Abstract
The upper aerodigestive tract (UAT) is the first line of defense against environmental stresses such as antigens, microbes, inhalants, foods, etc., and mucins, intracellular junctions, epithelial cells, and immune cells are the major constituents of this defensive mucosal barrier. Laryngopharyngeal reflux (LPR) is recognized as an independent risk factor for UAT mucosal disorders, and in this review, we describe the components and functions of the mucosal barrier and the results of LPR-induced mucosal inflammation in the UAT. We discuss the interactions between the refluxate and the mucosal components and the mechanisms through which these damaging events disrupt and alter the mucosal barriers. In addition, we discuss the dynamic alterations in the mucosal barrier that might be potential therapeutic targets for LPR-induced disorders.
Keywords: laryngopharyngeal reflux, LPR, upper aerodigestive tract, UAT, inflammatory response, mucosal barrier dysfunction
Introduction
As the junction of the respiratory system and digestive system, the upper aerodigestive tract (UAT) mucosa functions as the first line of defense against environmental stresses such as microbes, foreign antigens, tobacco smoke, pollutants, and laryngopharyngeal reflux (LPR). The UAT mucosal barrier is composed primarily of mucus, intracellular tight junctions of the epithelia, and the mucosal immune system. Increasing evidence indicates that mucosal barrier dysfunction may induce mucosal inflammation, including inflammation of the gastrointestinal tract (e.g. inflammatory bowel disease) and of the respiratory system (e.g. asthma).1 Therefore, it is very important to determine the mucosal barrier state in UAT mucosal inflammatory disorders.
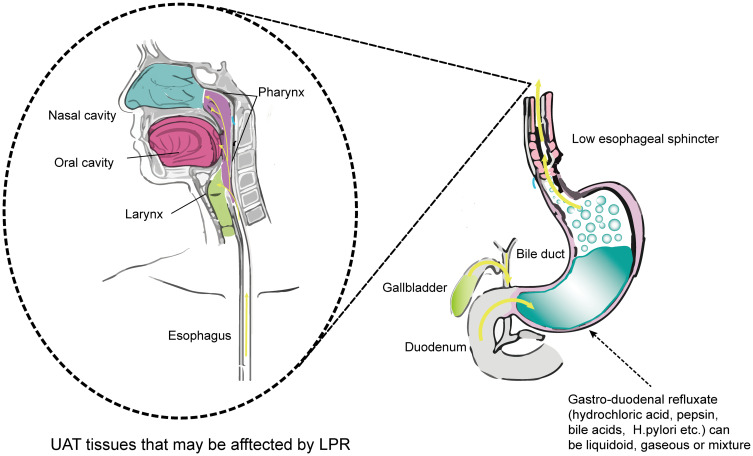
LPR is the result of the reflux of gastric contents into the laryngopharynx mucosa, where the refluxate comes into contact with the mucosa of the UAT,2,3 and LPR has been shown to play a role in the development of numerous UAT mucosal inflammatory disorders, including chronic laryngitis and pharyngitis,4 chronic rhinosinusitis,5 chronic otitis media,6–8 benign vocal fold lesions (BVFLs),9–11 and laryngeal mucosal precursor lesions (LMPLs)12–14 (Figure 1). LPR accounts for 4–10% of all otolaryngology clinic patients and leads to high costs for long-term anti-reflux therapy.15 It has been proposed that long-term stimulation from gastro-duodenal refluxate leads to the deficiency and/or dysregulation of UAT mucosal barrier function, which may eventually lead to mucosal inflammatory disorders or even to the malignant transformation of the mucosal epithelia. Recent studies have identified the inflammatory biomarkers of LPR-related mucosal disorders, and here we will review LPR-induced mucosal barrier dysfunction and the inflammatory response to LPR in the UAT mucosa.
Figure 1.
Mechanisms of LPR and LPR-related symptoms. Dysfunction of the lower esophageal sphincter causes the reflux of gastro-duodenal contents into the UAT mucosa. Reflux mainly injures the mucosa of the larynx and pharynx, but sometimes can even affect the middle ear through the eustachian tube. Refluxate containing multiple acids and proteases injures the UAT mucosa and causes later mucosal inflammation. The left part of Figure 1 has adapted from Douglas College Human Anatomy & Physiology II. Douglas College,New Westminster BC. Aug 31, 2017. Douglas College Human Anatomy and Physiology II (1st ed.) by Rice University is licensed under a Creative Commons Attribution 4.0 International License. 126
Anatomic and Physiological Characteristics of the UAT Mucosa
The UAT refers to the mixed airway/digestive tract, including the oral cavity, nasal cavity, paranasal sinuses, pharynx, larynx, and upper esophagus, and it serves the physical functions of respiration, swallowing, and speech production. UAT mucosal surfaces maintain homeostasis through dynamic responses, including the production of mucus, the regulation of epithelial intracellular junctions, and the modulation of the mucosal immune system,16,17 and the loss of mucosal integrity and impaired barrier function caused by external pathogens are prominent features of mucosal inflammatory disorders.1
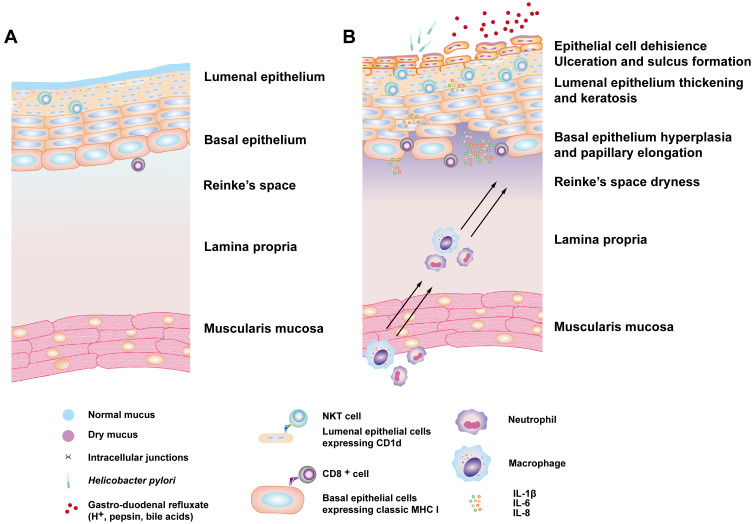
All mucosal surfaces are organized similarly. The first layer is mucus, which is secreted by epithelial mucus-secreting cells, and this forms a mucoid architecture covering the mucosal surfaces and provides defense against noxious agents and pathogens.18 Beneath the mucus layer, the epithelial cell layer serves as a major structural component of mucosal surfaces and is formed on top of an acellular basement membrane.17 Underneath this is a loose connective tissue layer, called the lamina propria, that includes blood vessels, lymphatics, immune cells, and other components. The mucus layer, mucosal epithelia, and immune cells constitute the physical and biological barrier against harmful stresses, and dysfunction of the mucosal barrier results in mucosal inflammatory disorders and diseases (Figure 2A).
Figure 2.
The normal state and the inflammatory changes that occur in LPR pathogenesis. (A) In the normal state, the MHC1high/CD1dlow basal epithelial cells gradually transition into the MHC 1low/CD1dhigh phenotype in the luminal layers, which can be recognized by NKT cells. (B) This phenomenon is more apparent during an LPR-induced inflammatory response in the squamous epithelium. The inflammatory mediators in this response also lead to changes in the extracellular mucus layer and to the subsequent chemoattraction and infiltration of immune cells followed by the keratinization and proliferative response of the mucosal epithelium.
Mucus Layer
The UAT epithelial surfaces (except for the oral cavity) are coated with a thick, viscoelastic layer of mucus. The extracellular mucus layer has two major components – mucins (Table 1) and anti-microbial molecules.18,19 The secreted and transmembrane mucins compose the major structure of mucus19 by forming a gel-like matrix to retain the anti-microbial components in the mucosal environment.18 Secreted mucins are large monomers that can be assembled into homo-oligomers to form an entangled mesh-like structure.18,19 Transmembrane mucins are anchored to the cell surface to form the glycocalyx and participate in intracellular signal transduction pathways, regulation of immune responses, cell differentiation, and cell proliferation.18 The anti-microbial components consist of nonspecific anti-microbials and specific anti-microbial immunoglobulins. Most nonspecific anti-microbials are microbicidal lectins or small peptides that can interact with and breach microbial cell membranes.18 The specific immunoglobins (IgA and IgG) are produced by B cells in the lamina propria and then transported into epithelial cells through the polymeric immunoglobulin receptor and are finally secreted by the epithelial cells into the mucus.20 In addition, the mucus also contains lipids, nucleic acids, and inorganic salts.18 The interplay among the mucus components forms a complex three-dimensional mucus shield to protect the mucosa surfaces against chemical, mechanical, and biological stresses. For example, the mucus layer has protective and lubricating functions when the UAT undergoes shearing actions such as coughing,18 and the gastric mucosa is resistant to the caustic effects of hydrochloric acid and pepsin, which is attributable to the impervious nature of the double-layer mucus.21 This architecture of the gastric mucus together with the bicarbonate buffer and lipids is responsible for preventing the back-diffusion of H+ and pepsin to the gastric mucosal surface.18,21 The nasal mucus layer can trap the inhaled or ingested particles (e.g. allergens and microbes) through their direct binding to mucins, and it performs mucosal clearance via anti-microbial molecules and the rapid turn-over of the mucus layer.19 Thus, the nasal mucus layer can respond in a dynamic manner towards different external stresses in order to maintain homeostasis.18,19,22
Table 1.
Molecular Alterations in UAT Mucosa Affected by LPR
| Molecular | Alteration in Anatomic Sites | References Number |
|---|---|---|
| Mucus layer | ||
| Muc1 | ↑laryngeal dysplasia and laryngeal cancer | [9] [88] |
| Muc2 | ↓laryngeal epithelium; ↑Barrett’s esophagus | [21,86] |
| MUC3 | ↓laryngeal epithelium | [86] |
| MUC4 | ↑Barrett’s esophagus | [21] |
| MUC5AC, MUC5B | ↓laryngeal epithelium | [86] |
| CA-III | ↑↑esophageal epithelium, –vocal fold, ↑laryngeal posterior commissure | [89–92] |
| Intracellular junctions | ||
| Claudin-1 | Esophageal epithelia (↑spinous and granular layers; ↓basal cells) | [98] |
| Claudin-3 | ↓esophageal epithelia | [96–98] |
| Claudin-4 | ↓esophageal epithelia | [96–98] |
| Occludin | Esophageal epithelia (↑spinous and granular layers; ↓basal layers) | [98] |
| ZO-1 | Esophageal epithelia (↑spinous and granular layers; ↓basal layers) | [98] |
| JAM-1 | Esophageal epithelia (↑spinous and granular layers; ↓basal layers) | [98] |
| Desmoglein 1 | Esophageal epithelia (↑spinous and granular layers; ↓basal layers) | [98] |
| E-cadherin | ↓pharyngeal, laryngeal and nasal epithelia; ↓head and neck squamous cell carcinoma | [86,99,100] |
| β-catenin | ↑laryngeal squamous carcinoma cell; | [104] |
| Immune system | ||
| CD1d | ↑laryngeal epithelia | [13] |
| TLR4 | ↑esophageal epithelia; ↑↑esophageal adenocarcinoma | [113] |
| IL-6 | ↑lung epithelium,↑middle ear, ↑ airway epithelium | [78,80,115,116] |
| IL-8 | ↑reflux esophagitis; ↑Barrett’s esophagus; ↑laryngeal carcinoma | [117,118] |
Notes: ↑Represents up-expression, ↑↑Represents higher up-expression, ↓Represents down-expression, –Represents depletion.
Epithelial Intracellular Junctions
Epithelial intercellular junctions can mediate adhesion and communication between adjoining epithelial cells and are a critical element in the barrier defense against external stresses. Apical junction complexes in the epithelia consist of the tight junctions (TJs) and the underlying adherent junctions (AJs).1 TJs form a barrier by regulating paracellular permeability and maintaining cell polarity,23 while AJs are critical for inhibiting epithelial cell growth and reducing the paracellular permeability of leukocytes and solutes.24
TJ proteins include claudins, occludins, junctional adhesion molecules (JAMs), and the scaffold proteins of the zonulae occludentes (ZO) (Supplementary Table 1). Claudins are the major components of TJs,1 and deletion of claudin-1 in mice compromises the epidermal barrier and causes excessive water loss.25 Occludins contribute to intercellular adhesion,26 and exogenous occludin expressed in fibroblasts localizes to the points of cell-cell contact in confluent cells and induces aggregation.27 JAMs and ZOs participate in the regulation of diverse functions such as intercellular adhesion, attachment to actin, establishment of cell polarity, transmembrane transport, cell signaling, and gene expression.26
AJs are another critical constituent of intracellular junctions and form below the TJs in the lateral membrane, and AJs are comprised of the cadherin, catenin, and nectin protein families.17 The key components of AJs are the members of the classical cadherin superfamily, such as epithelial cadherin (E-cadherin). Down regulation of E-cadherin and other junction proteins has been detected in the nasal epithelia of an ovalbumin-induced allergic rhinitis mouse model, and this accelerates epithelial barrier breakdown during nasal inflammation.17 By binding to specific regions of E-cadherin, proteins from the catenin family can regulate the stability and degradation of E-cadherin.17 β-catenin together with E-cadherin constitute the AJ complex and play a vital role in the maintenance of structural integrity and cell adhesion. Little is known about the functional role of the nectin family of proteins in epithelial cells.
The UAT Mucosal Immune System
Immune Cells
The mucosal immune system of the UAT can be roughly subdivided into two primary components – the organized mucosa-associated lympho-reticular tissues (MALTs), also known as Waldeyer’s ring (comprising the palatine tonsils, adenoids, and lingual tonsils), and the diffuse lamina propria or glandular tissues.28–30 MALTs and the lamina propria contain various immune cell types such as antigen-presenting cells (APCs), innate lymphoid cells, B cells, and T cells.31 Both innate and adaptive immunity are of great importance in barrier defense.
APCs such as dendritic cells (DCs), macrophages, and UAT epithelial cells can sense external threats and can quickly trigger innate immune responses via pattern recognition receptors32 such as the leucine-rich repeat domains that are seen in toll like receptors (TLRs), nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs), retinoic acid-inducible gene I (RIG-I)-like receptors (RLRs), and receptor-unidentified double-stranded DNA sensors.32–34 The activation of TLRs by pathogens results in the production of anti-microbial substances and inflammatory cytokines and chemokines to induce defensive responses.32 TLR2 and TLR4 in the upper airway mucosal basal cell layers are stimulated by pathogens to upregulate mucin expression.35 Furthermore, TLR2, 3, 4, and 7 in the human esophageal epithelial cell-line TE-1 promote the production of β-defensin 2 in response to luminal stimulation.36 The TLR-dependent production of inflammatory cytokines and chemokines is a double-edged sword, however. For example, these inflammatory mediators cause an influx of neutrophils, macrophages, and lymphocytes in order to facilitate bacterial clearance in the oral cavity, but they also recruit epithelial cells and macrophages to this site where they secrete TNF-α, which results in inflammation-related oral bone loss.35
The activation of the adaptive immune system mediated by T cells and B cells leads to an antigen-specific host response.37 Micro-fold cells, the professional antigen-sampling epithelial cells in the follicle-associated epithelium of the MALTs, are required for mucosal T cell-dependent IgA responses and have been recognized as a potential novel target for mucosal vaccines via nasal or oral administration.30 Increased numbers of Th17 cells and decreased numbers of Treg cells can be detected in both nasal polyp tissues and blood from chronic rhinosinusitis patients, and such alterations might be vital for the development of nasal polyps.38 Furthermore, the Th1/Th2 balance is critical in the mucosal immune response and tolerance,39 and it has been reported that HPV-6 and −11 can escape the virus clearance of the UAT mucosa through inhibition of Th1 and polarization of T cells into Tregs and memory Th2-like T cells.40
Cytokines and Chemokines
Cytokines and chemokines have complicated effects on cell growth, differentiation, and activation.41 APC-derived TNF-α, IL-6, and IL-8 are particularly effective in promoting the infiltration of immunocytes into the mucosal tissues where they can cause damage.41 Th1 cytokines predominate in tonsillar inflammatory disorders,42 and inhibition of Th1 responses might be a novel target for the treatment of chronic tonsillitis.43 Th2 cytokines are associated with allergic immune processes.41,44 For example, IL-5, secreted by Th2 cells, is essential for esophageal eosinophilia, and anti-IL-5 therapies targeting eosinophilic inflammation have been shown to decrease the numbers of esophageal eosinophils and mast cells.45 By down-regulating the IL-23/IL-17 proinflammatory axis of Th17 cells, the commensal and probiotic bacteria can inhibit pathogen colonization in the oral and pharyngeal mucosa and thus contribute to host defense.46
The chemokine superfamily consists of a large number of ligands and receptors divided into four subfamilies: CXC, CC, (X)C and CX3C.47 CCL25, CCL28, CXCL14, and CXCL17 are homeostatically expressed in mucosal tissues, and these mucosal chemokines exhibit broad anti-microbial activity and regulate the composition of the mucosal microbiome.48 The CXCL17/CXCR8 axis represents a powerful macrophage-recruitment mechanism that might be related to the functional macrophage subsets in mucosal tissues.48 Previous studies of the mucosal chemokines have primarily focused on the gastrointestinal mucosa, and their role in the UAT mucosa remains to be elucidated.
LPR-Related Mucosal Inflammatory Disorders
LPR
LPR is the backflow of gastroduodenal contents into the UAT, and it is involved in many ENT inflammatory, neoplastic disorders and is behind up to 50% of voice disorders.15,49–51 The most important mechanism of LPR-induced inflammation is the direct noxious effect of the gastroduodenal contents on the mucosa, causing swelling, mucus hypersecretion, and the secretion of inflammatory mediators. A second mechanism consists of triggering a vagal response supported by excessive vagal reactivity, and a hypothesized third mechanism postulates an association between Helicobacter pylori infection and mucosal inflammation (Figure 1). Laryngopharyngeal reflux differs from gastroesophageal reflux disease (GERD) in that it is not associated with heartburn and regurgitation symptoms, even though sharing some common pathophysiological mechanisms.2 The reflux episodes in GERD are more frequently liquid, recumbent, and nighttime, whereas the reflux episodes in LPR are mainly gaseous, upright, and daytime.52 In addition, unlike GERD that mainly presents as acidic reflux, LPR has acidic, nonacidic, and mixed subtypes, and these can be detected by multichannel intraluminal impedance-pH monitoring (MII-pH). In fact, recent studies have shown two main profiles of LPR patients according to MII-pH: patients with concomitant GERD and acidic LPR, and those without GERD who, in similar proportions, have nonacidic or mixed LPR.52–54 Although LPR and GERD have different molecular changes in the mucosa, they are interlinked conditions.52 Therefore, classification of LPR patients has great clinical significance in tailored treatments.
Treatments for LPR should include many aspects. Currently, the application of proton pump inhibitors has failed to show clear benefits in LPR.55 Anti-pepsin treatment, such as alginates, has gained more and more attention as an anti-acid therapy because pepsin is involved in multiple processes in LPR pathogenesis (discussed later). Further processing of alginates may improve their pharmacological properties of pepsin and bile acid absorption.55 Moreover, surgical treatment like laparoscopic fundoplication has also been applied in LPR patients, particularly those who are nonresponsive to anti-reflux medication.54 However, surgery’s role in the management of LPR remains uncertain because of a lack of reliable and reproducible results.4,12,54
LPR-Related UAT Mucosal Inflammation
Laryngitis and pharyngitis are the most common symptoms induced by LPR,4 and laryngeal erythema, posterior commissure hypertrophy, diffuse laryngeal edema, and pseudosulcus observed by laryngoscopy confirm the direct laryngopharyngeal irritation and inflammation. However, in vivo studies suggest that the subglottic mucosa and the vocal fold mucosa suffer the most from LPR, while LPR has minimal impact on the posterior commissure mucosa and ventricular mucosa2,3,54,56 or other parts of the larynx that are covered by squamous epithelia.54
Other tissues of the UAT can also be affected by gaseous reflux due to the large scope of gas diffusion.57 In recent clinical research, LPR has been correlated with chronic otitis media with effusion (OME), oral soft tissue disorders, chronic rhinosinusitis, and dacryostenosis,6,58–60 and patients with these diseases show non-specific laryngopharyngeal symptoms, positive results of MII-pH monitoring, or the detection of reflux agents in the secretions.6,61,62 Although the control of LPR in some of these diseases was considered beneficial, how LPR is involved in the diseases remains uncertain.61 Diseases such as oral soft tissue disorders and dental erosion are more widely accepted as extraesophageal symptoms of GERD63,64 because the gastro reflux can pass through the hypopharynx directly to the oral cavity. Nonetheless, LPR and GERD are mostly suggested to have a role in promoting local mucosal inflammation in the UAT,6,61–65 except for chronic sinusitis, in which reflux as a causal factor remains uncertain.66,67 The refluxate has also been suspected to act as an allergen in allergic diseases such as asthma because of the pepsin found in lung aspirate.54 Therefore, further research on the underlying mechanisms through which LPR is involved in these diseases is needed for the improvement of current therapies.
BVFLs
BVFLs consist of lesions involving vocal fold nodules, polyps, Reinke’s edema, sulcus vocalis, and cysts, most of which develop from the lamina propria.9 In addition to phonatory trauma and vocal misuse, a long-suspected causal factor of BVFLs is LPR,11 although the role of LPR in the pathogenesis of BVFLs remains unclear. Pepsin is frequently detected in polyp tissue from posterior commissure biopsies,11,68 which suggests the correlation between the inflammatory mucosal reaction of LPR and subsequent vocal cord disorders.69 It has been proposed that the vocal fold mucosa is more vulnerable to mechanical and biochemical stresses during normal and abnormal phonation processes in the context of reflux,11 which leads to the nodules, polyps, and Reinke’s edema.9 Typically, patients with these lesions also present with chronic mucosal irritation in the form of throat clearing or chronic cough as well as increased subglottic aerodynamic driving pressure, which in return increases the occurrence of microtraumas to the vocal fold.70
LMPLs
LMPLs often manifest as gross superficial mucosal appearances of leukoplakia, erythroplakia, or erythroleukoplakia,71 and they present with multiple pathologic changes ranging from hyperplasia or hyperkeratosis to severe dysplasia.49 Numerous challenges remain for laryngologists to provide accurate diagnoses and tailored treatments.
Premalignant and malignant LMPLs ultimately convert into laryngeal squamous cell carcinoma. Smoking and alcohol abuse are major risk factors for LMPLs,49 and LPR has recently been suggested to be an independent factor in LMPL development.49,71 It is hypothesized that the refluxate can lead to chronic mucosal inflammation and can trigger the development of laryngeal malignancies71 such as gastric cancer72 and hepatocellular carcinoma.73 Furthermore, GERD has been recognized as a key risk factor in the development of Barrett’s esophagus.74 Finally, a recent meta-analysis found an association between reflux diseases and laryngeal malignancy regardless of smoking and drinking histories.75 Further studies are needed to elucidate the pathological mechanisms of LPR-induced LMPLs, and such knowledge will likely be beneficial in clinical therapy.
LPR-Induced Mucosal Barrier Dysfunction
Damage from Gastric Refluxate
Gastric-duodenal refluxate contains gastric acid, proteases, and bile acids3,57 and has a pH ranging from 1.5 to 2.0,3,12,76 which is damaging to the luminal environment of the UAT that normally has a pH of 6.8–7.0.3,12 In healthy subjects, the amount of refluxate is usually small and can be cleared quickly by their innate defense mechanisms.57 While LPR leads to adverse clinical consequences, the nature of this evolving disease depends on the contents of the gastric refluxate, the duration of exposure, and the activity of the local defense mechanisms of the UAT mucosa.
Gastric Acid
The main component of gastric acid is hydrochloric acid. Large volumes of gastric acid result in chemical injury to the airway epithelium, while recurrent small-volume reflux may result in chronic injury.57 Damage to the UAT epithelia by acid reflux might be due to the toxicity of its low pH.3 Such caustic injury leads to disruption of the mucosal barrier, and the severity of such acid-induced damage depends on the level of exposure.3 Furthermore, different parts of the same organ react differently to acid exposure, and the subglottic columnar epithelium and vocal fold squamous epithelium are more sensitive to acid.3 The role of the acid in LPR remains uncertain because of the difficulty in quantifying acid exposure.2 Furthermore, the symptoms of LPR are not consistent with pH alterations induced by anti-reflux medication.12,54 In vitro studies suggest that the initial stimulus of gastric acid might trigger a cascade of inflammatory responses with the recruitment of immune cells and the release of various inflammatory mediators.12,57
Digestive Proteases
Pepsin is considered to be the most aggressive protease in the gastroduodenal refluxate.77 Previous studies have shown that acid alone cannot disrupt epithelial barrier integrity, but a combination of pepsin and acid results in significant damage to the laryngeal mucosa.78 In a porcine model, acid-pepsin causes the most severe damage at pH 2.0,3 although pepsin retains its activity and stability over a wide pH range of 1.5–7.5.12 In gaseous reflux, pepsin is brought into contact with the UAT mucosa, extending even as far as the middle ear,8,57 and may cause damage through subsequent acid reflux episodes.12 Furthermore, some studies showed that pepsin can be transported into epithelial cells79 and can be re-activated by intracellular structures such as Golgi bodies and lysosomes that have a lower pH of 5.0 and 4.0, respectively, thus leading to intracellular damage.12 Because pepsin is stable and detectable in multiple UAT organs and tissues,77 it plays a crucial role in reflux-related inflammation and injury and has been identified as a biomarker and potential therapeutic target of LPR.3,77,80
Bile Acid
Bile acid, one of the constitutes of gastroduodenal refluxate,12 is considered to be a pathogenic factor in numerous mucosal lesions.76,81–83 The conjugated and unconjugated bile acids in the human digestive tract have diverse activities at different pH values. Conjugated bile causes laryngeal injury at a low pH (1.2–1.5),12 while the unconjugated bile, like chenodeoxycholic acid, is activated at pH 7.0.12 Therefore, bile acids can cause mucosal injury in both acidic and non-acidic environments.
Mucosal Barrier Dysfunction
Dysfunction and Dysregulation of the Mucus Layer
Mucosal irritation leads to abnormal mucus secretion.54 Some LPR-related inflammatory diseases are accompanied with increased mucus secretion. In LPR-related OME, the pepsin level in the serous and ear mucoid subgroups is significantly increased compared to that in the dry ear subgroup, which can be interpreted to mean that the viscosity of the middle ear cavity fluid depends on the concentration of pepsin.60 In addition, low pH caused by acidic refluxate in the esophagus induces a neural reflex to aggregate the nasal mucus production via the vagus nerve.84 In GERD patients, it has been found that the salivary flow rate in response to esophageal acid infusion decreases, which explains why dry mouth is associated with burning symptoms in the oral cavity.85 However, the salivary secretion in LPR remains uncertain and needs further study.
Changes in mucus composition have also been observed. Secreted Muc2, 5AC, and 5B in the laryngeal mucus constitute the airway mucus gels,22 and the antagonistic effect of weak acid and pepsin was found to down-regulate mucin expression at low pH.22 In patients with LPR-related laryngitis, decreased expression of Muc2, 5AC, and 5B results in the reduction of secreted mucin by the laryngeal epithelia and thus reduces the protective effect of the mucosal surfaces.12 The sticky mucus triggers symptoms such as postnasal drip, globus sensation, and throat clearing. Muc3, a transmembrane mucin, is decreased in patients with LPR-associated inflammatory mucosa.22,86 Specifically, Muc3A is thought to be a key factor in the maintenance of the epithelium under hypoxic conditions and in the regulation of cell migration and apoptosis during wound healing.87 Muc1, another transmembrane mucin, has been shown to be highly expressed in patients with laryngeal dysplasia and laryngeal cancer.88 By binding with the epidermal growth factor family of receptor tyrosine kinases, Muc1 triggers cell proliferation. It also promotes metastasis in human cancer cells by binding with galectin-3, a member of the galectin family that regulates tissue fibrosis, immunity, and inflammatory response.22 Muc2 and Muc4 expression is up-regulated in intestinal metaplasia in Barrett’s esophagus;22 however, a retrospective study suggested that Muc4 is associated with better survival in patients with advanced non-metastatic laryngeal cancer.12 Thus, the function of mucins in LPR-related carcinogenesis needs further study. As an integral component of mucus, the secretion of bicarbonate is modulated by carbonic anhydrase (CA) and is critical in maintaining the mucosal luminal pH.12 In patients with GERD, increasing CA-III expression has been observed in inflamed esophageal squamous epithelial cells with a redistribution from the basal to the superficial cell layers,89,90 which protects against low pH damage from the refluxate. However, in patients with LPR, the expression of CA-III shows subsite specificity. CAIII expression is depleted in the vocal folds, but it is relatively higher in the posterior commissure. In addition to being activated in the acidic condition, the existence of pepsin alone can be a causal factor to CAIII depletion.12,89,91,92 Further research is needed into CA expression and its function in LPR-induced mucosal inflammation. β-defensins, which are anti-microbial peptides against a broad spectrum of microorganisms, are secreted by immune cells and epithelial cells into the mucus.93 Their expression can be stimulated by H. pylori infection, and their levels are significantly higher in vocal polyps than in vocal cord nodules.94 Apolipoprotein-A is a lipid component in the mucus18,95 and is negatively correlated with the formation of polyps.9 Because H. pylori can trigger the breakdown of the mucin-lipid network,18 decreased apolipoprotein-A expression appears to be associated with vocal polyps and H. pylori infection in the UAT mucosa as indicated by less favorable prognosis for vocal polyps compared to nodules.
Dysfunction of Epithelial Intercellular Junctions
Gastric refluxate has been reported to disrupt the cohesion between cells by digesting intracellular junctions,3 and the expression and the distribution of TJs are critical in the process of reflux diseases. Although there is limited literature in this field, research on TJs of the esophageal epithelium of GERD patients may be informative for future LPR therapy. Several studies based on a rat esophagitis model have shown the down-regulation of claudin-3 and claudin-4 in the plasma membrane of epithelial cells in the spinous and granular layers upon stimulation by acid, bile salts, and trypsin.96–98 However, the expression levels of claudin-1, occludin, ZO-1, JAM-1, and desmoglein 1(DSG-1) change under different pathological conditions. These proteins increased in both the cellular membrane and cytoplasm of spinous and granular layers around mucosal erosion, but decreased in individual cells of the same layer as hyperplasia continued in the basal cells.98 These findings suggest that elevated expression of these proteins to resist the damage from refluxate is an early molecular event in LPR pathogenesis. However, when the damage persists and the reflux starts to promote hyperplasia, the production of TJ proteins and desmosome proteins becomes insufficient, which results in the failure of this protein-compensating mechanism.98
For AJs, decreased E-cadherin expression in pharyngeal and nasal epithelia has been shown to occur in response to acid exposure.99,100 Decreased E-cadherin expression was also detected in biopsies from LPR patients.101 E-cadherin is one of the biomarkers of cancer progression and metastasis in neoplastic cells,102 and decreased E-cadherin expression is considered to be a poor prognostic factor in patients with head and neck squamous cell carcinoma.103 Because the loss of E-cadherin expression is a key initial step in tumor invasion,12 decreased E-cadherin expression in LPR-related chronic inflammation might not only play a role in the development of symptoms, but also might promote the development of dysplasia in the context of reflux.12 In addition, sustained loss of E-cadherin leads to epithelial differentiation into a mesenchymal phenotype, a process known as epithelial-mesenchymal transition,17 which is crucial in tumorigenesis. On the other hand, the expression of β-catenin, one of cytosolic plaque proteins, remains uncertain. It was found to be unchanged in laryngeal biopsies from LPR patients,101 but was elevated in laryngeal squamous carcinoma cell in the context of pepsin.104 The E-cadherin–catenin complexes are essential for the structural integrity of the epithelial tissues. Therefore, further research is needed into the roles of E-cadherin and β-catenin in the mucosal injury of LPR, especially dysplasia and tumorigenesis.
Dilation of intercellular spaces (DIS), presenting as a significantly greater intercellular space that can be measured by transmission electron microscopy, is reported to be an early morphological marker in GERD. This morphological alteration is considered to be an ultrastructural feature of acid damage in the esophageal squamous epithelium. However, whether this marker is useful in diagnosing LPR-related laryngitis remains controversial. Although Hu et al verified DIS as a morphologic marker for LPR in a rabbit reflux model,105 their method for generating artificial reflux lacked data about pH and so could not show that the reflux was acidic or not. On the other hand, Vaezi et al saw no increase in DIS of the distal esophagus or larynx among patients with GERD or those with reflux-related laryngitis.106 This difference compared to prior studies may suggest the diversity of reflux type among the GERD patients, even though the overwhelming majority of reflux in GERD is acidic. In addition, they also observed a possible graded distal esophageal variation of DIS that could be corrected by acid-suppressing therapy. Greater intracellular space and loss of intracellular junctions in the context of reflux leads to a disorganized barrier defense and increased permeability of the epithelium and may exacerbate mucosal damage and transformation. Future studies on mucosal morphology are needed to determine whether this transformation happens in LPR patients and how this transformation is distributed in LPR-related mucosa.
Dysregulation of the Mucosal Immune System
The infiltration of multiple immune cells has been observed in mucosal lesions in relation to LPR. In LPR-related laryngeal cancer, higher M2-macrophages polarization and increasing Treg cell numbers were detected in both tumor tissue and intact mucosa, with acidic reflux in particular.107 The “lipid index” of macrophages in induced sputum is a noninvasive marker of aspiration, and it was found that high lipid-laden macrophages (LLMI) may be related to higher incidence of acidic oropharyngeal reflux.108 Similar phenomena have been observed in patients with GERD-related asthma and chronic cough.109 However, LLMI was negatively correlated with GERD, tracheal aspirations, and respiratory diseases in children,110,111 which may be due to differences in the immune system between the children and adults.
Rees et al observed increased CD8+ T cell, natural killer T (NKT) cells, and elevated CD1d expression in the laryngeal mucosa in response to LPR.13 They also found a topographic switching in which major histocompatibility complex (MHC) 1high/CD1dlow epithelial cells in the basal layers of laryngeal mucosa gradually transition into the MHC 1low/CD1dhigh phenotype in the luminal layers.112 Upon activation, NKT cells rapidly produce high levels of IFN-γ and IL-4 in order to influence Th1/Th2 immune responses, and the switch from the MHC I–/CD8+ T cell axis at the basal layer to the CD1d/NKT cell or CD1d-responsive CD8+ T cell axis at the upper/superficial epithelial layer might play a central role in immunopathology during the chronic inflammatory challenge of LPR.13 (Figure 2B). This alteration may play a vital role in the maintenance of the balance of the upper airway tolerance and the inflammatory responses, which may be crucial in the larynx. Because the larynx is the junction of the IgA-dominated upper and IgG-dominated lower airways, it is confronted with a high density of inhaled/ingested challenges and thus contains a high density of immunologically active cells. Further exploration is needed to determine the exact role that the CD1d/NKT cell axis plays in LPR pathological processes and the response of the mucosal immune system in the upper and lower airway under these conditions.
The role that TLRs plays in LPR-related diseases remains uncertain, but the alteration of TLRs’ expression has been reported in GERD-related malignant complications such as esophageal adenocarcinoma and Barrett’s esophagus. The reflux stimulates TLR4 expression in normal esophageal cells, and TLR4 was found to be highly expressed in adenocarcinoma. The activation of TLR4 results in increased cell proliferation, which indicates an enhanced response in the process of inflammation-induced carcinogenesis.113 In addition, microbes, e.g., Streptococcus pneumonia, may be associated with the upregulation of TLRs that mediate the molecular changes that occur in esophageal adenocarcinoma carcinogenesis under reflux conditions.114
Reflux disease-related changes in IL-6 expression have been observed in several UAT tissues.12,78,80,115 IL-6 is a proinflammatory cytokine that functions in multiple immune processes, including acute-phase responses, inflammation, neutrophil recruitment, and Th17 cell differentiation,116 and it has been shown that IL-6 expression in the esophagus increases as the grade of reflux pathology increases and that it decreases upon therapy.12,115 Thus, IL-6 may be considered an indicator of LPR-related inflammation in the UAT mucosa. IL-8 has been suggested to play key roles in both acute inflammation and chronic inflammatory injury associated with GERD and LPR.12 IL-8 expression has been found to increase in more severe GERD patients, especially those with reflux-related complications,117 and to decrease significantly following anti-reflux surgery (e.g. laparoscopic fundoplication).115 Therefore, IL-8 is a potential indicator of reflux-related inflammation in the UAT mucosa. In addition, acidic pepsin and IL-8 expression are positively correlated with the development of laryngeal carcinoma,104 and tumor-derived IL-8 has been shown to activate epithelial cells in the tumor vasculature and to promote angiogenesis and to increase the proliferation and migration of cancer cells through the CXCR2 pathway.49,118 Moreover, tumor-associated macrophages are induced by IL-8 to secrete additional growth factors that increase cell proliferation.12 Increased Th2 cytokines, such as IL-5 and IL-13, are detected in allergic diseases and eosinophilic inflammation and are associated with non-specific reflux disease.119 It is believed that reflux disease-related allergic responses are associated with LPR, and these allergic symptoms are relieved after anti-reflux therapies,120,121 but the mechanisms by which the reflux induces the allergic responses need to be further studied.
Furthermore, many inflammatory mediators can interact with mucus and/or intracellular junctions in chronic mucosal inflammatory disorders. TNF-α, IL-6, and IL-8 upregulate Muc2, Muc4, Muc5AC, and Muc8 gene expression21,122 and IL-17 upregulates both Muc5B and Muc5AC,122,123 suggesting the differential and specific regulation of mucin genes by immune response mediators. The proinflammatory cytokines TNF-α, IL-4, IL-6, and IL-13 are known to increase the permeability of intestine epithelial cell monolayers, and this effect is related to the increased expression of claudin-2.124,125 Besides claudins, other TJ proteins have been shown to be regulated in inflammatory processes. The occludins and JAM-A are internalized in response to IFN-γ, thereby increasing the permeability of the mucosa,1 and defective E-cadherin expression in the airway can activate DCs and thus facilitate allergen presentation. Moreover, the loss of E-cadherin in cultured epithelial cells results in the increased production of thymic stromal lymphopoietin, which triggers an early step in breaking inhalational tolerance and leads to Th2 cell sensitization and asthma development.17 Future research on these interactions might be beneficial to understanding LPR-related mucosal inflammation.
Conclusions
LPR-related mucosal disorders are commonly encountered in the ENT department, presenting challenges in diagnosis and in optimizing treatment. The pathogenetic mechanism of mucosal inflammation is unclear, but it is believed to be caused by a mixture of acid and other injurious components, particularly pepsin. Pepsin has been implicated in multiple inflammatory processes and can work as a potential biomarker as well as a therapeutic target. Changes in UAT mucosal barrier function are a hallmark of LPR-related mucosal inflammatory disorders. CD8+ T cell infiltration, CD1d/NKT cell activation, and high expression of TNF-α, IL-6, and IL-8 have been observed in LPR-induced inflammatory processes, and further studies of these inflammatory cascades are needed to improve diagnosis and treatment. Further studies are also required to determine the mechanism of the mucosal immune response towards injuries caused by refluxate in order to provide a definitive diagnosis for laryngopharyngeal reflux and to improve current treatments.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos. 81970879, 81570913), Excellent Doctors-Excellent Clinical Researchers Program (No. SZA202002), “Pujing Talent Plan” (18PJ1401700) of Shanghai Science and Technology Committee (19441900200), and Research Projects of Shanghai Municipal Health Committee (2020YJZX0110).
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors have declared that no conflict of interest exists.
References
- 1.Georas SN, Rezaee F. Epithelial barrier function: at the front line of asthma immunology and allergic airway inflammation. J Allergy Clin Immunol. 2014;134(3):509–520. doi: 10.1016/j.jaci.2014.05.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ford CN. Evaluation and management of laryngopharyngeal reflux. JAMA. 2005;294(12):1534–1540. doi: 10.1001/jama.294.12.1534 [DOI] [PubMed] [Google Scholar]
- 3.Bulmer DM, Ali MS, Brownlee IA, Dettmar PW, Pearson JP. Laryngeal mucosa: its susceptibility to damage by acid and pepsin. Laryngoscope. 2010;120(4):777–782. doi: 10.1002/lary.20665 [DOI] [PubMed] [Google Scholar]
- 4.Wood JM, Athanasiadis T, Allen J. Laryngitis. BMJ. 2014;349(oct0921):g5827–g5827. doi: 10.1136/bmj.g5827 [DOI] [PubMed] [Google Scholar]
- 5.Southwood JE, Hoekzema CR, Samuels TL, et al. The impact of pepsin on human nasal epithelial cells in vitro: a potential mechanism for extraesophageal reflux induced chronic rhinosinusitis. Ann Otol Rhinol Laryngol. 2015;124(12):957–964. doi: 10.1177/0003489415593556 [DOI] [PubMed] [Google Scholar]
- 6.Iannella G, Di Nardo G, Plateroti R, et al. Investigation of pepsin in tears of children with laryngopharyngeal reflux disease. Int J Pediatr Otorhinolaryngol. 2015;79(12):2312–2315. doi: 10.1016/j.ijporl.2015.10.034 [DOI] [PubMed] [Google Scholar]
- 7.Abdel-aziz MM, El-Fattah AM, Abdalla AF. Clinical evaluation of pepsin for laryngopharyngeal reflux in children with otitis media with effusion. Int J Pediatr Otorhinolaryngol. 2013;77(10):1765–1770. doi: 10.1016/j.ijporl.2013.08.014 [DOI] [PubMed] [Google Scholar]
- 8.Gorecka-Tuteja A, Jastrzebska I, Skladzien J, Fyderek K. Laryngopharyngeal reflux in children with chronic otitis media with effusion. J Neurogastroenterol Motil. 2016;22(3):452–458. doi: 10.5056/jnm16013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Naunheim MR, Carroll TL. Benign vocal fold lesions: update on nomenclature, cause, diagnosis, and treatment. Curr Opin Otolaryngol Head Neck Surg. 2017;25(6):453–458. doi: 10.1097/MOO.0000000000000408 [DOI] [PubMed] [Google Scholar]
- 10.Bercin S, Kutluhan A, Yurttas V, Yalciner G, Bozdemir K, Sari N. Evaluation of laryngopharyngeal reflux in patients with suspected laryngopharyngeal reflux, chronic otitis media and laryngeal disorders. Eur Arch Otorhinolaryngol. 2008;265(12):1539–1543. doi: 10.1007/s00405-008-0710-0 [DOI] [PubMed] [Google Scholar]
- 11.Lechien JR, Hamdan AL, Saussez S. Laryngopharyngeal reflux and benign lesions of the vocal folds. Eur Arch Otorhinolaryngol. 2019;276(1):277–278. doi: 10.1007/s00405-018-5217-8 [DOI] [PubMed] [Google Scholar]
- 12.Wood JM, Hussey DJ, Woods CM, Watson DI, Carney AS. Biomarkers and laryngopharyngeal reflux. J Laryngol Otol. 2011;125(12):1218–1224. doi: 10.1017/S0022215111002234 [DOI] [PubMed] [Google Scholar]
- 13.Rees LE, Pazmany L, Gutowska-Owsiak D, et al. The mucosal immune response to laryngopharyngeal reflux. Am J Respir Crit Care Med. 2008;177(11):1187–1193. doi: 10.1164/rccm.200706-895OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.DeMeester SR. Laryngopharyngeal reflux: weak acid and weaker correlations. Dig Dis Sci. 2019;64(5):1070–1071. doi: 10.1007/s10620-019-5465-5 [DOI] [PubMed] [Google Scholar]
- 15.Lechien JR, Saussez S, Nacci A, et al. Association between laryngopharyngeal reflux and benign vocal folds lesions: a systematic review. Laryngoscope. 2019;129(9):E329–E341. doi: 10.1002/lary.27932 [DOI] [PubMed] [Google Scholar]
- 16.Sanchez de Medina F, Romero-Calvo I, Mascaraque C, Martinez-Augustin O. Intestinal inflammation and mucosal barrier function. Inflamm Bowel Dis. 2014;20(12):2394–2404. doi: 10.1097/MIB.0000000000000204 [DOI] [PubMed] [Google Scholar]
- 17.Buckley A, Turner JR. Cell biology of tight junction barrier regulation and mucosal disease. Cold Spring Harb Perspect Biol. 2018;10:1. doi: 10.1101/cshperspect.a029314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Taherali F, Varum F, Basit AW. A slippery slope: on the origin, role and physiology of mucus. Adv Drug Deliv Rev. 2018;124:16–33. doi: 10.1016/j.addr.2017.10.014 [DOI] [PubMed] [Google Scholar]
- 19.McGuckin MA, Linden SK, Sutton P, Florin TH. Mucin dynamics and enteric pathogens. Nat Rev Microbiol. 2011;9(4):265–278. doi: 10.1038/nrmicro2538 [DOI] [PubMed] [Google Scholar]
- 20.Strugnell RA, Wijburg OL. The role of secretory antibodies in infection immunity. Nat Rev Microbiol. 2010;8(9):656–667. doi: 10.1038/nrmicro2384 [DOI] [PubMed] [Google Scholar]
- 21.Niv Y, Fass R. The role of mucin in GERD and its complications. Nat Rev Gastroenterol Hepatol. 2012;9(1):55–59. doi: 10.1038/nrgastro.2011.211 [DOI] [PubMed] [Google Scholar]
- 22.Samuels TL, Handler E, Syring ML, et al. Mucin gene expression in human laryngeal epithelia: effect of laryngopharyngeal reflux. Ann Otol Rhinol Laryngol. 2008;117(9):688–695. doi: 10.1177/000348940811700911 [DOI] [PubMed] [Google Scholar]
- 23.Oshima T, Miwa H. Gastrointestinal mucosal barrier function and diseases. J Gastroenterol. 2016;51(8):768–778. doi: 10.1007/s00535-016-1207-z [DOI] [PubMed] [Google Scholar]
- 24.Pinheiro D, Bellaïche Y. Mechanical force-driven adherens junction remodeling and epithelial dynamics. Dev Cell. 2018;47(1):3–19. doi: 10.1016/j.devcel.2018.09.014 [DOI] [PubMed] [Google Scholar]
- 25.Gunzel D, Yu AS. Claudins and the modulation of tight junction permeability. Physiol Rev. 2013;93(2):525–569. doi: 10.1152/physrev.00019.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Runkle EA, Mu D. Tight junction proteins: from barrier to tumorigenesis. Cancer Lett. 2013;337(1):41–48. doi: 10.1016/j.canlet.2013.05.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bazzoni G, Dejana E. Endothelial cell-to-cell junctions: molecular organization and role in vascular homeostasis. Physiol Rev. 2004;84(3):869–901. doi: 10.1152/physrev.00035.2003 [DOI] [PubMed] [Google Scholar]
- 28.Kunisawa J, Fukuyama S, Kiyono H. Mucosa-associated lymphoid tissues in the aerodigestive tract: their shared and divergent traits and their importance to the orchestration of the mucosal immune system. Curr Mol Med. 2005;5(6):557–572. doi: 10.2174/1566524054863924 [DOI] [PubMed] [Google Scholar]
- 29.Kutta H, Steven P, Tillmann BN, Tsokos M, Paulsen FP. Region-specific immunological response of the different laryngeal compartments: significance of larynx-associated lymphoid tissue. Cell Tissue Res. 2003;311(3):365–371. doi: 10.1007/s00441-002-0692-y [DOI] [PubMed] [Google Scholar]
- 30.Yamamoto M, Pascual DW, Kiyono H. M cell-targeted mucosal vaccine strategies. Curr Top Microbiol Immunol. 2012;354:39–52. doi: 10.1007/82_2011_134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lima SF, Teixeira AG, Higgins CH, Lima FS, Bicalho RC. The upper respiratory tract microbiome and its potential role in bovine respiratory disease and otitis media. Sci Rep. 2016;6:29050. doi: 10.1038/srep29050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Geremia A, Biancheri P, Allan P, Corazza GR, Di Sabatino A. Innate and adaptive immunity in inflammatory bowel disease. Autoimmun Rev. 2014;13(1):3–10. doi: 10.1016/j.autrev.2013.06.004 [DOI] [PubMed] [Google Scholar]
- 33.Groeger S, Meyle J. Oral mucosal epithelial cells. Front Immunol. 2019;10:208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on toll-like receptors. Nat Immunol. 2010;11(5):373–384. doi: 10.1038/ni.1863 [DOI] [PubMed] [Google Scholar]
- 35.McClure R, Massari P. TLR-dependent human mucosal epithelial cell responses to microbial pathogens. Front Immunol. 2014;5:386. doi: 10.3389/fimmu.2014.00386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kauppila JH, Selander KS. Toll-like receptors in esophageal cancer. Front Immunol. 2014;5:200. doi: 10.3389/fimmu.2014.00200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bunte K, Beikler T. Th17 cells and the IL-23/IL-17 axis in the pathogenesis of periodontitis and immune-mediated inflammatory diseases. Int J Mol Sci. 2019;20(14):3394. doi: 10.3390/ijms20143394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shen Y, Tang XY, Yang YC, et al. Impaired balance of Th17/Treg in patients with nasal polyposis. Scand J Immunol. 2011;74(2):176–185. doi: 10.1111/j.1365-3083.2011.02546.x [DOI] [PubMed] [Google Scholar]
- 39.Neurath MF, Finotto S, Glimcher LH. The role of Th1/Th2 polarization in mucosal immunity. Nat Med. 2002;8(6):567–573. doi: 10.1038/nm0602-567 [DOI] [PubMed] [Google Scholar]
- 40.Bonagura VR, Hatam LJ, Rosenthal DW, et al. Recurrent respiratory papillomatosis: a complex defect in immune responsiveness to human papillomavirus-6 and −11. Apmis. 2010;118(6–7):455–470. doi: 10.1111/j.1600-0463.2010.02617.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Borish LC, Steinke JW 2. Cytokines and chemokines. J Allergy Clin Immunol. 2003;111(2 Suppl):S460–S475. doi: 10.1067/mai.2003.108 [DOI] [PubMed] [Google Scholar]
- 42.Todorović MM, Zvrko EZ. Immunoregulatory cytokines and chronic tonsillitis. Bosn J Basic Med Sci. 2013;13(4):230–236. doi: 10.17305/bjbms.2013.2330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Semberova J, Rychly B, Hanzelova J, Jakubikova J. The immune status in situ of recurrent tonsillitis and idiopathic tonsillar hypertrophy. Bratisl Lek Listy. 2013;114(3):140–144. doi: 10.4149/bll_2013_031 [DOI] [PubMed] [Google Scholar]
- 44.Roan F, Obata-Ninomiya K, Ziegler SF. Epithelial cell-derived cytokines: more than just signaling the alarm. J Clin Invest. 2019;129(4):1441–1451. doi: 10.1172/JCI124606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Choudhury S, Baker S. Eosinophilic Esophagitis: the potential role of biologics in its treatment. Clin Rev Allergy Immunol. 2019:1–10. [DOI] [PubMed] [Google Scholar]
- 46.Rizzo A, Losacco A, Carratelli CR, Domenico MD, Bevilacqua N. Lactobacillus plantarum reduces Streptococcus pyogenes virulence by modulating the IL-17, IL-23 and toll-like receptor 2/4 expressions in human epithelial cells. Int Immunopharmacol. 2013;17(2):453–461. doi: 10.1016/j.intimp.2013.07.005 [DOI] [PubMed] [Google Scholar]
- 47.Zlotnik A, Yoshie O. The chemokine superfamily revisited. Immunity. 2012;36(5):705–716. doi: 10.1016/j.immuni.2012.05.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hernandez-Ruiz M, Zlotnik A. Mucosal chemokines. J Interferon Cytokine Res. 2017;37(2):62–70. doi: 10.1089/jir.2016.0076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gale N, Gnepp DR, Poljak M, et al. Laryngeal squamous intraepithelial lesions: an updated review on etiology, classification, molecular changes, and treatment. Adv Anat Pathol. 2016;23(2):84–91. doi: 10.1097/PAP.0000000000000106 [DOI] [PubMed] [Google Scholar]
- 50.Sasaki CT, Doukas SG, Costa J, Vageli DP. Biliary reflux as a causal factor in hypopharyngeal carcinoma: new clinical evidence and implications. Cancer. 2019;125(20):3554–3565. doi: 10.1002/cncr.32369 [DOI] [PubMed] [Google Scholar]
- 51.Galli J, Cammarota G, Volante M, De Corso E, Almadori G, Paludetti G. Laryngeal carcinoma and laryngo-pharyngeal reflux disease. Acta Otorhinolaryngol Ital. 2006;26(5):260–263. [PMC free article] [PubMed] [Google Scholar]
- 52.Lechien JR, Bobin F, Muls V, et al. Gastroesophageal reflux in laryngopharyngeal reflux patients: clinical features and therapeutic response. Laryngoscope. 2020;130(8):E479–E489. doi: 10.1002/lary.28482 [DOI] [PubMed] [Google Scholar]
- 53.Lechien JR, Akst LM, Hamdan AL, et al. Evaluation and management of laryngopharyngeal reflux disease: state of the art review. Otolaryngol Head Neck Surg. 2019;160(5):762–782. doi: 10.1177/0194599819827488 [DOI] [PubMed] [Google Scholar]
- 54.Lechien JR, Saussez S, Karkos PD. Laryngopharyngeal reflux disease: clinical presentation, diagnosis and therapeutic challenges in 2018. Curr Opin Otolaryngol Head Neck Surg. 2018;26(6):392–402. doi: 10.1097/MOO.0000000000000486 [DOI] [PubMed] [Google Scholar]
- 55.Bardhan KD, Strugala V, Dettmar PW. Reflux revisited: advancing the role of pepsin. Int J Otolaryngol. 2012;2012:646901. doi: 10.1155/2012/646901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Qadeer MA, Swoger J, Milstein C, et al. Correlation between symptoms and laryngeal signs in laryngopharyngeal reflux. Laryngoscope. 2005;115(11):1947–1952. doi: 10.1097/01.mlg.0000176547.90094.ac [DOI] [PubMed] [Google Scholar]
- 57.Hunt EB, Sullivan A, Galvin J, MacSharry J, Murphy DM. Gastric aspiration and its role in airway inflammation. Open Respir Med J. 2018;12:1–10. doi: 10.2174/1874306401812010001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bobin F, Journe F, Lechien JR. Saliva pepsin level of laryngopharyngeal reflux patients is not correlated with reflux episodes. Laryngoscope. 2019;130(5):1278–1281. [DOI] [PubMed] [Google Scholar]
- 59.Weldon D. Laryngopharyngeal reflux and chronic sinusitis. Curr Allergy Asthma Rep. 2007;7(3):197–201. doi: 10.1007/s11882-007-0072-5 [DOI] [PubMed] [Google Scholar]
- 60.Luo H-N, Yang Q-M, Sheng Y, et al. Role of pepsin and pepsinogen: linking laryngopharyngeal reflux with otitis media with effusion in children. Laryngoscope. 2014;124(7):E294–E300. doi: 10.1002/lary.24538 [DOI] [PubMed] [Google Scholar]
- 61.Abd El-Fattah AM, Abdul Maksoud GA, Ramadan AS, Abdalla AF, Abdel Aziz MM. Pepsin assay: a marker for reflux in pediatric glue ear. Otolaryngol Head Neck Surg. 2007;136(3):464–470. [DOI] [PubMed] [Google Scholar]
- 62.Magliulo G, Plateroti R, Plateroti AM. Gastroesophageal reflux disease and the presence of pepsin in the tears. Med Hypotheses. 2013;80(2):129–130. doi: 10.1016/j.mehy.2012.11.008 [DOI] [PubMed] [Google Scholar]
- 63.Watanabe M, Nakatani E, Yoshikawa H, et al. Oral soft tissue disorders are associated with gastroesophageal reflux disease: retrospective study. BMC Gastroenterol. 2017;17(1):92. doi: 10.1186/s12876-017-0650-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Warsi I, Ahmed J, Younus A, et al. Risk factors associated with oral manifestations and oral health impact of gastro-oesophageal reflux disease: a multicentre, cross-sectional study in Pakistan. BMJ Open. 2019;9(3):e021458. doi: 10.1136/bmjopen-2017-021458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Halstead LA. Role of gastroesophageal reflux in pediatric upper airway disorders. Otolaryngol Head Neck Surg. 1999;120(2):208–214. doi: 10.1016/S0194-5998(99)70408-0 [DOI] [PubMed] [Google Scholar]
- 66.Dinis PB, Subtil J. Helicobacter pylori and laryngopharyngeal reflux in chronic rhinosinusitis. Otolaryngol Head Neck Surg. 2006;134(1):67–72. doi: 10.1016/j.otohns.2005.10.013 [DOI] [PubMed] [Google Scholar]
- 67.Hanna BC, Wormald PJ. Gastroesophageal reflux and chronic rhinosinusitis. Curr Opin Otolaryngol Head Neck Surg. 2012;20(1):15–18. doi: 10.1097/MOO.0b013e32834e8f11 [DOI] [PubMed] [Google Scholar]
- 68.Wang L, Tan JJ, Wu T, et al. Association between laryngeal pepsin levels and the presence of vocal fold polyps. Otolaryngol Head Neck Surg. 2017;156(1):144–151. doi: 10.1177/0194599816676471 [DOI] [PubMed] [Google Scholar]
- 69.Chung JH, Tae K, Lee YS, et al. The significance of laryngopharyngeal reflux in benign vocal mucosal lesions. Otolaryngol Head Neck Surg. 2009;141(3):369–373. doi: 10.1016/j.otohns.2009.05.033 [DOI] [PubMed] [Google Scholar]
- 70.Adessa M, Xiao R, Hull D, et al. Benign vocal fold lesions in patients with chronic cough. Otolaryngol Head Neck Surg. 2020;162(3):322–325. doi: 10.1177/0194599819900505 [DOI] [PubMed] [Google Scholar]
- 71.Ali SA, Smith JD, Hogikyan ND. The white lesion, hyperkeratosis, and dysplasia. Otolaryngol Clin North Am. 2019;52(4):703–712. doi: 10.1016/j.otc.2019.03.014 [DOI] [PubMed] [Google Scholar]
- 72.Yakirevich E, Resnick MB. Pathology of gastric cancer and its precursor lesions. Gastroenterol Clin North Am. 2013;42(2):261–284. doi: 10.1016/j.gtc.2013.01.004 [DOI] [PubMed] [Google Scholar]
- 73.Arzumanyan A, Reis HM, Feitelson MA. Pathogenic mechanisms in HBV- and HCV-associated hepatocellular carcinoma. Nat Rev Cancer. 2013;13(2):123–135. doi: 10.1038/nrc3449 [DOI] [PubMed] [Google Scholar]
- 74.Souza RF. The role of acid and bile reflux in oesophagitis and barrett’s metaplasia. Biochem Soc Trans. 2010;38(2):348–352. doi: 10.1042/BST0380348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Parsel SM, Wu EL, Riley CA, McCoul ED. Gastroesophageal and laryngopharyngeal reflux associated with laryngeal malignancy: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2019;17(7):1253–1264.e1255. doi: 10.1016/j.cgh.2018.10.028 [DOI] [PubMed] [Google Scholar]
- 76.Ali MS, Parikh S, Chater P, Pearson JP. Bile acids in laryngopharyngeal refluxate: will they enhance or attenuate the action of pepsin? Laryngoscope. 2013;123(2):434–439. doi: 10.1002/lary.23619 [DOI] [PubMed] [Google Scholar]
- 77.Johnston N, Dettmar PW, Ondrey FG, Nanchal R, Lee SH, Bock JM. Pepsin: biomarker, mediator, and therapeutic target for reflux and aspiration. Ann N Y Acad Sci. 2018;1434(1):282–289. doi: 10.1111/nyas.13729 [DOI] [PubMed] [Google Scholar]
- 78.Hurley BP, Jugo RH, Snow RF, et al. Pepsin triggers neutrophil migration across acid damaged lung epithelium. Sci Rep. 2019;9(1):13778. doi: 10.1038/s41598-019-50360-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Johnston N, Wells CW, Blumin JH, Toohill RJ, Merati AL. Receptor-mediated uptake of pepsin by laryngeal epithelial cells. Ann Otol Rhinol Laryngol. 2007;116(12):934–938. doi: 10.1177/000348940711601211 [DOI] [PubMed] [Google Scholar]
- 80.O’Reilly RC, Soundar S, Tonb D, et al. The role of gastric pepsin in the inflammatory cascade of pediatric otitis media. JAMA Otolaryngol Head Neck Surg. 2015;141(4):350–357. doi: 10.1001/jamaoto.2014.3581 [DOI] [PubMed] [Google Scholar]
- 81.Sereg-Bahar M, Jerin A, Hocevar-Boltezar I. Higher levels of total pepsin and bile acids in the saliva as a possible risk factor for early laryngeal cancer. Radiol Oncol. 2015;49(1):59–64. doi: 10.2478/raon-2014-0020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Roh J-L, Lee Y-W, Park HT. Effect of acid, pepsin, and bile acid on the stenotic progression of traumatized subglottis. Am J Gastroenterol. 2006;101(6):1186–1192. doi: 10.1111/j.1572-0241.2006.00594.x [DOI] [PubMed] [Google Scholar]
- 83.Aldhahrani A, Powell J, Ladak S, et al. The potential role of bile acids in acquired laryngotracheal stenosis. Laryngoscope. 2018;128(9):2029–2033. doi: 10.1002/lary.27105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wong IWY, Rees G, Greiff L, Myers JC, Jamieson GG, Wormald P-J. Gastroesophageal reflux disease and chronic sinusitis: in search of an esophageal–nasal reflex. Am J Rhinol Allergy. 2010;24(4):255–259. doi: 10.2500/ajra.2010.24.3490 [DOI] [PubMed] [Google Scholar]
- 85.Sreebny LM. Saliva in health and disease: an appraisal and update. Int Dent J. 2000;50(3):140–161. doi: 10.1111/j.1875-595X.2000.tb00554.x [DOI] [PubMed] [Google Scholar]
- 86.Wood JM, Hussey DJ, Woods CM, et al. Does gene expression in laryngeal subsites differ between patients with laryngopharyngeal reflux and controls? Clin Otolaryngol. 2018;43(1):158–163. doi: 10.1111/coa.12918 [DOI] [PubMed] [Google Scholar]
- 87.Ho SB, Dvorak LA, Moor RE, et al. Cysteine-rich domains of muc3 intestinal mucin promote cell migration, inhibit apoptosis, and accelerate wound healing. Gastroenterology. 2006;131(5):1501–1517. doi: 10.1053/j.gastro.2006.09.006 [DOI] [PubMed] [Google Scholar]
- 88.Paleri V, Pearson JP, Bulmer D, Jeannon JP, Wight RG, Wilson JA. Expression of mucin gene products in laryngeal squamous cancer. Otolaryngol Head Neck Surg. 2004;131(1):84–88. doi: 10.1016/j.otohns.2003.04.001 [DOI] [PubMed] [Google Scholar]
- 89.Johnston N, Bulmer D, Gill GA, et al. Cell biology of laryngeal epithelial defenses in health and disease: further studies. Ann Otol Rhinol Laryngol. 2003;112(6):481–491. doi: 10.1177/000348940311200601 [DOI] [PubMed] [Google Scholar]
- 90.Axford SE, Sharp N, Ross PE, et al. Cell biology of laryngeal epithelial defenses in health and disease: preliminary studies. Ann Otol Rhinol Laryngol. 2001;110(12):1099–1108. doi: 10.1177/000348940111001203 [DOI] [PubMed] [Google Scholar]
- 91.Johnston N, Knight J, Dettmar PW, Lively MO, Koufman J. Pepsin and carbonic anhydrase isoenzyme III as diagnostic markers for laryngopharyngeal reflux disease. Laryngoscope. 2004;114(12):2129–2134. doi: 10.1097/01.mlg.0000149445.07146.03 [DOI] [PubMed] [Google Scholar]
- 92.Min HJ, Hong SC, Yang HS, Mun SK, Lee SY. Expression of CAIII and Hsp70 Is Increased the mucous membrane of the posterior commissure in laryngopharyngeal reflux disease. Yonsei Med J. 2016;57(2):469–474. doi: 10.3349/ymj.2016.57.2.469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Pero R, Coretti L, Nigro E, et al. β-defensins in the fight against Helicobacter pylori. Molecules. 2017;22(3):3. doi: 10.3390/molecules22030424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Gu J, Huang Y. Beta-defensin-2 is overexpressed in human vocal cord polyps. Eur Arch Otorhinolaryngol. 2017;274(2):901–907. doi: 10.1007/s00405-016-4270-4 [DOI] [PubMed] [Google Scholar]
- 95.Tomazic PV, Birner-Gruenberger R, Leitner A, Darnhofer B, Spoerk S, Lang-Loidolt D. Apolipoproteins have a potential role in nasal mucus of allergic rhinitis patients: a proteomic study. Laryngoscope. 2015;125(3):E91–E96. doi: 10.1002/lary.25003 [DOI] [PubMed] [Google Scholar]
- 96.Björkman EVC, Edebo A, Oltean M, Casselbrant A. Esophageal barrier function and tight junction expression in healthy subjects and patients with gastroesophageal reflux disease: functionality of esophageal mucosa exposed to bile salt and trypsin in vitro. Scand J Gastroenterol. 2013;48(10):1118–1126. doi: 10.3109/00365521.2013.828772 [DOI] [PubMed] [Google Scholar]
- 97.Asaoka D, Miwa H, Hirai S, et al. Altered localization and expression of tight-junction proteins in a rat model with chronic acid reflux esophagitis. J Gastroenterol. 2005;40(8):781–790. doi: 10.1007/s00535-005-1628-6 [DOI] [PubMed] [Google Scholar]
- 98.Li F-Y, Li Y. Interleukin-6, desmosome and tight junction protein expression levels in reflux esophagitis-affected mucosa. World J Gastroenterol. 2009;15(29):3621–3630. doi: 10.3748/wjg.15.3621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kim B, Lee HJ, Im NR, et al. Effect of matrix metalloproteinase inhibitor on disrupted E-cadherin after acid exposure in the human nasal epithelium. Laryngoscope. 2018;128(1):E1–E7. doi: 10.1002/lary.26932 [DOI] [PubMed] [Google Scholar]
- 100.Im N-R, Lee DY, Kim B, et al. Role of matrix metalloproteinases 7 in the pathogenesis of laryngopharyngeal reflux: decreased E-cadherin in acid exposed primary human pharyngeal epithelial cells. Int J Mol Sci. 2019;20(21):21. doi: 10.3390/ijms20215276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Reichel O, Mayr D, Durst F, Berghaus A. E-cadherin but not beta-catenin expression is decreased in laryngeal biopsies from patients with laryngopharyngeal reflux. Eur Arch Otorhinolaryngol. 2008;265(8):937–942. doi: 10.1007/s00405-007-0568-6 [DOI] [PubMed] [Google Scholar]
- 102.Wong SHM, Fang CM, Chuah LH, Leong CO, Ngai SC. E-cadherin: its dysregulation in carcinogenesis and clinical implications. Crit Rev Oncol Hematol. 2018;121:11–22. doi: 10.1016/j.critrevonc.2017.11.010 [DOI] [PubMed] [Google Scholar]
- 103.Solomon B, Young RJ, Rischin D. Head and neck squamous cell carcinoma: genomics and emerging biomarkers for immunomodulatory cancer treatments. Semin Cancer Biol. 2018;52(Pt 2):228–240. doi: 10.1016/j.semcancer.2018.01.008 [DOI] [PubMed] [Google Scholar]
- 104.Tan -J-J, Wang L, Mo -T-T, Wang J, Wang M-G, Li X-P. Pepsin promotes IL-8 signaling-induced epithelial-mesenchymal transition in laryngeal carcinoma. Cancer Cell Int. 2019;19:64. doi: 10.1186/s12935-019-0772-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Hu Y, Xu X, Xu L, Lai C, Zhang T. Dilated intercellular space in the larynx and esophagus of a rabbit reflux model. Auris Nasus Larynx. 2013;40(4):379–382. doi: 10.1016/j.anl.2012.09.010 [DOI] [PubMed] [Google Scholar]
- 106.Vaezi MF, Slaughter JC, Smith BS, et al. Dilated intercellular space in chronic laryngitis and gastro-oesophageal reflux disease: at baseline and post-lansoprazole therapy. Aliment Pharmacol Ther. 2010;32(7):916–924. doi: 10.1111/j.1365-2036.2010.04420.x [DOI] [PubMed] [Google Scholar]
- 107.Kizim Y 1, Zabolotnyi D, Kizim V 1, Zabolotna D 2, Sulaieva O 3. Laryngopharyngeal reflux affects tumour immune microenvironment in carcinoma of larynx. Ann Oncol. 2020;31(S4):252Volume. doi: 10.1016/j.annonc.2020.08.179 [DOI] [Google Scholar]
- 108.Parameswaran K, Anvari M, Efthimiadis A, Kamada D, Hargreave FE, Allen CJ. Lipid-laden macrophages in induced sputum are a marker of oropharyngeal reflux and possible gastric aspiration. Eur Respir J. 2000;16(6):1119–1122. doi: 10.1034/j.1399-3003.2000.16f17.x [DOI] [PubMed] [Google Scholar]
- 109.Gibeon D, Zhu J, Sogbesan A, et al. Lipid-laden bronchoalveolar macrophages in asthma and chronic cough. Respir Med. 2014;108(1):71–77. doi: 10.1016/j.rmed.2013.10.005 [DOI] [PubMed] [Google Scholar]
- 110.Chang AB, Cox NC, Purcell J, et al. Airway cellularity, lipid laden macrophages and microbiology of gastric juice and airways in children with reflux oesophagitis. Respir Res. 2005;6:72. doi: 10.1186/1465-9921-6-72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Krishnan U, Mitchell JD, Tobias V, Day AS, Bohane TD. Fat laden macrophages in tracheal aspirates as a marker of reflux aspiration: a negative report. J Pediatr Gastroenterol Nutr. 2002;35(3):309–313. doi: 10.1097/00005176-200209000-00013 [DOI] [PubMed] [Google Scholar]
- 112.Fishelevich R, Malanina A, Luzina I, et al. Ceramide-dependent regulation of human epidermal keratinocyte CD1d expression during terminal differentiation. J Immunol. 2006;176(4):2590–2599. doi: 10.4049/jimmunol.176.4.2590 [DOI] [PubMed] [Google Scholar]
- 113.Kohtz PD, Halpern AL, Eldeiry MA, et al. Toll-like receptor-4 is a mediator of proliferation in Esophageal Adenocarcinoma. Ann Thorac Surg. 2019;107(1):233–241. doi: 10.1016/j.athoracsur.2018.08.014 [DOI] [PubMed] [Google Scholar]
- 114.Zaidi AH, Kelly LA, Kreft RE, et al. Associations of microbiota and toll-like receptor signaling pathway in esophageal adenocarcinoma. BMC Cancer. 2016;16:52. doi: 10.1186/s12885-016-2093-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hackett AP, Trinick RE, Rose K, Flanagan BF, McNamara PS. Weakly acidic pH reduces inflammatory cytokine expression in airway epithelial cells. Respir Res. 2016;17(1):82. doi: 10.1186/s12931-016-0399-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Hirano T. Interleukin 6 in autoimmune and inflammatory diseases: a personal memoir. Proc Jpn Acad Ser B Phys Biol Sci. 2010;86(7):717–730. doi: 10.2183/pjab.86.717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Oh DS, DeMeester SR, Vallbohmer D, et al. Reduction of interleukin 8 gene expression in reflux esophagitis and Barrett’s esophagus with antireflux surgery. Arch Surg. 2007;142(6):554–559; discussion 559–560. doi: 10.1001/archsurg.142.6.554 [DOI] [PubMed] [Google Scholar]
- 118.Parker NP. Vocal fold leukoplakia: incidence, management, and prevention. Curr Opin Otolaryngol Head Neck Surg. 2017;25(6):464–468. doi: 10.1097/MOO.0000000000000406 [DOI] [PubMed] [Google Scholar]
- 119.O’Shea KM, Aceves SS, Dellon ES, et al. Pathophysiology of Eosinophilic Esophagitis. Gastroenterology. 2018;154(2):333–345. doi: 10.1053/j.gastro.2017.06.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Solidoro P, Patrucco F, Fagoonee S, Pellicano R. Asthma and gastroesophageal reflux disease: a multidisciplinary point of view. Minerva Med. 2017;108(4):350–356. doi: 10.23736/S0026-4806.17.05181-3 [DOI] [PubMed] [Google Scholar]
- 121.Eryuksel E, Dogan M, Golabi P, Sehitoglu MA, Celikel T. Treatment of laryngopharyngeal reflux improves asthma symptoms in asthmatics. J Asthma. 2006;43(7):539–542. doi: 10.1080/02770900600857234 [DOI] [PubMed] [Google Scholar]
- 122.Ridley C, Thornton DJ. Mucins: the frontline defence of the lung. Biochem Soc Trans. 2018;46(5):1099–1106. doi: 10.1042/BST20170402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Rose MC, Voynow JA. Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol Rev. 2006;86(1):245–278. doi: 10.1152/physrev.00010.2005 [DOI] [PubMed] [Google Scholar]
- 124.Utech M, Mennigen R, Bruewer M. Endocytosis and recycling of tight junction proteins in inflammation. J Biomed Biotechnol. 2010;2010:484987. doi: 10.1155/2010/484987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Suzuki T, Yoshinaga N, Tanabe S. Interleukin-6 (IL-6) regulates claudin-2 expression and tight junction permeability in intestinal epithelium. J Biol Chem. 2011;286(36):31263–31271. doi: 10.1074/jbc.M111.238147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Douglas College Human Anatomy & Physiology II. Douglas College, New Westminster BC. Aug 31, 2017. [Google Scholar]