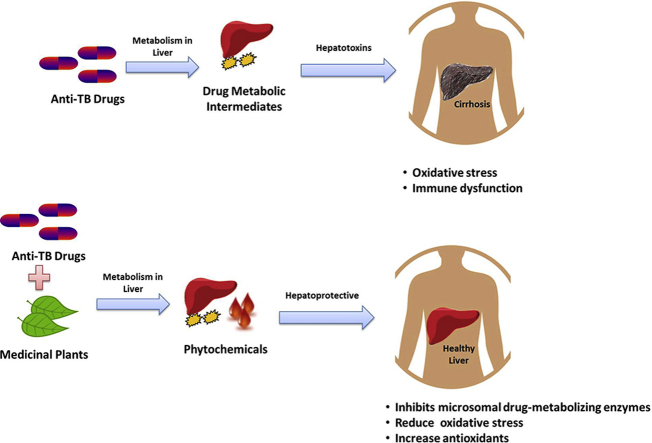
Abstract
The effectiveness of herbs for the management of chemically induced hepatotoxicity has been discussed by many researchers. However, there is a paucity of compressive literature on the significance of hepatoprotective plants for the management of anti-TB drug induced toxicity. Anti-TB drugs have been reported to causes hepatic damage, due to which, many patients across the globe discontinued the treatment. Medicinal plants have multiple therapeutic effects. The assessment of biological activity of plants against Mycobacterium and its use for hepatic recovery provides an effective treatment approach. Traditionally used medicinal plants are the rich source of phytochemicals and secondary metabolites. These compounds can restore normal function, enzymatic activity and structure of hepatic cells against anti-TB drug induced hepatotoxicity. The present review covers comprehensive details on different hepatoprotective and antimycobacterial plants studied during past few decades so that potential adjuvants can be studied for Tuberculosis chemotherapy.
Keywords: Anti-TB, Antimicrobial, Hepatoprotective, Phytochemicals, Secondary metabolites
Graphical abstract
1. Introduction
Tuberculosis (TB) is one of the leading causes of mortality worldwide. According to World Health Organization (WHO), 10.4 million TB patients were reported in 2016, with annual death of 1.7 million. Of global total, India had 26% of reported TB death in the same year [1]. Mycobacterium tuberculosis complex (MTC) includes related Mycobacterium strains i.e. M. tuberculosis, Mycobacterium bovis, M. africanum and other which cause TB disease [2]. Existing strategies to treat TB are for active tuberculosis and does not address associated side effects. Over the past few years, worldwide search for new anti-TB drug has increased due to emergence of MDR-TB and extensively-drug resistant (XDR) TB. The key objective is to discover and develop new drugs that can improve treatment strategy, enhance safety and address both infection and side effects [3]. In absence of effective therapeutic drugs for TB, hope is built on plant based natural products due to their chemical diversity and important role as phyto-drugs [4]. Medicinal plants are undying gift of nature that is used for the treatment of numerous diseases in human beings since time immemorial. According to an estimate of WHO, 80% population in developing countries rely on traditional medicines for the primary health care [5]. The use of medicinal plants extract and phytochemicals is of great significance for the treatment of various ailments including TB. Traditionally used medicinal plants and its parts i.e. root of Calpurnia aurea, seeds of Ocimum basilicum, leaves of Artemisia abyssinica, Croton macrostachyus, Artemisia afra and Eucalyptus camaldulensis possess anti-TB potential under in vitro conditions [2], [3].
The side effects of anti-TB drugs are GIT manifestations, hepatotoxicity, ototoxicity, nephrotoxicity, skin rashes, fever, peripheral neuritis and rarely psychotic changes [6]. The first line anti-TB drugs; rifampicin (RIF), isoniazid (INH) and pyrazinamide (PZA) are linked with toxicological responses inside tissues, predominantly in the liver, which lead to hepatitis [7]. Hepatotoxicity caused by anti-TB drugs is one of the major reason for the withdrawal of treatment by patients as well as development of Multi Drug Resistance (MDR) TB [8]. Management of patients with TB/MDR-TB necessitates multi-chemotherapy, which further have toxic and adverse effects on individual's health [9]. Anti-TB drugs such as RIF can cause hyperbilirubinemia by inhibiting bile salt export pump [10]. Moreover, it also affects the activity of key enzymes such as alkaline phosphatase (ALP), serum glutamic oxaloacetic transaminase (SGOT or AST), serum glutamic pyruvic transaminase (SGPT or ALT), and γ-Glutamyl transpeptidase (γ-GT) [11]. In case of any hepatic anomalies, the plant extract prevents cellular damage and can significantly restore normal level of hepatic enzymes. Many studies support hepatoprotective activities of glycosides, flavonoids, triterpenes and phenolic group of compounds extracted from plants [12], [13], [14]. These phytochemicals possess antioxidants and free radical scavenging properties which, prevent excessive lipid peroxidation in liver, decrease catalase, superoxide dismutase and glutathione [12]. The hepatoprotective activity of plant derived chemically defined molecules paved the way for the exploration of new effective hepatoprotective agents. The current review covers the use of such different hepatoprotective herbs which has a potential to be as anti-TB drugs.
2. Anti-TB drugs and hepatic damage
In a study, about 5–20% of individuals, under anti-TB medication were reported with hepatic complications. It also gets aggravated by drug combinations regime [13]. RIF, INH, PZA, ethionamide (EMB) and para-aminosalicylic acid (PAS) are reported to cause hepatitis anomalies [10]. Anti-TB drugs are one of the common causes for hepatotoxicity worldwide [12]. Clinical symptoms include jaundice, nausea, vomiting and abdominal pain along with elevated levels of bilirubin and hepatic transaminases [14]. The morbidity and mortality in TB increases due to anti-TB drug related hepatotoxicity. Under such condition, the mortality rate after onset of liver disease in TB patient is 6–12% if the use of drug is continued [15].
Liver is an important organs regulates various physiological processes. It contributes towards synthesis of vital molecules such as albumin, fibrinogen, cholesterol and bile acid. It is a source of enzymes proficient in transforming foreign molecules. As a result, it metabolizes and detoxifies numerous heterochthonous (drugs, insecticides, etc.) or autochthonous chemicals (steroids, fatty acids etc.) [16]. Using Cytochrome P450, detoxification of drug in the body ensues three phases i.e. transformation, conjugation and transportation into bile. Any deformity in either of the phases can be the basis of hepatotoxicity. The covalent binding of the drug or its metabolite with host proteins and the consequential oxidative stress, is foremost cause of hepatic injury [8], [10]. Oxidative stress could damage intracellular macromolecules such as glutathione, RNA, DNA, lipids, proteins and ATP [17]. Hepatotoxins also induces an inflammatory response in liver along with activation of MAPKs, NFκB and STAT3 signaling pathways [18].
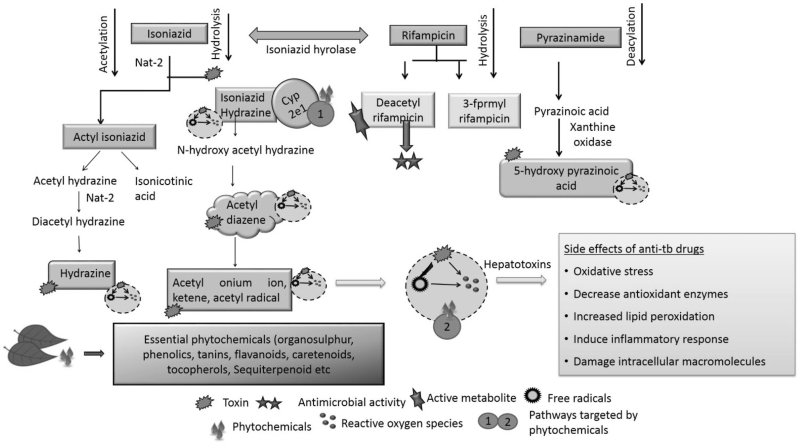
The metabolism of a drug produces many intermediates which cause hepatic damage. For instance, metabolism of INH forms acetyl diazine. This further produces acetyl onium ion, acetyl radical and ketene (Fig. 1). An enzyme, N-acetyltransferase 2 (NAT2) through acetylation, converts acetyl hydrazine to diacetyl hydrazine (DAH). DAH is not toxic to cells. In presence of INH, acetylation process is suppressed. As a result, non-acetylated hydrazines are produced, which can be toxic and cause liver injury [8], [19]. Similarly, PZA induces oxidative stress and hepatotoxicity via 5-hydroxypyrazinoic acid. PZA forms pyrazinoic acid by the activation of amidase. Pyrazinoic acid is then hydroxylated to 5-hydroxypyrazinoic acid through xanthine oxidase and cause hepatic distress [20]. RIF also cause toxicity in combination with other drugs. RIF is an inducer of enzymatic pathways such as Cytochrome P450 (CYP3A4) via hepatocyte Xeno Sensing Pregnane X Receptor (PXR). During anti-TB drug combination regime, RIF could increase the metabolism of INH leading to the generation of toxic intermediates. It also increases hydrazine production by inducing isoniazid hydrolases. Hydrazine in turn altered the expression of proteins related to lipid metabolism [21]. Prolong exposure to RIF can distress membrane permeability and considerably reduces the activity of glucose-6-phosphate. This could be one of the cause for the high lipid peroxidation in liver [22], [23], [24], [25]. Thus, collectively anti-TB drugs produce toxic metabolites, reactive oxygen species (ROS) and free radicals, the major sources of liver injury. Anti-TB drug also interferes with lipid deposition, induced by activating CYP2E1, fatty acid accumulation and LDL uptake [11], [25], [26].
Fig. 1.
Anti-TB drug toxic metabolic intermediates and protective effect of phytochemicals (adapted from Sharma and Sharma [14]).
3. Antimycobacterial and hepatoprotective activity of plants
Since ancient time, ethnobotanical knowledge subsists in India and people use herbs as a source of medicines, especially for primary healthcare. The country has about 45,000 plant species and many of them have been studied for their medicinal properties [16]. As mentioned above, due to acute toxicity and associated side effects of existing chemical medications there is an increase in inclination towards traditional source of medicines. Although the synthetic drugs are effective for the prevention, cure and management of countless diseases, but their use can cause surplus health vulnerabilities [27]. Plants are rich source of biologically active compounds, proven to be effective antimicrobial agents. Traditionally, many plants are used to treat Mycobacterium infections. Phytochemicals present in plants are reported to inhibits multidrug efflux system of microbes. In a study by Sharma et al. [28] piperine was reported to inhibit a putative multidrug efflux pump (Rv1258c) of M. tuberculosis. Another important phytochemical with anti-bacterial properties is Berberine. This compound has potential for the treatment of bacterial infection. Berberine can interrupt bacterial colonization or biofilm formation by binding with amyloid proteins. Berberine also competes with lipopolysaccharides for binding with TLR4/MD-2. This competence can result in an inhibition of inflammation during infection [29]. Saritha et al. [30] also reported mechanisms responsible for antibacterial activity of phytochemicals, which includes disruption of bacterial cell membrane, permeabilization and leakage of cellular contents. Thus, comparable to synthetic drug, phytochemicals also interfere with various cellular mechanisms to inhibit the growth of bacteria. Coumarin, allicin, andrographolide, glabridin and vasicine acetate are some examples of phytochemicals studied in recent years for their anti-mycobacterial properties [31], [32], [33], [34]. Plants are also effective against MDR-TB. Plant such as Acalypha indica, Adhatoda vasica, Allium sativum Andrographis paniculata and Cassia sophera had shown significant antimycobacterial activity against MDR-TB in vitro [35].
Traditional plants used for the treatment of TB are also a rich source of phytochemicals, which have several health benefits including on liver. Phytochemicals are well established hepatoprotective agents which can restore normal function, enzymatic activity and histology of hepatic cells against anti-TB drugs or chemical induced hepatotoxicity. The herbs can lower the elevated levels of serum enzymes, total bilirubin and protein. They can also restore the anomalous activity of enzymatic antioxidants and liver damage caused by anti-TB drugs [12], [24], [36]. For example, Withanolide from the plant Withania somnifera has anti-oxidant properties and reduces hepatocyte necrosis, serum ALT levels, and intrahepatic hemorrhage in vitro [37]. Apium graveolens, A. indica, A. paniculata, Ficus religiosa, Fumaria indica, Glycyrrhiza glabra, Syzygium aromaticum, W. somnifera, Tinospora cordifolia etc. are traditionally used plants for the treatment of TB which also possess hepatoprotective properties. These plants are rich source of alkaloids, flavonoids, diterpenoid, tannins, lipids, sterols etc. with anti-microbial and hepatic protective effects. Several traditionally used anti-TB plants possess anti-microbial activity against MTB-H37RV [31], [34], [37]. Supplementary Table S1 summarizes list of plant traditionally used for the treatment of TB and have antimicrobial, antitubercular and hepatoprotective activity.
4. Herbs: as an adjunct to tuberculosis chemotherapy to prevent hepatotoxicity
WHO recommended 6–9 months Directly Observed Therapy Short (DOTs) course for the treatment of TB. Many times, drugs may induce hepatic damage with DOTs combination regime. The prime cause of anti-TB drug hepatic induced damage is distinct metabolic reactions [38], [39]. At present, to decrease the thereafter effects and upsurge efficacy of medications is the basic constraint. Single or in combination, herbs are efficient in minimizing the side effects associated with drugs. The ethnomedicinal uses of plants against liver disease are well documented but its awareness is also important for the treatment [40]. Plants are rich source of broad spectrum secondary metabolites which can contribute towards the discovery of novel drug moiety for the target diseases [41].
The hepatoprotective activity of many plants against anti-TB drug induced hepatic damage studied in different animal models. Moringa oleifera leaf is rich source of phytochemicals such as alkaloids, flavonoids, carbohydrates, glycosides, saponins, tannins, and terpenoids. Pari and Kumar [42] reported that oral administration of M. oleifera leaf extract can repair normal liver activity in rat against INH, PZA and RIF induced hepatic damage. Leaf extract appears to improve the recovery from hepatic damage and restore regular functioning of enzymes ALS, AST, alkaline phosphatase, lipid peroxidation and bilirubin in the serum. Cassia auriculata root extract can significantly lower the elevated serum ALP, AST, ALT, total bilirubin, cholesterol and protein in anti-TB drug induced hepatotoxicity. It also maintains normal levels of enzymatic antioxidants and Malondialdehyde (MDA); a marker of oxidative stress [43]. Terminalia chebula a highly valued medicinal plant in Ayurvedic pharmacopeia has antioxidant and cell membrane stabilizing activities. It's fruits can prevent hepatotoxicity caused by the administration of anti-TB drug combination regime [7]. Herbal formulations containing T. chebula are also hepatoprotective. In a study, Sankar et al. [44] reported that polyherbal formulation of Eclipta alba, Tephrosia purpurea, Curcuma longa, Picrrohiza kurooa, W. somnifera, Phyllanthus amarus, Pinius succinifera, Pistacia lentiscus, Orchis mascula, and Cycas circinalis is effective against oxidative hepatic injury caused by RIF and INH in rats. Medicinal plants and TB drug combination regime to enhance the treatment efficacy is also supported by many clinical trials. In a study by Sharma et al. [12] three groups of patients receiving anti-TB drug were studied for 12 weeks. First group of patients received capsule containing extract of Aloe vera whole plant, Berberis aristata root and Solanum nigrum whole plant. Second and third groups were given decoction of Phyllanthus fraternus and placebo starch capsule respectively. At the end of trial, liver enzyme activity and level were at normal range in first and second group. However, in third group, a rise in ALT and AST was observed. Both ALT and ATS were used as marker enzymes for hepatotoxicity study. B. aristata is an active source of both antitubercular and hepatoprotective phytochemicals [45], [46], [47]. Whereas, S. nigrum is a potent antioxidant plant which can modulate the function of enzymes involved in the detoxification of chemicals and exhibits free radical scavenging properties [48]. Similar kind of hepatoprotective properties possessed by Phyllanthus fraternu [49]. In another study, Debnath et al. [50] used adjunct Ayurvedic therapy with Aswagandha for the management of pulmonary tuberculosis in patient receiving anti-TB drugs. The study claimed that Aswagandha can modulate immune system, restore normal SGPT and SGOT level, and also increase bioavailability of PYZ and INH within 28 days of treatment. Number of in vitro studies conducted on animal models using plant such as C. auriculata, F. religiosa, Lawsonia inermis, M. oleifera, T. chebula, T. cordifolia, W. somnifera etc. also supports the benefits of medicinal plants in reducing hepatic damage caused by anti-TB drugs (Table S1). Anti-TB and Ayurvedic drug combination regime can increase the survival rate in pulmonary TB patients. Some clinical study revealed that patients when received treatment with TB drugs, the cure and death rates of 11.42 % and 40.9 % respectively was observed. However, when the patients received TB drug and Ayurvedic drug combination regime the respective cure and death rate was 41.3% and 3.8% [50]. Few systematic studies also support effective use of Ayurveda for the management of pulmonary tuberculosis [51]. The herbal preparations can considerably avert hepatotoxicity, increase treatment outcome and its use is deprived of any toxicity or side effects [52].
5. Hepatoprotective phytochemicals and mode of action
Phytochemicals are effective in management of anti-TB drugs induced toxicity in many ways. Mainly they act on some major processes which are responsible for toxicity of drug. Phytochemicals act on Cytochrome P450, free radicals or reactive oxygen species (Fig. 1). These molecules are involved in the process of lipid peroxidation, oxidative stress and loss of membrane integrity which collectively are basis of liver injury [53]. For example; flavanoids, tannins and carotenoids act on reactive oxygen species. Phenolics, organosulphur compounds and electrophilic constituents such as picroside II and kurtkoside possess free radical scavenging properties. Organosulphur compounds and flavanoids also act on Cytochrome P450 activity there by affecting metabolism of drugs [12], [25], [39]. In this context phytochemicals from several plants can reduce hepatotoxicity (Table 1). Detail studies on blood components, liver function, histopathological and cellular machinery reveals the hepatoprotective activity of herbal extracts through different mechanism [16]. Curcumin, silymarin, stilbenes, quercetin, berberine and ursolic acid are some hepatoprotective natural products.
Table 1.
List of Hepatoprotective phytochemicals sources, structure and mode of action.
| Phytochemical | Structure | Source | Mode of action | References |
|---|---|---|---|---|
| Andrographolide | 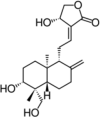 |
Andrographis paniculata |
|
Chen et al. [86] |
| Berberine | 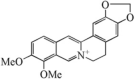 |
Berberis aristata |
|
Domitrovic and Iva Potocnjak [63] Potdar et al. [46] |
| Curcumin | 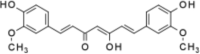 |
Curcuma longa |
|
Zhang et al. [79] Payton et al. [87] |
| Piperine | 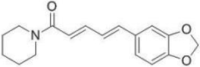 |
Piper longum, Piper nigrum, Acalypha indica etc. |
|
Sabina et al. [88] |
| Quercetin | 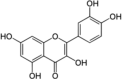 |
Vitis vinefera, Allium cepa, Camellia sinensis etc. |
|
Liu et al. [27] Nam et al. [55] |
| Resveratrol | 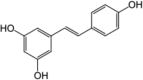 |
Paeonia lactiflora, Vitis vinifera, Arachis hypogaea etc. |
|
Nicoletti et al. [85] |
| Silymarin |
|
Silybum marianum |
|
Abouzid et al. [89] Zhang et al. [66] |
| Thymoquinone | 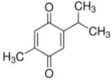 |
Nidorella anomala |
|
Raschi et al. [90] Mollazadeh and Hosseinzadeh [91] |
| Ursolic acid | 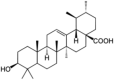 |
Bouvardia ternifolia, Byrsonima crassa, Calendula officinalis, etc. |
|
Liu et al. [56] Ma et al. [58] Singh et al. [24] |
Quercetin, a polyphenolic flavonol occurs in fruits and vegetables. It interact with various interacellar signaling cascades to provide protection from oxidative damages [54]. This compound activates Nrf2/HO-1 pathway and expression of oxidative stress-related genes. Thus helpful in overcoming oxidative hepatic damage [27], [54], [55].
Ursolic acid, a triterpenoid extracted from in plants Hedyotis corymbosa, Bouvardia ternifolia, Byrsonima crassa, Calendula officinalis, Mirabilis jalapa etc. It is a potent antioxidant and inhibitor of ROS production in mice. It suppresses MAPKs, CYP2E1 and NF-κB activation thereby protecting liver from stress [56], [57], [58], [59].
Berberine, an alkaloid possesses antioxidant properties and ameliorative effect on liver. It is found in plants such as B. aristata, Hydrastis canadensis, Coptis chinensis, Berberis aquifolium, Berberis vulgaris and Hydrastis Canadensis [60]. In vitro, berberine exhibits hepatoprotective action in experimental model partly through inhibition of microsomal drug-metabolizing enzymes and inhibitory action K/Ca currents [61], [62]. It also decreases oxidative stress by repressing TNF-α, COX-2 and iNOS expression [63].
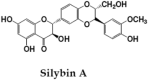
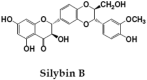
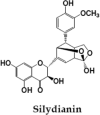
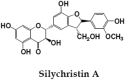
Silymarin, is a flavonoid-complex containing silybin, silydianin and silychristin [24]. It is one among the extensively studied hepatoprotective natural product. Seeds of Silybum marianum are source of Silymarin, which in turn is very effective against anti-TB drug induced toxicity and used as hepatoprotective reference compound in many experimental studies [64], [65]. It has cell protective properties due to its antioxidant potential and interactions with cell membrane components. This compound exhibits anti-inflammatory activities by down-regulating the expression of inflammatory genes; NF-κB, ICAM-1 and IL-6 [66].
Thymoquinone(TQ) is a monoterpenoid found in the seeds of Nigella sativa. It improves hepatic antioxidant level thereby preventing oxidative stress in liver. In experimental model, it increases the synthesis of chemoprotective enzyme glutathione peroxidase and superoxide dismutase. During drug induced inflammation, TNF-α, iNOS, COX2 and IL-1β gets activated. TQ can inactivate their activity, thereby minimizing hepatic damage. TQ interact with oxidative stress induced factors (e.g. glutathione S-transferase) and fix oxidative damage [67], [68], [69], [70], [71], [72], [73].
Curcumin is a polyphenolic compound obtained from rhizomes of Curcuma species. It exhibits hepatoprotection by activation of Keap1/Nrf2 pathway. The pathway regulates cellular protection under oxidative stress. Curcumin can also downregulates expression of NADPH oxidase in liver (NOX). This enzyme generates ROS, which in turn cause hepatic damage [74], [75]. Curcumin also induces enzymes such as heme oxygenase-1 and NAD(P)H quinone dehydrogenase 1. Both are phase II detoxification and antioxidant enzymes, which are involved in drug metabolism and detoxification [75], [76], [77]. Curcumin also reduces oxidative stress by reducing CYP2E1 as well as Prx1 expression. In vitro, attenuation of inflammatory responses in liver by curcumin is by down regulating the expression of NF-κB, TLR2 and TLR4 has been reported. This phytochemical also activates AMP-activated protein kinase (AMPK) pathway in liver cells [77], [78], [79], [80].
Andrographolide is a diterpenoid with antioxidant properties obtained from A. paniculata. It up-regulates the expression of hypoxia-inducible factor-1 alpha (HIF-1α), SOD-1, HO-1 and GST under oxidative stress [24], [81], [82]. Interaction of this diterpenoid with Glutathione (GSH) significantly induces CYP1A1 expression in experimental models. The expression of CYP1A1 play important role in drug metabolism, whereas GSH is an important defensive compound against oxidative stress in liver. Andrographolide synergistically induces CYP1A1 expression [83], [84].
Stilbenes are widely documented phytochemicals whose hepatoprotective mechanism is studied in vitro. Each of them keeps different machinery to protect hepatic cells from damage. Stilbenes such as trans-resveratrol and its glucoside are found in variety of plants such as Paeonia lactiflora, Vitis vinifera and Arachis hypogaea. Resveratrol (trans-3,5,4′-trihydroxystilbene,1) is most studied hepatoprotective phytochemical. Resveratrol prevents ISH and RIF induced hepatic damage in mice. The defensive effect of this phytochemical is due to modulation of SIRT1, PPAR-γ and PGC-1α mRNA expression in hepatic cells. SIRT1 is involved in the liver lipid metabolism pathways thereby by protecting liver from oxidative stress [63], [85].
6. Conclusion
Nature ensures a widespread stock of plants used to cure illness of mankind. As a primogenital and major framework of traditional medical systems, herbs have made a myriad impact to maintain the human health. The efficacy of chemically distinct molecules and plants as potential antimycobacterial and hepatoprotective agent exemplify progress in the search for epitome medicines. Thus, combining target specific properties of anti-TB drugs with multiple health benefits of medicinal plants could be a positive way out for the management of tuberculosis and associated side effects. In developing countries, many patients concurrently take prescribed drugs as well as herbal supplements. Consequently, this predominant prevalence need to be counterbalanced with relevant research. The underlying mechanism behind anti-TB drug and herbal constituent interaction has not been addressed widely. There exist huge knowledge gap regarding medical use of herbal adjuvant by the attending physician. The plants with inherited anti-oxidant and anti-tubercular properties may be explored for their active biomolecules and used for developing modern formulations forms, accepted to wider group of clinicians. Thus, to ensure safety and efficacy of such combinations there is need to precisely study herb–drug interaction in different standard experimental set-ups, before large scale human use.
Source(s) of funding
None declared
Conflict of interest
None
Acknowledgement
Authors are grateful to National Innovation Foundation India for providing facilities for Research and Development.
Footnotes
Peer review under responsibility of Transdisciplinary University, Bangalore.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jaim.2019.02.004.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Organization W.H. Geneva World Heal Organ; 2017. Global tuberculosis report 2017. [Google Scholar]
- 2.Gemechu A., Giday M., Worku A., Ameni G. In vitro Anti-mycobacterial activity of selected medicinal plants against Mycobacterium tuberculosis and Mycobacterium bovis Strains. BMC Complement Altern Med. 2013;13:291. doi: 10.1186/1472-6882-13-291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ntutela S., Smith P., Matika L., Mukinda J., Arendse H., Allie N. Efficacy of Artemisia afra phytotherapy in experimental tuberculosis. Tuberculosis. 2009;89:S33–S40. doi: 10.1016/S1472-9792(09)70009-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Patwardhan B., Vaidya A.D.B., Chorghade M. Ayurveda and natural products drug discovery. Curr Sci. 2004;86:789–799. [Google Scholar]
- 5.Ekor M. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014;4:177. doi: 10.3389/fphar.2013.00177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.El Din Tag M.A., El Maraghy A.A., Abdel Hay A.H. Adverse reactions among patients being treated for multi-drug resistant tuberculosis at Abbassia Chest Hospital. Egypt J Chest Dis Tuberc. 2015;64:939–952. [Google Scholar]
- 7.Tasduq S.A., Singh K., Satti N.K., Gupta D.K., Suri K.A., Johri R.K. Terminalia chebula (fruit) prevents liver toxicity caused by sub-chronic administration of rifampicin, isoniazid and pyrazinamide in combination. Hum Exp Toxicol. 2006;25:111–118. doi: 10.1191/0960327106ht601oa. [DOI] [PubMed] [Google Scholar]
- 8.Ramappa V., Aithal G.P. Hepatotoxicity related to anti-tuberculosis drugs: mechanisms and Management. J Clin Exp Hepatol. 2013;3:37–49. doi: 10.1016/j.jceh.2012.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kumar P., Balooni V., Sharma B.K., Kapil V., Sachdeva K.S., Singh S. High degree of multi-drug resistance and hetero-resistance in pulmonary TB patients from Punjab state of India. Tuberculosis. 2014;94:73–80. doi: 10.1016/j.tube.2013.10.001. [DOI] [PubMed] [Google Scholar]
- 10.Saukkonen J.J., Cohn D.L., Jasmer R.M., Schenker S., Jereb J.A., Nolan C.M. An official ATS statement: hepatotoxicity of antituberculosis therapy. Am J Respir Crit Care Med. 2006;174:935–952. doi: 10.1164/rccm.200510-1666ST. [DOI] [PubMed] [Google Scholar]
- 11.Anundi I., Lähteenmäki T., Rundgren M., Moldeus P., Lindros K.O. Zonation of acetaminophen metabolism and cytochrome P450 2E1-mediated toxicity studied in isolated periportal and perivenous hepatocytes. Biochem Pharmacol. 1993;45:1251–1259. doi: 10.1016/0006-2952(93)90277-4. [DOI] [PubMed] [Google Scholar]
- 12.Sharma Y.K., Singh H., Mehra B.L. Hepatoprotective effect of few Ayurvedic herbs in patients receiving antituberculus treatment. Indian J Tradit Knowl. 2004;4:391–396. [Google Scholar]
- 13.Arbex M.A., Varella M.D., Siqueira De H.R., Mello De F.A.F. Antituberculosis drugs: drug interactions, adverse effects, and use in special situations. Part 1: first-line drugs. J Bras Pneumol. 2010;36:626–640. doi: 10.1590/s1806-37132010000500016. [DOI] [PubMed] [Google Scholar]
- 14.Sharma R., Sharma V.L. Review: treatment of toxicity caused by anti-tubercular drugs by use of different herbs. Int J Pharma Sci Res. 2015;6:1288–1294. [Google Scholar]
- 15.Hassen Ali A., Belachew T., Yami A., Ayen W.Y. Anti-tuberculosis drug induced hepatotoxicity among TB/HIV co-infected patients at Jimma University Hospital, Ethiopia: nested case-control study. PLoS One. 2013;8:e64622. doi: 10.1371/journal.pone.0064622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bedi O., Bijjem K.R.V., Kumar P., Gauttam V. Herbal induced hepatoprotection and hepatotoxicity: a critical review. Indian J Physiol Pharmacol. 2016;60:6–21. [PubMed] [Google Scholar]
- 17.Bhattacharyya A., Chattopadhyay R., Mitra S., Crowe S.E. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev. 2014;94:329–354. doi: 10.1152/physrev.00040.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ambade A., Mandrekar P. Oxidative stress and inflammation: essential partners in alcoholic liver disease. Int J Hepatol. 2012;2012 doi: 10.1155/2012/853175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Scales M.D., Timbrell J.A. Studies on hydrazine hepatotoxicity. 1. Pathological findings. J Toxicol Environ Health. 1982;10:941–953. doi: 10.1080/15287398209530308. [DOI] [PubMed] [Google Scholar]
- 20.Shih T.-Y., Pai C.-Y., Yang P., Chang W.-L., Wang N.-C., Hu O.Y.-P. A novel mechanism underlies the hepatotoxicity of pyrazinamide. Antimicrob Agents Chemother. 2013;57:1685–1690. doi: 10.1128/AAC.01866-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Guldberg Klenø T., Rønnedal Leonardsen L., Ørsted Kjeldal H., Møller Laursen S., Nørregaard Jensen O., Baunsgaard D. Mechanisms of hydrazine toxicity in rat liver investigated by proteomics and multivariate data analysis. Proteomics. 2004;4:868–880. doi: 10.1002/pmic.200300663. [DOI] [PubMed] [Google Scholar]
- 22.Koster J.F., Slee R.G. Lipid peroxidation of rat liver microsomes. Biochim Biophys Acta (BBA) Lipids Lipid Metab. 1980;620:489–499. doi: 10.1016/0005-2760(80)90141-1. [DOI] [PubMed] [Google Scholar]
- 23.Saraswathy S.D., Shyamala Devi C.S. Modulating effect of Liv.100, an ayurvedic formulation on antituberculosis drug-induced alterations in rat liver microsomes. Phytherapy Res. 2001;15:501–505. doi: 10.1002/ptr.770. [DOI] [PubMed] [Google Scholar]
- 24.Singh D., Cho W.C., Upadhyay G. Drug-induced liver toxicity and prevention by herbal antioxidants: an Overview. Front Physiol. 2016;6:36. doi: 10.3389/fphys.2015.00363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Santhosh S., Sini T.K., Anandan R., Mathew P.T. Effect of chitosan supplementation on antitubercular drugs-induced hepatotoxicity in rats. Toxicology. 2006;219:53–59. doi: 10.1016/j.tox.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 26.Upadhyay G., Kumar A., Singh M.P. Effect of silymarin on pyrogallol- and rifampicin-induced hepatotoxicity in mouse. Eur J Pharmacol. 2007;565:190–201. doi: 10.1016/j.ejphar.2007.03.004. [DOI] [PubMed] [Google Scholar]
- 27.Liu X., Chen C., He W., Huang P., Liu M., Wang Q. Exploring anti-TB leads from natural products library originated from marine microbes and medicinal plants. Antonie Leeuwenhoek. 2012;102:447–461. doi: 10.1007/s10482-012-9777-0. [DOI] [PubMed] [Google Scholar]
- 28.Sharma S., Kumar M., Sharma S., Nargotra A., Koul S., Khan Piperine as an inhibitor of Rv1258c, a putative multidrug efflux pump of Mycobacterium tuberculosis. J Antimicrob Chemother. 2010:651694–651701. doi: 10.1093/jac/dkq186. [DOI] [PubMed] [Google Scholar]
- 29.Chu M., Ding R., Chu Z., Zhang M., Liu X., Xie S. Role of berberine in anti-bacterial as a high-affinity LPS antagonist binding to TLR4/MD-2 receptor. BMC Complement Altern Med. 2014;14:89. doi: 10.1186/1472-6882-14-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Saritha K., Rajesh A., Manjulatha K., Setty O.H., Yenugu S. Mechanism of antibacterial action of the alcoholic extracts of Hemidesmus indicus (L.) R. Br. ex Schult, Leucas aspera (Wild.), Plumbago zeylanica L., and Tridax procumbens (L.) R. Br. ex Schult. Front Microbiol. 2015;6:577. doi: 10.3389/fmicb.2015.00577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gupta V.K., Fatima A., Faridi U., Negi A.S., Shanker K., Kumar J.K. Antimicrobial potential of Glycyrrhiza glabra roots. J Ethnopharmacol. 2008;116:377–380. doi: 10.1016/j.jep.2007.11.037. [DOI] [PubMed] [Google Scholar]
- 32.Ignacimuthu S., Shanmugam N. Antimycobacterial activity of two natural alkaloids, vasicine acetate and 2-acetyl benzylamine, isolated from Indian shrub Adhatoda vasica Ness. leaves. J Biosci. 2010;35:565–570. doi: 10.1007/s12038-010-0065-8. [DOI] [PubMed] [Google Scholar]
- 33.Kontogiorgis C., Detsi A., Hadjipavlou-Litina D. Coumarin-based drugs: a patent review (2008–present) Expert Opin Ther Pat. 2012;22:437–454. doi: 10.1517/13543776.2012.678835. [DOI] [PubMed] [Google Scholar]
- 34.Singh R., Hussain S., Verma R., Sharma P. Anti-mycobacterial screening of five Indian medicinal plants and partial purification of active extracts of Cassia sophera and Urtica dioica. Asian Pac J Trop Med. 2013;6:366–371. doi: 10.1016/S1995-7645(13)60040-1. [DOI] [PubMed] [Google Scholar]
- 35.Subramani R., Narayanasamy M., Feussner K.D. Plant-derived antimicrobials to fight against multi-drug-resistant human pathogens. 3 Biotech. 2017;7:172. doi: 10.1007/s13205-017-0848-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Madrigal-Santillán E., Madrigal-Bujaidar E., Álvarez-González I., Sumaya-Martínez M.T., Gutiérrez-Salinas J., Bautista M. Review of natural products with hepatoprotective effects. World J Gastroenterol. 2014;20:14787–14804. doi: 10.3748/wjg.v20.i40.14787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jadeja R.N., Urrunaga N.H., Dash S., Khurana S., Saxena N.K. Withaferin-a reduces acetaminophen-induced liver injury in Mice. Biochem Pharmacol. 2015;97:122–132. doi: 10.1016/j.bcp.2015.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rivers E.C., Mancera R.L. New anti-tuberculosis drugs with novel mechanisms of action. Curr Med Chem. 2008;15:1956–1967. doi: 10.2174/092986708785132906. [DOI] [PubMed] [Google Scholar]
- 39.Sonika U., Kar P. Tuberculosis and liver disease: management issues. Trop Gastroenterol. 2012;33:102–106. doi: 10.7869/tg.2012.25. [DOI] [PubMed] [Google Scholar]
- 40.Amadi C.N., Orisakwe O.E. Herb-induced liver injuries in developing Nations: an update. Toxics. 2018;6:24. doi: 10.3390/toxics6020024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cragg G.M., Newman D.J. Natural products: a continuing source of novel drug leads. Biochim Biophys Acta Gen Subj. 2013;1830:3670–3695. doi: 10.1016/j.bbagen.2013.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pari L., Kumar N.A. Hepatoprotective activity of Moringa oleifera on antitubercular drug-induced liver damage in rats. J Med Food. 2002;5:171–177. doi: 10.1089/10966200260398206. [DOI] [PubMed] [Google Scholar]
- 43.Jaydeokar A.V., Bandawane D.D., Bibave K.H., Patil T.V. Hepatoprotective potential of Cassia auriculata roots on ethanol and antitubercular drug-induced hepatotoxicity in experimental models. Pharm Biol. 2014;52:344–355. doi: 10.3109/13880209.2013.837075. [DOI] [PubMed] [Google Scholar]
- 44.Sankar M., Rajkumar J., Sridhar D. Hepatoprotective activity of heptoplus on isoniazid and rifampicin induced liver damage in rats. Indian J Pharm Sci. 2015;77:556. doi: 10.4103/0250-474x.169028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mahapatra A., Maheswari V., Kalia N.P., Rajput V.S., Khan I.A. Synthesis and antitubercular activity of berberine derivatives. Chem Nat Compd. 2014;50:321–325. [Google Scholar]
- 46.Potdar D., Hirwani R.R., Dhulap S. Phyto-chemical and pharmacological applications of Berberis aristata. Fitoterapia. 2012;83:817–830. doi: 10.1016/j.fitote.2012.04.012. [DOI] [PubMed] [Google Scholar]
- 47.Unkeshwar P., Nasiruddin M., Fayazuddin M., Khan R.A., Khan A.A., Tajuddin Evaluation of hepatoprotective activity of Berberis aristata against carbon tetrachloride induced hepatotoxicity in rats. Int J Pharm Pharm Sci. 2013;5:107–110. [Google Scholar]
- 48.Lin H.M., Tseng H.C., Wang C.J., Lin J.J., Lo C.W., Chou F.P. Hepatoprotective effects of Solanum nigrum Linn extract against CCl4-iduced oxidative damage in rats. Chem Biol Interact. 2008;171:283–293. doi: 10.1016/j.cbi.2007.08.008. [DOI] [PubMed] [Google Scholar]
- 49.Bera T.K., Chatterjee K., De D., Ali K.M., Jana K., Maiti S. Hepatoprotective activity of Livshis, a polyherbal formulation in CCl4-induced hepatotoxic male Wistar rats: a toxicity screening approach. Genom Med Biomarkers Health Sci. 2011;3:103–110. [Google Scholar]
- 50.Debnath P., Mitra A., Alam M., Hazra J., Chattopadhyay J., Adhikari A. Adjunct therapy of Ayurvedic medicine with anti tubercular drugs on the therapeutic management of pulmonary tuberculosis. J Ayurveda Integr Med. 2012;3:141. doi: 10.4103/0975-9476.100180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Samal J. Ayurvedic management of pulmonary tuberculosis: a systematic review. J Intercult Ethnopharmacol. 2015;5(1):86–91. doi: 10.5455/jice.20151107020621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Adhvaryu M.R., Reddy N.M., Vakharia B.C. Prevention of hepatotoxicity due to anti tuberculosis treatment: a novel integrative approach. World J Gastroenterol. 2008;14:4753–4762. doi: 10.3748/wjg.14.4753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gu X., Manautou J.E. Molecular mechanisms underlying chemical liver injury. Expert Rev Mol Med. 2012;14:e4. doi: 10.1017/S1462399411002110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liu C.M., Ma J.Q., Xie W.R., Liu S.S., Feng Z.J., Zheng G.H. Quercetin protects mouse liver against nickel-induced DNA methylation and inflammation associated with the Nrf2/HO-1 and p38/STAT1/NF-kappaB pathway. Food Chem Toxicol. 2015;82:19–26. doi: 10.1016/j.fct.2015.05.001. [DOI] [PubMed] [Google Scholar]
- 55.Nam J.S., Sharma A.R., Nguyen L.T., Chakraborty C., Sharma G., Lee S.S. Application of bioactive quercetin in oncotherapy: from nutrition to nanomedicine. Molecules. 2016;21:E108. doi: 10.3390/molecules21010108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Liu J. Pharmacology of oleanolic acid and ursolic acid. J Ethnopharmacol. 1995;49:57–68. doi: 10.1016/0378-8741(95)90032-2. [DOI] [PubMed] [Google Scholar]
- 57.Kanter M., Meral I., Dede S., Cemek M., Ozbek H., Uygan I. Effects of Nigella sativa L. and Urtica dioica L. on lipid peroxidation, antioxidant enzyme systems and some liver enzymes in CCl4 treated rats. J Vet Med Ser A. 2003;50:264–268. doi: 10.1046/j.1439-0442.2003.00537.x. [DOI] [PubMed] [Google Scholar]
- 58.Ma J.Q., Ding J., Zhang L., Liu C.M. Protective effects of ursolic acid in an experimental model of liver fibrosis through Nrf2/ARE pathway. Clin Res Hepatol Gastroenterol. 2015;39:188–197. doi: 10.1016/j.clinre.2014.09.007. [DOI] [PubMed] [Google Scholar]
- 59.Kashyap D., Tuli H.S., Sharma A.K. Ursolic acid (UA): a metabolite with promising therapeutic potential. Life Sci. 2016;146:201–213. doi: 10.1016/j.lfs.2016.01.017. [DOI] [PubMed] [Google Scholar]
- 60.Asgari Z., J Selwyn B., Vonville H., L DuPont H. A systematic review of the evidence for use of herbal medicine for the treatment of acute diarrhea. Nat Prod J. 2012;2:1–8. [Google Scholar]
- 61.Gilani A.H., Janbaz K.H. Prevention of acetaminophen-induced liver damage by Berberis aristata leaves. Biochem Soc Trans. 1992;20:347S. doi: 10.1042/bst020347s. [DOI] [PubMed] [Google Scholar]
- 62.Wang F., Zhou H.Y., Zhao G., Fu L.Y., Cheng L., Chen J.G. Inhibitory effects of berberine on ion channels of rat hepatocytes. World J Gastroenterol. 2004;10:2842–2845. doi: 10.3748/wjg.v10.i19.2842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Domitrovic R., Potocnjak I. A comprehensive overview of hepatoprotective natural compounds: mechanism of action and clinical perspectives. Arch Toxicol. 2016;90:39–79. doi: 10.1007/s00204-015-1580-z. [DOI] [PubMed] [Google Scholar]
- 64.Eminzade S., Uraz F., Izzettin F.V. Silymarin protects liver against toxic effects of anti-tuberculosis drugs in experimental animals. Nutr Metab (Lond) 2008;5:18. doi: 10.1186/1743-7075-5-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Marjani M., Baghaei P., Dizaji M.K., Bayani P.G., Fahimi F., Tabarsi P. Evaluation of hepatoprotective effect of silymarin among under treatment tuberculosis patients: a randomized clinical trial. Iran J Pharm Res IJPR. 2016;15:247–252. [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang W., Hong R., Tian T. Silymarin's protective effects and possible mechanisms on alcoholic fatty liver for rats. Biomol Ther (Seoul) 2013;21:264–269. doi: 10.4062/biomolther.2013.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Al-Malki A.L., Sayed A.A.R. Thymoquinone attenuates cisplatin-induced hepatotoxicity via nuclear factor kappa-beta. BMC Complement Altern Med. 2014;14:282. doi: 10.1186/1472-6882-14-282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Khader M., Eckl P.M. Thymoquinone: an emerging natural drug with a wide range of medical applications. Iran J Basic Med Sci. 2014;17:950–957. [PMC free article] [PubMed] [Google Scholar]
- 69.Mansour M.A., Ginawi O.T., El-Hadiyah T., El-Khatib A.S., Al-Shabanah O.A., Al-Sawaf H.A. Effects of the volatile oil constituents of Nigella sativa on carbon tetrachloride induced hepatotoxicity in mice: evidence for antioxidant effects of thymoquinone. Res Commun Mol Pathol Pharmacol. 2001;110:239–251. [PubMed] [Google Scholar]
- 70.Prabhakar P., Reeta K.H., Maulik S.K., Dinda A.K., Gupta Y.K. Protective effect of thymoquinone against high-fructose diet-induced metabolic syndrome in rats. Eur J Nutr. 2015;54:1117–1127. doi: 10.1007/s00394-014-0788-7. [DOI] [PubMed] [Google Scholar]
- 71.Sayed-Ahmed M.M., Aleisa A.M., Al-Rejaie S.S., Al-Yahya A.A., Al-Shabanah O.A., Hafez M.M. Thymoquinone attenuates diethylnitrosamine induction of hepatic carcinogenesis through antioxidant signaling. Oxid Med Cell Longev. 2010;3:254–261. doi: 10.4161/oxim.3.4.12714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Noorbakhsh M.-F., Hayati F., Samarghandian S., Shaterzadeh-Yazdi H., Farkhondeh T. An overview of hepatoprotective effects of thymoquinone. Recent Pat Food, Nutr Agric. 2018;9:14–22. doi: 10.2174/2212798410666180221105503. [DOI] [PubMed] [Google Scholar]
- 73.Tekbas A., Huebner J., Settmacher U., Dahmen U. Plants and surgery: the protective effects of thymoquinone on hepatic injury-A systematic review of in vivo studies. Int J Mol Sci. 2018;1085 doi: 10.3390/ijms19041085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Palipoch S., Punsawad C., Koomhin P., Suwannalert P. Hepatoprotective effect of curcumin and alpha-tocopherol against cisplatin-induced oxidative stress. BMC Complement Altern Med. 2014;14:111. doi: 10.1186/1472-6882-14-111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Farombi E.O., Shrotriya S., Na H.K., Kim S.H., Surh Y.J. Curcumin attenuates dimethylnitrosamine-induced liver injury in rats through Nrf2-mediated induction of heme oxygenase-1. Food Chem Toxicol. 2008;46:1279–1287. doi: 10.1016/j.fct.2007.09.095. [DOI] [PubMed] [Google Scholar]
- 76.Gao S., Duan X., Wang X., Dong D., Liu D., Li X. Curcumin attenuates arsenic-induced hepatic injuries and oxidative stress in experimental mice through activation of Nrf2 pathway, promotion of arsenic methylation and urinary excretion. Food Chem Toxicol. 2013;59:739–747. doi: 10.1016/j.fct.2013.07.032. [DOI] [PubMed] [Google Scholar]
- 77.Xu D., Hu L., Su C., Xia X., Zhang P., Fu J. Tetrachloro-p-benzoquinone induces hepatic oxidative damage and inflammatory response, but not apoptosis in mouse: the prevention of curcumin. Toxicol Appl Pharmacol. 2014;280:305–313. doi: 10.1016/j.taap.2014.08.003. [DOI] [PubMed] [Google Scholar]
- 78.Bisht S., Khan M.A., Bekhit M., Bai H., Cornish T., Mizuma M. A polymeric nanoparticle formulation of curcumin (NanoCurc) ameliorates CCl4-induced hepatic injury and fibrosis through reduction of pro-inflammatory cytokines and stellate cell activation. Lab Investig. 2011;91:1383–1395. doi: 10.1038/labinvest.2011.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhang J., Xu L., Zhang L., Ying Z., Su W., Wang T. Curcumin attenuates d-galactosamine/lipopolysaccharide–induced liver injury and mitochondrial dysfunction in mice. J Nutr. 2014;144:1211–1218. doi: 10.3945/jn.114.193573. [DOI] [PubMed] [Google Scholar]
- 80.Liu Z., Cui C., Xu P., Dang R., Cai H., Liao D. Curcumin activates AMPK Pathway and regulates lipid metabolism in rats following prolonged clozapine exposure. Front Neurosci. 2017;11:558. doi: 10.3389/fnins.2017.00558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chen H.W., Huang C.S., Li C.C., Lin A.H., Huang Y.J., Wang T.S. Bioavailability of andrographolide and protection against carbon tetrachloride-induced oxidative damage in rats. Toxicol Appl Pharmacol. 2014;280:1–9. doi: 10.1016/j.taap.2014.07.024. [DOI] [PubMed] [Google Scholar]
- 82.Ye J.F., Zhu H., Zhou Z.F., Xiong R.B., Wang X.W., Su L.X. Protective mechanism of andrographolide against carbon tetrachloride-induced acute liver injury in mice. Biol Pharm Bull. 2011;34:1666–1670. doi: 10.1248/bpb.34.1666. [DOI] [PubMed] [Google Scholar]
- 83.Kondo S., Chatuphonprasert W., Jaruchotikamol A., Sakuma T., Nemoto N. Cellular glutathione content modulates the effect of andrographolide on β-naphthoflavone-induced CYP1A1 mRNA expression in mouse hepatocytes. Toxicology. 2011;280:18–23. doi: 10.1016/j.tox.2010.11.002. [DOI] [PubMed] [Google Scholar]
- 84.Jaruchotikamol A., Jarukamjorn K., Sirisangtrakul W., Sakuma T., Kawasaki Y., Nemoto N. Strong synergistic induction of CYP1A1 expression by andrographolide plus typical CYP1A inducers in mouse hepatocytes. Toxicol Appl Pharmacol. 2007;224:156–162. doi: 10.1016/j.taap.2007.07.008. [DOI] [PubMed] [Google Scholar]
- 85.Nicoletti N.F., Rodrigues-Junior V., Santos A.A., Jr., Leite C.E., Dias A.C.O., Batista E.L., Jr. Protective effects of resveratrol on hepatotoxicity induced by isoniazid and rifampicin via SIRT1 modulation. J Nat Prod. 2014;77:2190–2195. doi: 10.1021/np5003143. [DOI] [PubMed] [Google Scholar]
- 86.Chen H.W., Huang C.S., Liu P.F., Li C.C., Chen C.T., Liu C.T. Andrographis paniculata extract and andrographolide modulate the hepatic drug metabolism system and plasma tolbutamide concentrations in rats. Evid Based Complement Alternat Med. 2013;2013:982689. doi: 10.1155/2013/982689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Payton F., Sandusky P., Alworth W.L. NMR study of the solution structure of curcumin. J Nat Prod. 2007;70:143–146. doi: 10.1021/np060263s. [DOI] [PubMed] [Google Scholar]
- 88.Sabina E.P., Souriyan A.D.H., Jackline D., Rasool M.K. Piperine, an active ingredient of black pepper attenuates acetaminophen–induced hepatotoxicity in mice. Asian Pac J Trop Med. 2010;3:971–976. [Google Scholar]
- 89.Abouzid S.F., Chen S.N., Pauli G.F. Silymarin content in Silybum marianum populations growing in Egypt. Ind Crops Prod. 2016;83:729–737. doi: 10.1016/j.indcrop.2015.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Raschi A.B., Romano E., Benavente A.M., Altabef A Ben, Tuttolomondo M.E. Structural and vibrational analysis of thymoquinone. Spectrochim Acta Part A Mol Biomol Spectrosc. 2010;77:497–505. doi: 10.1016/j.saa.2010.06.026. [DOI] [PubMed] [Google Scholar]
- 91.Mollazadeh H., Hosseinzadeh H. The protective effect of Nigella sativa against liver injury: a review. Iran J Basic Med Sci. 2014;17:958–966. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.