamu Dufe V., Freitas S., Nur H., De Galan S., Gil G.A., Collet J.F., Mateos L.M., et al. Corynebacterium glutamicum survives arsenic stress with arsenate reductases coupled to two distinct redox mechanisms. Mol. Microbiol. 2011;82:998–1014. doi: 10.1111/j.1365-2958.2011.07882.x. [DOI] [PubMed] [Google Scholar]
- 230.Li Y., Hu Y., Zhang X., Xum H., Lescop E., Xia B., Jin C. Conformational fluctuations coupled to the thiol-disulfide transfer between thioredoxin and arsenate reductase in Bacillus subtilis. J. Biol. Chem. 2007;282:11078–11083. doi: 10.1074/jbc.M700970200. [DOI] [PubMed] [Google Scholar]
- 231.Ordóñez E., Van Belle K., Roos G., De Galan S., Letek M., Gil J.A., Wyns L., Mateos L.M., Messens J. Arsenate reductase, mycothiol, and mycoredoxin concert thiol/disulfide exchange. J. Biol. Chem. 2009;284:15107–15116. doi: 10.1074/jbc.M900877200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Poole K., Srikumar R. Multidrug efflux in Pseudomonas aeruginosa: Components, mechanisms and clinical significance. Curr. Top. Med. Chem. 2001;1:59–71. doi: 10.2174/1568026013395605. [DOI] [PubMed] [Google Scholar]
- 233.Rahman T., Yarnall B., Doyle D.A. Efflux drug transporters at the forefront of antimicrobial resistance. Eur. Biophys. J. 2017;46:647–653. doi: 10.1007/s00249-017-1238-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Nakaido H. Multidrug resistance in bacteria. Annu. Rev. Biochem. 2009;78:119–146. doi: 10.1146/annurev.biochem.78.082907.145923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Kuroda T., Tsuchiya T. Multidrug efflux transporters in the MATE family. Biochim. Biophys. Acta. 2009;1794:763–768. doi: 10.1016/j.bbapap.2008.11.012. [DOI] [PubMed] [Google Scholar]
- 236.Locher K.P. Review. Structure and mechanism of ATP-binding cassette transporters. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009;364:239–245. doi: 10.1098/rstb.2008.0125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Bay D.C., Rommens K.L., Turner R.J. Small multidrug resistance proteins: A multidrug transporter family that continues to grow. Biochim. Biophys. Acta. 2008;1778:1814–1838. doi: 10.1016/j.bbamem.2007.08.015. [DOI] [PubMed] [Google Scholar]
- 238.Yan N. Structural Biology of the Major Facilitator Superfamily Transporters. Annu. Rev. Biophys. 2015;44:257–283. doi: 10.1146/annurev-biophys-060414-033901. [DOI] [PubMed] [Google Scholar]
- 239.Getsin I., Nalbandian G.H., Yee D.C., Vastermark A., Paparoditis P.C.G., Reddy V.S., Saier M.H. Comparative genomics of transport proteins in developmental bacteria: Myxococcus xanthus and Streptomyces coelicolor. BMC Microbiol. 2013;13:279. doi: 10.1186/1471-2180-13-279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Schmidt A., Haferburg M., Sineriz M., Merten D., Büchel G., Kothe E. Heavy metal resistance mechanisms in actinobacteria for survival in AMD contaminated soils. Geochemistry. 2005;65:131–144. doi: 10.1016/j.chemer.2005.06.006. [DOI] [Google Scholar]
- 241.Raytapadar S., Datta R., Paul A.K. Effects of some heavy metals on growth, pigment and antibiotic production by Streptomyces albus. Acta Microbiol. Acta Microbiol. Immunol. Hung. 1995;42:171–177. [PubMed] [Google Scholar]
- 242.Mirimanoff N., Wilkinson K.J. Regulation of Zn accumulation by a freshwater Gram-positive bacterium (Rhodococcus opacus) Environ. Sci. Technol. 2000;34:616–622. doi: 10.1021/es990744g. [DOI] [Google Scholar]
- 243.Yang H.C., Fu H.L., Lin Y.F., Rosen B.P. Pathways of arsenic uptake and efflux. Curr. Top. Membr. 2012;69:325–358. doi: 10.1016/B978-0-12-394390-3.00012-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Fu H.L., Meng Y., Ordóñez E., Villadangons V.A., Bhattacharjee H., José A.G., Mateos L.M., Rosen B.P. Properties of arsenite efflux permeases (Acr3) from Alkaliphilus metalliredigens and Corynebacterium glutamicum. J. Biol. Chem. 2009;284:19887–19895. doi: 10.1074/jbc.M109.011882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Yumei L., Yamei L., Qiang L., Jie B. Rapid Biosynthesis of Silver Nanoparticles Based on Flocculation and Reduction of an Exopolysaccharide from Arthrobacter sp. B4: Its Antimicrobial Activity and Phytotoxicity. J. Nanomater. 2017;2017:9703614. doi: 10.1155/2017/9703614. [DOI] [Google Scholar]
- 246.Balagurunathan R., Radhakrishnan M., Babu Rajendran R., Velmurugan D. Biosynthesis of gold nanoparticles by actinomycete Streptomyces viridogens strain HM10. Indian J. Biochem. Biophys. 2011;48:331–335. [PubMed] [Google Scholar]
- 247.Kalabegishvili T.L., Kirkesali E.I., Rcheulishvili A.N., Ginturi E.N., Murusidze I.G., Pataraya D.T., Gurielidze M.A., Tsertsvadze G.I., Gabunia V.N., Lomidze L.G., et al. Synthesis of Gold Nanoparticles by Some Strains of Arthrobacter Genera. J. Mat. Sci. Eng. A. 2012;2:164–173. [Google Scholar]
- 248.Derakhshan F.K., Dehnad A., Salouti M. Extracellular Biosynthesis of Gold Nanoparticles by Metal Resistance Bacteria: Streptomyces griseus. Synth. React. Inorg. Met. Org. Nano Met. Chem. 2012;42:868–871. doi: 10.1080/15533174.2011.618484. [DOI] [Google Scholar]
- 249.Faghri Zonooz N., Salouti M., Shapouri R., Nasseryan J. Biosynthesis of Gold Nanoparticles by Streptomyces sp. ERI-3 Supernatant and Process Optimization for Enhanced Production. J. Clust. Sci. 2012;23:375–382. doi: 10.1007/s10876-012-0439-1. [DOI] [Google Scholar]
- 250.Verma V.C., Anand S., Ulrichs C., Singh S.K. Biogenic gold nanotriangles from Saccharomonospora sp., an endophytic actinomycetes of Azadirachta indica A. Juss. Intern. Nano Lett. 2013;3:21. doi: 10.1186/2228-5326-3-21. [DOI] [Google Scholar]
- 251.Waghmare S.S., Deshmukh A.M., Sadowski Z. Biosynthesis, optimization, purification and characterization of gold nanoparticles. Afr. J. Microbiol. Res. 2014;8:138–146. doi: 10.5897/AJMR10.143. [DOI] [Google Scholar]
- 252.Dehnad A., Hamedi J., Derakhshan-Khadivi F., Abuşov R. Green Synthesis of Gold Nanoparticles by a Metal Resistant Arthrobacter nitroguajacolicus Isolated from Gold Mine. IEEE Trans. NanoBiosci. 2015;14:393–396. doi: 10.1109/TNB.2014.2377232. [DOI] [PubMed] [Google Scholar]
- 253.Sowani H., Mohite P., Munot H., Shouche Y., Bapat T., Kumar A.R., Kulkarni M., Zinjarde S. Green synthesis of gold and silver nanoparticles by an actinomycete Gordonia amicalis HS-11: Mechanistic aspects and biological application. Proc. Biochem. 2016;51:374–383. doi: 10.1016/j.procbio.2015.12.013. [DOI] [Google Scholar]
- 254.Ranjitha V.R., Ray V.R. Actinomycetes mediated synthesis of gold nanoparticles from the culture supernatant of Streptomyces griseoruber with special reference to catalytic activity. 3 Biotech. 2017;7:299. doi: 10.1007/s13205-017-0930-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Tsibakhashvili N.Y., Kirkesali E.I., Pataraya D.T., Gurielidze M.A., Kalabegishvili T.L., Gvarjaladze D.N., Tsertsvadze G.I., Frontasyeva M.V., Zinicovscaia I.I., Wakstein M.S., et al. Microbial Synthesis of Silver Nanoparticles by Streptomyces glaucus and Spirulina platensis. Adv. Sci. Lett. 2011;4:3408–3417. doi: 10.1166/asl.2011.1915. [DOI] [Google Scholar]
- 256.Selvakumar P., Viveka S., Prakash S., Jasminebeaula S., Uloganathan R. Antimicrobial activity of extracellularly synthesized silver nanoparticles from marine derived Streptomyces rochei. Int. J. Pharm. Biol. Sci. 2012;3:188–197. [Google Scholar]
- 257.Otari S.V., Patil R.M., Nadaf N.H., Ghosh S.J., Pawar S.H. Green synthesis of silver nanoparticles by microorganism using organic pollutant: Its antimicrobial and catalytic application. Environ. Sci. Pollut. Res. 2014;21:1503–1513. doi: 10.1007/s11356-013-1764-0. [DOI] [PubMed] [Google Scholar]
- 258.Manivasagan P., Venkatesan J., Senthilkumar K., Sivakumar K., Kim S.K. Biosynthesis, antimicrobial and cytotoxic effect of silver nanoparticles using a novel Nocardiopsis sp. MBRC-1. BioMed. Res. Int. 2013;2013:1–9. doi: 10.1155/2013/287638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Priyaragini S., Sathishkumar S., Bhaskararao K. Biosynthesis of silver nanoparticles using actinobacteria and evaluating its antimicrobial and cytotoxicity activity. Int. J. Pharm. Pharm. Sci. 2013;5:709–712. [Google Scholar]
- 260.Babu Vimalanathan A., Ernest V., Arumugasamay K., Tyagi M.G. Biosynthesis of silver nano-particles by the bacterium Micrococcus luteus. Int. J. Appl. Biol. Pharm. Technol. 2013;4:77–83. [Google Scholar]
- 261.Shanmuga Priya T., Balasubramanian V. Enzyme Mediated Synthesis of Silver Nanoparticles using Marine Actinomycetes and Their Characterization. Biosci. Biotechnol. Res. Asia. 2014;11:159–165. doi: 10.13005/bbra/1405. [DOI] [Google Scholar]
- 262.Prakash N.K.U., Bhuvaneswari S., Prabha S.B., Kavitha K., Sandhya K.V., Sathyabhuvaneshwari P., Bharathiraja B. Green Synthesis of Silver Nanoparticles using Airborne Actinomycetes. Int. J. ChemTech. Res. 2014;6:4123–4127. [Google Scholar]
- 263.Karthik L., Kumar G., Vishnu Kirthi A., Rahuman A.A., Bhaskara Rao K.V. Streptomyces sp. LK3 mediated synthesis of silver nanoparticles and its biomedical application. Bioprocess Biosyst. Eng. 2014;37:261–267. doi: 10.1007/s00449-013-0994-3. [DOI] [PubMed] [Google Scholar]
- 264.Mohanta Y.K., Behera S.K. Biosynthesis, characterization and antimicrobial activity of silver nanoparticles by Streptomyces sp. SS2. Bioprocess Biosyst. Eng. 2014;37:2263–2269. doi: 10.1007/s00449-014-1205-6. [DOI] [PubMed] [Google Scholar]
- 265.Anasane N., Golinska P., Wypij M., Rathod D., Dahm H., Rai M. Acidophilic actinobacteria synthesised silver nanoparticles showed remarkable activity against fungi-causing superficial mycoses in humans. Mycoses. 2016;59:157–166. doi: 10.1111/myc.12445. [DOI] [PubMed] [Google Scholar]
- 266.Gowramma B., Keerthi U., Rafi M., Muralidhara Rao D. Biogenic silver nanoparticles production and characterization from native stain of Corynebacterium species and its antimicrobial activity. 3 Biotech. 2015;5:195–201. doi: 10.1007/s13205-014-0210-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Kamel Z., Saleh M., El Namoury N. Biosynthesis, Characterization, and Antimicrobial Activity of Silver Nanoparticles from Actinomycetes. Res. J. Pharm. Biol. Chem. Sci. 2016;7:119–127. [Google Scholar]
- 268.Raja M.M.M., John S.A. Biosynthesis of Silver Nanoparticles by Novel Isolate of Marine Micromonospora species (KU 867645) and its Antibacterial Activity against Multidrug Resistant Hospital-acquired Uropathogens in Reference with Standard Antibiotics. Indian J. Pharm. Sci. 2017;79:369–376. doi: 10.4172/pharmaceutical-sciences.1000239. [DOI] [Google Scholar]
- 269.Avilala J., Golla N. Antibacterial and antiviral properties of silver nanoparticles synthesized by marine Actinomycetes. Int. J. Pharm. Sci. Res. 2019;10:1223–1228. doi: 10.13040/IJPSR.0975-8232.10(3).1223-28. [DOI] [Google Scholar]
- 270.Hamed A.A., Kabary H., Khedr M., Emam A.N. Antibiofilm, antimicrobial and cytotoxic activity of extracellular green-synthesized silver nanoparticles by two marine-derived actinomycete. RSC Adv. 2020;10:10361–10367. doi: 10.1039/C9RA11021F. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Usha R., Prabu E., Palaniswamy M., Venil C.K., Rajendran R. Synthesis of Metal Oxide Nano Particles by Streptomyces sp. for Development of Antimicrobial Textiles. Glob. J. Biotech. Biochem. 2010;5:153–160. [Google Scholar]
- 272.Nabila M.I., Kannabiran K. Biosynthesis, characterization and antibacterial activity of copper oxide nanoparticles (CuO NPs) from actinomycetes. Biocatal. Agric. Biotechnol. 2018;15:56–62. doi: 10.1016/j.bcab.2018.05.011. [DOI] [Google Scholar]
- 273.Waghmare S., Deshmukh A., Kulkarni S.W., Oswaldo L. Biosynthesis and characterization of manganese and zinc nanoparticles. Univ. J. Environ. Res. Technol. 2011;1:64–69. [Google Scholar]
- 274.Rajamanickam U., Mylsamy P., Viswanathan S., Muthusamy P. Biosynthesis of Zinc Nanoparticles Using Actinomycetes for Antibacterial Food Packaging; Proceedings of the Internation Conference on Nutriotion and Food Science IPCBEE; Singapore. 23–24 July 2012; Singapore: IACSIT Press; 2012. pp. 195–199. [Google Scholar]
- 275.Piacenza E., Presentato A., Amborsi E., Speghini A., Turner R.J., Vallini G., Lampis S. Physical-Chemical Properties of Biogenic Selenium Nanostructures Produced by Stenotrophomonas maltophilia SeITE02 and Ochrobactrum sp. MPV1. Front. Microbiol. 2018;9:3178. doi: 10.3389/fmicb.2018.03178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Piacenza E., Presentato A., Heyne B., Turner R.J. Tunable photoluminescence properties of selenium nanoparticles: Biogenic versus chemogenic synthesis. Nanophotonics. 2020;9:3615–3628. doi: 10.1515/nanoph-2020-0239. [DOI] [Google Scholar]
- 277.Rahman M.M., Khan S.B., Jamal A., Faisal M., Aisiri A.M. Chapter 3: Iron Oxide Nanoparticles. In: Rhaman M.M., editor. Nanomaterials. IntechOpen; London, UK: 2011. pp. 43–66. [DOI] [Google Scholar]
- 278.Turner R.J. Metal-based antimicrobial strategies. Microb. Biotechnol. 2017;10:1062–1065. doi: 10.1111/1751-7915.12785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Bernardos A., Piacenza E., Sancenon F., Hamidi M., Maleki A., Turner R.J., Martinez-Manez R. Mesoporous Silica-Based Materials with Bactericidal Properties. Small. 2019;15:1900669. doi: 10.1002/smll.201900669. [DOI] [PubMed] [Google Scholar]