Abstract
COVID-19 has been a threat throughout the world since December 2019. In attempts to discover an urgent treatment regime for COVID-19, hydroxychloroquine (HCQ) and chloroquine (CQ) have been on solidarity clinical trial. However, many countries have pulled HCQ and CQ from their COVID-19 treatment regimens recently, some countries still continue using them for patients who have previously started HCQ and CQ and they may complete their course under the supervision of a doctor. HCQ and CQ are 4-aminoquinoline drugs and it is safe to use them for autoimmune diseases, rheumatoid arthritis, systemic lupus erythematosus and malaria as well. Determination of CQ, HCQ and their metabolites in biologic fluids and in pharmaceuticals has great importance, especially for pharmacokinetics, pharmacodynamics and epidemiological studies. In this review, liquid chromatographic methods developed in the last 10 years were summarized focusing on sample preparation and detection methods for HCQ and CQ determination in biological fluids and pharmaceutical preparations. It is hoped that this article could be helpful to facilitate the use of these drugs in clinical trials or drug research studies as it provides comprehensive information on the reported analytical methods.
Introduction
In Wuhan City, in December 2019, Chinese scientists have named a new coronavirus as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) following the pneumonia outbreak and it was subsequently named as novel coronavirus disease 2019 (COVID-19) by World Health Organization (WHO). Coronavirus is a group of viruses categorized under alpha and beta coronavirus (1). There were 15,538,736 confirmed cases with 634,325 deaths declared on 25 July 2020 (2). Many trials had been designed globally for COVID-19 treatment and chloroquine (CQ) and hydroxychloroquine (HCQ) were used as the two candidate molecules. CQ and HCQ or in some cases both in combination with other drugs were used globally (3). Even though in some countries it had stopped to be used in the treatment since the 17th of June, patients who have already started CQ/HCQ may complete their course under the supervision of the doctor. Drugs are accepted as safe to use in patients with autoimmune diseases or malaria (4).
CQ and its hydroxy-analogue HCQ are 4-aminoquinoline drugs. The exception of COVID-19 treatment trials, CQ/HCQ are the molecules that were used to cure malaria since 1955 (5). Due to its less toxicity as well as immunomodulatory and antithrombotic properties HCQ is used to treat antiphospholipid syndrome, rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE). HCQ also has a sensitizing effect on cancer cells when radiation and chemotherapy were used (6). Although the efficacy of CQ/HCQ has been attributed to different mechanisms, they were used in the treatment of coronaviruses like SARS-CoV, SARS-CoV-2 (1, 7, 8) due to the inhibition of the viral entry directly and spread in vitro and in vivo models. CQ/HCQ inhibits the terminal glycosylation of ACE2 protein acting as a receptor entering the cell by the new coronaviruses. SARS-CoV-2 spike protein interacts less with non-glycosylated ACE2, further inhibits viral entry (8). CQ/HCQ are heme polymerase inhibitors in Plasmodium species which prevent the conversion of heme to hemazoin. Diffusion of CQ/HCQ into lysosomes and endosomes results in protonation of drugs, their weak base character gives a rise in surrounding pH due to the accumulation of the drugs in lysosomes and subsequently prohibiting viruses to enter into the cells. If the pH of the lysosomes is altered, autoimmune diseases show reduced self-antigen presence and it is interfered that the plasmodia proteolysis the hemoglobin for the energy need. The dimerization of alpha and beta chains of the large histocompatibility complex (MHC) class II are prevented by antigen processing and the inflammatory response of the cell reduced. The recycling of MHC complexes is changed by the high pH in the vesicles. Due to the self-peptides bind to low-affinity MHC complexes, T cells play less role in the autoimmune system. Probably, interleukin-1 and tumor necrosis factor released as cytokines through Toll-like receptors are decreased by the accumulation of CQ/HCQ. CQ and HCQ affect the lysosome functions both in humans and plasmodia (9, 10).
HCQ was first synthesized in 1946 by the addition of a hydroxyl group to CQ to reduce toxicity. Experimental animal studies showed that CQ was 2–3 times more toxic than hydrochloroqune (11). CQ/HCQ both possess an asymmetric carbon atom resulting in two enantiomers and only their racemic mixtures are available commercially (12). Using racemic mixtures in therapy needs extra attention due to the possible cause of toxicity or adverse drug effects dealing with pharmacologically inactive or less active isomers (13). A toxicity data proved more toxicity for R- (+) isomer of CQ compared to its S- (+) form which has a very similar structure to HCQ. This may explain the less toxic effect of HCQ as well as its higher solubility compared to CQ (8, 11, 13–15). Retinopathy is one of the severe side effects of CQ/HCQ resulting with in visual impairment is possibly caused by free chloride radical formed by breaking the C-Cl bond (16–18). Other reported side effects are headache, various skin eruptions, drowsiness, visual disturbances, nausea, vomiting, cardiovascular collapse, shock, convulsions, respiratory arrest, cardiac arrest and hypokalemia (19).
CQ is N-dealkylated primarily by CYP2C8 and CYP3A4 to N-desethylchloroquine ((R)-CQ, N-desethyl—NDCQ). By using CYP3A5, CYP2D6, CQ is N-dealkylated to a less extent and it is even lesser when used CYP1A1. N-desethylchloroquine can be more N-dealkylated to N-bidesethylchloroquine (NBCQ), also by even more N-dealkylating it can be converted to 7-chloro-4-aminoquinoline (CAQ). In this manner, CYP3A4 can be used to N-dealkylate HCQ to get desethylhydroxychloroquine (DHCQ) which is the major metabolite as well as the inactive metabolites desethylchloroquine (DCQ) and bidesethylchloroquine (BDCQ) (20) (Figure 1). Some chemical, physical and pharmacological properties of CQ and HCQ are summarized in Table I.
Figure 1.
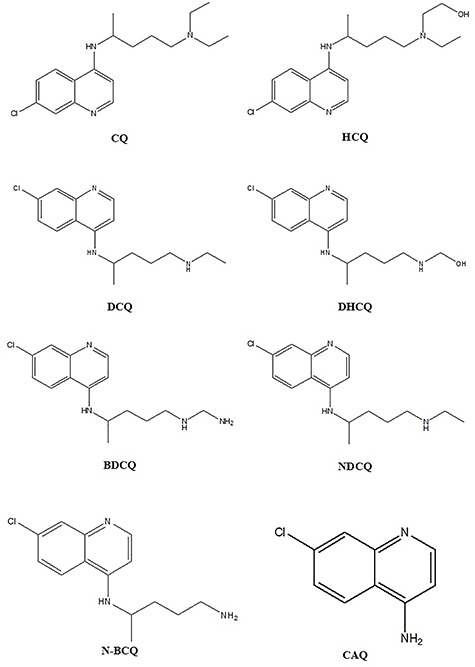
Chemical structures of CQ, HCQ and their related metabolites.
Table I.
Some Chemical, Physical and Pharmacological Properties of CQ and HCQ
| CQ | HCQ | |
|---|---|---|
| Molecular weight (g/mol) | 320 (21) | 336 (21) |
| Chemical formula | C18H26ClN3 | C18H26ClN3O |
| IUPAC name | 7-chloro-N-[5-(diethylamino)pentan-2-yl]quinolin-4-amine | 2-({4-[(7-chloroquinolin-4-yl)amino]pentyl}(ethyl)amino)ethan-1-ol |
| Solubility | Freely soluble in water as phosphate and sulfate form (12) | 2.61.10-2 g L−1 (in water as sulfate salt) (13, 22) |
| pKa | 4.0, 8.4 and 10.2 (23) | <4.0, 8.3 and 9.7 (23) |
| Log P | 3.85 (experimental) 4.12 (calculated from Clog P) (24) |
4.72 (experimental) 5.06 (calculated from Clog P) (24) |
| Absorption | 89 ± 16% (oral, fasting subjects) (21) | 74 ± 13% (oral, fasting subjects) (21) |
| Bioavailability | 52– 102% (mean 78%) as 300 mg oral solution form 67–114% (mean 89%) as 300 mg tablets form (25) |
Relative bioavailability was 109.5% (according to AUC0–60d) and 110.7% (according to AUC0–∞) (0.2 g HCQS tablets) (26). |
| C max | 122.10– 733.52 ng mL−1 (oral 150 mg CQ tablets) (27) | 135–422 ng mL−1 (oral 200 mg HCQS tablets) (28) |
| I max | 14.0–28.0 days (oral 150 mg CQ tablets) (27) | 1.5–7.0 h (oral 200 mg HCQS tablets) (28) |
| Volume of distribution | 65,000 L (29) | 47,257 L (29) |
| Protein Binding | 33–70% (plasma) (21) | 33–70% (plasma) (21) |
| Terminal elimination half-life | 20–60 days (21) | ~40 days (21) |
| Excretion | 40– 60% unchanged or metabolized drug through kidneys 8–25% unchanged or changed for in feces 5% sloughed off through skin 25–45% stored long-term in lean body tissues (21) |
40–60% unchanged or metabolized drug through kidneys 8–25% unchanged or changed for in feces 5% sloughed off through skin 25–45% stored long-term in lean body tissues (21) |
| Clearance | 0.10 L h−1 kg−1 from whole blood and 0.7 to 1 L h−1 kg−1 from plasma (30) | 15.5 L h−1 from blood (14) |
There are different reported analytical methods to determine CQ/HCQ and their metabolites to date but no review article is available to summarize the developed analytical methods focusing on liquid chromatography for the quantification of CQ/HCQ in different matrices. This review mainly relates the evaluation of sample preparation techniques and LC methods with several detections developed for CQ/HCQ focusing last 10 years. Sample preparation techniques, chromatographic conditions and validation results are summarized in Tables II and III, respectively. We believe it will be helpful to researchers who would like to develop new methods both in COVID-19 therapy and antimalarial use of CQ/HCQ.
Table II.
Summary of Sample Preparation Techniques and Chromatographic Conditions for Determination of HCQ and CQ in Biological Matrices and Pharmaceutical Dosage Forms
| Compound | Matrix/matrices | Sample preparation | Solvent(s) used in sample preparation | Mobile phase | LC column | Detection | References |
|---|---|---|---|---|---|---|---|
| HCQ, DCQ, BDCQ &DHCQ | 25 μL of mouse blood or 100 μL tissue homogenate | PP | ACN | Formic acid in W and 0.1% formic acid in MeOH | Thermo Aquasil C18 (50 × 4.6 mm; 3 μ) | MS/MS | (6) |
| HCQ | 100 μL serum | PP | Perchloric acid (0.33 mol/L) | W (or ACN) with 0.1% formic acid and 10 mmol/L ammonium formate. | Hypersil Gold C8 (50 × 2.1 mm; 5 μm) | MS/MS | (33) |
| HCQ | 50 μL of human blood | PP | ACN | 0.1% formic acid in W-ACN (94:6, v/v) | Agilent ZORBAX Eclipse XDB—C8 (50 × 2.1 mm; 5 μm) | MS/MS | (34) |
| HCQS | HCQS 200 mg gelatin hard capsules | – | Distilled water | MeOH:ACN:ammonium acetate 0.1 mol/L (45:15:40, v/v). | ZORBAX Extend C18 (150 x 4.6 mm; 5 μm) | FL | (35) |
| HCQ, DHCQ, DCQ & BDCQ | 10 μL of human whole blood | PP | Perchloric acid (70%) | 0.1% formic acid and 0.01% triethylamine in W (or ACN). | Kinetex C8 (2.1 × 50 mm; 2.6 μm) | MS/MS | (36) |
| CQ & DCQ | Human whole blood (50 μL) & plasma (100 μL) & DBS (50 μL) | DBS and SPE | Blood: 2% formic acid Plasma: ethyl acetate | ACN-ammonium formate 20 mM with 1% formic acid (15:85, v/v) | Zorbax SB-CN (50 mm × 4.6 mm; 3.5 μm) | MS/MS | (37) |
| CQ & DCQ | 150 μL human whole blood and 80 μL finger-pricked capillary blood spot | DBS and LLE | Hexane and tert-butyl methyl ether (1:, v/v) | 1% diethylamine, ACN and MeOH (20:55:25,v/v/v) | Thermo Hypersil Gold C18 (250 × 2.1 mm; 5 μm) | UV | (38) |
| HCQ, DCQ & DHCQ | 50 μL of human whole blood | PP | Zinc sulfate (0.1 M) and MeOH | Piperazine buffer (46.4 mM, pH = 9.8) and ACN | ACQUITY BEH Shield RP18 UPLC (1.7 × 100 mm; 2.1 μm) | FL | (40) |
| HCQ | 200 μL breast milk | PP | Methyl tert-butyl ether and sodium hydroxide | ACN-Phosphate buffer (13:87, v/v) | A Shim-pack CLC-C18 (6 mm × 15 cm; 5 μm) | FL | (41) |
| HCQ, DCQ & DHCQ | 200 μL of human whole blood | PP | Cold MeOH and cupric sulfate (3 mM) | Glycine buffer/NaCl (pH 9.7) and MeOH (46:54, v/v) | XTerra phenyl® (250 × 4.6 mm, 5 μm) | FL | (42) |
| HCQ, DHCQ,DCQ&BDCQ | 100 μL whole blood | PP | MeOH | W and MeOH with 0.1% formic acid | Hypersil Gold (50 mm × 3 mm; 3 μm) | MS/MS | (43) |
| CQ | 100 μL pig plasma | PP | ACN | MeOH-W (70:30, v/v, 3.5 mM ammonium formate and 0.2% formic acid) | HILIC Plus (100 × 4.6 mm; 3.5 μm) | MS/MS | (45) |
| HCQ, DHCQ, DCQ & BDCQ | 500 μL of human whole blood | LLE | NH3 and diethyl ether | W-MeOH–ACN (47:10:43, v/v/v) mixture containing 3.2 mM SDS | Luna C18 (150 × 4.6 mm; 5 μm) | FL | (46) |
| HCQ | 50 μL of human plasma | LLE | ACN | 0.6% formic acid aqueous solution:MeOH (80:20, v/v) | Phenomene × Kinetex C18 (50 × 2.1 mm; 2.6 μm) | MS/MS | (47) |
| HCQ | 100 μL of blood and 100 μL of plasma | LLE | Ammonium formate (pH 9.5) and methyl tert-butyl ether | ACN:20 mM ammonium formate (pH 3) | Phenomene × Gemini C18 (50 × 3 mm; 3 μm) | MS/MS | (48) |
| HCQ | 400 μL of animal plasma | LLE | Diethyl ether | W:(ACN:MeOH:50:50, v/v) (75:25, v/v) with sodium 1-pentanesulfonate and phosphoric acid | Hypersil C-18 (250 × 6 mm, 5 μm) | UV | (5) |
| HCQ | 180 μL human whole blood | LLE | Ammonium formate water soln containing 0.2% formic acid | Ammonium formate in MeOH or W; containing formic acid (70:30, v/v) | Phenomene × Kinetex PFP 100A (50 × 4.6 mm; 2.6 μm) | MS/MS | (49) |
| CQ & FSC | 100 μL liposome | LLE | Cold MeOH and chloroform | 0.2% formic acid in W & ACN | for CQ: Atlantis C18 (150 × 2.1 mm; 5 μm) for FSC: Luna C18 (150 × 2.0 mm; 5 μm) | MS/MS | (50) |
| HCQ | 1 mL of human blood serum | SPE | ACN | MeOH:W (70:30, v/v) | Eurospher II 100-5 C8 (150 x 4.6 mm; 5 μm) | UV | (53) |
| CQ & DCQ | 100 μL human plasma and 100 μL whole blood | SPE | Blood: ACN Plasma: 25 mM phosphate buffer (pH 7.0) | 25 mM potassium phosphate buffer pH 2.60:ACN (88:12) | Zorbax SB-CN (150 × 4.6 mm; 5 μm) | DAD | (54) |
| HCQ | Immediate-release formulation | – | 0.1 N HCl | W + Sodium Pentane Sulfonate+Phosphoric Acid (pH 2.5) and ACN | Kinetex XBC-18 (250 × 4.6 mm; 4 μm) | UV | (58) |
| HCQ | In-vitro: ester prodrugs of HCQ and five different aryl acetic acid NSAIDs In-vivo: wistar rats blood | – | Blood: EDTA Release: HCl buffer (pH 1.2) and phosphate buffer (pH 7.4) | Phosphate buffer (0.1 M, pH 3.0 ± 0.1):ACN (55:45, v/v) | Thermo Hypersil Gold C18 (250 × 4.36 mm; 5 μm) | UV | (61) |
| HCQ | 100 μL of human whole blood | – | 0.33 M perchloric acid | 10 mM ammonium formate and 0.1% formic acid in W or ACN. Isopropanol, ACN, acetone (40:40:20, v/v/v) | TFLC: Cyclone (50 × 0.5 mm) Analytical: Hypersil Gold C8 (50 × 2.1 mm; 3 μm). | MS/MS | (62) |
W = Water; ACN = Acetonitrile; MeOH = Methanol; API = active pharmaceutical ingredient.
Table III.
Analytical Method Validation Parameters for Determination of HCQ and/or CQ
| Method | Compound | LOD/LLOD | LOQ/LLOQ | Linearity | Recovery (%) | R 2 | Reference |
|---|---|---|---|---|---|---|---|
| LC–MS/MS | HCQ, DCQ, BDCQ & DHCQ | – | 1 ng mL−1 | 1–2,000 ng mL−1 | 84.1 ± 12.9 | 0.998 | (6) |
| LC–MS/MS | HCQ | – | – | 5.0–2,000 ng mL−1 | – | 0.998 | (33) |
| LC–MS/MS | HCQ | 2 ng mL−1 | 5 ng mL−1 | 5–2,000 ng mL−1 | >92 | 0.9999 | (34) |
| LC–FL | HCQS | 0.004 μg mL−1 (LOD) | 0.01 μg mL−1 (LOQ) | – | – | 0.9959 | (35) |
| LC–MS/MS | HCQ, DHCQ, DCQ & BDCQ | 5 ng mL−1 | 10 ng mL−1 (LLOQ) | – | – | 0.9907 | (36) |
| LC–MS/MS | CQ & DCQ | – | 1.82 ng mL−1 | – | 93–102 for whole blood 56–64 for DBS 69–80 for plasma | – | (37) |
| LC–UV | CQ & DCQ | – | 50 ng mL−1 for DBS 25 ng mL−1 for whole blood | 25–1,500 ng mL−1 | 74–87 | 0.997 | (38) |
| LC–MS/MS | CQ, Q, AQ, SD, MF, LF, PYM & PQ | 7 ng mL−1 | 20 ng mL−1 | – | 94.3–118.1 | >0.99 | (39) |
| UPLC–FL | HCQ, DCQ & DHCQ | 5 ng mL−1 | 10 ng mL−1 (LLOQ) | – | – | – | (40) |
| LC–FL | HCQ | – | – | 0.2–2 μg mL−1 | 90–110 | 0.9998 | (41) |
| LC–FL | HCQ, DCQ & DHCQ | 25 ng mL−1 | 50 ng mL−1 (LLOQ) | 50–4,000 ng mL−1 | ~55 | >0.995 | (42) |
| LC–MS/MS | HCQ, DHCQ, DCQ & BDCQ | – | 25 ng mL−1 | 25–2,000 ng mL−1 | >93 | >0.99 | (43) |
| LC–MS/MS | CQ & DCQ | – | 1.0 ng mL−1 | 1.0–500 ng mL−1 | 73.7 to 79.0 | >0.999 | (44) |
| HILIC–MS/MS | CQ | – | 1 ng mL−1 (LLOQ) | 1–100 ng mL−1 | 92–98 | >0.99 | (45) |
| RP LC–FL | HCQ, DHCQ, DCQ & BDCQ | 1 ng mL−1 (LLOD) | 20 ng mL−1 (LLOQ) | 1–2,500 ng mL−1 | – | – | (46) |
| LC–MS/MS | HCQ | – | 0.2 ng mL−1 (LLOQ) | 0.4–100 ng mL−1 | 91.87–97.84 | – | (47) |
| LC–MS/MS | HCQ | – | 1 ng mL−1 (LLOQ) | 1–1,000 ng mL−1 | – | 0.998 | (48) |
| LC–UV | HCQ | 240 ng mL−1 (LOD) | 840 ng mL−1 (LLOQ) | 100–2,000 ng mL−1 | > 95 | 0.9991 | (5) |
| LC–MS/MS | HCQ | – | – | 2–500 ng mL−1 | – | – | (49) |
| LC–MS/MS | CQ & FSC | 0.04 ppb | – | 0.1–100 ng mL−1 | – | 0.9942 | (50) |
| LC–DAD | CQ, DCQ & PQ | – | 10.32 ng mL−1 | 20–2,000 nM | 73–85.4 | 0.993 | (51) |
| UPLC–MS/MS | CQ & DCQ | – | 0.2 ng mL−1 | 0.2–1,000 ng mL−1 | 89.34–108.42 | 0.995 | (52) |
| LC–UV | HCQ | 0.2 μg L−1 | 0.67 μg L−1 | 1–300 μg L−1 | 96–103 | 0.9927 | (53) |
| LC–DAD | CQ & DCQ | 4 ng mL−1 | 10 ng mL−1 | 10–5,000 ng mL−1 | 77–82 for blood 92–89 for plasma | >0.999 | (54) |
| UPLC–DAD | CQ & PQ | 0.01 mg mL−1 | 0.03 mg mL−1 | 0.09–0.21 mg mL−1 | 98.0–102.2 | >0.99 | (59) |
| LC–DAD | CQ & PQ | 0.01 mg mL−1 | 0.03 mg mL−1 | 0.09–0.21 mg mL−1 | 98.0–102.2 | >0.99 | (59) |
| LC–DAD | CQ | 6 g mL−1 | 21 g mL−1 | 30–360 μg mL−1 | 98–100 | >0.99 | (60) |
| TFLC–MS/MS | HCQ | – | – | 15.7–4,000 ng mL−1 | – | – | (70) |
Methods
Sample preparation
For the sample preparation of CQ/HCQ different techniques were used including protein precipitation (PP) (6, 33, 34, 40–45) and several extraction methodologies like liquid–liquid extraction (LLE) (5, 38, 39, 46–52), solid-phase extraction (SPE) (37, 38, 53, 54), 96-well plate LLE (55, 56), microextraction techniques (57) as well as different sample collection techniques as volumetric absorptive microsampling (VAMS) (36) and dried blood spotting (DBS) (36–39).
Protein precipitation
PP is a commonly employed technique relatively crude but rapid sample clean-up can be accomplished by the addition of an organic solvent, a non-ionic surfactant and inorganic salts or certain metals, subsequently evaporation of supernatant under a nitrogen stream and reconstitution of the residue in the mobile phase (66, 67). For the analysis of HCQ and its metabolites in whole blood; zinc sulphate and methanol (40), perchloric acid (36) and methanol (43, 49) had been used as PP reagent. Determination of HCQ in breast milk was accomplished by using methyl tert-butyl ether and NaOH together with CQ as internal standard (41). Parvinizadeh et al. (53) applied PP by using acetonitrile to serum sample, the study also includes extraction processes of HCQ by using (Ni@MIL-100(Fe)@MIP) sorbent at pH 9, shaking followed by collecting the sorbent by an external magnet and removing aqueous solution, adding methanol as eluent solvent and subsequent analysis by LC. HCQ and DHCQ determined in human whole blood samples by LC–MS/MS using methanol as PP reagent (49). PP performed by dilution of the sample with 1% formic acid solution and addition of ice-cold ACN to mouse blood and tissue samples (6). Due to the quenching fluorescence of HCQ at high pH (42) cold MeOH and cupric sulphate (3 mM) were used as a PP agent instead of perchloric acid (33). One hundred percent ACN (45, 47) was preferred as PP reagent among 100% MeOH (44), 10% aqueous zinc sulphate solution in ACN and in MeOH (5: 95, v/v) solutions due the highest mass to signal ratio of the analyte (34).
Solid-phase extraction
SPE is a powerful sample preparation technique providing selectivity, flexibility, relatively simple procedures, availability of repacked cartridges and high automation potential (68) and could be both used for the enrichment of the analytes or the interferences. HCQ was determined in blood samples via a VAMS device (tip) made from a proprietary hydrophilic polymer served as a sorbent of the SPE (36). Both plasma and whole blood samples were extracted on 96-fixed well plates by pretreatment of ACN, further treatment with PB pH 7.0 and ACN mixture, finally elution with ACN-formic acid (95: 5, v/v) mixture (54). An automated liquid handler platform used to process whole blood, plasma or punched discs of DBS samples spiked into 96-well plates as well as the removal of phospholipids, solid–liquid extraction to determine CQ and DCQ (37).
Liquid–liquid extraction
LLE is a simple technique consisting of the transfer of a solute from one solvent to another and mixing followed by a phase separation. It provides high selectivity and clean sample extracts as well as simplicity and low cost, it is attractive and widely used method to separate compounds of different polarities (55, 69). HCQ and its metabolites were determined in whole blood by LLE using diethyl ether (46). Pharmacokinetics of HCQ in Japanese patients suffering SLE was carried out by using tert-butyl ether as extraction solvent and samples were further processed prior to LC–MS/MS analysis (48). In another study, HCQ samples were extracted with ammonium solution and diethyl ether where CQ was used as internal standard (5), similarly HCQ was determined in whole blood using 0.33 M perchloric acid (70). The 96-well LLE plate makes possible on-going and efficient extraction of analyte from an aqueous biological matrix and organic extraction solvent by using inert diatomaceous earth particles filled in a 96-well filter plate (56). 96-well LLE is superior in terms of price compared to SPE as well as LC–MS/MS compatibility and low consumption of organic solvents compared to SPE and LLE. PP with zinc sulphate and subsequent use of a 96-well filtration plate applied to whole blood samples containing HCQ and its metabolites. Lipids and phospholipids were removed with water prior to 96-well filtration (40). Following LLE with cold MeOH and chloroform final extracts of HCQ and its metabolites in liposome samples were processed through 96-well filtration prior to LC–MS/MS analysis (50). A mixture of n-hexane and ethyl acetate for CQ, DSCQ and PQ determination in human plasma (51); hexane and tert-butyl methyl ether mixture for the analysis of whole blood and DBS samples (38) and methyl t-butyl ether (MTBE)/isooctane for the extraction of plasma samples (52) were used as LLE solvents.
Volumetric absorptive microsampling
VAMS is a new microsampling technique using a device consisted of a holder and a tip (63). When the tip of the VAMS device is dipped into the blood, a fixed amount of it has been absorbed by capillary action. Qu et al. used the VAMS technique for the sampling of HCQ and its metabolites in capillary blood by fingerpick, device was dried on a dedicated rack, subsequently, samples were easily extracted from water by mixing with internal standard and perchloric acid as PP reagent. The use of VAMS device for the sample preparation of HCQ and its metabolites resulted in an accurate 10 μL sample collection (36).
Dried blood spotting
DBS is a non-invasive and simple blood sampling method comforting patient compared to conventional venous blood sampling (64). It is safe for both the subject and the worker as well as its storage and transportation which does not require cold chain or fragmentation, which is a big concern involved in plasma, serum or whole blood sampling (38). Besides, it can easily be used in children due to its applicability with a small amount of blood (37). DBS processing involves a collection of blood; preparation of blood spots; drying of blood spots; elution of analytes from DBS card and analyses of DBS eluates (65). Qu et al. (36) used both DBS and VAMS techniques for therapeutic drug monitoring (TDM) of HCQ and its metabolites. The capillary blood was collected from 44 patients using both techniques. Minimal volume of blood to fill DBS cards were not supplied by about 20% of the patients. However, all patients’ blood was collected successfully with the VAMS device, it could possibly due to a 10 μL small volume of blood enough for the VAMS device. It was also reported that due to difficulty in spreading the blood homogeneously in DBS cards and spot size’s being dependent on blood viscosity, the VAMS device was chosen over DBS cards (36). Cheomung et al. (38) worked with DBS cards to separate CQ and DCQ in whole blood, using hexane and tert-butyl methyl ether mixture. Five pressed discs were used which is equivalent to 15 μL of whole blood into 96-well plate (37). Several antimalarial drugs and two active metabolites were analyzed using 10 μL of DBS sample providing an advantage in transportation and storage (39).
Liquid chromatographic methods
Recently developed LC methods to determine CQ/HCQ and related metabolites in either biological fluids or in pharmaceutical preparations were categorized by detection types as UV spectrometric, fluorimetric and tandem mass spectrometric methods. Several different wavelengths were used for the UV detection such as 254 nm (53), 343 nm (5, 54), 240 nm (61), 254 nm (58), 343 nm (5), 331 nm (51), 256 nm (38), 260 nm (59), 250 nm (60) as well as different flow rates 0.8 mL (53, 61), 1.0 mL (38, 51, 58–60), 1.2 mL (54) and 2.0 mL (5). Fluorimetric detections were performed using 335 nm (40), 320 nm (46), 325 nm (35), 337 nm (41), 320 nm (42) for excitation and 390 nm (40), 370 nm (46), 375 nm (35), 371 nm (41) 380 nm (42) for emission wavelengths, respectively. Ambient temperature as well as 40°C (40, 46), 50°C (42) were used as column temperature to obtain better separation of analytes of interest. Electrospray ionization and multiple reaction monitoring were performed with positive mode in most of the developed LC–MS/MS methods (6, 33, 37, 39, 43–45, 47, 49, 50, 52, 70).
The overall chromatographic conditions of CQ/HCQ determination and the sample preparation procedures used by the researchers between the years [2010 and 2020] (including brackets) were summarized in Table II.
Results
For all the methods summarized in Table II, the analytical method validation parameters were given in Table III represented by linearity, recovery, LOD, LOQ and regression coefficient.
Liquid chromatographic methods based on ultraviolet-spectrometric detection
HCQ and CQ were well separated with good shape peaks. After single IV administration of drug, the mean values of different pharmacokinetic parameters were calculated and the calculated AUC was higher as compared to the given i.v. dose, so this difference proves the nonlinear pharmacokinetic (5). The developed methods for the determination of commonly prescribed DMARDs in biological fluids including HCQ were summarized (31). Dispersive microsolid-phase extraction (DMSPE) method was used for HCQ determination sulphate and a new MIP sorbent was synthesized. The effects of various parameters by a Box–Behnken design and combined by a desirability function. The method also facilitated the process by reducing time consumption applying to real human serum samples followed by LC-UV (53). To minimize the HCQ accumulation in non-targeted sites ester drugs were synthesized and them in vivo behavior, in vitro stability and hydrolysis kinetics were evaluated in aqueous buffers and tissue homogenates of rats. Intact pro-drug peaks were observed by LC analysis (61). The dissolution method in the USP monograph for HCQ was developed for water-based suspension formulation which was improved for precision in dosing and conformity in pediatric patients (58). CQ and DSCQ were determined in 9 P. vivax malaria patients administered 25 mg/kg of CQ. Extraction recoveries were found >70% for CQ and DSCQ. Stock solutions were stable for 20 days at −14°C as well as plasma samples for 6 h at room temperature and 90 days at −20°C (51). A SPE-LC method was developed for CQ and DCQ determination in whole blood and plasma samples obtained from 14 Plasmodium vivax patients given CQ at a dose of 25 mg base/kg. The optimum concentration of sodium perchlorate in mobile phase mixture was found as 2 mM, analytes were ideally separated in 10 min. The stability of drugs plasma samples was studied at ambient temperature in the autosampler and in freezer at −20 and − 80°C (54). Determination of CQ and its active plasma metabolite DECQ were performed by using finger-pricked capillary in blood samples of 10 Thai patients with P. vivax malaria given 2,000 mg CQP. UV detection was shown to be as sensitive as fluorescence detection (38). A UPLC-DAD method was developed for simultaneous quantitation of CQ and primaquine in tablet formulations and compared with LC. UPLC and HPLC techniques show very small differences between the results of retention time and tailing factor of peaks. UPLC method (1.5 min) took almost half of the time needed for the regular HPLC method (3.0 min) (59).
Liquid chromatographic methods based on fluorimetric detection
HCQ was separated from its two major metabolites and other unknown peaks in whole blood samples obtained from 37 lupus patients dosing with 200–600 mg/day HCQ. Analytical performance of the method was evaluated according to FDA guidelines. No significant carry-over observed following 2,000 ng mL−1 injection of HCQ, DCQ and DHCQ with satisfactory accuracy and precision results within 15% RSD. However, the method provides a relatively long runtime of 18 min compared to another method using 1.2 mL min−1 of flow rate. Cold MeOH and cupric sulphate mixture were used as PP reagent. Method was validated including freeze-thaw, short-term and postpreparative stability studies of HCQ and its metabolites (42). Quantification of HCQ and its metabolites in whole blood samples of 85 patients with lupus, administered 100–400 mg of HCQ was performed by UPLC. A simple PP was used by 96-well plate and satisfactory LLOQs as compared to LC–MS/MS allowing TDM in children (40). An ion-pairing LC method was developed for HCQ, DHCQ, DCQ and BDCQ, in whole blood samples obtained from SLE patients and undergoing HCQ treatment. Same samples were used by another LC/MS–MS method developed and two methods confirmed each other. Sodium dodecyl sulphate was used as ion-pairing reagent and the column oven temperature was set at 40°C to increase resolution and to reduce peak tailing. Whole blood from samples were diluted with water to lyse the erythrocytes and alkalized to increase the recovery; however, obtained recoveries were very low as between 25 and 60% compared to other techniques (46). The compatibility of HCQ with tablet excipients was evaluated using thermal and nonthermal stress testing including sample storage during a month under 75% RH and 40°C conditions and the developed LC-FL method was conducted for quantification (35). HCQ was determined in breast milk samples obtained from 33 patients after administration of 200–400 mg doses. No significant difference was obtained indicating linear PK process for HCQ; however milk composition, milk pH and polymorphism of CYP2D6 were not investigated in our study (41).
Liquid chromatographic methods based on mass spectrometric detection
A forced degradation with alkaline hydrolysis and oxidation was studied by UPLC-UV-MS/MS for CQ determination in tablets and resulted with the forming of two degradation products (47). A 7 min method was developed for the determination of HCQ in human blood samples by TFLC–MS/MS using a simple PP using perchloric acid. Observed carry-over was minimized by including a wash step after elution of the analytes (70). A bioequivalence study for two different formulations of HCQ, LC–MS/MS method was performed in human whole blood using pentafluorophenyl column with functional side chain interacting with quinoline group of HCQ to enhance the resolution (49). Mouse blood and several tissues were used in a pharmacokinetics and tissue distribution study following a single i.v. dose of HCQ (5 mg/kg). Obtained data showed the accumulation of HCQ in tissues; however, further toxicological studies are required to investigate the relation between drug concentration and effect both therapeutic and toxic, after repeated dosing (6). Another bioequivalence study of a generic with reference formulation of HCQ was performed in healthy subjects dosed with 0.2 g of HCQS tablets. The stability of HCQ in plasma samples was evaluated at room temperature and with a freeze-thaw study. Although the formulations were assumed bioequivalent as result the study was not performed in patients in clinical practice (47). In a clinical study blood and plasma drug concentration-time data of patients dosed with HCQS of 200–400 mg/day were analyzed using nonlinear mixed-effects model software and a stability study was performed as well (48). To reevaluate results obtained with plasma samples over whole blood, a turbulent flow LC–MS/MS method was developed for more robust assays. Online sample clean-up by TFLC allows depletion of high molecular weight matrix while retaining HCQ and metabolites. The developed method reduced total sample processing time to ~5 min with minimal carry-over increasing lab efficiency (34). To quantify HCQ concentration in whole blood samples of patients suffering small cell lung cancer LC–MS/MS was used in a phase-I clinical trial. The method offers a large linearity within the range of 5–2,000 ng mL−1 and short analysis with 3 min runtime (33). An overdose HCQ consumption in a suicide attempt was determined by an LC–MS/MS method using PP as a sample preparation technique. HCQ and its metabolites were quantified within a range of 75–97% recovery and a stability assay was performed (43). Antimalarial drug fosmidomycin and CQ was determined successfully in the liposomal preparations with a LOD level of 0.04 ppb (50). Simultaneous determination of CQ and DCQ in human plasma samples was performed with two sample preparation techniques. PP and LLE results were compared to minimize the carry-over and to increase recovery. PP was chosen by means of higher recovery and sensitivity, a stability and photodegradation study was conducted as well (44). HILIC–MS/MS was used to determine CQ in the plasma of miniature pigs 0.2 g CQ orally administered. Good peak shaped and resolution were obtained by the addition of ammonium formate and formic acid to the mobile phase mixture. Although the validation results were satisfactory, using two pigs not provided adequate data for a statistical pharmacokinetic study (45). CQ and DCQ were determined from whole blood, plasma and DBS samples using automated liquid handler platform into 96-well plates following a phree phospholipids removal 96-well plate. Samples were prepared with CBA-fixed SPE 96-well plate cartridges and solid–liquid extraction; 96-well plate and LLE for whole blood and plasma, respectively. Results obtained with DBS were higher due to the accumulation of CQ in red blood cells (37). Plasma samples obtained from 30 RA patients were prepared by LLE and the effect of lithium heparin and citrate phosphate dextrose as anticoagulants were compared and no significant difference was found (52). Simultaneous determination of seven antimalarial drugs including CQ in healthy subjects were performed by LC–MS/MS. DBS was used for sample preparation and effects of punching position, blood spot volume and hematocrit on DBS sampling were assessed. Hematocrit value should be standardized to get more accurate results with DBS (39).
Discussion
Sample preparation is the longest, the most time consuming and tedious process of an analysis. The easier technique of sample preparation methods is to convert a matrix into a sample suitable for analysis. This process is inevitably changing the interaction between components and their chemical environment which are detected through the physical and chemical properties of the analyte and matrix, affecting the recovery and reproducibility. The solubility of the molecule and the specific functional groups should be evaluated to find the appropriate sample preparation and chromatographic separation technique (31). Sulphate and hydrochloride form has the best solubility in water, respectively, for HCQ and CQ (12, 22, 31), and a better separation is obtained mostly when reversed-phase-based liquid chromatography is used due to the polar groups embedded in the chemical structure. The aqueous medium is the first solvent preferred to solve a drug substance due to the environmental friendly chemistry and the polar character of the small molecules. Since HCQ interacts with erythrocytes (32), in most of the studies (33, 34) the quantitation of HCQ and metabolites was performed in whole blood over plasma/serum, which is simpler matrix types compared to whole blood.
Determination of drugs in biologic fluids has a great importance due to TDM demonstrating therapeutic efficacy, pharmacokinetic and pharmacodynamics properties, severe side effects caused by elevated blood concentration as well as epidemiological studies. LC-based methods are primarily applied separation techniques for the analysis of CQ and HCQ as well as their metabolites from whole blood, plasma and/or pharmaceutical preparations. Different types of reversed-phase columns were used such as C18 (5, 6, 35, 38, 40, 41, 43, 44, 46, 50–52, 58–61, 70), C8 (33, 34, 36, 53), CN (37, 54), HILIC (45) and phenyl (42); however, C18 was the most used one. C18, C8 and octadecyl groups, including silica, can contribute to prevent sacrificing the peak shapes for CQ, HCQ and related metabolites.
Since CQ and HCQ possess conjugate double bonds and functional groups, they could be detected fluorimetrically and more sensitive methods were developed as compared to UV detections. The distribution of sample preparation techniques and LC methods with different detections are given in Figure 2.
Figure 2.

The distribution of sample preparation techniques and liquid chromatographic methods with different detections.
CQ and HCQ elute quickly (in 7 min or less) owing to the polar character and small molecule structure in most of the studies. Residual silanol groups in the silica-based column materials may interact with analyte molecules at neutral or low pH values thus resulting mostly in peak tailing (46). Since CQ, HCQ and related metabolites are basic compounds, the addition of trimethylamine (36, 40) or diethylamine (38) as a strong base to the mobile phase could reduce the charging of silanol groups with analytes having polar tertiary amine groups. However, this approach may change the character of the column and the use of strong basic mobile phases are needed for the elution of drugs which may deteriorate the column. To increase the ionic strength of the mobile phase phosphoric acid (5, 51, 58, 61) or phosphate buffer (41, 60, 61) could be added to the mobile phase which could prevent secondary retention. These two approaches with nonvolatile acids or buffers are only suitable with UV and FL detection could damage MS. Increasing the formic acid concentration in the mobile phase causes a decrease in the interaction with the column due to the basic character of the analytes (45). Since the retention of polar compounds are weak in reversed-phase LC and adding strong ion-pairing reagents to mobile phases could harm MS and column, HILIC was offered as an alternative with its higher separation power (45).
LC coupled UV methods are mostly used techniques in drug determination (71, 72) due to their ease to use and cheapness; however, low sensitivity and selectivity provided are not enough for clinical studies most of the time. FL detection is more selective and sensitive as compared to UV and may be as effective as mass spectrometric methods in biological fluid samples containing very low amounts of drug (73, 74). Although the instrument is relatively sophisticated and expensive, mass spectrometric detection provides a high-throughput analysis of large sample batches even without preconcentration with repeatable results since most of the drug substances do not have enough double bonds or functional groups to exhibit FL (75, 76). Solvent consumption could be reduced by downscaling conventional PP, LLE and SPE (57). Due to high sensitivity, low LLOQ, and short runtimes LC–MS/MS would be preferred over UV and DAD detections especially.
Conclusion
There is still no special drug or vaccine available to cure COVID-19; however, promising drug candidates and a variety of vaccines are currently in different stages of clinical trials. HCQ and CQ were used widely for the treatment although they have the limitations of toxicity. Liquid chromatographic techniques are the first methods preferred for quantitation, TDM as well as pharmacokinetics. Since many liquid chromatographic methods are described for the determination of HCQ/CQ in pharmaceutical dosage forms and biological samples, this review is intended to provide an overview of developed methods available between 2010 and 2020 focusing on different sample preparation techniques, detection modes and chromatographic conditions, which will help researchers on method development studies.
Manuscript Preparation Criterion
Electronic searches were conducted in PubMed, Google Scholar, Science Direct, ACS Publications, Web-of-Science and Research Gate databases by searching for keywords related to HCQ, CQ, determination of HCQ, determination of CQ on July 5–20, 2020. The electronic searches were limited only between the years 2010 and 2020, including the years 2010 and 2020. However, only some of these publications are used for bringing scientific relevance about the study topic. Sample preparation and detection methods were reviewed and broadly summarized in this article.
Acknowledgement
This review was prepared as a final project of a graduate course entitled “Sample Preparation in Pharmaceutical and Biological Samples, ANKI7052” in the Department of Analytical Chemistry, Faculty of Pharmacy at Istanbul University-Istanbul, Turkey.
Contributor Information
Zeynep Derya Bilgin, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Isil Evcil, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Didem Yazgi, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Gokce Binay, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Ceren Okuyucu Genc, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Busra Gulsen, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Aytaj Huseynova, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Ayse Zehra Ozdemir, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Emel Ozmen, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Yakup Usta, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Suade Ustun, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
Sena Caglar Andac, Department of Analytical Chemistry, Faculty of Pharmacy, Istanbul University, Suleymaniye, 7-1, 34116 Fatih/Istanbul, Turkey.
REFERENCES
- 1. Law S., Leung A.W., Xu C.; Severe acute respiratory syndrome (SARS) and coronavirus disease-2019 (COVID-19): From causes to preventions in Hong Kong; International Journal of Infectious Diseases, (2020); 94: 156–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. WHO ; WHO Coronavirus Disease (COVID-19) Dashboard Overview. (2020) https://covid19.who.int/ (accessed July 25, 2020).
- 3. Ferner R.E., Aronson J.K.; Chloroquine and Hydrochloroquine in covid 19; BMJ, (2020); 369: 1–2. [DOI] [PubMed] [Google Scholar]
- 4. WHO ; WHO Q&A: Hydroxychloroquine and COVID-19 (2020). https://www.who.int/news-room/q-a-detail/q-a-hydroxychloroquine-and-covid-19 (accessed July 25, 2020).
- 5. Singh A., Roopkishora S.C.L., Gupta R., Kumar S., Kumar M.; Development and validation of reversed-phase high performance liquid chromatographic method for hydroxychloroquine sulphate; Indian Journal of Pharmaceutical Sciences, (2015); 77: 586–591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chhonker Y.S., Sleightholm R.L., Li J., Oupicky D., Murry D.J.; Simultaneous quantitation of hydroxychloroquine and its metabolites in mouse blood and tissues using LC–ESI–MS/MS: An application for pharmacokinetic studies; Journal of Chromatography B, (2018); 1072: 320–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Drugbank ; Drugs. Hydroxychloroquine, (2020), https://go.drugbank.com/drugs/DB01611 (accessed July 2, 2020). Accession Number: DB01611.
- 8. Sahraei Z., Shabani M., Shokouhi S., Saffaei A.; Aminoquinolines against coronavirus disease 2019 (COVID-19): Chloroquine or hydroxychloroquine; International Journal of Antimicrobial Agents, (2020); 55: 4–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Salata C., Calistri A., Parolin C., Baritussio A., Palu G.; Antiviral activity of cationic amphiphilic drugs; Expert Review Anti Infective Theraphy, (2017); 15: 483–492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Vincent M.J., Bergeron E., Benjannet S.; Chloroquine is a potent inhibitor of SARS coronavirus infection and spread; Virology Journal, (2005); 2: 2–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Jordan P., Brookes J.G., Nikolic G., Le Couteur D.G.; Hydroxychloroquine overdose: Toxicokinetics and management; Journal of Toxicology-Clinical, (1999); 37: 861–864. [DOI] [PubMed] [Google Scholar]
- 12. Verbeeck R.K., Junginger H.E., Midh K.K., Shah V.P., Barends D.M.; Biowaiver monographs for immediate release solid oral dosage forms based on biopharmaceutics classification system (BCS) literature data: Chloroquine phosphate, chloroquine Sulfate, and chloroquine hydrochloride; Journal of Pharmaceutical Sciences, (2005); 94: 1389–1395. [DOI] [PubMed] [Google Scholar]
- 13. Moura Ferraz L.R., Santos F.L.A., Ataide Ferreira P., Junior R.T.L.M., Rosa T.A., Rolim L.A. et al. ; Clinical, pharmacokinetic and technological aspects of the hydroxychloroquine sulfat; IOSR Journal of Pharmacy, (2014); 4: 53–64. [Google Scholar]
- 14. Lim H., Im J., Cho J., Bae K., Klein T.A., Yeom J. et al. ; Pharmacokinetics of hydroxychloroquine and its clinical implications in chemoprophylaxis against malaria caused by plasmodium vivax; Antimicrobial Agents and Chemotheraphy, (2009); 53: 1468–1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Brocks D.R., Passutto F.M., Jamali F.; Analytical and semi-preparative high-performance liquid chromatographic separation and assay of hydroxychloroquine enantiomers; Journal of Chromatography-Biomedical Application, (1992); 581: 83–92. [DOI] [PubMed] [Google Scholar]
- 16. Moore D.E., Hemmens V.J.; Photosensitization by anti-malarial drugs; Photochemistry and Photobiology, (1982); 36: 71–77. [DOI] [PubMed] [Google Scholar]
- 17. Moore D.E., Tamat S.R.; Photosensitizing by drugs: Photolysis of some chlorine-containing drugs; Journal of Pharmacy and Pharmacology, (1980); 32: 172–177. [DOI] [PubMed] [Google Scholar]
- 18. Tønnesen H.H., Grislingaas A.L., Woo S.O., Karlsen J.; Photochemical stability of antimalarials; International Journal of Pharmaceutics, (1988); 43: 215–219. [Google Scholar]
- 19. Shravani P., Snigdha D.; Method development and validation for estimation Hydroxy chloroquine sulphate by UV-spectrometry; International Journal of Trends in Pharmacy and Life Sciences, (2016); 2: 1007–1017. [Google Scholar]
- 20. Drugbank ; Drugs. Chloroquine, (2020), https://go.drugbank.com/drugs/DB00608 (accessed July 15, 2020). Accession Number: DB00608.
- 21. Browning D.J.; Chapter 2: Pharmacology of chloroquine and hydroxychloroquine In Hydroxychloroquine and Chloroquine Retinopathy. Springer Science+Business Media, New York, NY, (2014), pp. 35–63. [Google Scholar]
- 22. Ph. Eur ; Hydroxychloroquine sulfate monograph, 01/2017:2849 corrected 10.0, 2017, pp. 2896–2897.
- 23. Schroeder R.L., Gerber J.P.; Chloroquine and hydroxychloroquine binding to melanin: Some possible consequences for pathologies; Toxicology Reports, (2014); 1: 963–968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Warhurst D.C., Steele J.C.P., Adagu I.S., Craig J.C., Cullander C.; Hydroxychloroquine is much less active than chloroquine against chloroquine-resistant plasmodium falciparum, in agreement with its physicochemical properties; Journal of Antimicrobial Chemotheraphy, (2003); 52: 188–193. [DOI] [PubMed] [Google Scholar]
- 25. Gustafsson L.L., Walker O., Alvan G., Beermann B., Estevez F., Gleisner L. et al. ; Disposition of chloroquine in man after single intravenous and oral doses; British Journal of Clinical Pharmacology, (1983); 15: 471–479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Fan H., Ma Z., Chen J., Yang X., Cheng J., Li Y.; Pharmacokinetics and Bioquivalence study of hydroxychloroquine Sulfate tablets in Chinese healthy volunteers by LC-MS/MS; Rheumatology Therapy, (2015); 2: 183195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Pereira D., Daher A., Zanini G., Maia I., Fonseca L., Pitta L. et al. ; Safety, efficacy and pharmacokinetic evaluations of a new coated chloroquine tablet in a single-arm open-label non-comparative trial in Brazil: A step towards a used-friendly malaria vivax treatment; Malaria Journal, (2016); 15: 1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Tett S., Day R., Cutler D.; Hydroxychloroquine relative bioavailability: Within subject reproducibility; British Journal of Clinical Pharmacology, (1996); 41: 244–246. [DOI] [PubMed] [Google Scholar]
- 29. Schrezenmeier E., Dörner T.; Mechanisms of action of hydroxychloroquine and chloroquine: Implications for rheumatology; Nature Reviews Rheumatology, (2020); 16: 155–166. [DOI] [PubMed] [Google Scholar]
- 30. Ducharme J., Farinotti R.; Clinical pharmacokinetics and metabolism of chloroquine, focus on recent advancements; Clinical Pharmacokinetics, (1996); 31: 257–274. [DOI] [PubMed] [Google Scholar]
- 31. Dogra A., Sharma A., Mandal U.K., Kotwal P., Bhatt S., Nandi U.; Liquid chromatography based methods for analysis of disease-modifying Antirheumatic drugs (DMARDs) in biological matrices; Critical Reviews in Analytical Chemistry, (2019); 49: 224–242. [DOI] [PubMed] [Google Scholar]
- 32. Ferrari V., Cutler D.J.; Uptake of chloroquine by human Erythrocyles; Biochemical Pharmacology, (1990); 39: 753–762. [DOI] [PubMed] [Google Scholar]
- 33. Füzéry A.K., Breaud A.R., Emezienna N., Schools S., Clarke W.A.; A rapid and reliable method for the quantitation of hydroxychloroquine in serum using turbulent flow liquid chromatography-tandem mass spectrometry; Clinica Chimica Acta, (2013); 421: 79–84. [DOI] [PubMed] [Google Scholar]
- 34. Wang L.Z., Ong R.Y.L., Chin T.M., Thuya W.L., Wan S.C., Wong A.L.A. et al. ; Method development and validation for rapid quantification of hydroxychloroquine in human blood using liquid chromatography–tandem mass spectrometry; Journal of Pharmaceutical and Biomedical Analysis, (2011); 61: 86–92. [DOI] [PubMed] [Google Scholar]
- 35. Moraes A.N.F., Silva L.A.D., Aparecida de Oliveira M., Magno de Oliveira E., Nascimento T.L., Lima E.M. et al. ; Compatibility study of hydroxychloroquine sulfate with pharmaceutical excipients using thermal and nonthermal techniques for the development of hard capsules; Journal of Thermal Analysis and Calorimetry, (2020); 140: 2283–2292. [Google Scholar]
- 36. Qu Y., Brady K., Apilado R., Q’Malley T., Reddy S., Chithara P. et al. ; Capillary blood collected on volumetric absorptive microsampling (VAMS) device for monitoring hydroxychloroquine in Rhematoid arthritis patients; Journal of Pharmaceutical and Biomedical Analysis, (2017); 140: 334–341. [DOI] [PubMed] [Google Scholar]
- 37. Kaewkhao K., Chotivanich K., Winterberg M., Day N.P.J., Tarning J., Blessborn D.; High sensitivity methods to quantify chloroquine and its metabolite in human blood samples using LC-MS/MS; Bioanalysis, (2019); 11: 333–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cheomung A., Na-Bangchang K.; HPLC with ultraviolet detection for the determination of chloroquine and desethylchloroquine in whole blood and finger prick capillary blood dried on filter paper; Journal of Pharmaceutical and Biomedical Analysis, (2011); 55: 1031–1040. [DOI] [PubMed] [Google Scholar]
- 39. Gallay J., Prod’hom S., Mercier T., Bardinet C., Spaggiari D., Pothin E. et al. ; LC–MS/MS method for the simultaneous analysis of 7 antimalarials and two active metabolites in dried blood spots for applications in field trials: Analytical and clinical validation; Journal of Pharmaceutical and Biomedical Analysis, (2018); 154: 263–277. [DOI] [PubMed] [Google Scholar]
- 40. Noé G., Amoura Z., Combarel D., Lori L., Tissot N., Seycha A. et al. ; Development and validation of a fast ultra-high performance liquid chromatography-fluorescent method for the quantification of hydroxychloroquine and its metabolites in patients with lupus; Therapeutic Drug Monitoring, (2019); 41: 476–482. [DOI] [PubMed] [Google Scholar]
- 41. Peng W., Liu R., Zhang L., Fu Q., Mei D., Du X.; Breast milk concentration of hydroxychloroquine in Chinese lactating women with connective tissue diseases; European Journal of Clinical Pharmacology, (2019); 75: 1547–1553. [DOI] [PubMed] [Google Scholar]
- 42. Qu Y., Noe G., Breaund A.R., Vidal M., Clarke W.A., Zahr N. et al. ; Development and validation of a clinical HPLC method for the quantification of hydroxychloroquine and its metabolites in whole blood; Future Science QA, (2015); 1: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Soichot M., Mégarbane B., Houzé P., Chevillard L., Fonsart J., Baud F.J. et al. ; Development, validation and clinical application of a LC-MS/MS method for the simultaneous quantification of hydroxychloroquine and its active metabolites in human whole blood; Journal of Pharmaceutical and Biomedical Analysis, (2014); 100: 131–137. [DOI] [PubMed] [Google Scholar]
- 44. Tang C., Sojinu O.S.; Simultaeous determination of chloroquine and its metabolite desethyl chloroquine in human plasma using liquid chromatography tandem mass spectrometry; Analytical Letters, (2012); 45: 2277–2289. [Google Scholar]
- 45. Wang Z.Z., Lu H.Y., Shang D.W., Ni X.J., Zhang M., Wen Y.G.; Development and validation of an HILIC–MS/MS method by one-step precipitation for chloroquine in miniature pig plasma; Bioanalysis, (2016); 8: 1159–1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Charlier B., Pingeon M., Piaz F.D., Conti V., Valentini G., Filippelli A. et al. ; Development of a novel ion-pairing HPLC-FL method for the separation and quantification of hydroxychloroquine and its metabolites in whole blood; Biomedical Chromatography, (2018); 32: 4258–4268. [DOI] [PubMed] [Google Scholar]
- 47. Fan H., Ma Z., Chen J., Yang X., Cheng J., Li Y.; Pharmacokinetics and bioequivalence study of hydroxychloroquine Sulfate tablets in Chinese healthy volunteers by LC–MS/MS; Rheumatoogy And Therapy, (2015); 2: 183–195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Morita S., Takahashi T., Yoshida Y., Yokota N.; Population pharmacokinetics of hydroxychloroquine in Japanese patients with cutaneous or systemic lupus erythematosus; Therapeutic Drug Monitoring, (2016); 38: 259–267. [DOI] [PubMed] [Google Scholar]
- 49. Park J.Y., Song H.H., Kwon Y.E., Kim S.J., Jang S., Joo S.S.; Development and validation of LC-MS/MS for bioanalysis of hydroxychloroquine in human whole blood; Journal of Biomedical Translational Research, (2018); 19: 130–139. [Google Scholar]
- 50. Urbán P., Estelrich J., Adeva A., Cortés A., Busquets X.F.; Study of the efficacy of antimalarial drugs delivered inside targeted immunoliposomal nanovectors; Nanoscale Research Letters, (2011); 6: 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Idárragaa L.Z., Jiméneza N.Y., Córdobab C.L., Trujillo S.B.; Validation of a method for the simultaneous quantification of chloroquine, desethylchloroquine and primaquine in plasma by HPLC-DAD; Journal of Pharmaceutical and Biomedical Analysis, (2014); 95: 200–206. [DOI] [PubMed] [Google Scholar]
- 52. Boonprasert R., Sri-in J., Pongnarin P., Chatsiricharoenkul S., Chandranipapongse W.; Development of the liquid chromatography tandem mass spectrometry method for determination of chloroquine and Desethylchloroquine in human plasma; Siriraj Med Journel, (2012); 64: 47–51. [Google Scholar]
- 53. Parvinizadeh F., Daneshfar A.; Fabrication of a magnetic metal–organic framework molecularly imprinted polymer for extraction of anti-malaria agent hydroxychloroquine; New Journal of Chemistry, (2019); 43: 8508–8516. [Google Scholar]
- 54. Pham T.V., Nguyen P.P., Khanh T.N.D., Thuy N.N.T., Nha C.N.T., Pouplin T. et al. ; An HPLC method with diode array detector for the simultaneous quantification of chloroquine and desethylchloroquine in plasma and whole blood samples from plasmodium vivax patients in Vietnam, using quinine as an internal standard; Biomedical Chromatography, (2016); 30: 1104–1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Steinborner S., Henion J.; Liquid-liquid extraction in the 96-well plate format with SRM LC/MS quantitative determination of methotrexate and its major metabolite in human plasma; Analytical Chemistry, (1999); 71: 2340–2345. [DOI] [PubMed] [Google Scholar]
- 56. Peng S.X., Branch T.M., King S.L.; Fully Auromated 96-well liquid-liquid extraction for analysis of biological samples by liquid chromatography with tandem mass spectrometry; Analytical Chemistry, (2001); 73: 708–714. [DOI] [PubMed] [Google Scholar]
- 57. Hansen F., Øiestad E.L., Bjergaard S.P.; Bioanalysis of pharmaceuticals using liquid-phase microextraction combined with liquid chromatography-mass spectrometry; Journal of Pharmaceutical and Biomedical Analysis, (2020); . doi: 10.1016/j.jpba.2020.113446 (available online). [DOI] [PubMed] [Google Scholar]
- 58. Pauli E., Joshi H., Vasavada A., Brackett J., Towa L.; Evaluation of and immediate-release formulation of hydroxychloroquine Sulfate with an interwoven Pediatric taste-masking system; Journal of Pharmaceutical Sciences, (2020); 109: 1493–1497. [DOI] [PubMed] [Google Scholar]
- 59. Silva P.H.R., Pianetti G.A., César I.C.; Simultaneous quantitation of chloroquine and primaquine by UPLC-DAD and comparison with a HPLC DAD method; Malaria Journal, (2015); 14: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Coelho A.S., Chagas C.A.P., Pádua R.M., Pianetti G.A., Fernandes C.; A comprehensive stability indicating HPLC method for determination of chloroquine in active pharmaceutical ingredient and tablets: Identification of oxidation impurities; Journal of Pharmaceutical and Biomedical Analysis, (2017); 145: 248–254. [DOI] [PubMed] [Google Scholar]
- 61. Poorvashree J., Suneela D.; Novel drug delivery of dual acting prodrugs of hydroxychloroquine with aryl acetic acid NSAIDs: Design, kinetics and pharmacological study; Drug Delivery and Translational Research, (2017); 7: 709–730. [DOI] [PubMed] [Google Scholar]
- 62. Chambliss A.B., Füzéry A.K., Clarke W.A.; Quantification of hydroxychloroquine in blood using turbulent flow liquid chromatography-tandem mass spectrometry (TFLC-MS/MS); clinical applications of mass spectrometry in drug analysis; Methods And Protocols, (2016); 1883: 177–184. [DOI] [PubMed] [Google Scholar]
- 63. Protti M., Mandrioli R., Mercolini L.; Tutorial: Volumetric absorptive microsampling (VAMS); Analytica Chimica Acta, (2019); 1046: 32–47. [DOI] [PubMed] [Google Scholar]
- 64. Wilhelm A.J., Burger J.C.G., Swart E.L.; Therapeutic drug monitoring by dried blood spot: Progress to date and future directions; Clinical Pharmacokinetics, (2014); 53: 961–973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Grüner N., Stambouli Q., Ross R.S.; Dried blood spots–preparing and processing for use in immunoassays and in molecular techniques; Jove-Journal of Visualized Experiments, (2015); 97: e52619. doi: 10.3791/52619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Marchi I., Rudaz S., Selman M., Veuthey J.; Evaluation of the influence of protein precipitation prior to on-line SPE-LC-API/MS procedures using multivariate data analysis; Journal of Chromatography B, (2007); 845: 244–252. [DOI] [PubMed] [Google Scholar]
- 67. Polson C., Sarkar P., Incledon B., Raguvaran V., Grant R.; Optimization of protein precipitation based upon effectiveness of protein removal and ionization effect in liquid chromatography-tandem mass spectrometry; Journal of Chromatography B, (2003); 785: 263–275. [DOI] [PubMed] [Google Scholar]
- 68. Wünsch U.J., Geuer J.K., Lechtenfeld O.J., Koch B.P., Murphy K.R., Stedmon C.A.; Quantifying the impact of solid-phase extraction on chromophoric dissolved organic matter composition; Marine Chemistry, (2018); 207: 33–41. [Google Scholar]
- 69. Reymond C., Dubuis A., Le Masle A., Colas C., Chahen L., Destandau E. et al. ; Characterization of liquid-liquid extraction fractions from lignocellulosic biomass by high performance liquid chromatography hypenated to tandem high-resolution mass spectrometry; Journal of Chromatography A, (1610); 2020: 460569. [DOI] [PubMed] [Google Scholar]
- 70. Chambliss A.B., Füzéry A.K., Clarke W.A.; Quantification of hydroxychloroquine in blood using turbulent flow liquid chromatography-tandem mass spectrometry (TFLC-MS/MS); Clinical Applications Of Mass Spectrometry In Drug Analysis: Methods And Protocols, (2016); 1383: 177–184. [DOI] [PubMed] [Google Scholar]
- 71. Caglar S., Alp A.R.; A validated high performance liquid chromatography method for the determination of Saxagliptin and metformin in bulk, a stability indicating study; Journal of Analytical & Bioanalytical Techniques, (2014); 12: 2–4. [Google Scholar]
- 72. Caglar S., Toker S.; Simultaneous determination of desloratadine and pseudoephedrine sulfate in tablets by high performance liquid chromatography and derivative spectrophotometry; Reviews in Analytical Chemistry, (2011); 30: 145–151. [Google Scholar]
- 73. Aydogmus Z., Caglar S., Toker S.; RP-HPLC method for determination of oseltamivir phosphate in capsules and spiked plasma; Analytical Letters, (2010); 43: 2200–2209. [Google Scholar]
- 74. Caglar S., Toker S.; Determination of rosuvastatin at picogram level in serum by fluorimetric derivatization with 9-Anthryldiazomethane using HPLC; Journal of Chromatographic Science, (2013); 51: 53–58. [DOI] [PubMed] [Google Scholar]
- 75. Caglar S., Morello R., Boos K.S.; Development and validation of an on-line multidimensional SPE-LC-MS/MS method for the quantitation of Tetrandrine in blood samples; Journal of Chromatography B-Analytical Technologies in the Biomedical and Life Sciences, (2015); 988: 25–32. [DOI] [PubMed] [Google Scholar]
- 76. Andac S.C.; Determination of drugs by online column-switching liquid chromatography; Journal of Chromatographic Science, (2016); 54: 1641–1647. doi: 10.1093/chromsci/bmw120. [DOI] [PubMed] [Google Scholar]


