Abstract
Background
Although physical activity has been consistently associated with reduced breast cancer mortality, evidence is largely based on data collected at one occasion. We examined how pre- and postdiagnosis physical activity was associated with survival outcomes in high-risk breast cancer patients.
Methods
Included were 1340 patients enrolled in the Diet, Exercise, Lifestyle and Cancer Prognosis (DELCaP) Study, a prospective study of lifestyle and prognosis ancillary to a SWOG clinical trial (S0221). Activity before diagnosis, during treatment, and at 1- and 2-year intervals after enrollment was collected. Patients were categorized according to the Physical Activity Guidelines for Americans as meeting the minimum guidelines (yes/no) and incrementally as inactive, low active, moderately active (meeting the guidelines), or high active.
Results
In joint-exposure analyses, patients meeting the guidelines before and 1 year after diagnosis experienced statistically significant reductions in hazards of recurrence (hazard ratio [HR] = 0.59, 95% confidence interval [CI] = 0.42 to 0.82) and mortality (HR = 0.51, 95% CI = 0.34–0.77); associations were stronger at 2-year follow-up for recurrence (HR = 0.45, 95% CI = 0.31 to 0.65) and mortality (HR = 0.32, 95% CI = 0.19 to 0.52). In time-dependent analyses, factoring in activity from all time points, we observed striking associations with mortality for low- (HR = 0.41, 95% CI = 0.24 to 0.68), moderate- (HR = 0.42, 95% CI = 0.23 to 0.76), and high-active patients (HR = 0.31, 95% CI = 0.18 to 0.53).
Conclusions
Meeting the minimum guidelines for physical activity both before diagnosis and after treatment appears to be associated with statistically significantly reduced hazards of recurrence and mortality among breast cancer patients. When considering activity from all time points, including during treatment, lower volumes of regular activity were associated with similar overall survival advantages as meeting and exceeding the guidelines.
Over the past decade, a large body of epidemiological evidence has demonstrated an inverse association between prediagnosis (1–14) and postdiagnosis (4,8,9,15–25) recreational physical activity (RPA) with mortality among breast cancer patients. Collectively, data show that patients reporting the highest levels of prediagnosis RPA experienced a 26%–27% reduction in mortality in comparison to their least active counterparts (26,27), whereas the highest levels of postdiagnosis RPA associated with even stronger protection, with reduced hazards of mortality ranging from 39% to 48% in comparison to the least active women (26,27). However, data describing the associations of RPA with breast cancer recurrence remains limited, conflicting, and not well understood (11,21,25,27,28).
An important limitation of the extant literature is that it is based almost entirely on data collected on one occasion, reflecting either pre- or postdiagnosis activity. Few publications have reported how activity measured on multiple occasions is associated with breast cancer survival (8,17), and none have described how pre- and postdiagnosis RPA, queried prospectively on 4 occasions before, during, and after chemotherapy, is associated with disease recurrence and mortality. Yet this remains a clinically significant area of inquiry given that many breast cancer patients decrease activity or become entirely inactive during or after cancer treatment (29–32). As such, we investigated the associations of pre- and postdiagnosis RPA with disease recurrence and mortality for patients with high-risk breast cancer. Specifically, we sought to examine whether meeting the Physical Activity Guidelines for Americans (PAGAs) before diagnosis, during treatment, and after treatment was associated with disease recurrence and/or mortality.
Methods
Study Population and Data Collection
The Diet, Exercise, Lifestyle and Cancer Prognosis Study (DELCaP) was a questionnaire-based study ancillary to a breast cancer intergroup phase III clinical trial (SWOG 0221; NCT00070564) led by SWOG (33). DELCaP was initiated to assess lifestyles of women with high-risk, pathologic stage I to III breast cancer at multiple times throughout survivorship, including at study enrollment (before treatment), during treatment, and after chemotherapy completion (34,35).
Patients were excluded from enrollment in the S0221 therapeutic trial if they received prior chemotherapy or radiation treatment, had any heart disease or abnormal organ function, were HIV positive, were pregnant, or had a Zubrod performance status greater than 1. Patients with a history of hypertension and/or patients ages 60 years and older must have undergone diagnostic testing to demonstrate at least a normal left ventricle ejection fraction. Patients experiencing unacceptable treatment toxicities or treatment delays (>3 weeks) were removed from the trial.
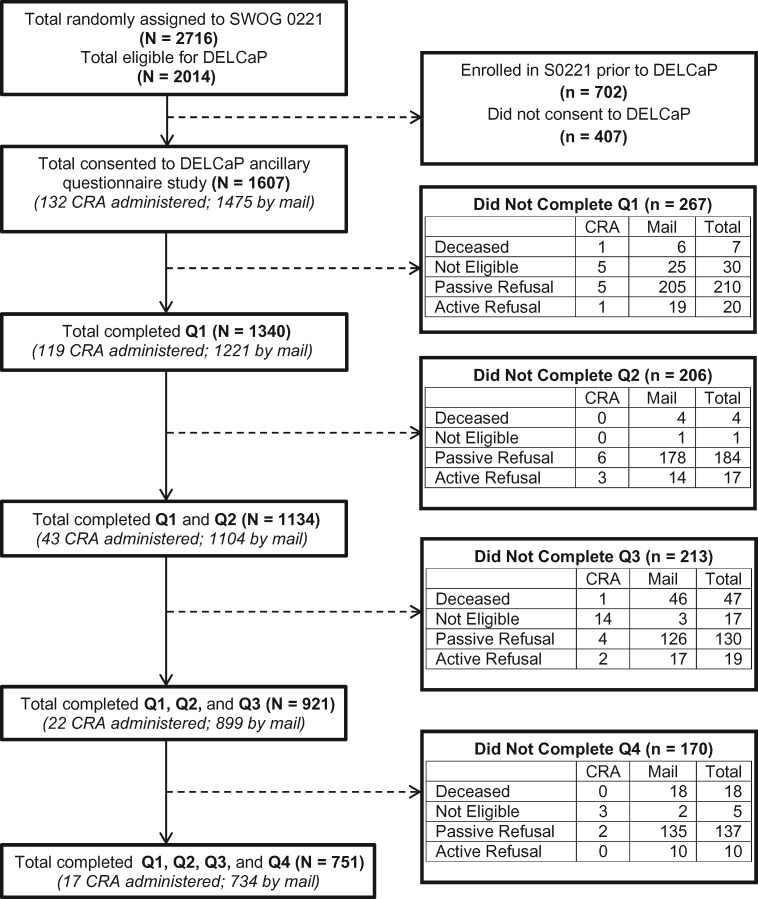
DELCaP was initiated after S0221 began, and formal approval to conduct the study was obtained in June 2005 from the institutional review boards at Roswell Park and all participating institutions that enrolled patients to S0221. As shown in Figure 1, a total of 2014 patients were eligible to participate in DELCaP, and 1607 (79.8%) consented to participate. All participants provided written informed consent.
Figure 1.
Study schema for participants included in the physical activity and survival analysis from the Diet, Exercise, Lifestyle and Cancer Prognosis (DELCaP) Study, an observational questionnaire study ancillary to S0221, a randomized treatment trial for high-risk breast cancer. Questionnaire 1 (Q1) was completed at the time of S0221 registration, prior to the initiation of chemotherapy. Q2 was completed at the completion of active treatment (approximately 6 months after trial registration). Q3 was completed 1 year after trial registration, approximately 6 months after treatment completion. Q4 was completed 2 years after trial registration, approximately 1 year after treatment completion. CRA = Clinical Research Associate.
DELCaP Questionnaire
The DELCaP questionnaire, a self-administered epidemiological survey assessing demographic and lifestyle factors, was adapted from an extensive instrument used with patients participating in the DataBank and BioRepository at Roswell Park Comprehensive Cancer Center (36). The baseline questionnaire (Q1) was administered at the time of study enrollment and queried lifestyle behaviors in the month prior to diagnosis. Among the 1607 consented patients, 1340 (83.4%) completed and returned Q1. The second questionnaire (Q2) assessed lifestyles during chemotherapy and was administered to patients returning Q1 6 months after study enrollment when treatment was scheduled to be completed; among the 1340 patients receiving Q2, 1134 (84.6%) completed the questionnaire. The third questionnaire (Q3) was administered to patients completing Q1 and Q2 approximately 1 year after study enrollment and assessed lifestyles in the preceding year; 921 patients (81.2%) completed Q3. Lastly, the fourth questionnaire (Q4) was administered 2 years after study enrollment to patients completing Q1–Q3 and queried lifestyles in the preceding year; 81.5% of eligible participants completed Q4.
Recreational Physical Activity Assessment
The DELCaP questionnaire assessed mode, frequency, and duration of RPA and was adapted from the Lifetime Physical Activity Questionnaire, a self-administered survey with established reliability among adult women (37). Total metabolic equivalent of task (MET) minutes/hours were calculated for each person at each time based on corresponding codes and MET values published in the Physical Activity Compendium (38). Activities were included in the analysis if performed at least once a week throughout the exposure window assessed and if the compendium MET value was at least 3.0 or higher (38–40).
We parameterized RPA as a categorical variable using three approaches. First, in light of emerging evidence showing that lower volumes of regular, weekly activity have been associated with decreased all-cause and cancer mortality in comparison to inactivity (41,42), we examined whether engaging in any regular, weekly RPA (i.e., at least once per week, yes/no) was associated with outcomes. We further examined relationships according to the minimum PAGAs (the MET hour equivalent of 150 minutes of moderate-intensity RPA per week, yes/no) (43). Lastly, in accordance with the incremental physical activity levels outlined in the PAGAs, we classified patients as inactive (no regular and/or weekly RPA); low active (some activity but insufficiently active according to minimum recommendations); moderate active (the equivalent of meeting the recommended range of RPA in the PAGAs); or high active (exceeding the minimum recommended range of RPA in the PAGAs) (43).
Clinical Outcome Ascertainment
The primary analytic outcomes were disease recurrence and all-cause mortality. Recurrence data were assessed via posttreatment follow-up visits in which patients underwent physical examination every 6 months for the first 5 years and annually for up to 15 years until death, whichever occurred first. Additional studies (eg, imaging, biopsy) to investigate and document suspected disease recurrence were performed as clinically indicated, and results were noted on the appropriate study forms. For disease recurrence, disease-free survival time was defined as time from randomization to first instance of disease recurrence, new breast primary tumor, or death from any cause, whichever came first. Vital status was ascertained from medical records, phone calls and/or letters to patient homes, obituaries, and national death records. For all-cause mortality, survival time was defined as time from randomization to death from any cause. Patients who had not recurred and who were still alive at the time of analysis were censored on the date of their last clinical contact.
Statistical Analysis
Standard Cox models were used to estimate associations of prediagnosis RPA with disease recurrence and mortality and to assess associations representing the joint exposure of pre- and postdiagnosis RPA with disease recurrence and mortality. Time-dependent models were used to consider RPA as a time-varying exposure across all exposure windows and to appropriately account for the possibility of immortal time bias (44,45). Sensitivity landmark analyses were conducted to further account for the possibility of immortal time bias and to examine associations at each independent exposure window. However, we used time-dependent analyses in the primary analyses because with each successive questionnaire, the landmark time became shorter and data points were lost, providing an incomplete picture of the exposure-outcome association (45).
We a priori defined age at baseline and a stratification factor corresponding to the original randomization treatment assignment from the SWOG clinical trial as important covariates in all multivariable analyses. We examined additional relevant prognostic variables for confounding using stepwise regression models and the 10% change-in-estimate method (46). Based on these approaches, we determined that body mass index, menopausal status, race, ethnicity, and education were not statistically significant factors. The number of positive nodes, HER2 status, and estrogen receptor and/orprogesterone receptor status were statistically significant factors in stepwise regression but did not change the minimally adjusted hazard ratios (HRs); thus, we present minimally adjusted models as our primary results.
We used standard diagnostic methods to examine our model-building process to detect any substantial departures from model assumptions that may have influenced our estimates (ie, examining residuals, ad hoc time-varying covariates of a discretized time scale, and Kaplan–Meier curves for the main effects under consideration). No interaction terms were statistically significant relative to our final conclusions.
Missing data from nonresponse (at least one missing survey) were assumed to be missing not at random. To account for the missing data mechanism, Taylor series variance estimation was employed and observations that had missing values were included in computing the degrees of freedom (47).
All statistical tests were two-sided, and a P value of less than .05 was considered statistically significant. All analyses were performed using SAS 9.4 (SAS Institute, Cary, NC).
Results
The demographic and clinical characteristics of the DELCaP study population according to overall and disease-free survival are summarized in Table 1. At the time of analysis, with a mean follow-up time of 89 months, 222 patients had died and 310 experienced recurrence. Table 2 characterizes the prevalence of RPA according to survival outcomes at each exposure window. We observed expected decreases in activity during chemotherapy, with 54.5% of patients reporting RPA, in contrast to 73.2% before diagnosis; the highest prevalence of activity was at 2-year follow-up (75.1%).
Table 1.
Baseline demographic and clinical characteristics of the Diet, Exercise, Lifestyle and Cancer Prognosis Study study population according to overall and disease-free survival status (n = 1340)
| Patient characteristic | Overall survival, No. (%) |
Disease-free survival, No. (%) |
||||
|---|---|---|---|---|---|---|
| Alive(n = 1118, 83%) | Deceased(n = 222, 17%) | P* | Disease-free(n = 1030, 77%) | Recurrence(n = 310, 23%) | P* | |
| Demographic characteristics | ||||||
| Age, mean (SD), y | 50.89 (9.72) | 53.39 (10.47) | <.001 | 50.87 (9.64) | 52.75 (10.56) | .005 |
| Body mass index, mean (SD) | 29.15 (6.77) | 30.49 (7.07) | .008 | 29.14 (6.75) | 30.16 (7.05) | .02 |
| Menopausal status | .006 | .006 | ||||
| Premenopausal | 545 (86.51) | 85 (13.49) | 506 (80.32) | 124 (19.68) | ||
| Postmenopausal | 563 (80.89) | 133 (19.11) | 515 (73.99) | 181 (26.01) | ||
| Self-reported race | .10 | .37 | ||||
| White | 937 (83.81) | 181 (16.19) | 864 (77.28) | 254 (22.72) | ||
| Black | 69 (73.40) | 25 (26.60) | 64 (68.09) | 30 (31.91) | ||
| Multiracial | 38 (84.44) | 7 (15.56) | 35 (77.78) | 10 (22.22) | ||
| American Indian | 11 (84.62) | 2 (15.38) | 10 (76.92) | 3 (23.08) | ||
| Asian/Pacific Islander | 44 (91.17) | 4 (.08) | 40 (83.33) | 8 (16.67) | ||
| Other | 15 (83.33) | 3 (16.67) | 14 (77.78) | 4 (22.22) | ||
| Self-reported ethnicity | .73 | .51 | ||||
| Non-Hispanic | 1059 (83.32) | 212 (16.68) | 978 (76.95) | 293 (23.05) | ||
| Hispanic | 51 (85.00) | 9 (15.00) | 44 (73.33) | 16 (26.67) | ||
| Education | .03 | .06 | ||||
| Grade school or some high school | 69 (74.19) | 24 (25.81) | 61 (65.59) | 32 (34.41) | ||
| High school graduate or GED | 234 (81.53) | 53 (18.47) | 219 (76.31) | 68 (23.69) | ||
| Some college or technical school | 412 (85.12) | 72 (14.88) | 380 (78.51) | 104 (21.49) | ||
| College graduate | 248 (86.71) | 38 (13.29) | 227 (79.37) | 59 (20.63) | ||
| Advanced degree | 149 (80.98) | 35 (19.02) | 137 (70.46) | 47 (25.54) | ||
| Clinical characteristics | ||||||
| Nodal status | <.001 | <.001 | ||||
| Negative | 309 (88.54) | 40 (11.46) | 290 (83.09) | 59 (16.91) | ||
| 1–3 positive nodes | 433 (86.25) | 69 (13.75) | 406 (80.88) | 96 (19.12) | ||
| ≥4 positive nodes | 374 (76.80) | 113 (23.20) | 332 (68.17) | 155 (31.83) | ||
| ER/PgR status | <.001 | .01 | ||||
| Positive (either or both positive) | 751 (86.03) | 122 (13.97) | 690 (79.04) | 183 (20.96) | ||
| Negative (both negative) | 365 (78.66) | 99 (21.34) | 338 (72.84) | 126 (27.16) | ||
| HER2 status | .03 | .007 | ||||
| Negative | 867 (82.18) | 188 (17.82) | 793 (75.17) | 262 (24.83) | ||
| Positive | 245 (87.81) | 34 (12.19) | 231 (82.80) | 48 (17.20) | ||
values reflect pooled t test for age and body mass index, otherwise χ2 test and are rounded to the nearest 100th place except where <0.01. ER = estrogen receptor; PgR = progesterone receptor.
Table 2.
The prevalence of regular, weekly pre- and postdiagnosis recreational physical activity as queried before, during, and after treatment in the Diet, Exercise, Lifestyle and Cancer Prognosis Study study population according to disease-free and overall survival
| Questionnaire/Time period (N)* | Regular RPA No. (%) | Disease-free survival |
Overall survival |
|||||
|---|---|---|---|---|---|---|---|---|
| Disease free No. (%) | Recurred No. (%) | P † | Alive No. (%) | Deceased No. (%) | P† | |||
| Q1, before diagnosis (n = 1340) | Yes | 981 (73.2) | 765 (74.3) | 216 (69.7) | .11 | 828 (84.4) | 153 (80.8) | .11 |
| No | 359 (26.8) | 265 (25.7) | 94 (30.3) | 290 (15.6) | 69 (19.2) | |||
| Q2, during treatment (n = 1147) | Yes | 625 (54.5) | 493 (55.3) | 132 (51.6) | .29 | 528 (54.7) | 97 (53.3) | .72 |
| No | 522 (45.5) | 398 (44.7) | 124 (48.4) | 437 (45.3) | 85 (46.7) | |||
| Q3, 1 year after enrollment (n = 936) | Yes | 640 (68.4) | 519 (69.2) | 121 (65.1) | .28 | 560 (69.3) | 80 (62.5) | .12 |
| No | 296 (31.6) | 231 (30.8) | 65 (34.9) | 248 (30.7) | 48 (37.5) | |||
| Q4, 2 years after enrollment (n = 769) | Yes | 577 (75.1) | 483 (76.4) | 94 (68.6) | .06 | 512 (75.5) | 65 (71.4) | .39 |
| No | 192 (24.9) | 149 (23.6) | 43 (31.4) | 166 (24.5) | 26 (28.6) | |||
*Total N for each questionnaire presented herein does not exactly coincide with Figure 1 because patients/deaths were excluded for the purposes of Landmark survival analyses for each successive exposure assessment and because 2 patients who completed Q3 did not complete Q2, and 5 patients who completed Q4 did not complete all prior questionnaires. RPA = recreational physical activity; Q = questionnaire.
values reflect two-sided χ2 test.
Table 3 presents the associations of prediagnosis RPA with cancer outcomes. In comparison to inactive patients, patients reporting any regular RPA experienced a 19% and 22% decreased hazard of recurrence and mortality, respectively, although estimates weren’t statistically significant. Patients meeting or exceeding the minimum PAGAs experienced a statistically significant reduced hazard of mortality compared with those not meeting the PAGAs (HR = 0.74, 95% CI = 0.56 to 0.96); there was a similar, non-statistically significant reduction in the hazard of recurrence (HR = 0.82, 95% CI = 0.66 to 1.03). When incremental activity levels were examined, only moderately active patients experienced a statistically significant reduced hazard of recurrence (HR = 0.62, 95% CI = 0.43 to 0.88) and mortality (HR = 0.48, 95% CI = 0.31 to 0.75).
Table 3.
Hazard ratios representing the associations of prediagnosis recreational physical activity with disease recurrence and mortality in the Diet, Exercise, Lifestyle and Cancer Prognosis Study (n = 1340)
| Multivariable models* | Parameterization of recreational physical activity† | Disease recurrence |
All-cause mortality |
||||
|---|---|---|---|---|---|---|---|
| No. of events/total | HR (95% CI)‡ | P | No. of Events/total | HR (95% CI)‡ | P | ||
| Minimally- adjusted models | Any regular, weekly RPA | ||||||
| No | 94/359 | 1.00 (Referent) | 69/359 | 1.00 (Referent) | |||
| Yes | 216/981 | 0.81 (0.64 to 1.03) | .08 | 153/981 | 0.78 (0.59 to 1.04) | .09 | |
| Meet the minimum PAGAs | |||||||
| No | 174/689 | 1.00 (Referent) | 131/689 | 1.00 (Referent) | |||
| Yes | 136/651 | 0.82 (0.66 to 1.03) | .09 | 91/651 | 0.74 (0.56 to 0.96) | .02 | |
| Incremental activity categories (PAGAs) | |||||||
| No weekly RPA | 94/359 | 1.00 (Referent) | 69/359 | 1.00 (Referent) | |||
| Low weekly activity | 80/330 | 0.87 (0.65 to 1.18) | .38 | 62/330 | 0.92 (0.65 to 1.29) | .61 | |
| Moderate activity | 45/257 | 0.62 (0.43 to 0.88) | .008 | 26/257 | 0.48 (0.31 to 0.75) | .001 | |
| High activity | 91/394 | 0.88 (0.66 to 1.18) | .39 | 65/394 | 0.87 (0.62 to 1.22) | .41 | |
| Fully-adjusted models | Any regular, weekly RPA | ||||||
| No | 94/359 | 1.00 (Referent) | 69/359 | 1.00 (Referent) | |||
| Yes | 216/981 | 0.80 (0.63 to 1.02) | .07 | 153/981 | 0.76 (0.57 to 1.01) | 0.06 | |
| Meet the minimum PAGAs | |||||||
| No | 174/689 | 1.00 (Referent) | 131/689 | 1.00 (Referent) | |||
| Yes | 136/651 | 0.84 (0.67 to 1.05) | .12 | 91/651 | 0.76 (0.58 to 0.99) | 0.04 | |
| Incremental activity categories (PAGAs) | |||||||
| No weekly RPA | 94/359 | 1.00 (Referent) | 69/359 | 1.00 (Referent) | |||
| Low weekly activity | 80/330 | 0.85 (0.63 to 1.15) | .38 | 62/330 | 0.87 (0.62 to 1.24) | 0.45 | |
| Moderate activity | 45/257 | 0.65 (0.46 to 0.93) | .01 | 26/257 | 0.51 (0.32 to 0.80) | 0.003 | |
| High activity | 91/394 | 0.85 (0.64 to 1.15) | .29 | 65/394 | 0.85 (0.60 to 1.19) | 0.33 | |
Minimally adjusted models are adjusted for age and stratified by treatment arm. Fully adjusted models are adjusted for age, HER2 status, hormone receptor status, number of positive nodes, and stratified by treatment arm. CI = confidence interval; HR = hazard ratio; MET = metabolic equivalent of task; PAGAs = Physical Activity Guidelines for Americans; RPA = recreational physical activity.
METs are expressed as average MET hours per week. Any regular, weekly RPA (yes/no) denotes at least 1 session per week throughout the exposure window assessed. Meeting the minimum PAGAs (yes/no) uses 8.3 MET hours per week as the cutoff and assumes the equivalent of 150 minutes per week of moderate-intensity activity, such as brisk walking at 3.0 miles per hour. Incremental physical activity levels are defined as inactive (reference group), low active (<8.3 MET hours per week), moderately active (8.3–16 MET hours per week), and high active (>16 MET hours per week).
Standard Cox models were used to estimate HRs and 95% CIs.
Joint exposure models representing the associations of pre- and postdiagnosis RPA with outcomes are presented in Table 4. The primary finding from these analyses is that patients meeting the minimum PAGAs (yes/no) before diagnosis and at 1-year follow-up experienced statistically significantly reduced hazards of recurrence (HR = 0.59, 95% CI = 0.42 to 0.82) and mortality (HR = 0.51, 95% CI = 0.34 to 0.77); observed associations became stronger at the 2-year follow-up for both recurrence (HR = 0.45, 95% CI = 0.31 to 0.65) and mortality (HR = 0.32, 95% CI = 0.19 to 0.52). Importantly, we also observed statistically significant reduced hazards of recurrence (46%) and mortality (43%) among patients not meeting the PAGAs before diagnosis but who met the PAGAs at the 2-year follow-up.
Table 4.
Multivariable models representing the joint exposure of prediagnosis and postdiagnosis recreational physical activity with disease recurrence and all-cause mortality in the Diet, Exercise, Lifestyle and Cancer Prognosis Study
| Physical activity parameterization* | Joint exposure time periods assessed† | Disease recurrence |
All-cause mortality |
||
|---|---|---|---|---|---|
| HR (95% CI)‡ | P | HR (95% CI)‡ | P | ||
| Any regular, weekly recreational physical activity | No regular RPA before diagnosis, No during treatment | 1.00 (Referent) | 1.00 (Referent) | ||
| No before diagnosis, Yes during treatment | 1.20 (0.74 to 1.96) | .46 | 1.44 (0.83 to 2.50) | .19 | |
| Yes before diagnosis, No during treatment | 0.85 (0.60 to 1.21) | .37 | 0.83 (0.54 to 1.27) | .38 | |
| Yes before diagnosis, Yes during treatment | 0.73 (0.53 to 1.01) | .06 | 0.74 (0.51 to 1.10) | .13 | |
| No regular RPA before diagnosis, No at 1 year | 1.00 (Referent) | 1.00 (Referent) | |||
| No before diagnosis, Yes at 1-year follow-up | 0.78 (0.45 to 1.36) | .38 | 0.69 (0.36 to 1.34) | .27 | |
| Yes before diagnosis, No at 1-year follow-up | 0.72 (0.44 to 1.17) | .19 | 0.63 (0.35 to 1.12) | .11 | |
| Yes before diagnosis, Yes at 1-year follow-up | 0.70 (0.48 to 1.03) | .07 | 0.57 (0.36 to 0.88) | .01 | |
| No regular RPA before diagnosis, No at 2 years | 1.00 (Referent) | 1.00 (Referent) | |||
| No before diagnosis, Yes at 2-year follow-up | 0.73 (0.39 to 1.35) | .31 | 0.99 (0.48 to 2.04) | .99 | |
| Yes before diagnosis, No at 2-year follow-up | 0.84 (0.46 to 1.53) | .56 | 0.71 (0.32 to 1.56) | .39 | |
| Yes before diagnosis, Yes at 2-year follow-up | 0.61 (0.39 to 0.97) | .04 | 0.59 (0.33 to 1.06) | .08 | |
| Met the minimum PAGAs | No before diagnosis, No during treatment | 1.00 (Referent) | 1.00 (Referent) | ||
| No before diagnosis, Yes during treatment | 1.18 (0.73 to 1.93) | .50 | 1.29 (0.75 to 2.21) | .36 | |
| Yes before diagnosis, No during treatment | 0.86 (0.67 to 1.11) | .25 | 0.78 (0.58 to 1.06) | .12 | |
| Yes before diagnosis, Yes during treatment | 0.78 (0.55 to 1.11) | .17 | 0.69 (0.45 to 1.06) | .09 | |
| No before diagnosis, No at 1-year follow-up | 1.00 (Referent) | 1.00 (Referent) | |||
| No before diagnosis, Yes at 1-year follow-up | 0.80 (0.54 to 1.20) | .29 | 0.81 (0.51 to 1.30) | .38 | |
| Yes before diagnosis, No at 1-year follow-up | 0.96 (0.74 to 1.25) | .76 | 0.86 (0.64 to 1.20) | .41 | |
| Yes before diagnosis, Yes at 1-year follow-up | 0.59 (0.42 to 0.82) | .001 | 0.51 (0.34 to 0.77) | .001 | |
| No before diagnosis, No at 2-year follow-up | 1.00 (Referent) | 1.00 (Referent) | |||
| No before diagnosis, Yes at 2-year follow-up | 0.54 (0.35 to 0.83) | .005 | 0.57 (0.35 to 0.94) | .03 | |
| Yes before diagnosis, No at 2-year follow-up | 0.94 (0.73 to 1.21) | .64 | 0.91 (0.68 to 1.23) | .55 | |
| Yes before diagnosis, Yes at 2-year follow-up | 0.45 (0.31 to 0.65) | <.001 | 0.32 (0.19 to 0.52) | <.001 | |
METs are expressed as average MET hours per week. Any regular, weekly RPA (yes/no) denotes at least 1 session per week throughout the exposure window assessed. Meeting the minimum PAGAs (yes/no) uses <8.3 MET hours per week as the cutoff for no, ≥8.3 for yes and assumes the equivalent of 150 minutes per week of moderate-intensity activity, such as brisk walking at 3.0 miles per hour. CI = confidence interval; HR = hazard ratio; MET = metabolic equivalent of task; PAGAs = Physical Activity Guidelines for Americans; RPA = recreational physical activity.
RPA exposure during four time points was considered in joint-exposure analyses as follows: RPA before diagnosis (Q1); during treatment (Q2); 1-year follow-up (Q3); and 2-year follow-up (Q4).
Multivariable hazard models are adjusted for age and stratified by treatment arm. Standard Cox models were used to estimate HRs and 95% CIs.
In time-dependent analyses, striking statistically significant inverse associations between RPA and mortality were observed, but the association for disease recurrence was attenuated (Table 5). In comparison to inactive patients, patients reporting any regular weekly activity experienced a 63% reduced hazard of mortality. Additionally, patients meeting the minimum PAGAs experienced a 60% reduced hazard of mortality in comparison to those who didn’t meet the PAGAs. When participants were categorized according to incremental activity levels, patients reporting low and moderate volumes of activity experienced similar overall survival advantages (HR = 0.41, 95% CI = 0.24 to 0.68 and HR = 0.42, 95% CI = 0.23 to 0.76, respectively). However, highly active patients experienced the greatest survival advantage with a 69% reduced hazard of mortality (HR = 0.31, 95% CI = 0.18 to 0.53).
Table 5.
Time-dependent multivariable risk models representing the associations of regular recreational physical activity sequentially measured before, during, and after treatment with disease recurrence and mortality in the Diet, Exercise, Lifestyle and Cancer Prognosis Study
| Multivariable models* | Parameterization of recreational physical activity† | Disease recurrence |
All-cause mortality |
||
|---|---|---|---|---|---|
| HR (95% CI)‡ | P | HR (95% CI)‡ | P | ||
| Minimally adjusted models | Any regular, weekly RPA | ||||
| No | 1.00 (Referent) | 1.00 (Referent) | |||
| Yes | 0.97 (0.72 to 1.29) | .81 | 0.37 (0.26 to 0.52) | <.001 | |
| Meet the minimum PAGAs | |||||
| No | 1.00 (Referent) | 1.00 (Referent) | |||
| Yes | 1.01 (0.72 to 1.42) | .95 | 0.40 (0.27 to 0.61) | <.001 | |
| Incremental activity categories (PAGAs) | |||||
| No weekly RPA | 1.00 (Referent) | 1.00 (Referent) | |||
| Low weekly activity | 0.90 (0.55 to 1.46) | .66 | 0.41 (0.24 to 0.68) | .001 | |
| Moderate activity | 1.01 (0.61 to 1.67) | .95 | 0.42 (0.23 to 0.76) | .004 | |
| High activity | 0.98 (0.63 to 1.54) | .95 | 0.31 (0.18 to 0.53) | <.001 | |
| Fully adjusted models | Any regular, weekly RPA | ||||
| No | 1.00 (Referent) | 1.00 (Referent) | |||
| Yes | 0.95 (0.71 to 1.29) | .75 | 0.38 (0.27 to 0.54) | <.001 | |
| Meet the minimum PAGAs | |||||
| No | 1.00 (Referent) | 1.00 (Referent) | |||
| Yes | 1.00 (0.70 to 1.42) | .99 | 0.41 (0.27 to 0.63) | <.001 | |
| Incremental activity categories (PAGAs) | |||||
| No weekly RPA | 1.00 (Referent) | 1.00 (Referent) | |||
| Low weekly activity | 0.89 (0.56 to 1.44) | .64 | 0.42 (0.25 to 0.69) | <001 | |
| Moderate activity | 1.06 (0.64 to 1.74) | .82 | 0.44 (0.24 to 0.80) | .007 | |
| High activity | 0.94 (0.60 to 1.49) | .80 | 0.31 (0.18 to 0.54) | <.001 | |
Minimally adjusted models are adjusted for age and stratified by treatment arm; fully adjusted models are adjusted for age, HER2 status, hormone receptor status, number of positive nodes, and stratified by treatment arm. CI = confidence interval; HR = hazard ratio; MET = metabolic equivalent of task; PAGAs = Physical Activity Guidelines for Americans; RPA = recreational physical activity.
METs are expressed as average MET hours per week. Any regular, weekly RPA (yes/no) denotes at least 1 session per week throughout the exposure window assessed. Meeting the minimum PAGAs (yes/no) uses 8.3 MET hours per week as the cutoff and assumes the equivalent of 150 minutes per week of moderate-intensity activty, such as brisk walking at 3.0 miles per hour. Incremental physical activity levels are defined as inactive (reference group), low active (<8.3 MET hours per week), moderately active (8.3–16 MET hours per week), and high active (>16 MET hours per week).
Time dependent models were used to estimate HRs and 95% CIs.
Sensitivity landmark analyses confirmed the validity of the inverse associations between RPA and mortality observed in time-dependent models. Patients meeting the PAGAs during treatment and at 1-year follow-up experienced a 44% and 36% reduced hazard of mortality, respectively, in comparison to those not meeting the PAGAs; but as expected, precision decreased as the landmark time became shorter and data points were lost (Supplementary Table 1, available online).
Discussion
In this prospective observational study embedded in a SWOG clinical trial, we made 4 key observations that expand the current physical activity and breast cancer literature. First, high-risk breast cancer patients meeting the minimum PAGAs, both before and after diagnosis, experienced greater than 50% reduced hazards of recurrence and mortality compared with those not meeting the PAGAs at either time point. Second, patients not meeting the minimum PAGAs prior to diagnosis, but who reported meeting the PAGAs after treatment (ie, 2-year follow-up) experienced statistically significantly reduced hazards of recurrence and mortality in comparison to patients not meeting the PAGAs at that time. These findings have important implications in the clinical oncology setting because they suggest that a cancer diagnosis may serve as an impetus for increasing physical activity in some patients, and among these patients, beginning an exercise program after treatment completion resulted in a survival advantage. Third, in time-dependent analyses considering activity from all time points, striking inverse associations remained at all activity levels for mortality demonstrating that patients who consistently engaged in lower volumes of regular, weekly RPA experienced similar survival advantages as patients who met or exceeded the PAGAs. Lastly, the strong inverse relationship between prediagnosis RPA and outcomes among those meeting the recommended range of activity reveals the broader health impact of these findings by suggesting that even though exercise may not prevent breast cancer in all women, it is consistently associated with a survival advantage.
This is the first report of how pre- and postdiagnosis RPA participation measured before, during, and after chemotherapy is associated with outcomes in high-risk breast cancer patients, expanding the current knowledge regarding the role of physical activity in breast cancer survivorship. The magnitudes of the associations observed herein for prediagnosis RPA are nearly identical to those previously reported (26,27). Additionally, time-dependent analyses showing patients meeting the minimum PAGAs experienced a 58%–60% reduced hazard of mortality are congruent with a report by Lahart et al. (27), who observed a 46% reduced hazard of all-cause mortality among survivors meeting the PAGAs after diagnosis.
Importantly, the finding suggesting that patients who began meeting the PAGAs after treatment experienced a statistically significant survival advantage coincides with previous work showing that inactive patients in the decade prior to diagnosis, who became active after diagnosis, experienced a statistically significantly reduced hazard of mortality in comparison to patients who remained inactive (41). Furthermore, the finding that lower volumes of activity were similarly protective as higher volumes of activity for overall survival is congruent with previous reports failing to show a linear dose–response association between RPA and cancer outcomes (41,48–50). Indeed, in the exercise science literature, it has been consistently reported that the association between RPA and health benefits is curvilinear (51), with the steepest increase in benefit occurring at the lower levels of activity and benefits plateauing or decreasing at higher levels of activity (51–53).
Few reports have described the association of pre- or postdiagnosis RPA with breast cancer recurrence, and the relationship remains poorly understood (11,21,25). The mixed findings regarding the RPA–disease recurrence association reported herein are not entirely surprising given that time-dependent models assess short-term effects (ie, HRs represent weighted averages of the association between RPA and recurrence for each exposure window prior to an event), whereas standard models assess long-term effects of RPA from one time point (44). Inconsistencies in the literature could also be the result of varying definitions of recurrence (ie, including or excluding mortality) or the general lack of well-designed prospective studies assessing this outcome. Notably, there is a growing recognition that recurrences are not suitable proxies for mortality because they are independent outcomes that may not associate with RPA through the same underlying mechanisms (28,54).
As previously summarized (41), the most commonly cited mechanisms explaining the associations of RPA with cancer outcomes include improved body composition, a decrease in the bioavailability of sex hormones, improved insulin sensitivity, decreased inflammation, improved adipokine milieu, improved immune surveillance, and improved DNA repair (28). Data from mechanistic studies have also demonstrated that aerobic exercise in tumor-bearing mice enhances sensitivity to chemotherapy through decreased hypoxia, resulting in the direct suppression of tumor growth and recurrence across several mouse models including mammary cancer (55,56). Additional studies suggest that myokines (eg, SPARC and calprotectin) secreted from contracting skeletal muscle prevent carcinogenesis through the promotion of autophagy, apoptosis, and antitumor immunity, while preventing invasion and metastases (57). It is also plausible that regular exercise decreases breast cancer recurrence and mortality by way of blunted adrenergic signaling and subsequently blunted immunosuppression (58,59). Research has also shown that RPA is associated with decreased treatment toxicities, decreased depressive symptoms, decreased cancer-related fatigue, decreased pain, and improved sleep and overall quality of life (60–62), all which have been shown to contribute to improved clinical outcomes in cancer patients.
A primary strength of our study was the availability of pre- and postdiagnosis RPA assessed at multiple times. Reliance on prediagnosis RPA alone would not account for changes in activity throughout follow-up, and reliance on postdiagnosis RPA cannot rule out a reverse causation bias. Thus, the incorporation of exposure data collected at multiple times throughout survivorship and the use of joint-exposure and time-dependent analyses helps offset some of the potential biases that might ensue from examining RPA at only one time.
An important limitation of our study remains the reliance on self-reported RPA data, which can result in recall error and misclassification. Despite this potential limitation, the DELCaP questionnaire yielded the expected prevalence of physical (in)activity (57), including decreased activity during treatment (31), and we observed similar associations as have been previously reported for pre- and postdiagnosis activity with all-cause mortality (44), thus reinforcing our confidence in these data.
Although we assessed the role of several potential confounders, we cannot entirely rule out the possibility that residual confounding by measured or unmeasured factors influenced our results. We cannot account for comorbidities such as cardiovascular disease, a major competing cause of death among older breast cancer patients, which may have developed after treatment completion (63). Relatedly, the primary outcome herein was all-cause mortality, which is also inversely associated with RPA. However, several recently published meta-analyses have demonstrated that the point estimates representing associations for all-cause and breast-cancer specific mortality with RPA are of similar magnitudes (26,27,64), suggesting that all-cause mortality is a suitable proxy for breast-cancer mortality. Because this was a clinical trial for high-risk patients who at enrollment had no comorbidities and had normal organ function and good performance status, competing causes of mortality may have been less likely to contribute to events in comparison to previous studies. However, we also acknowledge that because these were high-risk patients, RPA may have been unable to mitigate the biology of aggressive disease, while still exerting an overall survival advantage.
An additional limitation of the current work is that we cannot describe the clinical, demographic, or survival characteristics of the subcohort of patients who did not participate in DELCaP. It is possible that patients who did not enroll were sicker, less active, and more likely to experience an event, whereas patients who were more active and more likely to survive enrolled in the study. That is, fewer patients in the reference category would inflate the observed associations reported herein. To examine this possibility, we compared the 5-year survival experience of patients enrolled in DELCaP with the survival experience in the overall SWOG trial (33), and the differences between the 2 groups were slight. Survival rates were 89% in the trial compared with 88% in DELCaP, with event rates of 0.023 and 0.026, respectively. Thus, we observed no convincing evidence of a healthy survivor bias in the current analysis.
Consistently meeting the PAGAs before diagnosis and after treatment was associated with statistically significantly reduced hazards of recurrence and mortality among high-risk breast cancer patients enrolled in the DELCaP Study. When factoring in activity before diagnosis, during treatment, and at 1- and 2-year follow-ups, patients reporting lower volumes of activity experienced similar overall survival advantages as those who met the PAGAs. These findings have valuable clinical and public health implications because they provide additional evidence to solidify the message that low-volume activity is associated with statistically significant overall survival benefits and is superior to inactivity. This is particularly encouraging given that patients and survivors may be overwhelmed by the most current PAGAs recommending 150–300 minutes per week of moderate-intensity RPA or 3 days per week of at least 30 minutes of moderate-intensity structured aerobic exercise for cancer survivors (43,65).
Funding
This work was supported by R01 CA116395 (CBA), R01 CA139426 (CBA), the Breast Cancer Research Foundation (CBA), and Roswell Park Comprehensive Cancer Center and National Cancer Institute grant, P30CA016056. S0221 was supported, in part, by National Cancer Institute, Division of Cancer Prevention SWOG NCORP Research Base grant 5UG1CA189974-02; National Cancer Institute, National Clinical Trials Network: CA180888, CA180819, CA180863, CA180858, CA180828, CA180801, CA68183, CA04919, CA13612, CA46282; and in part by Amgen, Inc.
Notes
Role of the funder: The funding agencies and organizations including the National Cancer Institute, the Breast Cancer Research Foundation, Roswell Park Comprehensive Cancer Center, the National Clinical Trials Network, and Amgen, Inc, played no role in the design and conduct of the study; the collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Conflicts of interest: William E. Barlow Research Funding: AbbVie (Inst), Merck (Inst), AstraZeneca (Inst); Kara M. Kelly Research Funding: Merck (Inst), Travel, Accommodations, Expenses: Bristol-Myers Squibb; Dawn L. Hershman Consulting or Advisory Role: AIM Specialty Health; Halle C.F. Moore Research Funding: Puma Biotechnology (Inst), AbbVie (Inst); Claudine Isaacs Honoraria: Genentech, Roche, AstraZeneca, Pfizer, Consulting or Advisory Role: Pfizer, Genentech, Roche, Novartis, AstraZeneca, Medivation, NanoString Technologies, Syndax, Puma Biotechnology, Context Therapeutics, Speakers’ Bureau: Genentech, Pfizer, AstraZeneca, Research Funding: Novartis (Inst), Pfizer (Inst), Genentech (Inst), Tesaro (Inst), Patents, Royalties, Other Intellectual Property: McGraw Hill Publishing, UpToDate, Wolters Kluwer, Elsevier; Timothy J. Hobday Consulting or Advisory Role: AbbVie, Research Funding: Novartis (Inst); Gabriel N. Hortobagyi Consulting or Advisory Role: Novartis, Peregrine Pharmaceuticals, Agendia, Research Funding: Novartis (Inst), Travel, Accommodations, Expenses: Novartis; Julie R. Gralow Consulting or Advisory Role: Novartis, Genentech, Pfizer, Merck, Puma, Biotechnology, Sandoz, AstraZeneca, Immunomedics, Genomic Health; George T. Budd Research Funding: Genentech (Inst), Roche (Inst), TRACON Pharma (Inst), Daiichi Sankyo (Inst), Eli Lilly (Inst), Macrogenics (Inst), Ambrx (Inst), Deciphera (Inst); Kathy S. Albain Consulting or Advisory Role: Novartis, Pfizer, Myriad Genetics, Genomic Health, Agendia, Genentech, Roche; Research Funding: Seattle Genetics (Inst), Other Relationship: Puma Biotechnology; No other potential conflicts of interest were reported.
Role of the author: AH: Formal analysis. SD: Writing—review & editing. WM: Data curation; Formal analysis. SEM: Writing—review & editing. GRZ: Writing—review & editing. WB: Writing—review & editing. KMK: Writing—review & editing. CAD: Writing—review & editing. DLH: Writing—review & editing. JMU: Writing—review & editing. HCFM: Writing—review & editing. JAS: Writing—review & editing. CI: Writing—review & editing. TJH: Writing—review & editing. MS: Writing—review & editing. GNH: Writing—review & editing. JRG: Writing—review & editing. KSA: Writing—review & editing. GTB: Writing—review & editing. CBA: Conceptualization; Data curation; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Writing—review & editing.
Supplementary Material
References
- 1. Abrahamson PE, Gammon MD, Lund MJ, et al. Recreational physical activity and survival among young women with breast cancer. Cancer. 2006;107(8):1777–1785. [DOI] [PubMed] [Google Scholar]
- 2. Cleveland RJ, Eng SM, Stevens J, et al. Influence of prediagnostic recreational physical activity on survival from breast cancer. Eur J Cancer Prev. 2012;21(1):46–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Dal Maso L, Zucchetto A, Talamini R, et al. Effect of obesity and other lifestyle factors on mortality in women with breast cancer. Int J Cancer. 2008;123(9):2188–2194. [DOI] [PubMed] [Google Scholar]
- 4. de Glas NA, Fontein DB, Bastiaannet E, et al. Physical activity and survival of postmenopausal, hormone receptor-positive breast cancer patients: results of the tamoxifen exemestane adjuvant multicenter lifestyle study. Cancer. 2014;120(18):2847–2854. [DOI] [PubMed] [Google Scholar]
- 5. Emaus A, Veierod MB, Tretli S, et al. Metabolic profile, physical activity, and mortality in breast cancer patients. Breast Cancer Res Treat. 2010;121(3):651–660. [DOI] [PubMed] [Google Scholar]
- 6. Friedenreich CM, Gregory J, Kopciuk KA, et al. Prospective cohort study of lifetime physical activity and breast cancer survival. Int J Cancer. 2009;124(8):1954–1962. [DOI] [PubMed] [Google Scholar]
- 7. Hellmann SS, Thygesen LC, Tolstrup JS, et al. Modifiable risk factors and survival in women diagnosed with primary breast cancer: results from a prospective cohort study. Eur J Cancer Prev. 2010;19(5):366–373. [DOI] [PubMed] [Google Scholar]
- 8. Irwin ML, McTiernan A, Manson JE, et al. Physical activity and survival in postmenopausal women with breast cancer: results from the Women’s Health Initiative. Cancer Prev Res (Phila). 2011;4(4):522–529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Irwin ML, Smith AW, McTiernan A, et al. Influence of pre- and postdiagnosis physical activity on mortality in breast cancer survivors: the health, eating, activity, and lifestyle study. J Clin Oncol. 2008;26(24):3958–3964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Keegan TH, Milne RL, Andrulis IL, et al. Past recreational physical activity, body size, and all-cause mortality following breast cancer diagnosis: results from the breast cancer family registry. Breast Cancer Res Treat. 2010;123(2):531–542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Schmidt ME, Chang-Claude J, Vrieling A, et al. Association of pre-diagnosis physical activity with recurrence and mortality among women with breast cancer. Int J Cancer. 2013;133(6):1431–1440. [DOI] [PubMed] [Google Scholar]
- 12. Tao MH, Hainaut P, Marian C, et al. Association of prediagnostic physical activity with survival following breast cancer diagnosis: influence of TP53 mutation status. Cancer Causes Control. 2013;24(12):2177–2186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. West-Wright CN, Henderson KD, Sullivan-Halley J, et al. Long-term and recent recreational physical activity and survival after breast cancer: the California Teachers Study. Cancer Epidemiol Biomarkers Prev. 2009;18(11):2851–2859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Williams PT. Breast cancer mortality vs. exercise and breast size in runners and walkers. PLoS One. 2013;8(12):e80616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ammitzboll G, Sogaard K, Karlsen RV, et al. Physical activity and survival in breast cancer. Eur J Cancer. 2016;66:67–74. [DOI] [PubMed] [Google Scholar]
- 16. Beasley JM, Kwan ML, Chen WY, et al. Meeting the physical activity guidelines and survival after breast cancer: findings from the after breast cancer pooling project. Breast Cancer Res Treat. 2012;131(2):637–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bertram LA, Stefanick ML, Saquib N, et al. Physical activity, additional breast cancer events, and mortality among early-stage breast cancer survivors: findings from the WHEL Study. Cancer Causes Control. 2011;22(3):427–435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Borch KB, Braaten T, Lund E, et al. Physical activity before and after breast cancer diagnosis and survival - the Norwegian women and cancer cohort study. BMC Cancer. 2015;15(1):967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Bradshaw PT, Ibrahim JG, Khankari N, et al. Post-diagnosis physical activity and survival after breast cancer diagnosis: the Long Island Breast Cancer Study. Breast Cancer Res Treat. 2014;145(3):735–742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Holick CN, Newcomb PA, Trentham-Dietz A, et al. Physical activity and survival after diagnosis of invasive breast cancer. Cancer Epidemiol Biomarkers Prev. 2008;17(2):379–386. [DOI] [PubMed] [Google Scholar]
- 21. Holmes MD, Chen WY, Feskanich D, et al. Physical activity and survival after breast cancer diagnosis. JAMA. 2005;293(20):2479–2486. [DOI] [PubMed] [Google Scholar]
- 22. Maliniak ML, Patel AV, McCullough ML, et al. Obesity, physical activity, and breast cancer survival among older breast cancer survivors in the Cancer Prevention Study-II Nutrition Cohort. Breast Cancer Res Treat. 2018;167(1):133–145. [DOI] [PubMed] [Google Scholar]
- 23. Nelson SH, Marinac CR, Patterson RE, et al. Impact of very low physical activity, BMI, and comorbidities on mortality among breast cancer survivors. Breast Cancer Res Treat. 2016;155(3):551–557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Palesh O, Kamen C, Sharp S, et al. Physical activity and survival in women with advanced breast cancer. Cancer Nurs. 2018;41(4):E31–E38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Sternfeld B, Weltzien E, Quesenberry CP Jr, et al. Physical activity and risk of recurrence and mortality in breast cancer survivors: findings from the LACE study. Cancer Epidemiol Biomarkers Prev. 2009;18(1):87–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lee J. A meta-analysis of the association between physical activity and breast cancer mortality. Cancer Nurs. 2018;42(4):271–285. [DOI] [PubMed] [Google Scholar]
- 27. Lahart IM, Metsios GS, Nevill AM, et al. Physical activity, risk of death and recurrence in breast cancer survivors: A systematic review and meta-analysis of epidemiological studies. Acta Oncol. 2015;54(5):635–654. [DOI] [PubMed] [Google Scholar]
- 28. Friedenreich CM, Shaw E, Neilson HK, et al. Epidemiology and biology of physical activity and cancer recurrence. J Mol Med. 2017;95(10):1029–1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Branstrom R, Petersson LM, Saboonchi F, et al. Physical activity following a breast cancer diagnosis: implications for self-rated health and cancer-related symptoms. Eur J Oncol Nurs. 2015;19(6):680–685. [DOI] [PubMed] [Google Scholar]
- 30. De Groef A, Geraerts I, Demeyer H, et al. Physical activity levels after treatment for breast cancer: two-year follow-up. Breast. 2018;40:23–28. [DOI] [PubMed] [Google Scholar]
- 31. Devoogdt N, Van Kampen M, Geraerts I, et al. Physical activity levels after treatment for breast cancer: one-year follow-up. Breast Cancer Res Treat. 2010;123(2):417–425. [DOI] [PubMed] [Google Scholar]
- 32.National Cancer Institute. Cancer survivors and physical activity. 2018. https://progressreport.cancer.gov/after/physical_activity. Accessed February 2018.
- 33. Budd GT, Barlow WE, Moore HC, et al. SWOG S0221: a phase III trial comparing chemotherapy schedules in high-risk early-stage breast cancer. J Clin Oncol. 2015;33(1):58–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Zirpoli GR, Brennan PM, Hong CC, et al. Supplement use during an intergroup clinical trial for breast cancer (S0221). Breast Cancer Res Treat. 2013;137(3):903–913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Ambrosone CB, Zirpoli GR, Hutson AD, et al. Dietary supplement use during chemotherapy and survival outcomes of patients with breast cancer enrolled in a cooperative group clinical trial (SWOG S0221). J Clin Oncol. 2020;38(8):804–814. doi: 10.1200/jco.19.01203: Jco1901203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Luc L, Baumgart C, Weiss E, et al. Dietary supplement use among participants of a databank and biorepository at a comprehensive cancer centre. Public Health Nutr. 2015;18(5):916–926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Chasan-Taber L, Erickson JB, McBride JW, et al. Reproducibility of a self-administered lifetime physical activity questionnaire among female college alumnae. Am J Epidemiol. 2002;155(3):282–289. [DOI] [PubMed] [Google Scholar]
- 38. Ainsworth BE, Haskell WL, Herrmann SD, et al. 2011 Compendium of physical activities: a second update of codes and MET values. Med Sci Sports Exerc. 2011;43(8):1575–1581. [DOI] [PubMed] [Google Scholar]
- 39. Ainsworth BE, Haskell WL, Leon AS, et al. Compendium of physical activities: classification of energy costs of human physical activities. Med Sci Sports Exerc. 1993;25(1):71–80. [DOI] [PubMed] [Google Scholar]
- 40. Ainsworth BE, Haskell WL, Whitt MC, et al. Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exerc. 2000;32(9 suppl):S498–S504. [DOI] [PubMed] [Google Scholar]
- 41. Cannioto RA, Dighe S, Mahoney MC, et al. Habitual recreational physical activity is associated with significantly improved survival in cancer patients: evidence from the Roswell Park Data Bank and BioRepository. Cancer Causes Control. 2019;30(1):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. O’Donovan G, Lee I-M, Hamer M, et al. Association of “weekend warrior” and other leisure time physical activity patterns with risks for all-cause, cardiovascular disease, and cancer mortality. JAMA Intern Med. 2017;177(3):335–342. [DOI] [PubMed] [Google Scholar]
- 43.USDHHS. 2018 Physical Activity Guidelines for Americans Washington, DC: Office of Disease Prevention and Health Promotion; 2018.
- 44. Dekker FW, de Mutsert R, van Dijk PC, et al. Survival analysis: time-dependent effects and time-varying risk factors. Kidney Int. 2008;74(8):994–997. [DOI] [PubMed] [Google Scholar]
- 45. Agarwal P, Moshier E, Ru M, et al. Immortal time bias in observational studies of time-to-event outcomes: assessing effects of postmastectomy radiation therapy using the National Cancer Database. Cancer Control. 2018;25(1):107327481878935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Maldonado G, Greenland S.. Simulation study of confounder-selection strategies. Am J Epidemiol. 1993;138(11):923–936. [DOI] [PubMed] [Google Scholar]
- 47. Brick J, Kalton G.. Handling missing data in survey research. Stat Methods Med Res. 1996;5(3):215–238. [DOI] [PubMed] [Google Scholar]
- 48. Li T, Wei S, Shi Y, et al. The dose-response effect of physical activity on cancer mortality: findings from 71 prospective cohort studies. Br J Sports Med. 2016;50(6):339–345. [DOI] [PubMed] [Google Scholar]
- 49. Zhong S, Jiang T, Ma T, et al. Association between physical activity and mortality in breast cancer: a meta-analysis of cohort studies. Eur J Epidemiol. 2014;29(6):391–404. [DOI] [PubMed] [Google Scholar]
- 50. Li Y, Gu M, Jing F, et al. Association between physical activity and all cancer mortality: dose-response meta-analysis of cohort studies. Int J Cancer. 2016;138(4):818–832. [DOI] [PubMed] [Google Scholar]
- 51. Blair SN, Cheng Y, Holder JS.. Is physical activity or physical fitness more important in defining health benefits? Med Sci Sports Exerc. 2001;33(6 suppl):S379–99; discussion S419–420. [DOI] [PubMed] [Google Scholar]
- 52. Janssen I, Jolliffe CJ.. Influence of physical activity on mortality in elderly with coronary artery disease. Med Sci Sports Exerc. 2006;38(3):418–417. [DOI] [PubMed] [Google Scholar]
- 53. Sundquist K, Qvist J, Sundquist J, et al. Frequent and occasional physical activity in the elderly: a 12-year follow-up study of mortality. Am J Prev Med. 2004;27(1):22–27. [DOI] [PubMed] [Google Scholar]
- 54. Lafourcade A, His M, Baglietto L, et al. Factors associated with breast cancer recurrences or mortality and dynamic prediction of death using history of cancer recurrences: the French E3N cohort. BMC Cancer. 2018;18(1):171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Pedersen L, Idorn M, Olofsson GH, et al. Voluntary running suppresses tumor growth through epinephrine- and IL-6-dependent NK cell mobilization and redistribution. Cell Metab. 2016;23(3):554–562. [DOI] [PubMed] [Google Scholar]
- 56. Betof AS, Lascola CD, Weitzel D, et al. Modulation of murine breast tumor vascularity, hypoxia and chemotherapeutic response by exercise. J Natl Cancer Inst. 2015;107(5):djv040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Sanchis-Gomar F, Lucia A, Yvert T, et al. Physical inactivity and low fitness deserve more attention to alter cancer risk and prognosis. Cancer Prev Res (Phila). 2015;8(2):105–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Repasky EA, Eng J, Hylander BL.. Stress, metabolism and cancer: integrated pathways contributing to immune suppression. Cancer J. 2015;21(2):97–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Silverman MN, Deuster PA.. Biological mechanisms underlying the role of physical fitness in health and resilience. Interface Focus. 2014;4(5):20140040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Mishra SI, Scherer RW, Geigle PM, et al. Exercise interventions on health-related quality of life for cancer survivors. Cochrane Database Syst Rev. 2012;(8):CD007566. doi:10.1002/14651858.CD007566.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Duregon F, Vendramin B, Bullo V, et al. Effects of exercise on cancer patients suffering chemotherapy-induced peripheral neuropathy undergoing treatment: a systematic review. Crit Rev Oncol Hematol. 2018;121:90–100. [DOI] [PubMed] [Google Scholar]
- 62. Greenlee H, Hershman DL, Shi Z, et al. BMI, lifestyle factors and taxane-induced neuropathy in breast cancer patients: the pathways study. J Natl Cancer Inst. 2017;109(2):djw206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Mehta LS, Watson KE, Barac A, et al. Cardiovascular disease and breast cancer: where these entities intersect: a scientific statement from the American Heart Association. Circulation. 2018;137(8):e30–e66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Spei ME, Samoli E, Bravi F, et al. Physical activity in breast cancer survivors: a systematic review and meta-analysis on overall and breast cancer survival. Breast. 2019;44:144–152. [DOI] [PubMed] [Google Scholar]
- 65. Campbell KL, Winters-Stone KM, Wiskemann J, et al. Exercise guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51(11):2375–2390. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.