Abstract
Background
In addition to ovarian and breast cancers, loss-of-function mutations in BRCA1 and BRCA2 genes are also linked to an increased risk of pancreatic cancer, with ~ 4 to 7% of pancreatic cancer patients harboring germline BRCA mutations. Most BRCA alterations in pancreatic cancer are frame-shifting indels, stop-gain, and splice-site mutations, but single nucleotide substitutions are rare. Recent studies demonstrated a significant progression-free survival (PFS) benefit from maintenance olaparib, a poly (ADP-ribose) polymerase (PARP) inhibitor administered to patients with germline BRCA mutations and metastatic pancreatic cancer.
Case presentation
Here, we report a metastatic pancreatic cancer case who harbored a novel somatic BRCA2 c.6944T > C (p. I2315T) point mutation. After 6 weeks first-line chemotherapy, the patient was refractory to treatment and had a progressive disease. Due to the novel nonsynonymous BRCA2 point mutation, we decided to change the strategy by administering olaparib. The patient benefited from olaparib therapy and achieved a PFS of ~ 6.5 months.
Conclusions
We describe a patient carrying a novel somatic BRCA2 p. I2315T point mutation, which is first reported in metastatic pancreatic cancer. This case report indicates that a gene mutation-based strategy should be considered in the clinic to provide more effective treatment.
Keywords: BRCA2, Olaparib, Metastatic pancreatic cancer, PARP inhibitor, Somatic mutation
Background
Pancreatic cancer is the 7th leading cause of cancer-related deaths worldwide, with a 5-year survival rate of < 5% [1]. This devastating malignancy is generally diagnosed at metastatic stages in the clinic, indicative of its late detection and biological aggressiveness [1]. Tremendous efforts are ongoing with the aims of discovering early diagnostic markers and novel therapeutic avenues for pancreatic cancer; however, progress is remarkably hindered by the complicated heterogeneity within and between patient tumors.
BRCA1 and BRCA2 are essential players involved in homologous recombination repair of double-strand deoxyribonucleic acid (DNA) breaks [2]. BRCA inactivation due to somatic mutations or BRCA1 promoter methylation have been observed in various cancer types, including a small subgroup of metastatic pancreatic cancer [3–5]. Most BRCA alterations in pancreatic cancer are frame-shifting indels, stop-gain, and splice-site mutations, whereas single nucleotide substitutions are rarely identified [2, 4, 5].
A large body of evidence showed that the poly (ADP-ribose) polymerase (PARP) inhibitors are synthetically lethal in BRCA-mutated tumors with DNA repair defects and displayed potent anti-tumor activity when combined with DNA-damaging agents. Thus, patients carrying germline mutations in BRCA or the various patterns of somatic BRCA mutations that could result in the inactivation of BRCA are sensitive to PARP inhibitors [6].
A recent phase 3 POLO (Pancreas Cancer Olaparib Ongoing) trial demonstrated that the administration of a PARP inhibitor, olaparib, as a maintenance therapy significantly prolonged the median progression-free survival (PFS) of patients with germline BRCA mutations and metastatic pancreatic cancer who had not progressed during platinum-based chemotherapy compared to the placebo arm (7.4 months vs. 3.8 months) [7]. More importantly, olaparib treatment did not compromise health-related quality of life in those patients [8]. However, PARP inhibitor resistance is common due to homologous recombination repair restoration (HRR), epigenetic modification, reversion mutations, restoration of ADP-ribosylation (PARylation), and pharmacological alteration [9].
In this report, we present the case of a metastatic pancreatic cancer patient who had progressive disease (PD) following chemotherapy with gemcitabine and nab-paclitaxel. Mutational profiling analysis using targeted next-generation sequencing (NGS) revealed that the patient carried a novel somatic BRCA2 c.6944T > C (p. I2315T) point mutation. Olaparib was thereby administered in combination with a modified FOLFIRINOX regimen or as a monotherapy. The patient showed a significant response to this treatment strategy and exhibited stable disease and a PFS of ~ 6.5 months was observed.
Case presentation
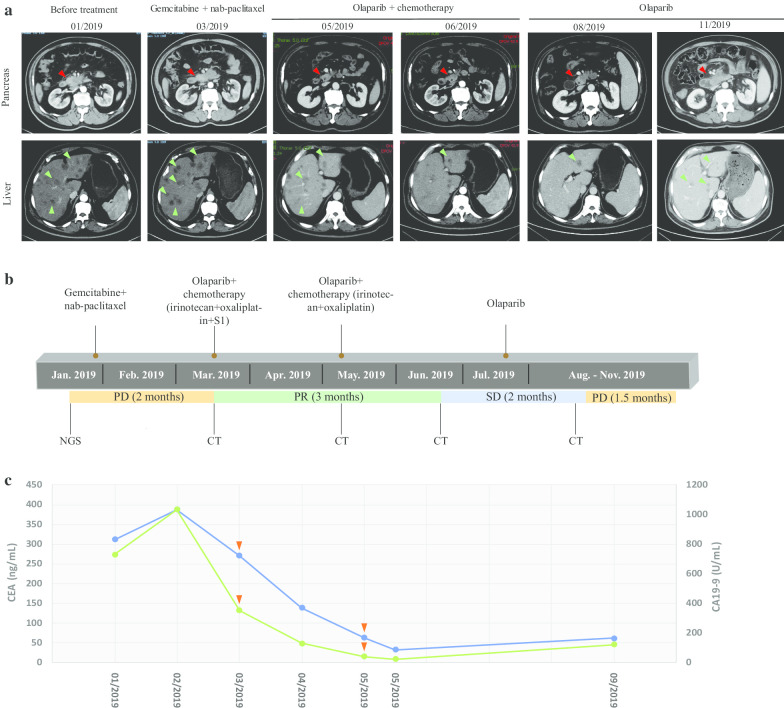
A 57-year-old Chinese male with no obvious symptoms was admitted to the hospital due to the identification of a low-density shadow in the liver during his physical examination. In January 2019, a computed tomography (CT) scan revealed a 5.0 × 3.0 cm tumor in the uncinate process of the pancreas with hepatic metastases (Fig. 1a). The patient had a family history of cancer: his mother was diagnosed with stomach cancer at the age of 65 years and his father had lung cancer. According to the National Comprehensive Cancer Network (NCCN) Guidelines, the patient first received gemcitabine treatment at a dose of 1.4 g once per week, and nab-paclitaxel at a dose of 200 mg every 2 weeks as a first-line chemotherapy in January 2019 (Fig. 1b). Unfortunately, a CT re-evaluation at 6 weeks post-treatment showed progressive enlargement of both pancreatic and hepatic lesions (Fig. 1a), accompanied with a marked elevation of serum carcinoembryonic antigen (CEA, 270.35 ng/mL) and carbohydrate antigen 19-9 (CA19-9, 352.56 U/mL) levels (Fig. 1c); thus, suggesting that the patient was refractory to treatment and had a PD.
Fig. 1.
CT images and measurements of serum tumor biomarkers during the course of treatment. a Serial CT scans demonstrated a reduction in the size of the pancreatic (red arrows) and metastatic hepatic lesions (green arrows) following olaparib therapy. b A timeline indicating the application of different therapeutic strategies and the patient’s response. c Measurements of serum CEA and CA19-9 levels at different treatment times. Blue line: CEA; green line: CA19-9; orange arrows: time points of the CT scans. PD progressive disease, PR partial response, SD stable disease
To determine a more effective and appropriate targeted therapy, we performed a targeted NGS analysis of 425 cancer-related genes on the patient’s plasma and tumor tissue biopsy samples. The cancer mutation panel test revealed multiple deleterious somatic mutations, such as the driver mutations, KRAS Q61R (c.182A > G), TP53 R110del (c.329_331delGTC), and APC S1465RfsX9 (c.4393_4394dupAG), and copy number gain of several genes (Table 1). Interestingly, we also observed a novel nonsynonymous BRCA2 c.6944T > C (p. I2315T) point mutation with a mutant allele frequency (MAF) of 28.0% in the plasma and 39.5% in the tumor biopsy specimens. The novel BRCA2 point mutation was not detected in white blood cells, and thus, it was confirmed as a somatic mutation.
Table 1.
Genetic alterations detected in the patient’s plasma and tumor biopsy specimens
| Genes | Alternations | Nucleotide change | MAF (plasma) | MAF (tumor) |
|---|---|---|---|---|
| BRCA2 | p. I2315T | c.6944T > C | 28% | 39.5% |
| KRAS | p. Q61R | c.182A > G | 27.4% | 43.2% |
| TP53 | p. R110del | c.329_331delGTC | 37.4% | 81.6% |
| APC | p. S1465RfsX9 | c.4393_4394dupAG | 26.7% | 71.7% |
| CCNE1 | Gene amplification | NA | 16.0-fold | 27.4-fold |
| CCNE1 | IGR (downstream UQCRFS1) ~ CCNE1 fusion | NA | 0.1% | – |
| PIK3CA | Gene amplification | NA | – | 1.9-fold |
| PKHD1 | p. R909X truncation | c.2725C > T | 4.9% | 16.9% |
| SOX2 | Gene amplification | NA | – | 2.0-fold |
| STMN1 | Gene amplification | NA | – | 1.8-fold |
| TERC | Gene amplification | NA | – | 1.9-fold |
| TUBB3 | Gene amplification | NA | – | 2.2-fold |
–, not detectable; NA, not applicable; MAF, mutant allele frequency. Gene amplification was presented as the relative fold change to normal controls
Given the clinical efficacy of olaparib in treating BRCA-mutated advanced solid tumors, we decided to revise the therapeutic strategy by administering olaparib at a dose of 150 mg twice per day in combination with a modified FOLFIRINOX regimen (irinotecan at a dose of 100 mg on days 1, 8 and 15; oxaliplatin at a dose of 150 mg on days 1 and 15; S-1 at a dose of 40 mg twice per day from day 1 to day 14) for one cycle beginning in March 2019 (Fig. 1b). However, due to the occurrence of adverse events (e.g., diarrhea, general weakness, fatigue, and loss of appetite) and myelosuppression, including grade 2–3 leukopenia and thrombocytopenia, chemotherapy was not given on day 15 and the frequency of irinotecan was reduced to only be administered on days 1 and 15 in the subsequent treatment. Additionally, megestrol acetate and granulocyte-colony stimulating factor (G-CSF) were prescribed to increase appetite and promote the recovery of myelosuppression. In May 2019, a reduction in the size of both pancreatic and hepatic lesions was revealed by the CT scan, while the levels of the serum tumor biomarkers, CEA (63.38 ng/mL) and CA19-9 (40.53 U/mL) were considerably reduced (Fig. 1a, c). Olaparib and modified FOLFIRINOX were thus continued with irinotecan and oxaliplatin being administered only on day 1 (Fig. 1b). After 2 weeks of treatment, CEA and CA19-9 levels decreased to 32.69 ng/mL and 25.57 U/mL, respectively (Fig. 1c). A CT scan performed in June 2019 showed a significant size reduction in the primary and metastatic tumors, which indicated a partial response (PR, Fig. 1a). Since the patient experienced severe marrow suppression during treatment, only olaparib monotherapy was administered since July 2019 to reduce adverse effects (Fig. 1b). Stable disease (SD) was subsequently observed in August 2019 (Fig. 1a). In September 2019, aside from an increase in CEA and CA19-9 levels (Fig. 1c), laboratory blood tests demonstrated that the patient underwent acquired granulocytopenia, anemia, and thrombocytopenia. Consequently, G-CSF, thrombopoietin, and erythropoietin were administered for symptom management. After olaparib treatment for another 1.5 months, PD was indicated by a CT scan (Fig. 1a). The patient achieved a PFS for ~ 6.5 months following olaparib combination and monotherapy.
Discussion and conclusions
Neoplastic cells lacking a functional homologous recombination repair system, such as those carrying BRCA mutations, are sensitive to PARP inhibition through accumulated DNA damage via multiple mechanisms [6]. As a new therapeutic concept, maintenance olaparib has shown promising results in the treatment of germline BRCA-mutated breast, ovarian, and metastatic pancreatic cancer [7, 10, 11]. To date, the majority of BRCA2 mutations identified in pancreatic cancer were frame-shifting indels (e.g., c.6174delT, c.6158insT) and splice-site mutations. In contrast, single point mutations have been rarely reported [4, 5, 12]. Mesman [13] recently assessed the potential pathogenic impact of a large set of BRCA2 missense variants using a mouse embryonic stem cell (mESC)-based functional assay, and found that BRCA2 missense mutations, such as c.93G > T (p. W31C) and c.8351G > A (p. R2784Q), were able to increase susceptibility to PARP inhibitor treatment.
The patient in our report harbored an unreported BRCA2 point mutation [c.6944T > C (p. I2315T)] located in exon 13 of BRCA2 between the G-CSF. Different germline BRCA2 mutations (e.g., p. I2315V, p. I2315L) at the same site have been documented with unknown functional significance [14, 15]. Although the underlying molecular mechanism(s) by which this novel BRCA2 p. I2315T mutation impairs DNA repair and sensitizes tumor cells to PARP inhibition remains to be elucidated. We postulate that it may be related to a change in the polarity of amino acid residues since isoleucine (I), valine (V) and leucine (L) are all non-polar and hydrophobic, while threonine (T) is hydrophilic. Thus, we hypothesize that the p. I2315T mutation is likely to cause a structural abnormality in the BRCA2 protein, which results in defective DNA double strand break (DSB) repair by homologous recombination (HR) and its sensitivity to olaparib. To test this hypothesis, we attempted to generate a three dimensional (3D) structural model of the BRCA2 p. I2315T mutation, but failed due to the absence of an established crystal structure of the full-length human BRCA2 due to its size and segmental nature [16].
In summary, we report a metastatic pancreatic cancer patient carrying a novel somatic BRCA2 p. I2315T point mutation. Furthermore, advances in NGS technology have provided a solid basis for precise detection of well-known driver mutations, and rare or novel mutations. Thus, NGS may provide clinicians with invaluable information (e.g., BRCA1/2 status of tumors) that can be leveraged for therapeutic decision making, and perform better evaluations of patients’ responses during the course of treatment.
Acknowledgements
We would like to thank the patient for providing written informed consent for publication.
Abbreviations
- PARP
Poly(ADP-ribose) polymerase
- PFS
Progression-free survival
- PD
Progressive disease
- DNA
Deoxyribonucleic acid
- POLO
Pancreas cancer olaparib ongoing
- CT
Computed tomography
- NCCN
The National Comprehensive Cancer Network
- CEA
Carcinoembryonic antigen
- CA19-9
Carbohydrate antigen 19-9
- NGS
Next-generation sequencing
- MAF
Mutant allele frequency
- FOLFIRINOX
FOL-folinic acid, F-fluorouracil, IRIN-irinotecan, OX-oxaliplatin
- G-CSF
Granulocyte-colony stimulating factor
- PR
Partial response
- mESC
Mouse embryonic stem cell
- DSBs
Double strand breaks
- HR
Homologous recombination
- 3D
Three dimensional
Authors' contributions
XL conceptualized and designed the manuscript. QT and DW carried out patient clinical management, sample collection and drafted the manuscript. RG analyzed the data. SL and MY revised the manuscript. All authors read and approved the final manuscript.
Funding
Supported by the Key Project of Zhenjiang City for Health Science and Technology (SH2016038 and SH2019046), and the Project of Young Medical Talents in Jiangsu Province (QNRC2016829), China. The funding body did not have any role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Availability of data and materials
The datasets generated and/or analysed during the current study are not publicly available in order to protect participant confidentiality but are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The study protocol was approved by the ethical committee of the Affiliated Hospital of Jiangsu University. Written informed consent was provided by the patient.
Consent for publication
Written informed consent was obtained from the patient for publication of the case and all accompanying tables/images. A copy of the written consent is available for review by the Editor of this journal.
Competing interests
Ruting Guan and Sisi Liu are the employees of Nanjing Geneseeq Technology Inc., China; Man Yu is an employee of Geneseeq Technology Inc., Canada. The remaining authors declare no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ducreux M, Seufferlein T, Van Laethem JL, et al. Systemic treatment of pancreatic cancer revisited. Semin Oncol. 2019;46:28–38. doi: 10.1053/j.seminoncol.2018.12.003. [DOI] [PubMed] [Google Scholar]
- 2.Gorodetska I, Kozeretska I, Dubrovska A. BRCA genes: the role in genome stability, cancer stemness and therapy Resistance. J Cancer. 2019;10:2109–2127. doi: 10.7150/jca.30410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Welcsh PL, King MC. BRCA1 and BRCA2 and the genetics of breast and ovarian cancer. Hum Mol Genet. 2001;10:705–713. doi: 10.1093/hmg/10.7.705. [DOI] [PubMed] [Google Scholar]
- 4.Ghiorzo P. Genetic predisposition to pancreatic cancer. World J Gastroenterol. 2014;20:10778–10789. doi: 10.3748/wjg.v20.i31.10778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Holter S, Borgida A, Dodd A, et al. Germline BRCA mutations in a large clinic-based cohort of patients with pancreatic adenocarcinoma. J Clin Oncol. 2015;33(28):3124–3129. doi: 10.1200/JCO.2014.59.7401. [DOI] [PubMed] [Google Scholar]
- 6.Faraoni I, Graziani G. Role of BRCA mutations in cancer treatment with poly(ADP-ribose) polymerase (PARP) inhibitors. Cancers. 2018;10:487. doi: 10.3390/cancers10120487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Golan T, Hammel P, Reni M, et al. Maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. N Engl J Med. 2019;381:317–327. doi: 10.1056/NEJMoa1903387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hammel P, Kindler HL, Reni M, et al. Health-related quality of life in patients with a germline BRCA mutation and metastatic pancreatic cancer receiving maintenance olaparib. Ann Oncol. 2019;30:1959–1968. doi: 10.1093/annonc/mdz406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li H, Liu Z-Y, Wu N, et al. PARP inhibitor resistance: the underlying mechanisms and clinical implications. Mol Cancer. 2020;19:107–107. doi: 10.1186/s12943-020-01227-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Robson M, Im S-A, Senkus E, et al. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Engl J Med. 2017;377:523–533. doi: 10.1056/NEJMoa1706450. [DOI] [PubMed] [Google Scholar]
- 11.Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379:2495–2505. doi: 10.1056/NEJMoa1810858. [DOI] [PubMed] [Google Scholar]
- 12.Martinez-Useros J, Garcia-Foncillas J. The role of BRCA2 mutation status as diagnostic, predictive, and prognosis biomarker for pancreatic cancer. Biomed Res Int. 2016;2016:1869304. doi: 10.1155/2016/1869304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mesman RLS, Calleja F, Hendriks G, et al. The functional impact of variants of uncertain significance in BRCA2. Genet Med. 2019;21:293–302. doi: 10.1038/s41436-018-0052-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Juwle A, Saranath D. BRCA1/BRCA2 gene mutations/SNPs and BRCA1 haplotypes in early-onset breast cancer patients of Indian ethnicity. Med Oncol. 2012;29:3272–3281. doi: 10.1007/s12032-012-0294-9. [DOI] [PubMed] [Google Scholar]
- 15.Borg A, Haile RW, Malone KE, et al. Characterization of BRCA1 and BRCA2 deleterious mutations and variants of unknown clinical significance in unilateral and bilateral breast cancer: the WECARE study. Hum Mutat. 2010;31:E1200–E1240. doi: 10.1002/humu.21202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shahid T, Soroka J, Kong E, et al. Structure and mechanism of action of the BRCA2 breast cancer tumor suppressor. Nat Struct Mol Biol. 2014;21:962–968. doi: 10.1038/nsmb.2899. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analysed during the current study are not publicly available in order to protect participant confidentiality but are available from the corresponding author on reasonable request.