Abstract
Objective: Wound healing without fibrosis remains a clinical challenge and a new strategy to promote the optimal wound healing is needed. Mesenchymal stem cells (MSCs) can completely regenerate tissue injury due to the robust MSCs ability in controlling inflammation niche leading to granulation tissue formation, particularly through a release of various growth factors including transforming growth factor-β (TGF-β). In response to TGF-β stimulation, fibroblasts differentiate into myofibroblast, marked by alpha-smooth muscle actin (α-SMA) that leads to wound healing acceleration. On the other hand, sustained activation of TGF-β in wound areas may contribute to fibrosis-associated scar formation. The aim of this study was to evaluate the α-SMA expression of myofibroblast induced by MSC-released TGF-β during wound healing process. Materials and Methods: Twenty-four full-thickness excisional rat wound models were randomly divided into four groups: sham (Sh), Control (C), and MSCs treatment groups; topically treated by the MSCs at doses 2x106 cells (T1) and 1x106 cells (T2), respectively. While the control group was treated with NaCl. TGF-β level was determined using ELISA assay, α-SMA expression of myofibroblast was analyzed by immunofluorescence staining, and wound size measurement was calculated using a standard caliper. Results: This study showed a significant increase in TGF-β levels in all treatment groups on days 3 and 6. This finding was consistent with a significant increase of α-SMA expression of myofibroblast at day 6 and wound closure percentage, indicating that MSCs might promote an increase of wound closure. Conclusion: MSCs regulated the release of TGF-β to induce α-SMA expression of myofibroblast for accelerating an optimal wound healing.
Keywords: MSCs, TGF-β, α-SMA, Myofibroblast, Wound healing
Introduction
An open wound healing is a highly organized physiological process to restore the integrity of the skin involving a complex interplay between various populations of cells particularly surrounding cutaneous and immune cell[1]. Under certain conditions such as diabetes and old patients, the wound healing is susceptible to interruption or failure which can lead nonhealing wounds[2]. On the other hand, the existence of mesenchymal stromal cells (MSCs) under normal skin and their crucial role in controlling the inflammation niche at the site of injuries, indicated that the exogenous MSCs administration may provide a promising breakthrough in reaching the optimal wound healing[3,4]. Under controlled inflammation, the release of platelet-derived growth factor (PDGF), epidermal growth factor (EGF), and transforming growth factor-β (TGF-β) may promote a granulation tissue formation characterized by an active fibroblast known as myofibroblast expressing alpha-smooth muscle actin (α-SMA). The TGF-β may induce myofibroblast to produce an excessive extracellular matrix (ECM) that potentially causes a scar formation[5].
Therefore, a better understanding of TGF-β and α-SMA expression following MSCs treatment during wounds healing process is needed.
MSCs are known as multipotent stromal progenitor cells isolated from the umbilical cord (UC), cord blood, placenta, bone marrow, mobilized peripheral blood, adipose tissue, and dental pulp. However, the umbilical-derived MSCs (UC-MSCs) show a more robust gene expression profile of stemness than the other sources of derived MSCs[6,7]. Moreover, in-vitro studies showed that the growth of UC-MSCs and their capacity to differentiate into multiple tissue-forming cell lineages, such as chondrocytes, osteoblasts, adipocytes, tenocytes, and myocytes are not age-related changes[8]. MSCs express various surface markers such as CD105, CD90, and CD73, and do not express CD11b, CD14, CD19 or CD79a, CD45, CD34, or Human Leukocyte Antigen (HLA) class II[9]. In numerous studies, UC-MSCs have been shown to promote tissue regeneration by multilineage differentiation and immumodulatory capability, such as increasing IL-10[10] and modulating regulatory T cell (T-reg) to control inflammatory cells[11,12], despite under hypoxia tissue injuries[13]. The immunosuppressive properties of MSCs in controlling the excessive inflammation in injury sites can trigger the shift of inflammation phase to the proliferation leading to granulation tissue formation[14,15].
The TGF-β has been identified as the main activator of fibroblast and therefore, once the dermal fibroblasts of an open wound tissue exposed by TGF-β, those cells proliferate and migrate from a wound margin into the provisional matrix at sites of injury. The activated fibroblast can trigger contractile myofibroblast formation characterized by α-SMA expression[16]. These indicate TGF-β has a crucial role in promoting the wound healing acceleration by stimulating myofibroblast to strengthen a repaired wound in addition, producing ECM to support granulation tissue formation. However, a sustained release of TGF-β can induce persistent myofibroblast in wound areas to produce excessively ECM contributing to fibrosis formation at the end of wound healing[17]. Moreover, our previous study found that MSCs administration can accelerate wound healing in an incisional rat wound model[18], however those studies have not yet investigated the role of TGF-β and α-SMA expression to wound healing. Therefore, evaluating the threshold of TGF-β and α-SMA expression following MSCs administration during wound healing process is crucial to be explored. The aim of this study was to evaluate the expression of α-SMA in myofibroblast induced by MSCreleased TGF-β during wound healing process in a full-thickness excisional rat wound model.
Materials and Methods
MSCs isolation, culture and characterization
Rat MSCs were isolated from a 19-day pregnant female rat. Briefly, donor rats were anesthetized, and the abdomens were dissected out. Under sterile conditions, the umbilical cord (UC) was collected and washed in phosphate-buffered saline (PBS). The UC artery and vein were removed, then the UC was cut into lengths of 2?5 mm using a sterile scalpel. The sections were then distributed evenly in T25 flasks using Dulbecco’s modified Eagle’s medium (DMEM) (Sigma-Aldrich, St. Louis, MO) supplemented with 10% PBS, 100 IU/ml penicillin/streptomycin (GIBCO, Invitrogen), then incubated at 37 °C with 5% CO2. The medium was renewed every 3 days, and the cells were passaged after reaching 80% confluence. UC-MSCs at passages 4?6 were employed for the following experiments.
MSCs surface antigens were analyzed by flow cytometry method at the fourth passage according to company protocols. Briefly, the cells were incubated in the dark with allophycocyanin (APC) mouse antihuman CD73, fluorescein isothiocyanate (FITC) mouse anti-human CD90, perCP-Cy5.5.1 mouse anti-human CD105 and phycoerythrin (PE) mouse anti-human Lin negative (CD45/CD34/CD11b/CD19/HLA-DR) antibodies. The analysis was performed using BD StemflowTM (BD Biosciences, San Jose, CA, USA). MSCs were stained with a specific antibody for 30 minutes at 4 °C, examined with a BD Accuri C6 Plus flow cytometer (BD Biosciences, San Jose, CA, USA) and analyzed with BD Accuri C6 Plus software (BD Biosciences, San Jose, CA, USA).
In-vitro osteogenic differentiation assay
The MSCs were cultured at a density of 1.5 x 104 cells/well. Cells were grown in a 24 well plate with standard medium containing DMEM (Sigma-Aldrich, Louis St, MO), supplemented with 10% FBS (Gibco™ Invitrogen, NY, USA) and 1% penicillin (100 U/mL)/streptomycin (100 μg/mL) (Gibco™ Invitrogen, NY, USA) at 37 °C, 5% CO2 and = 95% humidity. After 95% confluent, standard medium was aspirated and replaced with osteogenic differentiation medium containing Human MesenCult™ Osteogenic Differentiation Basal Medium (Stem Cell Technologies, Singapore), augmented with 20% Human MesenCult™ Osteogenic Differentiation 5X Supplement (Stem Cell Technologies, Singapore) and 1% L-Glutamine (Gibco™ Invitrogen, NY, USA). The differentiation medium was renewed every 3 days. After bone matrix formation occurred, osteogenic differentiation was visualized by staining with 1 ml of 2% alizarin red solution.
Full-Thickness Skin Wound Model and MSCs administration
Twenty-four male of the Wistar rats weighing 200 g were caged at 24 ± 2 °C and 60% relative humidity, with 12:12-hour light-dark cycle. To establish the animal model of a full-thickness skin wound, the rats were anesthetized by isofluorane inhalation, and then the dorsal skin was shaved and cleaned with tincture of iodine. One fullthickness circular 6 mm biopsy punch excision was done for each rat. The animals were randomly divided into four groups (n=6) T1 and T2 as treatment groups treated by topically MSCs at doses of 1x106 and 2x106 cells respectively, while the sham group was not treated and the control group was treated with NaCl.
Wound closure measurement
The wound measurement was taken at days 3 and 6. Wound areas were determined using a standard caliper. The percentage of wound closure was calculated using: ![]() , where A0 is the original wound area after creating a wound and At is the area of a wound at the time of measurement at days 3 and 6.
, where A0 is the original wound area after creating a wound and At is the area of a wound at the time of measurement at days 3 and 6.
Immunofluorescence
The expression of α-SMA in wound tissue was evaluated using the immunofluorescence technique with α-SMA-positive markers. The wound tissues of each animal on day 6 were fixed in 10% formaldehyde, embedded with paraffin, sectioned, and deparaffinized. We incubated the tissues with the anti-α-SMA as a primary antibody (LSBio, Seattle, USA) and the Alexa fluor 488 secondary antibodies (Abcam, Oregon, USA) according to the manufacturer’s protocol and then observed the tissues under the fluorescence microscope.
ELISA
TGF-β analysis using an ELISA kit was performed according to the manufacturer’s instructions at room temperature. Briefly, according to the manufacturer’s instructions (Fine Test, Wuhan, China), the colorimetric absorbance as the TGF-β concentration (pg/ml) was analyzed at a 450 nm wavelength using a microplate reader on days 3 and 6 of the experiment.
Statistical Analysis
Data are presented as the means ± standard deviation. All calculations were carried out using IBM SPSS 22.0 (IBM Corp., Armonk, NY, USA). The statistical significance of the differences between the groups was assessed using one way-ANOVA and continued with Fisher’s least significant difference (LSD) posthoc analysis. The differences between independent variables were analyzed using two way-ANOVA and continued with Duncan posthoc analysis. The correlation between α-SMA expression and wound closure percentage was done using one-tailed Pearson's test. P values: *, p < 0.05 and **, p < 0.001.
Result
Characteristics of MSCs based on cell morphology, differentiation capacity and immunophenotypic profile.
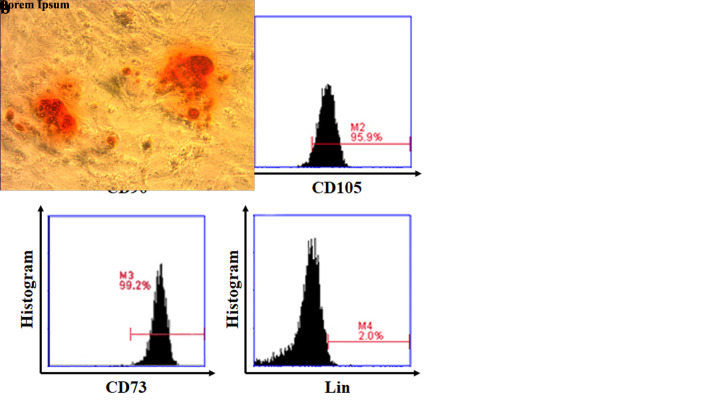
MSCs isolated and cultured from the umbilical cord were analyzed based on their plastic adherent capability under standard culture condition after 4 to 5 passages. In this study, we found the cells that showed typical monolayers of spindle-shaped fibroblast-like cells, with the capability to adhere to the plastic flask (Figure 1a). To confirm the in vitro differentiation potential of MSCs, we used osteogenic differentiation media to demonstrate that these MSCs can differentiate into osteogenic cells, characterized by the deposition of calcium. We found a red color at osteogenic differentiation assay as calcium deposition indicating the MSCs differentiate to osteogenic (Figure 1b). To characterize MSCs, we evaluated cell morphology and the expression of surface markers as indicated by the International Society for Stem Cell Therapy; the expression of CD73, CD105, CD90 and negative for Lin-(CD45/CD34/CD11b/CD19/HLA-DR)[9]. We found a high level of CD90 (99.9%), CD105 (95.9%), CD73 (99.2%) and lacked the expression of Lin (2.0%) as negative detection of hematopoietic lineage markers (Figure 1c).
Figure 1. Characterization of isolated MSCs. (a) Morphological MSCs. After the fourth passage, the cells appeared as homogeneous, spindle-shaped, fibroblast-like cells (200×magnification). (b) In vitro osteogenic differentiation assay. MSCs differentiate into osteogenic lineages indicating as red color in most MSC populations, following Alizarin Red staining (200×magnification). (c) Graphs displayed the phenotype of MSCs: CD90 (99.9%), CD105 (95.9%), CD73 (99.2%), and Lin (2.0%).
MSCs regulate the release of TGF-? to activate myofibroblast
After the initial inflammatory phase, the TGF-β rapidly upregulated and secreted by keratinocytes and macrophages for starting a granulation tissue formation[19]. The biologically active TGF-β in conjunction with the extradomain-A (ED-A)-containing fibronectin is one key regulator of myofibroblast differentiation, thus the TGF-β level is associated with wound contraction acceleration[20]. Therefore, to determine the role of MSCs in regulating TGF-β levels in full-thickness skin wound model, we assessed the concentration of TGF-β following MSCs administration using ELISA via the ophthalmic vein on day 3 and day 6.
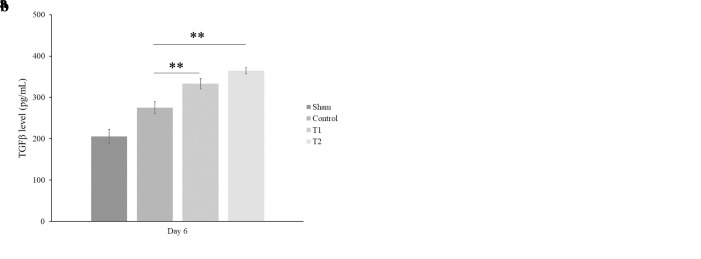
We found that there was a significant increase of TGF-β level in treatment groups (T1 and T2) at day 6 (333.2 ± 12.90 pg/mL; 364.5 ± 7.2 pg/mL, respectively, p < 0.001) compared to control groups (Figure 2(b)). In this study, we also found a significant increase of TGF-β level in all treatment groups (T1 and T2) starting in day 3 (309.9 ± 7.21 pg/mL and 315.7 ± 7.2 pg/mL, respectively, p < 0.001, Figure 2a)
Figure 2. MSCs increase the TGF-? concentration. TGF-? levels were quantified by ELISA assay at day 3 and day 6 after MSC treatment. Bars represent the mean ± SD. **, p<0.001. (a) There are a significant increase of TGF-β level at day 3, in which the higher dose of MSCs showed a higher level of TGF-β than the low dose of MSCs (315.7 ± 7.02 pg/mL. (b) A significant increase of TGF-β level at day 6 and the higher dose of MSCs showed a higher level of TGF-β than the low dose of MSCs (364.5 ± 7.2 pg/mL).
α-SMA expression correlates with wound closure
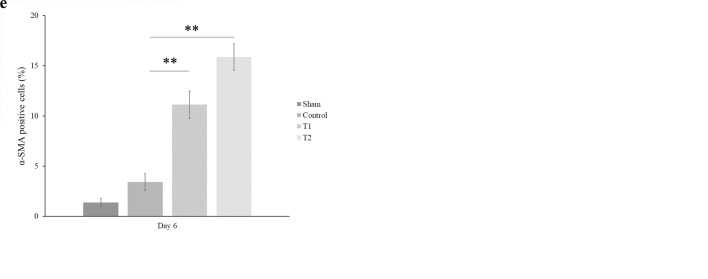
During wound healing, the activated fibroblasts cell as the one essential component cells of neo-formed connective tissue is modulated and turned into myofibroblast phenotype that characterized by the neo-expression of α-SMA[21]. Generally, about one week after wounding, the granulation tissue gradually started to be formed that marked by the myofibroblast-expressed α-SMA. To determine the role of MSCs in regulating the myofibroblast activity in wound healing, we assessed the expression of α-SMA using an immunofluorescence staining method. As shown in Figure 3, there was a significant increase of α-SMA expression at days 6 after MSCs administration in which the high-dose of MSCs resulted in a higher increase of α-SMA than the low-dose of MSCs (15,87 ± 1,32 % and 11,12 ± 1,37 %, respectively, p < 0.001).
Figure 3. MSCs enhance the percentage of α-SMA expression. A significant increase of ?-SMA expression was observed at day 6 following MSC administration in which there was a higher expression of α-SMA at the high-dose of MSCs (d) than low-dose (c) compared to control (b). Sham group (a) showed lack α-SMA expression. (e) The ?-SMA expression was analyzed using immunofluorescence. Bars represented the mean ± SD. ** p < 0.001.
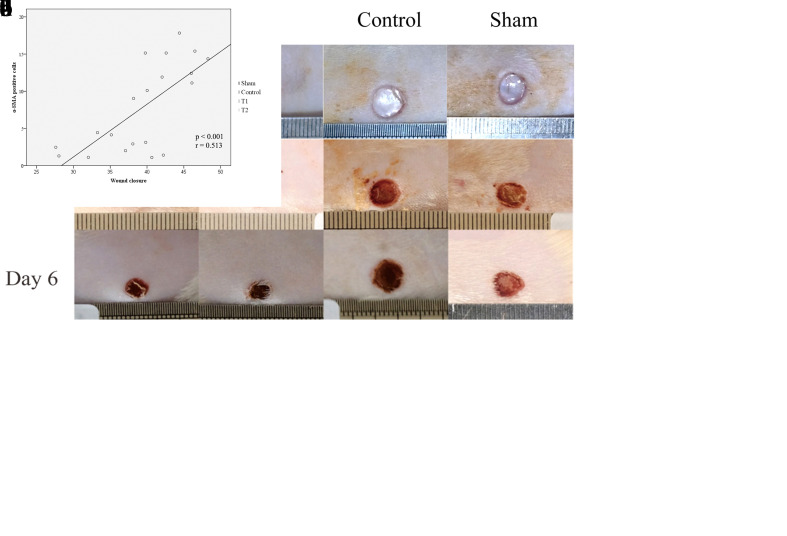
The α-SMA-positive cells appeared as spindle-shaped cell bodies with multiple cytoplasmic processes. Because α-SMA containing contractile apparatus to produce a strong contractile force, the de novo expression of α-SMA and incorporation into stress fibers in myofibroblast plays a central role in wound contraction. To find the correlation between α-SMA with wound closure acceleration, we initially measured the wound closure areas using a standard caliper and calculated the percentage of those wound closure. We found the significant increase of the wound closure percentage at day 6 after MSCs treatments compared to control groups, in which the highest wound closure percentage at high-dose of MSCs (44.31 ± 3.32%, p < 0.05, Figure 4c). With regard to wound healing, the increase of α-SMA was correlated with the wound closure percentage at day 6 (r=0.702, p<0.001, Figure 4d).
Figure 4. Differences in wound size of dorsal full-thickness skin wound of rat model following MSCs treatment. (a) The wound size were analyzed using the standard caliper. (b) A trend of the enhancement of the wound closure percentage at day 3 in all treament in which the highest wound closure percentage at highdose of MSCs (15.35 ± 5.08, p > 0.05). (c) A significant increase of the wound closure percentage at day 6 in all treament in which the highest wound closure percentage at high-dose of MSCs (44.31 ± 3.32%, p < 0.05). (d) A significant positive corelation between α-SMA expression and wound closure percentage at day 6 (r = 0.513, p < 0.001).
Discussion
Throughout a wound healing process, cellular interactions between keratinocytes with fibroblasts in the mid-and late phases of wound healing are crucial in inducing complete wound healing[22]. These processes are initiated by a release of various growth factors, including TGF-β, to induce myofibroblast for synthesizing ECM leading to accelerated granulation tissue formation[23]. A previous study reported that myofibroblast phenotype expressing α-SMA is governed by cell-matrix interactions such as fibronectin deposition and TGF-β stimulation. [14]A successful wound closure needs an effective wound contraction initiated by migrating fibroblast cells under TGF-β stimulation into a provisional matrix and then differentiating into myofibroblast for accelerating wound healing[23]. To evaluate the role of MSCs in regulating TGF-β and α-SMA expression during wound healing process, the rat fullthickness skin excisional wound was used as the animal model as in a previous study[18].
In this study, we found a significant increase of TGF-β levels in all treatment groups at day 3 (Figure 2a). Non-elevation of the TGF-β level in control group suggested that the wound groups without MSCs treatment were under inflammation condition. Several pro-inflammatory cytokines such as released by immune cells under inflammation phase might inhibit TGF-β to activate myofibroblast for producing ECM that contribute to non-optimal of wound healing[24]. These supported by a previous study that reported the endogenous inhibitors of TGF-β such as IL-1 and TNF as proinflammatory cytokines might inhibit myofibroblast differentiation during early stage of wound healing[18].
Therefore, the consequence of those findings, the myofibroblast differentiation occurred during late of wound healing process[25]. Under controlled inflammation, MSCs gradually accelerate the shift of inflammation phase to proliferation, thus promoting a release of TGF-β to modulate and differentiate fibroblasts into myofibroblast leading to wound healing acceleration[12]. Nevertheless, other study also reported that several growth factors such as PDGF might act as a strong mitogen to activate and differentiate fibroblasts into myofibroblast[26–28].
We also found a significant increase of TGF-β level in all treatment groups at day 6 (Figure 2b) that indicated a consistent release of TGF-β over the wound matrix could enhance the myofibroblast activity to generate an optimal tension within the wound, leading to wound contraction acceleration. The normal wound healing processes occur in the mid-and late phase of wound healing, in which gradually shifting the niche away from the inflammation phase to synthesis-driven granulation tissues leading to the remodelling phase[25]. The increase of TGF-β in the early phase of wound healing and constantly increase during the mid-phase at all treatment groups indicated that MSCs administration could accelerate the transition of the inflammation phase to the proliferation for initiating granulation tissues formation. myofibroblast, the main cells depositing granulation tissue in wound areas expressed the α-SMA for initiating wound closure, in addition, producing ECM[29].
This study was consistent with our other findings in which we also found a significant increase of α-SMA expression on all treatment groups at day 6 (Figure 3). The α-SMA expression following MSCs administration indicated a strong induction of contractile properties of the myofibroblast that align parallel to mechanical tension in granulation tissues. MSCs stimulating a release of TGF-β to induce the α-SMA expression of myofibroblast[30]. There are tight, controlled mutual interactions of the epithelial-mesenchymal layers to control the formation of skin architecture and appendages that are crucial in the wound closure[31]. However, the fibroblast is a major stromal cell in the connective tissue that may release several cytokines to modulate parenchymal cells and synthesize ECM to accelerate the optimal wound closure[32]. These findings were in line with the enhancement of wound percentage on all treatment groups at day 6 (Figure 4a and c), indicating that MSCs might accelerate the wound closure of full-thickness skin excisional wound. Furthermore, we assumed that the prolonged release of TGF-β that may continuously activate myofibroblast to produce ECM following MSCs treatment is not occured under controlled inflammation[26]. Therefore, the possibility of MSCs to induce scar formation in wound healing can be controlled.
The limit of this study is that we did not analyze the decrease of TGF-β level in late stage of wound healing, thus the effect of MSCs in controlling fibrosis formation remains unclear. We also did not analyze the PDGF as one of growth factor that involved in fibroblast activation. Hence, we have unclear observation regarding the role of PDGF in activating fibroblasts post-MSCs administration during wound healing process.
Conclusion
MSCs regulate the release of TGF-β level to induce α-SMA expression of the myofibroblast during wound healing. The enhanced TGF-β at day 6, in line with the expression of α-SMA indicated that there was optimally myofibroblast activity to accelerate wound closure. Thus, MSCs administration may be considered as the one of biological agents to accelerate optimal wound healing.
Acknowledgments
We would like to thank Stem Cell and Cancer Research Laboratory, Medical Faculty, Universitas Islam Sultan Agung Semarang for providing all facilities to finish this research
Glossary
Abbreviations
- APC:
Allophycocyanin
- CD:
Cluster of Differentiation
- DMEM:
Dulbecco’s Modified Eagle’s Medium
- ECM:
Extracellular Matrix
- FITC:
Fluorescein Isothiocyanate
- HLA:
Human Leukocyte Antigen
- IL-10:
Interleukine 10
- ISCT:
International Society of Cellular Therapy
- LSD:
Least Significant Difference
- MSCs:
Mesenchymal Stem Cells
- PBS:
Phosphate Buffered Saline
- PDGF:
Platelet-Derived Growth Factor
- PE:
Phycoerythrin
- TGF-β:
Transforming Growth Factor-β
- UC-MSCs:
Umbilical-Derived Mesenchymal Stem Cells
- α-SMA:
Alpha-Smooth Muscle Actin
Potential Conflicts of Interests
None
Sponsor/Funding
This work was supported by funding from Institute for Research and Community Service Unissula, Grants number 84/B.1/SA-LPPM/II/2019
References
- 1.Hu MS, Maan ZN, Wu JC, Rennert RC, Hong WX, Lai TS, Cheung AT, Walmsley GG, Chung MT, McArdle A, Longaker MT, Lorenz HP. Tissue engineering and regenerative repair in wound healing. Ann Biomed Eng. 2014;42((7)):1494–507. doi: 10.1007/s10439-014-1010-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dickinson LE, Gerecht S. Engineered Biopolymeric Scaffolds for Chronic Wound Healing. Front Physiol. 2016;7:341. doi: 10.3389/fphys.2016.00341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Usunier B, Benderitter M, Tamarat R, Chapel A. Management of fibrosis: the mesenchymal stromal cells breakthrough. Stem Cells Int. 2014;2014:340257. doi: 10.1155/2014/340257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Putra A, Rosdiana I, Darlan DM, Alif I, Hayuningtyas F, Wijaya I, Aryanti R, Makarim FR, Antari AD. Intravenous Administration is the Best Route of Mesenchymal Stem Cells Migration in Improving Liver Function Enzyme of Acute Liver Failure. Folia Med (Plovdiv). 2020;62((1)):52–58. doi: 10.3897/folmed..e47712. [DOI] [PubMed] [Google Scholar]
- 5.Mokos ZB, Jović A, Grgurević L, Dumić-Čule I, Kostović K, Čeović R, Marinović B. Current Therapeutic Approach to Hypertrophic Scars. Front Med (Lausanne). 2017;4:83. doi: 10.3389/fmed.2017.00083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Neri S. Genetic Stability of Mesenchymal Stromal Cells for Regenerative Medicine Applications: A Fundamental Biosafety Aspect. Int J Mol Sci. 2019;20((10)):2406. doi: 10.3390/ijms20102406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meng MY, Pang W, Jiang LH, Liu YH, Wei CY, Xie YH, Yu HD, Hou ZL. Stemness Gene Expression Profile Analysis in Human Umbilical Cord Mesenchymal Stem Cells. Experimental Biology and Medicine. 2012;237((6)):709–19. doi: 10.1258/ebm.2012.011429. [DOI] [PubMed] [Google Scholar]
- 8.Rohban R, Pieber TR. Mesenchymal Stem and Progenitor Cells in Regeneration: Tissue Specificity and Regenerative Potential. Stem Cells Int. 2017;2017:5173732. doi: 10.1155/2017/5173732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8((4)):315–7. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 10.Darlan DM, Munir D, Putra A, Jusuf NK. MSCs-released TGFβ1 generate CD4+CD25+Foxp3+ in T-reg cells of human SLE PBMC. J Formos Med Assoc. 2020;S0929-6646((20)):30288–6. doi: 10.1016/j.jfma.2020.06.028. [DOI] [PubMed] [Google Scholar]
- 11.Ikhsan R, Putra A, Munir D, Darlan DM, Suntoko B, Retno A. Mesenchymal stem cells induce regulatory T-cell population in human SLE. Bangladesh J Med Sci. 2020;19((4)):743–748. [Google Scholar]
- 12.Putra A, Ridwan FB, Putridewi AI, Kustiyah AR, Wirastuti K, Sadyah NAC, Rosdiana I, Munir D. The Role of TNF-α induced MSCs on Suppressive Inflammation by Increasing TGF-β and IL-10. Open Access Maced J Med Sci. 2018;6((10)):1779–1783. doi: 10.3889/oamjms.2018.404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Muhar AM, Putra A, Warli SM, Munir D. Hypoxia-Mesenchymal Stem Cells Inhibit Intra-Peritoneal Adhesions Formation by Upregulation of the IL-10 Expression. Open Access Maced J Med Sci. 2019;7((23)):3937–3943. doi: 10.3889/oamjms.2019.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gao F, Chiu SM, Motan DA, Zhang Z, Chen L, Ji HL, Tse HF, Fu QL, Lian Q. Mesenchymal stem cells and immunomodulation: current status and future prospects. Cell Death Dis. 2016;7((1)):e2062. doi: 10.1038/cddis.2015.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tracy LE, Minasian RA, Caterson EJ. Extracellular Matrix and Dermal Fibroblast Function in the Healing Wound. Adv Wound Care (New Rochelle). 2016;5((3)):119–136. doi: 10.1089/wound.2014.0561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Darby IA, Laverdet B, Bonté F, Desmoulière A. Fibroblasts and myofibroblasts in wound healing. Clin Cosmet Investig Dermatol. 2014;7:301–11. doi: 10.2147/CCID.S50046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shinde AV, Humeres C, Frangogiannis NG. The role of α-smooth muscle actin in fibroblast-mediated matrix contraction and remodeling. Biochim Biophys Acta Mol Basis Dis. 2017;1863((1)):298–309. doi: 10.1016/j.bbadis.2016.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nugraha A, Putra A. Tumor necrosis factor-α-activated mesenchymal stem cells accelerate wound healing through vascular endothelial growth factor regulation in rats. Universa Medicina. 2018;37((2)):135–142. [Google Scholar]
- 19.Liarte S, Bernabé-García Á, Nicolás FJ. Role of TGF-β in Skin Chronic Wounds: A Keratinocyte Perspective. Cells. 2020;9((2)):306. doi: 10.3390/cells9020306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Klingberg F, Chau G, Walraven M et al. The fibronectin ED-A domain enhances recruitment of latent TGF-β-binding protein-1 to the fibroblast matrix. J Cell Sci. 2018;131((5)):jcs201293. doi: 10.1242/jcs.201293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klingberg F, Hinz B, White ES. The myofibroblast matrix: implications for tissue repair and fibrosis. J Pathol. 2013;229((2)):298–309. doi: 10.1002/path.4104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wojtowicz AM, Oliveira S, Carlson MW, Zawadzka A, Rousseau CF, Baksh D. The importance of both fibroblasts and keratinocytes in a bilayered living cellular construct used in wound healing. Wound Repair Regen. 2014;22((2)):246–55. doi: 10.1111/wrr.12154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wipff PJ, Rifkin DB, Meister JJ, Hinz B. Myofibroblast contraction activates latent TGF-beta1 from the extracellular matrix. J Cell Biol. 2007;179((6)):1311–23. doi: 10.1083/jcb.200704042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Larouche J, Sheoran S, Maruyama K, Martino MM. Immune Regulation of Skin Wound Healing: Mechanisms and Novel Therapeutic Targets. Adv Wound Care (New Rochelle). 2018;7((7)):209–231. doi: 10.1089/wound.2017.0761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Landén NX, Li D, Ståhle M. Transition from inflammation to proliferation: a critical step during wound healing. Cell Mol Life Sci. 2016;73((20)):3861–85. doi: 10.1007/s00018-016-2268-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mulsow JJ, Watson RW, Fitzpatrick JM, O'Connell PR. Transforming growth factor-beta promotes pro-fibrotic behavior by serosal fibroblasts via PKC and ERK1/2 mitogen activated protein kinase cell signaling. Ann Surg. 2005;242((6)):880–7. doi: 10.1097/01.sla.0000189606.58343.cd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu X, Zheng L, Yuan Q, Zhen G, Crane JL, Zhou X, Cao X. Transforming growth factor-β in stem cells and tissue homeostasis. Bone Res. 2018;6:2. doi: 10.1038/s41413-017-0005-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rajkumar VS, Shiwen X, Bostrom M, Leoni P, Muddle J, Ivarsson M, Gerdin B, Denton CP, Bou-Gharios G, Black CM, Abraham DJ. Platelet-derived growth factor-beta receptor activation is essential for fibroblast and pericyte recruitment during cutaneous wound healing. Am J Pathol. 2006;169((6)):2254–65. doi: 10.2353/ajpath.2006.060196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Menon SN, Flegg JA, McCue SW, Schugart RC, Dawson RA, McElwain DL. Modelling the interaction of keratinocytes and fibroblasts during normal and abnormal wound healing processes. Proc Biol Sci. 2012;279((1741)):3329–38. doi: 10.1098/rspb.2012.0319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lecarpentier Y, Schussler O, Sakic A, Rincon-Garriz JM, Soulie P, Bochaton-Piallat ML, Kindler V. Human Bone Marrow Contains Mesenchymal Stromal Stem Cells That Differentiate In Vitro into Contractile Myofibroblast Controlling T Lymphocyte Proliferation. Stem Cells Int. 2018;2018:6134787. doi: 10.1155/2018/6134787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shirakihara T, Horiguchi K, Miyazawa K, Ehata S, Shibata T, Morita I, Miyazono K et al. TGF-β regulates isoform switching of FGF receptors and epithelial-mesenchymal transition. EMBO J. 2011;30((4)):783–95. doi: 10.1038/emboj.2010.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mescher AL. Macrophages and fibroblasts during inflammation and tissue repair in models of organ regeneration. Regeneration (Oxf). 2017;4((2)):39–53. doi: 10.1002/reg2.77. [DOI] [PMC free article] [PubMed] [Google Scholar]