Abstract
Background
Fistulas have puzzled us all the time and stem cell therapy for it is still in its infancy. We conducted a meta-analysis and systematic review to evaluate the efficacy of stem cells and its potential mechanisms in the management of Crohn’s fistula.
Methods
Electronic databases were searched comprehensively for studies reporting the efficacy and safety of stem cells in patients with any form of Crohn’s fistula. A random-effects model was used, and all outcomes were calculated by SPSS 24.0.
Results
Twenty-nine articles with 1252 patients were included. It showed that stem cell group had a higher rate of fistula healing compared to placebo group in patients of Crohn’s fistula (61.75% vs 40.46%, OR 2.21, 95% CI 1.19 to 4.11, P < 0.05). 3 × 107 cells/mL stem cell (SC) group had an advantage in fistula healing rate with 71.0% compared to other doses group of stem cells (RR 1.3, 95% CI 0.76 to 2.22). And the healing rates of patients with perianal and transsphincteric fistulas (77.95%, 76.41%) were higher than those with rectovaginal fistulas. It was an amazing phenomenon that CDAI and PDAI scores occurred an obviously transient rise with the use of stem cells after 1 month (both of P < 0.05), while they returned to the baseline level by giving stem cells 3 months later. Furthermore, the incidence rate of treatment-related adverse events in the stem cell group was significantly lower than in the placebo group (RR 0.58, 95% CI 0.30 to 1.14).
Conclusions
Our study has highlighted that stem cells was a promising method in the treatment of Crohn’s fistula based on its higher efficacy and lower incidence of adverse events, especially ADSCs and Cx601. While it also needs more clinical and pre-clinical studies to strengthen evidences in the future.
Keywords: Stem cells, Crohn’s fistula, Healing rate, Treatment-related adverse events
Introduction
Crohn’s disease (CD) is a chronic, idiopathic inflammation of the whole gastrointestinal tract [1–3]. One of its common and baffling complications is fistula, an abnormal, tunnel-like connection between bowel and nearby epithelial surfaces [4, 5]. And its incidence rate is up to 17 to 50% among CD patients in worldwide [6], in which, 18 to 50% of patients with Crohn’s fistula require surgical removal treatment according to population-based studies [7, 8]. And it is also characterized by facing a notoriously difficult surgical challenge of surgeons’ skills due to its high recurrence [9]. Moreover, since patients with Crohn’s fistula have suffered persistent fecal or urinary seepage, pain, and infection, Crohn’s fistula brought people severe and diverse somatic and social issues, such as sleep disturbance, sexual dysfunction, and bad personal hygiene which reduced the quality of life ultimately [10–12]. Previous studies have shown that impaired local immune system, mucosal and transmural inflammation, luminal bacteria disorder, and hereditary susceptibility might play important roles [13–17]. Yet the specific mechanism is uncertain.
According to the high incidence, mortality, and disability rate, a large amount of researches about treatment of Crohn’s fistula are on the way. The current conventional clinical managements include medications, endoscopic therapy, and surgery. Medications mainly include conventional anti-inflammatory agents such as 5-aminosalicylic acids (5-ASA) and systemic/local corticosteroids, as well as immunomodulators (azathioprine, 6-mercaptopurine (6-MP), cyclosporine, methotrexate, and anti-tumor necrosis factors (TNFs)) [18, 19]. Unfortunately, they have low rates of fistula closure like antibiotics of 21–48%, thiopurines of 20–40%, and infliximab of 23% [11] and have a recurrence rate of about 40.9% [20]. What is more, taking these drugs could lead to various adverse effects companied by lower compliance of patients [21–23]. With the development of diversities of endoscopy, they aimed at finding lesions and carrying drugs to the target in the patients of Crohn’s fistula [24–27]. Its functions are limited. By comparison, surgical removal or drainage of fistulas is the mainstay option to eliminate them at present [28, 29]. But its incidence of postoperative complications was up to 50%, supported by Patil and Cross [30]. And a multicenter trial reported that improvement of inflammatory bowel disease questionnaire (IBDQ) and the MOS item short from health survey score (SF-36) had no difference in the laparoscopic ileocecal resection group of fistula and in the infliximab group (178.10 vs 172.00, 112.10 vs 106.50, P > 0.05, [31]). There was no doubt that combination of medical and surgical treatment had been superior to single medicine treatment in fistula closure (53% vs 43%, P < 0.05) [16].
Stem cell therapy (SCT) has emerged as a novel significant approach to improve the clinical remission and response in a number of inflammatory diseases and tissue regeneration due to its properties of immunoregulatory and multiple differentiation potential by releasing various mediators, including immunosuppressive molecules, growth factors, exosomes, chemokines, complement components, and multiple metabolites [32–34]. As the American Society of Colon and Rectal Surgeons said, the goals of Crohn’s fistula management are alleviation of symptoms by eradication of the fistula, prevention of recurrence and preservation of sphincter integrity and continence [35]. Subsequently, SCT is envisaged as an effective alternative to patients of Crohn’s fistula. Some animal studies had already demonstrated that bone marrow-derived stem cells (BMSCs) were able to repair injured intestinal mucosa through downregulating the immune function of T lymphocytes [36]. Meanwhile, García-Olmo et al. conducted a phase I clinical trial in 2005, showing that the fistula healing rate (HR) reached to 60% with the use of adipose-derived mesenchymal stem cells (ADSCs) [37]. Since then, the application of ADSCs in Crohn’s fistula has been explored increasingly due to its advantages of easy acquisition, low cost-saving of adipose tissue, and minimal invasion [32, 38]. Our previous review also reported that approximately 62.52% of patients with Crohn’s fistula achieved complete clinical remission by using stem cells [39]. Until 2016, the study of Panés et al. (a randomized, double-blind, parallel-group, placebo-controlled trial) demonstrated that local allogeneic darvadstrocel (Cx601, Alofisel, Takeda) administration could be an effective and safe treatment for complex perianal fistulas in patients with Crohn’s disease. Expanded allogeneic adipose-derived mesenchymal stem cells had been a novel, minimally invasive, well-tolerated, and alternative option in the treatment of Crohn’s fistula in 2018, he said. To facilitate expanded development and application of stem cells in Crohn’s disease, we have made great progress over the past 20 years, while it is still short of reliable assessment about stem cells (SCs) in the process of fistula healing in clinic, our objective is to redefine the role of stem cells and elaborate the mechanisms after SCT in patients of Crohn’s fistula in this review.
Material and methods
Search strategy
We identified relevant studies by performing a comprehensive search of Pubmed and other databases (Cochrane Library and Embase) from the June 2005 to August 2020. The search was limited to clinical studies published in the English language. The search strategy was applied as below (all fields): (“mesenchymal stromal cells” OR “mesenchymal stem cells” OR “stem cells” OR “stroma cells”) AND (“fistula”) AND (“inflammatory bowel disease” OR “Crohn’s disease”), and any appropriate abbreviations.
Study selection
All study selections were conducted by two reviewers independently without any discrepancy. Finally, they satisfied the following inclusion criteria: (1) clinical trials, (2) randomized controlled trials (RCTs) or non-randomized experimental studies (cohorts), (3) articles in English with full texts, (4) established diagnosis of CD or inflammatory bowel disease (IBD), (5) applied stem cells treatment for fistula, and (6) efficacy and/or adverse events were reported. Exclusion criteria were as follows: (1) repeated studies, (2) case reports, (3) letters or/and comments, (4) reviews or/and meta-analysis, (5) related with the pregnancy patients, and (6) non-English language articles.
Data extraction and assessment of study quality
Eligible articles were reviewed, the data were extracted and checked, and these were performed by two reviewers in a blind manner. And any disagreement in this process was resolved by discussion. Assessment of the quality of each study included was performed using Newcastle Ottawa Quality Assessment Scale (NOS), which involves 9 items for clinical trials [40]. All answers generated the final scores for each study. A high-quality article scores 5–9 [41].
Statistical analysis
We evaluated the degree of heterogeneity between studies using inconsistency index (I2). Generally, the values of I2 equal to 25%, 50%, and 75% were considered to indicate low, moderate, and high heterogeneity, respectively [42]. We adopted a random-effects model in our analysis. Statistical meta-analysis was performed in Review Manager 5.3 (Cochrane Collaboration, Oxford, UK) to generate forest plots of pooled risk ratio (RR), odds ratio (OR) and mean with 95% confidence intervals (CIs), and SPSS 24.0 was used to assess the efficacy (fistula healing rate) and treatment-related adverse events (TRAEs). Subgroup data was analyzed using the Chi-square test at the subgroup analysis (dose of stem cells, TRAEs), and P < 0.05 was considered statistically significant. Prism 6.0 was used for drawing pictures. Fistula healing, scores of Crohn disease activity index (CDAI), perianal disease activity index (PDAI), inflammatory bowel disease questionnaire (IBDQ), and level of C-reactive protein (CRP) were assessed during the follow-up.
Results
Search results and study characters
A total of 901 articles were researched by our search strategy, of which 29 were included and a total of 1252 fistula patients were enrolled in our analysis by the eligibility criteria (Fig. 1), and study characteristics were shown in Table 1.There were 22 studies applied with ADSCs (four from Alofisel, Cx601, 16 from homemade cultures, one from Cx401, and one without a clear source), six with BMSCs and one with adipose tissue. The phases II–III studies accounted for approximately 44.83% (13/29) and phase I for 20.69% (6/29). Only 12 in 29 studies were RCTs: comparing applied with stem cells to placebo (e.g., Fibrin glue) [43–52], and comparing patients of Crohn’ fistula to those not with CD [43, 44, 53, 54]. The remaining received SCT with no control group. Eighteen studies used autologous stem cells [37, 44–47, 50, 55–66], seven with allogeneic stem cells [48, 49, 51, 52, 67–69], and both of them were used in other two studies [53, 54].
Fig. 1.
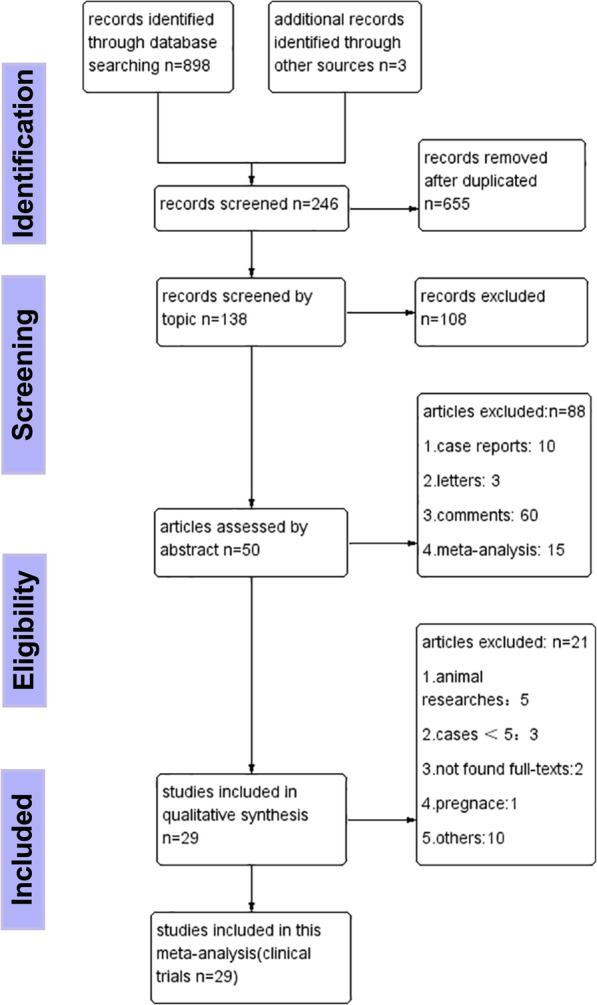
Flow chart for clinical trials of stem cells for the treatment of fistulas in this meta-analysis
Table 1.
Characteristics of clinical studies included
| Author | Year of publication | Phase | Age (M ± SD) | Sex (M:F) | Number of case included | Source of cell | Type of cell | Dose of cell injected | Donor | Follow-up* | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Both of CD and un-CD fistula | |||||||||||
| Garcia-Olmo et al. [43] | 2009 | II | 43.33 ± 9.9 | 24:25 | CD, 14 (ASC, 7; FG, 7); unCD, 35 (ASC, 17; FG, 18) | AT, Cx401 | ADSC | 1 × 107 cells/ml | NR | 7 M | FH, SF-12, SAE |
| Guadalajara et al. [44] | 2012 | II | 42.6 ± 10.9 | 24:25 | CD, 20 (ASC+FG, 17; FG, 3); unCD, 29 (ASC+FG, 7; FG, 22) | Liposuction, NR | ADSC | NR | Autologous | 5 Y | FH, MRI, SAE |
| Garcia-Olmo et al. [53] | 2015 | I–II–III | 49 | 8:2 | CD, 3; un-CD, 7 | AT, homemade | ADSC/SVF | 1–3 × 107 | Autologous/allogeneic | 1 Y | FH, Wexner scores, SAE |
| Herreros et al. [54] | 2019 | NR | 45 | 24:21 | CD, 18; un-CD, 24 | NR, homemade | ASC/SVF | 2 × 106 cells/ml | Autologous/allogeneic | 1 Y | FH, AE, SAE |
| Only un-CD fistula | |||||||||||
| Herreros et al. [45] | 2012 | III | 49.78 | NR | ASC, 64; ASC+FG, 60; FG, 59 | AT, homemade | ADSC | First, 2 × 107 cells/ml; second, 4 × 107 cells/ml | Autologous | 1 Y | SF-36Q, Wexner score 22, FCS, SAE |
| Choi et al. [55] | 2017 | II | 37.9 | 13:0 | 1 × 107 cells/mL, 5; 2 × 107 cells/mL, 8 | AT, homemade | ADSC | 2 × 107 or 4 × 107 cells/ml | Autologous | 6 M | FH, Wexner scale, SAEs |
| Dozois et al. [56] | 2019 | I | 39.8 | 7:8 | 15 | AT, homemade | AD-MSC | NR | Autologous | 6 M | FH, MRI, SAE |
| Topal et al. [57] | 2019 | NR | 47 ± 13.1 | 8:2 | 10 | AT, homemade | ADSC | NR | Autologous | 9 M | PE, SAE |
| Garcia-Arranz et al. [46] | 2020 | III | 50 ± 10 | 30:14 | 10 × 107 ASC+FG, 23; FG, 21 | NR, NR | ADSC | 10 × 107 cells/ml | Autologous | 2 Y | PE, MRI, SF-12, Wexner incontinence score, SAE |
| Only CD fistula | |||||||||||
| García-Olmo et al. [37] | 2005 | I | 35.1 ± 2.4 | 2:3 | 5 | AT, homemade | AD-MSC | 1–3 × 107 cells/ml | Autologous | 30 M | FH, MRI, SAE |
| Ciccocioppo et al. [58] | 2011 | NR | 32 | 8:4 | 12 | BM, homemade | BM-MSC | NR | Autologous | 1 Y | CDAI; PDAI; MRI; endoscopy; FoxP3; T cell; IL-2, 5, 10, and 12; IFN; TNF-a; SAE |
| Cho et al. [59] | 2013 | II | 26. ± 6.0 | 4:6 | 1 × 107 cells/ml, 3; 2 × 107 cells/ml, 4; 4 × 107 cells/ml, 3 | AT, homemade | ADSC | 1/2/4 × 107 cells/ml | Autologous | 8 M | FH, CD4/CD8 ratio, SAE |
| de la Portilla et al. [60] | 2013 | I/IIa | 36 ± 9.0 | 11:13 | 2 × 107 cells/ml, 9; 6 × 107 cells/ml, 15 | AT, homemade | ADSC | First, 2 × 107 cells/ml; second, 4 × 107 cells/ml | Autologous | 6 M | MRI, PDAI, CDAI, SAE |
| Lee et al. [61] | 2013 | II | 26.2 ± 5.4 | 30:13 | 42 | AT, homemade | ADSC | First, 15.8 × 107; second, 19.1 × 107 | Autologous | 1 Y | FH, SAE |
| Molendijk et al. [47] | 2015 | NR | 37.3 | 12:9 | 1 × 107 cells/ml, 5; 3 × 107 cells/ml, 5; 9 × 107 cells/ml, 5; placebo, 6 | BM, homemade | ADSC | 1/3/9 × 107 cells/ml | Autologous | 2 Y | FH, MRI, PDAI, CDAI, IBDQ, CDEIS, SES-CD, SF-36, CRP, IL-8, IL-1β, IL-6 and 10, TNF, IL-12p70, SAE |
| Ciccocioppo et al. [62] | 2015 | NR | 39.25 ± 14.32 | 5:3 | 8 | BM, homemade | BM-MSC | NR | Autologous | 6 Y | MRI, CDAI, PDAI, SAE |
| Cho et al. [63] | 2015 | II | 26.2 ± 5.5 | 28:13 | 41 | AT, homemade | ADSC | 3 × 107 cells/ml | Autologous | 2 Y | FH, AE, SAE |
| Park et al. [67] | 2016 | NR | 32.17 ± 7.96 | 4:2 | 1 × 107 cells/ml, 3; 3 × 107 cells/ml, 3 | AT, homemade | ADSC | 1 × 107 cells/ml, 3 × 107 cells/ml | Allogeneic | 6 M | FH, MRI, CD4/CD8 ratio, SAE |
| Panés et al. [48] | 2016 | III | 38 | 116:96 | ASC, 107; placebo, 105 | AT, alofisel | AD-MSC | 12 × 107 cells | Allogeneic | 6 M | MRI, PDAI, CDAI, IBDQ, van Assche score, IgG AE, SAE |
| García-Arranz et al. [68] | 2016 | I–IIa | 35 | NR | 10 | AT, homemade | ADSC | First, 2 × 107 cells; second, 4 × 107 cells | Allogeneic | 4.5 M | QoL, SF-36, fecal incontinence, severity index |
| Dietz et al. [64] | 2017 | I | 35 ± 14.21 | 6:6 | 12 | BM, homemade | MSC | 2 × 107 cells/ml | Autologous | 6 M | MRI, Van Assche score, SAE |
| Scott [69] | 2018 | III | 38 | NR | 24 | AT, alofisel | ADSC | 12 × 107 cells | Allogeneic | 13 M | IL-6, 12, and 10; TNF-α; TGF-β; PDAI; CDAI; SAE |
| Panés et al. [49] | 2018 | III | 38.3 | 114:98 | 12 × 107 cells, 107; placebo, 105 | AT, alofisel | ADSC | 12 × 107 cells | Allogeneic | 13 M | IBDQ, PDAI, CDAI, TNF, MRI, TEAE |
| Wainstein et al. [70] | 2018 | NR | 36 | 2:7 | 9 | AT, homemade | ADSC | NR | Autologous | 37 M | IBDQ, PDAI, SAE |
| Avivar-Valderas et al. [52] | 2019 | NR | NR | NR | ASC 58; placebo 42 | AT, Alofisel | AD-MSC | NR | Allogeneic | 13 M | DSA; HLA-I; CRPs; CD55, 46, and 59; AE; SAE |
| Dige et al. [66] | 2019 | NR | NR | 6:15 | 21 | AT, homemade | AT | NR | Autologous | 6 M | FH, complications |
| Zhou et al. [50] | 2020 | NR | 28.86 ± 10.13 | 21:1 | ADSC, 11; placebo, 11 | AT, homemade | ADSC | 5 × 106 cells/ml | NR | 1 Y | FH, MRI, ultrasonography, CDAI, PDAI, IBDQ, VAS, Wexner score, CRP, ESR, FC, SAE |
| Barnhoorn et al. [51] | 2020 | NR | 42 | 8:6 | 1 × 107 cells/ml, 5; 3 × 107 cells/ml, 5; 9 × 107 cells/ml, 5; placebo, 6 | BM homemade | BM-MSC | 1/3/9 × 107 cells/ml | Allogeneic | 4 Y | FH, HLA, MRI, rectoscopy, PDAI, CDAI, Vaizey, QoL, SF36, IBDQ, SAE |
| Lightner et al. [65] | 2020 | NR | 49 | 0:5 | 5 | BM, homemade | MSC | NR | Autologous | 6 M | FH, MRI, AE, SAE |
*Y = years and M = months
Abbreviations: NR not report, FG fibrin glue, AT adipose tissue, BM bone marrow, ADSC adipose-derived stem cells, SVF stromal vascular fraction, MSC mesenchymal stem cell, BM-MSC bone marrow-derived mesenchymal stromal cells, FH fistula healing, PE physical examination, QoF quality of life, MRI magnetic resonance imaging, FCS fistula complexity score, CDAI Crohn’s disease activity index, PDAI perianal disease activity index, CDEIS Crohn’s disease endoscopic index of severity, SES-CD simplified endoscopic activity score, CRP C-reactive protein, TGF-β transforming growth factor-β, DSA donor-specific antibodies, VSA pain scores with visual analog score, ESR erythrocyte sedimentation rate, FC fecal calprotectin, GF growth factor, BLI bioluminescence, TEAEs treatment-emergent adverse events
Quality assessment
The qualities of included studies were assessed by using the NOS, and we analyzed 29 clinical studies enrolled and finally had a total score of 175 with a mean of 6 and a range of 3 to 9 for each study (Supplement Table 1). In total, the quality of clinical trials reached the “high-quality study” level.
Primary outcomes
Efficacy of stem cells therapy for Crohn’ fistulas
Four of 12 RCTs reported fistula healing in Crohn’s fistula and un-CD fistula [43, 44, 53, 54]. Seven reported fistula healing administrated with stem cells and placebo (such as fibrin glue) [43, 44, 47–51]. The random-effects model was used to assess the differences in healing rate (HR) between the experiment group (Crohn’s fistulas, stem cell group) and control group (un-CD fistulas, placebo group). Regardless of the dose of stem cells injected and route of administration, the HRs of fistula in patients with CD and without CD were 58.71% and 58.72%, respectively (I2 = 0%, RR 0.00, 95% CI − 0.20 to 0.20, P>0.05) (Fig. 2a). Moreover, patients with Crohn’s fistula in the stem cell group had a higher HR of 61.75% than in the placebo group (40.46%). There was a significant statistical difference (I2 35%, RR 2.21, 95% CI 1.19 to 4.11, P = 0.01) (Fig. 2b). As for the source of stem cells, HR of Cx601 administration in patients of Crohn’s fistula was about 61.02%, which was higher than of ASCs from homemade cultures (51.43%, P < 0.05).
Fig. 2.
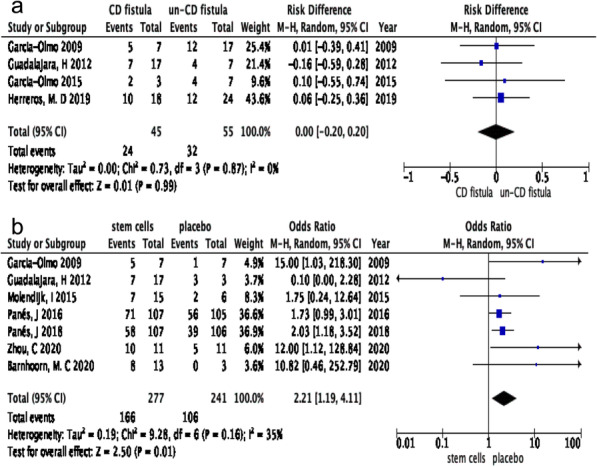
a CD fistulas versus un-CD fistulas for fistula healing administrated by stem cells. b Stem cells versus placebo administrated for fistula healing in the treatment of Crohn’s fistula
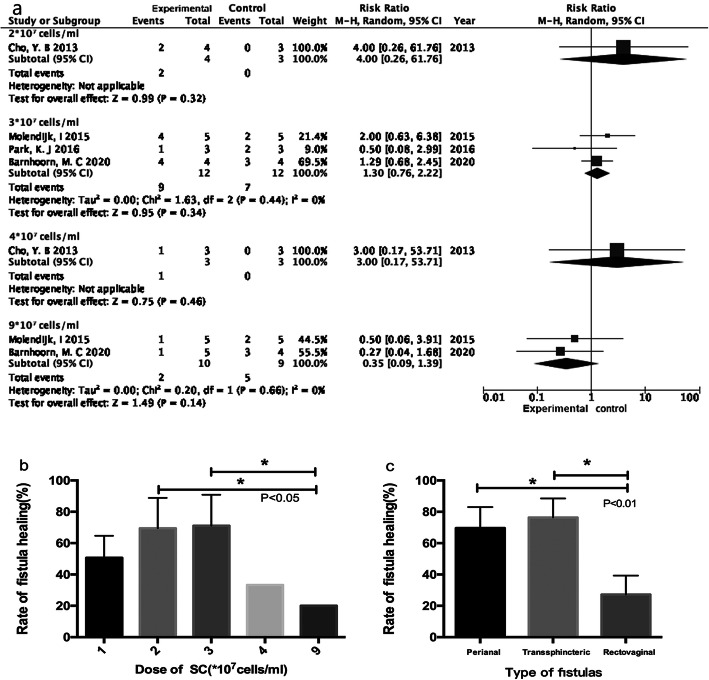
The doses of stem cells injected
There were four clinical trials involving the different doses of SCT [47, 51, 59, 67], and a total of 31 patients of Crohn’s fistula received the different doses of stem cells. They were divided into five subgroups: 1 × 107 cells/mL, 2 × 107 cells/mL, 3 × 107 cells/mL, 4 × 107 cells/mL, and 9 × 107 cells/mL groups, respectively. The patients injected with the dose of 1 × 107 cells/mL group were defined as the control group, and others were experimental groups. As Fig. 3a showed, only one study was treated with stem cells of doses of 2 × 107 cells/mL (n = 7) and 4 × 107 cells/mL (n = 6), which was lack of completely valid data [59], and three studies with stem cells of dose of 3 × 107 cells/mL (n = 24, [47, 51, 67]), two with 9 × 107 cells/mL (n = 19, [47, 51]). The dose of 3 × 107 cells/mL group had a stronger advantage in Crohn’ fistula healing compared to 1 × 107 cells/mL group (I2 0.00%, OR 1.30, 95% CI 0.76 to 2.22), but the dose of 9 × 107 cells/mL group was defeated by the 1 × 107 cells/mL group in terms of fistula healing (I2 0.00%, OR 0.35, 95% CI 0.09 to 1.39). The HRs of patients with Crohn’s fistula in 1 × 107 cells/mL, 2 × 107 cells/mL, 3 × 107 cells/mL, 4 × 107 cells/mL, and 9 × 107 cells/mL group were 50.60%, 69.50%, 71.00%, 33.00%, and 20.00%, respectively (showed in Fig. 3b). Compared to 4 × 107 cells/mL and 9 × 107 cells/mL groups, there were statistically significant differences in 2 × 107 cells/mL and 3 × 107 cells/mL groups, respectively (both of P < 0.05). Overall, the dose of 3 × 107 cells/mL stem cells had a higher superiority in patients of Crohn’s fistula.
Fig. 3.
a Forest plot of doses of stem cells implanted for Crohn’ fistula. b Fistula healing rate of dose of stem cells implanted for Crohn’s fistula. c Fistula healing rate of different fistula type administrated by stem cells
The types of Crohn’s fistula
Nine of 29 studies reported HRs based on fistula types with a total of 99 fistulas [37, 43, 44, 54, 64, 66–68, 70]. They mainly comprised perianal, transsphincteric, and rectovaginal fistulas. Our analysis involved in perianal fistula with seven studies, transsphincteric fistula with three studies, and rectovaginal fistula with seven. As Fig. 3c showed, the patients with perianal and transsphincteric fistulas had more apparent HRs compared to those with rectovaginal fistulas (77.95%, 76.41% vs. 27.18%, P < 0.01).
Secondary outcomes
Related scores and indicator assessment
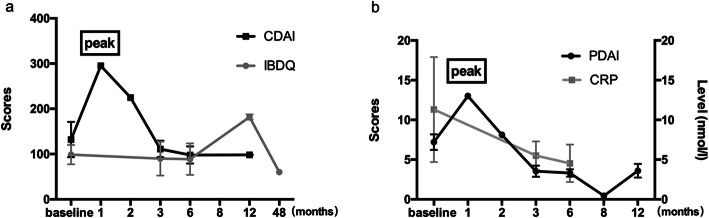
Most of clinical studies reported the detailed changes of scores and lab indicators, such as CDAI, PDAI, IBDQ, and CRP. Of them, four studies reported variation of CDAI after administrating stem cells [47, 49, 58, 60], seven articles were referred to changes of PDAI [47–50, 58, 60, 70], five to scores of IBDQ [47, 49–51, 70], and two to the level of CRP [47, 50]. For scores of CDAI, 1 month later, it occurred a transient rise among patients with the treatment of stem cell (295.00 vs. 132.10, P < 0.05), while 3 months later, it returned to the baseline score and last to endpoint (295.00 vs. 111.14, P < 0.05) (Fig. 4a). It had a mild difference in the variation of PDAI score—there were two rising tendency after 1 month and 12 months by SCT, respectively, and it downed to 3.54 and 3.31 until 3 and 6 months after giving SCs. There was a statistical significant difference between pre-stem cells and post-stem cells (P < 0.05) (Fig. 4b). Besides that, scores of IBDQ did not increase until 6 months later by SCT and reached a peak at 12 months with no significance difference (88.85 ± 69.96 vs. 181.95 ± 8.27, P > 0.05). Moreover, after stem cells therapy, the level of CRP (nmol/l) showed continuous drops (baseline vs. 6 months, 11.3 ± 13.23 vs. 4.5 ± 4.7, P < 0.05). These details could be found in Figs. 4a and b.
Fig. 4.
Different assessment index at follow-up. a CDAI and IBDQ. b PDAI and CRP
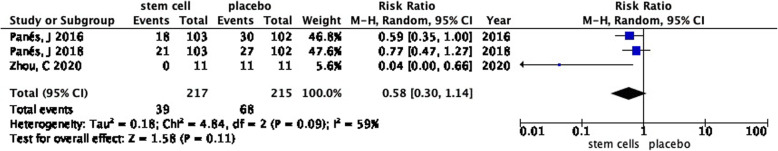
TRAEs analysis
All of studies showed the adverse events (AEs) and severe adverse events (SAEs). However, in terms of TRAEs, the details and accurate data are only reported in three RCTs [48–50] and eight cohort studies. Figure 5 showed the difference of the incidence rate of TRAEs between the stem cell group and placebo group, patients in the stem cell group had an advantage over in placebo group (I2 59%, RR 0.58, 95% CI 0.30 to 1.14). In cohort studies, only studies by de la Portilla et al. and Scott et al. [60, 69] reported five TRAEs.
Fig. 5.
Forest plot of TRAEs between stem cells and placebo in patients with Crohn’s fistula
Discussion
To our knowledge, this review summarized that SCT had a higher efficacy and lower incidence of adverse events in patients with Crohn’s fistula compared with conventional therapies. We revealed that the rate of fistula healing ranged from 58 to 62% after administrating SCs in patients of Crohn’s fistula, which was similar to our previous results with a clinical remission of 62.52% [39]. What is more, Cx601 (darvadstrocel) administration in patients of Crohn’s fistula had a higher HR of 61.02% compared to homemade cultures, it had strongly evidenced that it was necessary and important for a systematic, maturing and professional protocol in clinical work. Our analysis also found, whether patients suffered Crohn’s fistula or not (un-CD fistula), the HR had no statistically significant difference (58.71% vs. 58.72%). Considering the properties of immunoregulatory, anti-inflammatory, and multipotential differentiation of stem cells [33, 34, 71–73], which one dominated the important position and what the potential mechanism was? The possible explanations were presented as follows: primarily, SCs could alter immune reconstitution by stimulating de novo generation of an altered T cell repertoire and eliminating aberrant clones, it then would replace fistulas [74]. Secondly, SCs could release various mediators and multiple metabolites, such as growth factors, exosomes, and chemokines, to repair injured tissue and promote its regeneration by strengthening differentiation [75]. Plus, the study by Clevers et al. also had referred to “niche” of stem cells, and it was able to promote self-organization of multiple mammalian tissues by the self-renewal factors of Wnt signaling [76]. Conversely, stem cells’ fate was influenced by various growth factors to differentiate into normal tissues [77]. Obviously, the latter match more with roles of stem cells in this process among patients of Crohn’s fistula and un-CD fistula. It was, therefore, worth supposing that stem cells might work in patients of Crohn’s fistula and un-CD fistula by means of multi-directional differentiation to achieve tissue repair, rather than of immune reconstitution only.
With respect to the doses of SCT in patients of Crohn’s fistula in our review, data on the use of stem cells at different doses was limited. And a variety of articles consistently demonstrated that an optimal dose was the key point in the protocol of stem cells for Crohn’s fistula and was a focused problem for a long time in clinical and pre-clinical studies. In this study, only two clinical studies by Molendijk et al. and Barnhoorn et al. [47, 51] randomly assigned CD patients to three groups based on doses of stem cells: 1 × 107 cells/mL, 3 × 107 cells/mL, and 9 × 107 cells/mL, and they demonstrated that 3 × 107 cells/mL group had the highest HR. Meanwhile, the study of Scott reported that a single dose of 120 million stem cells (Darvadstrocel) administered into the perianal fistulas tissue was significantly more effective than placebo (saline) with a clinical remission of more than 50% [69]. Additionally, our published articles had put forward that the best range of dose of stem cells in Crohn’s fistula was 2–4 × 107 cells/mL with a HR of up to 80.07% [39]. Surprisingly, we found that stem cells at the dose of 3 × 107 cells/mL had the highest HR of 71.00% compared to other doses of cells in this meta-analysis, which was beneficial for improving the efficacy in Crohn’s fistula at a large degree and narrowed down the optimal dose of SCs further.
Given the classification of fistulas, they could be divided into simple and complex fistulas based on American Gastroenterological Association classification system [78, 79], as well as into intersphincteric, transsphincteric, extrasphincteric, or suprasphincteric fistulas by anatomy [80]. In our study, it referred mostly to perianal, rectovaginal, intersphincteric, and transsphincteric fistulas. Only nine of them reported the fistula healing rate based on the fistula types. Surprisingly, we noticed that patients of perianal and transsphincteric fistulas had a better reaction to SC implantation than of rectovaginal fistula with the efficacy of 77.95% and 76.41%, respectively. But the reason was not lucid. Among patients of perianal fistulas, the inflammation was extremely severe due to roles of intestinal bacterial and communication of excrement and perianal skin surface to trigger an immune response and cytokine production [43, 81]. As the study by Cellerix et al. supported, SCs could be stimulated by relatively high concentrations of pro-inflammatory cytokines (IFN-γ) produced by fistula lesions, and expressed indoleamine 2, 3-dioxygenase (IDO) of metabolizing tryptophan to kynurenine, which had an anti-inflammatory effect. So, let us make a hypothesis that in the relatively severe inflammatory microenvironment of perianal area, SCs play a remarkable role of anti-inflammatory, which was consistent with what we discussed previously (the “Discussion” section, paragraph 1, [82, 83]). Meanwhile, the second reason, perianal and transsphincteric fistulas themselves were also believed to own a relatively dry and comfortable local anatomical circumstance, which contribute to renovate impaired tissue being different from rectovaginal fistulas [54].
CDAI was defined to evaluate the efficacy and severity of Crohn’s disease, as well as PDAI [84]. In 2017, our study already showed that there was a different clinical remission between patients of CDAI baseline > 150 group and < 150 group by contrasting the change of level of CDAI after SCT [39]. While it did not refer to the change of CDAI at each time point—we solved it in this review. Coincidentally, we found a transient rise at 1 month extensively and reduced to a level below the baseline at 3 months after injecting SCs. Which of the mechanisms were playing significant roles in it? An animal experiment might have mirrored our results: once stem cells were injected into fistula walls among rats of Crohn’s fistula, effective stem cell mass was reduced from inception, and the detectable cells represented only 8.87% of their initial amount to take action at the first 30 days via monitoring the dynamics of bioluminescence (BLI) [85]. Moreover, the study of TiGenix et al. [86] had confirmed that ADSCs were present in the rectum and jejunum for ≥ 14 days and undetectable after 3 or 6 months in any of the tissues. Taken together, we can hypothesize that at the period of 1 month after SCT, up to 90% of stem cells vanished and destroyed by peoples’ immune clearance themselves; after 3 months, the remaining SCs (about 8%) were differentiated into functional epithelial cells or stromal cells; meanwhile, the phases of immune tolerance and reconstruction started up [74]. The defined and powerful mechanisms need further pre-clinical and clinical studies.
Limitations
Several limitations of our meta-analysis should be acknowledged: (1) In terms of animal researches, they were small-sized and lacked detailed data about mucous healing; assessment index, such as IL-2, IL-6, IL-8, and IL-10; and the adverse events, which failed to analyze systematically. (2) In clinical trials, they did not report the results about immunohistochemistry and endoscopy in quantification. (3) Subgroup analysis was inadequate owing to the majority of studies included about cohort studies. (4) Given the limited follow-up among included studies, we failed to elaborate the recurrent after administrating stem cells among CD patients. (5) We used random-effects model to account for the statistical heterogeneity conservatively in the pooled studies.
Conclusions
The utility of stem cells in patients of Crohn’s disease is a potential way, but still in the very early stages, particularly in Crohn’s fistula; the achievements are encouraging now but not comprehensive and systematic. Based on our review, SC treatment in treatment of Crohn’s fistula has a higher efficacy (fistula healing rate), as well as a lower TRAEs compared to other options. And the optimal dose of 3 × 107 cells/mL SCs injected has been determined. While a gold standard is still not be identified, we are eager to do more basic and clinical studies performed to further ascertain protocol and break current dilemmas.
Supplementary Information
Acknowledgements
Thanks a lot for the financial support by Henan Medical Science and Technology innovation talent in the study.
Abbreviations
- SCs
Stem cells
- SCT
Stem cell therapy
- RR
Risk ratios
- OR
Odds ratio
- CIs
Confidence intervals
- ADSCs
Adipose-derived stem cells
- RCTs
Random control trials
- HR
Healing rate
- CD
Crohn disease
- IBD
Inflammatory bowel disease
- 5-ASA
5-Aminosalicylic acids
- 6-MP
6-Mercaptopurine
- TNFs
Anti-tumor necrosis factors
- SF-36
MOS item short from health survey score
- BMSCs
Bone marrow-derived stem cells
- NOS
Newcastle Ottawa Quality Assessment Scale
- TRAEs
Treatment-related adverse events
- CDAI
Crohn disease activity index
- PDAI
Perianal disease activity index
- IBDQ
Inflammatory bowel disease questionnaire
- CRP
C-reactive protein
- AEs
Adverse events
- SAEs
Severe adverse events
Authors’ contributions
YC participated in the design of the study and drafted the manuscript. QS participated in the collection of study and performed the statistical analysis. BZ conceived of the study, and participated in its design and coordination and helped to draft and modify the manuscript. FS and SL revised the manuscript and polished the English language. All authors read and approved the final manuscript.
Funding
No.
Availability of data and materials
The data supporting the conclusions of this article are all online.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yantian Cao, Email: caoyantian15@163.com.
Qi Su, Email: suliner@outlook.com.
Bangjie Zhang, Email: zbjcx@126.com.
Fangfang Shen, Email: ffshen2010@163.com.
Shaoshan Li, Email: shaoshanlioo1@sina.com.
Supplementary information
Supplementary information accompanies this paper at 10.1186/s13287-020-02095-7.
References
- 1.Kappelman MD, Rifas-Shiman SL, Kleinman K, Ollendorf D, Bousvaros A, Grand RJ, Finkelstein JA. The prevalence and geographic distribution of Crohn’s disease and ulcerative colitis in the United States. Clin Gastroenterol Hepatol. 2007;5(12):1424–1429. doi: 10.1016/j.cgh.2007.07.012. [DOI] [PubMed] [Google Scholar]
- 2.Sonnenberg A, Genta RM. Geographic distributions of microscopic colitis and inflammatory bowel disease in the United States. Inflamm Bowel Dis. 2012;18(12):2288–2293. doi: 10.1002/ibd.22932. [DOI] [PubMed] [Google Scholar]
- 3.Loftus EV., Jr Clinical epidemiology of inflammatory bowel disease: incidence, prevalence, and environmental influences. Gastroenterology. 2004;126(6):1504–1517. doi: 10.1053/j.gastro.2004.01.063. [DOI] [PubMed] [Google Scholar]
- 4.Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn’s disease. Lancet. 2017;389(10080):1741–1755. doi: 10.1016/S0140-6736(16)31711-1. [DOI] [PubMed] [Google Scholar]
- 5.Lee MJ, Parker CE, Taylor SR, Guizzetti L, Feagan BG, Lobo AJ, Jairath V. Efficacy of medical therapies for fistulizing Crohn’s disease: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018;16(12):1879–1892. doi: 10.1016/j.cgh.2018.01.030. [DOI] [PubMed] [Google Scholar]
- 6.Schwartz DA, Loftus EV, Jr, Tremaine WJ, Panaccione R, Harmsen WS, Zinsmeister AR, Sandborn WJ. The natural history of fistulizing Crohn’s disease in Olmsted County, Minnesota. Gastroenterology. 2002;122(4):875–880. doi: 10.1053/gast.2002.32362. [DOI] [PubMed] [Google Scholar]
- 7.Bouguen G, Peyrin-Biroulet L. Surgery for adult Crohn’s disease: what is the actual risk? Gut. 2011;60(9):1178–1181. doi: 10.1136/gut.2010.234617. [DOI] [PubMed] [Google Scholar]
- 8.Fumery M, Singh S, Dulai PS, Gower-Rousseau C, Peyrin-Biroulet L, Sandborn WJ. Natural history of adult ulcerative colitis in population-based cohorts: a systematic review. Clin Gastroenterol Hepatol. 2018;16(3):343–356.e343. doi: 10.1016/j.cgh.2017.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Torres J, Billioud V, Sachar DB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis as a progressive disease: the forgotten evidence. Inflamm Bowel Dis. 2012;18(7):1356–1363. doi: 10.1002/ibd.22839. [DOI] [PubMed] [Google Scholar]
- 10.Kotze PG, Shen B, Lightner A, Yamamoto T, Spinelli A, Ghosh S, Panaccione R. Modern management of perianal fistulas in Crohn’s disease: future directions. Gut. 2018;67(6):1181–1194. doi: 10.1136/gutjnl-2017-314918. [DOI] [PubMed] [Google Scholar]
- 11.Gecse KB, Bemelman W, Kamm MA, Stoker J, Khanna R, Ng SC, Panés J, van Assche G, Liu Z, Hart A, et al. A global consensus on the classification, diagnosis and multidisciplinary treatment of perianal fistulising Crohn’s disease. Gut. 2014;63(9):1381–1392. doi: 10.1136/gutjnl-2013-306709. [DOI] [PubMed] [Google Scholar]
- 12.Williet N, Sarter H, Gower-Rousseau C, Adrianjafy C, Olympie A, Buisson A, Beaugerie L, Peyrin-Biroulet L. Patient-reported outcomes in a French nationwide survey of inflammatory bowel disease patients. J Crohns Colitis. 2017;11(2):165–174. doi: 10.1093/ecco-jcc/jjw145. [DOI] [PubMed] [Google Scholar]
- 13.Khor B, Gardet A, Xavier RJ. Genetics and pathogenesis of inflammatory bowel disease. Nature. 2011;474(7351):307–317. doi: 10.1038/nature10209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Steel AW, Mela CM, Lindsay JO, Gazzard BG, Goodier MR. Increased proportion of CD16(+) NK cells in the colonic lamina propria of inflammatory bowel disease patients, but not after azathioprine treatment. Aliment Pharmacol Ther. 2011;33(1):115–126. doi: 10.1111/j.1365-2036.2010.04499.x. [DOI] [PubMed] [Google Scholar]
- 15.Ballester Ferré MP, Boscá-Watts MM, Mínguez Pérez M. Crohn’s disease. Med Clin (Barc) 2018;151(1):26–33. doi: 10.1016/j.medcli.2017.10.036. [DOI] [PubMed] [Google Scholar]
- 16.Lopez N, Ramamoorthy S, Sandborn WJ. Recent advances in the management of perianal fistulizing Crohn’s disease: lessons for the clinic. Expert Rev Gastroenterology Hepatol. 2019;13(6):563–577. doi: 10.1080/17474124.2019.1608818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Haac BE, Palmateer NC, Seaton ME, Van YR, Fraser CM, Bafford AC. A distinct gut microbiota exists within Crohn’s disease-related perianal fistulae. J Surg Res. 2019;242:118–128. doi: 10.1016/j.jss.2019.04.032. [DOI] [PubMed] [Google Scholar]
- 18.Feuerstein JD, Cheifetz AS. Crohn disease: epidemiology, diagnosis, and management. Mayo Clin Proc. 2017;92(7):1088–1103. doi: 10.1016/j.mayocp.2017.04.010. [DOI] [PubMed] [Google Scholar]
- 19.Eustace GJ, Melmed GY. Therapy for Crohn’s disease: a review of recent developments. Curr Gastroenterol Rep. 2018;20(5):19. doi: 10.1007/s11894-018-0625-x. [DOI] [PubMed] [Google Scholar]
- 20.de Groof EJ, Sahami S, Lucas C, Ponsioen CY, Bemelman WA, Buskens CJ. Treatment of perianal fistula in Crohn’s disease: a systematic review and meta-analysis comparing seton drainage and anti-tumour necrosis factor treatment. Colorectal Dis. 2016;18(7):667–675. doi: 10.1111/codi.13311. [DOI] [PubMed] [Google Scholar]
- 21.Ruemmele FM, Veres G, Kolho KL, Griffiths A, Levine A, Escher JC, Amil Dias J, Barabino A, Braegger CP, Bronsky J, et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J Crohns Colitis. 2014;8(10):1179–1207. doi: 10.1016/j.crohns.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 22.Toruner M, Loftus EV, Jr, Harmsen WS, Zinsmeister AR, Orenstein R, Sandborn WJ, Colombel JF, Egan LJ. Risk factors for opportunistic infections in patients with inflammatory bowel disease. Gastroenterology. 2008;134(4):929–936. doi: 10.1053/j.gastro.2008.01.012. [DOI] [PubMed] [Google Scholar]
- 23.Ansari A, Patel N, Sanderson J, O’Donohue J, Duley JA, Florin TH. Low-dose azathioprine or mercaptopurine in combination with allopurinol can bypass many adverse drug reactions in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2010;31(6):640–647. doi: 10.1111/j.1365-2036.2009.04221.x. [DOI] [PubMed] [Google Scholar]
- 24.Fefferman DS, Farrell RJ. Endoscopy in inflammatory bowel disease: indications, surveillance, and use in clinical practice. Clin Gastroenterol Hepatol. 2005;3(1):11–24. doi: 10.1016/S1542-3565(04)00441-0. [DOI] [PubMed] [Google Scholar]
- 25.Leong RW, Ooi M, Corte C, Yau Y, Kermeen M, Katelaris PH, McDonald C, Ngu M. Full-spectrum endoscopy improves surveillance for dysplasia in patients with inflammatory bowel diseases. Gastroenterology. 2017;152(6):1337–1344.e1333. doi: 10.1053/j.gastro.2017.01.008. [DOI] [PubMed] [Google Scholar]
- 26.Le L, Fung BM, Tabibian JH. Capsule endoscopy for refractory Iron deficiency anemia in Crohn’s disease: captivating pathology hybrid therapy. Gastroenterology. 2018;155(2):276–277. doi: 10.1053/j.gastro.2018.01.055. [DOI] [PubMed] [Google Scholar]
- 27.Spiceland CM, Lodhia N. Endoscopy in inflammatory bowel disease: role in diagnosis, management, and treatment. World J Gastroenterol. 2018;24(35):4014–4020. doi: 10.3748/wjg.v24.i35.4014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Van Assche G, Dignass A, Panes J, Beaugerie L, Karagiannis J, Allez M, Ochsenkühn T, Orchard T, Rogler G, Louis E, et al. The second European evidence-based consensus on the diagnosis and management of Crohn’s disease: definitions and diagnosis. J Crohns Colitis. 2010;4(1):7–27. doi: 10.1016/j.crohns.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 29.Lee JL. Factors affecting surgical treatment with infliximab therapy in perianal fistula with Crohn disease. Ann Coloproctol. 2019;35(5):225–226. doi: 10.3393/ac.2019.10.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Patil SA, Cross RK. Medical versus surgical management of penetrating Crohn’s disease: the current situation and future perspectives. Expert Rev Gastroenterol Hepatol. 2017;11(9):843–848. doi: 10.1080/17474124.2017.1342536. [DOI] [PubMed] [Google Scholar]
- 31.Ponsioen CY, de Groof EJ, Eshuis EJ, Gardenbroek TJ, Bossuyt PMM, Hart A, Warusavitarne J, Buskens CJ, van Bodegraven AA, Brink MA, et al. Laparoscopic ileocaecal resection versus infliximab for terminal ileitis in Crohn’s disease: a randomised controlled, open-label, multicentre trial. Lancet Gastroenterol Hepatol. 2017;2(11):785–792. doi: 10.1016/S2468-1253(17)30248-0. [DOI] [PubMed] [Google Scholar]
- 32.Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12):4279–4295. doi: 10.1091/mbc.e02-02-0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, Lanino E, Sundberg B, Bernardo ME, Remberger M, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet. 2008, 371;(9624):1579–86. [DOI] [PubMed]
- 34.Forbes GM, Sturm MJ, Leong RW, Sparrow MP, Segarajasingam D, Cummins AG, Phillips M, Herrmann RP. A phase 2 study of allogeneic mesenchymal stromal cells for luminal Crohn’s disease refractory to biologic therapy. Clin Gastroenterol Hepatol. 2014;12(1):64–71. doi: 10.1016/j.cgh.2013.06.021. [DOI] [PubMed] [Google Scholar]
- 35.Whiteford MH, Kilkenny J, 3rd, Hyman N, Buie WD, Cohen J, Orsay C, Dunn G, Perry WB, Ellis CN, Rakinic J, et al. Practice parameters for the treatment of perianal abscess and fistula-in-ano (revised) Dis Colon Rectum. 2005;48(7):1337–1342. doi: 10.1007/s10350-005-0055-3. [DOI] [PubMed] [Google Scholar]
- 36.Song YP, Fang BJ, Wei XD, Zheng S: [A preliminary study on mechanisms for resistance of CML patient BM-derived bcr/abl+ and Flk1+CD31-CD34- stem cells to STI571 in vitro]. Zhong Shi Yan Xue Ye Xue Za Zhi 2005, 13(6):1004–1009. [PubMed]
- 37.García-Olmo D, García-Arranz M, Herreros D, Pascual I, Peiro C, Rodríguez-Montes JA. A phase I clinical trial of the treatment of Crohn’s fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum. 2005;48(7):1416–1423. doi: 10.1007/s10350-005-0052-6. [DOI] [PubMed] [Google Scholar]
- 38.da Silva ML, Chagastelles PC, Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119(Pt 11):2204–2213. doi: 10.1242/jcs.02932. [DOI] [PubMed] [Google Scholar]
- 39.Cao Y, Ding Z, Han C, Shi H, Cui L, Lin R. Efficacy of mesenchymal stromal cells for fistula treatment of Crohn’s disease: a systematic review and meta-analysis. Dig Dis Sci. 2017;62(4):851–860. doi: 10.1007/s10620-017-4453-x. [DOI] [PubMed] [Google Scholar]
- 40.Stang A, Jonas S, Poole C. Case study in major quotation errors: a critical commentary on the Newcastle-Ottawa scale. Eur J Epidemiol. 2018;33(11):1025–1031. doi: 10.1007/s10654-018-0443-3. [DOI] [PubMed] [Google Scholar]
- 41.Moskalewicz A, Oremus M. No clear choice between Newcastle-Ottawa scale and appraisal tool for cross-sectional studies to assess methodological quality in cross-sectional studies of health-related quality of life and breast cancer. J Clin Epidemiol. 2020;120:94–103. doi: 10.1016/j.jclinepi.2019.12.013. [DOI] [PubMed] [Google Scholar]
- 42.Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, Thomas J. Updated guidance for trusted systematic reviews: a new edition of the cochrane handbook for systematic reviews of interventions. Cochrane Database Syst Rev. 2019;10:Ed000142. doi: 10.1002/14651858.ED000142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Garcia-Olmo D, Herreros D, Pascual I, Pascual JA, Del-Valle E, Zorrilla J, De-La-Quintana P, Garcia-Arranz M, Pascual M. Expanded adipose-derived stem cells for the treatment of complex perianal fistula: a phase II clinical trial. Dis Colon Rectum. 2009;52(1):79–86. doi: 10.1007/DCR.0b013e3181973487. [DOI] [PubMed] [Google Scholar]
- 44.Guadalajara H, Herreros D, De-La-Quintana P, Trebol J, Garcia-Arranz M, Garcia-Olmo D. Long-term follow-up of patients undergoing adipose-derived adult stem cell administration to treat complex perianal fistulas. Int J Color Dis. 2012;27(5):595–600. doi: 10.1007/s00384-011-1350-1. [DOI] [PubMed] [Google Scholar]
- 45.Herreros MD, Garcia-Arranz M, Guadalajara H, De-La-Quintana P, Garcia-Olmo D. Autologous expanded adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistulas: a phase III randomized clinical trial (FATT 1: Fistula Advanced Therapy Trial 1) and long-term evaluation. Dis Colon Rectum. 2012;55(7):762–772. doi: 10.1097/DCR.0b013e318255364a. [DOI] [PubMed] [Google Scholar]
- 46.Garcia-Arranz M, Garcia-Olmo D, Herreros MD, Gracia-Solana J, Guadalajara H, de la Portilla F, Baixauli J, Garcia-Garcia J, Ramirez JM, Sanchez-Guijo F, et al. Autologous adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistula: a randomized clinical trial with long-term follow-up. Stem Cells Transl Med. 2020;9(3):295–301. doi: 10.1002/sctm.19-0271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Molendijk I, Bonsing BA, Roelofs H, Peeters KC, Wasser MN, Dijkstra G, van der Woude CJ, Duijvestein M, Veenendaal RA, Zwaginga JJ, et al. Allogeneic bone marrow-derived mesenchymal stromal cells promote healing of refractory perianal fistulas in patients with Crohn’s disease. Gastroenterology. 2015;149(4):918–927.e916. doi: 10.1053/j.gastro.2015.06.014. [DOI] [PubMed] [Google Scholar]
- 48.Panés J, García-Olmo D, Van Assche G, Colombel JF, Reinisch W, Baumgart DC, Dignass A, Nachury M, Ferrante M, Kazemi-Shirazi L, et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016;388(10051):1281–1290. doi: 10.1016/S0140-6736(16)31203-X. [DOI] [PubMed] [Google Scholar]
- 49.Panés J, García-Olmo D, Van Assche G, Colombel JF, Reinisch W, Baumgart DC, Dignass A, Nachury M, Ferrante M, Kazemi-Shirazi L, et al. Long-term efficacy and safety of stem cell therapy (cx601) for complex perianal fistulas in patients with Crohn’s disease. Gastroenterology. 2018;154(5):1334–1342.e1334. doi: 10.1053/j.gastro.2017.12.020. [DOI] [PubMed] [Google Scholar]
- 50.Zhou C, Li M, Zhang Y, Ni M, Wang Y, Xu D, Shi Y, Zhang B, Chen Y, Huang Y, et al. Autologous adipose-derived stem cells for the treatment of Crohn’s fistula-in-ano: an open-label, controlled trial. Stem Cell Res Ther. 2020;11(1):124. doi: 10.1186/s13287-020-01636-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Barnhoorn MC, Wasser M, Roelofs H, Maljaars PWJ, Molendijk I, Bonsing BA, Oosten LEM, Dijkstra G, van der Woude CJ, Roelen DL, et al. Long-term evaluation of allogeneic bone marrow-derived mesenchymal stromal cell therapy for Crohn’s disease perianal fistulas. J Crohns Colitis. 2020;14(1):64–70. doi: 10.1093/ecco-jcc/jjz116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Avivar-Valderas A, Martín-Martín C, Ramírez C, Del Río B, Menta R, Mancheño-Corvo P, Ortiz-Virumbrales M, Herrero-Méndez Á, Panés J, García-Olmo D, et al. Dissecting allo-sensitization after local administration of human allogeneic adipose mesenchymal stem cells in perianal fistulas of Crohn’s disease patients. Front Immunol. 2019;10:1244. doi: 10.3389/fimmu.2019.01244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Garcia-Olmo D, Guadalajara H, Rubio-Perez I, Herreros MD, de-la Quintana P, Garcia-Arranz M. Recurrent anal fistulae: limited surgery supported by stem cells. World J Gastroenterol. 2015;21(11):3330–3336. doi: 10.3748/wjg.v21.i11.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Herreros MD, Garcia-Olmo D, Guadalajara H, Georgiev-Hristov T, Brandariz L, Garcia-Arranz M. Stem cell therapy: a compassionate use program in perianal fistula. Stem Cells Int. 2019;2019:6132340. doi: 10.1155/2019/6132340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Choi S, Ryoo SB, Park KJ, Kim DS, Song KH, Kim KH, Chung SS, Shin EJ, Cho YB, Oh ST, et al. Autologous adipose tissue-derived stem cells for the treatment of complex perianal fistulas not associated with Crohn’s disease: a phase II clinical trial for safety and efficacy. Tech Coloproctol. 2017;21(5):345–353. doi: 10.1007/s10151-017-1630-z. [DOI] [PubMed] [Google Scholar]
- 56.Dozois EJ, Lightner AL, Mathis KL, Chua HK, Kelley SR, Fletcher JG, Dietz AB, Friton JJ, Butler GW, Faubion WA. Early results of a phase I trial using an adipose-derived mesenchymal stem cell-coated fistula plug for the treatment of transsphincteric cryptoglandular fistulas. Dis Colon Rectum. 2019;62(5):615–622. doi: 10.1097/DCR.0000000000001333. [DOI] [PubMed] [Google Scholar]
- 57.Topal U, Eray IC, Rencüzoğulları A, Yalav O, Alabaz Ö. Short-term results of adipose-derived stem cell therapy for the treatment of complex perianal fistula a single center experience. Ann Ital Chir. 2019;90:583–589. [PubMed] [Google Scholar]
- 58.Ciccocioppo R, Bernardo ME, Sgarella A, Maccario R, Avanzini MA, Ubezio C, Minelli A, Alvisi C, Vanoli A, Calliada F, et al. Autologous bone marrow-derived mesenchymal stromal cells in the treatment of fistulising Crohn’s disease. Gut. 2011;60(6):788–798. doi: 10.1136/gut.2010.214841. [DOI] [PubMed] [Google Scholar]
- 59.Cho YB, Lee WY, Park KJ, Kim M, Yoo HW, Yu CS. Autologous adipose tissue-derived stem cells for the treatment of Crohn’s fistula: a phase I clinical study. Cell Transplant. 2013;22(2):279–285. doi: 10.3727/096368912X656045. [DOI] [PubMed] [Google Scholar]
- 60.de la Portilla F, Alba F, García-Olmo D, Herrerías JM, González FX, Galindo A. Expanded allogeneic adipose-derived stem cells (eASCs) for the treatment of complex perianal fistula in Crohn’s disease: results from a multicenter phase I/IIa clinical trial. Int J Color Dis. 2013;28(3):313–323. doi: 10.1007/s00384-012-1581-9. [DOI] [PubMed] [Google Scholar]
- 61.Lee WY, Park KJ, Cho YB, Yoon SN, Song KH, Kim DS, Jung SH, Kim M, Yoo HW, Kim I, et al. Autologous adipose tissue-derived stem cells treatment demonstrated favorable and sustainable therapeutic effect for Crohn’s fistula. Stem Cells. 2013;31(11):2575–2581. doi: 10.1002/stem.1357. [DOI] [PubMed] [Google Scholar]
- 62.Ciccocioppo R, Gallia A, Sgarella A, Kruzliak P, Gobbi PG, Corazza GR. Long-term follow-up of Crohn disease fistulas after local injections of bone marrow-derived mesenchymal stem cells. Mayo Clin Proc. 2015;90(6):747–755. doi: 10.1016/j.mayocp.2015.03.023. [DOI] [PubMed] [Google Scholar]
- 63.Cho YB, Park KJ, Yoon SN, Song KH, Kim DS, Jung SH, Kim M, Jeong HY, Yu CS. Long-term results of adipose-derived stem cell therapy for the treatment of Crohn’s fistula. Stem Cells Transl Med. 2015;4(5):532–537. doi: 10.5966/sctm.2014-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Dietz AB, Dozois EJ, Fletcher JG, Butler GW, Radel D, Lightner AL, Dave M, Friton J, Nair A, Camilleri ET, et al. Autologous mesenchymal stem cells, applied in a bioabsorbable matrix, for treatment of perianal fistulas in patients with Crohn’s disease. Gastroenterology. 2017;153(1):59–62.e52. doi: 10.1053/j.gastro.2017.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lightner AL, Dozois EJ, Dietz AB, Fletcher JG, Friton J, Butler G, Faubion WA. Matrix-delivered autologous mesenchymal stem cell therapy for refractory rectovaginal Crohn’s fistulas. Inflamm Bowel Dis. 2020;26(5):670–677. doi: 10.1093/ibd/izz215. [DOI] [PubMed] [Google Scholar]
- 66.Dige A, Hougaard HT, Agnholt J, Pedersen BG, Tencerova M, Kassem M, Krogh K, Lundby L. Efficacy of injection of freshly collected autologous adipose tissue into perianal fistulas in patients with Crohn’s disease. Gastroenterology. 2019;156(8):2208–2216.e2201. doi: 10.1053/j.gastro.2019.02.005. [DOI] [PubMed] [Google Scholar]
- 67.Park KJ, Ryoo SB, Kim JS, Kim TI, Baik SH, Kim HJ, Lee KY, Kim M, Kim WH. Allogeneic adipose-derived stem cells for the treatment of perianal fistula in Crohn’s disease: a pilot clinical trial. Colorectal Dis. 2016;18(5):468–476. doi: 10.1111/codi.13223. [DOI] [PubMed] [Google Scholar]
- 68.García-Arranz M, Herreros MD, González-Gómez C, de la Quintana P, Guadalajara H, Georgiev-Hristov T, Trébol J, Garcia-Olmo D. Treatment of Crohn’s-related rectovaginal fistula with allogeneic expanded-adipose derived stem cells: a phase I-IIa clinical trial. Stem Cells Transl Med. 2016;5(11):1441–1446. doi: 10.5966/sctm.2015-0356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Scott LJ. Darvadstrocel: a review in treatment-refractory complex perianal fistulas in Crohn’s disease. BioDrugs. 2018;32(6):627–634. doi: 10.1007/s40259-018-0311-4. [DOI] [PubMed] [Google Scholar]
- 70.Wainstein C, Quera R, Fluxá D, Kronberg U, Conejero A, López-Köstner F, Jofre C, Zarate AJ. Stem cell therapy in refractory perineal Crohn’s disease: long-term follow-up. Colorectal Dis. 2018. [DOI] [PubMed]
- 71.Duijvestein M, Vos AC, Roelofs H, Wildenberg ME, Wendrich BB, Verspaget HW, Kooy-Winkelaar EM, Koning F, Zwaginga JJ, Fidder HH, et al. Autologous bone marrow-derived mesenchymal stromal cell treatment for refractory luminal Crohn’s disease: results of a phase I study. Gut. 2010;59(12):1662–1669. doi: 10.1136/gut.2010.215152. [DOI] [PubMed] [Google Scholar]
- 72.Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, Sun X, Chen J, Yang S, Cai J, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. Jama. 2012;307(11):1169–1177. doi: 10.1001/jama.2012.316. [DOI] [PubMed] [Google Scholar]
- 73.Reinders ME, Dreyer GJ, Bank JR, Roelofs H, Heidt S, Roelen DL, Zandvliet ML, Huurman VA, Fibbe WE, van Kooten C, et al. Safety of allogeneic bone marrow derived mesenchymal stromal cell therapy in renal transplant recipients: the neptune study. J Transl Med. 2015;13:344. doi: 10.1186/s12967-015-0700-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Muraro PA, Douek DC, Packer A, Chung K, Guenaga FJ, Cassiani-Ingoni R, Campbell C, Memon S, Nagle JW, Hakim FT, et al. Thymic output generates a new and diverse TCR repertoire after autologous stem cell transplantation in multiple sclerosis patients. J Exp Med. 2005;201(5):805–816. doi: 10.1084/jem.20041679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Shi Y, Wang Y, Li Q, Liu K, Hou J, Shao C, Wang Y. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat Rev Nephrol. 2018;14(8):493–507. doi: 10.1038/s41581-018-0023-5. [DOI] [PubMed] [Google Scholar]
- 76.Clevers H, Loh KM, Nusse R. Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science. 2014;346(6205):1248012. doi: 10.1126/science.1248012. [DOI] [PubMed] [Google Scholar]
- 77.Discher DE, Mooney DJ, Zandstra PW. Growth factors, matrices, and forces combine and control stem cells. Science. 2009;324(5935):1673–1677. doi: 10.1126/science.1171643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Panés J, Rimola J. Perianal fistulizing Crohn’s disease: pathogenesis, diagnosis and therapy. Nat Rev Gastroenterol Hepatol. 2017;14(11):652–664. doi: 10.1038/nrgastro.2017.104. [DOI] [PubMed] [Google Scholar]
- 79.Schwartz DA, Ghazi LJ, Regueiro M, Fichera A, Zoccali M, Ong EM, Mortelé KJ. Guidelines for the multidisciplinary management of Crohn’s perianal fistulas: summary statement. Inflamm Bowel Dis. 2015;21(4):723–730. doi: 10.1097/MIB.0000000000000315. [DOI] [PubMed] [Google Scholar]
- 80.American Gastroenterological Association medical position statement: perianal Crohn’s disease. Gastroenterology 2003, 125(5):1503–1507. [DOI] [PubMed]
- 81.Garcia-Olmo D, Garcia-Arranz M, Herreros D. Expanded adipose-derived stem cells for the treatment of complex perianal fistula including Crohn’s disease. Expert Opin Biol Ther. 2008;8(9):1417–1423. doi: 10.1517/14712598.8.9.1417. [DOI] [PubMed] [Google Scholar]
- 82.Mizuno H, Zuk PA, Zhu M, Lorenz HP, Benhaim P, Hedrick MH. Myogenic differentiation by human processed lipoaspirate cells. Plast Reconstr Surg. 2002;109(1):199–209. doi: 10.1097/00006534-200201000-00030. [DOI] [PubMed] [Google Scholar]
- 83.Vieira NM, Brandalise V, Zucconi E, Secco M, Strauss BE, Zatz M. Isolation, characterization, and differentiation potential of canine adipose-derived stem cells. Cell Transplant. 2010;19(3):279–289. doi: 10.3727/096368909X481764. [DOI] [PubMed] [Google Scholar]
- 84.Khanna R, Zou G, D’Haens G, Feagan BG, Sandborn WJ, Vandervoort MK, Rolleri RL, Bortey E, Paterson C, Forbes WP, et al. A retrospective analysis: the development of patient reported outcome measures for the assessment of Crohn’s disease activity. Aliment Pharmacol Ther. 2015;41(1):77–86. doi: 10.1111/apt.13001. [DOI] [PubMed] [Google Scholar]
- 85.Ryska O, Serclova Z, Mestak O, Matouskova E, Vesely P, Mrazova I. Local application of adipose-derived mesenchymal stem cells supports the healing of fistula: prospective randomised study on rat model of fistulising Crohn’s disease. Scand J Gastroenterol. 2017;52(5):543–550. doi: 10.1080/00365521.2017.1281434. [DOI] [PubMed] [Google Scholar]
- 86.Meng ZW, Baumgart DC. Darvadstrocel for the treatment of perianal fistulas in Crohn’s disease. Expert Rev Gastroenterol Hepatol. 2020;14(6):405–410. doi: 10.1080/17474124.2020.1764349. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data supporting the conclusions of this article are all online.