Abstract
Peritoneal carcinosarcoma is a highly aggressive and uncommon neoplasm that has carcinomatous and sarcomatous components; the malignancy rarely localizes to the omentum. We report a case of a bulky peritoneal carcinosarcoma with tiny high-grade serous carcinoma of the fallopian tube. A 60-year-old female with a huge pelvic mass (12 cm in diameter) underwent a total abdominal hysterectomy, bilateral salpingo-oophorectomy, and omentectomy for tumor debulking. Pathological findings showed minimally invasive high-grade serous carcinoma of the left fallopian tube and carcinosarcoma of the omentum. Similar p53 diffuse immunostaining in the omental carcinosarcoma and the tubal carcinoma provides evidence for a clonal relationship between the two neoplasias. This case suggests a small serous carcinoma, originating in the tubal mucosa, subsequently became implanted in the omentum and grew preferentially, converting to a carcinosarcoma at a remote site.
Keywords: Peritoneal carcinosarcoma, High-grade serous carcinoma, Serous tubal intraepithelial carcinoma, Fallopian tube, p53, Case report, Conversion theory
Introduction
Peritoneal carcinosarcoma is a highly aggressive, relatively unusual neoplasm composed of carcinomatous and sarcomatous components [1–5] that rarely occurs in the omentum [2, 3]. Recent evidence suggests peritoneal carcinoma may be derived from a defined precursor known as serous tubal intraepithelial carcinoma (STIC) of the fimbria of fallopian tube [6–8]. Coexistence of a peritoneal carcinosarcoma with a minute serous carcinoma (STIC) in the fimbria of fallopian tube has been reported [8]. The occurrence of STIC in pelvic carcinosarcoma has also been described [9]. In this latter case, a similar p53 immunostaining pattern and identical TP53 mutations were detected in the pelvic carcinosarcomas and their associated STICs [9].
Here, we report a rare case of a bulky carcinosarcoma of the omentum and tiny high-grade serous carcinoma of the fallopian tube. Following debulking surgery for omental carcinosarcoma, the patient was treated with paclitaxel–carboplatin adjuvant chemotherapy. We discuss the histogenesis of peritoneal carcinosarcoma, as the tumor may originate from STIC of the fallopian tube.
Case presentation
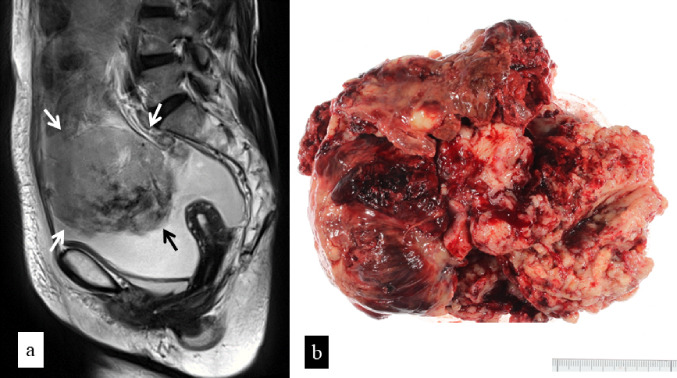
A 60-year-old woman (gravida 2, para 2) presented with a chief complaint of abdominal distension that had persisted for a month. Her past history was unremarkable, with no family history of malignant neoplasms. The serum cancer antigen 125 (CA125) level was 841.7 U/mL (normal, < 35 U/mL). Magnetic resonance imaging (MRI) revealed various sized masses (the largest measuring 12 cm) in the abdominal cavity, showing low and high signal intensities on T2-weighted imaging (Fig. 1a). Preoperative cytology of ascites was positive for malignancy. Surgery was planned according to the National Comprehensive Cancer Network (NCCN) guidelines for ovarian cancer [10]. Abdominal exploration revealed a bulky, cauliflower-like omental mass. The patient underwent a total hysterectomy, bilateral salpingo-oophorectomy, and omentectomy for tumor debulking. Tumors measuring ≥ 2 cm were left on the rectal serosal surface and Douglas’ pouch.
Fig. 1.
a Sagittal view of T2-weighted magnetic resonance imaging (MRI) image demonstrating a large solid mass with low- and high-signal intensity in the abdominal cavity (arrows). b Macroscopic view of the resected tumor from the omentum showing a cauliflower-like mass with marked hemorrhage
Grossly, the omental tumor (17 × 3 cm in size, 520 g) was grayish-white, solid, and friable with hemorrhagic necrosis (Fig. 1b). No visible tumors were detected on the uterine serosal surface and bilateral adnexa. Bilateral ovaries were not enlarged. A small tumor (1 cm) was found in the endometrium of the uterine corpus adjacent to a leiomyoma in the uterine myometrium. Histologically, the omental tumor was predominantly composed of spindle-shaped tumor cells, with a lesser amount of epithelial tumor component (Fig. 2a) that displayed an irregular tubular structure with markedly enlarged and pseudostratified nuclei, indicating adenocarcinoma (Fig. 2b). The spindle-cell component was arranged in interlacing bundles as features of leiomyosarcoma or fibrosarcoma with none of the heterologous elements that are associated with chondrosarcoma, osteosarcoma, or rhabdomyosarcoma (Fig. 2c). Immunohistochemically, the epithelial component was positive for cytokeratin AE1/AE3 and negative for vimentin (Fig. 3a), while the spindle-cell component showed a reciprocal staining pattern with the two antibodies (Fig. 3b). In addition, the epithelial component was CA125-positive (Fig. 3c) and BerEP4 and the spindle-cell component was positive for α-smooth muscle actin (Fig. 3d). Both components were negative for calretinin, Wilms’ tumor (WT)-1, PAX8, estrogen receptor (ER), hepatocyte nuclear factor-1β, MUC2, MUC6, napsin A, NSE or S-100, but were diffusely positive for p53 (Fig. 3e). The case was diagnosed as carcinosarcoma of homologous type. Even though WT-1 and PAX8 were negative, we considered morphologically as the epithelial component of carcinosarcoma to be suggestive of serous feature.
Fig. 2.
a Histologically, the tumor in the omentum consisted of a glandular tumor component located in the center and a spindle-cell tumor component surrounding the glandular component [hematoxylin–eosin (HE), original magnification ×40). b The carcinomatous component was composed of irregular glands of columnar cells with markedly enlarged nuclei (HE, ×400). c Sarcomatous component depicting spindle-cells in bundles (HE, ×400)
Fig. 3.
a AE1/AE3 immunopositivity was observed in the carcinomatous component, but not in the sarcomatous component (immunoperoxidase, ×40). b Vimentin reveals a reciprocal staining pattern against AE1/AE3 (immunoperoxidase, ×40). c CA125 immunoreactivity in the carcinomatous component (immunoperoxidase, ×200). d The spindle-cell component stained positive for α-smooth muscle actin (immunoperoxidase, ×200). e The carcinomatous and sarcomatous components were diffusely positive for p53 staining (upper half: carcinomatous component, lower half: sarcomatous component; immunoperoxidase, ×200)
We extensively sectioned and histologically examined both fallopian tubes and fimbriae, and subjected them to histological analysis. We found a 5-mm tumor in the fimbriae of the left fallopian tube that consisted of a low-papillary structure of secretory-type cells, located mainly in the epithelium (corresponding to STIC [6–8]) while slightly infiltrating the subepithelium (Fig. 4a). The tumor displayed nuclear enlargement, hyperchromasia, and a loss of nuclear polarity (Fig. 4b) and was diffusely positive for WT-1 (Fig. 4c), CA125, BerEP4 and p53 (Fig. 4d). The Ki-67 labeling index ranged from 30 to 40% (Fig. 4e). The morphological and immunohistochemical findings were consistent with a minimally invasive high-grade serous carcinoma of the left fallopian tube (“slightly advanced” STIC). Small implantation foci of serous carcinoma, without sarcomatous components, were detected around both adnexa, on the serosal surface of the uterine body, and in the endometrium. There was no evidence of stromal invasion of serous carcinoma in either ovary. The final diagnosis was carcinosarcoma in the omentum coexisting with “slightly advanced” STIC of the left fallopian tube, with implants to the uterus and adnexal surface (stage IIIC).
Fig. 4.
a Microscopic picture of a tumor in a fimbria of the fallopian tube: Left two-thirds of the area shows a serous intraepithelial carcinoma and the right one-third of the area shows a minimally invasive serous carcinoma [hematoxylin–eosin (HE), original magnification ×40). b A papillary carcinoma with enlarged and pseudostratified nuclei in the fallopian tube (HE, ×400). c, d, e The tumor was positive for WT-1 (c; immunoperoxidase, ×400), and p53 (d; immunoperoxidase, ×400) and high Ki-67 labeling (e; immunoperoxidase, ×400)
As postoperative adjuvant chemotherapy, six cycles of paclitaxel–carboplatin (paclitaxel, 175 mg/m2; carboplatin, mg in a dose equivalent to an area under the curve of 6) were administered. CA125 decreased to within the normal range, and a complete response (CR) was confirmed by abdominal imaging after adjuvant chemotherapy. However, 5 months after achieving a CR, the cancer recurred as a pelvic mass with massive ascites. Soon after, gemcitabine (1000 mg/m2) was started, but the patient unfortunately died a month after recurrence (11 months after surgery).
Discussion
Peritoneal carcinosarcoma is highly aggressive with a poor prognosis. Rajanbabu et al. [2] classified peritoneal carcinosarcoma cases into three groups based on tumor site: pelvic peritoneum (14 cases), peritoneum of the colorectal serosa (11 cases), and peritoneum of other sites, including omentum (11 cases). Patients in the first, second, and third groups survived on average for 21.5, 7.6, and 4.3 months, respectively. In a case report, patient with peritoneal carcinosarcoma (stage IIIc) was treated with cytoreductive surgery following adjuvant chemotherapy, including paclitaxel, carboplatin and bevacizumab, with lasting remission for 24 months [5]. We performed paclitaxel–carboplatin adjuvant chemotherapy followed by debulking surgery for carcinosarcoma; the patient unfortunately died 11 months after surgery.
Theoretically, the relationship between the two malignancies in this case could be explained in several ways. For example, the patient could have had a simultaneous double cancer, such as primary peritoneal carcinosarcoma and high-grade serous carcinoma of the fallopian tube. Alternatively, a small serous carcinoma that originated in the tubal mucosa may have subsequently implanted into the omentum, where it underwent conversion to a carcinosarcoma at a remote site. STIC occurs in the fallopian tubes of nearly half of patients with peritoneal serous carcinoma [6–8]. Accordingly, peritoneal and tubal carcinomas may be considered a disease entity of a pelvic high-grade serous carcinoma [8]. Huang and colleagues [5] reported a large (10 cm) peritoneal carcinosarcoma coexisting with a minute (1 mm) STIC in the fimbriated end of a fallopian tube. In a report by Ardighieri et al. [9], STIC was found in 10 of 16 patients (63%) with pelvic carcinosarcoma. We found a tiny (5 mm) tumor of the fallopian tube with a low-papillary structure of secretory-type pleomorphic cells limited mostly to the epithelium (corresponding to STIC [6–8]) with minimal invasion into subepithelium. The tumor was immunopositive for WT-1 and CA125, had diffuse and strong p53 nuclear staining, and a high Ki67 labeling index (> 10%), consistent with STIC. We, therefore, diagnosed a “slightly advanced” STIC of the fallopian tube.
The histogenetic hypotheses of carcinosarcoma has been proposed as collision, combination and conversion theories [10]; the latter two are supported by molecular genetic studies in most gynecological carcinosarcomas [11, 12]. Fujii et al. [11] analyzed allelic status using 41 microsatellite markers in 172 carcinomatous or sarcomatous foci microdissected from 17 gynecological carcinosarcomas: almost all materials shared allelic losses and retentions, highlighting the monoclonal origin of gynecological carcinosarcomas in line with combination or conversion theories.
In the present case, both the peritoneal carcinosarcoma and “slightly advanced” STIC demonstrated concordant diffuse p53 immunoreactivity. The occurrence of similar p53 immunostaining patterns and identical TP53 mutation status has been reported for both STIC and pelvic carcinosarcoma [9], suggesting their clonal relationship. Consistent with this, one of the tumor sites is metastatic, rather than a synchronous independently developed malignancy. Hence, we consider that our case originally developed as STIC (which arose after p53 alteration) that originated in the tubal mucosa and was implanted in omentum. This implanted tumor subsequently converted to carcinosarcoma (but retained the p53 alteration) in the omentum, in line with conversion theory.
In summary, we encountered an extremely rare case of a bulky carcinosarcoma in the omentum and “slightly advanced” STIC of the fallopian tube. The similar p53 staining pattern that we observed in omental carcinosarcoma and tubal carcinoma provides evidence for the monoclonality of these tumors.
Acknowledgements
The authors thank Masaru Isozaki, Kaoru Horii and Sayuri Miyazaki for their histological assistance.
Author contributions
AN: provided the concepts for the discussion and contributed to the finalization of the manuscript. YM: contributed to the original draft preparation. HS and TH: provided the study design and supervised the study. HM: contributed to pathological examination and review of the manuscript.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
For this type of study, formal consent is not required.
Informed consent
Informed consent was obtained from the patient included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Terada T. Carcinosarcoma in the pouch of Douglas. Arch Gynecol Obstet. 2010;281:345–348. doi: 10.1007/s00404-009-1132-y. [DOI] [PubMed] [Google Scholar]
- 2.Rajanbabu A, Zahoor Ahmad S, Vijaykumar DK, et al. The significance of the site of origin in primary peritoneal carcinosarcoma: case report and literature review. Ecancermedicalscience. 2013;7:295. doi: 10.3332/ecancer.2013.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang B, Ren KW, Yang YC, et al. Carcinosarcoma of the lesser omentum: a unique case report and literature review. Medicine (Baltimore) 2016;95:e3246. doi: 10.1097/MD.0000000000003246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Koyanagi T, To Y, Ando M, et al. Primary peritoneal carcinosarcoma arising from the Douglas pouch: a case report. MolClin Oncol. 2018;9:485–488. doi: 10.3892/mco.2018.1711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang C-C, Tsai H-J, Huang SH, Kok VC. A successfully treated primary peritoneal carcinosarcoma and serous carcinoma of stage IIIC rescued from hypovolemic shock due to tumor rupture. Taiwan J Obstet Gynecol. 2019;58:296–297. doi: 10.1016/j.tjog.2019.01.023. [DOI] [PubMed] [Google Scholar]
- 6.Carlson JW, Miron A, Jarboe EA, et al. Serous tubal intraepithelial carcinoma: its potential role in primary peritoneal serous carcinoma and serous cancer prevention. J Clin Oncol. 2008;26:4160–4165. doi: 10.1200/JCO.2008.16.4814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Seidman JD, Zhao P, Yemelyanova A. "Primary peritoneal" high-grade serous carcinoma is very likely metastatic from serous tubal intraepithelial carcinoma: assessing the new paradigm of ovarian and pelvic serous carcinogenesis and its implications for screening for ovarian cancer. Gynecol Oncol. 2011;120:470–473. doi: 10.1016/j.ygyno.2010.11.020. [DOI] [PubMed] [Google Scholar]
- 8.Kar T, Kar A, Dhal I, et al. Serous tubal carcinogenesis: the recent concept of origin of ovarian, primary peritoneal and fallopian tube high-grade serous carcinoma. J Obstet Gynecol India. 2017;67:432–441. doi: 10.1007/s13224-017-1009-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ardighieri L, More L, Conzadori S, et al. Identical TP53 mutations in pelvic carcinosarcomas and associated serous tubal intraepithelial carcinomas provide evidence of their clonal relationship. Virchows Arch. 2016;469:61–69. doi: 10.1007/s00428-016-1933-x. [DOI] [PubMed] [Google Scholar]
- 10.Pang A, Carbini M, Moreira AL, et al. Carcinosarcomas and related cancers: tumors caught in the act of epithelial-mesenchymal transition. J Clin Oncol. 2018;36:210–216. doi: 10.1200/JCO.2017.74.9523. [DOI] [PubMed] [Google Scholar]
- 11.Fujii H, Yoshida M, Gong ZX, et al. Frequent genetic heterogeneity in the clonal evolution of gynecological carcinosarcoma and its influence on phenotypic diversity. Cancer Res. 2000;60:114–120. [PubMed] [Google Scholar]
- 12.Jin Z, Ogata S, Tamura G, et al. Carcinosarcomas (malignant mullerian mixed tumors) of the uterus and ovary: a genetic study with special reference to histogenesis. Int J GynecolPathol. 2003;22:368–373. doi: 10.1097/01.pgp.0000092134.88121.56. [DOI] [PubMed] [Google Scholar]