Abstract
Epstein–Barr virus-associated diseases are important global health concerns. As a group I carcinogen, EBV accounts for 1.5% of human malignances, including both epithelial- and lymphatic-originated tumors. Moreover, EBV plays an etiological and pathogenic role in a number of non-neoplastic diseases, and is even involved in multiple autoimmune diseases (SADs). In this review, we summarize and discuss some recent exciting discoveries in EBV research area, which including DNA methylation alterations, metabolic reprogramming, the changes of mitochondria and ubiquitin-proteasome system (UPS), oxidative stress and EBV lytic reactivation, variations in non-coding RNA (ncRNA), radiochemotherapy and immunotherapy. Understanding and learning from this advancement will further confirm the far-reaching and future value of therapeutic strategies in EBV-associated diseases.
Subject terms: Cancer therapy, Cancer metabolism
Introduction
Epstein–Barr virus was the first tumorigenic DNA virus that persistently infects human and some additional primate B cells. It exists as a free particle with a genome length of about 172 kb.1 EBV is transmitted primarily through saliva, resulting in asymptomatic infection in about 95% of the world’s population. In a variety of tumor types, EBV can integrate into the host genome at a remarkable rate to promote tumor development.2 As early as 1997, EBV was classified as a group I carcinogen by the International Agency for Research on Cancer (IARC) and its associated tumors account for 1.5% of all human cancers. The etiology of EBV is primarily relevant to malignancies of epithelial and lymphatic origin, including nasopharyngeal carcinoma (NPC), gastric cancer (GC), pulmonary lymphoepithelioma-like carcinoma (LELC), breast cancer, tonsillar cancer, hepatobiliary system cancer, malignant salivary gland tumors, and thyroid cancer, Hodgkin’s lymphoma (HL), non-Hodgkin’s lymphomas (NHL), including Burkitt’s lymphoma (BL), extranodal NK/T-cell lymphoma, and post-transplant lymphoproliferative carcinoma (PTLD).3–5 Recently, the National Institutes of Health (NIH) identified EBV as an essential element in reducing the global cancer burden.5 Moreover, EBV plays an etiological and pathogenic role in a number of non-neoplastic diseases, including infectious mononucleosis (IM), chronic active EBV infection (CAEBV), EBV-associated hemophagocytic lymphohistiocytosis (EBV-HLH), and other systemic autoimmune diseases, such as systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), and Sjogren’s syndrome (SS).6
Notably, EBV infection of the host is predominantly latent. When the balance between host and virus is disturbed, EBV activation poses a risk of neoplasia. EBV has three latent infection types, type I is characterized by Burkitt’s lymphoma, which expresses only EBNA1 and the right fragment of BamHI A (BARTs) with the exception of EBERs. Type II includes NPC and Hodgkin’s lymphoma (HL) with EBV expressing EBNA1, LMP1, LMP2, BARTs, and EBERs. Type III includes lymphoproliferative diseases in immunosuppressed patients where EBV expresses all latent infection genes.7 LMP1 as a EBV encodes important oncogenic protein with specific functions occurring during latent infection and is essential for B-cell transformation in vitro.8 It mimics CD40 auto-oligomerization to continuously activate multiple intracellular signaling pathways that mediate various malignant phenotypes.9 LMP1 plays an important role in the development and progression of EBV-positive-associated tumors that are a prognostic marker for NPC patients.10,11 LMP2A and LMP2B were first discovered in 1990.12 LMP2A is a B-cell receptor (BCR) simulator that can protect BCR-depleted B cells from apoptosis.13 Epstein–Barr nuclear antigen 1 (EBNA1) is necessary to maintain stability of EBV particles, EBV replication,14 and also trigger B-cell lymphoma formation in transgenic mice.15 Studies have shown that EBNA1 causes genomic instability16 and promotes tumor cell survival.17 EBNA2 and EBNA3 are critical for B-cell transformation.18 Moreover, EBV has been shown to encode non-coding RNA,19 including BamHI-A rightward transcript microRNAs (miRNAs) (BART miRNAs), BHRF1 miRNAs, and EBV-encoded RNA 1 (EBER1) and EBER2, which interact with numerous proteins and facilitate the activation of innate immunity.
EBV-associated diseases
EBV and epithelial cancers
Nasopharyngeal carcinoma (NPC)
NPC is currently the most well-defined human epithelial cancer that is associated with EBV infection. In 1966, high titers of EBV-associated antibodies were found in NPCs.20 Studies have shown that an interaction of genetic, ethnic, and environmental factors may contribute to the development of NPC. In endemic areas, the EBV genome is detectable in almost all NPC tumor tissues. Subsequently, in the 1980s, serum levels of Epstein–Barr capsid antigen (VCA) immunoglobulin A (IgA) were established as one of the indicators for screening patients for NPC21 and the expression of latent genes in EBV-infected states was also discovered.22 With the use of qPCR to detect cell-free EBV DNA in 1999, a relationship between an increased risk of NPC due to EBV infection and poor prognosis was established.23 Since then, EBV DNA has become the gold standard for biological markers of NPC. Importantly, the genomic landscape of NPC has revealed that EBV latency genes provide growth and survival benefits, which result in the development of NPC. Therefore, with advances in NPC genomics, chromatin modifications enhance EBV load as a key feature of the NPC genome.24 Moreover, BALF2_CCT, as a high-risk subtype of EBV, promotes the progression of NPC in South China and facilitates early identification of high-risk individuals.25 In addition, Ephrin receptor A2 (EphA2), an epithelial growth factor-associated receptor, benefits EBV entry into epithelial cells.26 Importantly, common fragile regions are favored loci for EBV integration, which are genomic hotspots for DNA damage and vulnerable to genomic rearrangements. More recently, a study reports that EBV integration occur at 9.6% in NPC. These discoveries provide novel, potential intervention strategies for preventing EBV infection.
EBV-associated gastric cancer (EBVaGC)
Since 1992, the first demonstration of EBV in typical gastric adenocarcinomas occurred and facilitated the understanding of EBV in GC development.27 Approximately 9% of gastric cancer (GC) patients are EBV-positive, and these cases have a distinct molecular phenotype and clinical characteristics.28 Global epigenetic methylation and counteraction of the antitumor microenvironment are two major characteristics of this subtype of GC. With advances in the diagnosis and treatment of EBV infection, in situ hybridization (ISH) of EBER in biopsy specimens facilitate the diagnosis of EBVaGC.29 Recently, EBV can integrate into host genomes at 25.6% in GC tumorigenesis.2 In addition, immune checkpoint therapy for advanced stage or metastatic EBVaGC may be effective in treating this disease.30
Breast cancer
The first association of EBV infection with breast cancer was reported in 1995.31 Recently, several studies showed that EBV infection leads to more malignant transformation of breast epithelial cells and affects patient prognosis.32,33 EBV leads to a 4.74-fold increase in risk of breast cancer development compared with a control group.32 The potential oncogenesis mechanism of EBV is related to viral proteins, such as LMP1 that activate the Her2/Her3 signaling cascades.33
Several rare non-typical EBV-associated tumors, including pulmonary lymphoepithelioma-like carcinoma (LELC),34 tonsillar cancer,35 hepatobiliary system cancer,36 malignant salivary gland tumors,37 and thyroid cancer38 have also been reported. Among these, LELC is a rare and distinct subtype of primary lung cancer characterized by EBV infection and high LMP1 expression.34 These findings further expand our insight into the involvement of EBV in tumorigenesis.
EBV and lymphatic system cancers
HL and NHL
EBV-associated lymphomas consist of HL and NHL, of which the most widely studied NHL are Burkitt’s lymphoma (BL) and extranodal NK/T-cell lymphoma.39 EBV particles were first detected in BL back in 1964.40 The EBV genome was next revealed in BL41 and the relationship between EBV and lymphoma was successfully determined epidemiologically in the 1970s.42 Moreover, the identification of EBV DNA in HL serum43 suggests that EBV may serve as a biomarker for early disease detection and monitoring patient treatment. In the 1980s, the EBNA protein was detected in HRS cells of a HL patient44 and then EBV genome was sequenced in HL and NK/T-cell lymphoma.45 Also, ENK/T-cell lymphoma was characterized by EBV infection of T or NK cells, which express a latency II pattern with variable LMP1 expression.
Post-transplant lymphoproliferative carcinoma (PTLD)
PTLD is a serious complication that occurs after solid organ, bone marrow, or blood stem cell transplantation, and is associated with EB virus infection in 60–80% of patients.46 In post-transplant patients, immunosuppression leads to repression of T-cells with a subsequent uncontrolled proliferation of lymphoid cells.47 With the discovery of the EBV genome and antibodies in 1980,48 a majority of PTLD patients have been tested for the presence of LMP1 and EBNA2/EBNA3 expression. The first successful use of EBV-specific T-cell therapy in EBV-associated lymphocyte proliferation occurred in 1995.49 Therefore, monitoring of EBV infection in post-transplant patients is of paramount importance.
EBV chronic infectious diseases
Infectious mononucleosis (IM)
IM is a self-limiting disease caused by primary EBV infection, which was defined in 1968.50 Symptoms of IM are caused by an excessive immune response to EBV infection that presents with a “triad” of fever, pharyngitis, and lymphadenopathy, with a marked increase in peripheral blood lymphocytes and the appearance of heterogeneous lymphocytes.51
Chronic active EBV infection (CAEBV)
CAEBV is a serious EBV infection with clinical manifestations of persistent or recurrent IM-like symptoms.52 The main features of the advancement of the disease include dramatically enhanced levels of EBV DNA in the blood and infiltration of organs by positive EBER1-positive cells. Patients often exhibit fever, lymphadenopathy, splenomegaly, hepatitis, or pancytopenia.53,54 EBV infection of T cells and natural killer (NK) cells performs a pivotal contribution to the pathogenesis of CAEBV. Usually, T-cell infections have severe clinical symptoms and a poor prognosis. However, NK-cell-type infection patients favor for transformation into aggressive NK-cell leukemia or extranodal NK/T-cell lymphoma.55 Besides, in adult CAEBV, HLH may be the other clinical endpoint.56–58
EBV-associated hemophagocytic lymphohistiocytosis (EBV-HLH)
EBV-HLH is a severe, life-threatening hyperinflammatory syndrome of EBV infection, clinically characterized by abnormal proliferation and activation of lymphocytes and macrophages, fever, hepatosplenomegaly, blood cytopenias, hepatitis and/or hepatomegaly.59 Importantly, the timing and severity of these appearances can assist in distinguishing between EBV infection alone and HLH-complicated EBV infection.60
EBV-related autoimmune diseases
EBV has been shown to be the cause of several autoimmune diseases, and studies have shown that EBV-infected individuals have a higher frequency of disease, including SLE, RA, SS, compared to non-infected individuals.6 As EBV plays an etiological role in autoimmune diseases, serological measures of EBV reactivation provide additional biomarkers to identify high-risk individuals. The common features of these EBV-related autoimmune diseases are elevated plasma viral loads and levels of EBV-directed antibodies that result in decreased EBV-directed cell-mediated immunity, suggesting the presence of widespread lytic EBV infection in patients.61 Furthermore, EBV reactive and the host’s immune response result in different diseases patterns and clinical manifestations.
DNA methylation alterations and clinical applications in Ebv-associated malignancies
Regulation of DNA methylation by EBV
DNA methylation in mammalian organisms is a reversible process, which mostly occurs by the addition of a methyl group to the C-5 position of cytosine in the CpG sequence context. DNA methyltransferases (DNMTs) transfer methyl groups from S-adenosyl-L-methionone (SAM) to cytosine and create 5-methylcytosine (5mC). DNMT1 is the most abundant DNMT in mammalian cells and it maintains methylation status during cell replication.62 Ten-eleven translocation methylcytosine dioxygenases (TETs) oxidize 5-mC to 5-hydroxymethylcytosine (5hmC), which in turn undergo further oxidation into 5-formylcytosine (5fC) and 5-carboxylcytosine (5caC), thereby mediating DNA demethylation.63 The balance of DNA methylation- demethylation dynamics is maintained by DNMTs and TETs.
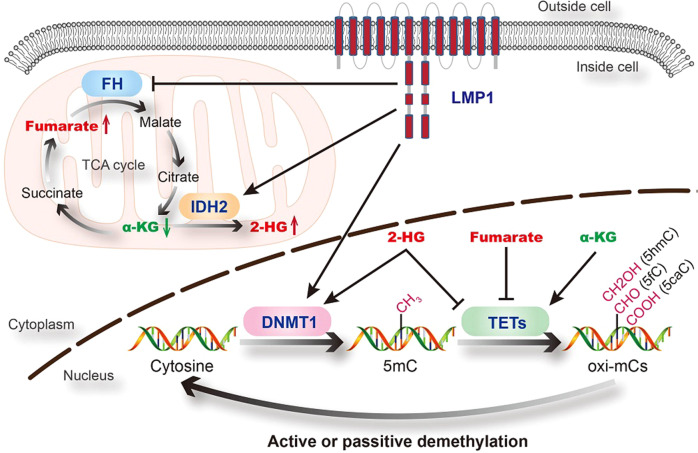
EBV infection is an epigenetic driver, which results in aberrant DNA methylation.64,65 First, EBV activates DNA methylation pathways. Tsai et al.66 reported that EBV-encoded latent membrane protein 1 (EBV-LMP1) enhances DNMT1 transcription and expression by activating the JNKs signaling pathway. We previously showed that EBV-LMP1 not only upregulates DNMT1 expression and activity, but also promotes its mitochondrial translocation.67 Secondly, EBV inhibits DNA demethylation pathway. TET family proteins, especially TET2, are downregulated during EBV infection, and TET2 may function as a resistance factor against EBV-induced DNA methylation acquisition.68 TET enzymes require α-ketoglutarate (α-KG) as an essential co-substrate to catalyze the conversion of 5-mC to 5-hmC. With a similar structure to α-KG, fumarate, succinate, and 2-HG could competitively inhibit the enzymatic activity of TETs.69 Recently, we revealed that EBV-LMP1 promotes the accumulation of fumarate and 2-HG, and the reduction of α-KG, which consequently leads to the inactivation of TETs.70,71 Moreover, 2-HG could also regulate DNMT1 activity by enhancing its binding to selected DNA regions.72 EBV infection might tip the balance in DNA methylation-demethylation dynamics (Fig. 1).
Fig. 1.
Regulation of DNA methylation by EBV (LMP1). The balance of DNA methylation- demethylation dynamics is maintained by DNMTs and TETs. DNMTs transfer methyl groups to cytosine and create 5mC. TETs iteratively oxidize 5mC to generate oxi-mCs (5hmC, 5fC, and 5caC). EBV (LMP1) enhances DNMT1 expression and activity, and also inhibit the enzymatic activity of TETs, which might tip the balance. FH fumarate hydratase, IDH2 isocitrate dehydrogenase 2
CpG island promoter methylation of tumor suppressor genes (TSGs) is one of the most characteristic abnormalities in EBV-associated malignancies. The CpG island methylation phenotype (CIMP) is defined as the high activity of global and nonrandom CpG island methylation. EBV infection mediates onco-epigenetic effects and is closely associated with CIMP.73 The most representative feature of EBV-positive GCs is an extensive hypermethylation phenotype, EBV-CIMP, which includes CDKN2A promoter hypermethylation.74 CIMP plays a driving role in EBV-associated malignancies, which leads to promoter hypermethylation and silencing of a series of functional genes (Tables 1–3).
Table 2.
Hypermethylated genes in EBV-associated GC
| Gene names | Full name | Gene function | Methylation status | Reference |
|---|---|---|---|---|
| AQP3 | Aquaporin 3 | Water channel | 95.8% (23/24) | 449 |
| B4GALNT2 | Beta-1,4-N-acetyl-galactosaminyltransferase 2 | Blood group systems biosynthesis | 50.8% (32/63) | 450 |
| CDKN2A | Cyclin-dependent kinase inhibitor 2A, P16 | Cell cycle control | 86.7% (13/15) | 451 |
| 68.2% (15/22) | 452 | |||
| CDKN2B | Cyclin-dependent kinase inhibitor 2B, P15 | Cell cycle control | 86.7% (13/15) | 451 |
| CDH1 | E-cadherin | Cell–cell adhesion | 100% (15/15) | 451 |
| CYLD | CYLD lysine 63 deubiquitinase | Deubiquitination | 56.0% (14/25) | 453 |
| DAPK1 | Death-associated protein kinase 1 | Apoptosis | 73.3% (11/15) | 451 |
| IRF5 | Interferon regulatory factor 5 | Transcription factor | 76.5% (13/17) | 454 |
| MARK1 | Microtubule affinity regulating kinase 1 | Cell migration | 68.0% (17/25) | 455 |
| MINT2 | amyloid beta precursor protein binding family A member 2 | Signal transduction | 92.0% (23/25) | 452 |
| PTEN | Phosphatase and tensin homolog | PI3K/AKT signaling | 64.3% (18/28) | 456 |
| REC8 | REC8 meiotic recombination protein | Meiosis | 92.3% (12/13) | 457 |
| RUNX3 | RUNX family transcription factor 3 | Transcription factor | 96.0% (24/25) | 452 |
| S100A4 | S100 calcium binding protein A4 | Signal transduction | 50.0% (12/24) | 455 |
| SCRN1 | Secernin 1 | Exocytosis | 48% (12/25) | 455 |
| SOX9 | SRY-box transcription factor 9 | Transcription factor | 85.7% (6/7) | 458 |
| SSTR1 | Somatostatin receptor 1 | G-protein-coupled receptor signaling | 75% (9/12) | 459 |
| ST3GAL6 | ST3 beta-galactoside alpha- 2,3- sialyltransferase 6 | Glycosphingolipids biosynthesis | 44.4% (28/63) | 450 |
| TP73 | Tumor protein p73 | Stress response | 96.0% (24/25) | 455 |
| 100% (15/15) | 451 | |||
| 96.0% (24/25) | 452 | |||
| WNT5A | Wnt family member 5A | WNT signaling | 91.3% (21/23) | 460 |
Table 1.
Hypermethylated genes in EBV-associated NPC
| Gene names | Full name | Gene function | Methylation status | Reference |
|---|---|---|---|---|
| APC | Regulator of WNT signaling pathway | WNT signaling | 34.0% (17/34) | 439 |
| CADM1 | Cell adhesion molecule 1 | Cell–cell adhesion | 69.8% (37/53) | 83 |
| 67.9% (19/28) | 440 | |||
| CDKN2A | Cyclin-dependent kinase inhibitor 2A, P16 | Cell cycle control | 66.0% (35/53) | 83 |
| CDH1 | E-cadherin | Cell–cell adhesion | 73.7% (18/28) | 441 |
| CDH13 | Cadherin 13 | Cell–cell adhesion | 77.4% (41/53) | 83 |
| CHFR | Checkpoint with forkhead and ring finger domains | Cell cycle control | 58.5% (31/53) | 83 |
| DAPK1 | Death-associated protein kinase 1 | Apoptosis | 27.3% (60/220) | 84 |
| DAB2 | DAB adaptor protein 2 | Proliferation | 65.2% (30/46) | 442 |
| DCC | DCC netrin 1 receptor | Signal transduction | 50.0% (23/46) | 439 |
| DLEC | DLEC1 cilia and flagella-associated protein | Proliferation | 60.4% (29/48) | 439 |
| DLC1 | DLC1 Rho GTPase activating protein | G-protein signaling | 76.9% (40/52) | 83 |
| 43.8% (21/48) | 439 | |||
| INPP4B | Inositol polyphosphate -4-phosphatase type II B | Phosphatidylinositol signaling | 60.0% (9/15) | 443 |
| ITGA9 | Integrin subunit alpha 9 | Cell–cell adhesion | 55.6% (20/36) | 444 |
| KIF1A | kinesin family member 1A | Membrane trafficking | 56.0% (28/50) | 439 |
| MAOA | Monoamine oxidase A | Amino acid metabolism | 40.7% (22/54) | 445 |
| miR-31 | microRNA 31 | Post-transcriptional regulation | 87.5% (14/16) | 446 |
| PRDM2 | PR/SET domain 2, RIZ1 | Histone methyltransferase | 56.6% (30/53) | 83 |
| PTEN | Phosphatase and tensin homolog | PI3K/AKT signaling | 80.0% (40/50) | 447 |
| RARB | Retinoic acid receptor beta | Nuclear receptor | 15.9% (35/220) | 84 |
| RASSF1A | Ras association domain family member 1 | Cell cycle control | 24.1% (53/220) | 84 |
| 75.5% (40/53) | 83 | |||
| 82.1% (23/28) | 440 | |||
| RASSF2A | Ras association domain family member 2 | Cell cycle control | 29.2% (14/48) | 83 |
| RRAD | Ras related glycolysis inhibitor and calcium channel regulator | Calcium channel | 74.3% (26/35) | 448 |
| UCHL1 | ubiquitin C-terminal hydrolase L1 | Protein ubiquitination | 64.0% (32/50) | 439 |
| WIF1 | WNT inhibitory factor 1 | WNT signaling | 51.8% (114/220) | 84 |
| 61.2% (30/49) | 83 | |||
| WNT7A | Wnt family member 7A | WNT signaling | 69.4% (25/36) | 444 |
Table 3.
Hypermethylated genes in other EBV-associated tumors
| Gene names | Full name | Gene function | Cancer type | Methylation status | Reference |
|---|---|---|---|---|---|
| BCL2L11 | BCL2 like 11 | Apoptosis | BL | 92.9% (13/14) | 461 |
| CDKN2A | Cyclin-dependent kinase inhibitor 2A, P16 | Cell cycle control | CSCC | 61.0% (25/41) | 462 |
| CDH1 | E-cadherin | Cell–cell adhesion | OSCC | 82.4% (56/68) | 463 |
| HACE1 | HECT domain and ankyrin repeat containing E3 ubiquitin protein ligase 1 | Protein ubiquitination | NKTCL | 100% (6/6) | 464 |
| MGMT | O-6-methylguanine-DNA methyltransferase | DNA repair | OSCC | 55.9% (38/68) | 463 |
| PRDM1 | PR/SET domain 1 | Transcriptional regulation | BL | 50.0% (11/22) | 465 |
BL Burkitt’s lymphoma, CSCC cervical squamous cell carcinoma, OSCC oral squamous cell carcinoma, NKTCL natural killer/T-cell lymphoma
Interestingly, DNA hypomethylation induced by EBV has also been observed in a few studies. For example, the S100 calcium binding protein A4 (S100A4) is overexpressed in LMP2A-positive NPC tissues and DNA hypomethylation plays a critical role in S100A4 regulation.75 In Hodgkin’s lymphoma, a set of tumor-related genes known to be silenced by promoter hypermethylation (e.g., RASSF1, DAPK, MGMT) are more frequently hypermethylated in EBV-negative compared to EBV-positive cases.76 EBV might also take part in the redistribution of DNA methylation modification sites.
The clinical significance of DNA methylation as a biomarker in EBV-associated malignancies
Abnormal DNA methylation has been investigated in NPC biopsies, nasopharyngeal brushings, and blood samples. Thus, aberrant DNA-methylated genes might be used as diagnostic or prognostic biomarkers.
In the EBV genome, DNA methylation takes an important part in lytic cycle regulation. The EBV genome is gradually methylated during the establishment of latency, while it becomes unmethylated in the viral lytic cycle. EBV DNA methylation could be used as a biomarker. For example, Zheng et al.77 found that a combination of the degree of EBV DNA methylation and EBV DNA load showed great potential for diagnosing NPC in nasopharyngeal brushing samples (sensitivity = 0.98, specificity = 0.95). The viral genome-wide methylation profiling of circulating EBV DNA in plasma demonstrated differential methylation patterns between NPC and non-NPC subjects. When combining the plasma EBV DNA load and methylation test pattern in plasma samples, the sensitivity was 0.97 and the specificity was 0.99 for population screening of NPC.78
Tumor-related genes are associated with EBV infection and carcinogenesis and some of these are summarized in Table 1. For instance, RASSF1A promoter methylation is a promising noninvasive biomarker for the diagnosis of NPC as determined from tissue and brushing samples (tissue: sensitivity = 0.72, specificity = 0.99, AUC = 0.98; brushing: sensitivity = 0.56, specificity = 1.00, AUC = 0.94).79 High levels of TIPE3 promoter methylation is associated with worse overall survival (OS), disease-free survival (DFS), and distant metastasis-free survival (DMFS), which could be an independent prognostic factor in NPC.80 The methylation level of the HOPX promoter is a potential prognostic biomarker for NPC and high levels are associated with poor prognosis.81 Patients with RAB37 hypermethylation are more sensitive to docetaxel-containing induction chemotherapy and RAB37 methylation levels might be used to predict chemotherapy sensitivity.82
Recently, panels of methylation genes, which are more sensitive than single biomarkers, have been reported. Hutajulu et al.83 found that combined analyses of five methylation markers (i.e., RASSF1A, p16, WIF1, CHFR, RIZ1) provided good discrimination between NPC and non-NPC patients with a detection rate of 98%. A panel of four methylated genes (i.e., RASSF1A, WIF1, DAPK1, RARβ2) in combination with the EBV DNA test increase the sensitivity for NPC detection at an early stage (sensitivity = 0.96, specificity = 0.65, AUC = 0.83).84 The genome-wide DNA methylation profiling study revealed a methylation gene panel (i.e., WIF1, UCHL1, RASSF1A, CCNA1, TP73, SFRP1) as a prognostic biomarker for OS and DFS, which was also associated with the efficacy of concurrent chemotherapy in NPC.85
Targeting DNA methylation for therapy against EBV-associated malignancies
DNA hypermethylation correlates with poor survival outcomes in patients with cancer, especially with EBV-associated tumors. DNMT1 (maintaining DNA hypermethylation of specific genes) is currently the most important target for epigenetic cancer therapy. DNMT inhibitors, such as Vidaza® (5-azacytidine, Celgene) and Dacogen® (decitabine or 5-aza-2’-deoxycytidine, SuperGen) have been approved by the FDA for cancer therapy.86 A few studies have focused on EBV-associated malignancies. In NPC preclinical studies, 5-azacytidine has shown the potential to improve outcomes with radiotherapy.87 However, in a phase II study, CC-486 (oral azacitidine) did not show sufficient clinical activity to support further development as a monotherapy in advanced NPC.88
Some natural compounds also showed efficacy as DNMT1 inhibitors. Previous studies from our group and other labs have reported that a farnesyl phenolic compound, grifolin, isolated from the fresh fruiting bodies of the mushroom Albatrellus confluens, exhibits extensive bioactivity.67,89–95 Most recently, we demonstrated that grifolin treatment inhibits DNMT1 expression and activity, as well as its mitochondrial translocation. Consequently, it results in demethylation of the pten gene and inhibition of downstream AKT signaling, which effectively attenuates glycolytic flux in NPC cells. Moreover, demethylation of the mtDNA D-loop region and upregulation of oxidative phosphorylation (OXPHOS) complexes are observed with grifolin treatment in NPC cells.67
Metabolic reprogramming and clinical application to Ebv-associated cancers
Glycolysis in NPC
Enhanced glucose metabolism is a common hallmark of cancer. Virus can increase aerobic glycolysis and use glucose biosynthetically.
EBV infection alters glucose metabolism and cause the Warburg Effect. For example, EBV upregulation of all glycolytic enzymes including rate-limiting enzyme hexokinase 2 (HK2), also EBV infection induced substantial relocalization of glucose transporter-1 (GLUT-1) and enhanced glycolytic flux. Elevated glycolysis has been demonstrated by EBV oncoprotein LMP1.96 An earlier study showed that GLUT-1 is highly expressed in the nasopharyngeal tissues of patients with NPC, and its expression is associated with clinical stage, lymph node metastasis, and EB virus infection.97 Some researchers suggested that Epstein–Barr virus-encoded LMP1 changes the metabolic profile and promotes increased glycolysis in NPC cells.98,99 According to Xiao et al.,98 a large-scale metabolic profiling approach showed that EBV (LMP1) regulated metabolic changes in NPC cells and, in particular, the promotion of glycolysis. HK2 was identified as a key modulator of LMP1-induced glycolysis, and conferred proliferative advantages and poor prognosis of NPC patients following radiation therapy. The LMP1-perturbed PI3-K/Akt-GSK3β-FBW7 signaling axis resulted in the stabilization of c-Myc, which was confirmed to be crucial for LMP1-induced glycolysis and confers NPC cells resistance to apoptosis.98 Lu et al.99 revealed a novel mechanism for LMP1-mediated radio-resistance by suppressing the DNA damage response (DDR) through DNA-PK/AMPK signaling. LMP1 reduced the phosphorylation of AMPK and disturbed its subcellular localization after irradiation. The findings showed that inhibition of AMPK signaling also accelerated glucose uptake and lactate production and conferred resistance of NPC cells to apoptosis induced by irradiation. Reactivation of AMPK by metformin, a pharmacological activator of AMPK, could substantially reverse radio-resistance of LMP1-positive NPC cells both in vitro and in vivo.99 EBV epigenetically reprogram several Hox genes expression in NPC cell lines and tumor biopsies. Ectopic expression of HoxC8 inhibits NPC cell growth in vitro and in vivo, modulates glycolysis and regulates the expression of TCA cycle-related genes.100 Recently, Luo et al.67 analyzed the metabolites of 13C6-d-glucose-traced NPC cells by using gas chromatography/mass spectrometry (GC/MS), and found that glycolytic flux in LMP1-overexpressing cells was markedly increased by about 90% compared to that in LMP1-negative cells. In addition, 13C-labeled pyruvate and lactate levels were markedly elevated, whereas 13C-labeled metabolite levels of the tricarboxylic acid cycle, including 13C-citrate, 13C-α-ketoglutarate, 13C-fumarate, and 13C-malate were significantly decreased.67 Importantly, our latest findings revealed that LMP1-positive NPC cells have a powerful mitochondrial fission function to sustain metabolic changes. Moreover, the phosphorylation of mitochondrial dynamin- protein Drp1 (Ser616 or Ser637) was indispensable for the increased glycolytic metabolism regulated by LMP1. The enhancement of glycolysis might be a crucial adaptive function of cancer cells under the high fission state of mitochondria induced by EBV infection stress.101 Increasing evidence suggests the progression of tumors might not only rely on the cancer cells themselves but also be influenced by the surrounding stroma cells. Extracellular vesicles packaged LMP1 activated normal fibroblasts (NFs) to cancer-associated fibroblasts (CAFs) via the NF-κB p65 signaling pathway, which promote tumor progression via autophagy and stroma-tumor metabolism coupling.102
Fibroblast growth factor 1 (FGFR1) signaling is a key pathway in LMP1-mediated growth transformation. LMP1-mediated FGFR1 activation contributes to aerobic glycolysis and transformation of epithelial cells, thereby implicating FGF2/FGFR1 signaling activation in the EBV-driven pathogenesis of NPC.103 EBV-LMP1 induces glycolytic addiction to enhanced cell motility in nasopharyngeal epithelial cells. EBV-LMP1 activates IGF1-mTORC2 signaling and nuclear acetylation of the Snail promoter by PDHE1α, an enzyme involved in glucose metabolism, to enhance cell motility, thereby driving cancer metastasis. The IGF1/mTORC2/PDHE1α/Snail axis correlates significantly with disease progression and poor prognosis in NPC patients.104
EBV infection and enhanced glucose metabolism might drive the development and progression of EBV-associated cancers. These findings provide a new perspective to reveal for the interplay between EBV infection and host metabolism-associated molecules in driving cancer pathogenesis. Understanding these key events might reveal novel therapeutic targets and potentially reverse resistance to treatment therapies in EBV-associated cancers patients.
Targeting glycolysis by natural compounds
Natural compounds derived from plants, fungi, and marine organisms are widely used as anticancer drugs in preclinical/clinical development or in clinic.105 A number of natural compounds that target metabolism-related pathways show inhibition activity on cancer cells.106 Neoalbaconol (NA), isolated from the fruiting body of the mushroom Albatrellus confluens, is a novel small-molecular compound with a drimane-type sesquiterpenoid structure.107 In response to various stimulations, PDK1 can activate phosphoinositide-3 kinase (PI3K) and then Akt, playing pivotal roles in energy metabolism, cell proliferation and migration. By using EBV-positive C666-1 cell line, we found that neoalbaconol inhibits phosphorylation of Akt and downstream targets, including TSC2, mTOR, and p70S6K1.108 Also, neoalbaconol inhibited the activity of key energy metabolic enzyme hexokinase 2 (HK2), which eventually resulted in a striking energy crisis. Furthermore, neoalbaconol treatment not only significantly decreased the glucose concentration in the media but also blocked ATP generation.108 Seahorse XFp cell energy phenotype test showed that cell oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) were decreased after neoalbaconol treatment. In addition, by using a nude mouse model, we found that neoalbaconol decreases C666-1 cell xenograft tumor growth and suppresses the PI3K/Akt pathway in vivo.108 In conclusion, neoalbaconol regulates PDK1/Akt/mTOR/p70S6K1 signaling pathway and glycolytic gene hexokinase, thus decreasing cell glycolysis and ATP synthesis to induce NPC cell death and inhibit tumor growth in vivo. This study is expected to contribute to the rational design and critical assessment of novel anticancer therapies based on natural compounds-mediated glycolysis reprogramming in EBV-related cancer.
Lipid alteration and fatty acid oxidation in EBV-associated cancers
Aberrant Lipid reprogramming is involved in promoting tumor progression and membrane homeostasis. EBVaGC has a different phospholipid and triacylglycerol (TG) expression pattern than EBV-negative gastric cancer. Fifteen of these lipid-related pathways were associated with EBVaGC metabolites. Analysis of the three major lipids, phospholipids, sphingolipids and neutral lipids revealed that TG and ceramide were downregulated, whereas phosphatidylcholine (PC) and phosphatidylethanolamine (PE) were upregulated in EBVaGC. These findings may help explain the mechanism of EBVaGC tumorigenesis.109 Furthermore, we know that in primary nasopharyngeal carcinoma, EBV-LMP1 induces lipid synthesis and lipid droplet formation. Lo et al.110 reported a novel function of LMP1 in promoting nascent lipogenesis. EBV-LMP1 increased the expression, maturation, and activation of steroid regulatory element-binding protein 1 (SREBP1) and its downstream target fatty acid synthase (FASN). In addition, elevated FASN expression was associated with poor survival in patients with invasive disease and NPC. Their findings suggest that LMP1 activation of SREBP1-mediated adipogenesis promotes tumor cell growth and is involved in the EBV-driven pathogenesis of NPC, which propose a new therapeutic protocol for the treatment of locally advanced or metastatic nasopharyngeal carcinoma using lipogenesis inhibitors.
Fatty acids are fundamental cellular components and energy sources. Fatty acid oxidation (FAO) is increasingly recognized as a crucial metabolic characteristic of cancer. For example, a group of the OXPHOS cluster diffuse large B-cell lymphomas (OXPHOS-DLBCL), which is significantly enriched in genes involved in mitochondrial oxidative phosphorylation, predicts potential differences in mitochondrial oxidative metabolism compared with other DLBCL groups. Notably, palmitate is a predominant respiratory fuel in OXPHOS-DLBCLs. NADPH and ATP is mainly supplied through the fatty acid β-oxidation and coupled oxidative phosphorylation, providing growth and proliferation advantages.111
Moreover, lipid reprogramming may play a vital role in mediating radiation resistance in NPC (Fig. 2). An active FAO is a vital signature of NPC radiation resistance, and the rate-limiting enzyme of FAO, carnitine palmitoyl transferase 1 A (CPT1A), was consistently upregulated in these cells.112 The PGC1α-CEBPB interaction promotes CPT1A transcription, which can facilitate FAO, and maximizes ATP and NADPH production, contributing to radiation resistance.113 Furthermore, the IHC scores in a tissue microarray indicated that PGC1α was highly expressed in non-keratinizing undifferentiated NPC, which corresponds to worse overall survival after radiation therapy in NPC.113 More interestingly, the CPT1A-Rab14 interaction plays roles in CPT1A-mediated radiation resistance by facilitating fatty acid trafficking. Inhibition of CPT1A resensitized NPC cells to radiation therapy by activating mitochondrial apoptosis both in vitro and in vivo.112 Targeting the PGC1α/CEBPB/CPT1A/FAO signaling axis by etomoxir (ETO) or genetic knockdown could be a potential strategy to improve the therapeutic effects of radiotherapy in NPC.113 Mechanistic studies of abnormal tumor fatty acid metabolism and radiotherapy resistance will help enrich our understanding of tumor lipid metabolism reprogramming and provide new perspectives and novel molecular targets for tumor radiotherapy resistance. However, how the EBV plays a role in this FAO need further exploration.
Fig. 2.

A schematic to illustrate CPT1A-mediated radiation resistance in NPC. PGC1α-CEBPB interaction promote the transcription, expression and enzyme activity of CPT1A, and CPT1A-Rab14 interaction promote fatty acid trafficking between lipid droplets and mitochondria, which facilitates fatty acid utilization and maximizes ATP and NADPH production, leading to resistance to radiation
Glutamine metabolism in NPC
Glutamine is one of the important precursors contributing to de novo nucleotide synthesis, which provides the nitrogen that is required for purine and pyrimidine nucleotide synthesis. Glutamine synthetase (GS) is responsible for glutamine metabolism in cancer therapeutic responses, in particular under irradiation-induced stress. GS is transcriptionally regulated by STAT5 and highly expressed in radiation-resistant NPC cells. High expression of GS drives metabolic flux toward nucleotide synthesis to enable efficient DNA repair, then acquired radiation resistance and survival advantages. Targeting GS and the associated pathways may provide therapeutic benefits to patients with both intrinsic and acquired radiation resistance.114
One-carbon metabolism in EBV-induced B-cell transformation
One-carbon (1C) has a critical contribution to speedy cell growth in support of embryonic progression, cancer and T-cell activation. Mitochondrial 1C metabolism stands out as one of the highest induced pathways for EBV infection. EBV triggers mitochondrial 1C metabolism in newly infected B cells, and 1C upregulation is necessary for efficient growth, survival, and transformation of these cells. Besides, EBV induced mitochondrial folate metabolism enzyme methylenetetrahydrofolate dehydrogenase 2 (MTHFD2), which was driven transcriptionally by EBV-encoded protein EBNA2 and MYC synergistically. EBV-induced mitochondrial 1C metabolism drives nucleotide, mitochondrial NADPH, and glutathione production. Which provide an attractive rationed for developing novel mitochondrial 1C metabolic inhibitors for the treatment of B lymphomas.115,116
Clinical application of metabolic analysis in EBV-related cancers
Serum metabolic profiling and radiotherapy in NPC
Based on gas chromatography-mass spectrometry (GC-MS) metabolomics coupled with partial least squares-discriminant analysis, an NPC discrimination model was established with a sensitivity of 88% and a specificity of 92%. Seven metabolites, including glucose, linoleic acid, stearic acid, arachidonic acid, proline, β-hydroxybutyrate, and glycerol 1-hexadecanoate, were identified as contributing most to the discrimination of NPC serum from healthy controls. Metabolic footprints of 20 NPC patients treated with standard radiotherapy are visually submitted to validate the model applied for therapeutic evaluation. The coincident rate of the trends of metabolic footprints to the actual clinical prognosis trend was ~80%. The metabolic profiling approach as a novel strategy may be capable of delineating the potential of metabolite alterations in discrimination and therapeutic evaluation of NPC patients.117
18F-FDG-based imaging in EBV-associated tumors
18F-Fluorodeoxyglucose (18F-FDG) positron emission tomography (PET), which identifies viable tumors by detecting enhanced tumor glycolysis, has long been used as an imaging modality for staging NPC and detecting recurrence of the disease. HK2 has been recognized as a therapeutic target. Two NPC clinical investigations showed that high 18F-FDG uptake indicates poor outcome in patients with NPC118,119, which is highly dependent on HK2. Xiao et al.98 suggested that therapeutic strategies targeting multifunctional metabolic genes, such as HK2, would provide more effective treatment options for NPC.
Previously, associations of combined 18F-FDG-PET and MRI parameters with histopathological features depended on tumor grading and correlated strongly with tumor cell expression of Ki-67 and HIF-1α in head and neck squamous cell carcinoma (HNSCC).120 Also, this metabolic imaging biomarker-based parameters are useful for evaluation of prognostic significance. Maximum and mean standardized uptake values (SUVmax, SUVmean) and total lesion glycolysis (TLG) of the primary tumor could predict a poor outcome in NPC patients.121
Recently, the plasma EBV DNA titer has been recognized as a particularly promising prognostic biomarker. For instance, investigations have compared the prognostic value of 18F-FDG PET parameters and the EBV DNA titer during radiotherapy in patients with primary M0 NPC. Combining 18F-FDG-PET- derived parameters and EBV DNA to subtype patients with primary NPC is more beneficent than the conventional TNM system. Furthermore, 18F-FDG PET-derived parameters and the EBV DNA titer can predict outcome in patients with primary NPC, which may represent an imaging-guided and biomarker-guided double evaluation strategy in the future.122
Thus, all the evidences point out that EBV infection effectively disrupts cellular metabolic homeostasis, and understanding these mechanism-based metabolic reprogramming-induced EBV will reveal new avenues for future diagnosis and treatment.
Mitochondria and Ebv-associated tumors
Mitochondria play an important role in cellular energy dynamics, and are the hubs that connect numerous signaling pathways in tumors. Mitochondrial DNA, mitochondrial fission and fusion dynamics, mitochondrial-related cell death are involved in coordinated processes of cellular physiological and pathological regulation, highlighting the multidimensional function of mitochondria in cancers (Fig. 3).
Fig. 3.
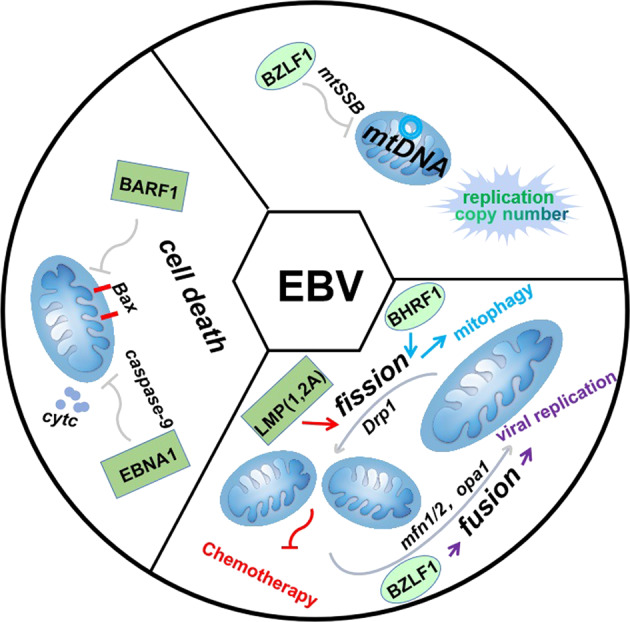
Mitochondria play an important etiological and pathogenesis role in the EBV-associated cancers. EBV has a new function in remodeling of mitochondrial dynamics during latency and lytic infection. EBV-encoded latent oncogene BARF1 and EBNA1 suppress the apoptotic pathways by decreasing Bax translocation to mitochondria and activating casepase-9. The BZLF1 protein encoded during EBV lytic activation inhibits the replication and copy number of mtDNA
Mitochondria DNA (mtDNA)
Mitochondria DNA (mtDNA) and NPC
Mitochondrial biosynthesis and function are, of course, largely controlled by mitochondrial DNA. Ethidium bromide can consume mitochondrial DNA and inhibit the proliferation of tumor cells, indicating that mitochondria are important contributors to tumor growth and invasion.123 One early study demonstrated that mutations in mitochondrial DNA may be involved in the development and progression of NPC. Mutations in mitochondrial DNA are mainly caused by the deletion of its common deletion (CD) of 4981 bp, the accumulation of which in patients might be involved in the development and progression of NPC linked with patient age and clinical stage.124 The mitochondrial genome is also considered to be one of the genetically susceptible areas in NPC. Research has indicated that NPC patients often exhibit higher mtDNA mutation frequency compared to normal nasopharyngeal epithelial tissues. Patients with NPC showed a high frequency of mutation at mtDNA np16362, np16519, and mtMSI D310 and have family heritability.125 The effect of mtDNA haplogroup on NPC occurrence was evaluated in 201 NPC patients with matched controls. Results indicated that patients with haplogroup R9, and its sub-haplogroup F1, in particular, exhibited the most vigorous development of NPC.126
Mitochondria DNA (mtDNA) and EBV
Mitochondria have been known to serve as targets for tumor virus aggression and also to intervene in viral tumorigenesis. BZLF1 is a DNA binding protein belonging to the basic leucine zipper transcription factor family.127 MtDNA replication is initiated by transcription-mediated priming facilitated by the mitochondrial single-stranded DNA binding protein (mtSSB). MtSSB is a tetramer composed of four 16 kDa subunits and binds to mitochondrial DNA at the transcription and replication initiation area.128 BZLF1 targets mtSSB and mediates the translocation of mtSSB from mitochondria into the nuclear compartment, thus inhibiting mtDNA replication, and decreasing mtDNA copy number.129
Mitochondrial dynamics triggered by EBV
Mitochondrial dynamics mainly consist of two mutually restrictive shaping processes: mitochondrial fission and fusion.130 Under severe stress, mitochondria tend to divide and exhibit rounded, fragmented morphology. When subjected to mild stimuli, mitochondria predominantly show a rod-shaped fused morphology.131 At present, mitochondrial dynamics have been used to classify tumors, predict clinical prognosis, and therapeutic response. The key regulators involved in mitochondrial remodeling proteins include mitochondrial dynamin-related protein (Drp1), mitochondrial outer membrane receptor proteins (Fis1, Mff, mid49/51) and mitochondrial fusion proteins (Mfn1/2, OPA1). These shaping proteins contribute more to mitochondrial function and cell metabolism.132 Drp1 is a factor in mitochondrial dynamics that is involved in tumor migration and pathogenesis.133–135 Crucially, phosphorylation of Drp1 has a vital role in the modulation of Drp1 activity. Increasing evidence indicates that Ser616 phosphorylation facilitates the movement of Drp1 from the cytoplasm to the outer mitochondrial membrane. However, Ser637 phosphorylation causes a reversal of this procedure.136
Mitochondrial dynamics and EBV latency infection
Mitochondrial heterogeneity may be responsible for the recurrence and treatment resistance of EBV-associated tumors.67,124 EBV encodes the oncogene LMP1 and latent membrane protein 2A (LMP2A), which play important roles in gene expression and are thought to be involved in EBV-mediated tumorigenesis.137 EBV expresses oncogenic proteins that can interfere with the function of CDKs and induce a number of signals that put their genomes replicate into the host’s cells.138 Recently, we revealed that CDK1 binds to Drp1 in the mitochondria to form a complex that enhances mitochondrial fission. Also, EBV-LMP1 has a novel function in mitochondrial remodeling as evidenced by enhanced mitochondrial fission and glycolytic metabolism through regulation of the cyclin B1/Cdk1/Drp1 signaling axes. This study implies that changes in mitochondrial morphology caused by EBV- encoded oncogenes promote mitochondrial function and reprogramming metabolism.101 Another latent protein, LMP2A, has also been reported to promote mitochondrial fission, induce cell migration, and epithelial–mesenchymal transition (EMT) in gastric and breast cancer cells, suggesting that EBV-driven mitochondrial fission promotes tumor cell survival and mediates drug-resistant therapy.139
Mitochondrial dynamics and EBV lytic infection
Zta (BZLF1 or EB1) and Rta (BRLF1) are immediate early response proteins, which are expressed at the early stage of EBV infection and have been reported to reorganize mitochondrial morphology during EBV lytic replication. Interestingly, a fusion event initiated by BZLF1 expression and the expression of BRLF1 resulted in the relocalization of mitochondria to one side of the nucleus.140 Furthermore, BHRF1 is expressed during the viral productive cycle and is able to translocate to mitochondria and shows strong functional homology with the human Bcl2 protein. BHRF1 prevents type I IFN pathway activation by encouraging mitochondrial fragmentation, which ultimately contributes to autophagosomes degradation by mitophagy.141 Thus, the loss of control of the EB virus stress over mitochondrial structure will be another checkpoint to overcome.
Targeting mitochondrial signaling
Targeting mitochondrial fission or fusion
Proteins involved in mitochondrial fusion and fission may participate in cancer cell resistance to apoptotic stimuli and serve as new therapeutic targets. Based on the observed high fission-to-fusion ratio in apoptotic cells, fission is believed to be a necessary prerequisite for apoptosis.142 The TP53-regulated inhibitor of apoptosis (TRIAP1) was discovered to be aberrantly overexpressed and associated with poor survival in NPC patients. Furthermore, miR-320b is a negative regulator of TRIAP1 and upregulation of miR-320b suppresses NPC cell proliferation and encourages mitochondrial fission and apoptosis.143 1,8-Dihydroxy-3-acetyl-6-methyl-9,10 anthraquinone (GXHSWAQ-1) targets mitochondrial proteins (CDH1, RAC1, CDC42) to enhance radiosensitization by altering the mitochondrial structure, including swollen volume, fragmented crista, and decreasing transmembrane potential.144
Furthermore, contrasting evidence points to Drp-mediated mitochondrial fission as an anti-cell death factor. Most recently, Drp1-regulated mitochondrial fission not only promotes the proliferation of tumor cells, but also participates in the maintenance of cell stemness, affecting tumor invasion and metastasis as well as tumor treatment.145 One study has shown that inhibiting Drp1 activity by interrupting the Cox-2/Drp1 axis, reducing the stemness of NPC cells, and helping to increase its sensitivity to resveratrol and 5-flumidine treatment,146 suggesting that targeting Drp1-dependent mitochondrial fission would be an effective treatment for NPC. In fact, the involvement of Drp1 in tumor pathogenesis and therapeutic sensitization was also confirmed in our report, demonstrating that mitochondrial fission can be used to classify cancers and play an important role in predicting clinical prognosis and therapeutic response of NPC. Notably, the protein level of p-Drp1 (Ser616) was positively related to the clinical stage of NPC. Patients with a high level of p-Drp1 (Ser616) showed shorter DFS and OS. In contrast, a high level of p-Drp1 (Ser637) in NPC patients is associated with a longer DFS and OS, suggesting that either serine 616 or serine 637 phosphorylation of Drp1 is a prognostic biomarker of NPC patients. What makes more sense is that metformin interrupts the AMPKɑ/ p-Drp1 Ser637 signaling pathway or cucurbitacin E targets the cyclinB1-Cdk1/p-Drp1 (Ser616) axis repressing mitochondrial fission.101 This discovery supports the mitochondrial dynamics markers Drp1 Ser616 and Ser637 as crucial targets for NPC in the future.
Targeting mitochondrial induces apoptosis
Previous studies focusing on the regulation of mitochondria in apoptosis indicated that pro-apoptotic proteins translocate to the mitochondrial outer membrane and trigger the source of reactive oxygen species (ROS) and apoptosis directly.147–149 Interrupting mitochondrial function to induce apoptosis may be a novel mechanism for targeted EBV-associated tumor therapy.
EBV-encoded oncogene BARF1 (BamH1-A Rightward Frame-1) is a survival factor by suppressing apoptosis pathways through modulation of the Bcl-2/Bax ratio. Knock down of BARF1 by siRNA induces mitochondrial-dependent cell death.150 In addition, Epstein–Barr nuclear antigen 1 (EBNA1) is the only protein expressed in all three types of latent infection, is also involved in the resistance to mitochondrial-dependent cell death. Triptolide, a diterpene epoxide of the tristeza extract, has been shown to target EBNA1, inhibit proliferation of EBV-positive NPC cells, and induce cell death through a caspase-9-dependent pathway.151 For example, extranodal natural killer/T-cell lymphoma (ENKTL) is characterized by EBV latent infection with the expression of EBNA1 and LMP1. Importantly, targeting hexokinase domain component 1 (HKDC1), which is highly expressed in ENKTL, is associated with mitochondrial dysfunction and oxidative stress, EBV replication, and P-glycoprotein (P-gp) expression.152 The above studies further clarify the role of mitochondria-related cell death in the EBV tumorigenesis process, which will help us to fully understand the molecular mechanism of mitochondria in the pathogenesis of EBV-related tumors, and will be beneficial to the discovery and application of targeted drugs.
Here, we summarize the compounds and gene modification interventions recently associated with mitochondrial-dependent cell death. Pectolinarigenin153 and cinobufagin,154 isolated from an active ingredient in medicinal plants, are novel effective antitumor drug candidates for NPC and mainly inhibit tumor growth by activating mitochondrial-related apoptosis and accumulation of ROS. Bufalin (C24H34O4), a cardiotonic steroid, has been shown to inhibit cell proliferation, decrease the number of total viable cells, and induce G2/M phase arrest and apoptosis in NPC-TW 076 cells through ROS-induced endoplasmic reticulum (ER) stress and TRAIL pathways.155 Celastrol is an effective antioxidant and anti-inflammatory natural compound that inhibits the activity of NPC cells, causes G2/M cycle arrest, and induces mitochondria-related apoptosis.156 Another compound, grape seed proanthocyanidin, has a similar effect in NPC.157 The plant extract, acetogenin, is characterized by a single tetrahydrofuran (THF), which causes G2/M phase arrest, mitochondrial damage and apoptosis, and increases cytosolic and mitochondrial Ca2+ levels in NPCs.158 This antitumor effect is similar to that of the natural product, tetrandrine (TET).159 An interesting study focusing on an essential trace element showed that Na2SeO3 causes cell cycle arrest and affects the mitochondrial-mediated intrinsic caspase pathway with significant anti-proliferative and apoptosis-inducing effects in CNE-2 NPC cells, suggesting that Na2SeO3 might have therapeutic potential in the treatment of NPC.160 Additionally, human nuclear factor with BRCT domain protein 1 (NFBD1), an amplifier of the p53 pathway and also known as a mediator of DNA damage checkpoint protein (MDC1), is vital for protecting cells from apoptosis in NPC cells mediated through the p53-ROS-mitochondrial pathway.161 Liver cancer-1 (DLC-1) is lost at high frequency, mainly due to aberrant promoter methylation, and its tumor suppressor roles are not just limited to liver cancer cells as confirmed in multiple cancers.162 In NPC cells, targeting DLC-1 induces mitochondrial-mediated apoptosis and EMT arrest through the EGFR/AKT/NF-κB signaling pathway.163 Generally, the mobility group box 1 (HMGB1) protein is involved in a variety of biological processes and plays an important role in inflammation, immunity and tumor development.164 One research study revealed that HMGB1 is overexpressed in the HONE-1 NPC cell line and depression of HMGB1 leads to mitochondrial-mediated apoptosis by inhibiting the HMGB1/RAGE pathways165 (Table 4).
Table 4.
Targeted mitochondria-related signaling in NPC
| Targeting molecules | Agents or regulators | Effects | Reference |
|---|---|---|---|
| Mitochondrial dynamics | |||
| AMPKɑ/Drp1 | Metformin | Decreases mitochondrial fission and increases chemotherapy sensitivity to cisplatin | 101 |
| CyclinB1-Cdk1/Drp1 | Cucurbitancin E | Decreases mitochondrial fission and increases chemotherapy sensitivity to cisplatin | 101 |
| COX-2/Drp1 | Resveratrol | Inhibits mitochondrial fission and 5-Fu-resistance | 146 |
| TRIAP1 | Knock down | Enhances mitochondrial fission and apoptosis | 143 |
| CDH1, RAC1, CDC42 | GXHSWAQ-1 | Enhances radiosensitisation by altering the mitochondrial structure | 144 |
| Mitochondrial-related cell death | |||
| Bcl-2 | Pectolinarigenin | Inhibits tumor growth by activating mitochondrial apoptosis and accumulation of ROS | 153 |
| Bcl-2 | Cinobufagin | Blocks cells at the S phase, increases intracellular ROS levels, and induces mitochondrial apoptosis pathway | 154 |
| TRAIL | Bufalin | Induces G2/M phase arrest and mitochondrial cell death | 155 |
| MAPK | Celastrol | Triggers G1 and G2/M phase cell cycle arrest and Fas-mediated apoptosis | 156 |
| Bax/Bcl-2 | GSPs | Induces apoptosis through the mitochondrial pathway and ultimately reduces cell viability | 157 |
| c-Jun/ERK | THF-ACGs | Inducers of the ESR that block cell proliferation | 158 |
| Bax, Bcl-xL, Bcl-2 | Tetrandrine | Causes G0/G1phase arrest; increases ROS and Ca2+ production and apoptotic cell death | 159 |
| Bax, Bak, Bcl-XL | Na2SeO3 | Inhibits cell proliferation and induces apoptosis | 160 |
| NFBD1/MDC1 | Knock down | Induces apoptosis through the p53/ROS/mitochondrial pathway | 161 |
| DLC-1 | Knock down | Leads mitochondrial apoptosis by targeting EGFR/AKT/NF-κB | 163 |
| HMGB1 | Knock down | Decreases mitochondrial fission and increases chemotherapy sensitivity to cisplatin | 165 |
Mitochondria play an important role in the pathogenesis of tumors by affecting mitochondrial DNA, dynamics, and cell death. The disruption of normal mitochondrial physiology is a hallmark of tumor development. Importantly, EBV- encoded proteins modulate mitochondrial function and thus alter bioenergetics and retrograde signaling pathways to promote tumorigenesis. Therefore, identification of effective mitochondrial molecules and signaling pathways in EBV-associated tumor is essential for developing novel therapeutic strategies.
EBV and the ubiquitin-proteasome system (Ups)
Post-translational modification of proteins by ubiquitin (Ub) is a key regulatory event in various cellular activities. Ubiquitination is a dynamic and reversible process, wherein ubiquitin chains are cleaved by deubiquitinase (DUB).166 Ubiquitin signaling comprises three major components of the ubiquitination process known as ubiquitin-activating enzyme (E1), ubiquitin-conjugating enzyme (E2), and ubiquitin ligase (E3). E1 activates and transfers ubiquitin to the E2 enzyme and then E2 interacts with the E3 ligase. The E3 ligase binds and transfers ubiquitin from the E2 enzyme to target-specific proteins for degradation. The deregulation of the ubiquitin system leads to human pathogeneses, especially cancer. Alterations in the ubiquitin system are known to occur during the initiation and progression of cancer, and this knowledge is starting to be exploited for the development of novel strategies to treat cancer.167
DUBs maintain ubiquitin system homeostasis by cleaving polyubiquitin chains or completely removing ubiquitin chains from ubiquitinated proteins and then generating and recycling free ubiquitin. Deubiquitination also has an important function in regulating ubiquitin-dependent pathways, including cell cycle regulation, cell death, protein degradation, protein function, gene expression, and signal transduction.168,169 Imbalances in DUB activities are involved in multiple diseases, including cancer, inflammation, neurological disorders, and microbial infections. DUB inhibitors (DIs) are becoming potential therapeutic approaches against cancer.170 Most DIs are small-molecular compounds, exerting their function by suppressing DUB activity. Thus far, about 100 DUBs have been identified and classified into two categories, including cysteine proteases and metalloproteases. The cysteine proteases comprise ubiquitin-specific protease (USP), ubiquitin C-terminal hydrolase (UCH), Machado-Josephin domain protease (MJD), ovarian tumor protease (OTU), and motif interacting with Ub-containing novel DUB family (MINDY). The metalloproteases comprise Jab1/Mov34/Mpr1 Pad1 N-terminal (MPN) (JAMM) domain protease.171,172
EBV-encoded proteins and ubiquitin system
All viruses need host machinery to maintain infection and replication. UPS is involved in all aspects of cellular activity. Oncoviruses rely on this system at many levels and even hijack the ubiquitin system to meet their survival needs. The same is also true for EBV. EBV nuclear antigen 1 (EBNA1) plays important roles in EBV latent gene expression. EBNA1 competitively interacts with the p53 binding site on ubiquitin carboxyl-terminal hydrolase 7 (USP7), which is also known as herpes virus-associated ubiquitin-specific protease (HAUSP). The EBNA1 and USP7 interaction can promote cell survival and contribute to EBNA1 functions at the EBV oriP and inhibit p53-mediated antiviral responses.173 EBV-encoded latent membrane protein 1 (LMP1) can induce the expression of ubiquitin carboxyl-terminal hydrolase isozyme L1 (UCHL1) and it may contribute to viral transformation and the progression of lymphoid malignancies.174 EBV early-lytic phase gene BRLF1 encoded Rta, which initial viral gene transcription. Rta can be ubiquitinated by E3 ubiquitin ligase TRIM5a. Overexpression of TRIM5a reduces the transactivating capabilities of Rta, whereas reducing TRIM5a expression enhances EBV lytic protein levels and DNA replication.175
Besides utilizing host DUBs, EBV can also encode the viral deubiquitinating enzyme (v-DUB), BPLF1, which is an immune evasion gene product that can suppress antiviral immune responses during primary infection. BPLF1 is expressed during the late phase of lytic EBV infection and is incorporated into viral particles. It can eliminate K63- and/or K48-linked ubiquitin chains and act as an active DUB during the productive lytic cycle and EBV infection.176
Disturbing of ubiquitin-proteasome system in EBV-related cancer
Cell cycle
Cell cycle regulation is one of the hallmarks of virus-mediated oncogenesis. EBV-encoded LMP1 is an important tumorigenic protein. Unlike many other cancers, p53 is highly expressed in NPC. LMP1 enhances p53 accumulation through two distinct ubiquitin modifications. LMP1 promotes p53 stability by suppressing K48-linked ubiquitination of p53 by E3 ligase murine double minute 2 (MDM2). LMP1 inhibits MDM2-mediated p53 ubiquitination associated with p53 Ser20 phosphorylation and also induces K63-linked ubiquitination of p53 by interacting with TRAF2, thus disrupting tumor cell apoptosis and cell cycle arrest.177 Another essential EBV latent protein 3C (EBNA3C) stabilizes Cyclin D2 to regulate cell cycle progression by directly binding to Cyclin D2, and co-localizing together in nuclear compartments.178 EBNA3C interacts with Bcl6 and facilitates its degradation through the ubiquitin-proteasome-dependent pathway, and suppresses Bcl6 mRNA expression by inhibiting the transcriptional activity of its promoter. EBNA3C-mediated Bcl6 regulation significantly promotes cell proliferation and cell cycle by targeting Bcl2 and CCND1.
During viral lytic replication, EBV protein kinase (PK) phosphorylates p27Kip1 so that it is ubiquitinated by the Skp1/Cullin/F-box protein (SCFSkp2) ubiquitin ligase and degraded in a proteasome-dependent manner. Unlike the cyclin E-CDK2 activity, the EBV PK activity is not inhibited by p27Kip1. Overall, EBV possesses its own strategy to degrade p27Kip1 upon onset of productive replication, contributing to provision of an S-phase-like cellular environment with high CDK activity.179
Cell death
Cell death is a critical component of the host immune response against invading microbial pathogens. As is discussed above, EBV-LMP1 disrupting tumor cell apoptosis through p53. EBV is also capable of preventing necroptosis. Necroptosis is a relatively newly discovered pathway of programmed cell death, the deregulation of which is related to various inflammatory diseases and cancer.180 LMP1 is armed with mechanisms independent of the RIP homotypic interaction motif (RHIM)-domain interactions to block necroptosis. First, LMP1 is able to interact with both receptor-interacting protein kinase 1 (RIPK1) and receptor-interacting protein kinase 3 (RIPK3) by C-terminal activation region 2 (CTAR2), which is necessary for this interaction. Breaking the combination of RIPK1 and RIPK3 inhibits the formation of necrosomes. Second, and more importantly, the ubiquitination of the two receptor-interacting proteins can be modulated by LMP1. LMP1-mediated promotion of K63-polyubiquitinated RIPK1, suppression of RIPK1 protein expression, and inhibition of K63-polyubiquitinated RIPK3 induces a switch in cell fate from necroptotic death to survival.181
DNA damage
DNA damage signaling and repair pathways are important determinants of both cancer development and cancer therapy. EBV nuclear protein expressed in gastric carcinomas (GC), EBNA1, focusing on promyelocytic leukemia (PML) nuclear bodies (NBs), which play important roles in apoptosis, p53 activation. EBV infection of GC cells leads to degradation of PML NBs through the action of EBNA1 interacting with USP7, resulting in impaired responses to DNA damage and promotion of cell survival. Therefore, PML disruption by EBNA1 is one mechanism by which EBV may contribute to the development of gastric cancer.182 O6-methylguanine-DNA methyltransferase (MGMT) is a key DNA repair enzyme contributing to the chemoresistance, which protects NPC cells from DNA damage by enhancing the capacity for DNA repair.183 Research findings showed that ubiquitin-conjugating enzyme E2 B (UBE2B) collaborates with E3 ubiquitin ligase RAD18 to mediated MGMT ubiquitination and degradation.184 Rad18 is also responsible for monoubiquitination of PCNA, which initiates several cellular DNA repair processes. V-DUB BPLF1 targets ubiquitinated PCNA and disrupts trans-lesion synthesis (TLS). Deubiquitination of PCNA by the viral DUB can disrupt repair of DNA damage by compromising recruitment of TLS polymerase to stalled replication forks.185 BPLF1 interacts directly with Rad18, and overexpression of BPLF1 results in increased levels of the Rad18 protein, suggesting that it stabilizes Rad18. Next, expression of functionally active BPLF1 causes relocalization of Rad18 into nuclear foci, which is consistent with sites of cellular DNA replication that occur during S phase, which contributes to virus replication and infectivity.186 Transcription factor p53 is a critical mediator that is directly phosphorylated and stabilized by DNA damage signaling kinases, which triggers the apoptotic program.187 Zhang et al.188 found that RING-finger domain-containing ubiquitin E3 ligase tripartite motif–containing 21 (TRIM21) repressed p53 expression by mediating guanine monophosphate synthase (GMPS) ubiquitination and degradation. TRIM21 protects NPC cells from radiation-induced apoptosis by manipulating the GMPS–p53 cascade.188 In addition, TRIM21 is also responsible for the polyubiquitination of prohibitin 1 (PHB1), a pleiotropic protein that functions as a tumor suppressor. Ubiquitination and degradation of PHB1 leads to transcriptional activation of NF-κB and STAT3.189 TRIM21 mediates small G-protein signaling modulator 1 (SGSM1) ubiquitination degradation and inhibits the MAPK pathway activation.190 Deubiquinase ubiquitin carboxyl-terminal hydrolase L1 (UCHL1) from the UCH family forms complexes with p53/MDM2/ARF and deubiquitinates p53 promoting p53 signaling, which is involved in NPC pathogenesis.191
Akt signaling
Deregulation of protein kinase B (Akt) signaling plays a vital role in the regulation of tumorigenesis in human cancers.192 K63-linked ubiquitination of Akt is required for Akt membrane translocation and activation.193 E3 ligase S-phase kinase-associated protein 2 (Skp2) overexpression is correlated with poor prognosis in NPC patients, Skp2-mediated ubiquitination and mitochondrial localization of Akt drive tumor growth and resistance to cisplatin.194,195 TNF-receptor-associated factor (TRAF) proteins are key adaptor molecules containing E3 ubiquitin ligase activity, which plays a critical role in multiple cell signaling pathways. TRAF6 binds to and ubiquitinates Krüppel-like factor 4 (KLF4), a complex transcription factor that acts as a tumor-promoting gene in NPC. This leads to KLF4 K32/K63-linked ubiquitination and stabilization, which results in NPC196 (Fig. 4).
Fig. 4.
EBV-encoded proteins modulate host ubiquitin signaling pathways. EBV regulates degradation and non-degradation ubiquitination of key molecules in cell signaling pathways through hijacking host ubiquitinase and deubiquitinase, or directly encoding viral deubiquitinase. The ubiquitinases in orange cycle represents activated by EBV protein, and the gray represents for suppressed ubiquitinase
The natural compound, neoalbaconol, targets the ubiquitin system in EBV-related cancer
The ubiquitin-proteasome system (UPS) is emerging as a novel target for anticancer drugs and molecular diagnostics.197 Escape from programmed cell death plays a critical role in cancer. Therefore, investigating the role of necroptosis in cancer has been of high interest. We found that necroptosis is one of the major cell death forms induced by neoalbaconol. Natural compounds derived from plants, fungi, and marine organisms are widely used as anticancer drugs in preclinical/clinical development or in clinic.105 Neoalbaconol down-regulates E3 ubiquitin ligases cellular inhibitors of apoptosis protein 1/2 (cIAP1/2) and TRAFs to abolish K63-linked ubiquitination on RIPK1.198 This induces the activation of the non-canonical NF-κB pathway and increases the transcription activation of TNFα. Moreover, neoalbaconol induces RIPK3-mediated reactive oxygen species (ROS) production that participates in necroptosis.198 RIPK1/NF-κB-dependent TNFα secretion and RIPK3-dependent ROS generation contribute to neoalbaconol-induced necroptosis.
Ubiquitinases and deubiquitinases are emerging as potential therapeutic targets in many diseases, especially cancer. In this chapter, we summarize the involvement of these enzymes in cancer signaling and cell regulation and illustrate the potential role of the ubiquitin system in response to cancer therapeutics (Table 5). We also discussed how the onco-virus EBV fights with the host by utilizing host ubiquitin signaling or even encoding v-DUB to hijack host cellular processes. Deciphering the various functions of ubiquitinases and deubiquitinases will be challenging but should help provide a greater understanding of how its deregulation results in tumorigenesis and development. As for the potential therapeutic targets, the ultimate challenge is to translate these basic research findings into novel and more effective cancer therapies.
Table 5.
Ubiquitinase in EBV-related tumorigenesis
| Type | Enzyme | Target | Function | Reference |
|---|---|---|---|---|
| Ubiquitin E3 ligase | UBE2B | MGMT | Chemoresistance | 184 |
| RAD18 | MGMT | Chemoresistance | 184 | |
| TRIM21 | GMPS | Apoptosis inhibition | 188 | |
| PHB1 | Cell proliferation | 189 | ||
| SGSM1 | MAPK pathway activation | 190 | ||
| Skp2 | Akt | Chemoresistance and cell proliferation | 193 | |
| TRAF6 | KLF4 | Tumorigenesis | 196 | |
| TRAF2 | P53 | Disrupting tumor cell apoptosis | 177 | |
| TRIM5a | Rta | Inhibit EBV lytic replication | 175 | |
| Deubiquintylase | UCHL1 | p53 | Tumorigenesis | 191 |
| USP7 | p53 | Inhibit antiviral responses | 173 | |
| v-Deubiquintylase | BPLF1 | PCNA | Disrupt repair of DNA damage | 185 |
Oxidative stress in EBV-associated diseases
The regulation of redox homeostasis is fundamental to maintaining normal cellular functions and ensuring cell survival. Cancer cells are characterized by increased aerobic glycolysis and high levels of oxidative stress.199 This oxidative stress is exerted by reactive oxygen species (ROS) that accumulate as a result of an imbalance between ROS generation and elimination.200 ROS are constantly produced by both enzymatic and non-enzymatic reactions.200 Enzyme-catalyzed reactions that generate ROS include those involving NADPH oxidase, xanthine oxidase, uncoupled endothelial nitric oxide synthase (eNOS), arachidonic acid and metabolic enzymes such as the cytochrome P450 enzymes, lipoxygenase and cyclooxygenase. The mitochondrial respiratory chain is a non-enzymatic source of ROS.200 The produced ROS include superoxide, hydrogen peroxide, singlet oxygen and reactive nitrogen species. Usually, determination of cellular redox status by a balance between levels of ROS inducers and ROS scavengers. The production of reactive oxygen species (ROS) can be induced by hypoxia, metabolic defects, endoplasmic reticulum (ER) stress and oncogenes. Conversely, ROS are eliminated by the activation of the transcription factor nuclear factor erythroid 2-related factor 2 (NRF2), the production of glutathione and NADPH, the activity of tumor suppressors (such as breast cancer susceptibility 1 (BRCA1), p53, phosphatase and tensin homolog (PTEN) and ataxia telangiectasia mutated (ATM)) and the action of dietary antioxidants.200 Nowadays, many compounds have been described as antioxidants. These include compounds with free sulfhydryl groups, including N-acetylcysteine (NAC) and lipoic acid, compounds with multiple double bonds and conjugation (e.g., carotenoids, tocopherols and retinoids, among others) and polyphenols (e.g., epicatechingallate and quercetin, among others). Compounds that inhibit reactive oxygen generation (i.e., NADPH oxidase inhibitors), xanthine oxidase inhibitors and compounds that induce oxidant defenses (e.g., Nrf2 activation-sulforaphane, among others) have also been allocated to this category.201
ROS-mediated oxidative stress has an increasingly well-recognized broad function in oncogenesis.202 Generating oxidative stress is a critical mechanism by which host cells defend against infection by pathogenic microorganisms.203 Several reports have shown that both DNA and RNA viruses can lead to oxidative stress by encoding various products and play roles in virus survival. For example, HBV encoded HBx204 and HSV-1 encoded UL12.5205 can induce oxidative stress by degradation of mtDNA; KSHV encoded Rac-1206 and HIV encoded Gp120, Tet or Nef207 can induce oxidative stress by regulating the NOX pathway.
EBV, a member of γ-herpesviruses, is an important cancer causing virus.4 NPC is an infection-associated cancer strongly driven by EBV.11,208 So far, several evidences showed increased levels of ROS in cells and in NPC patients. In vitro, EBV infection can induce an increase in malondialdehyde (MDA) levels and decreases in catalase and superoxide dismutase (SOD) activities. Additionally, a positive correlation was observed between BZLF1 and antioxidant enzyme genes expression, which also confirmed a role of EBV lytic reactivation in oxidative stress.209 Notably, reactive oxygen signaling can distinguish EBV-positive versus EBV-negative tumors based on the finding that elevated levels of ROS are observed in EBV-positive tumors but not in EBV-negative tumors.210 On the other hand, oxidative stress inducers, such as H2O2 or FeSO4, can cause EBV lytic cycle induction as demonstrated by BZLF1 gene expression.211 Moreover, various drugs that induce or inhibit EBV lytic reactivation also act by generating oxidative stress.212,213 Thus, a vicious cycle could be initiated whereby reactivation of EBV and ROS production amplify each another. In vivo, overexpression of iNOS and the accumulation of 8-OHdG, critical biomarkers of oxidative stress, were found in NPC patients.214,215 Huang et al.216 detected the serum 8-OHdG using ELISA in NPC patients and found that all cases of NPC were positive for 8-OHdG and therefore, 8-OHdG is proposed as a potential biomarker for evaluating the risk of NPC. Su et al.217 observed decreased SOD activity in NPC that is consistently associated with EBV. These observations indicate that NPC biopsies present a high level of oxidative stress.
Moreover, other EBV-associated tumors also present high levels of oxidative stress. For instance, Cerimele et al.210 demonstrated that EBV-positive BL expresses high levels of activated mitogen-activated protein kinase and reactive oxygen species (ROS), and that ROS directly regulate NF-κB activation. In EBV-associated gastric cancers, Kim et al.218 reported EBV induces high levels of oxidative stress and thus regulates cell viability.
Additionally, it is reported that several EBV-encoded products, including EBNA1, LMP1, and EBER are associated with oxidative stress. Possible mechanisms by which EBNA1 could induce ROS, include increasing levels of oxidases (e.g., NOX1 and NOX2) or affecting the levels of antioxidant enzymes, such as SOD1, peroxiredoxin 1, peroxiredoxin 6, or glutathione S-transferase.16,219,220 The mechanism of inducing oxidative stress by LMP1 was via activating of NOX2 and Nrf2 pathway (unpublished data). EBER that induces interleukin (IL)-10 and IL-10 has been implicated in the activation of mitochondrial ROS.210 Based on the reported preclinical and clinical evidence, it is indicated the oxidative stress is involved in EBV infection.
Bonner et al.201 has classified tumors by signaling pathways and proposed a concept of ROS-driven tumors. So far, several types of tumors have been classified into reactive oxygen species (ROS)-driven tumors and include EBV-associated Burkitt’s lymphoma, Hodgkin’s disease, gastric carcinoma, hepatitis C virus-related hepatocellular carcinoma, ultraviolet A-induced melanoma, inflammatory bowel disease-induced colon carcinoma, schistosomiasis-induced bladder cancer, tobacco-induced oral and lung carcinoma, and epidermolysis bullosa-associated squamous cell carcinoma. These cancers are characterized by activation of high expression levels of NF-κB, Akt and wild-type p53 and PTEN.201 According to the signaling events in the reactive oxygen-driven tumor and the fact of high oxidative stress in NPC, it is proposed that NPC is a reactive oxygen-driven tumor.
Regarding to the important role of ROS in EBV-associated diseases, the monitoring and detection of ROS is vital important to the therapy of EBV-related malignancies. So far, there are several ways to detect the level of intracellular oxidative stress. (a) Total ROS level (combined with ROS dye by flow cytometry); (b) mitochondria ROS level (using flow cytometry combined with mitochondrial-specific ROS dye MitoSox red); (c) intracellular NADP+/NADPH ratio (NADP+/NADPH kit); (d) intracellular GSSG/GSH ratio (GSSG/GSH kit); (e) Expression and activity of oxidoreductase. For the detection of oxidative stress level of tumor patients, pathological samples and serum samples can be collected, which can be reflected by detecting the content of oxidative stress marker 8-hydroxydeoxyguanosine (8-OHdG). 8-OHdG is one of the most common DNA damage products in response to oxidative stress, and has been recognized as a reliable and stable biomarker of oxidative stress.221,222 The level of 8-OHdG in tissue samples is positively correlated with the degree of oxidative stress, so the level of 8-OHdG in blood or pathological tissue samples can be detected by ELISA or immunohistochemistry to reflect the degree of oxidative stress.
Scavenging ROS might be a good way for NPC prevention and therapy. In our group, we demonstrated that scavenging ROS by NAC can decrease the level of ROS and increase the sensitivity of NPC to radiation (unpublished data). In addition, natural compounds, normally found in foods, are gaining increased attention due to their proposed cancer preventive and therapeutic activities and potential greater safety compared to synthetic compounds.223 In therapy of EBV-associated cancers, curcumin, resveratrol and EGCG reportedly show great potential.224–226
EBV lytic reactivation in EBV-associated diseases
Like all herpesviruses, EBV establishes a latent infection that is periodically reactivated into the productive lytic cycle.227 As showed in Fig. 5, EBV-encoded different products in latency and lytic phase. Following reactivation, the lytic genes of EBV are expressed in a temporally regulated manner, including immediate early (IE), early (E) and late (L). The latent-lytic switch is tightly controlled by both cellular and viral factors.227 About the history of EBV lytic research, there are several major milestones. In 1985, VCA-IgA levels detected in mass serological screening of NPC patients in China, confirming NPC risk.21 Countryman et al.228 demonstrated that BZLF1 is the switch from latent to lytic cycle. In 1987, EBV DNA was detected in HL biopsies.228 In 1989, Q-PCR was used to detect cell-free EBV DNA as NPC biomarker.23 The latest reports showed an important transcription factor MYC can also control the EBV lytic switch.229 EBV ncRNA from an NPC induces an inflammatory response that promotes virus production.230
Fig. 5.
EBV encodes different products in latent and lytic infection. During latency, a limited number of viral products, such as EBNAs, LMP1, LMP2A, LMP1 2B, EBERs are expressed and the viral genome exists in the nucleus as an episome. Upon reactivation, EBV briefly passes through three consecutive lytic phases, including immediate early (IE), early (E), and late (L). The viral IE genes BZLF1 and BRLF1 are first transcribed to encode the transactivators, Zta and Rta, respectively, followed by expression of the early genes required for EBV genome replication. After EBV DNA replication, late genes are expressed that encode mainly viral structural proteins, including capsid antigens and membrane proteins, followed by viral genome encapsulation and the production of mature virions
EBV lytic cycle and tumorigenesis
In recent years, evidence of a contribution of the lytic cycle to EBV-induced oncogenesis has emerged and has become a hot topic in this field.4,65,231 EBV lytic phase is associated with NPC, GC, and multiple lymphomas, including Burkitt lymphoma, Hodgkin lymphoma et al. and confers therapeutic resistance to tumor cells.4 In our lab, we also indicated that the EBV spontaneous lytic infection exists in NPC and lymphoma cells.36 Notably, in NPC, the expression of lytic EBV genes was observed in the upper layers of differentiated epithelium but not in the undifferentiated basal layers.10,34 Interestingly, Christian analyzed the degree of EBV lytic in EBV-related diseases. The sequence of lytic replication is: oral hairy leukoplakia > infectious mononucleosis > primary effusion lymphoma > nasopharyngeal carcinoma/gastric carcinoma > diffuse large B-cell lymphoma/Burkitt’s lymphoma/post-transplant lymphoproliferative disease/NK-cell or T-cell lymphoma > Hodgkin’s lymphoma.231
In addition, EBV lytic reactivation is also related to several other diseases, including multiple sclerosis (MS), systemic lupus erythematosus (SLE), rheumatoid arthritis (RA) and Sjogren’s syndrome (SS). MS is a chronic and disabling inflammatory and neurodegenerative disease of the central nervous system that affects 350,000 people in the USA and ~2 million worldwide. It is reported that MS caused an increased anti-EBV antibodies titer against multiple antigens and MS risk was up to 30-fold higher in individuals with the highest titers of anti-EBV antibodies as compared to those with the lowest.6,232 SLE is a multisystem autoimmune disease with varied presentations, progression, and symptoms experienced by patients. Patients with SLE have high frequencies of EBV-infected cells, higher plasma viral loads and higher early antigen IgG levels, which suggest active EBV lytic replication in SLE patients and serological measures of EBV reactivation may provide additional biomarkers to identify individuals at higher risk transitioning to clinical SLE.6,233 RA is a common autoimmune disease with a prevalence of ~1% and an incidence of 5–50 per 100,000 per year with three times more female than male patients. Common symptoms include arthritis, cardiovascular complications, metabolic syndrome, cognitive dysfunction, and depression. Presence of EBV DNA/RNA has been demonstrated in PBMCs, saliva, synovial fluid, and synovial membranes of RA patients. Furthermore, 10-fold higher frequencies of EBV-infected B-cells have been observed in RA patients compared to healthy controls. Interestingly, EBV DNA was found in many of the plasma cells producing cyclic citrullinated peptide (CCP) antibodies localized in synovial tissues of RA patients. These results indicate a widespread lytic EBV infection in RA patients.61,234 SS is a rather common autoimmune disease with a prevalence of about 0.5% and an incidence of 3–6 per 100,000 per year with a female preponderance (nine out of 10 SS patients are women). Increased viral load and EBV proteins have been found in salivary and lacrimal glands of SS patients indicating active infection, and elevated levels of EBV-directed antibodies have been found in the circulation.235 The above evidence demonstrates that the MS, SLE, RA, and SS patients have active EBV lytic replication. Elimination of the infection or reduction of the viral load by reducing the antigenic stimulation could reduce titers of autoantibodies and possibly have a clinical benefit.
Research findings have documented that EBV lytic reactivation is a major risk factor for the development of EBV-related diseases. They mainly contribute to the induction of genome instability, the counteraction of host immune responses, the resistance of cell death and the promotion of tumor development, progression and invasiveness.236,237 The EBV lytic proteins and tumorigenic functions are listed in Table 6. Additionally, induced activation of EBV contributes to carcinogenesis. It has been reported several lifestyle factors can induce transformation of EBV from the latent to the lytic stage and contribute to NPC occurrence, such as salted fish or preserved food.238 Besides, cigarette smoking can induce EBV lytic reactivation and increase the risk of NPC.239 Moreover, several cellular stresses were also reported to contribute to EBV reactivation and lead to an increase of NPC risk, such as oxidative stress, hypoxia and inflammation.237 In addition, lytic EBV replication might condition the tumor microenvironment for EBV-associated malignancies through attraction of monocytes via CCL5 and their differentiation into immune-suppressive tumor-associated macrophages (TAMs).231 The above-mentioned function of EBV lytic reactivation indicated that the lytic cycle plays an important role in the pathogenesis of EBV-related diseases.
Table 6.
EBV lytic proteins and tumorigenic functions
| Lytic protein | Lytic phase | Tumorigenic function |
|---|---|---|
| Zta | IE | Induction of IL-6, IL-8, IL-10, IL-13, and VEGF secretion |
| Rta | IE | Induction of genomic instability and progressive malignancy |
| BHRF1 | E | Anti-apoptosis |
| BARF1 | E | Anti-apoptosis |
| BCRF1 | E | Anti-apoptosis, homologous to IL-10 |
| BALF3 | E | Induction of genomic instability |
| BGLF4 | E | Induction of chromosomal abnormality and DNA damage |
| BGLF5 | E | Induction of genomic instability |
| DNase | E | Induction of genomic instability |
| BNLF2a | E | Immune evasion (inhibits the transporter of antigen processing) |
| BILF1 | E | Immune evasion (induces major histocompatibility complex (MHC) class I internalization and degradation) |
| BGLF5 | E | Immune evasion (mediates host shut‑off) |
| BNRF1 | L | Induction of centrosome amplification and chromosomal instability |
Currently, detection of EBV based on EBV lytic reactivation might be one of the most important approaches for the prevention and personalized treatment of EBV-associated cancers. For example, Rta-IgG, EAD-IgA, VCA-IgA, etc. are antibodies produced based on products of EBV lytic activation.4,240 In addition, in areas with high incidence of NPC, detection of EBNA1-IgA and VCA-IgA by ELISA has become a routine screening method for high-risk groups of NPC, and has been incorporated into the NPC screening standard of the National Health and Family Planning Commission.241 In addition, accumulating research data supports the use of plasma or serum EBV DNA as a tumor marker.242–246 Plasma or serum EBV DNA is gradually being adopted in clinical applications and has been shown to be correlated with tumor burden, TNM stage, response to therapy, and survival in NPC patients. Most notably, two phase 3 trials are under way, in which patients are randomly assigned to adjuvant regimens of differing intensities depending on whether EBV DNA is detectable after chemoradiotherapy (NCT02135042 and NCT00370890, Clinical Trails.gov).208 In addition, NP brush/swab sampling combined with EBV DNA detection has been a valuable supplement in the diagnosis of NPC because of its high sensitivity and specificity.247
EBV DNA detection also has a board application in other EBV-associated diseases except NPC. (1) For IM patients, the detection of EBV DNA may be helpful to cases in which EBV antibody detection results are difficult to explain. (2) For immunodeficiency patients related to EBV infection, such as XLPD and PTLD, duo to the insufficient of the antibody in immunodeficiency patients, the detection of EBV DNA is helpful for the diagnosis of primary EBV infection, and whole blood or PBMC specimens is recommended for EBV detection. (3) CAEBV (Chronic active EBV infection) is a serious EBV infection disease, and the EBV load in peripheral blood of CAEBV patients is significantly higher than that of healthy carriers. The EBV DNA in serum or plasma of patients with CAEBV was positive, or the EBV DNA in PBMC of peripheral blood was higher than 10^2.5 copies/μg DNA. So, the detection of EBV DNA is a good method for CAEBV. (4) For lymphoma patients, serum/plasma EBV DNA can be used as a marker of tumor burden in patients with EBV-related lymphoma, and may be used for treatment effect evaluation and prognosis judgment.
Antiviral agents targeting EBV lytic cycle
EBV infection has been shown to significantly contribute to carcinogenesis. Thus, Screening for anti-EBV agents have been developed as novel strategies for treatment of EBV-associated malignancies. So far, most of anti-EBV agents were targeting the proteins expressed in EBV lytic cycle. Valpromide (VPM), an amide derivative of valproic acid (VPA), has been proven to prevented expression of two EBV genes, BZLF1 and BRLF1, which mediate lytic reactivation. Unlike VPA, VPM did not activate cellular gene expression. VPM will be useful in probing the mechanism of EBV lytic reactivation and may have therapeutic application.248,249 Verma et al.250 have identified that Spironolactone, a mineralocorticoid receptor antagonist approved for clinical use, inhibits infectious EBV production by inhibiting EBV SM protein function. SM protein is a regulatory protein early in lytic replication of EBV, which is essential for efficient gene expression and infectious virion production. Maribavir (MBV), which is newly approved drug for use against human cytomegalovirus (HCMV) infection in allogeneic stem cell and bone-marrow transplant recipients, is of special interest because it is also a potent inhibitor of EBV replication.251 MBV on EBV lytic replication is produced mainly by inhibiting the enzymatic activity of EBV-PK and thus the viral DNA replication, as well as through suppression of EBV lytic gene expression.252,253 It’s worth noting that DNAzyme (DZ1) that was engineered to specifically target the LMP1 mRNA, which is a latent oncoprotein of EBV.254 As the research progresses, increasing evidence indicate that the high expression of LMP1 during EBV lytic cycle can promote the viral lytic replication. In our previous research, we have found that EBV DNA copy number was significantly decreased in DZ1-treated patients.255 Which consisted the critical role of LMP1 in EBV lytic reactivation. These suggesting that lytic inhibition may be an important mechanism for DZ1 against EBV-related cancer.
Currently, a wide variety of active phytochemicals or dietary ingredients have been identified as inhibitors of the EBV lytic cycle256–263 (Table 7). Most of the natural compounds act as antiviral agents by inhibiting lytic proteins. Our previous studies have shown that EGCG could effectively inhibit the constitutive lytic infection of EBV at the DNA, gene transcription and protein levels.264 And the molecular mechanism underlying EGCG inhibition of EBV lytic infection involves downregulation of LMP1.265 Through inhibiting the EBV lytic cycle, anti-EBV agents may decrease viral particle production, block the transmission of the virus from cell to cell and are, therefore, valuable in chemoprevention or clinical treatment of EBV-associated malignancies. Although these were still in vitro studies. These findings pave the way for development of antiviral drugs with new mechanisms of action directed against EBV in the future.
Table 7.
Antiviral agents targeting EBV lytic cycle in EBV-related diseases
| Agents | Mechanism | Model | Reference |
|---|---|---|---|
| Valproic acid | Inhibits Zta and Rta expression | In vitro (Burkitt’s lymphoma) | 248 |
| Valpromide | Inhibits Zta and Rta expression | In vitro (Burkitt’s lymphoma) | 249 |
| Maribavir | Inhibiting the enzymatic activity of EBV-PK | In vitro (Burkitt’s lymphoma) | 252,253 |
| Spironolactone | Inhibits SM expression | In vitro (Burkitt’s lymphoma, GC) | 250 |
| Dnazyme | Inhibits LMP1 expression | In vivo (NPC) | 254,255 |
| Epigallocatechin gallate | Inhibits EBV lytic gene expression | In vitro (Burkitt’s lymphoma, NPC) | 258,264,265 |
| Curcumin | Inhibits BZLF1 gene transcription | In vitro (Burkitt’s lymphoma) | 256 |
| Sulforaphane | Reduces the transactivation activity of the BRLF1 gene | In vitro (NPC) | 257 |
| Protoapigenone | Reduces the transcriptional function of Zta | In vitro (Burkitt’s lymphoma) | 259 |
| Moronic acid | Suppresses the transactivation function of Rta | In vitro (Burkitt’s lymphoma) | 260 |
| Emodin | Inhibits EBV IE protein expression and DNA replication | In vitro (Burkitt’s lymphoma) | 261 |
| Glycyrrhizic acid | Inhibits EBV IE protein expression and DNA replication | In vitro (Burkitt’s lymphoma) | 262 |
| Vitamin C | Reduces EBV EA IgG and VCA IgM antibody levels | In vivo (CAEBV) | 263 |
Recently, studies have showed that Ephrin receptor A2 (EphA2) is critical for EBV entry into the epithelial cells. EphA2 interacted with EBV entry proteins gH/gL and gB to facilitate EBV internalization and fusion. And the interaction is mediated by the EphA2 extracellular domain (ECD).266,267 In addition, Peng et al.268 have identified that small-molecular inhibitor of EphA2, ALW (ALW-II-41–27), strongly inhibited the proliferation of gastric cancer in vitro and in gastric cancer patient–derived xenografts. These data indicated that specifically inhibition of EphA2 may be an effective strategy in gastric cancer therapy. Which also opens up potential possibilities for antiviral agents targeting of EphA2. The discovery of EphA2 as an EBV epithelial cell receptor has important implications for EBV pathogenesis and may uncover new potential targets that can be used for the development of novel intervention strategies.
Non-coding RNA (ncRNA) and EBV-associated tumors
The role of EBV-encoded ncRNA
EBV was the first human virus found to encode microRNAs (miRNAs) since 2004.19 The EBV latent non-coding RNAs include BamHI-A rightward transcript miRNAs (BART miRNAs), BHRF1 miRNAs and EBER1 and EBER2.4 EBV-encoded miRNAs are generated from two regions of the viral genome, within the apoptosis regulator BHRF1 gene locus and near the BamHI A region in a latency type-dependent manner.269 EBV encodes 44 mature BART miRNAs, a number of which have been proven to promote carcinogenesis by targeting host genes or self-viral genes. BART miRNAs, which are encoded by the BamHI region of EBV, are reported to be abundant in NPC and have potential value in early diagnosis of NPC. Accumulating evidence suggests that EBV-miR-BARTs play a critical role in host cell survival, immune escape, cell proliferation, apoptosis, and cancer metabolism, promoting the generation of NPC.270
To understand the roles of EBV miRNAs in the pathogenesis of NPC, Chen et al.271 utilized deep sequencing technology to characterize the EBV miRNA transcriptome in clinical NPC tissues. Sequence analysis revealed that most of the highly abundant EBV miRNAs shared their seed sequences (nucleotides 2–7) with human miRNAs, suggesting that seed sequence content might be an important factor underlying the differential accumulation of EBV miRNAs. These observations not only provided a potential linkage between EBV miRNAs and human malignancy, but also suggested a highly coordinated mechanism through which EBV miRNAs might mimic or compete with human miRNAs to affect cellular functions. More importantly, EBV-encoded miRNAs compromise the attraction of CD8+ T cytotoxic lymphocytes into the tumor microenvironment by down-regulating CXCL11 expression and also inhibit antigen presentation on MHC class I molecules to these CD8+ T cells, rendering the microenvironment of EBV-associated malignancies immune-suppressive.231
EBV-miR-BART1–5p
EBV-encoded miR-BART1-5p detection by nasopharyngeal brush sampling could act as an efficient and less invasive method assisting clinical diagnosis of NPC (with 93.5% sensitivity and 100% specificity), and the miR-BART1-5p relative expression level in nasopharyngeal brush samples was reflective of NPC progression.272 EBV-miR-BART1-5p directly targeted the α1 catalytic subunit of AMP-activated protein kinase (AMPKα1) and consequently mediated the AMPK/mTOR/HIF-1 pathway, which promoted NPC cell anomalous aerobic glycolysis and angiogenesis, ultimately leading to uncontrolled growth of NPC.273 Also, EBV-miR-BART1 directly targeted the cellular tumor suppressor PTEN. Reduction of PTEN dosage by EBV-miR-BART1 activated PTEN-dependent pathways, including PI3-K/Akt, FAK-p130 (Cas), and Shc-MAPK/ERK1/2 signaling, drove EMT, and consequently increased migration, invasion, and metastasis of NPC cells, highlighting the important role of PTEN in EBV-miR-BART-driven metastasis in NPC.274
EBV-miR-BART2-5p
Circulating EBV miRNA BART2-5p has been reported to act as a valuable biomarker for early detection and screening of NPC.275
EBV-miR-BART5-3p
The upregulation of EBV-miR-BART5-3p promoted the growth of NPC and gastric carcinoma cells. BART5-3p directly targeted the tumor suppressor gene, TP53, on its 3ʹ-untranslated region (3ʹ-UTR) and consequently downregulated CDKN1A, BAX, and FAS expression, leading to acceleration of the cell cycle progress and inhibition of apoptosis. BART5-3p also contributed to resistance to chemotherapeutic drugs and ionizing irradiation-induced p53 increases.276
EBV-miR-BART6-3p
EBV-encoded miR-BART6-3p inhibits EBV-associated cancer cell migration and invasion by targeting and down-regulating a novel lncRNA LOC553103, providing a potential novel diagnostic and treatment biomarker for NPC and other EBV-related cancers.277 Through proteomics analysis, stathmin (STMN1) was determined to be affected by EBV-miR-BART6-3p and LOC553103. LOC553103 directly binds to and stabilizes the 3ʹ-UTR region of STMN1 mRNA. These results indicated that the EBV-miR-BART6-3p/LOC553103/STMN1 axis regulated the expression of cell cycle-associated proteins, which then inhibited NPC cell proliferation.278
EBV-miR-BART7-3p
EBV-miR-BART7-3p also targeted the human tumor suppressor PTEN, modulating PI3-K/Akt/GSK-3β signaling and eventually leading to high expression and nuclear accumulation of Snail and β-catenin, which favor EMT.279 Cai et al.280 identified SMAD7 as a novel target gene of EBV-miR-BART7-3p in addition to the PTEN gene that was previously reported; and this viral miRNA suppressed SMAD7, leading to activation of TGF-β signaling and eventually enhanced the stemness of NPC cells. Also, the EBV-miR-BART7 increased responsiveness of NPC to radiation treatment by targeting GFPT1/TGF-β1 signaling. Thus, detection of EBV-miR-BART7 might be a useful indicator for monitoring NPC progression and predicting radio-therapeutic outcomes.281
EBV-miR-BART8-3p
EBV-miR-BART8-3p promotes NPC cell proliferation in response to irradiation in vitro and is associated with the induction of cell cycle arrest at the G2/M phase, which is a positive factor for the DNA repair after radiation treatment. Treatment with KU60019 or AZD6738 increases the radiosensitivity of NPC by suppressing the expression of p-ATM and p-ATR, indicating that EBV-miR-BART8-3p promoted radioresistance in NPC by modulating the activity of the ATM/ATR signaling pathway.282
EBV-miR-BART13-3p
Vesicle-bound EBV-miR-BART13-3p in circulation distinguished NPC from other head and neck cancers and asymptomatic EBV-infections. The high specificity of circulating EBV-miR-BART13-3p (97%) for NPC detection is in agreement with active secretion from NPC tumor cells. Thus, EV-bound EBV-miR-BART13-3p in circulation is a promising, NPC-selective, biomarker that should be considered as part of a screening strategy to identify NPC in endemic regions.283
EBV-miR-BHRF1-1
The EBV-encoded miR-BHRF1-1 is barely expressed in most NPC cells with EBV latent infection. miR-BHRF1-1 has been shown to be involved in TPA-induced accumulation of EBV lytic proteins and viral copies in late lytic cycle. The miR-BHRF1-1-potentiated induction of EBV lytic replication is accompanied by inhibition of p53 expression. These results suggested that the EBV original pathogen miR-BHRF1-1 is involved in the control of EBV late lytic replication by directly targeting the host p53 gene.284
EBV-BART-lncRNAs
BART lncRNAs localize within the nucleus of EBV-infected cells and knockdown of BART lncRNAs substantially affected the expression of genes associated with cell adhesion, oxidoreductase activity, inflammation, and immunity. BART lncRNA is associated with the CBP/p300 complex and RNA polymerase II (Pol II) in the nucleus, suggesting that BART lncRNAs might mediate epigenetic regulation of gene expression by interacting with the chromatin remodeling machinery. EBV BART lncRNA expression modulates host gene expression and maintains EBV latency by interfering with histone methylation and acetylation processes. Thus, aberrant expression of affected host genes mediated by BART lncRNA might lead to immune evasion, progression, and metastasis of NPC.285 Verhoeven et al.286 showed that NF-κB activates the BART promoters and regulates expression of EBV-BART miRNAs and lncRNAs in NPC. The aberrant NF-κB signaling and expression of BARTs form an autoregulatory loop for maintaining EBV latency in NPC cells. Further exploration of how targeting NF-κB signaling interrupts EBV latency in NPC cells might reveal new options for NPC treatment.
EBV-encoded RNAs (EBERs)
Similar to EBV-BART miRNAs and lncRNAs, EBERs are another type of non-coding RNAs expressed by EBV and are highly expressed in all EBV infection programs. Owing to their most abundant viral transcripts and their own high abundance, in situ hybridization against EBERs still constitutes the gold standard for detecting EBV-infected cells. In contrast to the miRNAs, EBERs are confined to the nucleus and seem to interact with various RNA binding proteins. Transgenic overexpression of EBERs could lead to lymphoproliferations and, less frequently, B-cell lymphomas.231 Epstein–Barr-encoded RNA EBER2 increases CXCL8 expression, and this chemokine enhances spontaneous lytic replication levels in M81-infected B cells. The unique properties of M81 EBER2 could be ascribed to its unusually high expression level and to the ability of its single-stranded region to activate TLR7. Thus, M81 induces chronic inflammation in its target cells and resulting in increased virus production.230
EBV and exosomal miRNA
Interestingly, Zuo et al.287 revealed that aspirin was a promising drug for NPC therapy through its targeting of exosomal LMP1 (exo-LMP1) transfer and the regulatory effect of LMP1 on miR-203 expression. EBV could regulate its own tumorigenesis through the LMP1/NF-κB/exo-LMP1 axis, opening a new avenue for understanding the pathogenesis of this tumor virus and providing a rationale for the use of exo-LMP1 or exosomal miR-203 (exo-miR-203) in EBV-targeted therapy by aspirin in invasive NPC.
The interactions between miRNA and lncRNA/circRNA
Long non-coding RNAs (lncRNAs) and circular RNAs (circRNAs) have been reported to act as important regulators and also to play crucial roles in NPC. LINC00460 was reported to be markedly increased in NPC tissues and cells compared to their corresponding controls. LINC00460 contributes to the progression of NPC by regulating the miR-149-5p/IL-6 signaling pathway, suggesting that LINC00460 could be regarded as a novel prognostic biomarker and therapeutic target in NPC diagnosis and treatment.288 CircRNA ZNF609 (circ-ANF609) and miR-188 have been, respectively, reported to play a pro-cancer and anticancer role in NPC. Circ-ZNF609 depletion represses proliferation and cell cycle transition, and induces apoptosis of NPC cells through modulation of the miR-188/ELF2 axis, providing potential targets for the therapy of NPC.289 Moreover, circCRIM1 is upregulated in highly metastatic NPC cells and tissues, and its overexpression promotes NPC cell metastasis and EMT. Mechanistically, circCRIM1 competitively binds to miR-422a and prevents the suppressive effects of miR-422a on its target gene FOXQ1, which finally leads to NPC metastasis, EMT, and docetaxel chemoresistance.290
The combination of miRNA and transcriptome sequencing
Szeto et al.291 characterized mRNA and miRNA transcriptomes by RNA sequencing (RNA-Seq) of NPC model systems. They found 2812 genes and 149 miRNAs (human and EBV) to be differentially expressed in NP460, HK1, C666, and X666 with RNASeq and 533 miRNA-mRNA target pairs were inversely regulated in the three NPC cell lines compared to NP460 cells. Integrated mRNA/miRNA expression profiling and pathway analysis showed that extracellular matrix organization, beta-1 integrin cell surface interactions, and the PI3K/AKT, EGFR, ErbB, and Wnt pathways are potentially deregulated in NPC. This comprehensive characterization of mRNA and miRNA transcriptomes in NPC cell lines and xenografts provided insights on miRNA regulation of mRNA and valuable resources on transcript variation and regulation in NPC, which are potentially useful for mechanistic and preclinical studies.
The link between miRNA/lncRNA and epigenetic regulation
Increasing evidence supports the role of members of the polycomb group (PcG) gene family in tumor development and progression. Using NPC as a disease model, a comprehensive analysis was undertaken to examine the clinical significance of EZH2 expression, identification of the cellular processes regulated by EZH2, and the mechanisms of its deregulated expression. The combination of global miRNA profiling in primary NPC specimens, and in silico analyses provided several candidate miRNAs that could regulate EZH2. miR-26a, miR-101, and miR-98, which were validated as bona fide regulators of EZH2 expression. In particular, miR-98 was under-expressed in samples from relapsed patient, strongly suggesting an important role for the miR-98 and EZH2 axis in NPC biology.292 miR-26a strongly reduced the expression of the EZH2 oncogene in NPC cells. Similar to restoring miR-26a expression, EZH2 downregulation inhibited cell growth and cell-cycle progression, whereas EZH2 overexpression rescued the suppressive effect of miR-26a. These results indicated that miR-26a functions as a growth-suppressive miRNA in NPC, and that its suppressive effects are mediated chiefly by repressing EZH2 expression.293
The clinical significance of ncRNA in EBV-associated tumors
miRNAs act as biomarkers
Circulating miRNAs have become reliable sources of noninvasive biomarkers for cancer diagnosis. miRNA expression analysis in circulating blood for the identification of novel signatures might assist in the early detection of NPC patients. Five miRNAs in serum, including let-7b-5p, miR-140-3p, miR-192-5p, miR-223-3p, and miR-24-3p were found to be significantly upregulated in NPC patients and are potential biomarkers for NPC detection.294 The dynamic changes in plasma miRNAs (miR-9-3p, miR-124-3p, miR-892b, and miR-3676-3p) after treatment reflect the outcome of the disease and have the potential to monitor recurrence and metastasis in patients with NPC.295 Additionally, the high expression levels of miR-548q and miR-483-5p in plasma, cell lines, and clinical tissues of NPC patients indicates their significant roles as potential biomarkers for NPC.296 Twenty-three plasma miRNAs with differential expression levels were selected for qPCR analysis on an independent set, including 100 NPC patients and 55 healthy controls. NPC patients with low concentrations of miR-483-5p and miR-103 had better prognosis for 5-year OS than those with high concentrations. In contrast, those with low concentrations of miR-29a and let-7c had poorer prognosis. Thus, differentially expressed plasma miRNAs as identified by next-generation sequencing could be helpful for predicting survival in NPC patients.297 Moreover, four-serum miRNAs (miR-22, miR-572, miR-638, and miR-1234) were found to be differentially altered and were used to construct a miRNA signature for NPC. This four-serum miRNA signature adds prognostic value to the TNM staging system and provides information for personalized therapy in NPC.298
Additionally, miR-451 is significantly downregulated in NPC cell lines and clinical tissues. Patients with low expression of miR-451 had poorer OS and disease-free survival than patients with high expression, and miR-451 is an independent prognostic factor in NPC as shown by multivariate Cox regression analysis.299 Three NPC subtypes (immunogenic, classical, and mesenchymal) have been identified as molecularly distinct and clinically relevant, of which the mesenchymal subtype (~ 36%) is associated with poor prognosis, characterized by suppressing tumor suppressor miRNAs and the activation of EMT. Out of the 25 most differentially expressed miRNAs in the mesenchymal subtype, miR-142, miR-26a, miR-141, and let-7i had significant prognostic power for prediction of distant metastasis.300
EBV-associated gastric carcinoma (EBVaGC) is the most common malignancy caused by EBV infection. EBVaGC has definite histological characteristics similar to gastric carcinoma with lymphoid stroma. Clinically, EBVaGC has a significantly low frequency of lymph node metastasis compared with EBV-negative gastric cancer, resulting in a better prognosis. EBVaGC harbors a DNA methylation phenotype, PD-L1 and PD-L2 overexpression, and frequent alterations in the PIK3CA gene.301 In a study aiming to determine the potential utility of circulating cell-free EBV DNA as a biomarker for the detection and/or monitoring of therapeutic response in patients with EBVaGC, Shoda et al.302 showed that plasma EBV ratios were significantly correlated with the size of EBVaGC tumors. Patients with EBVaGC might have a better prognosis, but circulating cell-free EBV DNA had no or little impact on prognosis. In addition, repeated assessment of the plasma EBV ratio in EBVaGC showed a decrease in plasma EBV DNA after treatment and increase during tumor progression/recurrence.
ncRNAs and therapy-resistance
For the roles of ncRNA in chemotherapy resistance, miR-374a was observed to reduce NPC cell proliferation, migration, invasion, metastasis, and chemotherapy drug cisplatin (DDP) resistance through the miR-374a-CCND1-pPI3-K/AKT-c-Jun feedback loop.303 MiR-296-3p is negatively regulated by nicotine and directly targets MK2-induced Ras/Braf/ERK/Mek/c-Myc or PI3K/AKT/c-Myc signaling to suppress NPC cell proliferation, metastasis and chemotherapy resistance to DDP treatment.304 Also, a tumor suppressor miRNA, miR-3188, could directly target the mTOR-p-PI3K/AKT-c-Jun pathway and participate in FOXO1-mediated repression of cell growth, tumorigenesis, and NPC chemotherapy resistance to 5-FU.305
For the roles of ncRNA in radiotherapy resistance, a sequence polymorphism rs4919510C > G in miR-608 was reported to act as a simple marker to predict locoregional recurrence (LRR) in patients with radiotherapy-treated NPC.306 In addition, miR-19b-3p contributes to the radio-resistance of NPC by activating the TNFAIP3/NF-κB axis, and miR-19b-3p is a determinant of NPC radio-response and might serve as a potential therapeutic target in NPC treatment.307 MiR-138-5p enhances radio-sensitivity of NPC cells by targeting EIF4EBP1.308 MiR-483-5p decreases the radio-sensitivity and radiation-induced apoptosis of NPC cells by targeting DAPK1.309 MiR-504 affects the radio-resistance of NPC by down-regulating the expression of NRF1 and disturbing mitochondrial respiratory function through TFAM and OXPHOS complexes I, III, and IV. The serum miR-504 expression levels of NPC patients are elevated during different weeks of radiotherapy and correlate with tumor, lymph nodes, and metastasis (TNM) stages and total tumor volume. This indicates that high serum miR-504 expression levels are associated with tumor radio-resistance and poor radio-therapeutic effects. Therefore, miR-504 might become a promising biomarker of NPC radio-resistance and targeting miR-504 might improve tumor radiation response.310 Additionally, LncRNA ANCR promotes NPC cell growth and radiation resistance by repressing the expression of PTEN. This regulation relies on ANCR-mediated EZH2 binding and epigenetic regulation on the PTEN promoter.311 CircRNA_000543 knockdown sensitized NPC cells to irradiation by targeting the miR-9/platelet-derived growth factor receptor B (PDGFRB) axis. Thus, circRNA_000543 might be a potential therapeutic target for radio-resistant NPC.312
Radiochemotherapy in EBV-associated tumors
Radiotherapy (RT) alone is a good option for NPC patients with stage I disease, which is based on the eighth edition of the American Joint Committee on Cancer (AJCC) staging system.313 According to the National Comprehensive Cancer Network guidelines, the efficacious choice for patients with locoregionally advanced NPC (Stage II–IVa) is RT combined with chemotherapy. Depending on the tolerance and heterogeneity of patients, three regimens are used to treat locoregionally advanced NPC, including concurrent chemotherapy, adjuvant chemotherapy, and induction chemotherapy. Adjuvant chemotherapy and induction chemotherapy play a pivotal role in the development of concurrent chemotherapy, bringing more benefits to patients’ OS and PFS.314
Molecular-targeted therapy combined with chemotherapy
Cisplatin is the paramount and classical first-line concurrent chemotherapy drug, compared with fluorouracil, oxaliplatin, or nedaplatin. Clinical treatment helped popularize cisplatin as a backbone drug for most solid tumor therapeutic schedules. It has been first approved by Food and Drug Administration (FDA) to treat testicular cancer and bladder cancer in 1978. However, chemotherapy drugs are limited by their harsh toxicity and development of drug resistance in the process of treatment.315 Cisplatin-based chemotherapy benefits only 10%-20% of patients with squamous cell carcinomas of the head and neck (HNSCC). NPC is a subtype of HNSCC that shares similar characteristics.11,316 Currently, with the rapid progress of molecular diagnostic therapies, the development of more targeted drugs for the treatment of NPC to improve the prognosis of patients has become a promising prospect.
Several clinical trials have shown good contributions to patients’ OS and PFS by molecular-targeted drugs combined with concomitant radiochemotherapy in locoregionally advanced NPC. Epidermal growth factor receptor (EGFR) was defined as a driving oncogene with high mutations rate in many tumors, but not in NPC. EGFR is overexpressed in almost 80% of NPCs and is enhanced by the EBV-encoded oncoprotein, LMP1. Our previous studies have shown that nuclear EGFR/signal transducer and activator of transcription 3 (STAT3) can directly bind to and activate the cyclin D1 promoter under the regulation of LMP1, favoring cell proliferation and cycle progression.317,318 Monoclonal antibodies against EGFR, such as cetuximab and nimotuzumab are dominant among molecular-targeted therapies. Three phase II clinical trials (NCT01271439, NCT00700440, NCT01712919) have aimed to evaluate the toxicity and efficacy of cetuximab combined with chemotherapy in locoregionally advanced NPC. A phase II study showed the value of an intravenous immunoglobulin G1 monoclonal antibody (cetuximab) in combination with carboplatin in patients with recurrent or metastatic NPC.319 Significantly, another phase II study of an orally active EGFR tyrosine kinase inhibitor (gefitinib) in combination with cisplatin offers promising efficacy as shown by evaluation of plasma Epstein–Barr virus DNA and a manageable safety profile for patients with metastatic or locoregionally recurrent NPC.320 All of these trials will contribute to obtain a thoroughly understanding of the role of EGFR combined with concurrent chemotherapy in NPC. Moreover, vascular endothelial growth factor (VEGF) is another molecular target that is more active in combination chemotherapy treatment. A completed Phase II clinical trial (NCT00408694) covered the use of a monoclonal antibody (bevacizumab) together with cisplatin and fluorouracil aimed at stopping the growth of NPC by blocking blood flow to the tumor by targeting VEGF.
Biomarkers of chemotherapy for EBV-associated tumors
Additionally, numerous other molecular-targeting methods are in clinical trials, including interruption of compound and gene modifications. A novel and potent imidazo[4,5-c] quinolone derivative (NVP-BEZ235), which has entered phase I/II clinical trials in patients with advanced solid tumors, has a potential role in cisplatin-sensitive cancers by dual targeting of the PI3-K (phosphatidylinositol 3-kinase) and mTOR (mammalian target of rapamycin) kinases for NPC treatment.321 The potential ability of NPC stem cells to evade the apoptotic pathways can make them resistant to chemotherapy due to their potential ability to evade cell death. One study showed that overexpression of the inhibitory apoptotic protein protein (IAP) family in NPC stem cells may play a role in maintaining NPC stem cell characteristics. Targeting cancer stem cells (CSCs) by a novel antagonist of IAPs, APG-1387, induced autophagic degradation of sex-determining region Y (SRY)-box 2 (Sox2), suggesting that APG-1387 combined with chemotherapy might be a novel therapeutic strategy for the treatment of NPC.322 The search for anticancer agents originating from natural compounds has been a booming area of research in recent years due to their perceived low toxicity. Asiatic acid (AA), extracted from Centella asiatica, was found to have anticancer activity against various cancers, and appears to overcome cisplatin resistance by triggering apoptosis through the induction of p38 phosphorylation.323
Interfering with drug-resistant molecules through genetic modification is a common therapeutic strategy. Tumor cells can evade the killing effects of chemotherapy through molecular mechanisms of cell cycle blockade and DNA damage repair. The new 6q27 tumor suppressor disheveled-associated binding antagonist of beta-catenin 2 (DACT2), which is frequently silenced by CpG methylation, sensitizes NPC cells to paclitaxel and 5-FU toxicity through β-catenin/ Cdc25c signaling and G2/M arrest.324 Aryl hydrocarbon receptor nuclear translocator-like (ARNTL) hypermethylation promotes tumorigenesis and inhibits cisplatin sensitivity by activating cyclin-dependent kinase 5 (CDK5) transcription in NPC.325 Nuclear factor with BRCT domain protein 1 (NFBD1) functions in cell cycle checkpoint activation and DNA repair following DNA damage, which accompanying by cisplatin or 5-fluorouraci resistance in NPC.326 Indeed, the epithelial–mesenchymal transition (EMT) is considered to be the main driver of chemotherapy resistance in NPC. For example, forkhead box C2 (FOXC2)-mediated EMT may be an important mechanism through which cancer cells acquire and maintain drug resistance.327 Another report revealed that matrix metalloproteinase 1 promotes tumorigenesis and inhibits NPC’s sensitivity to 5-fluorouracil.328 Our previous study showed that EBV-LMP1 activates PKC to promote the nuclear entry of annexin A2 and induce DNA synthesis and cell proliferation.329 Moreover, human NPC cells with high ANXA2 expression can potentially resist cisplatin or 5-fluorouracil, docetaxel, and vincristine by upregulating EMT-associated signaling proteins. Therefore, annexin A2 might be a biomarker to evaluate the resistance of NPC during chemotherapy treatment.330 Recently, a growing number of studies show that triggering an immune response in cancer patients can make chemotherapy work better. Bone marrow stromal cell antigen 2 (BST2) is well-known as a natural immune factor in viral infections and is highly expressed in a majority of tumors. Knockdown of BST2 with siRNA leads to cisplatin resistance by activating the NF-κB signaling pathway and enhancing the expression of anti-apoptotic genes, such as Bcl-XL and livin.331 The CD271-associated protein, brain-expressed X-linked 3 (BEX3), is accompanied with high octamer-binding transcription factor 4 (OCT4) expression in cisplatin-resistant NPC, suggesting that targeting BEX3 in combination with cisplatin could have a synergistic effect on NPC.332 In addition, some EBV-driven treatments are forecasted to be effective only when lytic viral replication occurs. Cyclophosphamide can induce the lytic phase of EBV infection and is quite effective in reducing the number of EBER-expressing tumor cells within 5 days.333 DNA viral reactivation due to histone deacetylase inhibitor-induced immunosuppression or chromatin remodeling. In a phase II clinical trial of the HDAC inhibitor, romidepsin, in patients with T-cell lymphoma, 2 of 120 treated patients developed secondary EBV-associated lymphoproliferations. This trial suggests that reactivation of latent EBV is well characterized as an outcome of the immune suppression associated with systemic chemotherapy.334
All of these studies were aimed at confirming molecules related to chemoresistance. Notably, elucidating the molecular mechanisms of chemoresistance would benefit in discovering a personalized and accurate therapeutic approach for EBV-associated tumors.
EBV DNA as a biomarker in chemotherapy
Emerging evidence supports a role for EBV in NPC treatment. Significantly, an unfavorable EBV DNA response after induction chemotherapy or at the midpoint of RT is also shown to be an adverse prognosticator for clinical outcome. The plasma EBV DNA load at induction chemotherapy (ICT) completion (postICT-DNA) is a powerful and early outcome predictor in locoregionally advanced NPC, allowing comparison of the prognostic value of postICT-DNA and post-chemoradiation therapy (CCRT) DNA (postRT-DNA), which could facilitate further risk stratification and early treatment modification.335 Detectable EBV DNA after NACT in advanced-stage NPC is correlated with poor clinical outcome.336 EBV DNA level and tumor response after NACT may complement each other for assessing outcome in NPC patients who have received both NACT and CCRT. Two clinical trials (NCT02135042, NCT03668730) showed that EBV DNA could be used to identify patients at high risk for treatment failure and to perform further risk stratification, early treatment modification, or both, before the implementation of CCRT.243,337 Plasma EBV DNA is also of predictive value for prognosis in metastatic/recurrent NPC patients undergoing palliative chemotherapy.338 Plasma clearance of cell-free Epstein–Barr virus (pEBV) DNA could predict survival and subsequent response in patients with advanced or recurrent NPC. This dual endpoint is an innovative tool for assessing early drug responses.339 Furthermore, the influence of cumulative doses of cisplatin on the clinical outcome in NPC patients who received intensity-modulated radiotherapy (IMRT) has been investigated. A study suggested that the optimal cumulative dose of cisplatin in low-risk group patients (EBV DNA <4000 copies/ml) was adjusted by assessing OS.340 After initial treatment of advanced stage (stage III–IV) extranodal NK/T-cell lymphoma with cisplatin, dexamethasone, gemcitabine, or pegaspargase (DDGP), OS was found to be superior in the EBV-DNA-negative group compared to the EBV-DNA-positive group.341
EBV-activated chemotherapy-resistant signaling
The DNA damage response (DDR) is a surveillance mechanism to monitor genome integrity and coordinate different aspects of cellular response, including cell cycle progression, gene transcription, and DNA repair. DDR is dysregulated by EBV infection with EBV-encoded viral proteins.99 Studies have shown that EBV infection activates DNA damage checkpoints by promoting the phosphorylation of the ATM and CHK2 pathway. Phosphorylation of the ATR/checkpoint kinase 1 (CHK1) pathway proteins and gradually activated along with the duration of the EBV exposure in NPC cell lines. EBV activation of the ATR-mediated DNA damage response results in chemotherapy resistance to CDDP and 5-FU in NPC. Accordingly, ATR knockdown may serve as an effective treatment strategy for chemotherapy-resistant, EBV-positive NPC.342 Upregulation of NOTCH ligands (JAG1 or DLL4) and effector (HEY1) in the majority of EBV-positive tumor lines and primary tumors has been observed. NOTCH3 knockdown highly enhances the sensitivity of NPC cells to cisplatin treatment. Targeting NOTCH3 signaling may serve as a potential therapeutic approach for treating patients suffering from EBV-associated NPC.343
Recently, we revealed that the IC50 of cisplatin in EBV-LMP1-positive cells was significantly higher than that in EBV-LMP1-negative cells. The mechanisms behind this chemoresistant phenotype are closely related to the numerous EBV-mediated oncogenic signaling pathways, providing new approaches for preventing and reversing drug resistance in NPC. For example, EBV-LMP1 remodels the mitochondria by regulating the AMPK/Drp1 and cyclin B1/Cdk1/Drp1 axes,101 or decreases TRAIL-induced apoptosis by activating the PI3-K/AKT/FOXO3a signaling pathway.344 In addition, EBV-LMP1 modulates MAPK-mediated Op18/stathmin signaling to induce cell cycle progression and promote tumorigenesis.345 SiRNA-mediated silencing of stathmin may enhance the efficacy of paclitaxel in the treatment of NPC.346
EBV-miR-BART22 not only promoted tumor stemness and metastasis, but also enhanced the resistance to cisplatin (DDP) in vitro and in vivo. Cinobufotalin markedly reversed EBV-miR-BART22-induced cisplatin resistance by stimulating MAP2K4 to antagonize the non-muscle myosin heavy chain IIA/ glycogen synthase 3β/β-catenin signaling pathway.347
In summary, molecular-targeted therapy improves chemosensitivity and has the potential to interfere with tumor metastasis. EBV-mediated signaling pathways are important mechanisms of the phenotypic behavior of treatment resistance in EBV-related tumors. EBV DNA has been demonstrated to play a role as a prognosis biomarker to evaluate the efficacy of treatment in chemotherapy-based patients.
Radiation therapy in EBV-associated tumors
So far, the widespread application of intensity-modulated radiotherapy and optimization of chemotherapy strategies (induction, concurrent and adjuvant) are the main strategy in NPC clinical therapy and have contributed to improved survival with reduced toxicities.11 However, radiation resistance has been a major hurdle that limits therapeutic efficacy and results in recurrence.11,208
ROS-mediated oxidative stress in radiation therapy
So far, several specific signaling pathways contribute to cells gaining resistance against ionizing radiation. Among these signaling pathways, ROS play a significant but paradoxical role acting as a “double-edged sword” to regulate cellular response to radiation. On the one hand, ROS is a critical mediator of IR-induced cell killing. When ROS levels rise beyond a tolerable limit inside tumor cells, apoptosis is initiated and ROS-induced cell death may be either direct or indirect.348 High levels of ROS cause a cluster of lesions to form within DNA that are difficult to repair and lead directly to cell death. Indirect cell death involves altered cellular homeostasis and modified signaling pathways ultimately leading to apoptosis.349 On the other hand, ROS is a critical regulator of IR-resistant pathways.223 The tumor cell stemness, antioxidant enzymes expression and inflammation are the reason that related to the ROS-mediated IR-resistant.236 Presently, some biomarkers have been identified that enhance the radiosensitivity of NPC, and include COX-2, EGFR, Bcl-2, and VEGF. These markers were associated with cell cycle, apoptosis, angiogenesis, and DNA repair.350
LMP1 contributes to radioresistance
EBV-latent membrane protein 1 (LMP1), a driving oncogene in NPC, expresses in almost all primary NPC tissues.227 Our group have revealed that EBV (LMP1)-positive cells show a higher survival fraction were resistant to radiation therapy. The mechanism was that LMP1 appears to activate several oncogenic signaling axes, including LMP1/JNKs/c-Jun/HIF-1/VEGF,351 LMP1/NF-κB/ATM,352 LMP1/Akt/hTERTs,353 LMP1/PI3K/Akt/GSK3β/c-Myc/HK2,98 and LMP1/DNA-PK/AMPK/DDR,99 which mediate therapeutic resistance. Recently, we found that reactivation of EBV by LMP1-induced high oxidative stress could lead to radioresistance of NPC cells both in vitro and in vivo, suggesting that EBV reactivation plays an important role in NPC resistance (unpublished data). As LMP1 is an attractive and promising target for EBV-positive NPC, our group successfully developed a DNAzyme (DZ1) that was engineered to specifically target the LMP1 mRNA.255 DZ1 treatment can reverse malignant phenotypes caused by LMP1 and increase the radiosensitivity of NPC, indicating the potential for DZ1 therapeutic approaches for the treatment of EBV-related cancers.99,255,354,355
Other targets contribute to radioresistance
Furthermore, there are several other signaling pathways that are related to radioresistance. For example, the forkhead box O (FOXO) family of proteins is an important transcriptional regulator of pivotal proteins associated with many diverse cellular functions.356 Among them, FOXO3a has been extensively studied because of its unique role in the regulation of cell proliferation, apoptosis, metabolism, stress management, and longevity.357 In NPC, silencing FOXO3a promotes tumor radioresistance of NPC in vitro and in vivo through inducing EMT and activating Wnt/β-catenin signal pathway. It is supposed that FOXO3a can be a novel and reliable NPC marker and a potential therapeutic target against NPC.358 Guo et al.359 reported genetic variations in the PI3K/PTEN/AKT/mTOR pathway are associated with distant metastasis in nasopharyngeal carcinoma patients treated with intensity-modulated radiation therapy. Zhang et al.360 reported PTEN can regulate the radiation-resistant cancer stem-like cell properties through activity of nuclear β-catenin in nasopharyngeal carcinoma. In addition, inhibition of the Numb/Notch signaling pathway increases radiation sensitivity in human nasopharyngeal carcinoma cells.361 In this part, we primarily discuss the regulation of oxidative stress by EBV, the role of EBV lytic reactivation in EBV-associated diseases and the following radiation therapy. A better understanding of the relationship between them may lead to develop novel prevention and therapeutic strategies for EBV-associated diseases and facilitate the development of novel strategies against radiation-resistant tumors.
Immunotherapy for EBV-associated diseases
The expression of the EBV antigen and the dense interstitial infiltration of immune cells provide a unique and attractive target for immunotherapy in EBV-related tumors.7,11,362–364 The immunotherapy of EBV-associated malignancies primarily includes EBV-targeted vaccination and adoptive T-cell therapy, as well as immune checkpoint blockade, some of which show good antitumor activity in early clinical studies.
EBV-related vaccines
Two main types of EBV-related vaccines exist, including one that is preventive and the other that is therapeutic targeting EBV-related life cycle antigens. Gp350 belongs to the product of the virus lytic stage and is relatively easier to produce neutralizing antibodies, which makes it a candidate antigen for prophylactic vaccines.365 Most studies show that the gp350 prophylactic vaccine does not achieve satisfactory clinical results, which may be closely related to the number of clinical samples, research time, appropriate adjuvants, and vaccination time.366,367 No experimental data are yet available supporting the idea that the gp350 vaccine can prevent the occurrence of NPC, but some studies have confirmed that high levels of neutralizing antibodies are associated with a lower risk of NPC.368
The EBV latent proteins, EBNA1, LMP1, and LMP2, are continuously expressed in EBV-related tumor cells. These proteins are key factors in promoting the transformation of normal cells into tumor cells. Thus, therapeutic vaccines against these proteins has been widely studied in clinical trials and some results have been achieved369 (Table 8). EBNA1 and LMP2 generate the most robust T-cell immunity. Interestingly, the oncogenic protein, LMP1, is a very weak immunogen, generating very low levels of CD8+ T-cell immunity as both a standalone vaccine and as a part of a trivalent vaccine cocktail.370 LMP2 has a large number of CD8+ T-cell epitopes and a limited number of CD4+ T-cell epitopes, and vaccines against LMP2 have also been actively developed. The current clinical research on therapeutic vaccines for EBV- associated tumors focuses on NPC. Results from an experiment of transferring LMP2 antigen (rAd5-EBV-LMP2) by using recombinant adenovirus vector, phase I clinical trials of 3 different doses in patients with advanced NPC (D0, D7, D14, and D28), showed that the proportion of LMP2-specific CD3+ and CD4+ cells in peripheral blood of immunized patients increased in a dose-dependent manner within 28 days.371 In a phase II clinical trial evaluating the ability of dendritic cell (DC) vaccines to target LMP1 and LMP2 antigens in patients with advanced metastatic NPC, patients were vaccinated using adenovirus-transduced autologous DCs encoding truncated LMP1 (ΔLMP1) and full-length LMP2 (Ad-ΔLMP1-LMP2).372 Three patients had clinical responses showing specific T-cell activation, one partial response (7.5 months) and two stable disease (6.5 and 7.5 months), which means that Ad-ΔLMP1-LMP2 transduction of DC can be successfully generated and safely administered to patients with advanced NPC.372 Owing to limited efficacy, future studies should focus on administering DC vaccines with greater efficacy to subjects with less tumor burden. The carboxyl terminal and full-length LMP2 and EBNA1 were transmitted by a MVA vector as a vaccine to obtain fusion proteins (MVA-EL). The vaccine effectively expanded EBNA1 and LMP2-specific CD4+ and CD8+ cells from peripheral blood lymphocytes of healthy serum EBV-positive individuals in vitro.373 A phase I clinical trial of MVA-EL vaccine was conducted in Hong Kong, comprising NPC patients who were in remission at least 12 weeks after initial treatment and received three intradermal MVA-EL immunizations. The results showed that the vaccine was well tolerated and had no dose-dependent toxicity, and the T-cell response to one or two vaccine antigens tripled in 15 of the 18 patients who received treatment.374 Phase I clinical trials of the MVA-EL vaccine comprising 16 NPC patients in the UK also showed that 8 of 14 patients who received treatment had increased CD4+ and CD8+ T-cell responses to one or both antigens, and immunophenotypic analysis confirmed that vaccination induced differentiation and functional diversity of EBNA1- and LMP2-specific CD4+ and CD8+.375 Owing to the satisfactory results of two phase I trials, the Cancer Research UK will further evaluate adult patients with NPC (NCT01800071).
Table 8.
Clinical trials of EBV-related therapeutic vaccines
| Ref. | Conditions | Interventions | Enrollment | Clinical outcome |
|---|---|---|---|---|
| NCT01800071 | EBV-positive NPC | MVA-EBNA1/LMP2 vaccine | 22 | Immune memory and recall response to MVA-EBNA1/LMP2 vaccination, Measurement of EBV genome levels |
| NCT01094405 | EBV-positive NPC | Recombinant Epstein–Barr virus (EBV) vaccine | 25 | Objective Response Rate (ORR), Duration of Response (DR), Progression-free survival (PFS), Overall survival (OS) |
| NCT00078494 | Recurrence NPC | EBV-LMP-2 peptide | 99 | Evaluate how the vaccines affect immune system cells |
| NCT00478062 | After Initial Therapy of Hodgkin’s Lymphoma | Hodgkin’s antigens-GM-CSF-expressing cell vaccine | 35 | Determine immunologic responses |
| NCT01147991 | EBV-positive cancers | EBNA1 C-terminal/LMP2 chimeric protein | 16 | To determine safety and immunologic responses |
| Hui et al.374 NCT01256853 | EBV-positive NPC | MVA-EL, which encodes an EBNA1/LMP2 fusion protein | 18 | Toxicity of MVA-EBNA1/LMP2 vaccine, T-cell responses, and assess the changes of EBV genome |
| Si et al.371 | EBV-positive NPC | Recombinant adenoviral vaccine expressing EBV-LMP2 protein (rAd5-EBV-LMP2) | 24 | Proportion of CD3+ CD4+ cells in peripheral blood |
| Chia et al.372 | EBV-positive NPC | DCs transduced with adenovirus encoding a truncated LMP1 (DeltaLMP1) and full-length LMP2 (Ad-DeltaLMP1-LMP2) | 16 | Detected in tLMP1/2-specific T cell |
| Lin et al.372 | EBV-positive NPC | Autologous DCs pulsed with HLA-restricted epitope peptides from LMP2 | 16 | CD8+ T-cell responses |
| Li et al.466 | EBV-positive NPC | Autologous DCs pulsed with HLA-restricted epitope peptides from LMP2 | 16 | Patients responded to LMP2A peptides Serum EBV-DNA level |
The main goal of vaccine development is to prevent primary infection, the establishment of a latent period, and subsequently the development of EBV-associated tumors. To date, developing a vaccine against this virus has been difficult. Most vaccines are primarily designed to induce a protective antibody response, which is considered too risky even when used in an attenuated form due to the inherent carcinogenicity of EBV, which is a critical problem in developing a vaccine against the virus.376 Therefore, new therapeutic vaccines should be either recombinant viral vectors capable of inducing EBV-specific cellular immune responses or novel recombinant viral glycoprotein preparations capable of eliciting more efficient EBV-specific antibody responses.377–379
Adoptive immunotherapy in EBV-related cancers
EBV expresses potential tumor-associated virus antigens, making it a candidate focus for successive cell-based immunotherapy (CBI). Various preclinical and clinical studies have explored the application of cytotoxic T cells (CTLs), tumor-infiltrating lymphocytes (TIL), natural killer (NK) cells, and dendritic cells (DC) in the treatment of refractory and locally advanced NPC. Adoptive immunotherapy of EBV-associated cancers mainly works by bypassing antigen presentation steps, including direct activation of effector cells, especially CTLs and NK cells (Table 9).
Table 9.
Cellular-based immunotherapy clinical trials in EBV-related cancers
| NCT number | Study | Interventions | Phases | Enrollment |
|---|---|---|---|---|
| CTL trails | ||||
| NCT00516087 | LMP1- and LMP2-specific CTLs to patients with EBV-positive NPC (NATELLA) | Genetically modified CTLs | I | 23 |
| Louis et al.381 NCT00078546 | EBV-specific CTLs following CD45 antibody to patients with Epstein–Barr virus (EBV) + NPC | EBV-specific CTL Infusion and Anti CD45 monoclonal antibody | I | 12 |
| NCT00431210 | Epstein–Barr virus-specific immunotherapy for NPC | EBV-specific adoptive T-cells | I | 28 |
| NCT02578641 | Evaluating chemotherapy and immunotherapy for advanced NPC | Autologous EBV-specific cytotoxic T cells | III | 330 |
| NCT00834093 | Study of Epstein–Barr virus-specific immunotherapy for NPC | EBV-Specific Immunotherapy | II | 20 |
| NCT02065362 | TGF-β-resistant cytotoxic T-lymphocytes in treatment of EBV-positive NPC/RESIST-NPC | NPC-specific T cells | I | 14 |
| NCT01195480 | CD19-CAR immunotherapy for childhood acute lymphoblastic leukemia | EBV-specific CTLs | I/II | 29 |
| NCT00058604 | Prevention and treatment of Epstein–Barr virus (EBV) lymphoma following a solid organ transplant using EBV-specific cytotoxic T lymphocytes (CTLs) | EBV-specific CTLs | I | 12 |
| NCT00058617 | Epstein–Barr virus (EBV)-specific cytotoxic T-Cells, relapsed lymphoma, ANGEL | EBV-specific CTLs | I | 13 |
| NCT01447056 | Most closely HLA-matched CTLs for relapsed Epstein– Barr virus (EBV)-associated diseases (MALTED) | LMP-specific T cells | I | 37 |
| NCT01498484 | Therapeutic effects of Epstein–Barr virus immune T-lymphocytes derived from a normal HLA-compatible or partially matched third-party donor in the treatment of EBV lymphoproliferative disorders and EBV-associated malignancies | EBV-specific T cells | II | 87 |
| NCT01555892 | Cytotoxic T-lymphocytes for EBV-positive lymphoma, GRALE | EBV-specific T cells | I | 136 |
| NCT03988582 | Safety and effectiveness of EBV-specific cytotoxic T cells for the treatment for EBV lymphomas or other EBV-associated malignancies | EBV-specific T cells | II | 79 |
| Bollard et al.385 NCT00062868 | LMP-specific T-cells for patients with relapsed EBV-positive lymphoma (ALCI) | LMP1/2 CTLs | I | 74 |
| NK-cell trails | ||||
| NCT00717184 | A Pilot study of autologous ex vivo activated NK-cell infusion in the treatment of metastatic NPC | Interleukin, NK cells | I | Unknown |
| NCT02507154 | Reactivating NK cells in treating refractory head and neck cancer | Cetuximab + NK cells | I/II | 31 |
Adoptive T-cell therapy
Adoptive T-cell therapy against EBV antigens includes stimulating autologous CD8+ T cells in vitro and then infusing them intravenously into the patients. This treatment strategy has several advantages. First, T cells have a high affinity for binding to EBV antigens and can be expanded in vitro, which can easily reach the number needed for clinical treatment.380 In addition, patients can receive other treatments before transfusion of T cells to remove tumor-related suppressor immune cells, which is conducive to the growth and expansion of injected lymphocytes in vivo. Some clinical experiments have shown that adoptive T-cell therapy targeting EBV antigens has an exciting effect. Eight patients with refractory NPC were injected with increasing doses of a CD45 mAb, before receiving EBV-CTL. Results showed that after injection of the CD45 mAb, EBV peptide-specific T cells increased 2.8-fold within 8 weeks. In this study, the clinical benefit rate was 37.5%.381 Second, Secondino et al.382 conducted a similar study on 11 patients with refractory NPC treated with cyclophosphamide and fludarabine. Based on the standard of tumor regression or stability of more than 4 months, the disease control rate was 54.5%. Chia et al.383 further evaluated the effect of EBV-CTL infusion into patients with locally advanced NPC and treated with the standard first-line chemotherapy of gemcitabine and384 carboplatin. Most of the patients showed positive reactions, such as decreased tumor growth rate, reduced tumor size, and an extended period of disease stabilization. This study showed that in the treatment of patients with metastatic and/or locally recurrent NPC, the median OS time was 29.9 months, and the 2-year OS rate was 62.9%.383 Importantly, the observed clinical benefits were directly related to the infusion of LMP2-specific T cells in CTL, as evidenced by a significant improvement in OS compared with patients who received CTL that lacked LMP2 specificity. In response to these encouraging results, the third phase of the EBV-CTL study is currently under way (NCT02578641). Similarly, Bollard et al.385 studied 28 patients with high-risk or multiple recurrent EBV-positive lymphomas treated with LMP-CTLs adjuvant with a median remission period of 3.1 years after CTL infusion. Of the 21 patients with recurrent or refractory lymphomas, 13 had clinical responses and 11 had complete responses. LMP-specific T cells and non-viral tumor-associated antigens were detected in peripheral blood within 2 months of CTL infusion.
NK cells therapy
NK cells have the inherent ability to distinguish between viral infection and malignant transformed cells without prior antigen sensitization. Some ongoing clinical trials have also attempted to use NK cells as an alternative to adoptive cell therapy for NPC.386 Compared with CTL, NK cells have a much shorter lifespan and do not need suicide vectors to overcome the overexpansion of metastatic cells.387 However, NK cells account for only a small portion of the human lymphocyte population (5–20%), and their distorted phenotypic and functional impairment during cancer progression challenges the activation and expansion of clinical-grade purified NK cells in vitro to reach the optimal number of clinical transfusions.388 Therefore, several methods have been developed and optimized, including the addition of cytokines for treatment and long-term cultures,389 and the use of peripheral blood mononuclear cells, K562 cells, and lymphoblastoid cell lines (LCL) transformed by EBV as feeder cells.390 In addition, tumor-directed NK-cell immunotherapy can be accomplished by genetic engineering of NK cells expressing tumor antigen receptors, or by combining with monoclonal antibodies that give priority to killing tumors through extracellular Fc receptor-mediated antibody-dependent cytotoxic (ADCC).391 At present, phase I/II clinical trials of highly active NK cells and expanded NK cells combined with the EGFR monoclonal antibody, cetuximab, in the treatment of refractory cancers are under way (NCT02507154).
Immune checkpoint inhibitor therapy in EBV-associated cancers
Overexpression of programmed death ligand 1/2 (PD-L1/2) is a common event in EBV-associated tumors, suggesting the importance of immune evasion in tumorigenesis, which establishes the theoretical basis for the use of immune checkpoint inhibitors in EBV-positive tumors.29 Preliminary clinical data suggest that PD-1 inhibitors such as nivolumab, pembrolizumab, and avelumab are a promising therapeutic strategy for the treatment of certain EBV-associated tumors, particularly in recurrent or metastatic (R/M) EBV-positive tumors in combination with other treatment methods (e.g., radiotherapy, chemotherapy, or molecular-targeted drugs) showing promising clinical results392,393 (Table 10).
Table 10.
Clinical trials on the use of PD-1 inhibitors for recurrent/metastatic EBV-related tumors
| Trial | Phase | Setting | Enrollment | Agent | Combination therapy | Efficacy |
|---|---|---|---|---|---|---|
| Ma399 NCT02339558 | II | R/M NPC | 40 | Nivolumab | NO | Completed |
| NCT02054806 | Ib | R/M NPC | 44 | Pembrolizumab | NO | Completed |
| NCT02875613 | II | R/M NPC | 39 | Avelumab | NO | Ongoing |
| NCT03707509 | III | R/M NPC | 250 | Camrelizumab | Gemcitabine, Cisplatin | Ongoing |
| NCT02605967 | II | R/M NPC | 114 | PDR001 | Gemcitabine,capecitabine ordocetaxel | Ongoing |
| NCT03813394 | I/II | R/M NPC | 48 | pembrolizumab | bevacizumab | Ongoing |
| NCT02611960 | III | R/M NPC | 124 | Pembrolizumab | Gemcitabine, capecitabine | Ongoing |
| NCT03854838 | II | R NPC | 25 | Tolipalimab | IMRT | Ongoing |
| NCT03907826 | III | R NPC | 212 | Toripalimab | IMRT | Ongoing |
| NCT03930498 | II | R NPC | 43 | Toripalimab | Cisplatin and Gemcitabine IMRT | Ongoing |
| NCT03755440 | II | M GC | 20 | SHR-1210 | NO | Ongoing |
| NCT03015896 | I/II | R NHL/HL | 102 | Nivolumab | Lenalidomide | Ongoing |
| NCT02973113 | I | R/R HL | 8 | Nivolumab | EBVST Cells | Ongoing |
| NCT02950220 | I | R/R NHL | – | Pembrolizumab | Ibrutinib | Completed |
| Fang403 NCT03121716 | I | R/M NPC | 20 | SHR-1210 | Gemcitabine Cis-platinum | Completed |
R/M recurrent/metastatic, R recurrent, M metastatic, NPC nasopharyngeal carcinoma, GC gastric cancer, R/R relapsed/refractory, NHL non-Hodgkin lymphoma, HL Hodgkin lymphoma
NPC is characterized by high expression of PD-L1 (up to 90% of tumor cells) and a large amount of non-malignant lymphocyte infiltration (about 50% of the sample interstitial TIL > 70% or intratumoral TIL > 10%), making NPC patients suitable for PD-1/PD-L1 immune checkpoint blockade therapy.394–396 In a phase Ib clinical study, the objective response rate (ORR) for pembrolizumab in patients was 18%, the median PFS was 2 months, and the median OS was 13 months.397 For Hodgkin’s lymphoma, immune checkpoint blockade of PD-1 has also shown promising results (NCT01592370).392 Gastric cancer is currently classified into four subcategories, including EBV-positive, microsatellite instability (MSI), genomic stability, and chromosomal instability.29 In EBVaGC, the virus is cleverly positioned in the infected cells and microenvironment to counteract host immune cells through exosomes whereas cancer cells express PD-L1 and recruit PD-L1-positive immune cells to evade the host immune system.364 In a phase II clinical trial of pembrolizumab in metastatic gastric cancer, high MSI and the overall effectiveness rate of EBVaGC patients was 85.7 and 100%, respectively. The presence of EBV was also shown to be a biomarker for pembrolizumab treatment.398
Nivolumab was the first PD-1 monoclonal antibody approved for clinical use. The total ORR of 44 patients was 20.5%. The 1-year OS rate was 59%, the 1-year PFS rate was 19.3%, and the patients with high PD-L1 had better response to nivolumab, indicating that nivolumab has a good application prospect in NPC.399 PD-1 blockade also in relapsed/refractory B-cell non-Hodgkin’s lymphoma in a phase I trial of nivolumab showed promising results, including follicular lymphomas (which often show abundant PD-1 expression on intratumoral CD4+ T cells) and diffuse large B-cell lymphoma (variably expressing PD-1 and PD-L1).392,400 In a phase I trial of 27 patients with unresectable or metastatic NPC, these patients failed the previous standard treatment and produced an objective 26% response to pembrolizumab treatment, with a 1-year overall survival rate of 63%. In addition, the efficacy and safety of anti-PD-1 immune checkpoint inhibitors have been demonstrated in numerous clinical trials in recurrent or metastatic EBV-positive tumors. The unusually high response rate in phase I camrelizumab chemotherapy studies suggests that camrelizumab may have shed light on this potentially promising direction,401 and the combination of PD-1 inhibitors with other chemotherapeutic agents has also been shown to greatly improve overall survival.402 Fang et al403 reported that 23 patients with metastatic or recurrent NPC treated with a combination of camrelizumab and gemcitabine/cisplatin, the ORR was 91%, and the 1-year PFS was 61%. Camrelizumab combined with chemotherapy shows good tolerance and antitumor activity, and it is a potential treatment choice for patients with recurrent or metastatic NPC. Therefore, Sun Yat-sen University launched a Phase III clinical trial of camrelizumab combined with gemcitabine and cisplatin (NCT03707509). These results will demonstrate that a PD-1 monoclonal antibody is the first choice to prevent the failure of chemotherapy. Whether the PD-1 monoclonal antibody is better to use alone or in combination as a first-line drug treatment needs to be further explored.
EBV-related autoimmune diseases (SADs) and immunotherapy
SADs are a group of connective tissue diseases. EBV infection has an etiopathogenetic role in SADs, such as SLE, RA, and SS.61,404,405 RA patients have an increased risk of EBV-associated lymphoproliferative disease (LPD), and SS also increases the risk of developing EBV-associated malignancies.404 Patients with SADs exhibited EBV-specific T-cell defects, increased viral load, as well as high levels of EBV antibodies.406–408 The relationship between EBV and SADs is complex and involves different mechanisms. The proteins encoded by EBV are involved in immune evasion and suppression of apoptosis in transformed infected lymphocytes, leading to loss of immune tolerance and autoimmunity.409–411 In addition, molecular mimicry between EBV proteins and autoantigens provides another possible model for disease induction and immune escape.405 RA susceptibility is carried by the HLA-DRB1* allele, whose third highly variable region contains the QK/RRAA or RRRAA base sequence, which is known as a shared epitope.412 The QKRAA amino acid sequence of HLA-DRB1*04:01 can also be found in the EBV glycoprotein gp110, the molecular mimicry between these EBV epitopes and autoantigens could lead to tolerance and autoimmune system collapse in RA patients.413 Sequence similarity between EBNA1 and ANO2 that overlaps with the minimal epitope defined by ANO2 and is known to correlate with MS risk.405 This evidence suggested that viral infection may initiate an autoimmune response through molecular mimicry,414 thus providing some degree of mechanistic linkage between EBV and SADs.
A marked reduction in interferon gamma-producing CD8+ cytotoxic T cells is commonly observed in patients with SADs, which may be the result of defective or reduced EBV-specific cytotoxic T cells, resulting in poor control of EBV infection.406,415,416 Thus, cellular immunotherapy is currently one of the most promising treatment options for EBV-related autoimmune diseases.61 A phase I clinical trial of specific T cells targeting EBNA1, LMP1, and LMP2A was conducted in five patients with secondary progressive MS and five patients with primary progressive MS.417 Further clinical trials will help determine the efficacy of EBV-specific T cells in the treatment of SADs.
EBV and immune escape
Avoiding immune destruction is an important cancer hallmarker. EBV has developed powerful strategies to evade host immune surveillance, with varying mechanisms of immune escape in EBV-driven malignancies. EBV-associated tumors are often rich in lymphoid-like mesenchyme, and a recent study found EBV-positive was detected in 74.5% of hepatocellular carcinomas with immune cell stroma (HCC-IS).418–420 This subtype of hepatocellular carcinoma has high-density EB-associated TIL infiltration and poorer RFS and OS characteristics, which may be related to the upregulation of CD28 co-stimulatory signals, T-cell receptors and other signaling pathways in HCC-IS, leading to CD8+ T-cell depletion and thus induced immune escape420 in gastric cancer are silenced up to 75% EBVaGC exhibits a high frequency of DNA methylation, thus serving as a strategy for the virus to evade host immune regulation.421–423 Immune-related genes (IRGS) in EBVaGC are silenced up to 75% by methylation, such as metallothionein-1 (MT1) and homologous box A (HOXA) clusters, thereby promoting EBV cleavage replication and subviral particle release, as well as KSHV cleavage reactivation.421 Collectively,these results demonstrated that epigenetic silencing of IRGs is a viral strategy to escape immune surveillance and promote viral propagation, which is overall beneficial to viral oncogenesis of human gamma-herpesviruses, considering that these IRGs possess antiviral activities against these oncoviruses.421
Reports supporting the important role of the tumor microenvironment (TME) in the development of nasopharyngeal carcinoma, which is composed of cellular components and non-cellular ECM (extracellular matrix). A key feature of NPC-TME is the presence of a large number of tumor-infiltrating lymphocytes (TILs) in the NPC tumor stroma, and elevated levels of IL-6, IL-8, IL-10, and interferon-γ and decreased levels of IL-2 were commonly detected in biopsied tissues, which may be related to the recruitment and activity of these infiltrating leukocytes.424–427 EBV-encoded Zta can bind to the IL-8 promoter and induce expression of IL-8 by nasopharyngeal carcinoma cells.428 IL-8 is known to be highly chemotactic for neutrophils, which are thought to be the first immune cells to be activated and recruited to inflammatory tissues.429,430 Zta has also been shown to manipulate neighboring monocytes to release IL-10 into the TME.431 Zta upregulates two immunomodulators in nasopharyngeal cancer cells: granulocyte macrophage colony-stimulating factor (GM-CSF) and prostaglandin E2 (PGE2), both of which are released into the TME and have synergistic effects in promoting IL-10 production by monocytes.431 IL-10 has long been recognized as a potent immunosuppressive cytokine that effectively inhibits the cytotoxic function of activated CD8+ cells and promotes the survival of EBV-infected NPC cells.432 Also EBV latent membrane protein 1 (LMP1) induces tyrosine sulfation of CXCR4 via tyrosine protein sulfotransferase-1 (TPST-1), the enzyme responsible for catalyzing tyrosine sulfation in vivo, which may be related to the high metastatic properties of NPC.433,434
EBV cleavage replication is involved in the early stages of B-cell lymphangiogenesis by promoting the release of soluble factors from the microenvironment, directly promoting B-cell growth, suppressing the antitumor immune response, and stimulating neovascularization. LMP-1 is a major driver of increased expression and local release of cytokines and chemokines that may be functionally relevant to B-cell growth and survival, such as IL-6 and IL-10.435 LMP-2A may also induce B cells to produce more IL-10 through activation of PI3-kinase,436 and EBV may also interfere with the host cytokine environment by producing viral IL-10 homologs that activate autocrine signaling pathways involving JAK/STAT.437 Alternatively, EBNA1 may favor the formation of an immunosuppressive microenvironment by upregulating CCL20 to attract regulatory T (Treg) cells to the infected tissues.438
Evidence accumulated so far clearly indicates that EBV choreographs complex mechanisms that favor immune escape of tumor cells while creating an environment conducive to tumor cell growth and survival. The elucidation of the complex mechanisms between EBV infection and different tumors is one of the future research directions, which will provide powerful therapeutic targets for EBV-driven malignancies.
Future perspectives
EBV research highlights the insights showing that this virus continues to play an important etiological and pathogenetic role in EBV-associated cancers and other diseases. As an epigenetic driver, EBV mediates epigenetic effects, which are closely associated with a methylation-specific phenotype (EBV-CIMP). Also, EBV infection disrupts the balance in DNA methylation or demethylation. Development of panels of methylation genes and noninvasive DNA-based liquid biopsies, which exhibit disease-specific methylation patterns, will provide specific clinical significance for diagnosing these diseases. Discovery of promising demethylation agents for intervention in EBV-CIMP will be a new future direction.
Accumulating evidence confirms that EBV can target mitochondria directly or indirectly. The onco-signaling from EBV-encoded oncoproteins alter multiple mitochondrial functions, including metabolic reprogramming, cell death resistance, and mitochondrial dynamics. These finding provide new insights in understanding the role of mitochondria in EBV-associated carcinogenesis. Thus, targeting mitochondrial-related signaling will be a novel therapeutic strategy against EBV-associated diseases.
Lytic EBV replication contributes to viral-related tumorigenesis and systemic autoimmune diseases. The association of varying degrees of lytic EBV replication with EBV-related malignancies corresponds directly with the therapeutic resistance and prognosis of patients. Understanding new mechanisms of lytic replication and targeting lytic anti-genes will assist in early identification of high-risk individuals and is very meaningful in clinical settings, including staging and therapeutic responses.
EBV co-evolves with the host, which establishes a life-long interacting relationship between the pathogen and host. Interestingly, EBV relies on host machinery and hijacks the ubiquitin system, even encoding-DUB to meet its survival needs. Also, EBV-encoded ncRNA is a highly coordinated mechanism through which EBV might mimic a competition with human miRNAs to affect cellular functions by targeting host genes. Moreover, molecular mimicry between EBV and host proteins builds up a new link between EBV infection and SADs. The ultimate challenge is to translate these scientific findings into novel and more effective diagnosis and clinical therapeutic strategies in the future.
EBV has been shown to trigger a strong immune response, which provides a good basis for immunotherapy in EBV-related diseases. Further clinical trials will help determine the immunotherapy in the treatment of these diseases and provide a therapeutic option for patients. In addition, new vaccination strategies against EBV need to be further explored and will be a most important issue for EBV-associated diseases.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (81430064, 81602402, and 81672705, 81874172).
Competing interests
The authors declare no competing interests.
References
- 1.Lieberman PM. Virology. Epstein-Barr virus turns 50. Science. 2014;343:1323–1325. doi: 10.1126/science.1252786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xu M, et al. Genome-wide profiling of Epstein-Barr virus integration by targeted sequencing in Epstein-Barr virus associated malignancies. Theranostics. 2019;9:1115–1124. doi: 10.7150/thno.29622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Holmes D. The cancer-virus cures. Nat. Med. 2014;20:571–574. doi: 10.1038/nm0614-571. [DOI] [PubMed] [Google Scholar]
- 4.Young LS, Yap LF, Murray PG. Epstein-Barr virus: more than 50 years old and still providing surprises. Nat. Rev. Cancer. 2016;16:789–802. doi: 10.1038/nrc.2016.92. [DOI] [PubMed] [Google Scholar]
- 5.Klein G. Tumor associations of EBV-historical perspectives. Curr. Top. Microbiol. Immunol. 2015;390:17–22. doi: 10.1007/978-3-319-22822-8_2. [DOI] [PubMed] [Google Scholar]
- 6.Ascherio A, Munger KL. EBV and Autoimmunity. Curr. Top. Microbiol. Immunol. 2015;390:365–385. doi: 10.1007/978-3-319-22822-8_15. [DOI] [PubMed] [Google Scholar]
- 7.Münz C. Latency and lytic replication in Epstein-Barr virus-associated oncogenesis. Nat. Rev. Microbiol. 2019;17:691–700. doi: 10.1038/s41579-019-0249-7. [DOI] [PubMed] [Google Scholar]
- 8.Wang D, Liebowitz D, Kieff E. An EBV membrane protein expressed in immortalized lymphocytes transforms established rodent cells. Cell. 1985;43:831–840. doi: 10.1016/0092-8674(85)90256-9. [DOI] [PubMed] [Google Scholar]
- 9.Mosialos G, et al. The Epstein-Barr virus transforming protein LMP1 engages signaling proteins for the tumor necrosis factor receptor family. Cell. 1995;80:389–399. doi: 10.1016/0092-8674(95)90489-1. [DOI] [PubMed] [Google Scholar]
- 10.Dawson CW, Port RJ, Young LS. The role of the EBV-encoded latent membrane proteins LMP1 and LMP2 in the pathogenesis of nasopharyngeal carcinoma (NPC) Semin. Cancer Biol. 2012;22:144–153. doi: 10.1016/j.semcancer.2012.01.004. [DOI] [PubMed] [Google Scholar]
- 11.Chen YP, et al. Nasopharyngeal carcinoma. Lancet. 2019;394:64–80. doi: 10.1016/S0140-6736(19)30956-0. [DOI] [PubMed] [Google Scholar]
- 12.Longnecker R, Kieff E. A second Epstein-Barr virus membrane protein (LMP2) is expressed in latent infection and colocalizes with LMP1. J. Virol. 1990;64:2319–2326. doi: 10.1128/JVI.64.5.2319-2326.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Caldwell RG, Wilson JB, Anderson SJ, Longnecker R. Epstein-Barr virus LMP2A drives B cell development and survival in the absence of normal B cell receptor signals. Immunity. 1998;9:405–411. doi: 10.1016/S1074-7613(00)80623-8. [DOI] [PubMed] [Google Scholar]
- 14.Yates JL, Warren N, Sugden B. Stable replication of plasmids derived from Epstein-Barr virus in various mammalian cells. Nature. 1985;313:812–815. doi: 10.1038/313812a0. [DOI] [PubMed] [Google Scholar]
- 15.Wilson JB, Bell JL, Levine AJ. Expression of Epstein-Barr virus nuclear antigen-1 induces B cell neoplasia in transgenic mice. EMBO J. 1996;15:3117–3126. doi: 10.1002/j.1460-2075.1996.tb00674.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gruhne B, et al. The Epstein-Barr virus nuclear antigen-1 promotes genomic instability via induction of reactive oxygen species. Proc. Natl Acad. Sci. USA. 2009;106:2313–2318. doi: 10.1073/pnas.0810619106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kennedy G, Komano J, Sugden B. Epstein-Barr virus provides a survival factor to Burkitt’s lymphomas. Proc. Natl Acad. Sci. USA. 2003;100:14269–14274. doi: 10.1073/pnas.2336099100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tomkinson B, Robertson E, Kieff E. Epstein-Barr virus nuclear proteins EBNA-3A and EBNA-3C are essential for B-lymphocyte growth transformation. J. Virol. 1993;67:2014–2025. doi: 10.1128/JVI.67.4.2014-2025.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pfeffer S, et al. Identification of virus-encoded microRNAs. Science. 2004;304:734–736. doi: 10.1126/science.1096781. [DOI] [PubMed] [Google Scholar]
- 20.Old LJ, et al. Precipitating antibody in human serum to an antigen present in cultured burkitt’s lymphoma cells. Proc. Natl Acad. Sci. USA. 1966;56:1699–1704. doi: 10.1073/pnas.56.6.1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zeng Y, et al. Prospective studies on nasopharyngeal carcinoma in Epstein-Barr virus IgA/VCA antibody-positive persons in Wuzhou City, China. Int. J. Cancer. 1985;36:545–547. doi: 10.1002/ijc.2910360505. [DOI] [PubMed] [Google Scholar]
- 22.Rowe M, et al. Differences in B cell growth phenotype reflect novel patterns of Epstein-Barr virus latent gene expression in Burkitt’s lymphoma cells. Embo J. 1987;6:2743–2751. doi: 10.1002/j.1460-2075.1987.tb02568.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lo YM, et al. Quantitative and temporal correlation between circulating cell-free Epstein-Barr virus DNA and tumor recurrence in nasopharyngeal carcinoma. Cancer Res. 1999;59:5452–5455. [PubMed] [Google Scholar]
- 24.Lin DC, et al. The genomic landscape of nasopharyngeal carcinoma. Nat. Genet. 2014;46:866–871. doi: 10.1038/ng.3006. [DOI] [PubMed] [Google Scholar]
- 25.Xu M, et al. Genome sequencing analysis identifies Epstein-Barr virus subtypes associated with high risk of nasopharyngeal carcinoma. Nat. Genet. 2019;51:1131–1136. doi: 10.1038/s41588-019-0436-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang H, et al. Ephrin receptor A2 is an epithelial cell receptor for Epstein-Barr virus entry. Nat. Microbiol. 2018;3:1–8. doi: 10.1038/s41564-017-0093-3. [DOI] [PubMed] [Google Scholar]
- 27.Shibata D, Weiss LM. Epstein-Barr virus-associated gastric adenocarcinoma. Am. J. Pathol. 1992;140:769–774. [PMC free article] [PubMed] [Google Scholar]
- 28.Matsusaka K, et al. Classification of Epstein-Barr virus-positive gastric cancers by definition of DNA methylation epigenotypes. Cancer Res. 2011;71:7187–7197. doi: 10.1158/0008-5472.CAN-11-1349. [DOI] [PubMed] [Google Scholar]
- 29.Bass AJ, et al. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202–209. doi: 10.1038/nature13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cai L, et al. The better survival of MSI subtype is associated with the oxidative stress related pathways in gastric cancer. Front. Oncol. 2020;10:1269. doi: 10.3389/fonc.2020.01269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Labrecque LG, Barnes DM, Fentiman IS, Griffin BE. Epstein-Barr virus in epithelial cell tumors: a breast cancer study. Cancer Res. 1995;55:39–45. [PubMed] [Google Scholar]
- 32.Farahmand M, et al. Epstein-Barr virus and risk of breast cancer: a systematic review and meta-analysis. Future Oncol. 2019;15:2873–2885. doi: 10.2217/fon-2019-0232. [DOI] [PubMed] [Google Scholar]
- 33.Hu H, et al. Epstein-Barr virus infection of mammary epithelial cells promotes malignant transformation. EBioMedicine. 2016;9:148–160. doi: 10.1016/j.ebiom.2016.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hong S, et al. The genomic landscape of Epstein-Barr virus-associated pulmonary lymphoepithelioma-like carcinoma. Nat. Commun. 2019;10:3108. doi: 10.1038/s41467-019-10902-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mordasini V, et al. Activation of ATR-Chk1 pathway facilitates EBV-mediated transformation of primary tonsillar B-cells. Oncotarget. 2017;8:6461–6474. doi: 10.18632/oncotarget.14120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen ZX, et al. EBV as a potential risk factor for hepatobiliary system cancer: A meta-analysis with 918 cases. Pathol. Res. Pract. 2019;215:278–285. doi: 10.1016/j.prp.2018.10.037. [DOI] [PubMed] [Google Scholar]
- 37.Mozaffari HR, Ramezani M, Janbakhsh A, Sadeghi M. Malignant salivary gland tumors and Epstein-Barr Virus (EBV) infection: a systematic review and meta-analysis. Asian Pac. J. Cancer Prev.: APJCP. 2017;18:1201–1206. doi: 10.22034/APJCP.2017.18.5.1201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Almeida JFM, et al. Epstein-Barr virus and thyroid cancer. Crit. Rev. Oncogenesis. 2019;24:369–377. doi: 10.1615/CritRevOncog.2019031618. [DOI] [PubMed] [Google Scholar]
- 39.Murray PG, Young LS. An etiological role for the Epstein-Barr virus in the pathogenesis of classical Hodgkin lymphoma. Blood. 2019;134:591–596. doi: 10.1182/blood.2019000568. [DOI] [PubMed] [Google Scholar]
- 40.Epstein MA, Achong BG, Barr YM. Virus particles in cultured lymphoblasts from burkitt’s lymphoma. Lancet. 1964;1:702–703. doi: 10.1016/S0140-6736(64)91524-7. [DOI] [PubMed] [Google Scholar]
- 41.zur Hausen H, et al. EBV DNA in biopsies of Burkitt tumours and anaplastic carcinomas of the nasopharynx. Nature. 1970;228:1056–1058. doi: 10.1038/2281056a0. [DOI] [PubMed] [Google Scholar]
- 42.de-Thé G, et al. Epidemiological evidence for causal relationship between Epstein-Barr virus and Burkitt’s lymphoma from Ugandan prospective study. Nature. 1978;274:756–761. doi: 10.1038/274756a0. [DOI] [PubMed] [Google Scholar]
- 43.Weiss LM, Strickler JG, Warnke RA, Purtilo DT, Sklar J. Epstein-Barr viral DNA in tissues of Hodgkin’s disease. Am. J. Pathol. 1987;129:86–91. [PMC free article] [PubMed] [Google Scholar]
- 44.Poppema S, van Imhoff G, Torensma R, Smit J. Lymphadenopathy morphologically consistent with Hodgkin’s disease associated with Epstein-Barr virus infection. Am. J. Clin. Pathol. 1985;84:385–390. doi: 10.1093/ajcp/84.3.385. [DOI] [PubMed] [Google Scholar]
- 45.Jones JF, et al. T-cell lymphomas containing Epstein-Barr viral DNA in patients with chronic Epstein-Barr virus infections. N. Engl. J. Med. 1988;318:733–741. doi: 10.1056/NEJM198803243181203. [DOI] [PubMed] [Google Scholar]
- 46.Green M, Michaels MG. Epstein-Barr virus infection and posttransplant lymphoproliferative disorder. Am. J. Transplant. 2013;13:41–54. doi: 10.1111/ajt.12004. [DOI] [PubMed] [Google Scholar]
- 47.Al-Mansour Z, Nelson BP, Evens AM. Post-transplant lymphoproliferative disease (PTLD): risk factors, diagnosis, and current treatment strategies. Curr. Hematol. Malig. Rep. 2013;8:173–183. doi: 10.1007/s11899-013-0162-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nagington J, Gray J. Cyclosporin A immunosuppression, Epstein-Barr antibody, and lymphoma. Lancet. 1980;1:536–537. doi: 10.1016/S0140-6736(80)92784-1. [DOI] [PubMed] [Google Scholar]
- 49.Rooney CM, et al. Use of gene-modified virus-specific T lymphocytes to control Epstein-Barr-virus-related lymphoproliferation. Lancet (Lond., Engl.) 1995;345:9–13. doi: 10.1016/S0140-6736(95)91150-2. [DOI] [PubMed] [Google Scholar]
- 50.Griffin BE. Relation of Burkitt’s tumor-associated herpes-type virus to infectious mononucleosis. Rev. Med. Virol. 1998;8:61–66. doi: 10.1002/(SICI)1099-1654(199804/06)8:2<61::AID-RMV215>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- 51.Moss DJ, Lutzky VP. EBV-Specific immune response: early research and personal reminiscences. Curr. Top. Microbiol. Immunol. 2015;390:23–42. doi: 10.1007/978-3-319-22822-8_3. [DOI] [PubMed] [Google Scholar]
- 52.Tseng YJ, Ding WQ, Zhong L, Chen J, Luo ZG. Chronic active Epstein-Barr virus (CAEBV) enteritis. Int. J. Infect. Dis. 2019;82:15–17. doi: 10.1016/j.ijid.2019.02.020. [DOI] [PubMed] [Google Scholar]
- 53.Liu R, et al. The clinicopathologic features of chronic active Epstein-Barr virus infective enteritis. Mod. Pathol. 2019;32:387–395. doi: 10.1038/s41379-018-0144-1. [DOI] [PubMed] [Google Scholar]
- 54.Kimura H, Cohen JI. Chronic active Epstein-Barr virus disease. Front. Immunol. 2017;8:1867. doi: 10.3389/fimmu.2017.01867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kimura H, et al. Differences between T cell-type and natural killer cell-type chronic active Epstein-Barr virus infection. J. Infect. Dis. 2005;191:531–539. doi: 10.1086/427239. [DOI] [PubMed] [Google Scholar]
- 56.Sonke G, et al. Poor outcomes of chronic active Epstein-Barr virus infection and hemophagocytic lymphohistiocytosis in non-Japanese adult patients. Clin. Infect. Dis. 2008;47:105–108. doi: 10.1086/588790. [DOI] [PubMed] [Google Scholar]
- 57.Ohshima K, et al. Clinicopathological study of severe chronic active Epstein-Barr virus infection that developed in association with lymphoproliferative disorder and/or hemophagocytic syndrome. Pathol. Int. 1998;48:934–943. doi: 10.1111/j.1440-1827.1998.tb03864.x. [DOI] [PubMed] [Google Scholar]
- 58.Collins, P. et al. Characterising EBV-associated lymphoproliferative diseases and the role of myeloid-derived suppressor cells. Blood. 10.1182/blood.2020005611 (2020). [DOI] [PubMed]
- 59.Liu P, et al. Nivolumab treatment of relapsed/refractory Epstein-Barr virus-associated hemophagocytic lymphohistiocytosis in adults. Blood. 2020;135:826–833. doi: 10.1182/blood.2019003886. [DOI] [PubMed] [Google Scholar]
- 60.Henter JI, et al. HLH-2004: Diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatr. Blood Cancer. 2007;48:124–131. doi: 10.1002/pbc.21039. [DOI] [PubMed] [Google Scholar]
- 61.Draborg AH, Duus K, Houen G. Epstein-Barr virus in systemic autoimmune diseases. Clin. Dev. Immunol. 2013;2013:535738. doi: 10.1155/2013/535738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Dhe-Paganon S, Syeda F, Park L. DNA methyl transferase 1: regulatory mechanisms and implications in health and disease. Int. J. Biochem. Mol. Biol. 2011;2:58–66. [PMC free article] [PubMed] [Google Scholar]
- 63.Rasmussen KD, Helin K. Role of TET enzymes in DNA methylation, development, and cancer. Genes Dev. 2016;30:733–750. doi: 10.1101/gad.276568.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cao Y, DePinho RA, Ernst M, Vousden K. Cancer research: past, present and future. Nat. Rev. Cancer. 2011;11:749–754. doi: 10.1038/nrc3138. [DOI] [PubMed] [Google Scholar]
- 65.Cao Y. EBV based cancer prevention and therapy in nasopharyngeal carcinoma. NPJ Precis Oncol. 2017;1:10. doi: 10.1038/s41698-017-0018-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tsai CL, et al. Activation of DNA methyltransferase 1 by EBV LMP1 Involves c-Jun NH(2)-terminal kinase signaling. Cancer Res. 2006;66:11668–11676. doi: 10.1158/0008-5472.CAN-06-2194. [DOI] [PubMed] [Google Scholar]
- 67.Luo X, et al. DNMT1 mediates metabolic reprogramming induced by Epstein-Barr virus latent membrane protein 1 and reversed by grifolin in nasopharyngeal carcinoma. Cell Death Dis. 2018;9:619. doi: 10.1038/s41419-018-0662-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Namba-Fukuyo H, et al. TET2 functions as a resistance factor against DNA methylation acquisition during Epstein-Barr virus infection. Oncotarget. 2016;7:81512–81526. doi: 10.18632/oncotarget.13130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Koivunen P, Laukka T. The TET enzymes. Cell. Mol. Life Sci. 2018;75:1339–1348. doi: 10.1007/s00018-017-2721-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Shi F, et al. EBV(LMP1)-induced metabolic reprogramming inhibits necroptosis through the hypermethylation of the RIP3 promoter. Theranostics. 2019;9:2424–2437. doi: 10.7150/thno.30941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Shi F, et al. Wild-type IDH2 contributes to Epstein-Barr virus-dependent metabolic alterations and tumorigenesis. Mol. Metab. 2020;36:100966. doi: 10.1016/j.molmet.2020.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yang Z, et al. 2-HG inhibits necroptosis by stimulating DNMT1-dependent hypermethylation of the RIP3 promoter. Cell Rep. 2017;19:1846–1857. doi: 10.1016/j.celrep.2017.05.012. [DOI] [PubMed] [Google Scholar]
- 73.Lo KW, Chung GT, To KF. Deciphering the molecular genetic basis of NPC through molecular, cytogenetic, and epigenetic approaches. Semin Cancer Biol. 2012;22:79–86. doi: 10.1016/j.semcancer.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 74.Cancer Genome Atlas Research, N. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202–209. doi: 10.1038/nature13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lin Z, et al. S100A4 hypomethylation affects epithelial-mesenchymal transition partially induced by LMP2A in nasopharyngeal carcinoma. Mol. Carcinog. 2016;55:1467–1476. doi: 10.1002/mc.22389. [DOI] [PubMed] [Google Scholar]
- 76.Dhiab MB, et al. DNA methylation patterns in EBV-positive and EBV-negative Hodgkin lymphomas. Cell. Oncol. 2015;38:453–462. doi: 10.1007/s13402-015-0242-8. [DOI] [PubMed] [Google Scholar]
- 77.Zheng XH, et al. Detection of methylation status of Epstein-Barr virus DNA C promoter in the diagnosis of nasopharyngeal carcinoma. Cancer Sci. 2020;111:592–600. doi: 10.1111/cas.14281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lam WKJ, et al. Methylation analysis of plasma DNA informs etiologies of Epstein-Barr virus-associated diseases. Nat. Commun. 2019;10:3256. doi: 10.1038/s41467-019-11226-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ye M, Huang T, Ni C, Yang P, Chen S. Diagnostic capacity of RASSF1A promoter methylation as a biomarker in tissue, brushing, and blood samples of nasopharyngeal carcinoma. EBioMedicine. 2017;18:32–40. doi: 10.1016/j.ebiom.2017.03.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ren XY, et al. TIPE3 hypermethylation correlates with worse prognosis and promotes tumor progression in nasopharyngeal carcinoma. J. Exp. Clin. Cancer Res. : CR. 2018;37:227. doi: 10.1186/s13046-018-0881-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ren X, et al. HOPX hypermethylation promotes metastasis via activating SNAIL transcription in nasopharyngeal carcinoma. Nat. Commun. 2017;8:14053. doi: 10.1038/ncomms14053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Li Y, et al. RAB37 hypermethylation regulates metastasis and resistance to docetaxel-based induction chemotherapy in nasopharyngeal carcinoma. Clin. Cancer Res. 2018;24:6495–6508. doi: 10.1158/1078-0432.CCR-18-0532. [DOI] [PubMed] [Google Scholar]
- 83.Hutajulu SH, et al. Epigenetic markers for early detection of nasopharyngeal carcinoma in a high risk population. Mol. Cancer. 2011;10:48. doi: 10.1186/1476-4598-10-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yang X, et al. Epigenetic markers for noninvasive early detection of nasopharyngeal carcinoma by methylation-sensitive high resolution melting. Int. J. Cancer. 2015;136:E127–E135. doi: 10.1002/ijc.29192. [DOI] [PubMed] [Google Scholar]
- 85.Jiang W, et al. Genome-wide identification of a methylation gene panel as a prognostic biomarker in nasopharyngeal carcinoma. Mol. Cancer Ther. 2015;14:2864–2873. doi: 10.1158/1535-7163.MCT-15-0260. [DOI] [PubMed] [Google Scholar]
- 86.Zwergel C, Valente S, Mai A. DNA methyltransferases inhibitors from natural sources. Curr. Top. Med. Chem. 2016;16:680–696. doi: 10.2174/1568026615666150825141505. [DOI] [PubMed] [Google Scholar]
- 87.Jiang W, et al. 5-Azacytidine enhances the radiosensitivity of CNE2 and SUNE1 cells in vitro and in vivo possibly by altering DNA methylation. PLoS ONE. 2014;9:e93273. doi: 10.1371/journal.pone.0093273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mesia R, et al. Phase II study of CC-486 (oral azacitidine) in previously treated patients with locally advanced or metastatic nasopharyngeal carcinoma. Eur. J. Cancer. 2019;123:138–145. doi: 10.1016/j.ejca.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 89.Luo X, et al. Grifolin inhibits tumor cells adhesion and migration via suppressing interplay between PGC1alpha and Fra-1/LSF-MMP2/CD44 axes. Oncotarget. 2016;7:68708–68720. doi: 10.18632/oncotarget.11929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Luo X, et al. Grifolin directly targets ERK1/2 to epigenetically suppress cancer cell metastasis. Oncotarget. 2015;6:42704–42716. doi: 10.18632/oncotarget.5678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Luo XJ, et al. DAPK1 mediates the G1 phase arrest in human nasopharyngeal carcinoma cells induced by grifolin, a potential antitumor natural product. Eur. J. Pharm. 2011;670:427–434. doi: 10.1016/j.ejphar.2011.08.026. [DOI] [PubMed] [Google Scholar]
- 92.Luo XJ, et al. Grifolin, a potent antitumour natural product upregulates death-associated protein kinase 1 DAPK1 via p53 in nasopharyngeal carcinoma cells. Eur. J. Cancer. 2011;47:316–325. doi: 10.1016/j.ejca.2010.09.021. [DOI] [PubMed] [Google Scholar]
- 93.Ye M, et al. Grifolin, a potential antitumor natural product from the mushroom Albatrellus confluens, induces cell-cycle arrest in G1 phase via the ERK1/2 pathway. Cancer Lett. 2007;258:199–207. doi: 10.1016/j.canlet.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 94.Jin S, et al. Grifolin induces apoptosis via inhibition of PI3K/AKT signalling pathway in human osteosarcoma cells. Apoptosis. 2007;12:1317–1326. doi: 10.1007/s10495-007-0062-z. [DOI] [PubMed] [Google Scholar]
- 95.Ye M, et al. Grifolin, a potential antitumor natural product from the mushroom Albatrellus confluens, inhibits tumor cell growth by inducing apoptosis in vitro. FEBS Lett. 2005;579:3437–3443. doi: 10.1016/j.febslet.2005.05.013. [DOI] [PubMed] [Google Scholar]
- 96.Zhang J, Jia L, Tsang CM, Tsao SW. EBV infection and glucose metabolism in nasopharyngeal carcinoma. Adv. Exp. Med. Biol. 2017;1018:75–90. doi: 10.1007/978-981-10-5765-6_6. [DOI] [PubMed] [Google Scholar]
- 97.Zhou JC, et al. Expression of GLUT-1 in nasopharyngeal carcinoma and its clinical significance. Eur. Rev. Med. Pharm. Sci. 2017;21:4891–4895. [PubMed] [Google Scholar]
- 98.Xiao L, et al. Targeting Epstein-Barr virus oncoprotein LMP1-mediated glycolysis sensitizes nasopharyngeal carcinoma to radiation therapy. Oncogene. 2014;33:4568–4578. doi: 10.1038/onc.2014.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Lu J, et al. EBV-LMP1 suppresses the DNA damage response through DNA-PK/AMPK signaling to promote radioresistance in nasopharyngeal carcinoma. Cancer Lett. 2016;380:191–200. doi: 10.1016/j.canlet.2016.05.032. [DOI] [PubMed] [Google Scholar]
- 100.Jiang Y, et al. Repression of Hox genes by LMP1 in nasopharyngeal carcinoma and modulation of glycolytic pathway genes by HoxC8. Oncogene. 2015;34:6079–6091. doi: 10.1038/onc.2015.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Xie L, et al. Drp1-dependent remodeling of mitochondrial morphology triggered by EBV-LMP1 increases cisplatin resistance. Signal Transduct. Target Ther. 2020;5:56. doi: 10.1038/s41392-020-0151-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wu X, et al. Extracellular vesicle packaged LMP1-activated fibroblasts promote tumor progression via autophagy and stroma-tumor metabolism coupling. Cancer Lett. 2020;478:93–106. doi: 10.1016/j.canlet.2020.03.004. [DOI] [PubMed] [Google Scholar]
- 103.Lo AK, et al. Activation of the FGFR1 signalling pathway by the Epstein-Barr virus-encoded LMP1 promotes aerobic glycolysis and transformation of human nasopharyngeal epithelial cells. J. Pathol. 2015;237:238–248. doi: 10.1002/path.4575. [DOI] [PubMed] [Google Scholar]
- 104.Zhang J, et al. mTORC2-mediated PDHE1alpha nuclear translocation links EBV-LMP1 reprogrammed glucose metabolism to cancer metastasis in nasopharyngeal carcinoma. Oncogene. 2019;38:4669–4684. doi: 10.1038/s41388-019-0749-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lichota A, Gwozdzinski K. Anticancer activity of natural compounds from plant and marine environment. Int. J. Mol. Sci. 2018;19:3533. doi: 10.3390/ijms19113533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Guerra AR, Duarte MF, Duarte IF. Targeting tumor metabolism with plant-derived natural products: emerging trends in cancer therapy. J. Agric. Food Chem. 2018;66:10663–10685. doi: 10.1021/acs.jafc.8b04104. [DOI] [PubMed] [Google Scholar]
- 107.Hellwig V, et al. Activities of prenylphenol derivatives from fruitbodies of Albatrellus spp. on the human and rat vanilloid receptor 1 (VR1) and characterisation of the novel natural product, confluentin. Arch. Pharm. (Weinh.) 2003;336:119–126. doi: 10.1002/ardp.200390008. [DOI] [PubMed] [Google Scholar]
- 108.Deng Q, et al. Neoalbaconol induces energy depletion and multiple cell death in cancer cells by targeting PDK1-PI3-K/Akt signaling pathway. Cell Death Dis. 2013;4:e804. doi: 10.1038/cddis.2013.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yoon SJ, et al. Comprehensive multi-omics analysis reveals aberrant metabolism of Epstein-Barr-virus-associated gastric carcinoma. Cells. 2019;8:1220. doi: 10.3390/cells8101220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lo AK, et al. Activation of sterol regulatory element-binding protein 1 (SREBP1)-mediated lipogenesis by the Epstein-Barr virus-encoded latent membrane protein 1 (LMP1) promotes cell proliferation and progression of nasopharyngeal carcinoma. J. Pathol. 2018;246:180–190. doi: 10.1002/path.5130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Caro P, et al. Metabolic signatures uncover distinct targets in molecular subsets of diffuse large B cell lymphoma. Cancer Cell. 2012;22:547–560. doi: 10.1016/j.ccr.2012.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Tan Z, et al. Targeting CPT1A-mediated fatty acid oxidation sensitizes nasopharyngeal carcinoma to radiation therapy. Theranostics. 2018;8:2329–2347. doi: 10.7150/thno.21451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Du Q, et al. PGC1alpha/CEBPB/CPT1A axis promotes radiation resistance of nasopharyngeal carcinoma through activating fatty acid oxidation. Cancer Sci. 2019;110:2050–2062. doi: 10.1111/cas.14011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Fu S, et al. Glutamine synthetase promotes radiation resistance via facilitating nucleotide metabolism and subsequent DNA Damage repair. Cell Rep. 2019;28:1136–1143 e1134. doi: 10.1016/j.celrep.2019.07.002. [DOI] [PubMed] [Google Scholar]
- 115.Wang LW, et al. Epstein-Barr-virus-induced one-carbon metabolism drives B cell transformation. Cell Metab. 2019;30:539–555 e511. doi: 10.1016/j.cmet.2019.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Baratta MG. Virus-mediated hijack of one-carbon metabolism. Nat. Rev. Cancer. 2019;19:486. doi: 10.1038/s41568-019-0190-0. [DOI] [PubMed] [Google Scholar]
- 117.Yi L, et al. A metabolic discrimination model for nasopharyngeal carcinoma and its potential role in the therapeutic evaluation of radiotherapy. Metabolomics. 2014;10:12. doi: 10.1007/s11306-013-0606-x. [DOI] [Google Scholar]
- 118.Liu WS, et al. The role of pretreatment FDG-PET in nasopharyngeal carcinoma treated with intensity-modulated radiotherapy. Int J. Radiat. Oncol. Biol. Phys. 2012;82:561–566. doi: 10.1016/j.ijrobp.2010.11.055. [DOI] [PubMed] [Google Scholar]
- 119.Tang LQ, et al. Prospective study of tailoring whole-body dual-modality [18F]fluorodeoxyglucose positron emission tomography/computed tomography with plasma Epstein-Barr virus DNA for detecting distant metastasis in endemic nasopharyngeal carcinoma at initial staging. J. Clin. Oncol. 2013;31:2861–2869. doi: 10.1200/JCO.2012.46.0816. [DOI] [PubMed] [Google Scholar]
- 120.Surov A, Meyer HJ, Hohn AK, Sabri O, Purz S. Combined metabolo-volumetric parameters of (18)F-FDG-PET and MRI can predict tumor cellularity, ki67 level and expression of HIF 1alpha in head and neck squamous cell carcinoma: a pilot study. Transl. Oncol. 2019;12:8–14. doi: 10.1016/j.tranon.2018.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Alessi A, et al. Role of pretreatment 18F-FDG PET/CT parameters in predicting outcome of non-endemic EBV DNA-related nasopharyngeal cancer (NPC) patients treated with IMRT and chemotherapy. Radio. Med. 2019;124:414–421. doi: 10.1007/s11547-018-0980-6. [DOI] [PubMed] [Google Scholar]
- 122.Chen YH, et al. Value of early evaluation of treatment response using (18)F-FDG PET/CT parameters and the Epstein-Barr virus DNA load for prediction of outcome in patients with primary nasopharyngeal carcinoma. Eur. J. Nucl. Med. Mol. Imaging. 2019;46:650–660. doi: 10.1007/s00259-018-4172-3. [DOI] [PubMed] [Google Scholar]
- 123.Tan AS, et al. Mitochondrial genome acquisition restores respiratory function and tumorigenic potential of cancer cells without mitochondrial DNA. Cell Metab. 2015;21:81–94. doi: 10.1016/j.cmet.2014.12.003. [DOI] [PubMed] [Google Scholar]
- 124.Shao JY, et al. High frequency of common deletion (4981 bp) in mitochondrial DNA in nasopharyngeal carcinoma and its correlation with patient age and clinical stages. Cancer Biol. Ther. 2004;3:1270–1274. doi: 10.4161/cbt.3.12.1243. [DOI] [PubMed] [Google Scholar]
- 125.Peng Z, et al. Sequence variations of mitochondrial DNA D-loop region are associated with familial nasopharyngeal carcinoma. Mitochondrion. 2011;11:327–333. doi: 10.1016/j.mito.2010.12.008. [DOI] [PubMed] [Google Scholar]
- 126.Hu SP, Du JP, Li DR, Yao YG. Mitochondrial DNA haplogroup confers genetic susceptibility to nasopharyngeal carcinoma in Chaoshanese from Guangdong, China. PLoS ONE. 2014;9:e87795. doi: 10.1371/journal.pone.0087795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Chevallier-Greco A, et al. Both Epstein-Barr virus (EBV)-encoded trans-acting factors, EB1 and EB2, are required to activate transcription from an EBV early promoter. EMBO J. 1986;5:3243–3249. doi: 10.1002/j.1460-2075.1986.tb04635.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Miralles Fuste J, et al. In vivo occupancy of mitochondrial single-stranded DNA binding protein supports the strand displacement mode of DNA replication. PLoS Genet. 2014;10:e1004832. doi: 10.1371/journal.pgen.1004832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Wiedmer A, et al. Epstein-Barr virus immediate-early protein Zta co-opts mitochondrial single-stranded DNA binding protein to promote viral and inhibit mitochondrial DNA replication. J. Virol. 2008;82:4647–4655. doi: 10.1128/JVI.02198-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Caino MC, Altieri DC. Cancer cells exploit adaptive mitochondrial dynamics to increase tumor cell invasion. Cell Cycle. 2015;14:3242–3247. doi: 10.1080/15384101.2015.1084448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Senft D, Ronai ZA. Regulators of mitochondrial dynamics in cancer. Curr. Opin. Cell Biol. 2016;39:43–52. doi: 10.1016/j.ceb.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chen H, Chan DC. Emerging functions of mammalian mitochondrial fusion and fission. Hum. Mol. Genet. 2005;14:R283–R289. doi: 10.1093/hmg/ddi270. [DOI] [PubMed] [Google Scholar]
- 133.Zhao J, et al. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene. 2013;32:4814–4824. doi: 10.1038/onc.2012.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Ferreira-da-Silva A, et al. Mitochondrial dynamics protein Drp1 is overexpressed in oncocytic thyroid tumors and regulates cancer cell migration. PLoS ONE. 2015;10:e0122308. doi: 10.1371/journal.pone.0122308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Kashatus JA, et al. Erk2 phosphorylation of Drp1 promotes mitochondrial fission and MAPK-driven tumor growth. Mol. Cell. 2015;57:537–551. doi: 10.1016/j.molcel.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Zhu PP, et al. Intra- and intermolecular domain interactions of the C-terminal GTPase effector domain of the multimeric dynamin-like GTPase Drp1. J. Biol. Chem. 2004;279:35967–35974. doi: 10.1074/jbc.M404105200. [DOI] [PubMed] [Google Scholar]
- 137.Tsao SW, Tsang CM, Lo KW. Epstein-Barr virus infection and nasopharyngeal carcinoma. Philos Trans. R Soc. Lond. B Biol. Sci. 2017;372:20160270. doi: 10.1098/rstb.2016.0270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Tavakolian S, Goudarzi H, Faghihloo E. Cyclin-dependent kinases and CDK inhibitors in virus-associated cancers. Infect. Agents Cancer. 2020;15:27. doi: 10.1186/s13027-020-00295-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Pal AD, Basak NP, Banerjee AS, Banerjee S. Epstein-Barr virus latent membrane protein-2A alters mitochondrial dynamics promoting cellular migration mediated by Notch signaling pathway. Carcinogenesis. 2014;35:1592–1601. doi: 10.1093/carcin/bgu069. [DOI] [PubMed] [Google Scholar]
- 140.LaJeunesse DR, Brooks K, Adamson AL. Epstein-Barr virus immediate-early proteins BZLF1 and BRLF1 alter mitochondrial morphology during lytic replication. Biochem. Biophys. Res. Commun. 2005;333:438–442. doi: 10.1016/j.bbrc.2005.05.120. [DOI] [PubMed] [Google Scholar]
- 141.Vilmen G, et al. BHRF1, a BCL2 viral homolog, disturbs mitochondrial dynamics and stimulates mitophagy to dampen type I IFN induction. Autophagy. 2020;13:1–20. doi: 10.1080/15548627.2020.1758416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Karbowski M, et al. Spatial and temporal association of Bax with mitochondrial fission sites, Drp1, and Mfn2 during apoptosis. J. Cell Biol. 2002;159:931–938. doi: 10.1083/jcb.200209124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Li Y, et al. Overexpression of mitochondria mediator gene TRIAP1 by miR-320b loss is associated with progression in nasopharyngeal carcinoma. PLoS Genet. 2016;12:e1006183. doi: 10.1371/journal.pgen.1006183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Mo Y, et al. Mitochondrial protein targets of radiosensitisation by 1,8-dihydroxy-3-acetyl-6-methyl-9,10 anthraquinone on nasopharyngeal carcinoma cells. Eur. J. Pharm. 2014;738:133–141. doi: 10.1016/j.ejphar.2014.05.027. [DOI] [PubMed] [Google Scholar]
- 145.Chang CR, Blackstone C. Dynamic regulation of mitochondrial fission through modification of the dynamin-related protein Drp1. Ann. N.Y. Acad. Sci. 2010;1201:34–39. doi: 10.1111/j.1749-6632.2010.05629.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Zhou TJ, et al. Downregulation of mitochondrial cyclooxygenase-2 inhibits the stemness of nasopharyngeal carcinoma by decreasing the activity of dynamin-related protein 1. Theranostics. 2017;7:1389–1406. doi: 10.7150/thno.17647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Chan DC. Fusion and fission: interlinked processes critical for mitochondrial health. Annu. Rev. Genet. 2012;46:265–287. doi: 10.1146/annurev-genet-110410-132529. [DOI] [PubMed] [Google Scholar]
- 148.Westermann B. Mitochondrial fusion and fission in cell life and death. Nat. Rev. Mol. Cell Biol. 2010;11:872–884. doi: 10.1038/nrm3013. [DOI] [PubMed] [Google Scholar]
- 149.Grosse L, et al. Bax assembles into large ring-like structures remodeling the mitochondrial outer membrane in apoptosis. EMBO J. 2016;35:402–413. doi: 10.15252/embj.201592789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Mohidin TB, Ng CC. BARF1 gene silencing triggers caspase-dependent mitochondrial apoptosis in Epstein-Barr virus-positive malignant cells. J. Biosci. 2015;40:41–51. doi: 10.1007/s12038-015-9502-z. [DOI] [PubMed] [Google Scholar]
- 151.Zhou H, et al. Triptolide inhibits Epstein-Barr nuclear antigen 1 expression by increasing sensitivity of mitochondria apoptosis of nasopharyngeal carcinoma cells. J. Exp. Clin. Cancer Res. 2018;37:192. doi: 10.1186/s13046-018-0865-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Chen Q, et al. HKDC1 C-terminal based peptides inhibit extranodal natural killer/T-cell lymphoma by modulation of mitochondrial function and EBV suppression. Leukemia. 2020;34:2736–2748. doi: 10.1038/s41375-020-0801-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Wang C, et al. Pectolinarigenin suppresses the tumor growth in nasopharyngeal carcinoma. Cell Physiol. Biochem. 2016;39:1795–1803. doi: 10.1159/000447879. [DOI] [PubMed] [Google Scholar]
- 154.Pan Z, et al. Cinobufagin induces cell cycle arrest at the S phase and promotes apoptosis in nasopharyngeal carcinoma cells. Biomed. Pharmacother. 2020;122:109763. doi: 10.1016/j.biopha.2019.109763. [DOI] [PubMed] [Google Scholar]
- 155.Su EY, et al. Bufalin induces apoptotic cell death in human nasopharyngeal carcinoma cells through mitochondrial ROS and TRAIL pathways. Am. J. Chin. Med. 2019;47:237–257. doi: 10.1142/S0192415X19500125. [DOI] [PubMed] [Google Scholar]
- 156.Lin HF, et al. Celastrol-induced apoptosis in human nasopharyngeal carcinoma is associated with the activation of the death receptor and the mitochondrial pathway. Oncol. Lett. 2017;14:1683–1690. doi: 10.3892/ol.2017.6346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Yao K, et al. Grape seed proanthocyanidins induce apoptosis through the mitochondrial pathway in nasopharyngeal carcinoma CNE-2 cells. Oncol. Rep. 2016;36:771–778. doi: 10.3892/or.2016.4855. [DOI] [PubMed] [Google Scholar]
- 158.Juang SH, et al. Mechanistic study of tetrahydrofuran- acetogenins in triggering endoplasmic reticulum stress response-apotoposis in human nasopharyngeal carcinoma. Sci. Rep. 2016;6:39251. doi: 10.1038/srep39251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lin YJ, et al. Tetrandrine induces apoptosis of human nasopharyngeal carcinoma NPC-TW 076 cells through reactive oxygen species accompanied by an endoplasmic reticulum stress signaling pathway. Molecules. 2016;21:1353. doi: 10.3390/molecules21101353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zhang P, Wang P, Yan L, Liu L. Synthesis of gold nanoparticles with Solanum xanthocarpum extract and their in vitro anticancer potential on nasopharyngeal carcinoma cells. Int. J. Nanomed. 2018;13:7047–7059. doi: 10.2147/IJN.S180138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Wang Z, et al. Depletion of NFBD1/MDC1 induces apoptosis in nasopharyngeal carcinoma cells through the p53-ROS-mitochondrial pathway. Oncol. Res. 2017;25:123–136. doi: 10.3727/096504016X14732772150226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Liao YC, Lo SH. Deleted in liver cancer-1 (DLC-1): a tumor suppressor not just for liver. Int. J. Biochem. Cell Biol. 2008;40:843–847. doi: 10.1016/j.biocel.2007.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Huang W, et al. DLC-1 induces mitochondrial apoptosis and epithelial mesenchymal transition arrest in nasopharyngeal carcinoma by targeting EGFR/Akt/NF-kappaB pathway. Med. Oncol. 2015;32:115. doi: 10.1007/s12032-015-0564-4. [DOI] [PubMed] [Google Scholar]
- 164.Kang R, et al. HMGB1 in health and disease. Mol. Asp. Med. 2014;40:1–116. doi: 10.1016/j.mam.2014.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Peng T, et al. Effects of high‑mobility group box 1 knockdown on proliferation, migration and invasion of the HONE‑1 human nasopharyngeal carcinoma cell line. Mol. Med. Rep. 2015;12:7531–7537. doi: 10.3892/mmr.2015.4402. [DOI] [PubMed] [Google Scholar]
- 166.Oh E, Akopian D, Rape M. Principles of ubiquitin-dependent signaling. Annu. Rev. Cell Dev. Biol. 2018;34:137–162. doi: 10.1146/annurev-cellbio-100617-062802. [DOI] [PubMed] [Google Scholar]
- 167.Qi J, Ronai ZA. Dysregulation of ubiquitin ligases in cancer. Drug Resist. Updat. 2015;23:1–11. doi: 10.1016/j.drup.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Hanpude P, Bhattacharya S, Dey AK, Maiti TK. Deubiquitinating enzymes in cellular signaling and disease regulation. IUBMB Life. 2015;67:544–555. doi: 10.1002/iub.1402. [DOI] [PubMed] [Google Scholar]
- 169.Li Y, et al. The role of deubiquitinases in oncovirus and host interactions. J. Oncol. 2019;2019:2128410. doi: 10.1155/2019/2128410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Farshi P, et al. Deubiquitinases (DUBs) and DUB inhibitors: a patent review. Expert Opin. Ther. Pat. 2015;25:1191–1208. doi: 10.1517/13543776.2015.1056737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.D’Arcy P, Wang X, Linder S. Deubiquitinase inhibition as a cancer therapeutic strategy. Pharm. Ther. 2015;147:32–54. doi: 10.1016/j.pharmthera.2014.11.002. [DOI] [PubMed] [Google Scholar]
- 172.Clague MJ, Urbe S, Komander D. Breaking the chains: deubiquitylating enzyme specificity begets function. Nat. Rev. Mol. Cell Biol. 2019;20:338–352. doi: 10.1038/s41580-019-0099-1. [DOI] [PubMed] [Google Scholar]
- 173.Saridakis V, et al. Structure of the p53 binding domain of HAUSP/USP7 bound to Epstein-Barr nuclear antigen 1 implications for EBV-mediated immortalization. Mol. Cell. 2005;18:25–36. doi: 10.1016/j.molcel.2005.02.029. [DOI] [PubMed] [Google Scholar]
- 174.Bentz GL, et al. KSHV LANA and EBV LMP1 induce the expression of UCH-L1 following viral transformation. Virology. 2014;448:293–302. doi: 10.1016/j.virol.2013.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Huang HH, et al. TRIM5alpha promotes ubiquitination of Rta from Epstein-Barr virus to attenuate lytic progression. Front. Microbiol. 2016;7:2129. doi: 10.3389/fmicb.2016.02129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Whitehurst CB, et al. The Epstein-Barr virus (EBV) deubiquitinating enzyme BPLF1 reduces EBV ribonucleotide reductase activity. J. Virol. 2009;83:4345–4353. doi: 10.1128/JVI.02195-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Li L, et al. Viral oncoprotein LMP1 disrupts p53-induced cell cycle arrest and apoptosis through modulating K63-linked ubiquitination of p53. Cell Cycle. 2012;11:2327–2336. doi: 10.4161/cc.20771. [DOI] [PubMed] [Google Scholar]
- 178.Pei Y, et al. Epstein-Barr virus nuclear antigen 3C facilitates cell proliferation by regulating cyclin D2. J. Virol. 2018;92:e00663-18. doi: 10.1128/JVI.00663-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Iwahori S, et al. Phosphorylation of p27Kip1 by Epstein-Barr virus protein kinase induces its degradation through SCFSkp2 ubiquitin ligase actions during viral lytic replication. J. Biol. Chem. 2009;284:18923–18931. doi: 10.1074/jbc.M109.015123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Najafov A, Chen H, Yuan J. Necroptosis and cancer. Trends Cancer. 2017;3:294–301. doi: 10.1016/j.trecan.2017.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Liu X, et al. Epstein-Barr virus encoded latent membrane protein 1 suppresses necroptosis through targeting RIPK1/3 ubiquitination. Cell Death Dis. 2018;9:53. doi: 10.1038/s41419-017-0081-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Sivachandran N, et al. Contributions of the Epstein-Barr virus EBNA1 protein to gastric carcinoma. J. Virol. 2012;86:60–68. doi: 10.1128/JVI.05623-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Chen SH, et al. O(6) -methylguanine DNA methyltransferase repairs platinum-DNA adducts following cisplatin treatment and predicts prognoses of nasopharyngeal carcinoma. Int. J. Cancer. 2015;137:1291–1305. doi: 10.1002/ijc.29486. [DOI] [PubMed] [Google Scholar]
- 184.Hsu SH, Chen SH, Kuo CC, Chang JY. Ubiquitin-conjugating enzyme E2 B regulates the ubiquitination of O(6)-methylguanine-DNA methyltransferase and BCNU sensitivity in human nasopharyngeal carcinoma cells. Biochem. Pharm. 2018;158:327–338. doi: 10.1016/j.bcp.2018.10.029. [DOI] [PubMed] [Google Scholar]
- 185.Whitehurst CB, Vaziri C, Shackelford J, Pagano JS. Epstein-Barr virus BPLF1 deubiquitinates PCNA and attenuates polymerase eta recruitment to DNA damage sites. J. Virol. 2012;86:8097–8106. doi: 10.1128/JVI.00588-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Kumar R, Whitehurst CB, Pagano JS. The Rad6/18 ubiquitin complex interacts with the Epstein-Barr virus deubiquitinating enzyme, BPLF1, and contributes to virus infectivity. J. Virol. 2014;88:6411–6422. doi: 10.1128/JVI.00536-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Lanz MC, Dibitetto D, Smolka MB. DNA damage kinase signaling: checkpoint and repair at 30 years. EMBO J. 2019;38:e101801. doi: 10.15252/embj.2019101801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Zhang P, et al. TRIM21-SERPINB5 aids GMPS repression to protect nasopharyngeal carcinoma cells from radiation-induced apoptosis. J. Biomed. Sci. 2020;27:30. doi: 10.1186/s12929-020-0625-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Wang H, et al. LPLUNC1 stabilises PHB1 by counteracting TRIM21-mediated ubiquitination to inhibit NF-kappaB activity in nasopharyngeal carcinoma. Oncogene. 2019;38:5062–5075. doi: 10.1038/s41388-019-0778-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Zhang J, et al. Hypermethylation of SHISA3 promotes nasopharyngeal carcinoma metastasis by reducing SGSM1 stability. Cancer Res. 2019;79:747–759. doi: 10.1158/0008-5472.CAN-18-1754. [DOI] [PubMed] [Google Scholar]
- 191.Li L, et al. The tumor suppressor UCHL1 forms a complex with p53/MDM2/ARF to promote p53 signaling and is frequently silenced in nasopharyngeal carcinoma. Clin. Cancer Res. 2010;16:2949–2958. doi: 10.1158/1078-0432.CCR-09-3178. [DOI] [PubMed] [Google Scholar]
- 192.Manning BD, Toker A. AKT/PKB signaling: navigating the network. Cell. 2017;169:381–405. doi: 10.1016/j.cell.2017.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Chan CH, et al. The Skp2-SCF E3 ligase regulates Akt ubiquitination, glycolysis, herceptin sensitivity, and tumorigenesis. Cell. 2012;149:1098–1111. doi: 10.1016/j.cell.2012.02.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Yu X, et al. Skp2-mediated ubiquitination and mitochondrial localization of Akt drive tumor growth and chemoresistance to cisplatin. Oncogene. 2019;38:7457–7472. doi: 10.1038/s41388-019-0955-7. [DOI] [PubMed] [Google Scholar]
- 195.Fang FM, et al. Effect of S-phase kinase-associated protein 2 expression on distant metastasis and survival in nasopharyngeal carcinoma patients. Int. J. Radiat. Oncol. Biol. Phys. 2009;73:202–207. doi: 10.1016/j.ijrobp.2008.04.008. [DOI] [PubMed] [Google Scholar]
- 196.Mai J, et al. Polo-like kinase 1 phosphorylates and stabilizes KLF4 to promote tumorigenesis in nasopharyngeal carcinoma. Theranostics. 2019;9:3541–3554. doi: 10.7150/thno.32908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Narayanan S, et al. Targeting the ubiquitin-proteasome pathway to overcome anti-cancer drug resistance. Drug Resist. Updat. 2020;48:100663. doi: 10.1016/j.drup.2019.100663. [DOI] [PubMed] [Google Scholar]
- 198.Yu XDQ, et al. Neoalbaconol induces cell death through necroptosis by regulating RIPK-dependent autocrine TNFà and ROS production. Oncotarget. 2015;6:1995–2008. doi: 10.18632/oncotarget.3038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 200.Gorrini C, Harris IS, Mak TW. Modulation of oxidative stress as an anticancer strategy. Nat. Rev. Drug Discov. 2013;12:931–947. doi: 10.1038/nrd4002. [DOI] [PubMed] [Google Scholar]
- 201.Bonner MY, Arbiser JL. The antioxidant paradox: what are antioxidants and how should they be used in a therapeutic context for cancer. Future Med. Chem. 2014;6:1413–1422. doi: 10.4155/fmc.14.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Hussain SP, Hofseth LJ, Harris CC. Radical causes of cancer. Nat. Rev. Cancer. 2003;3:276–285. doi: 10.1038/nrc1046. [DOI] [PubMed] [Google Scholar]
- 203.Rada B, Leto TL. Oxidative innate immune defenses by Nox/Duox family NADPH oxidases. Contrib. Microbiol. 2008;15:164–187. doi: 10.1159/000136357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Marra M, et al. Molecular targets and oxidative stress biomarkers in hepatocellular carcinoma: an overview. J. Transl. Med. 2011;9:171. doi: 10.1186/1479-5876-9-171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Mathew SS, Bryant PW, Burch AD. Accumulation of oxidized proteins in Herpesvirus infected cells. Free Radic. Biol. Med. 2010;49:383–391. doi: 10.1016/j.freeradbiomed.2010.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Ma Q, et al. A role for virally induced reactive oxygen species in Kaposi’s sarcoma herpesvirus tumorigenesis. Antioxid. Redox Signal. 2013;18:80–90. doi: 10.1089/ars.2012.4584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Ivanov AV, et al. Oxidative Stress during HIV infection: mechanisms and consequences. Oxid. Med. Cell. Longev. 2016;2016:8910396. doi: 10.1155/2016/8910396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Chua MLK, Wee JTS, Hui EP, Chan ATC. Nasopharyngeal carcinoma. Lancet. 2016;387:1012–1024. doi: 10.1016/S0140-6736(15)00055-0. [DOI] [PubMed] [Google Scholar]
- 209.Gargouri B, et al. Reactive oxygen species production and antioxidant enzyme expression after Epstein-Barr virus lytic cycle induction in Raji cell line. Biol. Trace Elem. Res. 2011;144:1449–1457. doi: 10.1007/s12011-011-9135-5. [DOI] [PubMed] [Google Scholar]
- 210.Cerimele F, et al. Reactive oxygen signaling and MAPK activation distinguish Epstein-Barr Virus (EBV)-positive versus EBV-negative Burkitt’s lymphoma. Proc. Natl Acad. Sci. USA. 2005;102:175–179. doi: 10.1073/pnas.0408381102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Lassoued S, et al. Epstein-Barr virus induces an oxidative stress during the early stages of infection in B lymphocytes, epithelial, and lymphoblastoid cell lines. Mol. Cell Biochem. 2008;313:179–186. doi: 10.1007/s11010-008-9755-z. [DOI] [PubMed] [Google Scholar]
- 212.Zhao L, et al. Chlorpyrifos induces the expression of the Epstein-Barr virus lytic cycle activator BZLF-1 via reactive oxygen species. Oxid. Med. Cell Longev. 2015;2015:309125. doi: 10.1155/2015/309125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.De Leo A, et al. Resveratrol inhibits Epstein Barr Virus lytic cycle in Burkitt’s lymphoma cells by affecting multiple molecular targets. Antivir. Res. 2012;96:196–202. doi: 10.1016/j.antiviral.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 214.Jayasurya A, et al. Inducible nitric oxide synthase and bcl-2 expression in nasopharyngeal cancer: correlation with outcome of patients after radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2003;56:837–845. doi: 10.1016/S0360-3016(03)00122-6. [DOI] [PubMed] [Google Scholar]
- 215.Segawa Y, et al. Overexpression of inducible nitric oxide synthase and accumulation of 8-OHdG in nasopharyngeal carcinoma. Histopathology. 2008;52:213–223. doi: 10.1111/j.1365-2559.2007.02920.x. [DOI] [PubMed] [Google Scholar]
- 216.Huang YJ, et al. Nitrative and oxidative DNA damage as potential survival biomarkers for nasopharyngeal carcinoma. Med. Oncol. 2011;28:377–384. doi: 10.1007/s12032-010-9434-2. [DOI] [PubMed] [Google Scholar]
- 217.Su Y, et al. [Changes of superoxide dismutase (SOD) and metallothionien (MT) before, during, and after radiotherapy for nasopharyngeal carcinoma and their significance] Ai Zheng. 2003;22:629–633. [PubMed] [Google Scholar]
- 218.Kim SM, Hur DY, Hong SW, Kim JH. EBV-encoded EBNA1 regulates cell viability by modulating miR34a-NOX2-ROS signaling in gastric cancer cells. Biochem. Biophys. Res. Commun. 2017;494:550–555. doi: 10.1016/j.bbrc.2017.10.095. [DOI] [PubMed] [Google Scholar]
- 219.Kamranvar SA, Masucci MG. The Epstein-Barr virus nuclear antigen-1 promotes telomere dysfunction via induction of oxidative stress. Leukemia. 2011;25:1017–1025. doi: 10.1038/leu.2011.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Cao JY, Mansouri S, Frappier L. Changes in the nasopharyngeal carcinoma nuclear proteome induced by the EBNA1 protein of Epstein-Barr virus reveal potential roles for EBNA1 in metastasis and oxidative stress responses. J. Virol. 2012;86:382–394. doi: 10.1128/JVI.05648-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Valavanidis A, Vlachogianni T, Fiotakis C. 8-hydroxy-2’ -deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J. Environ. Sci. Health C. Environ. Carcinog. Ecotoxicol. Rev. 2009;27:120–139. doi: 10.1080/10590500902885684. [DOI] [PubMed] [Google Scholar]
- 222.Chuma M, et al. 8-Hydroxy-2’-deoxy-guanosine is a risk factor for development of hepatocellular carcinoma in patients with chronic hepatitis C virus infection. J. Gastroenterol. Hepatol. 2008;23:1431–1436. doi: 10.1111/j.1440-1746.2008.05502.x. [DOI] [PubMed] [Google Scholar]
- 223.Kim BM, et al. Therapeutic implications for overcoming radiation resistance in cancer therapy. Int. J. Mol. Sci. 2015;16:26880–26913. doi: 10.3390/ijms161125991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Na HK, Surh YJ. Modulation of Nrf2-mediated antioxidant and detoxifying enzyme induction by the green tea polyphenol EGCG. Food Chem. Toxicol. 2008;46:1271–1278. doi: 10.1016/j.fct.2007.10.006. [DOI] [PubMed] [Google Scholar]
- 225.Mileo AM, Miccadei S. Polyphenols as modulator of oxidative stress in cancer disease: new therapeutic strategies. Oxid. Med. Cell Longev. 2016;2016:6475624. doi: 10.1155/2016/6475624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Yu W, Fu YC, Wang W. Cellular and molecular effects of resveratrol in health and disease. J. Cell Biochem. 2012;113:752–759. doi: 10.1002/jcb.23431. [DOI] [PubMed] [Google Scholar]
- 227.Kenney SC, Mertz JE. Regulation of the latent-lytic switch in Epstein-Barr virus. Semin. Cancer Biol. 2014;26:60–68. doi: 10.1016/j.semcancer.2014.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Countryman J, Miller G. Activation of expression of latent Epstein-Barr herpesvirus after gene transfer with a small cloned subfragment of heterogeneous viral DNA. Proc. Natl Acad. Sci. USA. 1985;82:4085–4089. doi: 10.1073/pnas.82.12.4085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Guo R, et al. MYC controls the epstein-barr virus lytic switch. Mol. Cell. 2020;78:653–669 e658. doi: 10.1016/j.molcel.2020.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Li Z, et al. Epstein-Barr virus ncRNA from a nasopharyngeal carcinoma induces an inflammatory response that promotes virus production. Nat. Microbiol. 2019;4:2475–2486. doi: 10.1038/s41564-019-0546-y. [DOI] [PubMed] [Google Scholar]
- 231.Munz C. Latency and lytic replication in Epstein-Barr virus-associated oncogenesis. Nat. Rev. Microbiol. 2019;17:691–700. doi: 10.1038/s41579-019-0249-7. [DOI] [PubMed] [Google Scholar]
- 232.Capone G, Fasano C, Lucchese G, Calabro M, Kanduc D. EBV-associated cancer and autoimmunity: searching for therapies. Vaccines (Basel) 2015;3:74–89. doi: 10.3390/vaccines3010074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Jog NR, et al. Association of Epstein-Barr virus serological reactivation with transitioning to systemic lupus erythematosus in at-risk individuals. Ann. Rheum. Dis. 2019;78:1235–1241. doi: 10.1136/annrheumdis-2019-215361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Balandraud N, Roudier J. Epstein-Barr virus and rheumatoid arthritis. Jt. Bone Spine. 2018;85:165–170. doi: 10.1016/j.jbspin.2017.04.011. [DOI] [PubMed] [Google Scholar]
- 235.Maslinska M. The role of Epstein-Barr virus infection in primary Sjogren’s syndrome. Curr. Opin. Rheumatol. 2019;31:475–483. doi: 10.1097/BOR.0000000000000622. [DOI] [PubMed] [Google Scholar]
- 236.Hu J, et al. The role of oxidative stress in EBV lytic reactivation, radioresistance and the potential preventive and therapeutic implications. Int. J. Cancer. 2017;141:1722–1729. doi: 10.1002/ijc.30816. [DOI] [PubMed] [Google Scholar]
- 237.Li H, et al. Epstein-Barr virus lytic reactivation regulation and its pathogenic role in carcinogenesis. Int. J. Biol. Sci. 2016;12:1309–1318. doi: 10.7150/ijbs.16564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Jia WH, Qin HD. Non-viral environmental risk factors for nasopharyngeal carcinoma: a systematic review. Semin. Cancer Biol. 2012;22:117–126. doi: 10.1016/j.semcancer.2012.01.009. [DOI] [PubMed] [Google Scholar]
- 239.Xu FH, et al. An epidemiological and molecular study of the relationship between smoking, risk of nasopharyngeal carcinoma, and Epstein-Barr virus activation. J. Natl Cancer Inst. 2012;104:1396–1410. doi: 10.1093/jnci/djs320. [DOI] [PubMed] [Google Scholar]
- 240.Coghill AE, et al. Identification of a novel, EBV-based antibody risk stratification signature for early detection of nasopharyngeal carcinoma in Taiwan. Clin. Cancer Res. 2018;24:1305–1314. doi: 10.1158/1078-0432.CCR-17-1929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Liu Y, et al. Establishment of VCA and EBNA1 IgA-based combination by enzyme-linked immunosorbent assay as preferred screening method for nasopharyngeal carcinoma: a two-stage design with a preliminary performance study and a mass screening in southern China. Int. J. Cancer. 2012;131:406–416. doi: 10.1002/ijc.26380. [DOI] [PubMed] [Google Scholar]
- 242.Ji MF, et al. Evaluation of plasma Epstein-Barr virus DNA load to distinguish nasopharyngeal carcinoma patients from healthy high-risk populations in Southern China. Cancer. 2014;120:1353–1360. doi: 10.1002/cncr.28564. [DOI] [PubMed] [Google Scholar]
- 243.Liu LT, et al. The prognostic value of plasma Epstein-Barr viral DNA and tumor response to neoadjuvant chemotherapy in advanced-stage nasopharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2015;93:862–869. doi: 10.1016/j.ijrobp.2015.08.003. [DOI] [PubMed] [Google Scholar]
- 244.Xie P, Yue JB, Fu Z, Feng R, Yu JM. Prognostic value of 18F-FDG PET/CT before and after radiotherapy for locally advanced nasopharyngeal carcinoma. Ann. Oncol. 2010;21:1078–1082. doi: 10.1093/annonc/mdp430. [DOI] [PubMed] [Google Scholar]
- 245.Zhang L, et al. Plasma Epstein-Barr viral DNA complements TNM classification of nasopharyngeal carcinoma in the era of intensity-modulated radiotherapy. Oncotarget. 2016;7:6221–6230. doi: 10.18632/oncotarget.6754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Chan KC. Plasma Epstein-Barr virus DNA as a biomarker for nasopharyngeal carcinoma. Chin. J. Cancer. 2014;33:598–603. doi: 10.5732/cjc.013.10134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Peng H, et al. Prognostic impact of Plasma Epstein-Barr virus DNA in patients with nasopharyngeal carcinoma treated using intensity-modulated radiation therapy. Sci. Rep. 2016;6:22000. doi: 10.1038/srep22000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Daigle D, et al. Valproic acid antagonizes the capacity of other histone deacetylase inhibitors to activate the Epstein-barr virus lytic cycle. J. Virol. 2011;85:5628–5643. doi: 10.1128/JVI.02659-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Gorres KL, et al. Valpromide inhibits lytic cycle reactivation of Epstein-Barr virus. mBio. 2016;7:e00113. doi: 10.1128/mBio.00113-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Verma D, Thompson J, Swaminathan S. Spironolactone blocks Epstein-Barr virus production by inhibiting EBV SM protein function. Proc. Natl Acad. Sci. USA. 2016;113:3609–3614. doi: 10.1073/pnas.1523686113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Maertens J, et al. Maribavir for Preemptive Treatment of Cytomegalovirus Reactivation. N. Engl. J. Med. 2019;381:1136–1147. doi: 10.1056/NEJMoa1714656. [DOI] [PubMed] [Google Scholar]
- 252.Wang FZ, et al. Maribavir inhibits epstein-barr virus transcription in addition to viral DNA replication. J. Virol. 2009;83:12108–12117. doi: 10.1128/JVI.01575-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Whitehurst CB, et al. Maribavir inhibits Epstein-Barr virus transcription through the EBV protein kinase. J. Virol. 2013;87:5311–5315. doi: 10.1128/JVI.03505-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Yang L, Lu Z, Ma X, Cao Y, Sun LQ. A therapeutic approach to nasopharyngeal carcinomas by DNAzymes targeting EBV LMP-1 gene. Molecules. 2010;15:6127–6139. doi: 10.3390/molecules15096127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Cao Y, et al. Therapeutic evaluation of Epstein-Barr virus-encoded latent membrane protein-1 targeted DNAzyme for treating of nasopharyngeal carcinomas. Mol. Ther. 2014;22:371–377. doi: 10.1038/mt.2013.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Hergenhahn M, et al. The chemopreventive compound curcumin is an efficient inhibitor of Epstein-Barr virus BZLF1 transcription in Raji DR-LUC cells. Mol. Carcinog. 2002;33:137–145. doi: 10.1002/mc.10029. [DOI] [PubMed] [Google Scholar]
- 257.Wu CC, et al. Inhibition of Epstein-Barr virus reactivation in nasopharyngeal carcinoma cells by dietary sulforaphane. Mol. Carcinog. 2013;52:946–958. doi: 10.1002/mc.21926. [DOI] [PubMed] [Google Scholar]
- 258.Chang LK, et al. Inhibition of Epstein-Barr virus lytic cycle by (-)-epigallocatechin gallate. Biochem. Biophys. Res. Commun. 2003;301:1062–1068. doi: 10.1016/S0006-291X(03)00067-6. [DOI] [PubMed] [Google Scholar]
- 259.Tung CP, et al. Inhibition of the Epstein-Barr virus lytic cycle by protoapigenone. J. Gen. Virol. 2011;92:1760–1768. doi: 10.1099/vir.0.031609-0. [DOI] [PubMed] [Google Scholar]
- 260.Chang FR, et al. Inhibition of the Epstein-Barr virus lytic cycle by moronic acid. Antivir. Res. 2010;85:490–495. doi: 10.1016/j.antiviral.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 261.Yiu CY, et al. Inhibition of Epstein-Barr virus lytic cycle by an ethyl acetate subfraction separated from Polygonum cuspidatum root and its major component, emodin. Molecules. 2014;19:1258–1272. doi: 10.3390/molecules19011258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Lin J-C, et al. Inhibitory effects of some derivatives of glycyrrhizic acid against Epstein-Barr virus infection: Structure–activity relationships. Antivir. Res. 2008;79:6–11. doi: 10.1016/j.antiviral.2008.01.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Mikirova N, Hunninghake R. Effect of high dose vitamin C on Epstein-Barr viral infection. Med. Sci. Monit. 2014;20:725–732. doi: 10.12659/MSM.890423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Liu S, et al. (-)-Epigallocatechin-3-gallate inhibition of Epstein-Barr virus spontaneous lytic infection involves ERK1/2 and PI3-K/Aktsignaling in EBV-positive cells. Carcinogenesis. 2013;34:627–637. doi: 10.1093/carcin/bgs364. [DOI] [PubMed] [Google Scholar]
- 265.Liu S, Li H, Tang M, Cao Y. (-)-Epigallocatechin-3-gallate inhibition of Epstein-Barr virus spontaneous lytic infection involves downregulation of latent membrane protein 1. Exp. Ther. Med. 2018;15:1105–1112. doi: 10.3892/etm.2017.5495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Birsa N, Norkett R, Higgs N, Lopez-Domenech G, Kittler JT. Mitochondrial trafficking in neurons and the role of the Miro family of GTPase proteins. Biochem. Soc. Trans. 2013;41:1525–1531. doi: 10.1042/BST20130234. [DOI] [PubMed] [Google Scholar]
- 267.Chen J, et al. Ephrin receptor A2 is a functional entry receptor for Epstein–Barr virus. Nat. Microbiol. 2018;3:172–180. doi: 10.1038/s41564-017-0081-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Peng Q, et al. EPH receptor A2 governs a feedback loop that activates Wnt/beta-catenin signaling in gastric cancer. Cell Death Dis. 2018;9:1146. doi: 10.1038/s41419-018-1164-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Zhang X, et al. Implication of viral microRNAs in the genesis and diagnosis of Epstein-Barr virus-associated tumors. Oncol. Lett. 2019;18:3433–3442. doi: 10.3892/ol.2019.10713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Wang Y, et al. BART miRNAs: an unimaginable force in the development of nasopharyngeal carcinoma. Eur. J. Cancer Prev. 2017;26:144–150. doi: 10.1097/CEJ.0000000000000221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Chen SJ, et al. Characterization of Epstein-Barr virus miRNAome in nasopharyngeal carcinoma by deep sequencing. PLoS ONE. 2010;5:e12745. doi: 10.1371/journal.pone.0012745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Zheng XH, et al. Epstein-Barr virus mir-bart1-5p detection via nasopharyngeal brush sampling is effective for diagnosing nasopharyngeal carcinoma. Oncotarget. 2016;7:4972–4980. doi: 10.18632/oncotarget.6649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Lyu X, et al. EBV-miR-BART1-5P activates AMPK/mTOR/HIF1 pathway via a PTEN independent manner to promote glycolysis and angiogenesis in nasopharyngeal carcinoma. PLoS Pathog. 2018;14:e1007484. doi: 10.1371/journal.ppat.1007484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Cai L, et al. Epstein-Barr virus-encoded microRNA BART1 induces tumour metastasis by regulating PTEN-dependent pathways in nasopharyngeal carcinoma. Nat. Commun. 2015;6:7353. doi: 10.1038/ncomms8353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Jiang C, et al. Evaluation of circulating EBV microRNA BART2-5p in facilitating early detection and screening of nasopharyngeal carcinoma. Int. J. Cancer. 2018;143:3209–3217. doi: 10.1002/ijc.31642. [DOI] [PubMed] [Google Scholar]
- 276.Zheng X, et al. Epstein-Barr virus microRNA miR-BART5-3p inhibits p53 expression. J. Virol. 2018;92:e01022-18. doi: 10.1128/JVI.01022-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.He B, et al. Epstein-Barr virus-encoded miR-BART6-3p inhibits cancer cell metastasis and invasion by targeting long non-coding RNA LOC553103. Cell Death Dis. 2016;7:e2353. doi: 10.1038/cddis.2016.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Wang D, et al. Epstein-Barr virus-encoded miR-BART6-3p inhibits cancer cell proliferation through the LOC553103-STMN1 axis. FASEB J. 2020;34:8012–8027. doi: 10.1096/fj.202000039RR. [DOI] [PubMed] [Google Scholar]
- 279.Cai LM, et al. EBV-miR-BART7-3p promotes the EMT and metastasis of nasopharyngeal carcinoma cells by suppressing the tumor suppressor PTEN. Oncogene. 2015;34:2156–2166. doi: 10.1038/onc.2014.341. [DOI] [PubMed] [Google Scholar]
- 280.Cai L, et al. EBV-miR-BART7-3p imposes stemness in nasopharyngeal carcinoma cells by suppressing SMAD7. Front. Genet. 2019;10:939. doi: 10.3389/fgene.2019.00939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Gao W, et al. Epstein-Barr virus encoded microRNA BART7 regulates radiation sensitivity of nasopharyngeal carcinoma. Oncotarget. 2017;8:20297–20308. doi: 10.18632/oncotarget.15526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Zhou X, et al. EBV encoded miRNA BART8-3p promotes radioresistance in nasopharyngeal carcinoma by regulating ATM/ATR signaling pathway. Biosci. Rep. 2019;39:BSR20190415. doi: 10.1042/BSR20190415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Ramayanti O, et al. Vesicle-bound EBV-BART13-3p miRNA in circulation distinguishes nasopharyngeal from other head and neck cancer and asymptomatic EBV-infections. Int. J. Cancer. 2019;144:2555–2566. doi: 10.1002/ijc.31967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Li Z, et al. EBV encoded miR-BHRF1-1 potentiates viral lytic replication by downregulating host p53 in nasopharyngeal carcinoma. Int. J. Biochem Cell Biol. 2012;44:275–279. doi: 10.1016/j.biocel.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 285.Verhoeven RJA, et al. Epstein-Barr virus BART long non-coding rnas function as epigenetic modulators in nasopharyngeal carcinoma. Front. Oncol. 2019;9:1120. doi: 10.3389/fonc.2019.01120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Verhoeven RJ, et al. NF-kappaB signaling regulates expression of Epstein-Barr virus BART microRNAs and long noncoding RNAs in nasopharyngeal carcinoma. J. Virol. 2016;90:6475–6488. doi: 10.1128/JVI.00613-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Zuo L, et al. Targeting exosomal EBV-LMP1 transfer and miR-203 expression via the NF-kappaB pathway: the therapeutic role of aspirin in NPC. Mol. Ther. Nucleic Acids. 2019;17:175–184. doi: 10.1016/j.omtn.2019.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Kong YG, et al. LncRNA-LINC00460 facilitates nasopharyngeal carcinoma tumorigenesis through sponging miR-149-5p to up-regulate IL6. Gene. 2018;639:77–84. doi: 10.1016/j.gene.2017.10.006. [DOI] [PubMed] [Google Scholar]
- 289.Li M, Li Y, Yu M. CircRNA ZNF609 knockdown suppresses cell growth via modulating miR-188/ELF2 axis in nasopharyngeal carcinoma. Onco Targets Ther. 2020;13:2399–2409. doi: 10.2147/OTT.S234230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Hong X, et al. Circular RNA CRIM1 functions as a ceRNA to promote nasopharyngeal carcinoma metastasis and docetaxel chemoresistance through upregulating FOXQ1. Mol. Cancer. 2020;19:33. doi: 10.1186/s12943-020-01149-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Szeto CY, et al. Integrated mRNA and microRNA transcriptome sequencing characterizes sequence variants and mRNA-microRNA regulatory network in nasopharyngeal carcinoma model systems. FEBS Open Bio. 2014;4:128–140. doi: 10.1016/j.fob.2014.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Alajez NM, et al. Enhancer of Zeste homolog 2 (EZH2) is overexpressed in recurrent nasopharyngeal carcinoma and is regulated by miR-26a, miR-101, and miR-98. Cell Death Dis. 2010;1:e85. doi: 10.1038/cddis.2010.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Lu J, et al. MiR-26a inhibits cell growth and tumorigenesis of nasopharyngeal carcinoma through repression of EZH2. Cancer Res. 2011;71:225–233. doi: 10.1158/0008-5472.CAN-10-1850. [DOI] [PubMed] [Google Scholar]
- 294.Zou X, et al. MicroRNA expression profiling analysis in serum for nasopharyngeal carcinoma diagnosis. Gene. 2020;727:144243. doi: 10.1016/j.gene.2019.144243. [DOI] [PubMed] [Google Scholar]
- 295.Xu X, et al. Dynamic changes in plasma microRNAs have potential predictive values in monitoring recurrence and metastasis of nasopharyngeal carcinoma. Biomed. Res. Int. 2018;2018:7329195. doi: 10.1155/2018/7329195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Zheng XH, et al. Plasma microRNA profiling in nasopharyngeal carcinoma patients reveals miR-548q and miR-483-5p as potential biomarkers. Chin. J. Cancer. 2014;33:330–338. doi: 10.5732/cjc.013.10246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Wang HY, et al. Profiling plasma microRNA in nasopharyngeal carcinoma with deep sequencing. Clin. Chem. 2014;60:773–782. doi: 10.1373/clinchem.2013.214213. [DOI] [PubMed] [Google Scholar]
- 298.Liu N, et al. A four-miRNA signature identified from genome-wide serum miRNA profiling predicts survival in patients with nasopharyngeal carcinoma. Int. J. Cancer. 2014;134:1359–1368. doi: 10.1002/ijc.28468. [DOI] [PubMed] [Google Scholar]
- 299.Liu N, et al. MiR-451 inhibits cell growth and invasion by targeting MIF and is associated with survival in nasopharyngeal carcinoma. Mol. Cancer. 2013;12:123. doi: 10.1186/1476-4598-12-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Zhao L, Fong AHW, Liu N, Cho WCS. Molecular subtyping of nasopharyngeal carcinoma (NPC) and a microRNA-based prognostic model for distant metastasis. J. Biomed. Sci. 2018;25:16. doi: 10.1186/s12929-018-0417-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Nishikawa J, et al. Clinical importance of Epstein(-)Barr virus-associated gastric cancer. Cancers. 2018;10:167. doi: 10.3390/cancers10060167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Shoda K, et al. Clinical utility of circulating cell-free Epstein-Barr virus DNA in patients with gastric cancer. Oncotarget. 2017;8:28796–28804. doi: 10.18632/oncotarget.15675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Zhen Y, et al. miR-374a-CCND1-pPI3K/AKT-c-JUN feedback loop modulated by PDCD4 suppresses cell growth, metastasis, and sensitizes nasopharyngeal carcinoma to cisplatin. Oncogene. 2017;36:275–285. doi: 10.1038/onc.2016.201. [DOI] [PubMed] [Google Scholar]
- 304.Deng X, et al. miR-296-3p negatively regulated by nicotine stimulates cytoplasmic translocation of c-Myc via MK2 to suppress chemotherapy resistance. Mol. Ther. 2018;26:1066–1081. doi: 10.1016/j.ymthe.2018.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Zhao M, et al. miR-3188 regulates nasopharyngeal carcinoma proliferation and chemosensitivity through a FOXO1-modulated positive feedback loop with mTOR-p-PI3K/AKT-c-JUN. Nat. Commun. 2016;7:11309. doi: 10.1038/ncomms11309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Zheng J, et al. A sequence polymorphism in miR-608 predicts recurrence after radiotherapy for nasopharyngeal carcinoma. Cancer Res. 2013;73:5151–5162. doi: 10.1158/0008-5472.CAN-13-0395. [DOI] [PubMed] [Google Scholar]
- 307.Huang T, et al. MicroRNA-19b-3p regulates nasopharyngeal carcinoma radiosensitivity by targeting TNFAIP3/NF-kappaB axis. J. Exp. Clin. Cancer Res. 2016;35:188. doi: 10.1186/s13046-016-0465-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Gao W, et al. MicroRNA-138-5p controls sensitivity of nasopharyngeal carcinoma to radiation by targeting EIF4EBP1. Oncol. Rep. 2017;37:913–920. doi: 10.3892/or.2017.5354. [DOI] [PubMed] [Google Scholar]
- 309.Tian Y, et al. miR-483-5p decreases the radiosensitivity of nasopharyngeal carcinoma cells by targeting DAPK1. Lab Invest. 2019;99:602–611. doi: 10.1038/s41374-018-0169-6. [DOI] [PubMed] [Google Scholar]
- 310.Zhao L, et al. miR-504 mediated down-regulation of nuclear respiratory factor 1 leads to radio-resistance in nasopharyngeal carcinoma. Oncotarget. 2015;6:15995–16018. doi: 10.18632/oncotarget.4138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Ma X, et al. LncRNA ANCR promotes proliferation and radiation resistance of nasopharyngeal carcinoma by inhibiting PTEN expression. Onco Targets Ther. 2018;11:8399–8408. doi: 10.2147/OTT.S182573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Chen L, Zhou H, Guan Z. CircRNA_000543 knockdown sensitizes nasopharyngeal carcinoma to irradiation by targeting miR-9/platelet-derived growth factor receptor B axis. Biochem. Biophys. Res. Commun. 2019;512:786–792. doi: 10.1016/j.bbrc.2019.03.126. [DOI] [PubMed] [Google Scholar]
- 313.Au KH, et al. Treatment outcomes of nasopharyngeal carcinoma in modern era after intensity modulated radiotherapy (IMRT) in Hong Kong: A report of 3328 patients (HKNPCSG 1301 study) Oral. Oncol. 2018;77:16–21. doi: 10.1016/j.oraloncology.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 314.Peng L, Liu JQ, Chen YP, Ma J. The next decade of clinical trials in locoregionally advanced nasopharyngeal carcinoma. Br. J. Radiol. 2019;92:20181031. doi: 10.1259/bjr.20181031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Tang LQ, et al. Concurrent chemoradiotherapy with nedaplatin versus cisplatin in stage II-IVB nasopharyngeal carcinoma: an open-label, non-inferiority, randomised phase 3 trial. Lancet Oncol. 2018;19:461–473. doi: 10.1016/S1470-2045(18)30104-9. [DOI] [PubMed] [Google Scholar]
- 316.Jiffar T, et al. KiSS1 mediates platinum sensitivity and metastasis suppression in head and neck squamous cell carcinoma. Oncogene. 2011;30:3163–3173. doi: 10.1038/onc.2011.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Shi Y, et al. Nuclear epidermal growth factor receptor interacts with transcriptional intermediary factor 2 to activate cyclin D1 gene expression triggered by the oncoprotein latent membrane protein 1. Carcinogenesis. 2012;33:1468–1478. doi: 10.1093/carcin/bgs171. [DOI] [PubMed] [Google Scholar]
- 318.Xu Y, et al. Epstein-Barr Virus encoded LMP1 regulates cyclin D1 promoter activity by nuclear EGFR and STAT3 in CNE1 cells. J. Exp. Clin. Cancer Res. 2013;32:90. doi: 10.1186/1756-9966-32-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Chan AT, et al. Multicenter, phase II study of cetuximab in combination with carboplatin in patients with recurrent or metastatic nasopharyngeal carcinoma. J. Clin. Oncol. 2005;23:3568–3576. doi: 10.1200/JCO.2005.02.147. [DOI] [PubMed] [Google Scholar]
- 320.Ma B, et al. A phase II study of patients with metastatic or locoregionally recurrent nasopharyngeal carcinoma and evaluation of plasma Epstein-Barr virus DNA as a biomarker of efficacy. Cancer Chemother. Pharm. 2008;62:59–64. doi: 10.1007/s00280-007-0575-8. [DOI] [PubMed] [Google Scholar]
- 321.Yang F, et al. Dual phosphoinositide 3-kinase/mammalian target of rapamycin inhibitor NVP-BEZ235 has a therapeutic potential and sensitizes cisplatin in nasopharyngeal carcinoma. PLoS ONE. 2013;8:e59879. doi: 10.1371/journal.pone.0059879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Ji J, et al. XIAP limits autophagic degradation of Sox2 and is a therapeutic target in nasopharyngeal carcinoma stem cells. Theranostics. 2018;8:1494–1510. doi: 10.7150/thno.21717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Liu YT, et al. Asiatic acid, extracted from Centella asiatica and induces apoptosis pathway through the phosphorylation p38 mitogen-activated protein kinase in cisplatin-resistant nasopharyngeal carcinoma cells. Biomolecules. 2020;10:184. doi: 10.3390/biom10020184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 324.Zhang Y, et al. The new 6q27 tumor suppressor DACT2, frequently silenced by CpG methylation, sensitizes nasopharyngeal cancer cells to paclitaxel and 5-FU toxicity via beta-catenin/Cdc25c signaling and G2/M arrest. Clin. Epigenetics. 2018;10:26. doi: 10.1186/s13148-018-0459-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Peng H, et al. ARNTL hypermethylation promotes tumorigenesis and inhibits cisplatin sensitivity by activating CDK5 transcription in nasopharyngeal carcinoma. J. Exp. Clin. Cancer Res. 2019;38:11. doi: 10.1186/s13046-018-0997-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Zeng Q, et al. Knockdown of NFBD1/MDC1 enhances chemosensitivity to cisplatin or 5-fluorouracil in nasopharyngeal carcinoma CNE1 cells. Mol. Cell Biochem. 2016;418:137–146. doi: 10.1007/s11010-016-2739-5. [DOI] [PubMed] [Google Scholar]
- 327.Zhou Z, et al. FOXC2 promotes chemoresistance in nasopharyngeal carcinomas via induction of epithelial mesenchymal transition. Cancer Lett. 2015;363:137–145. doi: 10.1016/j.canlet.2015.04.008. [DOI] [PubMed] [Google Scholar]
- 328.Song L, Liu H, Liu Q. Matrix metalloproteinase 1 promotes tumorigenesis and inhibits the sensitivity to 5-fluorouracil of nasopharyngeal carcinoma. Biomed. Pharmacother. 2019;118:109120. doi: 10.1016/j.biopha.2019.109120. [DOI] [PubMed] [Google Scholar]
- 329.Luo W, et al. Epstein-Barr virus latent membrane protein 1 mediates serine 25 phosphorylation and nuclear entry of annexin A2 via PI-PLC-PKCalpha/PKCbeta pathway. Mol. Carcinogenesis. 2008;47:934–946. doi: 10.1002/mc.20445. [DOI] [PubMed] [Google Scholar]
- 330.Chen CY, et al. Targeting annexin A2 reduces tumorigenesis and therapeutic resistance of nasopharyngeal carcinoma. Oncotarget. 2015;6:26946–26959. doi: 10.18632/oncotarget.4521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Kuang CM, et al. BST2 confers cisplatin resistance via NF-kappaB signaling in nasopharyngeal cancer. Cell Death Dis. 2017;8:e2874. doi: 10.1038/cddis.2017.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Gao W, et al. BEX3 contributes to cisplatin chemoresistance in nasopharyngeal carcinoma. Cancer Med. 2017;6:439–451. doi: 10.1002/cam4.982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Tang W, et al. Viral response to chemotherapy in endemic burkitt lymphoma. Clin. Cancer Res. 2010;16:2055–2064. doi: 10.1158/1078-0432.CCR-09-2424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Ritchie D, et al. Reactivation of DNA viruses in association with histone deacetylase inhibitor therapy: a case series report. Haematologica. 2009;94:1618–1622. doi: 10.3324/haematol.2009.008607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Huang CL, et al. Plasma Epstein-Barr virus DNA load after induction chemotherapy predicts outcome in locoregionally advanced nasopharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2019;104:355–361. doi: 10.1016/j.ijrobp.2019.01.007. [DOI] [PubMed] [Google Scholar]
- 336.Liu LT, et al. Neoadjuvant or adjuvant chemotherapy plus concurrent CRT versus concurrent crt alone in the treatment of nasopharyngeal carcinoma: a study based on EBV DNA. J. Natl Compr. Cancer Netw. 2019;17:703–710. doi: 10.6004/jnccn.2018.7270. [DOI] [PubMed] [Google Scholar]
- 337.Chan ATC, et al. Analysis of plasma Epstein-Barr Virus DNA in nasopharyngeal cancer after chemoradiation to identify high-risk patients for adjuvant chemotherapy: a randomized controlled trial. J. Clin. Oncol. 2018;36:3091. doi: 10.1200/JCO.2018.77.7847. [DOI] [PubMed] [Google Scholar]
- 338.An X, et al. Plasma Epstein-Barr virus DNA level strongly predicts survival in metastatic/recurrent nasopharyngeal carcinoma treated with palliative chemotherapy. Cancer. 2011;117:3750–3757. doi: 10.1002/cncr.25932. [DOI] [PubMed] [Google Scholar]
- 339.Ma B, et al. Prospective evaluation of plasma Epstein-Barr virus DNA clearance and fluorodeoxyglucose positron emission scan in assessing early response to chemotherapy in patients with advanced or recurrent nasopharyngeal carcinoma. Br. J. Cancer. 2018;118:1051–1055. doi: 10.1038/s41416-018-0026-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Guo SS, et al. The impact of the cumulative dose of cisplatin during concurrent chemoradiotherapy on the clinical outcomes of patients with advanced-stage nasopharyngeal carcinoma in an era of intensity-modulated radiotherapy. BMC Cancer. 2015;15:977. doi: 10.1186/s12885-015-1964-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Zhao Q, et al. Clinical efficacy of cisplatin, dexamethasone, gemcitabine and pegaspargase (DDGP) in the initial treatment of advanced stage (stage III-IV) extranodal NK/T-cell lymphoma, and its correlation with Epstein-Barr virus. Cancer Manag. Res. 2019;11:3555–3564. doi: 10.2147/CMAR.S191929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Zhang B, et al. ATR activated by EB virus facilitates chemotherapy resistance to cisplatin or 5-fluorouracil in human nasopharyngeal carcinoma. Cancer Manag. Res. 2019;11:573–585. doi: 10.2147/CMAR.S187099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Man CH, et al. Inhibition of NOTCH3 signalling significantly enhances sensitivity to cisplatin in EBV-associated nasopharyngeal carcinoma. J. Pathol. 2012;226:471–481. doi: 10.1002/path.2997. [DOI] [PubMed] [Google Scholar]
- 344.Li SS, et al. Latent membrane protein 1 mediates the resistance of nasopharyngeal carcinoma cells to TRAIL-induced apoptosis by activation of the PI3K/Akt signaling pathway. Oncol. Rep. 2011;26:1573–1579. doi: 10.3892/or.2011.1423. [DOI] [PubMed] [Google Scholar]
- 345.Lin X, et al. Epstein-Barr virus-encoded LMP1 triggers regulation of the ERK-mediated Op18/stathmin signaling pathway in association with cell cycle. Cancer Sci. 2012;103:993–999. doi: 10.1111/j.1349-7006.2012.02271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Wu Y, et al. A combination of paclitaxel and siRNA-mediated silencing of Stathmin inhibits growth and promotes apoptosis of nasopharyngeal carcinoma cells. Cell. Oncol. (Dordr.) 2014;37:53–67. doi: 10.1007/s13402-013-0163-3. [DOI] [PubMed] [Google Scholar]
- 347.Liu Y, et al. Cinobufotalin powerfully reversed EBV-miR-BART22-induced cisplatin resistance via stimulating MAP2K4 to antagonize non-muscle myosin heavy chain IIA/glycogen synthase 3beta/beta-catenin signaling pathway. EBioMedicine. 2019;48:386–404. doi: 10.1016/j.ebiom.2019.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Dayal R, Singh A, Pandey A, Mishra KP. Reactive oxygen species as mediator of tumor radiosensitivity. J. Cancer Res Ther. 2014;10:811–818. doi: 10.4103/0973-1482.146073. [DOI] [PubMed] [Google Scholar]
- 349.Blyth BJ, Sykes PJ. Radiation-induced bystander effects: what are they, and how relevant are they to human radiation exposures? Radiat. Res. 2011;176:139–157. doi: 10.1667/RR2548.1. [DOI] [PubMed] [Google Scholar]
- 350.Chen W, Hu GH. Biomarkers for enhancing the radiosensitivity of nasopharyngeal carcinoma. Cancer Biol. Med. 2015;12:23–32. doi: 10.7497/j.issn.2095-3941.2014.0015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Yang L, et al. EBV-LMP1 targeted DNAzyme enhances radiosensitivity by inhibiting tumor angiogenesis via the JNKs/HIF-1 pathway in nasopharyngeal carcinoma. Oncotarget. 2015;6:5804–5817. doi: 10.18632/oncotarget.3331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Ma X, et al. Down-regulation of EBV-LMP1 radio-sensitizes nasal pharyngeal carcinoma cells via NF-kappaB regulated ATM expression. PLoS ONE. 2011;6:e24647. doi: 10.1371/journal.pone.0024647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Yang L, et al. Targeting EBV-LMP1 DNAzyme enhances radiosensitivity of nasopharyngeal carcinoma cells by inhibiting telomerase activity. Cancer Biol. Ther. 2014;15:61–68. doi: 10.4161/cbt.26606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Lu ZX, et al. DNAzymes targeted to EBV-encoded latent membrane protein-1 induce apoptosis and enhance radiosensitivity in nasopharyngeal carcinoma. Cancer Lett. 2008;265:226–238. doi: 10.1016/j.canlet.2008.02.019. [DOI] [PubMed] [Google Scholar]
- 355.Lu ZX, et al. Effect of EBV LMP1 targeted DNAzymes on cell proliferation and apoptosis. Cancer Gene Ther. 2005;12:647–654. doi: 10.1038/sj.cgt.7700833. [DOI] [PubMed] [Google Scholar]
- 356.Wang Y, Zhou Y, Graves DT. FOXO transcription factors: their clinical significance and regulation. Biomed. Res. Int. 2014;2014:925350. doi: 10.1155/2014/925350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.van der Vos KE, Coffer PJ. The extending network of FOXO transcriptional target genes. Antioxid. Redox Signal. 2011;14:579–592. doi: 10.1089/ars.2010.3419. [DOI] [PubMed] [Google Scholar]
- 358.Luo M, et al. FOXO3a knockdown promotes radioresistance in nasopharyngeal carcinoma by inducing epithelial-mesenchymal transition and the Wnt/beta-catenin signaling pathway. Cancer Lett. 2019;455:26–35. doi: 10.1016/j.canlet.2019.04.019. [DOI] [PubMed] [Google Scholar]
- 359.Guo Q, et al. Genetic variations in the PI3K-PTEN-AKT-mTOR pathway are associated with distant metastasis in nasopharyngeal carcinoma patients treated with intensity-modulated radiation therapy. Sci. Rep. 2016;6:37576. doi: 10.1038/srep37576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Zhang G, et al. Radiation-resistant cancer stem-like cell properties are regulated by PTEN through the activity of nuclear beta-catenin in nasopharyngeal carcinoma. Oncotarget. 2017;8:74661–74672. doi: 10.18632/oncotarget.20339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Shen ED, Zeng Q. Inhibition of the Numb/Notch signaling pathway increases radiation sensitivity in human nasopharyngeal carcinoma cells. Kaohsiung J. Med. Sci. 2019;35:474–485. doi: 10.1002/kjm2.12087. [DOI] [PubMed] [Google Scholar]
- 362.Chow JC, Ngan RK, Cheung KM, Cho WC. Immunotherapeutic approaches in nasopharyngeal carcinoma. Expert Opin. Biol. Ther. 2019;19:1165–1172. doi: 10.1080/14712598.2019.1650910. [DOI] [PubMed] [Google Scholar]
- 363.Le QT, et al. Current treatment landscape of nasopharyngeal carcinoma and potential trials evaluating the value of immunotherapy. J. Natl Cancer Inst. 2019;111:655–663. doi: 10.1093/jnci/djz044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Fukayama M, et al. Thirty years of Epstein-Barr virus-associated gastric carcinoma. Virchows Arch. Int. J. Pathol. 2020;476:353–365. doi: 10.1007/s00428-019-02724-4. [DOI] [PubMed] [Google Scholar]
- 365.Cohen JI, Fauci AS, Varmus H, Nabel GJ. Epstein-Barr virus: an important vaccine target for cancer prevention. Sci. Transl. Med. 2011;3:107fs107–107fs107. doi: 10.1126/scitranslmed.3002878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Moutschen M, et al. Phase I/II studies to evaluate safety and immunogenicity of a recombinant gp350 Epstein-Barr virus vaccine in healthy adults. Vaccine. 2007;25:4697–4705. doi: 10.1016/j.vaccine.2007.04.008. [DOI] [PubMed] [Google Scholar]
- 367.Sokal EM, et al. Recombinant gp350 vaccine for infectious mononucleosis: a phase 2, randomized, double-blind, placebo-controlled trial to evaluate the safety, immunogenicity, and efficacy of an Epstein-Barr virus vaccine in healthy young adults. J. Infect. Dis. 2007;196:1749–1753. doi: 10.1086/523813. [DOI] [PubMed] [Google Scholar]
- 368.Coghill AE, et al. High levels of antibody that neutralize B-cell infection of Epstein-Barr virus and that bind EBV gp350 are associated with a lower risk of nasopharyngeal carcinoma. Clin. Cancer Res. 2016;22:3451–3457. doi: 10.1158/1078-0432.CCR-15-2299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Dasari V, Sinha D, Neller MA, Smith C, Khanna R. Prophylactic and therapeutic strategies for Epstein-Barr virus-associated diseases: emerging strategies for clinical development. Expert Rev. Vaccines. 2019;18:457–474. doi: 10.1080/14760584.2019.1605906. [DOI] [PubMed] [Google Scholar]
- 370.Wojtak K, Perales-Puchalt A, Weiner DB. Novel synthetic DNA immunogens targeting latent expressed antigens of Epstein-Barr virus elicit potent cellular responses and inhibit tumor growth. Vaccines (Basel) 2019;7:44. doi: 10.3390/vaccines7020044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 371.Si Y, et al. The safety and immunological effects of rAd5-EBV-LMP2 vaccine in nasopharyngeal carcinoma patients: a phase I clinical trial and two-year follow-up. Chem. Pharm. Bull. (Tokyo) 2016;64:1118–1123. doi: 10.1248/cpb.c16-00114. [DOI] [PubMed] [Google Scholar]
- 372.Chia WK, et al. A phase II study evaluating the safety and efficacy of an adenovirus-ΔLMP1-LMP2 transduced dendritic cell vaccine in patients with advanced metastatic nasopharyngeal carcinoma. Ann. Oncol. 2012;23:997–1005. doi: 10.1093/annonc/mdr341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Taylor GS, et al. Dual stimulation of Epstein-Barr Virus (EBV)-specific CD4+- and CD8+-T-cell responses by a chimeric antigen construct: potential therapeutic vaccine for EBV-positive nasopharyngeal carcinoma. J. Virol. 2004;78:768–778. doi: 10.1128/JVI.78.2.768-778.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.Hui EP, et al. Phase I trial of recombinant modified vaccinia ankara encoding Epstein-Barr viral tumor antigens in nasopharyngeal carcinoma patients. Cancer Res. 2013;73:1676–1688. doi: 10.1158/0008-5472.CAN-12-2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Taylor GS, et al. A recombinant modified vaccinia ankara vaccine encoding Epstein-Barr Virus (EBV) target antigens: a phase I trial in UK patients with EBV-positive cancer. Clin. Cancer Res. 2014;20:5009–5022. doi: 10.1158/1078-0432.CCR-14-1122-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Sullivan NL, et al. Understanding the immunology of the Zostavax shingles vaccine. Curr. Opin. Immunol. 2019;59:25–30. doi: 10.1016/j.coi.2019.02.005. [DOI] [PubMed] [Google Scholar]
- 377.Rühl J, et al. Heterologous prime-boost vaccination protects against EBV antigen-expressing lymphomas. J. Clin. Investig. 2019;129:2071–2087. doi: 10.1172/JCI125364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Bu W, et al. Immunization with components of the viral fusion apparatus elicits antibodies that neutralize epstein-barr virus in B cells and epithelial cells. Immunity. 2019;50:1305–1316.e1306. doi: 10.1016/j.immuni.2019.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Kanekiyo M, et al. Rational design of an Epstein-Barr virus vaccine targeting the receptor-binding site. Cell. 2015;162:1090–1100. doi: 10.1016/j.cell.2015.07.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Houot R, Schultz LM, Marabelle A, Kohrt H. T-cell-based Immunotherapy: Adoptive Cell Transfer and Checkpoint Inhibition. Cancer Immunol. Res. 2015;3:1115–1122. doi: 10.1158/2326-6066.CIR-15-0190. [DOI] [PubMed] [Google Scholar]
- 381.Louis CU, et al. Enhancing the in vivo expansion of adoptively transferred EBV-specific CTL with lymphodepleting CD45 monoclonal antibodies in NPC patients. Blood. 2009;113:2442–2450. doi: 10.1182/blood-2008-05-157222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.Secondino S, et al. T-cell therapy for EBV-associated nasopharyngeal carcinoma: preparative lymphodepleting chemotherapy does not improve clinical results. Ann. Oncol. 2012;23:435–441. doi: 10.1093/annonc/mdr134. [DOI] [PubMed] [Google Scholar]
- 383.Chia W-K, et al. Adoptive T-cell transfer and chemotherapy in the first-line treatment of metastatic and/or locally recurrent nasopharyngeal carcinoma. Mol. Ther. 2014;22:132–139. doi: 10.1038/mt.2013.242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Peiris-Pagès M, Martinez-Outschoorn UE, Pestell RG, Sotgia F, Lisanti MP. Cancer stem cell metabolism. Breast Cancer Res. BCR. 2016;18:55. doi: 10.1186/s13058-016-0712-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Bollard CM, et al. Sustained complete responses in patients with lymphoma receiving autologous cytotoxic T lymphocytes targeting Epstein-Barr virus latent membrane proteins. J. Clin. Oncol. 2014;32:798–808. doi: 10.1200/JCO.2013.51.5304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Lanier LL. Up on the tightrope: natural killer cell activation and inhibition. Nat. Immunol. 2008;9:495–502. doi: 10.1038/ni1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Ames E, Murphy WJ. Advantages and clinical applications of natural killer cells in cancer immunotherapy. Cancer Immunol. Immunother. 2014;63:21–28. doi: 10.1007/s00262-013-1469-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Kim N, et al. Natural killer cells as a promising therapeutic target for cancer immunotherapy. Arch. Pharm. Res. 2019;42:591–606. doi: 10.1007/s12272-019-01143-y. [DOI] [PubMed] [Google Scholar]
- 389.McKenna DH, Jr., et al. Good manufacturing practices production of natural killer cells for immunotherapy: a six-year single-institution experience. Transfusion. 2007;47:520–528. doi: 10.1111/j.1537-2995.2006.01145.x. [DOI] [PubMed] [Google Scholar]
- 390.Lee AZE, Tan LSY, Lim CM. Cellular-based immunotherapy in Epstein-Barr virus induced nasopharyngeal cancer. Oral. Oncol. 2018;84:61–70. doi: 10.1016/j.oraloncology.2018.07.011. [DOI] [PubMed] [Google Scholar]
- 391.Rezvani K, Rouce R, Liu E, Shpall E. Engineering natural killer cells for cancer immunotherapy. Mol. Ther. 2017;25:1769–1781. doi: 10.1016/j.ymthe.2017.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Ansell SM, et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N. Engl. J. Med. 2015;372:311–319. doi: 10.1056/NEJMoa1411087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Xie M, Huang X, Ye X, Qian W. Prognostic and clinicopathological significance of PD-1/PD-L1 expression in the tumor microenvironment and neoplastic cells for lymphoma. Int. Immunopharmacol. 2019;77:105999. doi: 10.1016/j.intimp.2019.105999. [DOI] [PubMed] [Google Scholar]
- 394.Zhu Q, et al. Tumor cells PD-L1 expression as a favorable prognosis factor in nasopharyngeal carcinoma patients with pre-existing intratumor-infiltrating lymphocytes. Oncoimmunology. 2017;6:e1312240–e1312240. doi: 10.1080/2162402X.2017.1312240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Larbcharoensub N, et al. Characterization of PD-L1 and PD-1 expression and CD8+ tumor-infiltrating lymphocyte in epstein-barr virus-associated nasopharyngeal carcinoma. Am. J. Clin. Oncol. 2018;41:1204–1210. doi: 10.1097/COC.0000000000000449. [DOI] [PubMed] [Google Scholar]
- 396.Ono T, et al. Prognostic stratification of patients with nasopharyngeal carcinoma based on tumor immune microenvironment. Head. Neck. 2018;40:2007–2019. doi: 10.1002/hed.25189. [DOI] [PubMed] [Google Scholar]
- 397.Seiwert TY, et al. Safety and clinical activity of pembrolizumab for treatment of recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-012): an open-label, multicentre, phase 1b trial. Lancet Oncol. 2016;17:956–965. doi: 10.1016/S1470-2045(16)30066-3. [DOI] [PubMed] [Google Scholar]
- 398.Kim ST, et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat. Med. 2018;24:1449–1458. doi: 10.1038/s41591-018-0101-z. [DOI] [PubMed] [Google Scholar]
- 399.Ma BBY, et al. Antitumor activity of nivolumab in recurrent and metastatic nasopharyngeal carcinoma: An International, Multicenter Study of the Mayo Clinic Phase 2 Consortium (NCI-9742) J. Clin. Oncol. 2018;36:1412–1418. doi: 10.1200/JCO.2017.77.0388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Xu-Monette ZY, Zhou J, Young KH. PD-1 expression and clinical PD-1 blockade in B-cell lymphomas. Blood. 2018;131:68–83. doi: 10.1182/blood-2017-07-740993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Lin P, et al. Prognostic utility of (18)F-FDG PET-CT performed prior to and during primary radiotherapy for nasopharyngeal carcinoma: index node is a useful prognostic imaging biomarker site. Radiother. Oncol. 2016;120:87–91. doi: 10.1016/j.radonc.2016.05.021. [DOI] [PubMed] [Google Scholar]
- 402.Tao C-J, et al. Multi-subject atlas-based auto-segmentation reduces interobserver variation and improves dosimetric parameter consistency for organs at risk in nasopharyngeal carcinoma: a multi-institution clinical study. Radiother. Oncol. 2015;115:407–411. doi: 10.1016/j.radonc.2015.05.012. [DOI] [PubMed] [Google Scholar]
- 403.Fang W, et al. Camrelizumab (SHR-1210) alone or in combination with gemcitabine plus cisplatin for nasopharyngeal carcinoma: results from two single-arm, phase 1 trials. Lancet Oncol. 2018;19:1338–1350. doi: 10.1016/S1470-2045(18)30495-9. [DOI] [PubMed] [Google Scholar]
- 404.Maślińska M. The role of Epstein-Barr virus infection in primary Sjögren’s syndrome. Curr. Opin. Rheumatol. 2019;31:475–483. doi: 10.1097/BOR.0000000000000622. [DOI] [PubMed] [Google Scholar]
- 405.Tengvall K, et al. Molecular mimicry between Anoctamin 2 and Epstein-Barr virus nuclear antigen 1 associates with multiple sclerosis risk. Proc. Natl Acad. Sci. USA. 2019;116:16955–16960. doi: 10.1073/pnas.1902623116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Larsen M, et al. Exhausted cytotoxic control of Epstein-Barr virus in human lupus. PLoS Pathog. 2011;7:e1002328. doi: 10.1371/journal.ppat.1002328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Zandman-Goddard G, et al. Exposure to Epstein-Barr virus infection is associated with mild systemic lupus erythematosus disease. Ann. N.Y. Acad. Sci. 2009;1173:658–663. doi: 10.1111/j.1749-6632.2009.04754.x. [DOI] [PubMed] [Google Scholar]
- 408.Esen BA, et al. Serologic response to Epstein-Barr virus antigens in patients with systemic lupus erythematosus: a controlled study. Rheumatol. Int. 2012;32:79–83. doi: 10.1007/s00296-010-1573-4. [DOI] [PubMed] [Google Scholar]
- 409.Kanegane H, Wakiguchi H, Kanegane C, Kurashige T, Tosato G. Viral interleukin-10 in chronic active Epstein-Barr virus infection. J. Infect. Dis. 1997;176:254–257. doi: 10.1086/517260. [DOI] [PubMed] [Google Scholar]
- 410.Adler B, et al. Control of Epstein-Barr virus reactivation by activated CD40 and viral latent membrane protein 1. Proc. Natl Acad. Sci. USA. 2002;99:437–442. doi: 10.1073/pnas.221439999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Babcock GJ, Hochberg D, Thorley-Lawson AD. The expression pattern of Epstein-Barr virus latent genes in vivo is dependent upon the differentiation stage of the infected B cell. Immunity. 2000;13:497–506. doi: 10.1016/S1074-7613(00)00049-2. [DOI] [PubMed] [Google Scholar]
- 412.Winchester R, Dwyer E, Rose S. The genetic basis of rheumatoid arthritis. The shared epitope hypothesis. Rheum. Dis. Clin. North Am. 1992;18:761–783. [PubMed] [Google Scholar]
- 413.Roudier J, Petersen J, Rhodes GH, Luka J, Carson DA. Susceptibility to rheumatoid arthritis maps to a T-cell epitope shared by the HLA-Dw4 DR beta-1 chain and the Epstein-Barr virus glycoprotein gp110. Proc. Natl Acad. Sci. USA. 1989;86:5104–5108. doi: 10.1073/pnas.86.13.5104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Tan LC, et al. Specificity of T cells in synovial fluid: high frequencies of CD8(+) T cells that are specific for certain viral epitopes. Arthritis Res. 2000;2:154–164. doi: 10.1186/ar80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Kang I, et al. Defective control of latent Epstein-Barr virus infection in systemic lupus erythematosus. J. Immunol. (Baltim., Md. : 1950) 2004;172:1287–1294. doi: 10.4049/jimmunol.172.2.1287. [DOI] [PubMed] [Google Scholar]
- 416.Berner BR, et al. Phenotypic and functional analysis of EBV-specific memory CD8 cells in SLE. Cell. Immunol. 2005;235:29–38. doi: 10.1016/j.cellimm.2005.06.010. [DOI] [PubMed] [Google Scholar]
- 417.Pender MP, et al. Epstein-Barr virus-specific T cell therapy for progressive multiple sclerosis. JCI Insight. 2018;3:e124714. doi: 10.1172/jci.insight.124714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Thompson MP, Kurzrock R. Epstein-Barr virus and cancer. Clin. Cancer Res. 2004;10:803–821. doi: 10.1158/1078-0432.CCR-0670-3. [DOI] [PubMed] [Google Scholar]
- 419.Gullo I, et al. New insights into the inflamed tumor immune microenvironment of gastric cancer with lymphoid stroma: from morphology and digital analysis to gene expression. Gastric Cancer. 2019;22:77–90. doi: 10.1007/s10120-018-0836-8. [DOI] [PubMed] [Google Scholar]
- 420.Kang HJ, et al. Immunogenomic landscape of hepatocellular carcinoma with immune cell stroma and EBV-positive tumor-infiltrating lymphocytes. J. Hepatol. 2019;71:91–103. doi: 10.1016/j.jhep.2019.03.018. [DOI] [PubMed] [Google Scholar]
- 421.Fiches GN, et al. Profiling of immune related genes silenced in EBV-positive gastric carcinoma identified novel restriction factors of human gammaherpesviruses. PLoS Pathog. 2020;16:e1008778. doi: 10.1371/journal.ppat.1008778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Panda A, et al. Immune activation and benefit from avelumab in EBV-positive gastric cancer. J. Natl Cancer Inst. 2018;110:316–320. doi: 10.1093/jnci/djx213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Kim TS, da Silva E, Coit DG, Tang LH. Intratumoral immune response to gastric cancer varies by molecular and histologic subtype. Am. J. Surgical Pathol. 2019;43:851–860. doi: 10.1097/PAS.0000000000001253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Gourzones C, Barjon C, Busson P. Host-tumor interactions in nasopharyngeal carcinomas. Semin. Cancer Biol. 2012;22:127–136. doi: 10.1016/j.semcancer.2012.01.002. [DOI] [PubMed] [Google Scholar]
- 425.Chang KP, et al. Multiplexed immunobead-based profiling of cytokine markers for detection of nasopharyngeal carcinoma and prognosis of patient survival. Head. Neck. 2011;33:886–897. doi: 10.1002/hed.21557. [DOI] [PubMed] [Google Scholar]
- 426.Beck A, et al. Expression of cytokine and chemokine genes in Epstein-Barr virus-associated nasopharyngeal carcinoma: comparison with Hodgkin’s disease. J. Pathol. 2001;194:145–151. doi: 10.1002/path.867. [DOI] [PubMed] [Google Scholar]
- 427.Li J, et al. Functional inactivation of EBV-specific T-lymphocytes in nasopharyngeal carcinoma: implications for tumor immunotherapy. PLoS ONE. 2007;2:e1122. doi: 10.1371/journal.pone.0001122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 428.Hsu M, et al. Epstein-Barr virus lytic transactivator Zta enhances chemotactic activity through induction of interleukin-8 in nasopharyngeal carcinoma cells. J. Virol. 2008;82:3679–3688. doi: 10.1128/JVI.02301-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Weissmann G, Smolen JE, Korchak HM. Release of inflammatory mediators from stimulated neutrophils. N. Engl. J. Med. 1980;303:27–34. doi: 10.1056/NEJM198007033030109. [DOI] [PubMed] [Google Scholar]
- 430.Yoshimura T. Discovery of IL-8/CXCL8 (The Story from Frederick) Front. Immunol. 2015;6:278. doi: 10.3389/fimmu.2015.00278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Lee CH, et al. Epstein-Barr virus Zta-induced immunomodulators from nasopharyngeal carcinoma cells upregulate interleukin-10 production from monocytes. J. Virol. 2011;85:7333–7342. doi: 10.1128/JVI.00182-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 432.Lippitz BE. Cytokine patterns in patients with cancer: a systematic review. Lancet Oncol. 2013;14:e218–e228. doi: 10.1016/S1470-2045(12)70582-X. [DOI] [PubMed] [Google Scholar]
- 433.Xu J, et al. Tyrosylprotein sulfotransferase-1 and tyrosine sulfation of chemokine receptor 4 are induced by Epstein-Barr virus encoded latent membrane protein 1 and associated with the metastatic potential of human nasopharyngeal carcinoma. PLoS ONE. 2013;8:e56114. doi: 10.1371/journal.pone.0056114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 434.Hu J, et al. The expression of functional chemokine receptor CXCR4 is associated with the metastatic potential of human nasopharyngeal carcinoma. Clin. Cancer Res. 2005;11:4658–4665. doi: 10.1158/1078-0432.CCR-04-1798. [DOI] [PubMed] [Google Scholar]
- 435.Miyauchi K, Urano E, Yoshiyama H, Komano J. Cytokine signatures of transformed B cells with distinct Epstein-Barr virus latencies as a potential diagnostic tool for B cell lymphoma. Cancer Sci. 2011;102:1236–1241. doi: 10.1111/j.1349-7006.2011.01924.x. [DOI] [PubMed] [Google Scholar]
- 436.Incrocci R, McCormack M, Swanson-Mungerson M. Epstein-Barr virus LMP2A increases IL-10 production in mitogen-stimulated primary B-cells and B-cell lymphomas. J. Gen. Virol. 2013;94:1127–1133. doi: 10.1099/vir.0.049221-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Dolcetti R. Cross-talk between Epstein-Barr virus and microenvironment in the pathogenesis of lymphomas. Semin. Cancer Biol. 2015;34:58–69. doi: 10.1016/j.semcancer.2015.04.006. [DOI] [PubMed] [Google Scholar]
- 438.Baumforth KR, et al. Expression of the Epstein-Barr virus-encoded Epstein-Barr virus nuclear antigen 1 in Hodgkin’s lymphoma cells mediates Up-regulation of CCL20 and the migration of regulatory T cells. Am. J. Pathol. 2008;173:195–204. doi: 10.2353/ajpath.2008.070845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Loyo M, et al. A survey of methylated candidate tumor suppressor genes in nasopharyngeal carcinoma. Int. J. Cancer. 2011;128:1393–1403. doi: 10.1002/ijc.25443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 440.Zhou L, et al. Frequent hypermethylation of RASSF1A and TSLC1, and high viral load of Epstein-Barr Virus DNA in nasopharyngeal carcinoma and matched tumor-adjacent tissues. Neoplasia. 2005;7:809–815. doi: 10.1593/neo.05217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Niemhom S, Kitazawa S, Kitazawa R, Maeda S, Leopairat J. Hypermethylation of epithelial-cadherin gene promoter is associated with Epstein-Barr virus in nasopharyngeal carcinoma. Cancer Detection Prev. 2008;32:127–134. doi: 10.1016/j.cdp.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 442.Tong JH, et al. Putative tumour-suppressor gene DAB2 is frequently down regulated by promoter hypermethylation in nasopharyngeal carcinoma. BMC Cancer. 2010;10:253. doi: 10.1186/1471-2407-10-253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Yuen JW, et al. Epigenetic inactivation of inositol polyphosphate 4-phosphatase B (INPP4B), a regulator of PI3K/AKT signaling pathway in EBV-associated nasopharyngeal carcinoma. PLoS ONE. 2014;9:e105163. doi: 10.1371/journal.pone.0105163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 444.Nawaz I, et al. Integrin alpha9 gene promoter is hypermethylated and downregulated in nasopharyngeal carcinoma. Oncotarget. 2015;6:31493–31507. doi: 10.18632/oncotarget.5154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Lee HM, et al. Monoamine oxidase A is down-regulated in EBV-associated nasopharyngeal carcinoma. Sci. Rep. 2020;10:6115. doi: 10.1038/s41598-020-63150-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 446.Cheung CC, et al. miR-31 is consistently inactivated in EBV-associated nasopharyngeal carcinoma and contributes to its tumorigenesis. Mol. Cancer. 2014;13:184. doi: 10.1186/1476-4598-13-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Peng H, et al. Higher methylation intensity induced by EBV LMP1 via NF-kappaB/DNMT3b signaling contributes to silencing of PTEN gene. Oncotarget. 2016;7:40025–40037. doi: 10.18632/oncotarget.9474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Mo Y, et al. Promoter hypermethylation of Ras-related GTPase gene RRAD inactivates a tumor suppressor function in nasopharyngeal carcinoma. Cancer Lett. 2012;323:147–154. doi: 10.1016/j.canlet.2012.03.042. [DOI] [PubMed] [Google Scholar]
- 449.Wang J, et al. LMP2A induces DNA methylation and expression repression of AQP3 in EBV-associated gastric carcinoma. Virology. 2019;534:87–95. doi: 10.1016/j.virol.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 450.Kawamura YI, et al. DNA hypermethylation contributes to incomplete synthesis of carbohydrate determinants in gastrointestinal cancer. Gastroenterology. 2008;135:142–151.e143. doi: 10.1053/j.gastro.2008.03.031. [DOI] [PubMed] [Google Scholar]
- 451.Chang MS, et al. CpG island methylation status in gastric carcinoma with and without infection of Epstein-Barr virus. Clin. Cancer Res. 2006;12:2995–3002. doi: 10.1158/1078-0432.CCR-05-1601. [DOI] [PubMed] [Google Scholar]
- 452.Saito M, et al. Role of DNA methylation in the development of Epstein-Barr virus-associated gastric carcinoma. J. Med. Virol. 2013;85:121–127. doi: 10.1002/jmv.23405. [DOI] [PubMed] [Google Scholar]
- 453.Ghadami E, et al. Epigenetic alterations of CYLD promoter modulate its expression in gastric adenocarcinoma: A footprint of infections. J. Cell. Physiol. 2019;234:4115–4124. doi: 10.1002/jcp.27220. [DOI] [PubMed] [Google Scholar]
- 454.Dong SM, et al. Hypermethylation of the interferon regulatory factor 5 promoter in Epstein-Barr virus-associated gastric carcinoma. J. Microbiol. 2015;53:70–76. doi: 10.1007/s12275-014-4654-3. [DOI] [PubMed] [Google Scholar]
- 455.Okada T, et al. Identification of genes specifically methylated in Epstein-Barr virus-associated gastric carcinomas. Cancer Sci. 2013;104:1309–1314. doi: 10.1111/cas.12228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Hino R, et al. Activation of DNA methyltransferase 1 by EBV latent membrane protein 2A leads to promoter hypermethylation of PTEN gene in gastric carcinoma. Cancer Res. 2009;69:2766–2774. doi: 10.1158/0008-5472.CAN-08-3070. [DOI] [PubMed] [Google Scholar]
- 457.Yu J, et al. REC8 functions as a tumor suppressor and is epigenetically downregulated in gastric cancer, especially in EBV-positive subtype. Oncogene. 2017;36:182–193. doi: 10.1038/onc.2016.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 458.Sun M, et al. SOX9 expression and its methylation status in gastric cancer. Virchows Arch. : Int. J. Pathol. 2012;460:271–279. doi: 10.1007/s00428-012-1201-7. [DOI] [PubMed] [Google Scholar]
- 459.Zhao J, et al. Somatostatin receptor 1, a novel EBV-associated CpG hypermethylated gene, contributes to the pathogenesis of EBV-associated gastric cancer. Br. J. Cancer. 2013;108:2557–2564. doi: 10.1038/bjc.2013.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Liu X, et al. Epigenetic silencing of WNT5A in Epstein-Barr virus-associated gastric carcinoma. Arch. Virol. 2013;158:123–132. doi: 10.1007/s00705-012-1481-x. [DOI] [PubMed] [Google Scholar]
- 461.Paschos K, et al. Epstein-barr virus latency in B cells leads to epigenetic repression and CpG methylation of the tumour suppressor gene Bim. PLoS Pathog. 2009;5:e1000492. doi: 10.1371/journal.ppat.1000492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 462.Kim NR, Lin Z, Kim KR, Cho HY, Kim I. Epstein-Barr virus and p16INK4A methylation in squamous cell carcinoma and precancerous lesions of the cervix uteri. J. Korean Med. Sci. 2005;20:636–642. doi: 10.3346/jkms.2005.20.4.636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 463.Burassakarn A, et al. Aberrant gene promoter methylation of E-cadherin, p16 (INK4a), p14 (ARF), and MGMT in Epstein-Barr virus-associated oral squamous cell carcinomas. Med. Oncol. 2017;34:128. doi: 10.1007/s12032-017-0983-5. [DOI] [PubMed] [Google Scholar]
- 464.Sako N, Dessirier V, Bagot M, Bensussan A, Schmitt C. HACE1, a potential tumor suppressor gene on 6q21, is not involved in extranodal natural killer/T-cell lymphoma pathophysiology. Am. J. Pathol. 2014;184:2899–2907. doi: 10.1016/j.ajpath.2014.07.011. [DOI] [PubMed] [Google Scholar]
- 465.Zhang T, et al. Hypermethylation of the tumor suppressor gene PRDM1/Blimp-1 supports a pathogenetic role in EBV-positive Burkitt lymphoma. Blood Cancer J. 2014;4:e261. doi: 10.1038/bcj.2014.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 466.Li F, et al. Delayed-type hypersensitivity (DTH) immune response related with EBV-DNA in nasopharyngeal carcinoma treated with autologous dendritic cell vaccination after radiotherapy. J. Immunother. 2013;36:208–214. doi: 10.1097/CJI.0b013e31828bd87b. [DOI] [PubMed] [Google Scholar]