ABSTRACT
Chimeric antigen receptor (CAR)-T cell therapies have achieved remarkable success. However, application-related toxicities, such as cytokine release syndrome or neurotoxicity, moved natural killer (NK) cells into focus as novel players in immunotherapy. CAR-NK cells provide an advantageous dual killing-capacity by CAR-dependent and -independent mechanisms and induce few side effects. While the majority of trials still use CAR-T cells, CAR-NK cell trials are on the rise with 19 ongoing studies worldwide. This review illuminates the current state of research and clinical application of CAR-NK cells, as well as future developmental potential.
KEYWORDS: CAR, NK cells, NK-92 cells, cell therapy
Cytotoxic T cells and natural killer cells as immunotherapeutic tools
In addition to cytotoxic T lymphocytes (CTLs), natural killer (NK) cells have recently become a novel tool for immunotherapy. Although these cell types utilize similar killing mechanisms for eliminating malignant or virally infected cells, their target recognition mechanism differs significantly.1,2 CTLs, as part of the adaptive immune response, recognize their targets via a wide variety of clonally rearranged T cell receptors (TCRs) (Figure 1a), while NK cells, as innate lymphoid cells, integrate activating and inhibitory signals received by their germline-encoded receptor repertoire (Figure 1b). These distinct target recognition mechanisms allow complementary function and render NK cells as an attractive tool for immunotherapy.
Figure 1.
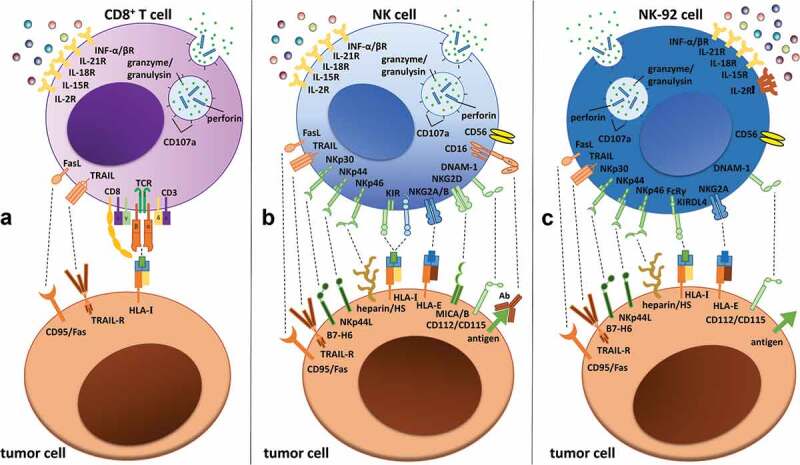
Receptor expression and interactions of (a) CD8+ T cells, (b) NK cells and (c) NK-92 cells with tumor target cells. T cells primarily recognize target cells via the TCR–HLA/MHC-I interaction, while NK and NK-92 cell activation depends on highly polymorphic activating and inhibitory germline-encoded receptors, such as KIRs or receptors of the NKG2-family. Activating receptors are shown in green, inhibitory receptors are shown in blue and killer receptors are shown in orange. Interleukin receptors are indicated in yellow, with the IL-2 receptor highlighted in orange on the NK-92 cell to illustrate its strong IL-2-dependency. Please note that only a selection of the most important receptors/ligands are shown, without claim of completeness.3–6Abbreviations: CD, cluster of differentiation; DNAM-1, DNAX accessory molecule-1; HS, heparan sulfate; HLA, human leukocyte antigen; IL, interleukin; INF, interferon; KIR, killer-cell immunoglobulin-like receptor; MHC, major histocompatibility complex; MIC, MHC class I chain-related protein; NKG2, natural killer group 2; TCR, T cell receptor; TRAIL, TNF-related apoptosis-inducing ligand.
Insights into NK and T cells – abundance and phenotype
NK cells are defined as CD56+CD3− lymphocytes and are traditionally classified into two main subsets based on the expression of CD56 and the low-affinity Fc gamma receptor 3A (FcγRIIIa, CD16). One population is primarily immunomodulatory with cells defined as CD56brightCD16dim while the other consists of CD56dimCD16bright NK cells displaying stronger cytotoxicity. Further functional subpopulations can be defined by the expression of several surface markers, e.g. chemokine receptors such as CXCR4, CX3CR1 or CCR7, which determine tissue distribution and homing capability as well as maturation markers such as CD27 or CD57, which define different developmental stages.7–9
The complete lack of TCR expression phenotypically and functionally distinguishes NK cells from CD3+ T lymphocytes. In contrast to T cells, which develop in the thymus, NK cells primarily develop and differentiate in the bone marrow and subsequently enter the circulation, where they account for up to 10–15% of peripheral blood mononuclear cells (PBMCs). T cells are much more abundant, with frequencies above 60%10,11 and mainly classified as CD4+ helper or CD8+ cytotoxic T cells. Additionally, T cells can be further divided into specific subsets, e.g., regulatory T cells (Treg, CD4+CD25+Foxp3+),12 memory T cells (CD3+CD45RO+),13 γδ T cells (TCR-γδ+),14 or NKT cells (CD3+CD16+CD56+). Of these, the NKT cell subset seems particularly interesting, as they show typical NK cell features while expressing the αβ-TCR to detect antigens displayed by CD1d, a monomorphic human leukocyte antigen (HLA)-like molecule, thereby potentially enabling allogeneic CAR-NKT cell therapies.15 However, in this review, we focus on the comparison of (CAR-)NK and (CAR-)T cells as being the major players in CAR-cell therapies at the moment.
Regulation of cytotoxicity – what causes the difference between T and NK cells?
T cells detect malignant or virus-infected cells through interactions between a wide variety of membrane-expressed TCRs and antigen loaded major histocompatibility complex (MHC) molecules present on the surface of all nucleated cells. A robust T cell response can take days to establish due to the necessity of encountering a T cell clone with the appropriate TCR-antigen specificity.
In contrast, NK cells do not require antigen-specific recognition to kill target cells. CD16 expression on NK cells (Figure 1b) enables these cells to mediate antibody-dependent cellular cytotoxicity (ADCC), which constitutes an activation-independent killing mechanism not available to T cells (Figure 1a).16 In addition, NK cells express a wide repertoire of highly polymorphic germline-encoded activating or inhibitory receptors that recognize distinct target cell ligands (Figure 1b).17,18 A number of inhibitory NK cell receptors, such as killer-immunoglobulin like receptors (KIRs) or the (C-type lectin-like) natural killer group 2 molecules NKG2A/B,18–20 bind to canonical and non-canonical HLA proteins, thereby inducing the cytolysis of cells lacking expression of these molecules. This process is called “missing-self” recognition.17 Downregulation of MHC-I/HLA-I protein expression represents one immune escape mechanism that tumor or viral-infected cells use to evade T cell recognition.21–23 However, MHC-independent inhibitory ligands, such as 2B4, KLRG1 or CEACAM1, can also be detected by NK cells.24 The major receptors responsible for NK cell activation are other C-type lectin-like molecules, such as NKG2D,19,25 DNAM-1 and the natural cytotoxicity receptors (NCRs) NKp30, NKp44 and NKp46. NCRs are lysis-triggering receptor proteins that are specific for non-MHC ligands and are localized in the cell membrane of NK cells in all higher mammals.5,26,27 The initial stochastically driven expression of these receptors is altered via an educational process that defines the final repertoire and activation status of each NK cell. Different models have been described for the process of maturation, such as the “licensing and arming”-model, the “disarming”-model and the “tuning”-model.28–30 Following activation, NK cells, similar to cytotoxic T cells, can mediate killing after forming a lytic synapse. Cell death is induced via either the death receptor pathway (e.g., through Fas/CD95 or TNF-related apoptosis-inducing ligand (TRAIL)) or the perforin granzyme pathway, which constitutes the major mode of killing by NK cells. The latter pathway utilizes the coordinated secretion of cytotoxic granules, which include pore-forming proteins, granulysin and/or perforin and combinations of effector proteases from the granzyme (Gzm) family (in humans: Gzm A, B, K, M and H). These molecules can enter the target cell and induce apoptosis, yet seem to be utilized differently in various T and NK cell lineages. Overall, NK cells exhibit the strongest expression of cytotoxic molecules, followed by CD8+ T cells and CD4+ T cells, for which only the TH1 subset expresses granulysin, granzyme and perforin.31
Different NK cell sources represent alternative therapeutic approaches
A common hurdle for NK cell-based therapies is the limited availability of primary NK cells, which could be overcome by improving NK cell expansion protocols using different cytokines, serum or feeder cells. Alternatively, different NK cell sources have been tested to overcome this limitation. Several NK cell lines exist, such as NK-92, NKL, YT, NK3.3 and NK-YS, but only the NK-92 cell line is approved for treatment of patients in clinical studies. NK-92 cells can be easily expanded under good manufacturing practice (GMP) conditions and show antitumor efficacy.32,33 These cells possess characteristics similar to those of activated primary NK cells but lack CD16 expression, which prevents them from inducing ADCC (Figure 1c). Notably, NK-92 cells have the advantage of lacking almost all inhibitory receptors (except for KIR2DL4 and CD94/NKG2A) and are therefore constitutively activated.6 Despite their relatively high cytotoxic activity and advantages regarding disposability and genetic manipulation, NK-92 cells face limitations in their clinical applicability. A constrain is that they originate from a lymphoma, which necessitates their irradiation prior to infusion to reduce their proliferation and persistence in vivo.34 Additionally, NK-92 cells are highly dependent on IL-2, which raises concern about toxicity if repeated IL-2 injections are required in NK-92-based cell therapies.35–37
Other alternatives are NK cells obtained from umbilical cord blood (UCB) or derived from either human embryonic or pluripotent stem cells (hESCs or hPSCs, respectively). The advantage of NK cells derived from UCB, hESCs or hPSCs is the wide availability of these cell sources and the ease of clinical-grade expansion under GMP conditions with 3- to 4-log expansion rates. In addition, UCB- or hPSC-derived NK cells demonstrated tolerance of long-term cryopreservation without loss of function, making them potential “off-the-shelf” products for NK cell-based immunotherapies.38,39
NK cells in the clinic: an overview of phase I/II trials
Patients with selective NK cell deficiencies suffer from uncontrolled viral infections, indicating the need for improved NK cell function.40 Clinical data from Ruggeri et al. in 2002 first highlighted the beneficial outcomes observed in acute myeloid leukemia (AML) patients in mismatched hematopoietic stem cell transplantation (allo-HSCT) who received alloreactive NK cells without an increased incidence of graft-versus-host disease (GvHD).41 In recent years, NK cell-based approaches have been developed for the treatment of not only AML but also other forms of cancer.42 Overall, there are currently more than 200 NK cell therapy trials under clinical evaluation worldwide (Figure 2a; for actual updates, see: www.ClinicalTrials.gov). However, divergent results have been reported by different studies.
Figure 2.
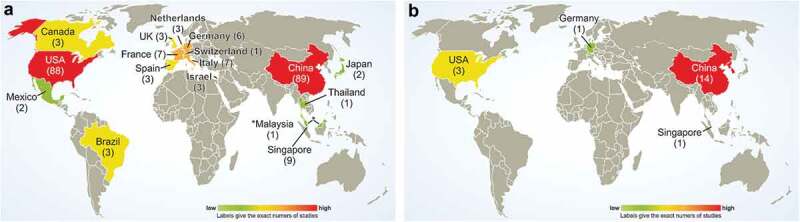
Geographical distribution of ongoing NK cell (a) and CAR-NK cell (b) therapy trials worldwide, as registered by mid February 2020 at https://www.clinicaltrials.gov/ with “Cancer (NK AND cell) OR NK-92 OR (natural AND killer AND cell)” as search terms and CAR-NK cell clinical trials as listed in Table 1.
Early approaches investigated the administration of autologous NK cells derived from peripheral blood (PB) in combination with high-dose IL-2. These so-called lymphokine-activated killer (LAK) cells showed severe side effects and limited antitumor activity.43 Although low-dose IL-2 has been indicated to be safe, IL-2 promotes not only the proliferation of NK cells but also the expansion of Tregs, which might inhibit immune function.44 Protocols to improve the persistence, activation and number of NK cells involve administration of different immune stimulants, including cytokines to augment NK cell activity, cytotoxicity and ex vivo expansion to produce sufficient NK cell numbers for adoptive transfer (for review, see Granzin et al.).45 For instance, several clinical trials have investigated the use of recombinant human IL-15 combined with different antibodies (NCT03759184, NCT02689453, NCT03388632, NCT03905135) or a superagonist of IL-15, ALT-803, to promote the proliferation of circulating NK cells (NCT01898793 and NCT02782546).46
The addition of monoclonal antibodies to NK cell-based immunotherapy can increase the cytolytic potential of NK cells by either promoting ADCC or blocking the interactions of inhibitory NK cell receptors with their respective tumor ligands. Some clinically approved antibodies that enhance ADCC are the anti-EGFR monoclonal antibody (mAb) cetuximab,47 the anti-Her2 mAb trastuzumab48 and the anti-CD20 mAb rituximab.49 Furthermore, bi- and tri-specific mAbs can be used as cross-linkers to form immunological synapses by binding both tumor antigens and NK cells.50 Checkpoint inhibitors, which have been very successfully used in T cell-based immunotherapy, can also be combined with adoptively transferred NK cells to overcome immunosuppression. The expression of the inhibitory receptor programmed cell death 1 (PD-1), for example, was found to be upregulated on NK cells from patients with multiple myeloma, and blockade of PD-1 led to enhanced NK cell cytotoxicity in these patients.51 Besides PD-1, other checkpoint inhibitors, including mAbs against KIR, NKG2A, CTLA-4, B7-H3, Siglec-7, TIGIT, TIM-3 and LAG-3 are under clinical evaluation in the context of NK cell-based immunotherapy (for review, see Khan et al.).52
The transfer of ex vivo expanded autologous NK cells has been found to be safe in a range of clinical trials for treating lymphoma, colon cancer, breast cancer and lung cancer patients. However, the effect on tumor suppression appeared to be low.53,54
To overcome the “missing-self” recognition of tumor cells, KIR-ligand mismatched allogeneic or haploidentical NK cell infusions are used. The persistence, in vivo expansion and increased antitumor activity of allogeneic NK cells have been demonstrated.55. The safety and feasibility of the adoptive transfer of allogeneic NK cells has been demonstrated in several phase I and II clinical trials, which showed no evidence of GvHD, cytokine release syndrome (CRS) or neurotoxicity.55–57 Only in very few cases NK cell infusion was suggested to be associated with GvHD.58 The contribution of blood-derived NK cells to GvHD is controversially discussed due to the fact that contaminating T cells can potentially indirectly contribute to GvHD (for review, see Lupo and Matosevic).59 To date, allogeneic NK cell infusions have been used in several tumor immunotherapy clinical trials of hematological malignancies and solid tumors such as melanoma, breast cancer, ovarian cancer, neuroblastoma, renal cell carcinoma, colorectal cancer and hepatocellular cancer. The results showed tremendous variations in clinical response among the different types of cancers.56 For example, in a phase I/II clinical trial (NCT00625729) of patients with relapsed non-Hodgkin lymphoma (NHL) or chronic lymphocytic leukemia (CLL), allogeneic NK cell infusions were given in combination with chemotherapy and rituximab. Four out of six patients showed complete or partial remission after 3 months, whereas two patients progressed after 6 months. In another phase II clinical trial evaluating the efficacy of allogeneic NK cell infusions for ovarian, fallopian tube, peritoneal and metastatic breast cancers, four out of 13 enrolled patients were alive 1 year after therapy (NCT01105650). Limitations in the treatment of solid tumors include the poor capability of NK cells to reach the tumor tissue, their inadequate in vivo expansion and persistence, and suppression mediated by the tumor microenvironment, which remains a major hurdle for the effectiveness of adoptive NK cell therapy. Cytokine-induced memory-like (CIML) NK cells, which can be generated by a pre-stimulatory cytokine protocol using IL-12, IL-15 and IL-18 may be a strategy to overcome some of these limitations based on their prolonged persistence in vivo.60 Several phase I or II clinical trials are elucidating CIML NK cells in the context of leukemia (e.g., NCT04354025, NCT03068819, NCT02782546) and head and neck cancer (NCT04290546).
NK-92 cells have been investigated in several clinical trials with variable clinical outcomes. For example, in a phase I clinical trial, patients with lymphoma or multiple myeloma who relapsed after autologous hematopoietic stem cell transplantation (AHSCT) were treated with NK-92 cell infusions. Two out of twelve patients showed a complete response, two patients showed minor responses, and one patient showed clinical improvement (NCT00990717).61 Of note, the success of NK-92 cell-based therapy is limited due to the need for prior irradiation resulting in a short lifespan in vivo. In addition, UCB-derived NK cells have been evaluated in several studies. In a phase II clinical trial, donor UCB NK cell infusions after chemotherapy in patients with refractory hematological cancer led to disease-free survival up to 6 months in two out of fifteen patients, with one patient remaining disease-free up to 12 months (NCT00354172).
From these studies, it can be concluded that the success of adoptive NK cell therapy depends on pretreatment with cytoreducing chemotherapy and activation of the NK cells with several cytokines alone or in combinations such as IL-2, IL-12, IL-15, IL-18, and IL-21, or ex vivo expansion using membrane bound (mb) IL-21-expressing feeder cells.55,60,62
A new tool for immunotherapy: CAR-NK cells
The development of genetically modified NK cells enabled a new approach for cancer immunotherapy. Based on the success of CAR-engineered autologous T cells, which achieved durable complete responses in patients with B-cell leukemia and lymphoma,63 and led to the approval of two CD19-targeted CAR-T cell products in the US and Europe (Kymriah® and Yescarta®), most studies performed to date have aimed to enhance the cytotoxicity of NK cells toward specific targets by the induced expression of CARs.
CARs are synthetically engineered molecules consisting of an extracellular binding domain, a hinge region, a transmembrane domain and one or more intracellular signaling domains. The extracellular antigen-binding domain is usually a single chain variable fragment (scFv) derived from a mouse antibody that recognizes a cognate tumor antigen. Recent studies have revealed the feasibility and efficacy of utilizing a Fab fragment, a nanobody or designed ankyrin repeat proteins (DARPins) instead of the scFv. 64–66
The extracellular binding domain allows MHC-independent target recognition and enables targeting of a very large repertoire of surface molecules, including proteins and carbohydrates.67 The hinge and transmembrane regions are commonly derived from CD8α or CD28. The design of the CAR intracellular domain varies according to the CAR generation. First-generation CARs include only the CD3ζ signaling domain, while second-generation CARs harbor an additional costimulatory domain. In third-generation CARs, two costimulatory domains are present in conjunction with the signaling domain of CD3ζ. Upon antigen contact, the intracellular signaling domains become activated, which in turn stimulates the downstream signaling cascade of the CAR-modified cell, leading to cancer cell killing. Different costimulatory domains such as CD28, 4–1BB (CD137), 2B4, OX40, DAP10 and DAP12 have been investigated, but CD28 and 4–1BB represent the most commonly used domains. Other approaches have been based on bispecific CARs that contain two distinct antigen-recognition domains either bound to one transmembrane domain and an intracellular signaling domain (tandem CAR) or to two single CAR constructs (dual CAR). These CARs exhibit several advantages in tumor killing, such as the avoidance of tumor immune escape due to the downregulation of antigen expression.68 Combinatorial CARs can function either in an AND-mode, requiring binding of both antigens, or in an OR-mode, where recognition of one antigen is sufficient. The combinatorial antigen-recognition strategy allows targeting of non-tumor specific antigens, since only the expression of both antigens leads to CAR-activation and thereby provides tumor specificity.69 New approaches utilize additional structural frameworks for CAR-cell therapy, such as recently developed fourth-generation CARs or so-called T cells redirected for universal cytokine killing (TRUCKs) that contain an additional inducible cytokine released upon T cell activation, thereby further modulating the tumor microenvironment.67 Furthermore, novel customized synthetic Notch (synNotch) receptors have been developed that release intracellular transcription factors upon antigen recognition and receptor activation.69 Turning an immunosuppressive signal into an activating signal can be achieved using inhibitory CARs that carry an inhibitory extracellular domain fused to an activating intracellular CAR domain.70
Additionally, split CAR technologies have evolved in recent years. Amongst these, the highly flexible adapter CAR technology, termed AdCAR, uses biotinylated mAbs and fragments as adapter molecules to quantitatively regulate immune effector cell function with on- and off-switch kinetics depending on the pharmacodynamics and pharmacokinetics of the utilized adapter molecule format. Separating antigen recognition and CAR activation facilitates universal, as well as combinatorial, immunotargeting.71,72
Generation of CAR-NK cells
Different systems, which can be classified as viral and nonviral technologies, can be used for the genetic modification of NK cells and NK cell lines. Many viral vector systems, including alpharetroviral (α-RV), gammaretroviral (γ-RV) and lentiviral (LV) systems have been developed, with retroviral vectors being the most commonly used (for review see Matosevic et al.).73 Transduction efficiencies differ between published studies and depend on the NK cell source, the viral vector system and the transduction enhancer used.73 For instance, γ-RV vector systems have achieved high transduction efficiencies in expanded PB- and UCB-derived NK cells.74–76 Interestingly, most protocols used gene-modified K562 feeder cells expressing membrane-bound cytokines (mbIL-21 or IL-15) and 4–1BB to enhance NK cell expansion and potentially improve the transduction efficiency. It is still unknown whether high transduction rates solely depend on the γ-RV vectors or whether other conditions play a significant role as well. Nevertheless, the use of γ-RV vectors is associated with the risk of insertional mutagenesis and oncogenesis. In a study of X-linked severe combined immunodeficiency (SCID), nine out of ten patients were cured using γ-RV-mediated gene therapy, but four of them developed T cell leukemia, indicating the need for viral vector systems with a safer integration pattern and a decreased risk of insertion mutation (i.e., LV and α-RV vectors or a nonviral approach).77,78
Further development of LV systems resulted in self-inactivating (SIN) LV vectors, which demonstrate a lower risk of insertional oncogenesis than γ-RV vectors.79,80 Additionally, LVs can infect nondividing cells, while γ- and α-RVs preferentially infect dividing cells. Subsequently, LV vectors have been successfully used to generate CAR-NK cells from a variety of sources.81 However, in the initial studies the transduction rate of PB-NK cells was limited to only 8−16%, while transduction rates of up to 70% were achieved for UCB-derived NK cells, highlighting the dependency of transduction efficiency on the NK cell origin and donor-dependent variation.82
The α-RV SIN vectors represent the newest generation of viral vectors used for NK cell transduction. It has been reported that α-RV vectors outperform both γ-RV and LV vectors in the transduction of PB-derived NK cells, exhibiting up to 60% transduction efficiency.83 Based on these findings, α-RV vectors have been used to successfully generate PB-derived CAR-NK cells redirected against CD19 and CD123.83–85 Additionally, α-RV vectors utilize a more neutral integration site than both γ-RV and LV vectors and can be considered safer, because insertional oncogenesis is less likely to occur.86,87 Recently, we and others demonstrated the dependency of the resulting transduction efficiency on the pseudotyping as RD114-TR- or Baboon-pseudotyped lentiviruses transduced NK cells via the surface entry receptor ASCT2 that is upregulated following cytokine stimulation.88,89
In addition, different transduction enhancers have been tested to increase the viral transduction efficiency of NK cells (e.g., polybrene, Retronectin, protamine sulfate, poly-L-lysine, DEAE-dextran, Vectofusin-1). The cationic polymers protamine sulfate and polybrene reduce the viral repulsion of NK cells, thereby enhancing the membrane fusion.90 Interestingly, the routinely used transduction enhancer polybrene showed some toxicity in NK-92 cells.73 The use of poly-L-lysine and DEAE-dextran has only been reported in a few NK cells transduction studies.91 We and others showed enhanced viral transduction efficiency for CAR-NK cells with not only Retronectin,88,92 but also Vectofusion-1, which has been reported as a promising alternative transduction enhancer.93 Our data showed that Vectofusin-1 was as effective as Retronectin using VSV-G-pseudotyped LV, but demonstrated higher transduction efficiency for the RD114-TR-pseudotyped α-RV.88 The use of the relatively new transduction enhancer Vectofucion-1 in combination with specific envelope glycoproteins needs further attention as high transduction efficiencies were also reported with Baboon-pseudotyped LV.89
Promising new tools for nonviral modification
Despite the current success of viral vectors, GMP-grade vector production for clinical therapy is cost intensive and requires high safety standards. Hence, nonviral alternatives for CAR-NK cell production have gained increasing attention. Compared to classic viral-transduction, nonviral approaches are numerous and mostly based on transient transfection. CAR-NK cells can be successfully obtained by electroporation,94,95 nucleofection,96,97 lipofection,98,99 trogocytosis100 or nanoparticle treatment.101,102 Additionally, genome editing using clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9, zinc finger nucleases (ZFNs) or transcriptional activator-like effector nucleases (TALENs) represent suitable methods for NK cell engineering.103,104
Electroporation utilizes an electric field to temporarily permeabilize the cell membrane via short electrical pulses and allows charged molecules to move into the cell (e.g., DNA and RNA) to induce transient gene expression. Virus-free cell membrane penetration avoids the integration of foreign genetic material and therefore unwanted replication in the cellular genome which is a possible risk with viral infection. One of the first NK cell electroporation approaches reported by Liu et al. in the 1990s showed successful nonviral modification of the NK cell line YT.105 From then on, electroporation has increasingly been used for NK cell genetic modification, and impacts of different peptide nucleic acid sources were shown.106 Significantly higher expression of CD19-CARs in NK cell lines was achieved by using mRNA instead of cDNA with minimal effects on cell viability.106 Further studies achieved high transfection efficiencies in primary human NK cells,95,107,108 which might be explained by the fact that both resting and stimulated primary NK cells can be transfected.107
Alternatively, nucleofection can be applied, which is a modified electroporation-technique able to transfer a gene directly into the nucleus without the need for cell division.109,110 Similar to electroporation, nucleofection achieves higher transfection efficiencies with RNA than DNA.106 To date, few preclinical studies have successfully reported CAR-NK-92 or NK cell generation by nucleofection.111–113
A few approaches for nonviral genomic modification of NK cells have been reported in recent years, for example, using electroporation-based piggyBac transposon systems. A modified transposon can integrate a genetically engineered gene-of-interest (GOI) into the cellular genome by recognizing transposon-specific inverted terminal repeat sequences. For NK-92-cells, a clinically safe, nonviral, NKG2D-specific CAR-NK cell system could be engineered based on the piggyBac transposon, which showed effective purinergic reprogramming and enhanced cytotoxicity against CD73+ solid tumors.114 Different CARs were also nucleofected with the piggyBac or SleepingBeauty transposons into induced pluripotent stem cell (iPSC)-derived NK cells, demonstrating NK cell production efficiency that was similar to non-CAR-expressing iPSCs.115 In this context, the expression of NKG2D was not compromised in CAR-NK-iPSCs and the iPSC-NK cells showed a similar phenotype compared to PB-NK cells.
Regarding CD19-CAR modification, the reported electroporation- and RV-based transduction efficiencies of NK cells and the resulting cytotoxicities seemed comparable.108 Whereas high viral transduction rates of 60% could be achieved with α-RV, even higher efficiencies of 80–100% were reported for mRNA transfection with electroporation.83,107,116 Of note, nonviral modification is less costly and time-consuming compared to viral transduction. Therefore, nonviral methods seem to be a promising tool for GMP clinical application. For example, the first GMP-compliant process for mRNA electroporation has been presented for the expression of the chemokine receptor CCR7 and antibody-binding receptor CD16 in NK cells.116–118
CAR-NK cells for clinical application
Regarding the clinical application of engineered immune cells, CAR-T cell therapies have recently achieved striking success, mainly for hematological malignancies, although severe side effects such as cytokine release syndrome (CRS), neurotoxicity, GvHD and on-target/off-tumor effects have been reported in some cases (for review, see Hartmann et al.).119 In contrast, compared to T cells, NK cells possess a relatively short life span and do not undergo clonal expansion, which lowers the risk of in vivo side effects. A major advantage of CAR-NK cell therapy is its dual killing capacity, as the engineered NK cells can kill in a CAR-dependent manner via antigen-recognition, as well as in the inherent CAR-independent manner, including the aforementioned antigen-independent killing mechanisms, e.g., by release of cytotoxic granules or via the TRAIL-pathway. Therefore, one major tumor escape mechanism, involving the downregulation of tumor antigens on the cell surface, might be overcome by CAR-NK cell therapies.
However, while a multitude of clinical trials with CAR-T cells have been performed, studies of CAR-NK cells are quite rare. Currently, 19 trials of CAR-NK cells are listed on https://www.clinicaltrials.gov/, including phase I, phase I/II and early phase I studies (Table 1). Eleven studies are evaluating CAR-NK cells for the treatment of hematological malignancies, whereas nine trials for the treatment of solid tumors. Most of the trials are being conducted in Asia (15 total) while only three trials are ongoing in the US and only one trial is being performed in Europe (Figure 2b).
Table 1.
Overview of CAR-NK cell trials worldwide listed on https://www.clinicaltrials.gov/. Trials are classified based on the source of NK cells used, and then within these sub-clusters based on their geographical distribution. Trials targeting hematological malignancies are highlighted in light blue and CAR-NK cell trials against solid tumors are highlighted in light gray.
 |
Abbreviations: C – Completed, E – Enrolling by invitation, EP – electroporation, LV – Lentiviral, N – Not yet recruiting, PB – Peripheral Blood, S – Suspended, R – Recruiting, RV – retroviral, U – Unknown, W – Withdrawn.
Seven of these 19 trials are investigating engineered NK-92 cells. Four phase I/II clinical trials with CAR-NK-92 cells targeting CD7- or CD19-positive leukemia or lymphoma (NCT02742727 and NCT02892695), MUC1-positive advanced refractory or relapsed solid tumors (NCT02839954) or relapsed/refractory CD33-expressing AML (NCT02944162) are being conducted by PersonGen BioTherapeutics (Suzhou, China). These studies are using 3rd-generation CARs consisting of CD28 and 4–1BB costimulatory domains in addition to the intracellular CD3ζ signaling domain. Furthermore, the Xinxiang Medical University (Henan, China) is investigating chimeric costimulatory converting receptor (CCCR)-modified NK-92 cells in pretreated non-small cell lung carcinoma patients (NCT03656705) and the Asklepios Technology Company Group (Suzhou, China) is supporting a clinical phase I/II trial testing BCMA-CAR-NK-92 cells for treatment of multiple myeloma at the Wuxi Hospital in Jiangsu, China (NCT03940833). The only CAR-NK cell trial in Europe at the Goethe University in Frankfurt, Germany, is evaluating the safety of ErbB2-specific NK-92 cells expressing a second-generation CAR (scFv (FRP5)/CD28/CD3ζ) in patients with recurrent HER2-positive glioblastoma in a phase I study (NCT03383978), based on promising preclinical data.120
With regard to primary NK cells as the source for adoptive CAR-NK therapy, two trials at the M.D. Anderson Cancer Center (Texas, US) are employing UCB-derived NK cells as the source of CAR-NK cells for targeting CD19-expressing B-cell malignancies. In a first trial, a CD19-CD28-zeta-2A-iCasp9-IL15 CAR construct was used, which contained the inducible Caspase 9 as a suicide gene and a gene to produce IL-15 ectopically in addition to the CD28 costimulatory and CD3ζ signaling domains (NCT03056339). In a murine lymphoma model, it was shown that the inclusion of IL-15 dramatically increased the in vivo persistence and cytotoxic potential of anti-CD19 CAR-NK cells.76 Recently, the clinical results of the first 11 patients have been published, which reported eight patients (73%) showing a clinical response. Of these patients, seven (four with lymphoma and three with CLL) had complete remission, and one had remission of Richter’s transformation. Responses were rapid and occurred within 30 days after infusion for all dose levels. The infused CAR-NK cells expanded and persisted at low levels for at least 12 months.121 A second trial in humans aimed to test a second-generation CD28-zeta-CAR targeting CD19-expressing B cell malignancies (NCT03579927), but it was withdrawn due to lack of funding.
Another clinical trial in the US, at the St. Jude Children’s Research Hospital (Memphis), completed a study investigating CD19-redirected donor NK cells after expansion in the presence of irradiated K562 cells expressing membrane bound IL-15 and 4–1BB ligand (K562-mb15-41BBL) for the treatment of B-lineage acute lymphoblastic leukemia (B-ALL) (NCT00995137). In Asia, a multitude of highly interesting clinical CAR-NK cell trials have been initiated. One study at the National University Health System Singapore, expanded donor-derived NK cells via coculture with K562-mb15-41BBL in the presence of IL-2. The expanded, activated CD19-targeting NK cells were infused into B-ALL-patients with persistent disease (NCT01974479). In addition to their safety and feasibility, the lifespan and phenotype of these redirected NK cells were analyzed. CAR-NK cells were produced using mRNA electroporation and CAR-NK cells were detectable in the peripheral blood for 2–3 days post-infusion (personal communication by D. Campana; results not yet published). Furthermore, two trials are employing ROBO1-specific CAR-NK cells for the treatment of pancreatic cancer and other solid tumors (NCT03940820 in Suzhou, Jiangsu, and NCT03941457 in Shanghai, China).
In a pilot study performed at Guangzhou Medical University in China, mRNA electroporation is being used to transiently enhance the specificity of autologous or allogeneic NK cells against NKG2D-ligand-bearing tumors (NCT03415100). To date, three patients with metastatic colorectal cancer have been treated. The first two patients were treated with an intraperitoneal infusion of low doses of NKG2DL-CAR-NK cells, and they showed a reduced tumor cell number in ascites samples as well as reduced ascites generation. A third patient receiving an intraperitoneal infusion of CAR-NK cells following ultrasound-guided percutaneous injection showed a fast regression of the tumor sites in the liver and a complete metabolic response.122
In 2019, the Allife Medical Science and Technology Co. Ltd. (China) launched five early phase I studies to evaluate the safety and efficacy of CAR-NK cells targeting several tumor antigens; however, they are not yet recruiting. They are investigating anti-prostate-specific membrane antigen (PSMA) CAR-NK cells for the treatment of castration-resistant prostate cancer (NCT03692663) and anti-mesothelin CAR-NK cells for the treatment of epithelial ovarian cancer (NCT03692637). Furthermore, three different constructs will be investigated for the treatment of refractory B cell lymphoma targeting CD19 or CD22 alone, or both CD19 and CD22 together (NCT03690310, NCT03692767 and NCT03824964, respectively).
Perspective – what will the future bring for CAR-NK cell-based therapy?
The impressive success of CAR-T cell-therapeutic approaches for the treatment of B cell malignancies has generated great enthusiasm for the genetic engineering of other immune cells. As described in detail, NK cells are a highly promising effector cell population for fighting cancer, as they possess broad intrinsic killing potential and can be applied as an off-the-shelf medicinal product with a low risk of side effects. In addition, rapid technological innovations, such as increasing NK cell specific signaling, the use of split or adapter CAR systems and involvement of CRISPR/Cas9-based genome editing to counteract tumor immune suppression, will make it possible to either enhance NK cell persistence and/or inhibit immune suppressive mechanisms.
Beginning in 2020, we look forward for new insights from the first clinical CAR-NK cell trials launched worldwide.
Acknowledgments
We thank D. Campana and K. Rezvani for personal communication and discussion of yet unpublished data from their ongoing clinical trials and we apologize to all investigators whose work was not cited in this article due to space limitations. The laboratory of EU has been supported by the FCI/DKTK (to EU), by the DFG (CRC 1292/IRTG) (to EU, LMR, NA, and TB), by the Faculty of Medicine of the Goethe University, Frankfurt (to SM), by the “Alfred & Angelika Gutermuth-Stiftung” and the “Menschen für Kinder e.V.”.
Funding Statement
This work was supported by the Deutsche Forschungsgemeinschaft [DFG, SFB/CRC/IRTG1292].
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
References
- 1.Cooper MA, Fehniger TA, Caligiuri MA.. The biology of human natural killer-cell subsets. Trends Immunol. 2001;22(11):633–12. doi: 10.1016/S1471-4906(01)02060-9. [DOI] [PubMed] [Google Scholar]
- 2.Wang M, Windgassen D, Papoutsakis ET. Comparative analysis of transcriptional profiling of CD3+, CD4+ and CD8+ T cells identifies novel immune response players in T-cell activation. BMC Genomics. 2008;9(1):225. doi: 10.1186/1471-2164-9-225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kruse PH, Matta J, Ugolini S, Vivier E. Natural cytotoxicity receptors and their ligands. Immunol Cell Biol. 2014;92(3):221–229. doi: 10.1038/icb.2013.98. [DOI] [PubMed] [Google Scholar]
- 4.Smyth MJ, Cretney E, Kelly JM, Westwood JA, Street SEA, Yagita H, Takeda K, Dommelen SLHV, Degli-Esposti MA, Hayakawa Y, et al. Activation of NK cell cytotoxicity. Mol Immunol. 2005;42(4):501–510. doi: 10.1016/j.molimm.2004.07.034. [DOI] [PubMed] [Google Scholar]
- 5.Luevano M, Daryouzeh M, Alnabhan R, Querol S, Khakoo S, Madrigal A, Saudemont A. The unique profile of cord blood natural killer cells balances incomplete maturation and effective killing function upon activation. Hum Immunol. 2012;73(3):248–257. doi: 10.1016/j.humimm.2011.12.015. [DOI] [PubMed] [Google Scholar]
- 6.Tonn T, Becker S, Esser R, Schwabe D, Seifried E. Cellular immunotherapy of malignancies using the clonal natural killer cell line NK-92. J Hematother Stem Cell Res. 2001;10(4):535–544. doi: 10.1089/15258160152509145. [DOI] [PubMed] [Google Scholar]
- 7.Freud AG, Mundy-Bosse BL, Yu J, Caligiuri MA. The broad spectrum of human natural killer cell diversity. Immunity. 2017;47(5):820–833. doi: 10.1016/j.immuni.2017.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Freud AG, Caligiuri MA. Human natural killer cell development. Immunol Rev. 2006;214(1):56–72. doi: 10.1111/j.1600-065X.2006.00451.x. [DOI] [PubMed] [Google Scholar]
- 9.Lopez-Verges S, Milush JM, Pandey S, York VA, Arakawa-Hoyt J, Pircher H, Norris PJ, Nixon DF, Lanier LL. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+ NK-cell subset. Blood. 2010;116(19):3865–3874. doi: 10.1182/blood-2010-04-282301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iannello A, Debbeche O, Samarani S, Ahmad A. Antiviral NK cell responses in HIV infection: I. NK cell receptor genes as determinants of HIV resistance and progression to AIDS. J Leukoc Biol. 2008;84(1):1–26. doi: 10.1189/jlb.0907650. [DOI] [PubMed] [Google Scholar]
- 11.Grievink HW, Luisman T, Kluft C, Moerland M, Malone KE. Comparison of three isolation techniques for human peripheral blood mononuclear cells: cell recovery and viability, population composition, and cell functionality. Biopreserv Biobank. 2016;14(5):410–415. doi: 10.1089/bio.2015.0104. [DOI] [PubMed] [Google Scholar]
- 12.Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol. 2003;4(4):330–336. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 13.Pan Y, Tian T, Park CO, Lofftus SY, Mei S, Liu X, Luo C, O’Malley JT, Gehad A, Teague JE, et al. Survival of tissue-resident memory T cells requires exogenous lipid uptake and metabolism. Nature. 2017;543(7644):252–256. doi: 10.1038/nature21379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ribot JC, deBarros A, Pang DJ, Neves JF, Peperzak V, Roberts SJ, Girardi M, Borst J, Hayday AC, Pennington DJ, et al. CD27 is a thymic determinant of the balance between interferon-γ- and interleukin 17–producing γδ T cell subsets. Nat Immunol. 2009;10(4):427–436. doi: 10.1038/ni.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kriegsmann K, Kriegsmann M, von Bergwelt-baildon M, Cremer M, Witzens-Harig M. NKT cells – new players in CAR cell immunotherapy? Eur J Haematol. 2018;101(6):750–757. doi: 10.1111/ejh.13170. [DOI] [PubMed] [Google Scholar]
- 16.Decaup E, Rossi C, Gravelle P, Laurent C, Bordenave J, Tosolini M, Tourette A, Perrial E, Dumontet C, Poupot M, et al. A tridimensional model for NK cell-mediated ADCC of follicular lymphoma. Front Immunol. 2019;10:1943. doi: 10.3389/fimmu.2019.01943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Freeman AJ, Vervoort SJ, Ramsbottom KM, Kelly MJ, Michie J, Pijpers L, Johnstone RW, Kearney CJ, Oliaro J. Natural killer cells suppress T cell-associated tumor immune evasion. Cell Rep. 2019;28(11):2784–94 e5. doi: 10.1016/j.celrep.2019.08.017. [DOI] [PubMed] [Google Scholar]
- 18.Mehta RS, Rezvani K. Chimeric antigen receptor expressing natural killer cells for the immunotherapy of cancer. Front Immunol. 2018;9:283. doi: 10.3389/fimmu.2018.00283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Iwaszko M, Bogunia-Kubik K. Clinical significance of the HLA-E and CD94/NKG2 interaction. Arch Immunol Ther Exp (Warsz). 2011;59(5):353–367. doi: 10.1007/s00005-011-0137-y. [DOI] [PubMed] [Google Scholar]
- 20.Smyth MJ, Cretney E, Kelly JM, Westwood JA, Shayna E A Street EA, Yagita H, Takeda K, van Dommelen LH, Degli-Esposti MA, Hayakawa Y. Activation of NK cell cytotoxicity. Mol Immunol. 2005;42(4):501–510. doi: 10.1016/j.molimm.2004.07.034. [DOI] [PubMed] [Google Scholar]
- 21.Algarra I, Garc�a-Lora A, Cabrera T, Ruiz-Cabello F, Garrido F. The selection of tumor variants with altered expression of classical and nonclassical MHC class I molecules: implications for tumor immune escape. Cancer Immunol Immunother. 2010;17(10):147–154. doi: 10.1007/s00262-004-0517-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bix M, Liao NS, Zijlstra M, Loring J, Jaenisch R, Raulet D. Rejection of class I MHC-deficient haemopoietic cells by irradiated MHC-matched mice. Nature. 1991;349(6307):329–331. doi: 10.1038/349329a0. [DOI] [PubMed] [Google Scholar]
- 23.Costello RT, Gastaut JA, Olive D. Tumor escape from immune surveillance. Arch Immunol Ther Exp (Warsz). 1999;47:83–88. [PubMed] [Google Scholar]
- 24.Kumar V, McNerney ME. A new self: MHC-class-I-independent Natural-killer-cell self-tolerance. Nat Rev Immunol. 2005;5(5):363–374. doi: 10.1038/nri1603. [DOI] [PubMed] [Google Scholar]
- 25.Ullrich E, Koch J, Cerwenka A, Steinle A. New prospects on the NKG2D/NKG2DL system for oncology. Oncoimmunology. 2013;2(10):e26097. doi: 10.4161/onci.26097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Arnon TI, Lev M, Katz G, Chernobrov Y, Porgador A, Mandelboim O. Recognition of viral hemagglutinins by NKp44 but not by NKp30. Eur J Immunol. 2001;31(9):2680–2689. doi:. [DOI] [PubMed] [Google Scholar]
- 27.Lakshmikanth T, Burke S, Ali TH, Kimpfler S, Ursini F, Ruggeri L, Capanni M, Umansky V, Paschen A, Sucker A, et al. NCRs and DNAM-1 mediate NK cell recognition and lysis of human and mouse melanoma cell lines in vitro and in vivo. J Clin Invest. 2009;119(5):1251–1263. doi: 10.1172/JCI36022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Raulet DH, Vance RE. Self-tolerance of natural killer cells. Nat Rev Immunol. 2006;6(7):520–531. doi: 10.1038/nri1863. [DOI] [PubMed] [Google Scholar]
- 29.Brodin P, Kärre K, Höglund P. NK cell education: not an on-off switch but a tunable rheostat. Trends Immunol. 2009;30(4):143–149. doi: 10.1016/j.it.2009.01.006. [DOI] [PubMed] [Google Scholar]
- 30.Pradeu T, Vivier E. The discontinuity theory of immunity. Sci Immunol. 2016;1(1):AAG0479. doi: 10.1126/sciimmunol.aag0479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bengsch B, Ohtani T, Herati RS, Bovenschen N, Chang KM, Wherry EJ. Deep immune profiling by mass cytometry links human T and NK cell differentiation and cytotoxic molecule expression patterns. J Immunol Methods. 2018;453:3–10. doi: 10.1016/j.jim.2017.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cheng M, Zhang J, Jiang W, Chen Y, Tian Z. Natural killer cell lines in tumor immunotherapy. Front Med. 2012;6(1):56–66. doi: 10.1007/s11684-012-0177-7. [DOI] [PubMed] [Google Scholar]
- 33.Tang X, Yang L, Li Z, Nalin AP, Dai H, Xu T, Yin J, You F, Zhu M, Shen W, et al. First-in-man clinical trial of CAR NK-92 cells: safety test of CD33-CAR NK-92 cells in patients with relapsed and refractory acute myeloid leukemia. Am J Cancer Res. 2018;8(6):1083–1089. [PMC free article] [PubMed] [Google Scholar]
- 34.Fang F, Xiao W, Tian Z. NK cell-based immunotherapy for cancer. Semin Immunol. 2017;31:37–54. doi: 10.1016/j.smim.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 35.Gong JH, Maki G, Klingemann HG. Characterization of a human cell line (NK-92) with phenotypical and functional characteristics of activated natural killer cells. Leukemia. 1994;8:652–658. [PubMed] [Google Scholar]
- 36.Klingemann H, Boissel L, Toneguzzo F. Natural killer cells for immunotherapy – advantages of the NK-92 cell line over blood NK cells. Front Immunol. 2016;7:91. doi: 10.3389/fimmu.2016.00091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Antony GK, Dudek AZ. Interleukin 2 in cancer therapy. Curr Med Chem. 2010;17(29):3297–3302. doi: 10.2174/092986710793176410. [DOI] [PubMed] [Google Scholar]
- 38.Nham T, Poznanski SM, Fan IY, Vahedi F, Shenouda MM, Lee AJ, Chew MV, Hogg RT, Lee DA, Ashkar AA, et al. Ex vivo-expanded natural killer cells derived from long-term cryopreserved cord blood are cytotoxic against primary breast cancer cells. J Immunother. 2018;41(2):64–72. doi: 10.1097/CJI.0000000000000192. [DOI] [PubMed] [Google Scholar]
- 39.Spanholtz J, Tordoir M, Eissens D, Preijers F, van der Meer A, Joosten I, Schaap N, de Witte TM, Dolstra H, et al. High log-scale expansion of functional human natural killer cells from umbilical cord blood CD34-positive cells for adoptive cancer immunotherapy. PLoS One. 2010;5(2):e9221. doi: 10.1371/journal.pone.0009221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mace EM, Orange JS. Genetic causes of human NK cell deficiency and their effect on NK cell subsets. Front Immunol. 2016;7. doi: 10.3389/fimmu.2016.00545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ruggeri L, Capanni M, Urbani E, Perruccio K, Shlomchik WD, Tosti A, Posati S, Rogaia D, Frassoni F, Aversa F, Martelli MF, Velardi A. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science. 2002;295(5562):2097. PMID: 11896281. doi: 10.1126/science.1068440. [DOI] [PubMed] [Google Scholar]
- 42.Terme M, Ullrich E, Delahaye NF, Chaput N, Zitvogel L. Natural killer cell-directed therapies: moving from unexpected results to successful strategies. Nat Immunol. 2008;9:486–494. doi: 10.1038/ni1580. [DOI] [PubMed] [Google Scholar]
- 43.Rosenberg SA. Immunotherapy of cancer by systemic administration of lymphoid cells plus interleukin-2. J Biol Response Mod. 1984;3:501–511. [PubMed] [Google Scholar]
- 44.Ghiringhelli F, Menard C, Terme M, Flament C, Taieb J, Chaput N, Puig PE, Novault S, Escudier B, Vivier E, et al. CD4+CD25+ regulatory T cells inhibit natural killer cell functions in a transforming growth factor-beta-dependent manner. J Exp Med. 2005;202(8):1075–1085. doi: 10.1084/jem.20051511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Granzin M, Wagner J, Köhl U, Cerwenka A, Huppert V, Ullrich E. Shaping of natural killer cell antitumor activity by ex vivo cultivation. Front Immunol. 2017;8. doi: 10.3389/fimmu.2017.00458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Romee R, Cooley S, Berrien-Elliott MM, Westervelt P, Verneris MR, Wagner JE, Weisdorf DJ, Blazar BR, Ustun C, DeFor TE, et al. First-in-human phase 1 clinical study of the IL-15 superagonist complex ALT-803 to treat relapse after transplantation. Blood. 2018;131(23):2515–2527. doi: 10.1182/blood-2017-12-823757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Roda JM, Joshi T, Butchar JP, McAlees JW, Lehman A, Tridandapani S, Carson WE. The activation of natural killer cell effector functions by cetuximab-coated, epidermal growth factor receptor–positive tumor cells is enhanced by cytokines. Clin Cancer Res. 2007;13(21):6419. doi: 10.1158/1078-0432.CCR-07-0865. [DOI] [PubMed] [Google Scholar]
- 48.Beano A, Signorino E, Evangelista A, Brusa D, Mistrangelo M, Polimeni M, Spadi R, Donadio M, Ciuffreda L, Matera L, et al. Correlation between NK function and response to trastuzumab in metastatic breast cancer patients. J Transl Med. 2008;6(1):25. doi: 10.1186/1479-5876-6-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wu L, Adams M, Carter T, Chen R, Muller G, Stirling D, Schafer P, Bartlett JB. Lenalidomide enhances natural killer cell and monocyte-mediated antibody-dependent cellular cytotoxicity of rituximab-treated CD20+ tumor cells. Clin Cancer Res. 2008;14(14):4650. doi: 10.1158/1078-0432.CCR-07-4405. [DOI] [PubMed] [Google Scholar]
- 50.Felices M, Lenvik TR, Davis ZB, Miller JS, Vallera DA. Generation of BiKEs and TriKEs to improve NK cell-mediated targeting of tumor cells. Methods Mol Biol. 2016;1441:333–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Benson DM Jr., Bakan CE, Mishra A, Hofmeister CC, Efebera Y, Becknell B, Baiocchi RA, Zhang J, Yu J, Smith MK, et al. The PD-1/PD-L1 axis modulates the natural killer cell versus multiple myeloma effect: a therapeutic target for CT-011, a novel monoclonal anti-PD-1 antibody. Blood. 2010;116(13):2286–2294. doi: 10.1182/blood-2010-02-271874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Khan M, Arooj S, Wang H. NK cell-based immune checkpoint inhibition. Front Immunol. 2020;11:167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Parkhurst MR, Riley JP, Dudley ME, Rosenberg SA. Adoptive transfer of autologous natural killer cells leads to high levels of circulating natural killer cells but does not mediate tumor regression. Clin Cancer Res. 2011;17(19):6287–6297. doi: 10.1158/1078-0432.CCR-11-1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sakamoto N, Ishikawa T, Kokura S, Okayama T, Oka K, Ideno M, Sakai F, Kato A, Tanabe M, Enoki T. Phase I clinical trial of autologous NK cell therapy using novel expansion method in patients with advanced digestive cancer. J Transl Med. 2015;13(1):277. doi: 10.1186/s12967-015-0632-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Miller JS, Soignier Y, Panoskaltsis-Mortari A, McNearney SA, Yun GH, Fautsch SK, McKenna D, Le C, Defor TE, Burns LJ, et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood. 2005;105(8):3051–3057. doi: 10.1182/blood-2004-07-2974. [DOI] [PubMed] [Google Scholar]
- 56.Rubnitz JE, Inaba H, Ribeiro RC, Jeffrey E. Rubnitz , Hiroto Inaba , Raul C. Ribeiro , Pounds S , Rooney B , BellChing-Hon Pui T , Leung W. NKAML: a pilot study to determine the safety and feasibility of haploidentical natural killer cell transplantation in childhood acute myeloid leukemia. J Clin Oncol. 2010;28(6):955–959. doi: 10.1200/JCO.2009.24.4590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shaffer BC, Le Luduec JB, Forlenza C, Jakubowski AA, Perales M-A, Young JW, Hsu KC. Phase II study of haploidentical natural killer cell infusion for treatment of relapsed or persistent myeloid malignancies following allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2016;22(4):705–709. doi: 10.1016/j.bbmt.2015.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shah NN, Baird K, Delbrook CP, Fleisher TA, Kohler ME, Rampertaap S, Lemberg K, Hurley CK, Kleiner DE, Merchant MS, et al. Acute GVHD in patients receiving IL-15/4-1BBL activated NK cells following T-cell–depleted stem cell transplantation. Blood. 2015;125(5):784–792. doi: 10.1182/blood-2014-07-592881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lupo KB, Matosevic S. Natural killer cells as allogeneic effectors in adoptive cancer immunotherapy. Cancers (Basel). 2019;11(6):769. doi: 10.3390/cancers11060769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Romee R, Rosario M, Berrien-Elliott MM, Wagner JA, Jewell BA, Schappe T, Leong JW, Abdel-Latif S, Schneider SE, Willey S, et al. Cytokine-induced memory-like natural killer cells exhibit enhanced responses against myeloid leukemia. Sci Transl Med. 2016;8(357):357ra123. doi: 10.1126/scitranslmed.aaf2341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Williams BA, Law AD, Routy B, denHollander N, Gupta V, Wang X-H, Chaboureau A, Viswanathan S, Keating A. A phase I trial of NK-92 cells for refractory hematological malignancies relapsing after autologous hematopoietic cell transplantation shows safety and evidence of efficacy. Oncotarget. 2017;8(51):89256–89268. doi: 10.18632/oncotarget.19204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ciurea SO, Schafer JR, Bassett R, Denman CJ, Cao K, Willis D, Rondon G, Chen J, Soebbing D, Kaur I, et al. Phase 1 clinical trial using mbIL21 ex vivo–expanded donor-derived NK cells after haploidentical transplantation. Blood. 2017;130(16):1857–1868. doi: 10.1182/blood-2017-05-785659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lim WA, June CH. The principles of engineering immune cells to treat cancer. Cell. 2017;168(4):724–740. doi: 10.1016/j.cell.2017.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.You F, Wang Y, Jiang L, Zhu X, Chen D, Yuan L, An G, Meng H, Yang L. A novel CD7 chimeric antigen receptor-modified NK-92MI cell line targeting T-cell acute lymphoblastic leukemia. Am J Cancer Res. 2019;9(1):64–78. eCollection 2019. PMID: 30755812 PMCID: PMC6356925. [PMC free article] [PubMed] [Google Scholar]
- 65.Duan H, Huang H, Jing G. An antibody fab fragment-based chimeric antigen receptor could efficiently eliminate human thyroid cancer cells. J Cancer. 2019;10(8):1890–1895. doi: 10.7150/jca.30163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Balakrishnan A, Rajan A, Salter AI, Kosasih PL, Wu Q, Voutsinas J, Jensen MC, Plückthun A, Riddell SR. Multispecific targeting with synthetic ankyrin repeat motif chimeric antigen receptors. Clin Cancer Res. 2019;25(24):7506. doi: 10.1158/1078-0432.CCR-19-1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chmielewski M, Hombach AA, Abken H. Of CARs and TRUCKs: chimeric antigen receptor (CAR) T cells engineered with an inducible cytokine to modulate the tumor stroma. Immunol Rev. 2014;257:83–90. doi: 10.1111/imr.12125. [DOI] [PubMed] [Google Scholar]
- 68.Grada Z, Hegde M, Byrd T, Shaffer DR, Ghazi A, Brawley VS, Corder A, Schönfeld K, Koch J, Dotti G, et al. TanCAR: a novel bispecific chimeric antigen receptor for cancer immunotherapy. Mol Ther Nucleic Acids. 2013;2:e105. doi: 10.1038/mtna.2013.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Roybal KT, Rupp LJ, Morsut L, Walker W, McNally K, Park J, Lim W. Precision tumor recognition by T cells with combinatorial antigen-sensing circuits. Cell. 2016;164(4):770–779. doi: 10.1016/j.cell.2016.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Fedorov VD, Themeli M, Sadelain M. PD-1– and CTLA-4–based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci Transl Med. 2013;5(215):215ra172. doi: 10.1126/scitranslmed.3006597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Grote S, Chan C-H, Baden C, Huber SM, Eckert F, Mittelstaet J, Kaiser A, Seitz C, Schlegel P, Handgretinger R and Schleicher S. Abstract B70: universal adapter CAR-engineered NK-92 cells target patient-derived glioblastoma cancer stem cells. Cancer Immunol Res. 2020;8:B70.doi: 10.1158/2326-6074.TUMIMM19-B70. Published March 2020 [Google Scholar]
- 72.Seitz CM, Kieble V, Illi C, Reiter S, Grote S, Mittelstaet J, Lock D, Kaiser A, Schleicher S, Handgretinger R, et al. Combinatorial targeting of multiple shared antigens by adapter-CAR-T Cells (aCAR-Ts) allows target cell discrimination and specific lysis based on differential expression profiles. Blood. 2018;132(Supplement 1):4543. doi: 10.1182/blood-2018-99-115630. [DOI] [Google Scholar]
- 73.Matosevic S. Viral and nonviral engineering of natural killer cells as emerging adoptive cancer immunotherapies. J Immunol Res. 2018;2018:4054815. doi: 10.1155/2018/4054815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chang YH, Connolly J, Shimasaki N, Mimura K, Kono K, Campana D. A chimeric receptor with NKG2D specificity enhances natural killer cell activation and killing of tumor cells. Cancer Res. 2013;73(6):1777–1786. doi:10.1158/0008-5472.CAN-12-3558. [DOI] [PubMed] [Google Scholar]
- 75.Imai C, Iwamoto S, Campana D. Genetic modification of primary natural killer cells overcomes inhibitory signals and induces specific killing of leukemic cells. Blood. 2005;106(1):376–383. doi: 10.1182/blood-2004-12-4797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Liu E, Tong Y, Dotti G, Shaim H, Savoldo B, Mukherjee M, Orange J, Wan X, Lu X, Reynolds A, et al. Cord blood NK cells engineered to express IL-15 and a CD19-targeted CAR show long-term persistence and potent antitumor activity. Leukemia. 2018;32(2):520–531. doi: 10.1038/leu.2017.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Hacein-Bey-Abina S, Garrigue A, Wang GP, Soulier J, Lim A, Morillon E, Clappier E, Caccavelli L, Delabesse E, Beldjord K, et al. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J Clin Invest. 2008;118(9):3132–3142. doi: 10.1172/JCI35700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hacein-Bey-Abina S, Von Kalle C, Schmidt M, S Hacein-Bey-Abina1, C Von Kalle, M Schmidt, McCormack MP, Wulffraat N, Leboulch P, Lim A, Osborne CS, Pawliuk R, E Morillon E, &, Cavazzana-Calvo M. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science. 2003;302(5644):415–419. PMID: 14564000 . doi: 10.1126/science.1088547. [DOI] [PubMed] [Google Scholar]
- 79.Modlich U, Navarro S, Zychlinski D, Maetzig T, Knoess S, Brugman MH, Schambach A, Charrier S, Galy A, Thrasher AJ, et al. Insertional transformation of hematopoietic cells by self-inactivating lentiviral and gammaretroviral vectors. Mol Ther. 2009;17(11):1919–1928. doi: 10.1038/mt.2009.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Montini E, Cesana D, Schmidt M, Sanvito F, Bartholomae CC, Ranzani M, Benedicenti F, Sergi LS, Ambrosi A, Ponzoni M, et al. The genotoxic potential of retroviral vectors is strongly modulated by vector design and integration site selection in a mouse model of HSC gene therapy. J Clin Invest. 2009;119(4):964–975. doi: 10.1172/JCI37630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rezvani K, Rouce R, Liu E, Shpall E. Engineering natural killer cells for cancer immunotherapy. Mol Ther. 2017;25(8):1769–1781. doi: 10.1016/j.ymthe.2017.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Boissel L, Betancur M, Lu W, Wels WS, Marino T, Van Etten RA, Klingemann H. Comparison of mRNA and lentiviral based transfection of natural killer cells with chimeric antigen receptors recognizing lymphoid antigens. Leuk Lymphoma. 2012;53(5):958–965. doi: 10.3109/10428194.2011.634048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Suerth JD, Morgan MA, Kloess S, Heckl D, Neudörfl C, Falk CS, Koehl U, Schambach A. Efficient generation of gene-modified human natural killer cells via alpharetroviral vectors. J Mol Med (Berl). 2016;94(1):83–93. doi: 10.1007/s00109-015-1327-6. [DOI] [PubMed] [Google Scholar]
- 84.Kloss S, Oberschmidt O, Morgan M, Dahlke J, Arseniev L, Huppert V, Granzin M, Gardlowski T, Matthies N, Soltenborn S, et al. Optimization of human NK cell manufacturing: fully automated separation, improved ex vivo expansion using IL-21 with autologous feeder cells, and generation of anti-CD123-CAR-expressing effector cells. Hum Gene Ther. 2017;28(10):897–913. doi: 10.1089/hum.2017.157. [DOI] [PubMed] [Google Scholar]
- 85.Oberschmidt O, Morgan M, Huppert V, Kessler J, Gardlowski T, Matthies N, Aleksandrova K, Arseniev L, Schambach A, Koehl U, et al. Development of automated separation, expansion, and quality control protocols for clinical-scale manufacturing of primary human NK cells and alpharetroviral chimeric antigen receptor engineering. Hum Gene Ther Methods. 2019;30(3):102–120. doi: 10.1089/hgtb.2019.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kaufmann KB, Brendel C, Suerth JD, Mueller-Kuller U, Chen-Wichmann L, Schwäble J, Pahujani S, Kunkel H, Schambach A, Baum C, et al. Alpharetroviral vector-mediated gene therapy for X-CGD: functional correction and lack of aberrant splicing. Mol Ther. 2013;21(3):648–661. doi: 10.1038/mt.2012.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Suerth JD, Maetzig T, Brugman MH, Heinz N, Appelt J-U, Kaufmann KB, Schmidt M, Grez M, Modlich U, Baum C, et al. Alpharetroviral self-inactivating vectors: long-term transgene expression in murine hematopoietic cells and low genotoxicity. Mol Ther. 2012;20(5):1022–1032. doi: 10.1038/mt.2011.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Müller S, Bexte T, Gebel V, Kalensee F, Stolzenberg E, Hartmann J, Koehl U, Schambach A, Wels WS, Modlich U, et al. High cytotoxic efficiency of lentivirally and alpharetrovirally engineered CD19-specific chimeric antigen receptor natural killer cells against acute lymphoblastic leukemia. Front Immunol. 2020;10. doi: 10.3389/fimmu.2019.03123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Bari R, Granzin M, Tsang KS, Roy A, Krueger W, Orentas R, Schneider D, Pfeifer R, Moeker N, Verhoeyen E, Dropulic B and Leung W. A distinct subset of highly proliferative and lentiviral vector (LV)-Transducible NK cells define a readily engineered subset for adoptive cellular therapy. Front Immunol. 2019;10. doi:10.3389/fimmu.2019.02001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Campbell KS, Dessing M, Lopez-Botet M, Cella M, Colonna M. Tyrosine phosphorylation of a human killer inhibitory receptor recruits protein tyrosine phosphatase 1C. J Exp Med. 1996;184(1):93–100. doi: 10.1084/jem.184.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Swaney WP, Sorgi FL, Bahnson AB, Barranger JA. The effect of cationic liposome pretreatment and centrifugation on retrovirus-mediated gene transfer. Gene Ther. 1997;4(12):1379–1386. doi: 10.1038/sj.gt.3300529. [DOI] [PubMed] [Google Scholar]
- 92.Lowe E, Truscott LC, De Oliveira SN. In vitro generation of human NK cells expressing chimeric antigen receptor through differentiation of gene-modified hematopoietic stem cells. In: Somanchi SS, editor. Natural killer cells: methods and protocols. New York: Springer New York; 2016. p. 241–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fenard D, Ingrao D, Seye A, Buisset J, Genries S, Martin S, Kichler A, Galy A. Vectofusin-1, a new viral entry enhancer, strongly promotes lentiviral transduction of human hematopoietic stem cells. Mol Ther Nucleic Acids. 2013;2:e90. doi: 10.1038/mtna.2013.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Chang Y-H, Connolly J, Shimasaki N, Mimura K, Kono K, Campana D. A chimeric receptor with NKG2D specificity enhances natural killer cell activation and killing of tumor cells. Cancer Res. 2013;73(6):1777. doi:10.1158/0008-5472.CAN-12-3558. [DOI] [PubMed] [Google Scholar]
- 95.Jiang H, Zhang W, Shang P, Zhang H, Fu W, Ye F, Zeng T, Huang H, Zhang X, Sun W, et al. Transfection of chimeric anti-CD138 gene enhances natural killer cell activation and killing of multiple myeloma cells. Mol Oncol. 2014;8(2):297–310. doi: 10.1016/j.molonc.2013.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Phatarpekar PV, Lee DA, Somanchi SS. Electroporation of siRNA to silence gene expression in primary NK cells. In: Somanchi SS, editor. Natural killer cells: methods and protocols. New York: Springer New York; 2016. p. 267–276. [DOI] [PubMed] [Google Scholar]
- 97.Maasho K, Marusina A, Reynolds NM, Coligan JE, Borrego F. Efficient gene transfer into the human natural killer cell line, NKL, using the Amaxa nucleofection system™. J Immunol Methods. 2004;284(1–2):133–140. doi: 10.1016/j.jim.2003.10.010. [DOI] [PubMed] [Google Scholar]
- 98.El Sobky SA, El-Ekiaby NM, Mekky RY, Elemam NM, Mohey Eldin MA, El-sayed M, Esmat G, Abdelaziz AI. Contradicting roles of miR-182 in both NK cells and their host target hepatocytes in HCV. Immunol Lett. 2016;169:52–60. doi: 10.1016/j.imlet.2015.10.013. [DOI] [PubMed] [Google Scholar]
- 99.Zhang J, Sun R, Wei H, Zhang J, Tian Z. Characterization of stem cell factor gene-modified human natural killer cell line, NK-92 cells: implication in NK cell-based adoptive cellular immunotherapy. Oncol Rep. 2004;11:1097–1106. [PubMed] [Google Scholar]
- 100.Somanchi SS, Somanchi A, Cooper LJN, Lee DA. Engineering lymph node homing of ex vivo-expanded human natural killer cells via trogocytosis of the chemokine receptor CCR7. Blood. 2012;119(22):5164–5172. doi: 10.1182/blood-2011-11-389924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Nakamura T, Kuroi M, Fujiwara Y, Warashina S, Sato Y, Harashima H. Small-sized, stable lipid nanoparticle for the efficient delivery of siRNA to human immune cell lines. Sci Rep. 2016;6(1):37849. doi: 10.1038/srep37849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Oyer JL, Pandey V, Igarashi RY, Somanchi SS, Zakari A, Solh M, Lee DA, Altomare DA, Copik AJ. Natural killer cells stimulated with PM21 particles expand and biodistribute in vivo: clinical implications for cancer treatment. Cytotherapy. 2016;18(5):653–663. doi: 10.1016/j.jcyt.2016.02.006. [DOI] [PubMed] [Google Scholar]
- 103.Daher M, Basar R, Shaim H, Gokdemir, E, Uprety, N, Kontoyiannis, A, Carolina Mendt M, Imahashi N.... & Rezvani K The TGF-β/SMAD signaling pathway as a mediator of NK cell dysfunction and immune evasion in myelodysplastic syndrome. Blood. 2017;130 (Supplement 1): 53.https://doi.org/10.1182/blood.V130.Suppl_1.53.53. [Google Scholar]
- 104.Dong G, Li Y, Jiang B, Yee J-K, Lee DA, McKeithan TW, Chan WC. Generation of natural killer cell lymphoma models in vitro by gene editing. Blood. 2016;128(22):2724. doi: 10.1182/blood.V128.22.2724.2724. [DOI] [Google Scholar]
- 105.Liu JH, Wei S, Blanchard DK, Djeu JY. Restoration of lytic function in a human natural killer cell line by gene transfection. Cell Immunol. 1994;156(1):24–35. doi: 10.1006/cimm.1994.1150. [DOI] [PubMed] [Google Scholar]
- 106.Boissel L, Betancur M, Wels WS, Tuncer H, Klingemann H. Transfection with mRNA for CD19 specific chimeric antigen receptor restores NK cell mediated killing of CLL cells. Leuk Res. 2009;33(9):1255–1259. doi: 10.1016/j.leukres.2008.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Li L, Liu LN, Feller S, Allen C, Shivakumar R, Fratantoni J, Wolfraim LA, Fujisaki H, Campana D, Chopas N, et al. Expression of chimeric antigen receptors in natural killer cells with a regulatory-compliant non-viral method. Cancer Gene Ther. 2010;17(3):147–154. doi: 10.1038/cgt.2009.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Shimasaki N, Fujisaki H, Cho D, Masselli M, Lockey T, Eldridge P, Leung W, Campana D. A clinically adaptable method to enhance the cytotoxicity of natural killer cells against B-cell malignancies. Cytotherapy. 2012;14(7):830–840. doi: 10.3109/14653249.2012.671519. [DOI] [PubMed] [Google Scholar]
- 109.Trompeter H-I, Weinhold S, Thiel C, Wernet P, Uhrberg M. Rapid and highly efficient gene transfer into natural killer cells by nucleofection. J Immunol Methods. 2003;274(1–2):245–256. doi: 10.1016/S0022-1759(02)00431-3. [DOI] [PubMed] [Google Scholar]
- 110.Somanchi SS, Denman CJ, Amritkar A, Senyukov V, Olivares S, Cooper LJN, Lee DA. Gene modification of human primary natural killer cells by electroporation with mRNA or DNA coding for an anti-GD2 chimeric antigen receptor. Blood. 2009;114(22):2467. doi: 10.1182/blood.V114.22.2467.2467.19608751 [DOI] [Google Scholar]
- 111.Elmacken M, Awasthi A, Ayello J, van de Ven C, Luo W, Liao Y, Riddell S, Cairo MS. Neuroblastoma and ewing’s sarcoma associated with ROR1 expression can be effectively targeted with NK cells modified to express an anti ROR1 chimeric antigen receptor. Biol Blood Marrow Transplant. 2015;21(2):S95–S7. doi: 10.1016/j.bbmt.2014.11.117. [DOI] [Google Scholar]
- 112.Liu H, Yang B, Sun T, Lin L, Hu Y, Deng M, Yang J, Liu T, Li J, Sun S, et al. Specific growth inhibition of ErbB2expressing human breast cancer cells by genetically modified NK92 cells. Oncol Rep. 2015;33(1):95–102. doi: 10.3892/or.2014.3548. [DOI] [PubMed] [Google Scholar]
- 113.Chu Y, Hochberg J, Yahr A, Ayello J, van de Ven C, Barth M, Czuczman M, Cairo MS. Targeting CD20+ aggressive b-cell non-hodgkin lymphoma by anti-CD20 CAR mRNA-modified expanded natural killer cells in vitro and in NSG mice. Cancer Immunol Res. 2015;3(4):333–344. doi: 10.1158/2326-6066.CIR-14-0114. [DOI] [PubMed] [Google Scholar]
- 114.Wang J, Lupo KB, Chambers AM, Matosevic S. Purinergic targeting enhances immunotherapy of CD73+ solid tumors with piggyBac-engineered chimeric antigen receptor natural killer cells. J ImmunoTher Cancer. 2018;6(1):136. doi: 10.1186/s40425-018-0441-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Li Y, Hermanson DL, Moriarity BS, Kaufman DS. Human iPSC-derived natural killer cells engineered with chimeric antigen receptors enhance anti-tumor activity. Cell Stem Cell. 2018;23(2):181–92.e5. doi: 10.1016/j.stem.2018.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Carlsten M, Levy E, Karambelkar A, Li L, Reger R, Berg M, Peshwa MV, Childs RW. Efficient mRNA-based genetic engineering of human NK cells with high-affinity CD16 and CCR7 augments rituximab-induced ADCC against lymphoma and targets NK cell migration toward the lymph node-associated chemokine CCL19. Front Immunol. 2016;7:105. doi: 10.3389/fimmu.2016.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Levy ER, Carlsten M, Childs RW. mRNA transfection to improve NK cell homing to tumors. In: Somanchi SS, editor. Natural killer cells: methods and protocols. New York: Springer New York; 2016. p. 231–240. [DOI] [PubMed] [Google Scholar]
- 118.Levy ER, Carlsten M, Childs RW. mRNA transfection to improve NK cell homing to tumors. Methods Mol Biol. 2016;1441:231–240. [DOI] [PubMed] [Google Scholar]
- 119.Hartmann J, Schussler-Lenz M, Bondanza A, Buchholz CJ. Clinical development of CAR T cells-challenges and opportunities in translating innovative treatment concepts. EMBO Mol Med. 2017;9(9):1183–1197. doi: 10.15252/emmm.201607485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Schonfeld K, Sahm C, Zhang C, Naundorf S, Brendel C, Odendahl M, Nowakowska P, Bönig H, Köhl U, Kloess S, et al. Selective inhibition of tumor growth by clonal NK cells expressing an ErbB2/HER2-specific chimeric antigen receptor. Mol Ther. 2015;23(2):330–338. doi: 10.1038/mt.2014.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Liu E, Marin D, Banerjee P, Macapinlac HA, Thompson P, Basar R, Nassif Kerbauy L, Overman B, Thall P, Kaplan M, et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N Engl J Med. 2020;382(6):545–553. doi: 10.1056/NEJMoa1910607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Xiao L, Cen D, Gan H, Sun Y, Huang N, Xiong H, Jin Q, Su L, Liu X, Wang K, et al. Adoptive transfer of NKG2D CAR mRNA-engineered natural killer cells in colorectal cancer patients. Mol Ther. 2019;27(6):1114–1125. doi: 10.1016/j.ymthe.2019.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]


