Abstract
The goal of this paper is to explore the ability of the human female urogenital sinus immediately below the bladder (proximal urethra) to undergo prostatic development in response to dihydrotestosterone (DHT). To establish this idea, xenografts of human fetal female proximal urethra were grown in castrated nude mouse hosts receiving a subcutaneous DHT pellet. To verify the prostatic nature of the resultant glands, DHT-treated human fetal female urethral xenografts were compared with human fetal prostatic xenografts (derived from male specimens) grown in untreated and DHT-treated castrated mouse hosts and human fetal female proximal urethral xenografts grown in untreated castrated hosts. The resultant glands observed in DHT-treated human fetal female proximal urethra expressed 3 prostate-specific markers, NKX3.1, prostate specific antigen and prostatic acid phosphatase as well as the androgen receptor. Glands induced by DHT exhibited a protein expression profile of additional immunohistochemical markers (seven keratins, RUNX1, ESR2, TP63 and FOXA1) consistent with the unique spatial pattern of these proteins in prostatic ducts. Xenografts of human fetal female proximal urethra grown in DHT-treated hosts expressed one of the salient features of prostatic development, namely androgen responsiveness. The experimental induction of prostatic differentiation from human fetal female proximal urethra makes possible future in-depth analysis of the molecular pathways directly involved in initiation of human prostatic development and subsequent epithelial differentiation, and more important whether the molecular pathways involved in human prostatic development are similar/identical versus different from that in murine prostatic development.
Keywords: Female prostate, urethra, androgen receptor, NKX3.1, development
Introduction
The prostate of all species develops from the urogenital sinus (UGS), a derivative of the ventral division of the cloaca (Liaw et al, 2018; Yamada et al, 2003). Solid prostatic buds emerge from human urogenital sinus epithelium (UGE) immediately below the developing bladder at about 10 weeks of gestation (Cunha et al, 2018). Prostatic development occurs in five stages: pre-bud, bud initiation, bud elongation, ductal branching and ductal canalization followed by cytodifferentiation of luminal and basal epithelial cells (Cunha et al, 2018). In the pre-bud stage the UGE is organized as an epithelial tube surrounded by urogenital sinus mesenchyme (UGM) which induces and specifies prostatic development (Cunha et al, 1987).
Prostatic development is dependent upon androgens which act via androgen receptors in the surrounding UGM (Cunha et al, 1987; Marker et al, 2003). With few species exceptions, females do not form prostate presumably due to insufficient androgen levels. The most compelling observation emphasizing the critical role of androgen action in prostatic development is the absence or minimal development of prostate in animals and humans secondary to absence or genetic defects in the gene encoding the androgen receptor (Ohno, 1979; Wilson, 1987; Wilson et al, 1983) and/or defects in androgen metabolism secondary to 5-alpha reductase type 2 deficiency (Wilson et al, 1993).
The embryonic UGS is a sexually indifferent structure that in males forms the bladder, urethra, prostate, and bulbourethral glands. In the course of development, the human fetal prostate can be recognized as a distinct bulge below the bladder in wholemount images (Fig. 1). The female UGS forms the bladder, urethra and contributes to the vagina (Robboy et al, 2017). Wholemount imaging of the developing female urogenital tract reveals an absence of the distinctive prostatic bulge (Fig. 1). We propose that the segment of the human female fetal urethra immediately below the bladder is anatomically homologous to the prostatic urethra. We designate this region of female developmental anatomy as “proximal urethra”. Given the likely homology between male and female urogenital sinuses immediately below the bladder, in this paper we expand on a previous preliminary observation (Cunha et al, 2018) to confirm that the human fetal female proximal urethra can form prostate when exposed to the androgen, dihydrotestosterone (DHT), and express prostate-specific markers.
Figure 1.
Wholemounts of the bladder, prostate and urethras of developing human male and female fetuses from 10 to 21 weeks of gestation. Note the prostatic bulge immediately below the bladder (top row) and its absence in female specimens (bottom row).
Materials and Methods
Human fetal prostates (9–21 weeks of gestation) and human female proximal urethras (11–14 weeks of gestation) were collected from specimens devoid of patient identifiers after elective termination of pregnancy (Committee on Human Research at UCSF, IRB# 12–08813). Gestational age of specimens was estimated using heel-toe length as described previously (Drey et al, 2005; Robboy et al, 2017). Specimens were fixed in formalin and processed for hematoxylin and eosin (H&E) or immunohistochemical staining. This study is based upon the analysis of 12 human fetal bladder/prostate wholemounts (also used for histologic analysis [Fig. 1]), 12 human fetal female proximal urethra wholemounts (Fig. 1), 15 xenografts of human fetal prostate (8 DHT-treated and 7 controls aged 12.5 to 14 weeks of gestation), and 14 xenografts of human female proximal urethra (8 DHT-treated and 6 controls) aged 10 to 14.5 weeks of gestation.
Human fetal specimens fixed in 10% buffered formalin were embedded in paraffin and serially sectioned at 7μm. Every 20th section was stained with H&E to assess histology. Intervening paraffin sections were immunostained with antibodies to a variety of proteins (Table 1) as described previously (Rodriguez et al, 2012). Immunostaining was detected using horseradish peroxidase-based Vectastain kits (Vector Laboratories, Burlingame, CA). For negative controls the primary antibodies were omited.
Table 1.
Antibodies used in this study
| Antibody | Source | Catalogue # | Concentration |
|---|---|---|---|
| α-actin | Sigma | A2547 | 1/2000 |
| GTF3C2 | Abcam | AB53218 | 1/100 |
| Estrogen receptor α (ESR1) | Abcam | Ab16660 | 1/100 |
| Estrogen receptor β (ESR2) | Leica | NCL-ER-beta | 1/50 |
| Keratin 6 | Acris Antibodies | AM21068PU-S | 1/200 |
| Keratin 7 | E.B. Lane | LP1K | 1/10 |
| Keratin 8 | E.B. Lane* | LE41 | 1/10 |
| Keratin 10 | Dako | M7002 | 1/50 |
| Keratin 14 | BioGenex | LL002 | 1/100 |
| Keratin 15 | Sigma | Sab4501658 | 1/50 |
| Keratin 19 | E.B. Lane* | LP2K | 1/10 |
| TP63 | Santa Cruz Biotechnology | Sc-8343 | 1/100 |
| Androgen receptor | Genetex | GTX62599 | 1/100 |
| RUNX1 | Abcam | Ab92336 | 1/100 |
| Uroplakin 1 | T. T.Sun** | 1/100 | |
| PAX2 | Abcam | Ab150391 | 1/50 |
| FOXA1 | Atlas Antibodies | HPA050505 | 1/500 |
| NKX3.1 | Santa Cruz Biotechnology | SC-393190 | 1/100 |
| PSA | Fisher Scientific | 01-673-879 | 1/1 |
| PAP | ThermoFisher | MA5-13078 | 1/3000 |
Institute of Medical Biology, Singapore
New York University, New York
Abbreviations: PSA= Prostate specific antigen, PAP= Prostatic acid phosphatase
For xenograft studies, 15 human fetal prostates at 12.5 to 14 weeks of gestation were surgically isolated from the bladder and the pelvic urethra and then transected in the midline to yield right and left halves which were transplanted under the renal capsules of castrated male athymic nude mice (CD-1 NU/NU, Charles River Laboratories, Wilmington, MA) as previously described (Cunha and Baskin, 2016). The IACUC committee at UCSF approved all grafting procedures. The castrated mouse hosts received a 20mg subcutaneous pellet of dihydrotestosterone (DHT) (A8380, Sigma-Aldrich, St. Louis, MO, USA) or were untreated (control), and thus were androgen deficient. Our selection of DHT, instead of testosterone, eliminates the possibility of aromatase-mediated conversion of testosterone to estradiol. Grafts were grown for 1 to 2 months, at which time the hosts were euthanized, and the grafts harvested and processed as described above for histology and immunohistochemistry.
Segments of 10 to 14.5 week human fetal female urethra immediately below the bladder, which is considered to be the anatomic homologue of the prostatic urethra, were also grafted into castrated male hosts that were either untreated (N=6) or DHT-treated (N=8) to determine whether DHT could induce prostatic development in the female proximal urethra. After 1 or 2 months of in vivo growth, the grafts were harvested and processed as above for histology and immunohistochemistry. This study confirms and extends an earlier preliminary study (Cunha et al, 2018).
C. Results
To determine whether human fetal female urethra can be induced by DHT to undergo prostatic development, the histologic and immunohistochemical profile of the donor human fetal female urethra was compared with that of the human fetal prostate. Moreover, the immunohistochemical profile of DHT-treated xenografts of human fetal prostate and human fetal female proximal urethra was assessed to determine whether the differentiation process in the xenografts conformed to that of normal prostatic development. To this end, we first defined the gross anatomy of male and female urogenital tracts, and then assessed the response of male and female target tissues to DHT.
A. Gross anatomy.
The ontogeny of human fetal prostate is depicted in gross anatomical wholemounts in figure 1 from 10 to 21 weeks of gestation. The human fetal prostate is recognized as a distinct bulge immediately below the urinary bladder (Fig. 1 top row). In contrast, such a bulge is absent in female specimens (Fig 1, bottom row). Human fetal female bladder and urethra are shown at higher magnification in figure 2. Note the absence of the prostatic bulge (Fig. 2A). The region between the white lines in figure 2A is designated as “female proximal urethra”. The histology of this region is depicted in figure 2B and shows the epithelium defining the urethral lumen and associated epithelial outpouchings that may represent urethral glands (glands of Littre) or may subsequently canalize to expand the urethral lumen.
Figure 2.
(A) Wholemount of a 13-week female bladder and urethra. The white lines designate the “proximal urethra”, whose histology is shown in (B). Note epithelial outpouchings (arrowheads). Adapted from Cunha et al 2018 with permission.
B. Histology and Immunohistochemistry of developing human prostate.
To determine whether human female proximal urethra can be induced by DHT to undergo prostatic development, normal prostatic differentiation was assessed via a constellation of immunohistochemical stains to define a set of proteins that are associated with prostatic development. Given the period of exposure of xenografts of human female proximal urethra to DHT (see below), it was appropriate to assess the process of prostatic development from the pre-bud stage (9 weeks) to advanced ductal elongation, branching, and canalization resulting in differentiation of definitive luminal and basal epithelial cells at 21 weeks of gestation. Morphological and immunohistochemical features of normal (non-grafted) human prostatic differentiation from pre-bud to differentiation of canalized ducts lined with luminal and basal cells are summarized in Table 2. Given that the prostatic urethra with associated prostatic ducts as well as the human female proximal urethra are both derived from similar (if not identical) portions of the UGS, we found that the immunohistochemical profile of the developing prostatic urethra was virtually identical to that of the developing female urethra (Table 2). However, the spatial ontogeny of marker expression in developing prostatic ducts is unique and highlights a critical distinction between the immunohistochemical profile of developing human prostatic ducts versus human fetal prostatic urethra and human female fetal urethra. For this reason, the unique ontogeny of immunohistochemical profile of developing prostatic ducts is emphasized.
Table 2.
Epithelial differentiation markers during human prostatic and female proximal urethral development 9 to 21 weeks of gestation (non-grafted specimens)
| Bud elongation & branching | Epithelial cells of canalized ducts | Prostatic urethra | Female proximal urethra | |||||
|---|---|---|---|---|---|---|---|---|
| Pre-bud # | Solid buds | Solid | Canalized | Basal cells | Luminal cells | |||
| Markers | ||||||||
| Keratin 6 | −/+ | + | + | + | + | − | + | + |
| Keratin 7 | −/+ | −/+ | −/+ | + | − | + | + | + |
| Keratin 8 | + | −/+ | −/+ | + | − | + | + | + |
| Keratin 10 | − | − | − | − | − | − | − | − |
| Keratin 14 | − | − | − | − | − | − | − | − |
| Keratin 15 | +/− | + | + | + | + | − | + | + |
| Keratin 19 | + | −/+ | + | + | − | + | + | + |
| RUNX1 | + | + | + | + | − | + | + | + |
| TP63 | + | + | + | + | + | − | + | + |
| FOXA1 | + | + | + | + | + | + | + | + |
| ESR1 | − | − | − | − | − | − | − | − |
| ESR2 | + | + | + | + | + | + | + | + |
| AR | + | −/+ | −/+ | + | − | + | + | + |
| NKX3.1 | − | − | − | − | − | − | − | − |
| Uroplakin | + | − | − | −/+ | − | − | + | + |
| PSA | − | − | − | − | − | − | − | − |
| PAP | − | − | − | +* | − | +* | − | − |
Pre-bud=UGE tube
21 week specimen only.
Abbreviations: PSA= Prostate specific antigen, PAP= Prostate acid phosphatase, AR=androgen receptor.
PAP was only observed in a 21-week human fetal prostatic specimen.
Figures 3–5 focus on several of the immunohistochemical features of human fetal prostate. Perhaps the most interesting area within the developing human prostate is the zone of ductal canalization in which solid prostatic ducts develop a lumen lined with luminal epithelial cells underlain by a continuous layer of basal epithelial cells. Such zones can be seen in specimens 11 to 21 weeks of gestation, indicating that the ductal canalization process occurs over a wide developmental period. From 11 to 21 weeks of gestation, elongation of solid ducts and their branching continues to occur distally (near the capsule), followed by a wave of ductal canalization which begins proximally (near the urethra) and extends distally into the branched ductal network (Cunha et al, 2018). The canalization process culminates in the differentiation of definitive luminal and basal epithelial cells (as well as neuroendocrine cells). Frequently, the various stages in the canalization process can be seen in a single section containing solid ducts, canalizing ducts and mature fully canalized ducts with definitive luminal and basal cells. Across the full range of epithelial proteins, a common pattern has emerged.
Figure 3.
Transverse sections of a 9-week urogenital sinus stained for ESR2 (A), androgen receptor (AR) (B), TP63 (C) and uroplakin (D). Epithelial ERβ stain is seen in the mid-dorsal UGE and UGM. Note the differential luminal versus basal expression for AR (B), TP63 (C) and uroplakin (D). FMD=fused Mullerian ducts, prostatic utricle. Adapted from Cunha et al 2018 with permission.
Figure 5.
Immunohistochemistry of human fetal prostate. Note luminal versus basal cell expression in canalized ducts (A-K). For solid ducts (E, black arrowhead; F. back arrowheads; J, black arrowhead)), proteins may be either undetectable (F, black arrowheads & I, asterisk), may be broadly and homogeneously expressed throughout solid prostatic ducts (E, J), or may contain a mixture of epithelial cells positive and negative for a particular protein (E, F, H, I, J, K, green arrowheads). A-C & E= 15-weeks, D=19 weeks, F=12 weeks, J=14 weeks G-I & K= 21 weeks.
The starting point for prostatic development is the pre-bud urogenital sinus, which is essentially prostatic urethra prior to prostatic bud initiation. At this stage (8–9 weeks), the male UGE is weakly ESR2−positive with ESR2 staining seen in the mid-dorsum of the UGE (Fig. 3A) and in mid-dorsal UGM between the UGE and the epithelia of the Wolffian ducts (WD) and fused Mullerian ducts (FMD, prostatic utricle). Androgen receptor (AR) and uroplakin were confined to the luminal epithelial layer (Figs. 3B & D), whereas TP63 was confined to the basal epithelial layer (Fig. 3C). A comparable region of the human female proximal urethra has an identical immunohistochemical profile (Table 2, not illustrated).
In older human fetal prostatic specimens (11–21 weeks) containing solid and canalized prostatic ducts, the androgen receptor (AR) was expressed broadly in the mesenchyme surrounding the epithelial ducts, in luminal cells of the urethra (Fig. 4A1–A2, red arrowheads), in well-differentiated luminal cells of canalized prostatic ducts (Fig. 4A1, red arrowheads), but not in basal epithelial cells of canalized ducts (Fig. 4A1–2). Androgen receptor immunostaining was also not detected in solid prostatic ducts (Fig. 4A2a & b). In zones where canalization was just initiating, undifferentiated luminal epithelial cells were also AR-negative (Fig. 4A2c, green arrowheads), suggesting that as lumen formation occurs, immature AR-negative luminal epithelial cells initially appear that subsequently express AR (Fig. 4A1–A2, red arrowheads). A similar pattern of expression is seen for RUNX1; luminal epithelial cells in canalized ducts are RUNX1-positive (Fig. 4B, red arrowheads), while epithelial cells of solid prostatic ducts are RUNX1-negative (Fig. 4B, green arrowheads).
Figure 4.
(A) Immunostaining for androgen receptor (AR) and (B) RUNX1 of a 14-week human fetal prostate. Images in both (A) and (B) are taken from a region within the prostate of transition between solid and canalizing ducts. The pattern of expression across this zone of canalization is similar for both proteins. Solid ducts (a-c in A) are negative for AR and RUNX1 (B), while canalized ducts are lined by AR- and RUNX1-positive epithelial cells (red arrowheads). Green arrowheads denote solid or canalized ducts lacking AR (A1 & A2) or RUNX1 (B) expression.
A similar pattern of expression in solid versus canalized prostatic ducts was seen for several keratins and TP63. For relatively mature canalized ducts, keratins 7 (Figs. 5B& I), 8 (Fig. 5C) and 19 (Fig. 5H) were expressed in luminal cells. Likewise in canalized ducts (but in obverse), keratin 6 (Fig. 5A), keratin 15 (Fig. 5G) and TP63 (Fig. 5D) were expressed in basal epithelial cells but were broadly expressed throughout solid prostatic ducts (Figs. 5E, black arrowhead, J–K, asterisks). For keratin 7, solid ducts (Fig. 5F, black arrowheads)were either negative or contained keratin-7-positive cells in the central core of solid ducts (Fig. 5F, green arrowhead), suggesting variability in the differentiation state, a pattern shared with keratins 8 and 19 (not illustrated). The pattern that emerges from observations of several proteins is a protracted differentiation process that begins with solid prostatic buds that are initially either negative or positive depending on the protein during the periods examined (9–21 weeks). Subsequently, in solid prostatic ducts protein expression becomes localized to central core epithelial cells for proteins destined to become luminal epithelial cells (Figs. 5F, H, I, keratin 7, keratin 19, keratin 8). For proteins destined to become basal cells, central core epithelial cells are devoid of immunohistochemical expression, while protein expression is prominent in basally located epithelial cells (Figs. 5D, E, G, J. K, TP63, keratin 6, keratin 15). The canalization/differentiation process culminates in restricted expression of proteins unique to luminal and basal cells. An exception to this pattern is seen for FOXA1, an endodermal marker and keratin 19, both of which are expressed in all cells of the prostatic urethra and in solid and canalized prostatic ducts (Table 2, not illustrated). While the developing prostatic urethra and the developing female proximal urethra express the same spectrum of proteins (Table 2), it is important to recognize the unique ontogenic spatial pattern of marker proteins in developing prostatic ducts.
Keratins 10 and 14, NKX3.1, and ERα were undetectable in developing prostate from 9 to 21 weeks of gestation (Table 2, not illustrated). Prostatic acid phosphatase (PAP) was detected luminal prostatic cells at 21 weeks, while PSA was not expressed at this stage (Fig. 6). Keratin 14 is a normal feature of adult prostatic basal epithelial cells (Hudson et al, 2001). The unexpected absence of keratin 14 immunostaining (Table 2) in developing human prostatic ducts may be a function of differentiation state of the epithelium.
Figure 6.
Section of human fetal prostate at 21 weeks of gestation immunostained for prostatic acid phosphatase (PAP) and prostatic specific antigen (PSA).
C. Histology and immunohistochemistry of the developing human female proximal urethra
The immunohistochemical profile of the developing human female proximal urethra is virtually identical to that of the developing human prostatic urethra and to a large extent prostatic ducts as well. Many of the keratins, as well as TP63, RUNX1, FOXA1, ESR2 and uroplakin are expressed in epithelium of both the developing human prostatic urethra and female proximal urethra (Tables 2 & 3). The most distinguishing feature between human fetal female proximal urethra and developing human fetal prostate is the formation of extensively branched ductal networks in the developing prostate and their absence in the developing human female proximal urethra. Epithelial outpouchings of the developing human female proximal urethra are present from at least 12 weeks of gestation (and most probably earlier) (Fig. 2B) and may represent rudiments of urethral glands (glands of Littre), although definitive differentiated urethral glands were not observed in the specimens examined. Urethral glands are diminutive in size and vastly simpler in morphology relative to the ductal-acinar networks of human prostate (Warick and Williams, 1973). The branched network of solid and canalized ducts of the developing prostate has a unique ontogenic pattern of expression of protein markers as described above not seen in the female proximal urethra.
Table 3.
Epithelial differentiation markers in non-grafted human fetal prostates versus non-grafted human fetal female proximal urethra.
| Human fetal prostatic urethra and ducts | Human female fetal proximal urethra | |
|---|---|---|
| Markers | ||
| Keratin 6 | + | + |
| Keratin 7 | + | + |
| Keratin 8 | + | + |
| Keratin 10 | − | − |
| Keratin 14 | − | − |
| Keratin 15 | + | −/+## |
| Keratin 18 | + | + |
| Keratin 19 | + | + |
| RUNX1 | + | + |
| TP63 | + | + |
| Uroplakin | −/+(#) | + |
| FOXA1 | + | + |
| ESR1 | − | − |
| ESR2 | + | + |
| AR | + | + |
| NKX3.1 | − | − |
| PSA | − | − |
| PAP | +* | − |
uroplakin is expressed in the prostatic urethra and not in prostatic ducts.
11-week specimen= K15-negative, 12-13 week specimen= K15-positive.
PAP was seen only in canalized ducts of 21-week human fetal prostate.
Abbreviations: PAP= Prostate acid phosphatase, PSA= Prostate specific antigen, AR= androgen receptor.
The androgen receptor is a feature common to both the prostatic urethra and the developing human female proximal urethra (Figs. 3B, 4A & 7). The epithelium of the prostatic urethra exhibits AR in luminal cells (Figs. 3B, 4A, 7B) as does the epithelium lining the female proximal urethra (Fig. 7A). However, the ontogeny of AR in prostatic ducts is a complicated and unique process. As solid prostatic buds emerge from the UGE (prostatic urethra), AR-positive epithelial cells are observed in the central core of the newly emergent prostatic buds at 11–12 weeks (Fig. 7B, asterisk). As newly emergent prostatic buds canalize, AR-positive luminal cells are apparent (Fig. 7B, arrowheads). Branched solid prostatic buds that have extended peripherally toward the capsule, are AR-negative (Figs. 4A2a–c & 7C, asterisk), but as ductal canalization occurs in a wave from the urethra outward, central core epithelial cells become AR-positive, and then the luminal epithelial cells express AR (Figs. 4A1–A2 & 7C). This complicated process of AR ontogeny occurs over an extended period from at least 11 to 21 weeks (Figs. 4 and 7B) and probably later, and does not occur in the female proximal urethra, even though solid outpouchings of the female proximal urethra express a core of scattered AR-positive cells (Fig. 7A, arrowheads).
Figure 7.
(A) Transverse section of a 12-week human female proximal urethra with two solid epithelial cords (arrowheads) that have extended peripherally from the urethral epithelium. (B) Transverse section of a 12-week human fetal prostate. New emerged prostatic buds have a central core of AR-positive epithelial cells (asterisk in B). Elongated canalized prostatic ducts (arrowheads) contain AR-positive luminal cells and AR-negative basal cells. Also note the distal solid tip of a prostatic duct that is AR-negative (green arrowheads). (C) Section of a 21-week prostate near the capsule where solid AR-negative prostatic ducts (*) are branching. Note AR-positive luminal cells.
D. Xenograft studies
1. Xenografts of human fetal prostate grown in untreated and DHT-treated castrated hosts.
a. General observations.
The rationale for including human fetal prostatic xenografts grown under androgenic and androgen-deprived conditions is that one unique feature of human fetal prostate is its responsiveness to androgens. Accordingly, if human fetal female proximal urethra can be induced to form prostate in response to DHT, the induced glands should exhibit responsiveness to androgenic conditions and manifest an ontogenic pattern of protein expression consistent with androgen-dependent prostatic differentiation. To assess androgen-dependent prostatic development, human fetal prostatic xenografts were grown in (a) untreated castrated athymic male mouse hosts (N=7) (androgen deficient) or in (b) DHT-treated castrated hosts (N=8). Many of the human fetal prostatic xenografts were represented as right-left pairs that were grown in untreated and DHT-treated hosts. These prostatic xenograft pairs were aged 11–14 weeks of gestation at the time of grafting, and thus prostatic buds were present at the time of grafting, and had an immunohistochemical profile consistent with this age range (see text and Tables) as described previously (Cunha et al, 2018). For each untreated/DHT-treated pair, the number of ducts observed in the DHT-treated xenografts greatly exceeded that of the androgen-deficient controls (Figs. 8A–B & D & F). Moreover, many of the ducts in the DHT-treated xenografts were canalized and contained differentiated luminal and basal epithelial cells (Figs. 8A, C, E, 9A & C), while these features were under-represented/absent in the control prostatic xenografts (Figs. 8B, F–G, 9B & D).
Figure 8.
Xenografts of a 14-week human fetal prostate grown for 8 weeks in DHT (A, C, D-E) and untreated (control) castrated athymic male mice (B, F-G). Note the substantial increase in prostatic ducts, many of which are canalized, in DHT-treated (A & D) versus the control (B & F) xenografts. (C) NKX3.1 (brown stain) is apparent in a subset of epithelial cells in canalized, but not in solid ducts (C, *) in prostatic xenografts treated with DHT. (D-G) Androgen receptor immunostained sections of a 14-week human fetal prostate grown for 4 weeks in a DHT-treated castrated mouse host (D-E) and an untreated (control) castrated mouse host (F-G). Note abundant AR-positive stromal cells and AR-positive luminal epithelial cells in the DHT-treated specimen (D-E) (but not in solid ducts (E, *), versus the absence of AR in the epithelium and reduction in AR-positive stromal cells in the control fetal prostatic graft (F-G) (* in G denotes a solid duct). AR-positive stromal cells are seen in the lower left quadrant of (G) far removed from the epithelium. Adapted in part from Cunha et al 2018 with permission.
Figure 9.
Human prostatic xenografts grown in DHT-treated (A & C) and androgen-deficient (B & D) hosts immunostained for PAP and PSA. Ages of the specimens at the time of grafting is given and ranges from 11 to 13 weeks. (A & C) Human fetal prostatic xenografts at the ages specified grown in DHT-treated hosts stained for PAP (A) and PSA (C). (B & D) Human fetal prostatic xenografts at the ages specified grown in untreated hosts stained for PAP (B) and PSA (D). Note canalized ducts in prostatic xenografts grown in DHT-treated hosts (A & C), and the absence of canalized ducts and PAP and PSA staining in prostatic xenografts grown in untreated hosts (B & D).
b. Immunohistochemical observations
The profile of the epithelial differentiation markers clearly segregated into two categories: androgen-independent and androgen-dependent. The first 12 epithelial markers (keratin 6 through ESR2) in Table 4 were androgen-independent as the expression profile was identical in prostatic grafts grown in both untreated and DHT-treated hosts. The last 4 epithelial markers in Table 4 (AR, NKX3.1, PSA and PAP) were expressed only in DHT-treated hosts and thus were androgen-dependent. Expression of epithelial AR and PAP in DHT-treated prostatic grafts greatly exceeded that in prostatic grafts grown in untreated hosts. NKX3.1 was only expressed in epithelium of prostatic grafts grown in DHT-treated hosts, albeit in 4/8 prostatic xenografts grown in DHT-treated castrated hosts, specifically in a subset of mostly luminal epithelial cells in canalized ducts, but never in solid ducts (asterisks, Fig. 8C, Table 4). Androgen receptor was broadly expressed in luminal epithelial cells of canalized prostatic ducts and in the surrounding stromal cells of DHT-treated xenografts, but was not expressed in solid prostatic ducts in the same section (Figs. 8D–E). Androgen receptor was minimally expressed in prostatic xenografts grown in untreated castrated hosts (androgen deficient) (Fig. 8F–G). Examination of non-grafted human fetal prostates of a comparable age of the xenografts indicated that epithelial and mesenchymal AR was present at (and before) the time of grafting, yet AR was absent/greatly diminished in prostatic xenografts grown in androgen-deficient hosts, suggesting that continued AR expression is dependent upon the presence of androgens. PAP was detected in canalized (but not solid) ducts of DHT-treated human fetal prostatic xenografts and never in prostatic xenografts grown in untreated hosts (Figs. 9A–B). PSA was weakly expressed in DHT-treated human fetal prostatic xenografts and never in prostatic xenografts grown in untreated hosts (Figs. 9C–D). Non-grafted prostatic specimens strongly expressed AR in both the epithelium and stroma (Figs. 3, 4 & 7B), while PAP (but not PSA and NKX3.1) was detected only in the 21-week non-grafted prostatic specimen (Fig. 6A and Table 4), perhaps suggesting different serum androgen thresholds or other factors required for expression of these proteins. It is notable that the “11 week” prostatic graft seen in figures 9B and 9D grown under androgen-deficient conditions (untreated castrated host) failed to form prostatic ducts. Perhaps this specimen was younger (pre-bud) than we estimated at the time of grafting.
Table 4.
Epithelial differentiation markers in non-grafted human fetal prostates and in human fetal prostatic xenografts grown in castrated DHT-treated and untreated (androgen-deficient) hosts.
| Non-grafted prostates | DHT-treated prostatic xenografts | Untreated prostatic xenografts | |
|---|---|---|---|
| Markers | |||
| Keratin 6 | + | + | + |
| Keratin 7 | + | + | + |
| Keratin 8 | + | + | + |
| Keratin 10 | − | − | − |
| Keratin 14 | − | − | − |
| Keratin 15 | + | + | + |
| Keratin 19 | + | + | + |
| RUNX1 | + | + | + |
| TP63 | + | + | + |
| FOXA1 | + | + | + |
| ESR1 | − | − | − |
| ESR2 | + | + | + |
| AR | + | + | − |
| NKX3.1 | − | + | − |
| PSA | − | + | − |
| PAP | −/+* | + | − |
Abbreviations: PSA= Prostate specific antigen, PAP= Prostate acid phosphatase, AR= androgen receptor.
PAP was detected in only the oldest (21-week) specimen.
(2). Xenografts of human fetal female proximal urethra grown in untreated and DHT-treated castrated hosts.
a. General observations.
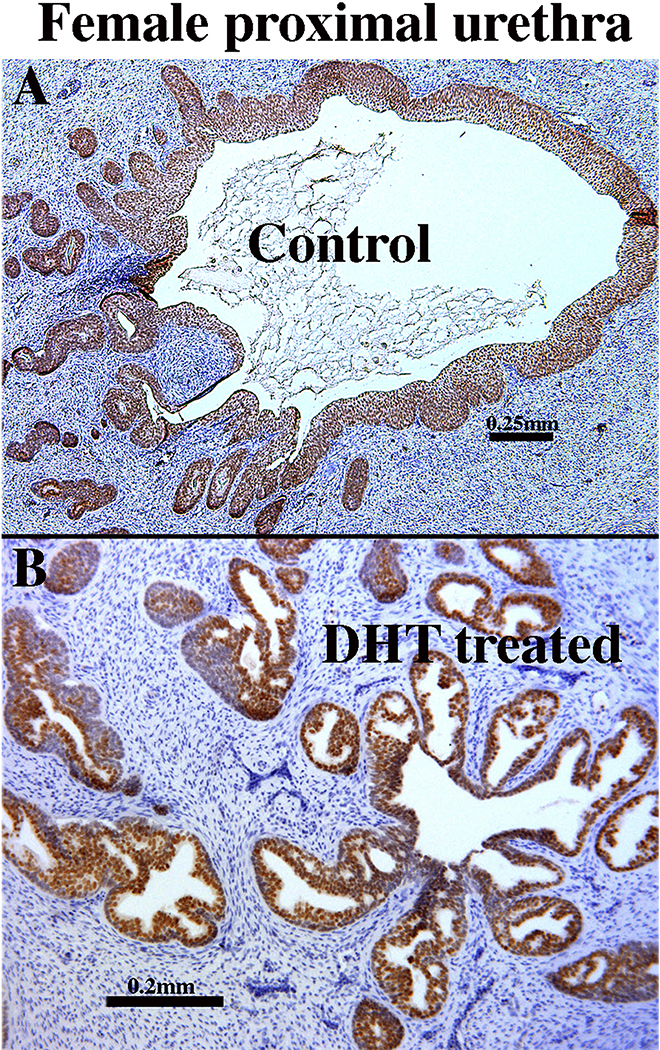
Following 4 or 8 weeks of in vivo growth, human female proximal urethral xenografts grown in androgen-deficient (untreated castrated) hosts maintained a histologic (Fig. 10A) and immunohistochemical profile consistent with a female urethral phenotype (Table 5). Xenografts of human female proximal urethras grown in untreated castrated hosts contained a lumen lined by stratified uroplakin-positive epithelium and associated solid epithelial outgrowths (Fig. 10A, Table 5) and an immunohistochemical profile identical to that of non-grafted human female proximal urethra and human fetal prostatic xenografts grown in untreated castrated hosts (not illustrated, see Tables 2–4). In contrast, xenografts of human female proximal urethra grown in DHT-treated hosts exhibited a histologic profile consistent with developing and differentiating prostate and thus contained an abundance of solid and canalized ducts containing luminal and basal epithelial cells (Fig. 10B).
Figure 10.
Xenografts of human female proximal urethra grown in untreated castrated hosts (control) (A) and in DHT-treated castrated hosts (B) both stained for FOXA1, an endodermal marker expressed in all epithelial cells. (A) The control grafts contained a urethra lined with a stratified epithelium and solid epithelial outgrowths. (B) Grafts grown in DHT-treated hosts contained prostate-like canalized and solid ducts.
Table 5.
Epithelial immunohistochemical profile of xenografts of human fetal prostate and human female proximal urethra.
| Markers | Prostatic xenografts (DHT) | Female urethral xenografts (DHT) | Prostatic xenografts (control) | Female urethral xenografts (control) |
|---|---|---|---|---|
| Morphology | Canalized ducts | Canalized ducts | Solid “ducts” | Stratified epi. |
| Keratin 6 | Basal cells only | Basal cells only | Basal cells only | Basal cells only |
| Keratin 7 | Luminal cells only | Luminal cells only | Central core cells | Central core cells |
| Keratin 8 | Luminal cells only | Luminal cells only | Central core cells | Central core cells |
| Keratin 10 | Negative | Negative | Negative | Negative |
| Keratin 14 | Negative | Negative | Negative | Negative |
| Keratin 15 | Basal cells only | Basal cells only | Basal cells only | Basal cells only |
| Keratin 19 | Luminal cells only | Luminal cells only | Central core cells | Central core cells |
| RUNX1 | Luminal cells only | Luminal cells only | Supra basal cells | Supra basal cells |
| TP63 | Basal cells only | Basal cells only | Basal & central cells | Basal & central cells |
| FOXA1 | All cells | All cells | All cells | All cells |
| ESR1 | Negative | Negative | Negative | Negative |
| ESR2 | Positive | Positive | Positive | Positive |
| AR | Luminal cells only | Luminal cells only | Negative | Negative |
| NKX3.1 | Luminal cells only | Luminal cells only | Negative | Negative |
| PSA | Positive | Positive | Negative | Negative |
| PAP | Positive | Positive 4 | Negative | Negative |
Abbreviations: PSA=prostate specific antigen, PAP=prostatic acid phosphatase, AR=androgen receptor, epi.= epithelium.
b. Immunohistochemical observations.
As described above the 12 androgen-independent markers (keratin 6 to ESR2 in Table 5) were present in both control and DHT-treated human female proximal urethral xenografts, but a distinctive prostatic expression pattern was evident for these proteins in solid and canalized ducts of DHT-treated specimens (8/8) (Figs. 11–13). Canalized and solid ducts exhibited keratins in a prostatic pattern as described above. Notably this included keratins 7 (Fig. 11A), keratin 8 (not illustrated), keratin 6 (Fig. 11B), keratin 15 (Fig. 11D), keratin 18 (Fig. 11E), keratin 19 (Fig. 11G). TP63 was expressed in basal cells of urethral epithelium, in basal cells of canalized ducts and throughout solid epithelial “ducts” of xenografts of DHT-treated human fetal female proximal urethra (Fig. 11F). Xenografts of human fetal female proximal urethra grown in DHT-treated hosts also expressed NKX3.1 in luminal cells of canalizing ducts (Fig. 12A) as did ESR2 (Fig 12B). The androgen receptor profile in grafts of DHT-treated of human fetal female proximal urethra (Fig. 12C) was identical to that of xenografts of DHT-treated human fetal prostate (compare Figs. 6C, & 8D-E with 12C) as well as non-grafted human fetal prostatic specimens described above (Fig. 4A, Table 2).
Figure 11.
Xenografts of human fetal female proximal urethra grown for 8 weeks in DHT-treated castrated mouse hosts immunostained as indicated. Age at the time of grafting is: (A) 11 weeks, (B-E) 10 weeks, and (F) 14 weeks. Note extensively branched ductal networks. The immunohistochemical profile for all markers is consistent with prostatic differentiation as described above (Table 5).
Figure 13.
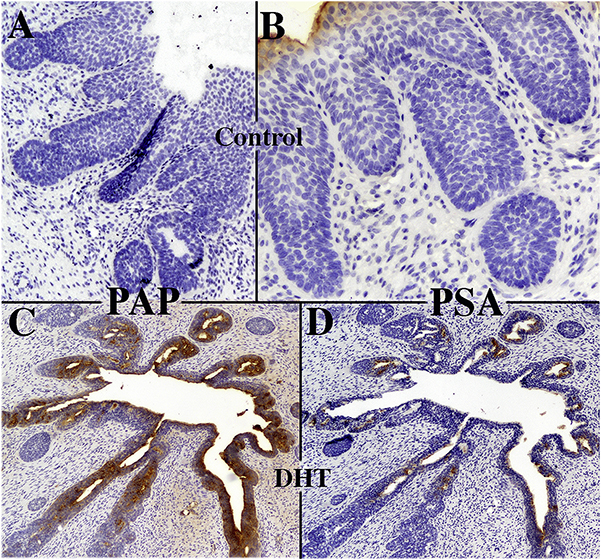
Xenografts of human fetal female proximal urethra grown for 8 weeks in DHT-treated castrated mouse hosts and untreated castrated (control) mouse hosts immunostained for PAP (A & C) and PSA (B & D).
Figure 12.
Xenografts of human fetal female proximal urethra grown for 8 weeks in DHT-treated castrated mouse hosts immunostained as indicated.
In DHT-treated xenografts of human fetal female proximal urethra the prostate-specific marker, NKX3.1, was expressed in luminal epithelial cells of canalized ducts (Fig. 12A), but not in solid epithelial cords and not in female urethral grafts grown in androgen-deficient hosts (not illustrated). ESR2 was likewise expressed in luminal epithelial cells of canalized ducts, but not in solid epithelial cords (Fig. 12B). The androgen receptor (AR) was broadly expressed throughout the stroma, as well as in luminal (but not basal) epithelial cells of canalized ducts (Fig. 12C) of human fetal female proximal urethra grown in DHT-treated hosts. Finally, PAP and PSA were both expressed in xenografts of human fetal female proximal urethra grown in DHT-treated hosts but not in human fetal female proximal urethra grown in androgen-deficient untreated castrated hosts, but only in canalized ducts (Fig. 13).
Discussion
The idea of prostatic development in xenografts of human fetal female proximal urethra can be inferred from studies in mice in which prostate can be induced when female urogenital sinuses are grafted into male hosts (Cunha, 1975) and in earlier animal studies in which female urogenital sinus formed prostatic buds when cultured in the presence of testosterone (Price and Ortiz, 1965; Takeda et al, 1986). Further support for prostatic development from human fetal urogenital sinus comes from radiographic evidence of prostate in a genetic XX female patient secondary to physiologic elevation in androgens due to congenital adrenal hyperplasia (Fang et al, 2013). Prostatic carcinogenesis has also been reported in genetic XX female patients also with congenital adrenal hyperplasia (Wesselius et al, 2017; Winters et al, 1996). The clinical implication is that patients with disorders of sex development with a XX genotype who are exposed in utero to high levels of androgens are at risk for formation of prostatic tissue as well as well documented genital virilization (Speiser et al, 2010). The implications for post-natal androgen exposure of XX genetic individuals such as in the transgender population remains less well defined (see discussion of “window of sensitivity” below) but should be monitored.
The significance of the present study is the reproducible experimental induction of prostatic morphogenesis and differentiation from human fetal female proximal urethra that includes expression of an epithelial immunohistochemical profile clearly indicative of prostatic ducts. This conclusion is supported by the expression of human prostate specific antigen (PSA), human prostatic acid phosphatase (PAP) and NKX3.1, known markers of human prostatic epithelial differentiation (Dema and Tudose, 1998; Irer et al, 2009; Lam et al, 1989). The experimental induction of prostatic differentiation from human fetal female proximal urethra makes possible future in-depth analysis of the molecular pathways directly involved in initiation of human prostatic development and subsequent epithelial differentiation, and more important whether the molecular pathways involved in human prostatic development are similar/identical versus different from that in murine prostatic development. The molecular biology of mouse prostatic development has advanced considerably in recent years (Abler et al, 2011; Georgas et al, 2015; Mehta et al, 2011; Toivanen and Shen, 2017) with the tacit, but unproven assumption that developmental pathways in mice are shared in human prostatic development. While many features of prostatic development, including certain molecular pathways, are clearly shared by these two species, as finer details of the molecular pathways are revealed in mice, it will now be possible to directly interrogate mouse versus human molecular pathways to determine whether species differences exist and to assess their biologic/therapeutic significance.
The human prostate develops from a restricted portion of the endodermal UGE, namely from the area immediately below the bladder. Accordingly, the same area in human females, which we designate the proximal urethra, is clearly homologous to the prostatic rudiment in males, and is thus capable of “female” prostatic development if exposed to androgens during the appropriate period of development. The ability of the human female UGS to form prostate in response to androgens is likely to be restricted temporally to a developmental window of sensitivity, after which the ability of the female UGS to form prostate is lost (Cunha, 1975). In this regard, the oldest female proximal urethra in the present study to undergo prostatic differentiation and express NKX3.1, PAP and PSA was derived from a 14.5 week fetus, 4–5 weeks later than the emergence of prostatic buds in male fetuses at 10 weeks of gestation (Cunha et al, 2018). Further studies will be required to determine when female urethra loses its ability to undergo prostatic development. The current study describes a simple experimental approach to reproducibly induce de novo prostatic development from female proximal urethra and documents the ontogeny of a variety proteins associated with and/or unique to prostatic differentiation.
Two critical questions deserving of utmost consideration are as follows: (a) Are the ductal structures induced by DHT in grafts of female proximal urethra actually prostatic; (b) Are the morphogenetic processes and the expression of immunohistochemical markers androgen-dependent.
The prostatic identity of ductal structures in grafts of female proximal urethra is supported by several observations. The overall morphogenetic process encompassing the formation of solid ducts, their canalization and differentiation of luminal and a continuous layer of basal cells compares favorably with human prostatic development (Cunha et al, 2018). The unique global spatial pattern of expression of 16 immunohistochemical markers in DHT-treated grafts of female proximal urethra is consistent with that of developing human prostate. Three prostate-specific markers (NKX3.1, prostate specific antigen [PSA] and prostatic acid phosphatase [PAP]) were expressed in DHT-treated grafts of female proximal urethra. NKX3.1 is a known specific marker of prostatic epithelium (Gelmann et al, 2003). Thus, the first question regarding prostatic identity of ductal structures induced by DHT in grafts of female proximal urethra is solidly supported by our data.
The second question, regarding the androgen-dependency of morphogenesis and expression of immunohistochemical markers is also solidly supported by many lines of evidence. The overall morphogenetic process (formation of solid ducts, subsequent canalization and differentiation of luminal and basal cells) is known to be androgen-dependent in the developing human prostate (Cunha et al, 2018; Wilson et al, 1981), and was precisely mimicked in DHT-treated (but not in control [non-androgenic]) grafts of female proximal urethra. Xenografts of human fetal prostate (13 and 14 weeks of gestation at the time of grafting) contained solid prostatic buds prior to transplantation. Prostatic ductal morphogenesis, epithelial differentiation and marker expression was enhanced (up-regulated) by DHT in these prostatic xenografts and was impaired as a result of androgen deficiency (untreated castrated hosts), thus confirming the prostatic morphogenesis is androgen-dependent. Several proteins expressed during prostatic development in grafts of female proximal urethra are known to be androgen-dependent (and prostate specific), namely, NKX3.1, PSA and PAP. NKX3.1 is an androgen-regulated homeobox gene (Korkmaz et al, 2004; Prescott et al, 1998) known to be expressed in benign and malignant human prostate (Gelmann et al, 2003) as well as mouse prostate (Bhatia-Gaur et al, 1999). The ontogeny of NKX3.1 in human fetal prostate has not been previously reported. NKX3.1 was not detected in non-grafted human fetal prostates from 9 to 21 weeks in solid and canalized prostatic ducts containing well differentiated luminal and basal cells. Whether NKX3.1 is expressed latter in the third trimester or whether its expression is delayed until puberty remains to be determined. NKX3.1 was also not detected in non-grafted human fetal female proximal urethra before or after growth in androgen-deficient hosts, but was detected in xenografts of human fetal female proximal urethra (and human fetal prostate) treated with DHT, thus confirming its androgen-dependence. NKX3.1 appeared to be expressed in luminal and perhaps in basal epithelial cells in xenografts of human fetal female proximal urethra, but in all cases, expression was seen only in canalizing or fully canalized ducts, and not in solid prostatic ducts of DHT-treated human fetal female proximal urethra. Whether NKX3.1 was expressed in luminal versus basal cells appears to be a function of the overall differentiation state of ducts transitioning from solid to fully canalized.
Prostate specific antigen (PSA) and prostatic acid phosphatase (PAP) are also know to the up-regulated by androgen in prostatic epithelial cells (Kim and Coetzee, 2004; Lin et al, 1993; Porvari et al, 1995). PSA and NKX3.1 were not observed in 21-week non-grafted prostates, while PAP was detected at 21-week human fetal prostates. Perhaps the androgenic threshold for expression of PAP is lower than that PSA and NKX3.1. The DHT pellets used in our xenograft studies may be releasing a supraphysiologic dose of androgen which is compatible with induction of these proteins. It is notable that PAP is expressed temporally before PSA in subcutaneous of human prostatic organoids grafted into male nude mice (Hayward, 1992). In any case the expression of PSA, PAP and NKX3.1 in DHT-treated xenografts of human fetal prostate and human fetal female proximal urethra is consistent with androgenic regulation of these proteins as these proteins were not detected under non-androgenic conditions. The expression of NKX3.1, PSA and PAP correlates with the presence of androgen receptors in the developing human prostate and human fetal female proximal urethra.
The androgen receptor (AR) is another known androgen-regulated protein. The androgen receptor is also known to be up-regulated in androgen target organs by testosterone and other androgens in vivo (Amet et al, 1986; Block et al, 1991; De Pergola et al, 1990). Thus, the broad presence of AR immunostaining in human fetal female proximal urethra grown in DHT-treated hosts (Fig. 12C) and the almost complete absence of AR in human fetal female proximal urethra grown in untreated castrated hosts is another manifestation of an androgen-dependent prostate-associated marker induced by DHT in human fetal female proximal urethra.
As an aside, our observations of NKX3.1 in the developing human prostate differ significantly from that reported for the developing mouse prostate (Bhatia-Gaur et al, 1999; Sciavolino et al, 1997). In mice NKX3.1 is expressed in the pre-bud male UGE at 14 days of gestation, in solid prostatic buds and subsequently in canalized prostatic ducts, while in human prostatic development NKX3.1 was only observed in canalized ducts, that is, in an advanced stage of prostatic development. Fourteen days of gestation in mice is a period when the fetal testes are producing testosterone (Bloch et al, 1971), and thus regulation of this androgen-dependent protein in mice may be exquisitely sensitive to androgen.
From a broader perspective, our statement above regarding so-called androgen-independent immunohistochemical markers in Tables 3–5 (keratin 6 to ESR2) requires qualification. The 12 immunohistochemical markers in question were said to be androgen-independent based upon the fact that they were equivalently expressed under androgenic and non-androgenic conditions in human prostatic and human fetal female proximal urethral grafts. However, the development of prostatic buds/ducts is known to be globally dependent upon androgens and virtually all immunohistochemical markers are expressed ontogenetically and spatially in developing prostatic ducts in a unique (androgen-dependent) pattern. Thus, from the more-narrow perspective of the developing prostatic ducts in DHT-treated female proximal urethra, nearly the entire spectrum of immunohistochemical markers can be considered to be androgen-dependent, a salient feature of prostatic development. Finally, grafts of human female urethra provide the opportunity of examining androgen-induced initiation of human fetal prostatic budding (with induction of NKX3.1, PSA and PAP) under controlled experimental conditions.
We have shown previously that rat and mouse UGM can induce prostatic development in human fetal and adult human urinary bladder epithelium (BLE), an event associated with the expression of PSA in the induced glands (Aboseif et al, 1999; Cunha et al, 1983). It is likely that such UGM+BLE tissue recombinants also express NKX3.1 and PAP, but this remains to be determined. These findings, coupled with the literature on human fetal prostatic xenografts and the findings reported herein, raise the possibility of using genetically altered mouse UGM in tissue recombinants composed of mutant mouse UGM plus human endodermal epithelium (from urinary bladder, UGE or female urethra) to define the role of specific mesenchymal genes in prostatic development.
Acknowledgements:
This study was supported by the following grant: K12DK083021.
Abbreviations:
- H&E
hematoxylin & eosin
- UGS
urogenital sinus
- UGE
urogenital sinus epithelium
- UGM
urogenital sinus mesenchyme
- MDE
Müllerian duct epithelium
- AR
androgen receptor
- DHT
dihydrotestosterone
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abler LL, Keil KP, Mehta V, Joshi PS, Schmitz CT and Vezina CM (2011) A high-resolution molecular atlas of the fetal mouse lower urogenital tract. Dev Dyn 240:2364–2377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aboseif S, El-Sakka A, Young P and Cunha G (1999) Mesenchymal reprogramming of adult human epithelial differentiation. Differentiation 65:113–118. [DOI] [PubMed] [Google Scholar]
- Amet Y, Abalain JH, Daniel JY, Di Stefano S and Floch HH (1986) Testosterone regulation of androgen receptor levels in the uropygial gland of quails (Coturnix coturnix): a further proof for the androgen dependency of the uropygial gland. Gen Comp Endocrinol 62:210–216. [DOI] [PubMed] [Google Scholar]
- Bhatia-Gaur R, Donjacour AA, Sciavolino PJ, Kim M, Desai N, Young P, Norton CR, Gridley T, Cardiff RD, Cunha GR, Abate-Shen C and Shen MM (1999) Roles for Nkx3.1 in prostate development and cancer. Genes Dev 13:966–977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloch E, Lew M and Klein M (1971) Studies on the inhibition of fetal androgen formation: testosterone synthesis by fetal and newborn mouse testes in vitro. Endocrinology 88:41–46. [DOI] [PubMed] [Google Scholar]
- Block LJ, Bartlett JM, Bolt-de Vries J, Themmen AP, Brinkmann AO, Weinbauer GF, Nieschlag E and Grootegoed JA (1991) Regulation of androgen receptor mRNA and protein in the rat testis by testosterone. J Steroid Biochem Mol Biol 40:343–347. [DOI] [PubMed] [Google Scholar]
- Cunha GR (1975) Age-dependent loss of sensitivity of female urogenital sinus to androgenic conditions as a function of the epithelial-stromal interaction. Endocrinology 95:665–673. [DOI] [PubMed] [Google Scholar]
- Cunha GR and Baskin L (2016) Use of sub-renal capsule transplantation in developmental biology. Differentiation 91:4–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunha GR, Donjacour AA, Cooke PS, Mee S, Bigsby RM, Higgins SJ and Sugimura Y (1987) The endocrinology and developmental biology of the prostate. Endocrine Rev. 8:338–362. [DOI] [PubMed] [Google Scholar]
- Cunha GR, Sekkingstad M and Meloy BA (1983) Heterospecific induction of prostatic development in tissue recombinants prepared with mouse, rat, rabbit, and human tissues. Differentiation 24:174–180. [DOI] [PubMed] [Google Scholar]
- Cunha GR, Vezina CM, Isaacson D, Ricke WA, Timms BG, Cao M, Franco O and Baskin LS (2018) Development of the human prostate. Differentiation 103:24–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Pergola G, Xu XF, Yang SM, Giorgino R and Bjorntorp P (1990) Upregulation of androgen receptor binding in male rat fat pad adipose precursor cells exposed to testosterone: study in a whole cell assay system. J Steroid Biochem Mol Biol 37:553–558. [DOI] [PubMed] [Google Scholar]
- Dema A and Tudose N (1998) Immunohistochemical expression of prostate specific antigen (PSA) in benign and malignant tumors of the prostate. Rom J Morphol Embryol 44:93–100. [PubMed] [Google Scholar]
- Drey EA, Kang MS, McFarland W and Darney PD (2005) Improving the accuracy of fetal foot length to confirm gestational duration. Obstet Gynecol 105:773–778. [DOI] [PubMed] [Google Scholar]
- Fang B, Cho F and Lam W (2013) Prostate gland development and adrenal tumor in a female with congenital adrenal hyperplasia: a case report and review from radiology perspective. J Radiol Case Rep 7:21–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelmann EP, Bowen C and Bubendorf L (2003) Expression of NKX3.1 in normal and malignant tissues. Prostate 55:111–117. [DOI] [PubMed] [Google Scholar]
- Georgas KM, Armstrong J, Keast JR, Larkins CE, McHugh KM, Southard-Smith EM, Cohn MJ, Batourina E, Dan H, Schneider K, Buehler DP, Wiese CB, Brennan J, Davies JA, Harding SD, Baldock RA, Little MH, Vezina CM and Mendelsohn C (2015) An illustrated anatomical ontology of the developing mouse lower urogenital tract. Development 142:1893–1908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayward SW The role of stroma in prostate epithelial function: Development of a model system. Imperial Cancer Research Fund. London: Council for National Academic Awards (UK), 1992.
- Hudson DL, Guy AT, Fry P, O’Hare MJ, Watt FM and Masters JR (2001) Epithelial cell differentiation pathways in the human prostate. Identification of intermediate phenotypes by keratin expression. J Histochem Cytochem 49:271–278. [DOI] [PubMed] [Google Scholar]
- Irer B, Toylu A, Aslan G, Celebi I, Yorukoglu K and Atabey N (2009) Increased expression of NKX3.1 in benign prostatic hyperplasia. Urology 73:1140–1144. [DOI] [PubMed] [Google Scholar]
- Kim J and Coetzee GA (2004) Prostate specific antigen gene regulation by androgen receptor. J Cell Biochem 93:233–241. [DOI] [PubMed] [Google Scholar]
- Korkmaz CG, Korkmaz KS, Manola J, Xi Z, Risberg B, Danielsen H, Kung J, Sellers WR, Loda M and Saatcioglu F (2004) Analysis of androgen regulated homeobox gene NKX3.1 during prostate carcinogenesis. J Urol 172:1134–1139. [DOI] [PubMed] [Google Scholar]
- Lam KW, Li CY, Yam LT, Sun T, Lee G and Ziesmer S (1989) Improved immunohistochemical detection of prostatic acid phosphatase by a monoclonal antibody. Prostate 15:13–21. [DOI] [PubMed] [Google Scholar]
- Liaw A, Cunha GR, Shen J, Cao M, Liu G, Sinclair A and Baskin LS (2018) Embryological Development of the Bladder and Ureterovesical Junction. Differentiation (This issue): [DOI] [PubMed] [Google Scholar]
- Lin MF, Garcia-Arenas R, Chao YC, Lai MM, Patel PC and Xia XZ (1993) Regulation of prostatic acid phosphatase expression and secretion by androgen in LNCaP human prostate carcinoma cells. Arch Biochem Biophys 300:384–390. [DOI] [PubMed] [Google Scholar]
- Marker PC, Donjacour AA, Dahiya R and Cunha GR (2003) Hormonal, cellular, and molecular control of prostatic development. Dev Biol 253:165–174. [DOI] [PubMed] [Google Scholar]
- Mehta V, Abler LL, Keil KP, Schmitz CT, Joshi PS and Vezina CM (2011) Atlas of Wnt and R-spondin gene expression in the developing male mouse lower urogenital tract. Dev Dyn 240:2548–2560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohno S (1979) Major Sex Determining Genes. Springer-Verlag, New York. [Google Scholar]
- Porvari K, Kurkela R, Kivinen A and Vihko P (1995) Differential androgen regulation of rat prostatic acid phosphatase transcripts. Biochem Biophys Res Commun 213:861–868. [DOI] [PubMed] [Google Scholar]
- Prescott JL, Blok L and Tindall DJ (1998) Isolation and androgen regulation of the human homeobox cDNA, NKX3.1. Prostate 35:71–80. [DOI] [PubMed] [Google Scholar]
- Price D and Ortiz E (1965) The role of fetal androgens in sex differentiation in mammals In: Ursprung RL and DeHaan H (ed) Organogenesis. Holt, Rinehart and Winston, New York, pp 629–652. [Google Scholar]
- Robboy SJ, Kurita T, Baskin L and Cunha GR (2017) New insights insights into human female reproductive tract development. Differentiation 97:9–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez E Jr., Weiss DA, Ferretti M, Wang H, Menshenia J, Risbridger G, Handelsman D, Cunha G and Baskin L (2012) Specific morphogenetic events in mouse external genitalia sex differentiation are responsive/dependent upon androgens and/or estrogens. Differentiation 84:269–279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciavolino PJ, Abrams EW, Yang L, Austenberg LP, Shen MM and Abate-Shen C (1997) Tissue-specific expression of murine Nkx3.1 in the male urogenital system. Dev Dyn 209:127–138. [DOI] [PubMed] [Google Scholar]
- Speiser PW, Azziz R, Baskin LS, Ghizzoni L, Hensle TW, Merke DP, Meyer-Bahlburg HF, Miller WL, Montori VM, Oberfield SE, Ritzen M, White PC and Endocrine S (2010) Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 95:4133–4160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeda I, Lasnitzki I and Mizuno T (1986) Analysis of prostatic bud induction by brief androgen treatment in the fetal rat urogenital sinus. J. Endocronol. 110:467–470. [DOI] [PubMed] [Google Scholar]
- Toivanen R and Shen MM (2017) Prostate organogenesis: tissue induction, hormonal regulation and cell type specification. Development 144:1382–1398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warick R and Williams PL (1973) Gray’s Anatomy W. B. Saunders, Philadelphia. [Google Scholar]
- Wesselius R, Schotman M, Schotman M and Pereira AM (2017) A Patient (46XX) With Congenital Adrenal Hyperplasia and Prostate Cancer: A Case Report. J Endocr Soc 1:1213–1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson J, George F and Griffin J (1981) The hormonal control of sexual development. Science 211:1278–1284. [DOI] [PubMed] [Google Scholar]
- Wilson J, Griffin J and Russell DW (1993) Steroid 5a-reductase deficiency. Endocrine Rev. 14:577–593. [DOI] [PubMed] [Google Scholar]
- Wilson JD (1987) Disorders of androgen action. Clinical research 35:1–12. [PubMed] [Google Scholar]
- Wilson JD, Griffin JE, Leshin M and P.C. M (1983) The androgen resistance syndromes: 5 alpha reductase deficiency, testicular feminization, and related disorders In: Stanbury JD, Wyngaarden JB, Fredrickson DS, Goldstein JD and Brown MS (ed) The Metabolic Basis of Inherited Disease. McGraw Hill, New York, pp 1001–1026. [Google Scholar]
- Winters JL, Chapman PH, Powell DE, Banks ER, Allen WR and Wood DP Jr. (1996) Female pseudohermaphroditism due to congenital adrenal hyperplasia complicated by adenocarcinoma of the prostate and clear cell carcinoma of the endometrium. Am J Clin Pathol 106:660–664. [DOI] [PubMed] [Google Scholar]
- Yamada G, Satoh Y, Baskin LS and Cunha GR (2003) Cellular and molecular mechanisms of development of the external genitalia. Differentiation; research in biological diversity 71:445–460. [DOI] [PubMed] [Google Scholar]