Abstract
Osteonecrosis of the femoral head (ONFH) is a progressive disease with a complex etiology and unclear pathogenesis, resulting in severe hip pain and dysfunction mainly observed in young patients. Although total hip arthroplasty (THA) is the most effective treatment for patients with ONFH in the terminal stage, the results of THA in young patients or active populations are often not favorable, with some complications related to the prosthesis. With the development of biotechnology, an increasing number of studies pay attention to use of stem cells for the treatment of ONFH. Stem cells are characterized by the ability to self-renew and differentiate into multiple cell types, including differentiation into osteoblasts and endothelial cells to mediate bone repair and angiogenesis. Furthermore, stem cells can offer growth factors to promote blood supply in the necrotic regions by paracrine effects. Therefore, stem cell therapy has become one of the hip-preserving alternatives for ONFH. This review summarized the current trends in stem cell therapy for ONFH, from clinical applications to related basic research, and showed that an increasing number of studies have confirmed the effectiveness of stem cell therapy in ONFH. However, many unsolved problems and challenges in practical applications of stem cell therapy still exist, such as patient selection, standardized procedures, safety assessment, and the fate of transplanted cells in the body. Additional studies are required to find ideal cell sources, appropriate transplantation methods, and the optimal number of cells for transplantation.
Keywords: Osteonecrosis, Femoral head, Cell therapy, Cell implantation, Stem cells, Tissue engineering
Abbreviations: ALP, alkaline phosphatase; AMSCs, adipose-derived MSCs; BCP, biphasic calcium phosphate; bFGF, basic fibroblast growth factor; BMC, bone marrow concentrate; BMMNCs, bone marrow mononuclear cells; BMP-2, bone morphogenetic protein-2; BMSCs, bone marrow-derived mesenchymal stem cells; CD, Core decompression; CPC, calcium phosphate; CSS, cap-shaped separation; DBM, demineralized bone matrix; HHS, Harris hip score; IP-CHA, interconnected porous calcium hydroxyapatite; MRI, magnetic resonance imaging; MSCs, Mesenchymal stem cells; MVD, microvessel density; ONFH, Osteonecrosis of the femoral head; PBMSCs, peripheral blood-derived MSCs; PLGA, poly lactide-co-glycolide; RCT, randomized controlled trial; SCPP, strontium-doped calcium polyphosphate; SVF, stromal vascular fractions; THA, total hip arthroplasty; TMCs, transformed mesenchymal cells; TNF, tumor necrosis factor; UCMSCs, umbilical cord-derived mesenchymal stem cells; VAS, visual analogue scale; VEGF, vascular endothelial growth factor; WOMAC, Western Ontario and McMaster Universities Arthritis Index; XACB, xenogeneic antigen-extracted cancellous bone; β-TCP, beta-tricalcium phosphate
Highlights
-
•
Diversities in repair processes present a challenge in the targeted treatment of ONFH.
-
•
Osteogenesis and angiogenesis are the primary mechanisms of MSCs treatment in ONFH.
-
•
Systematic safety assessment and cell tracing are necessary for stem cell therapy.
-
•
Optimal numbers and methods of cell transplantation need to be further confirmed.
1. Introduction
Osteonecrosis of the femoral head (ONFH) is a progressive disease that is characterized by interruption of blood supply, necrosis of subchondral bone, and the eventual bone collapse of the femoral head, resulting in severe hip pain and dysfunction mainly in young patients [1,2]. Although total hip arthroplasty (THA) is the most effective treatment for patients with ONFH in the terminal stages, the outcomes of THA in young adults or active populations are often not excellent, with a high failure rate caused by loosening of the prosthetic, excessive wear of polyethylene inserts, and peri-prosthetic infection [[3], [4], [5], [6]]. Therefore, hip-preserving treatments for ONFH has become a challenge in young patients.
Stem cells have been shown to have the ability to self-renew and to differentiate into multiple cell types. Mesenchymal stem cells (MSCs) are one of the most frequently used stem cell subsets, which are widely distributed in various tissues, such as bone marrow, peripheral blood, fat, umbilical cord, etc. [7]. MSCs can differentiate into osteoblasts and endothelial cells to affect bone repair and angiogenesis [8], and can produce growth factors to promote the blood supply to necrotic regions by paracrine effects [9,10]. Furthermore, MSCs not only play a very important role in the development of disease, but also are the main source of seed cells for regenerative therapy. At present, the pathogenesis of ONFH is considered to be related to bone marrow-derived mesenchymal stem cells (BMSCs), including cell apoptosis, decrease in cell numbers, and decline in osteogenic differentiation potential [11]. Thus, stem cell therapy has become one of the hip-preserving alternatives for ONFH. It has been nearly 20 years since the first report of stem cell therapy for ONFH [12]. With the rapid development of cell biotechnology and tissue engineering, stem cell therapy has shown promising results in the treatment of ONFH. However, there still are many unresolved problems and challenges in stem cell therapy for ONFH stemming from the lack of standardized procedures, optimal accumulation of cells, and systematic security assessment.
In this review, we summarized the current trends in stem cell therapy for ONFH, ranging from clinical applications to related basic research, and briefly analyzed the existing problems and challenges.
2. Problems of current ONFH treatment
2.1. Complicated and unclear pathogenesis for ONFH
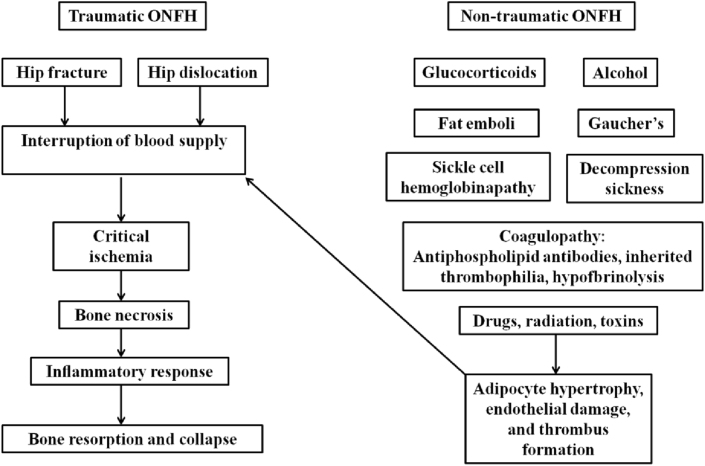
The etiology of ONFH is complicated, including impairment due to trauma, corticosteroids treatment, alcohol abuse, and connective tissue diseases [13], which ultimately leads to a disturbance in the microcirculation (Fig. 1). At present, the pathology of ONFH is considered to be related to damaged blood supply, abnormal lipid metabolism, weakened osteogenic ability of BMSCs, cell apoptosis, and gene polymorphism [11]. However, the specific pathological processes of ONFH are yet to be fully elucidated. Moreover, the repair process in different types of ONFH is inconsistent, leading to challenges in developing targeted treatments for ONFH.
Fig. 1.
Etiology and mechanisms of ONFH. Figure is from Ref. [13]. ONFH: osteonecrosis of the femoral head.
2.2. Insufficient early diagnosis of ONFH
In the early stages of ONFH, no obvious symptoms are observed except slight pain in the groin, and no obvious findings are observed in X-ray images, which often leads to difficulties in the early diagnosis of ONFH. In most of the patients with a definitive diagnosis, the disease has significantly progressed, presenting as obvious collapse of the femoral head, damage in the articular cartilage, narrowing of the joint space, and deterioration of the hip into serious osteoarthritis, thus presenting a great challenge for the treatment of ONFH.
2.3. Lack of more effective treatments for ONFH
Currently, THA is the most effective treatment for patients with ONFH in the terminal stages. However, most of the patients with ONFH range in age from 30 to 40 years, and the bilateral hip joints are involved in 75% of patients [3]. In these patients, THA is usually associated with a high failure rate due to loosening of the prosthetic and excessive wear of polyethylene inserts [[4], [5], [6]], along with surgical complications such as operative stress, peri-prosthetic infection, peri-prosthetic fracture, and prosthesis dislocation [[14], [15], [16]]. Therefore, it is important to seek more effective and safe treatments of ONFH.
3. The theoretical basis behind stem cell therapy for ONFH
ONFH is a process that is accompanied by osteonecrosis and bone reconstruction. However, the rate of bone reconstruction is not enough to compensate for osteonecrosis due to insufficient numbers of osteoblasts in the femoral head, inevitably leading to ONFH. Currently, the pathogenesis of ONFH is considered to be related to BMSCs, including cell apoptosis, decrease in cell numbers, weakened cellular growth ability, abnormal metabolism of stem cells, and decline in osteogenic differentiation potential [11,17]. MSCs are characterized by their ability to self-renew and their potential to differentiate in to multiple cell types, and can differentiate into osteoblasts and endothelial cells for bone repair and angiogenesis [8]. Furthermore, stem cells can offer growth factors to promote blood supply to necrotic regions by paracrine effects [9,10]. Therefore, osteogenesis and angiogenesis are some of the most important mechanisms of MSCs as a treatment for ONFH.
4. Current stem cell therapy for ONFH
4.1. Cell source
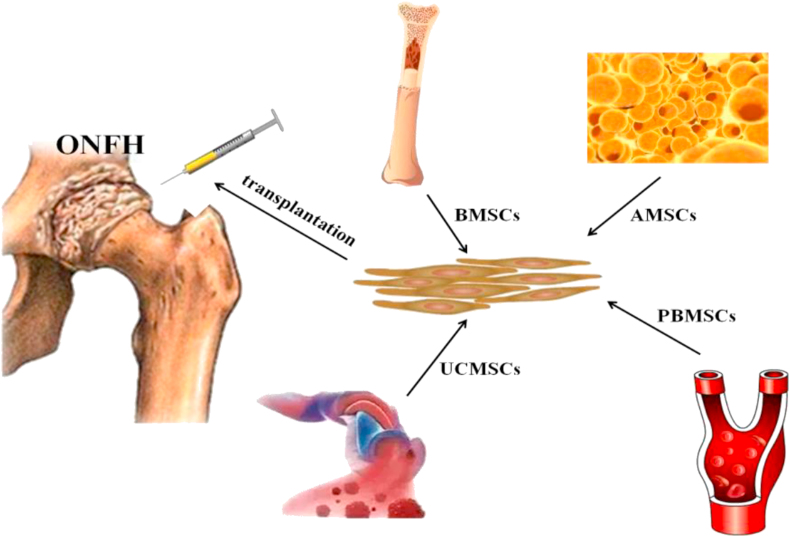
MSCs are one of the most frequently used stem cell subsets, and are widely distributed in the various tissues, such as bone marrow, peripheral blood, fat, umbilical cord, etc. [7]. Therefore, depending on the source of the tissue, MSCs are named bone marrow-derived MSCs (BMSCs), adipose-derived MSCs (AMSCs), peripheral blood-derived MSCs (PBMSCs) and umbilical cord-derived mesenchymal stem cells (UCMSCs) (Fig. 2).
Fig. 2.
The main source of stem cells for transplantation. ONFH: osteonecrosis of the femoral head. BMSCs: bone marrow-derived mesenchymal stem cells; AMSCs: adipose-derived mesenchymal stem cells; PBMSCs: peripheral blood-derived mesenchymal stem cells; UCMSCs: umbilical cord-derived mesenchymal stem cells.
4.1.1. BMSCs
Bone marrow is the most commonly used stem cell source in the treatment for ONFH. In a suitable environment, BMSCs can differentiate into musculoskeletal system cells, such as trabecular bone, articular cartilage, tendon, etc. [18]. At present, it would be of clinical interest to improve the osteogenic differentiation of BMSCs, which is influenced by growth factors, glucocorticoids, small-molecule drugs, and gene polymorphism. For example, the combination of bone morphogenetic protein-2 (BMP-2), vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) could promote the osteogenic differentiation of BMSCs [19]. Low-dose dexamethasone enhances the osteogenic differentiation potential of BMSCs by upregulating the expression of alkaline phosphatase (ALP), which is an early osteogenic differentiation marker [20]. Icariin can also promote osteogenesis in BMSCs by increasing the expression of P-glycoprotein [21]. In addition, the study by Li et al. has showed that miRNA-291a-3p promoted osteogenic differentiation of BMSCs by directly inhibiting the expression of DKK1 and activating the Wnt/β-catenin signaling pathway [22].
Notably, the clinical applications of BMSCs mainly include three forms: bone marrow concentrate (BMC), cultured cells, and bone marrow aspirates, with the mostly used form being BMC [23]. The study by Rastogi et al. [24] demonstrated that the injection of isolated mononuclear cells was more effective than direct injection of bone marrow in the treatment of ONFH, because isolated mononuclear cells had a higher density of cells at the implanted site.
4.1.2. AMSCs
Adipose tissue is another reliable stem cell source for the treatment for ONFH. AMSCs have some advantages such as easy accessibility, higher productivity, and similar differentiation potential as BMSCs [25,26]. In the induction of osteogenic differentiation, transplantation of autologous AMSCs can improve osteogenesis, and increase bone mass and bone density in rabbit model of ONFH [27]. The over-expression of VEGF could promote osteogenesis and angiogenesis by AMSCs [28]. According to the report by Abudusaimi et al. [29], core decompression combined with AMSCs transplantation improved the microstructure and osteogenesis of the necrotic area by increasing the expression of osteocalcin in rabbit models of ONFH. Furthermore, Pak et al. [30] injected autologous adipose stromal vascular fractions (SVF) percutaneously into hip joints, which improved patients’ symptoms and promoted bone regeneration in the area with osteonecrosis. However, most of these promising results are based on the basic research studies, and the evidence offered by clinical trials still requires further elucidation.
4.1.3. PBMSCs
Peripheral blood is also one of the sources of MSCs, and is considered to be superior to bone marrow. Compared with BMSCs, PBMSCs have many advantages, such as easy accessibility, higher cost-effectiveness, and lower risk of contamination from tumor cells [[31], [32], [33]]. The studies by Song et al. [34,35] demonstrated that transplantation of PBMSCs alleviated ischemic femoral head necrosis in the liquid-nitrogen refrigeration rabbit model by promoting new bone formation. In addition, Fu et al. [36] transplanted PBMSCs into the necrotic area in rabbit ONFH models. They found that PBMSCs transplantation could increase the bone density and improve the microstructure of the femoral head by upregulating the expression of BMP-2 and downregulating the expression of PPAR-γ. In the clinical trials, Mao et al. [37] combined PBMSCs with porous tantalum for the treatment of ONFH in the early and medium stages. This study showed that PBMSCs transplantation improved the efficacy of biomechanical support in the treatment of ONFH, compared to biomechanical support alone. However, this treatment combination is not suitable for patients with painful cap-shaped separation (CSS) ONFH [38].
4.1.4. UCMSCs
UCMSCs have apparent stem cell attributes and lower immunogenicity. UCMSCs are also easier to isolate and expand in vitro, and have a remarkable potential to promote tissue regeneration [39]. Kuang et al. [40] reported that exosomes derived from human UCMSCs could prevent the progress of glucocorticoid-induced ONFH in rats by reducing osteocyte apoptosis. Furthermore, this effect is achieved by regulating the miR-21-PTEN-AKT signaling pathway. In clinical trials, Chen et al. [41] grafted UC-MSCs to treat ONFH by intra-arterial infusion. On magnetic resonance imaging (MRI), they found that this method could reduce the necrotic volume of the femoral head, and increase the oxygen transport based on the oxygen delivery index analysis. The authors speculated that this result was due to the migration of UC-MSCs into the necrotic area and the osteogenic differentiation of UC-MSCs. Furthermore, Cai et al. [42] investigated the efficacy of the combination of allogeneic UCMSCs and autologous bone marrow mononuclear cells (BMMNCs) transplantation in the treatment of ONFH. The study demonstrated that this therapeutics approach gradually improved the clinical symptoms of patients, including pain and claudication.
4.2. Implantation methods
4.2.1. Combination of core decompression and MSC transplantation
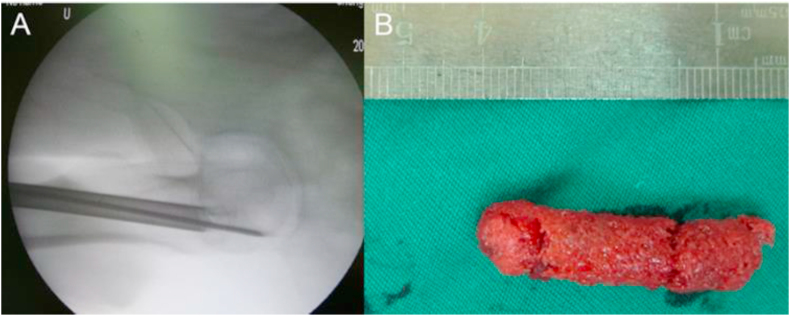
Core decompression (CD) is the most classical procedure for treatment of ONFH in the early stages, and reduces intraosseous pressure, removes the necrotic tissue, and stimulates the formation of new bone [43]. However, the efficacy of core decompression is variable, and favorable outcomes are observed only in patients with small necrotic lesions [44]. Therefore, a combination of core decompression and MSC transplantation has become a common approach in the treatment of ONFH. Briefly, stem cells are isolated from the relevant tissue, and then injected into the necrotic area of femoral head following core decompression. Tabatabaee et al. [45] performed the combination of core decompression and autologous bone marrow transplantation on patients with ONFH. They found that the combination treatment improved the Western Ontario and McMaster Universities Arthritis Index (WOMAC) and visual analogue scale (VAS) significantly, and promoted the repair of necrotic areas, as observed by MRI. In addition, the study by Rastogi et al. [24] also demonstrated that core decompression combined with autologous BMMNCs implantation could improve Harris hip score (HHS) and reduce the size of lesions in patients with ONFH. Sen et al. [46] performed a randomized control study, which showed that the clinical score and mean hip survival were significantly better in patients treated with the combination of core decompression and autologous bone marrow mononuclear cell transplantation, compared with patients treated with core decompression alone. Ma et al. [47] conducted a prospective, double-blinded, randomized controlled trial (RCT) to examine the efficacy of core decompression combined with bone marrow buffy coat grafting for the treatment of ONFH (Fig. 3). They found that the combination treatment improved the clinical symptoms and slowed the disease progression of ONFH.
Fig. 3.
Combination of core decompression and bone marrow buffy coat for the treatment of ONFH. (A) The X-ray showed that core decompression was performed by the Kirschner wire and 10-mm diameter trephine in the operation; (B) The long cylindrical bone obtained by core decompression was implanted into the bone tunnel after mixing with bone marrow. Figure is from Ref. [47]. ONFH: osteonecrosis of the femoral head.
In all, this combination treatment not only has some advantages of core decompression, but also provides seed cells to improve the repair and remodeling of the necrotic area. Therefore, in situ implantation of MSCs into the necrotic area can significantly improve the therapeutic efficacy of core decompression in ONFH.
4.2.2. MSCs arterial perfusion
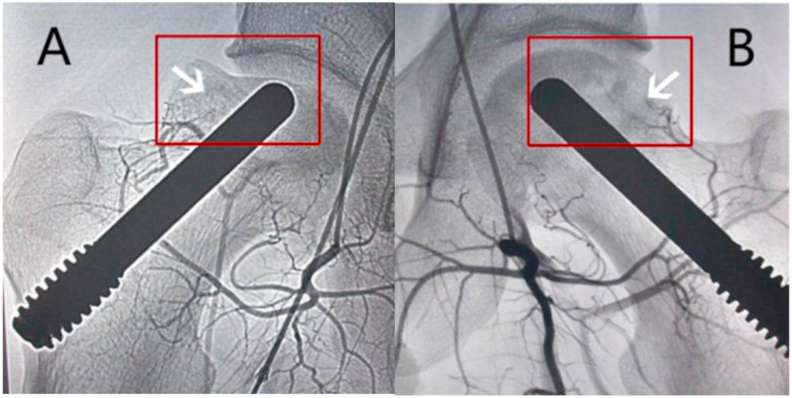
Arterial perfusion of MSCs is another treatment method for ONFH, the purpose of which is to improve the blood supply to the femoral head. The positive effect of MSCs on angiogenesis has been reported in previous studies. According to the study by Kinnaird et al. [48], MSCs augment arteriogenesis by inducing the release of vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF). Qian et al. [49] reported that the transplantation of autologous BMSCs could promote angiogenesis in rabbit ischemic hind limbs. Based on this theory, the efficacy of MSCs arterial perfusion in ONFH was explored in preclinical and clinical trials. Jin et al. [50] performed arterial perfusion of MSCs in the Beagle dog model of ONFH. They found that arterial perfusion of MSCs promoted the angiogenesis and vascular repair of the femoral head by increasing the expression of VEGF and by increasing microvessel density (MVD), which improved the blood supply and reconstruction of the necrotic area. However, arterial perfusion of MSCs is often combined with biomechanical support in clinical applications. Mao et al. [37] combined the targeted intra-arterial perfusion of PBMSCs with porous tantalum in the treatment of ONFH in the early and medium stages of the disease (Fig. 4). His study showed that arterial perfusion of PBMSCs improved the efficacy of biomechanical support in the treatment of ONFH, compared to biomechanical support alone. However, this method did not apply to severe lesions, but was applicable in early and medium stage ONFH. In addition, the study of Ying et al. [38] also indicated that this combination treatment was not suitable for patients with painful CSS ONFH.
Fig. 4.
Combination of arterial perfusion of PBMSCs and porous tantalum rod for the treatment of ONFH (36 months after operation). (A) Without vascular regeneration was observed in the control group, and the box showed the collapsed femoral head; (B) Vascular regeneration around the femoral head was found in the combination treatment group, and the box showed the intact and round femoral head. Figure is from Ref. [37]. PBMSCs: peripheral blood-derived mesenchymal stem cells; ONFH: osteonecrosis of the femoral head.
4.2.3. Cytokine pretreated or gene-modified MSCs transplantation
The current studies have demonstrated that many cytokines are involved in promoting the osteogenesis and angiogenesis of MSCs, and improving the repair of ONFH, including BMP, VEGF, bFGF, and tumor necrosis factor (TNF) [51]. Therefore, these cytokines are used to pretreat MSCs to establish an effective treatment of ONFH. Wang et al. [52] performed the transplantation of AMSCs pretreated with BMP-2 after drilling through the growth plate in a juvenile rabbit model of femoral head epiphyseal ischemic necrosis. This study showed that the combination treatment could increase new bone formation in the necrotic area and prevent the collapse of the femoral head.
Furthermore, with the rapid development of genetic engineering techniques, gene-modified MSCs have been widely employed as seed cells for the treatment of ONFH in preclinical trials. Ma et al. [53] investigated the effect of BMSCs modified by VEGF-165 and BMP-2 gene for the treatment of ONFH in a rabbit model. The results suggested that VEGF-165/BMP-2 gene transfection augmented the osteogenic effects of BMSCs, increased the number and quality of new bones, and accelerated the repair of ONFH. In addition, Peng et al. [54] combined the BMSCs transfected with BMP-2 and bFGF genes with a demineralized bone matrix (DBM) to repair ONFH in Beagle dogs. They found that the transplantation of BMP-2 and bFGF-transfected MSCs in conjunction with DBM could successfully promote the repair of the necrotic area by increasing osteogenesis and angiogenesis. The study by Zhang et al. [55] also demonstrated that the combination of BMSCs overexpressing FGF-2 gene and xenogeneic antigen-extracted cancellous bone (XACB) could effectively increase vascular regeneration, and promote the repair of ONFH in a rabbit model. Subsequently, Peng et al. [56] investigated the effects of the transplantation of bFGF-2 gene-transfected MSCs and XACB on TNF-α expression in a rabbit model of ONFH. They found that FGF-2/MSCs/XACB could promote the repair of ONFH via the inhibition of TNF-α expression.
4.2.4. MSCs transplantation and bone tissue engineering technology
Currently, bone tissue engineering technology is the main field of orthopedic research, including the culture of living cells in vitro, composite transplantation of cells in vivo, and the repair and reconstruction of bone tissue. Bone tissue engineering technology is characterized by seed cells, carriers and scaffolds. MSCs are the most ideal seed cells for bone tissue engineering due to their excellent differentiation capacity, strong proliferation activity, and low immunogenicity. The carriers or scaffolds can be used to regulate cellular function by not only integrating cells and receptors, but also by providing biomechanical support for the necrotic area of the femoral head. Therefore, the combination of MSCs and carriers or scaffolds has become a new approach for the treatment of ONFH.
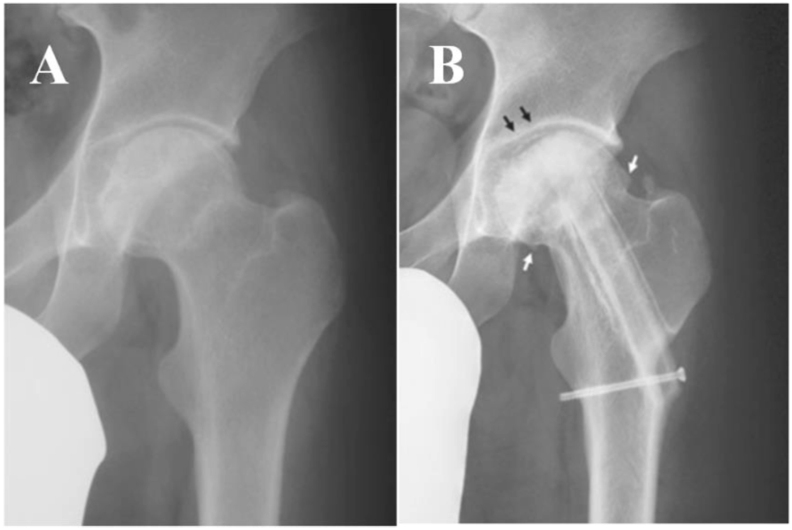
Autologous bone tissues are the common scaffolds for MSCs in the treatment of ONFH in the clinic. Kang et al. [57] combined autologous BMSCs transplantation with auto-iliac cancellous bone grafts to treat ONFH. They believed that this procedure was more suitable for patients who have medium sized lesions. Kawate et al. [58] combined autologous MSCs cultured with beta-tricalcium phosphate (β-TCP) ceramics with a free vascularized fibula to treat three patients with steroid-induced ONFH (Fig. 5). After treatment, osteonecrosis did not progress any further and early bone regeneration was observed. They observed that the tissue engineering approach had great potential for the treatment of osteonecrosis.
Fig. 5.
Combination of transplantation of autologous MSCs cultured with β-TCP ceramics and a free vascularized fibula for the treatment of ONFH. (A) A large necrotic area of femoral head was found in the preoperative X-ray. (B) Although the sclerosis area and osteophytes were found in the latest X-ray, the patient did not feel uncomfortable. Figure is from Ref. [58]. MSCs: mesenchymal stem cells; β-TCP: beta-tricalcium phosphate; ONFH: osteonecrosis of the femoral head.
In addition, synthetic scaffolds characterized by spatial structure and mechanical support similar to normal bone tissue have been applied to ONFH therapies, including DBM, XACB, tantalum rods, biphasic calcium phosphate (BCP) ceramic scaffolds [59], biomimetic synthetic scaffold poly lactide-co-glycolide (PLGA) [60], PLGA calcium phosphate (CPC) composite scaffold [61], and strontium-doped calcium polyphosphate (SCPP) [62]. Kang et al. [62] combined BMSCs with SCPP to treat ONFH in a rabbit model. They found that the combination treatment improved angiogenesis and remodeling into the new trabecular bone by increasing VEGF expression and promoting osteogenesis. Yamasaki et al. [63] evaluated the clinical results of the combination therapy comprising BMMNCs transplantation and interconnected porous calcium hydroxyapatite (IP-CHA) on early bone repair in ONFH. After treatment, the size of the osteonecrotic area in the BMMNC group was reduced, and hypertrophy of the bone was observed in the transition zone. Therefore, the study showed that combined BMMNCs and IP-CHA implantation could promote the repair of ONFH and prevent the collapse of the femoral head.
4.3. The number of transplanted MSCs
The efficacy of MSCs transplantation on ONFH is related to the number of cells in the local femoral head, which can affect the repair capacity of MSCs in the necrotic area. Nevertheless, the optimal number of transplanted cells is still undetermined. In theory, Hernigou et al. [64] suggested that the number of osteoblasts or osteocytes needed for bone reconstruction in ONFH was approximately 3 × 108. In fact, the realization of ideal cell numbers largely depends on the osteogenic differentiation ability of MSCs, and not only the number of transplanted cells. Remarkably, there is a decrease in the number of MSCs in the proximal femur of patients with corticosteroid-induced osteonecrosis [65], which means that the microenvironment of ischemia and hypoxia in the necrotic area can affect the proliferation and differentiation of MSCs. Moreover, the severity and extent of necrosis also need to be evaluated carefully before transplantation of MSCs. Although Hernigou et al. [66] believed that the number of MSCs transplanted should be as large as possible, the safety of maximum transplantation of MSCs needs to be investigated further. According to the current reports, the number of MSCs used in most of the studies ranged from 106 to 109, with the most commonly used cell number being 108 [24,67,68].
5. Comparison between stem cell therapy and other treatments for ONFH
5.1. Stem cell therapy and THA for ONFH - how should we select?
To date, THA, the most effective treatment for ONFH patients in the terminal stages, contributes to the excellent outcomes in pain relief and functional improvement of the hip joint. In the past, it was thought that prosthetic loosening and excessive wear of polyethylene inserts were the primary causes for high failure rate of THA in young patients [69,70]. However, the outcomes of THA in young patients with ONFH have been further augmented due to improvements in operative techniques, prosthesis design, and bearing materials. The study by Byun et al. [71] showed that utilization of third-generation ceramic-on-ceramic bearings in THA could provide excellent clinical results in active patients younger than 30 years. Similar results were found in other studies [72]. In addition, THA was demonstrated to have good implant survival and long-term outcomes for treatment of ONFH in young patients, with a mean time to follow-up of 14 years [73]. Nevertheless, several concerns still remain, such as the potential for ceramic fragmentation, prosthesis impingement, abnormal sounds, dislocation, difficulty and risk of revision. We should note that a 5% revision rate at 8.4 years in patients younger than 30 years after THA was shown in a systematic review and meta-analysis [74]. Thus, after THA, at least one or more revisions will be required in young patients during their lifespan. Meanwhile, a previous study also reported that the complication rate is high, and the survivorship is low in young patients after THA revision [75]. As a result, longer follow-up times and more systematic evaluation for THA in young patients with ONFH need to be performed in future studies.
Compared with THA, stem cell therapy serves as a minimally invasive technique which retains more joint function and facilitates postoperative recovery, in addition to the promotion of bone regeneration in necrotic areas. Importantly, the clinical results of future THA are not affected by stem cell therapy; therefore, this technique can be used as a part of a step-by-step treatment for ONFH.
Taken together, THA is suitable for terminal-stage ONFH with collapse of the femoral head, narrowing of joint space, and osteoarthritis or in patients who have failed joint-preserving treatment; meanwhile, stem cell therapy is considered as a hip-preserving alternative for ONFH.
5.2. Stem cell therapy and basic fibroblast growth factors (bFGFs) for ONFH, which is better?
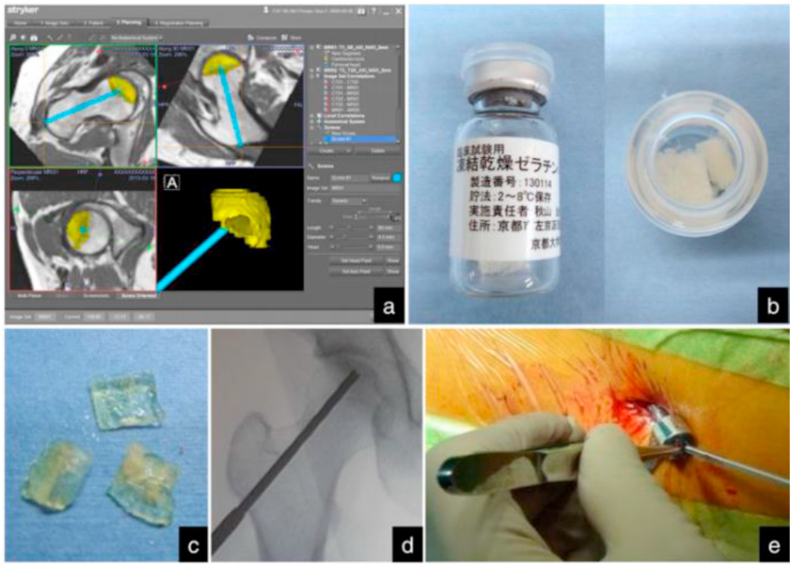
bFGF, also known as fibroblast growth factor-2 (FGF-2), is one of the peptides which exerts the beneficial effects on the modulation of bone remodeling and wound healing by promoting the proliferation and differentiation of MSCs and increasing the regeneration of capillary vasculature [76]. However, the effective half-life period of bFGF is relatively short in vivo due to protein instability, which leads to limitations in the conventional application of bFGF. As a result, a controlled release technique was used for the application of bFGF in previous studies. In an adult rabbit model of ONFH, the necrotic area was significantly repaired, and progression of the femoral head collapse was inhibited after the local injection of recombinant human FGF-2 (rhFGF-2) microspheres [77]. Furthermore, Kuroda et al. [78] used gelatin hydrogel as a slow-release carrier, and performed the direct administration of rhFGF-2-impregnated gelatin hydrogel for the treatment of collapsed ONFH. The authors found that the visual analogue scale (VAS) for pain scores, University of California, Los Angeles (UCLA) activity scores were significantly improved after treatment one year. Meanwhile, bone regeneration in the necrotic area of femoral head was investigated by computed tomography (Fig. 6). Moreover, Moncion et al. [79] used acoustically-responsive scaffolds to control the release of bFGF for angiogenesis. Nevertheless, the optimal concentration, explicit release time, the biologic stability, and long-term clinical results of bFGF need to be confirmed in future studies.
Fig. 6.
Controlled release of rhFGF-2 for the treatment of ONFH. (a) Pre-operative planning. (b) Preparation of rhFGF-2-impregnated gelatin hydrogel. (c) Pieces of the rhFGF-2-impregnated gelatin hydrogel. (d) Intra-operative fluoroscopic image after drilling. (e) Percutaneous administration of the gelatin hydrogel. Figure is from Ref. [78]. rhFGF-2: recombinant human fibroblast growth factor-2; ONFH: osteonecrosis of the femoral head.
Stem cell therapy appears to be simpler and more effective in theory because it is believed that MSCs can be easily harvested and expanded in vitro and be induced to differentiate into osteoblasts and chondrocytes. Furthermore, increasing attention has been paid to the biological function of exosomes secreted from MSCs in stem cell therapy. A number of studies have confirmed that exosomes secreted from MSCs could promote angiogenesis [80,81], osteogenesis [82], cell proliferation and inhibit cell apoptosis [83]. Thus, the paracrine effect of stem cells is another important advantage in the treatment of ONFH, compared with the single application of bFGF. In view of this, MSCs pretreated by cytokines or modified by genes related with osteogenesis and angiogenesis, such as BMP, VEGF, bFGF and TNF, have been widely employed as seed cells for treatment of ONFH in some preclinical trials [[51], [52], [53], [54], [55], [56]].
6. Challenges of stem cells therapy for ONFH
6.1. Confined patient selection
Many studies showed that the efficacy of stem cell therapy on ONFH was related to patient selection, which depends on the stage of ONFH [47]. The reports with a good outcome are mainly focused on patients in stage I and stage II, while the outcomes in stage III or IV cases, especially those with collapse of the femoral head, have proven to be poor [84]. In addition, etiology of ONFH is another important factor that affects clinical outcomes. It was reported that there were better outcomes of stem cell therapy in the patients with post-traumatic ONFH than in the non-traumatic cases [46]. Moreover, the modified Kerboul angle, which is relevant to the collapse of the femoral head, is a risk factor for the failure of stem cell therapy in ONFH [85,86]. In view of this, the decisions on how to improve the accuracy of early diagnosis in ONFH, select appropriate cases, and identify optimal indications remain the primary challenge for stem cell therapy in ONFH.
6.2. Lack of standardized procedures
A successful surgical technique depends on the presence of standardized procedures, and stem cell therapy in ONFH is no exception. Primarily, there is a lack of standardized cell management before operation, including the isolation, expansion, preservation, and the delivery of stem cells. Actually, most stem cells are prepared in research laboratories before clinical application, leading to a lack of sufficient manufacturing quality control. Therefore, the reproducibility of clinical results is not guaranteed. In addition, the surgical operation is not uniform and specific, and many controversies exist in the use of stem cell therapy for ONFH, such as how to choose the source of cells, how to adopt a suitable transplantation method, and how to determine the optimal number of cells for transplantation. Furthermore, the standard operation protocol also needs to be clarified when stem cell therapy is combined with other methods, including core decompression, carriers, and scaffolds.
6.3. Unclear safety
Although mild complications of stem cell therapy in ONFH have been reported [87], including hematoma, wound infection and pain at the site of bone marrow aspiration, the systematic assessment of safety in this technology in long-term follow-up should not be ignored.
Tumorigenicity is the most concerning safety issue. To some extent, some characteristics of stem cells are similar to cancer cells, such as the excellent viability, the relative ability to evade apoptosis, and the extensive proliferation capabilities [88]. As is known, the extensive expansion of MSCs in vitro is required usually to obtain sufficient cell numbers for clinical applications. However, the long-term expansion in vitro could alter the morphology and phenotype of MSCs, and could make MSCs highly tumorigenic, which have led to multiple fast-growing lung deposits after injection into immunodeficient mice [89]. The same result was confirmed by Ren et al. [90]. They found that the spontaneous transformation was observed in MSCs derived from adult cynomolgus monkey following culture in vitro. Importantly, these spontaneously transformed mesenchymal cells (TMCs) had a significant difference in growth patterns and morphology compared to normal MSCs, and had a high level of tumorigenicity, leading to the generation of subcutaneous tumors after injection into NOD/SCID mice. Nevertheless, there is still a lack of enough evidence for malignant transformation in vivo after MSCs transplantation in clinical studies, and the tumorigenicity of stem cells still needs to be elucidated. All in all, a systematic risk assessment in the body should be established to evaluate the safety of stem cell therapy in ONFH.
6.4. Unknown fate of transplanted cells in vivo
Although the efficacy of stem cell therapy in ONFH has been reported in preclinical and clinical trials, the fate of transplanted stem cells in vivo is still unknown. How are transplanted stem cells distributed in vivo? What is the number of transplanted cells in the necrotic area of the femoral head? In terms of arterial perfusion of stem cells, it is unknown whether the transplanted cells will spread with blood flow after transplantation. In addition, it is also not clear whether stem cells undergo mutation, apoptosis, or even necrosis, and how many stem cells differentiate into osteoblasts. In view of this, despite the insufficient research on stem cells tracing due to the limitation of tracer safety, effectiveness and activity, monitoring of transplanted stem cells via cell tracing technology must be pursued [91].
7. Conclusion
ONFH is a progressive disease with complex etiology and unclear pathogenesis and lacks optimal treatment, especially for young patients. With the development of biotechnology, stem cell therapy in ONFH has become the focus of current research. This review summarized the current trends in stem cell therapy in ONFH - from clinical application to related basic research - and showed that an increasing number of studies have confirmed the effectiveness of stem cell therapy in ONFH. However, there still are many unsolved problems and challenges in practical application, such as patient selection, standardized procedures, safety assessment, and the fate of the transplanted cells in the body. Therefore, more studies are required to find the ideal cell sources, appropriate transplantation methods, and optimal number of cells for transplantation. Moreover, systematic safety assessment and stem cell tracing in vivo are also necessary. With the resolution of the above problems, it is certain that stem cell therapy may become the most effective hip-preserving alternative to THA for the treatment of ONFH.
Declaration of competing interest
The authors declare that they have no conflict of interest to this study.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China; Grant number: 81802151; Shandong Province Natural Science Foundation; Grant number: ZR2016HQ05, ZR2017BH089, ZR2019MH012; China Postdoctoral Science Foundation; Grant number: 2018M642616; Qingdao Applied Foundational Research Youth Project; Grant number: 19-6-2-55-cg.
Footnotes
Peer review under responsibility of the Japanese Society for Regenerative Medicine.
Supplementary data related to this article can be found at https://doi.org/10.1016/j.reth.2020.11.003.
References
- 1.Feng Y., Yang S.H., Xiao B.J., Xu W.H., Ye S.N., Xia T. Decreased in the number and function of circulation endothelial progenitor cells in patients with avascular necrosis of the femoral head. Bone. 2010;46:32–40. doi: 10.1016/j.bone.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 2.Kamiya N., Yamaguchi R., Aruwajoye O., Adapala N.S., Kim H.K. Development of a mouse model of ischemic osteonecrosis. Clin Orthop Relat Res. 2015;473:1486–1498. doi: 10.1007/s11999-015-4172-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mont M.A., Hungerford D.S. Non-traumatic avascular necrosis of the femoral head. J Bone Joint Surg Am. 1995;77:459–474. doi: 10.2106/00004623-199503000-00018. [DOI] [PubMed] [Google Scholar]
- 4.Gardeniers J.W.M. Treatment of osteonecrosis by joint replacement. Hip Int. 1998;8:159–166. [Google Scholar]
- 5.Radl R., Hungerford M., Materna W., Rehak P., Windhager R. High failure rate and stem migration of an uncemented femoral component in patients with femoral head osteonecrosis than in patients with osteoarthrosis. Acta Orthop. 2005;76:49–55. doi: 10.1080/00016470510030319. [DOI] [PubMed] [Google Scholar]
- 6.Hartley W.T., McAuley J.P., Culpepper W.J., Engh C.A., Jr., Engh C.A., Sr. Osteonecrosis of the femoral head treated with cementless total hip arthroplasty. J Bone Joint Surg Am. 2000;82:1408–1413. doi: 10.2106/00004623-200010000-00006. [DOI] [PubMed] [Google Scholar]
- 7.Afizah H., Hui J.H. Mesenchymal stem cell therapy for osteoarthritis. J Clin Orthop Trauma. 2016;7:177–182. doi: 10.1016/j.jcot.2016.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee H.S., Huang G.T., Chiang H., Chiou L.L., Chen M.H., Hsieh C.H. Multipotential mesenchymal stem cells from femoral bone marrow near the site of osteonecrosis. Stem Cell. 2003;21:190–199. doi: 10.1634/stemcells.21-2-190. [DOI] [PubMed] [Google Scholar]
- 9.Li C., Li G., Liu M., Zhou T., Zhou H. Paracrine effect of inflammatory cytokine-activated bone marrow mesenchymal stem cells and its role in osteoblast function. J Biosci Bioeng. 2016;121:213–219. doi: 10.1016/j.jbiosc.2015.05.017. [DOI] [PubMed] [Google Scholar]
- 10.Haumer A., Bourgine P.E., Occhetta P., Born G., Tasso R., Martin I. Delivery of cellular factors to regulate bone healing. Adv Drug Deliv Rev. 2018;129:285–294. doi: 10.1016/j.addr.2018.01.010. [DOI] [PubMed] [Google Scholar]
- 11.Wang A., Ren M., Wang J. The pathogenesis of steroid-induced osteonecrosis of the femoral head: a systematic review of the literature. Gene. 2018;671:103–109. doi: 10.1016/j.gene.2018.05.091. [DOI] [PubMed] [Google Scholar]
- 12.Hernigou P., Beaujean F. Treatment of osteonecrosis with autologous bone marrow grafting. Clin Orthop Relat Res. 2002;405:14–23. doi: 10.1097/00003086-200212000-00003. [DOI] [PubMed] [Google Scholar]
- 13.Moya-Angeler J., Gianakos A.L., Villa J.C., Ni A., Lane J.M. Current concepts on osteonecrosis of the femoral head. World J Orthoped. 2015;6:590–601. doi: 10.5312/wjo.v6.i8.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Smith A.J., Dieppe P., Howard P.W., Blom A.W. Failure rates of metal-on-metal hip resurfacings: analysis of data from the National Joint Registry for England and Wales. Lancet. 2012;380:1759–1766. doi: 10.1016/S0140-6736(12)60989-1. [DOI] [PubMed] [Google Scholar]
- 15.Del Pozo J.L., Patel R. Infection associated with prosthetic joints. N Engl J Med. 2009;361:787–794. doi: 10.1056/NEJMcp0905029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ravi B., Pincus D., Wasserstein D., Govindarajan A., Huang A., Austin P.C. Association of overlapping surgery with increased risk for complications following hip surgery: a population based, matched cohort study. JAMA Intern Med. 2018;178:75–83. doi: 10.1001/jamainternmed.2017.6835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Houdek M.T., Wyles C.C., Packard B.D., Terzic A., Behfar A., Sierra R.J. Decreased osteogenic activity of mesenchymal stem cells in patients with corticosteroid-induced osteonecrosis of the femoral head. J Arthroplasty. 2016;31:893–898. doi: 10.1016/j.arth.2015.08.017. [DOI] [PubMed] [Google Scholar]
- 18.Zhu H., Guo Z.K., Jiang X.X., Li H., Wang X.Y., Yao H.Y. A protocol for isolation and culture of mesenchymal stem cells from mouse compact bone. Nat Protoc. 2010;5:550–560. doi: 10.1038/nprot.2009.238. [DOI] [PubMed] [Google Scholar]
- 19.Bai Y., Li P., Yin G., Huang Z., Liao X., Chen X. BMP-2, VEGF and bFGF synergistically promote the osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells. Biotechnol Lett. 2013;35:301–308. doi: 10.1007/s10529-012-1084-3. [DOI] [PubMed] [Google Scholar]
- 20.Xiao Y., Peperzak V., van Rijn L., Borst J., de Bruijn J.D. Dexamethasone treatment during the expansion phase maintains stemness of bone marrow mesenchymal stem cells. J Tissue Eng Regen Med. 2010;4:374–386. doi: 10.1002/term.250. [DOI] [PubMed] [Google Scholar]
- 21.Han N., Li Z., Cai Z., Yan Z., Hua Y., Xu C. P glycoprotein overexpression in bone marrow-derived multipotent stromal cells decreases the risk of steroid induced osteonecrosis in the femoral head. J Cell Mol Med. 2016;20:2173–2182. doi: 10.1111/jcmm.12917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li Z.H., Hu H., Zhang X.Y., Liu G.D., Ran B., Zhang P.G. MiR-291a-3p regulates the BMSCs differentiation via targeting DKK1 in dexamethasone-induced osteoporosis. Kaohsiung J Med Sci. 2020;36:35–42. doi: 10.1002/kjm2.12134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Andriolo L., Merli G., Tobar C., Altamura S.A., Kon E., Filardo G. Regenerative therapies increase survivorship of avascular necrosis of the femoral head: a systematic review and meta-analysis. Int Orthop. 2018;42:1689–1704. doi: 10.1007/s00264-018-3787-0. [DOI] [PubMed] [Google Scholar]
- 24.Rastogi S., Sankineani S.R., Nag H.L., Mohanty S., Shivanand G., Marimuthu K. Intralesional autologous mesenchymal stem cells in management of osteonecrosis of femur: a preliminary study. Musculoskelet Surg. 2013;97:223–228. doi: 10.1007/s12306-013-0273-0. [DOI] [PubMed] [Google Scholar]
- 25.Oedayrajsingh-Varma M.J., van Ham S.M., Knippenberg M., Helder M.N., Klein-Nulend J., Schouten T.E. Adipose tissue-derived mesenchymal stem cell yield and growth characteristics are affected by the tissue-harvesting procedure. Cytotherapy. 2006;8:166–177. doi: 10.1080/14653240600621125. [DOI] [PubMed] [Google Scholar]
- 26.Wyles C.C., Houdek M.T., Crespo-Diaz R.J., Norambuena G.A., Stalboerger P.G., Terzic A. Adipose-derived mesenchymal stem cells are phenotypically superior for regeneration in the setting of osteonecrosis of the femoral head. Clin Orthop Relat Res. 2015;473:3080–3090. doi: 10.1007/s11999-015-4385-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aimaiti A., Saiwulaiti Y., Saiyiti M., Wang Y.H., Cui L., Yusufu A. Therapeutic effect of osteogenically induced adipose derived stem cells on vascular deprivation-induced osteonecrosis of the femoral head in rabbits. Chin J Traumatol. 2011;14:215–220. [PubMed] [Google Scholar]
- 28.Wang H.J., Cai B., Zhao X.Y., Li S.Q., Feng W., Liu J.G. Repairing diabetic rats with bone defect by VEGF165 gene modified adipose-derived stem cells. Zhong Guo Gu Shang. 2017;30:545–551. doi: 10.3969/j.issn.1003-0034.2017.06.012. [DOI] [PubMed] [Google Scholar]
- 29.Abudusaimi A., Aihemaitijiang Y., Wang Y.H., Cui L., Maimaitiming S., Abulikemu M. Adipose-derived stem cells enhance bone regeneration in vascular necrosis of the femoral head in the rabbit. J Int Med Res. 2011;39:1852–1860. doi: 10.1177/147323001103900528. [DOI] [PubMed] [Google Scholar]
- 30.Pak J. Autologous adipose tissue-derived stem cells induce persistent bone-like tissue in osteonecrotic femoral heads. Pain Physician. 2012;15:75–85. [PubMed] [Google Scholar]
- 31.Jansen J., Hanks S., Thompson J.M., Dugan M.J., Akard L.P. Transplantation of hematopoietic stem cells from the peripheral blood. J Cell Mol Med. 2005;9:37–50. doi: 10.1111/j.1582-4934.2005.tb00335.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lie A.K., To L.B. Peripheral blood stem cells: transplantation and beyond. Oncol. 1997;2:40–49. [PubMed] [Google Scholar]
- 33.To L.B., Haylock D.N., Simmons P.J., Juttner C.A. The biology and clinical uses of blood stem cells. Blood. 1997;89:2233–2258. [PubMed] [Google Scholar]
- 34.Song H.J., Lan B.S., Cheng B., Zhang K.F., Yan H.W., Wang W.Z. Peripheral blood stem cell transplantation for ischemic femoral head necrosis. Transplant Proc. 2010;42:1862–1864. doi: 10.1016/j.transproceed.2010.02.077. [DOI] [PubMed] [Google Scholar]
- 35.Song H.J., Lan B.S., Cheng B., Zhang K.F., Yan H.W., Wang W.Z. Treatment of early avascular necrosis of femoral head by small intestinal submucosal matrix with peripheral blood stem cells. Transplant Proc. 2011;43:2027–2032. doi: 10.1016/j.transproceed.2010.12.060. [DOI] [PubMed] [Google Scholar]
- 36.Fu Q., Tang N.N., Zhang Q., Liu Y., Peng J.C., Fang N. Preclinical study of cell terapy for osteonecrosis of the femoral head with allogenic peripheral blood-derived mesenchymal stem cells. Yonsei Med J. 2016;57:1006–1015. doi: 10.3349/ymj.2016.57.4.1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mao Q., Wang W., Xu T., Zhang S., Xiao L., Chen D. Combination treatment of biomechanical support and targeted intra-arterial infusion of peripheral blood stem cells mobilized by granulocyte-colony stimulating factor for the osteonecrosis of the femoral head: a randomized controlled clinical trial. J Bone Miner Res. 2015;30:647–656. doi: 10.1002/jbmr.2390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ying J., Wang P., Ding Q., Shen J., O'Keefe R.J., Chen D. Peripheral blood stem cell therapy does not improve outcomes of femoral head osteonecrosis with cap-shaped separated cartilage defect. J Orthop Res. 2019;14:1–8. doi: 10.1002/jor.24471. [DOI] [PubMed] [Google Scholar]
- 39.Han Z.C. Umbilical cord mesenchymal stem cells (UC-MSC: biology, banking and clinical applications) Bull Acad Natl Med. 2009;193:545–547. [PubMed] [Google Scholar]
- 40.Kuang M.J., Huang Y., Zhao X.G., Zhang R., Ma J.X., Wang D.C. Exosomes derived from Wharton's jelly of human umbilical cord mesenchymal stem cells reduce osteocyte apoptosis in glucocorticoid induced osteonecrosis of the femoral head in rats via the miR-21-PTEN-AKT signalling pathway. Int J Biol Sci. 2019;15:1861–1871. doi: 10.7150/ijbs.32262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen C., Qu Z., Yin X., Shang C., Ao Q., Gu Y. Efficacy of umbilical cord-derived mesenchymal stem cell-based therapy for osteonecrosis of the femoral head: a three-year follow-up study. Mol Med Rep. 2016;14:4209–4215. doi: 10.3892/mmr.2016.5745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cai J., Wu Z., Huang L., Chen J., Wu C., Wang S. Cotransplantation of bone marrow mononuclear cells and umbilical cord mesenchymal stem cells in avascular necrosis of the femoral head. Transplant Proc. 2014;46:151–155. doi: 10.1016/j.transproceed.2013.06.021. [DOI] [PubMed] [Google Scholar]
- 43.Mont M.A., Ragland P.S., Etienne G. Core decompression of the femoral head for osteonecrosis using percutaneous multiple small-diameter drilling. Clin Orthop Relat Res. 2004;429:131–138. doi: 10.1097/01.blo.0000150128.57777.8e. [DOI] [PubMed] [Google Scholar]
- 44.Rajagopal M., Balch Samora J., Ellis T.J. Efficacy of core decompression as treatment for osteonecrosis of the hip: a systematic review. Hip Int. 2012;22:489–493. doi: 10.5301/HIP.2012.9748. [DOI] [PubMed] [Google Scholar]
- 45.Tabatabaee R.M., Saberi S., Parvizi J., Mortazavi S.M., Farzan M. Combining concentrated autologous bone marrow stem cells injection with Core decompression improves outcome for patients with early-stage osteonecrosis of the femoral head: a comparative study. J Arthroplasty. 2015;30:11–15. doi: 10.1016/j.arth.2015.06.022. [DOI] [PubMed] [Google Scholar]
- 46.Sen R.K., Tripathy S.K., Aggarwal S., Marwaha N., Sharma R.R., Khandelwal N. Early results of core decompression and autologous bone marrow mononuclear cells instillation in femoral head osteonecrosis: a randomized control study. J Arthroplasty. 2012;27:679–686. doi: 10.1016/j.arth.2011.08.008. [DOI] [PubMed] [Google Scholar]
- 47.Ma Y., Wang T., Liao J., Gu H., Lin X., Jiang Q. Efficacy of autologous bone marrow buffy coat grafting combined with core decompression in patients with avascular necrosis of femoral head: a prospective, double-blinded, randomized, controlled study. Stem Cell Res Ther. 2014;5:115. doi: 10.1186/scrt505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kinnaird T., Stabile E., Burnett M.S., Lee C.W., Barr S., Fuchs S. Marrow-derived stromal cells express genes encoding a broad spectrum of arteriogenic cytokines and promote in vitro and in vivo arteriogenesis through paracrine mechanisms. Circ Res. 2004;94:678–685. doi: 10.1161/01.RES.0000118601.37875.AC. [DOI] [PubMed] [Google Scholar]
- 49.Qian H.G., Zhu D.X., Zhu J.Z., Zhao J.D., Miao Z.N. Promotive effect of autologous bone marrow mesenchymal stem cell transplantation on angiogenesis in rabbit ischemic limbs. J Clin Rehabilitative Tissue Eng Res. 2008;12:10095–10097. [Google Scholar]
- 50.Jin H., Xia B., Yu N., He B., Shen Y., Xiao L. The effects of autologous bone marrow mesenchymal stem cell arterial perfusion on vascular repair and angiogenesis in osteonecrosis of the femoral head in dogs. Int Orthop. 2012;36:2589–2596. doi: 10.1007/s00264-012-1674-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zuo S.L., Gong Y.K. Application and prospect of bone marrow mesenchymal stem cells and cytokines in the treatment of femoral head necrosis. Chin J Tissue Eng Res. 2012;16:2621–26214. [Google Scholar]
- 52.Wang Z.L., He R.Z., Tu B., He J.S., Cao X., Xia H.S. Drilling combined with adipose-derived stem cells and bone morphogenetic protein-2 to treat femoral head epiphyseal necrosis in juvenile rabbits. Curr Med Sci. 2018;38:277–288. doi: 10.1007/s11596-018-1876-3. [DOI] [PubMed] [Google Scholar]
- 53.Ma X.W., Cui D.P., Zhao D.W. Vascular endothelial growth factor/bone morphogenetic protein-2 bone marrow combined modification of the mesenchymal stem cells to repair the avascular necrosis of the femoral head. Int J Clin Exp Med. 2015;8:15528–15534. [PMC free article] [PubMed] [Google Scholar]
- 54.Peng W.X., Wang L. Adenovirus-mediated expression of BMP-2 and BFGF in bone marrow mesenchymal stem cells combined with demineralized bone matrix for repair of femoral head osteonecrosis in Beagle dogs. Cell Physiol Biochem. 2017;43:1648–1662. doi: 10.1159/000484026. [DOI] [PubMed] [Google Scholar]
- 55.Zhang F., Peng W.X., Wang L., Zhang J., Dong W.T., Wu J.H. Role of FGF-2 transfected bone marrow mesenchymal stem cells in engineered bone tissue for repair of avascular necrosis of femoral head in rabbits. Cell Physiol Biochem. 2018;48:773–784. doi: 10.1159/000491906. [DOI] [PubMed] [Google Scholar]
- 56.Peng W., Dong W., Zhang F., Wang J., Zhang J., Wu J. Effects of transplantation of FGF-2-transfected MSCs and XACB on TNF-α expression with avascular necrosis of the femoral head in rabbits. Biosci Rep. 2019;39:1–10. doi: 10.1042/BSR20180765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kang J.S., Moon K.H., Kim B.S., Kwon D.G., Shin S.H., Shin B.K. Clinical results of auto-iliac cancellous bone grafts combined with implantation of autologous bone marrow cells for osteonecrosis of the femoral head: a minimum 5 year follow-up. Yonsei Med J. 2013;54 doi: 10.3349/ymj.2013.54.2.510. 510-505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kawate K., Yajima H., Ohgushi H., Kotobuki N., Sugimoto K., Ohmura T. Tissue-engineered approach for the treatment of steroid-induced osteonecrosis of the femoral head: transplantation of autologous mesenchymal stem cells cultured with beta-tricalcium phosphate ceramics and free vascularized fibula. Artif Organs. 2006;30:960–962. doi: 10.1111/j.1525-1594.2006.00333.x. [DOI] [PubMed] [Google Scholar]
- 59.Peng J., Wen C., Wang A., Wang Y., Xu W., Zhao B. Micro-CT-based bone ceramic scaffolding and its performance after seeding with mesenchymal stem cells for repair of load-bearing bone defect in canine femoral head. J Biomed Mater Res B Appl Biomater. 2011;96:316–325. doi: 10.1002/jbm.b.31770. [DOI] [PubMed] [Google Scholar]
- 60.Liao H., Zhong Z., Liu Z., Li L., Ling Z., Zou X. Bone mesenchymal stem cells co-expressing VEGF and BMP-6 genes to combat avascular necrosis of the femoral head. Exp Ther Med. 2018;15:954–962. doi: 10.3892/etm.2017.5455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang H.X., Zhang X.P., Xiao G.Y., Hou Y., Cheng L., Si M. In vitro and in vivo evaluation of calcium phosphate composite scaffolds containing BMP-VEGF loaded PLGA microspheres for the treatment of avascular necrosis of the femoral head. Mater Sci Eng C Mater Biol Appl. 2016;60:298–307. doi: 10.1016/j.msec.2015.11.055. [DOI] [PubMed] [Google Scholar]
- 62.Kang P., Xie X., Tan Z., Yang J., Shen B., Zhou Z. Repairing defect and preventing collapse of femoral head in a steroid-induced osteonecrotic of femoral head animal model using strontium-doped calcium polyphosphate combined BM-MNCs. J Mater Sci Mater Med. 2015;26:80. doi: 10.1007/s10856-015-5402-x. [DOI] [PubMed] [Google Scholar]
- 63.Yamasaki T., Yasunaga Y., Ishikawa M., Hamaki T., Ochi M. Bone-marrow-derived mononuclear cells with a porous hydroxyapatite scaffold for the treatment of osteonecrosis of the femoral head: a preliminary study. J Bone Joint Surg Br. 2010;92:337–341. doi: 10.1302/0301-620X.92B3.22483. [DOI] [PubMed] [Google Scholar]
- 64.Hernigou P., Trousselier M., Roubineau F., Bouthors C., Chevallier N., Rouard H. Stem cell therapy for the treatment of hip osteonecrosis: a 30-year review of progress. Clin Orthop Surg. 2016;8:1–8. doi: 10.4055/cios.2016.8.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hernigou P., Beaujean F., Lambotte J.C. Decrease in the mesenchymal stem-cell pool in the proximal femur in corticosteroid-induced osteonecrosis. J Bone Joint Surg Br. 1999;81:349–355. doi: 10.1302/0301-620x.81b2.8818. [DOI] [PubMed] [Google Scholar]
- 66.Zhao D., Cui D., Wang B., Tian F., Guo L., Yang L. Treatment of early stage osteonecrosis of the femoral head with autologous implantation of bone marrow-derived and cultured mesenchymal stem cells. Bone. 2012;50:325–330. doi: 10.1016/j.bone.2011.11.002. [DOI] [PubMed] [Google Scholar]
- 67.Pepke W., Kasten P., Beckmann N.A., Janicki P., Egermann M. Core decompression and autologous bone marrow concentrate for treatment of femoral head osteonecrosis: a randomized prospective study. Orthop Rev (Pavia) 2016;8:6162. doi: 10.4081/or.2016.6162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lim Y.W., Kim Y.S., Lee J.W., Kwon S.Y. Stem cell implantation for osteonecrosis of the femoral head. Exp Mol Med. 2013;45:e61. doi: 10.1038/emm.2013.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kim Y.H., Oh J.H., Oh S.H. Cementless total hip arthroplasty in patients with osteonecrosis of the femoral head. Clin Orthop Relat Res. 1995;320:73–84. [PubMed] [Google Scholar]
- 70.Stulberg B.N., Singer R., Goldner J., Stulberg J. Uncemented total hip arthroplasty in osteonecrosis: a 2- to 10-year evaluation. Clin Orthop Relat Res. 1997;334:116–123. [PubMed] [Google Scholar]
- 71.Byun J.W., Yoon T.R., Park K.S., Seon J.K. Third-generation ceramic-on-ceramic total hip arthroplasty in patients younger than 30 years with osteonecrosis of femoral head. J Arthroplasty. 2012;27:1337–1343. doi: 10.1016/j.arth.2011.07.004. [DOI] [PubMed] [Google Scholar]
- 72.Baek S.H., Kim S.Y. Cementless total hip arthroplasty with alumina bearings in patients younger than fifty with femoral head osteonecrosis. J Bone Joint Surg Am. 2008;90:1314–1320. doi: 10.2106/JBJS.G.00755. [DOI] [PubMed] [Google Scholar]
- 73.Swarup I., Shields M., Mayer E.N., Hendow C.J., Burket J.C., Figgie M.P. Outcomes after total hip arthroplasty in young patients with osteonecrosis of the hip. Hip Int. 2017;27:286–292. doi: 10.5301/hipint.5000457. [DOI] [PubMed] [Google Scholar]
- 74.Walker R.P., Gee M., Wong F., Shah Z., George M., Bankes M.J. Functional outcomes of total hip arthroplasty in patients aged 30 years or less: a systematic review and meta-analysis. Hip Int. 2016;26:424–431. doi: 10.5301/hipint.5000376. [DOI] [PubMed] [Google Scholar]
- 75.Girard J., Glorion C., Bonnomet F., Fron D., Migaud H. Risk factors for revision of hip arthroplasties in patients younger than 30 years. Clin Orthop Relat Res. 2011;469:1141–1147. doi: 10.1007/s11999-010-1669-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kuroda Y., Kawai T., Goto K., Matsuda S. Clinical application of injectable growth factor for bone regeneration: a systematic review. Inflamm Regen. 2019;39:20. doi: 10.1186/s41232-019-0109-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kuroda Y., Akiyama H., Kawanabe K., Tabata Y., Nakamura T. Treatment of experimental osteonecrosis of the hip in adult rabbits with a single local injection of recombinant human FGF-2 microspheres. J Bone Miner Metabol. 2010;28:608–616. doi: 10.1007/s00774-010-0172-5. [DOI] [PubMed] [Google Scholar]
- 78.Kuroda Y., Asada R., So K., Yonezawa A., Nankaku M., Mukai K. A pilot study of regenerative therapy using controlled release of recombinant human fibroblast growth factor for patients with pre-collapse osteonecrosis of the femoral head. Int Orthop. 2016;40:1747–1754. doi: 10.1007/s00264-015-3083-1. [DOI] [PubMed] [Google Scholar]
- 79.Moncion A., Lin M., O'Neill E.G., Franceschi R.T., Kripfgans O.D., Putnam A.J. Controlled release of basic fibroblast growth factor for angiogenesis using acoustically-responsive scaffolds. Biomaterials. 2017;140:26–36. doi: 10.1016/j.biomaterials.2017.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gong M., Yu B., Wang J., Wang Y., Liu M., Paul C. Mesenchymal stem cells release exosomes that transfer miRNAs to endothelial cells and promote angiogenesis. Oncotarget. 2017;8:45200–45212. doi: 10.18632/oncotarget.16778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Guo S.C., Tao S.C., Yin W.J., Qi X., Sheng J.G., Zhang C.Q. Exosomes from human synovial-derived mesenchymal stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head in the rat. Int J Biol Sci. 2016;12:1262–1272. doi: 10.7150/ijbs.16150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fang S., Li Y., Chen P. Osteogenic effect of bone marrow mesenchymal stem cell-derived exosomes on steroid-induced osteonecrosis of the femoral head. Drug Des Dev Ther. 2018;13:45–55. doi: 10.2147/DDDT.S178698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Spees J.L., Lee R.H., Gregory C.A. Mechanisms of mesenchymal stem/stromal cell function. Stem Cell Res Ther. 2016;7:125. doi: 10.1186/s13287-016-0363-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hauzeur J.P., De Maertelaer V., Baudoux E., Malaise M., Beguin Y., Gangji V. Inefficacy of autologous bone marrow concentrate in stage three osteonecrosis: a randomized controlled double-blind trial. Int Orthop. 2017;7:1429–1435. doi: 10.1007/s00264-017-3650-8. [DOI] [PubMed] [Google Scholar]
- 85.Houdek M.T., Wyles C.C., Collins M.S., Howe B.M., Terzic A., Behfar A. Stem cells combined with platelet-rich plasma effectively treat corticosteroid-induced osteonecrosis of the hip: a prospective study. Clin Orthop Relat Res. 2018;476:388–397. doi: 10.1007/s11999.0000000000000033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ha Y.C., Jung W.H., Kim J.R., Seong N.H., Kim S.Y., Koo K.H. Prediction of collapse in femoral head osteonecrosis: a modified Kerboul method with use of magnetic resonance images. J Bone Joint Surg Am. 2006;88:35–40. doi: 10.2106/JBJS.F.00535. [DOI] [PubMed] [Google Scholar]
- 87.Piuzzi N.S., Chahla J., Schrock J.B., LaPrade R.F., Pascual-Garrido C., Mont M.A. Evidence for the use of cell-based therapy for the treatment of osteonecrosis of the femoral head: a systematic review of the literature. J Arthroplasty. 2017;32:1698–1708. doi: 10.1016/j.arth.2016.12.049. [DOI] [PubMed] [Google Scholar]
- 88.Wang Y., Han Z.B., Song Y.P., Han Z.C. Safety of mesenchymal stem cells for clinical application. Stem Cell Int. 2012;2012:652034. doi: 10.1155/2012/652034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Rosland G.V., Svendsen A., Torsvik A., Sobala E., McCormack E., Immervoll H. Long-term cultures of bone marrow-derived human mesenchymal stem cells frequently undergo spontaneous malignant transformation. Canc Res. 2009;69:5331–5339. doi: 10.1158/0008-5472.CAN-08-4630. [DOI] [PubMed] [Google Scholar]
- 90.Ren Z., Wang J., Zhu W., Guan Y., Zou C., Chen Z. Spontaneous transformation of adult mesenchymal stem cells from cynomolgus macaques in vitro. Exp Cell Res. 2011;317:2950–2957. doi: 10.1016/j.yexcr.2011.09.008. [DOI] [PubMed] [Google Scholar]
- 91.Santelli J., Lechevallier S., Baaziz H., Vincent M., Martinez C., Mauricot R. Multimodal gadolinium oxysulfide nanoparticles: a versatile contrast agent for mesenchymal stem cell labeling. Nanoscale. 2018;10:16775–16786. doi: 10.1039/c8nr03263g. [DOI] [PubMed] [Google Scholar]