Abstract
Background: The distribution and connection of ventricular Purkinje fibers are known to be associated with idiopathic left ventricular arrhythmias. Unusual anatomy is one of the important factors associated with catheter ablation success rate. With the widefield high-speed, swept-source optical coherence microscopy (OCM) and light microscope, we visualized the left ventricular Purkinje fiber distribution. Methods: Left ventricular walls of five adult ovine hearts were incised from the mitral annulus to the apex. Using the widefield OCM technique and light microscopy, we observed the distribution, direction, depth, and dividing patterns of the Purkinje network with multiple tangential angles and without tissue destruction. Results: Widefield OCM was used to characterize the ovine heart Purkinje network system in a 4 × 4 mm2 field. Left ventricular Purkinje fibers traveled in the sub-endocardial area near the left-sided peri-membranous septal area and ran like a wide hair bundle. The distal branching fibers penetrated to the endocardium and connected to the contractile muscle. In this distal area, Purkinje fibers were connected to each other, forming multiple layers. Some Purkinje fibers were directly connected within the false tendon between the papillary muscles or between the trabeculations. Some free-running Purkinje fibers were directly connected to the papillary muscle from the left bundle. Conclusion: Using widefield OCM, we were able to observe the left bundle and its branching patterns in ovine left ventricle without tissue destruction. This might be applied to future cardiac ablation procedures.
Keywords: Purkinje fibers, optical microscopy, heart conduction system, cardiac arrhythmia
Introduction
The His-Purkinje system (HPS) is responsible for the rapid and efficient electrical conduction of the ventricles [1]. Cardiac Purkinje cells form a complex and dense network which cover the ventricular myocardium [2]. Continuous efforts have been made to visualize the structural and functional capacity of the HPS using optical mapping and dye injections, and the distribution of the HPS has been largely characterized due to its results [3,4]. Historically, techniques of injecting dye into the sheath or immunoenzyme labelling have been widely used, and this method can show the running distribution of the ventricular conduction system [5]. However, observations from light microscopy using various staining methods were not sufficient to visualize the entire system [3]. For instance, the sheath of the Purkinje fibers in humans is excessively thin and cannot easily tolerate the increasing perfusion pressure [6]. Imaging techniques such as micro computed tomography or bioprinting have been tried using a conduction system, but spatial resolution was limited [7,8]. Because the ungulate’s Purkinje fibers are piled up with thick sheaths, visualization is relatively easier than human HPS. Therefore the HPS was first discovered in the sheep heart [9].
This complex network has been known to be related to the mechanism of ventricular tachyarrhythmias [10,11]. The anatomic variation of the HPS and its relationship with papillary muscles have been associated with increased incidence of ventricular tachyarrhythmias [12,13]. Until now, there has been no way to observe the distribution of the HPS during catheter ablation for ventricular tachyarrhythmias.
Optical coherence microscopy (OCM) is an echo-based noninvasive non-destructive imaging modality that measures the time-of-flight of back-reflected light using low-coherence interferometry [14]. With its high spatial resolution, OCM is good for observing the HPS traveling in the subendocardial area of the shallow layer. In this study, we introduced the novel widefield high-speed, swept source OCM technique that allows easy and precise observation of the HPS located in the subendocardium. Unlike the traditional OCM equipment, this new OCM modality is suitable for observing widely traveling tissues such as HPS because it can capture a wide area with high spatial resolution from multiple different angles without tissue destruction. In this study, we visualized ovine HPS with the new OCM modality.
Using this new widefield OCM modality, we observed ovine Purkinje distribution and its anatomic relationship with ventricular papillary muscles.
Methods
Sheep heart preparation
Sheep hearts were commercially obtained from the slaughterhouse (Choongju, South Korea). IACUC (Institutional Animal Care and Use Committee) approval was not required when all procedures are being carried out on tissues obtained from a slaughterhouse, The researchers did not participate in the process of animal slaughter.
Five frozen adult sheep hearts were thawed in the laboratory for more than 12 hours at room temperature of 25°C. Thawed hearts were fixed in formalin solution for 48 h after gross anatomic evaluation. The lateral wall of the left ventricle was linearly incised from the mitral annulus to the apex so that the left ventricular endocardial wall was exposed between two papillary muscles. Then, the middle of the anterior mitral leaflet and the aortic annulus area between the left and non-coronary cusps of the aortic valve was incised so that the left-sided membranous septum was fully exposed (Supplementary Figure 1). Left ventricle surfaces were divided into 14 regions based on traditional 16 segments. Each segment was numbered sequentially from the posteromedial side. The left ventricular wall was cut out with 3 mm depth and observed by OCM (Supplementary Figure 2).
Optical coherence microscopy
A schematic of the built-in-house OCM system is shown in Figure 1. Light from a wavelength-swept source (HSL-20, Santec, repetition rate = 100 kHz; central wavelength = 1310 nm; bandwidth = 100 nm) was divided by a fiber coupler into reference and sample. Reflected light from the reference mirror was recombined with reflected light from the sample at the fiber coupler, and finally the interference signal was collected by a balanced photo detector (PDB470C, Thorlabs) which converts the light into an analogue electrical signal. Then the analogue electrical signal was converted to digital signal by a digitizer (APX-5050, AVALDATA). An objective lens (LSM03, Thorlabs; NA = 0.036) was used to focus the light on the specimen. The OCM system had lateral resolution of 7.8 μm and axial resolution of 11.4 μm in tissue. The sensitivity of the system is measured to be 105 dB. Field of view acquired by galvanometer scanning was 4 mm × 4 mm. Wide field of view images up to 20 mm × 20 mm were generated by stitching the multiple galvanometer scanning fields of view images, which could give a fast measurement time of about 12 seconds but a small measurement area limited by the small scanning angle of the galvanometer. To make a larger measurement area up to 20 mm × 20 mm, the widefield OCM method was used by moving the motorized two-axis translation sample stage. A total of 25 moving steps of the sample stage could achieve 20 mm × 20 mm measurement area. Supplementary Figure 3 shows photography of a frontal view and modules of the OCM system. Since OCM measures intensity of the scattering signal from the tissue, in the OCM image, white color corresponds to the highest scattering signal and black color to the lowest scattering signal from the tissue.
Figure 1.
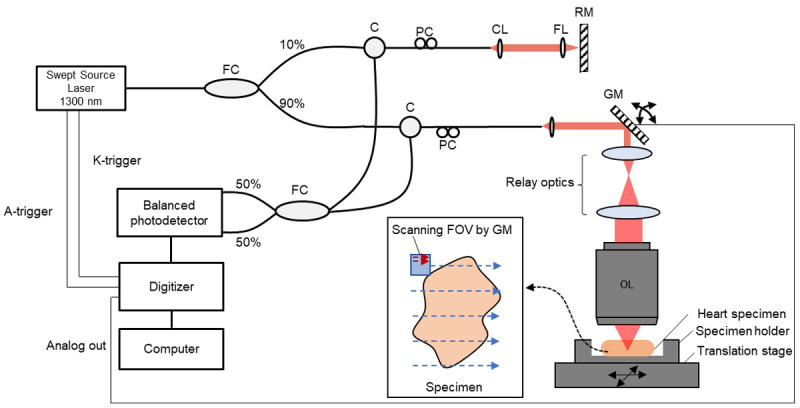
OCM system. This new optical coherence microscopy had lateral resolution of 7.8 μm and axial resolution of 11.4 μm in tissue. Field of view (FOV) from galvanometer scanning was 4 mm × 4 mm and wide FOV images up to 20 mm × 20 mm were generated by stitching and reconstructing the multiple galvanometer scanning FOV images acquired by moving the two-axis translation stage. FC, fiber coupler. C, circulator. PC, polarization controller. CL, collimation lens. GM, Galvanometer. RM, reference mirror. FL, focusing lens. OL, objective lens.
Light microscopic histology
The distribution of Purkinje fibers was observed by light microscopy after sectioning the myocardial tissue for OCM. The myocardial tissue was embedded en face such that the surface of the septal wall was sectioned first and then step sections were made at 4 μm intervals from the surface to the depth. Unstained sections were examined and then some of them were selected and stained by hematoxylin and eosin. Additional stains of Masson’s trichrome and reticulin were applied to reveal histologic details. Histologic sections were scanned by Aperio CS2 (Leica Biosystems Nussloch, Germany) and examined by Aperio Imagescope (leica Biosystems Nussloch, Germany). Semi-macroscopic distribution of Purkinje fibers and their histologic details were analyzed and compared with the OCM data.
Results
Gross anatomy of sheep hearts
The gross anatomy of the sheep heart was basically similar to that of the human heart although the inferior vena cava was longer and the aorta branched to the left and right. The two groups of papillary muscles were leaning to the wall without sub-papillary intertrabecular spaces. There was a muscular wall continuing to the subaortic part of the left ventricle between the right coronary cusp and the non-coronary cusp where a membranous septum was expected in a human heart. The whitish sheath traveled widely and was divided into several complicated divisions. Some of the narrow sheath bundles were free-running over the surface, not attaching the subendocardium, and met directly with the papillary muscle. Most bundles ran on the endomyocardial surface and traversed the crest of the trabeculation, and the valleys had no whitish membranous sheaths.
Optical coherence microscopic imaging of Purkinje system
By comparing images from OCM and standard light microscopy, the whitish sheaths were confirmed to be enclosing sheaths of Purkinje fibers (Figure 2). With OCM, Purkinje fibers were seen as low density and reticular sheath was observed as high density. Although the nucleus was not definitely distinguishable, OCM was enough to observe the distribution and running direction of HPS.
Figure 2.

OCM and conventional light microscopic observations of HPS in the same spot. A. OCM image. Purkinje fibers can be seen as low density and reticular sheath is observed as high density. It is suitable for observing the running direction of fibers in a wide area. B. Light microscope image. Purkinje fibers stained by hematoxylin and eosin have fewer myofibrils compared to other nearby myocardial tissues, have glycogen around the nucleus, and are larger and longer than cardiac myocytes.
Serial images were obtained in 5 μm increments from the surface without destruction of the tissue. The serial images of one tissue block are presented in Figure 3. As the depth of observation becomes deeper, the field becomes darker as the transmission of light decreases. HPS was located within 20 μm (base)~50 μm (apex) from the surface. The sites to be observed could be observed at different angle. Supplementary Figure 4 is an OCM image of the same site taken at different angles.
Figure 3.

Serial optical coherence microscopic images. With optical coherence microscopy, the region of interest can be observed at various desired depths without tissue destruction.
Subendocardial distribution of Purkinje fibers
The left bundle, which was separated from the His bundle, runs in the apical direction like a waterfall emerging from the myocardial peri-membranous septal wall (Figure 4A). The direction of this bundle is different from that of working myocardium in the endocardial area (Supplementary Figure 5). In the bundle (Figure 4B), each fiber splits into a thinner fascicle according to its destination (Supplementary Figure 6). Some of them are free-running and lead directly to papillary muscles (Figure 5).
Figure 4.

OCM images of left bundle branch and Purkinje bundles. A. Left bundle branch. The left bundle runs in the apical direction like a waterfall emerging from the myocardial peri-membranous septal wall. B. Bundle of Purkinje fibers. The direction of this bundle is different from that of the working myocardium in the endocardial area. Each fiber splits into a thinner fascicle according to its destination.
Figure 5.

Free-running Purkinje fibers. A. Gross findings. B. OCM findings. C. Histology. Some Purkinje bundles are free running without attaching with ventricular surface or directly connect between trabeculae and the papillary muscle. The Purkinje fibers are seen at most of the false tendon at the apical part of the septum. Only Purkinje cells without cardiac myocytes are in these false tendons.
The Purkinje fibers, which travel to the distal free wall or apex, move away from the base, and then either reverse direction or reach the destination and connect with myocardium. Besides running in the subendocardial area piled on the sheath, free running Purkinje fibers could be found within the false tendon connecting tissues. This enables efficient transmission of electrical signals to distant areas by using a fast-running free running structure without being affected by cardiac contraction. Especially, the Purkinje fibers inside the false tendon connected to the papillary muscles can be observed.
Observing the apical area connected to myocardium showed a more fractal appearance than the connected structure of the proximal part (Supplementary Figure 7). This might be because the Purkinje fibers in the distal area end in connection with the multiple working cardiomyocytes. Figure 6 is a schematic diagram of left ventricular Purkinje system we could observe with OCM.
Figure 6.

Schematic diagram of left ventricular Purkinje system. A. The left bundle branch is divided into two or four major fascicles. B. Main fascicles are divided into branches, but some branches meet again. C. Some fibers in the main fascicle are separated from the subendocardial area and connect directly to the papillary muscle. D. There are some free-running fibers connecting branched fascicles. E. Free-running fibers also branch or meet each other.
Histologic findings of the Purkinje system from light microscopy
The Purkinje fibers form an interlacing network from the beginning until their transition into the myocardial cells. They are surrounded by loose connective and fatty tissue at the proximal end but lose their covering sheath when transitioning into myocardial cells.
The Purkinje system starts at the level of the His bundle and is divided into the anterior, middle, and posterior fascicles at the basal septum. The largest bundles become medium- or small-sized bundles on their way to the mid- or apical septum. At the basal septum, Purkinje cells are seen as a large group of fibers. while they gradually become medium- to small-sized by the mid and apical septum.
The cardiac Purkinje cells run on the crest of the trabeculae, but the basal part of the groove between trabeculae is free of the Purkinje cells or less populated. Free-standing bundles of Purkinje fibers were observed at the false tendon as seen by OCM (Figure 5C). The most distal Purkinje cells merged with the myocardial cells predominantly at the mid- and apical myocardium but this transition was also found at the basal part.
Discussion
In this study, we could observe the running distribution and direction of Purkinje network system in the left ventricle using a widefield OCM technology. The ovine left ventricular Purkinje fibers were 1) traveling in the subendocardial area, 2) some free running fibers were directly connected to papillary muscles from left bundle branch, and 3) distally, Purkinje fibers become shorter and branched, forming multiple layers.
Although it is difficult to apply this study directly to the analysis of mechanism of human arrhythmia,s, we were able to get new information and ideas using the new imaging technique. Free-running fibers have the advantage of delivering electrical signal from the top to a remote location in the shortest time. Previous gross anatomy studies identified free-running fibers in 55% to 62% of human hearts, of which 60% carried conduction tissue fibers [15]. Previous studies have also reported connections between papillary muscles through false tendons, although Purkinje fibers inside the false tendon were not visualized [16]. In relation to the mechanism of arrhythmia, a circuit connecting the distal and proximal parts may be formed, or the abnormal electrical activity of myocardium may be easily transferred to the proximal. Although the papillary muscle plays an important role in the reentrant mechanism of ventricular arrhythmias, the connection between the papillary muscles and the electrical conduction system is not well known [17]. The association of papillary muscle with free-running Purkinje fibers is important information to understand idiopathic left ventricular arrhythmias [18]. In the swine heart model, left ventricular papillary muscles were related to fatal ventricular arrhythmias [19]. In addition, arrhythmia occurring in the papillary muscle and arrhythmia associated with the fascicle are difficult to evaluate because clinical symptoms are not significantly different [20]. Recently, Rivera et al. reported a muscular connection between papillary muscles, which are related to the electrocardiographic pattern in papillary muscle related ventricular tachyarrhythmias [12]. To ablate the focal area of fascicular reentrant or Purkinje fiber-related ventricular tachycardia, precise anatomic and electrical mapping is important [21].
We investigated the possibility of using a new widefield OCM for observation of the ovine HPS. The advantage of the widefield OCM is that 1) it can be visualized at tangential multi-sections and 3-dimensional reconstruction without tissue destruction, 2) it is suitable to observe a wide range of Purkinje fibers in one microscopic screen, and 3) it has a high spatial resolution. Although the resolution of OCM is inferior compared to the standard light microscope in resolution, widefield OCM is able to observe Purkinje fibers and free running fibers without tissue destruction, which might be important for use in catheter-based procedures.
In order to observe the tissue with a conventional optical microscope, the cutting direction of interest for slicing the tissue should be determined before microscopic observation. Once cut, the tissue cannot be observed again in the other direction. However, with OCM, we could observe the site of interest without tissue destruction. The HPS was especially suited to observe with OCM because it spreads widely in the very shallow layer from the surface. Previously, there were efforts to observe heart structures using OCM technique. Also, OCM is already used in clinical examinations for coronary atherosclerosis because of its high spatial resolution and its ability to observe tissue in real time in vivo [22]. The ability to apply OCM technology to catheter-based imaging is also a great advantage [22]. With advancements in OCM technology, in-vivo catheter-based observation of the HPS may be possible.
There are several considerations to be taken into account when interpreting this study. The configuration of the Purkinje network can be structurally different between animal species [23,24]. The observations in sheep cannot be directly applied to humans, as there may be inconsistencies in distribution of Purkinje fibers. Second, OCM cannot clarify the morphology of the membranous septum, because OCM had lateral resolution of 7.8 μm and axial resolution of 11.4 μm in tissue.
Acknowledgements
This work was supported by both a National Research Foundation of Korea grant funded by the Korean government (No. NRF-2020R1A2C1013832) and a grant KHRS 2018-2 from the Korean Heart Rhythm Society.
Disclosure of conflict of interest
None.
Abbreviations
- HPS
His-Purkinje system
- OCM
optical coherence microscopy
Supporting Information
References
- 1.Boyden PA. Purkinje physiology and pathophysiology. J Interv Card Electrophysiol. 2018;52:255–262. doi: 10.1007/s10840-018-0414-3. [DOI] [PubMed] [Google Scholar]
- 2.Duan D, Yu S, Cui Y, Li C. Morphological study of the atrioventricular conduction system and Purkinje fibers in yak. J Morphol. 2017;278:975–986. doi: 10.1002/jmor.20691. [DOI] [PubMed] [Google Scholar]
- 3.De Almeida MC, Lopes F, Fontes P, Barra F, Guimaraes R, Vilhena V. Ungulates heart model: a study of the Purkinje network using India ink injection, transparent specimens and computer tomography. Anat Sci Int. 2015;90:240–250. doi: 10.1007/s12565-014-0255-9. [DOI] [PubMed] [Google Scholar]
- 4.Xu Y, Peng Y, Qu R, Zheng G, Feng F, Feng Y, He L, He S, Li Z, Liu C, Xiao Z, Ouyang J, Dai J. Locating the human cardiac conduction system using a 3D model of its nutritious arteries. Sci Rep. 2017;7:344. doi: 10.1038/s41598-017-00504-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Atkinson A, Inada S, Li J, Tellez JO, Yanni J, Sleiman R, Allah EA, Anderson RH, Zhang H, Boyett MR, Dobrzynski H. Anatomical and molecular mapping of the left and right ventricular His-Purkinje conduction networks. J Mol Cell Cardiol. 2011;51:689–701. doi: 10.1016/j.yjmcc.2011.05.020. [DOI] [PubMed] [Google Scholar]
- 6.Davies F. The conducting system of the vertebrate heart. Br Heart J. 1942;4:66–76. doi: 10.1136/hrt.4.3.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stephenson RS, Boyett MR, Hart G, Nikolaidou T, Cai X, Corno AF, Alphonso N, Jeffery N, Jarvis JC. Contrast enhanced micro-computed tomography resolves the 3-dimensional morphology of the cardiac conduction system in mammalian hearts. PLoS One. 2012;7:e35299. doi: 10.1371/journal.pone.0035299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tracy EP, Gettler BC, Zakhari JS, Schwartz RJ, Williams SK, Birla RK. 3D bioprinting the cardiac purkinje system using human adipogenic mesenchymal stem cell derived Purkinje cells. Cardiovasc Eng Technol. 2020;11:587–604. doi: 10.1007/s13239-020-00478-8. [DOI] [PubMed] [Google Scholar]
- 9.Tawara S. Jena, Germany: Verlag von Gustav Fischer; 1906. Das reizleitungssystem de saugetierherzens: eine anatomich-hisologische studie uber das atrioventricularbundel und die purkinjeschen faden. [Google Scholar]
- 10.Haissaguerre M, Vigmond E, Stuyvers B, Hocini M, Bernus O. Ventricular arrhythmias and the His-Purkinje system. Nat Rev Cardiol. 2016;13:155–166. doi: 10.1038/nrcardio.2015.193. [DOI] [PubMed] [Google Scholar]
- 11.Wilde AAM, Garan H, Boyden PA. Role of the Purkinje system in heritable arrhythmias. Heart Rhythm. 2019;16:1121–1126. doi: 10.1016/j.hrthm.2019.01.034. [DOI] [PubMed] [Google Scholar]
- 12.Rivera S, Vecchio N, Ricapito P. Anatomical connections between the papillary muscles and the ventricular myocardium. Circ Arrhythm Electrophysiol. 2019;12:e007004. doi: 10.1161/CIRCEP.118.007004. [DOI] [PubMed] [Google Scholar]
- 13.Nogami A. Purkinje-related arrhythmias part I: monomorphic ventricular tachycardias. Pacing Clin Electrophysiol. 2011;34:624–650. doi: 10.1111/j.1540-8159.2011.03044.x. [DOI] [PubMed] [Google Scholar]
- 14.Huang D, Swanson EA, Lin CP, Schuman JS, Stinson WG, Chang W, Hee MR, Flotte T, Gregory K, Puliafito CA, et al. Optical coherence tomography. Science. 1991;254:1178–1181. doi: 10.1126/science.1957169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Loukas M, Louis RG Jr, Black B, Pham D, Fudalej M, Sharkees M. False tendons: an endoscopic cadaveric approach. Clin Anat. 2007;20:163–169. doi: 10.1002/ca.20347. [DOI] [PubMed] [Google Scholar]
- 16.Komatsu Y, Nogami A, Kurosaki K, Morishima I, Masuda K, Ozawa T, Kaneshiro T, Hanaki Y, Shinoda Y, Talib AK, Kowase S, Sekiguchi Y, Aonuma K. Fascicular ventricular tachycardia originating from papillary muscles: Purkinje network involvement in the reentrant circuit. Circ Arrhythm Electrophysiol. 2017;10:e004549. doi: 10.1161/CIRCEP.116.004549. [DOI] [PubMed] [Google Scholar]
- 17.Yamada T, McElderry HT, Okada T, Murakami Y, Doppalapudi H, Yoshida N, Allred JD, Murohara T, Kay GN. Idiopathic focal ventricular arrhythmias originating from the anterior papillary muscle in the left ventricle. J Cardiovasc Electrophysiol. 2009;20:866–872. doi: 10.1111/j.1540-8167.2009.01448.x. [DOI] [PubMed] [Google Scholar]
- 18.Nogami A. Purkinje-related arrhythmias part ii: polymorphic ventricular tachycardia and ventricular fibrillation. Pacing Clin Electrophysiol. 2011;34:1034–1049. doi: 10.1111/j.1540-8159.2011.03145.x. [DOI] [PubMed] [Google Scholar]
- 19.Kim YH, Xie F, Yashima M, Wu TJ, Valderrabano M, Lee MH, Ohara T, Voroshilovsky O, Doshi RN, Fishbein MC, Qu Z, Garfinkel A, Weiss JN, Karagueuzian HS, Chen PS. Role of papillary muscle in the generation and maintenance of reentry during ventricular tachycardia and fibrillation in isolated swine right ventricle. Circulation. 1999;100:1450–1459. doi: 10.1161/01.cir.100.13.1450. [DOI] [PubMed] [Google Scholar]
- 20.Good E, Desjardins B, Jongnarangsin K, Oral H, Chugh A, Ebinger M, Pelosi F, Morady F, Bogun F. Ventricular arrhythmias originating from a papillary muscle in patients without prior infarction: a comparison with fascicular arrhythmias. Heart Rhythm. 2008;5:1530–1537. doi: 10.1016/j.hrthm.2008.08.032. [DOI] [PubMed] [Google Scholar]
- 21.Takase S, Mukai Y, Honda N, Chishaki A, Sadamatsu K, Tsutsui H. Catheter ablation for verapamil-sensitive fascicular ventricular tachycardia guided by precise mapping using a multi-spline duodecapolar catheter with small electrodes and close spacings. HeartRhythm Case Rep. 2019;5:48–52. doi: 10.1016/j.hrcr.2018.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ambrosi CM, Moazami N, Rollins AM, Efimov IR. Virtual histology of the human heart using optical coherence tomography. J Biomed Opt. 2009;14:054002. doi: 10.1117/1.3213604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ono N, Yamaguchi T, Ishikawa H, Arakawa M, Takahashi N, Saikawa T, Shimada T. Morphological varieties of the Purkinje fiber network in mammalian hearts, as revealed by light and electron microscopy. Arch Histol Cytol. 2009;72:139–149. doi: 10.1679/aohc.72.139. [DOI] [PubMed] [Google Scholar]
- 24.Yoshimura A, Yamaguchi T, Kawazato H, Takahashi N, Shimada T. Immuno-histochemistry and three-dimensional architecture of the intermediate filaments in Purkinje cells in mammalian hearts. Med Mol Morphol. 2014;47:233–239. doi: 10.1007/s00795-014-0069-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


