Key Points
Question
Is there an association between fluoroquinolone use and the rate of aortic aneurysms in US adults?
Findings
This cohort study of 47 596 545 antibiotic prescription fills among US adults aged 18 to 64 years found an increased rate of aortic aneurysms within 90 days after fluoroquinolone use compared with alternative antibiotic use, and when stratified by age, an increased incidence of aneurysms was observed in adults 35 years or older. No differences were seen when stratifying by sex and common comorbidities (eg, hypertension and hyperlipidemia); rather, the association of fluoroquinolone use with the aneurysm rate was consistent, suggesting a risk of drug class among both healthy and unhealthy individuals.
Meaning
The results of this study suggested that fluoroquinolones should be used with caution among individuals aged 35 years or older, regardless of sex or comorbidities.
Abstract
Importance
Although fluoroquinolones are commonly prescribed antibiotics in the US, recent international studies have shown an increased risk of aortic aneurysm and dissection after fluoroquinolone use, leading to US Food and Drug Administration warnings limiting use for high-risk patients. It is unclear whether these data are true for the US population and who is truly high risk.
Objective
To assess aortic aneurysm and dissection risks in a heterogeneous US population after fluoroquinolone use.
Design, Setting, and Participants
Prescription fills for fluoroquinolones or a comparator antibiotic from 2005 to 2017 among commercially insured individuals aged 18 to 64 years were identified in this retrospective analysis of MarketScan health insurance claims. This cohort study included 27 827 254 US adults (47 596 545 antibiotic episodes), aged 18 to 64 years, with no known previous aortic aneurysm or dissection, no recent antibiotic exposure, and no recent hospitalization.
Exposures
Outpatient fill of an oral fluoroquinolone or comparator antibiotic (amoxicillin-clavulanate, azithromycin, cephalexin, clindamycin, and sulfamethoxazole-trimethoprim).
Main Outcomes and Measures
The 90-day incidence of aortic aneurysm and dissection. Inverse probability of treatment weighting in Cox regression was used to estimate the association between fluoroquinolone fill and 90-day aneurysm incidence. Interaction terms were used to assess the association of known risk factors (ie, sex, age, and comorbidities) with aneurysm after fluoroquinolone use. Data analysis was performed March 2019 to May 2020.
Results
Of 47 596 545 prescription fills, 9 053 961 (19%) were fluoroquinolones and 38 542 584 (81%) were comparator antibiotics. The median (interquartile range) age of adults with fluoroquinolone fills was 47 (36-57) years vs 43 (31-54) years with comparator antibiotic fills. Women comprised 61.3% of fluoroquinolone fills and 59.5% of comparator antibiotic fills. Before weighting, the 90-day incidence of newly diagnosed aneurysm was 7.5 cases per 10 000 fills (6752 of 9 053 961) after fluoroquinolones compared with 4.6 cases per 10 000 fills (17 627 of 38 542 584) after comparator antibiotics. After weighting for demographic characteristics and comorbidities, fluoroquinolone fills were associated with increased incidence of aneurysm formation (hazard ratio [HR], 1.20; 95% CI, 1.17-1.24). More specifically, compared with comparator antibiotics, fluoroquinolone fills were associated with increased 90-day incidence of abdominal aortic aneurysm (HR, 1.31; 95% CI, 1.25-1.37), iliac artery aneurysm (HR, 1.60; 95% CI, 1.33-1.91), and other abdominal aneurysm (HR, 1.58; 95% CI, 1.39-1.79), and adults were more likely to undergo aneurysm repair (HR, 1.88; 95% CI, 1.44-2.46). When stratified by age, all adults 35 years or older appeared at increased risk (18-34 years: HR, 0.99 [95% CI, 0.83-1.18]; 35-49 years: HR, 1.18 [95% CI, 1.09-1.28]; 50-64 years: HR, 1.24 [95% CI, 1.19-1.28]; P = .04).
Conclusions and Relevance
This study found that fluoroquinolones were associated with increased incidence of aortic aneurysm formation in US adults. This association was consistent across adults aged 35 years or older, sex, and comorbidities, suggesting fluoroquinolone use should be pursued with caution in all adults, not just in high-risk individuals.
This large cohort study uses data in the MarketScan Commercial Database to assess whether there is an association between fluoroquinolone use and the rate of aortic aneurysm overall, stratified by age, sex, and common comorbidities, in a heterogenous population of US adults.
Introduction
Fluoroquinolones are one of the most commonly prescribed antibiotic classes in the United States.1,2 Because of the broad-spectrum coverage, easy dosing, and historically low adverse effect profile, this antibiotic class is frequently prescribed.3,4 Recent international studies have called into question the safety of this class of antibiotics, with reports showing a possible association with acute aortic aneurysm and dissection development.5,6,7 Aortic aneurysms are a common disease, seen in 4% to 8% of the adult population, with mortality rates up to 53% in the US when associated with rupture.8 Therefore, aortic aneurysms require early diagnosis, close follow-up, and aggressive medical management to slow progression.7,9,10 Current known risk factors for aortic aneurysm development and rupture include age, male sex, tobacco use, atherosclerosis, hypertension, and family history.11
Recent studies have probed the safety of this antibiotic class by assessing populations in Taiwan, Sweden, and Canada—all countries with populations different from the United States.5,6,7 Although there are no US clinical studies exploring the association between fluoroquinolones and aortic aneurysm formation, the US Food and Drug Administration (FDA) warning was updated in 2018 to include that fluoroquinolone use in high-risk populations should be limited and to define high risk as the presence of known cardiovascular disease or aneurysms, certain genetic disorders that involve blood vessels, high blood pressure, and the elderly.12 Despite these studies and warnings, fluoroquinolones remain the most commonly prescribed antibiotic in long-term care facilities, suggesting minimal practice change.13 There is a paucity of data assessing the US population and the true extent of what defines those at highest risk.
The aim of this study was to evaluate the incidence of aortic aneurysm and dissection formation after fluoroquinolone prescription fills in a large cohort of US adults enrolled in commercial insurance plans. We hypothesized that fluoroquinolone use would increase the incidence of aortic aneurysm formation regardless of age and comorbidities.
Methods
Study Design and Population
This observational study was conducted using data from the IBM MarketScan Commercial Database. These data include health insurance claims across the continuum of care (eg, inpatient, outpatient, and outpatient pharmacy) as well as enrollment data from large employers and health plans across the United States that provide private health care coverage for 40 million employees, their spouses, and dependents each year. This administrative claims database includes a variety of fees for services, preferred provider organizations, and capitated health plans.14 After review by the institutional review board committee of the University of North Carolina at Chapel Hill, this study was determined to be exempt from review because a limited data set with inability to collect identifying patient data was used, and the requirement to obtain informed consent was waived.
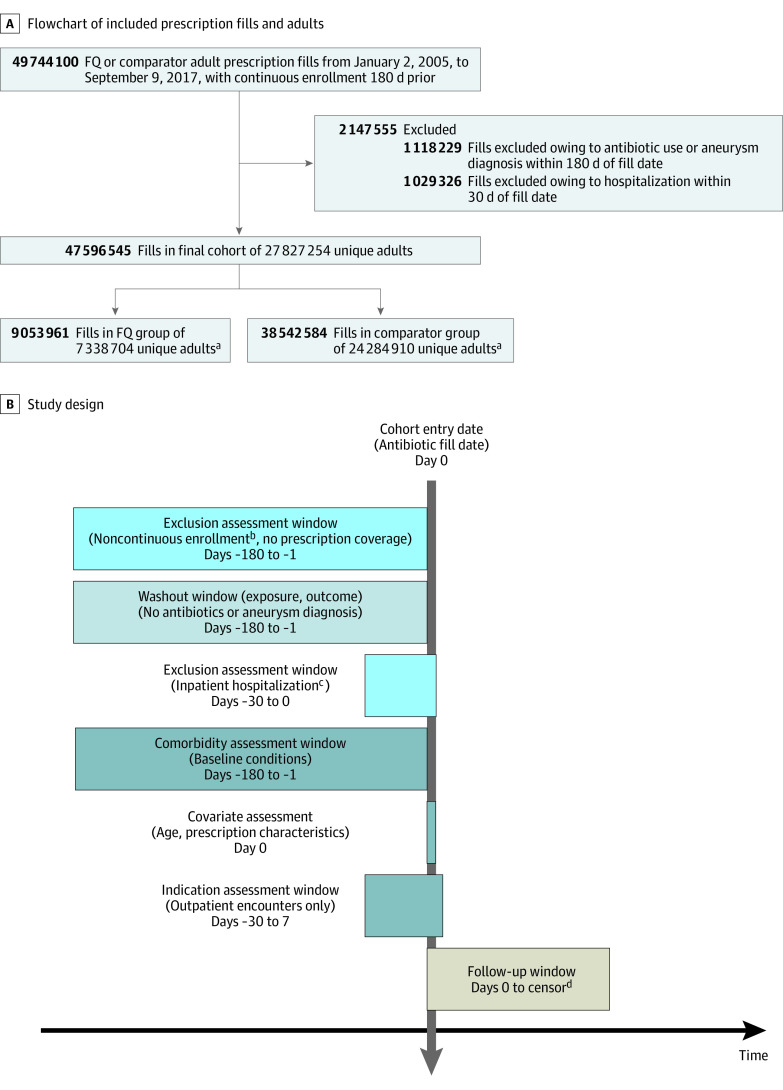
All prescription fills for either an oral fluoroquinolone or a comparator antibiotic (amoxicillin-clavulanate, azithromycin, cephalexin, clindamycin, and sulfamethoxazole-trimethoprim) between January 1, 2005, and September 30, 2017 (prior to US FDA warning), among adults aged 18 to 64 years were eligible for inclusion (eTable 1 in the Supplement). Comparator antibiotics were chosen based on commonly prescribed antibiotics for similar indications as those in the fluoroquinolone class. Of note, doxycycline was excluded from our comparator antibiotics because it is a known matrix metalloproteinase inhibitor.15 Because prior international studies have found that fluoroquinolone use only was associated with immediate and short-term aneurysm development (within 60-90 days from start of treatment),6,7 adults could contribute multiple episodes of antibiotic use and follow-up; each antibiotic episode was required to meet all inclusion and exclusion criteria to be included in the analysis (Figure 1). For each antibiotic episode, adults were required to have continuous enrollment in their insurance plan (including prescription coverage), with 8-day grace periods, for at least 180 days prior to their prescription fill (49 744 100 eligible fills). Antibiotic episodes in which a prior antibiotic fill (fluoroquinolone or comparator) or a preexisting diagnosis of aneurysm or dissection occurred within that 180-day lookback window were excluded (1 118 229 fills). Individuals were also excluded if they were hospitalized within 30 days of their index fill date (including the day of their prescription fill) (1 029 326 fills).
Figure 1. Flow Diagram and Study Design to Evaluate Episodes of Fluoroquinolone (FQ) or Comparator Antibiotic Fills in US Adults (Aged 18-64 Years) From 2005 to 2017.
A, Flow diagram. B, Study design; the color coding of the boxes is as follows: exclusion criteria (light blue), washout (light gray), covariates (dark gray), and follow-up (tan). The varying sizes of the boxes are due to the fact that they are to scale for the duration of the period (180-day exclusion period longer than 30-day washout period).
aIndividuals could contribute episodes to both FQ and the comparator group.
bUp to 8-day gaps in insurance enrollment allowed.
cAdults were excluded if they were admitted or discharged from a hospital within 30 days of their index fill date.
dFollowed up until insurance disenrollment, second antibiotic prescription fill, or end of follow-up 90 days after initial fill date.
Antibiotic indication was identified by capturing all outpatient International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) and International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) diagnoses classified as either “always” or “sometimes” treated with oral antibiotics according to the Chua et al16 classification scheme, documented between 30 days prior and 7 days after the index fill date. Diagnoses within 1 week after prescription fill were included to capture patients who were provided a prescription prior to their visit with a clinician (eg, patient had an antibiotic prescription called in prior to the clinic visit). Indications were then classified into discrete categories (eg, gastrointestinal or bacterial pneumonia) (eTable 2 in the Supplement). If multiple potential indications were identified, the diagnosis recorded in closest proximity to the index fill date was selected; priority was given to indications occurring before the fill date (eg, if a patient had an indication 2 days prior to fill and 2 days after fill, the diagnosis before the fill date was selected). Antibiotic fills without an identified indication were stratified into 2 groups—those with only noninfectious diagnoses (“no indication identified”) and those with no health care claims (“no billed health care”).
Adults were then followed up from their antibiotic fill date until insurance disenrollment, a second prescription fill for a fluoroquinolone or comparator drug (irrespective of their first medication type), or for 90 days to identify aneurysmal events.
Primary Outcome
The primary outcome was the 90-day incidence of aneurysm and aortic dissection per 10 000 antibiotic fills (eTable 3 in the Supplement). Patients could have diagnoses for different aneurysms during their follow-up period (ie, a diagnosis of an abdominal aortic and an iliac artery aneurysm could both be identified during the same follow-up period).
Comorbidity Assessment
Comorbidities were measured during the 180-day lookback window and identified using ICD-9-CM and ICD-10-CM codes as well as known comorbidity standard prescriptions (eTables 1 and 2 in the Supplement). The ICD-9-CM codes were identified using validated algorithms (eg, Deyo et al Charlson Comorbidity Index algorithm17) or clinical review; the ICD-10-CM codes were identified using forward and backward mapping to the General Equivalence Mappings published by the Centers for Medicare and Medicaid Services. Patients were considered users of comorbid condition medications if they had at least 1 prescription fill during the lookback window.
Statistical Analysis
Differences in demographic characteristics, comorbidities, and antibiotic indications between fluoroquinolones and the comparators were assessed using absolute standardized differences. The use of the standardized difference is preferable to the use of traditional tests (eg, χ2 or t test) because the former is independent of sample size.18 An absolute difference greater than 0.20 was considered a meaningful difference between groups.
Inverse probability of treatment weighting (IPTW) in Cox proportional hazards regression was used to estimate the association of fluoroquinolones compared with that of an antibiotic comparator with the 90-day incidence of aneurysm and aortic dissection. In brief, multivariable logistic regression, adjusting for age at fill, sex, Charlson Comorbidity Index components, other comorbidities, other medication use, region, indication for antibiotic use, and year and quarter of fill were used to estimate the probability (or propensity) of receiving a fluoroquinolone (or comparator). Age was modeled as a restricted quadratic spline. The propensity score was then scaled by the overall probability of receiving a fluoroquinolone (or comparator) in the cohort. The final IPTW ranged from 0.20 to 19.68. Robust sandwich estimators were used to account for weighting in the Cox models and multiple antibiotic fills among individuals.
Several sensitivity analyses were performed. First, fluoroquinolones were stratified by specific medication (ciprofloxacin, levofloxacin, and moxifloxacin) to assess whether the association was consistent across the entire fluoroquinolone class. A new IPTW for each medication fill was calculated using generalized logistic regression—which modeled the odds of receiving ciprofloxacin, levofloxacin, moxifloxacin or active comparator—and was then analyzed using the same methods described above. Second, interaction terms were used to assess potential effect measure modification (ie, is the incidence of aneurysm after fluoroquinolones different across subgroups) across sex, age (categorized as 18-34, 35-49, and 50-64 years), and comorbidity status (diabetes, hypertension, and hyperlipidemia). Third, we removed subsequent antibiotic fills (fluoroquinolone or comparator) that occurred after an individual had an antibiotic episode when they received a diagnosis of aneurysm to prevent overestimation of risk after antibiotic exposure. For example, if 1 adult contributed 4 antibiotic episodes and received a diagnosis of aneurysm during the follow-up of their second antibiotic, their third and fourth fills were excluded. If an adult contributed 4 antibiotic episodes and never received a diagnosis of aneurysm, no fills were removed. Fourth, we conducted an analysis in which we excluded patients with unknown indications (“no indication identified” and “no billed health care”).
A 2-sided P < .05 was considered statistically significant. All analyses were performed using SAS, version 9.3 (SAS Institute Inc) from March 2019 to May 2020.
Results
Patient Cohort
Overall, 47 596 545 antibiotic episodes (27 827 254 unique adults) were included. Of these, 9 053 961 (19%) were fluoroquinolone episodes, and 31 584 407 of 47 596 545 (66%) had complete 90-day follow-up (median, 90 days; interquartile range [IQR], 57-90 days). The mean number of included antibiotic fills per adult was 1.7 (median, 1; IQR, 1-2; range, 1-18), and among adults with multiple fills, the median time between included fills was 442 days (IQR, 306-669 days).
Adults with fluoroquinolone fills had a median (IQR) age of 47 (36-57) years vs a median (IQR) age of 43 (31-54) years in our comparator group (absolute standardized difference, 0.22; Table). Women comprised 61.3% of fluoroquinolone fills and 59.5% of comparator antibiotic fills. Minimal differences were seen across sex or comorbidities. Although the distribution of indications for fluoroquinolones and the comparator antibiotics differed before weighting, top antibiotic indications for both groups were the same: upper respiratory tract infection, urinary tract infection, and skin or soft tissue infection. After weighting, minimal differences were seen between groups (Table).
Table. Demographic and Clinical Characteristics of Patients With Treatment Episodes of Oral Fluoroquinolones or Comparator Antibiotics, Before and After Inverse Probability of Treatment Weighting.
| Characteristic | Crude | Inverse probability of treatment weighting | ||||
|---|---|---|---|---|---|---|
| No. (%) | Absolute standardized difference | No. (%) | Absolute standardized difference | |||
| Fluoroquinolones | Comparator | Fluoroquinolones | Comparator | |||
| Episodes, No. | 9 053 961 | 38 542 584 | NA | 9 064 727 | 38 545 278 | NA |
| Unique adults, No. | 7 388 704 | 24 284 910 | NA | NA | NA | NA |
| Age, median (IQR), y | 47 (36-57) | 43 (31-54) | 0.22 | 44 (32-55) | 44 (32-55) | 0.01 |
| Age categories, y | 0.20 | 0.01 | ||||
| 18-34 | 2 103 974 (23.2) | 11 993 236 (31.1) | NA | 2 748 149 (30.3) | 11 433 139 (29.7) | NA |
| 35-49 | 2 897 767 (32.0) | 12 595 603 (32.7) | 2 918 695 (32.2) | 12 537 584 (32.5) | ||
| 50-64 | 4 052 220 (36.2) | 13 953 745 (36.2) | 3 397 883 (37.5) | 14 574 555 (37.8) | ||
| Female | 5 550 141 (61.3) | 22 933 992 (59.5) | 0.04 | 5 522 620 (60.9) | 23 090 705 (59.9) | 0.02 |
| Indicationa | ||||||
| Upper respiratory tract infectionb | 1 470 567 (16.2) | 9 982 223 (25.9) | 0.24 | 2 097 414 (23.1) | 9 266 079 (24.0) | 0.02 |
| Skin/soft tissue/bone/lymph | 354 714 (3.9) | 3 349 658 (8.7) | 0.20 | 702 577 (7.8) | 2 999 538 (7.8) | <0.01 |
| Urethritis/UTI | 1 647 161 (18.2) | 1 489 697 (3.9) | 0.47 | 594 361 (6.6) | 2 531 527 (6.6) | <0.01 |
| Streptococcal/staphylococcusc | 37 835 (0.4) | 599 507 (1.6) | 0.12 | 116 425 (1.3) | 515 996 (1.3) | <0.01 |
| Gastrointestinal tract | 255 275 (2.8) | 182 493 (0.5) | 0.19 | 83 103 (0.9) | 354 847 (0.9) | <0.01 |
| Pneumonia | 195 332 (2.2) | 254 738 (0.7) | 0.13 | 84 585 (0.9) | 362 293 (0.9) | <0.01 |
| Pyelonephritis | 185 882 (2.1) | 57 589 (0.2) | 0.18 | 46 536 (0.5) | 206 112 (0.5) | <0.01 |
| Ocular | 41 249 (0.5) | 39 828 (0.1) | 0.07 | 15 966 (0.2) | 67 930 (0.2) | <0.01 |
| Cholecystitis/BTI | 19 713 (0.2) | 27 261 (0.1) | 0.04 | 9048 (0.1) | 38 279 (0.1) | <0.01 |
| Appendicitis | 8040 (0.1) | 10 266 (<0.1) | 0.03 | 3499 (<0.1) | 14 867 (<0.1) | <0.01 |
| Syphilis | 528 (<0.1) | 2205 (<0.1) | 0.00 | 520 (<0.1) | 2212 (<0.1) | <0.01 |
| Dental | 16 (<0.1) | 970 (<0.1) | 0.01 | 175 (<0.1) | 799 (<0.1) | <0.01 |
| Other indication | 21 330 (0.2) | 68 697 (0.2) | 0.01 | 17 074 (0.2) | 72 816 (0.2) | <0.01 |
| No indication identified | 3 816 829 (42.2) | 14 765 226 (38.3) | 0.08 | 3 561 058 (39.3) | 15 052 683 (39.1) | <0.01 |
| No billed health care | 999 490 (11.0) | 7 712 227 (20.0) | 0.25 | 1 732 388 (19.1) | 7 059 301 (18.3) | 0.02 |
| CCI, median (IQR), scored | 0 (0-0) | 0 (0-0) | 0.11 | 0 (0-0) | 0 (0-0) | <0.01 |
| Other comorbidities | ||||||
| Hypertension | 2 615 365 (28.9) | 9 311 008 (24.2) | 0.11 | 2 278 195 (25.1) | 9 658 144 (25.1) | <0.01 |
| Medically managed | 2 333 441 (25.8) | 8 348 296 (21.7) | NA | 2 030 844 (22.4) | 8 656 862 (22.5) | NA |
| Diagnosis only | 281 924 (3.1) | 962 712 (2.5) | 247 351 (2.7) | 1 001 282 (2.6) | ||
| Hyperlipidemia | 2 117 481 (23.4) | 7 360 818 (19.1) | 0.11 | 1 812 421 (20.0) | 7 667 094 (19.9) | 0.01 |
| Medically managed | 1 393 833 (15.4) | 4 860 737 (12.6) | NA | 1 177 331 (13.0) | 5 073 006 (13.2) | NA |
| Diagnosis only | 723 648 (8.0) | 2 500 081 (6.5) | 635 090 (7.0) | 2 594 088 (6.7) | ||
| Diabetes | 907 949 (10.1) | 3 115 705 (8.1) | 0.07 | 773 371 (8.5) | 3 265 212 (8.5) | <0.01 |
| Insulin-requiring | 196 253 (2.2) | 655 017 (1.7) | NA | 165 394 (1.8) | 692 038 (1.8) | NA |
| Oral medication | 524 049 (5.8) | 1 841 790 (4.8) | 451 478 (5.0) | 1 917 073 (5.0) | ||
| Diagnosis only | 187 647 (2.1) | 618 898 (1.6) | 156 499 (1.7) | 656 101 (1.7) | ||
| Atherosclerosis | 192 639 (2.1) | 614 754 (1.6) | 0.04 | 155 917 (1.7) | 655 940 (1.7) | <0.01 |
| Arrhythmia | 147 294 (1.6) | 533 387 (1.4) | 0.02 | 132 413 (1.5) | 553 536 (1.4) | <0.01 |
| Rheumatic disease, chronic | 97 094 (1.1) | 316 029 (0.8) | <0.01 | 6750 (0.1) | 28 128 (0.1) | <0.01 |
| Coronary artery disease | 39 476 (0.4) | 126 804 (0.3) | 0.02 | 32 349 (0.4) | 135 128 (0.4) | <0.01 |
| Angina | 32 265 (0.4) | 101 160 (0.3) | 0.02 | 26 055 (0.3) | 108 618 (0.3) | <0.01 |
| VTE | 29 557 (0.3) | 101 767 (0.3) | 0.01 | 26 476 (0.3) | 107 323 (0.3) | <0.01 |
| Cardiomyopathy | 23 717 (0.3) | 87 243 (0.2) | 0.01 | 21 535 (0.2) | 90 093 (0.2) | <0.01 |
| Pulmonary disease | 19 792 (0.2) | 64 732 (0.2) | 0.01 | 16 790 (0.2) | 69 031 (0.2) | <0.01 |
| Other valve diseases | 11 424 (0.1) | 38 854 (0.1) | 0.01 | 9759 (0.1) | 40 906 (0.1) | <0.01 |
| History of CVA | 3902 (<0.1) | 12 229 (<0.1) | <0.01 | 3186 (<0.1) | 13 189 (<0.1) | <0.01 |
| Pericarditis | 2820 (<0.1) | 14 080 (<0.1) | 0.01 | 3679 (<0.1) | 15 407 (<0.1) | <0.01 |
| Nonaortic, non–large-vessel aneurysmal diseasee | 3524 (<0.1) | 11 802 (<0.1) | <0.01 | 2964 (<0.1) | 12 472 (<0.1) | <0.01 |
| Endocarditis | 1638 (<0.1) | 6222 (<0.1) | <0.01 | 1582 (<0.1) | 6396 (<0.1) | <0.01 |
| Vasculopathy | 1133 (<0.1) | 3460 (<0.1) | <0.01 | 906 (<0.1) | 3762 (<0.1) | <0.01 |
| Rheumatic disease, acute | 480 (<0.1) | 1719 (<0.1) | <0.01 | 443 (<0.1) | 1789 (<0.1) | <0.01 |
| Myocarditis | 234 (<0.1) | 834 (<0.1) | <0.01 | 206 (<0.1) | 868 (<0.1) | <0.01 |
| Other medications | ||||||
| Corticosteroids (inhaled) | 826 885 (9.1) | 3 089 208 (8.0) | 0.04 | 763 616 (8.4) | 3 182 410 (8.3) | <0.01 |
| β-Agonist inhalants | 537 377 (5.9) | 1 959 141 (5.1) | 0.04 | 486 955 (5.4) | 2 030 644 (5.3) | <0.01 |
| Antiplatelets | 123 657 (1.4) | 400 895 (1.0) | 0.03 | 100 540 (1.1) | 425 551 (1.1) | <0.01 |
| Anticholinergics (inhaled) | 96 166 (1.1) | 250 753 (0.7) | 0.04 | 68 003 (0.8) | 284 437 (0.7) | <0.01 |
| Anticoagulants | 90 242 (1.0) | 382 727 (1.0) | <0.01 | 93 932 (1.0) | 383 995 (1.0) | <0.01 |
Abbreviations: BTI, biliary tract infection; CCI, Charlson Comorbidity Index; CVA, cerebrovascular accident; IQR, interquartile range; NA, not applicable; UTI, urinary tract infection; VTE, venous thromboembolism.
Captured using outpatient service records 30 days prior and 7 days after the index fill date; if a patient had multiple indications, the diagnosis recorded in closest proximity to the index fill date was selected; if patient had no indication but had 1 or more outpatient service records during the time frame, they were classified as “no indication identified”; patients with no records were classified as “no billed health care.”
Includes pharyngitis, sinusitis, laryngitis, and bronchitis.
Staphylococcus/streptococcus category International Classification of Diseases, Ninth Revision, Clinical Modification and International Classification of Diseases, Tenth Revision, Clinical Modification codes did not clarify specific site for categorization and thus is mutually exclusive from remaining infectious diagnoses.
Individual components of the CCI were included in the inverse probability of treatment weighting; diabetes was defined as no disease, diagnosis only, oral medication, or insulin-requiring, instead of as complicated and uncomplicated disease.
Includes retinal aneurysm, cerebral aneurysm, heart aneurysm, coronary aneurysm, and pulmonary aneurysm.
Primary Outcome
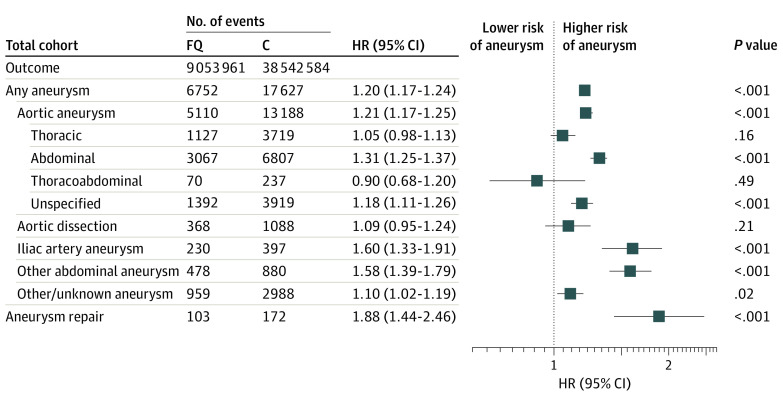
There were 24 141 aneurysm events (23 910 unique adults) diagnosed within 90 days of a fluoroquinolone or comparator antibiotic fill. The unweighted 90-day incidence of aneurysm (including dissection) in the fluoroquinolone group was 7.5 cases per 10 000 fills (6752 of 9 053 961) compared with 4.6 cases per 10 000 fills (17 627 of 38 542 584) after comparator antibiotics. After weighting, fluoroquinolones were still associated with a significant increase in the incidence of aneurysm formation (hazard ratio [HR], 1.20; 95% CI, 1.17-1.24; Figure 2). Specifically, fluoroquinolones were associated with a higher 90-day incidence of abdominal aortic aneurysms (HR, 1.31; 95% CI, 1.25-1.37), iliac artery aneurysms (HR, 1.60; 95% CI, 1.33-1.91), and other abdominal aneurysms (HR, 1.58; 95% CI, 1.39-1.79), compared with comparator antibiotics. In addition, those in the fluoroquinolone group were more likely to undergo an aneurysm repair in the 90-day period (HR, 1.88; 95% CI, 1.44-2.46). Fluoroquinolone use appeared to have little to no association with aortic dissection (HR, 1.09; 95% CI, 0.95-1.24) or thoracic (HR, 1.05; 95% CI, 0.98-1.13) and thoracoabdominal aortic (HR, 0.90; 95% CI, 0.68-1.20) aneurysms (Figure 2 and eTable 4 in the Supplement).
Figure 2. Associations Between 90-Day Incidence of Aneurysm Diagnosis After Fluoroquinolone (FQ) vs Antibiotic Comparator Prescription Fills.
Inverse probability of treatment weighting was used to account for potential confounding; weights and repeated observations in individuals were accounted for using robust sandwich estimators. C indicates comparator; HR, hazard ratio.
When evaluating episodes of primary interest by type of fluoroquinolone, ciprofloxacin and levofloxacin accounted for nearly 85% of prescriptions and 88% of aneurysm episodes, with all fluoroquinolone types closely correlated with a similar proportion of aneurysm events (Figure 3). After weighting, both ciprofloxacin and levofloxacin were associated with an increased incidence of aneurysm formation (eFigure 1 in the Supplement). These data suggest a trend toward an overall class association. When antibiotic fills after an aneurysm event were removed (13 732 antibiotic fills and 238 aneurysm events), similar results were observed (eFigure 2 in the Supplement).
Figure 3. Disaggregation of Cohort Showing Breakdown of the Fluoroquinolone (FQ) Group Into the Specific FQ Prescribed.
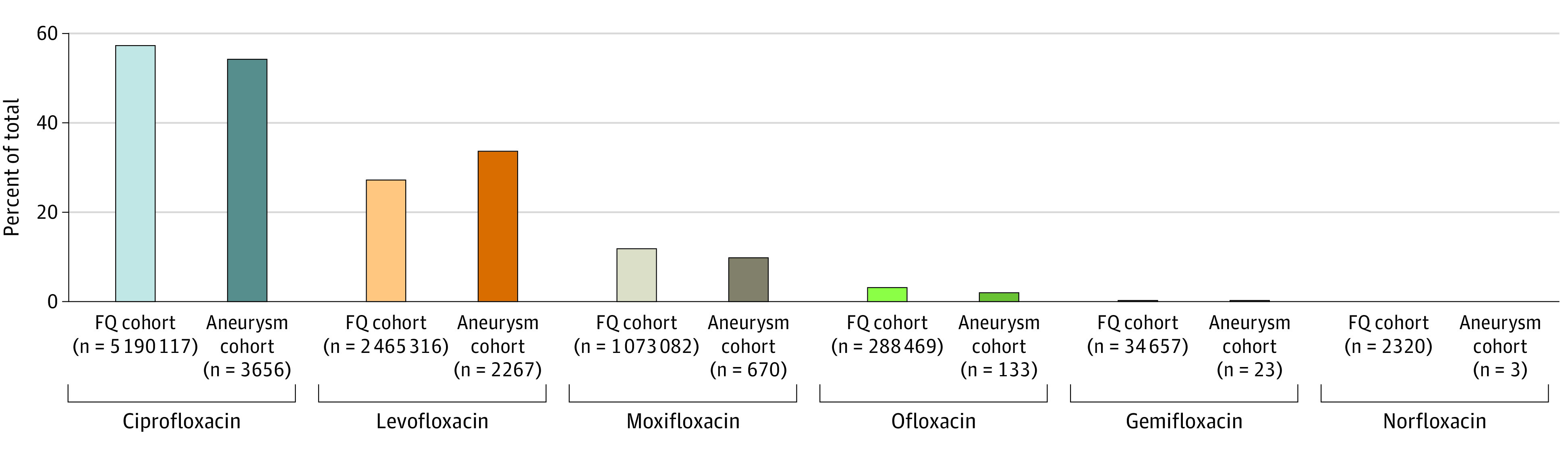
Ciprofloxacin and levofloxacin account for the majority of prescriptions.
Subgroup Analyses
When stratified by age, the association of fluoroquinolones with aneurysm was significantly different across age groups (18-34 years: HR, 0.99; 95% CI, 0.83-1.18; 35-49 years: HR, 1.18; 95% CI, 1.09-1.28; 50-64 years: HR, 1.24; 95% CI, 1.19-1.28; P = .04) (eFigure 3 in the Supplement). Minimal differences were seen when stratified by sex, diabetes, hypertension, and hyperlipidemia (Figure 4; eFigures 4, 5, 6, and 7 in the Supplement). These data collectively suggested that fluoroquinolone use was associated with an increased risk of aneurysm among all adults aged 35 years or older and that other traditional risk factors, such as sex and comorbidities, may also be associated with aneurysm among adults, although the association may be minimal.
Figure 4. Association of Fluoroquinolones vs Antibiotic Comparator With Aneurysm Risk Overall and Stratified by Sex, Age, and Comorbidity Status.
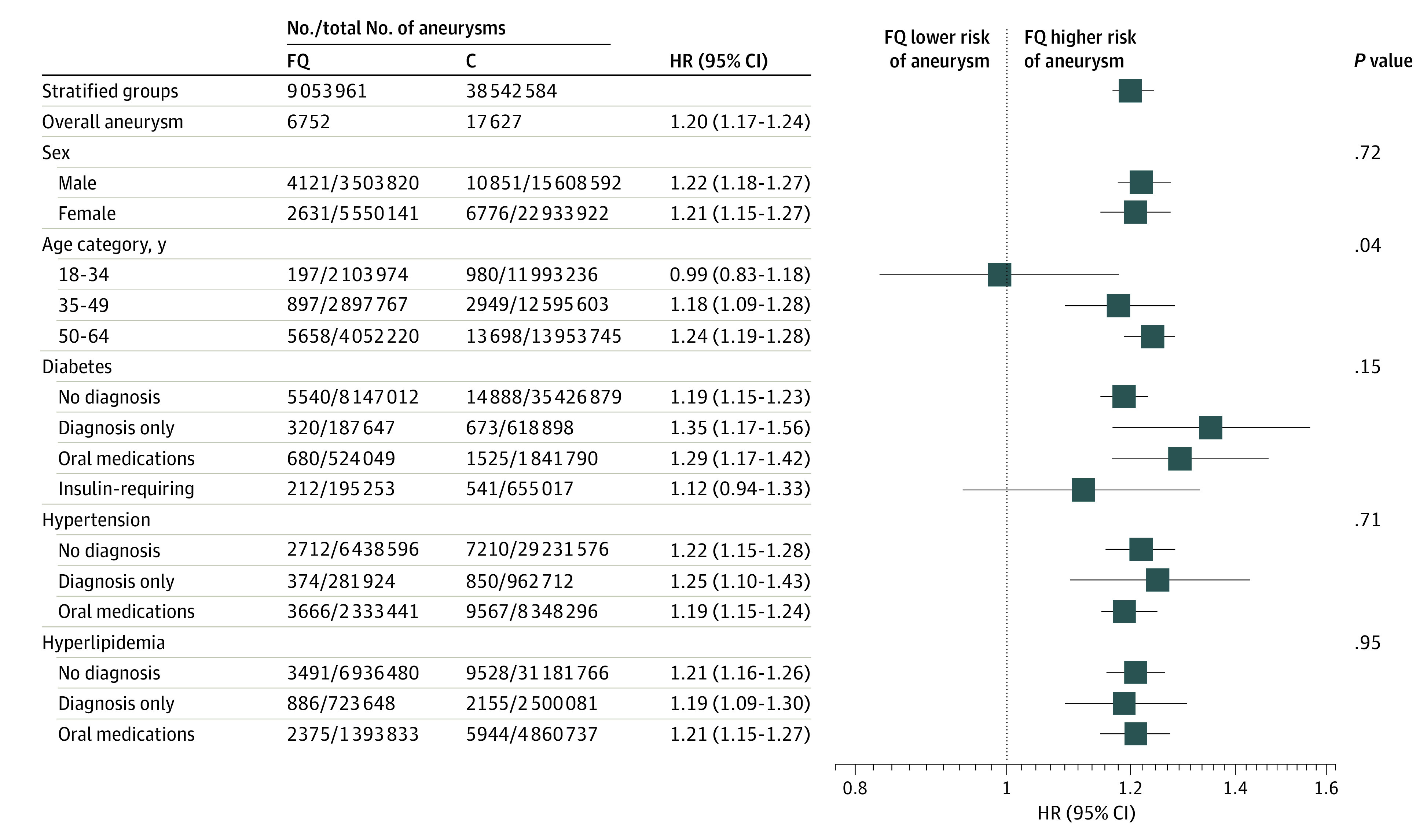
The only significant difference is the stratification by age (eFigure 3 in the Supplement). P values represent comparison of tiers within each stratified group. C indicates comparator; FQ, fluoroquinolone; and HR, hazard ratio.
Discussion
In this large cohort of commercially insured Americans aged 18 to 64 years, a fluoroquinolone prescription fill was associated with an increased incidence of aortic aneurysm development by 20% although the overall incidence of occurrence was low (unweighted: 7.5 cases per 10 000 prescription fills after fluoroquinolone vs 4.6 cases per 10 000 prescription fills after comparator antibiotics). More specifically, fluoroquinolone prescription fills were associated with an increased 90-day incidence of abdominal aortic aneurysms, iliac artery aneurysms, and other abdominal aneurysms, and those receiving fluoroquinolones were much more likely to undergo an aneurysm repair. The association between fluoroquinolone use and aneurysms was present in all adults aged 35 years or older and was consistent across standard aneurysm risk factors, such as sex and comorbidities. Fluoroquinolones did not appear to be associated with aortic dissection or thoracic aortic aneurysms.
Pasternak et al,6 in their 2018 study of a large Swedish cohort, found a similar association of aortic aneurysms with fluoroquinolone use compared with amoxicillin (HR, 1.66; 95% CI, 1.12-2.46). Their higher HRs may be due to the age group of their study population (≥50 years) and choice of comparator. Despite those differences, their study echoes our results in finding no association with aortic dissection and an unaffected risk of thoracic aortic aneurysm development compared with abdominal aortic aneurysms.
Other studies did not differentiate between dissection and aneurysm and instead evaluated them together, such as Lee et al, in their 2015 case-control Taiwanese study.5 They found an almost 3-fold increase in the odds of aortic aneurysm or aortic dissection after fluoroquinolone use (odds ratio, 2.71), although this was evaluated in an elderly population (mean age, 70.5 years) with more comorbidities (61% with hypertension, 20% with ischemic heart disease, 18% with chronic obstructive pulmonary disease, and 13% with diabetes), and lacked an active comparator. In a Canadian population study performed by Daneman et al,7 adults older than 65 years had an increased incidence of aortic aneurysm or dissection after fluoroquinolone exposure (HR, 2.24), which was also significantly higher than aneurysm or dissection risk after amoxicillin exposure (HR, 1.50).
Increased incidence of abdominal aortic aneurysms vs thoracic aortic aneurysms after fluoroquinolone use is of substantial relevance. Although each of these aneurysm types involve segments of a singular vessel, numerous studies have shown that there is significant heterogeneity in aneurysm structure, function, and response to physiological changes.19,20,21 Recent animal studies emphasize regional heterogeneity in aneurysm formation in the context of fluoroquinolone use. In a study performed by LeMaire et al,22 mice challenged with a high-fat diet and low-dose angiotensin infusion that received ciprofloxacin had an increased incidence in aneurysm formation in the thoracic aorta and suprarenal abdominal aorta but not in the infrarenal abdominal aorta. This was attributed to an increase in active matrix metalloproteinase 9 and decreased lysyl oxidase signaling. Alternatively, fluoroquinolones have been reported to affect circulating cytokine levels in vivo.23 One affected cytokine, interleukin 6, is elevated in patients with abdominal aortic aneurysms.24 When circulating interleukin 6 levels were increased in healthy wild-type mice, abdominal dilation occurred, while the thoracic aorta was not affected.25 This was attributed to increased macrophage recruitment in the abdominal aorta vs the thoracic aorta. Combined, these studies highlight the need for ongoing investigations that elucidate the mechanisms involved in aneurysm development with respect to physiologic location. As such, regional differences in the etiology, incidence, and clinical management of aortic disease in the thoracic vs abdominal aorta should be carefully considered.
Limitations
Although our study showed an association between fluoroquinolone use and aneurysm formation, we do recognize that there are limitations in the study. First, we were unable to capture undiagnosed aneurysms. In addition, abdominal imaging was not routinely performed. Thus, it is possible that aneurysms we classified as incident outcomes may have been preexisting, or that fluoroquinolones simply aggravated a preexisting condition rather than initiated a de novo aneurysm. Second, the definition of aneurysm relied on ICD-9-CM and ICD-10-CM codes. We did not have access to aneurysm size, modality used to diagnose the aneurysm (eg, ultrasonography and computed tomography), or indication for surgical intervention. Third, there were significant differences in documented indication between the fluoroquinolone and comparator groups. It is possible that some of the imaging performed for associated indications (eg, gastrointestinal infection) led to an incidental diagnosis of an aneurysm; however, we excluded patients hospitalized prior to their antibiotic fill and included indication in our propensity score model to minimize this potential confounding. Fourth, although we required no evidence of an aneurysm within the 6 months prior to the fill date, we found several adults in our cohort who had multiple antibiotic fills with an aneurysm event (n = 231), suggesting that some of our outcomes were indeed prevalent conditions. However, when we excluded subsequent antibiotic episodes after an aneurysm was identified, we found similar results. Fifth, the prevalence of comorbidities may be underestimated owing to being undiagnosed or not recently documented conditions; however, we do not expect misclassification of comorbidities to be differential across antibiotic groups. Sixth, several risk factors associated with aneurysm development, such as smoking, were not captured (or reliably captured) in claims data. This is an important risk factor that warrants further evaluation in future studies using alternative databases. Seventh, we were able to capture antibiotic fills but were unable to confirm that the prescriptions (fluoroquinolone or antibiotic comparator) were consumed by the individual or taken as prescribed; however, we assume unused fills would be minimal and similar between cohorts. In addition, more than 50% of fluoroquinolone and comparator antibiotic fills had no indication identified, despite using a broad list of infectious indications and a generous window for identification (from −30 to 7 days). Currently, there are no universal documentation requirements for antibiotic indications, making capturing indications and truly reflective antibiotic stewardship studies difficult to perform.13,16,26,27 However, when we restricted the analysis to individuals with indications identified, we found similar results (eTable 5 in the Supplement). Eighth, MarketScan data represent a younger, commercially insured population, and our results may not generalize to Medicare, Medicaid, or uninsured populations or to the entire US population.
Conclusions
In conclusion, fluoroquinolone use in the US and internationally has been associated with an increase in immediate incidence of aortic aneurysm formation. Although the overall incidence of aneurysm formation detected in the present study was low, the aneurysm incidence after a fluoroquinolone fill was 20% higher than that of comparator antibiotics, and consistent among all adults age 35 years or older. When examining specific anatomic sites, there was a 31% higher incidence of abdominal aortic aneurysm and a 60% higher incidence of iliac artery aneurysm after fluoroquinolone use. Contextualizing these data, we believe that the current US FDA black box warnings are warranted but may need to be expanded to include younger adults with other risk factors. Further studies will be needed to elucidate the mechanisms of fluoroquinolone-associated aneurysm development in humans in addition to studies that evaluate risk in the setting of known risk factors for aneurysmal disease or worse health care outcomes.
eTable 1. Generic Names Used to Capture Medications of Interest
eTable 2. ICD-9-CM and ICD-10-CM Codes Used to Capture Baseline Covariates and Indications for Antibiotic Use
eTable 3. ICD-9-CM, ICD-10-CM, and CPT Codes Used to Capture Outcomes of Interest
eTable 4. Outcomes of Entire Cohort on 90-Day Incidence of Aneurysm After Fluoroquinolone or Active Comparator, Stratified for Subanalysis
eTable 5. Association between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations After Restricting to Individuals with Indications Identified
eFigure 1. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Fluoroquinolone Type
eFigure 2. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Removing Repeated Observations
eFigure 3. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Age
eFigure 4. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Sex
eFigure 5. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Diabetes Status
eFigure 6. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Hypertension Status
eFigure 7. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Hyperlipidemia Status
References
- 1.Kabbani S, Palms D, Bartoces M, Stone N, Hicks LA. Outpatient antibiotic prescribing for older adults in the United States: 2011 to 2014. J Am Geriatr Soc. 2018;66(10):1998-2002. doi: 10.1111/jgs.15518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hicks LA, Bartoces MG, Roberts RM, et al. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin Infect Dis. 2015;60(9):1308-1316. doi: 10.1093/cid/civ076 [DOI] [PubMed] [Google Scholar]
- 3.Shapiro DJ, Hicks LA, Pavia AT, Hersh AL. Antibiotic prescribing for adults in ambulatory care in the USA, 2007-09. J Antimicrob Chemother. 2014;69(1):234-240. doi: 10.1093/jac/dkt301 [DOI] [PubMed] [Google Scholar]
- 4.Hersh AL, Gerber JS, Hicks LA, Pavia AT. Lessons learned in antibiotic stewardship: fluoroquinolone use in pediatrics. J Pediatric Infect Dis Soc. 2015;4(1):57-59. doi: 10.1093/jpids/piu044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee CC, Lee MT, Chen YS, et al. Risk of aortic dissection and aortic aneurysm in patients taking oral fluoroquinolone. JAMA Intern Med. 2015;175(11):1839-1847. doi: 10.1001/jamainternmed.2015.5389 [DOI] [PubMed] [Google Scholar]
- 6.Pasternak B, Inghammar M, Svanström H. Fluoroquinolone use and risk of aortic aneurysm and dissection: nationwide cohort study. BMJ. 2018;360:k678. doi: 10.1136/bmj.k678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Daneman N, Lu H, Redelmeier DA. Fluoroquinolones and collagen associated severe adverse events: a longitudinal cohort study. BMJ Open. 2015;5(11):e010077. doi: 10.1136/bmjopen-2015-010077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Karthikesalingam A, Holt PJ, Vidal-Diez A, et al. Mortality from ruptured abdominal aortic aneurysms: clinical lessons from a comparison of outcomes in England and the USA. Lancet. 2014;383(9921):963-969. doi: 10.1016/S0140-6736(14)60109-4 [DOI] [PubMed] [Google Scholar]
- 9.Davis FM, Rateri DL, Daugherty A. Mechanisms of aortic aneurysm formation: translating preclinical studies into clinical therapies. Heart. 2014;100(19):1498-1505. doi: 10.1136/heartjnl-2014-305648 [DOI] [PubMed] [Google Scholar]
- 10.Daugherty A, Cassis LA. Mechanisms of abdominal aortic aneurysm formation. Curr Atheroscler Rep. 2002;4(3):222-227. doi: 10.1007/s11883-002-0023-5 [DOI] [PubMed] [Google Scholar]
- 11.Schmitz-Rixen T, Keese M, Hakimi M, et al. Ruptured abdominal aortic aneurysm-epidemiology, predisposing factors, and biology. Langenbecks Arch Surg. 2016;401(3):275-288. doi: 10.1007/s00423-016-1401-8 [DOI] [PubMed] [Google Scholar]
- 12.US Food and Drug Administration. FDA warns about increased risk of ruptures or tears in the aorta blood vessel with fluoroquinolone antibiotics in certain patients. Published 2018. Accessed January 15, 2020. https://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-about-increased-risk-ruptures-or-tears-aorta-blood-vessel-fluoroquinolone-antibiotics
- 13.Blaszczyk AT, Anderson SS, Hall RG. Appropriateness of fluoroquinolone prescribing in the long-term care setting. J Am Geriatr Soc. 2020;68(3):661-663. doi: 10.1111/jgs.16324 [DOI] [PubMed] [Google Scholar]
- 14.IBM Watson Health. IBM MarketScan Research Databases for Health Services Researchers. Published 2019. Accessed April 7, 2020. https://www.ibm.com/downloads/cas/6KNYVVQ2
- 15.Castro MM, Tanus-Santos JE, Gerlach RF. Matrix metalloproteinases: targets for doxycycline to prevent the vascular alterations of hypertension. Pharmacol Res. 2011;64(6):567-572. doi: 10.1016/j.phrs.2011.04.002 [DOI] [PubMed] [Google Scholar]
- 16.Chua KP, Fischer MA, Linder JA. Appropriateness of outpatient antibiotic prescribing among privately insured US patients: ICD-10-CM based cross sectional study. BMJ. 2019;364:k5092. doi: 10.1136/bmj.k5092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45(6):613-619. doi: 10.1016/0895-4356(92)90133-8 [DOI] [PubMed] [Google Scholar]
- 18.Austin PC, Stuart EA. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat Med. 2015;34(28):3661-3679. doi: 10.1002/sim.6607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ruddy JM, Jones JA, Spinale FG, Ikonomidis JS. Regional heterogeneity within the aorta: relevance to aneurysm disease. J Thorac Cardiovasc Surg. 2008;136(5):1123-1130. doi: 10.1016/j.jtcvs.2008.06.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wolinsky H, Glagov S. Comparison of abdominal and thoracic aortic medial structure in mammals: deviation of man from the usual pattern. Circ Res. 1969;25(6):677-686. doi: 10.1161/01.RES.25.6.677 [DOI] [PubMed] [Google Scholar]
- 21.Wolinsky H. Comparison of medial growth of human thoracic and abdominal aortas. Circ Res. 1970;27(4):531-538. doi: 10.1161/01.RES.27.4.531 [DOI] [PubMed] [Google Scholar]
- 22.LeMaire SA, Zhang L, Luo W, et al. Effect of ciprofloxacin on susceptibility to aortic dissection and rupture in mice. JAMA Surg. 2018;153(9):e181804. doi: 10.1001/jamasurg.2018.1804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ogino H, Fujii M, Ono M, Maezawa K, Hori S, Kizu J. In vivo and in vitro effects of fluoroquinolones on lipopolysaccharide-induced pro-inflammatory cytokine production. J Infect Chemother. 2009;15(3):168-173. doi: 10.1007/s10156-009-0680-1 [DOI] [PubMed] [Google Scholar]
- 24.Dawson J, Cockerill GW, Choke E, Belli AM, Loftus I, Thompson MM. Aortic aneurysms secrete interleukin-6 into the circulation. J Vasc Surg. 2007;45(2):350-356. doi: 10.1016/j.jvs.2006.09.049 [DOI] [PubMed] [Google Scholar]
- 25.Akerman AW, Stroud RE, Barrs RW, et al. Elevated wall tension initiates interleukin-6 expression and abdominal aortic dilation. Ann Vasc Surg. 2018;46:193-204. doi: 10.1016/j.avsg.2017.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fischer MA, Mahesri M, Lii J, Linder JA. Non-infection-related and non-visit-based antibiotic prescribing is common among Medicaid patients. Health Aff (Millwood). 2020;39(2):280-288. doi: 10.1377/hlthaff.2019.00545 [DOI] [PubMed] [Google Scholar]
- 27.Ray MJ, Tallman GB, Bearden DT, Elman MR, McGregor JC. Antibiotic prescribing without documented indication in ambulatory care clinics: national cross sectional study. BMJ. 2019;367:l6461. doi: 10.1136/bmj.l6461 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eTable 1. Generic Names Used to Capture Medications of Interest
eTable 2. ICD-9-CM and ICD-10-CM Codes Used to Capture Baseline Covariates and Indications for Antibiotic Use
eTable 3. ICD-9-CM, ICD-10-CM, and CPT Codes Used to Capture Outcomes of Interest
eTable 4. Outcomes of Entire Cohort on 90-Day Incidence of Aneurysm After Fluoroquinolone or Active Comparator, Stratified for Subanalysis
eTable 5. Association between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations After Restricting to Individuals with Indications Identified
eFigure 1. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Fluoroquinolone Type
eFigure 2. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Removing Repeated Observations
eFigure 3. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Age
eFigure 4. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Sex
eFigure 5. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Diabetes Status
eFigure 6. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Hypertension Status
eFigure 7. Association Between Fluoroquinolone Use and 90-Day Incidence of Aneurysm Formations Stratified by Hyperlipidemia Status