Abstract
This is a retrospective chart review of 20 patients treated with a consensus-driven treatment algorithm in multisystem inflammatory syndrome in children patients across a wide clinical spectrum. Their treatments and clinical status are described as well as their favorable return to functional baseline by 30 days post presentation.
Keywords: COVID-19, hospital care, intensive care
Children with severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) infection, the virus responsible for coronavirus disease 2019 (COVID-19) disease, are often asymptomatic or have milder symptoms than infected adults. In a large case series from China, only 2 percent were below 19 years of age, indicating that children represent a minority of the affected population [1]. In late April, there emerged a new multisystem inflammatory condition temporally associated with SARS-CoV2 in children [2]. On May 14, 2020, the Centers for Disease Control and Prevention (CDC) disseminated a case definition of multisystem inflammatory syndrome in children (MIS-C), allowing for uniformity in the diagnosis [3].
Multicenter descriptive studies have depicted demographics, clinical features, and organ involvement in MIS-C [4, 5]. Treatment protocols from various institutions have circulated based on European data that suggest the efficacy of treatment for MIS-C utilizing intravenous immunoglobulin (IVIG) and steroids [4]. However, post-discharge outcomes are not yet well described. This study describes treatment for MIS-C with a consensus-driven algorithm and short-term outcomes for 20 patients during and shortly after a COVID-19 surge in New Jersey.
METHODS
Study Population
We retrospectively reviewed electronic medical records of 24 patients who met CDC criteria for MIS-C [3] who were admitted to the hospital between April 15, 2020, and June 20, 2020. Positivity for SARS-CoV-2 by reverse transcription-polymerase chain reaction (RT-PCR), serology, or evidence of SARS-CoV-2 exposure was required. We excluded patients who were transferred out of our institutions (n = 2) and those later discovered to have alternate diagnoses (n = 2).
Data Collection
Demographics, initial admission location (intensive care unit [ICU] or pediatric inpatient floor), hospital length of stay, results of laboratory and cardiac diagnostics, treatment, final disposition, and clinical status within 30 days post treatment were collected. Outcomes included final recorded clinical status and echocardiography, if done, within 30 days from treatment. If a patient had a follow-up visit within 30 days of treatment and had returned to their baseline, this was assumed to continue forward to 30 days. Institutional Review Board (IRB) approval and approval for waiver of consent were obtained.
Statistical Analysis
Descriptive analysis was performed with continuous variables summarized by mean (standard deviation) or median (interquartile range), depending on whether the data were normally distributed. Categorical variables were summarized as frequencies (percentage). Comparison of continuous variables between 2 groups was conducted using 2-sided t-test or Wilcoxon rank-sum test, as appropriate. A P-value < .05 was considered statistically significant. All data analysis was performed using SAS software v9.4.
RESULTS
Patient Characteristics
The baseline characteristics of all patients are listed in Table 1, including age, admission unit, days of symptoms prior to diagnosis, and hospital length of stay. The average Pediatric Early Warning Score (PEWS) on admission to the floor was 0.8, with a median of 1. The average PEWS score on admission to the ICU was 2.8, with a median of 3.
Table 1.
Demographics
| Characteristic | Statistic/Category | MIS-C (N = 20) |
|---|---|---|
| Gender | Female | 9 (45%) |
| Male | 11 (55%) | |
| BMI (if >2 y) N = 14 | Mean | 21 |
| Range | 14-32 | |
| Median | 21 | |
| Interquartile range | 6 | |
| Age in years | Mean | 7.3 |
| Range | 0.88-18.0 | |
| African American | 3 (15%) | |
| Race/ethnicity | Asian | 4 (20%) |
| Caucasian (non Hispanic) | 4 (20%) | |
| Hispanic | 8 (40%) | |
| Other | 1 (5%) | |
| Admission unit | Intensive Care | 10 (50%) |
| General Pediatrics | 10 (50%) | |
| Length of stay (d) | Mean | 4.6 |
| Range | 2-40a | |
| Median | 4 | |
| Days of symptoms prior to presentation | Mean | 5.4 |
| Range | 3-11 | |
| Median | 5 |
Abbreviations: BMI, body mass index; MIS-C, multisystem inflammatory syndrome in children.
aPatient who stayed for 40 days encompassed acute COVID-19 infection and MIS-C and was excluded from mean and median calculations.
Clinical Manifestations
All patients presented with subjective or measured fever. Eleven patients had gastrointestinal symptoms (55%), 11 had conjunctivitis (55%), and 10 had rash (50%). One patient initially had COVID-19 with acute respiratory distress syndrome (ARDS) and then developed MIS-C 4 weeks after admission. Cardiac abnormalities (defined as elevated brain natriuretic peptide [BNP] >150 pg/mL, dilation of coronary arteries, and/or decreased ejection fraction) were present in 13 patients (65%), as seen in Table 2. Nineteen patients (95%) were discharged home without activity limitation or supplemental oxygen. One patient (5%) required oxygen at discharge.
Table 2.
Echocardiogram Findings
| Study Findings | Initial Echocardiogram | Follow-up Echocardiogram | P-Value |
|---|---|---|---|
| N = 18 | N = 11 | ||
| Pericardial effusion | 2 (11%) | 0 (0%) | |
| Coronary artery dilation | 0 (0%) | 1 (9%) | |
| Valvulitis | 0 (0%) | 0 (0%) | |
| Decreased myocardial function | 7 (39%) | 1 (9%) | |
| Normal study | 9 (50%) | 9 (82%) | |
| Ejection fraction | 18 | 11 | |
| Mean ± SD | 57.9 ± 7.9 | 64.4 ± 7.1 | .03 |
| Range | 36.6-68.2 | 49-72.5 | |
| Difference in mean | 6.5 |
Treatments
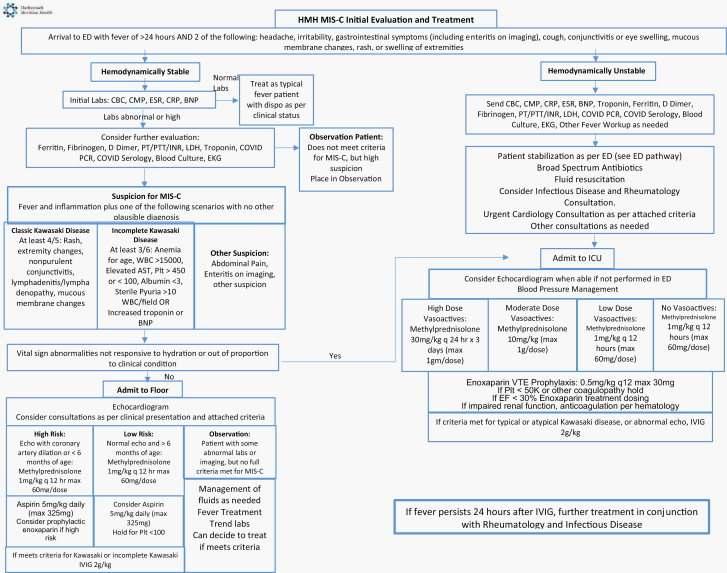
Our consensus protocol (Figure 1) included options for inpatient observation of patients with mild symptoms and corticosteroid and/or IVIG treatment for more severe cases. Ten patients (50%) received both corticosteroids and IVIG, 3 patients (15%) received corticosteroid monotherapy, and 2 patients (10%) received IVIG monotherapy. Five patients (25%) did not receive immunosuppressive therapy and improved with supportive care. Table 3 illustrates SARS-CoV-2 serology, initial PEWS score on admission, identified cardiac abnormality, and treatments for all patients. Patients who did not have positive PCR or serology testing were early in our treatment period and had household contacts with confirmed SARS-CoV-2 positivity. Patients treated with corticosteroids were discharged on a tapering schedule; 1 patient did not adhere closely to his/her prescribed tapering schedule. The MIS-C treatment protocol was applied in addition to treatments considered standard of care, including supportive care and empiric antibiotics if there was an initial concern for bacterial infection, and blood pressure support for hypotension or shock, which was required for 5 patients (25%).
Figure 1.
Treatment and evaluation algorithm.
Table 3.
Serology and Treatment of All Patients
| Patient | SARS-CoV-2 PCR or NAAT | SARS-CoV-2 IgG | Initial PEWS Score | Cardiac Abnormality | Treatment Received |
|---|---|---|---|---|---|
| 1 | Neg | Pos | 1 | Supportive | |
| 2 | Neg | Not Done | 0 | Supportive | |
| 3 | Neg | Pos | 3 | Y | IVIG |
| 4 | Neg | Pos | 3 | Y | Supportive |
| 5 | Neg | Neg | 0 | IVIG, Steroids | |
| 6 | Pos | Pos | 4 | Y | IVIG, Steroids |
| 7 | Pos | Pos | 3 | Y | IVIG, Steroids |
| 8 | Pos | Pos | 0 | Y | Steroids |
| 9 | Neg | Pos | 0 | Y | IVIG, Steroids |
| 10 | Pos | Pos | 4 | Y | IVIG, Steroids |
| 11 | Pos | Pos | 0 | Y | IVIG, Steroids |
| 12 | Neg | Pos | 2 | Steroids | |
| 13 | Neg | Neg | 2 | Y | IVIG |
| 14 | Pos | Pos | 0 | Y | Supportive |
| 15 | Neg | Neg | 0 | IVIG, Steroids | |
| 16 | Pos | Pos | 1 | IVIG, Steroids | |
| 17 | Neg | Pos | 3 | Y | IVIG, Steroids |
| 18 | Neg | Neg | 0 | Supportive | |
| 19 | Neg | Pos | 0 | Y | IVIG, Steroids |
| 20 | Neg | Neg | 0 | Y | Steroids |
Abbreviations: IgG, Immunoglobulin G; NAAT, Nucleic-Acid Amplification Test; Neg, negative; Pos, positive; PCR, polymerase chain reaction; PEWS, Pediatric Early Warning Score; IVIG, intravenous immunoglobulin.
Short-Term Outcomes
After discharge, all patients followed up with hospital-affiliated pediatric subspecialists at least once within 30 days of treatment, with an average of 3 visits within the 30-day period. The final follow-up appointment after discharge was a median of 29 days (range 14-30 days) and a mean of 27 days. One patient with both ARDS and MIS-C was discharged on supplemental oxygen, which was discontinued by day 30 post treatment. Of the 13 patients with initial cardiac involvement, 11 patients (85%) had a follow-up echocardiogram within 30 days following treatment (see Table 2). Nine patients (82%) had normal echocardiograms. One patient had a new finding of coronary artery dilation, 1 week after discharge (Z score = 2.89) that improved (Z score = 2.1) on a subsequent echocardiogram performed on day 15. One patient had a mild and clinically insignificant decline in left ventricular function on outpatient evaluation. This patient had not adhered to prescribed steroid tapering and had a history of chemotherapy, with last normal echocardiogram in 2014. An outpatient echocardiogram was not performed in 2 patients, due to normalization of echocardiogram while inpatient. By 30 days after treatment, all patients, including those transferred to an outside institution, had clinically improved and had returned to normal activity for age.
DISCUSSION
Since the description of MIS-C by the Royal College of Paediatrics and Child Health in mid-April [2], a variable clinical presentation with a wide range of clinical features has been described in the literature [6]. Our patients illustrate this variety of presentations, with differing illness severity and presenting manifestations.
Given that there is no consensus standard of care regarding treatment, multiple facilities have developed various treatment algorithms for MIS-C. Approaches have typically incorporated immunomodulatory treatments, such as steroids, IVIG, and biologics [7]. Treatment with biologics does have disadvantages, including cost, lack of availability, and unknown adverse effects in the MIS-C population. In June 2020, the American College of Rheumatology (ACR) issued guidance utilizing a stepwise approach [8]. The treatment protocol implemented in this study is similar to this guidance.
Some of the authors have noted similarities between MIS-C and Kawasaki disease [9], including some shared clinical manifestations and lab findings, as well as cardiac involvement and sequelae. Based on these common characteristics, treatments used in Kawasaki disease have been proposed for use in MIS-C. IVIG, the treatment with the best-documented efficacy in Kawasaki disease, already has evidence for good clinical outcomes when used for MIS-C [10]. Our patients had normalization of cardiac function at the time of follow-up, with or without the use of IVIG. One patient with evidence of left ventricular dysfunction after discharge was noncompliant with steroid taper; it is possible that this contributed to the decline in function. This highlights the importance of clear and understandable patient care instructions for continued clinical improvement following hospital discharge. Several patients did not have a repeat outpatient echocardiogram. This decision was individualized based on the patient’s clinical progress, inpatient echocardiogram, and physical exam and was deemed appropriate by our multidisciplinary task force. Clinical status and physical exam, combined with previous echocardiogram results, can help to determine whether follow-up with echocardiogram is necessary.
Longer-term description of MIS-C outcomes is limited because this illness has emerged so recently. However, as the COVID-19 pandemic progresses, new data on outcomes are appearing in the literature [11]. All of our patients, regardless of severity on presentation, returned to clinical baseline. This includes the 5 patients who received supportive care only.
Our population also highlights the variable presentation and severity of MIS-C. Earlier studies reported close to 80% requiring ICU admission and 48% requiring vasoactive support [5]. In our population, only 50% (n = 10) required ICU admission. Of those requiring ICU care, 5 patients required vasoactive support. Many of the sickest patients presented early in the study time period, prior to an education campaign in our region.
LIMITATIONS
Limitations to our study include the relatively small sample size and the retrospective study design. The first MIS-C patients in our institution were treated prior to the development of our institutional treatment protocol. This accounts for some of the variability in treatments received. However, all patients were treated based on the same principles detailed in our algorithm and in this study. Our task force agreed that 30 days was likely sufficient time to assess for the development of complications, based on experience with other viral-mediated myocarditis illnesses and with Kawasaki disease. Currently, there are limited data to determine whether this illness will have long-term outcomes different from other similar inflammatory illnesses.
CONCLUSIONS
COVID-19 has provided an epidemiologic, diagnostic, and management challenge since its emergence in China in the late part of 2019. MIS-C has likewise challenged clinicians because of limited data on optimal treatment and natural history in affected children. Even less is known about the comparable syndrome recently described in adults, MIS-A [12]. This descriptive study of a cohort of children treated with a multidisciplinary approach and consensus-driven algorithm adds to our understanding of the short-term, generally favorable outcomes of MIS-C.
Notes
Acknowledgments. We would like to thank Michelle Secic, MS, for her assistance with the statistics. Our task force and development of the algorithm included the following members, who contributed greatly to this multidisciplinary effort: Saranga Agarwal, MD; Okechukwu Anene, MD; Anna Gironella, MD, MS; Ginger Janow, MD, MPH; Yukiko Kimura, MD; Suzanne Li, MD; PhD; Mitchel Alpert, MD; Katherine Orellana, MD; Stacey Rifkin-Zenenberg, DO; Jeremy Schnall, MD; Pooja Shah, PharmD; Ariel Sherbany, MD; Anita Siu, PharmD; Meghan Tozzi, MD; Victor A. Uduaghan, MD; Jennifer Weiss, MD; and Austin Wong, MD.
Authors contributions. K. N. C., J. G., and S. M. B. conceptualized the initial study, participated in data collection and analysis, drafted the initial manuscript, and reviewed and revised the manuscript. R. L., C. B., and Z. M. participated in data collection, reviewed, and revised the manuscript. A. B., M. K., S. L., H. P., J. P., K. S., M .E. S., R. T., D. M. W., and J. A. conceptualized the initial study and reviewed and revised the manuscript, providing substantial changes important to the intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Potential conflicts of interest. The authors have no conflicts to disclose. All authors have submitted the ICMJE Form for Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020; 323:1239–42. [DOI] [PubMed] [Google Scholar]
- 2. Guidance—paediatric multisystem inflammatory syndrome temporally associated with COVID-19 Royal College of Paediatrics and Child Health Website. Published 2020.. www.rcpch.ac.uk/resources/guidance-paediatric-multisystem-inflammatory-syndrome-temporally-associated-covid-19; https://www.rcpch.ac.uk/sites/default/files/2020-05/COVID-19-Paediatric-multisystem-%20inflammatory%20syndrome-20200501.pdf. Accessed 2 August 2020.
- 3. Multisystem inflammatory syndrome in children (MIS-C) associated with coronavirus disease 2019 (COVID-19) Centers for Disease Control and Prevention Web site. Published 2020. https://emergency.cdc.gov/han/2020/han00432.asp. Accessed 2 August 2020.
- 4. Toubiana J, Poirault C, Corsia A, et al. Kawasaki-like multisystem inflammatory syndrome in children during the covid-19 pandemic in Paris, France: prospective observational study. BMJ 2020; 369:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Feldstein LR, Rose EB, Randolph AG. Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med 2020; 383:334–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Kaushik S, Aydin SI, Derespina KR, et al. Multisystem inflammatory syndrome in children (MIS-C) associated with SARS-CoV-2 infection: a multi-institutional study from New York City. J Pediatr 2020; 224:24–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hennon TR, Penque MD, Abdul-Aziz R, et al. COVID-19 associated multisystem inflammatory syndrome in children (MIS-C) guidelines; a Western New York approach. Prog Pediatr Cardiol 2020;101232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. American College of Rheumatology. Clinical guidance for pediatric patients with multisystem inflammatory syndrome in children (MIS-C) associated with SARS-CoV-2 and hyperinflammation in COVID-19 Published 2020. https://www.rheumatology.org/Portals/0/Files/ACR-COVID-19-Clinical-Guidance-Summary-MIS-C-Hyperinflammation.pdf. Accessed 2 August 2020.
- 9. Jones VG, Mills M, Suarez D, et al. COVID-19 and Kawasaki disease: novel virus and novel case. Hosp Pediatr 2020; 10:537–40. [DOI] [PubMed] [Google Scholar]
- 10. Belhadjer Z, Méot M, Bajolle F, et al. Acute heart failure in multisystem inflammatory syndrome in children (MIS-C) in the context of global SARS-CoV-2 pandemic. Circulation 2020; 142:429–36. [DOI] [PubMed] [Google Scholar]
- 11. Capone CA, Subramony A, Sweberg T, et al. Characteristics, cardiac involvement, and outcomes of multisystem inflammatory disease of childhood (MIS-C) associated with SARS-CoV-2 infection. J Pediatr 2020; 224:141–144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Morris SB, Schwartz NG, Patel P, et al. Case series of multisystem inflammatory syndrome in adults associated with SARS-CoV-2 infection—United Kingdom and United States, March–August 2020. MMWR Morb Mortal Wkly Rep 2020; 69:1450–6. [DOI] [PMC free article] [PubMed] [Google Scholar]