Abstract
Transforming growth factor-β (TGFβ) family members are structurally and functionally related cytokines that have diverse effects on the regulation of cell fate during embryonic development and in the maintenance of adult tissue homeostasis. Dysregulation of TGFβ family signaling can lead to a plethora of developmental disorders and diseases, including cancer, immune dysfunction, and fibrosis. In this review, we focus on TGFβ, a well-characterized family member that has a dichotomous role in cancer progression, acting in early stages as a tumor suppressor and in late stages as a tumor promoter. The functions of TGFβ are not limited to the regulation of proliferation, differentiation, apoptosis, epithelial–mesenchymal transition, and metastasis of cancer cells. Recent reports have related TGFβ to effects on cells that are present in the tumor microenvironment through the stimulation of extracellular matrix deposition, promotion of angiogenesis, and suppression of the anti-tumor immune reaction. The pro-oncogenic roles of TGFβ have attracted considerable attention because their intervention provides a therapeutic approach for cancer patients. However, the critical function of TGFβ in maintaining tissue homeostasis makes targeting TGFβ a challenge. Here, we review the pleiotropic functions of TGFβ in cancer initiation and progression, summarize the recent clinical advancements regarding TGFβ signaling interventions for cancer treatment, and discuss the remaining challenges and opportunities related to targeting this pathway. We provide a perspective on synergistic therapies that combine anti-TGFβ therapy with cytotoxic chemotherapy, targeted therapy, radiotherapy, or immunotherapy.
Subject terms: Cell biology, Molecular medicine
Introduction
Transforming growth factor-β (TGFβ) belongs to a family of multi-functional cytokines that includes TGFβs, bone morphogenetic proteins, activins, nodal, growth and differentiation factors, inhibins, lefty, and anti-Mullerian hormone. Members of this family are key regulators of embryonic development, tissue homeostasis, and regeneration, and their malfunction has been implicated in cancer, fibrosis, immune diseases, and many other pathologies.1–4 In this review, we focus on TGFβ, which is the prototypical member of the family. There are three highly structurally related mammalian TGFβ isoforms, that is, TGFβ1, TGFβ2, and TGFβ3.5,6 In vitro studies showed that all three isoforms elicit similar biological effects, with differences in potency in certain cell types.7 Mouse studies in which each specific isoform was knocked out revealed strikingly different phenotypes, indicating nonredundant in vivo functions for these three TGFβ isoforms. TGFβ1-deficient mice either die of vascular defects during embryogenesis or autoimmune disease postnatally.8,9 Mice deficient in TGFβ2 have defects in cardiac septation and valve remodeling,10 and mice lacking TGFβ3 have pulmonary defects and cleft palate.11,12 TGFβ1 is the most abundant and studied isoform, and it is particularly highly enriched in platelets and bone.13,14
TGFβ signaling has attracted the interest of cancer biologists because of its numerous roles in regulating cancer cell functions, including cell cycle progression, apoptosis, adhesion, and differentiation.3,15 The biphasic functions of TGFβ during cancer progression on tumor cells and other cells in the tumor microenvironment (TME) are summarized in Fig. 1. In different cell types and/or conditions, TGFβ can have different, even opposite, effects.16 In normal and premalignant cells, TGFβ predominantly acts as a tumor suppressor by inhibiting cell proliferation, promoting apoptosis, and maintaining genome stability.17 However, tumor cells can adapt to or selectively bypass the suppressive functions of TGFβ; they utilize TGFβ’s promotional roles to obtain a growth advantage and undergo processes, such as the epithelial-to-mesenchymal transition (EMT), that enable their migration, invasion, intravasation, and extravasation.18,19 Moreover, TGFβ can create a beneficial TME by acting in a paracrine manner to activate cancer-associated fibroblasts (CAFs), promote angiogenesis, produce extracellular matrix (ECM), and suppress anti-tumor immune reaction to trigger cancer progression and promote metastasis.3,4,20
Fig. 1.
Biphasic functions of TGFβ during tumor progression. TGFβ acts as a tumor suppressor in the initial stage of tumor progression by inducing cell cycle arrest and apoptosis of normal and pre-malignant epithelial cells. Upon activation of oncogenes and/or inactivation of tumor suppressor genes, tumor cells become insensitive to the TGFβ-induced cytostatic effects and undergo uncontrolled proliferation. TGFβ produced by tumor cells, fibroblasts, immune, and endothelial cells in the tumor microenvironment (TME) can trigger cancer cells to undergo an epithelial-to-mesenchymal transition (EMT). Thereby, late-stage cancer cells acquire the ability to escape from the primary niche, intravasate into the circulation, extravasate and localize to distant sites, and progress to form secondary tumors. Reciprocal TGFβ signaling between cancer cells and the TME contributes to cancer progression by activating cancer-associated fibroblasts (CAFs), stimulating angiogenesis, promoting protumor cytokine secretion, increasing extracellular matrix deposition, and evading an immune attack. In the metastatic sites, the mesenchymal tumor cells can undergo a mesenchymal-to-epithelial transition. Thus, thereby change back into an epithelial phenotype, which enables rapid outgrowth
Many preclinical results from in vitro cell models and in vivo animal models have shown the great potential of anti-tumor therapeutics with TGFβ-neutralizing antibodies and ligand traps that block the interaction of TGFβ with its receptors or selective small-molecule TGFβ receptor kinase inhibitors.21,22 However, rendering these pharmacological agents suitable for approved clinical use continues to be a challenge. Because TGFβ shows highly pleiotropic actions, in addition to the tumor itself, healthy tissues can be affected by the systemic inhibition of TGFβ, which can lead to unwanted side effects and safety concerns.23 Therefore, we need to better understand the underlying molecular mechanisms by which TGFβ signaling controls normal and malignant processes. Moreover, insight into stratifying tumor patients using biomarkers for selecting patients that may benefit from TGFβ targeting is urgently needed.24,25 In this review, we discuss recent clinical advancements and bottlenecks in anti-TGFβ cancer treatment, and provide perspectives of combined treatment to overcome chemo/targeted/radio-therapy resistance and increase the efficiency of immunotherapy responses in cancer patients.
Bioavailability and activation of TGFβ ligands
TGFβ is synthesized as a large precursor protein in the rough endoplasmic reticulum, it consists of a signal peptide, a large N-terminal pro-segment termed latency-associated peptide (LAP), and a short C-terminal mature peptide.26,27 The pro-peptide assembles into a homodimer in which the LAP portions link via two disulfide bonds and the mature TGFβ moieties’ interaction is stabilized through a disulfide bond.28 After cleavage of the precursor protein by the convertase furin in the Golgi complex, the LAP portions encircle the mature portions to form the small latent complex (SLC) and shield the mature TGFβ from binding to its receptors (Fig. 2).29,30
Fig. 2.
A schematic representation of the activation of latent TGFβ. The pro-TGFβ precursor is synthesized in the rough endoplasmic reticulum. It consists of an N-terminal signal peptide, latency-associated peptide (LAP), and a mature C-terminal TGFβ fragment. After cleavage by the convertase furin in the Golgi complex, the LAP dimer binds to mature TGFβ noncovalently, preventing its binding to cell surface receptors, and forms the small latent complex (SLC). There are three major mechanisms for activation of latent TGFβ. a Proteases (e.g., cathepsin, plasmin, matrix metalloproteinase 9/14 (MMP9/14)) in the extracellular matrix (ECM) cleave LAP and release active TGFβ. Also, thrombospondin (TSP) can induce activation by direct binding to LAP. b SLC is anchored to the ECM proteins (e.g., fibronectin and fibrillin) via latent TGFβ-binding protein (LTBP) and forms the so-called large latent complex (LLC). Active TGFβ can be released by cell contraction upon the interaction between LAP and integrins. c SLC binds to glycoprotein A repetition predominant protein (GARP) on the cell surface and can also mediate the release of active TGFβ upon interaction with integrins
For the activation of latent TGFβ, the mature TGFβ portions need to dissociate from the LAP portions. TGFβ can be activated in vitro upon heating or treating with a mild acid (pH 4.5) to dissociate the LAP portion from the mature protein.31,32 In addition, reactive oxygen species can activate TGFβ in vitro by destroying the ability of LAP to bind to mature TGFβ via oxidative modification.33–35 Several ECM serine proteases (plasmin/plasma kallikrein/cathepsin D) and matrix metalloprotease 9/14 (MMP9/14) can release active TGFβ via the proteolytic cleavage of LAP in vivo (Fig. 2a).3,31,36 Thrombospondin (TSP) is another activator of latent TGFβ that is present in the ECM; upon direct binding to LAP, TSP can induce the release of active TGFβ.37 In addition, the latent TGFβ binding protein (LTBP) can covalently bind to LAP, which facilitates the deposition of the SLC in the ECM and forms the large latent complex (LLC).38,39 LLC can form a covalent interaction with specific ECM proteins such as fibrillin and fibronectin via the N-terminal domain of LTBP (Fig. 2b).39,40 Moreover, the latent TGFβ can also interact with the transmembrane glycoprotein A repetition predominant protein (GARP), which is expressed on the cell surface of regulatory T (Treg) cells, platelets, and endothelial cells to facilitate latent TGFβ activation (Fig. 2c).41 Finally, integrins, which are cell adhesion receptors that control cell proliferation, survival migration, and invasion, also have a pivotal role in the activation of latent TGFβ.42,43 Integrins comprise α and β heterodimeric subunits that are both type I transmembrane receptors and are expressed in a wide range of cells.42 Certain integrins have been demonstrated to interact with the Arg-Gly-Asp (RGD) motif of LAP and lead to the mechanical release of LAP by cellular contractions.44–46
Functions of TGFβ signaling pathways during tumor progression
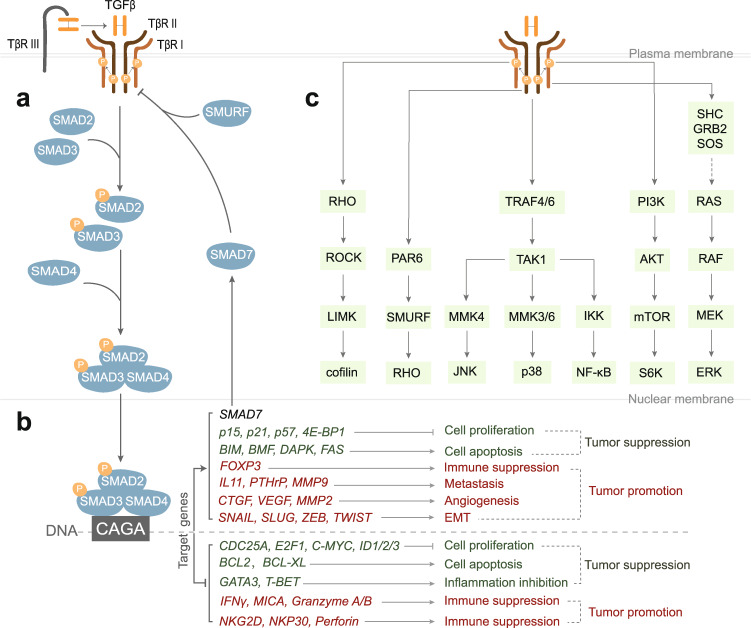
Regarding the canonical TGFβ pathway, active TGFβ initially binds to the low-affinity accessory TGFβ type III receptor (TβRIII), also named betaglycan, which is abundant on the surface of many cell types (Fig. 3a). Betaglycan can present TGFβ to the TGFβ receptor complex, which is present at low levels but high affinity, thereby facilitating signaling.47 The TGFβ2 isoform is, in particular, dependent on TβRIII for signaling. Consistent with this notion, endothelial cells that do not express TβRIII show weak responsiveness to TGFβ2 compared with that of TGFβ1 and TGFβ3.7 The TGFβ receptors complex is a tetramer consisting of two paired transmembrane serine/threonine protein kinases: two TβRIs and two TβRIIs.48–50 TGFβ binding triggers TβRII to transphosphorylate TβRI at specific serine/threonine residues that are located in the intracellular juxtamembrane region enriched with glycine and serine residues (the GS domain).51,52 After the extracellular signal is successfully transduced across the plasma membrane, activated TβRI initiates intracellular signaling by phosphorylating SMAD2/3 at their two extreme C-terminal serine residues.53,54 Thereafter, phosphorylated SMAD2/3 dissociates promptly from TβRI and assemble into a heteromeric complex with SMAD4. Subsequently, the SMAD2/3–SMAD4 complex can translocate into the nucleus to activate or repress the expression of target genes.55,56 SMAD7 is a target gene induced by TGFβ that encodes for a negative regulator of the TGFβ/SMAD signaling pathway by associating with activated TβRI, thereby blocking the interaction, phosphorylation, and activation of SMAD2. Subsequent steps in SMAD signaling, including SMAD2–SMAD4 complex formation and its translocation to the nucleus are also inhibited by SMAD7.57–59 SMAD7 can also antagonize TGFβ signaling in the nucleus by disrupting the formation of the functional SMAD–DNA complex.60,61 Moreover, SMAD7 recruits E3 ubiquitin ligases such as SMAD ubiquitination regulatory factor 1/2 (SMURF1/2), WW domain-containing protein 1 (WWP1), and neural precursor cell-expressed developmentally downregulated 4-2 (NEDD4-2) to TβRI, and thereby promotes its ubiquitination-mediated proteasomal and/or lysosomal degradation.62–64 Naturally, the ubiquitination of TβRI can be reversed by a set of deubiquitinating enzymes, including ubiquitin-specific protease 4 (USP4), USP11, USP15, and ubiquitin carboxyl-terminal hydrolase L1.65–68
Fig. 3.
Schematic of the TGFβ-induced canonical SMAD and noncanonical signaling pathways. a TβRIII presents TGFβ to TβRII. Thereafter, ligand occupied TβRII recruits and phosphorylates TβRI to trigger intracellular TGFβ signaling pathways. In the canonical pathway, activated TβRI phosphorylates SMAD2/3 and stimulates the formation of heteromeric complexes with SMAD4. These complexes are translocated into the nucleus and regulate target gene expression. One of the TGFβ/SMAD-induced target genes is SMAD7, of which the gene product participates in a negative feedback loop to regulate the duration and intensity of TGFβ signaling by recruiting E3 ubiquitin ligase SMURF to TβRI. b TGFβ pathway target genes relevant for tumor suppression are listed in green, and the target genes that encode proteins involved in the tumor promotion are listed in red. c TGFβ can also activate many noncanonical pathways, including RHO, JNK, p38, NF-κB, AKT, and ERK signaling components
The target genes of the canonical TGFβ pathway that play key roles in regulating tumor progression are summarized in Fig. 3b.69 In the early stage of tumorigenesis, TGFβ promotes the expression of cyclin-dependent kinase inhibitors, including p15, p21, p57, and eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1), to induce cell cycle arrest.70–74 TGFβ represses the expression of genes encoding several growth-promoting factors, such as cell division cycle 25a (CDC25A), E2F transcription factor 1 (E2F1) and C-MYC proto-oncogene, to induce an anti-mitogenic cellular response.75–79 Inhibitor of DNA binding 1 (ID1) protein functions as a cell differentiation inhibitor and stimulator of cell proliferation, and long-term stimulation of cells by TGFβ can silence the ID1 promoter and thereby drive epithelial cells to enter into a cytostatic program.80 ID1 was also shown to mediate the escape of pancreatic cancer from TGFβ tumor suppression.81 TGFβ can increase the expression of B-cell lymphoma 2 (BCL2)-interacting mediator of cell death (BIM), BCL2-modifying factor (BMF), FAS and death-associated protein kinase (DAPK) genes82–85 and decrease the expression of BCL2 and BCL-extra large (BCL-XL) genes to induce cell apoptosis.86,87 In addition, TGFβ can suppress inflammation in the early stage of tumorigenesis by downregulating the expression of GATA-binding protein 3 (GATA3) and transcription factor T-box expressed in T cells (T-BET) genes to inhibit T cell differentiation.88,89 In the malignant stage, tumor cells can become non-responsive to the TGFβ-induced cytostatic and proapoptotic effects when genes that encode key components of the TGFβ pathway are mutated, and this results in the functional inactivation of their corresponding gene products. For example, TβRII mutations are frequently found in colorectal cancer (CRC),90 SMAD2 mutations have been reported in CRC, hepatocellular carcinoma (HCC), and lung cancer,91,92 and the deletion or mutation of SMAD4 is often identified in pancreatic cancer, CRC, and HCC.93,94 There are several advanced tumors that still retain an intact canonical TGFβ pathway, such as glioma, melanoma, and breast cancer. These cancer cells can avert TGFβ-induced cytostatic functions through the acquisition of activating mutations in oncogenes, for example, rat sarcoma (RAS) and MYC, and inactivating mutations in tumor suppressor genes, for example, p53 and retinoblastoma protein (Rb). TGFβ can increase forkhead box P3 (FOXP3) gene expression,95 and decrease interferon-γ (IFNγ), MHC class I-chain-related molecules A (MICA), granzyme A/B, natural killer group 2 member D (NKG2D), natural cytotoxicity receptor 3 (NKP30), and Perforin gene expression to suppress immune function.96–99 Besides, TGFβ upregulates interleukin-11 (IL11), parathyroid hormone-related peptide (PTHrP), and MMP9 to facilitate metastasis to specific organs.100–102 Moreover, TGFβ can promote angiogenesis by upregulating connective tissue growth factor (CTGF), vascular endothelial growth factor (VEGF), and MMP2.103,104
In addition to the canonical SMAD pathway, TGFβ can also initiate multiple noncanonical signaling pathways downstream of TGFβ receptors (Fig. 3c).105 For example, TβRI activates RHO small GTPases and then regulates the activity of RHO-associated protein kinase and LIM kinase to phosphorylate cofilin, leading to actin cytoskeleton reorganization that regulates cell adhesion, motility, and growth.106 TβRII can directly phosphorylate the cell polarity regulator PAR6 that regulates tight junctions and cell migration.107 TGFβ also induces the activation of TGFβ-activated kinase 1 (TAK1) to stimulate c-Jun NH2-terminal kinase (JNK) and p38 mitogen-activated protein kinase (MAPK) and nuclear factor κ-light-chain enhancer of activated B cells (NF-κB) pathways. These responses are mediated by tumor necrosis factor-associated factor 4 (TRAF4) and TRAF6 that interact with TGFβ receptors.108–110 Serine/threonine kinase protein kinase B (AKT) signaling can also be activated by TGFβ in a phosphatidylinositol 3-kinase (PI3K)-dependent manner.111 Besides, TGFβ can induce the phosphorylation of Src homology domain 2-containing protein (SHC) and then recruit growth factor receptor-binding protein 2 (GRB2) and son of sevenless (SOS) to activate the extracellular signal-regulated kinase (ERK) pathway through the RAS, RAF, and mitogen-activated protein kinase (MEK) pathways.112 A recent study discovered that the RAS-responsive element-binding protein 1 (RREB1) provides a molecular link between RAS and TGFβ pathways for coordinated induction of a developmental and fibrogenic EMT pathway.113 These non-SMAD signaling pathways are not unique to TGFβ; some are tightly regulated by receptor tyrosine kinases (RTKs) and crosstalk with the canonical SMAD signaling pathway.114 Moreover, TGFβ can also indirectly activate these pathways by inducing the expression of secreted growth factors, for example, platelet-derived growth factor (PDGF), which then acts in an autocrine or paracrine manner via specific receptors endowed with intrinsic tyrosine kinase activity.115–117
Furthermore, TGFβ plays an important role in inducing the metastatic capacity of tumor cells by promoting the EMT through the upregulation of transcription factors such as SNAIL, SLUG, TWIST, and ZEB1/2 through either SMAD- or non-SMAD-dependent pathways.118–124 Accompanying the increase of TGFβ levels, EMT leads epithelial tumor cells to lose the capability of adhesion, polarity, and tight junctions by decreasing levels of tight-junction proteins zona occludence-1 (ZO-1), E-cadherin, and occludin, and acquire a highly migratory and invasive mesenchymal phenotype by the increased levels of fibronectin, vimentin, and N-cadherin (Fig. 4a).19 EMT has diverse phenotypic manifestations with intermediate epithelial/mesenchymal states and is a reversible process. This has been referred to as epithelial cell plasticity (EMP).125 TGFβ can induce different states of EMP and the cellular response to TGFβ is context-dependent; inflammatory factors, Wnt, Notch, Hippo, and Hedgehog interplay with TGFβ to orchestrate the EMP response.126 In Fig. 4b, we show that TGFβ promotes the EMT in non-transformed mouse mammary gland (NMuMG) epithelial cells.127 The NMuMG cell line is a frequently used model system to investigate TGFβ-induced EMT.128,129 The ability of TGFβ to promote the migration of the human mesenchymal triple-negative breast cancer (TNBC) MDA-MB-231 cells is shown in Fig. 4c.130 MDA-MB-231 is highly aggressive and its metastasis to bone occurs in a TGFβ/SMAD-dependent manner.101,119,131
Fig. 4.
The functions of TGFβ upon the epithelial–mesenchymal transition (EMT). a Schematic of TGFβ mediation of the EMT process. b TGFβ promotes the EMT in non-transformed NAMRU mouse mammary gland (NMuMG) epithelial cells, as visualized by immunofluorescent staining of cells with anti-E-cadherin antibody (red) and phalloidin (green) to measure filamentous actin expression in the absence and after treatment with 5 ng/ml TGFβ3 for 48 h. The typical morphological change from epithelial- to fibroblast-like cells, decreased E-cadherin, and accumulated striated fibers are observed in the NMuMG cells upon TGFβ stimulation. c TGFβ promotes the migration of MDA-MB-231 human breast cancer cells, as determined via real-time imaging of a wound-healing scratch assay. Left, graph showing the time-lapse relative migration rate; right, images of the cells taken when the initial scratch was made (0 h) and after 24 and 48 h in the absence or presence of 5 ng/ml TGFβ3
Functions of TGFβ in the TME
The TME consists of ECM, cytokines, and a large population of different cell types, including resident and infiltrating CAFs, immune-related cells, endothelial cells, and adipocytes that surround the tumor cells. The multipronged effects of TGFβ on tumor stroma cells built the TME, including its capacity to stimulate ECM production, activate CAFs, suppress the immune system, and promote angiogenesis. Notably, tumor stromal cells are also the main sources of inflammatory factors, including TGFβ. Reciprocally, the TME exerts profound effects on tumor growth and progression. In this section, we summarize the functions of TGFβ in the TME (Fig. 5).
Fig. 5.
Functions of TGFβ in the tumor microenvironment (TME). a TGFβ can activate/differentiate resident fibroblasts, mesenchymal stem cells, epithelial tumor cells, adipose tissue-derived stem cells, and endothelial cells into cancer-associated fibroblasts (CAFs) in the TME. b TGFβ promotes angiogenesis in the TME by acting directly and indirectly on endothelial cells stimulating their proliferation, migration, and sprouting. c TGFβ crucially suppresses the immune system by regulating the functions of immune cell populations in the TME. The specific actions of TGFβ are indicated in the boxes next to the different immune cells that are depicted
TGFβ and CAFs in the TME
CAFs are key components of the TME and play roles in providing a favorable environment to support tumor progression by producing ECM and cytokines, stimulating immune evasion, and promoting angiogenesis.132–134 Next to tumor-promoting CAFs, also tumor-suppressive CAFs have been identified.135 Within a tumor, the CAF population can be highly heterogeneous. For example in breast tumors, at least four distinct CAF subpopulations, that is, vascular CAFs, matrix CAFs, cycling CAFs, and developmental CAFs (dCAFs), were defined.136 In pancreatic tumors, CAFs have been classified into inflammatory CAFs, which are found distant from tumor foci with high IL6 and low α-smooth muscle actin (α-SMA) expression, and myofibroblastic CAFs that are located adjacent to tumor foci with high α-SMA expression and that demonstrate a strong TGFβ responsiveness.137–139 The characterization of molecular markers and mechanistic insights of different CAF subtypes may provide opportunities for their precise targeting in cancer therapy.140
During the formation of CAFs, TGFβ is a major driver in activating resident quiescent fibroblasts, and differentiating bone marrow-derived mesenchymal stem cells and adipose tissue-derived stem cells into CAFs (Fig. 5a).141–144 The activation of the TGFβ signaling pathway can also induce epithelial cancer cells into myofibroblasts via EMT, and differentiate endothelial cells into fibroblast-like cells by endothelial-mesenchymal transition (EndMT) (Fig. 5a).145,146 In turn, CAFs autocrine TGFβ can serve as an attractant to recruit more fibroblasts into the TME around the tumor invasion front and promote metastasis.147,148 CAFs surround and throughout the tumor often restrict the accessibility of anti-cancer drugs to tumor cells since the dense CAFs with stiff surrounding matrix can reduce the density of blood vessels and form a physical barrier surrounding the tumor cells.149 In hypoxic TME, CAF-secreted TGFβ2 can cooperate with hypoxia-inducible factor to increase stemness phenotype and induce robust chemotherapy of cancer cells.150 Radiation-induced TGFβ signaling can activate CAFs in the TME, which enhances the invasiveness of the associated tumor cells via promoting EMT.151 One recent study in which TβRII-mutant pancreatic cancer was investigated, found that the anti-tumor efficacy of TβRII blockade is due to the inhibition of the TGFβ signaling in CAFs.152 Furthermore, an increasing number of studies reported that the overactive TGFβ signaling in CAFs is a major reason of immunotherapy failure.153 Analysis of the single-cell landscape of CAFs in pancreatic cancer identified a TGFβ-driven CAF population expressing leucine-rich repeat-containing 15 (LRRC15).154 This transmembrane protein is associated with poor outcome in immunotherapy trial data.154 It will be interesting to explore if the targeting of LRRC15+ CAF in the TME will boost the response of cancer patients towards immune checkpoint blockade therapy.154
TGFβ and endothelial cells in the TME
Endothelial cells are located at the surface of blood and lymphatic vessels. Blood vessels nourish tumors by delivering blood/oxygen/nutrients, removing waste products, and mediating the entry and exit of immune cells and other substances.155–158 A positive correlation between the TGFβ level and microvessel density was observed in various types of tumors.159 Endothelial cells express two TβRIs (activin receptor-like kinase 1 (ALK1) and TβRI/ALK5). The TGFβ–ALK5 signaling axis can directly mediate inhibition of endothelial cell proliferation and migration (Fig. 5b).160,161 Indirectly, the expression of some key angiogenic factors, such as VEGF, CTGF, and fibroblast growth factor 2 (FGF2), can be induced by the TGFβ–ALK5 signaling axis.162–165 In the presence of endoglin, the TGFβ-ALK1 signaling axis can directly promote endothelial cell proliferation and migration.160,166 Moreover, the JNK pathway also mediates the proangiogenic response of TGFβ in endothelial cells.167 A recent study showed that TGFβ can promote VEGF-C production in tubular epithelial cells, macrophages, and mesothelial cells to induce lymphangiogenesis in renal and peritoneal fibrosis. However, the function of TGFβ in the formation of the cancer-associated lymphatic system is not well understood.168
TGFβ and immune-related cells in the TME
There are a variety of innate and adaptive immune cells dispersed throughout the TME (Fig. 5c). During tumorigenesis, myeloid cells (including myeloid-derived suppressor cells (MDSCs), macrophages, neutrophils) typically accumulate in the early stage of tumor outgrowth to suppress the T cell response and sustain an immunosuppressive environment.169 Dendritic cells (DCs) deliver tumor antigens to T cells and natural killer (NK) cells that exert antitumor cytotoxic effects.170 However, antitumor immune reactions often become suppressed during tumor development; TGFβ can exhibit pivotal immunosuppressive effects on the intrinsic antitumor potential of DCs and NK cells in the TME (Fig. 5c). At an early stage of cancer, TGFβ mitigates myeloid proliferation and differentiation by repressing the expression of cytokine IFNγ.171,172 Progressively, myeloid cells in advanced-stage cancer produce TGFβ and MMPs that further inhibit antitumor immune reactions and promote tumor metastasis.173–175 Indeed, experimentally induced inactivation of TGFβ signaling in myeloid cells can lead to an increase in antitumor activity.175,176 TGFβ blocks the activation of NK cells and suppresses their cytotoxic potential by inhibiting C-type lectin receptor NKG2D expression directly and indirectly.177 IL1 receptor-associated kinase M (IRAK-M) is a potent negative regulator of Toll-like receptor (TLR) signaling and is predominantly expressed in macrophages.178 By inducing IRAK-M expression and antagonizing TLR signaling, TGFβ protects tumors from the potential TLR-mediated antitumor activities of macrophages.179
TGFβ suppresses adaptive immunity during cancer progression mainly by inhibiting the activation, proliferation, differentiation, and migration of T cells. TGFβ can suppress the differentiation of naive CD4+ helper T cells into distinct effector subtypes. However, it induces the conversion of naive T cells into Treg cells (previously known as suppressor T cells) that suppress the immune response.180 A recent study showed that depletion of TβRII in CD4+ T cells suppresses cancer progression as a result of tissue healing and remodeling of the blood vasculature, causing cancer hypoxia and death in distant avascular regions.181 TGFβ can block the activation and maturation of cytotoxic CD8+ T cells by repressing the tumor antigen processing and presentation of DCs and inhibit CD8+ T cell proliferation through suppressing the expression of IFNγ and IL2.172,182–184 TGFβ promotes antigen-induced programmed cell death protein-1 (PD-1) expression in CD8+ T cells, which causes T cell exhaustion.185 Recently, researchers found that the TGFβ signal maintains the immune-suppressive identity of CD8+ Treg cells. Transcription factor Eomesodermin (Eomes) controls the follicular location of CD8+ Treg cells.186 Both TGFβ and Eomes coordinate to promote the homeostasis of CD8+ Treg cells.186 TGFβ can regulate the activation, proliferation, apoptosis of B cells, and stimulate the antibody switching in B cells. However, its function on B cell-mediated antitumor immunity is not well investigated.187–189
Pharmacological interventions of TGFβ in cancer therapy
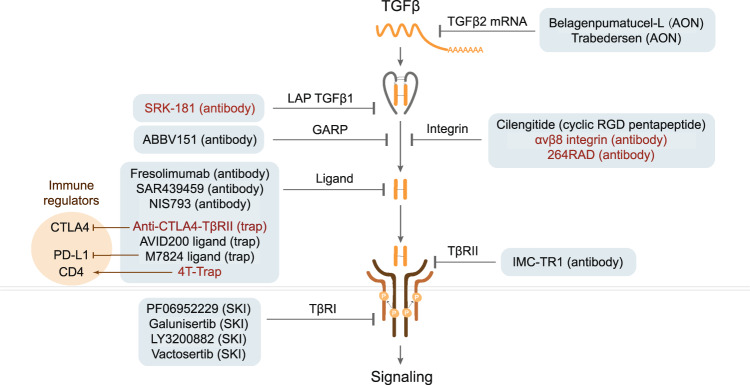
There are numerous anti-cancer pharmacological interventions that target specific mediators of TGFβ signaling pathway or TGFβ activators, which have been tested in human clinical trials or that displayed very promising results in pre-clinical animal models (Fig. 6). In this section, we introduce the recent advancements and bottlenecks of the main anti-TGFβ strategies, including neutralizing antibodies, ligand traps, small-molecule kinase inhibitors, and antisense oligonucleotides (AONs), and summarize the pharmacological interventions that have been or are currently being studied in clinical trials (Table 1).
Fig. 6.
Schematic of strategies utilized in (pre)clinical trials targeting TGFβ signal transduction for cancer therapy. Various pharmacological interventions are grouped into the targeting of different TGFβ signaling components, that is, TGFβ mRNA, GARP/integrins that are involved in activation of latent TGFβ, and ligands that interact with TGFβ receptors and TβRI kinase activity. The promising new targeting molecules that have been studied in pre-clinical models are highlighted in red color. Different strategies for targeting TGFβ signaling, including antisense oligonucleotide (AON), neutralizing antibody (antibody), cyclic RGD pentapeptide, TGFβ ligand trap (trap), and small-molecule kinase inhibitor (SKI) are indicated. The immune regulatory targets (CTLA4/PD-L1/CD4) of the bispecific molecules that sequester TGFβ with a TβRII extracellular domain containing trap are highlighted in the orange circle
Table 1.
Summary of pharmacological strategies targeting TGFβ for cancer therapy in clinical trials (information was obtained from https://www.clinicaltrials.gov/)
| Strategy | Drug | Target | Cancer | Identifier | Phase | Treatment | Status |
|---|---|---|---|---|---|---|---|
| Neutralizing antibody | Fresolimumab | TGFβ1/2/3 | Glioma | NCT01472731 | 2 | Monotherapy | Completed |
| Metastatic breast cancer | NCT01401062 | 2 | Combination with radiotherapy | Completed | |||
| Relapsed malignant pleural mesothelioma | NCT01112293 | 2 | Monotherapy | Completed | |||
| Renal cell carcinoma or malignant melanoma | NCT00356460 | 1 | Monotherapy | Completed | |||
| Early stage non-small cell lung cancer | NCT02581787 | 2 | Combination with radiotherapy | Recruiting | |||
| SAR439459 | TGFβ1/2/3 | Advanced solid tumors | NCT03192345 | 1 | Monotherapy/combination with cemiplimab | Recruiting | |
| NIS793 | TGFβ1/2/3 | Metastatic pancreatic ductal adenocarcinoma | NCT04390763 | 2 | Monotherapy/combination with spartalizumab/gemcitabine and paclitaxel | Active | |
| Breast/lung/hepatocellular/colorectal/pancreatic/renal cancer | NCT02947165 | 1 | Monotherapy/combination with spartalizumab | Recruiting | |||
| IMC-TR1 | TβRII | Advanced solid tumor | NCT01646203 | 1 | Monotherapy | Completed | |
| ABBV151 | GARP:TGFβ1 | Advanced/metastatic solid tumors | NCT03821935 | 1 | Monotherapy/combination with ABBV-181 | Recruiting | |
| Ligand trap | AVID200 | TGFβ1/3 | Advanced/metastatic solid tumors | NCT03834662 | 1 | Monotherapy | Active |
| M7824 | TGFβ/PD-L1 | Advanced non-small cell lung cancer | NCT03631706 | 3 | Monotherapy | Recruiting | |
| HPV-associated cancers | NCT04432597 | 2 | Combination with HPV vaccine PRGN-2009 | Recruiting | |||
| Cholangiocarcinoma/gallbladder cancer | NCT03833661 | 2 | Monotherapy | Active | |||
| Recurrent respiratory papillomatosis | NCT03707587 | 2 | Monotherapy | Active | |||
| Biliary tract cancer | NCT04066491 | 3 | Combination with gemcitabine and cisplatin | Recruiting | |||
| Relapsed small cell lung cancers | NCT03554473 | 2 | Combination with topotecan/temozolomide | Recruiting | |||
| Metastatic triple-negative breast cancer | NCT03579472 | 1 | Combination with eribulin mesylate | Recruiting | |||
| Thymoma/thymic carcinoma | NCT04417660 | 2 | Monotherapy | Active | |||
| Stage II–III HER2-positive breast cancer | NCT03620201 | 1 | Monotherapy | Recruiting | |||
| Advanced Kaposi sarcoma | NCT04303117 | 2 | Combination with M9241 | Recruiting | |||
| Advanced adenocarcinoma of the pancreas | NCT03451773 | 2 | Combination with gemcitabine | Completed | |||
| Metastatic colorectal cancer/advanced solid tumors | NCT03436563 | 2 | Monotherapy | Recruiting | |||
| Advanced pancreas cancer | NCT04327986 | 2 | Combination with M9241 and radiotherapy | Active | |||
| Metastatic non-prostate genitourinary malignancies | NCT04235777 | 1 | Combination with M9241/radiotherapy | Recruiting | |||
| Resectable head and neck squamous cell carcinoma | NCT04247282 | 2 | Monotherapy/combination with N-803 and TriAd Vaccine | Recruiting | |||
| Metastatic/locally advanced urothelial cancer | NCT04349280 | 1 | Monotherapy | Active | |||
| HER2-negative breast cancer | NCT03524170 | 1 | Combination with radiotherapy | Recruiting | |||
| Advanced-stage breast cancer | NCT04296942 | 1 | Combination with ado-trastuzumab emtansine, entinostat, and BN-brachyury vaccine | Recruiting | |||
| AT-hook 2 (HMGA2) expressing triple-negative breast cancer | NCT04489940 | 2 | Monotherapy | Active | |||
| Platinum-experienced cervical cancer | NCT04246489 | 2 | Monotherapy | Recruiting | |||
| Recurrent/second primary head and neck squamous cell cancer | NCT04220775 | 2 | Combination with radiotherapy | Recruiting | |||
| Advanced small bowel/colorectal cancers | NCT04491955 | 2 | Combination with N-803, M9241, and CV301 vaccine | Active | |||
| Small-molecule inhibitors | Galunisertib | TβRI | Advanced hepatocellular carcinoma | NCT02906397 | 1 | Combination with radiotherapy | Active |
| Metastatic castration-resistant prostate cancer | NCT02452008 | 2 | Combination enzalutamide | Recruiting | |||
| Rectal cancer | NCT02688712 | 2 | Combination fluorouracil/capecitabine and radiotherapy | Recruiting | |||
| Metastatic breast cancer | NCT02538471 | 2 | Combination with radiotherapy | Terminated | |||
| Metastatic pancreatic cancer | NCT02734160 | 1 | Combination with durvalumab | Terminated | |||
| Advanced refractory solid tumors | NCT02423343 | 1 | Combination with nivolumab | Active | |||
| Recurrent/refractory non-small cell lung cancer | NCT02423343 | 2 | Combination with nivolumab | Active | |||
| Hepatocellular carcinoma | NCT01246986 | 2 | Combination with sorafenib/ramucirumab | Active | |||
| Metastatic androgen receptor-negative triple-negative breast cancer | NCT02672475 | 1 | Combination with Paclitaxel | Recruiting | |||
| Newly diagnosed malignant glioma | NCT01220271 | 2 | Combination with temozolomide and radiotherapy | Completed | |||
| Carcinosarcoma of the uterus or ovary | NCT03206177 | 1 | Combination with paclitaxel/carboplatin | Recruiting | |||
| Advanced/metastatic unresectable pancreatic cancer | NCT01373164 | 2 | Combination gemcitabine/placebo | Completed | |||
| Vactosertib | TβRI | Advanced solid tumors | NCT02160106 | 1 | Monotherapy | Completed | |
| Refractory multiple myeloma | NCT03143985 | 1 | Combination with pomalidomide | Recruiting | |||
| Urothelial carcinoma | NCT04064190 | 2 | Combination with durvalumab | Active | |||
| Metastatic colorectal cancer | NCT03724851 | 1 | Combination with pembrolizumab | Recruiting | |||
| Advanced non-small cell lung cancer | NCT03732274 | 2 | Combination with durvalumab | Recruiting | |||
| Myeloproliferative neoplasm | NCT04103645 | 2 | Monotherapy | Active | |||
| Advanced desmoid tumor | NCT03802084 | 2 | Combination with imatinib | Recruiting | |||
| Metastatic gastric cancer | NCT03698825 | 2 | Combination with paclitaxel | Recruiting | |||
| Metastatic pancreatic ductal adenocarcinoma | NCT04258072 | 1 | Combination with irinotecan, fluorouracil, and leucovorin | Active | |||
| LY3200882 | TβRI | Solid tumors | NCT02937272 | 1 | Monotherapy | Active | |
| Activated colorectal cancer | NCT04031872 | 2 | Combination with capecitabine | Active | |||
| PF06952229 | TβRI | Advanced solid tumors | NCT03685591 | 1 | Monotherapy | Recruiting | |
| Cilengitide | αvβ3/5 | Diffuse intrinsic pontine glioma | NCT01165333 | 1 | Combination with radiotherapy | Completed | |
| Advanced non-small cell lung cancer | NCT00842712 | 2 | Combination with cetuximab and platinum-based chemotherapy | Completed | |||
| Brain metastases from lung cancer | NCT00884598 | 1 | Combination with radiotherapy | Completed | |||
| Advanced solid tumors/glioblastoma multiforme | NCT01122888 | 1 | Combination with sunitinib | Terminated | |||
| Metastatic prostate cancer | NCT00103337 | 2 | Monotherapy | Completed | |||
| Recurrent glioblastoma multiforme | NCT00093964 | 2 | Monotherapy | Completed | |||
| Diagnosed inoperable glioblastoma | NCT01558687 | 1 | Monotherapy | Terminated | |||
| Advanced solid tumors/lymphoma | NCT00077155 | 1 | Monotherapy | Completed | |||
| Childhood recurrent/progressive high-grade glioma | NCT00679354 | 2 | Monotherapy | Completed | |||
| Newly diagnosed glioblastoma | NCT00689221 | 3 | Combination temozolomide and radiotherapy | Completed | |||
| Childhood refractory primary brain tumors | NCT00063973 | 1 | Monotherapy | Completed | |||
| Prostate cancer | NCT00121238 | 2 | Monotherapy | Completed | |||
| Unresectable/metastatic melanoma | NCT00082875 | 2 | Monotherapy | Terminated | |||
| Acute myeloid leukemia | NCT00089388 | 2 | Monotherapy | Terminated | |||
| Advanced solid tumors | NCT00022113 | 1 | Monotherapy | Completed | |||
| Undergoing surgery for recurrent/progressive glioblastoma multiforme | NCT00112866 | 2 | Monotherapy | Terminated | |||
| HIV-related Kaposi’s sarcoma | NCT00006222 | 1 | Monotherapy | Terminated | |||
| Locally advanced/metastatic cancer | NCT00004258 | 1 | Monotherapy | Completed | |||
| Progressive/recurrent glioma | NCT00006093 | 2 | Monotherapy | Completed | |||
| Relapsed/refractory high-grade gliomas/diffuse intrinsic pontine gliomas | NCT01517776 | 2 | Combination with temozolomide and radiotherapy | Terminated | |||
| Recurrent/metastatic squamous cell carcinoma of the head and neck | NCT00705016 | 2 | Combination with cetuximab, cisplatin, and fluorouracil | Completed | |||
| Triple-negative breast cancer/advanced solid tumors that cannot be removed by surgery | NCT01276496 | 1 | Combination with paclitaxel | Completed | |||
| Newly diagnosed MGMT-promoter unmethylated glioblastoma | NCT01044225 | 3 | Combination with radiotherapy and temozolomide | Terminated | |||
| Antisense oligonucleotide | Trabedersen | TGFβ2 | Advanced tumors known to overproduce TGFβ2 | NCT00844064 | 1 | Monotherapy | Completed |
| Glioblastoma and anaplastic astrocytoma | NCT00761280 | 3 | Monotherapy | Terminated | |||
| Recurrent/refractory high-grade glioma | NCT00431561 | 2 | Monotherapy | Completed | |||
| Lucanix | TGFβ2 | Non-small cell lung cancer | NCT01058785 | 2 | Monotherapy | Completed | |
| Non-small cell lung cancer | NCT00676507 | 3 | Monotherapy | Completed |
Neutralizing antibodies
Neutralizing antibodies can be selectively directed against critical domains of native ligands and extracellular domains of receptors to block their biological activity. Fresolimumab (GC1008), a human IgG4κ monoclonal antibody that neutralizes TGFβ1, 2, and 3, demonstrated acceptable safety and antitumor activity in a phase 1 clinical trial for patients with malignant melanoma or renal carcinoma.190 Phase 2 clinical trials of this antibody have been conducted for patients with glioma, metastatic breast cancer, or relapsed malignant pleural mesothelioma, which showed good toleration.191–193 The phase 2 clinical evaluation of this antibody for the treatment of early-stage non-small cell lung cancer (NSCLC) is still ongoing (NCT02581787). LY3022859 is an anti-TβRII IgG1 monoclonal antibody blocking TGFβ binding to the ectodomain of TβRII that showed significant antitumor function against primary tumor growth and metastasis in several mice tumor models.194 When tested for use in the treatment of advanced solid tumors in a phase 1 clinical trial, the maximum-tolerated dose for this antibody was not determined since the patients suffered from uncontrolled cytokine release despite prophylaxis.195
Integrins are major activators of TGFβ ligand, including αvβ1, αvβ3, αvβ5, αvβ6, and αvβ8.46,196–199 An increase number of studies showed that integrin-induced TGFβ signaling pathway modulates the tumor stroma and in particular the immune microenvironment.200 Thus, besides their role to promote cell proliferation, survival, and migration, the ability of αv integrins to stimulate TGFβ activation contributes to cancer progression.201,202 Thus, integrins can be pharmacological targets for cancer treatment, and their targeting may enhance the efficacy of immune therapies.203 For example, previous research found that αvβ8 integrin-expressing tumor cells can evade host immunity by upregulating TGFβ signaling in immune cells.204 The engineered antibody against αvβ8 integrin can block the release of active TGFβ by cancer cells and promote the anti-tumor immune reaction in pre-clinical mice models of colon carcinoma and lung cancer.205 A recent study showed that αvβ6-neutralizing antibody 264RAD can suppress TGFβ signaling and significantly reduce tumor growth in αvβ6-positive human pancreatic ductal adenocarcinoma (PDAC) xenografts mice model and immunocompetent transgenic mice bearing αvβ6-positive PDAC tumors.206 Although most studies showed that anti-αvβ integrin therapy has promise in cancer treatment, an opposite finding was reported for antibody-mediated inhibition of αvβ6 that while leading to a strong decreased TGFβ signaling, it promoted pancreatic cancer progression.207
Ligand traps
TGFβ ligand traps are TGFβ receptor ectodomain-based chimeric fusion proteins that are rationally designed to prevent TGFβs from binding to their receptors. AVID200 is a selective trap of TGFβ1 and TGFβ3 that showed the capacity to enhance the anti-tumor efficacy by inhibiting the growth of syngeneic 4T1 TNBC homograft in immunocompetent host mice.187 A phase I clinical trial of AVID200 revealed that it was safe and well tolerated for patients with advanced solid tumors.208,209
Small-molecule inhibitors
Small-molecule kinase inhibitors of TβR kinases are undergoing intensively clinical trials for anti-TGFβ signaling in cancer treatment. Galunisertib (LY2157299) is an orally available small-molecule inhibitor that selectively binds to TβRI (and weakly to TβRII) and inhibits its kinase activity. It showed favorable preclinical results for antitumor growth, for example, in Calu6 and EMT6-LM2 mouse tumor models.210 In phase 1 clinical trials, this inhibitor exhibited acceptable safety and dose tolerability for patients with HCC/metastatic pancreatic cancer/malignant glioma/advanced solid tumors.211–214 Phase 1/2 clinical trials of this inhibitor for patients with metastatic prostate cancer/TNBC/advanced HCC/rectal cancer/recurrent glioblastoma are ongoing.213,215,216 A phase 2 study of this inhibitor for patients with advanced HCC showed acceptable safety and prolonged overall survival.215 However, in a phase 2 study of patients with recurrent glioblastoma, this inhibitor failed to improve the overall survival.217 Vactosertib (TEW-7197) is another orally bioavailable kinase inhibitor of TβRI that revealed promising antitumor function in a mouse model of myeloma.218 This inhibitor displayed a favorable safety profile and antitumor efficacy in phase 1 clinical trials for patients with advanced solid tumors or desmoid tumors.219,220 Phase 1 clinical trial investigation of this inhibitor for patients with refractory multiple myeloma/metastatic CRC/metastatic PDAC and phase 2 study for patients with metastatic gastric cancer/CRC/urothelial carcinoma/ NSCLC/myeloproliferative neoplasm/advanced desmoid tumors are ongoing.221–224 LY3200882 is a next-generation highly selective potent ATP-competitive TβRI inhibitor that showed antitumor efficacy in a preclinical mouse model of TNBC cancer.225 The phase 1 clinical trial results showed that LY3200882 had a tolerable safety profile and early signs of antitumor efficacy for patients with advanced or metastatic cancers (NCT04031872). PF06952229 is a selective and orally available TβRI inhibitor that is being studied in phase 1 clinical trials for patients with advanced/metastatic breast cancer/castration-resistant prostate cancer (NCT03685591).
Previous studies showed that genetically and pharmacologically inhibition of αvβ integrin can inhibit TGFβ signaling and suppress metastasis.201,203 For example, the cyclic RGD pentapeptide cilengitide (EMD121974) is a potent and selective inhibitor of integrin αvβ3/5 that showed a reduction of TGFβ1 and TGFβ2 messenger RNA (mRNA) and protein expression, SMAD2 phosphorylation, and TGFβ-mediated reporter gene activity in most glioma cells.226 The intracranial LN-308 glioma xenograft mice model also displayed decreased SMAD2 phosphorylation in response to cilengitide.226 This inhibitor has been evaluated in phase 1 clinical trials for patients with brain tumors/lung cancer/advanced solid tumor/lymphoma/HIV-related Kaposi’s sarcoma;227–230 in phase 2 clinical trials for patients with NSCLC/prostate cancer/metastatic melanoma/acute myeloid leukemia/head neck squamous cell carcinoma;231–237 and in phase 3 for patients with glioblastoma.238 The clinical trials of cilengitide were terminated for the treatment of patients with metastatic melanoma, acute myeloid leukemia, or HIV-related Kaposi’s sarcoma since it neither exhibited antitumor efficacy nor improved overall survival. However, the phase 3 clinical study of cilengitide in glioblastoma patients showed well toleration and single-agent activity.237,238
Antisense oligonucleotides
AONs are short oligonucleotides designed to suppress the expression of specific genes by blocking their translation.239 Trabedersen (AP12009) is a phosphonothioate antisense oligodeoxynucleotide that specifically targets TGFβ2 mRNA.240 Trabedersen was evaluated in the phase 1 clinical studies in patients with melanoma/pancreatic cancer/CRC, and showed good safety and encouraging survival results.241 The phase 2 clinical trials’ evaluation of this AON for patients with glioblastoma or anaplastic astrocytoma enabled the determination of the optimal dose for further clinical development.242 However, the phase 3 clinical trial of trabedersen for patients with glioblastoma was terminated because of the lack of patient recruitment.243–245 Lucanix (belagenpumatucel-L) is a vaccine expressing AON TGFβ2 RNA that was evaluated in clinical trial studies for NSCLC patients.246,247 This vaccine did not meet its survival endpoint in a phase 3 clinical trial for NSCLC patients; however, it improved the overall survival of patients who had completed chemotherapy within the previous 12 weeks.248
Targeting TGFβ in cancer therapy: challenges and opportunities
Overcoming the adverse effects of anti-TGFβ therapies
Cancer patients who were treated with blockers of TGFβ signaling can experience side effects if the function of TGFβ in physiological processes is compromised. For example, some TβRI kinase inhibitors showed therapeutic effects in cancer patients, but their cardiac toxicity at high doses (hemorrhagic, degenerative, and inflammatory lesions in heart valves) and skin toxicity (eruptive keratoacanthomas, hyperkeratosis, cutaneous squamous-cell carcinomas, and basal cell carcinoma) limits their safe therapy window.22 These adverse effects have (and continue to) challenge the clinical application of many other anti-TGFβ therapies. Careful dosing of TGFβ inhibitors to cancer patients may attenuate toxicity. Moreover, the adverse effects may be mitigated by pulsatile therapy, in which the patient has so-called “drug holidays” during the dosing period. Galunisertib (LY2157299) has been applied as a therapy regimen of 2 weeks on and 2 weeks off drug treatment to reduce the (cardiac) side effects.249 In addition, predictive biomarkers may aid in selecting patients who benefit most from treatment with anti-TGFβ agents. Transcriptional profiling of samples from patients with many different cancer types, including glioblastoma, pancreatic cancer, breast cancer, ovarian cancer, CRC, and NSCLC, revealed that cancer patients with mesenchymal subtypes have high expression of TGFβ target genes and that this correlates with poor prognosis. Therefore, subgroups of patients with cancers that carry a mesenchymal phenotype may particularly benefit from anti-TGFβ therapies.250–255
Perspectives on the synergy of combination therapies
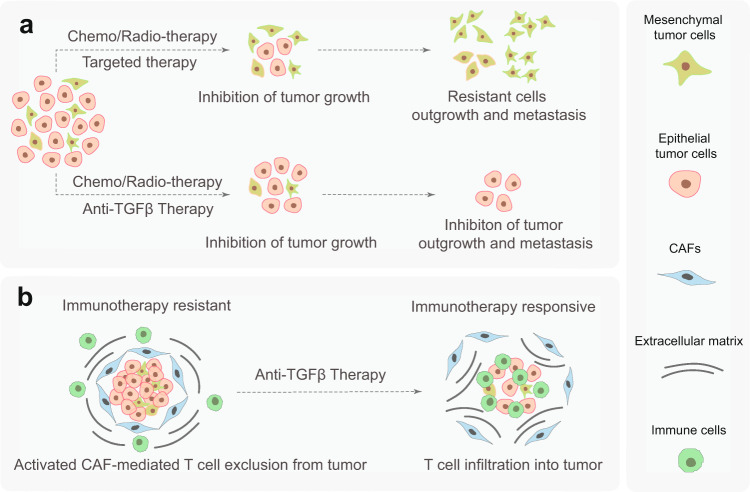
Increased TGFβ activity has been implicated in the resistance to various anticancer therapies, including cytotoxic chemotherapy, targeted therapy, radiotherapy, and immunotherapy.4 Therefore, combining anti-TGFβ therapy with these established strategies may dampen therapy resistance (Fig. 7). A future opportunity and challenge are to identify the optimal combination of a synergistic therapy regimen for each individual patient.
Fig. 7.
Sketch of synergistic combination therapies. a Chemo/radio/targeted therapy alone inhibits the growth of epithelial-like tumor cells, and in combination with anti-TGFβ therapy, invasive escape and resistance to these therapies are attenuated, and metastasis of mesenchymal tumor cells is restrained. b Activated CAFs mediated by high TGFβ activity suppresses immunotherapy efficacy by blocking T cell infiltration into tumors and inducing T cell dysfunction. In combination with anti-TGFβ therapy; however, T cell exclusion is inhibited, and the antitumor efficacy of the immunotherapy is improved
Anti-TGFβ therapy with chemotherapy, targeted therapy, or radiotherapy
Chemotherapy is a frequent standard first-line cancer therapy regimen consisting of one or more anticancer chemicals designed to stop and kill rapidly proliferating cancer cells. However, chemotherapy has strong side effects due to its toxicity to normal cells. Targeted therapy is performed by using small molecules that interfere with specific signaling pathways that promote cancer cell proliferation and survival.256 Examples of such small-molecule compounds are selective tyrosine kinase inhibitors.257 Selective small-molecule inhibitors have less severe side effects than standard chemotherapy, but both types of therapy can lead to therapy resistance and relapse, as some cells within the heterogeneous tumor population survive and proliferate after an initial beneficial anticancer response.258 TGFβ-induced cancer cell acquisition of a mesenchymal phenotype has emerged as a key mechanism that confers the therapy-resistant and stemness phenotype to cancer cells.22 Blocking TGFβ may kill two birds with one stone by impairing metastasis and preventing resistance against anticancer treatments.
TβRI kinase inhibitors, such as galunisertib (LY2157299), have been combined with multiple chemotherapies, including paclitaxel, sorafenib, or gemcitabine in patients with TNBC, glioblastoma, HCC, or PDAC to improve antitumor efficacy.215,216,259–261 LY3200882 is combined with gemcitabine/paclitaxel/cisplatin in an ongoing phase 1 trial for the treatment of patients with solid tumors (NCT02937272), or is tested in combination with capecitabine for patients with resistant metastatic CRC (NCT04031872). The efficacy of a combination of vactosertib (TEW-7197) and paclitaxel is explored in an ongoing phase 1/2 clinical trial for the treatment of metastatic gastric cancer (NCT03698825). NIS793 is a pan anti-TGFβ-neutralizing antibody that is currently being tested in a phase 2 clinical trial for patients with metastatic PDAC in combination with gemcitabine/nab-paclitaxel chemotherapy (NCT04390763).262 TβRI kinase inhibitors have also been combined with targeted therapies to overcome drug resistance in pre-clinical cancer models. For example, downregulation of mediator complex subunit 12 (MED12) leads to resistance to multiple tyrosine kinase inhibitors in multiple cancer cells. Mechanistically, MED12 depletion was found to increase TβRII protein levels, mediate MEK/ERK activation, and induce the expression of mesenchymal markers. Treatment with the TβRI kinase inhibitor galunisertib (LY2157299) sensitized the therapeutic response of metastatic non-small cell lung cancers with downregulated MED12 to RTK inhibitors.263
In radiotherapy, ionizing radiation is used to kill or control the growth of malignant cells. Some types of cancer are notably radioresistant, such as glioblastoma, which produces abundant TGFβ in the microenvironment that leads to the radioresistance of glioma-initiating cells.264 Radiation can also cause normal tissues to become more fibrotic by inducing the expression and release of active TGFβ.265 Therefore, the combination of anti-TGFβ therapy with radiotherapy may improve treatment by enhancing tumor cell radiosensitivity and protecting normal tissues.266,267 In a phase 2 clinical trial, the TGFβ-blocking antibody fresolimumab (GC1008) was combined with focal irradiation for the treatment of metastatic breast cancer, and it was found to prolong median overall survival and demonstrated a favorable systemic immune response.268,269 In addition, fresolimumab (GC1008) combined with stereotactic ablative radiotherapy is in a phase 2 clinical trial for early-stage NSCLC treatment (NCT02581787). The combination of the TβRI kinase inhibitor LY3200882 with intensity-modulated radiotherapy is currently being tested in a phase 1 clinical trial of patients with solid tumors (NCT02937272).
Anti-TGFβ therapy with immunotherapy
Immune checkpoints are crucial regulators in the process of the immune response. Two types of immune checkpoint signals have been described. Costimulatory signals are mediated by CD27, CD28, CD40, CD134, CD137, and so on, for cytotoxic effector T cell proliferation and their migration toward the tumor.270,271 Coinhibitory signals increase T cell exhaustion and dysfunction, and this process engages coinhibitory molecules, such as PD-1, cytotoxic T-lymphocyte-associated protein 4 (CTLA4), lymphocyte activation gene 3 (LAG3), T cell immunoglobulin domain and mucin domain 3 (TIM3), and sialic acid-binding immunoglobulin-type lectin 7 (SIGLEC7).270 Normal physiological context balance costimulatory and coinhibitory signals for maintaining self-tolerance and preventing autoimmunity. However, this balance is perturbed in the TME; coinhibitory signals are used by tumor cells to evade an immune attack. Therefore, costimulatory signal agonists and coinhibitory signal antagonists have been applied in cancer immunotherapy, of which CTLA4, PD-1, and its partner programmed death-ligand 1 (PD-L1) are the most studied.271 PD-L1 is a transmembrane ligand expressed on the surface of cancer cells, tumor-infiltrating DCs and macrophages, and binds to PD-1 receptor located in the plasma membrane of cytotoxic CD8+ T cells to suppress effector T cell antitumor function.272 CTLA4 is a receptor expressed on Treg cells and downregulates immune responses.273 Immune checkpoint inhibitors (ICIs) are currently at the forefront of cancer therapy, as they display successful long-lasting antitumor efficacy, even for certain metastatic cancers. However, only 15% of patients respond to immunotherapy.274 Anti-PD-1 therapy not only initiates a tumor rejection program, but also induces a competing TGFβ-driven immunosuppressive program.262 TGFβ has also been found to drive immune response evasion and attenuate the tumor response to anti-PD-L1 therapy by contributing to the exclusion of CD8+ effector T cells.275,276 In addition, the unsuccessful antitumor activity of chimeric antigen receptor (CAR) T cell therapy has been attributed in part to TGFβ-induced immunosuppression.277 Because of these insights into TGFβ’s immunosuppressive function, the combination of anti-TGFβ therapy and immunotherapy is under intensive investigation.
Many preclinical studies have shown a great success in the combination of immunotherapy with anti-TGFβ therapy (Fig. 6). For example, the TβRI kinase inhibitor galunisertib (LY2157299) has been combined with anti-PD-1/L1 immunotherapy in the treatment of breast cancer in a mouse model. The combined treatment showed an efficient antitumor immunity, leading to more persistent and complete responses.278 The anti-CTLA4-TβRII chimera has been studied in preclinical melanoma and TNBC tumor xenograft mouse models and demonstrated superior results with respect to antitumor efficacy than treatment with anti-CTLA4 antibody alone.279 The TGFβ ligand trap AVID200 combined with ICIs showed enhanced antitumor immunity in TNBC homograft models.280 Recently, researchers engineered a bispecific receptor decoy named CD4 TGFβ Trap that selectively blocks TGF-β signaling in CD4+ T cells and promotes reorganization of tumor vasculature and cancer cell death in a mouse mammary tumor virus promoter-driven Polyoma middle T-antigen transgenic mouse model of breast cancer.281 SRK-181-mIgG1 is a fully human antibody that selectively binds to latent TGFβ1 and inhibits its activation, which avoided toxicities observed with pan-TGFβ inhibitors.282 The coadministration of SRK-181-mIgG1 together with anti-PD-1 inhibitors has been applied in the treatment of checkpoint blockade-resistant syngeneic mouse tumors that displayed an increased influx of intratumoral CD8+ T cells and a lower level of immunosuppressive myeloid cells.282 Researchers designed pH-responsive clustered nanoparticles that can deliver TβRI kinase inhibitor galunisertib (LY2157299) and small interfering RNA targeting PD-L1 to the PDAC stroma microenvironment, and this regimen showed significant antitumor efficiency by both provoking antitumor immunity and suppressing tumor growth in PDAC mouse models.283 However, despite these highly encouraging results mentioned above, we like to provide a note of caution as different tumor models with different immunogenicity can exhibit totally different, sometimes even opposite, results.284
Some therapies in which anti-TGFβ and immunotherapy are combined have entered clinical evaluations with patients (Table 1). For example, the TβRI kinase inhibitor vactosertib (TEW-7197) was combined with the anti-PD-L1/PD-1 monoclonal antibody durvalumab in a phase 2 clinical trial for patients with advanced urothelial carcinoma or NSCLC (NCT03732274), and with the anti-PD-1 antibody pembrolizumab in a phase 1/2 clinical evaluation of patients with metastatic colorectal or gastric cancer (NCT03724851). The combination therapies showed a good safety profile and antitumor activity in both trials.285,286 SAR439459 is a pan-TGFβ ligand-neutralizing antibody, and the combination of SAR439459 and anti-PD1 antibody lead to tumor regression in the MC38 tumor-bearing mice model.287 SAR439459 in combination with the anti-PD1 antibody cemiplimab is now being tested in a phase 1 clinical trials for patients with advanced solid tumors (NCT03192345). NIS793 is a monoclonal antibody blocking TGFβ1 and TGFβ2 and is being tested in combination with the anti-PD1 antibody PDR001 in a phase 1 clinical trial of patients with advanced malignancies (NCT02947165).262 M7824 is a bifunctional fusion protein consisting of the ectodomain of TβRII, which serves as a TGFβ ligand trap, and a human monoclonal antibody against PD-L1.288 The fusion protein can target both TGFβ- and PD-L1-mediated signaling pathways, and it displayed promising results by eliciting antitumor activity in multiple mouse cancer models.289 M7824 showed a manageable safety profile and encouraging clinical efficacy in phase 1 clinical trials, including patients with advanced solid tumors, NSCLCs, recurrent glioblastoma, cervical cancer, metastatic TNBC, heavily pretreated CRC, or human papillomavirus (HPV)-associated cancers.290–296 M7824 is now being evaluated in a phase 2 clinical trial for patients with advanced/metastatic biliary tract cancer, gallbladder cancer, recurrent respiratory papillomatosis, thymoma, CRC, head and neck squamous cell cancer, advanced pancreas cancer, or recurrent prostate cancer.297 Moreover, the evaluation of M7824 for patients with advanced NSCLC or biliary tract cancer is now under investigation in phase 3 clinical trials.278,298 A recent study discovered that an anti-GARP:TGFβ1 mAb (ABBV151), which selectively blocks TGFβ1 production by Treg cells, can induce the regression of anti-PD-1 immunotherapy-resistant tumors in a mouse cancer model.299,300 The phase 1 clinical trial of an anti-GARP:TGFβ1 mAb (ABBV151) as monotherapy and in combination with the anti-PD1 mAb budigalimab (ABBV-181) were recently initiated for the evaluation of their safety and tolerability for patients with advanced solid tumors (NCT03821935).
CAR T cell therapy has demonstrated remarkable success by utilizing engineered T cells with tumor antigens that lead to the recognition and attack of tumor cells in blood cancers; however, this method still remains a big challenge for treating solid tumors partially due to TGFβ-mediated immunosuppression.301 Inhibiting TGFβ signaling in CART cells can boost their antitumor efficacy in solid tumors.302,303 Other strategies also reported overcoming CART cell repression by interfering with TGFβ signaling, such as coexpression of the dominant-negative TβRII,304 coexpression of TβRII-41BB to switch the TGFβ-suppressive signal to a 41BB stimulatory signal,305 coexpression of TGFβ binding single-chain variable fragment,306 and constitutively active AKT.307
Anti-TGFβ therapy can also be combined with cancer vaccines to enhance treatment efficacy.308 Vaccination against the EMT transcription factor TWIST1 can induce effector T cell responses and suppress murine tumor growth and spontaneous metastasis.309 The combination of an adenovirus cancer vaccine encoding tumor-associated antigen TWIST1 (Ad-TWIST) and bifunctional fusion protein M7824 that targets both TGFβ and PD-L1 displayed superior improvement of the antitumor efficacy as compared to Ad-TWIST monotherapy in multiple murine models of human solid tumors.310 In addition, a phase 2 clinical trial study of combination therapies with M7824 and therapeutic Tri-Ad (ETBX-011, ETBX-051, and ETBX-061) vaccine for patients with head and neck neoplasms is ongoing (NCT04247282). M7824 has also been combined with the HPV vaccine PRGN-2009 in phase 1/2 clinical trials for subjects with HPV-associated cancers (NCT04432597) and combined with cancer vaccines that target the EMT driver transcription factor brachyury in phase 1/2 clinical trials for patients with advanced breast cancer (NCT04296942) or metastatic castration-resistant prostate cancer (NCT03493945).291,311,312
Concluding remarks
TGFβ was discovered in the late 1970s/early 1980s,313,314 and its role as a multifunctional regulator of normal and cancer cell growth became apparent shortly thereafter.315 The purification of TGFβ and cloning of its complementary DNA, as well as the identification of TGFβ receptors, paved the way for in vitro and in vivo studies on its mechanism of action and revealed the pleiotropic roles of TGFβ in controlling pathophysiological processes.1–4,27,55,316–318 Moreover, these advances allowed the pharmacological interference of key pathways with neutralizing antibodies against TGFβ or TGFβ receptor kinase inhibitors.319,320 While preclinical studies in mouse models have shown great promise for TGFβ pharmacological agents, the role of TGFβ as a tumor suppressor and critical role in maintaining tissue homeostasis have made its clinical translation demanding and prevented TGFβ targeting strategies from reaching clinical approval for the treatment of cancer patients. Significant progress has been made to (potentially) make treatment with TGFβ-targeting agents more safer and effective. For example, by performing intermittent dosing strategies bypassing cardiovascular toxicity,321,322 by specifically inhibiting only the TGFβ1 isoform that has the strongest link with cancer progression,282 or by selecting cancer patients for treatment with a mesenchymal phenotype and high TGFβ activity that are likely to benefit most from the TGFβ targeting.22,250
We now know that cancer cells frequently escape from the TGFβ-induced cytostatic response and that TGFβ drives the EMT of cancer cells. Mesenchymal cancer cells have been linked to metastasis and chemotherapy, targeted therapy, and/or radiotherapy resistance. As most cancer patients die of metastasis and demonstrate relapse after chemotherapy, targeted therapy, and/or radiotherapy, the combination of these therapies with TGFβ inhibitors are being tested. Moreover, TGFβ strongly promotes cancer progression by acting on the TME, activating CAFs, stimulating angiogenesis, and eluding the immune system. How TGFβ inhibitors elicit their anticancer effect in patients is frequently unclear, but its reversal of immunosuppressive activity in the TME might be of key importance. Moreover, as the clinical failure of immune checkpoint inhibitors for cancer treatment has been linked to overactive TGFβ signaling activity (at least under some circumstances), we are witnessing a profound renewed interest in TGFβ as a target for cancer therapy. Thus, by combining chemotherapy, targeted therapy, radiotherapy, and immunotherapy with TGFβ-targeting drugs, treatments can be made more efficient by improving antitumor efficacy and reducing therapy resistance. One aspect remains of key importance, finding reliable biomarkers that enable clinicians to select the best (combinatorial) treatment for each individual cancer patient.
Acknowledgements
We apologize to authors whose papers could not be cited due to space limitations. We are grateful to Dieuwke Marvin and Adilson Fonseca Teixeira for a critical reading and comments. We thank Gerard van der Zon for the immunofluorescent staining images of NMuMG cells. Research on targeting TGFβ for cancer therapy in our laboratory is supported by the Cancer Genomics Centre Netherlands.
Author contributions
P.t.D. and S.L. conceived of the paper; S.L. wrote the initial draft of the paper and generated the figures and tables; P.t.D. and J.R. edited and revised the paper. All authors approved the final paper.
Competing interests
The authors declare no competing interests.
References
- 1.David CJ, Massagué J. Contextual determinants of TGFβ action in development, immunity and cancer. Nat. Rev. Mol. Cell Biol. 2018;19:419–435. doi: 10.1038/s41580-018-0007-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Moustakas A, Heldin CH. The regulation of TGFβ signal transduction. Development. 2009;136:3699–3714. doi: 10.1242/dev.030338. [DOI] [PubMed] [Google Scholar]
- 3.Batlle E, Massagué J. Transforming growth factor-β signaling in immunity and cancer. Immunity. 2019;50:924–940. doi: 10.1016/j.immuni.2019.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Derynck, R., Turley, S. J. & Akhurst, R. J. TGFβ biology in cancer progression and immunotherapy. Nat. Rev. Clin. Oncol. 10.1038/s41571-020-0403-1 (2020). [DOI] [PMC free article] [PubMed]
- 5.Flanders KC, et al. Quantitation of TGF-β proteins in mouse tissues shows reciprocal changes in TGF-β1 and TGF-β3 in normal vs neoplastic mammary epithelium. Oncotarget. 2016;7:38164–38179. doi: 10.18632/oncotarget.9416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ten Dijke P, Hansen P, Iwata KK, Pieler C, Foulkes JG. Identification of another member of the transforming growth factor type β gene family. Proc. Natl Acad. Sci. USA. 1988;85:4715–4719. doi: 10.1073/pnas.85.13.4715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cheifetz S, et al. Distinct transforming growth factor-β (TGF-β) receptor subsets as determinants of cellular responsiveness to three TGF-β isoforms. J. Biol. Chem. 1990;265:20533–20538. [PubMed] [Google Scholar]
- 8.Shull MM, et al. Targeted disruption of the mouse transforming growth factor-β1 gene results in multifocal inflammatory disease. Nature. 1992;359:693–699. doi: 10.1038/359693a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dickson MC, et al. Defective haematopoiesis and vasculogenesis in transforming growth factor-β 1 knock out mice. Development. 1995;121:1845–1854. doi: 10.1242/dev.121.6.1845. [DOI] [PubMed] [Google Scholar]
- 10.Sanford LP, et al. TGFβ2 knockout mice have multiple developmental defects that are non-overlapping with other TGFβ knockout phenotypes. Development. 1997;124:2659–2670. doi: 10.1242/dev.124.13.2659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Proetzel G, et al. Transforming growth factor-β3 is required for secondary palate fusion. Nat. Genet. 1995;11:409–414. doi: 10.1038/ng1295-409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kaartinen V, et al. Abnormal lung development and cleft palate in mice lacking TGF-β3 indicates defects of epithelial-mesenchymal interaction. Nat. Genet. 1995;11:415–421. doi: 10.1038/ng1295-415. [DOI] [PubMed] [Google Scholar]
- 13.Seyedin SM, et al. Cartilage-inducing factor-A. Apparent identity to transforming growth factor-β. J. Biol. Chem. 1986;261:5693–5695. doi: 10.1016/S0021-9258(17)38436-3. [DOI] [PubMed] [Google Scholar]
- 14.Assoian RK, Komoriya A, Meyers CA, Miller DM, Sporn MB. Transforming growth factor-β in human platelets. Identification of a major storage site, purification, and characterization. J. Biol. Chem. 1983;258:7155–7160. doi: 10.1016/S0021-9258(18)32345-7. [DOI] [PubMed] [Google Scholar]
- 15.Xie F, Ling L, van Dam H, Zhou F, Zhang L. TGF-β signaling in cancer metastasis. Acta Biochim. Biophys. Sin. 2018;50:121–132. doi: 10.1093/abbs/gmx123. [DOI] [PubMed] [Google Scholar]
- 16.Massagué J. TGFβ signalling in context. Nat. Rev. Mol. Cell Biol. 2012;13:616–630. doi: 10.1038/nrm3434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yang L, Moses HL. Transforming growth factor β: tumor suppressor or promoter? Are host immune cells the answer? Cancer Res. 2008;68:9107–9111. doi: 10.1158/0008-5472.CAN-08-2556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Derynck R, Muthusamy BP, Saeteurn KY. Signaling pathway cooperation in TGF-β-induced epithelial–mesenchymal transition. Curr. Opin. Cell Biol. 2014;31:56–66. doi: 10.1016/j.ceb.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hao Y, Baker D, Ten Dijke P. TGF-β-mediated epithelial-mesenchymal transition and cancer metastasis. Int. J. Mol. Sci. 2019;20:2767. doi: 10.3390/ijms20112767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen W, Ten Dijke P. Immunoregulation by members of the TGFβ superfamily. Nat. Rev. Immunol. 2016;16:723–740. doi: 10.1038/nri.2016.112. [DOI] [PubMed] [Google Scholar]
- 21.Huynh LK, Hipolito CJ, Ten Dijke P. A perspective on the development of TGF-β inhibitors for cancer treatment. Biomolecules. 2019;9:743. doi: 10.3390/biom9110743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Colak S, Ten Dijke P. Targeting TGF-β signaling in cancer. Trends Cancer. 2017;3:56–71. doi: 10.1016/j.trecan.2016.11.008. [DOI] [PubMed] [Google Scholar]
- 23.Teixeira AF, Ten Dijke P, Zhu H-J. On-target anti-TGF-β therapies are not succeeding in clinical cancer treatments: what are remaining challenges? Front. Cell Dev. Biol. 2020;8:605. doi: 10.3389/fcell.2020.00605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Farrington DL, et al. Development and validation of a phosphorylated SMAD ex vivo stimulation assay. Biomarkers. 2007;12:313–330. doi: 10.1080/13547500601162441. [DOI] [PubMed] [Google Scholar]
- 25.Cao Y, et al. NGS-based transcriptome profiling reveals biomarkers for companion diagnostics of the TGF-β receptor blocker galunisertib in HCC. Cell Death Dis. 2017;8:e2634. doi: 10.1038/cddis.2017.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gentry LE, Lioubin MN, Purchio AF, Marquardt H. Molecular events in the processing of recombinant type 1 pre-pro-transforming growth factor β to the mature polypeptide. Mol. Cell. Biol. 1988;8:4162–4168. doi: 10.1128/MCB.8.10.4162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Derynck R, et al. Human transforming growth factor-β complementary DNA sequence and expression in normal and transformed cells. Nature. 1985;316:701–705. doi: 10.1038/316701a0. [DOI] [PubMed] [Google Scholar]
- 28.Gray AM, Mason AJ. Requirement for activin A and transforming growth factor-β1 pro-regions in homodimer assembly. Science. 1990;247:1328–1330. doi: 10.1126/science.2315700. [DOI] [PubMed] [Google Scholar]
- 29.Dubois CM, Laprise MH, Blanchette F, Gentry LE, Leduc R. Processing of transforming growth factor β1 precursor by human furin convertase. J. Biol. Chem. 1995;270:10618–10624. doi: 10.1074/jbc.270.18.10618. [DOI] [PubMed] [Google Scholar]
- 30.Shi M, et al. Latent TGF-β structure and activation. Nature. 2011;474:343–349. doi: 10.1038/nature10152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lyons RM, Keski-Oja J, Moses HL. Proteolytic activation of latent transforming growth factor-β from fibroblast-conditioned medium. J. Cell Biol. 1988;106:1659–1665. doi: 10.1083/jcb.106.5.1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pfeilschifter J, Bonewald L, Mundy GR. Characterization of the latent transforming growth factor β complex in bone. J. Bone Miner. Res. 1990;5:49–58. doi: 10.1002/jbmr.5650050109. [DOI] [PubMed] [Google Scholar]
- 33.Barcellos-Hoff MH, Dix TA. Redox-mediated activation of latent transforming growth factor-β1. Mol. Endocrinol. 1996;10:1077–1083. doi: 10.1210/mend.10.9.8885242. [DOI] [PubMed] [Google Scholar]
- 34.Jobling MF, et al. Isoform-specific activation of latent transforming growth factor β (LTGF-β) by reactive oxygen species. Radiat. Res. 2006;166:839–848. doi: 10.1667/RR0695.1. [DOI] [PubMed] [Google Scholar]
- 35.Annes JP, Munger JS, Rifkin DB. Making sense of latent TGFβ activation. J. Cell Biol. 2003;116:217–224. doi: 10.1242/jcs.00229. [DOI] [PubMed] [Google Scholar]
- 36.Lyons RM, Gentry LE, Purchio AF, Moses HL. Mechanism of activation of latent recombinant transforming growth factor β 1 by plasmin. J. Cell Biol. 1990;110:1361–1367. doi: 10.1083/jcb.110.4.1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Crawford SE, et al. Thrombospondin-1 is a major activator of TGF-β1 in vivo. Cell. 1998;93:1159–1170. doi: 10.1016/S0092-8674(00)81460-9. [DOI] [PubMed] [Google Scholar]
- 38.Miyazono K, Olofsson A, Colosetti P, Heldin CH. A role of the latent TGF-β1-binding protein in the assembly and secretion of TGF-β1. EMBO J. 1991;10:1091–1101. doi: 10.1002/j.1460-2075.1991.tb08049.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Saharinen J, Taipale J, Keski-Oja J. Association of the small latent transforming growth factor-β with an eight cysteine repeat of its binding protein LTBP-1. EMBO J. 1996;15:245–253. doi: 10.1002/j.1460-2075.1996.tb00355.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zilberberg L, et al. Specificity of latent TGF-β binding protein (LTBP) incorporation into matrix: role of fibrillins and fibronectin. J. Cell Physiol. 2012;227:3828–3836. doi: 10.1002/jcp.24094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liénart S, et al. Structural basis of latent TGF-β1 presentation and activation by GARP on human regulatory T cells. Science. 2018;362:952–956. doi: 10.1126/science.aau2909. [DOI] [PubMed] [Google Scholar]
- 42.Bachmann M, Kukkurainen S, Hytönen VP, Wehrle-Haller B. Cell adhesion by integrins. Physiol. Rev. 2019;99:1655–1699. doi: 10.1152/physrev.00036.2018. [DOI] [PubMed] [Google Scholar]
- 43.Lodyga M, Hinz B. TGF-β1—a truly transforming growth factor in fibrosis and immunity. Semin. Cell Dev. Biol. 2020;101:123–139. doi: 10.1016/j.semcdb.2019.12.010. [DOI] [PubMed] [Google Scholar]
- 44.Humphries MJ. Monoclonal antibodies as probes of integrin priming and activation. Biochem. Soc. Trans. 2004;32:407–411. doi: 10.1042/bst0320407. [DOI] [PubMed] [Google Scholar]
- 45.Worthington JJ, Klementowicz JE, Travis MA. TGFβ: a sleeping giant awoken by integrins. Trends Biochem. Sci. 2011;36:47–54. doi: 10.1016/j.tibs.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 46.Mu D, et al. The integrin α(v)β8 mediates epithelial homeostasis through MT1-MMP-dependent activation of TGF-β1. J. Cell Biol. 2002;157:493–507. doi: 10.1083/jcb.200109100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.López-Casillas F, Wrana JL, Massagué J. Betaglycan presents ligand to the TGFβ signaling receptor. Cell. 1993;73:1435–1444. doi: 10.1016/0092-8674(93)90368-Z. [DOI] [PubMed] [Google Scholar]
- 48.Yamashita H, Ten Dijke P, Franzén P, Miyazono K, Heldin CH. Formation of hetero-oligomeric complexes of type I and type II receptors for transforming growth factor-β. J. Biol. Chem. 1994;269:20172–20178. doi: 10.1016/S0021-9258(17)42126-0. [DOI] [PubMed] [Google Scholar]
- 49.Weis-Garcia F, Massagué J. Complementation between kinase-defective and activation-defective TGF-β receptors reveals a novel form of receptor cooperativity essential for signaling. EMBO J. 1996;15:276–289. doi: 10.1002/j.1460-2075.1996.tb00358.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Luo K, Lodish HF. Signaling by chimeric erythropoietin-TGF-β receptors: homodimerization of the cytoplasmic domain of the type I TGF-β receptor and heterodimerization with the type II receptor are both required for intracellular signal transduction. EMBO J. 1996;15:4485–4496. doi: 10.1002/j.1460-2075.1996.tb00826.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wrana JL, Attisano L, Wieser R, Ventura F, Massagué J. Mechanism of activation of the TGF-β receptor. Nature. 1994;370:341–347. doi: 10.1038/370341a0. [DOI] [PubMed] [Google Scholar]
- 52.Wieser R, Wrana JL, Massagué J. GS domain mutations that constitutively activate TβRI, the downstream signaling component in the TGF-β receptor complex. EMBO J. 1995;14:2199–2208. doi: 10.1002/j.1460-2075.1995.tb07214.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Abdollah S, et al. TβRI phosphorylation of Smad2 on Ser465 and Ser467 is required for Smad2-Smad4 complex formation and signaling. J. Biol. Chem. 1997;272:27678–27685. doi: 10.1074/jbc.272.44.27678. [DOI] [PubMed] [Google Scholar]
- 54.Souchelnytskyi S, et al. Phosphorylation of Ser465 and Ser467 in the C terminus of Smad2 mediates interaction with Smad4 and is required for transforming growth factor-β signaling. J. Biol. Chem. 1997;272:28107–28115. doi: 10.1074/jbc.272.44.28107. [DOI] [PubMed] [Google Scholar]
- 55.Heldin CH, Moustakas A. Signaling receptors for TGF-β family members. Cold Spring Harb. Perspect. Biol. 2016;8:a022053. doi: 10.1101/cshperspect.a022053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Derynck R, Budi EH. Specificity, versatility, and control of TGF-β family signaling. Sci. Signal. 2019;12:aav5183. doi: 10.1126/scisignal.aav5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nakao A, et al. Identification of Smad7, a TGFβ-inducible antagonist of TGF-β signalling. Nature. 1997;389:631–635. doi: 10.1038/39369. [DOI] [PubMed] [Google Scholar]
- 58.Hayashi H, et al. The MAD-related protein Smad7 associates with the TGFβ receptor and functions as an antagonist of TGFβ signaling. Cell. 1997;89:1165–1173. doi: 10.1016/S0092-8674(00)80303-7. [DOI] [PubMed] [Google Scholar]
- 59.de Ceuninck van Capelle C, Spit M, Ten Dijke P. Current perspectives on inhibitory SMAD7 in health and disease. Crit. Rev. Biochem. Mol. Biol. 2020;55:691–715. doi: 10.1080/10409238.2020.1828260. [DOI] [PubMed] [Google Scholar]
- 60.Zhang S, et al. Smad7 antagonizes transforming growth factor β signaling in the nucleus by interfering with functional Smad-DNA complex formation. Mol. Cell Biol. 2007;27:4488–4499. doi: 10.1128/MCB.01636-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Shi X, et al. Study of interaction between Smad7 and DNA by single-molecule force spectroscopy. Biochem. Biophys. Res. Commun. 2008;377:1284–1287. doi: 10.1016/j.bbrc.2008.10.145. [DOI] [PubMed] [Google Scholar]
- 62.Kavsak P, et al. Smad7 binds to Smurf2 to form an E3 ubiquitin ligase that targets the TGFβ receptor for degradation. Mol. Cell. 2000;6:1365–1375. doi: 10.1016/S1097-2765(00)00134-9. [DOI] [PubMed] [Google Scholar]
- 63.Komuro A, et al. Negative regulation of transforming growth factor-β (TGF-β) signaling by WW domain-containing protein 1 (WWP1) Oncogene. 2004;23:6914–6923. doi: 10.1038/sj.onc.1207885. [DOI] [PubMed] [Google Scholar]
- 64.Kuratomi G, et al. NEDD4-2 (neural precursor cell expressed, developmentally down-regulated 4-2) negatively regulates TGF-β (transforming growth factor-β) signalling by inducing ubiquitin-mediated degradation of Smad2 and TGF-β type I receptor. Biochem. J. 2005;386:461–470. doi: 10.1042/BJ20040738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Liu S, de Boeck M, van Dam H, Ten Dijke P. Regulation of the TGF-β pathway by deubiquitinases in cancer. Int. J. Biochem. Cell Biol. 2016;76:135–145. doi: 10.1016/j.biocel.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 66.Liu S, et al. Deubiquitinase activity profiling identifies UCHL1 as a candidate oncoprotein that promotes TGFβ-induced breast cancer metastasis. Clin. Cancer Res. 2020;26:1460–1473. doi: 10.1158/1078-0432.CCR-19-1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Eichhorn PJ, et al. USP15 stabilizes TGF-β receptor I and promotes oncogenesis through the activation of TGF-β signaling in glioblastoma. Nat. Med. 2012;18:429–435. doi: 10.1038/nm.2619. [DOI] [PubMed] [Google Scholar]
- 68.Zhang L, et al. USP4 is regulated by AKT phosphorylation and directly deubiquitylates TGF-β type I receptor. Nat. Cell Biol. 2012;14:717–726. doi: 10.1038/ncb2522. [DOI] [PubMed] [Google Scholar]
- 69.Zhang Y, Alexander PB, Wang XF. TGF-β family signaling in the control of cell proliferation and survival. Cold Spring Harb. Perspect. Biol. 2017;9:a022145. doi: 10.1101/cshperspect.a022145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Seoane J, et al. TGFβ influences Myc, Miz-1 and Smad to control the CDK inhibitor p15INK4b. Nat. Cell Biol. 2001;3:400–408. doi: 10.1038/35070086. [DOI] [PubMed] [Google Scholar]
- 71.Seoane J, Le HV, Massagué J. Myc suppression of the p21(Cip1) Cdk inhibitor influences the outcome of the p53 response to DNA damage. Nature. 2002;419:729–734. doi: 10.1038/nature01119. [DOI] [PubMed] [Google Scholar]
- 72.Azar R, Alard A, Susini C, Bousquet C, Pyronnet S. 4E-BP1 is a target of Smad4 essential for TGF-β mediated inhibition of cell proliferation. EMBO J. 2009;28:3514–3522. doi: 10.1038/emboj.2009.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chen Z, et al. Targeted inhibition of p57 and p15 blocks transforming growth factor β-inhibited proliferation of primary cultured human limbal epithelial cells. Mol. Vis. 2006;12:983–994. [PMC free article] [PubMed] [Google Scholar]
- 74.Planas-Silva MD, Weinberg RA. The restriction point and control of cell proliferation. Curr. Opin. Cell Biol. 1997;9:768–772. doi: 10.1016/S0955-0674(97)80076-2. [DOI] [PubMed] [Google Scholar]
- 75.Kang SH, et al. Rapid induction of p21WAF1 but delayed down-regulation of Cdc25A in the TGF-β-induced cell cycle arrest of gastric carcinoma cells. Br. J. Cancer. 1999;80:1144–1149. doi: 10.1038/sj.bjc.6690478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bhowmick NA, et al. TGF-β-induced RhoA and p160ROCK activation is involved in the inhibition of Cdc25A with resultant cell-cycle arrest. Proc. Natl Acad. Sci. USA. 2003;100:15548–15553. doi: 10.1073/pnas.2536483100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Schwarz JK, et al. Expression of the E2F1 transcription factor overcomes type β transforming growth factor-mediated growth suppression. Proc. Natl Acad. Sci. USA. 1995;92:483–487. doi: 10.1073/pnas.92.2.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Pietenpol JA, Holt JT, Stein RW, Moses HL. Transforming growth factor β1 suppression of c-myc gene transcription: role in inhibition of keratinocyte proliferation. Proc. Natl Acad. Sci. USA. 1990;87:3758–3762. doi: 10.1073/pnas.87.10.3758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Chen C-R, Kang Y, Massagué J. Defective repression of c-myc in breast cancer cells: a loss at the core of the transforming growth factor β growth arrest program. Proc. Natl Acad. Sci. USA. 2001;98:992–999. doi: 10.1073/pnas.98.3.992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kang Y, Chen CR, Massagué J. A self-enabling TGFβ response coupled to stress signaling: Smad engages stress response factor ATF3 for Id1 repression in epithelial cells. Mol. Cell. 2003;11:915–926. doi: 10.1016/S1097-2765(03)00109-6. [DOI] [PubMed] [Google Scholar]
- 81.Huang Y-H, et al. ID1 mediates escape from TGFβ tumor suppression in pancreatic cancer. Cancer Discov. 2020;10:142–157. doi: 10.1158/2159-8290.CD-19-0529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wildey GM, Patil S, Howe PH. Smad3 potentiates transforming growth factor β (TGFβ)-induced apoptosis and expression of the BH3-only protein Bim in WEHI 231 B lymphocytes. J. Biol. Chem. 2003;278:18069–18077. doi: 10.1074/jbc.M211958200. [DOI] [PubMed] [Google Scholar]
- 83.Ramjaun A, Tomlinson S, Eddaoudi A, Downward J. Upregulation of two BH3-only proteins, Bmf and Bim, during TGFβ-induced apoptosis. Oncogene. 2007;26:970–981. doi: 10.1038/sj.onc.1209852. [DOI] [PubMed] [Google Scholar]
- 84.Ramesh S, Wildey GM, Howe PH. Transforming growth factor β (TGFβ)-induced apoptosis: the rise and fall of Bim. Cell Cycle. 2009;8:11–17. doi: 10.4161/cc.8.1.7291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Schrantz N, et al. 38-mediated regulation of an Fas-associated death domain protein-independent pathway leading to caspase-8 activation during TGFβ-induced apoptosis in human Burkitt lymphoma B cells BL41. Mol. Biol. Cell. 2001;12:3139–3151. doi: 10.1091/mbc.12.10.3139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Huang Y-L, Chou C-K. Bcl-2 blocks apoptotic signal of transforming growth factor-β in human hepatoma cells. J. Biomed. Sci. 1998;5:185–191. doi: 10.1007/BF02253468. [DOI] [PubMed] [Google Scholar]
- 87.Saltzman A, et al. Transforming growth factor-β-mediated apoptosis in the Ramos B-lymphoma cell line is accompanied by caspase activation and Bcl-XL downregulation. Exp. Cell Res. 1998;242:244–254. doi: 10.1006/excr.1998.4096. [DOI] [PubMed] [Google Scholar]
- 88.Heath VL, Murphy EE, Crain C, Tomlinson MG, O’Garra A. TGF-β1 down-regulates Th2 development and results in decreased IL-4-induced STAT6 activation and GATA-3 expression. Eur. J. Immunol. 2000;30:2639–2649. doi: 10.1002/1521-4141(200009)30:9<2639::AID-IMMU2639>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 89.Park IK, Shultz LD, Letterio JJ, Gorham JD. TGF-β1 inhibits T-bet induction by IFN-gamma in murine CD4+ T cells through the protein tyrosine phosphatase Src homology region 2 domain-containing phosphatase-1. J. Immunol. 2005;175:5666–5674. doi: 10.4049/jimmunol.175.9.5666. [DOI] [PubMed] [Google Scholar]
- 90.Markowitz S, et al. Inactivation of the type II TGF-β receptor in colon cancer cells with microsatellite instability. Science. 1995;268:1336–1338. doi: 10.1126/science.7761852. [DOI] [PubMed] [Google Scholar]
- 91.Riggins GJ, Kinzler KW, Vogelstein B, Thiagalingam S. Frequency of Smad gene mutations in human cancers. Cancer Res. 1997;57:2578–2580. [PubMed] [Google Scholar]
- 92.Eppert K, et al. MADR2 maps to 18q21 and encodes a TGFβ-regulated MAD-related protein that is functionally mutated in colorectal carcinoma. Cell. 1996;86:543–552. doi: 10.1016/S0092-8674(00)80128-2. [DOI] [PubMed] [Google Scholar]
- 93.Korkut A, et al. A pan-cancer analysis reveals high-frequency genetic alterations in mediators of signaling by the TGF-β superfamily. Cell Syst. 2018;7:422–437. doi: 10.1016/j.cels.2018.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hahn SA, et al. DPC4, a candidate tumor suppressor gene at human chromosome 18q21.1. Science. 1996;271:350–353. doi: 10.1126/science.271.5247.350. [DOI] [PubMed] [Google Scholar]
- 95.Zhou L, et al. TGF-β-induced Foxp3 inhibits TH-17 cell differentiation by antagonizing RORγt function. Nature. 2008;453:236–240. doi: 10.1038/nature06878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Trotta R, et al. TGF-β utilizes SMAD3 to inhibit CD16-mediated IFN-γ production and antibody-dependent cellular cytotoxicity in human NK cells. J. Immunol. 2008;181:3784–3792. doi: 10.4049/jimmunol.181.6.3784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Trinh TL, et al. Immune evasion by TGFβ-induced miR-183 repression of MICA/B expression in human lung tumor cells. Oncoimmunology. 2019;8:e1557372. doi: 10.1080/2162402X.2018.1557372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Thomas DA, Massagué J. TGF-β directly targets cytotoxic T cell functions during tumor evasion of immune surveillance. Cancer Cell. 2005;8:369–380. doi: 10.1016/j.ccr.2005.10.012. [DOI] [PubMed] [Google Scholar]
- 99.Castriconi R, et al. Transforming growth factor β1 inhibits expression of NKp30 and NKG2D receptors: consequences for the NK-mediated killing of dendritic cells. Proc. Natl Acad. Sci. USA. 2003;100:4120–4125. doi: 10.1073/pnas.0730640100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pollari S, et al. Identification of microRNAs inhibiting TGF-β-induced IL-11 production in bone metastatic breast cancer cells. PLoS ONE. 2012;7:e37361. doi: 10.1371/journal.pone.0037361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yin JJ, et al. TGF-β signaling blockade inhibits PTHrP secretion by breast cancer cells and bone metastases development. J. Clin. Invest. 1999;103:197–206. doi: 10.1172/JCI3523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wiercinska E, et al. The TGF-β/Smad pathway induces breast cancer cell invasion through the up-regulation of matrix metalloproteinase 2 and 9 in a spheroid invasion model system. Breast Cancer Res. Treat. 2011;128:657–666. doi: 10.1007/s10549-010-1147-x. [DOI] [PubMed] [Google Scholar]
- 103.Ruiz-Ortega M, Rodríguez-Vita J, Sanchez-Lopez E, Carvajal G, Egido J. TGF-β signaling in vascular fibrosis. Cardiovasc. Res. 2007;74:196–206. doi: 10.1016/j.cardiores.2007.02.008. [DOI] [PubMed] [Google Scholar]
- 104.Goumans M-J, Liu Z, Ten Dijke P. TGF-β signaling in vascular biology and dysfunction. Cell Res. 2009;19:116–127. doi: 10.1038/cr.2008.326. [DOI] [PubMed] [Google Scholar]
- 105.Zhang YE. Non-Smad signaling pathways of the TGF-β family. Cold Spring Harb. Perspect. Biol. 2017;9:a022129. doi: 10.1101/cshperspect.a022129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Vardouli L, Moustakas A, Stournaras C. LIM-kinase 2 and cofilin phosphorylation mediate actin cytoskeleton reorganization induced by transforming growth factor-β. J. Biol. Chem. 2005;280:11448–11457. doi: 10.1074/jbc.M402651200. [DOI] [PubMed] [Google Scholar]
- 107.Ozdamar B, et al. Regulation of the polarity protein Par6 by TGFβ receptors controls epithelial cell plasticity. Science. 2005;307:1603–1609. doi: 10.1126/science.1105718. [DOI] [PubMed] [Google Scholar]
- 108.Lee MK, et al. TGF-β activates Erk MAP kinase signalling through direct phosphorylation of ShcA. EMBO J. 2007;26:3957–3967. doi: 10.1038/sj.emboj.7601818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yamashita M, et al. TRAF6 mediates Smad-independent activation of JNK and p38 by TGF-β. Mol. Cell. 2008;31:918–924. doi: 10.1016/j.molcel.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sorrentino A, et al. The type I TGF-β receptor engages TRAF6 to activate TAK1 in a receptor kinase-independent manner. Nat. Cell Biol. 2008;10:1199–1207. doi: 10.1038/ncb1780. [DOI] [PubMed] [Google Scholar]
- 111.Chen RH, Su YH, Chuang RL, Chang TY. Suppression of transforming growth factor-β-induced apoptosis through a phosphatidylinositol 3-kinase/Akt-dependent pathway. Oncogene. 1998;17:1959–1968. doi: 10.1038/sj.onc.1202111. [DOI] [PubMed] [Google Scholar]
- 112.Zhang YE. Non-Smad pathways in TGF-β signaling. Cell Res. 2009;19:128–139. doi: 10.1038/cr.2008.328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Su J, et al. TGF-β orchestrates fibrogenic and developmental EMTs via the RAS effector RREB1. Nature. 2020;577:566–571. doi: 10.1038/s41586-019-1897-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lemmon MA, Schlessinger J. Cell signaling by receptor tyrosine kinases. Cell. 2010;141:1117–1134. doi: 10.1016/j.cell.2010.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Battegay EJ, Raines EW, Seifert RA, Bowen-Pope DF, Ross R. TGF-β induces bimodal proliferation of connective tissue cells via complex control of an autocrine PDGF loop. Cell. 1990;63:515–524. doi: 10.1016/0092-8674(90)90448-N. [DOI] [PubMed] [Google Scholar]
- 116.Bruna A, et al. High TGFβ-Smad activity confers poor prognosis in glioma patients and promotes cell proliferation depending on the methylation of the PDGF-B gene. Cancer Cell. 2007;11:147–160. doi: 10.1016/j.ccr.2006.11.023. [DOI] [PubMed] [Google Scholar]
- 117.Ikushima H, et al. An Id-like molecule, HHM, is a synexpression group-restricted regulator of TGF-β signalling. EMBO J. 2008;27:2955–2965. doi: 10.1038/emboj.2008.218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014;15:178–196. doi: 10.1038/nrm3758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Deckers M, et al. The tumor suppressor Smad4 is required for transforming growth factor β-induced epithelial to mesenchymal transition and bone metastasis of breast cancer cells. Cancer Res. 2006;66:2202–2209. doi: 10.1158/0008-5472.CAN-05-3560. [DOI] [PubMed] [Google Scholar]
- 120.Valcourt U, Kowanetz M, Niimi H, Heldin CH, Moustakas A. TGF-β and the Smad signaling pathway support transcriptomic reprogramming during epithelial-mesenchymal cell transition. Mol. Biol. Cell. 2005;16:1987–2002. doi: 10.1091/mbc.e04-08-0658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bakin AV, Tomlinson AK, Bhowmick NA, Moses HL, Arteaga CL. Phosphatidylinositol 3-kinase function is required for transforming growth factor β-mediated epithelial to mesenchymal transition and cell migration. J. Biol. Chem. 2000;275:36803–36810. doi: 10.1074/jbc.M005912200. [DOI] [PubMed] [Google Scholar]
- 122.Yu L, Hébert MC, Zhang YE. TGF-β receptor-activated p38 MAP kinase mediates Smad-independent TGF-β responses. EMBO J. 2002;21:3749–3759. doi: 10.1093/emboj/cdf366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Bhowmick NA, et al. Transforming growth factor-β1 mediates epithelial to mesenchymal transdifferentiation through a RhoA-dependent mechanism. Mol. Biol. Cell. 2001;12:27–36. doi: 10.1091/mbc.12.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Xie L, et al. Activation of the Erk pathway is required for TGF-β1-induced EMT in vitro. Neoplasia. 2004;6:603–610. doi: 10.1593/neo.04241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Yang J, et al. Guidelines and definitions for research on epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2020;21:341–352. doi: 10.1038/s41580-020-0237-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Derynck R, Muthusamy BP, Saeteurn KY. Signaling pathway cooperation in TGF-β-induced epithelial-mesenchymal transition. Curr. Opin. Cell Biol. 2014;31:56–66. doi: 10.1016/j.ceb.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Owens RB, Smith HS, Hackett AJ. Epithelial cell cultures from normal glandular tissue of mice. J. Natl Cancer Inst. 1974;53:261–269. doi: 10.1093/jnci/53.1.261. [DOI] [PubMed] [Google Scholar]
- 128.Miettinen PJ, Ebner R, Lopez AR, Derynck R. TGF-β induced transdifferentiation of mammary epithelial cells to mesenchymal cells: involvement of type I receptors. J. Cell Biol. 1994;127:2021–2036. doi: 10.1083/jcb.127.6.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Piek E, Moustakas A, Kurisaki A, Heldin CH, Ten Dijke P. TGF-β type I receptor/ALK-5 and Smad proteins mediate epithelial to mesenchymal transdifferentiation in NMuMG breast epithelial cells. J. Cell Sci. 1999;112:4557–4568. doi: 10.1242/jcs.112.24.4557. [DOI] [PubMed] [Google Scholar]
- 130.Cailleau R, Young R, Olivé M, Reeves WJ., Jr. Breast tumor cell lines from pleural effusions. J. Natl Cancer Inst. 1974;53:661–674. doi: 10.1093/jnci/53.3.661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Kang Y, et al. Breast cancer bone metastasis mediated by the Smad tumor suppressor pathway. Proc. Natl Acad. Sci. USA. 2005;102:13909–13914. doi: 10.1073/pnas.0506517102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kalluri R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer. 2016;16:582–598. doi: 10.1038/nrc.2016.73. [DOI] [PubMed] [Google Scholar]
- 133.Yu Y, et al. Cancer-associated fibroblasts induce epithelial–mesenchymal transition of breast cancer cells through paracrine TGF-β signalling. Br. J. Cancer. 2014;110:724–732. doi: 10.1038/bjc.2013.768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Calon A, et al. Dependency of colorectal cancer on a TGF-β-driven program in stromal cells for metastasis initiation. Cancer Cell. 2012;22:571–584. doi: 10.1016/j.ccr.2012.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Augsten M. Cancer-associated fibroblasts as another polarized cell type of the tumor microenvironment. Front. Oncol. 2014;4:62–62. doi: 10.3389/fonc.2014.00062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Bartoschek M, et al. Spatially and functionally distinct subclasses of breast cancer-associated fibroblasts revealed by single cell RNA sequencing. Nat. Commun. 2018;9:1–13. doi: 10.1038/s41467-018-07582-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Öhlund D, et al. Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J. Exp. Med. 2017;214:579–596. doi: 10.1084/jem.20162024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Elyada E, et al. Cross-species single-cell analysis of pancreatic ductal adenocarcinoma reveals antigen-presenting cancer-associated fibroblasts. Cancer Discov. 2019;9:1102–1123. doi: 10.1158/2159-8290.CD-19-0094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Pereira BA, et al. CAF subpopulations: a new reservoir of stromal targets in pancreatic cancer. Trends Cancer. 2019;5:724–741. doi: 10.1016/j.trecan.2019.09.010. [DOI] [PubMed] [Google Scholar]
- 140.Kanzaki R, Pietras K. Heterogeneity of cancer-associated fibroblasts: opportunities for precision medicine. Cancer Sci. 2020;111:2708–2717. doi: 10.1111/cas.14537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Evans RA, Tian YC, Steadman R, Phillips A. O TGF-β1-mediated fibroblast–myofibroblast terminal differentiation-the role of smad proteins. Exp. Cell Res. 2003;282:90–100. doi: 10.1016/S0014-4827(02)00015-0. [DOI] [PubMed] [Google Scholar]
- 142.Yang L, et al. Bone marrow-derived mesenchymal stem cells differentiate to hepatic myofibroblasts by transforming growth factor-β1 via sphingosine kinase/sphingosine 1-phosphate (S1P)/S1P receptor axis. Am. J. Pathol. 2012;181:85–97. doi: 10.1016/j.ajpath.2012.03.014. [DOI] [PubMed] [Google Scholar]
- 143.Jotzu C, et al. Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Anal. Cell Pathol. 2011;34:55–67. doi: 10.1007/s13402-011-0012-1. [DOI] [PubMed] [Google Scholar]
- 144.Calon A, Tauriello DVF, Batlle E. TGF-β in CAF-mediated tumor growth and metastasis. Semin. Cancer Biol. 2014;25:15–22. doi: 10.1016/j.semcancer.2013.12.008. [DOI] [PubMed] [Google Scholar]
- 145.Petersen OW, et al. Epithelial to mesenchymal transition in human breast cancer can provide a nonmalignant stroma. Am. J. Pathol. 2003;162:391–402. doi: 10.1016/S0002-9440(10)63834-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Zeisberg EM, Potenta S, Xie L, Zeisberg M, Kalluri R. Discovery of endothelial to mesenchymal transition as a source for carcinoma-associated fibroblasts. Cancer Res. 2007;67:10123–10128. doi: 10.1158/0008-5472.CAN-07-3127. [DOI] [PubMed] [Google Scholar]
- 147.Rønnov-Jessen L, Petersen OW. Induction of α-smooth muscle actin by transforming growth factor-β1 in quiescent human breast gland fibroblasts. Implications for myofibroblast generation in breast neoplasia. Lab. Invest. 1993;68:696–707. [PubMed] [Google Scholar]
- 148.Postlethwaite AE, Keski-Oja J, Moses HL, Kang AH. Stimulation of the chemotactic migration of human fibroblasts by transforming growth factor β. J. Exp. Med. 1987;165:251–256. doi: 10.1084/jem.165.1.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Olive KP, et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science. 2009;324:1457–1461. doi: 10.1126/science.1171362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Tang Y-A, et al. Hypoxic tumor microenvironment activates GLI2 via HIF-1α and TGF-β2 to promote chemoresistance in colorectal cancer. Proc. Natl Acad. Sci. USA. 2018;115:E5990–E5999. doi: 10.1073/pnas.1801348115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Li D, et al. Radiation promotes epithelial-to-mesenchymal transition and invasion of pancreatic cancer cell by activating carcinoma-associated fibroblasts. Am. J. Cancer Res. 2016;6:2192. [PMC free article] [PubMed] [Google Scholar]
- 152.Huang H, et al. Targeting TGFβR2-mutant tumors exposes vulnerabilities to stromal TGFβ blockade in pancreatic cancer. EMBO Mol. Med. 2019;11:e10515–e10515. doi: 10.15252/emmm.201910515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Chakravarthy A, Khan L, Bensler NP, Bose P, De Carvalho DD. TGF-β-associated extracellular matrix genes link cancer-associated fibroblasts to immune evasion and immunotherapy failure. Nat. Commun. 2018;9:1–10. doi: 10.1038/s41467-017-02088-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Dominguez CX, et al. Single-cell RNA sequencing reveals stromal evolution into LRRC15(+) myofibroblasts as a determinant of patient response to cancer immunotherapy. Cancer Dis. 2020;10:232–253. doi: 10.1158/2159-8290.CD-19-0644. [DOI] [PubMed] [Google Scholar]
- 155.Peske JD, Woods AB, Engelhard VH. Control of CD8 T-cell infiltration into tumors by vasculature and microenvironment. Adv. Cancer Res. 2015;128:263–307. doi: 10.1016/bs.acr.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Swartz MA, Lund AW. Lymphatic and interstitial flow in the tumour microenvironment: linking mechanobiology with immunity. Nat. Rev. Cancer. 2012;12:210–219. doi: 10.1038/nrc3186. [DOI] [PubMed] [Google Scholar]
- 157.Madri JA, Pratt BM, Tucker AM. Phenotypic modulation of endothelial cells by transforming growth factor-β depends upon the composition and organization of the extracellular matrix. J. Cell Biol. 1988;106:1375–1384. doi: 10.1083/jcb.106.4.1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Li GC, et al. Mesenchymal stem cells promote tumor angiogenesis via the action of transforming growth factor β1. Oncol. Lett. 2016;11:1089–1094. doi: 10.3892/ol.2015.3997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lin H, et al. High immunohistochemical expression of TGF-β1 predicts a poor prognosis in cervical cancer patients who harbor enriched endoglin microvessel density. Int. J. Gynecol. Pathol. 2012;31:482–489. doi: 10.1097/PGP.0b013e31824c23a4. [DOI] [PubMed] [Google Scholar]
- 160.Goumans MJ, et al. Balancing the activation state of the endothelium via two distinct TGF-β type I receptors. EMBO J. 2002;21:1743–1753. doi: 10.1093/emboj/21.7.1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Goumans MJ, Ten Dijke P. TGF-β signaling in control of cardiovascular function. Cold Spring Harb. Perspect. Biol. 2018;10:a022210. doi: 10.1101/cshperspect.a022210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Yang F, Strand DW, Rowley DR. Fibroblast growth factor-2 mediates transforming growth factor-β action in prostate cancer reactive stroma. Oncogene. 2008;27:450–459. doi: 10.1038/sj.onc.1210663. [DOI] [PubMed] [Google Scholar]
- 163.Chien W, et al. Expression of connective tissue growth factor (CTGF/CCN2) in breast cancer cells is associated with increased migration and angiogenesis. Int. J. Oncol. 2011;38:1741–1747. doi: 10.3892/ijo.2011.985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Breier G, et al. Transforming growth factor-β and Ras regulate the VEGF/VEGF-receptor system during tumor angiogenesis. Int. J. Mol. Sci. 2002;97:142–148. doi: 10.1002/ijc.1599. [DOI] [PubMed] [Google Scholar]
- 165.Risinger GM, Jr, Updike DL, Bullen EC, Tomasek JJ, Howard EW. TGF-β suppresses the upregulation of MMP-2 by vascular smooth muscle cells in response to PDGF-BB. Am. J. Physiol. Cell Physiol. 2010;298:C191–C201. doi: 10.1152/ajpcell.00417.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Oh SP, et al. Activin receptor-like kinase 1 modulates transforming growth factor-β1 signaling in the regulation of angiogenesis. Proc. Natl Acad. Sci. USA. 2000;97:2626–2631. doi: 10.1073/pnas.97.6.2626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Batlle R, et al. Regulation of tumor angiogenesis and mesenchymal-endothelial transition by p38α through TGF-β and JNK signaling. Nat. Commun. 2019;10:3071. doi: 10.1038/s41467-019-10946-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Kinashi H, Ito Y, Sun T, Katsuno T, Takei Y. Roles of the TGF-β–VEGF-C pathway in fibrosis-related lymphangiogenesis. Int. J. Mol. Sci. 2018;19:2487. doi: 10.3390/ijms19092487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Ginefra P, Lorusso G, Vannini N. Innate immune cells and their contribution to T-cell-based immunotherapy. Int. J. Mol. Sci. 2020;21:4441. doi: 10.3390/ijms21124441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Gonzalez H, Hagerling C, Werb Z. Roles of the immune system in cancer: from tumor initiation to metastatic progression. Genes Dev. 2018;32:1267–1284. doi: 10.1101/gad.314617.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Akhurst RJ, Fee F, Balmaint A. Localized production of TGF-β mRNA in tumour promoter-stimulated mouse epidermis. Nature. 1988;331:363–365. doi: 10.1038/331363a0. [DOI] [PubMed] [Google Scholar]
- 172.Naganuma H, et al. Transforming growth factor-β inhibits interferon-γ secretion by lymphokine-activated killer cells stimulated with tumor cells. Neurol. Med. Chir. 1996;36:789–795. doi: 10.2176/nmc.36.789. [DOI] [PubMed] [Google Scholar]
- 173.Bierie B, et al. Transforming growth factor–β regulates mammary carcinoma cell survival and interaction with the adjacent microenvironment. Cancer Res. 2008;68:1809–1819. doi: 10.1158/0008-5472.CAN-07-5597. [DOI] [PubMed] [Google Scholar]
- 174.Yang L, et al. Abrogation of TGFβ signaling in mammary carcinomas recruits Gr-1+CD11b+ myeloid cells that promote metastasis. Cancer Cell. 2008;13:23–35. doi: 10.1016/j.ccr.2007.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Pang Y, et al. TGF-β signaling in myeloid cells is required for tumor metastasis. Cancer Discov. 2013;3:936–951. doi: 10.1158/2159-8290.CD-12-0527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Novitskiy SV, et al. Deletion of TGF-β signaling in myeloid cells enhances their anti-tumorigenic properties. J. Leukoc. Biol. 2012;92:641–651. doi: 10.1189/jlb.1211639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Lazarova M, Steinle A. Impairment of NKG2D-mediated tumor immunity by TGF-β. Front. Immunol. 2019;10:2689. doi: 10.3389/fimmu.2019.02689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Kobayashi K, et al. IRAK-M is a negative regulator of Toll-like receptor signaling. Cell. 2002;110:191–202. doi: 10.1016/S0092-8674(02)00827-9. [DOI] [PubMed] [Google Scholar]
- 179.Standiford TJ, et al. TGF-β-induced IRAK-M expression in tumor-associated macrophages regulates lung tumor growth. Oncogene. 2011;30:2475–2484. doi: 10.1038/onc.2010.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Jiao S, et al. Differences in tumor microenvironment dictate T helper lineage polarization and response to immune checkpoint therapy. Cell. 2019;179:1177–1190. doi: 10.1016/j.cell.2019.10.029. [DOI] [PubMed] [Google Scholar]
- 181.Liu M, et al. TGF-β suppresses type 2 immunity to cancer. Nature. 2020;587:115–120. doi: 10.1038/s41586-020-2836-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Takeuchi M, Kosiewicz MM, Alard P, Streilein JW. On the mechanisms by which transforming growth factor-β2 alters antigen-presenting abilities of macrophages on T cell activation. Eur. J. Immunol. 1997;27:1648–1656. doi: 10.1002/eji.1830270709. [DOI] [PubMed] [Google Scholar]
- 183.Das L, Levine AD. TGF-β inhibits IL-2 production and promotes cell cycle arrest in TCR-activated effector/memory T cells in the presence of sustained TCR signal transduction. J. Immunol. 2008;180:1490–1498. doi: 10.4049/jimmunol.180.3.1490. [DOI] [PubMed] [Google Scholar]
- 184.McKarns SC, Schwartz RH. Distinct effects of TGF-β1 on CD4+ and CD8+ T cell survival, division, and IL-2 production: a role for T cell intrinsic Smad3. J. Immunol. 2005;174:2071–2083. doi: 10.4049/jimmunol.174.4.2071. [DOI] [PubMed] [Google Scholar]
- 185.Park BV, et al. TGFβ1-mediated SMAD3 enhances PD-1 expression on antigen-specific T cells in cancer. Cancer Discov. 2016;6:1366–1381. doi: 10.1158/2159-8290.CD-15-1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Mishra S, et al. TGF-β and Eomes control the homeostasis of CD8+ regulatory T cells. J. Exp. Med. 2021;218:e20200030. doi: 10.1084/jem.20200030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Sanjabi S, Oh SA, Li MO. Regulation of the immune response by TGF-β: from conception to autoimmunity and infection. Cold Spring Harb. Perspect. Biol. 2017;9:a022236. doi: 10.1101/cshperspect.a022236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Park SR, Seo GY, Choi AJ, Stavnezer J, Kim PH. Analysis of transforming growth factor-β1-induced Ig germ-line gamma2b transcription and its implication for IgA isotype switching. Eur. J. Immunol. 2005;35:946–956. doi: 10.1002/eji.200425848. [DOI] [PubMed] [Google Scholar]
- 189.Tamayo E, Alvarez P, Merino R. TGFβ superfamily members as regulators of b cell development and function—implications for autoimmunity. Int. J. Mol. Sci. 2018;19:3928. doi: 10.3390/ijms19123928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Morris JC, et al. Phase I study of GC1008 (fresolimumab): a human anti-transforming growth factor-β (TGFβ) monoclonal antibody in patients with advanced malignant melanoma or renal cell carcinoma. PLoS ONE. 2014;9:e90353. doi: 10.1371/journal.pone.0090353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Stevenson JP, et al. Immunological effects of the TGFβ-blocking antibody GC1008 in malignant pleural mesothelioma patients. Oncoimmunology. 2013;2:e26218. doi: 10.4161/onci.26218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.den Hollander MW, et al. TGF-β antibody uptake in recurrent high-grade glioma imaged with 89Zr-Fresolimumab PET. J. Nucl. Med. 2015;56:1310–1314. doi: 10.2967/jnumed.115.154401. [DOI] [PubMed] [Google Scholar]
- 193.Stevenson J, et al. Phase II trial of anti-transforming growth factor-β (TGFß) monoclonal antibody GC1008 in relapsed malignant pleural mesothelioma (MPM) J. Clin. Oncol. 2012;30:7077–7077. doi: 10.1200/jco.2012.30.15_suppl.7077. [DOI] [Google Scholar]
- 194.Zhong Z, et al. Anti-transforming growth factor β receptor II antibody has therapeutic efficacy against primary tumor growth and metastasis through multieffects on cancer, stroma, and immune cells. Clin. Cancer Res. 2010;16:1191–1205. doi: 10.1158/1078-0432.CCR-09-1634. [DOI] [PubMed] [Google Scholar]
- 195.Tolcher AW, et al. A phase 1 study of anti-TGFβ receptor type-II monoclonal antibody LY3022859 in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 2017;79:673–680. doi: 10.1007/s00280-017-3245-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Munger JS, Harpel JG, Giancotti FG, Rifkin DB. Interactions between growth factors and integrins: latent forms of transforming growth factor-β are ligands for the integrin αvβ1. Mol. Biol. Cell. 1998;9:2627–2638. doi: 10.1091/mbc.9.9.2627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Ludbrook SB, Barry ST, Delves CJ, Horgan CM. The integrin αvβ3 is a receptor for the latency-associated peptides of transforming growth factors β1 and β3. Biochem. J. 2003;369:311–318. doi: 10.1042/bj20020809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Asano Y, Ihn H, Yamane K, Jinnin M, Tamaki K. Increased expression of integrin αvβ5 induces the myofibroblastic differentiation of dermal fibroblasts. Am. J. Pathol. 2006;168:499–510. doi: 10.2353/ajpath.2006.041306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Munger JS, et al. The integrin αvβ6 binds and activates latent TGF β1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell. 1999;96:319–328. doi: 10.1016/S0092-8674(00)80545-0. [DOI] [PubMed] [Google Scholar]
- 200.Travis MA, Sheppard D. TGF-β activation and function in immunity. Annu. Rev. Immunol. 2014;32:51–82. doi: 10.1146/annurev-immunol-032713-120257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Brown NF, Marshall JF. Integrin-mediated TGFβ activation modulates the tumour microenvironment. Cancers. 2019;11:11091221. doi: 10.3390/cancers11091221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Khan Z, Marshall JF. The role of integrins in TGFβ activation in the tumour stroma. Cell Tissue Res. 2016;365:657–673. doi: 10.1007/s00441-016-2474-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Nolte M, Margadant C. Controlling immunity and inflammation through integrin-dependent regulation of TGF-β. Trends Cell Biol. 2020;30:49–59. doi: 10.1016/j.tcb.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 204.Stockis J, et al. Blocking immunosuppression by human Tregs in vivo with antibodies targeting integrin αVβ8. Proc. Natl Acad. Sci. USA. 2017;114:E10161–e10168. doi: 10.1073/pnas.1710680114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Takasaka N, et al. Integrin αvβ8-expressing tumor cells evade host immunity by regulating TGF-β activation in immune cells. JCI Insight. 2018;3:e122591. doi: 10.1172/jci.insight.122591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Reader CS, et al. The integrin αvβ6 drives pancreatic cancer through diverse mechanisms and represents an effective target for therapy. J. Pathol. 2019;249:332–342. doi: 10.1002/path.5320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Hezel AF, et al. TGF-β and αvβ6 integrin act in a common pathway to suppress pancreatic cancer progression. Cancer Res. 2012;72:4840–4845. doi: 10.1158/0008-5472.CAN-12-0634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Yap TA, et al. AVID200, first-in-class TGF-β 1 and 3 selective and potent inhibitor: safety and biomarker results of a phase I monotherapy dose-escalation study in patients with advanced solid tumors. J. Clin. Oncol. 2020;38:3587–3587. doi: 10.1200/JCO.2020.38.15_suppl.3587. [DOI] [Google Scholar]
- 209.Yap T, et al. P856 AVID200, first-in-class TGF-β1 and β3 selective inhibitor: results of a phase 1 monotherapy dose escalation study in solid tumors and evidence of target engagement in patients. J. Immunother. Cancer. 2020;8:A6–A7. doi: 10.1136/jitc-2020-SITC2020.0006. [DOI] [Google Scholar]
- 210.Yingling JM, et al. Preclinical assessment of galunisertib (LY2157299 monohydrate), a first-in-class transforming growth factor-β receptor type I inhibitor. Oncotarget. 2017;9:6659–6677. doi: 10.18632/oncotarget.23795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Ikeda M, et al. A phase 1b study of transforming growth factor-β receptor I inhibitor galunisertib in combination with sorafenib in Japanese patients with unresectable hepatocellular carcinoma. Invest. N. Drugs. 2019;37:118–126. doi: 10.1007/s10637-018-0636-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Ikeda M, et al. Phase 1b study of galunisertib in combination with gemcitabine in Japanese patients with metastatic or locally advanced pancreatic cancer. Cancer Chemother. Pharmacol. 2017;79:1169–1177. doi: 10.1007/s00280-017-3313-x. [DOI] [PubMed] [Google Scholar]
- 213.Wick A, et al. Phase 1b/2a study of galunisertib, a small molecule inhibitor of transforming growth factor-β receptor I, in combination with standard temozolomide-based radiochemotherapy in patients with newly diagnosed malignant glioma. Invest. N. Drugs. 2020;38:1570–1579. doi: 10.1007/s10637-020-00910-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Fujiwara Y, et al. Phase 1 study of galunisertib, a TGF-β receptor I kinase inhibitor, in Japanese patients with advanced solid tumors. Cancer Chemother. Pharmacol. 2015;76:1143–1152. doi: 10.1007/s00280-015-2895-4. [DOI] [PubMed] [Google Scholar]
- 215.Kelley RK, et al. A phase 2 study of galunisertib (TGF-β1 receptor type I inhibitor) and sorafenib in patients with advanced hepatocellular carcinoma. Clin. Transl. Gastroenterol. 2019;10:e00056. doi: 10.14309/ctg.0000000000000056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Melisi D, et al. Galunisertib plus gemcitabine vs. gemcitabine for first-line treatment of patients with unresectable pancreatic cancer. Br. J. Cancer. 2018;119:1208–1214. doi: 10.1038/s41416-018-0246-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Brandes AA, et al. A phase II randomized study of galunisertib monotherapy or galunisertib plus lomustine compared with lomustine monotherapy in patients with recurrent glioblastoma. Neuro Oncol. 2016;18:1146–1156. doi: 10.1093/neuonc/now009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Kim B-G, et al. Vactosertib, a TGF-β receptor I kinase/ALK5 inhibitor, diminishes tumor progression and bone disease in a mouse model of multiple myeloma and overcomes resistance to proteasome inhibitors. Blood. 2018;132:1918–1918. doi: 10.1182/blood-2018-99-117852. [DOI] [Google Scholar]
- 219.Keedy VL, et al. Association of TGF-β responsive signature with anti-tumor effect of vactosertib, a potent, oral TGF-β receptor type I (TGFBRI) inhibitor in patients with advanced solid tumors. J. Clin. Oncol. 2018;36:3031. doi: 10.1200/JCO.2018.36.15_suppl.3031. [DOI] [Google Scholar]
- 220.Kim HS, et al. A phase I study of TGF-β inhibitor, vactosertib in combination with imatinib in patients with advanced desmoid tumor (aggressive fibromatosis) J. Clin. Oncol. 2020;38:11557–11557. doi: 10.1200/JCO.2020.38.15_suppl.11557. [DOI] [Google Scholar]
- 221.Seufferlein, T. et al. In Translational Pancreatic Cancer Research, Molecular and Translational Medicine (eds Michalski, C. et al.) Ch. 15 (Humana, 2020).
- 222.Jung SY, et al. Pharmacokinetic characteristics of vactosertib, a new activin receptor-like kinase 5 inhibitor, in patients with advanced solid tumors in a first-in-human phase 1 study. Invest. N. Drugs. 2020;38:812–820. doi: 10.1007/s10637-019-00835-y. [DOI] [PubMed] [Google Scholar]
- 223.Kim HS, et al. A phase I study of TGF-β inhibitor, vactosertib in combination with imatinib in patients with advanced desmoid tumor (aggressive fibromatosis) J. Clin. Oncol. 2020;38:e11557. doi: 10.1200/JCO.2020.38.15_suppl.11557. [DOI] [Google Scholar]
- 224.Jung M, et al. Safety and preliminary antitumor activity of the transforming growth factor β (TGF-β) receptor I kinase inhibitor, vactosertib, in combination with paclitaxel in patients with metastatic gastric adenocarcinoma. J. Clin. Oncol. 2020;38:e16505. doi: 10.1200/JCO.2020.38.15_suppl.e16505. [DOI] [Google Scholar]
- 225.Pei H, et al. Abstract 955: LY3200882, a novel, highly selective TGFβRI small molecule inhibitor. Cancer Res. 2017;77:955–955. [Google Scholar]
- 226.Weller M, et al. Effect of the integrin inhibitor cilengitide on TGF-β signaling. J. Clin. Oncol. 2012;30:2055. doi: 10.1200/jco.2012.30.15_suppl.2055. [DOI] [Google Scholar]
- 227.Eskens FA, et al. Phase I and pharmacokinetic study of continuous twice weekly intravenous administration of Cilengitide (EMD 121974), a novel inhibitor of the integrins αvβ3 and αvβ5 in patients with advanced solid tumours. Eur. J. Cancer. 2003;39:917–926. doi: 10.1016/S0959-8049(03)00057-1. [DOI] [PubMed] [Google Scholar]
- 228.O’Donnell PH, et al. A phase I study of continuous infusion cilengitide in patients with solid tumors. Invest. N. Drugs. 2012;30:604–610. doi: 10.1007/s10637-010-9537-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Nabors LB, et al. Phase I and correlative biology study of cilengitide in patients with recurrent malignant glioma. J. Clin. Oncol. 2007;25:1651–1657. doi: 10.1200/JCO.2006.06.6514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Massabeau C, et al. Continuous infusion of cilengitide plus chemoradiotherapy for patients with stage III non-small-cell lung cancer: a phase I study. Clin. Lung Cancer. 2018;19:e277–e285. doi: 10.1016/j.cllc.2017.11.002. [DOI] [PubMed] [Google Scholar]
- 231.Raguse JD, Gath HJ, Bier J, Riess H, Oettle H. Cilengitide (EMD 121974) arrests the growth of a heavily pretreated highly vascularised head and neck tumour. Oral Oncol. 2004;40:228–230. doi: 10.1016/j.oraloncology.2003.08.003. [DOI] [PubMed] [Google Scholar]
- 232.Bradley DA, et al. Cilengitide (EMD 121974, NSC 707544) in asymptomatic metastatic castration resistant prostate cancer patients: a randomized phase II trial by the prostate cancer clinical trials consortium. Invest. N. Drugs. 2011;29:1432–1440. doi: 10.1007/s10637-010-9420-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Alva A, et al. Phase II study of cilengitide (EMD 121974, NSC 707544) in patients with non-metastatic castration resistant prostate cancer, NCI-6735. A study by the DOD/PCF prostate cancer clinical trials consortium. Invest. N. Drugs. 2012;30:749–757. doi: 10.1007/s10637-010-9573-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Stupp R, et al. Phase I/IIa study of cilengitide and temozolomide with concomitant radiotherapy followed by cilengitide and temozolomide maintenance therapy in patients with newly diagnosed glioblastoma. J. Clin. Oncol. 2010;28:2712–2718. doi: 10.1200/JCO.2009.26.6650. [DOI] [PubMed] [Google Scholar]
- 235.Vermorken JB, et al. Cisplatin, 5-fluorouracil, and cetuximab (PFE) with or without cilengitide in recurrent/metastatic squamous cell carcinoma of the head and neck: results of the randomized phase I/II ADVANTAGE trial (phase II part) Ann. Oncol. 2014;25:682–688. doi: 10.1093/annonc/mdu003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Vansteenkiste J, et al. Cilengitide combined with cetuximab and platinum-based chemotherapy as first-line treatment in advanced non-small-cell lung cancer (NSCLC) patients: results of an open-label, randomized, controlled phase II study (CERTO) Ann. Oncol. 2015;26:1734–1740. doi: 10.1093/annonc/mdv219. [DOI] [PubMed] [Google Scholar]
- 237.Kim KB, et al. A randomized phase II study of cilengitide (EMD 121974) in patients with metastatic melanoma. Melanoma Res. 2012;22:294–301. doi: 10.1097/CMR.0b013e32835312e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Stupp R, et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071-22072 study): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2014;15:1100–1108. doi: 10.1016/S1470-2045(14)70379-1. [DOI] [PubMed] [Google Scholar]
- 239.Juliano RL. Addressing cancer signal transduction pathways with antisense and siRNA oligonucleotides. NAR Cancer. 2020;2:zcaa025. doi: 10.1093/narcan/zcaa025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Schlingensiepen KH, et al. Targeted tumor therapy with the TGF-β2 antisense compound AP 12009. Cytokine Growth Factor Rev. 2006;17:129–139. doi: 10.1016/j.cytogfr.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 241.Oettle H, et al. Phase I/II study with trabedersen (AP 12009) monotherapy for the treatment of patients with advanced pancreatic cancer, malignant melanoma, and colorectal carcinoma. J. Clin. Oncol. 2011;29:2513–2513. doi: 10.1200/jco.2011.29.15_suppl.2513. [DOI] [Google Scholar]
- 242.Bogdahn U, et al. Randomized, active-controlled phase IIb study with trabedersen (AP 12009) in recurrent or refractory high-grade glioma patients: Basis for phase III endpoints. J. Clin. Oncol. 2009;27:2037–2037. doi: 10.1200/jco.2009.27.15_suppl.2037. [DOI] [Google Scholar]
- 243.Hwang L, Ng K, Wang W, Trieu V. Treatment with trabedersen, an anti-TGF-β2 antisense, primed tumors to subsequent chemotherapies. Cancer Res. 2016;76:3742. [Google Scholar]
- 244.Vallières L. Trabedersen, a TGFβ2-specific antisense oligonucleotide for the treatment of malignant gliomas and other tumors overexpressing TGFβ2. IDrugs. 2009;12:445–453. [PubMed] [Google Scholar]
- 245.Bogdahn U, et al. Targeted therapy for high-grade glioma with the TGF-β2 inhibitor trabedersen: results of a randomized and controlled phase IIb study. Neuro Oncol. 2011;13:132–142. doi: 10.1093/neuonc/noq142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Nemunaitis J, et al. Phase II study of belagenpumatucel-L, a transforming growth factor β-2 antisense gene-modified allogeneic tumor cell vaccine in non-small-cell lung cancer. J. Clin. Oncol. 2006;24:4721–4730. doi: 10.1200/JCO.2005.05.5335. [DOI] [PubMed] [Google Scholar]
- 247.Nemunaitis J, et al. Phase II trial of belagenpumatucel-L, a TGF-β2 antisense gene modified allogeneic tumor vaccine in advanced non small cell lung cancer (NSCLC) patients. Cancer Gene Ther. 2009;16:620–624. doi: 10.1038/cgt.2009.15. [DOI] [PubMed] [Google Scholar]
- 248.Giaccone G, et al. A phase III study of belagenpumatucel-L, an allogeneic tumour cell vaccine, as maintenance therapy for non-small cell lung cancer. Eur. J. Cancer. 2015;51:2321–23295. doi: 10.1016/j.ejca.2015.07.035. [DOI] [PubMed] [Google Scholar]
- 249.Rodon J, et al. First-in-human dose study of the novel transforming growth factor-β receptor I kinase inhibitor LY2157299 monohydrate in patients with advanced cancer and glioma. Clin. Cancer Res. 2015;21:553–560. doi: 10.1158/1078-0432.CCR-14-1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Fessler E, et al. TGFβ signaling directs serrated adenomas to the mesenchymal colorectal cancer subtype. EMBO Mol. Med. 2016;8:745–760. doi: 10.15252/emmm.201606184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Liu Q, et al. Genetic, epigenetic, and molecular landscapes of multifocal and multicentric glioblastoma. Acta Neuropathol. 2015;130:587–597. doi: 10.1007/s00401-015-1470-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Collisson EA, et al. Subtypes of pancreatic ductal adenocarcinoma and their differing responses to therapy. Nat. Med. 2011;17:500–503. doi: 10.1038/nm.2344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Lehmann BD, et al. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Invest. 2011;121:2750–2767. doi: 10.1172/JCI45014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Yang D, et al. Integrated analyses identify a master microRNA regulatory network for the mesenchymal subtype in serous ovarian cancer. Cancer Cell. 2013;23:186–199. doi: 10.1016/j.ccr.2012.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Kim HyunS, et al. Systematic identification of molecular subtype-selective vulnerabilities in non-small-cell lung cancer. Cell. 2013;155:552–566. doi: 10.1016/j.cell.2013.09.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Chae YK, et al. Path toward precision oncology: review of targeted therapy studies and tools to aid in defining “actionability” of a molecular lesion and patient management support. Mol. Cancer Ther. 2017;16:2645–2655. doi: 10.1158/1535-7163.MCT-17-0597. [DOI] [PubMed] [Google Scholar]
- 257.Pottier C, et al. Tyrosine kinase inhibitors in cancer: breakthrough and challenges of targeted therapy. Cancers. 2020;12:731. doi: 10.3390/cancers12030731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Mansoori B, Mohammadi A, Davudian S, Shirjang S, Baradaran B. The different mechanisms of cancer drug resistance: a brief review. Adv. Pharm. Bull. 2017;7:339. doi: 10.15171/apb.2017.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Bhola NE, et al. TGF-β inhibition enhances chemotherapy action against triple-negative breast cancer. J. Clin. Invest. 2013;123:1348–1358. doi: 10.1172/JCI65416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Capper D, et al. Biomarker and histopathology evaluation of patients with recurrent glioblastoma treated with galunisertib, lomustine, or the combination of galunisertib and lomustine. Int. J. Mol. Sci. 2017;18:995. doi: 10.3390/ijms18050995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Ungerleider N, Han C, Zhang J, Yao L, Wu T. TGFβ signaling confers sorafenib resistance via induction of multiple RTKs in hepatocellular carcinoma cells. Mol. Carcinog. 2017;56:1302–1311. doi: 10.1002/mc.22592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Dodagatta-Marri E, et al. α-PD-1 therapy elevates Treg/Th balance and increases tumor cell pSmad3 that are both targeted by α-TGFβ antibody to promote durable rejection and immunity in squamous cell carcinomas. J. Immunother. Cancer. 2019;7:62. doi: 10.1186/s40425-018-0493-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Huang S, et al. MED12 controls the response to multiple cancer drugs through regulation of TGF-β receptor signaling. Cell. 2012;151:937–950. doi: 10.1016/j.cell.2012.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Hardee ME, et al. Resistance of glioblastoma-initiating cells to radiation mediated by the tumor microenvironment can be abolished by inhibiting transforming growth factor-β. Cancer Res. 2012;72:4119–4129. doi: 10.1158/0008-5472.CAN-12-0546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Martin M, et al. Coactivation of AP-1 activity and TGF-β1 gene expression in the stress response of normal skin cells to ionizing radiation. Oncogene. 1997;15:981–989. doi: 10.1038/sj.onc.1201433. [DOI] [PubMed] [Google Scholar]
- 266.Andarawewa KL, Paupert J, Pal A, Barcellos-Hoff MH. New rationales for using TGFβ inhibitors in radiotherapy. Int. J. Radiat. Biol. 2007;83:803–811. doi: 10.1080/09553000701711063. [DOI] [PubMed] [Google Scholar]
- 267.Anscher MS. Targeting the TGF-β1 pathway to prevent normal tissue injury after cancer therapy. Oncologist. 2010;15:350–359. doi: 10.1634/theoncologist.2009-S101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Formenti SC, et al. Focal irradiation and systemic TGFβ blockade in metastatic breast cancer. Clin. Cancer Res. 2018;24:2493–2504. doi: 10.1158/1078-0432.CCR-17-3322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Formenti SC, et al. Baseline T cell dysfunction by single cell network profiling in metastatic breast cancer patients. J. Immunother. Cancer. 2019;7:177. doi: 10.1186/s40425-019-0633-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020;30:660–669. doi: 10.1038/s41422-020-0343-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Chen L, Flies DB. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat. Rev. Immunol. 2013;13:227–242. doi: 10.1038/nri3405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer. 2012;12:252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Leach DR, Krummel MF, Allison JP. Enhancement of antitumor immunity by CTLA-4 blockade. Science. 1996;271:1734–1736. doi: 10.1126/science.271.5256.1734. [DOI] [PubMed] [Google Scholar]
- 274.Herbst RS, et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature. 2014;515:563–567. doi: 10.1038/nature14011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Tauriello DVF, et al. TGFβ drives immune evasion in genetically reconstituted colon cancer metastasis. Nature. 2018;554:538–543. doi: 10.1038/nature25492. [DOI] [PubMed] [Google Scholar]
- 276.Mariathasan S, et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature. 2018;554:544–548. doi: 10.1038/nature25501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Hartley J, Abken H. Chimeric antigen receptors designed to overcome transforming growth factor‐β‐mediated repression in the adoptive T‐cell therapy of solid tumors. Clin. Transl. Immunol. 2019;8:e1064. doi: 10.1002/cti2.1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Ahn M-J, et al. Randomized open-label study of M7824 versus pembrolizumab as first-line (1L) treatment in patients with PD-L1 expressing advanced non-small cell lung cancer (NSCLC) J. Clin. Oncol. 2019;37:TPS9114. doi: 10.1200/JCO.2019.37.15_suppl.TPS9114. [DOI] [Google Scholar]
- 279.Ravi R, et al. Bifunctional immune checkpoint-targeted antibody-ligand traps that simultaneously disable TGFβ enhance the efficacy of cancer immunotherapy. Nat. Commun. 2018;9:741. doi: 10.1038/s41467-017-02696-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.O’Connor-McCourt MD, et al. Abstract 4688: AVID200: a novel computationally-designed TGF β trap promoting anti-tumor T cell activity. Cancer Res. 2017;77:4688–4688. [Google Scholar]
- 281.Li S, et al. Cancer immunotherapy via targeted TGF-β signalling blockade in TH cells. Nature. 2020;587:121–125. doi: 10.1038/s41586-020-2850-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Martin CJ, et al. Selective inhibition of TGFβ1 activation overcomes primary resistance to checkpoint blockade therapy by altering tumor immune landscape. Sci. Transl. Med. 2020;12:eaay8456. doi: 10.1126/scitranslmed.aay8456. [DOI] [PubMed] [Google Scholar]
- 283.Wang Y, et al. Co-inhibition of the TGF-β pathway and the PD-L1 checkpoint by pH-responsive clustered nanoparticles for pancreatic cancer microenvironment regulation and anti-tumor immunotherapy. Biomater. Sci. 2020;8:5121–5132. doi: 10.1039/D0BM00916D. [DOI] [PubMed] [Google Scholar]
- 284.Sow HS, Ren J, Camps M, Ossendorp F, Ten Dijke P. Combined inhibition of TGF-β signaling and the PD-L1 immune checkpoint is differentially effective in tumor models. Cells. 2019;8:320. doi: 10.3390/cells8040320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Han, J.-Y. et al. Safety and anti-tumor activity of the transforming growth factor β receptor I kinase inhibitor, vactosertib, in combination with durvalumab in patients with advanced non-small cell lung cancer (NSCLC). J. Immunother. Cancer7 (2019).
- 286.Lee, K.-W. et al. Safety and anti-tumor activity of the transforming growth factor β receptor I kinase inhibitor, vactosertib, in combination with pembrolizumab in patients with metastatic colorectal or gastric cancer. J. Immunother. Cancer7 (2019).
- 287.Gregory RC, et al. The anti-TGFβ neutralizing antibody, SAR439459, blocks the immunosuppressive effects of TGFβ and inhibits the growth of syngeneic tumors in combination with anti-PD1. Cancer Res. 2018;78:2790. [Google Scholar]
- 288.Lan Y, et al. Preclinical evaluation and mechanistic characterization of M7824 (MSB0011359C), a novel bifunctional fusion protein targeting the PD-L1 and TGFβ pathways. Cancer Res. 2017;77:2615. [Google Scholar]
- 289.Lan Y, et al. Enhanced preclinical antitumor activity of M7824, a bifunctional fusion protein simultaneously targeting PD-L1 and TGF-β. Sci. Transl. Med. 2018;10:eaan5488. doi: 10.1126/scitranslmed.aan5488. [DOI] [PubMed] [Google Scholar]
- 290.Strauss J, et al. Phase I trial of M7824 (MSB0011359C), a bifunctional fusion protein targeting PD-L1 and TGFβ, in advanced solid tumors. Clin. Cancer Res. 2018;24:1287–1295. doi: 10.1158/1078-0432.CCR-17-2653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Strauss J, et al. Phase I evaluation of M7824, a bifunctional fusion protein targeting TGF-β and PD-L1, in patients with human papillomavirus (HPV)-associated malignancies. Cancer Res. 2019;79:CT075. doi: 10.1136/jitc-2020-001395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Cho BC, et al. Bintrafusp α, a bifunctional fusion protein targeting TGF-β and PD-L1, in advanced squamous cell carcinoma of the head and neck: results from a phase I cohort. J. Immunother. Cancer. 2020;8:410. doi: 10.1136/jitc-2020-SITC2020.0385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Kopetz S, et al. M7824 (MSB0011359C), a bifunctional fusion protein targeting PD-L1 and TGF-β, in patients with heavily pretreated CRC: Preliminary results from a phase I trial. J. Clin. Oncol. 2018;36:377. doi: 10.1200/JCO.2018.36.4_suppl.764. [DOI] [Google Scholar]
- 294.Khasraw M, et al. Phase 1 study of M7824, a bifunctional fusion protein targeting TGF-β and PD-L1, among patients with recurrent glioblastoma. Neuro Oncol. 2018;20:vi4. doi: 10.1093/neuonc/noy148.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Allan S, et al. P37. Phase 1 evaluation of bintrafusp α (M7824), a bifunctional fusion protein targeting TGF-β and PD-L1, in cervical cancer. Int. J. Gynecol. Cancer. 2019;29:A72–A73. [Google Scholar]
- 296.Paz-Ares LG, et al. Results from a second-line (2L) NSCLC cohort treated with M7824 (MSB0011359C), a bifunctional fusion protein targeting TGF-β and PD-L1. J. Clin. Oncol. 2018;36:A72–A73. [Google Scholar]
- 297.Borad M, et al. P-20. Phase 2, open-label study of second-line M7824 treatment in patients with locally advanced or metastatic biliary tract cancer. Hepatobiliary Surg. Nutr. 2019;8:AB052. doi: 10.21037/hbsn.2019.AB052. [DOI] [Google Scholar]
- 298.Yoo C, et al. Phase I study of bintrafusp alfa, a bifunctional fusion protein targeting TGF-β and PD-L1, in patients with pretreated biliary tract cancer. J. Immunother. Cancer. 2020;8:e000564. doi: 10.1136/jitc-2020-000564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Liu T, et al. Cancer-associated fibroblasts: an emerging target of anti-cancer immunotherapy. J. Hematol. Oncol. 2019;12:86–86. doi: 10.1186/s13045-019-0770-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.de Streel G, et al. Selective inhibition of TGF-β1 produced by GARP-expressing Tregs overcomes resistance to PD-1/PD-L1 blockade in cancer. Nat. Commun. 2020;11:4545. doi: 10.1038/s41467-020-17811-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Hong M, Clubb JD, Chen YY. Engineering CAR-T cells for next-generation cancer therapy. Cancer Cell. 2020;38:473–788. doi: 10.1016/j.ccell.2020.07.005. [DOI] [PubMed] [Google Scholar]
- 302.Wang Z, et al. Galunisertib enhances chimeric antigen receptor-modified T cell function. Eur. J. Histochem. 2020;64:3122. doi: 10.4081/ejh.2020.3122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Tang N, et al. TGF-β inhibition via CRISPR promotes the long-term efficacy of CAR T cells against solid tumors. JCI Insight. 2020;5:e133977. doi: 10.1172/jci.insight.133977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Kloss CC, et al. Dominant-negative TGF-β receptor enhances PSMA-targeted human CAR T cell proliferation and augments prostate cancer eradication. Mol. Ther. 2018;26:1855–1866. doi: 10.1016/j.ymthe.2018.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Roth TL, et al. Pooled knockin targeting for genome engineering of cellular immunotherapies. Cell. 2020;181:728–744. doi: 10.1016/j.cell.2020.03.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Chang ZL, et al. Rewiring T-cell responses to soluble factors with chimeric antigen receptors. Nat. Chem. Biol. 2018;14:317–324. doi: 10.1038/nchembio.2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Sun J, et al. T cells expressing constitutively active Akt resist multiple tumor-associated inhibitory mechanisms. Mol. Ther. 2010;18:2006–2017. doi: 10.1038/mt.2010.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Disis ML. Enhancing cancer vaccine efficacy via modulation of the tumor microenvironment. Clin. Cancer Res. 2009;15:6476–6478. doi: 10.1158/1078-0432.CCR-09-2256. [DOI] [PubMed] [Google Scholar]
- 309.Ardiani A, et al. Vaccine-mediated immunotherapy directed against a transcription factor driving the metastatic process. Cancer Res. 2014;74:1945–1957. doi: 10.1158/0008-5472.CAN-13-2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Knudson KM, et al. M7824, a novel bifunctional anti-PD-L1/TGFβ Trap fusion protein, promotes anti-tumor efficacy as monotherapy and in combination with vaccine. Oncoimmunology. 2018;7:e1426519. doi: 10.1080/2162402X.2018.1426519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Redman JM, Steinberg SM, Gulley JL. A sequential cohort study of combination immunotherapy with BN-brachyury vaccine, M7824, ALT-803 and epacadostat in metastatic castration-resistant prostate cancer (mCRPC) (QuEST1) J. Clin. Oncol. 2018;36:TPS3130. doi: 10.1200/JCO.2018.36.15_suppl.TPS3130. [DOI] [Google Scholar]
- 312.Gatti-Mays ME, et al. A phase Ib trial of sequential combinations of BN-brachyury, entinostat, ado-trastuzumab emtansine (T-DM1) and bintrafusp α (M7824) in advanced stage breast cancer (BrEAsT) Cancer Res. 2020;80:OT1-08-01. [Google Scholar]
- 313.de Larco JE, Todaro GJ. Growth factors from murine sarcoma virus-transformed cells. Proc. Natl Acad. Sci. USA. 1978;75:4001–4005. doi: 10.1073/pnas.75.8.4001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Roberts AB, Anzano MA, Lamb LC, Smith JM, Sporn MB. New class of transforming growth factors potentiated by epidermal growth factor: isolation from non-neoplastic tissues. Proc. Natl Acad. Sci. USA. 1981;78:5339–5343. doi: 10.1073/pnas.78.9.5339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Shipley GD, Tucker RF, Moses HL. Type β transforming growth factor/growth inhibitor stimulates entry of monolayer cultures of AKR-2B cells into S phase after a prolonged prereplicative interval. Proc. Natl Acad. Sci. USA. 1985;82:4147–4151. doi: 10.1073/pnas.82.12.4147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Derynck R, Budi EHJSs. Specificity, versatility, and control of TGF-β family signaling. Sci. Signal. 2019;12:eaav5183. doi: 10.1126/scisignal.aav5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Lin HY, Wang XF, Ng-Eaton E, Weinberg RA, Lodish HF. Expression cloning of the TGF-β type II receptor, a functional transmembrane serine/threonine kinase. Cell. 1992;68:775–785. doi: 10.1016/0092-8674(92)90152-3. [DOI] [PubMed] [Google Scholar]
- 318.Franzén P, et al. Cloning of a TGF β type I receptor that forms a heteromeric complex with the TGF β type II receptor. Cell. 1993;75:681–692. doi: 10.1016/0092-8674(93)90489-D. [DOI] [PubMed] [Google Scholar]
- 319.Singh J, et al. Transforming the TGFβ pathway: convergence of distinct lead generation strategies on a novel kinase pharmacophore for TβRI (ALK5) Curr. Opin. Drug Discov. Dev. 2004;7:437–445. [PubMed] [Google Scholar]
- 320.Callahan JF, et al. Identification of novel inhibitors of the transforming growth factor β1 (TGF-β1) type 1 receptor (ALK5) J. Med. Chem. 2002;45:999–1001. doi: 10.1021/jm010493y. [DOI] [PubMed] [Google Scholar]
- 321.Kovacs RJ, et al. Cardiac safety of TGF-β receptor I kinase inhibitor LY2157299 monohydrate in cancer patients in a first-in-human dose study. Cardiovasc. Toxicol. 2015;15:309–323. doi: 10.1007/s12012-014-9297-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Rodón J, et al. Pharmacokinetic, pharmacodynamic and biomarker evaluation of transforming growth factor-β receptor I kinase inhibitor, galunisertib, in phase 1 study in patients with advanced cancer. Invest. N. Drugs. 2015;33:357–370. doi: 10.1007/s10637-014-0192-4. [DOI] [PMC free article] [PubMed] [Google Scholar]