Abstract
We report the clinical presentation and risk factors for survival in 175 patients with myeloproliferative neoplasms (MPN) and COVID-19, diagnosed between February and June 2020. After a median follow-up of 50 days, mortality was higher than in the general population and reached 48% in myelofibrosis (MF). Univariate analysis, showed a significant relationship between death and age, male gender, decreased lymphocyte counts, need for respiratory support, comorbidities and diagnosis of MF, while no association with essential thrombocythemia (ET), polycythemia vera (PV), and prefibrotic-PMF (pre-PMF) was found. Regarding MPN-directed therapy ongoing at the time of COVID-19 diagnosis, Ruxolitinib (Ruxo) was significantly more frequent in patients who died in comparison with survivors (p = 0.006). Conversely, multivariable analysis found no effect of Ruxo alone on mortality, but highlighted an increased risk of death in the 11 out of 45 patients who discontinued treatment. These findings were also confirmed in a propensity score matching analysis. In conclusion, we found a high risk of mortality during COVID-19 infection among MPN patients, especially in MF patients and/or discontinuing Ruxo at COVID-19 diagnosis. These findings call for deeper investigation on the role of Ruxo treatment and its interruption, in affecting mortality in MPN patients with COVID-19.
Subject terms: Infectious diseases, Myeloproliferative disease
Introduction
The SARS-CoV-2 coronavirus infection causing the coronavirus 2019 disease (COVID-19) is a highly contagious disease that appeared in China in December 2019 and is now extensively spreaded to multiple European and extra-European countries. The clinical spectrum of SARS-CoV-2 infection appears to be wide, encompassing asymptomatic infection, mild upper respiratory tract illness, and severe interstitial pneumonia with respiratory failure and high risk of death [1]. COVID-19 infected patients are also likely to be at increased risk of venous and arterial thromboembolism, as reported in China [2] and confirmed in autopsies of patients experiencing sudden death [3].
This infection has spread to Europe in February 2020 and, since then, thousands of studies addressing various aspects of COVID-19 have started and numerous clinical trials have been registered on ClinicalTrials.gov. However, there is limited information describing the presenting characteristics and outcomes of COVID-19 patients with myeloproliferative neoplasms (MPN), including essential thrombocythemia (ET), polycythemia vera (PV), and myelofibrosis (MF) [4, 5]. Natural history of MPN is marked by a high incidence of thrombosis and hemorrhagic complications and by a natural propensity to transform into overt MF and acute leukemia. Compared to the general population, overall expected survival is reduced, particularly in overt MF where it ranges from 2 to 5 years, depending on the stage of the disease [6].
MPN experts appointed by the American Society of Hematology (ASH) are updating their answers to some frequently asked clinical questions (https://www.hematology.org/COVID-19/COVID-19-and-myeloproliferative-neoplasms) regarding management of these patients under COVID-19; in particular, they discuss the role of antithrombotic drugs and how to handle the MPN-directed cytoreductive therapy before and after the onset of coronavirus infection. However, it should be acknowledged that these recommendations are derived from a consensus among experts in the MPN field, and that information on the real-world clinical practice of patients with MPN and CPVID-19 needs to be collected in order to assess the magnitude of the main outcomes’ frequency, such as survival and the corresponding risk factors, in the single MPN phenotype.
With this purpose, in May 2020, the European Leukemia Net (ELN), launched an observational study in Europe involving 38 centers from Italy, Spain, Germany, Poland, France, and United Kingdom. Here we report the first results of this study, focussing on clinical/laboratory presentation and risk factors for overall survival during the acute phase of SARS-CoV-2 infection.
Methods
Patients and COVID-19 diagnosis
This observational retrospective study (MPN-COVID) was promoted by ELN and had the endorsement of the European Haematology Association, GEMFIN Spanish network on MPN and HARMONY platform.
MPN-COVID study involved 180 patients recruited from 38 European hematology units. Sponsor of the study is the Foundation for Research (FROM) at Papa Giovanni XXIII Hospital in Bergamo, Italy, that obtained the approval by the Italian National Ethical Committee (Spallanzani Hospital, in Rome); all other centers had the approval by their respective local ethical committees.
We included consecutive adult patients with WHO-2016 diagnosed MPN from Spain (n = 81), Italy (n = 77), UK (n = 11), France (n = 7), Poland (n = 3), Germany (n = 1). Five patients were excluded from the analysis: three for acute leukaemia post-MPN and two for excess missing data. Therefore, the present analysis comprises 175 patients. Written informed consent of participants was collected where possible according to each Country regulation.
SARS-CoV-2 infection was ascertained by a positive real-time reverse transcriptase polymerase chain reaction from nasal swab (n = 155, 88.6%): for patients unable to swab (n = 20, 11.4%, COVID-19 diagnosis was based on presence of pneumonia and symptoms highly suggestive for COVID-19, such as fever, cough, oxygen saturation (O2 saturation) ≤93% at rest, dyspnea, diarrhea.
Data collection and outcome
The participating centers were asked to report in an electronic case report form) their consecutive MPN patients who developed COVID-19 in the period between 15 February 2020 and 31 May 2020. Data collected included patient demographics, comorbidities, medications, initial laboratory tests, chest radiographs, or CT scan prescribed by the treating physician, drug treatment for COVID-19, invasive mechanical ventilation, or noninvasive respiratory support in hospitalized general wards and intensive care unit (ICU) or in patients managed at home.
The additional information about MPN disease status concerned driver mutations (JAK2V617F, CALR and MPL), duration of MPN before COVID-19, presence of splenomegaly and cytoreductive drugs.
The study outcome was mortality from all-causes and was registered in hospitalized patients (regular wards or ICU) and in patients managed at home.
Statistical methods
Statistical analysis was carried out at the bio-statistical laboratory of the Foundation for Research (FROM) at Papa Giovanni XXIII Hospital in Bergamo.
Continuous variables were summarized with median along with interquartile range (IQR), and categorical ones were presented as frequencies and percentages. Characteristics of the study population were stratified for survival and differences between the two groups tested with the χ² test (or Fisher’s exact test where appropriate) or rank-sum test for categorical or continuous variables, respectively.
Overall survival was estimated by Kaplan–Meier method and compared according to Country, MF diagnosis, patient disposition, and Ruxolitinib (Ruxo) exposure and discontinuation with the log rank test. Using a multivariable logistic regression model, association with overall survival was evaluated for the following variables: age, sex, MF diagnosis, chronic heart disease, need of respiratory support, ICU admission, Ruxo exposure, and discontinuation.
A propensity score (PS) matching analysis [7] was performed to balance patients treated or not with Ruxo, by forming matched sets of 1 treated and 1 randomly-sampled non-treated patient (1:1 matching) who shared similar PS. The PS was estimated by logistic regression of exposure to Ruxo on the baseline covariates (MPN diagnosis, age, haemoglobin, neutrophilis/lymphocytes, platelets count, duration of MPN at COVID-19 onset, hospitalization for COVID-19). Matching was done using the nearest neighbor method with replacement and with caliper of width equal to 0.2 of the pooled standard deviation of the PS logit.
For all tested hypotheses, two-tailed p values < 0.05 were considered significant. Statistical analysis was performed using STATA Software, release 16 (StataCorp LP, College Station TX, USA).
Results
Survival and patient characteristics at baseline by mortality
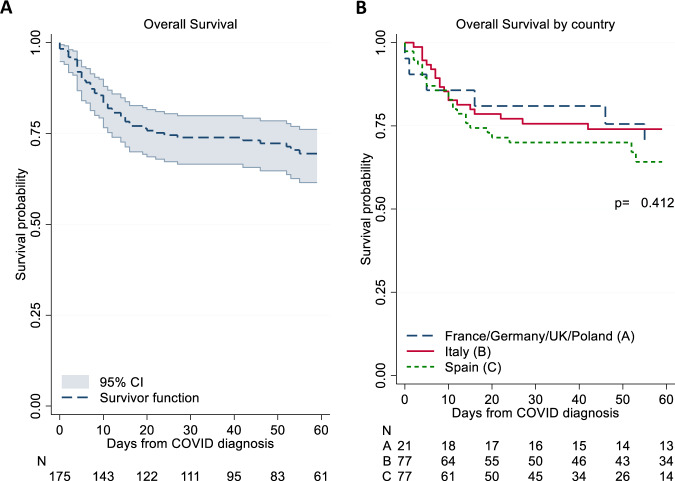
After diagnosis of COVID-19, 77% of the patients were hospitalized and 23% were followed at home; of the 59.2% who required respiratory support, 47.7% were managed with noninvasive respiratory support while 19 (11%) required ICU management. Overall, after a median follow-up of 50 days, 50 of the 175 MPN patients (28.6%) died at a median time of 9.5 days after diagnosis (Fig. 1A). This figure was not different among the participating Countries; in Italy and Spain, which were the major contributors of patients (44% and 44%, respectively), and in the rest of Europe (12% of cases) mortality was 32%, 25%, and 29%, respectively (Fig. 1B). Reported causes of death were multiorgan failure (42%), pneumonia (33%), or undefined (25%).
Fig. 1. Overall survival.
Kaplan Meier estimates for the overall survival (A), stratified by Country (B). P-values are calculated by log-rank test. CI confidence interval.
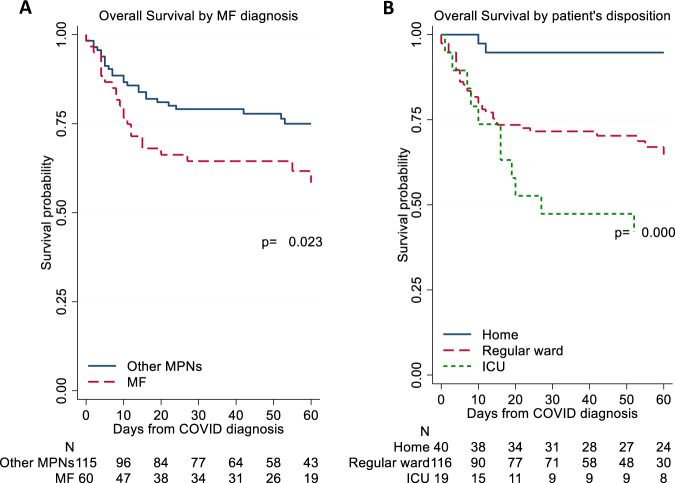
Table 1 shows characteristics of MPN patients stratified by survival. The diagnosis of ET, PV and pre-PMF did not influence the proportion of survivors versus non-survivors, while the MF phenotype was associated with a greater percentage of non-surviving patients (48.0% vs. 28.8%; p = 0.016). Figure 2A illustrates the probability of death in MF vs. the other phenotypes. Survival did not differ as regards to driver mutations, palpable splenomegaly, duration of MPN disease, or blood counts, with the exception of a decreased lymphocyte count and a higher neutrophil/lymphocyte ratio that was registered in non-survivors. Similar to what reported in general population, chronic heart failure, diabetes, older age, oxygen saturation at presentation, need of hospitalization, non-invasive and invasive respiratory support and need of being transferred to ICU were all associated with significantly lower proportion of survivors. In Fig. 2B, the survival curves in MPN patients according to the patient disposition are shown. As expected, the rate of death in ICU patients was 50%, compared to 25% and 6% for cases followed in the regular ward and at home, respectively (p < 0.001).
Table 1.
MPN-COVID clinical and laboratory characteristics in survivors and non-survivors.
| N non-missing | TOTAL | Survivors | Non-survivors | p value | |
|---|---|---|---|---|---|
| N = 175 | N = 125 | N = 50 | |||
| Sex (female/male), n (%) | 175 | 73/102 (41.7%/58.3%) | 58/67 (46.4%/53.6%) | 15/35 (30.0%/70.0%) | 0.062 |
| Age, years, median (IQR) | 174 | 71.0 (60.0–79.9) | 69.5 (57.6–78.9) | 74.1 (69.6–82.1) | 0.001 |
| <60 years, n (%) | 42 (24.1%) | 40 (32.0%) | 2 (4.1%) | <0.001 | |
| 60–70 years, n (%) | 37 (21.3%) | 24 (19.2%) | 13 (26.5%) | ||
| >70 years, n (%) | 95 (54.6%) | 61 (48.8%) | 34 (69.4%) | ||
| MPN type, n (%) | 175 | ||||
| Essential thrombocythemia | 51 (29.1%) | 38 (30.4%) | 13 (26.0%) | 0.56 | |
| Polycythemia vera | 46 (26.3%) | 37 (29.6%) | 9 (18.0%) | 0.12 | |
| Myelofibrosis | 60 (34.3%) | 36 (28.8%) | 24 (48.0%) | 0.016 | |
| Prefibrotic primary myelofibrosis | 18 (10.3%) | 14 (11.2%) | 4 (8.0%) | 0.53 | |
| Time from MPN to COVID-19 diagnosis, years, median (IQR) | 174 | 6.0 (3.1–11.0) | 6.0 (3.1–10.4) | 5.8 (3.2–11.5) | 0.81 |
| JAK2V617F, n (%) | 170 | 123 (72.4%) | 90 (73.2%) | 33 (70.2%) | 0.70 |
| CALR, n (%) | 95 | 27 (28.4%) | 20 (30.3%) | 7 (24.1%) | 0.54 |
| MPL, n (%) | 90 | 5 (5.6%) | 2 (3.2%) | 3 (10.7%) | 0.15 |
| Palpable splenomegaly, n (%) | 153 | 41 (26.8%) | 28 (26.2%) | 13 (28.3%) | 0.92 |
| MPN drugs, n (%) | |||||
| Hydroxyurea | 175 | 79 (45.1%) | 60 (48.0%) | 19 (38.0%) | 0.23 |
| Discontinued after COVID-19 onset | 52 | 9 (17.3%) | 7 (21.2%) | 2 (10.5%) | 0.33 |
| Ruxolitinib | 175 | 45 (25.7%) | 25 (20.0%) | 20 (40.0%) | 0.006 |
| Discontinued after COVID-19 onset | 45 | 11 (24.4%) | 2 (8.0%) | 9 (45.0%) | 0.004 |
| Interferon | 175 | 4 (2.3%) | 4 (3.2%) | 0 (0.0%) | 0.20 |
| Anagrelide | 175 | 8 (4.6%) | 5 (4.0%) | 3 (6.0%) | 0.57 |
| Other | 175 | 5 (2.3%) | 4 (3.2%) | 1 (2.0%) | 0.67 |
| Comorbidities, n (%) | |||||
| Cerebrovascular disease | 174 | 23 (13.2%) | 17 (13.7%) | 6 (12.0%) | 0.76 |
| Chronic dialysis/Kidney disease | 174 | 19 (10.9%) | 9 (7.3%) | 10 (20.0%) | 0.015 |
| Chronic heart failure | 173 | 25 (14.5%) | 13 (10.6%) | 12 (24.0%) | 0.023 |
| Chronic obstructive pulmonary disease | 174 | 25 (14.4%) | 16 (12.9%) | 9 (18.0%) | 0.39 |
| Current/former tobacco smoker | 152 | 35 (23.0%) | 22 (20.2%) | 13 (30.2%) | 0.19 |
| Hyperlipidemia | 168 | 47 (28.0%) | 36 (29.8%) | 11 (23.4%) | 0.41 |
| Hypertension | 171 | 104 (60.8%) | 70 (57.9%) | 34 (68.0%) | 0.22 |
| Diabetes mellitus | 172 | 23 (13.4%) | 12 (9.8%) | 11 (22.4%) | 0.027 |
| Blood values at COVID-19 diagnosis, median (IQR) | |||||
| Hemoglobin, g/dL | 145 | 12.4 (10.0–13.6) | 12.6 (10.4–13.5) | 11.7 (9.5–13.9) | 0.37 |
| White blood cells, ×109/L | 145 | 6.5 (4.6–10.1) | 6.4 (4.5–9.2) | 7.1 (5.0–11.7) | 0.14 |
| Lymphocytes, ×109/L | 130 | 0.9 (0.6–1.6) | 0.9 (0. 6–1.6) | 0.7 (0.6–1.1) | 0.098 |
| Neutrophils/lymphocytes ratio | 125 | 5.4 (3.4–8.9) | 5.0 (3.3–8.6) | 7.3 (4.5–9.9) | 0.029 |
| Platelets, ×109/L | 143 | 252.0 (152.0–394.0) | 260.5 (170.5–437.5) | 245.0 (121.0–338.0) | 0.14 |
| O2 saturation, %, median (IQR) | 114 | 93.0 (88.0–96.0) | 94.0 (91.0–96.0) | 90.0 (88.0–93.0) | <0.001 |
| Disposition, n (%) | 175 | <0.001 | |||
| Home | 40 (22.9%) | 38 (30.4%) | 2 (4.0%) | ||
| Regular ward | 116 (66.3%) | 80 (64.0%) | 36 (72.0%) | ||
| Intensive care unit | 19 (10.9%) | 7 (5.6%) | 12 (24.0%) | ||
| Main symptoms, n (%) | |||||
| Fever | 175 | 140 (80.0%) | 99 (79.2%) | 41 (82.0%) | 0.68 |
| Dispnea | 175 | 97 (55.4%) | 66 (52.8%) | 31 (62.0%) | 0.27 |
| Gastrointestinal | 175 | 22 (12.6%) | 12 (9.6%) | 10 (20.0%) | 0.061 |
| Need of respiratory support, n (%) | 174 | 103 (59.2%) | 59 (47.2%) | 44 (89.8%) | <0.001 |
| None | 71 (40.8%) | 66 (52.8%) | 5 (10.2%) | <0.001 | |
| Non-invasive | 83 (47.7%) | 53 (42.4%) | 30 (61.2%) | ||
| Invasive | 20 (11.5%) | 6 (4.8%) | 14 (28.6%) | ||
| COVID-19 drugs, n (%) | |||||
| Steroid | 162 | 45 (27.8%) | 28 (23.7%) | 17 (38.6%) | 0.060 |
| Antibiotic | 162 | 114 (70.4%) | 77 (65.8%) | 37 (82.2%) | 0.040 |
| Hydroxychloroquine | 166 | 100 (60.2%) | 73 (59.8%) | 27 (61.4%) | 0.86 |
| Antiviral | 166 | 57 (34.3%) | 43 (35.8%) | 14 (30.4%) | 0.51 |
| Lopinavir/Ritonavir | 53 | 46 (86.8%) | 33 (82.5%) | 13 (100.0%) | 0.17 |
| Other | 53 | 7 (13.2%) | 7 (17.5%) | 0 (0.0%) | |
| Experimental | 170 | 19 (11.2%) | 11 (8.9%) | 8 (17.0%) | 0.13 |
| Tocilizumab | 19 | 15 (78.9%) | 7 (63.6%) | 8 (100.0%) | 0.39 |
| Ruxolitinib | 19 | 2 (10.5%) | 2 (18.2%) | 0 (0.0%) | |
| Other | 19 | 2 (10.5%) | 2 (18.2%) | 0 (0.0%) | |
| Antithrombotic | 166 | 93 (56.0%) | 70 (57.9%) | 23 (51.1%) | 0.44 |
| Direct oral anticoagulants | 92 | 2 (2.2%) | 2 (2.9%) | 0 (0.0%) | 1.00 |
| Low molecular weight heparin | 92 | 89 (96.7%) | 66 (95.7%) | 23 (100.0%) | |
| Warfarin | 92 | 1 (1.1%) | 1 (1.4%) | 0 (0.0%) | |
Univariate analysis.
MPN myeloproliferative neoplasms.
Fig. 2. Survival by MPN phenotypes and patients disposition.
Kaplan Meier survival estimates stratified by MPN phenotypes (A) and patients disposition (B). P-values are calculated by log-rank test. MF myelofibrosis, MPNs myeloproliferative neoplasms, ICU intensive care unit.
Role of MPN-directed therapy on survival
At the time of COVID-19 diagnosis, 45.1% of the patients were receiving hydroxyurea (HU), 25.7% ruxolitinib, 2.3% interferon, 4.6% anagrelide, and 2.3% other drugs, while 19.4% were not receiving MPN-directed therapies. Stratification of MPN-directed ongoing treatment at COVID-19 diagnosis by survival category revealed a significantly higher proportion of non-survivors (40%) among the 45 patients (ET n = 2; PV n = 8; MF n = 34; pre-PMF n = 1) who were on Ruxo (p = 0.006).
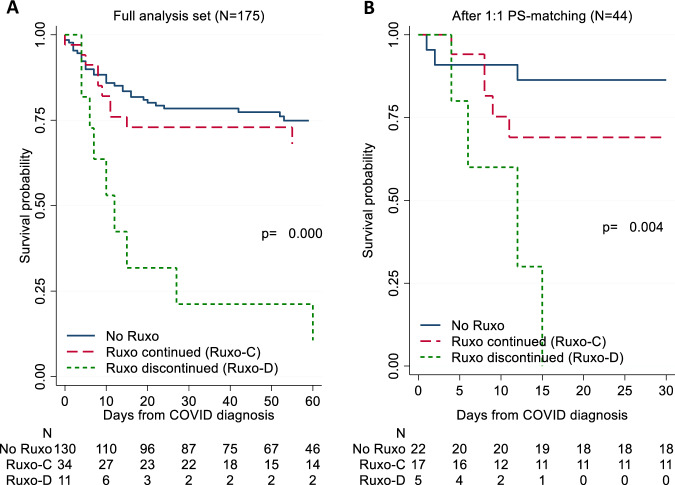
After COVID-19 diagnosis, 11.4% and 24.4% of the patients on HU and Ruxo, respectively, stopped treatment. Of note, median time from COVID-19 diagnosis to Ruxo discontinuation was 0.5 days, i.e. immediately subsequent to diagnosis. By analysing survival of patients who continued vs. those who stopped Ruxo therapy during the infection course, we found that the latter had higher mortality (Fig. 3A). Survival probabilities at 60 days from COVID-19 diagnosis were 75%, 68%, and 11% in no Ruxo, Ruxo continued, and Ruxo discontinued groups, respectively (p < 0.001). We found no such difference for the patients who stopped HU after COVID-19 diagnosis.
Fig. 3. Survival by Ruxolitinib exposure and discontinuation.
Kaplan Meier survival estimates stratified by the use and discontinuation of Ruxo in the full analysis set (A) and after PS matching (B). P-values are calculated by log-rank test. PS propensity score, Ruxo ruxolitinib.
Risk factors for mortality
We included in a logistic multivariable model the most relevant risk factors for survival found in univariate analysis, i.e. age, sex, MF phenotype, chronic heart disease, need of respiratory support, ICU admission, Ruxo treatment and Ruxo discontinuation during the coronavirus infection. Of these, age (OR = 1.07, p = 0.003), male gender (OR = 2.48, p = 0.047), admission to ICU (OR = 3.6, p = 0.037), need of respiratory support (OR = 10.4, p < 0.001) and Ruxo discontinuation (OR = 8.4, p = 0.040) were statistically significant; however, some confidence intervals (CIs), in particular the one for Ruxo discontinuation, were wide, likely due to the low number of available events compared to number of confounders (Table 2). Nevertheless, these significances were statistically robust to inclusion of both the known relevant COVID-19-related and patient-related confounders.
Table 2.
Multivariable analysis of risk factors for mortality after COVID-19 diagnosis.
| OR (95% CI) | p value | |
|---|---|---|
| Age | 1.07 (1.02–1.11) | 0.003 |
| Male sex | 2.48 (1.01–6.07) | 0.047 |
| MF diagnosis | 1.69 (0.61–4.66) | 0.315 |
| Chronic heart disease | 2.18 (0.64–7.36) | 0.210 |
| Respiratory support | 10.0 (2.94–34.0) | <0.001 |
| ICU admission | 4.93 (1.36–17.9) | 0.015 |
| Ruxolitinib administration | 2.40 (0.72–8.02) | 0.154 |
| Ruxolitinib discontinuation | 8.51 (1.14–63.4) | 0.037 |
OR odds ratio, CI confidence interval, MF myelofibrosis, ICU intensive care unit.
Moreover, to provide more robust evidence of this finding, we performed a sensitivity analysis by PS matching. Baseline characteristics before and after PS matching (Table S1) showed that those factors linked to the severity of MPN and those associated with COVID-19 were well balanced in the two groups. Patients for whom no match was found were excluded, leading to a reduced number of matched pairs (22 exposed/nonexposed pairs). The overall picture of comparative profiles is well illustrated in Fig. 3B, confirming the negative effect of Ruxo discontinuation found in the analysis on the full patient set (Fig. 3A).
Discussion
In this multicenter European study, we describe a relatively large MPN series of 175 patients with MPN and COVID-19, collected under conditions of exceptional COVID-19 lethality, from February to June 2020, and report estimates and risk factors of mortality. In our sample, death occurred more frequently among males, and, as expected, in older aged patients. Of note, MF was the most represented among the MPNs (34%) and HU was the most common treatment (45.1%). Ruxo was used in 25% of cases, a figure that is consistent with recent reports on real-world clinical treatment of MPNs [8, 9] and in agreement with the high prevalence of MF in our cohort.
All the 135 hospitalized patients and 20 (50%) of those treated at home had COVID-19 diagnosis confirmed by a positive result on polymerase chain reaction testing of a nasopharyngeal sample; the remaining 20 patients managed at home were diagnosed based on highly suggestive symptomatology. In this regard, it should be noted that during the period in which the study was conducted, the possibility of carrying out diagnostic tests with nasopharyngeal swab was limited, especially in patients at home.
Concerning COVID-19 presentation and therapy, our MPN patients did not differ from the general non-MPN population under the profile of inflammatory markers or comorbidities, and drug treatment was consistent with therapeutic standards at the time. In particular, the use of steroids was very limited. Estimates of mortality were higher than in the general population and did not differ by Country. Comparable figures of mortality were reported in a recent Italian study on COVID-19 and hematological malignancies, in which 27 of 83 patients with MPN died (33%) [10]. Notably, mortality in our patients with MF was higher (48%) in comparison to ET, PV, or pre-PMF (~25%), suggesting a higher vulnerability of patients with MF to COVID-19. As expected, the highest rate of mortality was in patients admitted to ICU (50%), while it was only 25 and 6% in patients admitted to regular ward or at home. When compared with mortality rate found in the general population in Italy and Spain, the two Countries with the highest number of patients in this study, these rates exceed the overall case-fatality rate reported in Italy (12.8%) in the same period and are comparable to those observed in Spain (23%) only for ET, PV, and pre-PMF but not for MF (48%) [11, 12].
An unexpected result of this study was the association between Ruxo and overall mortality that was found in 40% of the 45 cases treated with this drug. In further investigations by multivariable analysis and PS matching, we highlighted that this effect was due not so much to drug exposure but to its rapid discontinuation (median 0.5 days), that accounted for 75% of deaths. In fact, in a multivariable analysis we identified a set of variables that were significantly correlated with mortality, even adjusting for known confounders (age, gender, respiratory support and admission to ICU). Contrary to what observed in univariate analysis, in logistic regression we found no effect of Ruxo treatment alone versus the other treatments; instead, we confirmed a significant increase in mortality related to its discontinuation. Treatment stop might be due to several reasons, such as uncertainties about possible adverse events, risk of interactions with other drugs, and possibly missing interactions between care managing physicians and referring hematologist. This is a potential bias in our analysis; however, a lot of effort was put into adjusting for all measured known confounders, which led us to perform a sensitivity analysis by PS matching which confirmed the previous findings.
The biological plausibility of this effect may be related to the devastating enhancement of inflammation (“cytokine storm”) that follows the abrupt suspension of Ruxo, leading to the “ruxolitinib discontinuation syndrome”, which in turn may lead to fatal clinical complications and multiorgan failure [13, 14]. On the other hand, in observational studies, Ruxo itself has proven effective in improving survival in COVID-19 patients from the general population [15–17].
The main constraint of this study is the limited number of patients and, consequently, of events; this holds particularly true for the discontinued Ruxo group, which only consisted of 11 patients. However, we believe this work suggests that Ruxo treatment in MPN patients who are already assuming it at COVID-19 diagnosis should be continued, in agreement with the ASH recommendations (https://www.hematology.org/covid-19/covid-19-and-myeloproliferative-neoplasms). Of course, we acknowledge that this is a limitation that can hardly be avoided when dealing with both a rare condition, such as the MPN, and an exceptional situation of emergency, such as the COVID-19 crisis. Moreover, we stress that this study was originally designed to assess mortality and other outcomes in a vulnerable subpopulation of patients with COVID-19, and as such has not adequate power to test specific hypotheses about treatment effects. Yet, the effect of Ruxo discontinuation in our analysis was striking and robust to multiple confounders correction and sensitivity analysis.
This preliminary observation is, in our opinion, worthy of dissemination amongst patients and clinician communities, although it needs to be confirmed in a study with a longer follow-up and a sample size adequate to test the hypothesis. Efforts are currently undergoing to put in motion an observational study that meets these requirements.
Supplementary information
Acknowledgements
The study was supported by a research grant from the COVID “3 × 1 project”, BREMBO S.p.A., Bergamo, Italy.
Author contributions
TB conceived and designed the study, supervised the analysis, and wrote the paper. CH, AMV, AR, VDS, AAL revised the study and contributed to manuscript writing. AM directed the project. AC, AG, AF performed statistical analysis. AAL, AI, GR, EE, MMAC, MGK, JJK, FP, GB, VGG, MLF, MAF, CMM, ER, SO, PP, MBo, KSQC, MSS, GCT, MAS, FL, AP, BNE, AA, EMM, SK, MR, BC, JCHB, ELA, BXC, PG, MG, DC, RD, FC, BB, LB, NCG, MBe, SB collected data. All authors revised and approved the final version of the manuscript.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version of this article (10.1038/s41375-020-01107-y) contains supplementary material, which is available to authorized users.
References
- 1.Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA. 2020;323:2052–9. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xu J, Wang L, Zhao L, Li F, Liu J, Zhang L, et al. Risk assessment of venous thromboembolism and bleeding in COVID-19 patients. Research Square 2020. On line publication. 10.21203/rs.3.rs-18340/v1. [DOI] [PMC free article] [PubMed]
- 3.Carsana L, Sonzogni A, Nasr A, Rossi RS, Pellegrinelli A, Zerbi P, et al. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: a two-centre descriptive study. Lancet Infect Dis. 2020;20:1135–40. doi: 10.1016/S1473-3099(20)30434-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Breccia M, Piciocchi A, De Stefano V, Finazzi G, Iurlo A, Fazi P, et al. COVID-19 in Philadelphia-negative myeloproliferative disorders: a GIMEMA survey. Leukemia. 2020;34:2813–4. doi: 10.1038/s41375-020-01032-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Palandri F, Piciocchi A, De Stefano V, Breccia M, Finazzi G, Iurlo A, et al. How the coronavirus pandemic has affected the clinical management of Philadelphia-negative chronic myeloproliferative neoplasms in Italy-a GIMEMA MPN WP survey. Leukemia. 2020;34:2805–8. doi: 10.1038/s41375-020-0953-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barbui T, Tefferi A, Vannucchi AM, Passamonti F, Silver RT, Hoffman R, et al. Philadelphia chromosome-negative classical myeloproliferative neoplasms: revised management recommendations from European LeukemiaNet. Leukemia. 2018;32:1057–69. doi: 10.1038/s41375-018-0077-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70:41–55. doi: 10.1093/biomet/70.1.41. [DOI] [Google Scholar]
- 8.Vannucchi AM, Sordi B, Morettini A, Nozzoli C, Poggesi L, Pieralli F, et al. Compassionate use of JAK1/2 inhibitor ruxolitinib for severe COVID-19: a prospective observational study. Leukemia. 2020;19:1–13. doi: 10.1038/s41375-020-01018-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pemmaraju N, Yu J, Parasuraman S, Paranagama D, Kish J, Visaria J, et al. Ruxolitinib (RUX) retreatment in patients (Pts) with myelofibrosis (MF): Real-world evidence on pt characteristics and outcomes. J Clin Onc. 2020;38:e19535.
- 10.Passamonti F, Cattaneo C, Arcaini L, Bruna R, Cavo M, Merli F, et al. Clinical characteristics and risk factors associated with COVID-19 severity in patients with haematological malignancies in Italy: a retrospective, multicentre, cohort study. Lancet Haematol. 2020;7:e737–45. doi: 10.1016/S2352-3026(20)30251-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA. 2020;323:1775–6. doi: 10.1001/jama.2020.4683. [DOI] [PubMed] [Google Scholar]
- 12.Posso M, Comas M, Román M, Domingo L, Louro J, González C, et al. Comorbidities and mortality in patients with COVID-19 aged 60 years and older in a university hospital in Spain. Arch Bronconeumol. 2020;S0300-2896:30215–5. doi: 10.1016/j.arbres.2020.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tefferi A, Pardanani A. Serious adverse events during ruxolitinib treatment discontinuation in patients with myelofibrosis. Mayo Clin Proc. 2011;86:1188–91. doi: 10.4065/mcp.2011.0518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Coltro G, Mannelli F, Guglielmelli P, Pacilli A, Bosi A, Vannucchi AM. A life-threatening ruxolitinib discontinuation syndrome. Am J Hematol. 2017;92:833–8. doi: 10.1002/ajh.24775. [DOI] [PubMed] [Google Scholar]
- 15.La Rosée F, Bremer HC, Gehrke I, Kehr A, Hochhaus A, Birndt S, et al. The Janus kinase 1/2 inhibitor ruxolitinib in COVID-19 with severe systemic hyperinflammation. Leukemia. 2020;34:1805–15. doi: 10.1038/s41375-020-0891-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heidel F, Hochhaus A. Holding CoVID in check through JAK? The MPN-approved compound ruxolitinib as a potential strategy to treat SARS-CoV-2 induced systemic hyperinflammation. Leukemia. 2020;34:1723–5. doi: 10.1038/s41375-020-0898-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Koschmieder S, Jost E, Cornelissen C, Müller T, Schulze-Hagen M, Bickenbach J, et al. Favorable COVID-19 course despite significant comorbidities in a ruxolitinib-treated patient with primary myelofibrosis. Eur J Haematol. 2020;105:655–8. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.