Abstract
Non-alcoholic fatty liver disease (NAFLD) is considered the hepatic manifestation of metabolic syndrome and has become the major cause of chronic liver disease, especially in western countries. NAFLD encompasses a wide spectrum of hepatic histological alterations, from simple steatosis to steatohepatitis and cirrhosis with a potential development of hepatocellular carcinoma. Non-alcoholic steatohepatitis (NASH) is characterized by lobular inflammation and fibrosis. Several studies reported that insulin resistance, redox unbalance, inflammation, and lipid metabolism dysregulation are involved in NAFLD progression. However, the mechanisms beyond the evolution of simple steatosis to NASH are not clearly understood yet. Recent findings suggest that different oxidized products, such as lipids, cholesterol, aldehydes and other macromolecules could drive the inflammation onset. On the other hand, new evidence indicates innate and adaptive immunity activation as the driving force in establishing liver inflammation and fibrosis. In this review, we discuss how immunity, triggered by oxidative products and promoting in turn oxidative stress in a vicious cycle, fuels NAFLD progression. Furthermore, we explored the emerging importance of immune cell metabolism in determining inflammation, describing the potential application of trained immune discoveries in the NASH pathological context.
Keywords: non-alcoholic fatty liver disease, trained immunity, oxidative stress, hepatocellular carcinoma
1. Introduction
Non-alcoholic fatty liver disease (NAFLD) is defined as the pathological hepatocyte fat accumulation in >5% of liver tissue, in absence of viral hepatopathy, alcohol consumption, drug intake, and other secondary causes [1]. A wide spectrum of conditions exists, ranging from the “simple” hepatocyte fat accumulation to a more complex histologic picture characterized by cytolytic damage, lobular inflammation and fibrosis [2], namely non-alcoholic steatohepatitis (NASH). It can be complicated by the development of cirrhosis and/or hepatocellular carcinoma (HCC), determining hugely the worsening of the prognosis [1,2]. Therefore, due to the high incidence in western countries and the intricate clinical management, NAFLD has become one of the most important health care burden of the 21st century [1,2].
NAFLD has been studied as the hepatic complication of the metabolic syndrome (MS) as well as an independent risk factor for the development of cardiovascular diseases [1,3,4]. In fact, the major risk factors for NAFLD development and progression represented the pathologic background that defines MS, determining a further increase of the cardiovascular risk in a vicious cycle [4,5,6]. Therefore, in a scenario in which the prevalence of MS increases [7] and viral hepatitis are efficiently contrasted, NAFLD has become the first cause of liver transplantation in the western countries [1,7,8]. Nowadays it constitutes the global predominant hepatopathy showing a prevalence of 6–35% worldwide (25–26% in Europe), with wide variations due to the ethnicity [8,9].
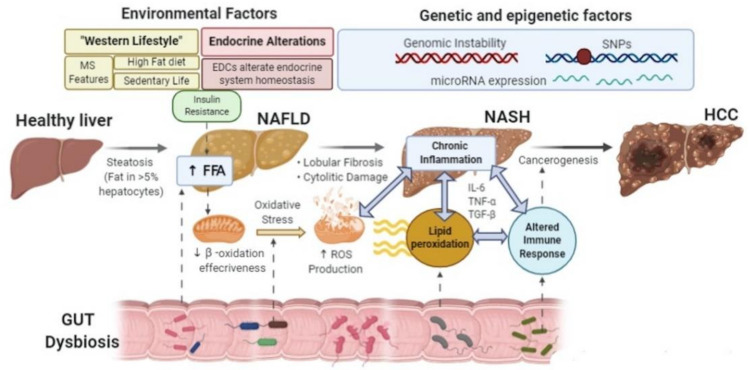
From a pathogenetic standpoint, NAFLD is considered the result of a multifactorial interaction among genetic background (including genomic instability, single nucleotide polymorphisms (SNPs), microRNA expression), environmental factors and dysmetabolism, with a prominent role played by insulin resistance (IR) [10,11] (Figure 1).
Figure 1.
The complexity of NAFLD pathogenesis as the result of multifactorial disease.SNPs: single nucleotide polymorphisms; miRNA: microRNA expression; MS: metabolic syndrome; FFAs: free fatty acids; ROS: reactive oxygen species; EDCs: endocrine-disrupting compounds; IL-6: Interleukin-6; TNF-α: rumor necrosis factor-α; TGF-β: transforming growth factor-β; NAFLD: non-alcoholic fatty liver disease; NASH: non-alcoholic steatohepatitis; HCC: hepatocellular carcinoma. Several factors (genetic, epigenetic, and environmental) are involved in the genesis and progression of NAFLD. The onset of an insulin resistance (IR) status is a critical event contributing to the beginning as well as the worsening of the disease. In this complex scenario, oxidative stress, inflammation, impaired immune response, and intestinal dysbiosis appear altogether to be able to work in concert and promote progression, impacting the chances of an HCC occurrence.
Accordingly, a recent expert consensus suggested that metabolic (dysfunction) associated fatty liver disease (MAFLD) would be a more appropriate definition [12]. The main factor in NAFLD pathogenesis has been identified in the hepatic accumulation of free fatty acids (FFAs) [13]. Mitochondria activity counteracts this FFAs overload, increasing the inner membrane permeability with the loss of its biologic function [14]. The direct consequence of this impairment is the mitochondrial dysfunction associated with the reactive species production, responsible in turn, of several consequences such as mtDNA mutations, lipotoxic intermediates accumulation, pro-inflammatory cytokines production, which promote IR, immune response and hence disease progression [15]. Moreover, new evidence indicates innate and adaptive immunity activation as the driving forces in establishing liver inflammation and fibrosis [16,17] (Figure 1).
In this review, we discuss the mechanisms underlying the crosstalk between oxidative stress and immune dysfunction in NAFLD evolution with new insights in the concepts of immune cell metabolism and trained immunity.
1.1. Overview on Molecular Mechanisms of Oxidative Stress in NAFLD
Chronic liver diseases are characterized by excessive production of reactive species and redox imbalance [18]. NAFLD is a very complex disease that involves several factors, genetic, epigenetic and environmental. Currently, the “multiple hit” pathogenesis is the accepted theory to explain its development, giving a mechanistic overview in which these factors act in parallel [19]. In this model, oxidative stress works as a cornerstone in NAFLD progression. Normally reactive species mediate a plethora of cellular functions such as apoptosis, proliferation, metabolism and immune defense regulation [20,21]. Conversely, the excessive reactive oxygen species (ROS) production leads to oxidative stress, compromising the physiological redox signaling with the consequent cellular damage [22,23]. This phenomenon acquires even more relevance in case of powerful ROS production, generated in a tissue oxidative microenvironment [24]. A typical example is represented by the iron overload which occurs in some NASH patients, facilitating the Fenton reaction that generates the extremely toxic HO from H2O2 [14].
Liver possesses potent scavenging systems to counteract the production of ROS deriving especially from mitochondria and cytochrome P450 (CYP) as well as other enzymes. However, when the ROS production overcomes the scavenging capacity, a redox imbalance occurs and superoxide anion radicals (O2·−) and hydrogen peroxide (H2O2) are continuously formed as bioproducts of energetic metabolism [14,25]. This involves hepatocytes, Kupffer cells (KCs), neutrophils and other liver cell types, underlying the crosstalk between oxidative stress and immunity in the progression of the disease. Moreover, FFAs deriving from diet, released by the adipose tissue or produced by de novo lipogenesis, overload the liver, serving as substrate for production of lipotoxic substances (e.g., ceramides, lysophosphatidylcholines and diacylglycerols) especially by mitochondria and CYP activities [26,27,28,29,30]. This metabolic stress promotes pro-inflammatory cytokine release and cell death that, in turn, represents one of the most important mechanisms of damage in the NAFLD histologic picture [31,32]. ROS can be generated in several organelles, such as mitochondria, endoplasmic reticulum (ER) and peroxisomes, but also by a lot of enzymes such as xanthine oxidase, CYP2E1, cyclooxygenases, NADPH oxidase (NOX) and lipoxygenase [20,33].
1.1.1. Oxidative Stress Derived from Mitochondrial Dysfunction
The most important source of ROS in NAFLD is represented by mitochondria, that also act by responsible of lipid metabolism dysregulation in case of impaired biologic functioning typically highlighted in metabolic disorders [34,35]. Accordingly, alterations in oxygen consumption, electron transport chain (ETC) activity and fatty acid oxidation (FAO) are widely described in literature. In fact, during early stages of NAFLD, the liver faces a high influx of external FAs and several studies showed the increase of FAO in different animal models of NASH [36,37,38,39,40], although some contrasting data exist in human ones [41,42,43,44,45,46,47,48,49,50,51,52]. A recent study, for instance, described the raising of FAO after 4 weeks in high fat diet (HFD)-fed mice, while after 8 weeks the normal β-oxidation was restored [53].
In the lipid-rich condition indeed, the liver tries to counteract the excessive production of lipotoxic molecules, by accumulating triglycerides and controlling FAO [24,54,55], even if several factors such as IR, leptin and enterokines might also play a role in this scenario [56]. ETC results also altered by FAs overload as the presence of FAs, especially Polyunsaturated fatty acids (PUFAs), generates O2·− and inactivates complex I and III [57,58,59,60], by direct interaction with ETC components or interference with membrane fluidity which provokes proton leakage [57,58,59,60,61,62]. The role of oxysterols is NAFLD progression should not be underestimated as suggested by novel evidence [63]. In fact, in a recent study it was shown that cholesterol supplementation in HFD-fed mice determined the hepatic accumulation of specific oxysterols (e.g., 7-hydroxycholesterol, 7-Ketocholesterol, and 5-cholestane-3, 5, 6-triol), which acted synergistically with FAs to impair mitochondrial function and biogenesis, facilitating NAFLD progression [27].
Several further discrepancies exist in literature about the mitochondrial dysfunction in simple steatosis and NASH such as alterations of cytochrome c oxidase (COX) activity [41,64,65,66,67,68,69], oxygen consumption of isolated mitochondria [70,71,72], oxidative phosphorylation (OXPHOS) efficiency [68,71,72] and ATP levels [72,73,74,75,76], either in patients and murine models. In particular, COX activity resulted unchanged, reduced or increased; ATP levels have been described as normal or reduced; mitochondrial oxygen uptake was augmented, normal or largely decreased; OXPHOS resulted augmented or unchanged [64,65,66,67,68,69,70,72,73,74,75,76].
1.1.2. ER Contribution to Oxidative Stress
An important contribute to oxidative stress derives also from ER stress. For instance, the production of H2O2 derives from transfer of electrons to molecular oxygen operated by ER oxidoreductase-1, which in turn received electrons from protein disulphide isomerase in the process of protein folding [24]. Several animal studies showed that the increased activity of C/EBP homologous protein (CHOP) is involved in the UPR-mediated ROS production during prolonged ER stress [77,78]. Disturbances in GSH/GSSH ratio in ER lumen entails protein misfolding and ROS production [79,80,81]. Furthermore, RNA-dependent protein kinase-like ER eukaryotic initiation factor-2 α kinase (eIF2a) phosphorylates nuclear factor erythroid 2–related factor 2 (Nrf2), promoting its translocation to the nucleus and the expression of antioxidants weapons such as hemeoxygenase-1 (HO-1) [82]. Therefore, the crosstalk between ER stress and mitochondrial dysfunction is of current interest and needs further studies to assess its specific contribution in this context.
2. Redox Biology and Immune Regulation in NAFLD Progression
Redox balance is primary involved in both adaptive and innate immunity, contributing to macrophage, lymphocyte and dendritic cell (DC) signaling and in cytokine response modulation [83]. The first evidence of oxidant role in immune activity was described in 1991, when it was shown that H2O2 at micromolar concentrations induced nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) transcription in human T lymphocytes [84].
In several liver diseases the crosstalk between redox imbalance and immune response has been described [85]. Inflammation and oxidative stress occur simultaneously in the liver, fueling each other. Moreover, persistent oxidative stress promotes the establishment of a chronic inflammation that represents the fil rouge for the detrimental interconnection between the liver disease and its systemic complications.
In fact, reactive species activate several transcriptional factors and receptors such as NF-κB, activator protein-1 (AP-1), p53, hypoxia-inducible factor 1-alpha (HIF-1α), peroxisome proliferator-activated receptor γ(PPAR-γ), β-catenin/Wnt, and Nrf2, which in turn modulate the expression of molecules involved in inflammation [86]. Of note, NF-κB is a key-mediator of ROS proinflammatory effect, being activated directly by reactive species and indirectly by ROS-damaged DNA [87].
In NAFLD, the excessive lipid accumulation is the primum movens of metabolic stress, which promotes innate immune activation, the driving force of inflammation in NASH [88,89,90]. In particular, KCs are activated by ROS and other stimuli and become the main hepatic producers of ROS, as well as cytokines, growth factors and chemokines [91,92,93].
2.1. The Role of PRRs
In NAFLD, the liver is continuously invaded by pathogen associated molecular patterns (PAMPs) (e.g., bacterial products, lipopolysaccharides—LPS), damage-associated molecular patterns (DAMPs) and metabolites (e.g., FFAs, mitochondrial DNA, lipotoxic products) which are sensed by pattern recognition receptors (PRRs), such as toll-like receptors (TLRs) and nucleotide-binding oligomerization domain-like receptors (NLRs) with the consequent activation of pro-inflammatory cascades [94,95,96,97]. The most known DAMP released by damaged cells is high mobility group box 1 protein (HMGB1), which activates TLR4 with the consequent myd-88 dependent NF-kB pathway induction [98,99]. In a murine model of ischemia/reperfusion damage, superoxide anion induced NOX activation by TLR4 in neutrophils [100]. Several experimental murine models showed that interaction between mtDNA and TLR-9 induced cytokine and fibrogenic responses in KCs and hepatic stellate cells (HSCs) [101,102,103]. However, a group of cytoplasmic PRRs is constituted by oligoadenylate synthase (OAS)-like receptors (OLRs), sensors of nucleic acids. In case of damage, dsDNA, microbial and mtDNA can be detected by OLRs, which activate transcription of proinflammatory genes by inhibitor of nuclear factor kappa-B kinase subunit ε (IKKε) and activator protein 1 (TBK1) signaling [17].
In hepatic inflamed areas and on HSCs there is a higher expression of receptors for advanced glycation end-products (RAGEs), a class of PRRs [104]. The AGEs formation promotes the generation of a considerable quantity of ROS; furthermore, oxidized RAGEs activate NOX1, contributing to ROS production [105]. NLR family pyrin domain containing 3 (NLRP3) is a PRR that interacts with PAMPs and DAMPs and is involved in generation of inflammasome complexes. In particular, ROS produced by mitochondria and NOXs promote NLRP3 inflammasome formation [106], while scavengers such as catalase and superoxide dismutase (SOD) inhibit its activation [107]. Accordingly, it has been suggested that the crystal structure of NLRP3 is very sensitive to red ox balance variations [108].
Overall, PRRs activate several intracellular targets such as Transforming Growth Factor-β (TGF-β) activated kinase 1 (TAK1) and apoptosis signal-regulating kinase 1 (ASK1), both members of mitogen-activated protein kinase kinase kinases (MAP3K) family, which activate in turn other kinases such as c-Jun-N-terminal chinasi (JNK), adenosine monophosphate-activated protein kinase (AMPK) and IkB, hence with a key role in NASH pathogenesis [109,110,111].
Interestingly, apart from receptor interaction, several reports described that ROS could influence downstream innate immunity signaling [112]. For instance, H2O2 increases activity of MAPKs (e.g., p38) and therefore, JNK pathway, which in turn promotes the production of further ROS [113,114,115]. Moreover, NF-kB, AP-1 and PPARs can be directly modulated by oxidative species, inducing transcription of cytokines [116,117,118,119]. ROS and lipid peroxidation products such as hydroxinonenal- (HNE-) and malondialdehyde- (MDA-) adducts can activate NF-kB directly or by inhibition of IKKs phosphorylation in innate immune cells [120,121,122]. Therefore, the natural consequence of persistent stimuli is the chronic inflammation, activation of HSCs with collagen deposition, increased IR and further aberrant expression of pro- and antioxidant enzymes [123,124,125]. However, the presence of redox imbalance and lipid peroxidation is a common characteristic of NAFLD and NASH [26] and the involvement of adaptive immunity seems to be a key factor in NAFLD progression. Accordingly, recently Azzimato V. et al. described that liver macrophages are proinflammatory in NASH, but not in NAFLD and obesity, although sings of oxidative stress are present in both conditions as NRF2 is downregulated [126].
2.2. The Role of OSEs
Oxidative stress has been described as the main trigger in stimulating adaptive immunity, especially through oxidized phospholipids and aldehydes generated by lipid peroxidation such as MDA, malondialdehyde-acetaldehyde (MAA) and 4-HNE-protein adducts, formally named oxidation-specific epitopes (OSEs) [127,128,129]. OSEs are implicated in several systemic diseases and atherosclerosis, by stimulating both innate and adaptive immunity [127,130].
Specific anti-OSE immunoglobulin G (IgG) are highly prevalent (about 40%) in adult with NAFLD or NASH and in children with NASH (60%). Moreover, high they have been associated with fibrosis, severity of hepatic lobular inflammation and intrahepatic infiltrate of B cell and T cell aggregates [128,131,132]. These observations were corroborated in animal models of NAFLD and NASH. In particular, titers of anti-OSE IgG correlated with B cell maturation in plasma cells, while antioxidant treatment with N-acetylcysteine prevented antibody response [131,133,134]. Mice pre-immunized with protein MAA-adducts present a Th1 polarization of CD4+ T lymphocytes in the liver, which produce IFN-γ, promoting KCs M1 activation [131]. Accordingly, in patients with NASH anti-OSEs IgG correlate with circulating Interferon γ (IFN-γ) levels [133]. In fact, the involvement of Th1 and Th17 lymphocytes in NAFLD evolution to NASH has been widely reported [131,135,136,137,138]. In parallel, in NAFLD murine models the quantity of hepatic CD4+ regulatory T cells (Tregs) lymphocytes is reduced [139,140]. Treg cells are typically involved in maintaining self-tolerance and controlling excessive T-cell activation in the liver [141,142]. It seems that ROS promote Treg cells apoptosis, hence facilitating Th1 and Th17 proinflammatory activity [139,140]. Therefore, the B cell activity with anti-OSE IgG production mirrors the presence of oxidative stress and inflammation. By contrast, it has been described that anti-OSEs IgM decrease in NAFLD patients compared to controls and inversely correlate with obesity and liver damage [143]. Accordingly, T15 natural IgM against oxidized phosphatidylcholine protect against NASH in LDLR KO mice fed with HF-HC diet, probably by preventing the oxidized-LDL-induced activation of KCs [144].
Overall, these findings underline how oxidative imbalance behaves as the cornerstone of NAFLD progression, fueling innate immunity to produce cytokines and new reactive species which perpetuate inflammation thorough adaptive immune cells (summary in Table 1).
Table 1.
Immune system-oxidative stress crosstalk main mechanisms in NAFLD progression.
| Main Mechanisms and Activities | Main Effects and Epiphenomena |
|---|---|
| Redox unbalance: ROS 11 production overcomes the scavenging capacity so that superoxide anion radicals (O2·−) and hydrogen peroxide (H2O2) are continuously formed. As a consequence: |
Perpetuation of chronic inflammation Creation of a vicious circle where the chronic inflammation and the oxidative stress fuelling each other, involving adaptive immune cell mechanisms, contribute to the disease progression. Increased insulin resistance Chronic inflammation worsens insulin resistance, FFAs 1 accumulation and thus NAFLD evolution. |
| |
| |
| |
| OSEs 3: oxidative stress promotes the production of oxidised phospholipids and aldehydes generated by lipid peroxidation such as MAA 4 and 4-HNE 5-protein adducts. As a consequence: |
Inflammation and Liver Damage Pro-inflammatory cytokine release and cell death promote the worsening of liver histological damage |
| |
| PAMPs 13 and DAMPs 14 hepatic invasion:13 PAMPs (e.g., LPS 15), 14 DAMPs (e.g., HMGB1, mtDNA, lipotoxic products, and other metabolites) continuously invade the liver in NAFLD 16. As a consequence: |
Inflammation and Liver Damage Pro-inflammatory cytokine release promotes the worsening of liver histological damage. Fibrosis By HSCs fibrogenic response. |
| |
|
1 FFAs: Free Fatty Acids; KCs: 2 Kupffer cells; 3 OSEs: oxidation-specific epitopes; 4 MAA: malondialdehyde-acetaldehyde; 5 HNE: dydroxinonenal; 6 NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; 7 AP-1: activator protein-1; 8 HIF-1α: hypoxia-inducible factor 1-alpha; 9 PPAR: peroxisome proliferator-activated receptor; 10 Nrf2: nuclear factor erythroid 2–related factor 2; 11 ROS: reactive oxygen species; 12 Tregs: regulatory T cells; 13 PAMPs: Pathogen Associated Molecular Patterns; 14 DAMPs: damage-associated molecular patterns; 15 LPS: lipopolysaccharides; NAFLD: 16 non-alcoholic fatty liver disease; 17 HSCs: hepatic stellate cells; 18 TLRs: toll-like receptors; 19 NLRS: nucleotide-binding oligomerization domain-like receptors; 20 OLRs: oligoadenylate synthase (OAS)-like receptors; 21 RAGEs: receptors for advanced glycation end products; 22 PRRs: pattern recognition receptors.
3. The Immune Response in HCC Development
Nowadays HCC represents the fifth most common tumor and the third leading cause of cancer-related death worldwide, resulting in over 600,000 deaths annually [145]. It can occur in the NAFLD context independently to the surrounding advanced fibrosis or cirrhosis, complicating the course of the disease and worsening hugely the prognosis [146].
HCC pathogenesis still remains extremely complex, embracing various and not completely clarified biological dynamics. In the last few decades, in parallel to the genetic and epigenetic mechanisms surrounding HCC development and worsening, several findings revealed three linked elements creating a tumoral microenvironment: the chronic phlogosis, the oxidative stress and the immune system dysregulation [146,147,148].
Normally, inflammation represents the adaptive response to tissue damage, involving different mechanisms (blood vessel dilatation, immune cell recruitment, and cytokines production) and aiming to damage resolution and homeostasis recovering [149].
We have previously described the importance of the bidirectional relationship between oxidative stress and immunity in NAFLD progression. Moreover, when the inflammatory stimuli persist or the regulatory mechanisms are disrupted, a “non-resolving inflammation” occurs, determining pathological consequences such as fibrosis and metaplasia, as described in several human diseases, including HCC [149,150]. Therefore, chronic inflammation is critical for HCC development so that this tumor represents a classic paradigm of inflammation-linked cancer [151]. In line with the current scientific knowledge, several studies revealed that more than 90% of HCCs arise in the context of hepatic inflammation and, interestingly, the use of non-steroidal anti-inflammatory agents decreases its incidence and/or recurrence [149,152,153,154].
The innate immune system is triggered by oxidative stress and persistent injury to react not only against “non-self” potential pathogens but also to DAMPS, encouraging thus the emerging concept of “sterile inflammation” that seems to be very crucial in HCC development [155,156].
As previously mentioned, most of DAMPs are produced under condition of oxidative stress and/or mitochondrial dysfunction [156,157]. The high ROS production induces mtDNA damage, triggering mitochondrial dysfunction and generating thus a vicious cycle in which a deficient production of enzymes involved in the respiratory electron transport chain occurs, worsening thus ROS production [158]. After being released into the cytosol and extracellular environment, mtDNA acts as DAMP, since it contains the unmethylated CpG DNA motifs [159,160].
Furthermore, mitochondrial dysfunction is also associated to dysregulation of mitochondrial fission, mitochondrial permeability transition (MPT) pores, and necroptosis (a form of programmed cellular death), all involved in NASH and liver cancer progression [158,161,162,163].
As already described, an important DAMP is HMGB1, a ubiquitous nuclear non-histone protein that participates in DNA replication, transcription, and repair [164]. The biologic activity of released HMGB1 depends on its redox state (fully oxidized or disulphide), which is modulated by the oxidative features of the tumoral microenvironment [165].
The fully oxidized HMGB1 form promotes immunotolerance, reducing the maturation and the activation of DCs and favoring thus tumor immune escape mechanisms [165].
DCs are an important hepatic cellular population, whose alterations, in terms of reduced functionality such as reduced maturation and failure of HCC-associated antigens presentation, are involved in tumor pathogenesis [166,167,168]. In HCC patients, the amount of activated DCs (CD83-positive DCs) in hepatic nodules is lower than in cirrhotic or healthy control tissues [169]. Moreover, the lower DCs expression of human leukocyte antigen (HLA) class-Ⅰ molecules implies the loss of antigen presentation activity, thus contributing to impaired recruitment of tumor-specific lymphocytes [170]. Considering the major function of DCs in the interplay between innate and adaptive immune reactions, the disruption of the efficiency of these mechanisms causes a weak T cell immune response against cancer [171]. Accordingly, CD14+ cytotoxic T-lymphocyte-associated protein (CTLA)-4+ DCs, a subset recently identified, are over-represented in tumoral tissue of HCC patients and are involved in the production of some regulatory cytokines (e.g., IL-10), which suppress the CD4+ T-cell immune response, facilitating the tumor progression [172].
As already discussed, HMGB1 disulphide form behaves as a DAMP, activating TLR4, and promoting innate immune response with pro-inflammatory and chemoattractant effects [173].
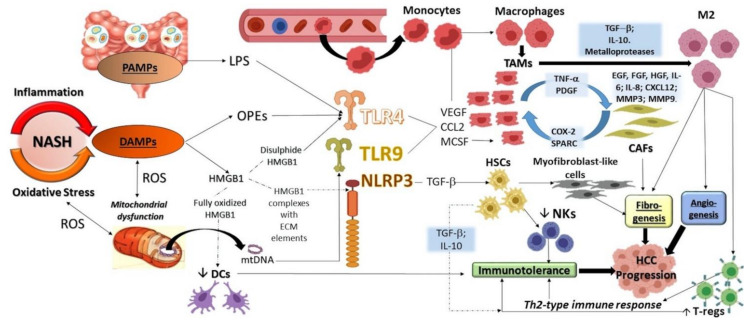
The activation of TLRs, through the stimulation of JNK and NF-kB pathways, leads to different consequences, including among others, the production of Tumor Necrosis Factor-α (TNF-α) (able to induce the proliferation of tumor cells) as well as many other mediators involved in the recruitment and polarization of different inflammatory cell types [174,175,176,177]. In particular, high levels of macrophage colony-stimulating factor (MCSF) and chemokine (C-C motif) ligand 2 (CCL2), vascular endothelial growth factor (VEGF), and TGF-β induce macrophages recruitment and infiltration in peri-tumoral tissue, their polarization in M2 phenotype as well as the differentiation in tumor-associated macrophages (TAMs) [178,179] (Figure 2).
Figure 2.
Oxidative stress, inflammation, and immune dysregulation in HCC progression. NASH: non-alcoholic steatohepatitis; ROS: reactive oxygen species; DAMPs: damage-associated molecular patterns; PAMPs: pathogen-associated molecular patterns; LPS: lipopolysaccharide; OPEs: oxidation-specific epitopes; HMGB1: High Mobility Group Box 1; ECM: extracellular matrix; NLRP3: NOD-like receptor family pyrin domain containing-3; TLR: Toll-like receptor; DCs: dendritic cells; NKs: natural killer cells; HSCs: hepatic stellate cells; CAFs: cancer-associated fibroblasts; MCSF: macrophage colony-stimulating factor; CCL2: chemokine (C-C motif) ligand 2; VEGF: vascular endothelial growth factor; EGF: Epidermal growth factor; HGF: hepatocyte growth factor; FGF: fibroblast growth factor; IL-6: interleukin 6; IL-8: Interleukin 8 chemokine; CXCL12: C-X-C motif ligand 12; MMP3: matrix metalloproteases 3; MMP-9: matrix metalloproteases 9; TGF-β: transforming growth factor-β; IL-10: Interleukin 10. Solid lines indicate direct and stable mechanisms; dashed lines indicate indirect and potential ones.
Inflammation enchained with oxidative stress (both constantly present in the NASH context), mitochondrial dysfunction (including mtDNA releasing), and gut microbiota alterations (particularly, the action of LPS) all promote the creation of DAMPS and PAMPS. These molecules, in turn, are able to stimulate and activate TLRs and Inflammasomes resulting in cytokines, chemokines, and growth factors production. These events implicate several consequences and in particular: (i) monocytes recruitment, activation, and differentiation in TAMs, cells able to contribute to the angiogenesis and fibrosis, working in concert with CAFs; (ii) HSCs activation and differentiation in myofibroblast-like cells worsening thus the fibrogenesis. Moreover, activated HSCs contribute to the immuno-tolerance, disrupting NKs functions (particularly, NKs capability to induce HSCs apoptosis) and favoring T-regs activation. These last events are facilitated also by the abovementioned TAMs able to promote a Th2 immune response, thus leading to an immuno-tolerance condition. Altogether, the above-illustrated mechanisms contribute to the HCC worsening and progression.
Moreover, ROS production represents a crucial point in the process of macrophagic differentiation into M2 and TAMs, and in fact, antioxidant treatment, reducing superoxide O2- production, inhibits TAMs differentiation and tumorigenesis in mouse models of cancer [180,181].
Moreover, TAMs can further differentiate in immunotolerant M2 macrophages, whose presence in HCC is associated with poor survival rate after curative resection [182]. These cells, in fact, through the secretion of cytokines (such as TGF-β and IL-10), chemokines, growth factors, and matrix metalloproteases, can contribute to fibrosis, tumor growth and progression, intrahepatic metastasis development, angiogenesis, and, most notably, immune suppression by promoting Th2-type immune response [182,183].
The gene expression profile of HCC patients reveals a shift from a Th1 to a Th2 immune response, suggesting a central role played by cellular immune dysregulation in hepatic tumor appearance [184]. The mechanisms underlying this change are not completely clarified yet, although different mediators produced by either stromal cells and tumour cells, such as IL-10, glucocorticoid hormones, ROS, apoptotic cells, and immune complex contribute to create a particular microenvironment that facilitates the polarization of T cells in Th2 lymphocytes [185]. This shift entails an increase of Th2 cytokines levels (i.e., IL-4, IL-5, and IL-10) and a reduction of Th1 ones (i.e., IFN-γ, IL-8, IL-15, IL-18, and IL-2). This is relevant as CD4+ Th1 lymphocytes, through production IFN-γ, enhance anti-tumoral response in HCC and accordingly, a significant decrease of CD4+ Th1-cells in patients with liver cirrhosis and HCC has been reported, highlighting their importance in liver cancer biology [186,187].
Interestingly, M2 differentiated TAMs promote immune tolerance also favoring the T-regs recruitment [178]. Tregs, characterized by the CD25 expression on their surface and by the activity of intracellular transcriptional factor forkhead box P3 (Foxp3), physiologically ensure self-tolerance by suppressing self-reactive immune cells. In particular, HCC is associated with the presence of a particular T-regs subset, namely induced T-regs (iTregs), both in the peripheral blood and in tumoral microenvironment. The iTregs depletion seems to be able to determine anti-tumoral immune responses [188,189] In this regard, the production of TGF-β and the induction of T-regs seems to be favored by myeloid-derived suppressor cells (MDSCs), a subset of inflammatory monocytes highly represented in tumoral tissue of HCC patients [190].
Moreover, an aberrant cooperation between TAMs and liver mesenchymal cells seems to be involved in the generation of a vicious cycle triggering the tumor onset. Cancer-associated fibroblasts (CAFs) can produce epidermal growth factor (EGF), hepatocyte growth factor (HGF), fibroblast growth factor (FGF), IL-6, IL-8, chemokine C-X-C motif ligand 12 (CXCL12), and matrix metalloproteases (MMP-3 and MMP-9) [179,191,192]. Through cyclooxygenase 2 (COX-2) and secreted protein acidic rich in cysteine (SPARC), CAFs recruit and stimulate TAMs, which can further increase the activation of fibroblasts through the secretion of TNF-α and Platelet-Derived Growth Factor (PDGF), generating a biologic mechanism by which phlogosis and fibrosis stimulate each other with the involvement of HSCs [193] (Figure 2). Once again, interestingly, the oxidative stress plays a central role in the biologic dynamics surrounding this process as proved by various clinical and animal models that suggest the ROS and lipoperoxidation derived products (particularly H2O2 and MDA-adducts) ability to promote the HSCs activation [194].
The activation of HSCs represents a pivotal moment in this picture. Indeed, activated HSCs can differentiate in a senescent profile, inducing the recruitment and the activation of immune cells from the bloodstream, including TAMs, as well as increasing the growth factor receptor signaling, in particular PDGF and TGF-β [195]. In line whit this, recent findings suggest the TGF-β ability to inhibit the activation and functions of Natural Killer (NK) cells by repressing the mammalian target of rapamycin (mTOR) pathway [196]. NK cells normally mediate their functions producing cytolytic granules containing perforin, granzymes, tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), and IFN-γ, therefore playing a double role: cytolytic action and regulation of the other immune cells [197].
In NK cells the receptor activity, modulated by activated HSCs, inhibits the normal functioning of hepatic NK cells contributing thus to the development and progression of the liver cancer [198]. Even more in the HCC context, the NKs deficit of function may play a critical role, since these cells are able to induce apoptosis of activated HSCs, resulting by the end to exert an anti-fibrotic effect [199]. Moreover, persistent inflammation and oxidative stress generates apoptosis-resistant activated HSCs, enhancing fibrosis deposition [200].
Activated HSCs are also implicated in the promotion of immunotolerance mechanisms, secreting cytokines, which induce MDSC expansion and T-regs recruitment in tumor tissue [201]. As already mentioned, T regs in turn are able to interfere with the innate immunity, regulating NK cells and DCs activity (primarily through anti-inflammatory cytokines production such as TGF-β and IL-10) [202]. Moreover, T-regs could also disrupt the adaptive immunity response, by suppressing T-cell proliferation, CD8+ T-cells infiltration in cancer tissue, and IFN-γ secretion [203,204]. As known, IFN-γ secretion represents a fundamental moment to promote the activation of naive CD4+ and CD8+ T cells against pathogens and cancer [205]. For effective T-cell activation, in fact, the antigen presentation by Antigen Presenting Cells (APCs) to the CD4/CD8+ T-cell receptor is not sufficient [206] and the interplay between lymphocytes costimulatory receptors (g. e. CD28, ICOS, CD137, OC40, and CD27) and their relative ligands (g. e. CD80/86, B7RP1, CD137L, OC40L, and CD70) on APCs results essential [207]. Meanwhile, the physiological modulation of T-lymphocyte response is mediated by co-inhibitory receptors activated by other ligands [207]. These interactions, including CTLA-4-CD80/86 and programmed death 1 (PD-1)-PD-L1, represent the so-called “immune checkpoints”. Relevantly, chronic inflammation in NASH creates a tissue microenvironment that favors exhausted T-cells development: these cells express high levels of coinhibitory receptors (e.g. TLA-4 and PD-1) and low effector cytokines, with consequent impaired anticancer cytotoxicity [208].
Therefore, given this evidence, the possibility to regulate the immune system, nowadays is considered a very fascinating therapeutic frontier. In particular, the administration of immune checkpoint inhibitors (ICI), as anti-CTLA-4 and PD1 drugs, represents an encouraging therapeutic strategy in patients with this neoplasm [209].
Overall, these studies show how the immune system dysregulation in HCC, in terms of quantity and function alterations of immune cells, contribute to creating a tumoral microenvironment in which cancer development and progression are out of control.
In this heterogeneous scenario embracing very different cell populations, the oxidative imbalance could represent the fil rouge that connect all the elements of this inflammatory network.
4. Future Perspectives: The Role of Trained Immunity
By the years, the NAFLD has become one of the most important health problems in the era of obesity pandemic and strength scientific efforts have been oriented in understanding the mechanisms underlying its appearance and evolution, in which the interpretation seems to be represented by inflammation persistence, largely linked to the macrophages activation [210]. As previously mentioned, these cells could be basically involved in driving this complicated pathological picture in developing several metabolic consequences like IR, obesity, type II diabetes mellitus and atherosclerosis, all processes ground on an impaired control of the oxidative stress imbalance [211].
The medicine progress and the new advanced scientific knowledge have changed the old definition of immune system functioning. Multiple contacts with some antigens (e.g., PAMPs, DAMPs), as well as metabolic stimuli, would be able to produce a secondary immune response not exclusively made by adaptive immune cells [212,213]. According to the international scientific community, the term “Trained Immunity” (TI) represents a totality of immunological processes, which involve cells of non-specific immunity, in particular monocytes and NKs, deriving from a second antigenic contact [212]. This second contact would be able to produce the activation of several signaling pathways, which have different effects on the cellular genic expression that in turn, are mainly oriented to produce a more powerful immune reaction against antigens in comparison to the first contact [212,214].
Differently from immune adaptation which occurs in B lymphocyte, through the production of bone marrow cell clusters able to elaborate a specific response against a given antigen, this type of adaptation is mainly based on epigenetic changes, linked to a different histone’s acetylation or to the action of some siRNA like miR-155 and, therefore, it has a duration that can vary from few days to some years [212,215,216]. A TI response is not “antigen-specific” and can be transmitted to new cell generation in medullar maturation, giving them the possibility to react against the antigens with a typical “trained response” [217,218,219].
However, to better clarify the reason of a possible involvement of TI in the complex NAFLD scenario, one of the most important aspect is the interconnection between TI response and cell metabolism [220,221]. The immunometabolism represents the cornerstone research field in understanding the regulation of the inflammatory response (such as in M1 macrophages), which is supported mainly by the glycolytic activity and with a consequent stop of the Krebs cycle. Some metabolic products of this cycle such as succinate or alpha ketoglutarate play important roles as regulating mediators in this process [222].
Moreover, some enzymes of mitochondrial ETC are involved in the TI, including succinate dehydrogenase (SDH), that would represent one of the most important biological targets of itaconate [223]. Altogether, these scientific evidences confirmed the crucial role of the mitochondria in this complex biologic network by which the pathogenesis of some immune-metabolic-mediated diseases, not completely known yet, could be better clarified.
The balance between succinate and alpha ketoglutarate determines the regulation of some effectors of TI including: jumonji domain-containing (JMJ) and ten eleven translocation (TET), which would mainly act through the modification of the histones acetylation, like H3K4 [224]. To this purpose, the investigation of the monocyte mitochondrial metabolism and the cellular switch from the ETC to glycolytic prevalent energetic pathway in animal (high fat diet model) and clinical models of NAFLD could be the useful routes to fully understand the involvement of TI in this complex pathologic picture.
Moreover, another important point that could be assessed in this context is represented by the Itaconate activity, one the principal regulating mediator of TI, deriving directly from the conversion of cis-aconitate made by immune responsive gene 1 (IRG1) enzyme [225,226].
IRG-1 expression is genetically and epigenetically regulated by LPS, that encourages its expression through a pathway that involves protein kinase C (PKC) activation [227]. The miR93 whereas, decreases IRG-1 expression, by acting on the transcription factor Interferon Regulatory Factor-9 [228].
Itaconate act as an anti-inflammatory mediator, ready to regulate the unjustified and excessive activation of inflammatory cascade, acting through different mechanisms: stops the activity of SDH, involved in the Coenzyme Q synthesis and then inflammasome activation and fructose-6-phosphate kinase; forms complexes with glutathione; inhibits the transcription IkB-zeta factor, involved in the synthesis of IL-6; activates the Nrf2 through the kelch-like ECH associated protein 1 (KEAP1) alkylation [229,230]. LPS is able to induce an increase of proinflammatory cytokines production, soon followed by a stimulus for itaconate synthesis through the IRG-1 activation. In a paper recently published, it has been highlighted the importance of the TI in a mouse model of ischemia/reperfusion liver damage, used as well-known model of liver oxidative stress damage, in which the Nrf2 activity seems to be essential in this setting [231,232]. Due to the main role of macrophages in NAFLD and their essential involvement in the regulation of oxidative stress imbalance, not only in the liver but also in peripheral tissues like adipose and vascular ones, it seems to be reasonable to explore the possible involvement of the TI in this context. The assessment of Itaconate synthesis, IRG-1 expression as well as the synthesis and the biologic response derived from the activity of the above-mentioned transcription factors and/or the investigation of the effect derived from the administration, in animal models, of the itaconate, represent some important research points that are waiting to be addressed.
Moreover, in the light of the increased intestinal permeability to several immune-triggering antigens in NAFLD patients, this hypothesis acquires even more interest. This could give the opportunity not only to understand better the pathogenetic process surrounding NAFLD, but could drive the scientific community to the comprehension of its natural history and complications onset, as well as to evaluate it as a potential therapeutic target in the actual burden of the optimal medical management [233,234]
5. Conclusions
The complexity of NAFLD pathogenesis does not permit to individualize mechanisms incontrovertibly responsible of disease development and progression. Consequently, specific and efficient treatments are not available yet, making NAFLD a health care burden worldwide. However, a determinant contribute to the knowledge is deriving from studies on redox biology and hepatic immunity. It is well known that redox imbalance is involved in chronic liver diseases and all NAFLD stages, highlighting redox alterations as the triggering force of inflammatory response, although it is still not clear why this happens only to a part of patients. The potential involvement of pre-existing immunity factors should be explored to explain whether redox imbalance affects a susceptible immune system.
Moreover, through studies on redox biology in the immune cells, very recent reports are shedding light on the importance of immune cell metabolism, developing new concepts like trained immunity and indicating new potential therapeutic targets. Therefore, a deeper understanding of molecular mechanism underlying the crosstalk between redox biology and immunity are eagerly needed to provide a significant contribute to novel therapeutic approaches.
Acknowledgments
The representation of cellular structures, molecular pathways, and other elements in the figures was created using the graphic support and the available icons offered by the BioRender software (biorender.com).
Abbreviations
| AMPK | Adenosine monophosphate-activated protein kinase |
| AP-1 | Activator protein-1 |
| APCs | Antigen Presenting Cells |
| ASK 1 | Apoptosis signal-regulating kinase 1 |
| CAFs | Cancer-associated fibroblasts |
| CCL-2 | Chemokine (C-C motif) ligand 2 |
| CHOP | C/EBP Homologous Protein |
| COX | Cytochrome c oxidase |
| COX-2 | Cyclooxygenase 2 |
| CTLA | CD14+ cytotoxic T-lymphocyte-associated protein |
| CXCL12 | Chemokine C-X-C motif ligand 12 |
| CYP | Cytochrome P450 |
| DAMPs | Damage-associated molecular patterns |
| DC | Dendritic cell |
| EGF | Epidermal growth factor |
| eIF2a | RNA-dependent protein kinase-like ER eukaryotic initiation factor-2 α kinase |
| ER | Endoplasmic Reticulum |
| ETC | Electron transport chain |
| FAO | Fatty acid oxidation |
| FFAs | Free fatty acids |
| FGF | Fibroblast growth factor |
| FoxP3 | Forkhead box P3 |
| HCC | Hepatocellular Carcinoma |
| HFD | High fat diet |
| HGF | Hepatocyte growth factor |
| HIF-1α | Hypoxia-inducible factor 1-alpha |
| HLA | Human leukocyte antigen |
| HMGB1 | high mobility group box 1 protein |
| HNE | Dydroxinonenal |
| HO-1 | Hemeoxygenase-1 |
| HSCs | Hepatic stellate cells |
| ICI | Immune checkpoint inhibitors |
| IFN-γ | Interferon γ |
| IKK | Inhibitor of nuclear factor kappa-B kinase |
| IR | Insulin Resistance |
| IRG1 | Immune responsive gene 1 |
| JMJ | Jumonji domain-containing |
| JNK | c-Jun-N-terminal kinase |
| KCs | Kupffer cells |
| KEAP1 | Kelch-like ECH associated protein 1 |
| LPS | Lipopolysaccharides |
| MAA | Malondialdehyde-acetaldehyde |
| MAFLD | Metabolic (dysfunction) associated fatty liver disease |
| MAP3K | Mitogen-activated protein kinase kinase kinase |
| MAPK | Mitogen activated protein kinase. |
| MCSF | Macrophage colony-stimulating factor |
| MDA | Malondialdehyde |
| MDSCs | Myeloid-derived suppressor cells |
| MMP | Matrix metalloproteases |
| MPT | Mitochondrial permeability transition |
| MS | Metabolic Syndrome |
| mTOR | Mammalian target of rapamycin |
| NAFLD | Non-alcoholic fatty liver disease |
| NASH | Non-alcoholic steatohepatitis |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NK | Natural Killer |
| NLRP | NLR family pyrin domain containing |
| NLRS | Nucleotide-binding oligomerization domain-like receptors |
| NOX | NADPH oxidase |
| Nrf2 | Nuclear factor erythroid 2–related factor 2 |
| OLRs | Oligoadenylate synthase (OAS)-like receptors |
| OSEs | Oxidation-specific epitopes |
| OXPHOS | Oxidative phosphorylation |
| PAMPs | Pathogen Associated Molecular Patterns |
| PDGF | Platelet-Derived Growth Factor |
| PPARs | Peroxisome proliferator-activated receptor |
| PD1 | Programmed cell Death 1 |
| PRRs | Pattern recognition receptors |
| PUFAs | Polyunsaturated fatty acids |
| RAGEs | Receptors for advanced glycation end products |
| ROS | Reactive oxygen species |
| SDH | Succinate dehydrogenase |
| SNPs | Single Nucleotide Polymorphisms |
| SOD | Superoxide dismutase |
| SPARC | Secreted protein acidic rich in cysteine |
| TAK 1 | TGF-β-activated kinase 1 |
| TAMs | Tumour-associated macrophages |
| TET | Ten eleven translocation |
| TGF-β | Transforming Growth Factor-β |
| TI | Trained Immunity |
| TLRs | Toll-like receptors |
| TNF-α | Tumor Necrosis Factor-α |
| TRAIL | Tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) |
| Tregs | Regulatory T cells |
| VEGF | Vascular endothelial growth factor |
Author Contributions
Conceptualization, M.D. and A.F.; Resources, C.L. and G.S.; Writing—Original Draft Preparation, M.D., M.S., M.R.; Visualization, C.L. and A.F.; Supervision, R.V. and A.D.R.; Project Administration, A.F. and M.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bellentani S. The epidemiology of non-alcoholic fatty liver disease. Liver international. Off. J. Int. Assoc. Study Liver. 2017;37(Suppl. 1):81–84. doi: 10.1111/liv.13299. [DOI] [PubMed] [Google Scholar]
- 2.Lindenmeyer C.C., McCullough A. The Natural History of Nonalcoholic Fatty Liver Disease—An Evolving View. Clin. Liver Dis. 2018;22:11–21. doi: 10.1016/j.cld.2017.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Abenavoli L., Boccuto L., Federico A., Dallio M., Loguercio C., Di Renzo L., De Lorenzo A. Diet and Non-Alcoholic Fatty Liver Disease: The Mediterranean Way. Int. J. Environ. Res. Public Heal. 2019;16:3011. doi: 10.3390/ijerph16173011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wijarnpreecha K., Aby E.S., Ahmed A., Kim D. Evaluation and Management of Extrahepatic Manifestations of Nonalcoholic Fatty Liver Disease. Clin. Mol. Hepatol. 2020;14, 2:168–178. doi: 10.3350/cmh.2020.0239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Samuel V.T., Shulman G.I. Nonalcoholic Fatty Liver Disease as a Nexus of Metabolic and Hepatic Diseases. Cell Metab. 2018;27:22–41. doi: 10.1016/j.cmet.2017.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Federico A., Dallio M., Masarone M., Persico M., Loguercio C. The epidemiology of non-alcoholic fatty liver disease and its connection with cardiovascular disease: Role of endothelial dysfunction. Eur. Rev. Med. Pharmacol. Sci. 2016;20:4731–4741. [PubMed] [Google Scholar]
- 7.Saklayen M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018;20:1–8. doi: 10.1007/s11906-018-0812-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Masarone M., Federico A., Abenavoli L., Loguercio C., Persico M. Non Alcoholic Fatty Liver: Epidemiology and Natural History. Rev. Recent Clin. Trials. 2015;9:126–133. doi: 10.2174/1574887109666141216111143. [DOI] [PubMed] [Google Scholar]
- 9.Younossi Z.M., Blissett D., Blissett R., Henry L., Stepanova M., Younossi Y., Racila A., Hunt S., Beckerman R. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology. 2016;64:1577–1586. doi: 10.1002/hep.28785. [DOI] [PubMed] [Google Scholar]
- 10.Sherlock S. In: Sherlock’s Diseases of the Liver and Biliary System. 13th ed. Dooley J.S., Garcia-Tsao G., Pinzani M., editors. Volume 28. John Wiley & Sons Ltd; Hoboken, NJ, USA: 2018. pp. 540–560. [DOI] [Google Scholar]
- 11.Dallio M., Masarone M., Errico S., Gravina A.G., Nicolucci C., Di Sarno R., Gionti L., Tuccillo C., Persico M., Stiuso P., et al. Role of bisphenol A as environmental factor in the promotion of non-alcoholic fatty liver disease: In vitro and clinical study. Aliment. Pharmacol. Ther. 2018;47:826–837. doi: 10.1111/apt.14499. [DOI] [PubMed] [Google Scholar]
- 12.Eslam M., Sanyal A.J., George J. International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158:1999–2014.e1. doi: 10.1053/j.gastro.2019.11.312. [DOI] [PubMed] [Google Scholar]
- 13.Pierantonelli I., Svegliati-Baroni G. Nonalcoholic Fatty Liver Disease: Basic Pathogenetic Mechanisms in the Progression from NAFLD to NASH. Transplantation. 2019;103:e1–e13. doi: 10.1097/TP.0000000000002480. [DOI] [PubMed] [Google Scholar]
- 14.Masarone M., Rosato V., Dallio M., Gravina A.G., Aglitti A., Loguercio C., Federico A., Persico M. Role of Oxidative Stress in Pathophysiology of Nonalcoholic Fatty Liver Disease. Oxidative Med. Cell. Longev. 2018;2018:1–14. doi: 10.1155/2018/9547613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Patterson R.E., Kalavalapalli S., Williams C.M., Nautiyal M., Mathew J.T., Martinez J., Reinhard M.K., McDougall D.J., Rocca J.R., Yost R.A., et al. Lipotoxicity in steatohepatitis occurs despite an increase in tricarboxylic acid cycle activity. Am. J. Physiol. Metab. 2016;310:E484–E494. doi: 10.1152/ajpendo.00492.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sutti S., Albano E. Adaptive immunity: An emerging player in the progression of NAFLD. Nat. Rev. Gastroenterol. Hepatol. 2019;17:81–92. doi: 10.1038/s41575-019-0210-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cai J., Zhang X.-J., Li H. Role of Innate Immune Signaling in Non-Alcoholic Fatty Liver Disease. Trends Endocrinol. Metab. 2018;29:712–722. doi: 10.1016/j.tem.2018.08.003. [DOI] [PubMed] [Google Scholar]
- 18.Jadeja R.N., Devkar R., Nammi S. Oxidative Stress in Liver Diseases: Pathogenesis, Prevention, and Therapeutics. Oxidative Med. Cell. Longev. 2017;2017:1–2. doi: 10.1155/2017/8341286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Buzzetti E., Pinzani M., Tsochatzis E. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD) Metab. Clin. Exp. 2016;65:1038–1048. doi: 10.1016/j.metabol.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 20.Forrester S.J., Kikuchi D.S., Hernandes M.S., Xu Q., Griendling K.K. Reactive Oxygen Species in Metabolic and Inflammatory Signaling. Circ. Res. 2018;122:877–902. doi: 10.1161/CIRCRESAHA.117.311401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Serviddio G., Bellanti F., Vendemiale G. Free radical biology for medicine: Learning from nonalcoholic fatty liver disease. Free. Radic. Biol. Med. 2013;65:952–968. doi: 10.1016/j.freeradbiomed.2013.08.174. [DOI] [PubMed] [Google Scholar]
- 22.Bigarella C.L., Liang R., Ghaffari S. Stem cells and the impact of ROS signaling. Development. 2014;141:4206–4218. doi: 10.1242/dev.107086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mates J.M., Sanchez-Jimenez F. Antioxidant enzymes and their implications in pathophysiologic processes. Frontiers in bioscience. J. Virtual Libr. 1999;4:D339–D345. doi: 10.2741/A432. [DOI] [PubMed] [Google Scholar]
- 24.Zhang L., Wang X., Cueto R., Effi C., Zhang Y., Tan H., Qin X., Ji Y., Yang X., Wang H. Biochemical basis and metabolic interplay of redox regulation. Redox Biol. 2019;26:101284. doi: 10.1016/j.redox.2019.101284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Takaki A., Kawai D., Yamamoto K. Multiple Hits, Including Oxidative Stress, as Pathogenesis and Treatment Target in Non-Alcoholic Steatohepatitis (NASH) Int. J. Mol. Sci. 2013;14:20704–20728. doi: 10.3390/ijms141020704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bellanti F., Villani R., Facciorusso A., Vendemiale G., Serviddio G. Lipid oxidation products in the pathogenesis of non-alcoholic steatohepatitis. Free. Radic. Biol. Med. 2017;111:173–185. doi: 10.1016/j.freeradbiomed.2017.01.023. [DOI] [PubMed] [Google Scholar]
- 27.Bellanti F., Villani R., Tamborra R., Blonda M., Iannelli G., Di Bello G., Facciorusso A., Poli G., Iuliano L., Avolio C., et al. Synergistic interaction of fatty acids and oxysterols impairs mitochondrial function and limits liver adaptation during nafld progression. Redox Biol. 2017;15:86–96. doi: 10.1016/j.redox.2017.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sangineto M., Villani R., Cavallone F., Romano A.D., Loizzi D., Serviddio G. Lipid Metabolism in Development and Progression of Hepatocellular Carcinoma. Cancers. 2020;12:1419. doi: 10.3390/cancers12061419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Serviddio G., Bellanti F., Vendemiale G., Altomare E. Mitochondrial dysfunction in nonalcoholic steatohepatitis. Expert Rev. Gastroenterol. Hepatol. 2011;5:233–244. doi: 10.1586/egh.11.11. [DOI] [PubMed] [Google Scholar]
- 30.Serviddio G., Giudetti A.M., Bellanti F., Priore P., Rollo T., Tamborra R., Siculella L., Vendemiale G., Altomare E., Gnoni G.V. Oxidation of Hepatic Carnitine Palmitoyl Transferase-I (CPT-I) Impairs Fatty Acid Beta-Oxidation in Rats Fed a Methionine-Choline Deficient Diet. PLoS ONE. 2011;6:e24084. doi: 10.1371/journal.pone.0024084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Friedman S.L., Neuschwander-Tetri B.A., Rinella M., Sanyal A.J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 2018;24:908–922. doi: 10.1038/s41591-018-0104-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen Z., Yu Y., Cai J., Li H. Emerging Molecular Targets for Treatment of Nonalcoholic Fatty Liver Disease. Trends Endocrinol. Metab. 2019;30:903–914. doi: 10.1016/j.tem.2019.08.006. [DOI] [PubMed] [Google Scholar]
- 33.Schieber M., Chandel N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014;24:R453–R462. doi: 10.1016/j.cub.2014.03.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mansouri A., Gattolliat C.-H., Asselah T. Mitochondrial Dysfunction and Signaling in Chronic Liver Diseases. Gastroenterology. 2018;155:629–647. doi: 10.1053/j.gastro.2018.06.083. [DOI] [PubMed] [Google Scholar]
- 35.Simões I.C., Fontes A., Pinton P., Zischka H., Wieckowski M.R. Mitochondria in non-alcoholic fatty liver disease. Int. J. Biochem. Cell Biol. 2018;95:93–99. doi: 10.1016/j.biocel.2017.12.019. [DOI] [PubMed] [Google Scholar]
- 36.García-Ruiz I., Rodríguez-Juan C., Díaz-Sanjuan T., Del Hoyo P., Colina F., Muñoz-Yagüe T., Solís-Herruzo J.A. Uric acid and anti-TNF antibody improve mitochondrial dysfunction in ob/ob mice. Hepatology. 2006;44:581–591. doi: 10.1002/hep.21313. [DOI] [PubMed] [Google Scholar]
- 37.Garcia-Ruiz I., Rodriguez-Juan C., Diaz-Sanjuan T., Martinez M.A., Munoz-Yague T., Solis-Herruzo J.A. Ef-fects of rosiglitazone on the liver histology and mitochondrial function in ob/ob mice. Hepatology. 2007;46:414–423. doi: 10.1002/hep.21687. [DOI] [PubMed] [Google Scholar]
- 38.Li Z., Yang S., Lin H., Huang J., Watkins P.A., Moser A.B., DeSimone C., Song X., Diehl A.M. Probiotics and antibodies to TNF inhibit inflammatory activity and improve nonalcoholic fatty liver disease. Hepatology. 2003;37:343–350. doi: 10.1053/jhep.2003.50048. [DOI] [PubMed] [Google Scholar]
- 39.Rizki G., Arnaboldi L., Gabrielli B., Yan J., Lee G.S., Ng R.K., Turner S.M., Badger T.M., Pitas R.E., Maher J.J. Mice fed a lipogenic methionine-choline-deficient diet develop hypermetabolism coincident with hepatic suppression of SCD-1. J. Lipid Res. 2006;47:2280–2290. doi: 10.1194/jlr.M600198-JLR200. [DOI] [PubMed] [Google Scholar]
- 40.Romestaing C., Piquet M.A., Letexier D., Rey B., Mourier A., Servais S., Belouze M., Rouleau V., Dautresme M., Ollivier I., et al. Mitochondrial adaptations to steatohepatitis in-duced by a methionine- and choline-deficient diet. American journal of physiology. Endocrinol. Metab. 2008;294:E110–E119. doi: 10.1152/ajpendo.00407.2007. [DOI] [PubMed] [Google Scholar]
- 41.Koliaki C., Szendroedi J., Kaul K., Jelenik T., Nowotny P., Jankowiak F., Herder C., Carstensen-Kirberg M., Krausch M., Knoefel W.T., et al. Adaptation of Hepatic Mitochondrial Function in Humans with Non-Alcoholic Fatty Liver Is Lost in Steatohepatitis. Cell Metab. 2015;21:739–746. doi: 10.1016/j.cmet.2015.04.004. [DOI] [PubMed] [Google Scholar]
- 42.Bugianesi E., Gastaldelli A., Vanni E., Gambino R., Cassader M., Baldi S., Ponti V., Pagano G., Ferrannini E., Rizzetto M. Insulin resistance in non-diabetic patients with non-alcoholic fatty liver disease: Sites and mechanisms. Diabetologia. 2005;48:634–642. doi: 10.1007/s00125-005-1682-x. [DOI] [PubMed] [Google Scholar]
- 43.Chalasani N., Gorski J.C., Asghar M.S., Asghar A., Foresman B., Hall S.D., Crabb D.W. Hepatic cytochrome P450 2E1 activity in nondiabetic patients with nonalcoholic steatohepatitis. Hepatology. 2003;37:544–550. doi: 10.1053/jhep.2003.50095. [DOI] [PubMed] [Google Scholar]
- 44.Dasarathy S., Kasumov T., Edmison J.M., Gruca L.L., Bennett C., Duenas C., Marczewski S., McCullough A.J., Hanson R.W., Kalhan S. Glycine and urea kinetics in nonalcoholic steatohepatitis in human: Effect of intralipid infusion. Am. J. Physiol. Liver Physiol. 2009;297:G567–G575. doi: 10.1152/ajpgi.00042.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dasarathy S., Yang Y., McCullough A.J., Marczewski S., Bennett C., Kalhan S.C. Elevated hepatic fatty acid oxidation, high plasma fibroblast growth factor 21, and fasting bile acids in nonalcoholic steatohepatitis. Eur. J. Gastroenterol. Hepatol. 2011;23:382–388. doi: 10.1097/MEG.0b013e328345c8c7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hodson L., McQuaid S.E., Humphreys S.M., Milne R., Fielding B.A., Frayn K.N., Karpe F. Greater dietary fat oxidation in obese compared with lean men: An adaptive mechanism to prevent liver fat accumulation? Am. J. Physiol. Metab. 2010;299:E584–E592. doi: 10.1152/ajpendo.00272.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Miele L., Grieco A., Armuzzi A., Candelli M., Forgione A., Gasbarrini A., Gasbarrini G. Hepatic mitochondrial beta-oxidation in patients with nonalcoholic steatohepatitis assessed by 13C-octanoate breath test. Am. J. Gastroenterol. 2003;98:2335–2336. doi: 10.1111/j.1572-0241.2003.07725.x. [DOI] [PubMed] [Google Scholar]
- 48.Sanyal A.J., Campbell–Sargent C., Mirshahi F., Rizzo W.B., Contos M.J., Sterling R.K., Luketic V.A., Shiffman M.L., Clore J.N. Nonalcoholic steatohepatitis: Association of insulin resistance and mitochondrial abnormalities. Gastroenterology. 2001;120:1183–1192. doi: 10.1053/gast.2001.23256. [DOI] [PubMed] [Google Scholar]
- 49.Schneider A.R.J., Kraut C., Lindenthal B., Braden B., Caspary W.F., Stein J. Total body metabolism of 13C-octanoic acid is preserved in patients with non-alcoholic steatohepatitis, but differs between women and men. Eur. J. Gastroenterol. Hepatol. 2005;17:1181–1184. doi: 10.1097/00042737-200511000-00005. [DOI] [PubMed] [Google Scholar]
- 50.Mawatari H., Inamori M., Fujita K., Yoneda M., Iida H., Endo H., Hosono K., Nozaki Y., Yoneda K., Akiyama T., et al. The continuous real-time 13C-octanoate breath test for patients with nonalcoholic steatohepatitis using the BreathID system. Hepatogastroenterology. 2009;56:1436–1438. [PubMed] [Google Scholar]
- 51.Pérez-Carreras M., Del Hoyo P., Martín M.A., Rubio J.C., Martín A., Castellano G., Colina F., Arenas J., Solis-Herruzo J.A. Defective hepatic mitochondrial respiratory chain in patients with nonalcoholic steatohepatitis. Hepatology. 2003;38:999–1007. doi: 10.1002/hep.1840380426. [DOI] [PubMed] [Google Scholar]
- 52.Petersen K.F., Befroy D.E., Dufour S., Rothman D.L., Shulman G.I. Assessment of Hepatic Mitochondrial Oxidation and Pyruvate Cycling in NAFLD by 13 C Magnetic Resonance Spectroscopy. Cell Metab. 2016;24:167–171. doi: 10.1016/j.cmet.2016.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kakimoto P.A., Chausse B., Caldeira da Silva C.C., Donato Junior J., Kowaltowski A.J. Resilient hepatic mi-tochondrial function and lack of iNOS dependence in diet-induced insulin resistance. PLoS ONE. 2019;14:e0211733. doi: 10.1371/journal.pone.0211733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.George J., Liddle C. Nonalcoholic fatty liver disease: Pathogenesis and potential for nuclear receptors as thera-peutic targets. Mol. Pharm. 2008;5:49–59. doi: 10.1021/mp700110z. [DOI] [PubMed] [Google Scholar]
- 55.Yamaguchi K., Yang L., McCall S., Huang J., Yu X.X., Pandey S.K., Bhanot S., Monia B.P., Li Y.-X., Diehl A.M. Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology. 2007;45:1366–1374. doi: 10.1002/hep.21655. [DOI] [PubMed] [Google Scholar]
- 56.Satapati S., Sunny N.E., Kucejova B., Fu X., He T.T., Méndez-Lucas A., Shelton J.M., Perales J.C., Browning J.D., Burgess S.C. Elevated TCA cycle function in the pathology of diet-induced hepatic insulin resistance and fatty liver. J. Lipid Res. 2012;53:1080–1092. doi: 10.1194/jlr.M023382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cocco T., Di Paola M., Papa P., Lorusso M. Arachidonic acid interaction with the mitochondrial electron transport chain promotes reactive oxygen species generation. Free. Radic. Biol. Med. 1999;27:51–59. doi: 10.1016/S0891-5849(99)00034-9. [DOI] [PubMed] [Google Scholar]
- 58.Loskovich M.V., Grivennikova V.G., Cecchini G., Vinogradov A.D. Inhibitory effect of palmitate on the mi-tochondrial NADH:ubiquinone oxidoreductase (complex I) as related to the active-de-active enzyme transition. Biochem. J. 2005;387:677–683. doi: 10.1042/BJ20041703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Schönfeld P., Reiser G. Rotenone-like Action of the Branched-chain Phytanic Acid Induces Oxidative Stress in Mitochondria. J. Biol. Chem. 2006;281:7136–7142. doi: 10.1074/jbc.M513198200. [DOI] [PubMed] [Google Scholar]
- 60.Schönfeld P., Wojtczak L. Fatty acids decrease mitochondrial generation of reactive oxygen species at the reverse electron transport but increase it at the forward transport. Biochim. Biophys. Acta Bioenerg. 2007;1767:1032–1040. doi: 10.1016/j.bbabio.2007.04.005. [DOI] [PubMed] [Google Scholar]
- 61.Gille L., Nohl H. The Ubiquinol/bc1 Redox Couple Regulates Mitochondrial Oxygen Radical Formation. Arch. Biochem. Biophys. 2001;388:34–38. doi: 10.1006/abbi.2000.2257. [DOI] [PubMed] [Google Scholar]
- 62.Schönfeld P., Struy H. Refsum disease diagnostic marker phytanic acid alters the physical state of membrane proteins of liver mitochondria. FEBS Lett. 1999;457:179–183. doi: 10.1016/S0014-5793(99)01009-1. [DOI] [PubMed] [Google Scholar]
- 63.Serviddio G., Blonda M., Bellanti F., Villani R., Iuliano L., Vendemiale G. Oxysterols and redox signaling in the pathogenesis of non-alcoholic fatty liver disease. Free. Radic. Res. 2013;47:881–893. doi: 10.3109/10715762.2013.835048. [DOI] [PubMed] [Google Scholar]
- 64.Aoun M., Fouret G., Michel F., Bonafos B., Ramos J., Cristol J.P., Carbonneau M.-A., Coudray C., Feillet-Coudray C. Dietary fatty acids modulate liver mitochondrial cardiolipin content and its fatty acid composition in rats with non alcoholic fatty liver disease. J. Bioenerg. Biomembr. 2012;44:439–452. doi: 10.1007/s10863-012-9448-x. [DOI] [PubMed] [Google Scholar]
- 65.Flamment M., Arvier M., Gallois Y., Simard G., Malthièry Y., Ritz P., Ducluzeau-Fieloux P.-H. Fatty liver and insulin resistance in obese Zucker rats: No role for mitochondrial dysfunction. Biochimie. 2008;90:1407–1413. doi: 10.1016/j.biochi.2008.05.003. [DOI] [PubMed] [Google Scholar]
- 66.Flamment M., Rieusset J., Vidal H., Simard G., Malthièry Y., Fromenty B., Ducluzeau P.-H. Regulation of hepatic mitochondrial metabolism in response to a high fat diet: A longitudinal study in rats. J. Physiol. Biochem. 2012;68:335–344. doi: 10.1007/s13105-012-0145-3. [DOI] [PubMed] [Google Scholar]
- 67.Nadal-Casellas A., Amengual-Cladera E., Proenza A.M., Lladó I., Gianotti M. Long-term High-fat-diet Feeding Impairs Mitochondrial Biogenesis in Liver of Male and Female Rats. Cell. Physiol. Biochem. 2010;26:291–302. doi: 10.1159/000320552. [DOI] [PubMed] [Google Scholar]
- 68.Vial G., Dubouchaud H., Couturier K., Cottet-Rousselle C., Taleux N., Athias A., Galinier A., Casteilla L., Leverve X.M. Effects of a high-fat diet on energy metabolism and ROS production in rat liver. J. Hepatol. 2011;54:348–356. doi: 10.1016/j.jhep.2010.06.044. [DOI] [PubMed] [Google Scholar]
- 69.Wardlaw G.M., Kaplan M.L. Oxygen Consumption and Oxidative Capacity of Hepatocytes from Young Male Obese and Nonobese Zucker Rats. Exp. Biol. Med. 1986;183:199–206. doi: 10.3181/00379727-183-42405. [DOI] [PubMed] [Google Scholar]
- 70.Poussin C., Ibberson M., Hall D., Ding J., Soto J., Abel E.D., Thorens B. Oxidative Phosphorylation Flexibility in the Liver of Mice Resistant to High-Fat Diet-Induced Hepatic Steatosis. Diabetes. 2011;60:2216–2224. doi: 10.2337/db11-0338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Satapati S., Kucejova B., Duarte J.A., Fletcher J.A., Reynolds L., Sunny N.E., He T., Nair L.A., Livingston K.A., Fu X., et al. Mitochondrial metabolism mediates oxidative stress and inflammation in fatty liver. J. Clin. Investig. 2015;125:4447–4462. doi: 10.1172/JCI82204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Valdecantos M.P., Pérez-Matute P., González-Muniesa P., Prieto-Hontoria P.L., Moreno-Aliaga M.J., Martínez J.A. Lipoic acid administration prevents nonalcoholic steatosis linked to long-term high-fat feeding by modulating mitochondrial function. J. Nutr. Biochem. 2012;23:1676–1684. doi: 10.1016/j.jnutbio.2011.11.011. [DOI] [PubMed] [Google Scholar]
- 73.Carmiel-Haggai M., Cederbaum A.I., Nieto N. A high-fat diet leads to the progression of non-alcoholic fatty liver disease in obese rats. FASEB J. 2004;19:136–138. doi: 10.1096/fj.04-2291fje. [DOI] [PubMed] [Google Scholar]
- 74.Cortez-Pinto H., Chatham J., Chacko V.P., Arnold C., Rashid A., Diehl A.M. Alterations in liver ATP homeostasis in human nonalcoholic steatohepatitis: A pilot study. JAMA. 1999;282:1659–1664. doi: 10.1001/jama.282.17.1659. [DOI] [PubMed] [Google Scholar]
- 75.Jiang Y., Zhao M., An W. Increased hepatic apoptosis in high-fat diet-induced NASH in rats may be associated with downregulation of hepatic stimulator substance. J. Mol. Med. 2011;89:1207–1217. doi: 10.1007/s00109-011-0790-y. [DOI] [PubMed] [Google Scholar]
- 76.Mingorance C., Duluc L., Chalopin M., Simard G., Ducluzeau P.-H., Herrera M.D., De Sotomayor M.A., Andriantsitohaina R. Propionyl-L-carnitine Corrects Metabolic and Cardiovascular Alterations in Diet-Induced Obese Mice and Improves Liver Respiratory Chain Activity. PLoS ONE. 2012;7:e34268. doi: 10.1371/journal.pone.0034268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Fuchs C.D., Claudel T., Scharnagl H., Stojakovic T., Trauner M. FXR controls CHOP expression in steatohepatitis. FEBS Lett. 2017;591:3360–3368. doi: 10.1002/1873-3468.12845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Song B., Scheuner D., Ron D., Pennathur S., Kaufman R.J. Chop deletion reduces oxidative stress, improves β cell function, and promotes cell survival in multiple mouse models of diabetes. J. Clin. Investig. 2008;118:3378–3389. doi: 10.1172/JCI34587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ashraf N.U., Sheikh T.A. Endoplasmic reticulum stress and Oxidative stress in the pathogenesis of Non-alcoholic fatty liver disease. Free. Radic. Res. 2015;49:1405–1418. doi: 10.3109/10715762.2015.1078461. [DOI] [PubMed] [Google Scholar]
- 80.Cao S.S., Kaufman R.J. Endoplasmic Reticulum Stress and Oxidative Stress in Cell Fate Decision and Human Disease. Antioxid. Redox Signal. 2014;21:396–413. doi: 10.1089/ars.2014.5851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Malhotra J.D., Kaufman R.J. Endoplasmic Reticulum Stress and Oxidative Stress: A Vicious Cycle or a Double-Edged Sword? Antioxid. Redox Signal. 2007;9:2277–2294. doi: 10.1089/ars.2007.1782. [DOI] [PubMed] [Google Scholar]
- 82.Cullinan S.B., Diehl J.A. PERK-dependent Activation of Nrf2 Contributes to Redox Homeostasis and Cell Survival following Endoplasmic Reticulum Stress. J. Biol. Chem. 2004;279:20108–20117. doi: 10.1074/jbc.M314219200. [DOI] [PubMed] [Google Scholar]
- 83.Yang Y., Bazhin A.V., Werner J., Karakhanova S. Reactive Oxygen Species in the Immune System. Int. Rev. Immunol. 2013;32:249–270. doi: 10.3109/08830185.2012.755176. [DOI] [PubMed] [Google Scholar]
- 84.Schreck R., Rieber P., Baeuerle P.A. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J. 1991;10:2247–2258. doi: 10.1002/j.1460-2075.1991.tb07761.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Li S., Hong M., Tan H.-Y., Wang N., Feng Y. Insights into the Role and Interdependence of Oxidative Stress and Inflammation in Liver Diseases. Oxidative Med. Cell. Longev. 2016;2016:1–21. doi: 10.1155/2016/4234061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mittal M., Siddiqui M.R., Tran K., Reddy S.P., Malik A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxid. Redox Signal. 2014;20:1126–1167. doi: 10.1089/ars.2012.5149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Bubici C., Papa S., Pham C.G., Zazzeroni F., Franzoso G. The NF-kappaB-mediated control of ROS and JNK signaling. Histol. Histopathol. 2006;21:69–80. doi: 10.14670/HH-21.69. [DOI] [PubMed] [Google Scholar]
- 88.Zhu L.H., Wang A., Luo P., Wang X., Jiang D.-S., Deng W., Zhang X., Wang T., Liu Y., Gao L., et al. Mindin/Spondin 2 inhibits hepatic steatosis, insulin resistance, and obesity via interaction with peroxisome proliferator-activated receptor α in mice. J. Hepatol. 2014;60:1046–1054. doi: 10.1016/j.jhep.2014.01.011. [DOI] [PubMed] [Google Scholar]
- 89.Wang X.-A., Deng S., Jiang D., Zhang R., Zhang S., Zhong J., Yang L., Wang T., Hong S., Guo S., et al. CARD3 Deficiency Exacerbates Diet-Induced Obesity, Hepatosteatosis, and Insulin Resistance in Male Mice. Endocrinology. 2013;154:685–697. doi: 10.1210/en.2012-1911. [DOI] [PubMed] [Google Scholar]
- 90.Tong J., Han C.J., Zhang J.Z., He W.Z., Zhao G.J., Cheng X., Zhang L., Deng K.Q., Liu Y., Fan H.F., et al. Hepatic Interferon Regulatory Factor 6 Alleviates Liver Steatosis and Metabolic Disorder by Transcriptionally Suppressing Peroxisome Proliferator-Activated Receptor gamma in Mice. Hepatology. 2019;69:2471–2488. doi: 10.1002/hep.30559. [DOI] [PubMed] [Google Scholar]
- 91.Wang P.-X., Zhang R., Huang L., Zhu L.-H., Jiang D.-S., Chen H.-Z., Zhang Y., Tian S., Zhang X.-F., Liu D., et al. Interferon regulatory factor 9 is a key mediator of hepatic ischemia/reperfusion injury. J. Hepatol. 2015;62:111–120. doi: 10.1016/j.jhep.2014.08.022. [DOI] [PubMed] [Google Scholar]
- 92.Hu J., Zhu X.-H., Zhang X.-J., Wang P.-X., Zhang R., Zhang P., Zhao G.-N., Gao L., Tian S., Li H. Targeting TRAF3 signaling protects against hepatic ischemia/reperfusions injury. J. Hepatol. 2016;64:146–159. doi: 10.1016/j.jhep.2015.08.021. [DOI] [PubMed] [Google Scholar]
- 93.Chen L., Huang J., Ji Y.X., Mei F., Wang P.X., Deng K.Q., Jiang X., Ma G., Li H. Tripartite Motif 8 Contributes to Pathological Cardiac Hypertrophy Through Enhancing Transforming Growth Factor beta-Activated Kinase 1-Dependent Signaling Pathways. Hypertension. 2017;69:249–258. doi: 10.1161/HYPERTENSIONAHA.116.07741. [DOI] [PubMed] [Google Scholar]
- 94.Zhao G.-N., Zhang P., Gong J., Zhang X.-J., Wang P.-X., Yin M., Jiang Z., Shen L.-J., Ji Y.-X., Tong J., et al. Tmbim1 is a multivesicular body regulator that protects against non-alcoholic fatty liver disease in mice and monkeys by targeting the lysosomal degradation of Tlr4. Nat. Med. 2017;23:742–752. doi: 10.1038/nm.4334. [DOI] [PubMed] [Google Scholar]
- 95.Yan Z., Huang Y., Wang X., Wang H., Ren F., Tian R., Cheng X., Cai J., Zhang Y., Zhu X., et al. Integrated Omics Reveals Tollip as an Regulator and Therapeutic Target for Hepatic Ischemia-Reperfusion Injury in Mice. Hepatology. 2019;70:1750–1769. doi: 10.1002/hep.30705. [DOI] [PubMed] [Google Scholar]
- 96.Cai J., Xu M., Zhang X., Li H. Innate Immune Signaling in Nonalcoholic Fatty Liver Disease and Cardiovascular Diseases. Annu. Rev. Pathol. Mech. Dis. 2019;14:153–184. doi: 10.1146/annurev-pathmechdis-012418-013003. [DOI] [PubMed] [Google Scholar]
- 97.Federico A., Caprio G.G., Ormando V.M., Loguercio C. Gut Microbiota and Gastrointestinal Tract, Liver and Pancreas: From Physiology to Pathology. Minerva Gastroenterol. Dietol. 2017;63:385–398. doi: 10.23736/S1121-421X.17.02375-3. [DOI] [PubMed] [Google Scholar]
- 98.Luedde T., Schwabe R.F. NF-kappaB in the liver-linking injury, fibrosis and hepatocellular carcinoma. Nature reviews. Gastroenterol. Hepatol. 2011;8:108–118. doi: 10.1038/nrgastro.2010.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Li L., Gao J., Li J. Emerging role of HMGB 1 in fibrotic diseases. J. Cell. Mol. Med. 2014;18:2331–2339. doi: 10.1111/jcmm.12419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Al-Khafaji A.B., Tohme S., Yazdani H.O., Miller D., Huang H., Tsung A. Superoxide induces Neutrophil Extracellular Trap Formation in a TLR-4 and NOX-Dependent Mechanism. Mol. Med. 2015;22:621–631. doi: 10.2119/molmed.2016.00054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Garcia-Martinez I., Santoro N., Chen Y., Hoque R., Ouyang X., Caprio S., Shlomchik M.J., Coffman R.L., Candia A., Mehal W.Z. Hepatocyte mitochondrial DNA drives nonalcoholic steatohepatitis by activation of TLR9. J. Clin. Investig. 2016;126:859–864. doi: 10.1172/JCI83885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Gan L.T., Van Rooyen D.M., Koina M.E., McCuskey R.S., Teoh N.C., Farrell G.C. Hepatocyte free cholesterol lipotoxicity results from JNK1-mediated mitochondrial injury and is HMGB1 and TLR4-dependent. J. Hepatol. 2014;61:1376–1384. doi: 10.1016/j.jhep.2014.07.024. [DOI] [PubMed] [Google Scholar]
- 103.Begriche K., Igoudjil A., Pessayre D., Fromenty B. Mitochondrial dysfunction in NASH: Causes, consequences and possible means to prevent it. Mitochondrion. 2006;6:1–28. doi: 10.1016/j.mito.2005.10.004. [DOI] [PubMed] [Google Scholar]
- 104.Yu Y., Cai J., She Z., Li H. Insights into the Epidemiology, Pathogenesis, and Therapeutics of Nonalcoholic Fatty Liver Diseases. Adv. Sci. 2019;6:1801585. doi: 10.1002/advs.201801585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Yuan T., Yang T., Chen H., Fu D., Hu Y., Wang J., Yuan Q., Yu H., Xu W., Xie X. New insights into oxidative stress and inflammation during diabetes mellitus-accelerated atherosclerosis. Redox Biol. 2019;20:247–260. doi: 10.1016/j.redox.2018.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Doridot L., Jeljeli M., Chêne C., Batteux F. Implication of oxidative stress in the pathogenesis of systemic sclerosis via inflammation, autoimmunity and fibrosis. Redox Biol. 2019;25:101122. doi: 10.1016/j.redox.2019.101122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Abais J.M., Xia M., Li G., Gehr T.W.B., Boini K.M., Li P.-L. Contribution of endogenously produced reactive oxygen species to the activation of podocyte NLRP3 inflammasomes in hyperhomocysteinemia. Free. Radic. Biol. Med. 2014;67:211–220. doi: 10.1016/j.freeradbiomed.2013.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Bae J.Y., Park H.H. Crystal Structure of NALP3 Protein Pyrin Domain (PYD) and Its Implications in Inflammasome Assembly. J. Biol. Chem. 2011;286:39528–39536. doi: 10.1074/jbc.M111.278812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yan F.-J., Zhang X.-J., Wang W.-X., Ji Y.-X., Wang P.-X., Yang Y., Gong J., Shen L.-J., Zhu X.-Y., Huang Z., et al. The E3 ligase tripartite motif 8 targets TAK1 to promote insulin resistance and steatohepatitis. Hepatology. 2017;65:1492–1511. doi: 10.1002/hep.28971. [DOI] [PubMed] [Google Scholar]
- 110.Xiang M., Wang P.-X., Wang A.-B., Zhang X.-J., Zhang Y., Zhang P., Mei F.-H., Chen M.-H., Li H. Targeting hepatic TRAF1-ASK1 signaling to improve inflammation, insulin resistance, and hepatic steatosis. J. Hepatol. 2016;64:1365–1377. doi: 10.1016/j.jhep.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 111.Wang Y., Wen H., Fu J., Cai L., Li P., Zhao C., Dong Z., Ma J.P., Wang X., Tian H., et al. Hepatocyte TNF Receptor–Associated Factor 6 Aggravates Hepatic Inflammation and Fibrosis by Promoting Lysine 6–Linked Polyubiquitination of Apoptosis Signal-Regulating Kinase 1. Hepatology. 2020;71:93–111. doi: 10.1002/hep.30822. [DOI] [PubMed] [Google Scholar]
- 112.Blaser H., Dostert C., Mak T.W., Brenner D. TNF and ROS Crosstalk in Inflammation. Trends Cell Biol. 2016;26:249–261. doi: 10.1016/j.tcb.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 113.Son Y., Cheong Y.-K., Kim N.-H., Chung H.-T., Kang D.G., Pae H.-O. Mitogen-Activated Protein Kinases and Reactive Oxygen Species: How Can ROS Activate MAPK Pathways? J. Signal. Transduct. 2011;2011:1–6. doi: 10.1155/2011/792639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kamata H., Honda S., Maeda S., Chang L., Hirata H., Karin M. Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell. 2005;120:649–661. doi: 10.1016/j.cell.2004.12.041. [DOI] [PubMed] [Google Scholar]
- 115.Win S., Than T.A., Fernández-Checa J.C., Kaplowitz N. JNK interaction with Sab mediates ER stress induced inhibition of mitochondrial respiration and cell death. Cell Death Dis. 2014;5:e989. doi: 10.1038/cddis.2013.522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhao G.-N., Jiang D.-S., Li H. Interferon regulatory factors: At the crossroads of immunity, metabolism, and disease. Biochim. Biophys. Acta Mol. Basis Dis. 2015;1852:365–378. doi: 10.1016/j.bbadis.2014.04.030. [DOI] [PubMed] [Google Scholar]
- 117.Zhang Y., Zhang X.-J., Wang P.-X., Zhang P., Li H. Reprogramming Innate Immune Signaling in Cardiometabolic Disease. Hypertension. 2017;69:747–760. doi: 10.1161/HYPERTENSIONAHA.116.08192. [DOI] [PubMed] [Google Scholar]
- 118.Zhang X.-J., Li H. Targeting Interferon Regulatory Factor for Cardiometabolic Diseases: Opportunities and Challenges. Curr. Drug Targets. 2017;18:1754–1778. doi: 10.2174/1389450116666150804110412. [DOI] [PubMed] [Google Scholar]
- 119.Wang X.A., Zhang R., She Z.G., Zhang X.F., Jiang D.S., Wang T., Gao L., Deng W., Zhang S.M., Zhu L.H., et al. Interferon regulatory factor 3 constrains IKKbeta/NF-kappaB signaling to alleviate hepatic steatosis and insulin resistance. Hepatology. 2014;59:870–885. doi: 10.1002/hep.26751. [DOI] [PubMed] [Google Scholar]
- 120.Yadav U.C., Ramana K.V. Regulation of NF-kappaB-induced inflammatory signaling by lipid peroxidation-derived aldehydes. Oxidative Med. Cell. Longev. 2013;2013:690545. doi: 10.1155/2013/690545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Morgan M.J., Liu Z.G. Crosstalk of reactive oxygen species and NF-kappaB signaling. Cell Res. 2011;21:103–115. doi: 10.1038/cr.2010.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Furnkranz A., Leitinger N. Regulation of inflammatory responses by oxidized phospholipids: Structure-function relationships. Curr. Pharm. Des. 2004;10:915–921. doi: 10.2174/1381612043452929. [DOI] [PubMed] [Google Scholar]
- 123.Wang P., Zhang X., Li H. Liver capsule: IRFs in hepatocytes: Pathophysiology. Hepatology. 2016;63:1706. doi: 10.1002/hep.28433. [DOI] [PubMed] [Google Scholar]
- 124.Luo P., Wang P.X., Li Z.Z., Zhang X.J., Jiang X., Gong J., Qin J.J., Guo J., Zhu X., Yang S., et al. Hepatic Oncostatin M Receptor beta Regulates Obesity-Induced Steatosis and Insulin Resistance. Am. J. Pathol. 2016;186:1278–1292. doi: 10.1016/j.ajpath.2015.12.028. [DOI] [PubMed] [Google Scholar]
- 125.Bai L., Li H. Innate immune regulatory networks in hepatic lipid metabolism. J. Mol. Med. 2019;97:593–604. doi: 10.1007/s00109-019-01765-1. [DOI] [PubMed] [Google Scholar]
- 126.Azzimato V., Jager J., Chen P., Morgantini C., Levi L., Barreby E., Sulen A., Oses C., Willerbrords J., Xu C., et al. Liver macrophages inhibit the endogenous antioxidant response in obesity-associated insulin resistance. Sci. Transl. Med. 2020;12:eaaw9709. doi: 10.1126/scitranslmed.aaw9709. [DOI] [PubMed] [Google Scholar]
- 127.Milicevic N.P., Busch C.J.-L., Binder C.J. Malondialdehyde Epitopes as Targets of Immunity and the Implications for Atherosclerosis. Dev. Funct. Myeloid Subsets. 2016;131:1–59. doi: 10.1016/bs.ai.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Nobili V., Parola M., Alisi A., Marra F., Piemonte F., Mombello C., Sutti S., Povero D., Maina V., Novo E., et al. Oxidative stress parameters in paediatric non-alcoholic fatty liver disease. Int. J. Mol. Med. 2010;26:471–476. doi: 10.3892/ijmm_00000487. [DOI] [PubMed] [Google Scholar]
- 129.Chou M.-Y., Fogelstrand L., Hartvigsen K., Hansen L.F., Woelkers D., Shaw P.X., Choi J., Perkmann T., Bäckhed F., Miller Y.I., et al. Oxidation-specific epitopes are dominant targets of innate natural antibodies in mice and humans. J. Clin. Investig. 2009;119:1335–1349. doi: 10.1172/JCI36800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Smallwood M.J., Nissim A., Knight A.R., Whiteman M., Haigh R., Winyard P.G. Oxidative stress in autoimmune rheumatic diseases. Free. Radic. Biol. Med. 2018;125:3–14. doi: 10.1016/j.freeradbiomed.2018.05.086. [DOI] [PubMed] [Google Scholar]
- 131.Sutti S., Jindal A., Locatelli I., Vacchiano M., Gigliotti L., Bozzola C., Albano E. Adaptive immune responses triggered by oxidative stress contribute to hepatic inflammation in NASH. Hepatology. 2014;59:886–897. doi: 10.1002/hep.26749. [DOI] [PubMed] [Google Scholar]
- 132.Albano E., Mottaran E., Vidali M., Reale E., Saksena S., Occhino G., Burt A.D., Day C.P. Immune response towards lipid peroxidation products as a predictor of progression of non-alcoholic fatty liver disease to advanced fibrosis. Gut. 2005;54:987–993. doi: 10.1136/gut.2004.057968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Bruzzì S., Sutti S., Giudici G., Burlone M.E., Ramavath N.N., Toscani A., Bozzola C., Schneider P., Morello E., Parola M., et al. B2-Lymphocyte responses to oxidative stress-derived antigens contribute to the evolution of nonalcoholic fatty liver disease (NAFLD) Free. Radic. Biol. Med. 2018;124:249–259. doi: 10.1016/j.freeradbiomed.2018.06.015. [DOI] [PubMed] [Google Scholar]
- 134.Baumgardner J.N., Shankar K., Hennings L., Albano E., Badger T.M., Ronis M.J. N-acetylcysteine attenuates progression of liver pathology in a rat model of nonalcoholic steatohepatitis. J. Nutr. 2008;138:1872–1879. doi: 10.1093/jn/138.10.1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Rolla S., Alchera E., Imarisio C., Bardina V., Valente G., Cappello P., Mombello C., Follenzi A., Novelli F., Carini R. The balance between IL-17 and IL-22 produced by liver-infiltrating T-helper cells critically controls NASH development in mice. Clin. Sci. 2015;130:193–203. doi: 10.1042/CS20150405. [DOI] [PubMed] [Google Scholar]
- 136.Rau M., Schilling A.-K., Meertens J., Hering I., Weiss J., Jurowich C., Kudlich T., Hermanns H.M., Bantel H., Beyersdorf N., et al. Progression from Nonalcoholic Fatty Liver to Nonalcoholic Steatohepatitis Is Marked by a Higher Frequency of Th17 Cells in the Liver and an Increased Th17/Resting Regulatory T Cell Ratio in Peripheral Blood and in the Liver. J. Immunol. 2015;196:97–105. doi: 10.4049/jimmunol.1501175. [DOI] [PubMed] [Google Scholar]
- 137.Inzaugarat M.E., Solari N.E.F., Billordo L.A., Abecasis R., Gadano A.C., Cherñavsky A.C. Altered Phenotype and Functionality of Circulating Immune Cells Characterize Adult Patients with Nonalcoholic Steatohepatitis. J. Clin. Immunol. 2011;31:1120–1130. doi: 10.1007/s10875-011-9571-1. [DOI] [PubMed] [Google Scholar]
- 138.Solari N.E.F., Inzaugarat M.E., Baz P., De Matteo E., Lezama C., Galoppo M., Galoppo C., Cherñavsky A.C. The Role of Innate Cells Is Coupled to a Th1-Polarized Immune Response in Pediatric Nonalcoholic Steatohepatitis. J. Clin. Immunol. 2012;32:611–621. doi: 10.1007/s10875-011-9635-2. [DOI] [PubMed] [Google Scholar]
- 139.Ma X., Hua J., Mohamood A.R., Hamad A.R.A., Ravi R., Li Z. A high-fat diet and regulatory T cells influence susceptibility to endotoxin-induced liver injury. Hepatology. 2007;46:1519–1529. doi: 10.1002/hep.21823. [DOI] [PubMed] [Google Scholar]
- 140.Ma C., Kesarwala A.H., Eggert T., Medina-Echeverz J., Kleiner D.E., Jin P., Stroncek P.J.D.F., Terabe M., Kapoor V., Elgindi M., et al. NAFLD causes selective CD4+ T lymphocyte loss and promotes hepatocarcinogenesis. Nat. Cell Biol. 2016;531:253–257. doi: 10.1038/nature16969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Van Herck M.A., Weyler J., Kwanten W.J., Dirinck E.L., De Winter B.Y., Francque S.M., Vonghia L. The Differential Roles of T Cells in Non-alcoholic Fatty Liver Disease and Obesity. Front. Immunol. 2019;10:82. doi: 10.3389/fimmu.2019.00082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Laurent S., Carrega P., Saverino D., Piccioli P., Camoriano M., Morabito A., Dozin B., Fontana V., Simone R., Mortara L., et al. CTLA-4 is expressed by human monocyte-derived dendritic cells and regulates their functions. Hum. Immunol. 2010;71:934–941. doi: 10.1016/j.humimm.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 143.Hendrikx T., Watzenböck M.L., Walenbergh S.M.A., Amir S., Gruber S., Kozma M.O., Grabsch H.I., Koek G.H., Pierik M.J., Staufer K., et al. Low levels of IgM antibodies recognizing oxidation-specific epitopes are associated with human non-alcoholic fatty liver disease. BMC Med. 2016;14:107. doi: 10.1186/s12916-016-0652-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Bieghs V., Van Gorp P.J.J., Walenbergh S.M., Gijbels M.J., Verheyen F., Buurman W.A., Briles D.E., Hofker M.H., Binder C.J., Shiri-Sverdlov R. Specific immunization strategies against oxidized low-density lipoprotein: A novel way to reduce nonalcoholic steatohepatitis in mice. Hepatology. 2012;56:894–903. doi: 10.1002/hep.25660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Kulik L., El-Serag H.B. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology. 2019;156:477–491.e1. doi: 10.1053/j.gastro.2018.08.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Margini C., Dufour J.-F. The story of HCC in NAFLD: From epidemiology, across pathogenesis, to prevention and treatment. Liver Int. 2015;36:317–324. doi: 10.1111/liv.13031. [DOI] [PubMed] [Google Scholar]
- 147.Sachdeva M., Chawla Y.K., Arora S.K. Immunology of hepatocellular carcinoma. World J. Hepatol. 2015;7:2080–2090. doi: 10.4254/wjh.v7.i17.2080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Fu Y., Chung F.-L. Oxidative stress and hepatocarcinogenesis. Hepatoma Res. 2018;4:4. doi: 10.20517/2394-5079.2018.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Yu L.-X., Ling Y., Wang H. Role of nonresolving inflammation in hepatocellular carcinoma development and progression. npj Precis. Oncol. 2018;2:1–10. doi: 10.1038/s41698-018-0048-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428–435. doi: 10.1038/nature07201. [DOI] [PubMed] [Google Scholar]
- 151.El–Serag H.B., Rudolph K.L. Hepatocellular Carcinoma: Epidemiology and Molecular Carcinogenesis. Gastroenterology. 2007;132:2557–2576. doi: 10.1053/j.gastro.2007.04.061. [DOI] [PubMed] [Google Scholar]
- 152.Sahasrabuddhe V.V., Gunja M.Z., Graubard B.I., Trabert B., Schwartz L.M., Park Y., Hollenbeck A.R., Freedman N.D., McGlynn K.A. Nonsteroidal Anti-inflammatory Drug Use, Chronic Liver Disease, and Hepatocellular Carcinoma. J. Natl. Cancer Inst. 2012;104:1808–1814. doi: 10.1093/jnci/djs452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Yeh C.C., Lin J.T., Jeng L.B., Ho H.J., Yang H.R., Wu M.S., Kuo K.N., Wu C.Y. Nonsteroidal anti-inflammatory drugs are associated with reduced risk of early hepatocellular carcinoma recurrence after curative liver resection: A nationwide cohort study. Ann. Surg. 2015;261:521–526. doi: 10.1097/SLA.0000000000000746. [DOI] [PubMed] [Google Scholar]
- 154.Zappavigna S., Vanacore D., Lama S., Potenza N., Russo A., Ferranti P., Dallio M., Federico A., Loguercio C., Sperlongano P., et al. Silybin-Induced Apoptosis Occurs in Parallel to the Increase of Ceramides Synthesis and miRNAs Secretion in Human Hepatocarcinoma Cells. Int. J. Mol. Sci. 2019;20:2190. doi: 10.3390/ijms20092190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Matzinger P. The Danger Model: A Renewed Sense of Self. Science. 2002;296:301–305. doi: 10.1126/science.1071059. [DOI] [PubMed] [Google Scholar]
- 156.Mihm S. Danger-Associated Molecular Patterns (DAMPs): Molecular Triggers for Sterile Inflammation in the Liver. Int. J. Mol. Sci. 2018;19:3104. doi: 10.3390/ijms19103104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Cruz C.S.D., Kang M.-J. Mitochondrial dysfunction and damage associated molecular patterns (DAMPs) in chronic inflammatory diseases. Mitochondrion. 2018;41:37–44. doi: 10.1016/j.mito.2017.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Zhang X., Wu X., Hu Q., Wu J., Wang G., Hong Z., Ren J.-A., Infections L.F.T.A.S. Mitochondrial DNA in liver inflammation and oxidative stress. Life Sci. 2019;236:116464. doi: 10.1016/j.lfs.2019.05.020. [DOI] [PubMed] [Google Scholar]
- 159.Fang C., Wei X., Wei Y. Mitochondrial DNA in the regulation of innate immune responses. Protein Cell. 2016;7:11–16. doi: 10.1007/s13238-015-0222-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.West A.P., Shadel G.S. Mitochondrial DNA in innate immune responses and inflammatory pathology. Nat. Rev. Immunol. 2017;17:363–375. doi: 10.1038/nri.2017.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Bao D., Zhao J., Zhou X., Yang Q., Chen Y., Zhu J., Yuan P., Yang J., Qin T., Wan S., et al. Mitochondrial fission-induced mtDNA stress promotes tumor-associated macrophage infiltration and HCC progression. Oncogene. 2019;38:5007–5020. doi: 10.1038/s41388-019-0772-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Riley J.S., Quarato G., Cloix C., Lopez J., O’Prey J., Pearson M., Chapman J., Sesaki H., Carlin L.M., Passos J.F., et al. Mitochondrial inner membrane permeabilisation enables mtDNA release during apoptosis. EMBO J. 2018;37:1–16. doi: 10.15252/embj.201899238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kaczmarek A., Vandenabeele P., Krysko D.V. Necroptosis: The Release of Damage-Associated Molecular Patterns and Its Physiological Relevance. Immunity. 2013;38:209–223. doi: 10.1016/j.immuni.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 164.Andersson U., Tracey K.J. HMGB1 Is a Therapeutic Target for Sterile Inflammation and Infection. Annu. Rev. Immunol. 2011;29:139–162. doi: 10.1146/annurev-immunol-030409-101323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Kazama H., Ricci J.-E., Herndon J.M., Hoppe G., Green D.R., Ferguson T.A. Induction of Immunological Tolerance by Apoptotic Cells Requires Caspase-Dependent Oxidation of High-Mobility Group Box-1 Protein. Immunity. 2008;29:21–32. doi: 10.1016/j.immuni.2008.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Yang T., Zhang W., Wang L., Xiao C., Gong Y., Huang D., Guo B., Li Q., Xiang Y., Nan Y. Co-culture of dendritic cells and cytokine-induced killer cells effectively suppresses liver cancer stem cell growth by inhibiting pathways in the immune system. BMC Cancer. 2018;18:984. doi: 10.1186/s12885-018-4871-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Dou L., Ono Y., Chen Y.-F., Chen X.-P., Thomson A.W. Hepatic Dendritic Cells, the Tolerogenic Liver Environment, and Liver Disease. Semin. Liver Dis. 2018;38:170–180. doi: 10.1055/s-0038-1646949. [DOI] [PubMed] [Google Scholar]
- 168.Streba L.A.M., Streba L.A.M., Sandulescu D.L., Vere C.C., Mitrut P., Cotoi B.V., Popescu L.N., Ion D.A. Dendritic cells and hepatocellular carcinoma. Romanian J. Morphol. Embryol. Rev. Roum. Morphol. Embryol. 2014;55:1287–1293. [PubMed] [Google Scholar]
- 169.Chen S., Akbar S.M., Tanimoto K., Ninomiya T., Iuchi H., Michitaka K., Horiike N., Onji M. Absence of CD83-positive mature and activated dendritic cells at cancer nodules from patients with hepatocellular carcinoma: Relevance to hepatocarcinogenesis. Cancer Lett. 2000;148:49–57. doi: 10.1016/S0304-3835(99)00312-2. [DOI] [PubMed] [Google Scholar]
- 170.Matsui M., Machida S., Itani-Yohda T., Akatsuka T. Downregulation of the proteasome subunits, transporter, and antigen presentation in hepatocellular carcinoma, and their restoration by interferon-gamma. J. Gastroenterol. Hepatol. 2002;17:897–907. doi: 10.1046/j.1440-1746.2002.02837.x. [DOI] [PubMed] [Google Scholar]
- 171.Roliński J., Hus I. Breaking immunotolerance of tumors: A new perspective for dendritic cell therapy. J. Immunotoxicol. 2014;11:311–318. doi: 10.3109/1547691X.2013.865094. [DOI] [PubMed] [Google Scholar]
- 172.Onishi Y., Fehervari Z., Yamaguchi T., Sakaguchi S. Foxp3+ natural regulatory T cells preferentially form aggregates on dendritic cells in vitro and actively inhibit their maturation. Proc. Natl. Acad. Sci. USA. 2008;105:10113–10118. doi: 10.1073/pnas.0711106105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Tang D., Kang R., Livesey K.M., Zeh H.J., Lotze M.T. High mobility group box 1 (HMGB1) activates an autophagic response to oxidative stress. Antioxid. Redox Signal. 2011;15:2185–2195. doi: 10.1089/ars.2010.3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Szlosarek P.W., Balkwill F.R. Tumour necrosis factor alpha: A potential target for the therapy of solid tumours. Lancet Oncol. 2003;4:565–573. doi: 10.1016/S1470-2045(03)01196-3. [DOI] [PubMed] [Google Scholar]
- 175.Schrader J., Iredale J.P. The inflammatory microenvironment of HCC-the plot becomes complex. J. Hepatol. 2010;54:853–855. doi: 10.1016/j.jhep.2010.12.014. [DOI] [PubMed] [Google Scholar]
- 176.Wu S.-D., Ma Y.-S., Fang Y., Liu L.-L., Fu D., Shen X. Role of the microenvironment in hepatocellular carcinoma development and progression. Cancer Treat. Rev. 2012;38:218–225. doi: 10.1016/j.ctrv.2011.06.010. [DOI] [PubMed] [Google Scholar]
- 177.Maeda S. NF-κB, JNK, and TLR Signaling Pathways in Hepatocarcinogenesis. Gastroenterol. Res. Pr. 2010;2010:1–10. doi: 10.1155/2010/367694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Capece D., Fischietti M., Verzella D., Gaggiano A., Cicciarelli G., Tessitore A., Zazzeroni F., Alesse E. The Inflammatory Microenvironment in Hepatocellular Carcinoma: A Pivotal Role for Tumor-Associated Macrophages. BioMed Res. Int. 2012;2013:1–15. doi: 10.1155/2013/187204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Komohara Y., Takeya M. CAFs and TAMs: Maestros of the tumour microenvironment. J. Pathol. 2016;241:313–315. doi: 10.1002/path.4824. [DOI] [PubMed] [Google Scholar]
- 180.Oršolić N., Kunštić M., Kukolj M., Gračan R., Nemrava J. Oxidative stress, polarization of macrophages and tumour angiogenesis: Efficacy of caffeic acid. Chem. Biol. Interact. 2016;256:111–124. doi: 10.1016/j.cbi.2016.06.027. [DOI] [PubMed] [Google Scholar]
- 181.Zhang Y., Choksi S., Chen K., Pobezinskaya Y.L., Linnoila I., Liu Z.-G. ROS play a critical role in the differentiation of alternatively activated macrophages and the occurrence of tumor-associated macrophages. Cell Res. 2013;23:898–914. doi: 10.1038/cr.2013.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zhu X.-D., Zhang J.-B., Zhuang P.-Y., Zhu H.-G., Zhang W., Xiong Y.-Q., Wu W.-Z., Wang L., Tang Z.-Y., Sun H.-C. High Expression of Macrophage Colony-Stimulating Factor in Peritumoral Liver Tissue Is Associated With Poor Survival After Curative Resection of Hepatocellular Carcinoma. J. Clin. Oncol. 2008;26:2707–2716. doi: 10.1200/JCO.2007.15.6521. [DOI] [PubMed] [Google Scholar]
- 183.Budhu A., Forgues M., Ye Q.-H., Jia H., He P., Zanetti K.A., Kammula U.S., Chen Y., Qin L., Tang Z.-Y., et al. Prediction of venous metastases, recurrence, and prognosis in hepatocellular carcinoma based on a unique immune response signature of the liver microenvironment. Cancer Cell. 2006;10:99–111. doi: 10.1016/j.ccr.2006.06.016. [DOI] [PubMed] [Google Scholar]
- 184.Kudo M. Immuno-Oncology in Hepatocellular Carcinoma: 2017 Update. Oncology. 2017;93:147–159. doi: 10.1159/000481245. [DOI] [PubMed] [Google Scholar]
- 185.Makarova-Rusher O.V., Medina-Echeverz J., Duffy A.G., Greten T.F. The yin and yang of evasion and immune activation in HCC. J. Hepatol. 2015;62:1420–1429. doi: 10.1016/j.jhep.2015.02.038. [DOI] [PubMed] [Google Scholar]
- 186.Attallah A.M., Tabll A.A., El-Sadany M., Ibrahim T.A., El-Dosoky I. Dysregulation of blood lymphocyte subsets and natural killer cells in schistosomal liver cirrhosis and hepatocellular carcinoma. Z. Die Gesamte Exp. Med. 2003;3:181–185. doi: 10.1007/s10238-003-0023-y. [DOI] [PubMed] [Google Scholar]
- 187.Zhang X. NAFLD Related-HCC: The Relationship with Metabolic Disorders. Adv. Exp. Med. Biol. 2018;1061:55–62. doi: 10.1007/978-981-10-8684-7_5. [DOI] [PubMed] [Google Scholar]
- 188.Ormandy L.A., Hillemann T., Wedemeyer H., Manns M.P., Greten T.F., Korangy F. Increased Populations of Regulatory T Cells in Peripheral Blood of Patients with Hepatocellular Carcinoma. Cancer Res. 2005;65:2457–2464. doi: 10.1158/0008-5472.CAN-04-3232. [DOI] [PubMed] [Google Scholar]
- 189.Greten T.F., Ormandy L.A., Fikuart A., Höchst B., Henschen S., Hörning M., Manns M.P., Korangy F. Low-dose Cyclophosphamide Treatment Impairs Regulatory T Cells and Unmasks AFP-specific CD4+ T-cell Responses in Patients with Advanced HCC. J. Immunother. 2010;33:211–218. doi: 10.1097/CJI.0b013e3181bb499f. [DOI] [PubMed] [Google Scholar]
- 190.Hoechst B., Ormandy L.A., Ballmaier M., Lehner F., Krüger C., Manns M.P., Greten T.F., Korangy F. A New Population of Myeloid-Derived Suppressor Cells in Hepatocellular Carcinoma Patients Induces CD4+CD25+Foxp3+ T Cells. Gastroenterology. 2008;135:234–243. doi: 10.1053/j.gastro.2008.03.020. [DOI] [PubMed] [Google Scholar]
- 191.Pietras K., Östman A. Hallmarks of cancer: Interactions with the tumor stroma. Exp. Cell Res. 2010;316:1324–1331. doi: 10.1016/j.yexcr.2010.02.045. [DOI] [PubMed] [Google Scholar]
- 192.Affo S., Yu L.X., Schwabe R.F. The Role of Cancer-Associated Fibroblasts and Fibrosis in Liver Cancer. Annu. Rev. Pathol. 2017;12:153–186. doi: 10.1146/annurev-pathol-052016-100322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Zhang Y., Yang B., Du Z., Bai T., Gao Y.T., Wang Y.J., Lou C., Wang F.M., Bai Y. Aberrant methylation of SPARC in human hepatocellular carcinoma and its clinical implication. World J. Gastroenterol. 2012;18:2043–2052. doi: 10.3748/wjg.v18.i17.2043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Nieto N., Friedman S.L., Cederbaum A.I. Stimulation and proliferation of primary rat hepatic stellate cells by cytochrome P450 2E1-derived reactive oxygen species. Hepatology. 2002;35:62–73. doi: 10.1053/jhep.2002.30362. [DOI] [PubMed] [Google Scholar]
- 195.Barry A.E., Baldeosingh R., Lamm R., Patel K., Zhang K., Dominguez D.A., Kirton K.J., Shah A.P., Dang H. Hepatic Stellate Cells and Hepatocarcinogenesis. Front. Cell Dev. Biol. 2020;8:709. doi: 10.3389/fcell.2020.00709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Viel S., Marçais A., Guimaraes F.S.-F., Loftus R., Rabilloud J., Grau M., Degouve S., Djebali S., Sanlaville A., Charrier E., et al. TGF-β inhibits the activation and functions of NK cells by repressing the mTOR pathway. Sci. Signal. 2016;9:ra19. doi: 10.1126/scisignal.aad1884. [DOI] [PubMed] [Google Scholar]
- 197.Vermijlen D., Luo D., Froelich C.J., Medema J.P., Kummer J.A., Willems E., Braet F., Wisse E. Hepatic natural killer cells exclusively kill splenic/blood natural killer-resistant tumor cells by the perforin/granzyme pathway. J. Leukoc. Biol. 2002;72:668–676. [PubMed] [Google Scholar]
- 198.Li T., Yang Y., Song H., Li H., Cui A., Liu Y., Su L., Crispe I.N., Tu Z. Activated NK cells kill hepatic stellate cells via p38/PI3K signaling in a TRAIL-involved degranulation manner. J. Leukoc. Biol. 2019;105:695–704. doi: 10.1002/JLB.2A0118-031RR. [DOI] [PubMed] [Google Scholar]
- 199.Singh H.D., Otano I., Rombouts K., Singh K.P., Peppa D., Gill U.S., Böttcher K., Kennedy P.T.F., Oben J., Pinzani M., et al. TRAIL regulatory receptors constrain human hepatic stellate cell apoptosis. Sci. Rep. 2017;7:5514. doi: 10.1038/s41598-017-05845-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Radaeva S., Wang L., Radaev S., Jeong W.-I., Park O., Gao B. Retinoic acid signaling sensitizes hepatic stellate cells to NK cell killing via upregulation of NK cell activating ligand RAE1. Am. J. Physiol. Liver Physiol. 2007;293:G809–G816. doi: 10.1152/ajpgi.00212.2007. [DOI] [PubMed] [Google Scholar]
- 201.Zhao W., Zhang L., Xu Y., Zhang Z., Ren G., Tang K., Kuang P., Zhao B., Yin Z., Wang X. Hepatic stellate cells promote tumor progression by enhancement of immunosuppressive cells in an orthotopic liver tumor mouse model. Lab. Investig. 2014;94:182–191. doi: 10.1038/labinvest.2013.139. [DOI] [PubMed] [Google Scholar]
- 202.Ghiringhelli F., Ménard C., Terme M., Flament C., Taieb J., Chaput N., Puig P.E., Novault S., Escudier B., Vivier E., et al. CD4+CD25+ regulatory T cells inhibit natural killer cell functions in a transforming growth factor-beta-dependent manner. J. Exp. Med. 2005;202:1075–1085. doi: 10.1084/jem.20051511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Fu J., Xu D., Liu Z., Shi M., Zhao P., Fu B., Zhang Z., Yang H., Zhang H., Zhou C., et al. Increased Regulatory T Cells Correlate With CD8 T-Cell Impairment and Poor Survival in Hepatocellular Carcinoma Patients. Gastroenterology. 2007;132:2328–2339. doi: 10.1053/j.gastro.2007.03.102. [DOI] [PubMed] [Google Scholar]
- 204.Ju M.-J., Qiu S.-J., Gao Q., Fan J., Cai M.-Y., Li Y.-W., Tang Z.-Y. Combination of peritumoral mast cells and T-regulatory cells predicts prognosis of hepatocellular carcinoma. Cancer Sci. 2009;100:1267–1274. doi: 10.1111/j.1349-7006.2009.01182.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Curtsinger J.M., Schmidt C.S., Mondino A., Lins D.C., Kedl R.M., Jenkins M.K., Mescher M.F. Inflammatory cytokines provide a third signal for activation of naive CD4+ and CD8+ T cells. J. Immunol. 1999;162:3256–3262. [PubMed] [Google Scholar]
- 206.Lever M., Maini P.K., Van Der Merwe P.A., Dushek O. Phenotypic models of T cell activation. Nat. Rev. Immunol. 2014;14:619–629. doi: 10.1038/nri3728. [DOI] [PubMed] [Google Scholar]
- 207.Pardoll D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer. 2012;12:252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Jiang Y., Li Y., Zhu B. T-cell exhaustion in the tumor microenvironment. Cell Death Dis. 2015;6:e1792. doi: 10.1038/cddis.2015.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.El Dika I., Khalil D.N., Abou-Alfa G.K. Immune checkpoint inhibitors for hepatocellular carcinoma. Cancer. 2019;125:3312–3319. doi: 10.1002/cncr.32076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Ritz T., Krenkel O., Tacke F. Dynamic plasticity of macrophage functions in diseased liver. Cell. Immunol. 2018;330:175–182. doi: 10.1016/j.cellimm.2017.12.007. [DOI] [PubMed] [Google Scholar]
- 211.Devisscher L., Verhelst X., Colle I., Van Vlierberghe H., Geerts A. The role of macrophages in obesity-driven chronic liver disease. J. Leukoc. Biol. 2016;99:693–698. doi: 10.1189/jlb.5RU0116-016R. [DOI] [PubMed] [Google Scholar]
- 212.Bekkering S., Blok B.A., Joosten L.A., Riksen N.P., van Crevel R., Netea M.G. In Vitro Experimental Model of Trained Innate Immunity in Human Primary Monocytes. Clin. Vaccine Immunol. 2016;23:926–933. doi: 10.1128/CVI.00349-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Xue J., Schmidt S.V., Sander J., Draffehn A., Krebs W., Quester I., De Nardo D., Gohel T.D., Emde M., Schmidleithner L., et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity. 2014;40:274–288. doi: 10.1016/j.immuni.2014.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Mourits V.P., Arts R.J., Novakovic B., Matzaraki V., De Bree L.C.J., Koeken V.A., Moorlag S.J., Van Puffelen J.H., Groh L., Van Der Heijden C.D., et al. The role of Toll-like receptor 10 in modulation of trained immunity. Immunology. 2019;159:289–297. doi: 10.1111/imm.13145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.O’Connell R.M., Chaudhuri A.A., Rao D.S., Baltimore D. Inositol phosphatase SHIP1 is a primary target of miR-155. Proc. Natl. Acad. Sci. USA. 2009;106:7113–7118. doi: 10.1073/pnas.0902636106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Quintin J., Saeed S., Martens J.H., Giamarellos-Bourboulis E.J., Ifrim D.C., Logie C., Jacobs L., Jansen T., Kullberg B.-J., Wijmenga C., et al. Candida albicans Infection Affords Protection against Reinfection via Functional Reprogramming of Monocytes. Cell Host Microbe. 2012;12:223–232. doi: 10.1016/j.chom.2012.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Yona S., Kim K.W., Wolf Y., Mildner A., Varol D., Breker M., Strauss-Ayali D., Viukov S., Guilliams M., Misharin A., et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity. 2013;38:79–91. doi: 10.1016/j.immuni.2012.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Kleinnijenhuis J., Quintin J., Preijers F., Joosten L.A.B., Ifrim D.C., Saeed S., Jacobs C., Van Loenhout J., De Jong D., Stunnenberg H.G., et al. Bacille Calmette-Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc. Natl. Acad. Sci. USA. 2012;109:17537–17542. doi: 10.1073/pnas.1202870109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Morgillo F., Dallio M., Morgillo F., Gravina A.G., Viscardi G., Loguercio C., Ciardiello F., Federico A. Carcinogenesis as a Result of Multiple Inflammatory and Oxidative Hits: A Comprehensive Review from Tumor Microenvironment to Gut Microbiota. Neoplasia. 2018;20:721–733. doi: 10.1016/j.neo.2018.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Ganeshan K., Chawla A. Metabolic Regulation of Immune Responses. Annu. Rev. Immunol. 2014;32:609–634. doi: 10.1146/annurev-immunol-032713-120236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Sangineto M., Grabherr F., Adolph T.E., Grander C., Reider S., Jaschke N., Mayr L., Schwärzler J., Dallio M., Moschen A.R., et al. Dimethyl fumarate ameliorates hepatic inflammation in alcohol related liver disease. Liver Int. 2020;40:1610–1619. doi: 10.1111/liv.14483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Tannahill G.M., Curtis A.M., Adamik J., Palsson-McDermott E.M., McGettrick A.F., Goel G., Frezza C., Bernard N.J., Kelly B., Foley N.H., et al. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature. 2013;496:238–242. doi: 10.1038/nature11986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Lampropoulou V., Sergushichev A., Bambouskova M., Nair S., Vincent E.E., Loginicheva E., Cervantes-Barragan L., Ma X., Huang S.C.-C., Griss T., et al. Itaconate Links Inhibition of Succinate Dehydrogenase with Macrophage Metabolic Remodeling and Regulation of Inflammation. Cell Metab. 2016;24:158–166. doi: 10.1016/j.cmet.2016.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Jha A.K., Huang S.C.-C., Sergushichev A., Lampropoulou V., Ivanova Y., Loginicheva E., Chmielewski K., Stewart K.M., Ashall J., Everts B., et al. Network Integration of Parallel Metabolic and Transcriptional Data Reveals Metabolic Modules that Regulate Macrophage Polarization. Immunity. 2015;42:419–430. doi: 10.1016/j.immuni.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 225.Michelucci A., Cordes T., Ghelfi J., Pailot A., Reiling N., Goldmann O., Binz T., Wegner A., Tallam A., Rausell A., et al. Immune-responsive gene 1 protein links metabolism to immunity by catalyzing itaconic acid production. Proc. Natl. Acad. Sci. USA. 2013;110:7820–7825. doi: 10.1073/pnas.1218599110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Dominguez-Andres J., Novakovic B., Li Y., Scicluna B.P., Gresnigt M.S., Arts R.J., Oosting M., Moorlag S.J., Groh L.A., Zwaag J., et al. The Itaconate Pathway Is a Central Regulatory Node Linking Innate Immune Tolerance and Trained Immunity. Cell Metab. 2019;29:211–220.e5. doi: 10.1016/j.cmet.2018.09.003. [DOI] [PubMed] [Google Scholar]
- 227.Xiao W., Wang L., Xiao R., Wu M., Jinquan T., He Y. Expression profile of human immune-responsive gene 1 and generation and characterization of polyclonal antiserum. Mol. Cell. Biochem. 2011;353:177–187. doi: 10.1007/s11010-011-0784-7. [DOI] [PubMed] [Google Scholar]
- 228.Ganta V.C., Choi M.H., Kutateladze A., Fox T.E., Farber C.R., Annex B.H. A MicroRNA93–Interferon Regulatory Factor-9–Immunoresponsive Gene-1–Itaconic Acid Pathway Modulates M2-Like Macrophage Polarization to Revascularize Ischemic Muscle. Circulation. 2017;135:2403–2425. doi: 10.1161/CIRCULATIONAHA.116.025490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Bambouskova M., Gorvel L., Lampropoulou V., Sergushichev A., Loginicheva E., Johnson K., Korenfeld D., Mathyer M.E., Kim H., Huang L.H., et al. Electrophilic properties of itaconate and derivatives regulate the IκBζ-ATF3 inflammatory axis. Nature. 2018;556:501–504. doi: 10.1038/s41586-018-0052-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Costa A.S.H., Higgins M., Hams E., Szpyt J., Runtsch M.C., King M.S., McGouran J.F., Fischer R., Kessler B.M., McGettrick A.F., et al. Itaconate is an anti-inflammatory metabolite that activates Nrf2 via alkylation of KEAP1. Nature. 2018;556:113–117. doi: 10.1038/nature25986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Cordes T., Lucas A., Divakaruni A.S., Murphy A.N., Cabrales P., Metallo C.M. Itaconate modulates tricarboxylic acid and redox metabolism to mitigate reperfusion injury. Mol. Metab. 2020;32:122–135. doi: 10.1016/j.molmet.2019.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Yi Z., Deng M., Scott M.J., Fu G., Loughran P.A., Lei Z., Li S., Sun P., Yang C., Li W., et al. IRG1/Itaconate Activates Nrf2 in Hepatocytes to Protect Against Liver Ischemia-Reperfusion Injury. Hepatology. 2020;72:1394–1411. doi: 10.1002/hep.31147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Lefere S., Tacke F. Macrophages in obesity and non-alcoholic fatty liver disease: Crosstalk with metabolism. JHEP Rep. 2019;1:30–43. doi: 10.1016/j.jhepr.2019.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Federico A., Dallio M., Di Sarno R., Giorgio V., Miele L. Gut microbiota, obesity and metabolic disorders. Minerva Gastroenterol. Dietol. 2017;63:337–344. doi: 10.23736/S1121-421X.17.02376-5. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.