Abstract
Background
Postural orthostatic tachycardia syndrome (POTS) is characterized by excessive heart rate increase on standing and orthostatic intolerance. Previous data indicate autoimmune involvement. We studied serum activity against G protein–coupled receptors in relation to symptoms in patients with POTS and controls using a commercial cell‐based assay.
Methods and Results
Forty‐eight patients with POTS (aged 28.6±10.5 years; 44 women) and 25 healthy individuals (aged 30.7±8.6 years; 21 women) were included. The 10‐item Orthostatic Hypotension Questionnaire (OHQ) was completed by 33 patients with POTS and all controls. Human embryonic kidney 293 cells overexpressing one G protein–coupled receptor: adrenergic α1 receptor, adrenergic β2 receptor, cholinergic muscarinic type 2 receptor, and opioid receptor‐like 1 were treated with sera from all patients. Receptor response was analyzed using a β‐arrestin–linked transcription factor driving transgenic β‐lactamase transcription by fluorescence resonance energy transfer method. Receiver operating characteristic curves were constructed. G protein–coupled receptor activation was related to OHQ indices in linear regression models. Sera from patients with POTS activated all 4 receptors to a higher degree compared with controls (P<0.01 for all). The area under the curve was 0.88 (0.80–0.97, P<0.001) combining all 4 receptors. Adrenergic α1 receptor activation associated with OHQ composite score (β=0.77 OHQ points per SD of activity, P=0.009) and with reduced tolerability for prolonged standing (P=0.037) and walking for short (P=0.042) or long (P=0.001) periods. All 4 receptors were associated with vision problems (P<0.05 for all).
Conclusions
Our results indicate the presence of circulating proteins activating adrenergic, muscarinic, and nociceptin receptors in patients with POTS. Serum‐mediated activation of these receptors has high predictive value for POTS. Activation of adrenergic α1 receptor is associated with orthostatic symptoms severity in patients with POTS.
Keywords: adrenergic receptors, autoimmunity, G protein–coupled receptors, orthostatic intolerance, postural orthostatic tachycardia syndrome
Subject Categories: Autonomic Nervous System
Nonstandard Abbreviations and Acronyms
- ADRA1
adrenergic α1 receptor
- ADRB2
adrenergic β2 receptor
- CHRM2
cholinergic muscarinic type 2 receptor
- FRET
fluorescence resonance energy transfer
- GPCR
G protein–coupled receptor
- HR
heart rate
- IVIG
intravenous immunoglobulin
- OHDAS
Orthostatic Hypotension Daily Activity Scale
- OHQ
Orthostatic Hypotension Questionnaire
- OHSA
Orthostatic Hypotension Symptom Assessment
- OPRL1
opioid receptor‐like 1
- POTS
postural orthostatic tachycardia syndrome
Clinical Perspective
What Is New?
Serum of patients with postural orthostatic tachycardia syndrome (POTS) demonstrates activity against cardiovascular and nociceptive G protein–coupled receptors, and such activation is highly predictive of POTS diagnosis.
Serum‐mediated adrenergic α1 receptor activity is associated with the severity of orthostatic symptoms in POTS, independently of the orthostatic hemodynamic response.
What Are the Clinical Implications?
Our findings provide new insights in the pathophysiology of POTS and prompt further research on a possible autoimmune involvement in POTS.
Measurement of G protein–coupled receptor activity may be added as a diagnostic tool for POTS, even though the optimal panel of receptors and specific cutoff values are yet to be determined.
POTS is likely to be a heterogeneous disease, and it remains to be explored whether detection of autoimmune G protein–coupled receptor activity may identify different subtypes of POTS, potentially responding to different treatments.
Postural orthostatic tachycardia syndrome (POTS) is a disorder of unknown cause characterized by orthostatic intolerance and increased heart rate (HR) of 30 beats per minute during orthostasis, in the absence of orthostatic hypotension. 1 In addition to orthostatic intolerance, patients with POTS may experience debilitating symptoms only partly related or unrelated to orthostasis, including light‐headedness, nausea, blurred vision, fatigue, mental confusion (“brain fog”), chest pain, and gastrointestinal problems. 2 Syncope may occur although presyncopal symptoms are more common. Several potential underlying mechanisms have been suggested for POTS including autonomic denervation, hypovolemia, hyperadrenergic stimulation, and autoantibodies against adrenergic receptors. 2 , 3 , 4 However, none of these proposed mechanisms has yet led to effective treatment.
It has been observed that some patients develop POTS following an infection. This observation has given rise to the hypothesis of an autoimmune‐mediated cause of POTS. The fact that the majority of patients with POTS are women, who are more susceptible to developing autoimmune diseases, further supports the autoimmune hypothesis. 2 Additionally, nearly one fourth of patients with POTS have positive antinuclear antibodies and a higher prevalence of autoimmune diseases such as Hashimoto disease and Sjögren syndrome. 5 Finally, we and others have previously demonstrated presence of antibodies against adrenergic and cholinergic GPCRs (G protein–coupled receptors) in POTS. 6 , 7 , 8 , 9
GPCRs constitute a large family of receptors that detect molecules outside the cell and activate internal signaling pathways, ultimately leading to cellular responses. 10 , 11 Over time, antibodies against GPCRs have been linked to a spectrum of conditions, including POTS. 10 , 12 Thus, we aimed to expand previous observations by studying serum activity against specific GPCRs, cardiovascular ADRA1 (adrenergic α1 receptor), ADRB2 (adrenergic β2 receptor), CHRM2 (cholinergic muscarinic type 2 receptor), and nociception‐related OPRL1 (opioid receptor‐like 1) in patients with POTS and matched controls, and relate the serum‐mediated activation to specific orthostatic symptoms.
Methods
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Study Population
A total of 48 patients with POTS and 25 healthy controls were recruited for study from a tertiary referral center incorporating both syncope unit and cardiovascular autonomic laboratory at Skåne University Hospital, Malmö, Sweden. The patients and controls were recruited between January and December 2018. All patients with POTS had confirmed diagnoses by one of us with special expertise in POTS (A.F.). Blood samples were collected from all 73 patients and sent for analysis at the Center for Apheresis and Stem Cell Handling at Karolinska University Hospital in Stockholm, Sweden. Of the 73 participants, 48 (33 POTS and 25 controls) performed orthostatic tests and answered the Orthostatic Hypotension Questionnaire (OHQ) during their blood sample collection visit at the Clinical Research Unit at Skåne University Hospital in Malmö. Blood samples from the other 15 patients with POTS were collected and sent to Karolinska University Hospital from local hospitals and primary care facilities around Sweden, which is why these 15 participants did not complete the OHQ or orthostatic test at the time of their blood sample collection visit. All patients with POTS had a previous positive tilt test, which qualified them as confirmed cases in this study. Controls did not perform a formal tilt testing. Active standing tests were performed in both patients and controls during the study visit when the blood samples were collected and the study questionnaire was filled in. Controls’ active standing test results were negative. The study population is depicted in Figure 1. All patients provided written informed consent. The study was approved by the regional ethical review board in Lund (DNR 08/82 and 17/295) and all procedures were performed in accordance with the Declaration of Helsinki.
Figure 1. Study population flow chart.
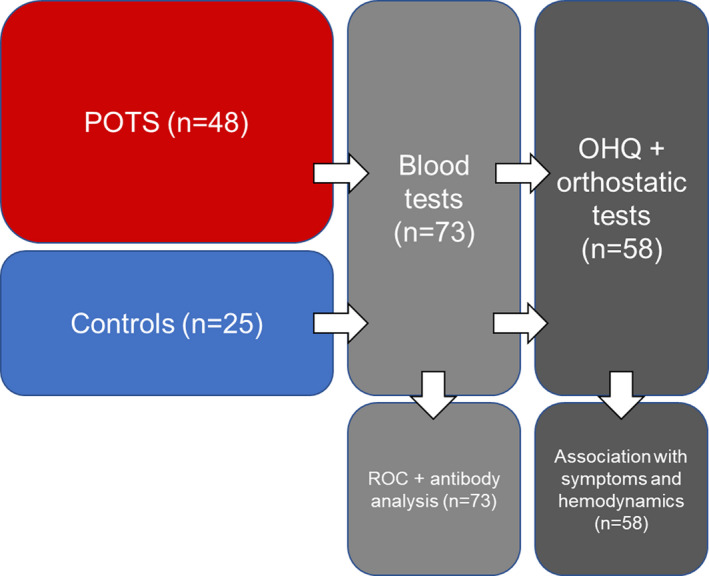
Orthostatic Hypotension Questionnaire (OHQ) and orthostatic test were completed by 33 patients with postural orthostatic tachycardia syndrome (POTS) and 25 controls at the time of blood sampling. ROC indicates receiver operating characteristic.
Orthostatic Hypotension Questionnaire
The OHQ is a questionnaire that has been previously validated and used for orthostatic hypotension 13 but has also been used for quantification of POTS‐related symptoms. 2 , 14 The OHQ is divided into 2 subgroups: Orthostatic Hypotension Symptom Assessment (OHSA) and Orthostatic Hypotension Daily Activity Scale (OHDAS). OHSA consists of 6 questions: (1) dizziness, light‐headedness, feeling faint, or feeling like you might blackout; (2) problems with vision (eg, blurring, seeing spots, and tunnel vision); (3) generalized weakness; (4) fatigue; (5) trouble concentrating; and (6) head/neck discomfort. OHDAS consists of 4 questions that assess the impact of symptoms on daily activities (standing for long and short duration, walking for short or long duration). The recall period is “over the past week.” The items are scored on a scale from 0 to 10, with 0 indicating no symptoms and 10 indicating the worst possible symptoms. The composite OHQ score is calculated by averaging the OHSAS and the OHDAS. Activities that are marked as zero or “cannot be done for other reasons” at baseline are not included in the scoring system. In this study, the OHQ was translated into Swedish and validated by an expert in the field of health status assessment.
Measurement of GPCR Activity
Sera from all patients with POTS and controls were analyzed by a fluorescence resonance energy transfer (FRET)–based reporter system (Tango GeneBLAzer, Thermo Fisher Scientific) based on a β2‐arrestin–linked transcription factor driving transgenic β‐lactamase transcription. The FRET‐based method has been previously described in greater detail. 15 Human embryonic kidney 293 cells overexpressing one of the GPCRs, ADRA1, ADRB2, CHRM2, and OPRL1, were plated and allowed to reattach during 48 hours. Cells were treated with 10% sera diluted in Roswell Park Memorial Institute medium for 5 hours, followed by addition of the FRET substrate, incubation for 60 minutes, and quantification analysis in a CLARIO Star multipurpose plate reader. GPCR activity was measured as the ratio between emission of cleaved and noncleaved FRET substrate. The selection of the ADRB2 over the ADRB1 was based on our previous results, 8 which showed that the combination of the 2 adrenergic receptors, α1 and β2, provides the highest discriminative efficacy in regard to patients with POTS (≈94%).
Statistical Analysis
OHQ scores were compared according to the median of serum activation, using independent samples Student t test. Receiver operating characteristic curves were constructed to analyze the predictive value of GPCR activity for POTS. A logistic model with all 4 GPCRs as POTS predictors was performed and a predicted value for every individual was calculated. Quantification of the activation of the GPCRs was log‐transformed and related to the OHQ composite and individual items scores in age‐adjusted linear regression models. In addition, the relationship between GPCR and the OHQ scores were tested in linear regression models, including the change in HR and systolic blood pressure after 3 minutes of orthostatic test as additional covariates. Data were analyzed using SPSS software version 25 (IBM). A 2‐sided P value <0.05 was considered significant for all tests. P values are displayed unadjusted for multiple testing; however, all results were interpreted accounting for multiple testing.
Results
Study Population Characteristics
The mean age in the POTS and control groups was 28.6±10.5 years and 30.7±8.6 years, respectively. Among patients with POTS, 44 (91.7%) were women, and among controls, 21 (84%) were women. Mean OHQ score was 6.36±1.68 in patients with POTS and 0.67±1.03 in controls (P<0.001). A total of 36 patients with POTS were treated with HR‐regulating and/or vasoactive agents by the time of the completion of the symptom questionnaire and when blood was drawn (Table S1). By inclusion criteria, no control patients were taking HR‐regulating or vasoactive medications. Three patients with POTS and 2 controls reported use of levothyroxine for hypothyroidism (P for difference in proportions between groups=0.799). Exclusion of these 5 patients from the analyses did not substantially change the results, which is why they are included in the following results. Study population characteristics are shown in Table 1.
Table 1.
Characteristics of the Study Population
| Patients With POTS (n=48) | Controls (n=25) | P Value | |
|---|---|---|---|
| Age, y | 28.6±10.5 | 30.7±8.6 | 0.394 |
| Women, n. | 91.7 | 84.0 | 0.320 |
| SBP, mm Hg | 116.73±12.46* | 113.60±8.33 | 0.283 |
| HR, beats per min | 69.39±12.27* | 65.14±9.87 | 0.161 |
| ΔSBP, 3 min | −1.00±7.91† | 1.24±5.97 | 0.252 |
| ΔHR, 3 min | 26.32±11.75‡ | 17.64±9.72 | 0.005 |
| OHQ score | 6.36±1.68* | 0.67±1.03 | < 0.001 |
Age, heart rate (HR), and systolic blood pressure (SBP) are expressed as mean±SD, whereas the proportion of women and different medications are expressed as percentages of total within each group. ΔHR and ΔSBP refer to the change in HR and SBP from supine to 3 minutes of active standing. P values denote P for independent samples t test or chi‐square test, respectively. OHQ indicates Orthostatic Hypotension Questionnaire; and POTS, postural orthostatic tachycardia syndrome.
Missing values: *n=15; †n=19; ‡n=17.
Receptor Activity in Patients With POTS Compared With Controls
The mean receptor activity was significantly higher in patients with POTS compared with controls for all 4 receptors (Figure 2A through 2D; Table S2). The obtained area under the curve was 0.88 (0.80–0.97, P<0.001) when analyzing all 4 receptors (Figure 3A). The area under the curve when analyzing receptors individually was 0.72 (0.58–0.85, P<0.001) for ADRA1 was 0.76 (0.64–0.88, P<0.001), for ADRB2 was 0.73 (0.60–0.87, P<0.001) for CHRM2, and 0.75 (0.62–0.88, P<0.001) for OPRL1, respectively (Figure 3B through 3E). Some GPCR activity was seen in all patients with POTS and all controls. However, 42 patients with POTS (87.5%) had at least one value above the 75th percentile in control patients for the respective receptor (Figure S1). Correlations for receptor activity in patients with POTS and controls are shown in Tables S3 and S4.
Figure 2. Receptor activation (y axis) shown as the ratio between emitted light from cleaved substrate and noncleaved substrate.
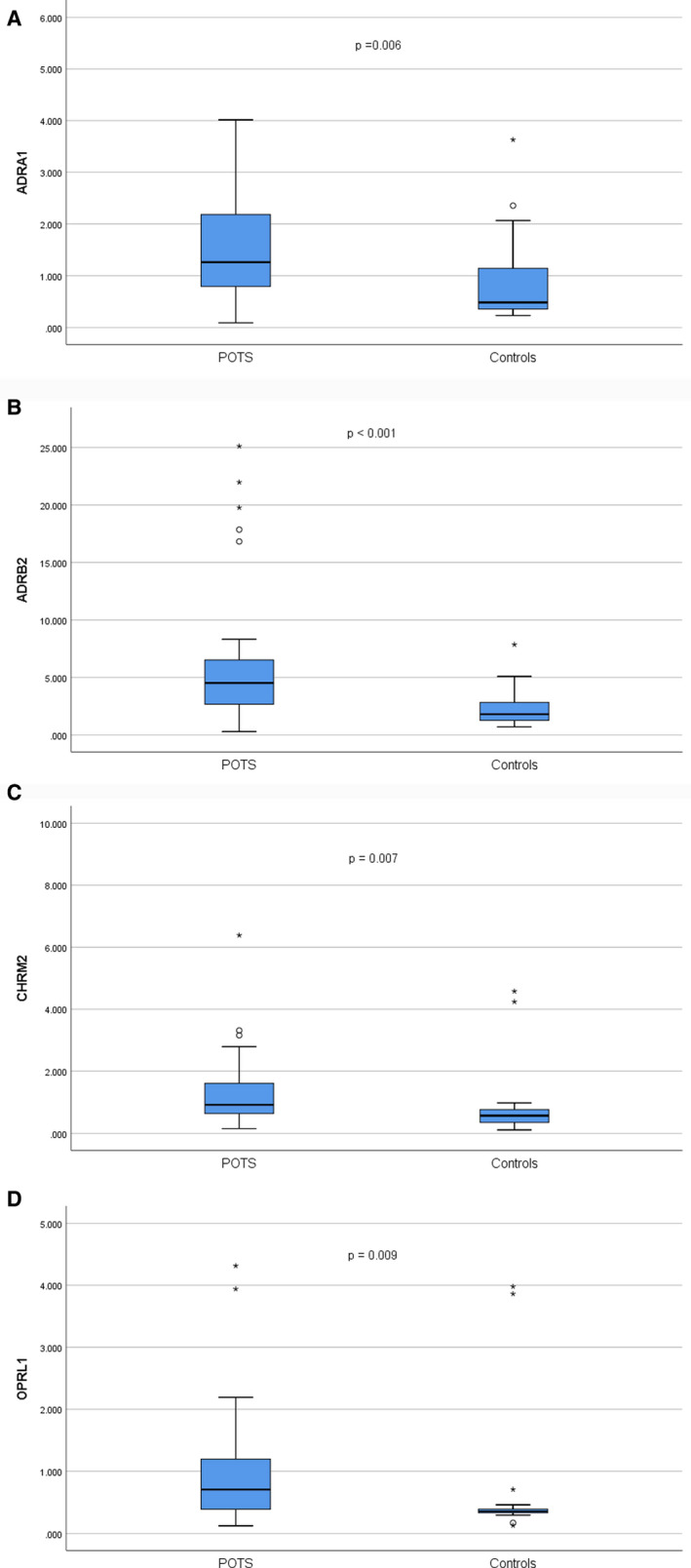
A, ADRA1 (adrenergic α1 receptor) activation in patients with postural orthostatic tachycardia syndrome (POTS) and controls. B, ADRB2 (adrenergic β2 receptor) activation in patients with POTS and controls. C, CHRM2 (cholinergic muscarinic 2 receptor) activation in patients with POTS and controls. D, OPRL1 (opioid‐receptor‐like 1) activation in patients with POTS and controls. P values denote the difference between mean values, using independent samples t test for the log‐transformed receptor activity. Please note that by design, one extreme outlier in the POTS group with a value of 14.838 for ADRA1 activity and 26.709 for CHRM2 activity, respectively, is not displayed in the figures.
Figure 3. The predictive value of specific G protein–coupled receptors (GPCRs) in postural orthostatic tachycardia syndrome (POTS).
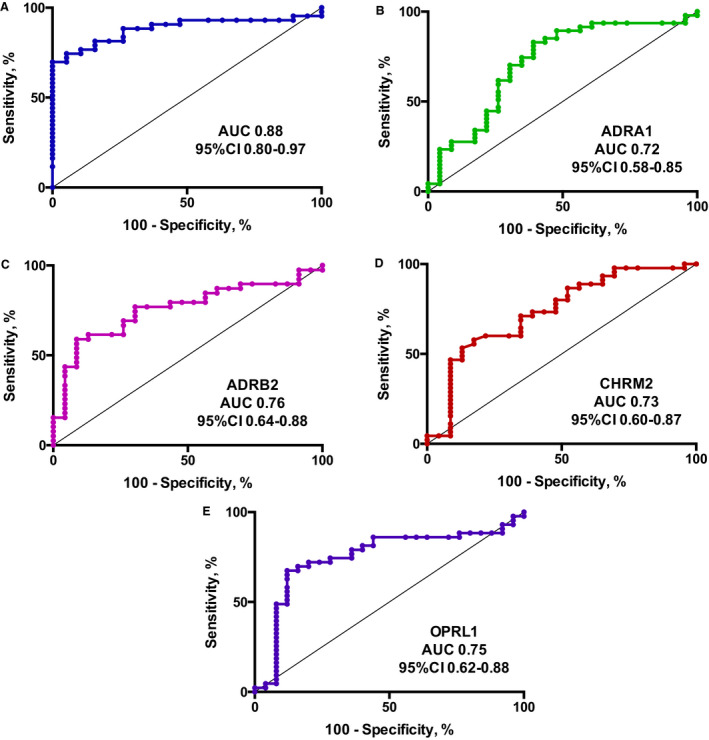
Receiver operating characteristic (ROC) curves for all 4 GPCRs (A) and the individual receptors (B through E) for the diagnosis of POTS in the 73 patients. A, ROC curve for all 4 GPCRs (ADRA1 [adrenergic α1 receptor], ADRB2 [adrenergic β2 receptor], CHRM2 [cholinergic muscarinic 2 receptor], and OPRL1 [opioid receptor‐like 1]) for the diagnosis of POTS. B, ROC curve for ADRA1 for the diagnosis of POTS. C, ROC curve for ADRB2 for the diagnosis of POTS. D, ROC curve for CHRM2 for the diagnosis of POTS. E, ROC curve for OPRL1 for the diagnosis of POTS. AUC indicates area under curve.
Association Between Receptor Activity and OHQ Score in Patients With POTS
The OHQ composite score was higher in those patients with above the median serum ADRA1 activation (P=0.043), but not in ADRB2, CHRM2, or OPRL1 in patients with POTS (Table 2). There were no significant associations in the control group.
Table 2.
Severity of Symptoms in Relation to Receptor Activity in Patients With POTS (n=33)
| ADRA1 Above the Median | ADRA1 Below the Median | P Value | |
|---|---|---|---|
| OHQ score | 6.94±1.18 | 5.74±1.95 | 0.043* |
| ADRB2 Above the Median | ADRB2 Below the Median | ||
|---|---|---|---|
| OHQ score | 6.42±1.41 | 6.14±1.88 | 0.638* |
| CHRM2 Above the Median | CHRM2 Below the Median | ||
|---|---|---|---|
| OHQ score | 6.62±1.30 | 5.95±1.93 | 0.260* |
| OPRL1 Above the Median | OPRL1 Below the Median | ||
|---|---|---|---|
| OHQ score | 6.39±1.67 | 6.32±1.74 | 0.904 |
The composite Orthostatic Hypotension Questionnaire (OHQ) score in relation to specific receptor activation. ADRA1 indicates adrenergic α1 receptor; ADRB2, adrenergic β2 receptor; CHRM2, cholinergic muscarinic 2 receptor; OPRL1, opioid receptor‐like 1; and POTS, postural orthostatic tachycardia syndrome.
Missing values: *n=1.
Serum ADRA1 activation was associated with the OHQ composite score (β=0.77, OHQ points per SD of activity; P=0.009), whereas there were no significant associations among controls (P=0.953). The association between ADRA1 and total OHQ also remained significant after adjusting for increase in HR and decrease in systolic blood pressure after 3 minutes (P=0.031). ADRA1 activation was associated with symptoms during prolonged standing (P=0.037) and walking for short (P=0.042) or long (P=0.001) periods. All 4 receptors were associated with a higher score for vision problems (ADRA1, P<0.001; ADRB2, P=0.011; CHRM2, P=0.014; and OPRL1, P=0.003). In addition, OPRL1 activity was associated with symptoms during prolonged walking (P=0.035). The activity of ADRA1, ARB2, CHRM2, and OPRL1 was not associated with OHQ composite score in controls. The full results are shown in Table S5.
A detailed clinical assessment revealed that 2 patients in the control group had treated hypothyroidism and another patient had rheumatoid arthritis. However, results did not differ when we excluded these 3 patients from the control group (data not shown).
Discussion
We report that sera from patients with POTS activate the 4 GPCRs, ADRA1A, ADRB2, CHRM2, and OPRL1, to a significantly higher degree compared with sera from controls. We also show that such GPCR activity is highly predictive of POTS, as indicated by receiver operating characteristic analyses. The ADRA1 activity is associated with severity of orthostatic symptoms, measured by the OHQ score, in patients with POTS and this association is partly independent of the hemodynamic response during orthostasis. Moreover, activity in all 4 GPCRs correlates specifically with symptoms of disturbed vision.
Role of GPCRs in POTS
The present results are in line with previous research indicating that there may be autoimmune involvement in POTS, targeting specific GPCRs. Previous studies have detected various autoantibodies in POTS. 6 , 7 , 9 , 16 , 17 In the present study, activity towards specific GPCRs rather than presence of autoantibodies was measured. The FRET‐based method detects changes in receptor conformation, which can be caused by allosteric or orthosteric binding of a ligand or antibody. The hypothesis is that these conformational changes seen in the present study could be caused by autoantibodies. After binding to GPCR, autoantibodies can yield stimulatory and inhibitory effects. 11 Previous studies have detected increased levels of autoantibodies against ADRA1 and ADRAB1/2 in patients with POTS compared with healthy controls. 6 , 7 Interestingly, IgG from POTS was found to shift the ADRA1 dose‐response curve to the right after phenylephrine administration, indicating a partial antagonistic effect on ADRA1. This could potentially block the effects of endogenous norepinephrine on ADRA1, which, in turn, would lead to impaired vasoconstriction and increased baroreceptor activation, consecutively leading to increased sympathetic activity. In contrast, the ADRB1/2 effect was found to be the opposite and shifted the ADRB1/2 dose‐response curve to the left after isoproterenol administration; hence, the IgG from POTS had a stimulatory effect on ADRB1/2. The relatively unprotected ADRB1/2 would respond to increased sympathetic activity and circulating catecholamines with reflex tachycardia. 6 , 7 These inhibitory and stimulatory effects provide an interesting pathophysiological explanation for the cardiovascular effects associated with upright posture in patients with POTS.
The predictive value of GPCR activity for a diagnosis of POTS was analyzed using receiver operating characteristic, showing an excellent prediction of the diagnosis in POTS (area under the curve, 0.88) when combining all 4 receptors. The area under the curve for the individual receptors were between 0.72 and 0.76. These findings indicate that measurement of GPCR activity may be added as a diagnostic tool for POTS, even though the optimal panel of receptors and specific cutoff values are yet to be determined. POTS is likely to be a heterogeneous disease, and it remains to be explored whether detection of autoimmune GPCR activity may identify different subtypes of POTS, potentially responding to different treatments.
Orthostatic Symptoms in Relation to Receptor Activity
A correlation between the severity of symptoms and the presence of autoantibodies in POTS has been observed in a previous study by Gunning et al. This study used a method of calculating symptom severity in OH, another form of orthostatic intolerance with overlap in clinical presentation, revealing a weak correlation with all 9 receptor subtypes of autoantibody concentration (ADRA1/2, ADRB1/2, and CHRM1–5) and the severity of orthostatic symptoms. 9 The same study indicated that 89% of patients with POTS had autoantibodies against ADRA1 whereas antibodies against other adrenergic and muscarinic antibodies were less prevalent. Interestingly, there was a tendency that subtypes of both adrenergic (α2, β1, and β2) and muscarinic receptor antibodies were not detected in the sera of patients with POTS, unless autoantibodies were expressed against the α1 adrenergic receptor subtype. 9 In our study, ADRA1 activity was more strongly associated with the orthostatic symptoms compared with ADRB2, CHRM2 and OPRL1. Thus, ADRA1 may play a particularly important role in POTS. It should be emphasized that in the study by Gunning et al, 9 the authors applied a different antibody detection method, ELISA, which does not include the whole cell assay but only specific isolated epitopes derived from GPCRs.
In addition to adrenergic receptors, we included CHRM2 and OPRL1. CHRM2 autoantibodies were first detected in patients with Chagas disease and have later been found in patients with dilated cardiomyopathy. 11 The CHRM2 was reported to have a negative chronotropic effect in cultured cardiomyocytes. 18 In our study, CHRM2 activity was greater in patients with POTS compared with controls but did not correlate with symptom severity. This is in contrast to a previous study, where symptoms correlated with all 5 different muscarinic receptors. 9 The strongest correlation in that study; however, was seen in CHRM4, which was not included in our study. 9 In contrast to adrenergic and muscarinic receptors, there are no published studies regarding the role of OPRL1 in POTS and other syndromes of orthostatic intolerance. The OPRL1 is involved in pain perception in humans. 19 Thus, OPRL1 may provide a clue to the symptom of chronic pain at various locations, which is often described by patients with POTS. 2 OPRL1 activity was greater in patients with POTS compared with controls and was associated with vision problems and walking for long distances. However, the associations were not strong. It is still unclear whether the increased activities seen in CHRM2 and OPRL1 are caused by stimulatory or inhibitory effects and the specific role of these 2 receptors in POTS is yet to be further explored.
As demonstrated in the present study, there was strong association between vision disturbances and all 4 GPCRs in patients with POTS. Disturbed vision is a common symptom of orthostatic intolerance. The retina is more susceptible to hypoperfusion than the brain, because of the presence of the intraocular pressure, which adds an impediment to eye perfusion that is not present in cerebral circulation. 20 However, the decrease in blood pressure when upright in patients with POTS is usually only modest or nonexistent, unless the patient experiences presyncope/syncope, caused by vasovagal reflex activation. 3 The autonomic nervous system influences numerous ocular functions such as controlling pupil size, accommodation of the lens, regulation of ocular blood flow, and intraocular pressure. 21 This may explain why patients with autonomic dysfunction, such as those with POTS, may experience impaired vision. Interestingly, OPRL1 was also associated with impaired vision. Of interest in relation to our findings, opioid receptors have been implicated in the regulation of iris function 22 and regulation of intraocular pressure, 23 which could explain why patients with increased activity in OPRL1 might have vision disturbances.
Immunomodulatory Treatments in Patients With POTS
The present results support the hypothesis that POTS is an autoimmune disease, which, in turn, may suggest that some patients with POTS could benefit from immunomodulatory therapies. Controlled treatment trials are in progress to determine whether immunomodulatory therapies may be effective in certain POTS subgroups. 12 At this time, a few case reports have been published describing improvements in POTS symptoms after intravenous immunoglobulin (IVIG), rituximab, autologous adipose stem cell infusions, and plasmapheresis in highly selected cases with comorbid autoimmunity. 24 , 25 , 26 , 27 , 28 A case report on a 32‐year‐old patient with POTS demonstrated a positive response to plasma exchange and improved OHQ score. 29
A retrospective study 30 including 38 patients with dysautonomia of various types, including 26 patients with POTS, reported that 83.5% improved while taking IVIG. The mean time to the first sign of response was 5.3 weeks and the study reported no serious adverse events. GPCRs were not analyzed but antiphospholipid antibodies and novel Sjögren antibodies were often found to be present and correlated with a high response rate to IVIG. There is increasing evidence that IVIG is safe and effective in a subset of patients with autonomic disorders and evidence of autoimmunity. According to this study, 29 a 4‐month IVIG trial should be considered in severely affected patients who are refractory to lifestyle and pharmacological therapies. Taken together, these above reports on efficacy of immunomodulatory therapy in select patients with POTS lack both systematic assessment of serum positivity for autoantibodies against GPCRs as entrance criterion and randomization of patients to a placebo‐controlled arm.
Limitations
Our study has a number of important limitations. First, the sample size is small and our findings should be externally validated in larger cohorts. Second, the OHQ was previously validated in orthostatic hypotension and not specifically in POTS. In addition to symptoms of orthostatic intolerance, patients with POTS may demonstrate a number of additional symptoms, including cognitive impairment, gastrointestinal problems, and unexplained pain, 2 which are not specifically captured by the OHQ. Third, as already described in the Methods section, we did not measure ADRB1 activity. Fourth, the majority of patients with POTS were treated with HR‐regulating and/or vasoactive medications when completing symptom questionnaire and when the blood samples were drawn. However, these patients still reported significant symptoms, as indicated by the OHQ. Finally, as previously mentioned, receptor activity rather than antibodies was measured in the current study, and it is possible that the conformational changes observed in the GPCR could be the result of something other than autoantibodies. However, since several previous studies have identified antibodies against these GPCRs, it may be permissible to assume that the increased activity in the specific receptors is a consequence of autoimmune disease.
Conclusions
Serum of patients with POTS demonstrates activity against cardiovascular and nociceptive GPCRs and such activation is highly predictive of POTS diagnosis. Serum‐mediated ADRA1A activity is associated with the severity of orthostatic symptoms in patients with POTS, independently of the orthostatic hemodynamic response. These findings provide new insights into the pathophysiology of POTS and prompt further research on possible autoimmune involvement in patients with POTS.
Sources of Funding
This study was supported by grants from the Swedish Heart‐Lung Foundation, Solidex, the Swedish Heart and Lung Association, the Medical Faculty of Lund University, ALF Funds, Skåne University Hospital Funds, the Crafoord Foundation, Ernhold Lundströms Research Foundation, Region Skåne, Hulda and Conrad Mossfelt Foundation, and Anna‐Lisa and Sven Eric Lundgren Foundation for Medical Research.
Disclosures
Fedorowski reports personal fees from Medtronic Inc and Biotronik outside the submitted work. Hamrefors reports an educational congress grant from Boston Scientific Inc outside of the submitted work. Sutton reports personal fees and other from Medtronic Inc. and St. Jude Medical Inc. (Abbott Laboratories) outside the submitted work; is a member of the Speakers’ Bureau at St. Jude Medical/Abbott Inc.; is a shareholder in Boston Scientific Inc. and Edwards Lifesciences Inc; and performs consultancy for Medtronic Inc. The remaining authors have no disclosures to report.
Supporting information
Tables S1–S5
Figure S1
Acknowledgments
We would like to thank Jenny Persson‐Tholin and her colleagues at the Clinical Research Unit at Skåne University Hospital in Malmö for their devoted assistance in this study.
Serum Activity Against G Protein–Coupled Receptors and Severity of Orthostatic Symptoms in Postural Orthostatic Tachycardia Syndrome. (J Am Heart Assoc. 2020;9:e015989 DOI: 10.1161/JAHA.120.015989.)
For Sources of Funding and Disclosures, see page 9.
References
- 1. Schondorf R, Low PA. Idiopathic postural orthostatic tachycardia syndrome: an attenuated form of acute pandysautonomia? Neurology. 1993;43:132–137. [DOI] [PubMed] [Google Scholar]
- 2. Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019;285:352–366. [DOI] [PubMed] [Google Scholar]
- 3. Sheldon RS, Grubb BP, Olshansky B, Shen WK, Calkins H, Brignole M, Raj SR, Krahn AD, Morillo CA, Stewart JM, et al. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12:e41–e63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Arnold AC, Ng J, Raj SR. Postural tachycardia syndrome—diagnosis, physiology, and prognosis. Auton Neurosci. 2018;215:3–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Blitshteyn S. Autoimmune markers and autoimmune disorders in patients with postural tachycardia syndrome (POTS). Lupus. 2015;24:1364–1369. [DOI] [PubMed] [Google Scholar]
- 6. Li H, Yu X, Liles C, Khan M, Vanderlinde‐Wood M, Galloway A, Zillner C, Benbrook A, Reim S, Collier D, et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc. 2014;3:e000755 DOI: 10.1161/JAHA.113.000755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Fedorowski A, Li H, Yu X, Koelsch KA, Harris VM, Liles C, Murphy TA, Quadri SMS, Scofield RH, Sutton R, et al. Antiadrenergic autoimmunity in postural tachycardia syndrome. Europace. 2017;19:1211–1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Yu X, Li H, Murphy TA, Nuss Z, Liles J, Liles C, Aston CE, Raj SR, Fedorowski A, Kem DC. Angiotensin II type 1 receptor autoantibodies in postural tachycardia syndrome. J Am Heart Assoc. 2018;7:e008351 DOI: 10.1161/JAHA.117.008351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Gunning WT III, Kvale H, Kramer PM, Karabin BL, Grubb BP. Postural orthostatic tachycardia syndrome is associated with elevated G‐protein coupled receptor autoantibodies. J Am Heart Assoc. 2019;8:e013602 DOI: 10.1161/JAHA.119.013602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Luft FC. Activating autoantibodies and cardiovascular disease. Physiology (Bethesda). 2013;28:254–261. [DOI] [PubMed] [Google Scholar]
- 11. Wallukat G, Schimke I. Agonistic autoantibodies directed against G‐protein‐coupled receptors and their relationship to cardiovascular diseases. Semin Immunopathol. 2014;36:351–363. [DOI] [PubMed] [Google Scholar]
- 12. Vernino S, Stiles LE. Autoimmunity in postural orthostatic tachycardia syndrome: current understanding. Auton Neurosci. 2018;215:78–82. [DOI] [PubMed] [Google Scholar]
- 13. Kaufmann H, Malamut R, Norcliffe‐Kaufmann L, Rosa K, Freeman R. The Orthostatic Hypotension Questionnaire (OHQ): validation of a novel symptom assessment scale. Clin Auton Res. 2012;22:79–90. [DOI] [PubMed] [Google Scholar]
- 14. Wells R, Spurrier AJ, Linz D, Gallagher C, Mahajan R, Sanders P, Page A, Lau DH. Postural tachycardia syndrome: current perspectives. Vasc Health Risk Manag. 2018;14:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hanson BJ, Wetter J, Bercher MR, Kopp L, Fuerstenau‐Sharp M, Vedvik KL, Zielinski T, Doucette C, Whitney PJ, Revankar C. A homogeneous fluorescent live‐cell assay for measuring 7‐transmembrane receptor activity and agonist functional selectivity through beta‐arrestin recruitment. J Biomol Screen. 2009;14:798–810. [DOI] [PubMed] [Google Scholar]
- 16. Li J, Zhang Q, Liao Y, Zhang C, Hao H, Du J. The value of acetylcholine receptor antibody in children with postural tachycardia syndrome. Pediatr Cardiol. 2015;36:165–170. [DOI] [PubMed] [Google Scholar]
- 17. Watari M, Nakane S, Mukaino A, Nakajima M, Mori Y, Maeda Y, Masuda T, Takamatsu K, Kouzaki Y, Higuchi O, et al. Autoimmune postural orthostatic tachycardia syndrome. Ann Clin Transl Neurol. 2018;5:486–492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wallukat G, Nissen E, Morwinski R, Muller J. Autoantibodies against the beta‐ and muscarinic receptors in cardiomyopathy. Herz. 2000;25:261–266. [DOI] [PubMed] [Google Scholar]
- 19. Al‐Hasani R, Bruchas MR. Molecular mechanisms of opioid receptor‐dependent signaling and behavior. Anesthesiology. 2011;115:1363–1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wieling W, Thijs RD, van Dijk N, Wilde AA, Benditt DG, van Dijk JG. Symptoms and signs of syncope: a review of the link between physiology and clinical clues. Brain. 2009;132:2630–2642. [DOI] [PubMed] [Google Scholar]
- 21. McDougal DH, Gamlin PD. Autonomic control of the eye. Compr Physiol. 2015;5:439–473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Murray RB, Adler MW, Korczyn AD. The pupillary effects of opioids. Life Sci. 1983;33:495–509. [DOI] [PubMed] [Google Scholar]
- 23. Drago F, Panissidi G, Bellomio F, Dal Bello A, Aguglia E, Gorgone G. Effects of opiates and opioids on intraocular pressure of rabbits and humans. Clin Exp Pharmacol Physiol. 1985;12:107–113. [DOI] [PubMed] [Google Scholar]
- 24. Adamec I, Bilic E, Lovric M, Habek M. Postural orthostatic tachycardia syndrome (POTS) as presenting symptom of CIDP. Neurol Sci. 2016;37:1163–1166. [DOI] [PubMed] [Google Scholar]
- 25. Weinstock LB, Brook JB, Myers TL, Goodman B. Successful treatment of postural orthostatic tachycardia and mast cell activation syndromes using naltrexone, immunoglobulin and antibiotic treatment. BMJ Case Rep. 2018;2018:bcr2017221405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hendrickson JE, Hendrickson ET, Gehrie EA, Sidhu D, Wallukat G, Schimke I, Tormey CA. Complex regional pain syndrome and dysautonomia in a 14‐year‐old girl responsive to therapeutic plasma exchange. J Clin Apher. 2016;31:368–374. [DOI] [PubMed] [Google Scholar]
- 27. Numan MT, Kamdar A, Young J, Butler IJ. Autologous adipose stem cell therapy for autonomic nervous system dysfunction in two young patients. Stem Cells Dev. 2017;26:391–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Blitshteyn S, Brook J. Postural tachycardia syndrome (POTS) with anti‐NMDA receptor antibodies after human papillomavirus vaccination. Immunol Res. 2017;65:282–284. [DOI] [PubMed] [Google Scholar]
- 29. Wells R, Hissaria P, Elliott AD, Sanders P, Page A, Baumert M, Lau DH. Plasma exchange therapy in postural tachycardia syndrome: a novel long‐term approach? Am J Med. 2020;133:e157–e159. [DOI] [PubMed] [Google Scholar]
- 30. Schofield JR, Chemali KR. Intravenous immunoglobulin therapy in refractory autoimmune dysautonomias: a retrospective analysis of 38 patients. Am J Ther. 2019;26:570–582. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Tables S1–S5
Figure S1


