Abstract
COVID-19, a new pandemic caused by SARS-CoV-2, was first identified in 2019 in Wuhan, China. The novel corona virus SARS-CoV-2 and the 2002 SARS-CoV have 74% identity and use similar mechanisms to gain entry into the cell. Both the viruses enter the host cell by binding of the viral spike glycoprotein to the host receptor, angiotensin converting enzyme 2 (ACE2). Targeting entry of the virus has a better advantage than inhibiting the later stages of the viral life cycle. The crystal structure of the SARS-CoV (6CRV: full length S protein) and SARS-CoV-2 Spike proteins (6M0J: Receptor binding domain, RBD) was used to determine potential small molecule inhibitors. Curcumin, a naturally occurring phytochemical in Curcuma longa, is known to have broad pharmacological properties. In the present study, curcumin and its derivatives were docked, using Autodock 4.2, onto the 6CRV and 6M0J to study their capability to act as inhibitors of the spike protein and thereby, viral entry. The curcumin and its derivatives displayed binding energies, ΔG, ranging from −10.98 to −5.12 kcal/mol (6CRV) and −10.01 to −5.33 kcal/mol (6M0J). The least binding energy was seen in bis-demethoxycurcumin with: ΔG = −10.98 kcal/mol (6CRV) and −10.01 kcal/mol (6M0J). A good binding energy, drug likeness and efficient pharmacokinetic parameters suggest the potential of curcumin and few of its derivatives as SARS-CoV-2 spike protein inhibitors. However, further research is necessary to investigate the ability of these compounds as viral entry inhibitors.
Communicated by Ramaswamy H. Sarma
Keywords: Curcumin, SARS-CoV-2, SARS-CoV, spike protein
Introduction
Coronaviruses are enveloped positive-sense single stranded RNA viruses that belong to the family coronaviridae. They usually infect birds and mammals and cause mild respiratory diseases (Kahn & McIntosh, 2005). However, in the recent past, these viruses have caused lethal endemics such as Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) endemics (Kahn & McIntosh, 2005). In December 2019, Wuhan city in China became the centre of a surge in cases of pneumonia by an unknown cause (Zhu et al., 2020). A novel corona virus, called SARS-CoV-2, was isolated from these pneumonia patients in January 2020 and the disease was called the Coronavirus disease 2019 (COVID-19) by the World Health Organization in February, 2020 (Gorbalenya et. al, 2020; Jiang et al., 2020; Who, 2020). Even though the epidemic may have started from a zoonotic transmission in a seafood market, that also sold wild animals, it became clear that this disease was transmitted from person to person (Li et al., 2020). The clinical characteristics of this disease are broad, constituting asymptomatic infections, mild respiratory disease, severe pneumonia with respiratory failure and even death (Chen et al., 2020; Huang et al., 2020; Wang et al., 2020). The COVID-19 pandemic poses a significant challenge to global public health (Phelan et al., 2020), appealing for the development of safe and successful prophylactics and therapies against infection of its causative agent, the SARS-CoV-2 virus.
SARS-CoV-2 has a genome of ∼30 kilobases, which codes for multiple structural and non-structural proteins (Chan et al., 2020). The structural proteins, present on the surface of the mature virion, include the spike protein, the membrane protein, the envelope protein and the nucleocapsid protein (Chan et al., 2020). The Spike protein, of the beta coronaviruses SARS-CoV-2 and SARS-CoV, enables the attachment of the virus to the cells of the lower respiratory tract of humans to gain entry into the lung tissue (Hoffmann et al., 2020). Apart from attachment, the spike glycoprotein also appears to play a role in fusion and entry of the virus into the host (Chen et al., 2020; Hoffmann et al., 2020). The spike protein, of the novel coronavirus, utilizes the SARS-CoV receptor, ACE2 for entry (Chen et al., 2020) and is primed by the cellular protease, TMPRSS2 (Hoffmann et al., 2020). A serine protease inhibitor, which can act on TMPRSS2, has been shown to inhibit novel coronavirus entry (Hoffmann et al., 2020). Therefore, the spike protein of the novel coronavirus is a good drug target and identifying small molecules that bind to S protein would inhibit viral recognition of host cells and disrupt viral-host interactions.
Curcumin, a naturally occurring phytochemical and principal component of Curcuma longa, has exhibited broad pharmacological properties including antioxidant, anti-inflammatory, anti-cancer and anti-viral effects (Khor et al., 2019; Kocaadam & Şanlier, 2017; Lal et al., 2016; Wiggers et al., 2017). Curcumin and its derivatives, due to its rich conventional medicinal interest, has undergone comprehensive in vitro and in vivo studies. It has, therefore, been associated with more than 100 cellular targets, including cytokines, proteins, transcription factors, and receptors. Previous studies have shown the potential of curcumin as a treatment against Influenza A virus infection, by an effect mediated by modulating immune response to prevent injury to the lung tissue (Han et al., 2018). Curcumin has also been shown to have anti neuraminidase (NA) activity for the influenza virus NA protein (Richart et al., 2018). Therefore, in the present study, curcumin and its derivatives were docked onto the spike protein of the SARS-CoV and the SARS-CoV-2 to predict the binding interactions.
We found that curcumin and few of its derivatives showed promising results to be potential spike protein inhibitors. One of the derivatives, bis-demethoxycurcumin, showed the best binding affinity to the spike protein of both the SARS-CoV and the novel corona virus, SARS-CoV-2. The possibility for few of the curcumin derivatives, that showed good binding affinity, could be tested for further therapeutic use against COVID-19.
Experimental section
Sequence analysis
The sequences of the SARS-CoV-2 and SARS-CoV were downloaded from National Center for Biotechnology Information. Multiple sequence analysis and pairwise sequence identity was determined using the Clustal Omega server at the European Bioinformatics Institute (Sievers et al., 2011).
Preparation of coordinate file
The X-ray crystal structure of spike surface glycoprotein of SARS-CoV (PDB entry: 6CRV, resolution = 3.2 Å) and co-crystallized structure of SARS-CoV-2 RBD with human ACE2 Protein (PDB entry: 6M0J, resolution = 2.45 Å) was retrieved from Protein Data Bank (https:/www.rscb.org/pdb). The protein structure was prepared using the Discovery Studio Visualizer (version 3.1) and AutoDock Tools (ADT; version 1.5.4) through different steps viz. removal of water molecules and co-crystallized ligand, addition of missing hydrogen atoms, addition of Gasteiger-Marsili and Kollman charges, merging of non-polar hydrogens, and assignment of rotatable bonds. The file was then saved in pdbqt file format for further analysis.
Preparation of ligands
The chemical structures of curcumin derivatives were constructed using Chem3D 15.0 module of ChemOffice 15.0 and saved in PDB format. The structures were optimized using “Prepare Ligands” in the AutoDock 4.2, flexible torsions were assigned and the acyclic dihedral angles were allowed to rotate freely. The file was then saved as pdbqt file format for further analysis.
Molecular docking
Molecular docking simulations were conducted on the curcumin derivatives using the AutoDock 4.2 to get insight into their binding preferences within the active site of the receptor. The molecular docking simulations were performed on the PC based machines running on Windows 7 (x86) as operating system. The software included MGL tools 1.5.4 based AutoDock 4.2 (www.scripps.edu) which uses Python 2.7 language - Cygwin C:\program (www.cygwin.com) and Python 2.5 (www.python.com) (Morris et al., 2009).
Docking methodology
The flexible docking was performed using the refined spike protein of SARS-CoV (6CRV) and RBD domain of SARS-CoV-2 (6M0J). The grid maps of the interaction energies of various atom types were pre-calculated using AutoGrid 4.2. In each docking for spike surface glycoprotein and spike RBD, a grid box was created using a grid map of 45 × 45 × 45 points, 60 × 60 × 60 points with grid spacing of 0.375 Å and 0.420 Å respectively. The grid maps were centred on the corresponding ligand binding site within the protein structure.
Lamarckian Genetic Algorithm (LGA) was adopted to perform docking simulations using the following default parameters, viz. 100 independent runs with step sizes of 0.2 Å for translations and 5 Å for orientations and torsions, an initial population of random individuals with a population size of 150 individuals, a maximum of 2.5 × 106 energy evaluations, maximum number of generations of 27,000; mutation and crossover rates of 0.02 and 0.8 respectively and an elitism value of 1. All the computations were carried out on Cygwin and was used to generate both grid parameter file (.gpf file) and docking parameter file (.dpf file) for each ligand. The docked conformations of each ligand were ranked into clusters based on the binding energy and the top ranked conformations were used for further study. The pose with the lowest ΔG-score was considered the best fitted one and was further analyzed for Ligand-receptor interactions.
In-silico drug-likeness and pharmacokinetic property prediction
The in-silico prediction studies were performed, using pkCSM online prediction platforms (Pires et al., 2015), to assess the theoretical pharmacokinetic parameters of the ligands to predict the drug-likeness of ligands. The software calculated pharmaceutically relevant properties such as H-bond donor, H-bond acceptor, octanol-water partition coefficient (LogP), surface area, and number of rotatable bonds. In addition the ADME parameters of the curcumin analogs such as water solubility, Caco2 permeability, human intestinal absorption, skin permeability, P-glycoprotein I and II inhibition, volume of distribution, fraction of unbound drug, Blood Brain Barrier and CNS permeability, cytochrome P450 (CYP3A4 and CYP2C9 inhibition) inhibition, total clearance, action as renal OCT2 (organic cation transporter 2) substrate were analysed.
Results
Sequence alignment of the SARS-CoV-2 and SARS-CoV spike protein sequences
Prior to performing the docking studies the multiple sequence alignment was carried out for the spike proteins of the novel corona virus, SARS-CoV-2 and the 2002 SARS-CoV (Supplementary Figure 1 and Table 1). These results show that there is a very high percentage identity (greater than 99%) among the spike protein of the novel corona virus SARS-CoV-2 strains. There was around 74% identity of the SARS-CoV-2 spike proteins with the 2002 SARS-CoV. Therefore, the SARS-CoV-2 spike protein is very much closer to SARS-CoV than to the other corona viruses as shown in other studies (Ahmed et al., 2020).
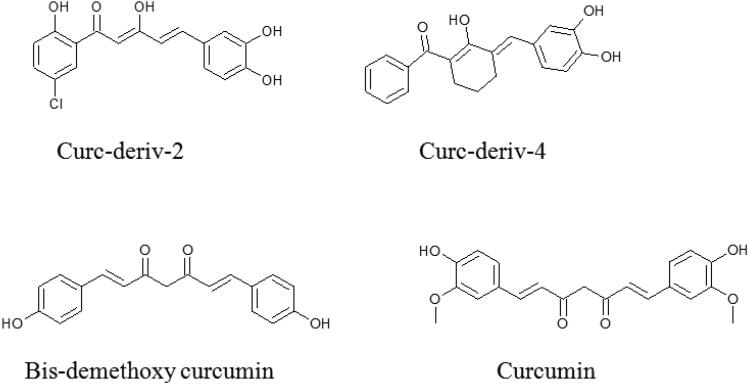
Figure 1.
The curcumin and its derivatives which displayed high binding affinity for SARS-CoV-2 and SARS-CoV spike proteins.
Table 1.
Percentage identity Matrix of Spike glycoprotein of SARS-CoV-2 and SARS-CoV. Indian 1 and 2 (QHS34546.1 and QIA98583.1), Korea (QHZ00379.1), Brazil (QIG55994.1), USA (QHO60594.1), Wuhan (QHD43416.1), Pakistan (QIQ22760.1), Italy (QIA98554.1), Australia (QHR84449.1) are spike proteins from SARS-CoV-2. SARS-6CRV is the spike protein from the 2002 SARS-CoV spike protein (6CRV).
| Viral Strain | SARS-CoV | Indian1 | Korea | Indian2 | Brazil | USA | Wuhan | Pakistan | Italy | Australia |
|---|---|---|---|---|---|---|---|---|---|---|
| SARS-CoV | 100 | 73.75 | 73.77 | 73.69 | 73.77 | 73.77 | 73.77 | 73.77 | 73.77 | 73.77 |
| Indian1 | 73.75 | 100 | 99.84 | 99.84 | 99.92 | 99.92 | 99.92 | 99.92 | 99.92 | 99.84 |
| Korea | 73.77 | 99.84 | 100 | 99.84 | 99.92 | 99.92 | 99.92 | 99.92 | 99.92 | 99.84 |
| Indian2 | 73.69 | 99.84 | 99.84 | 100 | 99.92 | 99.92 | 99.92 | 99.92 | 99.92 | 99.84 |
| Brazil | 73.77 | 99.92 | 99.92 | 99.92 | 100 | 100 | 100 | 100 | 100 | 99.92 |
| USA | 73.77 | 99.92 | 99.92 | 99.92 | 100 | 100 | 100 | 100 | 100 | 99.92 |
| Wuhan | 73.77 | 99.92 | 99.92 | 99.92 | 100 | 100 | 100 | 100 | 100 | 99.92 |
| Pakistan | 73.77 | 99.92 | 99.92 | 99.92 | 100 | 100 | 100 | 100 | 100 | 99.92 |
| Italy | 73.77 | 99.92 | 99.92 | 99.92 | 100 | 100 | 100 | 100 | 100 | 99.92 |
| Australia | 73.77 | 99.84 | 99.84 | 99.84 | 99.92 | 99.92 | 99.92 | 99.92 | 99.92 | 100 |
Molecular docking studies
The three-dimensional structure of the SARS-CoV spike protein and SARS-CoV-2 Spike protein RBD has been solved. The PDB files of the SARS-CoV spike protein (6CRV) and the Spike protein RBD of the SARS-CoV-2 (6M0J) was downloaded from the PDB databank. These structures were used to predict the ability of the binding of curcumin and its derivatives to the spike proteins of the two corona viruses. Curcumin and 24 of its derivatives (Figure 1 and Supplementary Figure 2) were docked on to 6CRV and 6M0J. Tables 2 and 3 gives the binding energies of curcumin and its derivatives with 6CRV (binding energies ranged from −10.98 to −5.12 kcal/mol) and with 6M0J (binding energies ranged −10.01 to −5.33 kcal/mol) respectively. Visual examination of the computationally docked optimal binding poses of curcumin and its derivatives on 6CRV and 6MOJ revealed the important role of various types of interactions viz. hydrogen bonding and hydrophobic interactions, including π–π stacking, π–cation, and π–σ interactions in the stability of the binding of the curcumin/derivatives to the spike protein (6CRV and 6M0J). All compounds showed one or more hydrogen bonds with 6CRV and 6M0J except a few.
Figure 2.
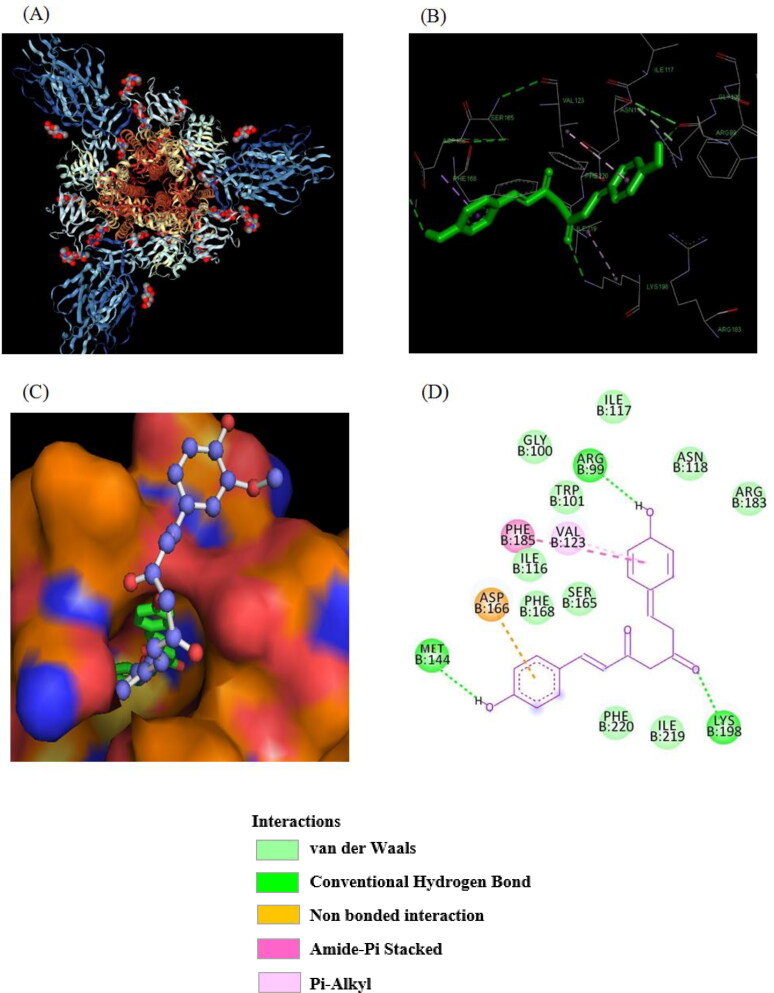
Molecular docking of Bis-demethoxy curcumin on SARS-CoV spike protein (6CRV). (A) 3 D structure of SARS-CoV Spike Glycoprotein Macromolecule (6CRV). (B) Binding Interaction of Bis-demethoxy curcumin with amino acid residues of SARS-CoV Spike glycoprotein (PDB ID-6CRV). (C) Superimposed binding mode of Bis-demethoxy curcumin with SARS-CoV Spike protein. (D) 2 D representation of the binding interaction of Bis-demethoxy curcumin with SARS-CoV Spike protein.
Table 2.
Molecular docking results of curcumin and its derivatives with SARS-CoV Spike protein (6CRV). The top hits with the binding energy lesser than -8.4 are in bold.
| Compound | Binding Energy (ΔG) | Ligand Efficiency | Intermolecular energy | XP H-Bond |
|---|---|---|---|---|
| Curcumin | –10.1 | –0.14 | –8.24 | –10.9724 |
| BHBC | –8.28 | –0.13 | –7.87 | –3.42239 |
| MNC | –8.01 | –0.43 | –6.98 | –8.01452 |
| BDMC | –10.98 | –0.18 | –8.94 | –14.0467 |
| 1 | –5.12 | –0.25 | –4.17 | 0 |
| 2 | –8.55 | –0.41 | –6.56 | –18.4317 |
| 3 | –6.56 | –0.21 | –4.92 | –11.1148 |
| 4 | –8.69 | –0.13 | –6.94 | –19.1539 |
| 5 | –7.03 | –0.19 | –5.93 | –8.47153 |
| 6 | –6.59 | –0.36 | –5.12 | –7.41531 |
| 7 | –7.96 | –0.21 | –6.14 | –7.98548 |
| 8 | –7.55 | –0.19 | –5.14 | –6.14537 |
| 9 | –6.91 | –0.21 | –5.18 | 0 |
| 10 | –7.75 | –0.21 | –6.14 | –6.73473 |
| 11 | –8.02 | –0.19 | –6.19 | –17.2465 |
| 12 | –7.85 | –0.38 | –6.25 | –14.6321 |
| 13 | –5.94 | –0.29 | –4.12 | –11.3576 |
| 14 | –8.75 | –0.25 | –7.19 | –21.1412 |
| 15 | –7.87 | –0.38 | –7.26 | –0.473228 |
| 16 | –8.47 | –0.25 | –6.88 | –8.458912 |
| DMC | –7.89 | –0.21 | –6.41 | –10.47057 |
| FAC | –7.17 | –0.36 | –5.15 | –9.14873 |
| IBC | –7.91 | –0.23 | –7.17 | –6.90341 |
| IVC | –7.43 | –0.44 | –6.67 | –19.9864 |
| SYC | –6.82 | –0.26 | –5.45 | –9.44678 |
Abbreviations: BHBC: 3-5-di-tert-butyl-4-hydroxybenzaldehyde curcumin, MNC: 4-methoxy-1-naphthaldehyde curcumin, BDMC: Bis-demethoxy curcumin, DMC: Demethoxy curcumin, FAC: Ferulic-acid curcumin, IBC: Ibuprofen curcumin, IVC: Isovanillin curcumin, SYC: Syringaldehyde curcumin.
Table 3.
Molecular docking analysis of curcumin and its derivatives with SARS-CoV-2 Spike protein Receptor binding domain (6M0J). The top hits with the binding energy lesser than -8.4 are in bold.
| Compound | Binding Energy (ΔG) | Ligand Efficiency | Intermolecular energy | XP H-Bond |
|---|---|---|---|---|
| Curcumin | –9.81 | –0.15 | –8.35 | –15.1512 |
| BHBC | −5.33 | –0.13 | –7.96 | 0 |
| MNC | –7.96 | –0.27 | –7.38 | –4.24687 |
| BDMC | –10.01 | –0.12 | –9.18 | –13.9824 |
| 1 | –7.58 | –0.36 | –6.19 | –18.3489 |
| 2 | –8.81 | –0.31 | –8.38 | 1.2654 |
| 3 | –6.14 | –0.34 | –5.45 | –6.95102 |
| 4 | –8.88 | –0.18 | –7.98 | –14.0321 |
| 5 | –6.23 | –0.26 | –5.88 | –3.5891 |
| 6 | –7.16 | –0.37 | –6.15 | –10.1688 |
| 7 | –6.89 | –0.25 | –5.98 | –9.12515 |
| 8 | –5.49 | –0.12 | –3.90 | –5.92265 |
| 9 | –6.92 | –0.18 | –6.22 | –6.96719 |
| 10 | –7.03 | –0.24 | –6.33 | –7.0 |
| 11 | –6.88 | –0.27 | –6.23 | –6.44363 |
| 12 | –7.95 | –0.41 | –5.70 | –2.5581 |
| 13 | –6.11 | –0.39 | –5.42 | –7.34943 |
| 14 | –7.45 | –0.19 | –6.40 | –10.5367 |
| 15 | –5.74 | –0.39 | –5.51 | –2.36748 |
| 16 | –7.83 | –0.27 | –7.48 | –6.64321 |
| DMC | –5.98 | –0.19 | –5.38 | –5.99913 |
| FAC | –5.85 | –0.38 | –5.25 | –6.46613 |
| IBC | –6.65 | –0.15 | –6.20 | –1.52841 |
| IVC | –8.96 | –0.43 | –7.73 | –12.2707 |
| SYC | –5.87 | –0.29 | –5.43 | –4.4411 |
Abbreviations: BHBC: 3-5-di-tert-butyl-4-hydroxybenzaldehyde curcumin, MNC: 4-methoxy-1-naphthaldehyde curcumin, BDMC: Bis-demethoxy curcumin, DMC: Demethoxy curcumin, FAC: Ferulic-acid curcumin, IBC: Ibuprofen curcumin, IVC: Isovanillin curcumin, SYC: Syringaldehyde curcumin.
In 6CRV, it was found that ARG99, GLY100, TRP 101, ILE116, ILE117, ASN118, VAL123, MET144, SER165, ASP166, PHE168, ARG183, PHE185, LYS198, ILE219, PHE220 were the major amino acids involved in hydrogen bonding, hydrophobic and van der Waals interaction with most of the curcumin compounds. However, majority of the curcumin compounds exhibited hydrogen bonding with ARG99, MET144, LYS198 except compound 1 and 9. In addition, all the compounds exhibited hydrophobic (π–π stacking, π–cation, and π–σ) interactions with ASP166, VAL123, PHE185 and van der Waals interaction with GLY100, TRP 101, ILE117, ASN118, ARG183, ILE116, PHE168, SER165, PHE220, ILE219.
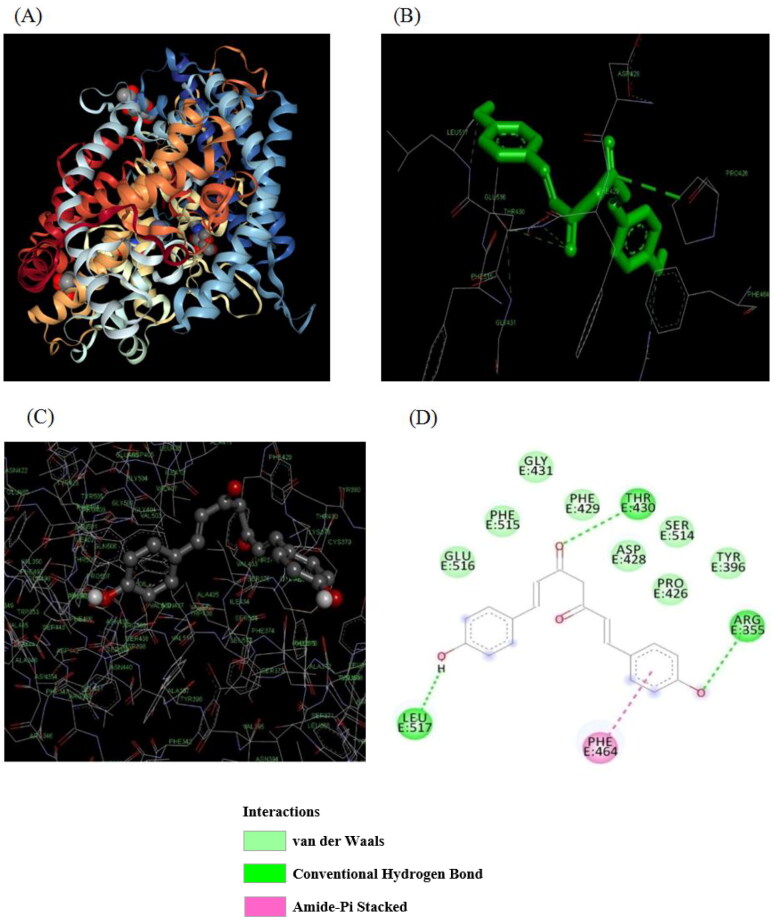
All compounds showed one or more hydrogen bonding interactions with spike RBD of the SARS-CoV-2 (6M0J) except 3-5-di-tert-butyl-4-hydroxybenzaldehyde curcumin. It was found that ARG355, TYR396, PRO426, ASP428, THR430, GLY431, PHE429, PHE 464, GLU516, SER514, LEU517 and PHE515 were the major amino acids involved in hydrogen bonding hydrophobic and van der Waals interaction with the all of curcumin compounds. However, majority of the curcumin compounds exhibit hydrogen bonding with ARG355, LEU517 and THR430. In addition, the compounds exhibited hydrophobic (π–π stacking, π–cation, and π–σ) and van der Waals interactions with PHE464, GLY431, GLU516, SER514, PHE515, TYR396, PRO426, ASP428 and PHE429.
The curcumin derivative with the best binding affinity was Bis-demethoxycurcumin for both 6CRV (ΔG = −10.98 kcal/mol) and 6M0J (ΔG = −10.01 kcal/mol). The native compound, curcumin, demonstrated a binding energy of −9.81 kcal/mol for 6M0J and −10.1 kcal/mol for 6CRV. Binding mode of Bis-demethoxycurcumin with SARS-CoV Spike protein and SARS-CoV-2 Spike protein RBD is shown in Figure 2 and 3, respectively.
Figure 3.
Molecular docking of Bis-demethoxy curcumin on SARS-CoV-2 spike protein receptor binding domain (6M0J). (A) 3D structure of SARS-CoV-2 spike protein RBD bound with ACE2 protein Macromolecule. (B) Binding Interaction of Bis-demethoxy curcumin with amino acid residue of spike protein RBD of SARS-CoV-2 (PDB ID-6M0J). (C) Superimposed binding mode of Bis-demethoxy curcumin, docked with SARS-CoV-2 spike protein RBD. (D) 2D representation of the binding interaction of Bis-demethoxy curcumin with SARS-CoV-2 spike RBD.
In-silico drug-likeness and pharmacokinetic property prediction
All the structures used for docking were analysed for in silico drug-likeness based on the Lipinski’s rules using pkCSM server (Table 4). The lipophilicity (expressed as LogP) predicted for all the compounds were found to be well above the traditionally cut-off value of 5 used for drug design. Curcumin and its derivatives, used in this study, show suitable MW values (MW < 500) essential for a successful penetration through biological membranes. The surface area (SA) for all the compounds was observed to be in the range 115.89 − 240.65 Å2 which is well within the limit. All compounds, except 5-di-tert-butyl-4-hydroxybenzaldehyde curcumin (BHBC), 4-methoxy-1-naphthaldehyde curcumin (MNC), Syringaldehyde curcumin (SYC) and compound-16, fall into the appropriate range indicating good bioavailability of the candidate molecule. The number of hydrogen bond acceptors (HBA, ≤10) and donors (HBD, ≤5) for all the compounds were in accordance with the Lipinski’s rule of five.
Table 4.
In-silico prediction of drug-likeness for curcumin derivatives[a].
| Compound | MW | LogP | SA | HBA | HBD | nviolations | Rotatable bonds |
|---|---|---|---|---|---|---|---|
| BHBC | 534.78 | 8.93 | 236.0 | 4 | 3 | 1 | 6 |
| MNC | 538.59 | 6.14 | 232.11 | 7 | 2 | 1 | 11 |
| BDMC | 308.33 | 3.83 | 133.51 | 4 | 3 | 5 | |
| 1 | 270.353 | 4.21 | 115.89 | 3 | 1 | 4 | |
| 2 | 332.739 | 3.79 | 136.57 | 5 | 4 | 4 | |
| 3 | 314.337 | 3.77 | 133.64 | 5 | 4 | 4 | |
| 4 | 322.36 | 4.36 | 139.56 | 3 | 3 | 4 | |
| 5 | 364.441 | 4.93 | 159.62 | 4 | 0 | 5 | |
| 6 | 320.388 | 4.95 | 141.45 | 4 | 3 | 1 | |
| 7 | 325.32 | 3.60 | 138.02 | 5 | 1 | 6 | |
| 8 | 329.193 | 4.45 | 125.75 | 2 | 1 | 4 | |
| 9 | 462.181 | 5.24 | 164.71 | 7 | 2 | 0 | |
| 10 | 351.358 | 4.13 | 149.74 | 4 | 5 | 1 | |
| 11 | 360.375 | 5.07 | 149.89 | 2 | 1 | 3 | |
| 12 | 419.565 | 6.24 | 185.40 | 9 | 4 | 1 | |
| 13 | 389.495 | 5.22 | 171.67 | 4 | 4 | 1 | |
| 14 | 342.778 | 3.81 | 143.82 | 4 | 2 | 2 | |
| 15 | 469.629 | 4.38 | 208.20 | 6 | 5 | 1 | |
| 16 | 486.564 | 6.19 | 211.58 | 11 | 6 | 1 | 0 |
| DMC | 342.391 | 3.20 | 146.43 | 5 | 2 | 9 | |
| FAC | 550.604 | 4.7 | 232.81 | 9 | 2 | 15 | |
| IBC | 556.655 | 5.24 | 240.62 | 7 | 1 | 15 | |
| IVC | 504.535 | 4.69 | 214.22 | 8 | 3 | 11 | |
| SYC | 430.453 | 3.77 | 180.12 | 10 | 8 | 1 | 3 |
MW = Molecular weight, LogP = octanol-water partition coefficient, SA = Surface Area, HBA = Number of hydrogen bond acceptor, HBD = Number of hydrogen bond donor, nviolations = violations from Lipinski’s rule. BHBC: 3-5-di-tert-butyl-4-hydroxybenzaldehyde curcumin, MNC: 4-methoxy-1-naphthaldehyde curcumin, BDMC: Bis-demethoxy curcumin, DMC: Demethoxy curcumin, FAC: Ferulic-acid curcumin, IBC: Ibuprofen curcumin, IVC: Isovanillin curcumin, SYC: Syringaldehyde curcumin.
Additionally, a variety of key ADMET (Absorption, Distribution, Metabolism and Excretion) properties have also been calculated with the aid of pkCSMserver. The results are listed in Table 5. All curcumin derivatives showed moderate to high water solubility ranging from −2.91 log mol/L (BHBC) to −6.99 log mol/L (compound-12), in addition to high Caco-2 permeability (permeation > 0.90) except compound BHBC and MNC (permeation = 0.67 and 0.43 respectively) which showed moderate permeability. Intestinal absorption (IA) has been found to be greater than 85% indicating good permeation across the intestinal membrane. Further, all curcumin compounds showed good permeation through skin (permeation > −2.5). Additionally, all curcumin compounds showed no inhibition towards P-glycoprotein I and P-glycoprotein II, except compounds BHBC, MNC, compound-13 and Ferrulic acid curcumin (FAC) which demonstrated inhibition towards P-glycoprotein I. Furthermore, all curcumin compounds showed poor BBB permeability and moderate CNS permeability except Isovanillin curcumin (IVC) and SYC. All showed inhibition towards the metabolizing enzyme CYP3A4 except BHBC, compounds-8, -11, -14, -15 and -16, while except compound-15, all showed inhibition towards CYP2C9. All curcumin derivatives were found to show the total clearance in the range 0.10 log mL/min/kg (compound-1 and -4) to 1.01 log mL/min/kg (compound-15). Further, all curcumin derivatives except compound 15 were found to act as OCT2 substrate, thus indicating that these compounds will not have any adverse interactions and no negative effect on renal clearance.
Table 5.
In-silico ADME prediction for curcumin derivatives[a].
| Comp | Absorption |
Distribution |
Metabolism |
Excretion |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WS | CP | IA | SP | PI-1 | PI-2 | VD | FU | BBB | CNS | CI-1 | CI-2 | TC | RS | |
| BHBC | –2.91 | 0.67 | 88.62 | –2.73 | Yes | Yes | –0.39 | 0.31 | –0.67 | –0.85 | No | Yes | 0.15 | No |
| MNC | –3.64 | 0.43 | 100 | –2.73 | Yes | Yes | –1.45 | 0.33 | –0.39 | –2.73 | Yes | Yes | 0.26 | No |
| BDMC | –4.43 | 1.01 | 95.92 | –2.99 | No | No | –0.22 | 0.08 | –0.72 | –2.11 | Yes | Yes | 0.12 | No |
| 1 | –4.66 | 1.25 | 91.80 | –2.51 | No | No | 0.18 | 0.06 | 0.01 | –1.45 | Yes | Yes | 0.10 | No |
| 2 | –3.23 | 0.93 | 89.16 | –2.76 | No | No | –0.07 | 0.12 | –1.07 | –2.29 | Yes | Yes | 0.05 | No |
| 3 | –3.19 | 0.94 | 89.70 | –2.76 | No | No | –0.02 | 0.13 | –1.06 | –2.27 | Yes | Yes | 0.06 | No |
| 4 | –4.11 | 0.92 | 87.64 | –3.07 | No | No | 0.09 | 0 | –0.74 | –1.85 | Yes | Yes | 0.10 | No |
| 5 | –6.39 | 1.06 | 94.85 | –2.49 | No | No | 0.25 | 0 | –0.03 | –1.30 | Yes | Yes | 0.22 | No |
| 6 | –5.42 | 1.43 | 90.49 | –2.73 | No | No | 0.05 | 0 | –0.33 | –1.29 | Yes | Yes | 0.23 | No |
| 7 | –4.54 | 1.04 | 91.97 | –2.73 | No | No | –0.37 | 0 | 0.28 | –2.20 | Yes | Yes | 0.13 | No |
| 8 | –5.09 | 1.66 | 90.44 | –2.29 | No | No | 0.27 | 0 | 0.16 | –1.47 | No | Yes | 0.14 | No |
| 9 | –7.03 | 1.13 | 93.34 | –2.51 | No | No | 0.37 | 0 | 0.46 | –1.26 | Yes | Yes | 0.26 | No |
| 10 | –4.89 | 0.94 | 91.50 | –2.74 | No | No | –0.04 | 0 | –0.24 | –1.93 | Yes | Yes | 0.11 | No |
| 11 | –5.93 | 1.28 | 89.16 | –2.59 | No | No | 0.36 | 0 | 0.66 | –1.40 | No | Yes | 0.13 | No |
| 12 | –6.99 | 1.02 | 91.30 | –2.75 | No | No | 0.66 | 0 | –0.32 | –1.85 | Yes | Yes | 1.04 | No |
| 13 | –5.42 | 1.27 | 92.76 | –2.80 | Yes | Yes | 0.54 | 0 | –0.02 | –1.58 | Yes | Yes | 0.26 | No |
| 14 | –4.20 | 1.12 | 90.31 | –2.92 | No | No | –0.04 | 0.01 | –0.31 | –1.33 | No | Yes | 0.08 | No |
| 15 | –3.76 | 0.97 | 88.86 | –2.79 | No | No | 1.90 | 0.16 | –0.20 | –1.26 | No | No | 1.01 | Yes |
| 16 | –6.92 | 0.90 | 94.67 | –2.73 | No | No | –0.59 | 0.07 | –0.72 | –2.62 | No | Yes | 0.41 | No |
| DMC | –3.95 | 0.99 | 92.03 | –2.74 | No | No | –0.06 | 0.12 | –0.23 | –2.66 | Yes | Yes | 0.32 | No |
| FAC | –4.02 | 1.41 | 83.68 | –2.76 | Yes | Yes | –0.58 | 0.24 | –0.91 | –3.26 | Yes | Yes | 0.55 | No |
| IBC | –4.36 | 0.91 | 91.72 | –2.73 | No | No | –0.88 | 0.18 | –0.53 | –2.77 | Yes | Yes | 0.62 | No |
| IVC | –3.95 | 0.96 | 89.31 | –2.73 | No | No | –0.60 | 0.24 | –1.37 | –3.02 | Yes | Yes | 0.16 | No |
| SYC | –3.89 | 1.01 | 89.15 | –2.73 | No | No | 0.28 | 0.17 | –1.47 | –3.14 | Yes | Yes | 0.23 | No |
Abbreviations: WS—Water solubility (log mol/L), CP—Caco2 permeability (log Papp in 10-6 cm/s), IA—Human intestinal absorption (% Absorbed), SP—Skin permeability (log Kp),PI-1—P-glycoprotein I inhibitor, PI-2—P-glycoprotein II inhibitor, VD—Human volume ofdistribution (log L/kg), FU—Fraction unbound (human) (Fu), BBB—BBB permeability (logBB), CNS—CNS permeability (log PS), CI-1—CYP3A4 inhibitor, CI-2—CYP2C9 inhibitor,TC—Total clearance (log mL/min/kg), RS—Renal OCT2 (organic cation transporter 2) substrate. BHBC: 3-5-di-tert-butyl-4-hydroxybenzaldehyde curcumin, MNC: 4-methoxy-1-naphthaldehyde curcumin, BDMC: Bis-demethoxy curcumin, DMC: Demethoxy curcumin, FAC: Ferulic-acid curcumin, IBC: Ibuprofen curcumin, IVC: Isovanillin curcumin, SYC: Syringaldehyde curcumin.
Conclusion and discussion
The spike proteins of corona viruses are essential for entry of the virus into the target cells. The spike protein exists as a trimer on the surface of the virus with one of the monomer in up conformation and the other two in down conformation (Wrapp et al., 2020). The N-terminal region (S1) of the S protein is important for binding to the cellular receptor ACE2 (Hoffmann et al., 2020; Tai et al., 2020). The S protein undergoes priming by cellular protease, TMPRSS2 and the S2 region of the protein is responsible for fusion of viral and cellular membrane (Hoffmann et al., 2020). Therefore, identifying therapeutics for the S protein of the novel corona virus could potentially target the critical process of entry and fusion of the virus.
Curcumin and its derivatives are known for their many biological activities, one of them is its antiviral activity. Therefore, here, we examined the potential of curcumin, and its derivatives, to bind to the SARS-CoV and SARS-CoV-2 spike protein. From our computational molecular docking approach (using auto dock 4.2, PDB ID - 6CRV, 6M0J) and in-silico ADMET tool, we predicted that Bis-demethoxy curcumin, compound-4 and compound-2 were the most recommended curcumin compounds which bind to RBD domain of the SARS-CoV-2 Spike protein and SARS-CoV spike protein efficiently in in silico studies.
Curcumin has earlier shown to have specific inhibitory effect on the NA activity in influenza virus (Chen et al., 2013; Richart et al., 2018) and modulating immune response to prevent injury to the lung tissue (Han et al., 2018). Till date, Bis-demethoxy curcumin has shown to have anti-cancer and hepato-protective activities (Kumaravel et al., 2013; Rajagopalan et al., 2010). This is the first time; it is predicted to have a potential anti-viral activity. Therefore, these curcumin derivatives, which have been predicted to bind to the SARS-CoV-2 spike protein, could be explored as probable inhibitors of COVID-19 spike protein through experimental studies.
Supplementary Material
Acknowledgements
We acknowledge the support rendered by the Department of Biochemistry, Indian Academy Degree College - Autonomous, Bengaluru and Ramanbhai Patel College of Pharmacy, Charotar University of Science and Technology, Gujarat for their support in carrying out this work.
Disclosure statement
The authors declare that they have no conflict of interests
References
- Chan, J. F., Kok, K. H., Zhu, Z., Chu, H., To, K. K., Yuan, S., Yuen, K. Y. (2020). Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan. Emerg Microbes Infect, 9(1), 221–236. 10.1080/22221751.2020.1719902. Erratum in: Emerg Microbes Infect. PMID: 3198700 [DOI] [PMC free article] [PubMed]
- Chen, Y., Guo, Y., Pan, Y., & Zhao, Z. J. (2020). Structure analysis of the receptor binding of 2019-nCoV. Biochemical and Biophysical Research Communications, 525(1), 135–140. 10.1016/j.bbrc.2020.02.071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorbalenya, A. E., Baker, S. C., Baric, R. S., de Groot, R. J., Drosten, C., Gulyaeva, A. A., Haagmans, B. L., Lauber, C., Leontovich, A. M., Neuman, B. W., Penzar, D., Perlman, S., Poon, L. L. M., Samborskiy, D. V., Sidorov, I. A., Sola, I., Ziebuhr, J., Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. (2020). The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nature Microbiology, 5, 536–544. 10.1038/s41564-020-0695-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han, S., Xu, J., Guo, X., & Huang, M. (2018). Curcumin ameliorates severe influenza pneumonia via attenuating lung injury and regulating macrophage cytokines production. Clincal and Experimental Pharmacology Physiology, 45(1), 84–93. 10.1111/1440-1681.12848 [DOI] [PubMed] [Google Scholar]
- Hoffmann, M., Kleine-Weber, H., Schroeder, S., Krüger, N., Herrler, T., Erichsen, S., Schiergens, T. S., Herrler, G., Wu, N. H., Nitsche, A., Müller, M. A., Drosten, C., Pöhlmann, S. (2020). SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell,181(2), 271-280.e8. 10.1016/j.cell.2020.02.052 [DOI] [PMC free article] [PubMed]
- Huang, C., Wang, Y., Li, X., Ren, L., Zhao, J., Hu, Y., Zhang, L., Fan, G., Xu, J., Gu, X., Cheng, Z., Yu, T., Xia, J., Wei, Y., Wu, W., Xie, X., Yin, W., Li, H., Liu, M., … Cao, B. (2020). Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet, 395(10223), 497–506. 10.1016/S0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang, S., Shi, Z., Shu, Y., Song, J., Gao, G. F., Tan, W., & Guo, D. (2020). A distinct name is needed for the new coronavirus. Lancet (London, England), 395(10228), 949. 10.1016/S0140-6736(20)30419-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn, J. S., & McIntosh, K. (2005). History and recent advances in coronavirus discovery. Pediatr Infect Dis, J24, S223–S227. discussion S226. [DOI] [PubMed] [Google Scholar]
- Khor, P. Y., Aluwi, M. F. F. M., Rullah, K., & Lam, K. W. (2019). Insights on the synthesis of asymmetric curcumin derivatives and their biological activities. European Journal of Medicinal Chemistry, 183, 111704. 10.1016/j.ejmech.2019.111704 [DOI] [PubMed] [Google Scholar]
- Kocaadam, B., & Şanlier, N. (2017). Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Critical Reviews in Food Science and Nutrition, 57(13), 2889–2895. 10.1080/10408398.2015.1077195 [DOI] [PubMed] [Google Scholar]
- Kumaravel, M., Sankar, P., Latha, P., Benson, C. S., & Rukkumani, R. (2013). Antiproliferative effects of an analog of curcumin in Hep-2 cells: A comparative study with curcumin. Natural Product Communications, 8(2), 183–186. [PubMed] [Google Scholar]
- Lal, J., Gupta, S. K., Thavaselvam, D., & Agarwal, D. D. (2016). Synthesis and pharmacological activity evaluation of curcumin derivatives. Chinese Chemical Letters, 27(7), 1067–1072. 10.1016/j.cclet.2016.03.032 [DOI] [Google Scholar]
- Li, Q., Guan, X., Wu, P., Wang, X., Zhou, L., Tong, Y., Ren, R., Leung, K. S. M., Lau, E. H. Y., Wong, J. Y., Xing, X., Xiang, N., Wu, Y., Li, C., Chen, Q., Li, D., Liu, T., Zhao, J., Liu, M., … Feng, Z. (2020). Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. New England Journal of Medicine, 382(13), 1199–1207. 10.1056/NEJMoa2001316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris, G. M., Huey, R., Lindstrom, W., Sanner, M. F., Belew, R. K., Goodsell, D. S., & Olson, A. J. (2009). AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. Journal of Computational Chemistry, 30(16), 2785–2791. 10.1002/jcc.21256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phelan, A. L., Katz, R., & Gostin, L. O. (2020). The novel coronavirus originating in Wuhan, China: Challenges for global health governance. JAMA, 323(8), 709–710. 10.1001/jama.2020.1097 [DOI] [PubMed] [Google Scholar]
- Pires, D. E. V., Blundell, T. L., & Ascher, D. B. (2015). pkCSM: Predicting Small-Molecule Pharmacokinetic and Toxicity Properties Using Graph-Based Signatures. Journal of Medicinal Chemistry, 58(9), 4066–4072. 10.1021/acs.jmedchem.5b00104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajagopalan, R., Sridharana, S., & Menon, V. P. (2010). Hepatoprotective role of bis-demethoxy curcumin analog on the expression of matrix metalloproteinase induced by alcohol and polyunsaturated fatty acid in rats. Toxicology Mechanisms and Methods, 20(5), 252–259. 10.3109/15376516.2010.482961 [DOI] [PubMed] [Google Scholar]
- Richart, S. M., Li, Y. L., Mizushina, Y., Chang, Y. Y., Chung, T. Y., Chen, G. H., Tzen, J. T., Shia, K. S., & Hsu, W. L. (2018). Synergic effect of curcumin and its structural analogue (Monoacetylcurcumin) on anti-influenza virus infection. Journal of Food and Drug Analysis, 26(3), 1015–1023. 10.1016/j.jfda.2017.12.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sievers, F., Wilm, A., Dineen, D., Gibson, T. J., Karplus, K., Li, W., Lopez, R., McWilliam, H., Remmert, M., Soding, J., Thompson, J. D., & Higgins, D. G. (2011). Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Molecular Systems Biology, 7, 539 10.1038/msb.2011.75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tai, W., He, L., Zhang, X., Pu, J., Voronin, D., Jiang, S., Zhou, Y., & Du, L. (2020). Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: Implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cellular & Molecular Immunology, 17(6), 613–620. 10.1038/s41423-020-0400-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, D., Hu, B., Hu, C., Zhu, F., Liu, X., Zhang, J., Wang, B., Xiang, H., Cheng, Z., Xiong, Y., Zhao, Y., Li, Y., Wang, X., & Peng, Z. (2020). Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA, 323(11), 1061. 10.1001/jama.2020.1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Who (2020). Coronavirus disease 2019 (COVID-19) situation report-56, WHO.
- Wiggers, H. J., Zaioncz, S., Cheleski, J., Mainardes, R. M., Khalil, N. M., & Atta Ur, R. (2017). Chapter 7 - Curcumin, a Multitarget Phytochemical: Challenges and Perspectives. Studies in Natural Products Chemistry, Elsevier. 53, 243–276. [Google Scholar]
- Wrapp, D., Wang, N., Corbett, K. S., Goldsmith, J. A., Hsieh, C.-L., Abiona, O., Graham, B. S., & McLellan, J. S. (2020). Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science, 367(6483), 1260–1263. 10.1126/science.abb2507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, N., , Zhang, D., , Wang, W., , Li, X., , Yang, B., , Song, J., , Zhao, X., , Huang, B., , Shi, W., , Lu, R., , Niu, P., , Zhan, F., , Ma, X., , Wang, D., , Xu, W., , Wu, G., , Gao, G. F., & , Tan, W. (2020). A Novel Coronavirus from Patients with Pneumonia in China, 2019. The New England Journal of Medicine, 382(8), 727–733. 10.1056/NEJMoa2001017 31978945 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.