Abstract
Background
Prunella vulgaris, commonly known as self-heal, has been extensively used in the traditional system of medicines. The plant has been found to contain a number of bioactive molecules including those having radical scavenging property which indicates its potential for the treatment of those diseases which are induced by free radical damage like drug-induced hepatotoxicity.
Objective
The current study was undertaken to investigate the flavonoid and total phenolic content and evaluate the hepatoprotective potential of various extracts obtained from floral spikes of P. vulgaris.
Material and methods
Flavonoid and otal phenolic contents were obtained from the standard curves of Gallic acid as per the reported methods. The extent of hepatotoxicity induced by paracetamol (500 mg/kg b.w, p.o daily for 14 days), hepatoprotective potential of extracts (200 mg/kg b.w/day, orally) and standard drug silymarin (50 mg/kg b.w/day, orally) were evaluated by analyzing various biochemical parameters like Serum Glutamic Oxaloacetic Transaminase, Serum Glutamic Pyruvic Transaminase, Alkaline Phosphatase, Total Proteins, Total and Direct Bilirubin and detailed histopathology of rat livers.
Results
Methanolic extract showed higher quantity of flavonoids and total phenolic content followed by ethanolic, hydroalcoholic and aqueous extracts. Treatment of rats with extracts showed a highly significant reduction in the enzyme activities of Serum Glutamic Oxaloacetic Transaminase, Serum Glutamic Pyruvic Transaminase, Alkaline Phosphatase, and serum levels of Total, Direct Bilirubin (P < 0.01) and highly significant elevation in Total Proteins (P < 0.01) when compared with the toxic control group. This was further confirmed by histopathological evaluation, where almost normal hepatic architecture or very less hepatic damage was observed in groups treated with extracts and silymarin compared to paracetamol treated group. Results from biochemical and histopathological evaluation indicated that among the extracts methanolic extract was most effective.
Conclusion
From the results, it can be concluded that the extracts obtained from floral spikes of P. vulgaris possess highly significant hepatoprotective activity which could be attributed to its radical scavenging potential and hepatic regeneration. This is further authenticated by the presence of phenolic and flavonoids which are known to possess radical scavenging properties.
Keywords: Prunella vulgaris, Hepatoprotective potential, Extracts, Paracetamol, Silymarin
1. Introduction
Herbal medicines play an important role in the primary health sector, mainly in the developing countries, which may be attributed to a number of factors such as better cultural acceptability, biological friendliness or better compatibility with the human body and lesser side effects. However, this could be further contributed to a lack of access to the modern system of medicines [1]. There are a number of metabolic disorders like hepatotoxicity, atherosclerosis, diabetes, obesity etc. for which no satisfactory treatment is available under the modern system of medicine. Research work on natural products including traditional herbal medicines is mainly focussed with the aim of providing scientific validation about the potency and efficacy of the drugs used in traditional medicine and provides a rationale about the use of these drugs for the treatment of such disorders [2]. Liver, being a major metabolizing and detoxifying organ, is liable to injury from an enormous array of therapeutic and environmental chemicals. Poor prognosis and high mortality rates are because of the lack of effective treatment options. These factors prompt to screen herbal drugs with the aim of producing new hepatoprotective drugs with better biological activity and lesser side effects [3]. Paracetamol, a hepatotoxic agent, is commonly used as a model for evaluating the hepatoprotective effect of a drug [4,5]. Its metabolism produces toxic intermediates like N-acetyl-para-benzoquinoneimine (NAPQI), which under normal conditions conjugates with glutathione. However, at supratherapeutic doses, metabolizing enzymes become saturated resulting in an increase in the concentration of NAPQI [6]. Hepatocellular injury is caused by binding of NAPQI to cell proteins which are followed by a cascade of intracellular events including the production of reactive oxidative ions resulting in mitochondrial oxidative stress, activating stress proteins. Besides the immune system of the liver may be activated inducing further damage to the hepatocellular system. Gene transcription may also produce such mediators which may incur further damage. Under such circumstances, protective and destructive pathways operate simultaneously and the type of balance between the two would ultimately decide whether there is recovery or cell death [7,8]. Silymarin, a flavonoid isolated from Silybum marianum, is used in clinical practice as an effective antidote and hepatoprotective drug. Thus it has been used as a standard drug in the screening of various agents as possible hepatoprotective drugs [9,10].
Prunella vulgaris commonly known as self-heal, a low growing perennial herb, has been extensively used for treating various ailments like inflammation, eye pain, headache, sore throat, herpetic keratitis, dizziness and wound healing in traditional system of medicine [11,12]. The plant has been evaluated for different biological activities. Aqueous extract of P. vulgaris has been found to have a protective effect on insulin-secreting cell line and reducing their apoptosis. Besides, it has been found to reduce inflammatory cytokine expression in these cells [13]. Caffeic acid ethylene ester isolated from the plant has been reported to show the anti-diabetic effect [14]. Aqueous and ethanol extracts from the plant have shown anti-inflammatory effect by reducing levels of interleukins, nitric oxide, and prostaglandins. Rosamarinic acid, isolated from the plant, has also been found to inhibit COX-2 expression [15,16]. Besides in-vitro free radical scavenging activity, the plant has been reported to contain different phenolic compounds thus empowering the plant with antioxidant properties [17,18]. Different extracts from the plant have shown activity against HIV which has been achieved by a different mechanism like inhibition of integrase activity [19,20]. The plant has been also found to be active against HSV and lentivirus [21,22]. Due to the anti-estrogenic activity plant has been found to be active against breast and uterine cancers [23]. Ethanolic extract and a polysaccharide P32, isolated from aqueous extract of the plant, have been found to have anti-lung cancer activity [24,25]. Methanolic extract with rosamarinic acid as one the component has been found to induce T cell activation [26]. Aqueous extract from the plant has shown macrophage activation in in-vitro studies indicating the immunomodulatory property of the plant [27]. Herbal based formulation of the plant has been found effective against gingivitis in a clinical study [28]. Studies have shown memory-boosting properties of ethanolic extract of the plant which has been attributed to decreasing acetylcholine esterase activity by the extract [12].
Phytochemical analysis of the plant revealed the presence of Alkaloids, Phenols, Tannins, Saponins, Carbohydrates, Lipids, Proteins [29]. Other studies on Phytochemical analysis have indicated the presence of Terpenoids [30], Saponins [31], Sterols and steroids [32], Polysaccharides [33], Flavonoids [34], Coumarins [35].
In the current study, an effort has been made to find out the therapeutic effectiveness of P. vulgaris as a hepatoprotective agent.
2. Materials and methods
2.1. Collection and identification of plant material
The whole plant of P. vulgaris was collected from the Gulmargh area of district Baramullah, Jammu & Kashmir, India and was authenticated by the curator Akhtar H Malik, Department of Taxonomy, University of Kashmir, under voucher specimen No. 1899-KASH Herbarium, University of Kashmir, 16/07/2013. Gulmargh.
2.2. Preparation of extracts
After collection, floral spikes of the plant were cut off and dried in shade for 20 days. The dried plant material was coarsely powdered and was then extracted with water, ethanol, hydroalcohol (1:1) and methanol using a fresh portion of plant powder each time adopting cold percolation method. The method involved placing a weighed amount of powdered plant material in a percolator and adding solvent to it. This process of extraction was continued for 72 h with occasional shaking. Thereafter, the solvent was removed from the percolator and filtered. The resultant filtrate was distilled under reduced pressure, using a rotary evaporator, in order to remove the solvent completely and the dry extract was obtained. The same process was repeated with all the solvents using a fresh portion of plant powder each time and different extracts were obtained. The extracts so obtained were weighed and their percentage yield was calculated.
2.3. Determination of flavonoids
1 ml of sample (1 mg/ml) was mixed with 3 ml of methanol, 0.2 ml of 10% aluminum chloride, 0.2 ml of 1 m potassium acetate and 5.6 ml of distilled water and kept at room temperature for 30 min. The absorbance of the reaction mixture was measured at 420 nm with a UV–Visible spectrophotometer. The content of flavonoids was determined by the extrapolation of the calibration curve (or using the equation of straight line) which was made by preparing different concentrations of Gallic acid solution (1 mg %) in distilled water [36].
2.4. Determination of total phenol
5 ml of 10% Folin-ciocaltue reagent and 4 ml of 7.5% sodium bicarbonate was added to 1 ml of sample (3 replicates) of plant extract (1 mg/ml). The final volume was made 10 ml with distilled water. The resulting mixture was incubated at 45 °C with shaking for 45 min. The absorbance of the sample was measured at 765 nm using UV spectrophotometer. The concentration of the test solution was obtained using the calibration curve of the standard solution of Gallic acid (10 mg %) [37].
2.5. Experimental animals
For accessing the hepatoprotective activity Male Albino Wistar rats with weight ranging from 150 to 200 g were used. The animals were maintained at a temperature of 25 °C–28 °C, relative humidity of 40–70% and 12 h light and dark cycles. They were fed with standard rat diet and water and subjected to 12h fasting before the experimentation. Studies were performed after obtaining due approval (Approval No. F-IAEC (Pharm.Sc.)APPROVAL/23).
2.6. Acute oral toxicity
Dose selection was done according to OECD 425 guidelines (2008) following limit test using Wistar albino rats. On the basis of acute toxicity study, 1/10th of the dose was selected for further experimental evaluation [38].
2.7. Experimental design
Forty-two rats (180–200 g) were randomly divided into seven groups and each group consisted of six rats. Overall treatment was carried out for a total of 14 days. 2% w/v gum acacia in distilled water was used as a vehicle for the oral administration of extracts, standard drug silymarin, and paracetamol in the form of suspension. The following experimental protocol was followed for evaluating the hepatoprotective activity of P. vulgaris:
Group – I (Normal control): Vehicle (2% gum acacia) treated rats were kept on a normal diet (standard pellet diet and water) and served as the control for 14 days. This group was given neither paracetamol nor treatment.
Group – II (Toxic control): Rats received paracetamol 500 mg/kg body weight (b.w), p.o daily for 14 days.
Group – III (Standard control): Rats of this group received standard drug silymarin (50 mg/kg body weight (b.w)/day, orally) and paracetamol as group II, for 14 days.
Group – IV (Ethanol fraction): Rats received the ethanolic extract of P. vulgaris (200 mg/kg b.w/day, orally) and paracetamol as group II, for 14 days.
Group – V (Aqueous fraction): Rats received the aqueous extract of P. vulgaris (200 mg/kg b.w/day, orally) and paracetamol as group II, for 14 days.
Group – VI (Hydroalcoholic fraction): Rats received the hydroalcoholic extract of P. vulgaris (200 mg/kg b.w/day, orally) and paracetamol as group II, for 14 days.
Group – VII (Methanolic fraction): Rats received the methanolic extract of P. vulgaris (200 mg/kg b.w/day, orally) and paracetamol as group II, for 14 days.
2.8. Determination of biochemical parameters
At the end of the treatment, the animals were subjected to fasting for 24 h. Blood was collected by retro-orbital puncture and allowed to clot for 1 h at room temperature and serum was separated by centrifugation at 3500 rotations per minute at room temperature for 20 min. Biochemical parameters like Serum Glutamic Oxaloacetic Transaminase (SGOT) [39], Serum Glutamic Pyruvic Transaminase (SGPT) [39], Alkaline Phosphatase (ALP) [40], Total Proteins (Biuret method), Total Bilirubin and Direct Bilirubin [41] were carried out by reported methods.
2.9. Histopathological studies
For histopathological examination, the animals were sacrificed and livers were dissected out, washed with saline and were transferred and stored in 10% neutral formalin solution. The formalin-fixed samples were stained with hematoxylin-eosin. The sections were examined microscopically for changes in histopathological architecture.
2.10. Statistical analysis
Data are expressed as mean ± S.E.M and statistical analysis was carried out using one-way ANOVA followed by Dunnett's post hoc test of significance using Graph Pad Instat program. P > 0.05 was considered statistically significant.
3. Results
3.1. Percentage yield of extracts
The data on the percentage yield of extract was given in Table 1.
Table 1.
| S. No. | Type of extract | Percentage Yield |
|---|---|---|
| I. | Aqueous | 8.04 |
| II. | Ethanolic | 4.63 |
| III. | Hydroalcoholic | 6.62 |
| IV. | Methanolic | 6.20 |
Abbreviations: SGOT- Serum glutamate oxaloacetate transaminase; SGPT- Serum glutamate pyruvate transaminase; ALP-Alkaline phosphatase; TP, Total protein; DB Direct Bilirubin p.o., per-oral. Values are mean ± S.E.M of six animals. ∗∗P < 0.01; Highly significant vs Paracetamol; One way ANOVA followed by Dunnett's test.
3.2. Flavonoid content
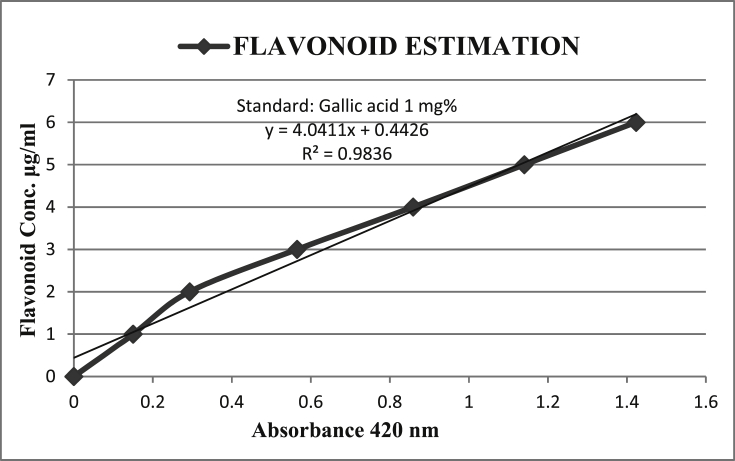
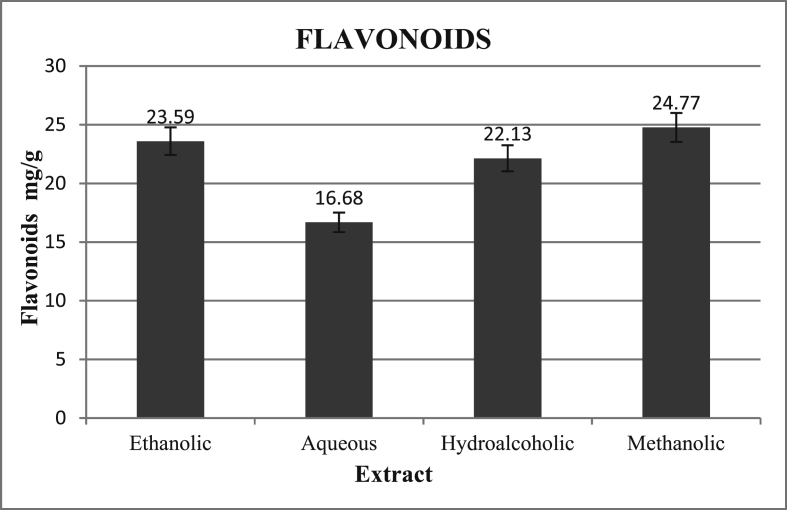
The flavonoid content has been obtained from the calibration curve Fig.1 and the results have been expressed graphically in Fig. 2.
Fig. 1.
Calibration curve for the estimation of Flavonoids Using Gallic acid as Standard.
Fig. 2.
Flavonoids (mg/g) in terms of Gallic Acid equivalents (GAE) in different extracts of floral spikes of P. vulgaris.
3.3. Total phenolic content
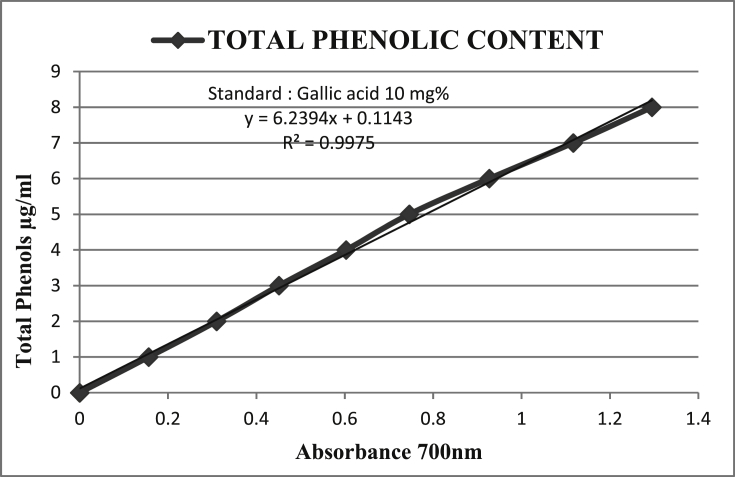
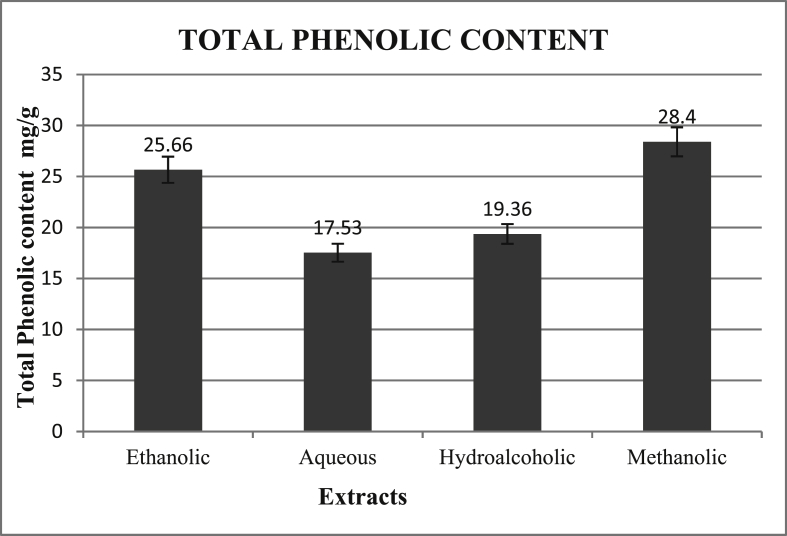
The total phenolic content has been obtained from the calibration curve Fig. 3 and the results have been expressed graphically in Fig. 4.
Fig. 3.
Calibration curve for the estimation of Total Phenols Gallic acid as standard.
Fig. 4.
Total Phenolic Content (mg/g) in terms of Gallic Acid equivalents (GAE) in different extracts of floral spikes of P. vulgaris.
3.4. Acute toxicity
All the extracts were found to be safe. No mortality was observed for 14 days of treatment with a limit dose of 2000 mg/kg body weight. All the rats tolerated the extract with no signs of toxicity. 1/10th of the dose i.e 200 mg/kg of body weight was selected for further experimental evaluation.
3.5. Hepatoprotective activity
3.5.1. Serum analysis
The detailed effect of various extracts on serum enzymatic activity in paracetamol-induced liver damage in rats is elucidated clearly by Table 2.
Table 2.
Effect of various extracts of floral spikes of P. vulgaris on serum enzymatic activity in paracetamol-induced liver damage in rats.
| S No. | Treatment | Dose | SGOT (U/L) | SGPT (U/L) | ALP (U/L) | TP (mg/dl) | TB (mg/dl) | DB (mg/dl) |
|---|---|---|---|---|---|---|---|---|
| I. | Normal(control) | 10 ml/kg | 37.51 ± 2.46 | 29.31 ± 2.02 | 57.12 ± 3.30 | 9.92 ± 0.58 | 0.44 ± 0.03 | 0.26 ± 0.02 |
| II. | Toxic (control) | 500 mg/kg | 125.12 ± 6.02∗∗ | 144.39 ± 6.56∗∗ | 243.83 ± 9.09∗∗ | 5.07 ± 0.40∗∗ | 2.58 ± 0.45∗∗ | 0.96 ± 0.03∗∗ |
| III. | Silymarin | 50 mg/kg | 57.47 ± 3.16∗∗ | 52.13 ± 2.67∗∗ | 114.16 ± 6.47∗∗ | 8.97 ± 0.26∗∗ | 0.57 ± 0.04∗∗ | 0.41 ± 0.02∗∗ |
| IV. | Ethanolic | 200 mg/kg | 76.24 ± 2.19∗∗ | 75.45 ± 1.78∗∗ | 127.00 ± 5.89∗∗ | 8.70 ± 0.09∗∗ | 0.78 ± 0.03∗∗ | 0.60 ± 0.01∗∗ |
| V. | Aqueous | 200 mg/kg | 85.30 ± 2.79∗∗ | 88.56 ± 3.12∗∗ | 154.33 ± 9.27∗∗ | 7.67 ± 0.35∗∗ | 0.94 ± 0.02∗∗ | 0.70 ± 0.03∗∗ |
| VI. | Hydroalcoholic | 200 mg/kg | 83.38 ± 1.87∗∗ | 76.35 ± 2.87∗∗ | 142.00 ± 2.80∗∗ | 8.60 ± 0.26∗∗ | 0.84 ± 0.02∗∗ | 0.62 ± 0.02∗∗ |
| VII. | Methanolic | 200 mg/kg | 69.56 ± 2.06∗∗ | 63.59 ± 3.80∗∗ | 126.00 ± 5.56∗∗ | 8.65 ± 0.24∗∗ | 0.69 ± 0.03∗∗ | 0.50 ± 0.02∗∗ |
Abbreviations: SGOT- Serum glutamate oxaloacetate transaminase; SGPT- Serum glutamate pyruvate transaminase; ALP-Alkaline phosphatase; TP, Total protein; DB Direct Bilirubin p.o., per-oral. Values are mean ± S.E.M of six animals. ∗∗P < 0.01; Highly significant vs Paracetamol; One way ANOVA followed by Dunnett's test.
3.5.2. Histopathology
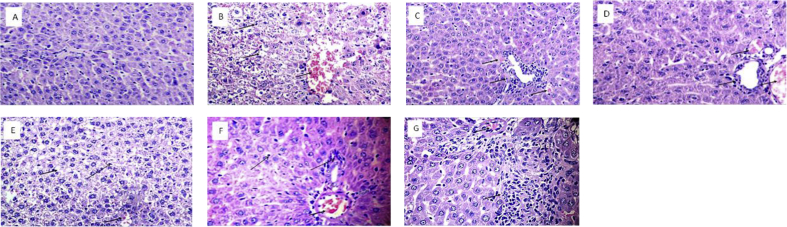
Histopathological evaluation of livers of rats in normal control, toxic control and treatment groups is depicted in Fig. 5.
Fig. 5.
Histopathology of liver of rats from different groups (H&E 100). A: Liver of normal rat showing normal hepatic architecture. B: Liver of rat administered paracetamol showing severe degeneration, nuclear pyknosis, necrotic cells, congestion and infiltration throughout the liver. C: Silymarin treated rat liver showing normal hepatocytes with mild infiltration and very mild congestion in portal areas. D: Ethanolic extract treated rat liver showing mild areas of congestion with normal hepatocytes and intact hepatic cords. E: Aqueous extract treated rat liver showing congestion and areas of degeneration. F: Hydroalcoholic extract treated rat liver showing mild infiltration and congestion in portal areas. G: Methanolic extract treated rat liver showing mild infiltration and congestion without any degeneration. Hepatocytes are normal and hepatic cords are intact.
4. Discussion
The liver is a target for toxicity because of its role in metabolizing and detoxifying substances. Paracetamol in overdoses is capable of causing liver toxicity because of NAPQI, an oxidized metabolite of paracetamol, which covalently binds with sulfhydryl groups of protein resulting in lipid peroxidation and cell necrosis [42]. Abnormally higher activities of serum enzymes SGOT, SGPT, ALP after paracetamol administration is an indication of hepatic injury, which is responsible for the leakage of cellular enzymes into the blood [43]. Bilirubin, on the other hand, is produced by the enzymatic breakdown of heme within the reticuloendothelial system. It is an important parameter and a marker to assess the functioning of the liver but seems to have little role in determining the extent of hepatocellular injury [44]. Decreased total serum proteins in rats administered with paracetamol is an indication of damage to liver tissues and impaired protein synthesis [45]. Plant-based secondary metabolites have been found effective in the treatment of different disorders including that of the liver. However, among different secondary metabolites flavonoids and phenolic compounds, because of their radical scavenging properties, have been found very potential hepatoprotective agents [46,47].
In the current study determination of flavonoids revealed that methanolic extract showed the highest amount of flavonoids followed by ethanolic, hydroalcoholic and aqueous extract (Fig. 2). The results for the estimation of Total Phenolic content revealed a similar order with methanolic extract having the highest amount of phenolic contents as evident from (Fig. 4). However a study on phytochemical analysis of whole plant extract of P. vulgaris by Rasool et al. showed a much lower quantity of flavonoids (0.56 mg/g of extract) and absence of phenolic compounds [29]. Another study by Hwang et al. ethanolic and aqueous extracts of the plant showed Total Phenolic contents equal to 303.66 and 322.80 mg GAE/g dry weight respectively [18]. The quantitative difference between the results from different studies could be attributed to the fact that in our study extracts from floral spikes were analyzed instead of whole plant extract used by Rasool et al. and Hwang et al.,
Further, the extracts administered to counter the damage induced by paracetamol were able to bring serum enzyme activities, Total Bilirubin, Direct Bilirubin, and Total Proteins levels back to normal (P < 0.01) as evident from Table 2. It has been observed that the administration of paracetamol (toxic control) resulted in the elevation of activities of SGOT, SGPT, ALP elevation in the serum levels of Direct bilirubin and Total bilirubin and reduction in the levels of Total proteins. However, the administration of extracts was able to restore the enzyme activities of SGOT, SGPT, ALP and serum levels of Direct bilirubin, Total bilirubin and Total proteins. The biochemical analysis further revealed the better hepatoprotective activity of silymarin as compared to other extracts. This could be attributed to the presence of a high quantity of flavonolignans and its ability to improve the antioxidant defense of the body by various mechanisms [48]. It is again evident from Table 2 that methanolic extract could afford better hepatoprotection as compared to other extracts. This can be attributed to the greater antioxidant potential of methanolic extract as indicated by the presence of higher amounts of flavonoids and total phenols. Histopathological evaluation of livers of rats supported the biochemical results since normal livers were seen in the normal control group (Fig. 5A), while severe degeneration, congestion throughout the liver, infiltration and nuclear pyknosis were observed in rats in which paracetamol was administered (Fig. 5B) However, the livers of extract-treated rats from groups IV, V, VI, and VII showed only mild congestion, infiltration and also mild degeneration (Fig. 5 D–G)Silymarin treated rats showed almost normal biochemical results and normal livers as indicated by histopathology (Fig. 5C) revealing its greater hepatoprotective potential as compared to all the extracts. The biochemical parameters and histopathological results revealed that ethanolic, aqueous, hydroalcoholic and methanolic extracts of P. vulgaris possessed a varied degree of hepatoprotective activity. However, the methanolic extract showed more hepatoprotective potential as evidenced by biochemical estimation and histopathology of livers compared to other groups. Accelerated recovery of hepatocytes as evidenced from histopathological examination suggested protection of hepatocellular architecture, cellular membrane and thus reducing leakage of marker enzymes into the circulation. This kind of protection could be attributed to the presence of antioxidants like phenols, flavonoids in P. vulgaris and its ability to scavenge free radicals. In a study involving hepatoprotective activity of Hibiscus esculentus commonly known as Okra, it was concluded that the possible mechanisms of hepatoprotection against CCl4 induced toxicity in rats, could be contributed to different factors such as stabilizing the cell membrane of hepatocytes, inhibition of peroxidation of lipids, prevention of cytochrome P 450-dependent oxidative processes and increase in levels of protective total proteins with anti-oxidative potential [49]. This interpretation is more or less similar to our study where it is believed that hepatic protection was possibly achieved due to the stimulatory effect on hepatic regeneration as indicated by histopathological results. In another hepatoprotective study of Terminalia paniculata on wistar rats by Eesha et al. using paracetamol-induced hepatocellular damage it was found that ethanolic extract was able to lower the activities of liver function enzymes highlighting its hepatic protective potential. The activity was attributed to antioxidants like flavonoids [50] The pattern of study is somewhat similar to our in the senses that in both cases paracetamol was used as a hepatotoxic agent and silymarin was used as standard although the plant understudy in two cases is different. Both the studies showed hepatoprotective action as indicated by lowering of liver function enzymes when compared to toxic control. However, in our study histopathological evaluation was also done to find out the nature of damage by the hepatotoxic agent and the capability of the plant in restoring hepatic architecture. A number of studies have also indicated a direct correlation between hepatoprotection and antioxidant property [51]. Thus inhibitory effects of lipid peroxidation and free radical scavenging could be other possible reasons for the hepatoprotective action of the plant because of the anti-oxidant nature of the plant [52].
5. Conclusion
The current study provided concrete evidence about the hepatoprotective potential of extracts obtained from floral spikes P. vulgaris with methanolic extract having maximum activity as compared to other extracts. This could be attributed to the fact the methanolic extract contains more free radical scavenging constituents like flavonoids and total phenols. However, more work can be done in this direction so as to isolate pure active principle(s) responsible for hepatoprotective potential which may be a step ahead in this direction for getting a new effective hepatoprotective agent(s).
Source(s) of funding
None.
Conflict of interest
None
Footnotes
Peer review under responsibility of Transdisciplinary University, Bangalore.
References
- 1.Kamboj V.P. Herbal medicine. Curr Sci. 2000;78:35–39. [Google Scholar]
- 2.Hurkadale P.J., Shelar P.A., Palled S.G., Mandavkar Y.D., Khedkar A.S. Hepatoprotective activity of Amorphophallus paeoniifolius tubers against paracetamol-induced liver damage in rats. Asian Pac J Trop Biomed. 2012;2:S238–S242. [Google Scholar]
- 3.Fakurazi S., Sharifudin S.A., Arulselvan P. Moringa oleifera hydroethanolic extracts effectively alleviate acetaminophen-induced hepatotoxicity in experimental rats through their antioxidant nature. Molecules. 2012;17:8334–8350. doi: 10.3390/molecules17078334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grace-Lynn C., Chen Y., Latha L.Y., Kanwar J.R., Jothy S.L., Vijayarathna S. Evaluation of the hepatoprotective effects of lantadene A, a pentacyclic triterpenoid of Lantana plants against acetaminophen-induced liver damage. Molecules. 2012;17:13937–13947. doi: 10.3390/molecules171213937. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 5.Singh D., Gupta R.S. Hepatoprotective activity of methanol extract of Tecomella undulata against alcohol and paracetamol induced hepatotoxicity in rats. Life Sci Med Res. 2011;26:1–8. [Google Scholar]
- 6.Gelotte C.K., Auiler J.F., Lynch J.M., Temple A.R., Slattery J.T. Disposition of acetaminophen at 4, 6, and 8 g/day for 3 days in healthy young adults. Clin Pharmacol Ther. 2007;81:840–848. doi: 10.1038/sj.clpt.6100121. [DOI] [PubMed] [Google Scholar]
- 7.Mitchell J.R., Jollow D.J., Potter W.Z., Gillette J.R., Brodie B.B. Acetaminophen-induced hepatic necrosis. IV. Protective role of glutathione. J Pharmacol Exp Ther. 1973;187:211–217. [PubMed] [Google Scholar]
- 8.Jones D.P., Lemasters J.J., Han D., Boelsterli U.A., Kaplowitz N. Mechanisms of pathogenesis in drug hepatotoxicity putting the stress on mitochondria. Mol Interv. 2010;10:98. doi: 10.1124/mi.10.2.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fraschini F., Demartini G., Esposti D. Pharmacology of silymarin. Clin Drug Investig. 2002;22:51–65. [Google Scholar]
- 10.Sidana J., Deswal G., Nain P., Arora K. Liver toxicity and hepatoprotective herbs. Int J Pharm Sci Rev Res. 2011;9:116–121. [Google Scholar]
- 11.Psotová J., Kolář M., Soušek J., Švagera Z., Vičar J., Ulrichová J. Biological activities of Prunella vulgaris extract. Phyther Res An Int J Devoted to Pharmaco Toxico Eva Nat Prod Deriv. 2003;17:1082–1087. doi: 10.1002/ptr.1324. [DOI] [PubMed] [Google Scholar]
- 12.Park S.J., Kim D.H., Lee I.K., Jung W.Y., Park D.H., Kim J.M. The ameliorating effect of the extract of the flower of Prunella vulgaris var. lilacina on drug-induced memory impairments in mice. Food Chem Toxicol. 2010;48:1671–1676. doi: 10.1016/j.fct.2010.03.042. [DOI] [PubMed] [Google Scholar]
- 13.Wu H., Gao M., Ha T., Kelley J., Young A., Breuel K. Prunella vulgaris aqueous extract attenuates IL-1β-induced apoptosis and NF-κ B activation in INS-1 cells. Exp Ther Med. 2012;3:919–924. doi: 10.3892/etm.2012.524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li H.M., Kim J.K., Jang J.M., Kwon S.O., Cui C.B., Lim S.S. The inhibitory effect of Prunella vulgaris L. on aldose reductase and protein glycation. Biomed Res. 2012;2012 doi: 10.1155/2012/928159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Huang N., Hauck C., Yum M.Y., Rizshsky L., Widrlechner M.P., McCoy J.A. Rosmarinic acid in Prunella vulgaris ethanol extract inhibits lipopolysaccharide-induced prostaglandin E2 and nitric oxide in RAW 264.7 mouse macrophages. J Agric Food Chem. 2009;57:10579–10589. doi: 10.1021/jf9023728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Park S.H., Koo H.J., Sung Y.Y., Kim H.K. The protective effect of Prunella vulgaris ethanol extract against vascular inflammation in TNF-α-stimulated human aortic smooth muscle cells. BMB Rep. 2013;46:352. doi: 10.5483/BMBRep.2013.46.7.214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang G., He L., Hu M. Optimized ultrasonic-assisted extraction of flavonoids from Prunella vulgaris L. and evaluation of antioxidant activities in vitro. Innov Food Sci Emerg Technol. 2011;12:8–25. [Google Scholar]
- 18.Hwang Y.J., Lee E.J., Kim H.R., Hwang K.A. In vitro antioxidant and anticancer effects of solvent fractions from Prunella vulgaris var. lilacina. BMC Complement Altern Med. 2013;13:310. doi: 10.1186/1472-6882-13-310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Au T.K., Lam T.L., Ng T.B., Fong W.P., Wan D.C.C. A comparison of HIV-1 integrase inhibition by aqueous and methanol extracts of Chinese medicinal herbs. Life Sci. 2001;68:1687–1694. doi: 10.1016/s0024-3205(01)00945-6. [DOI] [PubMed] [Google Scholar]
- 20.Lam T.L., Lam M.L., Au T.K., Ip D.T.M., Ng T.B., Fong W.P. A comparison of human immunodeficiency virus type-1 protease inhibition activities by the aqueous and methanol extracts of Chinese medicinal herbs. Life Sci. 2000;67:2889–2896. doi: 10.1016/s0024-3205(00)00864-x. [DOI] [PubMed] [Google Scholar]
- 21.Xu H.X., Lee S.H.S., Lee S.F., White R.L., Blay J. Isolation and characterization of an anti-HSV polysaccharide from Prunella vulgaris. Antivir Res. 1999;44:43–54. doi: 10.1016/s0166-3542(99)00053-4. [DOI] [PubMed] [Google Scholar]
- 22.Brindley M.A., Widrlechner M.P., McCoy J.A., Murphy P., Hauck C., Rizshsky L. Inhibition of lentivirus replication by aqueous extracts of Prunella vulgaris. Virol J. 2009;6:8. doi: 10.1186/1743-422X-6-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Collins N.H., Lessey E.C., DuSell C.D., McDonnell D.P., Fowler L., Palomino W.A. Characterization of antiestrogenic activity of the Chinese herb, Prunella Vulgaris, using in vitro and in vivo (Mouse Xenograft) models. Biol Reprod. 2009;80:375–383. doi: 10.1095/biolreprod.107.065375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Feng L., Jia X.B., Shi F., Chen Y. Identification of two polysaccharides from Prunella vulgaris L. and evaluation on their anti-lung adenocarcinoma activity. Molecules. 2010;15:5093–5103. doi: 10.3390/molecules15085093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Feng L., Au-Yeung W., Xu Y.H., Wang S.S., Zhu Q., Xiang P. Oleanolic acid from Prunella vulgaris L. Induces SPC-A-1 cell line apoptosis via regulation of Bax, Bad and Bcl-2 expression. Asian Pac J Cancer Prev. 2011;12:403–408. [PubMed] [Google Scholar]
- 26.Ahn S.C., Oh W.K., Kim B.Y., Kang D.O., Kim M.S., Heo G.Y. Inhibitory effects of rosmarinic acid on Lck SH2 domain binding to a synthetic phosphopeptide. Planta Med. 2003;69:642–646. doi: 10.1055/s-2003-41111. [DOI] [PubMed] [Google Scholar]
- 27.Han E.H., Choi J.H., Hwang Y.P., Park H.J., Choi C.Y., Chung Y.C. Immunostimulatory activity of aqueous extract isolated from Prunella vulgaris. Food Chem Toxicol. 2009;47:62–69. doi: 10.1016/j.fct.2008.10.010. [DOI] [PubMed] [Google Scholar]
- 28.Adámková H., Vicar J., Palasová J., Ulrichová J., Simanek V. Macleya cordata and Prunella vulgaris in oral hygiene products-their efficacy in the control of gingivitis. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2004;148:103–105. doi: 10.5507/bp.2004.019. [DOI] [PubMed] [Google Scholar]
- 29.Rasool R., Ganai B.A. Prunella vulgaris L.: a literature review on its therapeutic potentials. Pharmacologia. 2013;4:441–448. [Google Scholar]
- 30.Jeong G.S., An R.B., Pae H.O., Oh G.S., Chung H.T., Kim Y.C. Heme oxygenase-1 inducing constituent of Prunella vulgaris in HepG2 cells. Biol Pharm Bull. 2008;31:531–533. doi: 10.1248/bpb.31.531. [DOI] [PubMed] [Google Scholar]
- 31.Zhang L.Z., Guo Y.J., Tu G.Z., Shi R.B., Qin W., Ba Y.Y. A novel triterpenoid saponin from Prunella vulgaris. Yao xue xue bao Acta Pharm Sin. 2008;43:169–172. [PubMed] [Google Scholar]
- 32.Kojima H., Sato N., Hatano A., Ogura H. Sterol glucosides from Prunella vulgaris. Phytochemistry. 1990;29:2351–2355. [Google Scholar]
- 33.Tabba H.D., Chang R.S., Smith K.M. Isolation, purification, and partial characterization of prunellin, an anti-HIV component from aqueous extracts of Prunella vulgaris. Antivir Res. 1989;11:263–273. doi: 10.1016/0166-3542(89)90036-3. [DOI] [PubMed] [Google Scholar]
- 34.Xia B.H., Xiong S.H., Tang J., Zhang Z.M., Li Y.M., Li M.J. Extraction of flavonoids in Prunella vulgaris based on deep eutectic solvent method: application of new green solvent. China J Chin Mater Med. 2018;43:3484–3492. doi: 10.19540/j.cnki.cjcmm.20180702.002. [DOI] [PubMed] [Google Scholar]
- 35.Dmitruk S.I. Coumarins of Prunella vulgaris. Chem Nat Compd. 1986;22:480. [Google Scholar]
- 36.Aiyegoro O.A., Okoh A.I. Preliminary phytochemical screening and in vitro antioxidant activities of the aqueous extract of Helichrysum longifolium DC. BMC Complement Altern Med. 2010;10:21. doi: 10.1186/1472-6882-10-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Spanos G.A., Wrolstad R.E., Heatherbell D.A. Influence of processing and storage on the phenolic composition of apple juice. J Agric Food Chem. 1990;38:1572–1579. [Google Scholar]
- 38.Erhirhie E.O., Ekene N.E., Ajaghaku D.L. Guidelines on dosage calculation and stock solution preparation in experimental animals studies. J Nat Sci Res. 2014;4:100–106. [Google Scholar]
- 39.Reitman S., Frankel S. A colorimetric method for the determination of serum glutamic oxaloacetic and glutamic pyruvic transaminases. Am J Clin Pathol. 1957;28:56–63. doi: 10.1093/ajcp/28.1.56. [DOI] [PubMed] [Google Scholar]
- 40.Tietz N.W., Rinker A.D., Shaw L.M. IFCC methods for the measurement of catalytic concentration of enzymes Part 5. IFCC method for alkaline phosphatase (orthophosphoric-monoester phosphohydrolase, alkaline optimum, EC 3.1. 3.1. J Clin Chem Clin Biochem. 1983;21:731. [PubMed] [Google Scholar]
- 41.Jendrassik L., Grof P. Colorimetric method of determination of bilirubin. Biochem Z. 1938;297:81–82. [Google Scholar]
- 42.Tabassum N., Agrawal S.S. Hepatoprotective activity of Eclipta alba Hassk. against paracetamol induced hepatocellular damage in mice. JK Pract. 2004;11:278–280. [Google Scholar]
- 43.Center S.A. Interpretation of liver enzymes. Vet Clin North Am Small Anim Pract. 2007;37:297–333. doi: 10.1016/j.cvsm.2006.11.009. [DOI] [PubMed] [Google Scholar]
- 44.Sasidharan S., Aravindran S., Latha L.Y., Vijenthi R., Saravanan D., Amutha S. In vitro antioxidant activity and hepatoprotective effects of Lentinula edodes against paracetamol-induced hepatotoxicity. Molecules. 2010;15:4478–4489. doi: 10.3390/molecules15064478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Payasi A., Chaudhary M., Singh B.M., Gupta A., Sehgal R. Sub-acute toxicity studies of paracetamol infusion in albino wistar rats. Platelets. 2010;10:105. [Google Scholar]
- 46.Shehab N.G., Abu-Gharbieh E., Bayoumi F.A. Impact of phenolic composition on hepatoprotective and antioxidant effects of four desert medicinal plants. BMC Complement Altern Med. 2015;15:401. doi: 10.1186/s12906-015-0919-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ved A., Gupta A., Rawat A.K.S. Antioxidant and hepatoprotective potential of phenol-rich fraction of Juniperus communis Linn. leaves. Pharmacogn Mag. 2017;13:108. doi: 10.4103/0973-1296.197648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Surai P. Silymarin as a natural antioxidant: an overview of the current evidence and perspectives. Antioxidants. 2015;4(1):204–247. doi: 10.3390/antiox4010204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Alqasoumi S.I. Okra Hibiscus esculentus L.: a study of its hepatoprotective activity. Saudi Pharm J. 2012;20:135–141. doi: 10.1016/j.jsps.2011.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Eesha B.R., Mohanbabu A.V., Meena K.K., Vijay M., Lalit M., Rajput R. Hepatoprotective activity of Terminalia paniculata against paracetamol induced hepatocellular damage in Wistar albino rats. Asian Pac J Trop Med. 2011;4:466–469. doi: 10.1016/S1995-7645(11)60127-2. [DOI] [PubMed] [Google Scholar]
- 51.Celik I., Temur A., Isik I. Hepatoprotective role and antioxidant capacity of pomegranate (Punica granatum) flowers infusion against trichloroacetic acid-exposed in rats. Food Chem Toxicol. 2009;47:145–149. doi: 10.1016/j.fct.2008.10.020. [DOI] [PubMed] [Google Scholar]
- 52.Ben-Hsouna A., Dhibi S., Dhifi W., Mnif W., Ben-Nasr H., Hfaiedh N. Chemical composition and hepatoprotective effect of essential oil from Myrtus communis L. flowers against CCl4-induced acute hepatotoxicity in rats. RSC Adv. 2019;9:3777–3787. doi: 10.1039/c8ra08204a. [DOI] [PMC free article] [PubMed] [Google Scholar]