Abstract
Purpose
The optimal timing of surgery for congenital diaphragmatic hernia (CDH) is controversial. We aimed to validate our protocol for the timing of CDH repair using the quantified patent ductus arteriosus (PDA) flow pattern.
Methods
This retrospective comparative study analyzed patients with a prenatal diagnosis of isolated CDH between 2007 and 2020. We defined the “LR ratio” as the percentage of velocity–time integral (VTI) of the left-to-right flow of PDA against overall VTI on echocardiography. Since 2010, we followed the decision criterion of performing surgery when LR ratio of > 50% has been achieved in the patients (protocol group). The protocol group (2010–2020) was compared with the historical control group (2007–2009).
Results
The average age at surgery was 104.1 ± 175.9 and 37.3 ± 30.6 h in the control and protocol groups, respectively (p = 0.11). Survival rate (88.9% vs. 95.0%, p = 0.53) and the rate of worsening of pulmonary hypertension within 24 h after surgery (22.2% vs. 10.0%, p = 0.57) were not different between the groups. The protocol group had a significantly shorter duration of tracheal intubation (26.9 ± 21.1 vs. 13.3 ± 9.5 days, p = 0.03).
Conclusion
Our decision criterion might have the advantage of facilitating early and safe surgery for patients with CDH.
Keywords: Congenital diaphragmatic hernia, Patent ductus arteriosus, Pulmonary hypertension, Echocardiography, Surgery
Introduction
Despite significant advances in neonatal intensive care, the prognosis of neonates prenatally diagnosed with a congenital diaphragmatic hernia (CDH) remains unacceptably high, with mortality rates ranging from 20 to 40% [1–3]. Pulmonary hypertension (PH) is one of the key factors influencing prognosis [4]. Neonatal PH is commonly treated with inhaled nitric oxide, medication, and sedation. In fact, good controlled sedation is crucial, because PH is worsened by various kinds of stresses, with surgery being the major cause of stress. Therefore, in the perioperative management of CDH, it is important to choose the right time for surgery and to avoid the acute deterioration of PH, also known as “PH attack.”
In recent years, the strategy of delayed surgery after stabilization has been broadly accepted [5, 6]. Meanwhile, some researchers emphasized the harmful effects of prolonged compression of the lung and left ventricle by the abdominal viscera in the intrathoracic space [7, 8]. These authors suggested that earlier surgery may help to increase the pulmonary vascular bed volume and improve the function of the left ventricle owing to the elimination of the mass effect [9, 10]. The timing of surgery is a prominent part of the management of CDH patients; however, there has been no consensus regarding the optimal timing of surgery.
In our institution, we have determined the timing of surgery based on the flow patterns of the patent ductus arteriosus (PDA) by echocardiography since the 1990s. The flow patterns of PDA were categorized into three groups: left-to-right, bidirectional, and right-to-left. Tanabe et al. reported 13 cases of CDH, and indicated that patients with the bidirectional and right-to-left pattern were required to be stabilized before CDH repair, because all patients who were found to have the right-to-left pattern at surgery died [11]. The left-to-right pattern is considered the index of stabilization. Patients with the left-to-right pattern have been the only surgical candidates. This method had the advantages of being clear-cut, easily applicable, and reliable. We are also aware that the weakness of this method is the completely subjective interpretation of the flow pattern. Therefore, since 2010, we have quantified this PDA flow pattern using the planimetric method of left-to-right flow. This objective index was used as the criterion of stabilization, in which the surgery can be performed safely.
The purpose of the present study was to validate our decision protocol for the timing of surgery for CDH.
Methods
Patients
This retrospective comparative study using a historical cohort was conducted at a single tertiary referral center, from January 2007 to March 2020. Infants with a prenatal diagnosis of isolated CDH were included in this study. The patients with chromosomal anomaly and severe cardiac malformations were excluded. The patients requiring support with extracorporeal membrane oxygenation (ECMO) were also excluded, because PDA flow cannot be precisely assessed under ECMO circulation.
Preoperative management
All patients were treated with the same strategy soon after birth. All were delivered via cesarean section, except for six infants who were delivered vaginally. The neonates were intubated in the delivery room and immediately transferred to the intensive-care unit. The first ventilator strategy used was the conventional method (Babylog VN500, Drägerwerk AG & Co., Lübeck, Germany), and high-frequency oscillation (Humming V, Metran Co., Ltd. Co., Kawaguchi, Japan) was used as the second line. Sedation was achieved with morphine and midazolam, and we tried to minimize the use of muscle-paralyzing agents. Persistent PH was evaluated by echocardiography and was managed mainly by nitric oxide inhalation. ECMO was used in patients who failed to respond to conventional therapy. Patients underwent surgical repair only after respiratory and hemodynamic stabilization had been achieved. Exogenous surfactant therapy was not routinely applied. Extubation was performed after appropriate weaning from ventilatory support.
Quantification of PDA flow
Echocardiographic examinations (Vivid S6, GE Healthcare, Chicago, USA) were performed daily after birth by a pediatric cardiologist or a neonatologist. Doppler samples of PDA flow were obtained using two-dimensional echocardiographic imaging. The velocity–time integral (VTI) of each of the left-to-right and right-to-left flows was calculated. We defined the LR ratio as the percentage of VTI of the left-to-right flow of PDA against the overall VTI consisting of left-to-right and right-to-left flows (Fig. 1). The LR ratio of > 50% was considered as “left-to-right dominant.” When the PDA had already been closed, the LR ratio was set at 100% for the purpose of convenience.
Fig. 1.
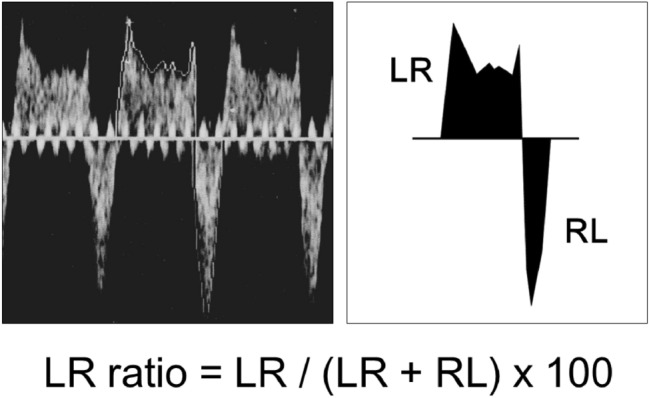
Quantification of patent ductus arteriosus (PDA) flow. The LR ratio is defined as the percentage of velocity–time integral (VTI) of the left-to-right (LR) flow of PDA against the overall VTI containing the LR and right-to-left (RL) flows on echocardiography
Risk stratification by postnatal parameters
The Japanese Congenital Diaphragmatic Hernia Study Group proposed a postnatal risk stratification system using the 1-min Apgar score and the best oxygenation index within 24 h after birth (Table 1) [12]. This classification predicts mortality well by simple prognostic factors. The 90-day survival rates in categories 1, 2, and 3 were found to be 100, 88, and 52%, respectively. All patients were classified into categories 1–3 and were evaluated the change of LR ratio according to these categories.
Table 1.
Postnatal risk stratification system for congenital diaphragmatic hernia
| Definition | |
|---|---|
| Category 0 | No need for respiratory care within 24 h after birth |
| Category 1 | Neither Ap1 of 0–4 nor best OI ≥ 8.0 |
| Category 2 | Either Ap1 of 0–4 or best OI ≥ 8.0 |
| Category 3 | Both Ap1 of 0–4 and best OI ≥ 8.0 |
| Category 4 | No OI due to death prior to intubation (including fetal death and dead birth) |
Ap1 Apgar score at 1 min; OI oxygenation index
This table has been adapted from Terui et al. [12]
Timing of surgery
During the period from 2007 to 2009, the timing of surgery was subjectively determined by the attending physician. All factors, including blood pressure, pre-ductal and post-ductal oxygen saturation, and urinary output, might be considered. Since 2010, we have followed a strict protocol for the timing of surgery, that is, to perform surgery as soon as possible after the LR ratio exceeded 50%.
Comparison between protocol and historical control groups
The patients were divided into the following two groups according to the timing of their surgery: the control group consisting of patients who underwent surgery between 2007 and 2009 and those whose timing of surgery was judged subjectively, and the protocol group, consisting of patients who underwent surgery between 2010 and 2020 using the surgical criterion of LR ratio > 50%.
The following factors were extracted from the medical records: background information including gestational age and birth weight; Kitano group, a prenatal severity grade that includes the liver and stomach positions [13]; postnatal category; the size of the diaphragmatic defect [14]; timing of surgery; echocardiographic factors including LR ratio; and outcome, including the rate of acute worsening of PH within 24 h after surgery, duration of tracheal intubation, and survival rate at 90 days of age.
Statistical analyses
The summary statistics were determined using frequencies and proportions for categorical data and means ± standard deviations, if appropriate, for continuous variables. Univariate analyses were performed using the t test for continuous variables and Fisher’s exact test for categorical variables. P < 0.05 was defined as statistically significant. All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is designed for the R statistical package. More precisely, it is a modified version of the R commander designed to add statistical functions frequently used in biostatistics [15].
Results
Patients
A total of 51 infants were born with CDH and received treatment at our institution. Patients with chromosome abnormalities (n = 1), severe cardiac anomalies (n = 5), and other severe anomalies (n = 8), and those requiring extracorporeal membrane oxygenation (n = 8) were excluded. A total of 29 patients were included in the present study and were dichotomized into the control (n = 9) and protocol (n = 20) groups.
Change in LR ratio
The daily changes in the LR ratio in each case are shown in Fig. 2. The end of the polygonal line represented the timing of surgery, but one patient (with an asterisk in Fig. 2b) underwent surgery at 24 days of age. In non-severe cases assigned to Category 1 (n = 21), the LR ratio increased immediately after birth (Fig. 2a). All of the non-severe cases had an LR ratio of > 50% until 2 days of age (Table 2). PDA was closed until the surgery in 5 cases (24% of non-severe cases). In severe cases assigned to Category 2 or 3 (n = 8), the LR ratio increased over time (Fig. 2b). It took 4 days after birth to obtain an LR ratio of > 50% in all severe cases. In three cases assigned to Categories 2–3, the LR ratio that had once reached > 50% peaked out. Two out of these three patients died from persistent PH († in Fig. 2b).
Fig. 2.
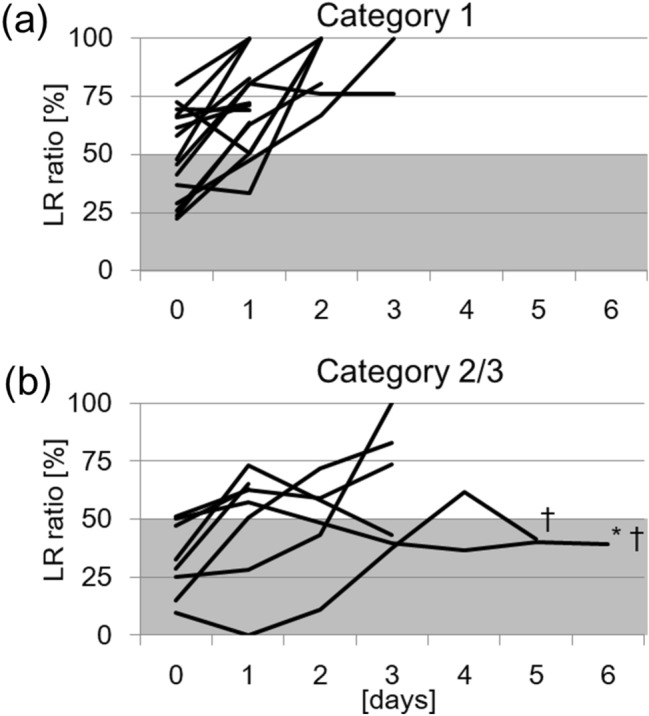
Daily changes in the LR ratio in each case. a Non-severe cases are assigned to Category 1 (n = 19). b Severe cases were assigned to Category 2 or 3 (n = 8). The end of the polygonal line representing the timing of surgery, but one patient (asterisk) underwent surgery at 24 days of age. Two patients are dead (†)
Table 2.
Comparison of achievement of the LR ratio of > 50% across the different categories
| The number of cases achieving the LR ratio of > 50% | ||||
|---|---|---|---|---|
| Category 1 (n = 21) | Category 2 − 3 (n = 8) | |||
| Day 0 | 13 | (61.9%) | 2 | (25.0%) |
| Day 1 | 6 | (28.6%) | 4 | (50.0%) |
| Day 2 | 2 | (9.5%) | 0 | (0.0%) |
| Day 3 | 0 | (0.0%) | 1 | (12.5%) |
| Day 4 | 0 | (0.0%) | 1 | (12.5%) |
LR ratio, the percentage of velocity–time integral (VTI) of the left-to-right (LR) flow of patent ductus arteriosus against the overall VTI containing the LR and right-to-left (RL) flows by echocardiography
The age of achieving the LR ratio of > 50% was significantly different between the groups (Category 1: 0.5 ± 0.7 days vs. Categories 2–3: 1.4 ± 1.4 days, p = 0.03 (Fig. 3a). The age of surgery was also significantly different between the groups (Category 1: 1.1 ± 1.0 days vs. Categories 2–3: 5.4 ± 7.6 days, p = 0.02) (Fig. 3b).
Fig. 3.
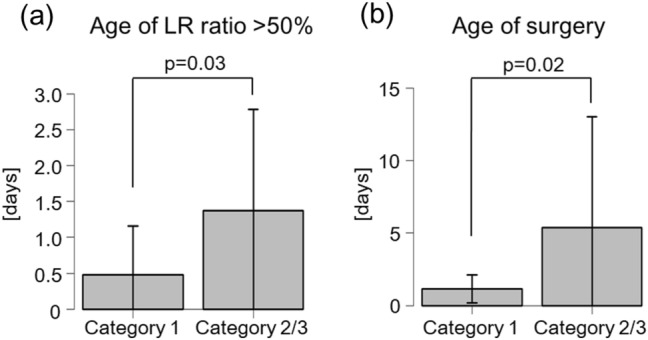
Comparison of the median age of achieving the LR ratio of > 50% (a) and the median age of surgery (b) between non-severe (Category 1) and severe cases (Category 2 or 3). The patient who underwent surgery at 24 days of age is plotted outside the area
Comparison between the control and protocol groups
Demographic data, severity grade, clinical parameters, and outcomes were compared between the control and protocol groups (Table 2). Although the gestational age showed no statistical difference between the two groups, birth weight was lower in the protocol group than in the control group. There was no difference in the severity grade between the prenatal and postnatal states in both groups. Age at surgery was 4.3 ± 7.3 and 1.6 ± 1.3 days in the control and protocol groups, respectively (p = 0.11). In the protocol group, surgery was performed in 55% of the patients at the first day of achieving an LR ratio of > 50%, and, in 90% of patients, surgery was performed at the following day after the day of achieving an LR ratio of > 50%. The LR ratio at each postnatal time was not significantly different in the protocol and control groups. The rates of acute worsening of PH within 24 h after surgery were not different between the groups (22.2% vs. 10.0%, p = 0.57). The survival rate was not significantly different between the two groups, as well (88.9% vs. 95.0%, p = 0.53). The protocol group had a significantly shorter duration of tracheal intubation (26.9 ± 21.1 vs. 13.3 ± 9.5 days, p = 0.03) (Table 3).
Table 3.
Comparisons of preoperative conditions and outcomes between the control and protocol groups
| Control group (n = 9) | Protocol group (n = 20) | P value | |
|---|---|---|---|
| Gestational age at birth (weeks) | 37.7 ± 1.1 | 37.6 ± 0.8 | 0.77 |
| Birth weight (g) | 2,679 ± 426 | 2,439 ± 283 | 0.09 |
| Gestational age at diagnosis (weeks) | 30.9 ± 4.4 | 27.6 ± 5.8 | 0.14 |
| Kitano group (I/II/III) | 8/0/0 | 15/3/2 | 0.57 |
| Category group (1/2/3) | 5/4/0 | 16/3/1 | 0.23 |
| Defect size (A/B/C/D) | 3/5/1/0 | 8/7/5/0 | 0.67 |
| Closure (direct/patch) | 8/1 | 13/7 | 0.37 |
| Age at surgery (days) | 4.3 ± 7.3 | 1.6 ± 1.3 | 0.11 |
| LR ratio (%) | |||
| Day 0 | 47.7 ± 17.2 | 51.8 ± 25.6 | 0.66 |
| Day 1 | 68.6 ± 10.0 | 60.5 ± 29.1 | 0.43 |
| Day 2 | 82.6 ± 15.2 | 70.0 ± 31.9 | 0.54 |
| Day 3 | 60.3 ± 22.2 | 77.7 ± 29.7 | 0.38 |
| Outcomes | |||
| 90-day survival (%) | 88.9 | 95.0 | 0.53 |
| Postoperative PH attack (%) | 22.2 | 10.0 | 0.57 |
| Ventilation time (days) | 26.9 ± 21.1 | 13.3 ± 9.5 | 0.03 |
| Ventilation time after surgery (days) | 25.0 ± 20.8 | 12.1 ± 9.0 | 0.03 |
Postoperative PH attack, acute worsening of pulmonary hypertension within 24 h after surgery
Discussion
The principal finding of this study is that the proposed criterion of performing surgery when the LR ratio of > 50% is achieved did not worsen the treatment outcome or increase the incidence of adverse events. Furthermore, the duration of respiratory care, especially after the surgery, was shortened significantly. We considered that the optimal timing of repairing CDH causes early improvement of pulmonary and cardiac functions. Delaying surgery is a broadly accepted policy, because neonatal PH typically improves with time [16]. Meanwhile, the affected lung of CDH patients continues to be compressed by the herniated viscera. A release from this compression may result in an increased area of gas exchange and the volume of the pulmonary vascular bed. Although the former effect is trivial, especially in a hypoplastic lung, the latter is indispensable; the increase in pulmonary vascular bed volume results in increased left ventricle diastolic performance, consequently leading to improvement in the left cardiac function and relief of PH. In fact, especially in severe CDH patients, an excessive delay of surgery carries the risk of worsening of PH due to the limited pulmonary vascular bed and prolonged diastolic left-ventricular failure [9, 10]. We also experienced a similar case in a patient who had once achieved an LR ratio of > 50% at 1 day of age, but did not maintain a > 50% LR ratio from 2 days of age (the patient is marked with an asterisk in Fig. 3b). The patient finally underwent surgery at 24 days of age, but she died at 65 days of age from respiratory failure. In retrospect, we missed the chance of performing the surgery, and should not have postponed the surgery unnecessarily. To avoid errors in judgment, it is crucial to know the details of PH status over time.
Deeney et al. based on a similar concept, reported that the use of a protocol, requiring that the echocardiogram-estimated PAP should decrease to < 80% of the systemic blood pressure before surgery, was associated with a significant reduction in acute postoperative clinical decompensation [17]. Although the main result was similar to that of our study, the age of surgery was completely different between the two studies. In Deeney et al.’s protocol group and the control group, the mean age at surgery was 6.8 days and 5.2 days, respectively. However, in our study, the infants in the protocol group were younger at the time of surgery compared to those in the control group (1.6 days in the protocol group and 4.3 days in the control group). Furthermore, ventilation time was not significantly different between the two groups in Deeney et al.’s study, which might be because Deeney et al.’s criteria for the surgery were stricter than ours. Although either strategy may work in almost all CDH patients, we emphasize the risk of missing the optimal timing of surgery. Therefore, the LR ratio is more suitable to pursue our original concept, in which the surgery should be performed as soon as possible without excessive delays.
The most precise method for estimating PH is by performing a catheterization study. Practically, ultrasonic echocardiography is broadly applied because of its convenience. Ultrasonic echocardiography permits repeated bedside testing. Most commonly, systolic pulmonary artery pressure (PAP) can be estimated in the following manner: (1) tricuspid regurgitation pressure gradient (TRPG), measured from the tricuspid regurgitant velocity (TRV) with the use of the modified Bernoulli’s equation; (2) right-ventricular systolic pressure (RVSP) calculated by adding TRPG to the estimated right atrial pressure (RAP); (3) RVSP can be taken as systolic PAP, provided that the patient has no stenoses in the right-ventricular outflow or pulmonary valve; and (4) the configuration of the right ventricle and ventricular septum, thickness of the right-ventricular wall, and the diameter of the inferior vena cava were also evaluated simultaneously [18]. This method is highly standardized and broadly used for the screening of PH [17, 19], but it has some limitations: (1) all estimations are based on the absolute value of flow velocity; therefore, measurement error can occur due to the trivial changes in the angle or the area of measurement; (2) situations such as the presence of patent foramen ovale and septal defect increase the complexity; and (3) for patients with right cardiac failure, PH cannot be estimated because of decreased cardiac output and pulmonary artery flow. On the other hand, the LR ratio has the following advantages: (1) measurement is easily performed with ultrasonic echocardiography without any particular calculation; (2) measurement error can be minimized, because relative values, not absolute values, are used; (3) right-to-left shunt, which is the worst situation, can be clearly manifested; and (4) LR ratio represents a balance between the left-to-right and right-to-left shunts, reflecting not only PH but also systemic blood pressure. These advantages enabled us to easily share the recent status in real time with each member of the medical care team. Furthermore, this may be a useful parameter when the preoperative status is discussed among several medical teams. Although not observed in our cohort, there may exist rare cases in which the LR ratio could never exceed 50% within a week. Stabilization will probably not improve PH in such cases, and the indication of surgery on an ECMO standby must be discussed. During the target period, there were three cases of inducted ECMO therapy. Their first LR ratios were 17.5%, 23.5%, and 33.4%. Because all of them were inducted ECMO on the first day, the change in the LR ratio was unclear.
The CDH EURO Consortium Consensus proposed the following factors for the indication of the surgery: mean arterial blood pressure should be normal for gestational age, pre ductal saturation levels of 85–95% on FiO2 < 50%, lactate levels < 3 mmol/l, and urine output > 1 ml/kg/h [5]. In the protocol group of our cohort, the patients’ preoperative status met these criteria, except FiO2 (data not shown). We may need to manage the patients with higher FiO2 to obtain a better LR ratio. Our protocol should include a higher limit for the level of FiO2 when assessing the LR ratio.
The current study has several limitations. First, our work had a retrospective design with a relatively small number of patients included. Second, we also performed a comparative analysis using a historical cohort, although we did not change our treatment for CDH during the study period. Further prospective studies to validate our protocol are needed.
In conclusion, our decision criterion of using the LR ratio > 50% for the surgery of CDH facilitates early and safe surgery. This criterion may be useful to avoid missing the optimal timing of surgery, especially in severe cases.
Author contributions
K.T. designed the study and Y.S. wrote the initial draft of the manuscript. Y.O. and M.E. performed echocardiography and contributed to the data collection. All of the other authors have contributed to the analysis and interpretation of data as well as the critical review of the manuscript. The final version of the manuscript was approved by all authors.
Funding
None.
Code availability
The datasets during the current study are available from the corresponding author on reasonable request.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The study design was approved by the Research Ethics Committee of the Graduate School of Medicine, Chiba University (no. 509) and the independent ethics committees of the other four participating institutions. The study was performed in accordance with the principles of the Declaration of Helsinki and the Ethical Guidelines for Medical and Health Research Involving Human Subjects.
Informed consent
Signed informed consent was not required as this was a retrospective study with de-identified data.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Javid PJ, Jaksic T, Skarsgard ED, Lee S, Network CN. Survival rate in congenital diaphragmatic hernia: the experience of the Canadian neonatal network. J Pediatr Surg. 2004;39:657–660. doi: 10.1016/j.jpedsurg.2004.01.022. [DOI] [PubMed] [Google Scholar]
- 2.Ontario Congenital Anomalies Study Group Apparent truth about congenital diaphragmatic hernia: a population-based database is needed to establish benchmarking for clinical outcomes for CDH. J Pediatr Surg. 2004;39:661–665. doi: 10.1016/j.jpedsurg.2004.01.032. [DOI] [PubMed] [Google Scholar]
- 3.Nagata K, Usui N, Kanamori Y, Takahashi S, Hayakawa M, Okuyama H, Inamura N, Fujino Y, Taguchi T. The current profile and outcome of congenital diaphragmatic hernia: a nationwide survey in Japan. J Pediatr Surg. 2013;48:738–744. doi: 10.1016/j.jpedsurg.2012.12.017. [DOI] [PubMed] [Google Scholar]
- 4.Vijfhuize S, Schaible T, Kraemer U, Cohen-Overbeek TE, Tibboel D, Reiss I. Management of pulmonary hypertension in neonates with congenital diaphragmatic hernia. Eur J Pediatr Surg. 2012;22:374–383. doi: 10.1055/s-0032-1329531. [DOI] [PubMed] [Google Scholar]
- 5.Reiss I, Schaible T, van Den Hout L, Capolupo I, Allegaert K, van Heijst A, Gorett Silva M, Greenough A, Tibboel D, CDH EURO Consortium Standardized postnatal management of infants with congenital diaphragmatic hernia in Europe: the CDH EURO consortium consensus. Neonatology. 2010;98:354–364. doi: 10.1159/000320622. [DOI] [PubMed] [Google Scholar]
- 6.Snoek KG, Reiss IKM, Greenough A, Capolupo I, Urlesberger B, Wessel L, Storme L, Deprest J, Schaible T, van Heijst A, Tibboel D, CDH EURO Consortium Standardized postnatal management of infants with congenital diaphragmatic hernia in Europe: the CDH EURO consortium consensus - 2015 Update. Neonatology. 2016;110:66–74. doi: 10.1159/000444210. [DOI] [PubMed] [Google Scholar]
- 7.Kinsella JP, Steinhorn RH, Mullen MP, Hopper RK, Keller RL, Ivy DD, Austin ED, Krishnan US, Rosenzweig EB, Fineman JR, Everett AD, Hanna BD, Humpl T, Raj JU, Abman SH, Pediatric Pulmonary Hypertension Network (PPHNet) The left ventricle in congenital diaphragmatic hernia: Implications for the management of pulmonary hypertension. J Pediatr. 2018;197:17–22. doi: 10.1016/j.jpeds.2018.02.040. [DOI] [PubMed] [Google Scholar]
- 8.Altit G, Bhombal S, Van Meurs K, Tacy TA. Diminished cardiac performance and left ventricular dimensions in neonates with congenital diaphragmatic hernia. Pediatr Cardiol. 2018;39:993–1000. doi: 10.1007/s00246-018-1850-7. [DOI] [PubMed] [Google Scholar]
- 9.Okuyama H, Kubota A, Oue T, Kuroda S, Ikegami R, Kamiyama M, Kitayama Y, Yagi M. Inhaled nitric oxide with early surgery improves the outcome of antenatally diagnosed congenital diaphragmatic hernia. J Pediatr Surg. 2002;37:1188–1190. doi: 10.1053/jpsu.2002.34469. [DOI] [PubMed] [Google Scholar]
- 10.Inamura N, Kubota A, Nakajima T, Kayatani F, Okuyama H, Oue T, Kawahara H. A proposal of new therapeutic strategy for antenatally diagnosed congenital diaphragmatic hernia. J Pediatr Surg. 2005;40:1315–1319. doi: 10.1016/j.jpedsurg.2005.05.018. [DOI] [PubMed] [Google Scholar]
- 11.Tanabe M, Yoshida H, Iwai J, Takahashi H, Ohnuma N, Terai M. Doppler flow patterns through the ductus arteriosus in patients with congenital diaphragmatic hernia. Eur J Pediatr Surg. 2000;10:92–95. doi: 10.1055/s-2008-1072333. [DOI] [PubMed] [Google Scholar]
- 12.Terui K, Nagata K, Kanamori Y, Takahashi S, Hayakawa M, Okuyama H, Inamura N, Yoshida H, Taguchi T, Usui N. Risk stratification for congenital diaphragmatic hernia by factors within 24 h after birth. J Perinatol. 2017;37:805–808. doi: 10.1038/jp.2017.11. [DOI] [PubMed] [Google Scholar]
- 13.Kitano Y, Okuyama H, Saito M, Usui N, Morikawa N, Masumoto K, Takayasu H, Nakamura T, Ishikawa H, Kawataki M, Hayashi S, Inamura N, Nose K, Sago H. Re-evaluation of stomach position as a simple prognostic factor in fetal left congenital diaphragmatic hernia: a multicenter survey in Japan. Ultrasound Obstet Gynecol. 2011;37:277–282. doi: 10.1002/uog.8892. [DOI] [PubMed] [Google Scholar]
- 14.Lally KP, Lasky RE, Lally PA, Bagolan P, Davis CF, Frenckner BP, Hirschl RM, Langham MR, Buchmiller TL, Usui N, Tibboel D, Wilson JM, Congenital Diaphragmatic Hernia Study Group Standardized reporting for congenital diaphragmatic hernia - An international consensus. J Pediatr Surg. 2013;48:2408–2415. doi: 10.1016/j.jpedsurg.2013.08.014. [DOI] [PubMed] [Google Scholar]
- 15.Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48:452–458. doi: 10.1038/bmt.2012.244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Boloker J, Bateman DA, Wung JT, Stolar CJ. Congenital diaphragmatic hernia in 120 infants treated consecutively with permissive hypercapnea/spontaneous respiration/elective repair. J Pediatr Surg. 2002;37:357–366. doi: 10.1053/jpsu.2002.30834. [DOI] [PubMed] [Google Scholar]
- 17.Deeney S, Howley LW, Hodges M, Liechty KW, Marwan AI, Gien J, Kinsella JP, Crombleholme TM. Impact of objective echocardiographic criteria for timing of congenital diaphragmatic hernia repair. J Pediatr. 2018;192:99–104.e4. doi: 10.1016/j.jpeds.2017.09.004. [DOI] [PubMed] [Google Scholar]
- 18.de Boode WP, Singh Y, Molnar Z, Schubert U, Savoia M, Sehgal A, Levy PT, McNamara PJ, El-Khuffash A, European Special Interest Group ‘Neonatologist Performed Echocardiography’ (NPE) Application of neonatologist performed echocardiography in the assessment and management of persistent pulmonary hypertension of the newborn. Pediatr Res. 2018;84:68–77. doi: 10.1038/s41390-018-0082-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, Simonneau G, Peacock A, Vonk Noordegraaf A, Beghetti M, Ghofrani A, Gomez Sanchez MA, Hansmann G, Klepetko W, Lancellotti P, Matucci M, McDonagh T, Pierard LA, Trindade PT, Zompatori M, Hoeper M, ESC Scientific Document Group 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: the joint task force for the diagnosis and treatment of pulmonary hypertension of the european society of cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: association for European paediatric and congenital cardiology (AEPC), international society for heart and lung transplantation (ISHLT) Eur Heart J. 2016;37:67–119. doi: 10.1093/eurheartj/ehv317. [DOI] [PubMed] [Google Scholar]
- 20.Collaborative CCDH, Puligandla PS, Skarsgard ED, Offringa M, Offringa M, Adatia I, Baird R, Bailey M, Brindle M, Chiu P, Cogswell A, Dakshinamurti S, Flageole H, Keijzer R, McMillan D, Oluyomi-Obi T, Pennaforte T, Perreault T, Piedboeuf B, Riley SP, Ryan G, Synnes A, Traynor M. Diagnosis and management of congenital diaphragmatic hernia: a clinical practice guideline. CMAJ. 2018;190:E103–E112. doi: 10.1503/cmaj.170206. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets during the current study are available from the corresponding author on reasonable request.


