Abstract
Mesenchymal stem cells (MSCs) are the most frequently used stem cells in clinical trials due to their easy isolation from various adult tissues, their ability of homing to injury sites and their potential to differentiate into multiple cell types. However, the realization that the beneficial effect of MSCs relies mainly on their paracrine action, rather than on their engraftment in the recipient tissue and subsequent differentiation, has opened the way to cell-free therapeutic strategies in regenerative medicine. All the soluble factors and vesicles secreted by MSCs are commonly known as secretome. MSCs secretome has a key role in cell-to-cell communication and has been proven to be an active mediator of immune-modulation and regeneration both in vitro and in vivo. Moreover, the use of secretome has key advantages over cell-based therapies, such as a lower immunogenicity and easy production, handling and storage. Importantly, MSCs can be modulated to alter their secretome composition to better suit specific therapeutic goals, thus, opening a large number of possibilities. Altogether these advantages now place MSCs secretome at the center of an important number of investigations in different clinical contexts, enabling rapid scientific progress in this field.
Keywords: Mesenchymal stem cells, Secretome, Soluble factors, Extra-cellular vesicles, Exosomes, Bone regeneration
Core Tip: Mesenchymal stem cells (MSCs) produce a high number of bioactive molecules and extracellular vesicles, known as secretome, which exerts important paracrine effects on neighbouring cells and tissues. The use of MSCs secretome in tissue regeneration therapies would circumvent the problems linked to MSCs-based therapies, such as low cell survival and engraftment, which importantly limit their therapeutic efficacy, or the negative side effects associated with the administration of these cells.
INTRODUCTION
Over the last few years, mesenchymal stem/stromal cells (MSCs) have emerged as a promising therapeutic option, being the focus of many studies in the field of regenerative medicine. This specific type of stem cells has unparalleled features that make them a valuable tool for tissue repair. MSCs are fibroblast-like cells, present in various adult tissues that have retained a high degree of plasticity, and are able to differentiate into various mesodermal, ectodermal and endodermal lineages[1]. Although initially it was precisely this differentiation potential, together with the ability of homing to injury sites, that attracted the interest of the scientific community, it has been shown that the regenerative potential of MSCs is overall very limited, particularly in pathological environments. MSCs are present in many tissues, but in very low numbers, making necessary an expansion period upon harvesting in order to be able to obtain the number of cells needed for the subsequent procedure. Even when this expansion step is successful and a high number of cells are transplanted in the procedure, the cells frequently have very reduced viability and low engraftment in the recipient tissue[2]. Factors such as the age of the donor, the number of passages during the in vitro expansion, the culture conditions, the administration procedure and the pathological microenvironment encountered by the transplanted cells, adversely impact the cells ability to survive and engraft in the recipient tissue. Even after successful engraftment, MSCs seem to be unable to properly direct angiogenesis, a crucial step for successful tissue regeneration[3]. Currently many studies are trying to overcome these drawbacks by genetically modifying MSCs and optimizing their culture conditions[4], but overall, important limitations still remain concerning MSC biosafety. Recent studies have shown the potential tumorigenicity of MSCs and the risk of infection during the isolation and administration procedures[5,6], the possibility of transmission of zoonotic diseases due to the use of fetal bovine serum during the in vitro expansion stage[7], their putative pro-fibrogenic potential and lung-entrapment related embolism after systemic administration of MSCs[8,9] and the heterogeneity of the MSC population in terms of differentiation potential[10]. Despite all the constraints linked to the therapeutic procedures using MSCs, these cells could still be highly useful in the regenerative medicine field due to other important qualities. While initially the techniques related to MSCs in tissue engineering have been based on their cellular aspects, many studies now support that the beneficial effect of transplanted MSCs reported in some applications is related to their important paracrine activity rather than to their ability to differentiate towards specific cell lineages. In fact, as much as 80% of the regenerative potential of transplanted MSCs has been linked to that paracrine activity[11]. MSCs produce and secrete a wide variety of bioactive molecules in response to different microenvironment conditions and are even referred to as “trophic factories”. The combination of all the trophic factors or molecules secreted by these cells to the extracellular space is known as secretome. The term secretome includes two different components: A soluble fraction, mainly constituted by cytokines, chemokines, immuno-modulatory molecules and growth factors[12], and a vesicular fraction, comprised of different types of vesicles with a crucial role in the delivery of microRNAs and proteins involved in cell-to-cell communication[13].
The use of the MSCs-derived secretome in tissue engineering has many important advantages over stem-cell based applications[14]. Firstly, it avoids the surgical intervention needed to source the cells and the associated risks involved as well as the subsequent in vitro expansion of those cells. Secondly, the use of secretome also prevents all the aforementioned drawbacks linked to the administration of MSCs. Moreover, the secretome can be considered a pharmaceutical agent and, thus, it might be evaluated in the same way for dosage and safety. However, most importantly, the composition of the secretome is notably affected by diverse stimuli present in the microenvironment, allowing us to alter it to better suit specific therapeutic goals.
Although the research on the regenerative capacity of MSCs secretome is scarce compared to that on the regenerative potential of MSCs, the preclinical tests performed up to date have already shown significant positive results and few adverse effects linked to the use of this product. The current review aims to assess the role of MSCs secretome as the main trigger of their regenerative activity, describing the main components of the soluble and vesicular parts and the key biological processes in tissue regeneration positively affected by these bioactive components.
MSCs-DERIVED SOLUBLE FACTORS
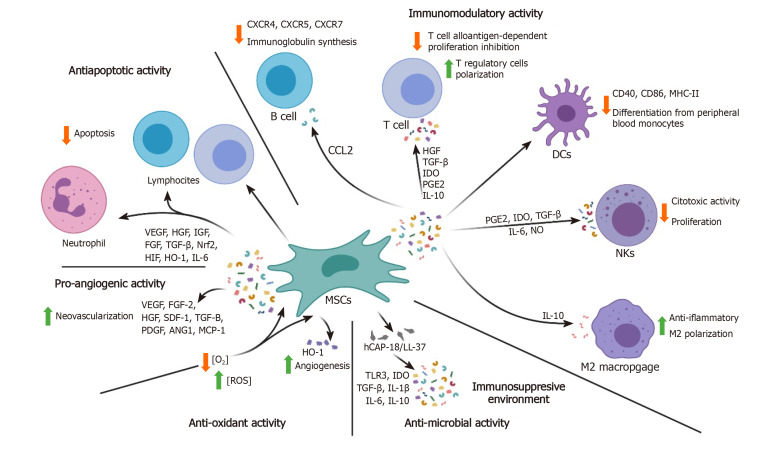
MSCs are able to secrete a variety of autocrine and paracrine factors including cytokines, chemokines, extracellular matrix (ECM) proteases and growth factors, enabling the possibility to use them as a potential cell-free based therapy source[15]. The bone marrow (BM)-MSCs derived biomolecules have been widely investigated to better clarify their potential[16]. In fact, proteomic analyses have revealed the presence of up to 1533 proteins[17,18] that participate in different biological processes. A summary of this information is displayed in Figure 1.
Figure 1.
Summary of the various soluble factors secreted by mesenchymal stem cells and their functions. HGF: Hepatocyte growth factor; TGF-β: Transforming growth factor–β; IDO: Indoleamine 2,3-dioxygenase; PGE2: Prostaglandin E2; IL: Interleukin; DCs: Dendritic cells; CCL: CC-chemokine ligand; NKs: Natural killer cells; MSCs: Mesenchymal stem cells; CXCR: C-X-C chemokine receptor type; VEGF: Vascular endothelial growth factor; IGF: Insulin-like growth factor; FGF: Fibroblast growth factor; Nrf2: Nuclear factor erythroid-related factor 2; HIF: Hypoxia-inducible factor; SDF: Stromal cell-derived factor; PDGF: Platelet-derived growth factor; ANG1: Angiogenesis 1; MCP-1: Monocyte chemotactic protein-1; ROS: Reactive oxygen species; hCAP: Human cathelicidin anti-microbial peptide; HO-1: Heme oxygenase; NO: Nitric oxide.
Modulation of the immune system and inflammation
MSCs are able to affect innate and adaptive immune system responses to different stimuli by means of cell-to-cell contact, but also through the secretion of specific factors. There is mounting evidence of a relation between MSCs soluble factors and immune cells such as lymphocytes, dendritic cells (DCs), natural killer (NK) cells and macrophages. MSCs have an immunosuppressive role towards myeloid DCs. MSCs down-regulate the surface expression of co-stimulatory molecules such as, CD40 and CD86, and the major histocompatibility complex II (MHC-II), and hamper the initial differentiation of DCs from peripheral blood monocytes, producing, overall hypo-responsive T-cells[19]. The secretion of prostaglandin E2 (PGE2), indoleamine 2,3-dioxygenase (IDO), transforming growth factor–β1 (TGF-β1), interleukin-6 (IL-6) and nitric oxide (NO) by MSCs has shown to strongly weaken NK cytotoxic activity and proliferation[20].
MSCs also have an important role in the regulation of macrophages, whose action is critical for inflammation response and tissue regeneration. Macrophages can generate from monocytes through a classical activation pathway (M1) or by an alternative pathway (M2). Whereas M1 macrophages are characterized by the production and release of pro-inflammatory cytokines, M2 macrophages are considered to have an anti-inflammatory role and are responsible for the release of IL-10. MSCs promote the M2 polarization of macrophages both in vivo and in vitro[21] via production of anti-inflammatory mediators such as IL-10, and inflammatory cytokines down-modulation[22].
Concerning the adaptive immune response, MSCs can also have an effect on T and B-lymphocytes activation, proliferation and differentiation. MSCs can inhibit T-cell alloantigen-dependent proliferation in a dose-dependent manner. Among the secreted factors affecting T-cell biology, we can distinguish hepatocyte growth factor (HGF), TGF-β1, IDO, PGE2 and IL-10. The differentiation pathway is also polarized toward T regulatory cells, one of the CD4+ subclasses of T lymphocytes involved in the cellular immune response and preventing autoimmune diseases[20,23]. Moreover, MSCs can encourage regulatory T type 1 (Tr1)-like cells that are characterized by interferon-γ (IFN-γ) and IL-10 secretion[24]. Concordant with these data, graft-versus-host disease patients who received treatment with MSCs showed a subsequent increase in CD4+ CD25+ Foxp3+ and Tr1 populations[25]. With regard to B cells, their humoral activity is also affected by MSCs. MSCs can diminish the amount of chemokine receptors such as C-X-C chemokine receptor type 4 (CXCR4), CXCR5 and CXCR7, decreasing their overall activation rate, although the expression of molecules implicated in the antigen presentation step is not affected. Also, MSCs-derived CC-chemokine ligand-2 suppresses signal transducer and activator of transcription 3 (STAT3) activity, resulting in down-regulation of Paired box 5 totally inhibiting normal immunoglobulin synthesis. Therefore, B-cells chemotactic activity and antibody production are extremely affected after MSCs injection[26].
One aspect worth stressing is the fact that MSCs immunomodulation is not permanently effective but rather relies on the strength and type of inflammatory stimulation. In fact, MSCs can sense the surrounding microenvironment and increase or diminish their activity[27] driving a dual effect regarding inflammation modulation. Initially, damaged tissue needs an acute inflammation response. However, the subsequent repair phase, is favoured by an immunosuppressive state. Toll-like receptors (TLRs) guide MSCs during these phases with TLR4 triggering a pro-inflammatory response and TLR3 repression of immune system activity. The interaction between MSCs and inflammatory molecules can affect MSCs paracrine effects[28,29], where an abundance of pro-inflammatory mediators [e.g. tumor necrosis factor-α (TNF-α) and IFN-γ], typical of tissue injuries, will facilitate a MSCs-dependent pro-inflammatory response. On the other hand, the activation of cytokines such as IL-1 and IL-6 supports the down-regulation of B and T cells activity[30].
Anti-apoptotic activity
In conditions characterized by a strong inflammatory response, such as sepsis and cell injuries, endothelial, epithelial and immune cells undergo apoptosis by activation of caspase signaling. The impact on the host is intense immunosuppression and lymphopenia. In several studies, it has been demonstrated that MSCs are capable of modulating apoptotic signals through the secretion of paracrine mediators such as vascular endothelial growth factor (VEGF), HGF, insulin-like growth factor (IGF), fibroblast growth factor (FGF), TGF, nuclear factor erythroid-related factor 2, hypoxia-inducible factor (HIF) and heme oxygenase (HO-1), but also through direct contact with the affected cell types, such as T and B cells[31]. MSCs can delay lymphocytes and neutrophil apoptosis through an IL-6-mediated mechanism that ultimately induces STAT3 nuclear translocation and decreases reactive oxygen species (ROS)[19,31,32]. The anti-apoptotic role of MSCs in combination with the immunomodulation of pro-inflammatory signals and molecules has been associated with faster bone repair, mainly through the down-regulation of IL-6 and TNF-α, thus, reducing the pro-apoptotic effects taking place in bone cells[29].
Pro-angiogenic activity
MSCs promote neovascularization of injured tissues through the release of paracrine factors such as VEGF, basic FGF2, HGF, stromal cell-derived factor-1 (SDF-1), TGF-β, platelet-derived growth factor (PDGF), angiopoietin-1 (Ang-1) and monocyte chemotactic protein-1 (MCP-1)[33,34]. These factors are capable of ameliorating tissue vascularity and of promoting endothelial progenitor cells differentiation. This property could be exploited to sustain tissue/organ perfusion in ischemic settings and, through a synergistic effect between IGF-1 and VEGF, also in bone regeneration[35,36]. Hypoxia levels strictly control the secretion of angiogenic factors by MSCs. It has been shown that MSCs are able to increase the secretion of angiogenic molecules when oxygen supply decreases. Moreover, lipopolysaccharide and the inflammatory mediators normally found in sepsis are able to up-regulate MSCs-dependent neovascularization[12].
Anti-microbial activity
The protective effect of MSCs in sepsis conditions also includes an anti-microbial activity against harmful agents that can be attributed to the release of human cathelicidin anti-microbial peptide (hCAP-18/LL-37)[19], a host defense peptide that seems to be able to enhance the expression of TLR3 as well as IDO, IL-10, TGF-β, IL-6, and IL-1β, thus providing an immunosuppressive environment that could be useful in settings where the immune system response is abnormally intensified, as previously mentioned[37]. Interestingly, in an experiment carried out by Krasnodembskaya et al[38], MSCs and their conditioned media were able to decrease both Gram+ (Staphylococcus aureus) and Gram- (Escherichia coli and Pseudomonas aeruginosa) growth in vitro[38].
Anti-oxidant activity
Oxidative stress, due to an imbalance between oxidants and antioxidants, is a major problem in cell survival. The presence of insults such as UV irradiation, heavy metals, pathogens, hypoxia or xenobiotics, leads to the generation of ROS that can induce damage to proteins and nucleic acids and produce mutations leading to carcinogenesis[39-41]. In the case of hypoxia-induced injury and ROS accumulation, MSCs are able to up-regulate the expression of HO-1, an enzyme with antioxidative, anti-inflammatory and antiapoptotic effects[42]. HO-1 up-regulation appears to correlate with the release of pro-angiogenic growth factors, such as VEGF-A, SDF-1 and HGF[42].
Autophagy modulation
Autophagy can be defined as the removal of macromolecules and organelles from their original localization to the lysosome and their subsequent recycling to maintain cell homeostasis. This “self-eating” process is induced during fasting and stress conditions and is essential for cell survival. Autophagy has two different types of implications regarding MSCs. MSCs autophagy can be modulated, but also autophagy of other cells can be modulated by MSCs. Firstly, autophagy in MSCs can actually affect MSC functions, and in particular their differentiation potential. Undifferentiated MSCs contain more autophagosomes than differentiated cells, due to a malfunction of the autophagosome-lysosome fusion. During the differentiation process there is a transient activation of autophagy that will allow the cells to obtain all the energy and molecules necessary to support the changes associated with cell differentiation[43,44]. There are also some data linking MSCs autophagy with their capacity to modulate immune response. According to the work performed by Pantovic et al[44], in a mouse model of autoimmune encephalomyelitis, inhibition of autophagy increases the immunosuppressive action of mouse MSCs. On the other hand, MSCs can also modulate autophagy of cells residing in the same tissue or of cells that have been recruited to that tissue. However, these results are at odds with those presented by Gao et al[45] that correlate MSCs immunosuppressive actions with their autophagy levels. There are also conflicting data regarding the effect of MSC conditioned media on autophagy. The secretion of the neurotropic nerve growth factor (NGF) was found to be associated with autophagy abrogation in n-hexane-induced neuropathy and hexanedione-induced autophagy via the NGF-PI3K/Akt/mTOR signaling pathway[46,47]. On the other hand, it was demonstrated to exert a paracrine effect on hypoxia/reoxygenation-induced damage of myocardial cells by modulating the Notch2/mTOR/autophagy pathway, thus protecting them from cell death. Modulating autophagy ability is related to stemness, anti-oxidative stress, anti-apoptosis, and pro-survival capacity of MSCs, therefore representing a key characteristic for cellular or tissue injury therapies[48,49].
Induction of cell proliferation
MSCs have also been shown to produce factors that can induce cell proliferation in keratinocytes, endothelial cells, skin fibroblasts and lung fibroblasts[50-53]. Analysis of the mitogenic factors present in the conditioned media produced by cord blood MSCs and BM-MSCs showed the presence of PDGF-AA; however, since this proliferation was not affected by the presence of anti-PDGF antibodies or PDGF receptor antibodies, the induction of proliferation could not be directly attributed to the presence of this factor. Interestingly, this mitogenic effect seems to be blocked by Wnt antagonists, such as secreted frizzle related protein-1 (SFRP-1) indicating a relevant role of the secreted Wnt proteins present in the conditioned media (1, 2 and 7b) in promoting cell proliferation[52].
All the features explained in this section apply in general to MSCs but it is now accepted that MSC phenotypic and functional heterogeneity corresponds to several population subsets strongly performing in unique modalities under identical conditions. It is also important to highlight that MSCs effects differ significantly based on the anatomical source, isolation methodology, age of the donor, culture medium choice, etc[54-56].
EXTRACELLULAR VESICLES
In the last few years both industry and academia are focusing their interest on the insoluble part of the secretome, the Extracellular Vesicles (EVs) due to their particular properties that make them invaluable tools for therapeutic applications[57-59].
Characteristics and classification of EVs
Whereas cytokines and growth factors constitute the soluble part of the secretome, the non-soluble part is constituted by a variety of nano- and micro-sized vesicles known altogether as EVs. Although they were first described in 1946 by Chargaff and colleagues[60], it was not until 2011 that the term extracellular vesicles was proposed to refer to these particular membrane-enclosed structures[61]. EVs are secreted by a variety of cell types and can be found in the majority of body fluids, such as blood, semen, saliva, plasma, urine, cerebrospinal fluid, epididymal fluid, amniotic fluid, malignant and pleural effusions of ascites, bronchoalveolar lavage fluid, synovial fluid, and breast milk[62-64]. Although the general term of EVs refers to all lipidic vesicles secreted by the cells, they can be classified according to different criteria, such as size morphology, densities, cargo and biogenesis.
EVs were initially organized into two size categories: 1 nm diameter and 100 nm diameter vesicles[65]. Later, with the arrival of improved visualizing techniques, it was shown that the small vesicles can have a much broader size range and density and can display up to 9 different morphologies such as simple or double vesicles, tubular or oval, among others[64]. On the other hand, three different subpopulations of EVs can be differentiated according to their densities: Low-density (1.01-1.06 g/cm³), medium-density (1.08-1.14 g/cm³) and high-density (1.16-1.28 g/cm³)[66]. Interestingly, these differences in density were also accompanied by differences in their surface markers and in the proliferative and anti-apoptotic effects on renal tubular epithelial cells[66].
Another parameter that could serve to classify EVs is their cargo. Lunavat et al[67] were able to discriminate between different subpopulations of EVs secreted by melanoma according to their different profiles of micro RNAs (miRNA) and non-coding RNAs (ncRNA)[67]. This difference in miRNA cargo profiles has also been described in EVs secreted by BM-MSCs[66]. Wang et al[68] also observed differences in miRNA profiles secreted by MSCs at different stages of osteogenic differentiation[68], highlighting the fact that the cargo of EVs is determined by the physiological state of the cells.
Information on the DNA cargo is still scarce. EVs can harbour single or double stranded DNA or even mitochondrial DNA[69,70] that can be located inside the vesicle or associated externally to their lipidic membrane[63,70]. Similar to the RNA cargo, it is possible to discriminate different subpopulations of EVs according to their DNA content[70,71].
Protein markers are extremely useful in discriminating specific subpopulations of EVs. Tetraspanins, such as CD9, CD63 and CD81, together with ALIX and tumor susceptibility gene 101 (TSG101), are mainly, but not exclusively, expressed in small EVs, recovered by centrifugation at 10000 × g[72,73]. On the other hand, potential surface markers for higher EVs, isolated at 2000 × g or 10000 × g, are actinin-4 and mitofilin[72]. The physiological state of the cells also influences their protein cargo, as shown in DCs at different stages of maturation[74].
Currently, the main criterion in EVs classification is their biogenesis. According to this, we can differentiate three main classes of EVs, designated as exosomes (Exos), microvesicles (MVs) and apoptotic bodies (ApoBDs).
Exos, with a diameter range between 30 and 150 nm, are produced by the endosomal pathway. In brief, after the inward budding of the plasmatic cell membrane, the early endosome matures into multivesicular bodies (MVBs) by the inward movement of its membrane and the presence of intraluminal vesicles[63,75-77]. Whether MVBs are degraded by the lysosome or fused with the plasma membrane, seems to be determined by the cholesterol levels in these particles, since cholesterol presence is necessary to avoid lysosomal degradation[75,78]. It has been suggested that cellular homeostasis is also key in determining the fate of MVBs and that exosomes could be essential in protecting the cells against intracellular stress[79-81]. Exos will be released later when MVBs fuse with the plasma membrane. Exos production is mainly driven by the endosomal sorting complex required for transport (ESCR) proteins and their accessory proteins such as ALIX, TSG101, vacuolar protein sorting-associated protein 4 and soluble NSF attachment protein receptors (SNAREs) among others[76,77,82]. In addition, Exos can also be produced by an ESCRT-independent pathway. In cells that lack ESCRT machinery, Exos production depends on ceramides and tetraspanins (CD9, CD63, and CD81)[76,77].
MVs and ApoBDs originate from outward budding of the extracellular membrane, by the action of the cytoskeleton. The difference between these two particles is the viability of the cell[75,77,83]. MVs are produced by healthy cells, whereas ApoBDs are released by dying cells. MVs are middle size EVs with a diameter ranging from 50 nm to 1 μm. The release of MVs depends on the calcium levels inside the cell, since their production requires the action of amino phospholipid translocases, scramblases, and calpain, all of which are calcium-dependent proteins[77]. Cholesterol level and cytoskeleton components are important in MVs release[77,78]. ApoBDs are the largest EVs, which can reach 5 μm in diameter. ApoBDs formation is mediated by factors such as Rho-associated protein kinase and the plasma membrane channel Pannexin 1. The first step in ApoBDs formation is the emergence of blebs in the plasmatic membrane that later become protrusions, ending up in the release of the ApoBDs[83].
Role of EVs in cell-to-cell communication
Although EVs were first considered to simply perform a waste excretory role[82], eventually, their essential role in cell-to-cell communication was underscored. EVs encapsulate part of the cytosol giving rise to a population of phospholipid-walled particles that will play an important role in cell-to-cell communication in normal and pathological conditions.
Once released into the extracellular medium, EVs interact with both the ECM and the cells. Interestingly, EVs can carry matrix-remodelling enzymes, such as metalloproteinases[84], as well as their regulators, or can directly induce the release of the ECM remodelling enzymes from surrounding cells, overall contributing to modification of the composition of the ECM[85]. Surface molecules such as integrins, tetraspanins and proteoglycans drive the interaction between EVs and cells, being the ones on the cell surface driving the interaction[86]. This EVs-cell interaction is also influenced by the size of the EVs, and the physiological state of the acceptor cell[77,82].
Two different types of EVs mediating cell-to-cell communication can be distinguished. EVs can induce a response on a target cell by activating specific receptors on the cell surface or by directly delivering their cargo into the cell. As an example of these mechanisms, EVs secreted by activated macrophages are recognized by TLR9 on the surface of inactive macrophages, triggering the release of chemokine TNF-α. In addition, these EVs transfer cell division control protein 42 homolog (Cdc42) into recipient cells triggering the release of more EVs[87]. A direct interaction between cell surface molecules and the EVs is needed for EVs uptake. In fact, inhibitors of these surface molecules have been shown to decrease EVs uptake[86]. EVs uptake requires energy and is also cholesterol-dependent[88,89]. Endocytic uptake after EVs recognition can be as diverse as the diversity of EVs and acceptor cells. Verdera et al[88] described a reduction in uptake of EVs by epithelial cells after inhibition of macropinocytosis in 2D and 3D cell cultures[88]. Phagocytosis of EVs by monocytes is described to decrease in preeclampsia due to higher expression of CD47, which inhibits EVs uptake[90]. However, the main pathways described for EVs uptake are caveolar[88,89,91] and clathrin endocytic pathways, being the last pathways mediated by integrins[86] and by the dynamin GTPase[92]. In addition, other investigations show that EVs cargo can also be delivered by lipid raft endocytosis[82,89] and by membrane fusion[82,93].
The functions of EVs are very diverse. They take part in processes such as immune response, organogenesis, tissue repair and tissue calcification[74,85,94]. EVs perform different roles in the immune system. It has been proven that EVs secreted by immune cells are able to present antigens leading to an immune response[74]. As previously mentioned, they might also function as mediators of the immune response activating macrophages after recognition by TLR9 on the cell surface[87]. EVs secreted by active dendrocytes, might lead to the activation of immune humoral response and activation of CD4+ and CD8+ T cells. Finally, it has been shown that EVs secreted by immunosuppressor-DCs seem to be able to reduce systematic inflammation[74]. EVs are key mediators in the critical cell-to-cell communication among different cell types, even from different embryonic layers, during organogenesis[83,94]. Hayashi et al[95] observed that MSCs-derived EVs carry miRNA-135b-3p to epithelial cells, which is essential for their proliferation[95]. On the other hand, epithelial cells secrete EVs capable of inducing mineralization and dentin production driven by MSCs during teeth development[83].
EVs can also have a marked influence in disease development, including cancer. EVs secreted by cancerous cells can actually help prepare a pre-metastatic niche and seem to be able to induce epidermal-mesenchymal transitions, favouring tumour spreading[85].
In 2010, Lai et al[96] identified MSCs-derived Exos as one of the main active factors in MSCs-secretome[96,97] having important messenger functions. The effect of MSCs-derived Exos was first studied in inflammatory diseases such as rheumatoid arthritis[98]. It was observed that Exos secreted by MSCs, and not MSCs themselves, were effective for immunomodulation, with an immunosuppressive effect decreasing T and B-cells proliferation. The therapeutic effect of EVs secreted by MSCs has also been described in dental pulp stem cells (DPSCs)[91] where Exos derived from DPSCs undergoing osteogenic differentiation are able to interact with fibronectin and collagen type I fibres of the ECM and promote teeth regeneration[91]. Importantly, there seems to be marked differences, probably due to changes on EVs cargo, in the effect over acceptor cells between Exos secreted by naïve and differentiated cells[89]. According to Narayanan et al[99,100], Exos from MSCs growing in osteogenic differentiation media were the most effective in inducing osteogenic differentiation to other MSCs[99,100].
ApoBDs also have a role in cell-to-cell communication. These vesicles are known to carry a large cargo of molecules, mainly RNA[101,102], but also DNA and proteins[103]. Due to the size of this cargo, the impact of ApoBDs on their target cells should be important, however, there is currently little information on their function.
Although the biological effects of EVs have been intensively studied in the last few years, the role of these vesicles in vivo is still poorly understood and the major effects of the EVs may still be unknown.
BM-MSC-DERIVED EVs ON BONE REGENERATION
Although bone tissue engineering has rapidly evolved in the last few years, the application of bone regeneration techniques in some scenarios still encounters several drawbacks. The effectiveness of BM-MSCs-derived extracellular vesicles (MSC-EVs) in bone tissue regeneration has been tested in the last few years[104,105]. To better understand how MSC-EVs improve the bone healing process, first, it is necessary to outline the different stages of this process and then, how MSC-EVs could positively influence this process. The most important proteins and miRNAs in MSC-EVs cargo involved in this process and their mechanism of action are highlighted in the next section.
Role of MSC-EVs on bone healing enhancement
Bone healing occurs through a classical cascade characterized by an initial inflammatory stage followed by the repair/remodelling process. Rather than occur successively, these two phases partially overlap in time[106]. On the other hand, fracture healing can be divided into two major categories: Primary (direct or cortical) bone healing and secondary (indirect or spontaneous) bone healing. Since the latter is the most common this will be discussed here.
The three stages in fracture healing are haematoma, callus formation and bone remodelling[107]. Once fracture occurs, disruption of the vascular supply results in haematoma formation and initiation of the acute inflammatory response. This phase is essential, since immune cells attracted to the injury site are actually responsible for the initiation of the repair cascade by stimulating angiogenesis, attracting and promoting MSCs differentiation and enhancing ECM deposition[106]. At this stage, the immune response must be properly modulated. A prolonged activation or attenuation of inflammation may cause excessive bone tissue damage or necrotic bone accumulation respectively, and thus, delay fracture healing or even non-union. In 2016, Chen et al[108] showed that culturing T cells in the presence of Exos derived from MSCs isolated from healthy individuals, significantly suppresses the secretion of pro-inflammatory factors TNF-α and IL-1β, and increases the secretion of anti-inflammatory factors TGF-β during in vitro culture, suggesting that MSCs-derived Exos are in charge of maintaining the immune balance crucial for bone healing. During the second stage of bone healing, known as callus formation, cells from the innate immune system, such as macrophages, start to secrete chemotactic mediators that recruit fibroblasts, MSCs, endothelial cells/endothelial progenitors and osteoprogenitor cells from local niches (e.g. cambium layer). When the wound site becomes progressively vascularized, these cells are responsible for the formation of the callus: First, the expansion and chondrogenic differentiation of progenitor cells takes place, which leaves a cartilaginous callus. This will then be completely replaced by bone tissue through intramembranous and endochondral osteogenesis processes[107,109]. When optimal blood supply or mechanical stability is not achieved, atrophic fibrous non-union, hypertrophic fibrous non-union or pseudoarthrosis can develop[110].
Bone remodelling is the final phase of fracture healing. After a bony callus is formed, a remodelling process needs to take place to ultimately achieve healing. As the callus is composed of woven or fibrous bone, mechanically-weak bone, it has to be replaced by mechanically-strong lamellar bone. To achieve this, osteoblasts and osteoclasts coordinate bone formation and bone resorption. If the two processes involved in bone remodelling are not balanced, all biomechanical properties of normal bone will not be fully restored[106,107,110]. MSCs-EVs also have a positive role in promoting proliferation[111] and differentiation[112] of osteoblastic cells, thus, also contributing in this sense to bone regeneration.
Altogether, different studies have clearly shown that MSCs-EVs can improve every step of the bone healing process. Many efforts are now directed at identifying the molecules responsible to the aforementioned effects. Some of the information regarding the EVs cargo and its relation to bone regeneration activity is discussed in the following sections.
Cargo proteins involved in bone homeostasis
Many pieces of evidence support the role of EV proteins in bone regeneration. In 2016, the work of Furuta et al[104] reported the presence of MCP-1, MCP-3 and SDF-1 in MSC-EVs. These factors have very important functions that would aid fracture healing. MCP-1 induces angiogenesis while MCP-3 promotes cell proliferation. In addition, both MCP-1 and MCP-3 can recruit MSCs to the injury site[113]. SDF-1 seems to increase MSCs chemotaxis and thus, the recruitment of these cells to participate in bone regeneration[114]. Using a cytokine antibody array, Nakamura et al[115] confirmed the presence of IL-6, FGF-2, and PDGF-BB in MSC-Exos[115]. All these molecules are involved in endothelial cell proliferation, angiogenesis and osteogenesis[109,113,116,117]. Besides, PDGF is a key regulator of fracture healing, initiating callus formation, potentiating cartilage and intramembranous bone formation and stimulating angiogenic pathways with pro-angiogenic direct and indirect (through VEGF up-regulation) effects[106,109]. Albeit at low concentrations, all these factors were detected in MSC-derived Exos, suggesting that the positive effect of these particles on bone regeneration also relies on their protein cargo[104,115].
Although VEGF is present in the soluble part of the secretome, in 2019 Takeuchi et al[105] highlighted the presence of VEGF in MSC-EVs at similar levels to those found in the rest of the BM-MSCs secretome. VEGF acts as an effective growth factor for bone regeneration by enhancing the migration of endogenous vascular endothelial and stem cells[118]. VEGF also improves the cellular activity of osteoblasts, overall promoting bone regeneration. These functions reflect the close relationship between osteogenesis and vascularization and the important role of cell-to-cell communication between vascular endothelial cells and osteoblasts.
Proteomic analyses have provided further information on the protein contents of EVs in the last few years. In 2019, Munshi et al[119] carried out a quantitative proteomic analysis comparing the protein profile of MSC-EVs and their parental cells. This study established a list of 270 proteins enriched in the EVs by at least two-fold. In this list we can identify important proteins for bone healing such as angiopoietin-related protein 2 (ANGPTL2), fibronectin 1, IGF-I and TGFβ-1. ANGPTL2 potentiates sprouting in endothelial cells[120]. Fibronectin collaborates in cell movement and migration and plays an important role in providing provisional fibres in cartilaginous matrices. IGF-I induces collagen synthesis, reduces collagen degradation, promotes clonal expansion of chondrocytes and proliferation of pre-osteoblastic cells and regulates migration of osteoblasts and MSCs. It has been demonstrated that IGF-I sustained systemic or local infusion promotes bone formation[109,118]. On the other hand, TGFβ-1 acts as a potent chemoattractant for macrophages and enhances migration, proliferation and differentiation of osteoprogenitor cells and cellular matrix production[109,118,120,121].
Some membrane-bound proteins in MSC-EVs may also contribute to bone healing. Here, we highlight neuropilin 1 (NRP1), a glycoprotein which complexes with both VEGF receptors 1 and 2 (VEGFR-1 and VEGFR-2) and increases the binding of VEGF-A. Although the main function of NRP1 is related to neural development, vascular development involvement has been proved as well, since its interaction with several members of the VEGF family induce cell migration resulting in pro-angiogenic activity. NRP1 also regulates platelet-derived growth factor receptors (PDGFR) and NRP-1/PDGFR cross-talk is crucial for vascular remodelling[119]. A summary of the main proteins in the EVS cargo with a role in bone homeostasis is shown in Table 1.
Table 1.
The main proteins and miRNAs having an effect on bone homeostasis are shown in the table as well as their putative function in this process and the corresponding references where this activity is described
|
MSC-EVs CARGO
| ||
|
Proteins
|
Bone healing functions
|
Ref.
|
| MCP-1 | Induces angiogenesis and MSC recruitment to injury site | [107] |
| MCP-3 | Promotes cell proliferation and MSC recruitment to injury site | [107] |
| SDF-1 α | Recruits osteoprogenitor cells to wound area | [100] |
| IL-6 | Induces endothelial/endothelial progenitor cell proliferation angiogenesis and osteogenesis | [107] |
| FGF-2 | Fosters proliferation, differentiation and migration of vascular cells, chondrocytes and osteoblasts | [103,110,111] |
| PDGF-B | Promotes mesenchymal proliferation, potentiates cartilage and intramembranous bone formation and stimulates angiogenic pathways | [100,103,114] |
| VEGF | Promotes revascularization, regulation of vascular endothelial cell migration, proliferation, and capillary production, and improves the cellular activity of osteoblasts | [99] |
| ANGPTL2 | Potentiates sprouting in endothelial cells | [114] |
| Fibronectin | Collaborates in cell movement and migration and provides provisional fibers in cartilaginous matrices | [103] |
| IGF-I | Induces collagen synthesis, reduces collagen degradation, promotes clonal expansion of chondrocytes and proliferation of preosteoblastic cells, and regulates migration of osteoblasts and MSCs | [103] |
| TGF-β1 | Chemoattraction of macrophages, enhances migration, proliferation and differentiation of osteoprogenitor cells and cellular matrix production | [103,112,114] |
| NRP1 | Induces endothelial cell migration and regulates other proangiogenic actions through the VEGF family and PDGFR | [113] |
| microRNAs | Bone healing function | Ref. |
| miR-10a | Improves mesenchymal stem cell differentiation capacity of hBM-MSCs and reduces cell senescence | [118] |
| miR-10b | Increases in vitro migration of BM-MSCs | [119] |
| miR-218, miR-92a and miR-199b | Enhance osteoblast differentiation of BM-MSCs | [120,122] |
| miR-217 and miR-34 | Promote proliferation and osteoblast differentiation of BM-MSCs | [120,121] |
| miR-375, miR-216a, let-7c and miR-22 | Stimulate osteogenic differentiation of hAD-MSCs | [124,125,127] |
| miR-196a | Increases osteogenic differentiation of hAD-MSCs and osteogenic activity of osteoblasts | [106,126] |
| miR-494 | Induces endothelial cell migration | [109] |
| miR-129 and miR-136 | Promote endothelial cell proliferation | [129] |
| miR-130a, miR-135b, let-7f and miR27b | Promote vascular tube formation | [117,130,132] |
| miR-1246 | Enhances endothelial migration and tube formation | [131] |
| miR-23a and miR-424 | Cause migration, proliferation and tube formation of endothelial cells | [133,134] |
| miR-214 | Fosters prevention of cell senescence, migration and tube formation in endothelial cells, and RANKL-induced osteoclast differentiation in BMMs | [136,137] |
| miR-148a | Stimulates osteoclastogenesis in early osteoclast progenitors | [138] |
| miR-27a, miR-206, miR-29b, miR-181a and miR-302a | Induce osteogenic activity of osteoblasts | [106,139-141] |
| miR-21 | Assists osteogenic differentiation, migration and senescence prevention in BM-MSCs; migration and proliferation of fibroblasts; migration, proliferation and ability to form endothelial tubes in endothelial cells; and RANKL-induced osteoclastogenesis in BMMs | [142-146] |
MSC: Mesenchymal stem cell; EVs: Extracellular vesicles; MCP: Monocyte chemotactic protein; SDF-1 α: Stromal cell-derived factor-1 α; IL-6: Interleukin-6; FGF-2: Fibroblast growth factor-2; VEGF: Vascular endothelial growth factor; ANGPTL: Angiopoietin-related protein; IGF: Insulin-like growth factor; TGF-β1: Transforming growth factor–β1; NRP1: Neuropilin 1; hAD: Human adipose; BM: Bone marrow; BMM: Bone marrow monocytes; RANKL: Receptor activator of nuclear factor kappa ligand.
Cargo miRNAs involved in bone homeostasis
The presence of several miRNAs with bone pro-regenerative properties in MSC-EVs has been described[104,112,115,121-123]. The mechanism of action of those miRNAs will also be discussed; in order to explain why this content, at least in part, is able to develop the well-orchestrated response needed to improve bone healing[104].
MiR-10a, one of the miRNAs commonly found in the EVs cargo, is able to improve the capacity of hBM-MSCs to produce osteogenic, adipogenic and chondrogenic lineages, while simultaneously reducing cell senescence[124]. MiR-10b, on the other hand, has been shown to increase in vitro migration of BM-MSCs[125]. MiR-34a, miR-92a, miR-199b, and miR-217 enhance osteoblast differentiation of BM-MSCs. In addition, miR-217 and miR-34a also promote BM-MSCs proliferation[126,127]. MiR-218, also found in EVs cargo, is a potent activator of the ostegenic lineage leading to osteoblastic differentiation of BM-MSCs and osteogenic differentiation of osteoblasts[128]. In 2018, Shi et al[129] proved the important role of this miRNA when they showed that local administration of BM-MSCs infected with a virus expressing miR-218 clearly improved the quality and reduced the time of newly formed bone in a mouse femoral fracture model. MiR-375, miR-216a and miR-196a stimulate ostegenic differentiation of human adipose (hAD)-MSCs in vitro and two of them (miR-375 and miR-216a) can also promote bone formation in vivo. Moreover, miR-216a rescued bone formation and the osteoblastogenesis inhibitory effects of dexamethasone demonstrating its ability to prevent or treat osteoporosis and bone metabolism-related diseases[130-132]. Let-7c and miR-22 are able to unbalance hAD-MSC differentiation inhibiting adipogenic fate and promoting osteoblast formation in vitro[133]. Let-7c was also able to induce ectopic bone formation in vivo[134].
Angiogenesis is a essential step in almost every repair process, including bone formation. Some of the miRNAs found in MSC-EVs are involved in that specific function. To list a few, miR-494 induces endothelial cell migration[115], miR-129 and miR-136 promote endothelial cell proliferation[135], and let-7f and miR27b assist in sprout formation in vitro[136]. Furthermore, mir-1246 enhances endothelial migration and tube formation in vitro[137]. Mir-135b also promotes tube formation in vitro in addition to being able to induce neovascularization in vivo[138]. Two miRNAs, miR-23a and miR-424, carry out a broad set of pro-angiogenic functions, causing migration, proliferation and tube formation of endothelial cells in vitro and neovascularization in vivo[139,140]. miR-130a, is a potent inducer of angiogenesis due to the inhibitory role on two anti-angiogenic factors: Growth arrest-specific homeobox (GAX) and homeobox A5 (HOXA5). Dose-dependent improvement in all endothelial tube formation endpoints, such as total tube length and mesh index are promoted by this miRNA[123,141]. Besides, miR-214 also plays a role in preventing cell senescence, and promoting migration and tube formation in endothelial cells in vitro and blood vessel formation in vivo[142]. On the other hand, miR-214 also enhances, both in vivo and in vitro, osteoclast differentiation of bone marrow monocytes mediated by receptor activator of nuclear factor kappa ligand (RANKL)[143], which is essential for bone remodelling, the final step in bone repair.
MiR-214 is not alone in its ability to support the final stages of bone repair. miR-148a also stimulates osteoclastogenesis in early osteoclast progenitors in vitro[144]. Conversely, miR-196a, miRNA-27a, and miRNA-206 enhance osteogenic activity of osteoblasts[112,132] and miR-29b, miR-181a and miR-302a induce osteoblast differentiation in vitro[145-147].
MiR-21 deserves a special mention in this section. This microRNA appeared at the highest concentrations in MSC-EVs in the assays carried out by Furuta et al[104] and Nakamura et al[115], and several bone healing-related funcions have been attributed to mir-21 including anti-apoptotic activity and prevention of H2O2-induced cell death in vitro[148]. It has also been demonstrated that miR-21 is able to induce migration and proliferation of fibroblasts and induce migration, proliferation and the ability to form endothelial tubes in human microvascular endothelial cells[149]. These functions are accomplished throught two different pathways, which are stimulated by the active inhibition of phosphate and tensin homolog (PTEN) and sprouty homolog 1 (SPRYL). These exact same mechanisms are also responsible for osteogenic differentiation of BM-MSCs[150,151]. Specifically, Yang et al[151] proved in 2019 that miR-21 not only stimulates osteogenic differentiation and migration of BM-MSCs in vitro, but also activates the expression of angiogenic related genes (HIF-1α and VEGF). In fact, improved new bone formation in critical size defects was detected in rat and canine animal models treated with miR-21-modified BM-MSCs seeded in β-tricalcium phosphate (β-TCP) scaffolds. Furthermore, the presence of miR-21 has been established as a prerequisite for RANKL-induced osteoclastogenesis in bone marrow-derived monocytes/macrophages precursors, a crucial activity in bone repair[152]. A summary of the main miRNAs in the EVs cargo with a role in bone homeostasis is shown in Table 1.
Developing MSC secretome-based therapies to improve bone regeneration
Being aware of the beneficial properties of the content of the MSC-EVs, for the process of bone regeneration, several researchers have worked to establish different methods that would highlight MSC-EVs as a valid and successful bone therapy.
In 2020, Jia et al[153] reported that MSC-EVs from young rats could induce in vitro proliferation and osteogenic differentiation of old rat BM-MSCs in a dose-dependent manner. This study also showed that, in vivo, MSC-EVs not only accelerated bone regeneration at five weeks after distraction, but also improved the mechanical properties of the newly formed bone compared to the control group.
An important study carried out by Takeuchi et al[105], in 2019, demonstrated that treatment with MSC-EVs could up-regulate the expression of angiogenesis (VEGF, ANG1, and ANG2) and osteogenesis-related genes [collagen I (COLI), alkaline phosphatase (ALPL), osteocalcin (OCN) and osteopontin (OPN)] in human BM-MSCs in vitro, as well as induce bone and blood vessel formation in critical-sized defects in Wistar rat parietal bones, as shown by haematoxylin and eosin staining and CD31 immunostaining, respectively. Takeuchi confirmed that, in terms of angiogenesis and osteogenesis, MSC-EVs and whole MSC secretome showed similar levels of restoration, both with approximately half of the defect covered with newly formed bone after four weeks of treatment. In the same line, Furuta et al[104] showed that, in a femur fracture model of CD9 mice, lacking MSC-EVs production, there was a significantly lower bone union due to retardation of callus formation. However, this retardation was rescued by local injection of MSC-EVs. This fact clearly indicates that MSC-EVs lead to cell proliferation and prevention of cell apoptosis[107]. Interestingly, this study also revealed that MSC-EVs-depleted secretome was not able to promote fracture healing, while the use of MSC-EVs reduced the healing time compared to a wild-type control and demonstrated an improvement in the process. Together, these studies suggest that the powerful bone regenerative characteristics associated with the whole BM-MSC secretome in the past[15,113], could really be referred to these vesicles, and then, shed light on their crucial role.
In a recent study, Zhang et al[154] demonstrated in vitro that MSC-EVs were able to internalize and promote proliferation and migration of endothelial cells (HUVEC) and osteoblast precursor cells (MC3TE-E1Cs). Moreover, MSC-EVs enhanced the angiogenic tube formation ability of HUVECs and osteogenic ability of MC3TE-E1Cs. In vivo analyses in a rat model of femoral fracture showed that weekly transplantation of MSC-EVs into the injury produced a significant enhancement of angiogenesis, osteogenesis and bone healing compared to the control group. A third group, the MSC-EVs-depleted secretome, produced significantly lower effects than the MSC-EVs group.
It is worth noting an interesting study using Exos derived from human-induced pluripotent stem cell-derived mesenchymal stem cells (hiPS-MSC-Exos) that incorporates the advantages of both MSCs and iPSCs[155]. This analysis demonstrated in vitro that hiPSC-MSC-Exos could be released from β-TCP scaffolds and were internalized by hBM-MSCs, then stimulating proliferation, migration and osteogenic differentiation of these cells. These activities clearly support recruitment of hBM-MSCs to the injury site, thus, promoting new bone formation. In a very similar study, Qi et al[156] applied hiPSC-MSC-Exos together with β-TCP scaffolds to critical-size calvaria defects in osteoporotic model rats. The results revealed a significant dose-dependent increase in bone mineral density, bone volume, mineralization level, newly formed bone area, new blood vessel area and vessel number, and levels of osteogenic (OCN and OPN) and vascular markers (CD31).
Martins et al[157] demonstrated that cultivating hBM-MSCs in osteogenic medium as well as transfecting runt-related transcription factor 2 (RUNX2) gene into these cells, made them produce MSC-EVs, which potentiated the osteogenic commitment of naïve hBM-MSCs in vitro. In a similar approach, Narayanan et al[99] confirmed the results obtained by Martins in terms of induction with osteogenic medium in vitro, and proved the beneficial effects of this method in 3D cultures in vitro and in vivo, the latter even with capillaries and blood vessels development within the scaffolds, indicating neovascularization.
In 2019, Liang et al[158] preconditioned hBM-MSCs with a low dose of dimethyloxal-oylglycine (DMOG), a small molecule that stabilizes HIF-1α, thus mimicking a hypoxia microenvironment in cells. These MSC-EVs enhanced migration and tube formation of HUVECs in vitro, and bone healing and blood vessel formation in critical-sized calvarial defects in rat models. In a similar study, Li et al[159] showed that the intrinsic regenerative characteristics of MSC-EVs could be improved by adding an overexpressing HIF-1α mutation to the parental cells. This mutation considerably boosted matrix mineralization, ALP activity and osteogenesis-related genes expression in BM-MSCs and migration, proliferation and tube formation of HUVECs in vitro in a dose-dependent manner; in addition to quickening trabecula bone regeneration in necrotic femoral heads in rabbits, then promoting both bone regeneration and neovascularization in vivo.
DISCUSSION
This year marks the twenty-fifth anniversary of the publication of the first MSC test in humans[160]. Since then, activity in the field has been very intense, as indicated by the high number of registered clinical trials. Despite this, only two drugs, TEMCELL®, for the treatment of acute graft-versus-host disease, and Alofisel® (Darvadstrocel), for the treatment of fistulae in Crohn’s disease, have been approved by regulatory agencies. For various reasons, MSCs approval as a drug is proving elusive[161,162] and, from the outside, it might seem that the field is in a period of stagnation.
Nevertheless, in the last years there have been substantial changes in our understanding of the mechanisms by which MSCs assist tissue recovery. According to the new paradigm, after preventing an exaggerated immune response, MSCs release a plethora of molecules which inhibit apoptosis and scar formation, and stimulate angiogenesis and mitosis of resident stem cells[163]. This paradigm change has led many groups to shift the focus of their research to MSC secretions, and more specifically, to EVs.
The healing mechanism induced by MSCs and EVs is essentially the same, but some differences need to be taken into consideration when designing therapies. From a practical point of view, EVs are easier to handle and transport. In the treatment of acute syndromes, where pre-banked cells are the only viable cellular alternative, EVs have an additional advantage, since thawed MSCs require an adaptive period to recover their full therapeutic potential[164]. Response to intravenous administration is another of the differences between EVs and MSCs. While a high percentage of MSCs are retained in the lungs[9], the small size of the EVs allows them to overcome these barriers, and they are detectable in blood only a few minutes after administration. However, the clearance of EVs by macrophages of the endothelial reticulum system is very rapid, and EVs accumulate in macrophage-rich tissues such as liver, spleen and lungs[165,166]. Coating of EVs with polyethylene glycol (PEG) polymers might impede their clearance by macrophages. PEG polymers could also serve to bind tissue-specific ligands to the particles, allowing EVs direct to the target tissue[167,168].
A post-secretion alteration of the surface is not the only way of manipulating EVs. The pioneering work by Gnecchi et al[169] already suggested the possibility of changing EVs cargo, when they showed that conditioned medium from MSCs overexpressing Akt, limited infarct size and improved ventricular function in an in vivo model. Changes in culture conditions alter the angiogenic properties of the secretome[170]. Conditioned medium from MSCs cultured in the presence of pro-inflammatory cytokines shows more osteogenic ability[171]. Enrichment in a particular miRNA is also possible by directly transfecting EVs with a miRNA mimic[172]. There seems to be no limit to the possibility of tailoring EVs to adapt them to the particular needs of different pathologies.
One of the main concerns of therapies involving living cells is the possibility of infused cells becoming malignant. Malignancy of MSCs is a highly unlikely event, but the role of MSCs in cancer progression and metastasis is a disputed issue. Whereas some studies show proliferative effects in the progression of some cancers, others show inhibitory effects[173]. As far as EVs are concerned, results are also inconclusive. For instance, EVs from human Wharton's jelly MSCs have been shown to retard the growth of bladder cancer cells[174], and to promote renal cancer cell growth[175]. On the other hand, Peak et al[176] suggested the use of EVs as an anti-cancer therapy as they found that EVs from placental MSCs inhibit the growth of aggressive prostate cancer cells in culture[176]. However, the possibility of promoting cancer growth and metastasis remains a significant concern for EV-based therapies, especially for long-term procedures that would require repeated infusions of EVs.
Another possible advantage of EVs relates to immune tolerance. MSCs express molecules able to inhibit both innate and adaptive immune response. The initial interpretation of the absence of immune rejection of MSCs was that MSCs might have a singular immune privilege. However, recent data indicate that allogeneic MSCs can elicit an immune response which can result in rejection[177]. Like all somatic cells, MSCs constitutively express MHC class I and, upon induction, also MHC class II molecules. After accumulation within the lungs, MSCs can initially evade the immune system by a macrophage-driven, apoptosis-dependent process known as efferocytosis[178]. However, there is no guarantee that repeated cell infusions do not result in rejection[177].
EVs from MSCs are very heterogeneous in size and composition. A mass spectrometry proteome analysis of conditioned media from MSCs isolated from three tissue sources identified a total of 1533 proteins, and only 999 of them were common among samples[18]. Differences in EVs' working protocols often complicate the comparison of results. The Extracellular RNA Communication Consortium (ERCC1)[179] and the International Society for Extracellular Vesicles (ISEV)[180] have released guidelines to assist researchers in standardizing the isolation and characterization of EVs. However, the heterogeneity in size, composition, and, most importantly, biological activity can also be due to donor-to-donor variability. MSCs have limited expansion capability, and mass production of EVs is only possible by the constant renovation of cell sources. Even MSCs derived from human induced pluripotent stem cells need to be repeatedly renewed. This continuous change in cell sources compromises the reproducibility of Exo production. Several immortal cell lines, constructed by overexpressing hTERT, maintain most characteristics of primary cells, including immunosuppression[181]. Also, EVs from a MYC-immortalized cell line retain their therapeutic capabilities, despite their adipogenic potential being lost[182].
The use of EVs has several advantages over MSCs in terms of handling, safety and the possibility of standardization. Pre-clinical studies show that EVs have a therapeutic effect in fracture healing[183] and that they are also applicable in chronic pathologies such as osteoporosis[156]. Nevertheless, more studies are needed to determine if EVs can replace all the effects of MSCs. If so, EVs could be a viable alternative to MSCs in bone regenerative medicine.
CONCLUSION
MSCs produce a high number of bioactive molecules and extracellular vesicles, known as secretome, which exerts important paracrine effects on neighbouring cells and tissues. The use of MSCs secretome in tissue regeneration therapies would circumvent the problems linked to MSCs-based therapies, such as low cell survival and engraftment, which importantly limit their therapeutic efficacy, or the negative side effects associated with the administration of these cells.
Footnotes
Conflict-of-interest statement: None of the authors have any conflicts of interest relevant to this study.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Sociedad Española de Investigacion osea y del metabolismo Mineral SEIOMM; Sociedad Española de Bioquimica y Biologia Molecular.
Peer-review started: June 29, 2020
First decision: September 24, 2020
Article in press: November 11, 2020
Specialty type: Cell and tissue engineering
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Brody AR S-Editor: Fan JR L-Editor: Webster JR P-Editor: Li JH
Contributor Information
Alberto González-González, Department of Molecular Biology_IDIVAL, Faculty of Medicine, University of Cantabria, Santander 39011, Cantabria, Spain.
Daniel García-Sánchez, Department of Molecular Biology_IDIVAL, Faculty of Medicine, University of Cantabria, Santander 39011, Cantabria, Spain.
Monica Dotta, Department of Molecular Biology_IDIVAL, Faculty of Medicine, University of Cantabria, Santander 39011, Cantabria, Spain.
José C Rodríguez-Rey, Department of Molecular Biology_IDIVAL, Faculty of Medicine, University of Cantabria, Santander 39011, Cantabria, Spain.
Flor M Pérez-Campo, Department of Molecular Biology_IDIVAL, Faculty of Medicine, University of Cantabria, Santander 39011, Cantabria, Spain. f.perezcampo@unican.es.
References
- 1.Bianco P, Gehron Robey P. Marrow stromal stem cells. J Clin Invest. 2000;105:1663–1668. doi: 10.1172/JCI10413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Haque N, Kasim NH, Rahman MT. Optimization of pre-transplantation conditions to enhance the efficacy of mesenchymal stem cells. Int J Biol Sci. 2015;11:324–334. doi: 10.7150/ijbs.10567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rezaie J, Mehranjani MS, Rahbarghazi R, Shariatzadeh MA. Angiogenic and Restorative Abilities of Human Mesenchymal Stem Cells Were Reduced Following Treatment With Serum From Diabetes Mellitus Type 2 Patients. J Cell Biochem. 2018;119:524–535. doi: 10.1002/jcb.26211. [DOI] [PubMed] [Google Scholar]
- 4.García-Sánchez D, Fernández D, Rodríguez-Rey JC, Pérez-Campo FM. Enhancing survival, engraftment, and osteogenic potential of mesenchymal stem cells. World J Stem Cells. 2019;11:748–763. doi: 10.4252/wjsc.v11.i10.748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barkholt L, Flory E, Jekerle V, Lucas-Samuel S, Ahnert P, Bisset L, Büscher D, Fibbe W, Foussat A, Kwa M, Lantz O, Mačiulaitis R, Palomäki T, Schneider CK, Sensebé L, Tachdjian G, Tarte K, Tosca L, Salmikangas P. Risk of tumorigenicity in mesenchymal stromal cell-based therapies--bridging scientific observations and regulatory viewpoints. Cytotherapy. 2013;15:753–759. doi: 10.1016/j.jcyt.2013.03.005. [DOI] [PubMed] [Google Scholar]
- 6.Neri S. Genetic Stability of Mesenchymal Stromal Cells for Regenerative Medicine Applications: A Fundamental Biosafety Aspect. Int J Mol Sci. 2019;20:2406. doi: 10.3390/ijms20102406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lepperdinger G, Brunauer R, Jamnig A, Laschober G, Kassem M. Controversial issue: is it safe to employ mesenchymal stem cells in cell-based therapies? Exp Gerontol. 2008;43:1018–1023. doi: 10.1016/j.exger.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 8.Russo FP, Alison MR, Bigger BW, Amofah E, Florou A, Amin F, Bou-Gharios G, Jeffery R, Iredale JP, Forbes SJ. The bone marrow functionally contributes to liver fibrosis. Gastroenterology. 2006;130:1807–1821. doi: 10.1053/j.gastro.2006.01.036. [DOI] [PubMed] [Google Scholar]
- 9.Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue H, Savitz SI, Laine GA, Cox CS Jr. Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 2009;18:683–692. doi: 10.1089/scd.2008.0253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McLeod CM, Mauck RL. On the origin and impact of mesenchymal stem cell heterogeneity: new insights and emerging tools for single cell analysis. Eur Cell Mater. 2017;34:217–231. doi: 10.22203/eCM.v034a14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maguire G. Stem cell therapy without the cells. Commun Integr Biol. 2013;6:e26631. doi: 10.4161/cib.26631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Madrigal M, Rao KS, Riordan NH. A review of therapeutic effects of mesenchymal stem cell secretions and induction of secretory modification by different culture methods. J Transl Med. 2014;12:260. doi: 10.1186/s12967-014-0260-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Teixeira FG, Salgado AJ. Mesenchymal stem cells secretome: current trends and future challenges. Neural Regen Res. 2020;15:75–77. doi: 10.4103/1673-5374.264455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Baglio SR, Pegtel DM, Baldini N. Mesenchymal stem cell secreted vesicles provide novel opportunities in (stem) cell-free therapy. Front Physiol. 2012;3:359. doi: 10.3389/fphys.2012.00359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gunawardena TNA, Rahman MT, Abdullah BJJ, Abu Kasim NH. Conditioned media derived from mesenchymal stem cell cultures: The next generation for regenerative medicine. J Tissue Eng Regen Med. 2019;13:569–586. doi: 10.1002/term.2806. [DOI] [PubMed] [Google Scholar]
- 16.Heo JS, Choi Y, Kim HS, Kim HO. Comparison of molecular profiles of human mesenchymal stem cells derived from bone marrow, umbilical cord blood, placenta and adipose tissue. Int J Mol Med. 2016;37:115–125. doi: 10.3892/ijmm.2015.2413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baberg F, Geyh S, Waldera-Lupa D, Stefanski A, Zilkens C, Haas R, Schroeder T, Stühler K. Secretome analysis of human bone marrow derived mesenchymal stromal cells. Biochim Biophys Acta Proteins Proteom. 2019;1867:434–441. doi: 10.1016/j.bbapap.2019.01.013. [DOI] [PubMed] [Google Scholar]
- 18.Tachida Y, Sakurai H, Okutsu J, Suda K, Sugita R, Yaginuma Y, Ogura Y, Shimada K, Isono F, Kubota K, Kobayashi H. Proteomic comparison of the secreted factors of mesenchymal stem cells from bone marrow, adipose tissue and dental pulp. J Proteomics Bioinform . 2015;8:266–273. [Google Scholar]
- 19.Saeedi P, Halabian R, Imani Fooladi AA. A revealing review of mesenchymal stem cells therapy, clinical perspectives and Modification strategies. Stem Cell Investig. 2019;6:34. doi: 10.21037/sci.2019.08.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gao F, Chiu SM, Motan DA, Zhang Z, Chen L, Ji HL, Tse HF, Fu QL, Lian Q. Mesenchymal stem cells and immunomodulation: current status and future prospects. Cell Death Dis. 2016;7:e2062. doi: 10.1038/cddis.2015.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li W, Zhang Q, Wang M, Wu H, Mao F, Zhang B, Ji R, Gao S, Sun Z, Zhu W, Qian H, Chen Y, Xu W. Macrophages are involved in the protective role of human umbilical cord-derived stromal cells in renal ischemia-reperfusion injury. Stem Cell Res. 2013;10:405–416. doi: 10.1016/j.scr.2013.01.005. [DOI] [PubMed] [Google Scholar]
- 22.Gao S, Mao F, Zhang B, Zhang L, Zhang X, Wang M, Yan Y, Yang T, Zhang J, Zhu W, Qian H, Xu W. Mouse bone marrow-derived mesenchymal stem cells induce macrophage M2 polarization through the nuclear factor-κB and signal transducer and activator of transcription 3 pathways. Exp Biol Med (Maywood) 2014;239:366–375. doi: 10.1177/1535370213518169. [DOI] [PubMed] [Google Scholar]
- 23.Weiss ARR, Dahlke MH. Immunomodulation by Mesenchymal Stem Cells (MSCs): Mechanisms of Action of Living, Apoptotic, and Dead MSCs. Front Immunol. 2019;10:1191. doi: 10.3389/fimmu.2019.01191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Castro-Manrreza ME, Montesinos JJ. Immunoregulation by mesenchymal stem cells: biological aspects and clinical applications. J Immunol Res. 2015;2015:394917. doi: 10.1155/2015/394917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blazar BR, MacDonald KPA, Hill GR. Immune regulatory cell infusion for graft-versus-host disease prevention and therapy. Blood. 2018;131:2651–2660. doi: 10.1182/blood-2017-11-785865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhou Y, Yamamoto Y, Xiao Z, Ochiya T. The Immunomodulatory Functions of Mesenchymal Stromal/Stem Cells Mediated via Paracrine Activity. J Clin Med. 2019;8 doi: 10.3390/jcm8071025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Renner P, Eggenhofer E, Rosenauer A, Popp FC, Steinmann JF, Slowik P, Geissler EK, Piso P, Schlitt HJ, Dahlke MH. Mesenchymal stem cells require a sufficient, ongoing immune response to exert their immunosuppressive function. Transplant Proc. 2009;41:2607–2611. doi: 10.1016/j.transproceed.2009.06.119. [DOI] [PubMed] [Google Scholar]
- 28.Liu H, Li D, Zhang Y, Li M. Inflammation, mesenchymal stem cells and bone regeneration. Histochem Cell Biol. 2018;149:393–404. doi: 10.1007/s00418-018-1643-3. [DOI] [PubMed] [Google Scholar]
- 29.Oryan A, Kamali A, Moshiri A, Baghaban Eslaminejad M. Role of Mesenchymal Stem Cells in Bone Regenerative Medicine: What Is the Evidence? Cells Tissues Organs. 2017;204:59–83. doi: 10.1159/000469704. [DOI] [PubMed] [Google Scholar]
- 30.MacFarlane RJ, Graham SM, Davies PS, Korres N, Tsouchnica H, Heliotis M, Mantalaris A, Tsiridis E. Anti-inflammatory role and immunomodulation of mesenchymal stem cells in systemic joint diseases: potential for treatment. Expert Opin Ther Targets. 2013;17:243–254. doi: 10.1517/14728222.2013.746954. [DOI] [PubMed] [Google Scholar]
- 31.Xu G, Zhang Y, Zhang L, Ren G, Shi Y. The role of IL-6 in inhibition of lymphocyte apoptosis by mesenchymal stem cells. Biochem Biophys Res Commun. 2007;361:745–750. doi: 10.1016/j.bbrc.2007.07.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Raffaghello L, Bianchi G, Bertolotto M, Montecucco F, Busca A, Dallegri F, Ottonello L, Pistoia V. Human mesenchymal stem cells inhibit neutrophil apoptosis: a model for neutrophil preservation in the bone marrow niche. Stem Cells. 2008;26:151–162. doi: 10.1634/stemcells.2007-0416. [DOI] [PubMed] [Google Scholar]
- 33.Ge Q, Zhang H, Hou J, Wan L, Cheng W, Wang X, Dong D, Chen C, Xia J, Guo J, Chen X, Wu X. VEGF secreted by mesenchymal stem cells mediates the differentiation of endothelial progenitor cells into endothelial cells via paracrine mechanisms. Mol Med Rep. 2018;17:1667–1675. doi: 10.3892/mmr.2017.8059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tao H, Han Z, Han ZC, Li Z. Proangiogenic Features of Mesenchymal Stem Cells and Their Therapeutic Applications. Stem Cells Int. 2016;2016:1314709. doi: 10.1155/2016/1314709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cunningham CJ, Redondo-Castro E, Allan SM. The therapeutic potential of the mesenchymal stem cell secretome in ischaemic stroke. J Cereb Blood Flow Metab. 2018;38:1276–1292. doi: 10.1177/0271678X18776802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Osugi M, Katagiri W, Yoshimi R, Inukai T, Hibi H, Ueda M. Conditioned media from mesenchymal stem cells enhanced bone regeneration in rat calvarial bone defects. Tissue Eng Part A. 2012;18:1479–1489. doi: 10.1089/ten.tea.2011.0325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Oliveira-Bravo M, Sangiorgi BB, Schiavinato JL, Carvalho JL, Covas DT, Panepucci RA, Neves FA, Franco OL, Pereira RW, Saldanha-Araujo F. LL-37 boosts immunosuppressive function of placenta-derived mesenchymal stromal cells. Stem Cell Res Ther. 2016;7:189. doi: 10.1186/s13287-016-0448-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Krasnodembskaya A, Song Y, Fang X, Gupta N, Serikov V, Lee JW, Matthay MA. Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37. Stem Cells. 2010;28:2229–2238. doi: 10.1002/stem.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Redza-Dutordoir M, Averill-Bates DA. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim Biophys Acta. 2016;1863:2977–2992. doi: 10.1016/j.bbamcr.2016.09.012. [DOI] [PubMed] [Google Scholar]
- 40.Tafani M, Sansone L, Limana F, Arcangeli T, De Santis E, Polese M, Fini M, Russo MA. The Interplay of Reactive Oxygen Species, Hypoxia, Inflammation, and Sirtuins in Cancer Initiation and Progression. Oxid Med Cell Longev. 2016;2016:3907147. doi: 10.1155/2016/3907147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Idelchik MDPS, Begley U, Begley TJ, Melendez JA. Mitochondrial ROS control of cancer. Semin Cancer Biol. 2017;47:57–66. doi: 10.1016/j.semcancer.2017.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhang ZH, Zhu W, Ren HZ, Zhao X, Wang S, Ma HC, Shi XL. Mesenchymal stem cells increase expression of heme oxygenase-1 leading to anti-inflammatory activity in treatment of acute liver failure. Stem Cell Res Ther. 2017;8:70. doi: 10.1186/s13287-017-0524-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nuschke A, Rodrigues M, Stolz DB, Chu CT, Griffith L, Wells A. Human mesenchymal stem cells/multipotent stromal cells consume accumulated autophagosomes early in differentiation. Stem Cell Res Ther. 2014;5:140. doi: 10.1186/scrt530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pantovic A, Krstic A, Janjetovic K, Kocic J, Harhaji-Trajkovic L, Bugarski D, Trajkovic V. Coordinated time-dependent modulation of AMPK/Akt/mTOR signaling and autophagy controls osteogenic differentiation of human mesenchymal stem cells. Bone. 2013;52:524–531. doi: 10.1016/j.bone.2012.10.024. [DOI] [PubMed] [Google Scholar]
- 45.Gao L, Cen S, Wang P, Xie Z, Liu Z, Deng W, Su H, Wu X, Wang S, Li J, Ouyang Y, Wu Y, Shen H. Autophagy Improves the Immunosuppression of CD4+ T Cells by Mesenchymal Stem Cells Through Transforming Growth Factor-β1. Stem Cells Transl Med. 2016;5:1496–1505. doi: 10.5966/sctm.2015-0420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hao J, Li S, Shi X, Qian Z, Sun Y, Wang D, Zhou X, Qu H, Hu S, Zuo E, Zhang C, Hou L, Wang Q, Piao F. Bone marrow mesenchymal stem cells protect against n-hexane-induced neuropathy through beclin 1-independent inhibition of autophagy. Sci Rep. 2018;8:4516. doi: 10.1038/s41598-018-22857-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhang X, Kong Y, Sun Y, Qian Z, Gao C, Shi X, Li S, Piao Y, Piao F. Bone marrow mesenchymal stem cells conditioned medium protects VSC4.1 cells against 2,5-hexanedione-induced autophagy via NGF-PI3K/Akt/mTOR signaling pathway. Brain Res. 2018;1696:1–9. doi: 10.1016/j.brainres.2018.04.028. [DOI] [PubMed] [Google Scholar]
- 48.Hu C, Zhao L, Wu D, Li L. Modulating autophagy in mesenchymal stem cells effectively protects against hypoxia- or ischemia-induced injury. Stem Cell Res Ther. 2019;10:120. doi: 10.1186/s13287-019-1225-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li X, Xie X, Yu Z, Chen Y, Qu G, Yu H, Luo B, Lei Y, Li Y. Bone marrow mesenchymal stem cells-derived conditioned medium protects cardiomyocytes from hypoxia/reoxygenation-induced injury through Notch2/mTOR/autophagy signaling. J Cell Physiol. 2019;234:18906–18916. doi: 10.1002/jcp.28530. [DOI] [PubMed] [Google Scholar]
- 50.Jones S, Horwood N, Cope A, Dazzi F. The antiproliferative effect of mesenchymal stem cells is a fundamental property shared by all stromal cells. J Immunol. 2007;179:2824–2831. doi: 10.4049/jimmunol.179.5.2824. [DOI] [PubMed] [Google Scholar]
- 51.Potapova IA, Gaudette GR, Brink PR, Robinson RB, Rosen MR, Cohen IS, Doronin SV. Mesenchymal stem cells support migration, extracellular matrix invasion, proliferation, and survival of endothelial cells in vitro. Stem Cells. 2007;25:1761–1768. doi: 10.1634/stemcells.2007-0022. [DOI] [PubMed] [Google Scholar]
- 52.Salazar KD, Lankford SM, Brody AR. Mesenchymal stem cells produce Wnt isoforms and TGF-beta1 that mediate proliferation and procollagen expression by lung fibroblasts. Am J Physiol Lung Cell Mol Physiol. 2009;297:L1002–L1011. doi: 10.1152/ajplung.90347.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Arno AI, Amini-Nik S, Blit PH, Al-Shehab M, Belo C, Herer E, Tien CH, Jeschke MG. Human Wharton's jelly mesenchymal stem cells promote skin wound healing through paracrine signaling. Stem Cell Res Ther. 2014;5:28. doi: 10.1186/scrt417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kozlowska U, Krawczenko A, Futoma K, Jurek T, Rorat M, Patrzalek D, Klimczak A. Similarities and differences between mesenchymal stem/progenitor cells derived from various human tissues. World J Stem Cells. 2019;11:347–374. doi: 10.4252/wjsc.v11.i6.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ratushnyy A, Ezdakova M, Buravkova L. Secretome of Senescent Adipose-Derived Mesenchymal Stem Cells Negatively Regulates Angiogenesis. Int J Mol Sci. 2020;21 doi: 10.3390/ijms21051802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Romanov YA, Volgina NE, Vtorushina VV, Romanov AY, Dugina TN, Kabaeva NV, Sukhikh GT. Comparative Analysis of Secretome of Human Umbilical Cord- and Bone Marrow-Derived Multipotent Mesenchymal Stromal Cells. Bull Exp Biol Med. 2019;166:535–540. doi: 10.1007/s10517-019-04388-1. [DOI] [PubMed] [Google Scholar]
- 57.Gudbergsson JM. Extracellular vesicles for targeted drug delivery: triumphs and challenges. Future Med Chem. 2020;12:1285–1287. doi: 10.4155/fmc-2020-0117. [DOI] [PubMed] [Google Scholar]
- 58.Elsharkasy OM, Nordin JZ, Hagey DW, de Jong OG, Schiffelers RM, Andaloussi SE, Vader P. Extracellular vesicles as drug delivery systems: Why and how? Adv Drug Deliv Rev. 2020 doi: 10.1016/j.addr.2020.04.004. [DOI] [PubMed] [Google Scholar]
- 59.Villa F, Quarto R, Tasso R. Extracellular Vesicles as Natural, Safe and Efficient Drug Delivery Systems. Pharmaceutics. 2019;11 doi: 10.3390/pharmaceutics11110557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.CHARGAFF E, WEST R. The biological significance of the thromboplastic protein of blood. J Biol Chem. 1946;166:189–197. [PubMed] [Google Scholar]
- 61.György B, Szabó TG, Pásztói M, Pál Z, Misják P, Aradi B, László V, Pállinger E, Pap E, Kittel A, Nagy G, Falus A, Buzás EI. Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell Mol Life Sci. 2011;68:2667–2688. doi: 10.1007/s00018-011-0689-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200:373–383. doi: 10.1083/jcb.201211138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Simon B, Bolumar D, Amadoz A, Jimenez-Almazán J, Valbuena D, Vilella F, Moreno I. Identification and Characterization of Extracellular Vesicles and Its DNA Cargo Secreted During Murine Embryo Development. Genes (Basel) 2020;11 doi: 10.3390/genes11020203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zabeo D, Cvjetkovic A, Lässer C, Schorb M, Lötvall J, Höög JL. Exosomes purified from a single cell type have diverse morphology. J Extracell Vesicles. 2017;6:1329476. doi: 10.1080/20013078.2017.1329476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gould SJ, Raposo G. As we wait: coping with an imperfect nomenclature for extracellular vesicles. J Extracell Vesicles. 2013;2 doi: 10.3402/jev.v2i0.20389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Collino F, Pomatto M, Bruno S, Lindoso RS, Tapparo M, Sicheng W, Quesenberry P, Camussi G. Exosome and Microvesicle-Enriched Fractions Isolated from Mesenchymal Stem Cells by Gradient Separation Showed Different Molecular Signatures and Functions on Renal Tubular Epithelial Cells. Stem Cell Rev Rep. 2017;13:226–243. doi: 10.1007/s12015-016-9713-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lunavat TR, Cheng L, Kim DK, Bhadury J, Jang SC, Lässer C, Sharples RA, López MD, Nilsson J, Gho YS, Hill AF, Lötvall J. Small RNA deep sequencing discriminates subsets of extracellular vesicles released by melanoma cells--Evidence of unique microRNA cargos. RNA Biol. 2015;12:810–823. doi: 10.1080/15476286.2015.1056975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang X, Omar O, Vazirisani F, Thomsen P, Ekström K. Mesenchymal stem cell-derived exosomes have altered microRNA profiles and induce osteogenic differentiation depending on the stage of differentiation. PLoS One. 2018;13:e0193059. doi: 10.1371/journal.pone.0193059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Thakur BK, Zhang H, Becker A, Matei I, Huang Y, Costa-Silva B, Zheng Y, Hoshino A, Brazier H, Xiang J, Williams C, Rodriguez-Barrueco R, Silva JM, Zhang W, Hearn S, Elemento O, Paknejad N, Manova-Todorova K, Welte K, Bromberg J, Peinado H, Lyden D. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 2014;24:766–769. doi: 10.1038/cr.2014.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lázaro-Ibáñez E, Lässer C, Shelke GV, Crescitelli R, Jang SC, Cvjetkovic A, García-Rodríguez A, Lötvall J. DNA analysis of low- and high-density fractions defines heterogeneous subpopulations of small extracellular vesicles based on their DNA cargo and topology. J Extracell Vesicles. 2019;8:1656993. doi: 10.1080/20013078.2019.1656993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Vagner T, Spinelli C, Minciacchi VR, Balaj L, Zandian M, Conley A, Zijlstra A, Freeman MR, Demichelis F, De S, Posadas EM, Tanaka H, Di Vizio D. Large extracellular vesicles carry most of the tumour DNA circulating in prostate cancer patient plasma. J Extracell Vesicles. 2018;7:1505403. doi: 10.1080/20013078.2018.1505403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kowal J, Arras G, Colombo M, Jouve M, Morath JP, Primdal-Bengtson B, Dingli F, Loew D, Tkach M, Théry C. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc Natl Acad Sci USA. 2016;113:E968–E977. doi: 10.1073/pnas.1521230113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Willms E, Johansson HJ, Mäger I, Lee Y, Blomberg KE, Sadik M, Alaarg A, Smith CI, Lehtiö J, El Andaloussi S, Wood MJ, Vader P. Cells release subpopulations of exosomes with distinct molecular and biological properties. Sci Rep. 2016;6:22519. doi: 10.1038/srep22519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lindenbergh MFS, Stoorvogel W. Antigen Presentation by Extracellular Vesicles from Professional Antigen-Presenting Cells. Annu Rev Immunol. 2018;36:435–459. doi: 10.1146/annurev-immunol-041015-055700. [DOI] [PubMed] [Google Scholar]
- 75.Doyle LM, Wang MZ. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells. 2019;8:727. doi: 10.3390/cells8070727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lässer C, Jang SC, Lötvall J. Subpopulations of extracellular vesicles and their therapeutic potential. Mol Aspects Med. 2018;60:1–14. doi: 10.1016/j.mam.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 77.van Niel G, D'Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19:213–228. doi: 10.1038/nrm.2017.125. [DOI] [PubMed] [Google Scholar]
- 78.Lundell G, Holm LE, Ljunggren JG, Wasserman J. Incidence of hypothyroidism after 131I therapy for hyperthyroidism. Relation to pretherapy serum levels of T3, T4 and thyroid antibodies. Acta Radiol Oncol. 1981;20:225–230. doi: 10.3109/02841868109130200. [DOI] [PubMed] [Google Scholar]
- 79.Baixauli F, López-Otín C, Mittelbrunn M. Exosomes and autophagy: coordinated mechanisms for the maintenance of cellular fitness. Front Immunol. 2014;5:403. doi: 10.3389/fimmu.2014.00403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Desdín-Micó G, Mittelbrunn M. Role of exosomes in the protection of cellular homeostasis. Cell Adh Migr. 2017;11:127–134. doi: 10.1080/19336918.2016.1251000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Tian J, Popal MS, Zhao Y, Liu Y, Chen K, Liu Y. Interplay between Exosomes and Autophagy in Cardiovascular Diseases: Novel Promising Target for Diagnostic and Therapeutic Application. Aging Dis. 2019;10:1302–1310. doi: 10.14336/AD.2018.1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Mathieu M, Martin-Jaular L, Lavieu G, Théry C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21:9–17. doi: 10.1038/s41556-018-0250-9. [DOI] [PubMed] [Google Scholar]
- 83.Jiang L, Paone S, Caruso S, Atkin-Smith GK, Phan TK, Hulett MD, Poon IKH. Determining the contents and cell origins of apoptotic bodies by flow cytometry. Sci Rep. 2017;7:14444. doi: 10.1038/s41598-017-14305-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Shimoda M, Khokha R. Metalloproteinases in extracellular vesicles. Biochim Biophys Acta Mol Cell Res. 2017;1864:1989–2000. doi: 10.1016/j.bbamcr.2017.05.027. [DOI] [PubMed] [Google Scholar]
- 85.Nawaz M, Shah N, Zanetti BR, Maugeri M, Silvestre RN, Fatima F, Neder L, Valadi H. Extracellular Vesicles and Matrix Remodeling Enzymes: The Emerging Roles in Extracellular Matrix Remodeling, Progression of Diseases and Tissue Repair. Cells. 2018;7:167. doi: 10.3390/cells7100167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Nicholson C, Shah N, Ishii M, Annamalai B, Brandon C, Rodgers J, Nowling T, Rohrer B. Mechanisms of extracellular vesicle uptake in stressed retinal pigment epithelial cell monolayers. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165608. doi: 10.1016/j.bbadis.2019.165608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zhang Y, Jin X, Liang J, Guo Y, Sun G, Zeng X, Yin H. Extracellular vesicles derived from ODN-stimulated macrophages transfer and activate Cdc42 in recipient cells and thereby increase cellular permissiveness to EV uptake. Sci Adv. 2019;5:eaav1564. doi: 10.1126/sciadv.aav1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Costa Verdera H, Gitz-Francois JJ, Schiffelers RM, Vader P. Cellular uptake of extracellular vesicles is mediated by clathrin-independent endocytosis and macropinocytosis. J Control Release. 2017;266:100–108. doi: 10.1016/j.jconrel.2017.09.019. [DOI] [PubMed] [Google Scholar]
- 89.Huang CC, Kang M, Narayanan R, DiPietro LA, Cooper LF, Gajendrareddy P, Ravindran S. Evaluating the Endocytosis and Lineage-Specification Properties of Mesenchymal Stem Cell Derived Extracellular Vesicles for Targeted Therapeutic Applications. Front Pharmacol. 2020;11:163. doi: 10.3389/fphar.2020.00163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kovács ÁF, Láng O, Turiák L, Ács A, Kőhidai L, Fekete N, Alasztics B, Mészáros T, Buzás EI, Rigó J Jr, Pállinger É. The impact of circulating preeclampsia-associated extracellular vesicles on the migratory activity and phenotype of THP-1 monocytic cells. Sci Rep. 2018;8:5426. doi: 10.1038/s41598-018-23706-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Huang CC, Narayanan R, Alapati S, Ravindran S. Exosomes as biomimetic tools for stem cell differentiation: Applications in dental pulp tissue regeneration. Biomaterials. 2016;111:103–115. doi: 10.1016/j.biomaterials.2016.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Shah N, Ishii M, Brandon C, Ablonczy Z, Cai J, Liu Y, Chou CJ, Rohrer B. Extracellular vesicle-mediated long-range communication in stressed retinal pigment epithelial cell monolayers. Biochim Biophys Acta Mol Basis Dis. 2018;1864:2610–2622. doi: 10.1016/j.bbadis.2018.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Keshtkar S, Azarpira N, Ghahremani MH. Mesenchymal stem cell-derived extracellular vesicles: novel frontiers in regenerative medicine. Stem Cell Res Ther. 2018;9:63. doi: 10.1186/s13287-018-0791-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Rackov G, Garcia-Romero N, Esteban-Rubio S, Carrión-Navarro J, Belda-Iniesta C, Ayuso-Sacido A. Vesicle-Mediated Control of Cell Function: The Role of Extracellular Matrix and Microenvironment. Front Physiol. 2018;9:651. doi: 10.3389/fphys.2018.00651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hayashi T, Lombaert IM, Hauser BR, Patel VN, Hoffman MP. Exosomal MicroRNA Transport from Salivary Mesenchyme Regulates Epithelial Progenitor Expansion during Organogenesis. Dev Cell. 2017;40:95–103. doi: 10.1016/j.devcel.2016.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lai RC, Arslan F, Lee MM, Sze NS, Choo A, Chen TS, Salto-Tellez M, Timmers L, Lee CN, El Oakley RM, Pasterkamp G, de Kleijn DP, Lim SK. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4:214–222. doi: 10.1016/j.scr.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 97.Phan J, Kumar P, Hao D, Gao K, Farmer D, Wang A. Engineering mesenchymal stem cells to improve their exosome efficacy and yield for cell-free therapy. J Extracell Vesicles. 2018;7:1522236. doi: 10.1080/20013078.2018.1522236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Cosenza S, Toupet K, Maumus M, Luz-Crawford P, Blanc-Brude O, Jorgensen C, Noël D. Mesenchymal stem cells-derived exosomes are more immunosuppressive than microparticles in inflammatory arthritis. Theranostics. 2018;8:1399–1410. doi: 10.7150/thno.21072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Narayanan R, Huang CC, Ravindran S. Hijacking the Cellular Mail: Exosome Mediated Differentiation of Mesenchymal Stem Cells. Stem Cells Int. 2016;2016:3808674. doi: 10.1155/2016/3808674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Narayanan K, Kumar S, Padmanabhan P, Gulyas B, Wan ACA, Rajendran VM. Lineage-specific exosomes could override extracellular matrix mediated human mesenchymal stem cell differentiation. Biomaterials. 2018;182:312–322. doi: 10.1016/j.biomaterials.2018.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bayraktar R, Van Roosbroeck K, Calin GA. Cell-to-cell communication: microRNAs as hormones. Mol Oncol. 2017;11:1673–1686. doi: 10.1002/1878-0261.12144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Crescitelli R, Lässer C, Szabó TG, Kittel A, Eldh M, Dianzani I, Buzás EI, Lötvall J. Distinct RNA profiles in subpopulations of extracellular vesicles: apoptotic bodies, microvesicles and exosomes. J Extracell Vesicles. 2013;2 doi: 10.3402/jev.v2i0.20677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Blander JM. The many ways tissue phagocytes respond to dying cells. Immunol Rev. 2017;277:158–173. doi: 10.1111/imr.12537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Furuta T, Miyaki S, Ishitobi H, Ogura T, Kato Y, Kamei N, Miyado K, Higashi Y, Ochi M. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem Cells Transl Med. 2016;5:1620–1630. doi: 10.5966/sctm.2015-0285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Takeuchi R, Katagiri W, Endo S, Kobayashi T. Exosomes from conditioned media of bone marrow-derived mesenchymal stem cells promote bone regeneration by enhancing angiogenesis. PLoS One. 2019;14:e0225472. doi: 10.1371/journal.pone.0225472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Haumer A, Bourgine PE, Occhetta P, Born G, Tasso R, Martin I. Delivery of cellular factors to regulate bone healing. Adv Drug Deliv Rev. 2018;129:285–294. doi: 10.1016/j.addr.2018.01.010. [DOI] [PubMed] [Google Scholar]
- 107.Gao M, Gao W, Papadimitriou JM, Zhang C, Gao J, Zheng M. Exosomes-the enigmatic regulators of bone homeostasis. Bone Res. 2018;6:36. doi: 10.1038/s41413-018-0039-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Chen W, Huang Y, Han J, Yu L, Li Y, Lu Z, Li H, Liu Z, Shi C, Duan F, Xiao Y. Immunomodulatory effects of mesenchymal stromal cells-derived exosome. Immunol Res. 2016;64:831–840. doi: 10.1007/s12026-016-8798-6. [DOI] [PubMed] [Google Scholar]
- 109.Azari K, Doll BA, Sfeir C, Mu Y, Hollinger JO. Therapeutic potential of bone morphogenetic proteins. Expert Opin Investig Drugs. 2001;10(9):1677–1686. doi: 10.1517/13543784.10.9.1677. [DOI] [PubMed] [Google Scholar]
- 110.Marsell R, Einhorn TA. The biology of fracture healing. Injury. 2011;42:551–555. doi: 10.1016/j.injury.2011.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Zhao P, Xiao L, Peng J, Qian YQ, Huang CC. Exosomes derived from bone marrow mesenchymal stem cells improve osteoporosis through promoting osteoblast proliferation via MAPK pathway. Eur Rev Med Pharmacol Sci. 2018;22:3962–3970. doi: 10.26355/eurrev_201806_15280. [DOI] [PubMed] [Google Scholar]
- 112.Qin Y, Wang L, Gao Z, Chen G, Zhang C. Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo. Sci Rep. 2016;6:21961. doi: 10.1038/srep21961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Ando Y, Matsubara K, Ishikawa J, Fujio M, Shohara R, Hibi H, Ueda M, Yamamoto A. Stem cell-conditioned medium accelerates distraction osteogenesis through multiple regenerative mechanisms. Bone. 2014;61:82–90. doi: 10.1016/j.bone.2013.12.029. [DOI] [PubMed] [Google Scholar]
- 114.Liu X, Zhou C, Li Y, Ji Y, Xu G, Wang X, Yan J. SDF-1 promotes endochondral bone repair during fracture healing at the traumatic brain injury condition. PLoS One. 2013;8:e54077. doi: 10.1371/journal.pone.0054077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Nakamura Y, Miyaki S, Ishitobi H, Matsuyama S, Nakasa T, Kamei N, Akimoto T, Higashi Y, Ochi M. Mesenchymal-stem-cell-derived exosomes accelerate skeletal muscle regeneration. FEBS Lett. 2015;589:1257–1265. doi: 10.1016/j.febslet.2015.03.031. [DOI] [PubMed] [Google Scholar]
- 116.Mori S, Hatori N, Kawaguchi N, Hamada Y, Shih TC, Wu CY, Lam KS, Matsuura N, Yamamoto H, Takada YK, Takada Y. The integrin-binding defective FGF2 mutants potently suppress FGF2 signalling and angiogenesis. Biosci Rep. 2017;37:BSR20170173. doi: 10.1042/BSR20170173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Song R, Wang D, Zeng R, Wang J. Synergistic effects of fibroblast growth factor-2 and bone morphogenetic protein-2 on bone induction. Mol Med Rep. 2017;16:4483–4492. doi: 10.3892/mmr.2017.7183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Katagiri W, Watanabe J, Toyama N, Osugi M, Sakaguchi K, Hibi H. Clinical Study of Bone Regeneration by Conditioned Medium From Mesenchymal Stem Cells After Maxillary Sinus Floor Elevation. Implant Dent. 2017;26:607–612. doi: 10.1097/ID.0000000000000618. [DOI] [PubMed] [Google Scholar]
- 119.Munshi A, Mehic J, Creskey M, Gobin J, Gao J, Rigg E, Muradia G, Luebbert CC, Westwood C, Stalker A, Allan DS, Johnston MJW, Cyr T, Rosu-Myles M, Lavoie JR. A comprehensive proteomics profiling identifies NRP1 as a novel identity marker of human bone marrow mesenchymal stromal cell-derived small extracellular vesicles. Stem Cell Res Ther. 2019;10:401. doi: 10.1186/s13287-019-1516-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.The UniProt Consortium. UniProt: the universal protein knowledgebase. Nucleic Acids Res. 2017;45:D158–D169. doi: 10.1093/nar/gkw1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Baglio SR, Rooijers K, Koppers-Lalic D, Verweij FJ, Pérez Lanzón M, Zini N, Naaijkens B, Perut F, Niessen HW, Baldini N, Pegtel DM. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res Ther. 2015;6:127. doi: 10.1186/s13287-015-0116-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Xu JF, Yang GH, Pan XH, Zhang SJ, Zhao C, Qiu BS, Gu HF, Hong JF, Cao L, Chen Y, Xia B, Bi Q, Wang YP. Altered microRNA expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS One. 2014;9:e114627. doi: 10.1371/journal.pone.0114627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ferguson SW, Wang J, Lee CJ, Liu M, Neelamegham S, Canty JM, Nguyen J. The microRNA regulatory landscape of MSC-derived exosomes: a systems view. Sci Rep. 2018;8:1419. doi: 10.1038/s41598-018-19581-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Li J, Dong J, Zhang ZH, Zhang DC, You XY, Zhong Y, Chen MS, Liu SM. miR-10a restores human mesenchymal stem cell differentiation by repressing KLF4. J Cell Physiol. 2013;228:2324–2336. doi: 10.1002/jcp.24402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zhang F, Jing S, Ren T, Lin J. MicroRNA-10b promotes the migration of mouse bone marrow-derived mesenchymal stem cells and downregulates the expression of E-cadherin. Mol Med Rep. 2013;8:1084–1088. doi: 10.3892/mmr.2013.1615. [DOI] [PubMed] [Google Scholar]
- 126.Wang J, Liu S, Li J, Zhao S, Yi Z. Roles for miRNAs in osteogenic differentiation of bone marrow mesenchymal stem cells. Stem Cell Res Ther. 2019;10:197. doi: 10.1186/s13287-019-1309-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kang H, Chen H, Huang P, Qi J, Qian N, Deng L, Guo L. Glucocorticoids impair bone formation of bone marrow stromal stem cells by reciprocally regulating microRNA-34a-5p. Osteoporos Int. 2016;27:1493–1505. doi: 10.1007/s00198-015-3381-x. [DOI] [PubMed] [Google Scholar]
- 128.Hassan MQ, Maeda Y, Taipaleenmaki H, Zhang W, Jafferji M, Gordon JA, Li Z, Croce CM, van Wijnen AJ, Stein JL, Stein GS, Lian JB. miR-218 directs a Wnt signaling circuit to promote differentiation of osteoblasts and osteomimicry of metastatic cancer cells. J Biol Chem. 2012;287:42084–42092. doi: 10.1074/jbc.M112.377515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Shi L, Feng L, Liu Y, Duan JQ, Lin WP, Zhang JF, Li G. MicroRNA-218 Promotes Osteogenic Differentiation of Mesenchymal Stem Cells and Accelerates Bone Fracture Healing. Calcif Tissue Int. 2018;103:227–236. doi: 10.1007/s00223-018-0410-8. [DOI] [PubMed] [Google Scholar]
- 130.Chen S, Zheng Y, Zhang S, Jia L, Zhou Y. Promotion Effects of miR-375 on the Osteogenic Differentiation of Human Adipose-Derived Mesenchymal Stem Cells. Stem Cell Reports. 2017;8:773–786. doi: 10.1016/j.stemcr.2017.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Li H, Li T, Fan J, Li T, Fan L, Wang S, Weng X, Han Q, Zhao RC. miR-216a rescues dexamethasone suppression of osteogenesis, promotes osteoblast differentiation and enhances bone formation, by regulating c-Cbl-mediated PI3K/AKT pathway. Cell Death Differ. 2015;22:1935–1945. doi: 10.1038/cdd.2015.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Liao YH, Chang YH, Sung LY, Li KC, Yeh CL, Yen TC, Hwang SM, Lin KJ, Hu YC. Osteogenic differentiation of adipose-derived stem cells and calvarial defect repair using baculovirus-mediated co-expression of BMP-2 and miR-148b. Biomaterials. 2014;35:4901–4910. doi: 10.1016/j.biomaterials.2014.02.055. [DOI] [PubMed] [Google Scholar]
- 133.Huang S, Wang S, Bian C, Yang Z, Zhou H, Zeng Y, Li H, Han Q, Zhao RC. Upregulation of miR-22 promotes osteogenic differentiation and inhibits adipogenic differentiation of human adipose tissue-derived mesenchymal stem cells by repressing HDAC6 protein expression. Stem Cells Dev. 2012;21:2531–2540. doi: 10.1089/scd.2012.0014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Wei J, Li H, Wang S, Li T, Fan J, Liang X, Li J, Han Q, Zhu L, Fan L, Zhao RC. let-7 enhances osteogenesis and bone formation while repressing adipogenesis of human stromal/mesenchymal stem cells by regulating HMGA2. Stem Cells Dev. 2014;23:1452–1463. doi: 10.1089/scd.2013.0600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Qin Y, Sun R, Wu C, Wang L, Zhang C. Exosome: A Novel Approach to Stimulate Bone Regeneration through Regulation of Osteogenesis and Angiogenesis. Int J Mol Sci. 2016;17:712. doi: 10.3390/ijms17050712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kuehbacher A, Urbich C, Zeiher AM, Dimmeler S. Role of Dicer and Drosha for endothelial microRNA expression and angiogenesis. Circ Res. 2007;101:59–68. doi: 10.1161/CIRCRESAHA.107.153916. [DOI] [PubMed] [Google Scholar]
- 137.Yamada N, Tsujimura N, Kumazaki M, Shinohara H, Taniguchi K, Nakagawa Y, Naoe T, Akao Y. Colorectal cancer cell-derived microvesicles containing microRNA-1246 promote angiogenesis by activating Smad 1/5/8 signaling elicited by PML down-regulation in endothelial cells. Biochim Biophys Acta. 2014;1839:1256–1272. doi: 10.1016/j.bbagrm.2014.09.002. [DOI] [PubMed] [Google Scholar]
- 138.Umezu T, Tadokoro H, Azuma K, Yoshizawa S, Ohyashiki K, Ohyashiki JH. Exosomal miR-135b shed from hypoxic multiple myeloma cells enhances angiogenesis by targeting factor-inhibiting HIF-1. Blood. 2014;124:3748–3757. doi: 10.1182/blood-2014-05-576116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Bao L, You B, Shi S, Shan Y, Zhang Q, Yue H, Zhang J, Zhang W, Shi Y, Liu Y, Wang X, Liu D, You Y. Metastasis-associated miR-23a from nasopharyngeal carcinoma-derived exosomes mediates angiogenesis by repressing a novel target gene TSGA10. Oncogene. 2018;37:2873–2889. doi: 10.1038/s41388-018-0183-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Ghosh G, Subramanian IV, Adhikari N, Zhang X, Joshi HP, Basi D, Chandrashekhar YS, Hall JL, Roy S, Zeng Y, Ramakrishnan S. Hypoxia-induced microRNA-424 expression in human endothelial cells regulates HIF-α isoforms and promotes angiogenesis. J Clin Invest. 2010;120:4141–4154. doi: 10.1172/JCI42980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Zimta AA, Baru O, Badea M, Buduru SD, Berindan-Neagoe I. The Role of Angiogenesis and Pro-Angiogenic Exosomes in Regenerative Dentistry. Int J Mol Sci. 2019;20:406. doi: 10.3390/ijms20020406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.van Balkom BW, de Jong OG, Smits M, Brummelman J, den Ouden K, de Bree PM, van Eijndhoven MA, Pegtel DM, Stoorvogel W, Würdinger T, Verhaar MC. Endothelial cells require miR-214 to secrete exosomes that suppress senescence and induce angiogenesis in human and mouse endothelial cells. Blood. 2013;121:3997–4006, S1. doi: 10.1182/blood-2013-02-478925. [DOI] [PubMed] [Google Scholar]
- 143.Zhao C, Sun W, Zhang P, Ling S, Li Y, Zhao D, Peng J, Wang A, Li Q, Song J, Wang C, Xu X, Xu Z, Zhong G, Han B, Chang YZ, Li Y. miR-214 promotes osteoclastogenesis by targeting Pten/PI3k/Akt pathway. RNA Biol. 2015;12:343–353. doi: 10.1080/15476286.2015.1017205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Cheng P, Chen C, He HB, Hu R, Zhou HD, Xie H, Zhu W, Dai RC, Wu XP, Liao EY, Luo XH. miR-148a regulates osteoclastogenesis by targeting V-maf musculoaponeurotic fibrosarcoma oncogene homolog B. J Bone Miner Res. 2013;28:1180–1190. doi: 10.1002/jbmr.1845. [DOI] [PubMed] [Google Scholar]
- 145.Li Z, Hassan MQ, Jafferji M, Aqeilan RI, Garzon R, Croce CM, van Wijnen AJ, Stein JL, Stein GS, Lian JB. Biological functions of miR-29b contribute to positive regulation of osteoblast differentiation. J Biol Chem. 2009;284:15676–15684. doi: 10.1074/jbc.M809787200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bhushan R, Grünhagen J, Becker J, Robinson PN, Ott CE, Knaus P. miR-181a promotes osteoblastic differentiation through repression of TGF-β signaling molecules. Int J Biochem Cell Biol. 2013;45:696–705. doi: 10.1016/j.biocel.2012.12.008. [DOI] [PubMed] [Google Scholar]
- 147.Kang IH, Jeong BC, Hur SW, Choi H, Choi SH, Ryu JH, Hwang YC, Koh JT. MicroRNA-302a stimulates osteoblastic differentiation by repressing COUP-TFII expression. J Cell Physiol. 2015;230:911–921. doi: 10.1002/jcp.24822. [DOI] [PubMed] [Google Scholar]
- 148.Lv C, Hao Y, Han Y, Zhang W, Cong L, Shi Y, Tu G. Role and mechanism of microRNA-21 in H2O2-induced apoptosis in bone marrow mesenchymal stem cells. J Clin Neurosci. 2016;27:154–160. doi: 10.1016/j.jocn.2015.07.029. [DOI] [PubMed] [Google Scholar]
- 149.Hu Y, Rao SS, Wang ZX, Cao J, Tan YJ, Luo J, Li HM, Zhang WS, Chen CY, Xie H. Exosomes from human umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function. Theranostics. 2018;8:169–184. doi: 10.7150/thno.21234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Yang N, Wang G, Hu C, Shi Y, Liao L, Shi S, Cai Y, Cheng S, Wang X, Liu Y, Tang L, Ding Y, Jin Y. Tumor necrosis factor α suppresses the mesenchymal stem cell osteogenesis promoter miR-21 in estrogen deficiency-induced osteoporosis. J Bone Miner Res. 2013;28:559–573. doi: 10.1002/jbmr.1798. [DOI] [PubMed] [Google Scholar]
- 151.Yang C, Liu X, Zhao K, Zhu Y, Hu B, Zhou Y, Wang M, Wu Y, Zhang C, Xu J, Ning Y, Zou D. miRNA-21 promotes osteogenesis via the PTEN/PI3K/Akt/HIF-1α pathway and enhances bone regeneration in critical size defects. Stem Cell Res Ther. 2019;10:65. doi: 10.1186/s13287-019-1168-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Sugatani T, Vacher J, Hruska KA. A microRNA expression signature of osteoclastogenesis. Blood. 2011;117:3648–3657. doi: 10.1182/blood-2010-10-311415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Jia Y, Qiu S, Xu J, Kang Q, Chai Y. Exosomes Secreted by Young Mesenchymal Stem Cells Promote New Bone Formation During Distraction Osteogenesis in Older Rats. Calcif Tissue Int. 2020;106:509–517. doi: 10.1007/s00223-019-00656-4. [DOI] [PubMed] [Google Scholar]
- 154.Zhang L, Jiao G, Ren S, Zhang X, Li C, Wu W, Wang H, Liu H, Zhou H, Chen Y. Exosomes from bone marrow mesenchymal stem cells enhance fracture healing through the promotion of osteogenesis and angiogenesis in a rat model of nonunion. Stem Cell Res Ther. 2020;11:38. doi: 10.1186/s13287-020-1562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Zhang J, Liu X, Li H, Chen C, Hu B, Niu X, Li Q, Zhao B, Xie Z, Wang Y. Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signaling pathway. Stem Cell Res Ther. 2016;7:136. doi: 10.1186/s13287-016-0391-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Qi X, Zhang J, Yuan H, Xu Z, Li Q, Niu X, Hu B, Wang Y, Li X. Exosomes Secreted by Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Repair Critical-Sized Bone Defects through Enhanced Angiogenesis and Osteogenesis in Osteoporotic Rats. Int J Biol Sci. 2016;12:836–849. doi: 10.7150/ijbs.14809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Martins M, Ribeiro D, Martins A, Reis RL, Neves NM. Extracellular Vesicles Derived from Osteogenically Induced Human Bone Marrow Mesenchymal Stem Cells Can Modulate Lineage Commitment. Stem Cell Reports. 2016;6:284–291. doi: 10.1016/j.stemcr.2016.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Liang B, Liang JM, Ding JN, Xu J, Xu JG, Chai YM. Dimethyloxaloylglycine-stimulated human bone marrow mesenchymal stem cell-derived exosomes enhance bone regeneration through angiogenesis by targeting the AKT/mTOR pathway. Stem Cell Res Ther. 2019;10:335. doi: 10.1186/s13287-019-1410-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Li H, Liu D, Li C, Zhou S, Tian D, Xiao D, Zhang H, Gao F, Huang J. Exosomes secreted from mutant-HIF-1α-modified bone-marrow-derived mesenchymal stem cells attenuate early steroid-induced avascular necrosis of femoral head in rabbit. Cell Biol Int. 2017;41:1379–1390. doi: 10.1002/cbin.10869. [DOI] [PubMed] [Google Scholar]
- 160.Lazarus HM, Haynesworth SE, Gerson SL, Rosenthal NS, Caplan AI. Ex vivo expansion and subsequent infusion of human bone marrow-derived stromal progenitor cells (mesenchymal progenitor cells): implications for therapeutic use. Bone Marrow Transplant. 1995;16:557–564. [PubMed] [Google Scholar]
- 161.Caplan AI. Cell-Based Therapies: The Nonresponder. Stem Cells Transl Med. 2018;7:762–766. doi: 10.1002/sctm.18-0074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Galipeau J, Sensébé L. Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities. Cell Stem Cell. 2018;22:824–833. doi: 10.1016/j.stem.2018.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Caplan AI. Adult Mesenchymal Stem Cells: When, Where, and How. Stem Cells Int. 2015;2015:628767. doi: 10.1155/2015/628767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Moll G, Geißler S, Catar R, Ignatowicz L, Hoogduijn MJ, Strunk D, Bieback K, Ringdén O. Cryopreserved or Fresh Mesenchymal Stromal Cells: Only a Matter of Taste or Key to Unleash the Full Clinical Potential of MSC Therapy? Adv Exp Med Biol. 2016;951:77–98. doi: 10.1007/978-3-319-45457-3_7. [DOI] [PubMed] [Google Scholar]
- 165.Bala S, Csak T, Momen-Heravi F, Lippai D, Kodys K, Catalano D, Satishchandran A, Ambros V, Szabo G. Biodistribution and function of extracellular miRNA-155 in mice. Sci Rep. 2015;5:10721. doi: 10.1038/srep10721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Yáñez-Mó M, Siljander PR, Andreu Z, Zavec AB, Borràs FE, Buzas EI, Buzas K, Casal E, Cappello F, Carvalho J, Colás E, Cordeiro-da Silva A, Fais S, Falcon-Perez JM, Ghobrial IM, Giebel B, Gimona M, Graner M, Gursel I, Gursel M, Heegaard NH, Hendrix A, Kierulf P, Kokubun K, Kosanovic M, Kralj-Iglic V, Krämer-Albers EM, Laitinen S, Lässer C, Lener T, Ligeti E, Linē A, Lipps G, Llorente A, Lötvall J, Manček-Keber M, Marcilla A, Mittelbrunn M, Nazarenko I, Nolte-'t Hoen EN, Nyman TA, O'Driscoll L, Olivan M, Oliveira C, Pállinger É, Del Portillo HA, Reventós J, Rigau M, Rohde E, Sammar M, Sánchez-Madrid F, Santarém N, Schallmoser K, Ostenfeld MS, Stoorvogel W, Stukelj R, Van der Grein SG, Vasconcelos MH, Wauben MH, De Wever O. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015;4:27066. doi: 10.3402/jev.v4.27066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Di Rocco G, Baldari S, Toietta G. Towards Therapeutic Delivery of Extracellular Vesicles: Strategies for In Vivo Tracking and Biodistribution Analysis. Stem Cells Int. 2016;2016:5029619. doi: 10.1155/2016/5029619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Zou J, Shi M, Liu X, Jin C, Xing X, Qiu L, Tan W. Aptamer-Functionalized Exosomes: Elucidating the Cellular Uptake Mechanism and the Potential for Cancer-Targeted Chemotherapy. Anal Chem. 2019;91:2425–2430. doi: 10.1021/acs.analchem.8b05204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Gnecchi M, He H, Noiseux N, Liang OD, Zhang L, Morello F, Mu H, Melo LG, Pratt RE, Ingwall JS, Dzau VJ. Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. FASEB J. 2006;20:661–669. doi: 10.1096/fj.05-5211com. [DOI] [PubMed] [Google Scholar]
- 170.Patel DB, Gray KM, Santharam Y, Lamichhane TN, Stroka KM, Jay SM. Impact of cell culture parameters on production and vascularization bioactivity of mesenchymal stem cell-derived extracellular vesicles. Bioeng Transl Med. 2017;2:170–179. doi: 10.1002/btm2.10065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Li C, Li G, Liu M, Zhou T, Zhou H. Paracrine effect of inflammatory cytokine-activated bone marrow mesenchymal stem cells and its role in osteoblast function. J Biosci Bioeng. 2016;121:213–219. doi: 10.1016/j.jbiosc.2015.05.017. [DOI] [PubMed] [Google Scholar]
- 172.Zhang D, Lee H, Zhu Z, Minhas JK, Jin Y. Enrichment of selective miRNAs in exosomes and delivery of exosomal miRNAs in vitro and in vivo. Am J Physiol Lung Cell Mol Physiol. 2017;312:L110–L121. doi: 10.1152/ajplung.00423.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Lin W, Huang L, Li Y, Fang B, Li G, Chen L, Xu L. Mesenchymal Stem Cells and Cancer: Clinical Challenges and Opportunities. Biomed Res Int. 2019;2019:2820853. doi: 10.1155/2019/2820853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Wu S, Ju GQ, Du T, Zhu YJ, Liu GH. Microvesicles derived from human umbilical cord Wharton's jelly mesenchymal stem cells attenuate bladder tumor cell growth in vitro and in vivo. PLoS One. 2013;8:e61366. doi: 10.1371/journal.pone.0061366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Du T, Ju G, Wu S, Cheng Z, Cheng J, Zou X, Zhang G, Miao S, Liu G, Zhu Y. Microvesicles derived from human Wharton's jelly mesenchymal stem cells promote human renal cancer cell growth and aggressiveness through induction of hepatocyte growth factor. PLoS One. 2014;9:e96836. doi: 10.1371/journal.pone.0096836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Peak TC, Praharaj PP, Panigrahi GK, Doyle M, Su Y, Schlaepfer IR, Singh R, Vander Griend DJ, Alickson J, Hemal A, Atala A, Deep G. Exosomes secreted by placental stem cells selectively inhibit growth of aggressive prostate cancer cells. Biochem Biophys Res Commun. 2018;499:1004–1010. doi: 10.1016/j.bbrc.2018.04.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Ankrum JA, Ong JF, Karp JM. Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol. 2014;32:252–260. doi: 10.1038/nbt.2816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Elliott MR, Koster KM, Murphy PS. Efferocytosis Signaling in the Regulation of Macrophage Inflammatory Responses. J Immunol. 2017;198:1387–1394. doi: 10.4049/jimmunol.1601520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Das S Extracellular RNA Communication Consortium; Ansel KM; Bitzer M; Breakefield XO; Charest A; Galas DJ; Gerstein MB; Gupta M; Milosavljevic A; McManus MT; Patel T; Raffai RL; Rozowsky J; Roth ME; Saugstad JA; Van Keuren-Jensen K; Weaver AM; Laurent LC. The Extracellular RNA Communication Consortium: Establishing Foundational Knowledge and Technologies for Extracellular RNA Research. Cell. 2019;177:231–242. doi: 10.1016/j.cell.2019.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, Antoniou A, Arab T, Archer F, Atkin-Smith GK, Ayre DC, Bach JM, Bachurski D, Baharvand H, Balaj L, Baldacchino S, Bauer NN, Baxter AA, Bebawy M, Beckham C, Bedina Zavec A, Benmoussa A, Berardi AC, Bergese P, Bielska E, Blenkiron C, Bobis-Wozowicz S, Boilard E, Boireau W, Bongiovanni A, Borràs FE, Bosch S, Boulanger CM, Breakefield X, Breglio AM, Brennan MÁ, Brigstock DR, Brisson A, Broekman ML, Bromberg JF, Bryl-Górecka P, Buch S, Buck AH, Burger D, Busatto S, Buschmann D, Bussolati B, Buzás EI, Byrd JB, Camussi G, Carter DR, Caruso S, Chamley LW, Chang YT, Chen C, Chen S, Cheng L, Chin AR, Clayton A, Clerici SP, Cocks A, Cocucci E, Coffey RJ, Cordeiro-da-Silva A, Couch Y, Coumans FA, Coyle B, Crescitelli R, Criado MF, D'Souza-Schorey C, Das S, Datta Chaudhuri A, de Candia P, De Santana EF, De Wever O, Del Portillo HA, Demaret T, Deville S, Devitt A, Dhondt B, Di Vizio D, Dieterich LC, Dolo V, Dominguez Rubio AP, Dominici M, Dourado MR, Driedonks TA, Duarte FV, Duncan HM, Eichenberger RM, Ekström K, El Andaloussi S, Elie-Caille C, Erdbrügger U, Falcón-Pérez JM, Fatima F, Fish JE, Flores-Bellver M, Försönits A, Frelet-Barrand A, Fricke F, Fuhrmann G, Gabrielsson S, Gámez-Valero A, Gardiner C, Gärtner K, Gaudin R, Gho YS, Giebel B, Gilbert C, Gimona M, Giusti I, Goberdhan DC, Görgens A, Gorski SM, Greening DW, Gross JC, Gualerzi A, Gupta GN, Gustafson D, Handberg A, Haraszti RA, Harrison P, Hegyesi H, Hendrix A, Hill AF, Hochberg FH, Hoffmann KF, Holder B, Holthofer H, Hosseinkhani B, Hu G, Huang Y, Huber V, Hunt S, Ibrahim AG, Ikezu T, Inal JM, Isin M, Ivanova A, Jackson HK, Jacobsen S, Jay SM, Jayachandran M, Jenster G, Jiang L, Johnson SM, Jones JC, Jong A, Jovanovic-Talisman T, Jung S, Kalluri R, Kano SI, Kaur S, Kawamura Y, Keller ET, Khamari D, Khomyakova E, Khvorova A, Kierulf P, Kim KP, Kislinger T, Klingeborn M, Klinke DJ 2nd, Kornek M, Kosanović MM, Kovács ÁF, Krämer-Albers EM, Krasemann S, Krause M, Kurochkin IV, Kusuma GD, Kuypers S, Laitinen S, Langevin SM, Languino LR, Lannigan J, Lässer C, Laurent LC, Lavieu G, Lázaro-Ibáñez E, Le Lay S, Lee MS, Lee YXF, Lemos DS, Lenassi M, Leszczynska A, Li IT, Liao K, Libregts SF, Ligeti E, Lim R, Lim SK, Linē A, Linnemannstöns K, Llorente A, Lombard CA, Lorenowicz MJ, Lörincz ÁM, Lötvall J, Lovett J, Lowry MC, Loyer X, Lu Q, Lukomska B, Lunavat TR, Maas SL, Malhi H, Marcilla A, Mariani J, Mariscal J, Martens-Uzunova ES, Martin-Jaular L, Martinez MC, Martins VR, Mathieu M, Mathivanan S, Maugeri M, McGinnis LK, McVey MJ, Meckes DG Jr, Meehan KL, Mertens I, Minciacchi VR, Möller A, Møller Jørgensen M, Morales-Kastresana A, Morhayim J, Mullier F, Muraca M, Musante L, Mussack V, Muth DC, Myburgh KH, Najrana T, Nawaz M, Nazarenko I, Nejsum P, Neri C, Neri T, Nieuwland R, Nimrichter L, Nolan JP, Nolte-'t Hoen EN, Noren Hooten N, O'Driscoll L, O'Grady T, O'Loghlen A, Ochiya T, Olivier M, Ortiz A, Ortiz LA, Osteikoetxea X, Østergaard O, Ostrowski M, Park J, Pegtel DM, Peinado H, Perut F, Pfaffl MW, Phinney DG, Pieters BC, Pink RC, Pisetsky DS, Pogge von Strandmann E, Polakovicova I, Poon IK, Powell BH, Prada I, Pulliam L, Quesenberry P, Radeghieri A, Raffai RL, Raimondo S, Rak J, Ramirez MI, Raposo G, Rayyan MS, Regev-Rudzki N, Ricklefs FL, Robbins PD, Roberts DD, Rodrigues SC, Rohde E, Rome S, Rouschop KM, Rughetti A, Russell AE, Saá P, Sahoo S, Salas-Huenuleo E, Sánchez C, Saugstad JA, Saul MJ, Schiffelers RM, Schneider R, Schøyen TH, Scott A, Shahaj E, Sharma S, Shatnyeva O, Shekari F, Shelke GV, Shetty AK, Shiba K, Siljander PR, Silva AM, Skowronek A, Snyder OL 2nd, Soares RP, Sódar BW, Soekmadji C, Sotillo J, Stahl PD, Stoorvogel W, Stott SL, Strasser EF, Swift S, Tahara H, Tewari M, Timms K, Tiwari S, Tixeira R, Tkach M, Toh WS, Tomasini R, Torrecilhas AC, Tosar JP, Toxavidis V, Urbanelli L, Vader P, van Balkom BW, van der Grein SG, Van Deun J, van Herwijnen MJ, Van Keuren-Jensen K, van Niel G, van Royen ME, van Wijnen AJ, Vasconcelos MH, Vechetti IJ Jr, Veit TD, Vella LJ, Velot É, Verweij FJ, Vestad B, Viñas JL, Visnovitz T, Vukman KV, Wahlgren J, Watson DC, Wauben MH, Weaver A, Webber JP, Weber V, Wehman AM, Weiss DJ, Welsh JA, Wendt S, Wheelock AM, Wiener Z, Witte L, Wolfram J, Xagorari A, Xander P, Xu J, Yan X, Yáñez-Mó M, Yin H, Yuana Y, Zappulli V, Zarubova J, Žėkas V, Zhang JY, Zhao Z, Zheng L, Zheutlin AR, Zickler AM, Zimmermann P, Zivkovic AM, Zocco D, Zuba-Surma EK. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750. doi: 10.1080/20013078.2018.1535750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Wolbank S, Stadler G, Peterbauer A, Gillich A, Karbiener M, Streubel B, Wieser M, Katinger H, van Griensven M, Redl H, Gabriel C, Grillari J, Grillari-Voglauer R. Telomerase immortalized human amnion- and adipose-derived mesenchymal stem cells: maintenance of differentiation and immunomodulatory characteristics. Tissue Eng Part A. 2009;15:1843–1854. doi: 10.1089/ten.tea.2008.0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Chen TS, Arslan F, Yin Y, Tan SS, Lai RC, Choo AB, Padmanabhan J, Lee CN, de Kleijn DP, Lim SK. Enabling a robust scalable manufacturing process for therapeutic exosomes through oncogenic immortalization of human ESC-derived MSCs. J Transl Med. 2011;9:47. doi: 10.1186/1479-5876-9-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Zhang Y, Hao Z, Wang P, Xia Y, Wu J, Xia D, Fang S, Xu S. Exosomes from human umbilical cord mesenchymal stem cells enhance fracture healing through HIF-1α-mediated promotion of angiogenesis in a rat model of stabilized fracture. Cell Prolif. 2019;52:e12570. doi: 10.1111/cpr.12570. [DOI] [PMC free article] [PubMed] [Google Scholar]