Abstract
Introduction: Malignant granular cell tumor (MGCT) of the esophagus is an extremely rare malignancy with a poor prognosis. Literature describing this condition is not sufficient, especially regarding long-term survival.
Presentation of Case: A 52-year-old woman presented with dyspnea and slow onset dysphagia. The endoscopy, endoscopic ultrasound (EUS), bronchoscopy, and positron emission tomography (PET)/computed tomography (CT) supported the suspicion of esophageal gastrointestinal stromal tumor (GIST). Open wedge esophagectomy and tracheal resection were performed. The histology proved periodic acid–Schiff (PAS)-positive granules in epithelial cells, hyperchromatic nuclei and the positivity of Protein soluble in 100% ammonium sulfate (S-100), vimentin, neuron-specific enolase, laminin, and myelinic proteins. Local recurrence after 10 months required a two-phase esophagectomy with retrosternal gastroplasty. Bone, liver, and mediastinal metastases occurred 6 months later, with overall survival of 34 months.
Discussion: Preoperative histological confirmation is often not reliable. Tracheal invasion increases the perioperative risk and the probability of an unsuccessful resection. Esophagectomy or radical R0 local resection is the only known curative therapy. Repeated resections may increase survival in case of locoregional recurrence. Radiotherapy has a potential for palliative care.
Conclusion: Esophageal MGCT requires a detailed presentation including long-term survival. Early surgical removal of intramural esophageal neoplasms with potentially malignant features is highly recommended. Radical and/or repeated esophageal resections are the only known therapies with curative potential.
Keywords: granular cell tumor, esophageal neoplasm, esophagectomy, neoplasm recurrence, local case report
Introduction
Malignant granular cell tumor (GCT, Abrikossoff tumor) of the esophagus is an extremely rare malignancy, originating from pacemaker neuroectodermal cells of the esophageal submucosal plexus. The incidence of GCT reaches 0.002–0.03% of all described malignant tumors,1) mostly affecting women in their fifties. Multiple occurrences in one patient (6–14%) or organs (5–12%) have been observed. The most common site is the tongue (40%), the skin (30%), and breast (15%); the digestive tract is affected in 2.7–8.1% of the cases (30% of them in the esophagus). Rare locations include the bronchi and the urogenital tract.2) The evidence is limited and literature describing long-term survival are rare.
Case Report
A 52-year-old woman, without past history of family illnesses neither smoking history, was presented with a chronic cough and dysphagia without weight loss (body mass index [BMI] 24). Expansion of the retrotracheal space with tracheal and esophageal compression was found on high-resolution computed tomography (CT) scans. The positron emission tomography (PET) CT showed increased 18F-fludeoxyglucose uptake in the retrotracheal enlargement sized 27 × 23 × 22 mm (Fig. 1A and 1B). The submucosal expansion with preserved esophageal lumen; localized 23–25 cm from the incisors was later detected by esophagoscopy (Fig. 2A). The endoscopic ultrasound (EUS) confirmed a tumorous mass emerging from the muscular layer of the esophageal wall, extending from submucosa to periesophageal tissue, sized 25 × 25 mm (Fig. 2B). The structure was heterogenic and hypoechoic, no lymph nodes were detected. Fine needle aspiration biopsy (FNAB) was not performed. The localized expansion of the membranous part of the trachea was detected by bronchoscopy. Tumor markers including neuron-specific enolase (NSE), carcinoembryonic antigen (CEA), and chromogranin A were all negative.
Fig. 1. PET CT of the esophageal granular cell tumor: (A) lateral view and (B) transverse section. PET CT: positron emission tomography computed tomography.
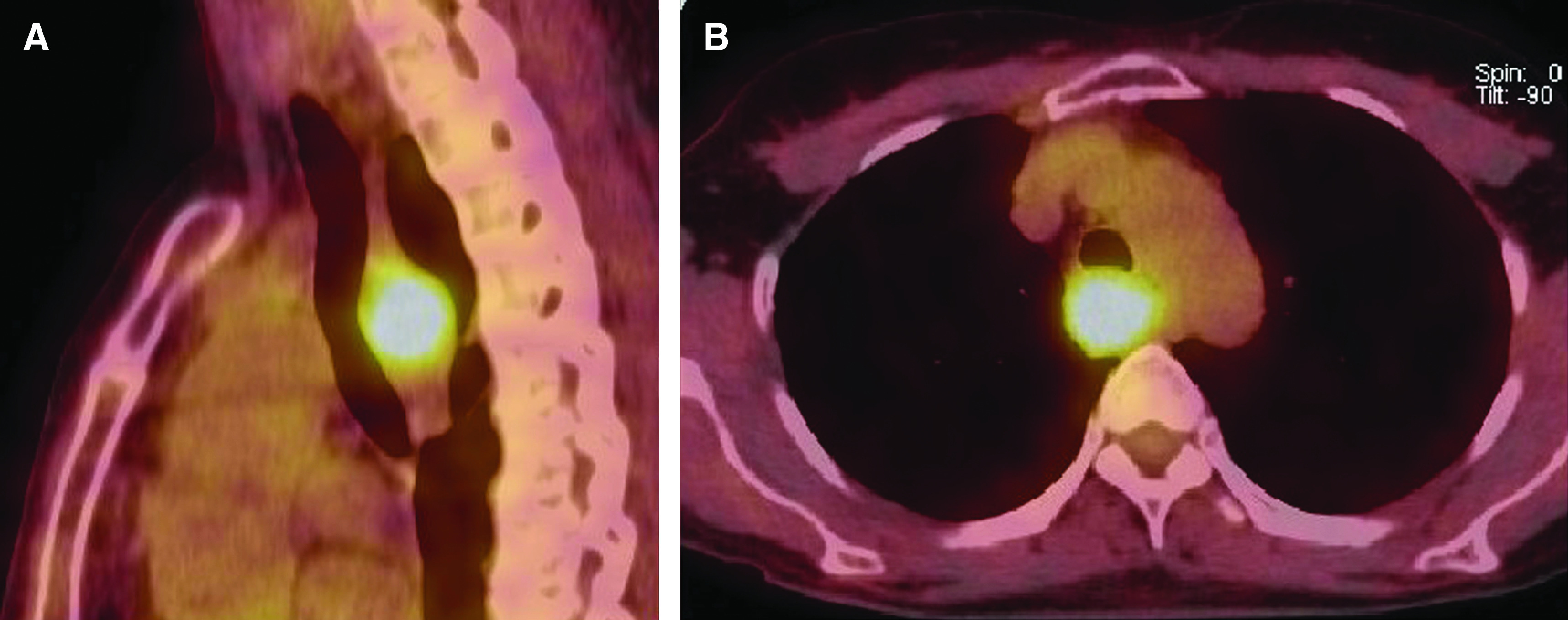
Fig. 2. Clinical appearance of malignant granular cell tumor: (A) endoscopic appearance, (B) EUS confirming partial growth to mediastinum surrounding the aorta, and (C) resected tumor. EUS: endoscopic ultrasound.
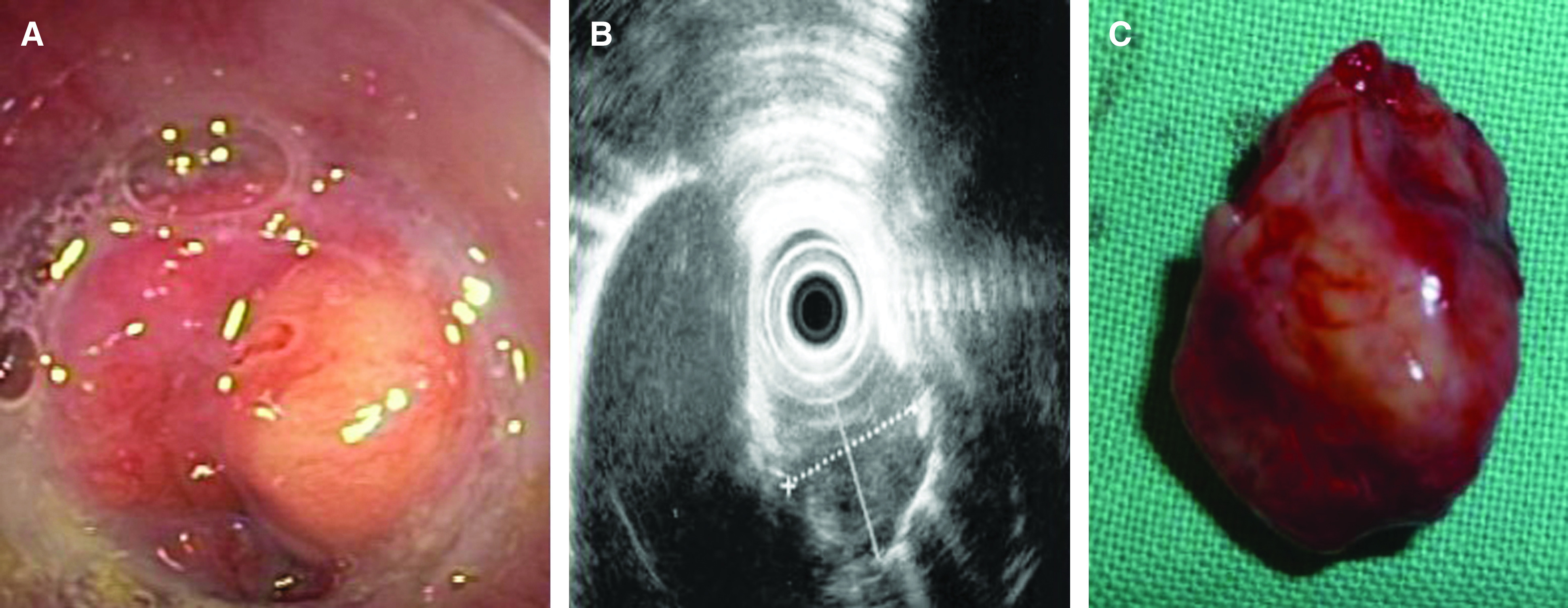
The patient was referred to surgery 5 weeks after the onset of symptoms. The unexpected progression of the tumor was discovered during the intraoperative endoscopy and thoracoscopy, which visualized an ingrowth of the tumor to the trachea and the right bronchus. The thoracoscopic approach in a left lateral decubitus position was converted to thoracotomy in the 5th intercostal space. Partial esophageal wedge resection with transverse resection of the membranous part of the trachea was made and the right bronchus sutured together with a pedicled periostomuscular flap, supplied by the intercostal artery. The resection was performed with negative margins on intraoperative histology (Fig. 2C). The patient healed primarily and experienced a left recurrent nerve palsy.
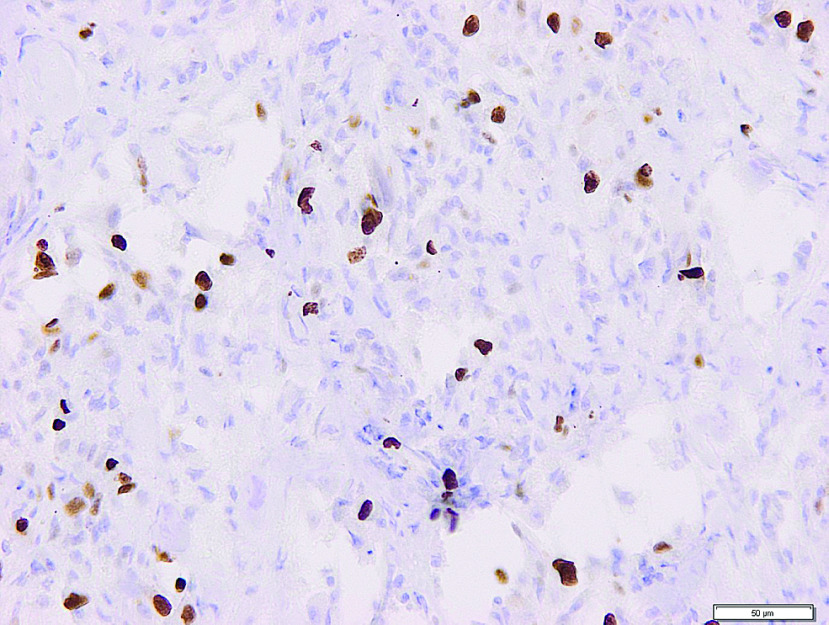
The pathology slides described streaks and nests of the epithelioid and spindle cells with a central hyperchromatic vesicular and granular eosinophilic cytoplasm with periodic acid–Schiff (PAS)-positive granula and prominent nucleoli (Fig. 3A and 3B). Pseudoepitheliomatous hyperplasia was found on the adjacent mucosal surface. The mitotic activity was 2/10 high-power field (HPF), Ki67 (MIB1) reached 10% (Fig. 4), without observed angioinvasion. The adventitia was discontinued with tumor ingrowth to the mediastinal structures. Immunohistochemistry proved the positivity of Protein soluble in 100% ammonium sulfate (S-100) protein, vimentin, NSE, laminin, and the myelinic proteins. Smooth muscle actin (SMA), pancytokeratin, MelanA, Human Melanoma Black-45 (HMB-45), cytokeratin AE1-AE3 (AE1-AE3), chromogranin, synaptophysin, cluster of differentiation (CD) 56, CD 34 and CD 117 were negative.
Fig. 3. Histology. (A) Hematoxylin-eosin staining, enlarged 400×. (B) PAS staining, enlargement 200×. PAS: periodic acid–Schiff.
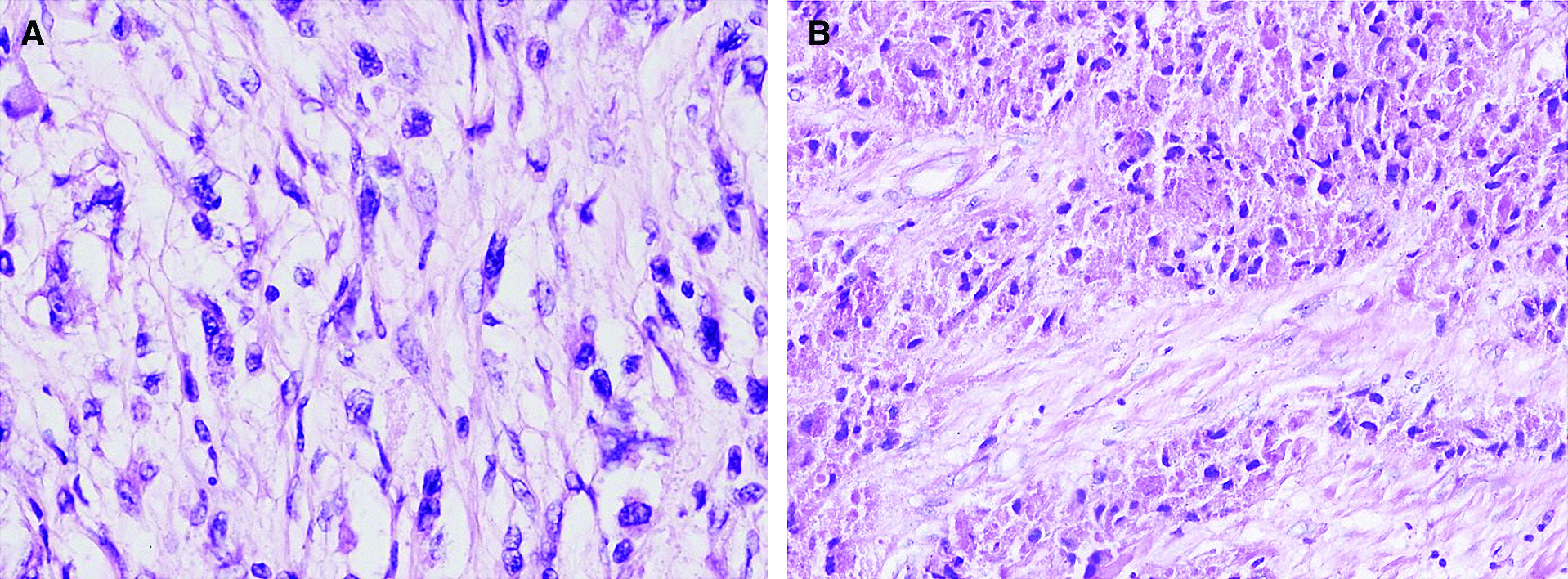
Fig. 4. Immunohistochemistry of granular cell tumor expressing 10% Ki67 (MIB1).
Further oncologic therapy was not indicated due to expected radioresistance and chemoresistance. Surveillance and monitoring of the patient involved PET CT every 4–6 months. Local recurrence was diagnosed 10 months later; the patient was indicated for a transthoracic esophagectomy. An esophagostomy and gastrostomy were made due to significant intraoperative bleeding. Restoration with gastroplasty and retrosternal placement with cervical anastomosis was constructed 17 days after the first operation. The patient was discharged home on the 11th postoperative day after reconstruction and with no restrictions of peroral intake.
Distant metastases were found in the liver and in the lateral part of the sacral bone. Diagnosed by a PET CT, 6 months later. Symptomatic relief with palliative radiotherapy of the osteolytic lesions in the sacrum was indicated. 23 months after the second operation, the patient developed a traumatic right hip fracture, followed by a gradual progression of dysphagia and malignant cachexia. The overall survival of the patient was 34 months, and an autopsy was not performed.
Discussion
To date, the literature evidence contains 15 reports on malignant esophageal GCT.3–17) Main features of published case reports are summarized in Table 1. The tumor in the esophageal location usually presents as oligosymptomatic or with non-specific symptoms: chest pain, slow-onset dysphagia, dyspepsia, diffuse localized back pain, dyspnea, or a palpable cervical resistance. 75% of benign GCTs are smaller than 2 cm in diameter, most commonly occurring in the distal esophagus (65%), followed by the middle esophagus in 20 % and in the proximal esophagus in 15%.
Table 1. Basic features of malignant esophageal granular cell tumors: literature overview.
| Basic characteristics | No. of references in total | Results | No. |
|---|---|---|---|
| Age | ∑ = 16 | 52,3 (21–81, median 60) | |
| Sex (M/F) | ∑ = 16 | 4/12 | |
| Symptoms | ∑ = 11 | Dyspnoea | 4 |
| Dysphagia | 5 | ||
| Cough, hoarse voice | 2 | ||
| Back pain | 1 | ||
| Palpable resistance | 1 | ||
| Asymptomatic | 1 | ||
| Size (mm) | ∑ = 11 | 70,5 (12–150, median 54) | |
| Location | ∑ = 15 | Cervical | 2 |
| Upper thoracic | 2 | ||
| Middle thoracic | 5 | ||
| Lower thoracic | 4 | ||
| Mediastinum | 3 | ||
| Invasion | ∑ = 11 | Trachea | 5 |
| Pulmonary hilum, pleura | 2 | ||
| Soft tissue | 6 | ||
| Esophageal wall (preserved adventitia) | 5 | ||
| Therapy | ∑ = 13 | Esophagectomy | 2 |
| Esophageal resection | 9 | ||
| Mediastinal, lung and chest wall resection | 1 | ||
| Inoperable | 1 | ||
| Alternative | 1 | ||
| Surveillance (months) | ∑ = 11 | 18,3 (7–52, median 14) | |
| Disease-free | 2 (surveillance 10 months) | ||
| Metastasis site | ∑ = 13 | Liver | 4 |
| Bone | 3 | ||
| Pleura | 2 | ||
| Lung | 2 | ||
| Cervical lymph nodes | 1 | ||
| Not reported | 5 |
Isolated and stiff sessile submucosal nodules covered with whitish intact mucosa are usually found during endoscopy. Rare forms are found, including circular obstructing tumor, giant tumor or the infiltrative variant, which are not necessarily malignant.18)
EUS usually describes a homogeneous hypoechoic lesion with regular margins. The relation to the esophageal wall and adventitia, the definition of the regularity of the border, echogenicity, lesion size, relationship to adjacent structures, and presence of lymph node enlargement should be evaluated. The contribution of PET CT helps to diagnose metastatic lymph nodes and distant metastasis. The preoperative biopsy is difficult due to frequently intact mucosa and with the risk of the ulceration and subsequent inability to provide enucleation. FNAB is not reliable enough to specify the tumor type and the malignant potential19); however, there is a risk of undesirable tumor seeding.
Generally, a lesion with the diameter above 2 cm and rapid growth on EUS or endoscopy should be the indication for an intramural tumor removal even in cases of unknown histology.
The tumor is composed of the stripes and nests of epithelioid cells and fusiform cells in a different representation. The cells have central hyperchromatic nuclei and granular eosinophilic cytoplasm with PAS-positive granules. Pseudoepitheliomatous hyperplasia can be present in the proximity of the tumor. Immunohistochemistry usually proves positive expression of S-100 protein, vimentin, neuron-specific enolase, laminin, myelin proteins, and negative expression of SMA, pancytokeratin, Melan-A, HMB-45, chromogranin, synaptophysin, CD 56, CD 34, and CD 117.
Differential diagnosis includes myogenic tumors (leiomyoma, leiomyosarcoma, rhabdomyosarcoma—SMA positivity), neuroendocrine tumors (chromogranin, synaptophysin, and NSE positivity), gastrointestinal stromal tumor (GIST), especially mixed type (Discovered On GIST1 [DOG1] and CD 117 positivity), gastrointestinal autonomous nerve tumor (typical morphology), sarcomatoid variant of malignant melanoma (S-100 protein, SRY-related HMG-box transcription factor 10 [SOX-10] and anti-high molecular weight cytokeratin antibodies [AA-HMW] positivity) and granular form of basaloid carcinoma. The coincidence of epithelial malignancies, especially squamous cell carcinomas should be excluded.
The criteria for deciding on the biological nature of the tumor include necrosis, spindling of tumor cells, vesicular nuclei with large nucleoli, high nuclear-cytoplasmic ratio, pleomorphism, and increased mitotic rates (w2/10 HPFs at 6200 magnification) with the antigen identified by monoclonal antibody Ki-67 (Ki-67) index higher than 10 %. Three or more of the above-mentioned criteria confirm the malignancy.20) The presence of more than 50% of p53-positive cells may be an additive diagnostic criterion. Adverse prognostic factors include local recurrence after local therapy, metastasis, tumor size (more than 4 cm), and older patients.
Therapy of malignant GCT is mainly surgical. The indication for surgical therapy includes malignant appearance, invasion of the deep layers (sm3 and deeper), confirmed malignancy, and multiple symptomatic tumors with contraindication of endoscopic therapy. Local growth (relation to the trachea, vertebral bodies, pulmonary hilum, aorta, and superior vena cava) and the presence of simultaneous metastatic lesions play a crucial role in the decision-making algorithm. The possible surgical therapy is partial esophageal resection in case of small tumors, and esophagectomy with debulking in case of advanced tumors. The approach (left-sided or right-sided video-assisted thoracoscopy/conventional thoracotomy) and intraoperative endoscopic assistance remain to be decided by the surgeon. Even pathology-confirmed R0 resection does not exclude the risk of local recurrence. Preserved adventitia with the tumor limited to the esophageal wall is linked to disease-free survival in published observations (two of five cases demonstrating no recurrence during postoperative surveillance).
In case of local recurrence after partial esophageal resection, salvage esophagectomy may be an alternative to achieve longer survival. Surgical revision carries a higher technical risk, such as severe intraoperative bleeding from the pulmonary artery with subsequent enforced two-phase esophagectomy, which was observed in our case. During the reconstruction phase, the tubulized stomach was placed on the retrosternal position for the predicted longer patency in comparison to posterior mediastinal placement. The metastatic pattern involves early hematogenic spread even in small-sized tumors and has a high potential for locoregional recurrence.
Oncological therapy has a palliative indication only. The tumor is characterized by chemoresistance. A palliative indication of radiotherapy is a feasible option for bone metastases.
The prognosis of malignant GCT of the esophagus is serious. The variable length of follow-up in previously published case reports together with incomplete information does not provide reliable results regarding the presented long-term survival. A thorough collection of further information is necessary.
Conclusion
Malignant GCT of the esophagus has a poor prognosis and complicated diagnostic and therapeutic strategy. The only known therapeutic modality with curative potential is radical surgical therapy with repeated procedures in selected cases. Options for oncological therapy are limited. The future publications on malignant GCT require a detailed presentation including long-term survival. Early surgical removal of intramural esophageal neoplasms with potentially malignant features is highly recommended.
Disclosure Statement
The authors declare no relevant conflict of interest.
References
- 1). Simsir A, Osborne BM, Greenebaum E. Malignant granular cell tumor: a case report and review of the recent literature. Hum Pathol 1996; 27: 853– 8. [DOI] [PubMed] [Google Scholar]
- 2). Johnston J, Helwig EB. Granular cell tumors of the gastrointestinal tract and perianal region: a study of 74 cases. Dig Dis Sci 1981; 26: 807– 16. [DOI] [PubMed] [Google Scholar]
- 3). Crawford ES, De Bakey ME. Granular-cell myoblastoma; two unusual cases. Cancer 1953; 6: 786– 9. [DOI] [PubMed] [Google Scholar]
- 4). Obiditsch-Mayer I, Salzer-Kuntschik M. [Malignant "granular cell neuroma", so-called "myoblastmyoma" of the esophagus]. Beitr Pathol Anat 1961; 125: 357– 73. (in German) [PubMed] [Google Scholar]
- 5). Harrer WV, Patchefsky AS. Malignant granular-cell myoblastoma of the posterior mediastinum. Chest 1972; 61: 95– 6. [DOI] [PubMed] [Google Scholar]
- 6). Sasaki T, Aoki H, Kasahara M. Granular cell tumor of esophagus with malignancy on histological findings. J Jpn Soc Clin Surg 1981; 36: 1645– 9. (in Japanese) [Google Scholar]
- 7). Ohmori T, Arita N, Uraga N, et al. Malignant granular cell tumor of the esophagus. A case report with light and electron microscopic, histochemical, and immunohistochemical study. Acta Pathol Jpn 1987; 37: 775– 83. [PubMed] [Google Scholar]
- 8). Wyatt MG, O'Donoghue DS, Clarke TJ, et al. Malignant granular cell tumour of the oesophagus. Eur J Surg Oncol 1991; 17: 388– 91. [PubMed] [Google Scholar]
- 9). Iwase H, Morise K, Hirouchi Y, et al. Malignant granular cell tumor of the esophagus, report of a case. Stomach and Intestine 1991; 26: 661– 8. (in Japanese) [Google Scholar]
- 10). Yang SW, Hong SW, Cho MY, et al. Malignant granular cell tumor at the retrotracheal space. Yonsei Med J 1999; 40: 76– 9. [DOI] [PubMed] [Google Scholar]
- 11). Yoshizawa A, Ota H, Sakaguchi N, et al. Malignant granular cell tumor of the esophagus. Wirchows Arch 2004; 444: 304– 6. [DOI] [PubMed] [Google Scholar]
- 12). Loo CK, Santos LD, Killingsworth MC. Malignant oesophageal granular cell tumour: a case report. Pathology 2004; 36: 506– 8. [DOI] [PubMed] [Google Scholar]
- 13). Isogai K, Yamamoto E, Yamaoka S, et al. A case report of the malignant granular cell tumor coexisting with squamous cell carcinoma of the esophagus. Jpn J Gastroenterol Surg 2005; 102: 1025– 9. (in Japanese) [PubMed] [Google Scholar]
- 14). Christopher PR, Kingsley PA, Singh Bedi H, et al. Large mid-esophageal granular cell tumor: benign versus malignant. Rare Tumors 2015; 7: 5772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15). Takebayashi K, Kinoshita Y, Ozawa T, et al. A case of malignant granular cell tumor of the esophagus with multiple liver metastasis. Jpn J Gastroenterol Surg 2012; 45: 23– 9. (in Japanese) [Google Scholar]
- 16). Nakao M, Hishida T, Ishii G, et al. Malignant granular cell tumor of the posterior mediastinum with dissemination. Asian Cardiovasc Thorac Ann 2012; 20: 71– 3. [DOI] [PubMed] [Google Scholar]
- 17). De Luca G, Luciano A, Benincasa G, et al. Giant malignant granular cell tumor (GCT) of the posterior mediastinum. J Thorac Oncol 2013; 8: 1107– 8. [DOI] [PubMed] [Google Scholar]
- 18). Goldblum JR, Rice TW, Zuccaro G, et al. Granular cell tumors of the esophagus: a clinical and pathologic study of 13 cases. Ann Thorac Surg 1996; 62: 860– 5. [DOI] [PubMed] [Google Scholar]
- 19). Smith AR, Gilbert CF, Strausbauch P, et al. Fine needle aspiration cytology of a mediastinal granular cell tumor with histologic confirmation and ancillary studies. A case report. Acta Cytol 1998; 42: 1011– 6. [DOI] [PubMed] [Google Scholar]
- 20). Fanburg-Smith JC, Meis-Kindblom JM, Fante R, et al. Malignant granular cell tumor of soft tissue: diagnostic criteria and clinicopathologic correlation. Am J Surg Pathol 1998; 22: 779– 94. [DOI] [PubMed] [Google Scholar]