Abstract
Background
Emerging evidence suggests that sodium-glucose cotransporter-2 (SGLT-2) inhibitors and glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are associated with decreased risk of cardiovascular and renal events in type 2 diabetes mellitus (DM) patients. However, no study to date has compared the effect of SGLT-2 inhibitors with that of GLP-1 RAs in type 2 DM patients with chronic kidney disease (CKD). We herein investigated the benefits of SGLT-2 inhibitors and GLP-1 RAs in CKD patients.
Methods
We performed a systematic literature search through November 2020. We selected randomized control trials that compared the risk of major adverse cardiovascular events (MACE) and a composite of renal outcomes. We performed a network meta-analysis to compare SGLT-2 inhibitors with GLP-1 RAs indirectly. Risk ratios (RRs) with corresponding 95% confidence intervals (CI) were synthesized.
Results
Thirteen studies were selected with a total of 32,949 patients. SGLT-2 inhibitors led to a risk reduction in MACE and renal events (RR [95% CI]; 0.85 [0.75–0.96] and 0.68 [0.59–0.78], respectively). However, GLP-1 RAs did not reduce the risk of cardiovascular or renal adverse events (RR 0.91 [0.80–1.04] and 0.86 [0.72–1.03], respectively). Compared to GLP-1 RAs, SGLT-2 inhibitors did not demonstrate a significant difference in MACE (RR 0.94 [0.78–1.12]), while SGLT-2 inhibitors were associated with a lower risk of renal events compared to GLP-1 RAs (RR 0.79 [0.63–0.99]). A sensitivity analysis revealed that GLP-1 analogues significantly decreased MACE when compared to placebo treatment (RR 0.81 [0.69–0.95]), while exendin-4 analogues did not (RR 1.03 [0.88–1.20]).
Conclusions
In patients with type 2 DM and CKD, SGLT-2 inhibitors were associated with a decreased risk of cardiovascular and renal events, but GLP-1 RAs were not. SGLT-2 inhibitors significantly decreased the risk of renal events compared to GLP-1 RAs. Among GLP-1 RAs, GLP-1 analogues showed a positive impact on cardiovascular and renal outcomes, while exendin-4 analogues did not.
Keywords: SGLT2 inhibitors, GLP-1 receptor agonist, Meta-analysis, Cardiovascular disease, Renal outcomes, Diabetes mellitus, Chronic kidney disease
Background
Diabetes mellitus (DM) is a major public health problem with a high prevalence. The International Diabetes Federation estimated that 351.7 million people of working age (20–64 years) had DM in 2019, and this number is expected to increase to 417.3 million by 2030 [1]. Type 2 DM is the leading cause of chronic kidney disease (CKD), accounting for roughly 36% of adult CKD in the United States [2]. CKD with DM can progress to end-stage renal disease (ESRD) [3], which confers a poor overall prognosis. Moreover, type 2 DM and CKD increase the risk of cardiovascular disease [4, 5]. Therefore, the prevention of CKD progression and cardiovascular events is essential for the management of patients with type 2 DM and CKD.
Sodium-glucose cotransporter-2 (SGLT-2) inhibitors are a new class of glucose-lowering agents. SGLT-2 inhibitors function through reducing renal tubular glucose reabsorption, thereby lowering blood glucose without stimulating insulin release. Several large cohort studies and randomized controlled trials (RCTs) have demonstrated favorable cardiovascular and renal outcomes associated with SGLT-2 inhibitors [6–10]. Moreover, recent RCTs revealed that SGLT-2 inhibitors are associated with favorable cardiovascular outcomes in CKD patients [11, 12].
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) decrease hemoglobin A1c by both stimulating glucose-dependent insulin secretion and reducing glucagon secretion [13]. GLP-1 RAs are known to reduce blood pressure (BP) [14] and body weight [15]. Large cohort studies and meta-analyses of RCTs demonstrated that GLP-1 RAs improve cardiovascular outcomes [16–20] The American Diabetes Association (ADA) recommends SGLT-2 inhibitors or GLP-1 RAs in type 2 DM patients who have atherosclerotic cardiovascular disease or kidney disease [21]. However, it remains unclear if GLP-1 RAs are beneficial to type 2 DM patients with CKD as well.
Several studies have investigated the impact of these novel agents on cardiovascular disease prevention; they show that SGLT-2 inhibitors and GLP-1 are comparable in their ability to decrease cardiovascular and renal events [18, 22]. However, no study has compared the effect of SGLT-2 inhibitors on renal and cardiovascular diseases with that of GLP-1 RAs in CKD patients, who are at a high risk of morbidity. We herein investigate the benefits of SGLT-2 inhibitors and GLP-1 RAs in CKD patients by network meta-analysis.
Methods
Literature search
The search strategy was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) extension statement for network meta-analysis [23, 24]. We performed a systematic search of PubMed, Medline, EMBASE, and the Cochrane Library from inception to November 20, 2020. The following keywords were applied: (“sodium-glucose cotransporter-2 inhibitors” [MeSH] OR “SGLT-2 inhibitor” OR SGLT-2 OR “empagliflozin” OR “dapagliflozin” OR “canagliflozin” OR “luseogliflozin” OR “ertugliflozin”) OR (GLP-1 OR “glucagon-like peptide-1 receptor “[MeSH] OR “glucagon-like peptide-1 receptor agonist” OR “exenatide” OR “liraglutide” OR “lixisenatide” OR “semaglutide” OR “dulaglutide” OR “albiglutide”) AND (“diabetes mellitus, type 2” [MeSH] OR “diabetes mellitus type 2” OR “type 2 diabetes mellitus” OR DM or diabetes) AND (“renal insufficiency, chronic” [MeSH] or CKD or “chronic kidney disease” or “kidney disease” or “kidney failure” or CKF or “chronic kidney failure” or “renal failure” or CRF or CRD or “chronic renal disease”) AND (“myocardial infarction” [MeSH] OR MI OR “coronary artery disease” [MeSH] OR “cardiovascular disease” [MeSH] OR “cerebrovascular disorders” [MeSH] OR “stroke” [MeSH] OR CVA OR “cerebrovascular accident” OR MACE OR “major adverse cardiovascular event” OR “death” OR mortality [MeSH] OR “all-cause mortality” OR “cardiovascular mortality” OR “heart failure” OR “end stage renal disease” OR “renal failure” OR “kidney failure” OR ESRD OR “renal death” OR “albuminuria” OR “urine albumin” OR “proteinuria” OR “urine protein”).
We restricted the search to human studies. Reference lists included in meta-analysis studies were reviewed to minimize missing relevant studies. Two independent and blinded authors (TY and AB) reviewed the search results separately to select studies based on inclusion and exclusion criteria. When a consensus was not reached between the two authors, a third author (MW) was consulted to reach a decision.
Study selection
Studies were selected if they met the following criteria: (1) they were published in peer-reviewed journals; (2) they included adult patients (≥ 18 years old) with type 2 DM; (3) they were RCTs that compared SGLT-2 inhibitors or GLP-1 RAs with a placebo; (4) they compared the risk of major adverse cardiovascular events (MACE) between treatment and placebo groups; (5) they compared the risk of renal outcomes; and (6) they showed an incidence of MACE and a composite of renal outcomes in patients with CKD (defined as estimated glomerular filtration rate (eGFR) < 60 ml/min/1.73 m2). There was no restriction on publication language. Studies were excluded if (1) they included non-human subjects and (2) there was insufficient data for estimating the risk ratio (RR) even after contacting the authors.
Outcomes
The primary efficacy outcome of this analysis was 3-point MACE (MACE-3), including cardiovascular death, myocardial infarction (MI), and stroke. The secondary outcome was a composite of renal outcomes, including ESRD, a decline in kidney function (such as worsening of eGFR or increasing creatinine), albuminuria, and renal or cardiovascular death. For studies reporting multiple renal outcomes, renal outcomes without albuminuria were prioritized for consistency.
Data extraction and quality assessment
All data from eligible studies were abstracted independently by two investigators (TY and AB). Any conflicts in data extraction or quality assessment were resolved by a third reviewer (MW). For each study, data regarding the incidence of MACE and a composite of renal outcomes in each group were abstracted. We used the Cochrane risk of bias assessment to explore sources of bias in the RCTs included in this analysis [25]. Applying this tool, we evaluated the risk of bias in random sequence generation, allocation concealment, the blinding of participants and researchers, the blinding of outcome assessments, selective reporting, incomplete outcome data, and other metrics.
Statistical analysis
We performed a network meta-analysis using the “netmeta” package (version 1.1-0) and R programming language (R Foundation for Statistical Computing, Vienna, Austria). The RRs and 95% confidence intervals (CIs) were estimated using Mantel–Haenszel methods. A random-effects model was used for the analysis. Heterogeneity was assessed by the probability value of the I2 variable [26, 27]. Heterogeneity was considered to be low, moderate, or high if I2 was 25%, 50%, or 75%, respectively.
Results
Literature search and included studies
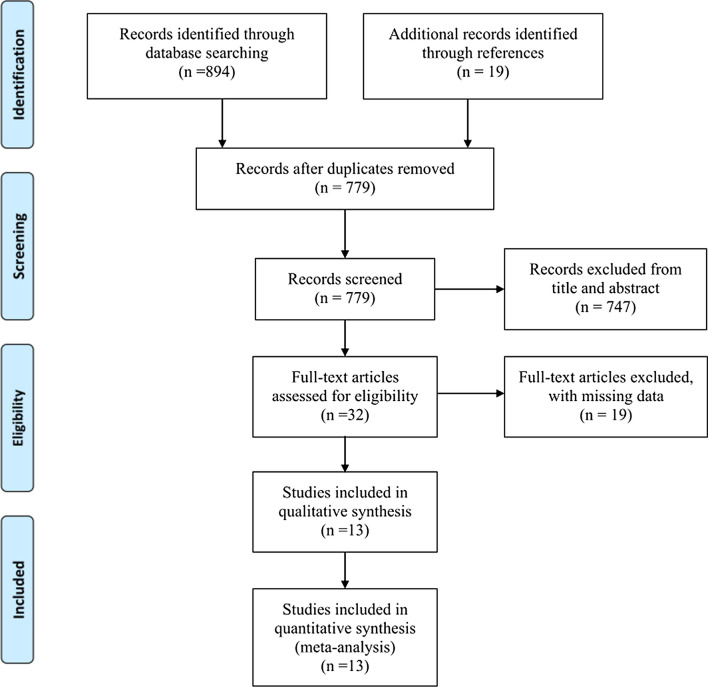
A diagram of the study selection is shown in Fig. 1. Initially, a total of 894 studies were obtained in the primary search from databases, and nineteen additional studies were identified through references. We removed 134 duplicate studies; 779 studies were screened. By screening titles and abstracts, 747 papers were excluded because they did not meet the inclusion criteria. By assessing full-text articles, nineteen additional studies were excluded due to missing data. Finally, thirteen studies published up to November 20, 2020, were selected for our meta-analysis according to the inclusion criteria [12, 28–39]. Out of thirteen studies, six studies were RCTs that compared SGLT-2 inhibitors (Canagliflozin [12, 29], Dapagliflozin [31], Empagliflozin [30], Ertugliflozin [28], and Sotagliflozin [32]) with placebo; seven studies compared GLP-1 RAs (Albiglutide [34], Dulaglutide weekly [33], Exenatide weekly [35], Liraglutide [37], Lixisenatide [39], Semaglutide subcutaneously weekly [38], and Semaglutide oral [36]) with placebo. The pooled population consisted of 20,106 patients in SGLT-2 inhibitor studies (10,716 in the group treated with SGLT-2 inhibitors and 9390 in the control group) and 12,843 patients in GLP-1 RA studies (6364 in the group treated with GLP-1 RAs and 6479 in the control group). Hemoglobin A1c ranged from 6.5 to 12%. eGFR ranged from 15 to 59 ml/min/1.73 m2 in one study [33], 25 to 59 ml/min/1.73 m2 in one study, [32] and from 30 to 59 ml/min/1.73 m2 in other studies, with two studies that not mentioned their lower limit [31, 38]. The median length of follow-up ranged from 16.0 months to 50.4 months in SGLT-2 inhibitor studies and 19.2 months to 64.8 months in GLP-1 RA studies. All studies defined MACE-3 as a composite outcome comprised of cardiovascular death, myocardial infarction, and stroke, except ELIXA, [39] which also includes unstable angina. For renal outcomes, all studies included ESRD; five studies included renal death; [28–30, 33, 35] two studies included renal or cardiovascular death; [12, 31] one study defined reduced kidney function as a decrease in eGFR ≥ 30% [33], three as a decrease in eGFR ≥ 40% [29, 31, 35], one as a decrease in eGFR ≥ 50% [11], and four as a doubling of creatinine [12, 28, 30, 37].
Fig. 1.
Flow diagram for study selection
Study characteristics and quality assessment
The definitions of terms, including a composite of renal outcomes and characteristics of the included studies, are listed in Table 1. Table 2 highlights the demographics of included studies. All studies defined CKD as eGFR < 60 ml/min/1.73 m2. The quality evaluation of the included studies is shown in Fig. 2. Subclasses of GLP-1 RAs (exendin-4 analogues or human GLP-1 RA analogues) in included studies are outlined in Additional file 1: Table S1.
Table 1.
Definitions of terms in included studies
| Study | Study design | Setting | Drug dose (mg/day) | Median follow up (months) | eGFR (ml/min/1.73 m2) | Range of Hgb A1c | Primary outcome | Definition of renal outcomes |
|---|---|---|---|---|---|---|---|---|
| SGLT2i vs placebo | ||||||||
| CANVAS Program | RCT | Multinational | Canagliflozin 300/100 | 29.0 | 30–59 | 7.0–10.5 | MACE | ≥ 40% eGFR decline, ESRD, renal death |
| CREDENCE | RCT | Multinational | Canagliflozin 100 | 31.4 | 30–59 | 6.5–12.0 | Renal outcomes | Doubing creatinine, ESRD, renal or CV death |
| DECLARE-TIMI 58 | RCT | Multinational | Dapagliflozin 10 | 50.4 | <60 | 6.5–12.0 | MACE | ≥ 40% eGFR decline, ESRD, renal or CVdeath |
| EMPA-REG OUTCOME | RCT | Multinational | Empagliflozin 10/25 | 37.2 | 30–59 | 7.0–9.0 | MACE | Doubling creatinine, ESRD, renal death |
| SCORED | RCT | Multinational | Sotagliflozin 400 | 16.0 | 25–60 | > 7.0 | MACE | ≥ 50% eGFR decline, ESRD |
| VERTIS-CV | RCT | Multinational | Ertugliflozin 5/15 | 36.0 | 30–59 | 7.0–10.5 | MACE | Doubing creatinine, ESRD, renal death |
| GLP-1 RA vs placebo | ||||||||
| ELIXA | RCT | Multinational | Lixisenatide 20 mcg | 25.2 | 30–59 | 5.5–11.0 | MACE (including unstable angina) | N/A |
| EXSCEL | RCT | Multinational | Exenatide 2 (weekly) | 38.4 | 30–59 | 6.5–10 | MACE | ≥ 40% EGFR decline, ESRD, renal death |
| HARMONY Outcomes | RCT | Multinational | Albiglutide 30/50 | 19.2 | 30–59 | > 7.0 | MACE | N/A |
| LEADER | RCT | Multinational | Liraglutide 1.8 | 45.6 | 30–59 | > 7.0 | MACE | Doubling of serum creatinine, ESRD |
| PIONEER-6 | RCT | Multinational | Semaglutide 14 (oral) | 15.9 | 30–59 | N/A | MACE | N/A |
| REWIND | RCT | Multinational | Dulaglutide 1.5 (weekly) | 64.8 | 15–59 | < 9.5 | MACE | ≥ 30% eGFR decline, ESRD, renal death |
| SUSTAIN-6 | RCT | Multinational | Semaglutide 0.5/1 (weekly) | 25.2 | <60 | > 7.0 | MACE | N/A |
SGLT2i sodium-glucose cotransporter-2, GLP-1 RA glucagon-like peptide-1 receptor agonist, RCT randomized control study, eGFR estimated glomerular filtration rate, Hgb A1c hemoglobin A1c, ESRD end-stage renal disease, MACE major adverse cardiovascular events, RCT randomized control trial, CV cardiovascular
Table 2.
Baseline characteristics of included studies in patients with GFR < 60 ml/min/1.73 m2
| Study | Number of patients (GFR < 60) | Age | Male (%) | BMI | sBP (mmHg) | dBP (mm Hg) | HgbA1c (%) | GFR < 30 (%) | GFR 30-45 (%) | GFR 45-60 (%) | Median GFR in CKD | UACR (mg/g) | Noormo-albuminuria (%) | Microalbuminuria (%) | Macroalbuminuria (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SGLT2i vs placebo | |||||||||||||||
| CANVAS Program | 2039 | 67.6 | 58.2 | 32.3 | 138 | 75 | 8.2 | 0.0 | 27.2 | 72.8 | 49.1 | 24.4 | 55.4 | N/A | N/A |
| CREDENCE | 2592 | 63.8 | 66.5 | 31.3 | 140 | 78 | 8.2 | 0.0 | 45.3 | 48.1 | 45.1 | 1012 | N/A | N/A | N/A |
| DECLARE-TIMI 58 | 1265 | 67.3 | 64.3 | 34.5 | 134 | 75 | 8.2 | N/A | N/A | N/A | 51.4 | N/A | 55.6 | 30.9 | 13.5 |
| EMPA-REG OUTCOME | 1819 | 67.1 | 72.8 | 30.6 | 138 | 76 | 8.1 | 0 | 31.3 | 68.7 | N/A | N/A | 47.7 | 33.9 | 18.4 |
| SCORED | 10,584 | 69.0 | 55.1 | 31.8 | 138 | 78 | 8.3 | 7.7 | 43.9 | 48.3 | 44.5 | 74.5 | 35.1 | 33.9 | 31.0 |
| VERTIS-CV | 1807 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| Weighted average (SGLT-2 inhibitors) | 67.8 | 59.5 | 31.9 | 137.9 | 77.2 | 8.2 | 40.8 | 53.4 | 40.8 | 33.6 | 27.7 | ||||
| GLP-1 RA vs placebo | |||||||||||||||
| ELIXA | 1399 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | 59.7 | 25.5 | 14.8 |
| EXSCEL | 3177 | 66.5 | 57.1 | 32.8 | N/A | N/A | 8.1 | 0 | 28.0 | 72.0 | 49.2 | N/A | 71.4 | 19.6 | 9.0 |
| HARMONY Outcomes | 2222 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| LEADER | 2158 | 67.3 | 61.3 | 32.7 | 136.4 | 75.1 | 8.6 | 10.4 | 29.0 | 60.6 | 45.6 | 49.5 | 47.6 | 29 | 23.4 |
| PIONEER-6 | 856 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| REWIND | 2199 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| SUSTAIN-6 | 832 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| Weighted average (GLP-1 RAs) | 66.8 | 58.8 | 32.8 | 8.3 | 28.4 | 67.4 | 61.3 | 23.8 | 14.8 | ||||||
Normoalbuminuria, microlabuminuria and macroalbuminuria defined as < 30, 30–299, and ≥ 300 mg/g creatinine, respectively
SGLT2i sodium-glucose cotransporter-2 inhibitors, GLP-1 RAs glucagon-like peptide-1 receptor agonists, GFR glomerular filtration rate, BMI body mass index, sBP systolic blood pressure, dBP diastolic blood pressure, Hgb A1c hemoglobin A1c, UACR urine albumin-to-creatinine ratio, N/A not available
Fig. 2.

Quality assessment (Cochrane risk of bias tool) for included RCTs. RCT randomized control study
Network meta-analysis of treatment groups
Mace-3
Network plots were shown in Fig. 3. SGLT-2 inhibitors were associated with a decreased risk of MACE-3 compared with placebo (RR [95% CI]; 0.85 [0.75–0.96]), but GLP-1 RAs were not (RR 0.91 [0.80–1.04]). Compared to GLP-1 RAs, SGLT-2 inhibitors did not show a significant difference in the risk of MACE-3 (RR 0.94 [0.78–1.12]) (Fig. 4). However, there was significant heterogeneity (I2= 47.8%, p = 0.039).
Fig. 3.
Network plot for MACE. SGLT-2 sodium-glucose cotransporter 2, GLP-1 RA glucagon-like peptide-1 receptor agonist, MACE major adverse cardiovascular events
Fig. 4.
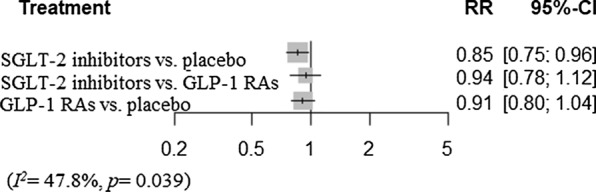
Network meta-analysis reporting risk ratio (RR) for MACE in CKD patients. SGLT-2 sodium-glucose cotransporter 2, GLP-1 RA glucagon-like peptide-1 receptor agonist, MACE major adverse cardiovascular events, CKD chronic kidney disease
Renal outcomes
We also performed a network meta-analysis of the risk of renal events and found that SGLT-2 inhibitors significantly decreased renal events (RR 0.68 [0.59–0.78]), while the impact on renal events of GLP-1 RAs was not statistically significant (RR 0.86 [0.72–1.03]). SGLT-2 inhibitors were also associated with lower risk compared to GLP-1 RAs (RR 0.79 [0.63–0.99]) (Fig. 5). There was no heterogeneity (I2= 0%, p = 0.92).
Fig. 5.
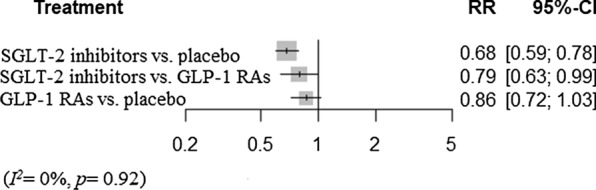
Network meta-analysis reporting risk ratio (RR) for renal outcomes in CKD patients. SGLT-2 sodium-glucose cotransporter 2, GLP-1 RA glucagon-like peptide-1 receptor agonist, CKD chronic kidney disease
Sensitivity analyses
Mace-3
The results of sensitivity analyses are summarized in Table 3. First, we performed a sensitivity analysis for studies that defined MACE-3 as the primary outcome. Compared to placebo, SGLT-2 inhibitors had a tendency to decrease a risk of MACE-3 (RR 0.87 [0.76–1.00]), while GLP-1 RAs did not (RR 0.91 [0.79–1.04]). There was no significant difference between SLGT-2 inhibitors and GLP-1 RA (RR 0.96 [0.79–1.17]). There was significant heterogeneity (I2= 50.4%, p = 0.033).
Table 3.
The summary of sensitivity analyses
| Sensitivity analysis | Subjects | ||||||
|---|---|---|---|---|---|---|---|
| SGLT-2 inhibitors | GLP-1 RAs | Comparison | Risk ratio | 95% CI | I2 (%) | p value | |
| MACE-3 | |||||||
| MACE-3 as the primary outcome | 17514 | 10644 | SGLT-2 inhibitors vs. placebo | 0.87 | 0.76–1.00 | 50.4 | 0.033 |
| GLP-1 RAs vs. placebo | 0.91 | 0.79–1.04 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.96 | 0.79–1.17 | |||||
| Without ELIXA | 20106 | 14007 | SGLT-2 inhibitors vs. placebo | 0.85 | 0.75–0.97 | 49.5 | 0.037 |
| GLP-1 RAs vs. placebo | 0.88 | 0.76–1.03 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.97 | 0.79–1.18 | |||||
| Daily GLP-1 RAs | 20106 | 6635 | SGLT-2 inhibitors vs. placebo | 0.84 | 0.75–0.96 | 47.7 | 0.053 |
| GLP-1 RAs vs. placebo | 0.86 | 0.73–1.02 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.99 | 0.80–1.21 | |||||
| Weekly GLP-1 RAs | 20106 | 4009 | SGLT-2 inhibitors vs. placebo | 0.85 | 0.76–0.95 | 31.8 | 0.19 |
| GLP-1 RAs vs. placebo | 1.01 | 0.83–1.22 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.85 | 0.68–1.05 | |||||
| GLP-1 analogues | 20106 | 6068 | SGLT-2 inhibitors vs. placebo | 0.85 | 0.76–0.95 | 30.6 | 0.17 |
| GLP-1 RAs vs. placebo | 0.81 | 0.69–0.95 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 1.05 | 0.86–1.27 | |||||
| Exendin-4 analogues | 20106 | 4576 | SGLT-2 inhibitors vs. placebo | 0.85 | 0.77–0.94 | 28.7 | 0.21 |
| GLP-1 RAs vs. placebo | 1.03 | 0.88–1.20 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.83 | 0.69–0.99 | |||||
| Renal outcomes | |||||||
| Renal endpoints as secondary outcomes | 18583 | 7534 | SGLT-2 inhibitors vs. placebo | 0.67 | 0.55–0.83 | 0 | 0.86 |
| GLP-1 RAs vs. placebo | 0.86 | 0.72–1.03 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.78 | 0.59–1.03 | |||||
| Macroalbuminuria | 1505 | 7534 | SGLT-2 inhibitors vs. placebo | N/A | N/A | 0 | 0.73 |
| GLP-1 RAs vs. placebo | 0.91 | 0.81–1.02 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | N/A | N/A | |||||
| Weekly GLP-1 RAs | 20088 | 5376 | SGLT-2 inhibitors vs. placebo | 0.65 | 0.55–0.78 | 0 | 0.73 |
| GLP-1 RAs vs. placebo | 0.91 | 0.81–1.02 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.71 | 0.58–0.89 | |||||
| GLP-1 analogues | 20088 | 4357 | SGLT-2 inhibitors vs. placebo | 0.68 | 0.59–0.78 | 0 | 0.94 |
| GLP-1 RAs vs. placebo | 0.82 | 0.66–1.01 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.83 | 0.65–1.07 | |||||
SGLT2i sodium-glucose cotransporter-2 inhibitors, GLP-1 RAs glucagon-like peptide-1 receptor agonists, CI confidence intervals, MACE-3 3-point major adverse cardiovascular event, vs. versus, N/A not available
Next, we conducted another sensitivity analysis excluding ELIXA, [39] as ELIXA includes unstable angina in its definition of adverse events in addition to MACE-3. The results were consistent: GLP-1 RAs did not show a significant difference when compared to placebo (RR 0.88 [0.76–1.03]) with significant heterogeneity (I2= 49.5%, p = 0.037). SGLT-2 inhibitors did not reduce MACE-3 significantly when compared to GLP-1 RAs (RR 0.97 [0.79–1.18]).
Third, we performed another analysis based on the frequency of GLP-1 RA dosing. Daily GLP-1 RAs tended to reduce the risk of MACE-3 when compared to placebo (RR 0.86 [0.73–1.02]), while weekly GLP-1 RAs did not (RR 1.01 [0.83–1.22]). GLP-1 RAs were comparable to SGLT-2 inhibitors (RR 0.99 [0.80–1.21] and 0.85 [0.68–1.05], respectively). Both analyses showed moderate heterogeneity (I2= 47.7% and 31.8%, respectively).
Lastly, we divided GLP-1 RAs into two subclasses, GLP-1 analogues and exendin-4 analogues, and found that GLP-1 analogues displayed significantly lower risk than placebo (RR 0.81 [0.69-0.95]), although there was moderate heterogeneity (I2= 30.6%, p = 0.17). The risk reduction between SGLT-2 inhibitors and GLP-1 analogues was similar (RR 1.05 [0.86-1.27]). Exendin-4 analogues, on the other hand, were not associated with a decreased risk of MACE-3 compared to placebo (RR 1.03 [0.88–1.20]), with moderate heterogeneity (I2= 28.7%, p = 0.21). SGLT-2 inhibitors were associated with significantly lower risk compared to exendin-4 analogues (RR 0.83 [0.69–0.99]).
Renal outcomes
Since most studies classify renal outcomes as secondary endpoints, we conducted a sensitivity analysis excluding the study classifying renal events as primary outcomes [12]. When compared to placebo, SGLT-2 inhibitors reduced the incidence of renal outcomes (RR 0.67 [0.55–0.83]). GLP-1 RAs did not show a statistical difference when compared to placebo (RR 0.86 [0.72–1.03]). SGLT-2 inhibitors tended to reduce the risk when compared to GLP-1 RAs (RR 0.78 [0.59–1.03]). No heterogeneity was appreciated (I2= 0%, p = 0.86).
Since GLP-1 RA studies included macroalbuminuria as a prespecified renal outcome, we performed a sensitivity analysis of studies that included macroalbuminuria as a renal outcome. The prespecified definitions of renal outcomes are shown in Additional file 1: Table S2. GLP-1 RAs were not associated with significantly reduced risk compared to placebo (RR 0.91 [0.81–1.02]). Only one SGLT-2 study included macroalbuminuria in renal outcomes.
Additional analysis based on GLP-1 RA frequency showed similar results. Weekly GLP-1 RAs did not decrease renal events compared to placebo (RR 0.91 [0.81-1.02]). There was no heterogeneity (I2= 0%, p = 0.73). SGLT-2 inhibitors were superior to weekly GLP-1 RAs (RR 0.71 [0.58–0.89]). Only one study investigated daily GLP-1 RAs.
We performed another analysis for GLP-1 analogues. GLP-1 analogues had a trend towards a reduction in renal events compared to placebo (RR 0.82 [0.66-1.01]) without heterogeneity (I2= 0%, p = 0.94). Compared to GLP-1 analogues, SGLT-2 inhibitors were associated with lower risk (RR 0.68 [0.59–0.78]). There was only one study that investigated renal risks for exendin-4 analogues.
Subgroup analysis
The results of subgroup analyses are summarized in Table 4. We conducted a subgroup analysis based on eGFR: 30-44 ml/min/1.73 m2 and 45-59 ml/min/1.73 m2. In a subgroup of eGFR: 30-44 ml/min/1.73 m2 patients, SGLT-2 inhibitors reduced MACE-3 significantly (RR 0.73 [0.54-0.97]), but GLP-1 RAs did not (RR 1.02 [0.78-1.33]). There was high heterogeneity (I2= 57.1%, p = 0.063). SGLT-2 inhibitors also showed beneficial effects on renal outcomes compared with placebo (RR 0.75 [0.62-0.91]), while GLP-1 RAs did not (RR 0.78 [0.46-1.32]). There was no heterogeneity (I2= 0%, p = 0.69). Compared to GLP-1 RAs, SGLT-2 inhibitors did not achieve a statistically significant difference (RR 0.96 [0.55–1.29]).
Table 4.
The summary of subgroup analyses
| Subgroup analysis | Subjects | ||||||
|---|---|---|---|---|---|---|---|
| SGLT-2 inhibitors | GLP-1 RAs | Comparison | Risk ratio | 95% CI | I2 (%) | p value | |
| MACE-3 | |||||||
| eGFR 30-44 | 2479 | 1934 | SGLT-2 inhibitors vs. placebo | 0.73 | 0.54–0.97 | 57.1 | 0.063 |
| GLP-1 RAs vs. placebo | 1.02 | 0.78–1.33 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.72 | 0.48–1.06 | |||||
| eGFR 45-59 | 3974 | 4576 | SGLT-2 inhibitors vs. placebo | 0.82 | 0.66–1.01 | 44.8 | 0.12 |
| GLP-1 RAs vs. placebo | 0.85 | 0.71–1.03 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.96 | 0.72–1.27 | |||||
| Renal outcomes | |||||||
| eGFR 30-44 | 2420 | 889 | SGLT-2 inhibitors vs. placebo | 0.75 | 0.62–0.91 | 0 | 0.69 |
| GLP-1 RAs vs. placebo | 0.78 | 0.46–1.32 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.96 | 0.55–1.29 | |||||
| eGFR 45-59 | 3976 | 2288 | SGLT-2 inhibitors vs. placebo | 0.61 | 0.48–0.77 | 0 | 0.52 |
| GLP-1 RAs vs. placebo | 1.18 | 0.76–1.84 | |||||
| SGLT-2 inhibitors vs. GLP-1 RAs | 0.52 | 0.31–0.85 | |||||
SGLT2i sodium-glucose cotransporter-2 inhibitors, GLP-1 RAs glucagon-like peptide-1 receptor agonists, eGFR estimated glomerular filtration rate, CI confidence intervals, MACE-3 3-point major adverse cardiovascular event, vs. versus, N/A not available
For patients with eGFR 45-59 ml/min/1.73 m2, both SGLT-2 inhibitors and GLP-1 RAs had a similar tendency to reduce MACE-3 compared to placebo (RR 0.82 [0.66-1.01] and 0.85 [0.71–1.03], respectively). There was moderate heterogeneity (I2= 44.8%, p = 0.12). In terms of renal outcomes, SGLT-2 inhibitors reduced renal outcomes (RR 0.61 [0.48-0.77), but GLP-1 RAs did not (RR 1.18 [0.76–1.84]). A comparison between SGLT-2 inhibitors and GLP-1 RAs was not statistically significant (RR 0.52 [0.31–0.85]). No heterogeneity was observed (I2= 0%, p = 0.52).
Discussion
Our study revealed that SGLT-2 inhibitors decrease the risk of cardiovascular and renal events in type 2 DM patients with CKD, which is compatible with RCTs of CKD patients [11, 12]. On the other hand, GLP-1 RAs did not lead to significantly lower cardiovascular or renal endpoints, although they showed numerically better results. An indirect comparison of SGLT-2 inhibitors with GLP-1 RAs revealed that SGLT-2 inhibitors significantly decreased the risk of renal outcomes. Sensitivity analyses showed a similar tendency. Interestingly, a sensitivity analysis among GLP-1 RA subclasses revealed that GLP-1 analogues significantly reduced MACE-3 and renal events, while exendin-4 analogues did not.
Several mechanisms have been proposed for the positive impact of SGLT-2 inhibitors. First, SGLT-2 inhibitors have mild natriuretic and diuretic effects [40]. Recent RCTs reveal that in patients with heart failure with reduced ejection fraction, SGLT-2 inhibitors were associated with a lower risk of cardiovascular death or hospitalization for heart failure [41, 42]. This can be at least partially attributed to diuretic effects, which lead to BP reduction and thereby confer cardiovascular and renoprotective benefits. Second, SGLT-2 inhibitors mitigate low-grade inflammation. SGLT-2 inhibitors prevent glucose entry into proximal tubular cells, which limits glucotoxicity, potentially leading to less oxidative stress [43]. Third, SGLT-2 inhibitors block sodium reuptake in the proximal tubule; an increased delivery of sodium to the macula densa leads to afferent arteriolar constriction and a reduction in intraglomerular pressure [44, 45].
Since SGLT-2 inhibitors antagonize glucose reabsorption in the renal tubule, we can anticipate that the effect of SGLT-2 inhibitors is dependent on eGFR. However, a pooled analysis of clinical trials revealed that SGLT-2 inhibitors decrease body weight, BP, and albuminuria regardless of eGFR, although glucose-lowering effects decreased as eGFR declined [11, 46]. Our study also revealed beneficial effects of SGLT-2 inhibitors in both eGFR 30-44 ml/min/1.73 m2 and 45–59 ml/min/1.73 m2 groups.
In addition to glycemic control, several mechanisms explain the beneficial effects of GLP-1 RAs. First, GLP-1 RAs lead to BP reduction, [14] which can be attributed to natriuresis [47]. Other possible mechanisms are the reduction of reactive oxygen species and inflammation [48, 49] and improvement of endothelial function [50]. However, our study did not find a significant difference between GLP-1 RAs and placebo. A subgroup analysis of a prior meta-analysis by Kristensen et al. [17] also revealed that GLP-1 RAs were not associated with a significantly reduced risk of MACE, although the RR was 0.88. Our sensitivity analysis among GLP-1 RAs drug subclasses revealed a beneficial effect of GLP-1 analogues, while exendin-4 analogues were not. Zelniker et al. revealed a similar tendency [18]. There are several differences between these two subclasses. First, exendin-4 analogues are metabolized and eliminated by the kidneys. GLP-1 analogues, in contrast, are endogenously metabolized [51]. Second, exendin-4 analogues are resistant to inactivation by dipeptidyl peptidase-4. Conversely, GLP-1 analogues can be partially metabolized to the metabolite, which could have an additional cardioprotective effect [52]. As far as we know, there are no studies that evaluate the difference in the cardiovascular benefits between these subclasses. Further investigation is warranted to explore the differences among GLP-1 RAs subclasses.
The major strength of our analysis is that this is the first study that investigates the effect of SGLT-2 inhibitors and GLP-1 RAs on cardiovascular and renal events in type 2 DM patients with CKD [53]. The ADA preferably recommends SGLT-2 inhibitors over GLP-1 RAs in CKD patients; [21] our study supports their recommendations, with additional evidence that GLP-1 analogues can be an alternative option. Our large sample size was another strength of our analysis, allowing us to tease out statistically significant differences among interventions.
Our meta-analysis has several limitations. First, there is still a concern that CKD patients may not be fully randomized since the studies included are subgroup analyses of RCTs. Second, high heterogeneity was observed, and this heterogeneity persisted in most sensitivity and subgroup analyses. As shown in Table 2, the proportions of advanced CKD stages and macroalbuminuria are noticeably different. Population variety among studies may be a source of this heterogeneity. The results should be interpreted with caution. Third, the patients included in each trial may have achieved different levels of hemoglobin A1c, which may be subject to bias as well. Fourth, definitions of renal outcomes are not consistent across studies, though we prioritized renal outcomes without macroalbuminuria in an attempt to reduce inconsistency. Since most GLP-1 RAs studies included macroalbuminuria as a prespecified renal outcome, analyzing renal outcomes without macroalbuminuria may lead to bias. Lastly, we defined CKD as eGFR < 60 ml/min/1.73 m2 and excluded patients with albuminuria. Therefore, we were not able to investigate the effects of SGLT-2 inhibitors and GLP-1 RAs for those who have albuminuria and eGFR > 60 ml/min/1.73 m2. Ongoing RCTs, including EMPA-KIDNEY, SOUL, and FLOW, will provide better insight into the cardiorenal effects of SGLT-2 inhibitors and GLP-1 RAs [54–56]. Further research, including head-to-head comparison, is warranted to explore the effects of SGLT-2 inhibitors and GLP-1 RAs in CKD patients [57].
Conclusion
In patients with type 2 DM and CKD defined as eGFR < 60 ml/min/1.73 m2, SGLT-2 inhibitors were associated with decreased risk of cardiovascular and renal events. GLP-1 RAs did not lead to significantly lower cardiovascular or renal endpoints, although they showed numerically better results. SGLT-2 inhibitors significantly decrease the risk of renal events compared to GLP-1 RAs. Sensitivity analyses were consistent. Among GLP-1 RAs, GLP-1 analogues showed a positive impact, while exendin-4 analogues did not.
Supplementary information
Additional file 1. Supplementary tables.
Acknowledgements
Financial Support: This work was supported by grants from the Japan Society for the Promotion of Science, Salt Science Research Foundation (20C4), and Japan Agency for Medical Research and Development (AMED).
Abbreviations
- DM
Diabetes mellitus
- CKD
Chronic kidney disease
- ESRD
End-stage renal disease
- SGLT-2
Sodium-glucose cotransporter-2
- RCT
Randomized controlled trial
- GLP-1 RA
Glucagon-like peptide-1 receptor agonist
- ADA
American Diabetes Association
- BP
Blood pressure
- PRISMA
Preferred reporting items for systematic reviews and meta-analyses
- MACE
Major adverse cardiovascular events
- eGFR
Estimated glomerular filtration rate
- RR
Risk ratio
- 3-point MACE
MACE-3
- MI
Myocardial infarction
- CI
Confidence interval
Authors’ contributions
TY Designed the study, collected the data, contributed to the statistical analysis, and served as the primary author of the manuscript. MW Collected the data, contributed to the statistical analysis, and served as an author of the manuscript (equivalent contributor). AB Collected the data and contributed to the statistical analysis. NC Contributed to the statistical analysis and assisted with the writing of the manuscript. HM Contributed to the writing of the manuscript and provided critical feedback to shape the manuscript. TM Contributed to the writing of the manuscript and provided critical feedback to shape the manuscript. HU Contributed to the writing of the manuscript and provided critical feedback to shape the manuscript. TF Contributed to the writing of the manuscript and provided critical feedback to shape the manuscript. YS Contributed to the data collection and data analysis. TY Contributed to the data collection and assisted with the writing of the manuscript. KA Contributed to the statistical analysis and assisted with the writing of the manuscript. SU, TS, EA Contributed to the data collection and assisted with the writing of the manuscript. HW Contributed to the statistical analysis and assisted with the writing of the manuscript. KT Contributed to the statistical analysis and assisted with the writing of the manuscript. All authors read and approved the final manuscript.
Funding
The author (KT) received a fund from the Japan Society for the Promotion of Science, and Japan Agency for Medical Research and Development (AMED). The funding agency had no role in designing the study, conducting the analysis, interpreting the data or writing the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Takayuki Yamada and Mako Wakabayashi contributed equally to this work
Supplementary Information
The online version contains supplementary material available at 10.1186/s12933-020-01197-z.
References
- 1.Federation ID: IDF Diabetes Atlas—9th Edition. 2019.
- 2.Saran R, Robinson B, Abbott KC, Bragg-Gresham J, Chen X, Gipson D, Gu H, Hirth RA, Hutton D, Jin Y et al. US renal data system 2019 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2020;75(1S1):A6–A7. [DOI] [PubMed]
- 3.Jiang G, Luk AOY, Tam CHT, Xie F, Carstensen B, Lau ESH, Lim CKP, Lee HM, Ng ACW, Ng MCY, et al. Progression of diabetic kidney disease and trajectory of kidney function decline in Chinese patients with type 2 diabetes. Kidney Int. 2019;95(1):178–187. doi: 10.1016/j.kint.2018.08.026. [DOI] [PubMed] [Google Scholar]
- 4.Yandrapalli S, Jolly G, Horblitt A, Sanaani A, Aronow WS. Cardiovascular benefits and safety of non-insulin medications used in the treatment of type 2 diabetes mellitus. Postgrad Med. 2017;129(8):811–821. doi: 10.1080/00325481.2017.1358064. [DOI] [PubMed] [Google Scholar]
- 5.Briasoulis A, Bakris GL. Chronic kidney disease as a coronary artery disease risk equivalent. Curr Cardiol Rep. 2013;15(3):340. doi: 10.1007/s11886-012-0340-4. [DOI] [PubMed] [Google Scholar]
- 6.Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377(7):644–657. doi: 10.1056/NEJMoa1611925. [DOI] [PubMed] [Google Scholar]
- 7.Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, Mattheus M, Devins T, Johansen OE, Woerle HJ, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117–2128. doi: 10.1056/NEJMoa1504720. [DOI] [PubMed] [Google Scholar]
- 8.Shao SC, Chang KC, Lin SJ, Chien RN, Hung MJ, Chan YY, Kao Yang YH, Lai EC. Favorable pleiotropic effects of sodium glucose cotransporter 2 inhibitors: head-to-head comparisons with dipeptidyl peptidase-4 inhibitors in type 2 diabetes patients. Cardiovasc Diabetol. 2020;19(1):17. doi: 10.1186/s12933-020-0990-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bonaca MP, Wiviott SD, Zelniker TA, Mosenzon O, Bhatt DL, Leiter LA, McGuire DK, Goodrich EL, De Mendonca Furtado RH, Wilding JPH, et al. Dapagliflozin and cardiac, kidney, and limb outcomes in patients with and without peripheral artery disease in DECLARE-TIMI 58. Circulation. 2020;142(8):734–747. doi: 10.1161/CIRCULATIONAHA.119.044775. [DOI] [PubMed] [Google Scholar]
- 10.Wanner C, Inzucchi SE, Zinman B, Koitka-Weber A, Mattheus M, George JT, von Eynatten M, Hauske SJ, Investigators E-RO. Consistent effects of empagliflozin on cardiovascular and kidney outcomes irrespective of diabetic kidney disease categories: Insights from the EMPA-REG OUTCOME trial. Diabetes Obes Metab. 2020. [DOI] [PubMed]
- 11.Heerspink HJL, Stefansson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, Mann JFE, McMurray JJV, Lindberg M, Rossing P, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383(15):1436–1446. doi: 10.1056/NEJMoa2024816. [DOI] [PubMed] [Google Scholar]
- 12.Mahaffey KW, Jardine MJ, Bompoint S, Cannon CP, Neal B, Heerspink HJL, Charytan DM, Edwards R, Agarwal R, Bakris G, et al. Canagliflozin and cardiovascular and renal outcomes in type 2 diabetes mellitus and chronic kidney disease in primary and secondary cardiovascular prevention groups. Circulation. 2019;140(9):739–750. doi: 10.1161/CIRCULATIONAHA.119.042007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology. 2007;132(6):2131–2157. doi: 10.1053/j.gastro.2007.03.054. [DOI] [PubMed] [Google Scholar]
- 14.Sun F, Wu S, Guo S, Yu K, Yang Z, Li L, Zhang Y, Quan X, Ji L, Zhan S. Impact of GLP-1 receptor agonists on blood pressure, heart rate and hypertension among patients with type 2 diabetes: a systematic review and network meta-analysis. Diabetes Res Clin Pract. 2015;110(1):26–37. doi: 10.1016/j.diabres.2015.07.015. [DOI] [PubMed] [Google Scholar]
- 15.Shah M, Vella A. Effects of GLP-1 on appetite and weight. Rev Endocr Metab Disord. 2014;15(3):181–187. doi: 10.1007/s11154-014-9289-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bethel MA, Patel RA, Merrill P, Lokhnygina Y, Buse JB, Mentz RJ, Pagidipati NJ, Chan JC, Gustavson SM, Iqbal N, et al. Cardiovascular outcomes with glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes: a meta-analysis. Lancet Diabetes Endocrinol. 2018;6(2):105–113. doi: 10.1016/S2213-8587(17)30412-6. [DOI] [PubMed] [Google Scholar]
- 17.Kristensen SL, Rorth R, Jhund PS, Docherty KF, Sattar N, Preiss D, Kober L, Petrie MC, McMurray JJV. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7(10):776–785. doi: 10.1016/S2213-8587(19)30249-9. [DOI] [PubMed] [Google Scholar]
- 18.Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Furtado RHM, Bonaca MP, Mosenzon O, Kato ET, Cahn A, et al. Comparison of the effects of glucagon-like peptide receptor agonists and sodium-glucose cotransporter 2 inhibitors for prevention of major adverse cardiovascular and renal outcomes in type 2 diabetes mellitus. Circulation. 2019;139(17):2022–2031. doi: 10.1161/CIRCULATIONAHA.118.038868. [DOI] [PubMed] [Google Scholar]
- 19.Longato E, Di Camillo B, Sparacino G, Tramontan L, Avogaro A, Fadini GP. Better cardiovascular outcomes of type 2 diabetic patients treated with GLP-1 receptor agonists versus DPP-4 inhibitors in clinical practice. Cardiovasc Diabetol. 2020;19(1):74. doi: 10.1186/s12933-020-01049-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yang CT, Yang CY, Ou HT, Kuo S. Comparative cardiovascular safety of GLP-1 receptor agonists versus other glucose-lowering agents in real-world patients with type 2 diabetes: a nationwide population-based cohort study. Cardiovasc Diabetol. 2020;19(1):83. doi: 10.1186/s12933-020-01053-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.American Diabetes A: 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020, 43(Suppl 1):S98-S110. [DOI] [PubMed]
- 22.Thein D, Christiansen MN, Mogensen UM, Bundgaard JS, Rorth R, Madelaire C, Fosbol EL, Schou M, Torp-Pedersen C, Gislason G, et al. Add-on therapy in metformin-treated patients with type 2 diabetes at moderate cardiovascular risk: a nationwide study. Cardiovasc Diabetol. 2020;19(1):107. doi: 10.1186/s12933-020-01078-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. doi: 10.1136/bmj.b2535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, Ioannidis JP, Straus S, Thorlund K, Jansen JP, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med. 2015;162(11):777–784. doi: 10.7326/M14-2385. [DOI] [PubMed] [Google Scholar]
- 25.Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–1558. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 28.Cannon CP, Pratley R, Dagogo-Jack S, Mancuso J, Huyck S, Masiukiewicz U, Charbonnel B, Frederich R, Gallo S, Cosentino F, et al. Cardiovascular outcomes with ertugliflozin in type 2 diabetes. N Engl J Med. 2020;383(15):1425–1435. doi: 10.1056/NEJMoa2004967. [DOI] [PubMed] [Google Scholar]
- 29.Neuen BL, Ohkuma T, Neal B, Matthews DR, de Zeeuw D, Mahaffey KW, Fulcher G, Desai M, Li Q, Deng H, et al. Cardiovascular and renal outcomes with canagliflozin according to baseline kidney function. Circulation. 2018;138(15):1537–1550. doi: 10.1161/CIRCULATIONAHA.118.035901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wanner C, Lachin JM, Inzucchi SE, Fitchett D, Mattheus M, George J, Woerle HJ, Broedl UC, von Eynatten M, Zinman B, et al. Empagliflozin and clinical outcomes in patients with type 2 diabetes mellitus, established cardiovascular disease, and chronic kidney disease. Circulation. 2018;137(2):119–129. doi: 10.1161/CIRCULATIONAHA.117.028268. [DOI] [PubMed] [Google Scholar]
- 31.Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380(4):347–357. doi: 10.1056/NEJMoa1812389. [DOI] [PubMed] [Google Scholar]
- 32.Bhatt DL, Szarek M, Pitt B, Cannon CP, Leiter LA, McGuire DK, Lewis JB, Riddle MC, Inzucchi SE, Kosiborod MN et al. Sotagliflozin in patients with diabetes and chronic kidney disease. N Engl J Med. 2020. [DOI] [PubMed]
- 33.Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, Probstfield J, Botros FT, Riddle MC, Ryden L, et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet. 2019;394(10193):131–138. doi: 10.1016/S0140-6736(19)31150-X. [DOI] [PubMed] [Google Scholar]
- 34.Hernandez AF, Green JB, Janmohamed S, D’Agostino RB, Sr, Granger CB, Jones NP, Leiter LA, Rosenberg AE, Sigmon KN, Somerville MC, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): a double-blind, randomised placebo-controlled trial. Lancet. 2018;392(10157):1519–1529. doi: 10.1016/S0140-6736(18)32261-X. [DOI] [PubMed] [Google Scholar]
- 35.Holman RR, Bethel MA, Mentz RJ, Thompson VP, Lokhnygina Y, Buse JB, Chan JC, Choi J, Gustavson SM, Iqbal N, et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2017;377(13):1228–1239. doi: 10.1056/NEJMoa1612917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, Franco DR, Jeppesen OK, Lingvay I, Mosenzon O, Pedersen SD, et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019;381(9):841–851. doi: 10.1056/NEJMoa1901118. [DOI] [PubMed] [Google Scholar]
- 37.Mann JFE, Fonseca V, Mosenzon O, Raz I, Goldman B, Idorn T, von Scholten BJ, Poulter NR. Effects of liraglutide versus placebo on cardiovascular events in patients with type 2 diabetes mellitus and chronic kidney disease. Circulation. 2018;138(25):2908–2918. doi: 10.1161/CIRCULATIONAHA.118.036418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jodar E, Leiter LA, Lingvay I, Rosenstock J, Seufert J, Warren ML, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834–1844. doi: 10.1056/NEJMoa1607141. [DOI] [PubMed] [Google Scholar]
- 39.Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Kober LV, Lawson FC, Ping L, Wei X, Lewis EF, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015;373(23):2247–2257. doi: 10.1056/NEJMoa1509225. [DOI] [PubMed] [Google Scholar]
- 40.Imprialos KP, Sarafidis PA, Karagiannis AI. Sodium-glucose cotransporter-2 inhibitors and blood pressure decrease: a valuable effect of a novel antidiabetic class? J Hypertens. 2015;33(11):2185–2197. doi: 10.1097/HJH.0000000000000719. [DOI] [PubMed] [Google Scholar]
- 41.Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, Januzzi J, Verma S, Tsutsui H, Brueckmann M, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413–1424. doi: 10.1056/NEJMoa2022190. [DOI] [PubMed] [Google Scholar]
- 42.McMurray JJV, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA, Ponikowski P, Sabatine MS, Anand IS, Belohlavek J, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008. doi: 10.1056/NEJMoa1911303. [DOI] [PubMed] [Google Scholar]
- 43.Ojima A, Matsui T, Nishino Y, Nakamura N, Yamagishi S. Empagliflozin, an inhibitor of sodium-glucose cotransporter 2 exerts anti-inflammatory and antifibrotic effects on experimental diabetic nephropathy partly by suppressing AGEs-Receptor axis. Horm Metab Res. 2015;47(9):686–692. doi: 10.1055/s-0034-1395609. [DOI] [PubMed] [Google Scholar]
- 44.Cherney DZ, Perkins BA, Soleymanlou N, Maione M, Lai V, Lee A, Fagan NM, Woerle HJ, Johansen OE, Broedl UC, et al. Renal hemodynamic effect of sodium-glucose cotransporter 2 inhibition in patients with type 1 diabetes mellitus. Circulation. 2014;129(5):587–597. doi: 10.1161/CIRCULATIONAHA.113.005081. [DOI] [PubMed] [Google Scholar]
- 45.Kitada M, Hirai T, Koya D. Significance of SGLT2 inhibitors: lessons from renal clinical outcomes in patients with type 2 diabetes and basic researches. Diabetol Int. 2020;11(3):245–251. doi: 10.1007/s13340-020-00444-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Petrykiv S, Sjostrom CD, Greasley PJ, Xu J, Persson F, Heerspink HJL. Differential effects of dapagliflozin on cardiovascular risk factors at varying degrees of renal function. Clin J Am Soc Nephrol. 2017;12(5):751–759. doi: 10.2215/CJN.10180916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lovshin JA, Barnie A, DeAlmeida A, Logan A, Zinman B, Drucker DJ. Liraglutide promotes natriuresis but does not increase circulating levels of atrial natriuretic peptide in hypertensive subjects with type 2 diabetes. Diabetes Care. 2015;38(1):132–139. doi: 10.2337/dc14-1958. [DOI] [PubMed] [Google Scholar]
- 48.Chaudhuri A, Ghanim H, Vora M, Sia CL, Korzeniewski K, Dhindsa S, Makdissi A, Dandona P. Exenatide exerts a potent antiinflammatory effect. J Clin Endocrinol Metab. 2012;97(1):198–207. doi: 10.1210/jc.2011-1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nonomura K, Iizuka K, Kuwabara-Ohmura Y, Yabe D. SGLT2 inhibitor and GLP-1 receptor agonist combination therapy substantially improved the renal function in a patient with type 2 diabetes: implications for additive renoprotective effects of the two drug classes. Intern Med. 2020;59(12):1535–1539. doi: 10.2169/internalmedicine.4323-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nandy D, Johnson C, Basu R, Joyner M, Brett J, Svendsen CB, Basu A. The effect of liraglutide on endothelial function in patients with type 2 diabetes. Diab Vasc Dis Res. 2014;11(6):419–430. doi: 10.1177/1479164114547358. [DOI] [PubMed] [Google Scholar]
- 51.Gupta V. Glucagon-like peptide-1 analogues: an overview. Indian J Endocrinol Metab. 2013;17(3):413–421. doi: 10.4103/2230-8210.111625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gorriz JL, Soler MJ, Navarro-Gonzalez JF, Garcia-Carro C, Puchades MJ, D’Marco L, Martinez Castelao A, Fernandez-Fernandez B, Ortiz A, Gorriz-Zambrano C et al. GLP-1 receptor agonists and diabetic kidney disease: a call of attention to nephrologists. J Clin Med. 2020, 9:4. [DOI] [PMC free article] [PubMed]
- 53.Dardano A, Miccoli R, Bianchi C, Daniele G, Del Prato S. Invited review. Series: implications of the recent CVOTs in type 2 diabetes: which patients for GLP-1RA or SGLT-2 inhibitor? Diabetes Res Clin Pract. 2020, 162:108112. [DOI] [PubMed]
- 54.Herrington WG, Preiss D, Haynes R, von Eynatten M, Staplin N, Hauske SJ, George JT, Green JB, Landray MJ, Baigent C, et al. The potential for improving cardio-renal outcomes by sodium-glucose co-transporter-2 inhibition in people with chronic kidney disease: a rationale for the EMPA-KIDNEY study. Clin Kidney J. 2018;11(6):749–761. doi: 10.1093/ckj/sfy090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.A Heart Disease Study of Semaglutide in Patients With Type 2 Diabetes (SOUL). https://clinicaltrials.gov/ct2/show/NCT03914326. Accessed 28 Oct 2020
- 56.A Research Study to See How Semaglutide Works Compared to Placebo in People With Type 2 Diabetes and Chronic Kidney Disease (FLOW). https://clinicaltrials.gov/ct2/show/NCT03819153. Accessed 28 Oct 2020
- 57.Ludwig L, Darmon P, Guerci B. Computing and interpreting the number needed to treat for cardiovascular outcomes trials: perspective on GLP-1 RA and SGLT-2i therapies. Cardiovasc Diabetol. 2020;19(1):65. doi: 10.1186/s12933-020-01034-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Supplementary tables.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.