Abstract
As of November 25, 2020, over 60 million people have been infected worldwide by COVID-19, causing almost 1.43 million deaths. Puzzling low incidence numbers and milder, non-fatal disease have been observed in Thailand and its Southeast (SE) Asian neighbors. Elusive genetic mechanisms might be operative, as a multitude of genetic factors are widely shared between the SE Asian populations, such as the more than 60 different thalassemia syndromes (principally dominated by the HbE trait). In this study, we have plotted COVID-19 infection and death rates in SE Asian (SEA) countries against heterozygote HbE and thalassemia carrier prevalence. COVID-19 infection and death incidence numbers appear inversely correlated with the prevalence of HbE and thalassemia heterozygote populations. We posit that the evolutionary protective effect of the HbE and other thalassemic variants against malaria and the dengue virus may extend its advantage to resistance to COVID-19 infection, as HbE heterozygote population prevalence appears to be positively correlated with immunity to COVID-19. Host immune system modulations induce antiviral interferon responses and alter structural protein integrity, thereby inhibiting cellular access and viral replication. These changes are possibly engendered by HbE carrier miRNAs. Proving this hypothesis is important, as it may shed light on the mechanism of viral resistance and lead to novel antiviral treatments. This development can thus guide decision-making and action to prevent COVID-19 infection.
MeSH Keywords: Asia, Southeast; beta-Thalassemia; COVID-19; Hemoglobinopathies; Thailand; Thalassemia
Background
The coronavirus pandemic (COVID-19) has to date infected over 60 million people worldwide, causing more than 1.43 million deaths [1]. The latest COVID-19 statistics for the Kingdom of Thailand as of November 25, 2020, were 3942 confirmed cases and 60 deaths in a total population of 66.6 million [2,3]. These numbers translate to 56.25 cases and 0.86 deaths per million inhabitants, respectively, both being puzzlingly low among countries with reliable statistical information [4]. The whole of the Mekong River basin and other Southeast (SE) Asian (SEA) neighbors seems to share Thailand’s low infection and death rates. Myanmar, Laos, Cambodia, and China’s Yunnan province all report similarly minimal infection rates (Table 1, Figures 1, 2) [4,5]. Many attribute this low incidence to Thai social and cultural practices promoting an inherent social distancing. When greeting one another, Thais do not shake hands or hug, but use the “wai”, a prayer-like motion. An outdoor lifestyle combined with mask wearing practices due to pre-existing environmental pollution was already in place before the pandemic [5]. Thailand’s efficient, robust, and responsive grassroots-level universal health care system, directed by able scientists, has greatly impacted preventive and treatment measures [6] even with a rising prevalence of pre-existing comorbidities like obesity, diabetes, and hypertension in the whole of the Mekong river region [7,8]. Others might argue that Thailand’s low testing frequency (19 tests per 1000 people) might underestimate the magnitude of the COVID-19 situation [3]. Here, we need to bear in mind that while all returning residents are tested and quarantined, community testing only occurs when tracing contacts or for symptomatic cases seeking treatment (virtually non-existent in the community). Another way of looking at the extent of testing relative to the scale of the outbreak is to ask: how many tests does a country do to find 1 COVID-19 case? The WHO has suggested around 10–30 tests per confirmed case as a general benchmark of adequate testing, and the Thai average as of November 5, 2020 was 649.7 tests per 1 positive case [9]. With her porous land border crossings, Thailand allows for population movements. Furthermore, COVID-19 infection rates have increased in its western neighbors. Yet Thailand has not seen any increase in the number of local COVID19 cases. After several months without local infections, single-digit indigenous re-emergences have died out spontaneously both in Thailand and Vietnam [10]. Elusive locally-shared, genetic determinants may provide an explanation to the above observations.
Table 1.
HbE and any Thalassemia carrier prevalence are given and opposed against COVID-19 infections and deaths per one million (M) population [1,3,4,13,15–32].
| Prevalence/Country | HbE carrier (%) | Any thalassemia carrier (%) | HERD immunity limit (%) | COVID-19 deaths per M | COVID-19 cases per M |
|---|---|---|---|---|---|
| Thailand | 53 | 52.6 | 43 | 0.86 | 55.76 |
| Laos | 53 | 52.6 | 43 | 0 | 3.3 |
| Cambodia | 62 | 62.7 | 43 | 0 | 18.18 |
| Myanmar | 42 | 44.1 | 43 | 30.8 | 1376.26 |
| Indonesia | 33 | 56 | 43 | 57.03 | 1757.74 |
| Yunnan Province (China) | 39.1 | 21.7 | 43 | 0.43 | 4.09 |
| Vietnam | 35.6 | 30 | 43 | 0.36 | 13.4 |
| India | 30 | 21 | 43 | 95.77 | 6524.88 |
| Bangladesh | 8.68 | 11.89 | 43 | 38.28 | 2678.73 |
| Malaysia | 3.8 | 16.25 | 43 | 10.07 | 1596.74 |
| Sri Lanka | 0.5 | 15.8 | 43 | 3.41 | 879.88 |
| Hong Kong | 2 | 10 | 43 | 1.84 | 269.44 |
| Philippines | 2 | 8.2 | 43 | 72.99 | 3772.82 |
| Pakistan | 0.4 | 9 | 43 | 34.23 | 1668.98 |
| Taiwan | 0.027 | 8.4 | 43 | 0.29 | 25.57 |
| Singapore | 0.64 | 3.85 | 43 | 4.79 | 9937.71 |
| Brazil | 1.81 | 43 | 790.65 | 28785.87 | |
| Spain | 0.92 | 43 | 933.98 | 34110.80 | |
| France | 3.2 | 43 | 769.64 | 32996.77 | |
| Italy | 12 | 43 | 848.57 | 24065.13 | |
| Greece | 15 | 43 | 129.23 | 9127.55 | |
| Cyprus | 35 | 43 | 52.52 | 10502.35 |
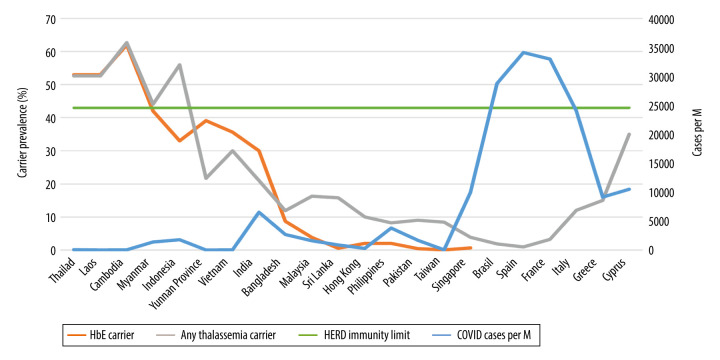
Figure 1.
HbE and any thalassemia carrier prevalence (%) versus COVID-19 cases per million (M) people on November 25, 2020.
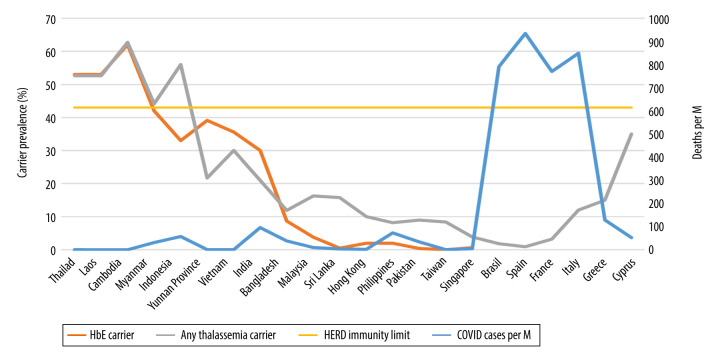
Figure 2.
HbE and any thalassemia carrier prevalence (%) versus COVID-19 confirmed deaths per million (M) people on November 25, 2020.
Epidemiology of HbE and Thalassemia Traits in Southeast Asia
SE Asia is the third most populous region in the world [11], encompassing 10 countries with more than 650 million people inhabiting an area over 4.5 million km2. SE Asia is a genetic cauldron of different ethnicities descending from diverse and complex origins [11,12]. Shaped by local malarial environmental pressures, SE Asia’s common genetic denominator is thalassemia, presenting with over 60 different allelic variants, but mainly dominated by 4 major thalassemic syndromes [13]. The α-thalassemias, with a gene prevalence approaching 40% in northern Thailand and Laos, are considerably less common in Malaysia and the Philippines (around 5%) [13]. The β-thalassemias showing local variations in genetic prevalence account for up to 9% of all cases. Thirdly, Hb Constant Spring [CS] is also seen, with gene frequencies of up to 8% [13]. HbE, the thalassemic symbol of SE Asia, is increasingly encountered worldwide. HbE gene prevalence may reach up to 70% locally in northeastern Thailand and and up to 62% at the triple border region between Thailand, Cambodia, and Laos [13–15]. Apart from SE Asia, thalassemia genes (including HbE) are also present with diminishing gene frequency in Yunnan, Nepal, Northeastern India, Pakistan, Sri Lanka, Bangladesh, Hong Kong, and Taiwan [13–24].
Epidemiological Evidence of HbE Protection Against COVID-19
Intriguing epidemiological observations suggest, and biological evidence supports, an inverse correlation between increasing HbE/thalassemia trait prevalence rates and decreased susceptibility to, and fatality from, COVID-19 infections (Table 1, Figures 1, 2). Local COVID-19 infections within Thailand vary 6-fold between the northeast (2.84% of total cases) versus the south (19%), in tandem with regional variations in HbE gene frequency (70% in the northeast compared to 12% in the south) [3,13,25]. Similarly, Malaysia, which borders southern Thailand, has an HbE trait frequency of only 3.8% [26] but reports substantially higher COVID19 infections compared to Thailand [4]. Interestingly, in Cambodia and Laos (where zero COVID-19 deaths are reported) (Table 1), thalassemia mutation rates are similar to Thailand [13]; for Cambodia, the thalassemia prevalence is 62.7% (HbE being the most prevalent, at 56%) [27]. In Myanmar (HbE carrier rates vary between 1.9 and 42% depending on ethnic group and geography), low infection and death rates are also seen (Table 1) [28]. Vietnam and the neighboring Chinese province of Yunnan both report low COVID-19 infection rates, with locally high prevalence of HbE trait in line with other mainland SE Asian nations (Table 1) [15–24,29–31]. In Vietnam, the Mon-Khmer-speaking ethnic groups are genetically closer to the Thai-Lao-Cambodian border populations and report HbE gene prevalence up to 36% [29]. Singapore, where the prevalence of β-thalassemia trait is 0.9% and HbE trait is 0.55% [32], was severely hit by COVID-19 compared to neighboring Thailand, but the death rates were very low [4]. The high number of cases in Singapore seems to stem from clusters in male migrant workers living in close proximity in crowded living quarters, promoting higher infectious spread [33]. These migrant workers are from various ethnic groups (e.g., Bengali and Tamil) with higher HbE/thalassemia trait prevalence but also higher glucose-6-phosphate dehydrogenase (G6PD) deficiency rates, compared to resident Singaporeans [32,34,35]. G6PD-deficient patients have been shown to be highly susceptible to enteroviruses, human dengue virus, and human coronavirus 229E infection. They also tend to develop severe pneumonia after microbial infection [36]. Consequently, close proximity overrides the protective effects of HbE and/or selectively potentiates the risk to infect the ones without the hypothetically protective trait, especially from patients carrying a higher viral load. Thailand’s and Singapore’s effective 14-day quarantine policy of all arrivals have now virtually eradicated local COVID-19 cases [37]. In the remainder of SE Asia and South Asian territories, the trend continues with increasing COVID-19 infection and death rates where HbE/thalassemia trait prevalence is low (Table 1, Figures 1, 2) [14–23]. In Brazil, a country severely hit by COVID-19, despite heavy early Italian migrations, less than 2% of Brazilian blood donors exhibit hemoglobinopathies [38]. Similarly, Spain, also severely affected by COVID-19, has a thalassemia trait of only 0.92% [39]. Despite its higher thalassemia trait prevalence, Italy was severely hit by COVID-19, initially in Lombardy, with a traditionally low carrier rate compared to islands and coastal areas [40]. Greece [40] and Cyprus [41], with an effective lock-down, a distancing strategy, and a higher, homogenous carrier rate, seem to be experiencing lower COVID-19 rates compared to their European peers, even during the second wave (Table 1, Figures 1, 2).
How is the HbE Trait Protective?
The hypothesis we posit is that thalassemia traits in general, and particularly HbE, are protective against COVID-19 infection in similar ways to the numerous thalassemia traits conferring protection against malaria [30,42] and the dengue virus [43]. Herd immunity development traditionally requires that 60% of the population becomes homogeneously infected by or vaccinated against an infectious disease. However, recent mathematical models introducing age and activity heterogeneities into population models predict that herd immunity can be achieved at 43% [44], a level substantially lower than the observed prevalence of HbE in some areas of SE Asia [13–15]. In fact, in countries where the HbE trait is above 43%, the numbers of both COVID-19 infections and deaths have been minimal (Table 1, Figures 1, 2).
The genetic and molecular mechanisms of this hypothesized protection are as yet elusive, but biological evidence is suggestive. Hemoglobinopathy-induced red blood cell structural modifications hinder invasion, growth, and migration of plasmodium [42,45]. Recent studies in thalassemias have pointed to the involvement of microRNAs (miRNAs) in malarial pathogenesis and anti-plasmodial defense [45]. MicroRNAs (miRNAs) are 18–25 nucleotide long, small, non-coding RNA molecules whose production is strictly regulated and abundant in all human cells [46]. miRNAs can downregulate gene expression in translational repression and target around 60% of all genes [46]. They exhibit decisive regulatory functions associated with a variety of disease processes, including microbial defense [46]. In dengue virus infection, which is perennially endemic in SE Asia, red blood cell precursors in Thai carriers of thalassemia and HbE trait were significantly less susceptible to the dengue virus compared to normal controls [43]. This was the first report documenting an antiviral effect of the HbE trait akin to its anti-malarial effect [43]. A large number of miRNAs have also been implicated in dengue virus defense via structural protein integrity alterations inhibiting access of viral replication machinery to the cytoskeletal apparatus (miR-223) [47], but also through modulations of the host immune interferon response (miR-155) [48]. Remarkably, increased levels of the latter molecule have been reported in β-thalassemia/HbE patients and linked to BACH1 downregulation [49]. The same miRNA molecule was also reported to inhibit dengue virus replication by inducing antiviral interferon responses through the same BACH1 pathway downregulation and heme oxygenase-1-(HO-1) induction [48]. Rare putative loss-of-function variants of X-chromosomal toll-like receptor 7 (TLR7) causing immunological defects in type I and II interferon production have been very recently identified in 4 young male patients with severe COVID-19 [50]. Type I and II interferon (IFN) responses have been implicated in the initiation of an early immune response to clear the SARS-CoV-2 coronavirus and prevent the development of COVID-19 [50]. Moreover, the addition of interferon beta-1b to other antivirals in the clinical setting was also more effective in treating COVID-19 patients and rendering them noninfectious [51]. HO-1 pathway derangements have been implicated in severe COVID-19 infection [52] and in causing exhaustion of hematopoietic stem cells, possibly leading to immune system failure [53]. Similar structural and molecular mechanisms might be operative in immune effector cells of HbE heterozygotes.
The hypothesized conferred protection against COVID-19 could also derive from local SE Asian HLA-class allotypes, possibly in linkage disequilibrium with the thalassemia mutations. Protection from and resistance to severe malaria has been described in association with HLA antigens in the African continent [54,55] and the ABO blood group system [56]. However, no HLA associations with COVID-19 infection have been noted in a recent report [57] but an association with the ABO blood group system was confirmed, with bearers of the blood group A phenotype showing an increased risk for COVID-19 infection compared to blood group O [58]. The O phenotype appears protective to malaria [56] and its worldwide distribution seems to have been shaped by the parasite’s selective genetic pressure [56]. The frequency of the O blood group in Thailand is 40.5% [59] and its relatively high prevalence along with the high HbE heterozygote frequency could further potentiate the hypothesized antiviral protective effect.
A similarly intriguing hypothesis querying whether Italian β-thalassemia subjects are immunized against COVID-19 has been put forward [60]. We believe that the diluted carrier population sizes available in southern European countries are not of the magnitude presently observed in SE Asia and could lead to erroneous interpretation [60,61].
Limitations and Proving the HbE Protection Hypothesis
We are not claiming causality at this stage but are hypothesizing an interesting association that needs to be tested with regard to causality. Simple hemoglobin electrophoresis of the approximately 4000 infected Thai COVID-19 cohort patients as a case control study will reveal whether the prevalence of thalassemia traits in general (and HbE in particular) differs between infected and uninfected populations. Advanced molecular analyses can be performed on the affected cases. G6PD and ABO blood group analyses could also be performed to elucidate whether the COVID-19-infected cohort has a higher representation of these genetic determinants. Furthermore, miRNA analyses and genetic polymorphism and molecular studies of the BACH1/HO-1 pathway and interferons would elucidate the mechanism of resistance and eventually lead to novel treatment modalities via miRNA decoys or miRNA mimetics or interferon administration [46,51].
Conclusions
We theorize that another “Amazing Thailand” attribute [62], an evolutionary HbE variant arising in Thailand through natural selection possibly around 2000 years ago in a malaria-endemic region, might hypothetically be providing its carriers survival advantages with resistance to malaria, dengue virus, and possibly also to COVID-19 infection [63]. Host immune system modulations induce antiviral interferon responses as well as alter structural protein integrity and inhibit cellular access and viral replication. These changes are possibly engendered by HbE/thalassemia carrier miRNAs. Proving the hypothesis is of importance, as it may shed light on the mechanisms of viral resistance and lead to novel antiviral treatments. These developments can guide decision-making and action to prevent COVID-19 infection.
Acknowledgements
We are thankful to James T.A. Marshall and Professor Bengt Hallengren for their invaluable input on the manuscript.
Footnotes
Conflicts of interest
None.
Source of support: Departmental sources
References
- 1.https://ourworldindata.org/grapher/total-confirmed-cases-of-covid-19-per-million-people?tab=table
- 2.https://www.boi.go.th/index.php?page=demographic
- 3.https://covid19.ddc.moph.go.th/en
- 4.https://ourworldindata.org/grapher/total-confirmed-cases-of-covid-19-per-million-people?tab=table
- 5.https://www.nytimes.com/2020/07/16/world/asia/coronavirus-thailand-photos.html
- 6.https://www.bangkokpost.com/thailand/general/1746289/thailands-healthcare-ranked-sixth-best-in-the-world
- 7.Sakboonyarat B, Pornpongsawad C, Sangkool T, et al. Trends, prevalence and associated factors of obesity among adults in a rural community in Thailand: Serial cross-sectional surveys, 2012 and 2018. BMC Public Health. 2020;20(1):850. doi: 10.1186/s12889-020-09004-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chavasit V, Kriengsinyos W, Photi J, Tontisirin K. Trends of increases in potential risk factors and prevalence rates of diabetes mellitus in Thailand. Eur J Clin Nutr. 2017;71(7):839–43. doi: 10.1038/ejcn.2017.52. [DOI] [PubMed] [Google Scholar]
- 9.https://ourworldindata.org/coronavirus-testing#tests-per-confirmed-case
- 10.https://www.bbc.com/news/world-asia-53690711
- 11.https://www.usasean.org/why-asean/what-is-asean
- 12.HUGO Pan-Asian SNP Consortium. Abdulla MA, Ahmed I, et al. Mapping human genetic diversity in Asia. Science. 2009;326(5959):1541–45. doi: 10.1126/science.1177074. [DOI] [PubMed] [Google Scholar]
- 13.Fucharoen S, Winichagoon P. Haemoglobinopathies in southeast Asia. Indian J Med Res. 2011;134(4):498–506. [PMC free article] [PubMed] [Google Scholar]
- 14.Bachir D, Galacteros F. Hemoglobin E disease. Orphanet Encyclopedia. 2004 Nov; http://www.orpha.net/data/patho/GB/uk-HbE.pdf. [Google Scholar]
- 15.Viprakasit V, Lee-Lee C, Chong QT, et al. Iron chelation therapy in the management of thalassemia: The Asian perspectives. Int J Hematol. 2009;90(4):435–45. doi: 10.1007/s12185-009-0432-0. [DOI] [PubMed] [Google Scholar]
- 16.Patne SC, Shukla J. Hemoglobin E disorders in Eastern Uttar Pradesh. Indian J Pathol Microbiol. 2009;52(1):110–12. doi: 10.4103/0377-4929.44991. [DOI] [PubMed] [Google Scholar]
- 17.Hossain MS, Raheem E, Sultana TA, et al. Thalassemias in South Asia: Clinical lessons learnt from Bangladesh. Orphanet J Rare Dis. 2017;12(1):93. doi: 10.1186/s13023-017-0643-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Noor FA, Sultana N, Bhuyan GS, et al. Nationwide carrier detection and molecular characterization of β-thalassemia and hemoglobin E variants in Bangladeshi population. Orphanet J Rare Dis. 2020;15(1):15. doi: 10.1186/s13023-020-1294-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ansari SH, Shamsi TS, Ashraf M, et al. Molecular epidemiology of β-thalassemia in Pakistan: Far reaching implications. Int J Mol Epidemiol Genet. 2011;2(4):403–8. [PMC free article] [PubMed] [Google Scholar]
- 20.Moiz B, Hashmi MR, Nasir A, et al. Hemoglobin E syndromes in Pakistani population. BMC Blood Disord. 2012;12:3. doi: 10.1186/1471-2326-12-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee CK, Wong HK, Hong J, et al. A study of the predonation hemoglobin and iron status among Hong Kong Chinese blood donors. Transfusion. 2013;53(2):322–27. doi: 10.1111/j.1537-2995.2012.03788.x. [DOI] [PubMed] [Google Scholar]
- 22.Capanzana MV, Mirasol LMA, Smith G, et al. Thalassemia and other hemoglobinopathies among anemic individuals in Metro Manila, Philippines and their intake of iron supplements. Asia Pac J Clin Nutr. 2018;27(3):519–26. doi: 10.6133/apjcn.092017.01. [DOI] [PubMed] [Google Scholar]
- 23.Black ML, Sinha S, Agarwal S, et al. A descriptive profile of β-thalassaemia mutations in India, Pakistan and Sri Lanka. J Community Genet. 2010;1(3):149–57. doi: 10.1007/s12687-010-0026-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Timan IS, Aulia D, Atmakusma D, et al. Some hematological problems in Indonesia. Int J Hematol. 2002;76(Suppl 1):286–90. doi: 10.1007/BF03165264. [DOI] [PubMed] [Google Scholar]
- 25.Nuinoon M, Kruachan K, Sengking W, et al. Thalassemia and hemoglobin e in southern Thai blood donors. Adv Hematol. 2014;2014 doi: 10.1155/2014/932306. 932306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jameela S, Sabirah SO, Babam J, et al. Thalassaemia screening among students in a secondary school in Ampang, Malaysia. Med J Malaysia. 2011;66(5):522–24. [PubMed] [Google Scholar]
- 27.Munkongdee T, Tanakulmas J, Butthep P, et al. Molecular epidemiology of hemoglobinopathies in Cambodia. Hemoglobin. 2016;40(3):163–67. doi: 10.3109/03630269.2016.1158723. [DOI] [PubMed] [Google Scholar]
- 28.Bancone G, Gilder ME, Chowwiwat N, et al. Prevalences of inherited red blood cell disorders in pregnant women of different ethnicities living along the Thailand-Myanmar border. Wellcome Open Res. 2017;2:72. doi: 10.12688/wellcomeopenres.12338.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.O’Riordan S, Hien TT, Miles K, et al. Large-scale screening for haemoglobin disorders in southern Vietnam: implications for avoidance and management. Br J Haematol. 2010;150(3):359–64. doi: 10.1111/j.1365-2141.2010.08237.x. [DOI] [PubMed] [Google Scholar]
- 30.Deng Z, Li Q, Yi H, et al. Hemoglobin E protects against acute Plasmodium vivax infections in a Kachin population at the China-Myanmar border. J Infect. 2018;77(5):435–39. doi: 10.1016/j.jinf.2018.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu H, Huang K, Liu S, et al. Gene frequency and haplotype distribution of hemoglobin E among seven minority groups of Yunnan, China. Am J Hum Biol. 2016;28(6):927–31. doi: 10.1002/ajhb.22868. [DOI] [PubMed] [Google Scholar]
- 32.National Thalassaemia Registry Statistics. Singapore: 2011. [Google Scholar]
- 33.Koh D. Migrant workers and COVID-19. Occup Environ Med. 2020;77(9):634–36. doi: 10.1136/oemed-2020-106626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sasi S, Yassin MA, Nair AP, Al Maslamani MS. A case of COVID-19 in a patient with asymptomatic hemoglobin D rhalassemia and glucose-6-phosphate dehydrogenase deficiency. Am J Case Rep. 2020;21:e925788. doi: 10.12659/AJCR.925788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vick DJ. Glucose-6-phosphate dehydrogenase deficiency and COVID-19 infection. Mayo Clin Proc. 2020;95(8):1803–4. doi: 10.1016/j.mayocp.2020.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wu YH, Chiu DT, Lin HR, et al. Glucose-6-phosphate dehydrogenase enhances antiviral response through downregulation of NADPH sensor HSCARG and upregulation of NF-κB signaling. Viruses. 2015;7(12):6689–706. doi: 10.3390/v7122966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.https://www.bangkokpost.com/world/2025347/singapore-nearly-virus-free-after-local-cases-and-clusters-cease
- 38.Lisot CL, Silla LM. [Screening for hemoglobinopathies in blood donors from Caxias do Sul, Rio Grande do Sul, Brazil: Prevalence in an Italian colony]. Cad Saude Publica. 2004;20(6):1595–601. doi: 10.1590/s0102-311x2004000600017. [in Portuguese] [DOI] [PubMed] [Google Scholar]
- 39.Casado A, Casado MC, Lopez-Fernandez ME, Venarucci D. Thalassemia and G6PD deficiency in Spanish blood donors. Panminerva Med. 1997;39(3):205–7. [PubMed] [Google Scholar]
- 40.World Health Organization. Management of haemoglobin disorders: Report of a joint WHO-TIF meeting; 2007 November 16–18; Nicosia, Cyprus. https://apps.who.int/iris/handle/10665/43969. [Google Scholar]
- 41.Kountouris P, Kousiappa I, Papasavva T, et al. The molecular spectrum and distribution of haemoglobinopathies in Cyprus: A 20-year retrospective study. Sci Rep. 2016;20:26371. doi: 10.1038/srep26371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ha J, Martinson R, Iwamoto SK, Nishi A. Hemoglobin E, malaria and natural selection. Evol Med Public Health. 2019;2019(1):232–41. doi: 10.1093/emph/eoz034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sornjai W, Khungwanmaythawee K, Svasti S, et al. Dengue virus infection of erythroid precursor cells is modulated by both thalassemia trait status and virus adaptation. Virology. 2014;471–73:61–71. doi: 10.1016/j.virol.2014.10.004. [DOI] [PubMed] [Google Scholar]
- 44.Britton T, Ball F, Trapman P. A mathematical model reveals the influence of population heterogeneity on herd immunity to SARS-CoV-2. Science. 2020;369(6505):846–49. doi: 10.1126/science.abc6810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chakrabarti M, Garg S, Rajagopal A, et al. Targeted repression of Plasmodium apicortin by host microRNA impairs malaria parasite growth and invasion. Dis Model Mech. 2020;13(6):dmm042820. doi: 10.1242/dmm.042820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Papadopoulos KI, Wattanaarsakit P, Prasongchean W, Narain R. 10 – Gene therapies in clinical trials. In: Narain R, editor. Polymers and nanomaterials for gene therapy. Woodhead Publishing; 2016. pp. 231–56. [Google Scholar]
- 47.Wong RR, Abd-Aziz N, Affendi S, Poh CL. Role of microRNAs in antiviral responses to dengue infection. J Biomed Sci. 2020;27(1):4. doi: 10.1186/s12929-019-0614-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Su Y-C, Huang Y-F, Wu Y-W, et al. MicroRNA-155 inhibits dengue virus replication by inducing heme oxygenase-1-mediated antiviral interferon responses. FASEB J. 2020;34:7283–94. doi: 10.1096/fj.201902878R. [DOI] [PubMed] [Google Scholar]
- 49.Srinoun K, Nopparatana C, Wongchanchailert M, Fucharoen S. MiR-155 enhances phagocytic activity of β-thalassemia/HbE monocytes via targeting of BACH1. Int J Hematol. 2017;106(5):638–47. doi: 10.1007/s12185-017-2291-4. [DOI] [PubMed] [Google Scholar]
- 50.van der Made CI, Simons A, Schuurs-Hoeijmakers J, et al. Presence of genetic variants among young men with severe COVID-19. JAMA. 2020;324(7):1–11. doi: 10.1001/jama.2020.13719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hung IF-N, Lung K-C, Tso EY-K, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395(10238):1695–704. doi: 10.1016/S0140-6736(20)31042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hooper PL. COVID-19 and heme oxygenase: novel insight into the disease and potential therapies. Cell Stress Chaperones. 2020;25(5):707–10. doi: 10.1007/s12192-020-01126-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Szade K, Zukowska M, Szade A, et al. Heme oxygenase-1 deficiency triggers exhaustion of hematopoietic stem cells. EMBO Rep. 2020;21(2):e47895. doi: 10.15252/embr.201947895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hill AV, Allsopp CE, Kwiatkowski D, et al. Common west African HLA antigens are associated with protection from severe malaria. Nature. 1991;352(6336):595–600. doi: 10.1038/352595a0. [DOI] [PubMed] [Google Scholar]
- 55.Hill AV, Elvin J, Willis AC, et al. Molecular analysis of the association of HLA-B53 and resistance to severe malaria. Nature. 1992;360(6403):434–39. doi: 10.1038/360434a0. [DOI] [PubMed] [Google Scholar]
- 56.Cserti Christine M, Dzik Walter H. The ABO blood group system and Plasmodium falciparum malaria. Blood. 2007;110(7):2250–58. doi: 10.1182/blood-2007-03-077602. [DOI] [PubMed] [Google Scholar]
- 57.Kaser A. Genetic risk of severe Covid-19. N Engl J Med. 2020;383:1590–91. doi: 10.1056/NEJMe2025501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.The Severe Covid-19 GWAS Group. Genomewide association study of severe Covid-19 with respiratory failure. N Engl J Med. 2020;383:1522–34. doi: 10.1056/NEJMoa2020283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nathalang O, Kuvanont S, Punyaprasiddhi P, et al. A preliminary study of the distribution of blood group systems in Thai blood donors determined by the gel test. Southeast Asian J Trop Med Public Health. 2001;32(1):204–7. [PubMed] [Google Scholar]
- 60.Lansiaux E, Pébaÿ PP, Picard JL, Son-Forget J. COVID-19: beta-thalassemia subjects immunised? Med Hypotheses. 2020;142:109827. doi: 10.1016/j.mehy.2020.109827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Drouin E. beta-thalassemia may protect against COVID 19. Med Hypotheses. 2020;143:110014. doi: 10.1016/j.mehy.2020.110014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.https://www.tourismthailand.org/
- 63.Ohashi J, Naka I, Patarapotikul J, et al. Extended linkage disequilibrium surrounding the hemoglobin E variant due to malarial selection. Am J Hum Genet. 2004;74(6):1198–208. doi: 10.1086/421330. [DOI] [PMC free article] [PubMed] [Google Scholar]