Abstract
Objective
The aim of this study was to determine the causes leading to a first revision of primary total knee arthroplasty (TKA) in a specialized knee centre and compare the results with previously published data.
Methods
Prospectively collected data of a consecutive number of 195 patients after primary TKA and who underwent first revision surgery after completing the diagnostic algorithm for persistent knee pain were included. Data was prospectively collected from a specialized knee centre in which the patients presented between 2015 and 2020 and retrospectively analysed. Indications for revision surgery were categorized using all available information from patients’ records. Patients were divided into early (up to two years) and late revision (more than two years).
Results
Overall mean time from index to revision surgery was 3.6 years. 49% of knee revisions occurred in the first two years, 51% after two years. 86% of the patients were referred to the knee centre from other surgeons. The most frequent reason for revision was instability, followed by patellofemoral problems, extensor mechanism insufficiency and malalignment. The most frequently performed revision was complete removal and re-implantation of a semi constrained implant design (52.5%) followed by revision using a full constrained implant design (16%). Secondary patella-resurfacing as part of complete revision was carried out in 71.5% of the cases. The majority of the patients showed concurrent reasons for TKA failure with significant correlations amongst another. Furthermore, correlations were identified between indications for revision surgery and revision implant designs.
Conclusion
In a specialized knee centre the most common indications for the first TKA revision were instability and patellofemoral and/or extensor mechanism insufficiency followed by malalignment. In most patients there was not only one failure mode, but a combination of many. It is important to establish a standardized diagnostic algorithm to facilitate comprehensive and efficient diagnostics and the optimal treatment.
Keywords: Total knee arthroplasty, Failure, Indication, Revision, Implants, Total knee replacement, Switzerland
1. Introduction
Total knee arthroplasty (TKA) is the treatment of choice for patients with an advanced osteoarthritis (OA) of the knee. Most patients after TKA recover well and experience pain relief within 3–6 months.1 Nevertheless, about 10–30% of the patients report ongoing or recurrent pain or are not satisfied and require revision surgery.2 The number of revision TKA is rising in many countries, with 10′507 in the United States,3 6′357 in United Kingdom,4 25′567 in Australia,5 13′378 in Germany6 and 2′284 in Switzerland7 in 2018. National registries report revision rates between 3 and 12%, depending on the length of the observed period and included interventions.3,5,7
Previous studies have analysed failure modes after primary TKA. According to those results, the most frequent causes of failure are infection, loosening, instability, arthrofibrosis, and osteolysis.8, 9, 10, 11, 12, 13, 14 Some authors differentiated between early (within the first two years after primary TKA) and late (thereafter) revisions and found polyethylene wear and accordingly aseptic loosening as most common failure mode for late revisions.11,15 Fehring et al. reported that infection and instability were the most common indications for revision in the early failure group.9 However, modes of failure have changed over the last decade. Whereas revisions due to polyethylene wear and subsequent osteolysis became less prevalent as a result of improved polyethylene quality and manufacturing, infection rates on the contrary were increasing.8,11,16, 17, 18, 19
Most data on the causes of TKA failure and further revision surgery are obtained from national joint registries, health care providers and multicentre studies.9,11,14,20 These data are not very specific and provided from many different persons who might have different judgments for assessing the revision causes. Neither was a thorough and standardized diagnostic workup, including a detailed patient history, clinical examination, radiological, serological and microbiological investigations, carried out systematically in the majority of the patients. This impedes the careful identification or exclusion of all possible failure modes and thus select the right strategy for the revision surgery. In particular, many of these studies report only one main reason which indicated revision surgery and fail to illustrate a more detailed picture of the revision causes.
The aim of this study was to assess the most common reasons for failure of primary TKA by reporting the indications for first TKA revision surgery in a large single-centre series. It was hypothesised that the modes of failure differ from previously published data, especially regarding the number of concurrent reasons for failure in one patient.
2. Methods
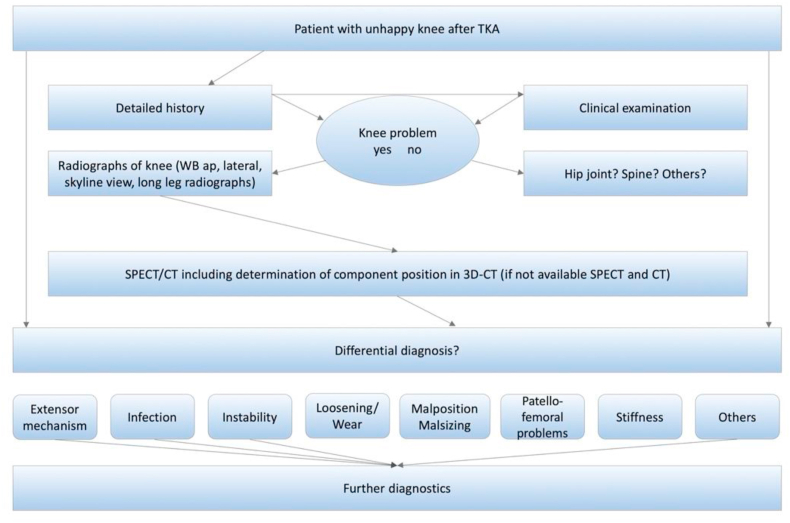
A prospectively collected consecutive number of 195 patients after primary TKA (27% (N = 54) primary patellar resurfacing) who complained about persistent knee pain and subsequently underwent first revision surgery were included in this retrospective cohort study. The indication for primary TKA was in 86% (N = 172) primary OA and in 14% (N = 28) secondary OA. Data was prospectively collected from a specialized knee centre in which the patients presented between 2015 and 2020 due to persistent pain after primary TKA. All patients followed a previously published, standardized diagnostic algorithm (Fig. 1)21 including a detailed patient history and clinical examination, blood tests, joint aspiration in the operating room (OR), intraoperative findings, culture and histology results, and various imaging modalities depending on the evaluated failure mode(s) (Table 1). The latter include standardized (anterior-posterior and lateral weight bearing, patellar skyline view) and stress radiographs, conventional computerised tomography (CT), magnetic resonance imaging (MRI) and combined single-photon emission computed tomography/computed tomography using technetium-99 m hydroxymethylene diphosphonate (99m-Tc-HDP-SPECT/CT). At the end of this standardized clarification procedure, the revision reason(s) was/were determined for all patients by one expert knee surgeon (senior orthopaedic consultant, MTH) for each patient in a standardized manner and documented in the consultation and surgery report. The failure categories were aseptic loosening, periprosthetic infection, instability, polyethylene wear/osteolysis, arthrofibrosis/stiffness, malalignment, patellofemoral and/or extensor mechanism insufficiency, periprosthetic fracture, irritation of iliotibial band (ITB) and others. In case of more than one causes for revision all causes were reported. A member of the study group (L.L.) reviewed all available reports and collected the aforementioned revision reasons and revision interventions performed.
Fig. 1.
The “Bruderholz” standardized diagnostic algorithm for patients with pain after total knee arthroplasty. WB, weight bearing; SPECT/CT, single photon emission computed tomography/computer tomography.
Table 1.
Revision causes and time to first revision. SD, standard deviation. OA, osteoarthritis. PE, polyethylene. PF, patellofemoral. Sy, syndrome. PCL, posterior cruciate ligament.
|
Failure mode |
Overall N (%) N = 200 |
Time to revision (yrs), mean ± SD (range) | Time to Failurea |
|
|---|---|---|---|---|
| Early N (%) N = 98 |
Late N (%) N = 102 |
|||
| Aseptic loosening | 28 (14) | 3.5 ± 3.3 (0.5–17.4) | 12 (12.2) | 16 (15.7) |
| femoral | 9 (4.5) | 3.6 ± 2.3 (0.5–8) | 2 (2) | 7 (6.9) |
| tibial | 25 (12.5) | 3.7 ± 3.4 (1.1–17.4) | 11 (11.2) | 14 (13.7) |
| Periprosthetic infection | 9 (4.5) | 6.9 ± 8.2 (0.6–24.2) | 4 (4.1) | 5 (4.9) |
| High grade | 6 (3) | 9.2 ± 9.3 (0.6–24.2) | 2 (2) | 4 (3.9) |
| Low grade | 3 (1.5) | 2.2 ± 2 (1–4.5) | 2 (2) | 1 (1) |
| Instability (I.) | 116 (58) | 3.3 ± 3.3 (0.7–16.5) | 58 (59.2) | 58 (56.9) |
| Multidirectional I. | 16 (8) | 2.9 ± 1.9 (0.7–7.3) | 7 (7.1) | 9 (8.8) |
| Anteroposterior I. | 30 (15) | 2.7 ± 2.2 (0.7–9.8) | 15 (15.3) | 15 (14.7) |
| Medial I. | 23 (11.5) | 3.2 ± 3.0 (0.8–12.9) | 11 (11.2) | 12 (11.8) |
| Lateral I. | 51 (25.5) | 3.4 ± 3.7 (0.7–16.5) | 25 (25.5) | 26 (25.5) |
| Posterior I. | 19 (9.5) | 3.1 ± 3.3 (1.1–15.4) | 7 (7.1) | 12 (11.8) |
| Flexion I. | 57 (28.5) | 3.1 ± 3.0 (0.8–16.5) | 30 (30.6) | 27 (26.5) |
| Extension I. | 10 (5) | 5.2 ± 4.8 (0.9–15.7) | 2 (2) | 8 (7.8) |
| Posterior dislocation | 1 (0.5) | 15.4 | 0 (0) | 1 (1) |
| Polyethylene wear/osteolysis | 4 (2) | 13.6 ± 8.2 (2.5–21.1) | 0 (0) | 4 (3.9) |
| Arthrofibrosis/stiffness | 28 (14) | 2.3 ± 2.1 (0.3–9.6) | 19 (19.4) | 9 (8.8) |
| Malalignment | 53 (26.5) | 3 ± 2.9 (0.3–13.9) | 29 (29.6) | 24 (23.5) |
| Patellofemoral/extensor mechanism insufficiency | 100 (50) | 3 ± 2.6 (0.3–15.7) | 48 (49) | 52 (51) |
| Progression OA patella | 13 (6.5) | 3.3 ± 2.9 (0.8–11.6) | 5 (5.1) | 8 (7.8) |
| Patella baja | 24 (12) | 2.8 ± 3.1 (0.3–15.7) | 15 (15.3) | 9 (8.8) |
| Patella maltracking | 5 (2.5) | 1.8 ± 1.1 (0.3–2.8) | 2 (2) | 3 (2.9) |
| Patella dislocation | 1 (0.5) | 10 | 0 (0) | 1 (1) |
| Insufficiency/rupture tendon (patellar/quadriceps) | 4 (2) | 3.1 ± 2.5 (0.8–6.6) | 1 (1) | 3 (2.9) |
| PE dislocation patella | 1 (0.5) | 5 | 0 (0) | 0 (0) |
| PF overstuffing | 72 (36) | 3.6 ± 2.8 (1.4–11.1) | 35 (35.7) | 37 (36) |
| Periprosthetic fracture | 6 (3) | 5.2 ± 6.1 (1.6–17.4) | 1 (1) | 5 (4.) |
| Irritation iliotibial band | 11 (5.5) | 3.2 ± 2.1 (1.2–7.6) | 4 (4.1) | 7 (6.9) |
| Unexplained pain | 6 (3) | 5.5 ± 5.7 (0.9–12.9) | 3 (3.1) | 3 (2.9) |
| Others | 11 (5.5) | 4.2 ± 5.4 (0–18.1) | 5 (5.1) | 6 (5.9) |
| Painful ossification | 2 (1) | 1.7 ± 0.2 (1.6–1.8) | 2 (2) | 0 (0) |
| Overloading Pes anserine | 1 (0.5) | 2.5 | 0 (0) | 1 (1) |
| Metallosis | 1 (0.5) | 18.1 | 0 (0) | 1 (1) |
| Infrapatellar branch of saphenous nerve sy. | 2 (1) | 1.5 ± 1.4 (0.6–2.5) | 1 (1) | 1 (1) |
| Calcinosis cutis | 1 (0.5) | 9.6 | 0 (0) | 1 (1) |
| Rupture PCL | 1 (0.5) | 1.1 | 1 (1) | 0 (0) |
| End of stem pain | 1 (0.5) | 2.2 | 0 (0) | 1 (1) |
| Intercondylar cement | 1 (0.5) | 0 | 1 (1) | 0 (0) |
Early failure was defined as failures that occurred two years and less after primary TKA, late failure thereafter.
Revision was defined according to the Swiss National Registry: “A revision procedure is a secondary surgical procedure of a patient's knee joint whereby the complete primary implant or parts thereof are replaced by new components”.7 Secondary patella-resurfacing due to progression of OA was also considered as revision procedure. Only first revisions after primary TKA were included. Patients, who have suffered a trauma, underwent revision surgery in other hospitals between primary TKA and presentation at our knee centre were excluded from this study (N = 19).
The study was approved by the local ethical committee (2017–02048) and was performed in accordance with the ethical standards of the responsible committee and with the guidelines of the Helsinki Declaration of 1975, as revised in 2008. A written informed consent was signed by every patient.
Aseptic loosening is described as the failure of the integration between a prosthesis and bone in the absence of infection. Loosening reported in the first few years of implantation of a prosthesis is most likely due to failure of the implant to gain fixation, however, loosening in later years likely reflects loss of fixation due to bone resorption.22 A recent meta-analysis suggested that SPECT/CT is the most diagnostically accurate modality for the detection of aseptic loosening in TKA.22 Therefore, the activity of bone tracer uptake (BTU) in SPECT/CT was evaluated in this study to diagnose aseptic loosening.23 All patients with suspicious component loosening underwent 99m-Tc-HDP-SPECT/CT imaging following a standardized protocol.23, 24, 25, 26 These patients received a commercial 700 MBq (18.92 mCi) 99m-Tc-HDP injection (Malinckrodt, Wollerau, Switzerland). SPECT/CT was performed using a hybrid system (Symbia T16, Siemens, Erlangen, Germany), which consists of a pair of low-energy, high-resolution collimators and a dual-head gamma camera with an integrated 16-slice CT scanner (collimation of 16 × 0.75 mm). Planar scintigraphic images were taken in the perfusion phase (immediately after injection), the soft tissue phase (1–5 min after injection) and the delayed metabolic phase (2 h after injection). SPECT/CT was performed with a matrix size of 128 × 128, an angle step of 32, and a time per frame of 25 s 2 h after injection. BTU activity was volumetrically measured in 3D in various anatomical areas according to a previously validated localization scheme.23
Periprosthetic infection was diagnosed based on the 2018 definition of periprosthetic hip and knee infection by the Infectious Diseases Society (IDSA).27 A revision involving two stages or more because of infection was considered as two events.
The failure mode instability was assessed by positive clinical history, physical examination and suitable radiological imaging. Clinical history includes recurrent knee swelling, a sense of knee instability with or without giving-way, in particular instability that was worse on descending stairs and pain in general. Then, an objective physical examination was performed by the expert knee surgeon (MTH) that included the observation of the gait (including varus-thrusting), the evaluation of the limb alignment in a standing position and testing of varus-valgus and anteroposterior stability at full extension and 30°/90° flexion, and the drawer test. Radiological assessment involved stress radiographs in the anterior–posterior projection with full extension and 30° flexion for assessment of varus and valgus laxity as well as in lateral projection in 15° and 90° flexion to assess for anterior and posterior laxity using a Telos stress device with 15 N force. In addition, for the morphological assessment of the ligaments and surrounding soft tissue, MRI was performed in selected cases.
Polyethylene wear/osteolysis was assessed by macroscopic findings on the insert and microscopic report, according to type I of the classification system describe by Krenn et al.28
Arthrofibrosis/stiffness was defined as a limited range of motion (ROM) in flexion and/or extension, that is not attributable to an osseous or prosthetic block to movement from malaligned, malpositioned or incorrectly sized components, metal hardware, ligament reconstruction, infection, pain, chronic regional pain syndrome (CRPS) or other specific causes, but due to soft-tissue fibrosis that was not present pre-operatively.29
Malalignment and malpositioning of the TKA components were assessed radiologically with SPECT/CT. All patients with suspicious component positioning or alignment underwent 99m-Tc-HDP-SPECT/CT imaging following a standardized protocol.23, 24, 25 Mechanical alignment and TKA position were assessed using a customized validated 3D-software.23, 24, 25 The BTU values and measurements of the positioning of the TKA components was interpreted in accordance with the findings published by Awengen et al., in 2016.30
Patellofemoral problems and extensor mechanism insufficiency include progression of patella OA, patellar dislocation or instability, maltracking, lateral patellar facet impingement, patella baja or alta, patella clunk syndrome, patellar component wear and loosening, tendon rupture or extensor insufficiency, patellofemoral overstuffing and avascular necrosis.31 The diagnosis was based on patient's history, detailed clinical examination of the extensor mechanism and patellofemoral joint and imaging diagnostics (e.g. conventional radiographs including tangential radiographs of the patella, ultrasound, MRI,CT and SPECT/CT).
Periprosthetic fractures were assessed by conventional radiographs or computerised tomography.
Revision involving isolated secondary resurfacing of the patella was only categorized as a failure due to progression of OA, if the patient reported retropatellar pain, especially in flexion, with pathological findings on a series of tangential radiographs of the patella. Revisions were categorized into an early and a late failure group. An interval of two years between primary TKA and first revision was considered as cut-off between the early and late failure modes.11
2.1. Statistical analysis
Results are presented as mean, standard deviation (SD) and range or number and percentage. To compare group differences, t-tests for independent samples or Chi-square tests were calculated. To test for relationships between the variables, Pearson-correlations (which are identical to phi when calculated between two dichotomous variables) are used. Two-sided p values < 0.05 were considered significant. All data were analysed by an independent professional statistician using IBM SPSS Statistics for Windows, version 26.0 (Armonk, NY: IBM Corp, USA).
3. Results
This study group consisted of 200 knees in 195 patients who had first revision TKA (right: left 49.5% (N = 99): 50.5% (N = 101)). The mean age of the total cohort at the time of revision was 66.3 years (standard deviation (SD) ± 10.7, range 24–87). Men comprised 39% (N = 76) of the knee failures and women 61% (N = 119). The mean of the patients had mild to moderate systemic diseases (ASA grade II; mean, SD 2.4 ± 0.6, range grade I-IV) and a body mass index of 29.3 kg/m2 (SD ± 5.5, range 18–50). Overall mean time from index to revision surgery was 3.6 years (SD ± 3.9, range 0.2–24.2). 49% (N = 98) of knee revisions occurred in the first two years, 51% (N = 102) after 2 years. Due to the set-up in a specialized knee centre with focus on painful TKA most of the included patients were referred from other surgeons to our clinic (86%). Therefore, a total of 87 different surgeons have performed primary arthroplasty.
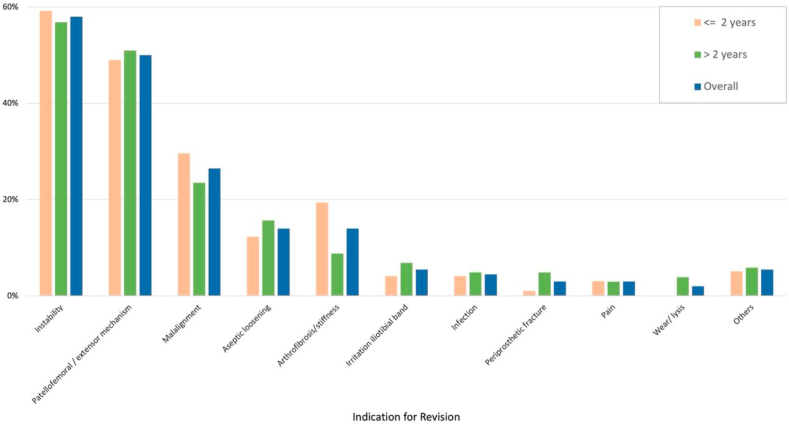
The most frequent reason for revision was instability (overall 58% (N = 116), early revisions 59.2% (N = 58), late revisions 56.9% (N = 58)) followed by patellofemoral and/or extensor mechanism insufficiency (overall 50% (N = 100), early revisions 49% (N = 48), late revisions 51% (N = 52)), see Table 1 and Fig. 2 for details. The majority of the patients (59%) showed several causes for failure, only 41% presented one underlying failure mode. Although the early and late failure groups were very equally balanced (N = 98 and 102) significant differences between the two groups were not found, with the exception of wear/osteolysis and arthrofibrosis/stiffness. Wear/osteolysis was the more common indication for late revisions and arthrofibrosis/stiffness for early revisions (p < 0.05).
Fig. 2.
The percentages of patients with each revision indication stratified into early, late an overall subgroup are shown.
The most frequently performed revision intervention was complete removal and re-implantation of a semi constrained implant design (52.5%, N = 105), followed by revision with re-implantation of a full constrained implant design (16%, N = 32); the revision was limited to the isolated exchange of the femoral, tibial, patellar or insert component in 1% (N = 2), 1% (N = 2), 2% (N = 4) and 5.5% (N = 11) of the cases. In six patients (3%) the components were removed and replaced by a posterior-stabilized TKA. Secondary patellar resurfacing due to OA progression of the patella was performed as isolated intervention in 18% (N = 36) and as part of complete revision in 71.5% (N = 143) of the cases; the patella was left unresurfaced during revision in only 2% (N = 4). In two patients (1%) a two-stage revision using a cement spacer was performed. All revisions were carried out by one senior revision TKA surgeon (MTH).
Table 3 demonstrates correlations amongst various failure modes and between failure modes and surgical interventions. The most outstanding ones include: A positive correlation between patellofemoral/extensor mechanism insufficiency and malalignment (p < 0.01) and wear/osteolysis and “unexplained” pain (p < 0.01) was found. On the contrary, patellofemoral problems and/or extensor mechanism insufficiency shows negative correlations with instability (p < 0.001), infection (p < 0.01) and aseptic loosening (p < 0.05). A rather intuitive finding was that secondary patellar resurfacing as isolated intervention was only carried out when the patients' complaints were attributed to patellofemoral and/or extensor mechanism insufficiency and significantly less when other causes were present.
Table 3.
Pearson correlation amongst various failure modes and surgical interventions. Significant correlations in bold when positive and in italic when negative, *p < 0.05, **p < 0.01, ***p < 0.001. ITB, iliotibial band; TKA, total knee arthroplasty; PS, posterior stabilized.
| Pearson | Instability | PF/extensor mechanism insufficiency | Malalignment | Infection | Aseptic loosening | Wear/osteolysis | Arthro-fibrosis/stiffness | Fracture | Irritation ITB | Pain | Others |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Instability | 1*** | −0.34*** | −0.06 | −0.21** | 0.05 | −0.1 | −0.1 | −0.15* | 0.07 | −0.09 | −0.11 |
| PF/extensor mechanism insufficiency | −0.34*** | 1*** | 0.22** | −0.22** | −0.14* | −0.07 | 0 | 0 | −0.11 | 0 | −0.02 |
| Malalignment | −0.06 | 0.22** | 1*** | −0.08 | −0.05 | −0.09 | 0.02 | −0.04 | −0.05 | 0.03 | −0.05 |
| Infection | −0.21** | −0.22** | −0.08 | 1*** | −0.02 | −0.03 | −0.02 | −0.04 | −0.05 | −0.04 | −0.05 |
| Aseptic loosening | 0.05 | −0.14* | −0.05 | −0.02 | 1*** | 0.05 | −0.08 | 0.01 | −0.1 | 0.01 | −0.03 |
| Wear/osteolysis | −0.1 | −0.07 | −0.09 | −0.03 | 0.05 | 1*** | −0.06 | −0.03 | −0.03 | 0.18** | 0.12 |
| Arthrofibrosis/stiffness | −0.1 | 0 | 0.02 | −0.02 | −0.08 | −0.06 | 1*** | −0.07 | −0.1 | 0.1 | 0.03 |
| Fracture | −0.15* | 0 | −0.04 | −0.04 | 0.01 | −0.03 | −0.07 | 1*** | −0.04 | −0.03 | −0.04 |
| Irritation ITB | 0.07 | −0.11 | −0.05 | −0.05 | −0.1 | −0.03 | −0.1 | −0.04 | 1*** | −0.04 | 0.13 |
| Pain | −0.09 | 0 | 0.03 | −0.04 | 0.01 | 0.18** | 0.1 | −0.03 | −0.04 | 1*** | −0.04 |
| Others | −0.11 | −0.02 | −0.05 | −0.05 | −0.03 | 0.12 | 0.03 | −0.04 | 0.13 | −0.04 | 1*** |
| Primary patella resurf. | 0.11 | −0.32*** | −0.01 | 0.3*** | −0.08 | −0.01 | 0.01 | 0.09 | −0.05 | −0.11 | 0.1 |
| Secondary patella resurf. | −0.13 | 0.32*** | 0.03 | −0.29*** | 0.13 | 0.01 | 0 | −0.08 | 0.06 | 0.11 | −0.09 |
| Semi-constrained TKA design | 0.33*** | −0.09 | 0.23*** | −0.18* | 0.27*** | −0.15* | −0.02 | −0.13 | 0.01 | −0.01 | −0.08 |
| Fully-constrained TKA design | 0.21** | −0.19** | −0.05 | 0.04 | −0.06 | 0.23*** | 0.06 | −0.08 | 0.07 | 0.08 | 0.07 |
| PS TKA design | −0.03 | −0.06 | −0.04 | 0.1 | 0.01 | −0.03 | −0.07 | −0.03 | −0.04 | −0.03 | 0.09 |
| Isolated tibial insert exchange | −0.02 | −0.11 | −0.1 | 0.16* | −0.1 | 0.12 | −0.03 | 0.09 | −0.06 | −0.04 | −0.06 |
| Isolated secondary patella resurf. | −0.5*** | 0.39*** | −0.16* | −0.1 | −0.19** | −0.07 | 0 | 0.07 | 0 | −0.01 | 0.06 |
4. Discussion
The most important findings and implications of this study were the following:
Firstly, the most common indications for TKA revision in this study cohort were instability and patellofemoral and/or extensor mechanism insufficiency. These findings are different to most published data (Table 2). This is not surprising since the majority of all revisions in this study (86%) were referred to a specialized knee centre after primary TKA performed elsewhere. Due to the standardized diagnostics following a validated algorithm for painful patients after TKA in the setting of a knee centre (Fig. 1), it is likely that the identified failure modes might differ from those based on registries and studies carried out in less specialized institutions. In 2002, Sharkey et al. found polyethylene wear (25%) as the most prevalent mechanism for TKA revision11 and 12 years later they identified aseptic loosening (39.9%) as most common failure mode.32 This shift of failure modes is most likely a result of improved polyethylene quality and manufacturing, better seating and anchorage mechanisms, and more highly polished surfaces.8,16, 17, 18,33 Thiele et al. reported aseptic loosening (21.8%), instability (21.8%) and malalignment (20.7%) as the most common indications for revision.19 In the Swiss National joint registry aseptic loosening (34.5%) followed by patellofemoral and/or extensor mechanism insufficiency (25.7%) and instability (15.9%) are the most frequent indications for TKA revision.7 The reason for the high frequency of related revisions due to patellofemoral problems and/or extensor mechanism insufficiency in our cohort but also generally in Switzerland might be due to the relatively low percentage of patients who undergo primary patellar resurfacing (26.5%).7 Subsequently, secondary patellar resurfacing has been performed in the vast majority of the patients in this cohort (71.5%). The discrepancy between this high number of secondary patellar resurfacing (71.5%) and the relatively few revision indications due to progression of patellar OA (6.5%) are explained by the fact, that with a revision TKA it is generally indicated to resurface the patella due to the less patella friendly design features of a revision TKA.34
Table 2.
Comparison of previous reports and the present study on failure modes (%) after TKA. In some authors the percentages totalled >100% because some knees had more than one mode of failure recorded.
| Ref. and publication year | Origin | N | Mean time to failure | Aseptic Loosening | Wear/osteolysis | Instability | Infection | Patello-femoral/extensor mechanisma | Mal-alignment |
|---|---|---|---|---|---|---|---|---|---|
| Fehring et al.,9 2001 | US | 279 | <5 yr | 3 | 7 | 26 | 38 | 8 | 12 |
| Sharkey et al.,11 2002 | US | 212 | NR (1.1–28 yr) | 17/34 | 12/44 | 21/22 | 25/7.8 | 10/2 | 12/12 |
| Schroer et al.,16 2013 | US | 844 | 5.9 yr (10d–31 yr) | 19/31 | 1/10 | 25/19 | 23/16 | 7/6 | 8/7 |
| Le et al.,35 2014 | US | 253 | 2.9 yr (SD ± 2.2) | 14/13 | 2/9 | 26/18 | 24/25 | 4 | 7 |
| Pitta el al,36 2018 | US | 405 | 2.1 yr (SD ± 1.7) | 21.2 | 2.5 | 24.4 | 25.7 | 3 | 2.5 |
| Postler et al.,45 2018 | GER | 402 | 6.2 yr (0.1–24.2 yr) | 21.6 | 5.2 | 6.7 | 36.3 | 3.7 | NR |
| Thiele et al.,19 2015 | GER | 358 | 1.5 yr/7.9 yr | 12.7/34.7 | 0/18.5 | 23.9/18.5 | 26.8/8.9 | 11.3/3.2 | 18.3/10.5 |
| Hossain et al.,10 2010 | UK | 349 | 7 yr (0.1–13,9) | 3/12 | 1/12 | 4/3 | 12/21 | 3/2 | 4/3 |
| National Registry,7 2019 | CH | 8340 | 1.6 yr (SD ± 1.17) | 34.5 | 5.8 | 15.9 | 17 | 25.7 | 10.4 |
| Present study | CH | 200 | 3.6 yr (0.2–24.2) | 14 | 2 | 58 | 4.5 | 50 | 26.5 |
Sharkey et al.,11 Hossain et al.10 and Le et al.35: First number is early (<2years) failures, second number is late failures; Schroer et al.16: First number is early (<2years) failures, second is overall failures. Thiele et al.19: First number is early (<1year), second is late ((≥3 years).
N, qualified for revision surgery. NR, not reported.
US, United States; UK, United Kingdom; GER, Germany; CH, Switzerland.
Includes patellar dislocation or instability, maltracking, progression of osteoarthritis, lateral patellar facet impingement, patella baja/alta, patella clunk syndrome, patellar component wear and loosening, tendon rupture or extensor insufficiency, patellofemoral overstuffing and avascular necrosis.
The high incidence of instability in this cohort (58%) was observed, albeit to a lesser extent, in other contemporary studies.16,19,35,36 While other common modes of failure such as aseptic loosening and prosthetic infection have been studied extensively, there is a lack of evidence in relation to instability as a mode of failure. A considerable number of definitions for knee instability exist in the current literature.37,38 An assessment of an unstable knee requires standardized and comprehensive diagnostics including clinical and radiological screenings to obtain the correct diagnosis.39, 40, 41, 42 Prerequisites, which in modern medicine, also as a result of technical advances, are much more fulfilled than in the past.
The same applies to malalignment, which was found in 26.5% of all patients. Our study group demonstrated in 2011 that 3D-CT is the most accurate method to determine TKA component position relating to the mechanical axis. However, most authors use axial 2D-CT slices for measurement of component position, although it has been shown that these measurements are very variable and less reliable.43,44 It is therefore not surprising that in this study cohort, in which 3D-CT is regularly used as a firm component of a standardized algorithm (Fig. 1) malalignment is diagnosed with increasing frequency. However, one has to differentiate symptomatic from asymptomatic malalignment. Only when the symptoms of the patients match the imaging findings it is a symptomatic malalignment.
Aseptic loosening and periprosthetic joint infection might be regarded as “simple” revisions treating with the exchange of the loose component and debridement, antibiotics, implant retention (DAIR), respectively. These cases might be treated in smaller hospitals whereas complex revisions for instability, patellofemoral and/or extensor mechanism insufficiency and malalignment were more frequently referred to our department.
Secondly, for the majority of the patients in this cohort more than one reason for failure was found. It appears to be common in literature, if failure was due to multiple reasons, only the dominant cause was mentioned.9,16,19,35,45,46 However, the few authors who have listed more than one cause of failure have not reported the percentages of multiple reasons.7,11 Only a very recent study of Postler et al. has published numbers on concurrent reasons for TKA failure in one patient. They found in 88.6% of all patients more than one reason indicating revision surgery.45 These results in combination with our findings underline the general opinion that TKA revision surgery is complex and challenging and require a lot of experience.47,48 It is therefore crucial to identify all possible failure modes in order to treat the patient adequately.
Thirdly, significant differences in terms of demographics, indications and performed revision procedures could not be found between the early and late failure group. The only exception constitutes wear/osteolysis with higher incidence for late revisions (3.9%) and arthrofibrosis/stiffness for early revisions (19.4%). This distribution is in accordance with previous findings of Postler et al., Thiele et al. and Sharkey et al.19,32,45 According to their findings wear/osteolysis accounts for 8.2%, 18.5% and 5% of late revisions and arthrofibrosis/stiffness for 9.2%, 7% and 10% of early revisions. There is a lack of consensus on the diagnostic criteria for arthrofibrosis of the knee, which obscures its true prevalence after surgical procedures.29 That might be an explanation why the incidence varies widely in literature (1%–13% postoperatively) including the findings of this study (9%–19%).49 Nevertheless, arthrofibrosis represents 28% of hospital readmissions due to surgical complications within 90 days of discharge, and constitutes a serious early complication after TKA.11,35,36,46
With regard to wear/osteolysis, however, the literature proves to be very clear and states this problem unsurpassed as a long-term complication with the incidence in most series beyond 10-years.50 Our findings with a mean time to revision of 13.6 ± 8.2 years are thus in line with the common tenet.
Fourthly, 105 of the 200 included first revisions were treated with complete removal and re-implantation of a semi constrained implant design (52.5%), followed by isolated secondary patellar resurfacing (18%, N = 36) and complete revision with re-implantation of a full constrained implant design (16%, N = 32). Previously published data reported lower frequencies for complete revisions (approx. 30–35%), but a higher proportion of isolated tibial insert exchange (approx. 14%) due to larger percentage of infection in their cohorts.7,51 The choice of implant design was based on thorough preoperative and intraoperative evaluation. The surgical goals of revision TKA are to obtain stable fixation of the prosthesis to host bone, to restore the pre-arthroplasty height of the joint line, to obtain a stable range of motion compatible with the patient's activities of daily living, and to achieve these goals while using the least degree of prosthetic constraint so that soft tissues may share in load transfer. Thus, the surgeon is faced with the question, “How much constraint is needed?”
However, our choice of implant can be compared with other findings in literature. Hossain et al. retrospectively reviewed 349 revision knee arthroplasties and found a similar distribution between semi (43%) and full constrained (21%) implant design.10
Finally, pain after TKA is usually the main reason for choosing a knee specialist. The causes for persistent or recurrent pain after TKA are manifold and sometimes difficult to identify. Nevertheless, surgeons should never fail to make every effort to find the cause of the pain. It is the common sense, that revision operations should only be performed if the cause(s) of the complaints described have been identified and fit the clinical picture, as revision surgery for unexplained pain has consistently been shown to result in poor outcomes.47,48,52,53 Thus, the diagnosis “unexplained pain” should not be an indication for a TKA revision but rather the symptom that has an underlying cause. According to this, in our study pain was only diagnosed in connection with a “true” indication, in particular with wear/lysis (significant) and arthrofibrosis/stiffness (tendency).
Several limitations of the present study have to be acknowledged. Most patients of this study (86%) were referred to our specialized knee centre for their first TKA revision and thus, this cohort is heterogeneous in terms of number of different surgeons and mean time to failure. This most probably caused a selection of more complicated revisions and therefore the frequencies differ from other studies or registries. Another limitation of this study is the fact that functional failure, which includes patients with low functional and clinical scores for the operated knee who did not have any further interventions, were not taken into consideration.
The strength of this study compared to registry data is that the causes for failure are much more likely to be accurate as a dedicated research fellow was meticulously combing through several reports of consultation and surgery to collect the necessary data related to revision surgery.
5. Conclusion
This study illustrated the main indications for the first TKA revisions in a specialized knee centre in Switzerland. The most frequent reasons for revision were instability, patellofemoral and/or extensor mechanism insufficiency and malalignment. The majority of the patients showed concurrent reasons for TKA failure with significant correlations amongst another. Only if we understand why a TKA fails one can tailor the diagnostic and treatment algorithm in this direction. In the current climate of increasingly short resources an efficient diagnostic and treatment algorithm should be able to detect the most important failure modes in TKA.
Declaration of competing interest
None declared.
References
- 1.Vilardo L., Shah M. Chronic pain after hip and knee replacement. Tech Reg Anesth Pain Manag. 2011;15:110–115. [Google Scholar]
- 2.Beswick A.D., Wylde V., Gooberman-Hill R., Blom A., Dieppe P. What proportion of patients report long-term pain after total hip or knee replacement for osteoarthritis? A systematic review of prospective studies in unselected patients. BMJ Open. 2012;2(1) doi: 10.1136/bmjopen-2011-000435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.American joint replacement registry (AJRR). Sixth AJRR annual report 2019 on hip and knee arthroplasty data. 2019. https://connect.ajrr.net/hubfs/PDFs%20and%20PPTs/AAOS_AJRR_2019_Annual_Report_Update_FINAL_150DPI.pdf?utm_campaign=2019%20AJRR%20AR&utm_medium=email&_hsenc=p2ANqtz-_R9e0yFLoCd0ilZVdi-Kx2fWPDNQKU55Z-MaFp5uUfeJUAIm2rJh-Lca146_R2hFgmvbRSs0CNPzjVEuIFjUh5cnj8k4QZYaNRm3lAyiu-JH3Pc2s&_hsmi=79114016&utm_content=79114016&utm_source=hs_automation&hsCtaTracking=f8fa4e79-6709-45d2-b51b-908382b7d5a7%7Cb61796dc-af0d-4a90-9b72-871845865e0b No authors listed.
- 4.National joint registry for england, Wales, Northern Ireland and the Isle of Man. 16th annual report 2019. 2019. https://reports.njrcentre.org.uk/Portals/0/PDFdownloads/NJR%2016th%20Annual%20Report%202019.pdf No authors listed.
- 5.Australian orthopaedic association (AOA). National joint replacement registry (NJRR) hip and knee arthroplasty annual report 2019. 2019. https://aoanjrr.sahmri.com/documents/10180/668596/Hip%2C+Knee%26+Shoulder+Arthroplasty/c287d2a3-22df-a3bb-37a2-91e6c00bfcf0 No authors listed.
- 6.Endoprothesenregister deutschland (EPRD) Jahresbericht. 2019 https://www.eprd.de/fileadmin/user_upload/Jahresbericht_2019_doppelseite_2.0.pdf No authors listed. [Google Scholar]
- 7.Swiss national joint registry. 2019. https://www.siris-implant.ch/de/index.php?section=Downloads&download=145 No authors listed. SIRIS Report 2019.
- 8.Bozic K.J., Kurtz S.M., Lau E. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop Relat Res. 2010;468(1):45–51. doi: 10.1007/s11999-009-0945-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fehring T.K., Odum S., Griffin W.L., Mason J.B., Nadaud M. Early failures in total knee arthroplasty. Clin Orthop Relat Res. 2001;(392):315–318. doi: 10.1097/00003086-200111000-00041. [DOI] [PubMed] [Google Scholar]
- 10.Hossain F., Patel S., Haddad F.S. Midterm assessment of causes and results of revision total knee arthroplasty. Clin Orthop Relat Res. 2010;468(5):1221–1228. doi: 10.1007/s11999-009-1204-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sharkey P.F., Hozack W.J., Rothman R.H., Shastri S., Jacoby S.M. Insall Award paper. Why are total knee arthroplasties failing today? Clin Orthop Relat Res. 2002;404:7–13. doi: 10.1097/00003086-200211000-00003. [DOI] [PubMed] [Google Scholar]
- 12.Vince K.G. Why knees fail. J Arthroplasty. 2003;18(3 Suppl 1):39–44. doi: 10.1054/arth.2003.50102. [DOI] [PubMed] [Google Scholar]
- 13.Callaghan J.J., O'Rourke M.R., Saleh K.J. Why knees fail: lessons learned. J Arthroplasty. 2004;19(4 Suppl 1):31–34. doi: 10.1016/j.arth.2004.02.015. [DOI] [PubMed] [Google Scholar]
- 14.Vessely M.B., Whaley A.L., Harmsen W.S., Schleck C.D., Berry D.J. The Chitranjan Ranawat Award: long-term survivorship and failure modes of 1000 cemented condylar total knee arthroplasties. Clin Orthop Relat Res. 2006;452:28–34. doi: 10.1097/01.blo.0000229356.81749.11. [DOI] [PubMed] [Google Scholar]
- 15.Khan M., Osman K., Green G., Haddad F.S. The epidemiology of failure in total knee arthroplasty: avoiding your next revision. Bone Joint Lett J. 2016;98-B(1 Suppl A):105–112. doi: 10.1302/0301-620X.98B1.36293. [DOI] [PubMed] [Google Scholar]
- 16.Schroer W.C., Berend K.R., Lombardi A.V. Why are total knees failing today? Etiology of total knee revision in 2010 and 2011. J Arthroplasty. 2013;28(8 Suppl):116–119. doi: 10.1016/j.arth.2013.04.056. [DOI] [PubMed] [Google Scholar]
- 17.Kasahara Y., Majima T., Kimura S., Nishiike O., Uchida J. What are the causes of revision total knee arthroplasty in Japan? Clin Orthop Relat Res. 2013;471(5):1533–1538. doi: 10.1007/s11999-013-2820-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Koh I.J., Cho W.S., Choi N.Y., Kim T.K., Kleos Korea Research G. Causes, risk factors, and trends in failures after TKA in Korea over the past 5 years: a multicenter study. Clin Orthop Relat Res. 2014;472(1):316–326. doi: 10.1007/s11999-013-3252-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thiele K., Perka C., Matziolis G., Mayr H.O., Sostheim M., Hube R. Current failure mechanisms after knee arthroplasty have changed: polyethylene wear is less common in revision surgery. J Bone Joint Surg Am. 2015;97(9):715–720. doi: 10.2106/JBJS.M.01534. [DOI] [PubMed] [Google Scholar]
- 20.Mulhall K.J., Ghomrawi H.M., Scully S., Callaghan J.J., Saleh K.J. Current etiologies and modes of failure in total knee arthroplasty revision. Clin Orthop Relat Res. 2006;446:45–50. doi: 10.1097/01.blo.0000214421.21712.62. [DOI] [PubMed] [Google Scholar]
- 21.Hirschmann M.T., Henckel J., Rasch H. SPECT/CT in patients with painful knee arthroplasty-what is the evidence? Skeletal Radiol. 2013;42(9):1201–1207. doi: 10.1007/s00256-013-1657-9. [DOI] [PubMed] [Google Scholar]
- 22.Barnsley L., Barnsley L. Detection of aseptic loosening in total knee replacements: a systematic review and meta-analysis. Skeletal Radiol. 2019;48(10):1565–1572. doi: 10.1007/s00256-019-03215-y. [DOI] [PubMed] [Google Scholar]
- 23.Hirschmann M.T., Amsler F., Rasch H. Clinical value of SPECT/CT in the painful total knee arthroplasty (TKA): a prospective study in a consecutive series of 100 TKA. Eur J Nucl Med Mol Imag. 2015;42(12):1869–1882. doi: 10.1007/s00259-015-3095-5. [DOI] [PubMed] [Google Scholar]
- 24.Rasch H., Falkowski A.L., Forrer F., Henckel J., Hirschmann M.T. 4D-SPECT/CT in orthopaedics: a new method of combined quantitative volumetric 3D analysis of SPECT/CT tracer uptake and component position measurements in patients after total knee arthroplasty. Skeletal Radiol. 2013;42(9):1215–1223. doi: 10.1007/s00256-013-1643-2. [DOI] [PubMed] [Google Scholar]
- 25.Hirschmann M.T., Wagner C.R., Rasch H., Henckel J. Standardized volumetric 3D-analysis of SPECT/CT imaging in orthopaedics: overcoming the limitations of qualitative 2D analysis. BMC Med Imag. 2012;12:5. doi: 10.1186/1471-2342-12-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Murer A.M., Hirschmann M.T., Amsler F., Rasch H., Huegli R.W. Bone SPECT/CT has excellent sensitivity and specificity for diagnosis of loosening and patellofemoral problems after total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2020;28(4):1029–1035. doi: 10.1007/s00167-019-05609-w. [DOI] [PubMed] [Google Scholar]
- 27.Parvizi J., Tan T.L., Goswami K. The 2018 definition of periprosthetic hip and knee infection: an evidence-based and validated criteria. J Arthroplasty. 2018;33(5):1309–1314 e1302. doi: 10.1016/j.arth.2018.02.078. [DOI] [PubMed] [Google Scholar]
- 28.Morawietz L., Krenn V. [The spectrum of histomorphological findings related to joint endoprosthetics] Pathologe. 2014;35(Suppl 2):218–224. doi: 10.1007/s00292-014-1976-1. [DOI] [PubMed] [Google Scholar]
- 29.Kalson N.S., Borthwick L.A., Mann D.A. International consensus on the definition and classification of fibrosis of the knee joint. Bone Joint Lett J. 2016;98-B(11):1479–1488. doi: 10.1302/0301-620X.98B10.37957. [DOI] [PubMed] [Google Scholar]
- 30.Awengen R., Rasch H., Amsler F., Hirschmann M.T. Symptomatic versus asymptomatic knees after bilateral total knee arthroplasty: what is the difference in SPECT/CT? Eur J Nucl Med Mol Imag. 2016;43(4):762–772. doi: 10.1007/s00259-015-3278-0. [DOI] [PubMed] [Google Scholar]
- 31.Parker D.A., Dunbar M.J., Rorabeck C.H. Extensor mechanism failure associated with total knee arthroplasty: prevention and management. J Am Acad Orthop Surg. 2003;11(4):238–247. doi: 10.5435/00124635-200307000-00003. [DOI] [PubMed] [Google Scholar]
- 32.Sharkey P.F., Lichstein P.M., Shen C., Tokarski A.T., Parvizi J. Why are total knee arthroplasties failing today--has anything changed after 10 years? J Arthroplasty. 2014;29(9):1774–1778. doi: 10.1016/j.arth.2013.07.024. [DOI] [PubMed] [Google Scholar]
- 33.Cerquiglini A., Henckel J., Hothi H. Retrieval analysis of contemporary antioxidant polyethylene: multiple material and design changes may decrease implant performance. Knee Surg Sports Traumatol Arthrosc. 2019;27(7):2111–2119. doi: 10.1007/s00167-019-05387-5. [DOI] [PubMed] [Google Scholar]
- 34.Parvizi J., Mortazavi S.M., Devulapalli C., Hozack W.J., Sharkey P.F., Rothman R.H. Secondary resurfacing of the patella after primary total knee arthroplasty does the anterior knee pain resolve? J Arthroplasty. 2012;27(1):21–26. doi: 10.1016/j.arth.2011.04.027. [DOI] [PubMed] [Google Scholar]
- 35.Le D.H., Goodman S.B., Maloney W.J., Huddleston J.I. Current modes of failure in TKA: infection, instability, and stiffness predominate. Clin Orthop Relat Res. 2014;472(7):2197–2200. doi: 10.1007/s11999-014-3540-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pitta M., Esposito C.I., Li Z., Lee Y.Y., Wright T.M., Padgett D.E. Failure after modern total knee arthroplasty: a prospective study of 18,065 knees. J Arthroplasty. 2018;33(2):407–414. doi: 10.1016/j.arth.2017.09.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wilson C.J., Theodoulou A., Damarell R.A., Krishnan J. Knee instability as the primary cause of failure following Total Knee Arthroplasty (TKA): a systematic review on the patient, surgical and implant characteristics of revised TKA patients. Knee. 2017;24(6):1271–1281. doi: 10.1016/j.knee.2017.08.060. [DOI] [PubMed] [Google Scholar]
- 38.Longo U.G., Candela V., Pirato F., Hirschmann M.T., Becker R., Denaro V. Midflexion instability in total knee arthroplasty: a systematic review. Knee Surg Sports Traumatol Arthrosc. 2020 doi: 10.1007/s00167-020-05909-6. [DOI] [PubMed] [Google Scholar]
- 39.Petrie J.R., Haidukewych G.J. Instability in total knee arthroplasty : assessment and solutions. Bone Joint Lett J. 2016;98-B(1 Suppl A):116–119. doi: 10.1302/0301-620X.98B1.36371. [DOI] [PubMed] [Google Scholar]
- 40.Cottino U., Sculco P.K., Sierra R.J., Abdel M.P. Instability after total knee arthroplasty. Orthop Clin N Am. 2016;47(2):311–316. doi: 10.1016/j.ocl.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 41.Hirschmann M.T., Becker R. Springer International Publishing; 2015. The Unhappy Total Knee Replacement: A Comprehensive Review and Management Guide. [Google Scholar]
- 42.Murer M., Falkowski A.L., Hirschmann A., Amsler F., Hirschmann M.T. Threshold values for stress radiographs in unstable knees after total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2020 doi: 10.1007/s00167-020-05964-z. [DOI] [PubMed] [Google Scholar]
- 43.Hirschmann M.T., Konala P., Amsler F., Iranpour F., Friederich N.F., Cobb J.P. The position and orientation of total knee replacement components: a comparison of conventional radiographs, transverse 2D-CT slices and 3D-CT reconstruction. J Bone Joint Surg Br. 2011;93(5):629–633. doi: 10.1302/0301-620X.93B5.25893. [DOI] [PubMed] [Google Scholar]
- 44.Skowronek P., Arnold M., Starke C. Intra- and postoperative assessment of femoral component rotation in total knee arthroplasty: an EKA knee expert group clinical review. Knee Surg Sports Traumatol Arthrosc. 2020 doi: 10.1007/s00167-020-06006-4. [DOI] [PubMed] [Google Scholar]
- 45.Postler A., Lutzner C., Beyer F., Tille E., Lutzner J. Analysis of total knee arthroplasty revision causes. BMC Muscoskel Disord. 2018;19(1):55. doi: 10.1186/s12891-018-1977-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dalury D.F., Pomeroy D.L., Gorab R.S., Adams M.J. Why are total knee arthroplasties being revised? J Arthroplasty. 2013;28(8 Suppl):120–121. doi: 10.1016/j.arth.2013.04.051. [DOI] [PubMed] [Google Scholar]
- 47.Hofmann S., Seitlinger G., Djahani O., Pietsch M. The painful knee after TKA: a diagnostic algorithm for failure analysis. Knee Surg Sports Traumatol Arthrosc. 2011;19(9):1442–1452. doi: 10.1007/s00167-011-1634-6. [DOI] [PubMed] [Google Scholar]
- 48.Mandalia V., Eyres K., Schranz P., Toms A.D. Evaluation of patients with a painful total knee replacement. J Bone Joint Surg Br. 2008;90(3):265–271. doi: 10.1302/0301-620X.90B3.20140. [DOI] [PubMed] [Google Scholar]
- 49.Cheuy V.A., Foran J.R.H., Paxton R.J., Bade M.J., Zeni J.A., Stevens-Lapsley J.E. Arthrofibrosis associated with total knee arthroplasty. J Arthroplasty. 2017;32(8):2604–2611. doi: 10.1016/j.arth.2017.02.005. [DOI] [PubMed] [Google Scholar]
- 50.Naudie D.D., Ammeen D.J., Engh G.A., Rorabeck C.H. Wear and osteolysis around total knee arthroplasty. J Am Acad Orthop Surg. 2007;15(1):53–64. doi: 10.5435/00124635-200701000-00006. [DOI] [PubMed] [Google Scholar]
- 51.Delanois R.E., Mistry J.B., Gwam C.U., Mohamed N.S., Choksi U.S., Mont M.A. Current epidemiology of revision total knee arthroplasty in the United States. J Arthroplasty. 2017;32(9):2663–2668. doi: 10.1016/j.arth.2017.03.066. [DOI] [PubMed] [Google Scholar]
- 52.Phillips J.R., Hopwood B., Stroud R., Dieppe P.A., Toms A.D. The characterisation of unexplained pain after knee replacement. Br J Pain. 2017;11(4):203–209. doi: 10.1177/2049463717719774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Jacofsky D.J., Della Valle C.J., Meneghini R.M., Sporer S.M., Cercek R.M. Revision total knee arthroplasty: what the practicing orthopaedic surgeon needs to know. Instr Course Lect. 2011;60:269–281. [PubMed] [Google Scholar]