Abstract
Some environmental factors are associated with an increased risk of multiple sclerosis (MS). Air pollution could be a main one. This study was conducted to investigate the association of particulate matter 2.5 (PM2.5) concentrations with MS prevalence in the province of Pavia, Italy. The overall MS prevalence in the province of Pavia is 169.4 per 100,000 inhabitants. Spatial ground-level PM2.5 gridded data were analysed, by municipality, for the period 2010–2016. Municipalities were grouped by tertiles according to PM2.5 concentration. Ecological regression and Bayesian statistics were used to analyse the association between PM2.5 concentrations, degree of urbanization, deprivation index and MS risk. MS risk was higher among persons living in areas with an average winter PM2.5 concentration above the European annual limit value (25 μg/m3). The Bayesian map revealed sizeable MS high-risk clusters. The study found a relationship between low MS risk and lower PM2.5 levels, strengthening the suggestion that air pollution may be one of the environmental risk factors for MS.
Keywords: Multiple sclerosis, Epidemiology, PM2.5, Air pollution, Bayesian mapping, Ecological study
Introduction
Multiple sclerosis (MS) is a complex disorder, likely arising from interactions between numerous genetic factors and several still incompletely clarified environmental factors (Thompson et al. 2018).
These environmental factors include diet and vitamin D levels (Ascherio and Munger 2007), both of which have been widely studied. However, air pollution, too, might influence the pathogenesis of immune and inflammatory processes in MS through various mechanisms, such as the development of oxidative stress and proinflammatory milieu (Veldhoen et al. 2008) (Odoardi et al. 2012), blood-brain barrier breakdown (Minagar and Alexander 2003; Kooij et al. 2010; Oppenheim et al. 2013), microglia activation (Calderón-Garcidueñas et al. 2002; Li et al. 2005) and vitamin D hypovitaminosis (Hosseinpanah et al. 2010).
Air quality is a major concern in northern Italy, especially in the Po Valley (Pianura Padana), a flat area particularly prone to high levels of particulate matter (PM). There are several reasons for this: high anthropogenic emissions, the natural geography of the area (with the Alps forming a barrier to the north) and its typically low wind speed that favours the formation and accumulation of pollutants.
The existence of possible links between air pollution and the epidemiology of MS has been investigated in few studies. It was found indeed an association between PM10 levels and MS prevalence (Gregory et al. 2008), as well as links with the occurrence of MS clinical relapses (Koski et al. 2003; Roux et al. 2017; Jeanjean et al. 2018) and MRI ‘activity’ (Bergamaschi et al. 2018).
A multicentre study in Italy found a 42% increase in hospital admissions for MS in Lombardy after elevated daily PM10 levels (Angelici et al. 2016). In another Italian study, the prevalence of MS in the north-eastern province of Padua (Veneto region) was demonstrated to be higher in urban areas with increased PM2.5 levels than in rural villages/localities (Tateo et al. 2019).
From an epidemiological point of view, it is appealing to analyse possible relationships between air pollution and MS occurrence in relatively small areas, with a view to enabling future research to focus on specific clusters of interest.
The present ecological study was conducted to investigate the spatial distribution of MS risk across the administrative areas of the province of Pavia (northern Italy), based on air pollution distribution (PM2.5 concentrations).
Materials and methods
Study population
MS cases were ascertained employing the sources and inclusion criteria already used in the previous research conducted by our group (Bergamaschi et al. 2006).
Briefly, the primary source was the Pavia Multiple Sclerosis Registry (PREMS) kept by the MS Centre at the C. Mondino Foundation in Pavia; this registry has been active since 1990 and has been part of the ‘Italian Multiple Sclerosis Registry’ since 2015. Data were also drawn from the data warehouse of the local Health Protection Agency (HPA), Department of Hygiene and Health Prevention of Pavia, specifically searching for exemption code (046.340) and the ICD-9-CM code for MS (340). Data were subsequently cleaned of duplicates.
The clinical documentation of the patients found through the HPA data (i.e. patients not followed by the MS Centre at the Mondino Foundation and not included in the PREMS) were reviewed by a senior neurologist (RB) to verify the validity of the diagnosis. Each patient’s life status was checked by consulting the HPA mortality registry.
All residents of the province of Pavia with a diagnosis of MS according to the 2010 revised McDonald diagnostic criteria (Polman et al. 2011) were included in the study. The mean delay in reporting MS cases was taken into account (Esbjerg et al. 1999) by setting 31 December 2016 as the prevalence day. On this day, the total count of MS patients in the province of Pavia was 927, and the overall prevalence of MS was 169.4 per 100,000 inhabitants (95% CI: 158.8–180.6) (Bergamaschi et al. 2019).
Study area
The province of Pavia is one of the 12 provinces in the north-western Italian region of Lombardy. It covers an area of 2965 km2. The province is subdivided into 188 administrative areas called municipalities, and data from the 2017 inter-census ISTAT survey (http://demo.istat.it/pop2017/) reported 547,251 inhabitants (266,487 males and 280,764 females).
In the EU, local administrative units (LAUs) can be classified, for statistical stratification analysis purposes, according to their degree of urbanization (Eurostat 2018) as: 1-Cities (densely populated areas), 2-Towns and suburbs (intermediate density areas) and 3-Rural areas (thinly populated areas). Since Pavia and Vigevano are the only municipalities in the province of Pavia classifiable as cities (with 72,612 and 63,153 inhabitants, respectively), we re-classified the province’s LAUs into two categories: cities/towns and suburbs (LAU categories 1 and 2) vs rural areas (LAU category 3).
In addition, the deprivation index developed by Caranci et al. 2010 was used as a measure of socioeconomic status, calculated for municipalities and standardized at provincial level. This index is based on five main factors that represent the ‘multidimensionality of the social and material deprivation concept’ (namely, low education level, lack of employment, percentage of occupied rented houses, single-parent families, high housing density), and it provides accurate profiles of socioeconomic-health inequalities (Caranci et al. 2010).
Environmental data source
Spatial information on pollutants was gathered from the database of the European Monitoring and Evaluation Programme (EMEP), a scientifically based and policy-driven programme under the Convention on Long-range Transboundary Air Pollution, signed by member states of the United Nations Economic Commission for Europe. The EMEP aims to cut down polluting emissions from several sources.
The database is constituted by gridded emissions and concentrations of several air pollutants (SOx, NH3, CO, NOx, PM2.5, PM10) in 0.1° × 0.1° (long-lat) resolution for the whole time series, from the year 2000 onwards. The EMEP grid domain covers the geographical area between 30°N-82°N latitude and 30°W-90°E longitude. The emission distribution is based on reported gridded emissions. Additional data were also obtained from the Emissions Database for Global Atmospheric Research https://edgar.jrc.ec.europa.eu.
In accordance with the purpose of the study, gridded data on PM2.5 for the province of Pavia were extracted. The grid was further subdivided into the province’s 188 municipalities. For each municipality, average seasonal (winter/summer) concentrations of PM2.5 were extracted and grouped by tertiles: lowest tertile 6.74–25 μg/m3, middle tertile 25.1–31.9 μg/m3 and highest tertile 32–46.6 μg/m3. The EU currently stipulates an annual PM2.5 limit value of 25 μg/m3 (https://www.eea.europa.eu/themes/air/air-quality-concentrations/air-quality-standards).
Statistical analysis
Expected counts were calculated using direct standardization methods based on age- and sex-specific prevalence rates observed in the study area/period. Standardized morbidity ratios (SMRs) were calculated as ratios between observed and expected counts. To compare maximum likelihood estimates of prevalence rates and SMR differences, we used beta-binomial and gamma-Poisson distributions, respectively, in a hierarchical Bayesian fashion.
Cases were assigned the ecological characteristics (PM2.5 concentrations, degree of urbanization, deprivation index) of the geographical unit (municipality) they were resident in.
The SMR is unable to provide reliable spatial distribution of the disease on a small geographical grid, as high estimates are often obtained for small, sparsely inhabited areas. Hence, a Bayesian two-stage modelling approach was employed in order to draw MS relative risk (RR) maps. In the first stage, a Poisson log-linear model for count data was used (Agresti 2012); in the second stage, a log-linear hierarchical generalized mixed model (HGLMM) with reparametrized Besag York and Mollié random effects (BYM2) was implemented. The BYM2 method uses informative prior distributions (conditioning for the previous results) and both spatially structured and exchangeable error terms, thereby accounting for the degree of variability not explained by predictors in the first stage model (Besag et al. 1991; Riebler et al. 2016). The proposed final full Bayesian HGLMM model related the observed count of MS cases for each area to spatially structured random effects and PM2.5 levels, degree of urbanization and deprivation index.
We used medians of marginal posterior distributions of RR as estimators of the ‘true risk’. Moreover, to assess the presence of disease risk clusters and to evaluate our uncertainty about our risk estimates, we used posterior probabilities (PPs), PP being defined as the relative frequency of samples with RR > 1 for each municipality. The PP range (0–1) was divided into five intervals (< 0.10, 0.10–0.25, 0.25–0.75, 0.75–0.90, > 0.90). A PP value higher than or equal to 0.90 strongly indicated an ‘area of high risk’, while a PP value smaller than or equal to 0.10 strongly indicated an ‘area of low risk’. Similarly, PP values falling within the fourth interval (0.75–0.90) and second interval (0.10–0.25) merely indicated, respectively, areas of high and low risk of MS. As regards PP values in the central interval (0.25–0.75), the degree of evidence was considered insufficient to allow judgement. In conclusion, the map of PPs can be used to evaluate how confident we should be when we analyse area-specific RRs.
Calculations were carried out using statistical software, specifically the Stata 15, R version 3.6.1, Stan 2.17.3 and R packages.
Results
Distribution of PM2.5 concentrations in the province of Pavia
The mean concentrations of PM2.5 across the 7-year time series (2010–2016) were calculated for each municipality of the province.
The analysis focused on PM2.5 levels in winter, as it is the season with the highest pollutant concentrations. In terms of air pollution, the province of Pavia appeared to be divided into three strips (Fig. 1). The northernmost municipalities, being closer to the Milan area, demonstrated levels of PM2.5 within the range of the highest tertile. The southernmost municipalities in the rural area called Oltrepò appeared to be the least polluted, with levels of PM2.5 in the lowest tertile. Lying between these two strips, a central portion of the province showed intermediate PM2.5 levels, which were nevertheless above the European Commission threshold of 25 μg/m3. This portion of the territory includes both of the province’s two urban cities (Pavia and Vigevano).
Fig. 1.
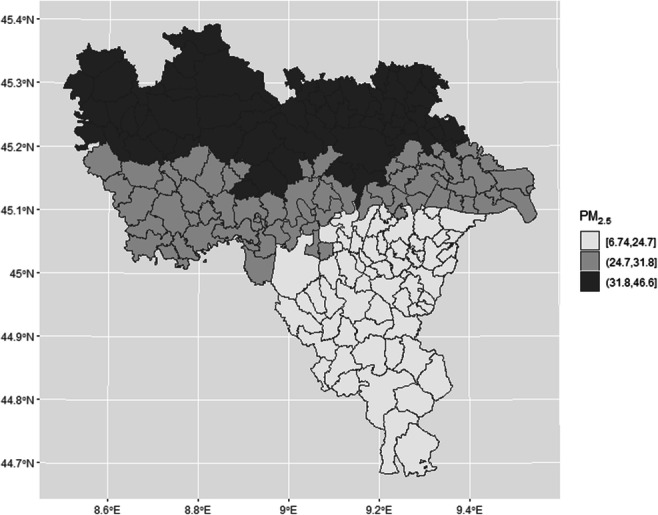
Average winter concentrations of PM2.5 across the 188 municipalities in the province of Pavia, divided into tertiles
PM2.5 and MS risk
Geographical comparisons of specific MS prevalence estimates and SMRs between the area around Pavia (identified as a high MS risk cluster) and the low polluted area of Oltrepò (identified as a low MS risk cluster) revealed differences. Specifically, Pavia and its hinterland (the city and the 10 peri-urban bordering municipalities) had an MS prevalence of 196.4 per 100,000 inhabitants (95% CI: 171.8–224.6), while Oltrepò had an MS prevalence of 157.9 per 100,000 inhabitants (95% CI: 137.7–181.5) (p = 0.027). The SMR of Pavia and its hinterland was 1.14 (95% CI: 1.04–1.25), while in the Oltrepò area the SMR was 0.96 (95% CI: 0.87–1.03) (p = 0.0045).
A Bayesian model analysed area-specific RR of MS in relation to levels of PM2.5 exposure (divided into tertiles), degree of urbanization and deprivation index. It showed that MS risk, after adjustment for urbanization degree and deprivation index, was higher among persons residing in areas with a PM2.5 concentration in the middle tertile with respect to persons living in areas with a PM2.5 in the lowest tertile. The former had a 29% increased risk (RR = 1.29, 95% CI: 1.11–1.49, p value = 0.0001). Overall, MS risk was found to be higher in urban than in rural areas (RR = 1.16, 95%CI: 1.04–1.30, p value = 0.003). Deprivation index did not modify MS risk.
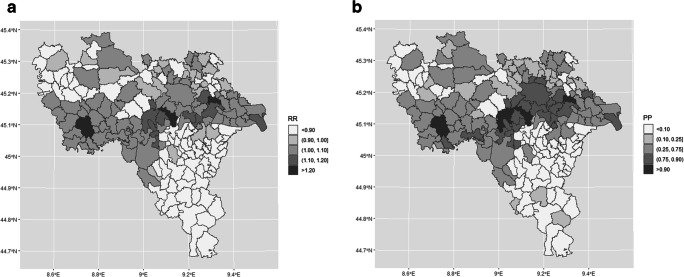
The geographical distributions of RR and PP across the province of Pavia are depicted in Fig. 2. As seen in Fig. 2a, the municipalities associated with higher RRs (darker tones) are distributed around the centre of the map, in the area corresponding to intermediate winter concentrations of PM2.5. A similar PP distribution can be seen in Fig. 2b. This strip of territory roughly extends from the city of Pavia to the western portion of the province. The less polluted rural area of Oltrepò to the south showed consistently low RR levels.
Fig. 2.
a Map of the relative risk (RR) of developing MS in association with average winter PM2.5 levels, after adjustment for degree of urbanization and deprivation index, across the 188 municipalities in the province of Pavia. Darker areas have higher RR values (gradients of grey). b Posterior probabilities (PPs) in association with average winter PM2.5 levels. The highest PPs were consistently identified in the areas also characterized by the highest RRs
Discussion
The present study aimed to add to current knowledge on the multifactorial aetiology of MS, by exploring through an advanced statistical approach, the relationship between MS risk distribution and environmental air pollution in a well-defined geographical area.
To date, few studies have explored the distribution of MS in small geographical areas (Bergamaschi et al. 2006; Cocco et al. 2011). However, the province of Pavia, in Lombardy, has previously been investigated from this perspective. In 1976, it had an estimated MS prevalence of 16 cases per 100,000 inhabitants (Bergamaschi et al. 2006), and in 2000, this was found to have risen to 94 cases per 100,000 inhabitants (Bergamaschi et al. 2006). The latest estimate (169.4 per 100,000) (Bergamaschi et al. 2019) strongly indicates an increasing prevalence of the disease, in accordance with other literature data (Bezzini and Battaglia 2017; Grassivaro et al. 2019).
The increasing prevalence of MS can be explained in part by improved detection and longer survival of cases, which reflects the growing availability and efficacy of disease-modifying therapies. However, the increase in MS prevalence could also be explained by greater exposure to risk factors.
The availability of reliable estimates of MS distribution in small geographical areas would be crucial for identifying spatial clusters of MS risk. These clusters could then serve as the basis for ecological studies focusing on genetic and environmental factors. Unfortunately, the application of conventional statistical methods is probably unsuitable for analyses of this type. The main problem with a classical frequentist approach is that it produces ‘crude’ maps of the distribution of a disease that are strongly affected by random variation. This holds especially true in the presence of a relatively small number of cases that are in turn divided into numerous subgroups, e.g. small geographical units as in the present study. Under these conditions, the resulting frequentist map of the geographical disease distribution resembles a patchwork, being strongly subject to random errors. Therefore, these maps are difficult to interpret because the real effects we are interested in could be masked by random noise, and conversely, random noise could be mistaken for real effects. This issue can be overcome by building geographical distribution maps with a Bayesian method that separately models the random and the true variations. When the random noise from the final Bayesian map is filtered out, the map shows the true underlying variations in MS prevalence and can finally be taken as a reliable estimate of area-specific prevalence rates.
The clusters that emerged from Bayesian disease mapping in our study suggest that people living in the area with the lowest levels of pollutants (first tertile) have a low MS risk. The area in question, known as Oltrepò, is a hilly territory rich in vineyards, with no industrial settlements. With respect to rural dwellers, individuals residing in urban areas were found to have an increased risk of MS independently of PM2.5 levels. The highest risk was observed among individuals residing in more densely populated areas, especially those around the city of Pavia, which showed intermediate PM2.5 levels (the middle strip in Fig. 1).
We should ask why the highest MS risk was not observed in the more polluted northernmost municipalities. This finding could be partially due to the limitations of our study, which correction via the deprivation index failed to address completely. First, our analysis was based on formal residential addresses, which do not always correspond to where subjects actually live. Second, we did not collect data on occupation and therefore failed to take into account the fact that residents of a certain municipality may not necessarily spend most of their outdoor time there. Third, no data on indoor pollution were collected. Finally, although we analysed the prevalence and RR of MS, incidence data may be more useful in evaluating the relationship between disease risk and PM exposure.
Conclusions
Albeit partly biased by the aforementioned issues, our study detected a lower risk of MS in individuals residing in rural areas with low levels of PM2.5, which suggests that reducing air pollution contributes to reducing the risk of developing MS.
In view of our epidemiological observation of an increasing trend in MS prevalence and our detection of clusters with an unexpected excess number of MS cases, we are currently carrying out specific analytical studies in these ‘high risk’ areas, with the aim of analysing multiple environmental factors, air pollution included, possibly related to the heterogeneous distribution of MS risk.
Acknowledgements
Open access funding provided by Università degli Studi di Pavia within the CRUI-CARE Agreement.
Authors’ contributions
All authors provided critical feedback and helped shape the research, analysis and manuscript. RB and CM conceived the original idea; RB and MCM took the lead in writing the manuscript, together with CM who designed the experimental plan. RB, GM and LG contributed to sample preparation and revision of medical records collection; LG took care of the quality control of the data. MCM, CM and EP verified the analytical methods. MCM, LT and LG performed the computations; LT verified the theoretical formalism and performed the numerical simulations; CM and RB supervised the findings of this work. RB, MCM, CM, GM, LT and EP contributed to the interpretation of the results.
All authors participated to the revision of the final manuscript and approved it.
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Compliance with ethical standards
Conflict of interest
Bergamaschi R has received funding for congress/travel/accommodation expenses for scientific meetings and honoraria for speaking from Almirall, Bayer Schering, Biogen-Idec, Merck Serono, Novartis, Sanofi-Genzyme and Teva; he has served on scientific Advisory Boards for Almirall, Biogen, Merck Serono, Novartis, Sanofi-Genzyme, Teva; he has received research grants for his department from Biogen, Merck Serono, Sanofi-Genzyme and Teva.
Mallucci G has received support for congress/travel/accommodation expenses related to scientific meetings from Biogen, Genzyme, Merck Serono, Novartis, Roche, Sanofi-Aventis and Teva; he has received research grants for his department from Biogen and has served on scientific advisory boards for Biogen, Genzyme and Merck Serono.
Monti MC, Trivelli L, Gerosa L, Pisoni E and Montomoli C. have nothing to disclose.
Ethics approval
The study was carried out according to the Declaration of Helsinki and its later amendments following the national ethical guidelines for ecological studies with retrospective data collection.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Maria Cristina Monti and Leonardo Trivelli contributed equally to this work.
References
- Agresti A (2012) Categorical data analysis, 3rd edition, Wiley
- Angelici L, Piola M, Cavalleri T, Randi G, Cortini F, Bergamaschi R, Baccarelli AA, Bertazzi PA, Pesatori AC, Bollati V. Effects of particulate matter exposure on multiple sclerosis hospital admission in Lombardy region, Italy. Environ Res. 2016;145:68–73. doi: 10.1016/j.envres.2015.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ascherio A, Munger KL (2007) Environmental risk factors for multiple sclerosis. Part II: Noninfectious factors. Ann Neurol 1(6):504–513. 10.1002/ana.21141 [DOI] [PubMed]
- Bergamaschi R, Cortese A, Pichiecchio A, Berzolari FG, Borrelli P, Mallucci G, Bollati V, Romani A, Nosari G, Villa S, Montomoli C. Air pollution is associated to the multiple sclerosis inflammatory activity as measured by brain MRI. Mult Scler J. 2018;24:1578–1584. doi: 10.1177/1352458517726866. [DOI] [PubMed] [Google Scholar]
- Bergamaschi R, Monti MC, Trivelli L, Introcaso VP, Mallucci G, Borrelli P, Gerosa L, Montomoli C. Increased prevalence of multiple sclerosis and clusters of different disease risk in northern Italy. Neurol Sci. 2019;41:1089–1095. doi: 10.1007/s10072-019-04205-7. [DOI] [PubMed] [Google Scholar]
- Bergamaschi R, Montomoli C, Candeloro E, Monti MC, Cioccale R, Bernardinelli L, Fratino P, Cosi V, PREMS (Pavia REgister of Multiple Sclerosis) Group Bayesian mapping of multiple sclerosis prevalence in the province of Pavia, northern Italy. J Neurol Sci. 2006;244:127–131. doi: 10.1016/J.JNS.2006.01.013. [DOI] [PubMed] [Google Scholar]
- Besag J, York J, Mollié A. Bayesian image restoration, with two applications in spatial statistics. Ann Inst Stat Math. 1991;43:1–20. doi: 10.1007/BF00116466. [DOI] [Google Scholar]
- Bezzini D, Battaglia MA (2017) Multiple sclerosis epidemiology in Europe. Adv Exp Med Biol 958:141–159. 10.1007/978-3-319-47861-6_9 [DOI] [PubMed]
- Calderón-Garcidueñas L, Azzarelli B, Acuna H, Garcia R, Gambling TM, Osnaya N, Monroy S, del Rosario Tizapantzi M, Carson JL, Villarreal-Calderon A, Rewcastle B. Air pollution and brain damage. Toxicol Pathol. 2002;30:373–389. doi: 10.1080/01926230252929954. [DOI] [PubMed] [Google Scholar]
- Caranci N, Biggeri A, Grisotto L, Pacelli B, Spadea T, Costa G (2010) L’indice di deprivazione italiano a livello di sezione di censimento: definizione, descrizione e associazione con la mortalità [The Italian deprivation index at census block level: definition, description and association with general mortality]. Epidemiol Prev 34(4):167–176 [PubMed]
- Cocco E, Sardu C, Massa R, Mamusa E, Musu L, Ferrigno P, Melis M, Montomoli C, Ferretti V, Coghe G, Fenu G, Frau J, Lorefice L, Carboni N, Contu P, Marrosu MG. Epidemiology of multiple sclerosis in south-western Sardinia. Mult Scler J. 2011;17:1282–1289. doi: 10.1177/1352458511408754. [DOI] [PubMed] [Google Scholar]
- Esbjerg S, Keiding N, Koch-Henriksen N. Reporting delay and corrected incidence of multiple sclerosis. Stat Med. 1999;18:1691–1706. doi: 10.1002/(SICI)1097-0258(19990715)18:13<1691::AID-SIM160>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- Eurostat (2018) Degree of urbanisation classification - 2011 revision - Statistics Explained. https://ec.europa.eu/eurostat/statisticsexplained/pdfscache/31486.pdf. Accessed 21 April 2020
- Grassivaro F, Puthenparampil M, Pengo M, Saiani M, Venturini M, Stropparo E, Perini P, Rinaldi F, Freddi N, Cadaldini M, Colledan L, Piccinno MG, Pedrazzoli E, Gallo P. Multiple sclerosis incidence and prevalence trends in the province of Padua, Northeast Italy, 1965–2018. Neuroepidemiology. 2019;52:41–46. doi: 10.1159/000493857. [DOI] [PubMed] [Google Scholar]
- Gregory AC, Shendell DG, Okosun IS, Gieseker KE. Multiple sclerosis disease distribution and potential impact of environmental air pollutants in Georgia. Sci Total Environ. 2008;396:42–51. doi: 10.1016/j.scitotenv.2008.01.065. [DOI] [PubMed] [Google Scholar]
- Hosseinpanah F, Pour SH, Heibatollahi M, et al. The effects of air pollution on vitamin D status in healthy women: a cross sectional study. BMC Public Health. 2010;10:519. doi: 10.1186/1471-2458-10-519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeanjean M, Bind M-A, Roux J, Ongagna JC, de Sèze J, Bard D, Leray E. Ozone, NO2 and PM10 are associated with the occurrence of multiple sclerosis relapses. Evidence from seasonal multi-pollutant analyses. Environ Res. 2018;163:43–52. doi: 10.1016/J.ENVRES.2018.01.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kooij G, van Horssen J, de Lange ECM, Reijerkerk A, van der Pol SMA, van het Hof B, Drexhage J, Vennegoor A, Killestein J, Scheffer G, Oerlemans R, Scheper R, van der Valk P, Dijkstra CD, de Vries HE. T lymphocytes impair P-glycoprotein function during neuroinflammation. J Autoimmun. 2010;34:416–425. doi: 10.1016/j.jaut.2009.10.006. [DOI] [PubMed] [Google Scholar]
- Koski K, Oikonen M, Lilius E-M, et al. Ambient air quality and occurrence of multiple sclerosis relapse. Neuroepidemiology. 2003;22:95–99. doi: 10.1159/000067108. [DOI] [PubMed] [Google Scholar]
- Li J, Baud O, Vartanian T, Volpe JJ, Rosenberg PA. Peroxynitrite generated by inducible nitric oxide synthase and NADPH oxidase mediates microglial toxicity to oligodendrocytes. Proc Natl Acad Sci U S A. 2005;102:9936–9941. doi: 10.1073/pnas.0502552102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minagar A, Alexander JS. Blood-brain barrier disruption in multiple sclerosis. Mult Scler. 2003;9:540–549. doi: 10.1191/1352458503ms965oa. [DOI] [PubMed] [Google Scholar]
- Odoardi F, Sie C, Streyl K, Ulaganathan VK, Schläger C, Lodygin D, Heckelsmiller K, Nietfeld W, Ellwart J, Klinkert WEF, Lottaz C, Nosov M, Brinkmann V, Spang R, Lehrach H, Vingron M, Wekerle H, Flügel-Koch C, Flügel A. T cells become licensed in the lung to enter the central nervous system. Nature. 2012;488:675–679. doi: 10.1038/nature11337. [DOI] [PubMed] [Google Scholar]
- Oppenheim HA, Lucero JA, Guyot AC, Herbert LM, McDonald JD, Mabondzo A, Lund AK. Exposure to vehicle emissions results in altered blood brain barrier permeability and expression of matrix metalloproteinases and tight junction proteins in mice. Part Fibre Toxicol. 2013;10:62. doi: 10.1186/1743-8977-10-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polman CH, Reingold SC, Banwell B, Clanet M, Cohen JA, Filippi M, Fujihara K, Havrdova E, Hutchinson M, Kappos L, Lublin FD, Montalban X, O'Connor P, Sandberg-Wollheim M, Thompson AJ, Waubant E, Weinshenker B, Wolinsky JS. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol. 2011;69:292–302. doi: 10.1002/ana.22366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riebler A, Sørbye SH, Simpson D, Rue H (2016) An intuitive Bayesian spatial model for disease mapping that accounts for scaling. Stat Methods Med Res 25(4):1145-1165. 10.1177/0962280216660421 [DOI] [PubMed]
- Roux J, Bard D, Le Pabic E, et al. Air pollution by particulate matter PM10 may trigger multiple sclerosis relapses. Environ Res. 2017;156:404–410. doi: 10.1016/j.envres.2017.03.049. [DOI] [PubMed] [Google Scholar]
- Tateo F, Grassivaro F, Ermani M, Puthenparampil M, Gallo P. PM2.5 levels strongly associate with multiple sclerosis prevalence in the province of Padua, Veneto region, north-east Italy. Mult Scler J. 2019;25:1719–1727. doi: 10.1177/1352458518803273. [DOI] [PubMed] [Google Scholar]
- Thompson AJ, Baranzini SE, Geurts J, Hemmer B, Ciccarelli O. Multiple sclerosis. Lancet. 2018;391:1622–1636. doi: 10.1016/S0140-6736(18)30481-1. [DOI] [PubMed] [Google Scholar]
- Veldhoen M, Hirota K, Westendorf AM, Buer J, Dumoutier L, Renauld JC, Stockinger B. The aryl hydrocarbon receptor links TH17-cell-mediated autoimmunity to environmental toxins. Nature. 2008;453:106–109. doi: 10.1038/nature06881. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.