Abstract
Objective
To compare the postoperative inflammation and pain response between medial pivot (MP) and posterior stabilized (PS) prostheses among total knee arthroplasty (TKA) patients.
Methods
A prospective cohort study was conducted from January 2019 to May 2019 at the Affiliated Hospital of Qingdao University. The study included patients diagnosed with stage III or IV Kellgren–Lawrence knee osteoarthritis (KOA) who had failed conservative treatment, had undergone no previous knee surgeries, had varus substantial deformities (11°–20° deviation), and had received their first unilateral TKA. A total of 109 patients who underwent PS prosthesis TKA and 98 patients who underwent MP prosthesis TKA were continuously enrolled. Inflammation biomarkers, such as leukocyte (white blood cells), erythrocyte sedimentation rate (ESR), and C‐reactive protein (CRP), together with hemoglobin (Hb), the visual analog pain score (VAS) and range of motion (ROM) were compared between the two groups. The Student t‐test was applied to analyze continuous parameters, and the χ2‐test was used for categorical parameters. The linear mixed model was used for the repeated measurement data from the follow‐up visits. Multivariate backward logistic and linear regression models were used to determine the factors potentially influencing prostheses and VAS scores.
Results
All these enrolled patients were followed up at 2, 4, 7, and 30 days after TKA. There were no significant differences between the PS group and the MP group in body mass index (BMI), gender, laterality, usage of nonsteroidal anti‐inflammatory drugs (NSAIDs) and opioids, and drain tube extubation time (P > 0.05). Compared with the PS group, the MP group were older (67.5 years vs 65.4 years), and had a higher mid‐vastus approach rate (67.3% vs 26.6%), a shorter tourniquet duration (68.3 ± 10.2 h vs 73.9 ± 11.2 h), a larger prosthetic pad (10.8 ± 1.2 mm vs 10.4 ± 1.2 mm), and a lower drain tube diversion volume (187.6 ± 119.3 mL vs 234.0 ± 155.7 mL). In the linear mixed model, MP prostheses had less CRP and ESR elevation and less Hb decrease than PS prostheses (P for group × time < 0.001). There were no significant differences in the changing trends between MP and PS prostheses by time for VAS scores and ROM. In the multivariate logistic regression model, MP prostheses showed significant differences compared with PS prostheses in treatment approach (odds ratio [OR] = 3.371, 95% confidence interval [CI]: 1.953–7.127; P < 0.001), ultrasound treatment start time (OR = 2.669, 95% CI: 1.385–5.141; P = 0.003), and tourniquet duration (OR = 0.954, 95% CI: 0.925–0.984; P = 0.003). Higher VAS scores on the second day postoperatively were related to high VAS scores preoperatively, use of opioids, high drain tube diversion, long tourniquet duration, and long drain tube extubation (P < 0.05), respectively.
Conclusion
The MP prostheses showed potential advantages compared with PS prostheses in TKA in inflammatory responses.
Keywords: Inflammation, Medial pivot prostheses, Posterior stabilized prostheses, Postoperative effects, Total knee arthroplasty

There were no significant differences in WBC changes in the PS group and the MP group over time. However, the MP group had lower increases in ESR and CRP and greater decreases in Hb after TKA than the PS group.
Introduction
Total knee arthroplasty (TKA) is the most effective surgical intervention for terminal stage knee osteoarthritis (KOA) 1 . With China's aging population, the prevalence of symptomatic KOA has risen to 8.1% according to a survey published in 2016, potentially increasing the candidates for TKA 2 . Today, with the availability of multiple prosthesis designs and the advances in surgical techniques and instruments, patients with KOA have the opportunity to have their pain relieved, deformities corrected, and their quality of their life improved. During the past two decades, prostheses designs have undergone many revisions and modifications. However, among the variated prostheses, which design represents the best solution for KOA patients is still under debate 3 .
The posterior‐stabilized TKA (PS‐TKA) was developed in 1978 to address the issue of abnormal posterior cruciate ligament morphology 4 . Some studies have reported that posterior stabilized (PS) prostheses have better postoperative mobility, and that it is easier to achieve soft tissue balance during operations with PS prostheses. Moreover, the PS‐TKA relies on a femoral cam that articulates with a tibial post, improving femoral rollback and increasing the anteroposterior and translational stability of the knee 5 . However, disadvantages of PS prostheses include central tibial impingement and large osteotomy 6 . Mahringer‐Kunz et al. reported that blood loss was significantly higher for PS‐TKA due to the box preparation that exposes more cancellous femoral bone, which may also increase postoperative bleeding 7 . Meneghini et al. suggest that advantages of avoiding a PS‐TKA include eliminating the risk of box cut‐induced femoral condylar fracture, improving operative efficiency by removing procedural steps, removing the articulation that is a source of wear, post‐deformation, breakage and dislocation, and eliminating patella clunk 8 .
Normal knees show a medial pivot kinematic pattern, characterized by femoral external rotation relative to the tibia with movement from full extension to mid‐flexion 9 . Based on these biomechanical findings, the medial pivot (MP‐TKA) design was developed, aimed at better mimicking the natural kinematics of the knee, with more natural femoral rollback. The MP‐TKA functions like a ball and socket joint, with the lateral femoral condyle translating in an anteroposterior direction and rotating around the medial compartment in flexion 10 . The medial compartment can move in a small range, and the lateral compartment can slide back and forth. At the same time, the larger area of the medial compartment tibial pad and the femoral condyle reduce the wear of the pad. Moreover, the MP‐TKA design features a deeper, highly conforming medial compartment and a less congruent lateral component to allow relative freedom of anteroposterior movement of the lateral condyle 11 .
There have been comparative studies of medial pivot (MP) and PS prostheses considering patient‐reported outcomes, radiographic outcomes, and safety. Samy et al. reported that those in the MP group did statistically and significantly better in terms of the forgotten joint score compared with the PS group 11 . Yuan et al. compared 49 MP‐TKA with 51 PS‐TKA cases and concluded that MP‐TKA is a safe and effective method for Chinese patients 12 . However, some studies have found no statistical difference between these two designs with respect to clinical outcomes. Lee et al. reported that there was no evidence of the superiority of MPTKA over PSTKA in terms of preference and satisfaction 13 . Lin et al. reached similar conclusions in a retrospective study 14 . Kulshrestha et al. reported that patients who underwent MP‐TKA were better at getting up from a chair and had a faster walking speed, while PS‐TKA resulted in better knee flexion 15 .
The inflammatory state of patients with KOA is an indicator of the degree of KOA disease. The inflammatory state of patients undergoing TKA may, therefore, be a predictor of postoperative recovery 16 . Surgery induces a marked immune response; the immune response to trauma is described as pro‐inflammatory followed by an anti‐inflammatory response. Surgery‐induced immune response can also predict postoperative recovery by reflecting the extent of tissue damage 17 . Studies have also proved that erythrocyte sedimentation rate (ESR) and C‐reactive protein level (CRP) and their variations could be suitable factors to detect probable infections in patients undergoing TKA operations 18 . Besides differences in kinematics, morphology, and materials, different prosthesis designs may also lead to varying intraoperative maneuvers, including the surgical approach, soft tissue procedures, and bone cuts, which may cause variation in inflammation markers and pain scores. Few studies have focused on these issues.
Therefore, this prospective follow‐up study evaluated the clinical outcomes of inflammation, including leukocyte (white blood cell [WBC]) level, ESR level, and CRP level. The hemoglobin (Hb) level, which represents blood loss, the visual analog scale (VAS) score, which represents pain, and the range of motion (ROM), which represents function, were also compared between PS‐TKA and MP‐TKA.
The purpose of the current study was to provide surgeons with an understanding of: (i) the different clinical manifestations in patients using PS and MP prostheses; (ii) the different early stage postoperative inflammation and pain effects caused by the two surgical techniques and philosophies of design between PS‐TKA and MP‐TKA; and (iii) the TKA‐related factors for postoperative pain.
Methods and Materials
Ethics Statement
This study was conducted in accordance with the Declaration of Helsinki (Ethical Principles for Medical Research Involving Human Subjects) and was approved by the Medical Ethics Committee of the Affiliated Hospital of Qingdao University (No. QYFYKYLL‐2018‐01‐15‐10).
Inclusion and Exclusion Criteria
Patient eligibility criteria were as follows: (i) patients diagnosed with Kellgren–Lawrence stage III or IV KOA for whom conservative treatment had failed and who had varus substantial deformities (11°–20°) 19 , 20 ; (ii) patients who had not had previous knee surgeries and who had undergone their first unilateral TKA; (iii) patients who had undergone PS‐TKA or MP‐TKA; (iv) inflammation biomarkers were available; and (v) informed consent had been provided for participation in the prospective cohort study.
The following were grounds for exclusion: (i) any metabolic disease of bone or severe osteoporosis; (ii) severe cardiopulmonary dysfunction, diabetes, cerebrovascular disease, rheumatism, or immunity disorder; (iii) history of infectious, mental, or neurologic disease; (iv) use of potentially interfering medications; (v) osteogenic or osteoclastic tumors; (vi) partial use of tourniquet or did not receive ultrasound treatment; and (vii) participation in other research experiments or poor compliance.
Surgical Procedures
All surgeries were performed by one arthroplasty surgeon in operation rooms with the same laminar airflow. All patients were placed in supine position and underwent general anesthesia combined with peripheral nerve blocks. A thigh tourniquet was applied and inflated to 100 mm Hg above systolic in all patients. Exposure of the surgical site was obtained by either a standard medial parapatellar approach or a mid‐vastus approach 21 , 22 , the patella was translated laterally instead of everted, and severe cartilage wear and massive osteophytes were found and removed in all patients intraoperatively. In the PS group, a Zimmer NexGen prosthesis (NexGen LPS Flex, Zimmer, Warsaw) was applied for each patient. In the MP group, a Wright Advance prosthesis was used. Antibiotic prophylaxis was given prior to inflation of the tourniquet and two doses were administered postoperatively. All patients received a tourniquet with postoperative ultrasound, and NSAIDS or opioids for pain relief after TKA if necessary.
Data Collection and Postoperative Visits
All patients were followed up at 2, 4, 7, and 30 days after the TKA, and the VAS and the ROM were recorded at each visit. Patients also received three blood tests, respectively, on the day before the TKA, and 2 and 4 days after the TKA.
Visual Analog Scale
The VAS score is used widely in social and behavioral sciences to measure pain. The VAS score assesses pain based on a score (0–10) that patients assign themselves 23 . A score of 0 represents no pain; a score of 1–3 represents mild pain that could be tolerated, with sleep not affected; a score of 4–6 means that pain could be endured and sleep was affected; a score of 7–10 represents severe pain that could not be tolerated.
Range of Motion
The ROM of the knee was measured by one physician using a goniometer. The angle is considered 0° when the knee is in full extension without hyperextension or flexion contraction, the angle is considered positive in flexion, and negative if hyperextension exists 24 .
Inflammation Biomarkers
The recorded inflammation blood biomarkers include CRP, ESR, and WBC. CRP is a protein produced in the liver due to an inflammation in the body, which could be used as a highly sensitive test to detect infections 25 . CRP was measured using the particle‐enhanced light‐scattering immunoassay on the TBA‐200FR system (Toshiba, Tokyo, Japan) according to the manufacturer's instructions. ESR is a hematological condition, which increases when one's body is affected by an infection, inflammation, major stressors, and autoimmune diseases 25 . ESR was measured using the photometric kinetic analysis of capillary‐stopped flow on the Test‐1 system (Alifax, Padova, Italy). A WBC count measures the total number of the WBC in blood, which is usually stated as the number in one cubic millimeter.
Hemoglobin
Hemoglobin is an iron‐containing protein in the red blood cells (erythrocytes) that transports oxygen to the tissues. Hb forms an unstable reversible bond with oxygen. In this study, the level of Hb reflects the postoperative blood loss and recovery.
Statistical Analysis
The patients were divided into two groups based on the prosthesis used: the PS group and the MP group. The demographic characteristics of continuous variables (e.g. age, body mass index [BMI], tourniquet duration, prosthetic pad, and drain tube diversion) were described as means with standard deviations (SD), and compared between PS and MP groups using the Student t‐test. The categorical variables were described as frequency and percentage. χ2‐tests were used to compare the categorical variables between PS and MP groups. The linear mixed model was used for the repeated measurement data from the follow‐up visits. The univariate and multivariate backward logistic and linear regression models were used to determine the factors potentially influencing prostheses and VAS scores. Statistical analyses were performed using SAS software, version 9.4 (SAS Institute, Cary, NC, USA) and P‐values <0.05 were considered statistically significant.
Results
Patient Demography
In total, 207 patients requiring unilateral TKA at the Affiliated Hospital of Qingdao University from January 2019 to May 2019 were continuously recruited (Fig. 1). There were 109 patients who received PS‐TKA (Fig. 2) and 98 patients who received MP‐TKA (Fig. 3).
Fig. 1.
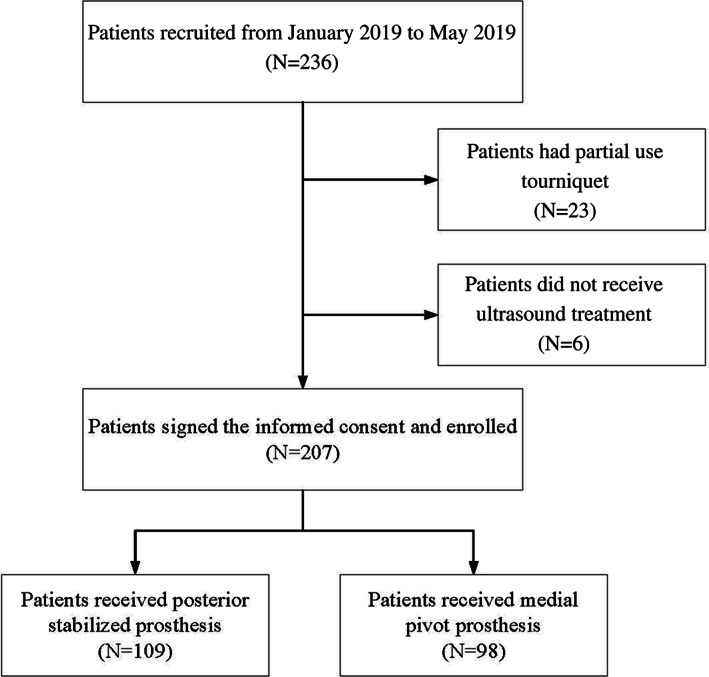
The flow chart shows the patient enrollment process.
Fig. 2.
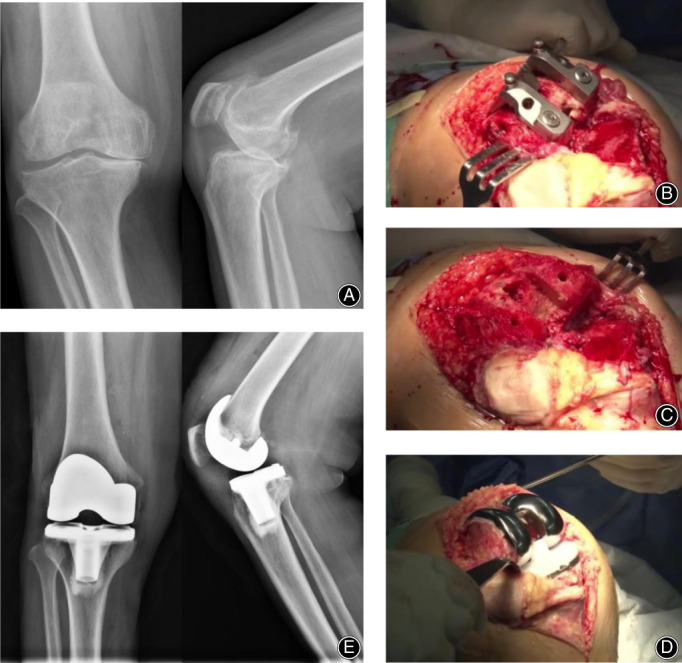
A case of a 67‐year woman who underwent posterior stabilized total knee arthroplasty. (A) Anteroposterior X‐ray of knee demonstrated right knee osteoarthritis of Kellgren–Lawrence stage IV. (B) A posterior stabilized (PS) box cutting jig was placed after femoral cut, (C) Image of bone after box cutting. (D) The whole package of prosthesis and polyethylene with a cam was properly placed. (E) Postoperative anteroposterior X‐ray of the knee.
Fig. 3.
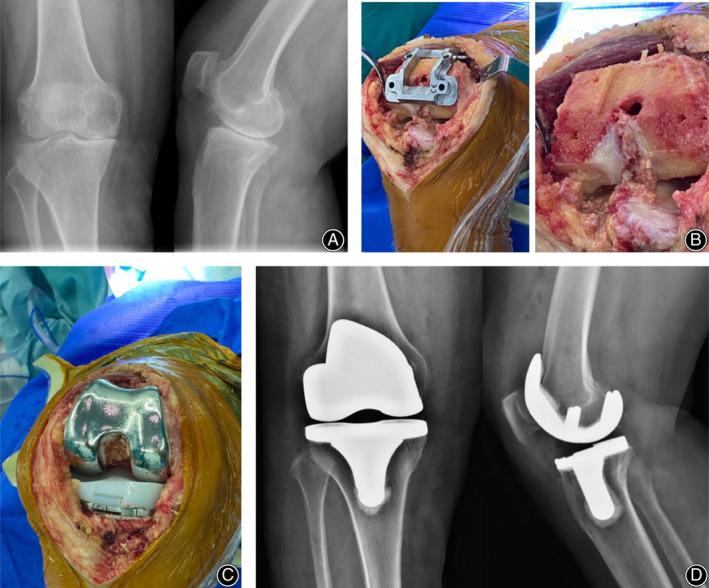
A 65‐year woman who underwent medial pivot total knee arthroplasty. (A) Preoperative anteroposterior X‐ray of the knee demonstrated right knee osteoarthritis of Kellgren–Lawrence stage IV. (B) Due to a different design of prosthesis less box cutting was done to compare the posterior stabilized total knee arthroplasty and more bone was preserved. (C) Polyethylene without a cam was placed after cementing of the femoral and tibial components. (D) Postoperative anteroposterior X‐ray of the patient.
The average ages of the enrolled patients were 65.4 ± 6.2 years in the PS group and 67.5 ± 6.5 years in the MP group (P = 0.015). All enrolled patients were followed up for 30 days after TKA. As shown in Table 1, there were no significant differences between the PS group and the MP group in BMI, gender, laterality, usage of NSAIDS and opioids, and drain tube extubation time. Compared with the PS group, those in the MP group were older (67.5 vs 65.4 years), and had a higher mid‐vastus approach rate (67.3% vs 26.6%), a shorter tourniquet duration (68.3 ± 10.2 h vs 73.9 ± 11.2 h), a larger prosthetic pad (10.8 ± 1.2 mm vs 10.4 ± 1.2 mm), and a lower drain tube diversion volume (187.6 ± 119.3 mL vs 234.0 ± 155.7 mL).
TABLE 1.
Demographic and surgical characteristics of enrolled patients (mean ± SD)
| Indexes | PS prosthesis (n = 109) | MP prosthesis (n = 98) | P‐value | |
|---|---|---|---|---|
| Age (years) | 65.4 ± 6.2 | 67.5 ± 6.5 | 0.015 | |
| BMI (kg/m2) | 27.6 ± 3.0 | 27.3 ± 3.0 | 0.397 | |
| Gender (n [%]) | Female | 80 (73.4) | 74 (75.5) | 0.728 |
| Male | 29 (26.6) | 24 (24.5) | ||
| Laterality (n [%]) | Left knee | 48 (44.0) | 47 (48.0) | 0.572 |
| Right knee | 61 (56.0) | 51 (52.0) | ||
| Surgical methods (n [%]) | Medial parapatellar approach | 80 (73.4) | 32 (32.7) | <0.001 |
| Mid‐vastus approach | 29 (26.6) | 66 (67.3) | ||
| Usage of NSAIDS (n [%]) | No | 6 (5.5) | 5 (5.1) | 0.897 |
| Yes | 103 (94.5) | 93 (94.9) | ||
| Usage of Opioids (n [%]) | No | 40 (36.7) | 36 (36.7) | 0.996 |
| Yes | 69 (63.3) | 62 (63.3) | ||
| Ultrasound treatment start time (n [%]) | 1 day after surgery | 75 (68.8) | 37 (37.8) | <0.001 |
| 2 days after surgery | 34 (31.2) | 61 (62.2) | ||
| Drain tube extubation time (n [%]) | 1 day after surgery | 44 (40.4) | 27 (27.6) | 0.053 |
| 2 days after surgery | 65 (59.6) | 71 (72.4) | ||
| Tourniquet duration (h) | 73.9 ± 11.2 | 68.3 ± 10.2 | <0.001 | |
| Prosthetic pad (mm) | 10.4 ± 1.2 | 10.8 ± 1.2 | 0.017 | |
| Drain tube diversion (mL) | 234.0 ± 155.7 | 187.6 ± 119.3 | 0.017 | |
BMI, body mass index; MP, medial pivot; NSAIDS, nonsteroidal anti‐inflammatory drugs; PS, posterior stabilized; SD, standard deviation.
Changes in Blood Markers between Posterior Stabilized and Medial Pivot Groups
The leukocyte (WBC) level, the ESR, and CRP levels were low before the TKA in both the MP group and the PS group (Fig. 4). The WBC level, the ESR, and the CRP level all rose to be relatively high by the second day after TKA. The patients' WBC decreased in both groups by the fourth day (Fig. 4A). The patients' ESR remained raised on the fourth day after TKA (Fig. 4B). However, the CRP remained stable from day 2 to day 4 (Fig. 4C). The Hb level slightly decreased after TKA (Fig. 4D). In the linear mixed model, all the blood markers changed significantly among different time points (all P‐values for time < 0.001). The P‐value for group × time was 0.329 for WBC, which meant there was no significant difference in WBC changes in the PS group and the MP group over time. The results showed different changing trends of ESR, which increased more rapidly in the PS group than in the MP group (P‐value for group × time < 0.001). The CRP in the PS group also increased more rapidly in the PS group than in the MP group (P‐value for group × time < 0.001). The Hb level in the MP group decreased slower than in the PS group (P‐value for group × time < 0.001, Fig. 4D).
Fig. 4.
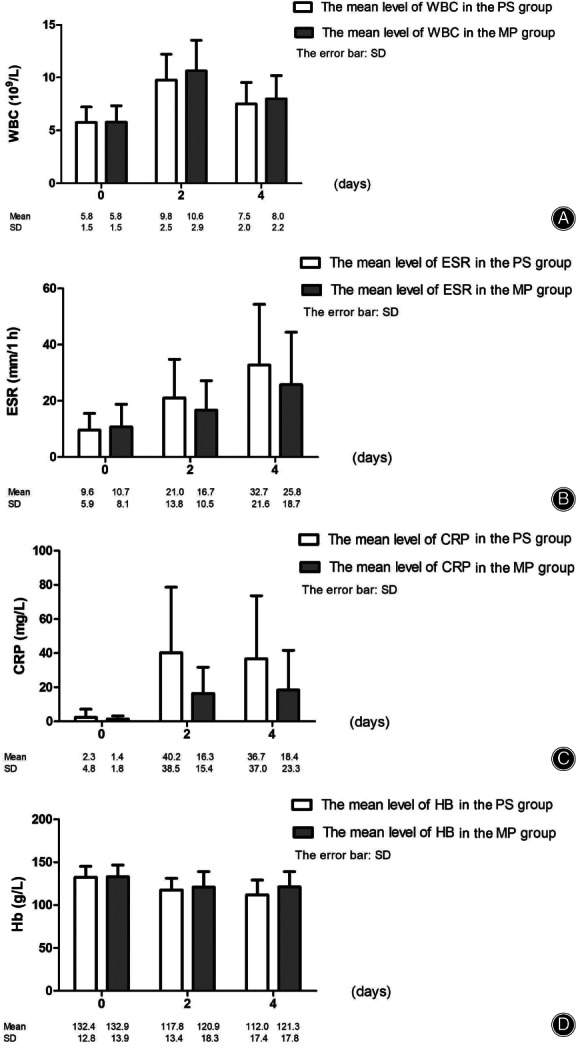
The graphs show the changes of means and standard deviations (SD) in leukocyte (white blood cells) levels (A), erythrocyte sedimentation rate (ESR) (B), C‐reactive protein (CRP) levels (C), and hemoglobin (Hb) levels (D) after total knee arthroplasty (TKA) in medial pivot (MP) and posterior stabilized (PS) groups.
Changes in Visual Analog Pain Score and Range of Motion between Posterior Stabilized and Medial Pivot Groups
In Fig. 5, the VAS scores showed slight fluctuation within 1 week after TKA and decreased by day 30 in both PS and MP groups (P‐value for group = 0.333 and P‐value for group × time = 0.158). The flexion range of the knee in both groups showed an increase after TKA with no significant difference (P‐value for group = 0.491 and P‐value for group × time = 0.595). The extension range in the MP group showed a more rapid decrease than that of the PS group after TKA (P‐value for group = 0.007 and P‐value for group × time = 0.210). The changing trends in MP and PS groups over time were similar for VAS scores, flexion, and extension.
Fig. 5.
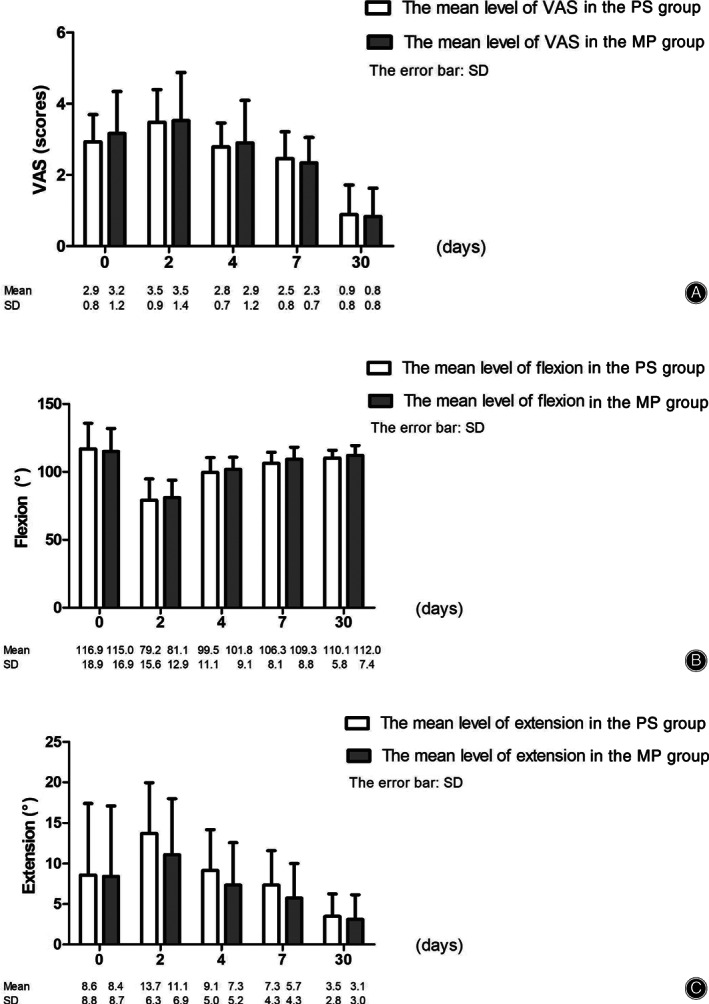
The graphs show the changes of means and standard deviations (SD) in visual analog pain score (VAS) (A) and range of motion (ROM) (B and C) after total knee arthroplasty (TKA) among medial pivot (MP) and posterior stabilized (PS) groups.
Potential Influencing Factors on Visual Analog Pain Score on the Second Day after Total Knee Arthroplasty
In the multivariate logistic regression model (Table 2), MP prostheses showed significant differences compared with PS prostheses in treatment approach (OR = 3.371, 95% CI: 1.953–7.127; P < 0.001), ultrasound treatment start time (OR = 2.669, 95% CI: 1.385–5.141; P = 0.003), and tourniquet duration (OR = 0.954, 95% CI: 0.925–0.984; P = 0.003).
TABLE 2.
Multivariate logistic regression of factors on medial pivot (MP) and posterior stabilized (PS) prostheses
| Univariate regression | Multivariate regression | |||
|---|---|---|---|---|
| Indexes | OR (95% CI) | P | OR (95% CI) | P |
| Age | 1.055 (1.010–1.103) | 0.017 | — | — |
| BMI | 0.961 (0.877–1.053) | 0.396 | — | — |
| Gender (female vs male) | 0.895 (0.478–1.674) | 0.728 | — | — |
| Laterality left knee vs right knee | 0.854 (0.494–1.477) | 0.572 | — | — |
| Medial parapatellar approach vs mid‐vastus approach | 5.690 (3.126–10.356) | <0.001 | 3.371 (1.953–7.127) | <0.001 |
| NSAIDS (no vs yes) | 1.083 (0.320–3.667) | 0.898 | — | — |
| Opioids (no vs yes) | 0.998 (0.567–1.758) | 0.996 | — | — |
| Ultrasound treatment start time 1 day vs 2 days | 3.636 (2.045–6.464) | <0.001 | 2.669 (1.385–5.141) | 0.003 |
| Drain tube extubation 1 day vs 2 days | 1.780 (0.991–3.197) | 0.054 | — | — |
| Tourniquet duration | 0.951 (0.926–0.978) | <0.001 | 0.954 (0.925–0.984) | 0.003 |
| Prosthetic pad | 1.333 (1.048–1.695) | 0.019 | — | — |
| Drain tube diversion | 0.998 (0.996–1.000) | 0.020 | — | — |
BMI, body mass index; CI, confidence interval; NSAIDS, nonsteroidal anti‐inflammatory drugs; OR, odds ratio
—, the factors were removed in multivariate logistic regression model.
The VAS reached the peak by the second day after TKA, which revealed great pain for the patients. As a result, factors that potentially influence the VAS scores on the second day after TKA should be considered. In Table 3, the univariate linear regression models showed that MP and PS prostheses did not influence the VAS on the second day after TKA (P = 0.786). In multivariate regression, the higher VAS before TKA influenced the high VAS on the second day after TKA (P < 0.001). This could also explain the higher rates of opioid use (P < 0.001). The higher VAS on the second day after TKA also related to the tourniquet duration, the drain tube extubation time, and drain tube diversion (P = 0.002, P = 0.025 and P = 0.027).
TABLE 3.
Multivariate linear regression of factors on VAS scores 2 days after TKA
| Indexes | Univariate regression | Multivariate regression | ||
|---|---|---|---|---|
| β | P | β | P | |
| PS vs MP | 0.043 | 0.786 | — | — |
| VAS before TKA | 0.610 | <0.001 | 0.591 | <0.001 |
| Age | −0.006 | 0.624 | — | — |
| BMI | −0.006 | 0.809 | — | — |
| Gender (Female vs Male) | 0.168 | 0.358 | — | — |
| Laterality Left knee vs Right knee | −0.209 | 0.192 | — | — |
| Medial parapatellar approach vs mid‐vastus approach | −0.100 | 0.211 | ||
| NSAIDS (no vs yes) | 0.526 | 0.139 | — | — |
| Opioids (no vs yes) | 0.620 | <0.001 | 0.643 | <0.001 |
| Ultrasound treatment start time 1 day vs 2 days | −0.239 | 0.135 | — | — |
| Drain tube extubation (1 day vs 2 days) | 0.479 | 0.004 | 0.323 | 0.025 |
| Tourniquet duration | 0.008 | 0.295 | 0.019 | 0.002 |
| Prosthetic pad | 0.063 | 0.350 | — | — |
| Drain tube diversion | 0.001 | 0.010 | 0.001 | 0.027 |
BMI, body mass index; MP, medial pivot; NSAIDS, nonsteroidal anti‐inflammatory drugs; PS, posterior stabilized; TKA, total knee arthroplasty; VAS, visual analog scale score
—, the factors were removed in multivariate logistic regression model.
Complications
No complications, such as wound infection, loosening of prostheses, or dislocation of polyethylene, were found in either group during the follow‐up.
Discussion
This prospective cohort study showed that the MP prosthesis had potential advantages compared with the PS prosthesis. The MP prosthesis started being used in TKA more than 10 years later than the PS prosthesis 4 , 26 . Previous studies have shown that PS and MP prostheses both have advantages in improving femoral rollback 5 , 27 . The PS prosthesis can increase anteroposterior and translational stability of the knee, while the MP prosthesis allows relative freedom of anteroposterior movement of the lateral condyle 19 . To date, many studies have focused on the PS prosthesis, but few have compared the MP prosthesis with the PS prosthesis 11 , 28 , 29 .
Previous studies comparing the MP prosthesis and the PS prosthesis retrospectively reviewed patients for 6 weeks to 5 years. Our study first prospectively recorded the inflammation and pain manifestation in a cohort study, with the follow up visits at postoperative 2, 4, 7, and 30 days. Samy et al. reported that patients who underwent the MP‐TKA scored better than those who underwent the PS‐TKA, particularly regarding deep knee flexion and stability of the prosthesis 11 , which was similar to our results. Another two studies showed that there were no significant differences in midterm outcomes in patients receiving MP or PS prostheses 12 , 28 .
Differences on Inflammation Biomarkers Changes
Although there were no significant differences in VAS scores between PS and MP prostheses among unilateral TKA patients, the postoperative serum inflammatory markers of ESR and CRP highlighted the potential advantages of MP prosthesis. The ESR and CRP could reflect the body's inflammatory response, especially among the KOA patients. Xu et al. evaluated the prevalence of preoperatively elevated serum inflammatory markers to determine the association with periprosthetic joint infection in patients with osteoarthritis undergoing TKA 29 and found that the rate of periprosthetic joint infection was significantly higher in the both elevated ESR and CRP groups (12.5%) compared with both normal group (1.4%) and high group (0.9%) (P < 0.001). Sanchez‐Ramirez et al. found that the elevated CRP values are related to a lower gain in muscle strength over time in patients with established KOA 30 . Our results showed that the MP group had lower extension than the PS group, which meant that patients who received MP would recover faster in regard to straightening function. Combining these results, there is a potential better degree of recovery for patients receiving MP prostheses.
Differences in Blood Loss
The lower drain tube diversion of the MP group (187.6 vs 234.0 mL) also highlighted the potential advantages. The higher Hb level in the MP prosthesis group than in the PS prosthesis group postoperatively verified the results. Four days after the TKA, the Hb was still slowly decreasing in the PS prosthesis group but remained stable, slightly increasing, in the MP prosthesis group, with the average Hb level of 112.0 ± 17.4 g/L vs 121.3 ± 17.8 g/L. These results demonstrated the potential better inflammation responses in the MP group, which might be due to less trauma of the MP group and patients might experience less pain or faster recovery.
Total Knee Arthroplasty‐Related Factors for Postoperative Pain
In the multivariate linear regression analysis, the results demonstrated that we should pay more attention to patients with high VAS scores before TKA and longer tourniquet duration. Previous studies on the timing of releasing tourniquets showed that releasing the tourniquet before the wound recovery would cause an increase in total blood loss but decrease the risk of complications 31 , 32 . In contrast, in a meta‐analysis, the authors found that the mean pain scores of patients treated with tourniquets were significantly higher than those treated without tourniquets, with the mean pain scores of 5.23 ± 1.94 cm and 3.78 ± 1.61 cm, respectively 33 . Similar with this meta‐analysis, our results showed that the higher VAS on the second day after TKA related to the tourniquet duration, the drain tube extubation time, and drain tube diversion (P = 0.002, P = 0.025 and P = 0.027), which indicated that the tourniquet should be release as early as possible to relieve pain.
Differences on Clinical Manifestations
The design of PS and MP prostheses are based on different kinematics and philosophies, which lead to different instrumentation and surgical techniques. In our experience, more procedures are required for PS prosthesis due to more complex instruments; thus, a prolonged tourniquet duration is needed, which might potentially cause further muscular damage. In addition, more bone cutting is necessary is made when using a PS prosthesis to achieve better femoral roll back. Moreover, in this study we found that the instrument design for MP prostheses was more suitable for the mid‐vastus approach in practice. In contrast, the results in Table 3 showed that tourniquet duration was independently related to the postoperative VAS scores and the postoperative pain. These results may explain the phenomenon of MP TKA having significantly shorter tourniquet duration, smaller inflammation effects, and a higher rate of mid‐vastus approach adoption.
Limitations
Some limitations of this study should be addressed. First, this was a single center prospective cohort study, and the follow‐up visits were within the postoperative 1 month. Multi‐center studies with long‐term follow up are needed. Second, our study only evaluated the VAS; however, the American Knee Society score and the Western Ontario and McMaster Universities osteoarthritis index should also be considered as endpoints in such research. Finally, this study enrolled 207 patients; a larger sample size could provide more powerful results.
Conclusion
The MP prostheses showed potential advantages compared with PS prostheses in TKA in inflammatory responses.
Acknowledgments
This study was funded by the Beijing Union Medical College “Central University Basic Research Business Fees” Project (No. 3332018004). The authors thank all the participants enrolled in this study.
Disclosure: The authors declare no conflict of interest.
Contributor Information
Hui‐jun Bai, Email: huijunbai@aliyun.com.
Ying‐zhen Wang, Email: 18661808238@163.com.
References
- 1. Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res, 2016, 28: 1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Tang X, Wang S, Zhan S, et al The prevalence of symptomatic knee osteoarthritis in China: results from the China health and retirement longitudinal study. Arthritis Rheumatol, 2016, 68: 648–653. [DOI] [PubMed] [Google Scholar]
- 3. Bontempi M, Roberti di Sarsina T, Marcheggiani Muccioli GM, et al J‐curve design total knee arthroplasty: the posterior stabilized shows wider medial pivot compared to the cruciate retaining during chair raising. Knee Surg Sports Traumatol Arthrosc, 2020, 28: 2883–2892. [DOI] [PubMed] [Google Scholar]
- 4. Dorr LD, Ochsner JL, Gronley J, Perry J. Functional comparison of posterior cruciate‐retained versus cruciate‐sacrificed total knee arthroplasty. Clin Orthop Relat Res, 1988, 236: 36–43. [PubMed] [Google Scholar]
- 5. Ranawat CS, Komistek RD, Rodriguez JA, Dennis DA, Anderle M. In vivo kinematics for fixed and mobile‐bearing posterior stabilized knee prostheses. Clin Orthop Relat Res, 2004, 418: 184–190. [DOI] [PubMed] [Google Scholar]
- 6. Anderson MJ, Becker DL, Kieckbusch T. Patellofemoral complications after posterior‐stabilized total knee arthroplasty: a comparison of 2 different implant designs. J Arthroplasty, 2002, 17: 422–426. [DOI] [PubMed] [Google Scholar]
- 7. Mähringer‐Kunz A, Efe T, Fuchs‐Winkelmann S, et al Bleeding in TKA: posterior stabilized vs. cruciate retaining. Arch Orthop Trauma Surg, 2015, 135: 867–870. [DOI] [PubMed] [Google Scholar]
- 8. Meneghini RM, Stefl MD, Hodge WA, Banks SA. A cam‐post mechanism is no longer necessary in modern primary Total knee arthroplasty. J Knee Surg, 2019, 32: 710–713. [DOI] [PubMed] [Google Scholar]
- 9. Tanifuji O, Sato T, Kobayashi K, et al Three‐dimensional in vivo motion analysis of normal knees employing transepicondylar axis as an evaluation parameter. Knee Surg Sports Traumatol Arthrosc, 2013, 21: 2301–2308. [DOI] [PubMed] [Google Scholar]
- 10. Bae DK, Cho SD, Im SK, Song SJ. Comparison of midterm clinical and radio‐ graphic results between total knee arthroplasties using medial pivot and posterior‐stabilized prosthesis‐a matched pair analysis. J Arthroplasty, 2016, 31: 419–424. [DOI] [PubMed] [Google Scholar]
- 11. Samy DA, Wolfstadt JI, Vaidee I, Backstein DJ. A retrospective comparison of a medial pivot and posterior‐stabilized Total knee arthroplasty with respect to patient‐reported and radiographic outcomes. J Arthroplasty, 2018, 33: 1379–1383. [DOI] [PubMed] [Google Scholar]
- 12. Yuan D, Zhang QS, Zhang K, et al Total knee arthroplasty using a medial pivot or posterior cruciate‐stabilizing prosthesis in Chinese patients. J Knee Surg, 2020, 33: 892–898. [DOI] [PubMed] [Google Scholar]
- 13. Lee QJ, Wai Yee EC, Wong YC. No difference in patient preference for medial pivot versus posterior‐stabilized design in staged bilateral total knee arthroplasty: a prospective study. Knee Surg Sports Traumatol Arthrosc, 2020;Jan 28 10.1007/s00167-020-05867-z. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 14. Lin Y, Chen X, Li L, Li Z, Zhang Y, Fan P. Comparison of patient satisfaction between medial pivot prostheses and posterior‐stabilized prostheses in Total knee arthroplasty. Orthop Surg, 2020, 12: 836–842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kulshrestha V, Sood M, Kanade S, Kumar S, Datta B, Mittal G. Early outcomes of medial pivot Total knee arthroplasty compared to posterior‐stabilized design: a randomized controlled trial. Clin Orthop Surg, 2020, 12: 178–186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Soysal P, Stubbs B, Lucato P, et al Corrigendum toInflammation and frailty in the elderly: a systematic review and meta‐analysis” [Ageing Res Rev. 31 (2016) 1–8]. Ageing Res Rev, 2017, 35: 364–365. [DOI] [PubMed] [Google Scholar]
- 17. Langkilde A, Jakobsen TL, Bandholm TQ, et al Inflammation and post‐operative recovery in patients undergoing total knee arthroplasty‐secondary analysis of a randomized controlled trial. Osteoarthritis Cartilage, 2017, 25: 1265–1273. [DOI] [PubMed] [Google Scholar]
- 18. Nazem K, Motififard M, Yousefian M. Variations in ESR and CRP in total knee arthroplasty and total hip arthroplasty in Iranian patients from 2009 to 2011. Adv Biomed Res, 2016, 5: 148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kellgren JH, Lawrence JS. Radiological assessment ofosteo‐arthrosis. Ann Rheum Dis, 1957, 16: 494–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. De Muylder J, Victor J, Cornu O, et al Total knee arthroplasty in patients with substantial deformities using primary knee components. Knee Surg Sports Traumatol Arthrosc, 2015, 23: 3653–3659. [DOI] [PubMed] [Google Scholar]
- 21. Klein GR, Hartzband MA. Surgical approaches to total knee arthroplasty In: Par‐vizi J, Klatt BA, eds. Essentials in Total Knee Arthroplasty. Thorofare, NJ: Slack, Inc, 2011; 65–71. [Google Scholar]
- 22. Reid JB 3rd, Guttmann D, Ayala M, Lubowitz JH. Minimally invasive surgery‐total knee arthroplasty. Art Ther, 2004, 20: 884–889. [DOI] [PubMed] [Google Scholar]
- 23. Pérez de la Cruz S. Effectiveness of aquatic therapy for the control of pain and increased functionality in people with Parkinson's disease: a randomized clinical trial. Eur J Phys Rehabil Med, 2017, 53: 825–832. [DOI] [PubMed] [Google Scholar]
- 24. Benner RW, Shelbourne KD, Bauman SN, Norris A, Gray T. Knee osteoarthritis: alternative range of motion treatment. Orthop Clin North Am, 2019, 50: 425–432. [DOI] [PubMed] [Google Scholar]
- 25. Honsawek S, Deepaisarnsakul B, Tanavalee A, et al Relationship of serum IL‐6, C‐reactive protein, erythrocyte sedimentation rate, and knee skin temperature after total knee arthroplasty: a prospective study. Int Orthop, 2011, 35: 31–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Dalury DF, Barrett WP, Mason JB, Goldstein WM, Murphy JA, Roche MW. Midterm survival of a contemporary modular total knee replacement: a multicentre study of 1970 knees. J Bone Joint Surg Br, 2008, 90: 1594–1596. [DOI] [PubMed] [Google Scholar]
- 27. Ranawat CS, Komistek RD, Rodriguez JA, Dennis DA, Anderle M. Fluoroscopic analyses of cruciate‐retaining and medial pivot knee implants. Clin Orthop Relat Res, 2003, 410: 139–147. [DOI] [PubMed] [Google Scholar]
- 28. Bae DK, Cho SD, Im SK, Song SJ. Comparison of midterm clinical and radiographic results between total knee arthroplasties using medial pivot and posterior‐stabilized prosthesis‐a matched pair analysis. J Arthroplasty, 2016, 31: 419–424. [DOI] [PubMed] [Google Scholar]
- 29. Xu C, Guo H, Qu P, Fu J, Kuo FC, Chen JY. Preoperatively elevated serum inflammatory markers increase the risk of periprosthetic joint infection following total knee arthroplasty in patients with osteoarthritis. Ther Clin Risk Manag, 2018, 14: 1719–1724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Sanchez‐Ramirez DC, van der Leeden M, van der Esch M, et al Elevated C‐reactive protein is associated with lower increase in knee muscle strength in patients with knee osteoarthritis: a 2‐year follow‐up study in the Amsterdam osteoarthritis (AMS‐OA) cohort. Arthritis Res Ther, 2014, 16: R123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Zhang P, Liang Y, He J, Fang Y, Chen P, Wang J. Timing of tourniquet release in total knee arthroplasty: a meta‐analysis. Medicine (Baltimore), 2017, 96: e6786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Huang Z, Ma J, Zhu Y, et al Timing of tourniquet release in total knee arthroplasty. Orthopedics, 2015, 38: 445–451. [DOI] [PubMed] [Google Scholar]
- 33. McCarthy Deering E, Hu SY, Abdulkarim A. Does tourniquet use in TKA increase postoperative pain? A systematic review and meta‐analysis. Clin Orthop Relat Res, 2019, 477: 547–558. [DOI] [PMC free article] [PubMed] [Google Scholar]


