Abstract
Purpose
Mortality in infective endocarditis (IE) is still high, and the long term prognosis remains uncertain. This study aimed to identify predictors of long-term mortality for any cause, adverse event rate, relapse rate, valvular and ventricular dysfunction at follow-up, in a real-world surgical centre.
Methods
We retrospectively analyzed 363 consecutive episodes of IE (123 women, 34%) admitted to our department with a definite diagnosis of non-device-related IE. Median follow-up duration was 2.9 years. Primary endpoints were predictors of mortality, recurrent endocarditis, and major non-fatal adverse events (hospitalization for any cardiovascular cause, pace-maker implantation, new onset of atrial fibrillation, sternal dehiscence), and ventricular and valvular dysfunction at follow-up.
Results
Multivariate analysis independent predictors of mortality showed age (HR per unit 1.031, p < 0.003), drug abuse (HR 3.5, p < 0.002), EUROSCORE II (HR per unit 1.017, p < 0.0006) and double valve infection (HR 2.3, p < 0.001) to be independent predictors of mortality, while streptococcal infection remained associated with a better prognosis (HR 0.5, p < 0.04). Major non-fatal adverse events were associated with age (HR 1.4, p < 0.022). New episodes of infection were correlated with S aureus infection (HR 4.8, p < 0.001), right-sided endocarditis (HR 7.4, p < 0.001), spondylodiscitis (HR 6.8, p < 0.004) and intravenous drug abuse (HR 10.3, p < 0.001). After multivariate analysis, only drug abuse was an independent predictor of new episodes of endocarditis (HR 8.5, p < 0.001). Echocardiographic follow-up, available in 95 cases, showed a worsening of left ventricular systolic function (p < 0.007); severe valvular dysfunction at follow-up was reported only in 4 patients, all of them had mitral IE (p < 0.03).
Conclusions
The present study highlights some clinical, readily available factors that can be useful to stratify the prognosis of patients with IE.
Keywords: Infective endocarditis, Prognostic factors, Outcome, Valvular dysfunction, Endocarditis, Mortality, Prognosis
Background
Despite recent diagnostic and therapeutic advances, the mortality of IE remains high in most series, with in-hospital mortality even of 24% [1], and three-year mortality over 30% [2]. The literature reports some prognostic factors associated with higher mortality, such as advanced age, female gender [1–3], prosthetic valve endocarditis [4], Staphylococcus aureus aetiology [5], comorbidity [6–12], leucocytosis, hypoalbuminemia, C-reactive protein levels, elevated ERS [13], and IE complications [6].
Most published series are retrospective studies, each focusing on specific aspects. A multicenter cohort study evaluated predictors of hospital re-admissions in patients with infective endocarditis [14], one study included only patients with prosthetic valve IE [15], and another study included only surgically treated patients [16]. Still, there is a lack of consensus about which characteristics individuate patients with an adverse prognostic profile. The epidemiology of the disease has changed in recent decades. A progressive increase of the average patient age, a higher prevalence of cardiac devices and prostheses, and a rising incidence of nosocomial or procedure-related endocarditis have changed the microbiologic yield of the disease [17]. Therefore, an update on prognostic factors of infective endocarditis is necessary to consider the contemporary epidemiologic metamorphosis of this pathology. An accurate risk stratification could individuate patients who might benefit from a more aggressive strategy. This study aimed to investigate factors associated with a worse prognosis in a referral surgical centre.
Methods
Patient selection
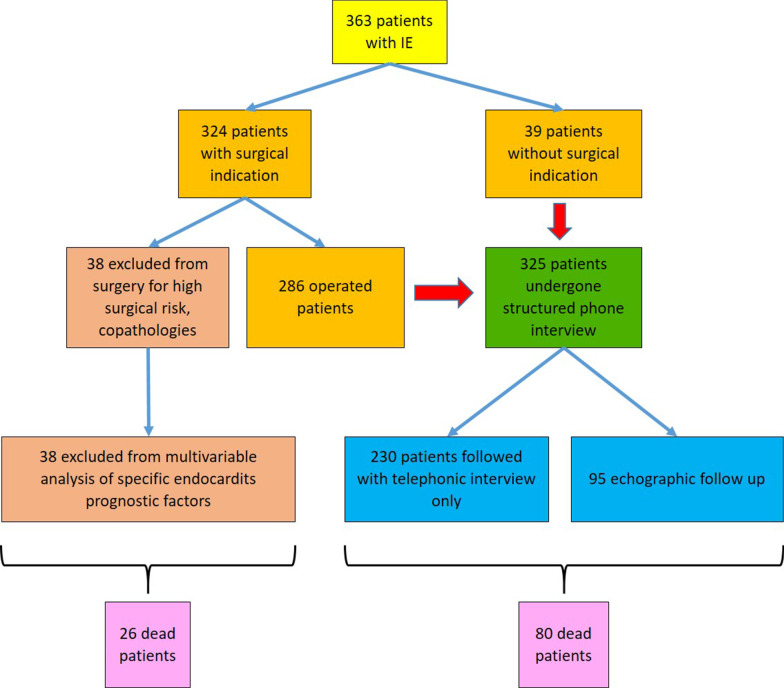
We have created a local registry of patients affected by non-device-related IE, collecting retrospectively all the cases admitted to our department between January 2013 and December 2019 with a definite diagnosis of IE. Device-related IE was defined as an infection on cardiac devices, implants and grafts, other than valve prosthesis, documented by echocardiography. We excluded all patients with electro-catheter vegetation or cutaneous infection over the pocket area of the implanted device. The registry comprises 363 patients and 294 variables. We got data for analysis from electronic hospital charts and wholly anonymized them, as reported elsewhere [18]. The local Ethics Committee approved the study. We followed the current international IE guidelines for diagnostic work-up and treatment strategies [19]. Of the 363 patients with IE, 39 received only medical therapy because of the absence of surgical indication, 286 underwent surgical intervention associated with antibiotic treatment, and 38 were excluded from surgery despite surgical indication because of prohibitive general conditions. We excluded the latter group from the multivariate analysis of prognostic factors specific of IE since it identifies patients with extremely compromised conditions aside from IE, and their prognosis depended on critical basal conditions more than on IE itself (Fig. 1). Multivariate analysis of mortality was adjusted for the treatment received: medical therapy only, early surgery, or delayed surgery. We excluded procalcitonin and TAPSE from multivariate analysis, because of a high percentage of missing values (22 and 26% respectively). Variables included in the multivariate analysis were gender, age, diabetes, renal failure, history of drug abuse, the microbiologic agent involved, ejection fraction, the type of valve (native or prosthetic), double valve infection, the paravalvular extension of infection, severe valvular dysfunction, oral anticoagulant therapy, brain embolism at admission, EUROSCORE II and the presence of a pacemaker. We defined renal failure as GFR < 60 ml/min/1.73 m2.
Fig. 1.
Flow chart of patient selection
Follow-up
We calculated follow-up duration from the time of IE diagnosis. A structured phone interview updated the follow-up of all patients to July 2020. In a subset of 95 cases of the 325 patients included in the multivariate analysis, both echocardiographic and clinical evaluation were available (Fig. 1). Median follow-up duration was 2.9 years (SD 1.5), minimum 0.3, maximum 7 years.
Study endpoints
Primary endpoints were predictors of mortality, recurrent endocarditis, major non-fatal adverse events (hospitalization for any cardiovascular cause, pace-maker implantation, new onset of atrial fibrillation, sternal dehiscence), worsening of left ventricular ejection fraction (LVEF), and valvular dysfunction during follow-up.
Statistical analysis
We used the chi-square and the Mann–Whitney or Kruskal–Wallis tests to compare respectively proportions and continuous variables with normal or non-normal distributions. We performed univariate and multivariate analyses using logistic regression and general linear models. We used the Kaplan–Meier method to estimate the univariate cumulative incidence of events and event-free survival. All tests were 2-sided, and statistical significance was defined as a p-value < 0.05. We performed the analyses with SPSS 23.0 and R 3.6.3.
Results
Patient characteristics
We included 363 consecutive episodes of IE (123 women, 34%). Median follow-up was three years (0.3–7). We summarized the principal clinical and demographic characteristics of the 363 patients in Table 1. We reported in Table 2 the underlying valvular disease and the degree of valvular dysfunction.
Table 1.
Demographic, clinical and echocardiographic characteristics of 363 patients with IE
| Demographic and clinical characteristics of 363 patients with IE | |
|---|---|
| Age (years), mean ± SD | 65 ± 15 |
| Gender (women), n (%) | 123 (34) |
| BMI, mean ± SD | 25.0 ± 4.1 |
| Renal failure, n (%) | 96 (26) |
| Mild n (%) | 41 (43) |
| Moderate n (%) | 34 (35) |
| Severe n (%) | 14 (15) |
| On dialysis n (%) | 7 (7) |
| Arterial hypertension, n (%) | 212 (59) |
| Previous malignancies, n (%) | 79 (22) |
| Drug abuse, n (%) | 43 (12) |
| Diabetes, n (%) | 68 (18) |
| Dyslipidemia, n (%) | 109 (30) |
| Pacemaker, n (%) | 41 (11) |
| Oral anticoagulant therapy, n (%) | 91 (25) |
| Clinical and echocardiographic characteristics of 363 episodes of IE | |
| Prosthetic valve, n (%) | 132 (36) |
| First episode of IE, n (%) | 330 (90) |
| Systemic embolism, n (%) | 141 (39) |
| Cerebral embolism, n (%) | 67 (47) |
| Perivalvular extension, n (%) | 84 (23) |
| Severe valvular dysfunction, n (%) | 172 (47) |
| Vegetation length (mm), mean ± SD | 9.1 ± 7.6 |
| Ejection Fraction (%), mean ± SD | 56.7 ± 10.0 |
| TAPSE (mm), mean ± SD | 20.8 ± 5.6 |
| Euroscore II | 12.4 ± 16.5 |
Table 2.
Underlying valvular disease in 363 episodes of IE
| Type of valve n (%) | Site of infection n (%) | Degree of valvular dysfunction (stenosis/regurgitation) | ||
|---|---|---|---|---|
| Absent n (%) | Mild or moderate n (%) | Severe n (%) | ||
|
Native 231 (63%) |
Aortic 106 (29%) | 17 (5) | 30 (8) | 59 (16) |
| Mitral 108 (29%) | 10 (3) | 34 (9) | 64 (17) | |
| Tricuspid 17 (5%) | 2 (1) | 6 (2) | 9 (2) | |
|
Prosthetic 132 (37%) |
Aortic 87 (24%) | 23 (6) | 38 (11) | 26 (7 |
| Mitral 39 (11%) | 8 (2) | 19 (5) | 12 (3) | |
| Tricuspid 6 (2%) | 0 (0) | 4 (2) | 2 (1) | |
Microbiology
Blood cultures were positive in 80% of cases. We summarized the microbiologic yield in Table 3. A presumable bacterial access location was present in 241 (66%) patients. The access location was cutaneous in 71 patients (29%), nosocomial in 57 (24%), gastroenteric in 51 (21%), urinary in 34 (14%), and oropharyngeal in 28 (12%). Since the prevalence of streptococcal infection was 16%, oropharyngeal access was probably underdiagnosed.
Table 3.
Microbiological yield of blood cultures in 363 episodes of IE
| Etiologic agent | N (%) |
|---|---|
| Streptococcus Viridans | 62 (17) |
| Streptococcus Bovis | 25 (7) |
| Staphylococcus Aureus | 64 (18) |
| Negative Coagulase Staphylococci | 46 (13) |
| Enterococci | 65 (18) |
| Other | 27 (7) |
| Negative cultures | 74 (20) |
| Total | 363 (100) |
Surgical treatment
A total of 286 (79%) episodes of IE underwent surgical intervention; 246 (86%) underwent valve replacement and 40 valve repair (14%). Seventy-seven (21%) patients were treated conservatively, 39 due to the absence of surgical indication, and 38 due to prohibitive surgical risk. Surgery was performed within 14 days from diagnosis in 222 (78%) patients and later in the remaining 64 (22%); in these patients, surgery was delayed because of neurologic complications or a delayed referral from peripheral hospitals.
The purpose of this study was to evaluate prognostic factors at baseline, rather than studying a specific treatment or therapeutic strategy. We didn’t deepen comparison among medical therapy, early surgery and delayed surgery. On the other hand, exclusion from surgery in patients with surgical indication had disproportionately high mortality: in this subgroup, mortality at 30 days extrapolated from Kaplan–Meier curves was 33.8% (95% CI 17–47.1), and one-year mortality was 64.9% (95% CI 46.1–77.2). Indeed, these patients had prohibitive general conditions, aside from IE. As stated in the method section, we, therefore, excluded this subgroup of patients from the multivariate analysis of mortality.
Surgical intervention was performed less frequently in women (p < 0.027), elderly (p < 0.037) and in patients with a low ejection fraction (p < 0.001); at multivariate analysis female gender was an independent factor for exclusion from surgery for absence of surgical indications (OR 0.44, 95% CI 0.21–0.91, p < 0.028), while ejection fraction (OR per increasing unit 0.94, 95% CI 0.91–0.97, p < 0.001) and age (OR per increasing unit 1.05, 95% CI 1.02–1.08, p < 0.001) were independent predictors for exclusion from surgery when indicated, with an inverse and direct relation respectively.
Mortality
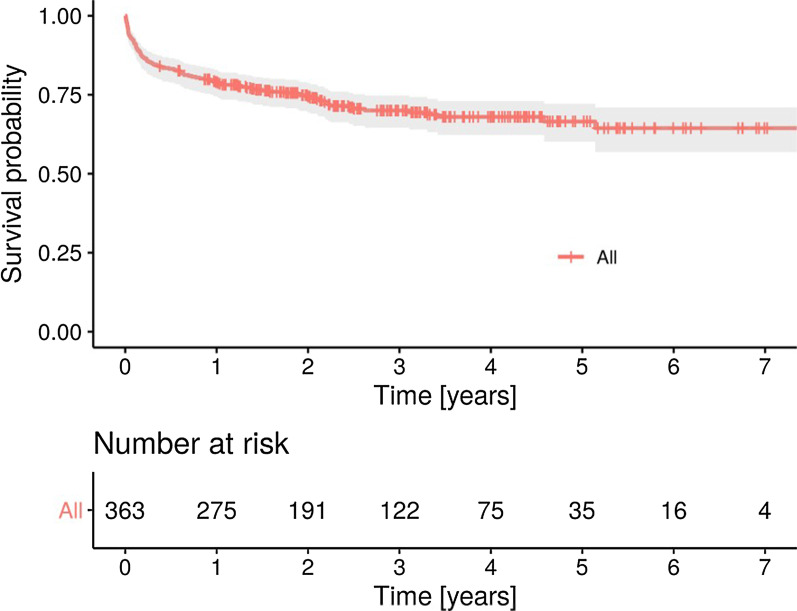
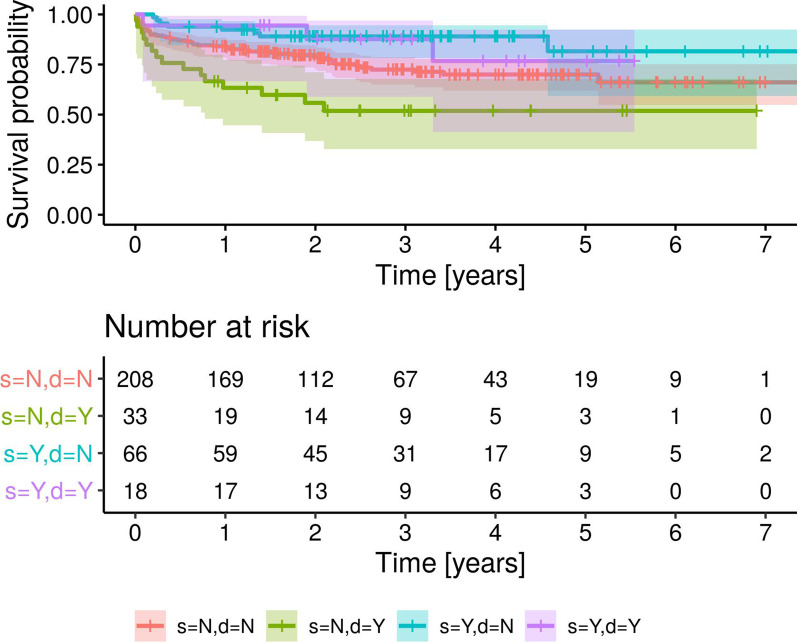
After extrapolation from Kaplan–Meier curves, overall all-cause thirty-day mortality was 7.7% (95% CI 4.9–10.4), and all-cause three-year mortality was 29.9% (95% CI 24.7–34.8) (Fig. 2). We reported the predictors of death for any cause at univariate analysis in Table 4. The independent predictors of mortality were age, drug abuse, EUROSCORE II and double valve infection, while streptococcal infection confirmed to be associated with a better prognosis (Table 4). Given a type I error rate of 0.05, the detected differences had a power level > 0.8, except for the association of streptococcal infection with lower mortality, that was characterized by a power of 0.5. Figure 3 shows the multivariate survival curves of double valve infection and streptococcal infection.
Fig. 2.
Kaplan–Meier analysis of survival probability of 363 patients with infective endocarditis
Table 4.
Univariate and multivariate analysis of predictors of mortality for any cause
| HR | 95% CI | p | ||
|---|---|---|---|---|
| Univariate analysis (n = 363) | ||||
| Female gender | 1.2 | 1.0 | 1.4 | 0.023 |
| Age > 70 years | 2.1 | 1.4 | 3.2 | 0.001 |
| Euroscore II > 10 | 3.1 | 2.1 | 4.6 | 0.001 |
| Chronic renal failure | 1.9 | 1.3 | 2.9 | 0.001 |
| Diabetes | 1.7 | 1.1 | 2.7 | 0.009 |
| Oral anticoagulant therapy | 1.7 | 1.2 | 2.7 | 0.005 |
| Presence of pacemaker | 2.2 | 1.4 | 3.6 | 0.001 |
| Ejection fraction < 40% | 3.1 | 1.8 | 5.4 | 0.001 |
| TAPSE < 19 | 2.4 | 1.5 | 3.9 | 0.001 |
| Double valve infection | 1.8 | 1.2 | 2.9 | 0.007 |
| Prosthetic infection | 1.7 | 1.1 | 2.5 | 0.015 |
| Procalcitonin levels > 0.22 mg/dl | 1.9 | 1.2 | 3.0 | 0.003 |
| PCR > 83 mg/dl | 1.6 | 1.0 | 2.5 | 0.037 |
| Enterococcal infection | 1.6 | 1.0 | 2.5 | 0.022 |
| Streptococcal infection | 0.34 | 0.1 | 0.6 | 0.002 |
| Exclusion from surgery if indicated | 7.4 | 3.2 | 17.3 | 0.001 |
| Multivariate analysis (n = 325) | ||||
| Age* | 1.033 | 1.01 | 1.056 | 0.003 |
| Drug abuse | 3.40 | 1.41 | 8.13 | 0.006 |
| EUROSCORE II* | 1.017 | 1.009 | 1.025 | 0.0006 |
| Double valve infection | 2.07 | 1.21 | 3.54 | 0.008 |
| Streptococcal infection | 0.49 | 0.26 | 0.92 | 0.028 |
*HR per unit
Fig. 3.
Multivariate Kaplan–Meier analysis of survival probability of 325 patients with infective endocarditis with (Y) or without (N) double valve infection (d) and with (Y) or without (N) Streptococcal infection (s)
Major non-fatal adverse event rate and new infection rate
During follow-up, 112 (34%) patients had had one or more major non-fatal adverse events. Age, hypertension, and history of cancer predicted major non-fatal adverse events at three years. At multivariate analysis, only age was independently associated with major non-fatal adverse events (Table 5). Nineteen patients (6%) had one or more recurrent episodes of IE. S aureus infection, right-sided endocarditis, spondylodiscitis, and drug abuse predicted recurrent infection. After multivariate analysis, only drug abuse was an independent predictor of new episodes of endocarditis (Table 5).
Table 5.
Univariate and multivariate analysis of predictors of major non-fatal adverse events and episodes of recurrent infective endocarditis
| HR | 95% CI | p | ||
|---|---|---|---|---|
| Major non-fatal adverse events (112 patients), univariate analysis | ||||
| Age | 1.02 | 1.00 | 1.03 | 0.001 |
| Hypertension | 1.46 | 1.06 | 2.03 | 0.02 |
| History of cancer | 1.56 | 1.11 | 2.20 | 0.009 |
| Major non-fatal adverse events (112 patients), multivariate analysis | ||||
| Age | 1.49 | 1.06 | 2.118 | 0.022 |
| Recurrent episodes of IE (19 patients), univariate analysis | ||||
| Staphylococcus aureus infection | 4.8 | 1.9 | 11.9 | 0.001 |
| Right-sided endocarditis | 7.4 | 2.9 | 18.9 | 0.001 |
| Spondylodiscitis | 6.8 | 2.6 | 17.8 | 0.004 |
| Drug abuse | 10.3 | 4.1 | 25.6 | 0.001 |
| Recurrent episodes of IE (19 patients), multivariate analysis | ||||
| Drug abuse | 8.5 | 2.3 | 31 | 0.001 |
Echocardiographic follow-up
We performed echocardiographic follow-up in a subset of 95 patients. We used the PISA method for quantitative assessment of valve regurgitation and the Simpson method from the apical 4-chamber view for the quantitative evaluation of Left ventricular ejection fraction (LVEF). We reported clinical and echocardiographic data of this subset of patients in Table 6. The subset of patients undergone echographic follow-up is not clinically representative of the whole cohort, since patients willing to come to the visit were younger and had better general conditions (synthetically expressed by EUROSCORE II at diagnosis). On the other hand, echocardiographic parameters were not significantly different between the two groups (Table 7), and results are extensible to the entire cohort. The average EF at baseline was 59.8 (SD 8.13) while the follow-up EF was 57.46 (SD 8.88); the EF distributions are not normal (Shapiro test p = 0.036 and p = 0.003). These differences are not significant considering baseline and follow-up measures as independent (Mann–Whitney test, p < 0.093), while they are significant when considering repeated measures (Wilcoxon signed-rank test, p < 0.007). Predictors of worsening EF will be the object of investigation in a thorough study dedicated to echocardiographic follow-up. Only four patients reported severe valvular dysfunction at follow-up, all of them had mitral IE (p < 0.03), one treated conservatively because of refusal of surgery, one undergone valve repair, and two valve replacement.
Table 6.
Clinical and echocardiographic data of patients undergone to echocardiographic follow-up (n = 95)
| Site of infection | p | |||
|---|---|---|---|---|
| Aortic (n = 50) | Mitral (n = 39) | Tricuspid (n = 6) | ||
| Gender (women), n (%) | 9 (18) | 7 (18) | 0 (0) | 0.5 |
| Age (years), mean ± SD | 61.5 ± 14.8 | 60.3 ± 12.2 | 61.6 ± 12.5 | 0.7 |
| Severe valvular dysfunction, n (%) | 23 (46) | 25 (64) | 3 (50) | 0.2 |
| Paravalvular extension, n (%) | 13 (26) | 5 (13) | 0 (0) | 0.1 |
| Vegetation length (mm), mean ± SD | 7.7 ± 6.2 | 10.2 ± 7.9 | 26.3 ± 13.3 | 0.008 |
| Second infected valve, n (%) | 4 (8) | 5 (13) | 1 (17) | 0.6 |
| Ejection Fraction (%), mean ± SD | 59.1 ± 8.0 | 60.9 ± 8.5 | 58.3 ± 6.6 | 0.5 |
| TAPSE (mm), mean ± SD | 20.8 ± 6.2 | 23.2 ± 5.3 | 21.2 ± 6.9 | 0.2 |
| Euroscore II | 7.3 ± 6.3 | 5.6 ± 9.1 | 4.3 ± 2.5 | 0.035 |
| Surgery, n (%) | 42 (84) | 34 (87) | 6 (100) | 0.5 |
| Ejection Fraction (%) at follow-up, mean ± SD | 58.9 ± 8.3 | 55.9 ± 9.1 | 55.1 ± 11.0 | 0.2 |
| Severe valvular dysfunction at follow-up, n (%) | 0 (0) | 4 (10) | 0 (0) | 0.03 |
Table 7.
Comparison between patients undergone or not to echocardiographic follow-up
| Echocardiographic follow-up | No (n = 268) | Yes (n = 95) | p value |
|---|---|---|---|
| Female gender, n (%) | 107 (40) | 16 (17) | < 0.001 |
| Age (years), mean ± SD | 66.8 ± 15.1 | 61.0 ± 13.5 | < 0.001 |
| Euroscore II, mean ± SD | 14.5 ± 18.2 | 6.4 ± 7.5 | < 0.001 |
| Site of infection | NS | ||
| Aortic | 143 (53) | 50 (53) | NS |
| Mitral | 108 (40) | 41 (41) | NS |
| Tricuspidal | 17 (7) | 6 (6) | NS |
| Ejection Fraction (%), mean ± SD | 55.6 ± 10.3 | 59.8 ± 8.1 | NS |
| TAPSE (mm), mean ± SD | 20.3 ± 5.4 | 21.9 ± 5.9 | NS |
| Vegetation length (mm), mean ± SD | 8.8 ± 7.2 | 9.9 ± 8.6 | NS |
Discussion
The three-year mortality rate of IE in the literature often exceeds 30% [2]. The overall mortality rate at three years in our series was 29%. We found several, commonly available predictors of higher mortality in endocarditis, that can be useful for clinicians to stratify patient prognosis better. The principal adverse prognostic factors belong to three categories: clinical features, echocardiographic parameters, and microbiologic aetiology.
Clinical features
Clinical characteristics associated with an adverse prognosis were female gender, age > 70 years, EUROSCORE II, chronic renal failure, diabetes, and drug abuse. Gender differences are attributable to a variety of factors, including comorbidities or inherent physiologic differences [1]. IE occurs in males more frequently, with a 2:1 to 9:1 ratio. Estrogens may play a protective role against endothelial damage. Moreover, females tend to encounter heart disease at an older age and have a higher incidence of comorbid conditions, which may result in a worse outcome [2, 3]. Deepening the analysis, we found that the female gender was not an independent predictor of adverse prognosis, as opposed to age and EUROSCORE II, that confirmed to be independent predictors at multivariate analysis. Despite the availability of recent IE-specific scores and considering the trade-off between the indexes, the logistic EUROSCORE II seemed to be the best predictor of mortality risk in the literature [5–8]. Relatively to age, the epidemiology of IE patients has changed. Currently, most of IE patients are elderly, frail, and with multiple comorbidities [9–12]. Drug addiction is also an independent predictor of adverse prognosis in IE and the major risk factor for recurring IE, consistently with other studies [13–21].
Echocardiographic parameters
Echocardiographic predictors of a worse prognosis were double valve infection, ejection fraction < 40%, and prosthetic infection. The value of echocardiographic findings in predicting outcome in infective endocarditis is emergent [22]. Heart failure is associated with a worse outcome in IE [15, 23], as well as echocardiographic parameters showing right or left ventricular dysfunction [24–26]. Prosthetic infection is a known adverse prognostic factor, both for biological and mechanical valves [16, 19]. Double valve endocarditis is a rare condition that is independently associated with an adverse outcome. This association was previously described in surgical series [27, 28], while we found this is a marker of worse outcome independently of surgery. The mechanisms of the spread of the infection differ whether endocarditis is only left-sided or bilateral (also involving tricuspid valve). Left-sided bivalvular endocarditis is often due to a secondary mitral lesion following primary aortic endocarditis. Multivalvular endocarditis is frequently responsible for severe heart failure. Coherently with our data, the study of Sulton-Suty proofs the importance of early diagnosis of endocarditis to avoid the spread of the infection to more than one valve [29]. The involvement of multiple valves leads to a higher rate of complications and thus, mortality. Moreover, biventricular IE is an uncommon condition with no specific guidelines for treatment [30].
Echocardiographic follow-up showed a mild but significant average worsening of left ventricular systolic function; we will address predictors of lowering of LVEF in an incoming study.
Microbiologic aetiology
Finally, microbiologic aetiology can identify patients with different prognosis. Enterococcal infection is associated with a worse outcome, while streptococcal IE seems to be protective. Chirouze’s study [31] found that enterococcal IE is healthcare-associated in 25% of cases and involves prosthetic valves in 30%. In our series, the enterococcal infection had a worse prognosis but was not an independent predictor of mortality at multivariate analysis, reflecting the specific epidemiology of this germ. On the other hand, Streptococcal infection was independently associated with a better prognosis, suggesting a less aggressive infection of this pathogen. These data emphasize the prognostic relevance of microorganism characterization in IE.
Our study has some limitations: first, changes in the clinical management of IE may have occurred during the long study period. Second, it is a real-world single-centre experience. Finally, our study has a potential referral bias, since we have conducted it in a high-volume surgical centre; therefore, the percentage of patients with a surgical indication in disproportionately high.
Conclusions
In our series, the independent predictors of mortality were age, drug abuse, EUROSCORE II, and double valve infection, while the streptococcal infection had a better prognosis. Age predicted major non-fatal adverse events. Drug abuse was the only independent predictor of new episodes of endocarditis. Our results demonstrate some readily available parameters that identify IE patients at an increased risk of adverse prognosis. Accurate stratification of the prognosis could orient towards more aggressive management in selected patients.
Acknowledgements
Authors are grateful to ‘Fondazione A.R.Card Onlus’ for its unconditional support.
Authors’ contributions
VS projected the study, analyzed and interpreted the data and wrote the manuscript. IM collected the data for analysis and contributed writing the manuscript. RM revised the manuscript. NZ carried out the statistical analysis. PLS performed the surgical interventions, and was a major contributor in writing the manuscript. NM and IO revised the final version of the manuscript. SDP, NC, VA and BA performed clinical follow-up and contributed collecting the data for analysis. All authors read and approved the final manuscript.
Funding
None to declare.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The study was approved by the local ethics Committee (Comitato Etico Regionale per la Sperimentazione Clinica della Regione Toscana, Sezione: AREA VASTA CENTRO, n 12113_oss), that, in keeping with statements by the Italian Regulatory Authorities for retrospective, observational studies (https://www.garanteprivacy.it/web/guest/home/docweb/-/docweb-display/docweb/5805552), granted a waiver of informed consent from study participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gabbieri D, Dohmen PM, Linneweber J, Grubitzsch H, von Heymann C, Neumann K, et al. Early outcome after surgery for active native and prosthetic aortic valve endocarditis. J Heart Valve Dis. 2008;17(5):508–524. [PubMed] [Google Scholar]
- 2.Dohmen PM, Binner C, Mende M, Daviewala P, Etz CD, Borger MA, et al. Gender-based long-term surgical outcome in patients with active infective aortic valve endocarditis. Med Sci Monit. 2016;22:2520–2527. doi: 10.12659/MSM.899360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Elamragy AA, Meshaal MS, El-Kholy AA, Rizk HH. Gender differences in clinical features and complications of infective endocarditis: 11-year experience of a single institute in Egypt. Egypt Heart J. 2020;72(1):5. doi: 10.1186/s43044-020-0039-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McDonald JR. Acute infective endocarditis. Infect Dis Clin North Am. 2009;23(3):643–664. doi: 10.1016/j.idc.2009.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Selton-Suty C, Hoen B, Grentzinger A. Clinical and bacteriological characteristics of infective endocarditis. Heart (British Cardiac Society) 1997;77(3):260–263. doi: 10.1136/hrt.77.3.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hoen B, Duval X. Infective endocarditis. N Engl J Med. 2013;368:1425–1433. doi: 10.1056/NEJMcp1206782. [DOI] [PubMed] [Google Scholar]
- 7.Nashef SAM, Roques F, Sharples LD, Nilsson J, Smith C, Goldstonee AR, et al. EuroSCORE II. Eur J Cardiothorac Surg. 2012;41(4):734–744. doi: 10.1093/ejcts/ezs043. [DOI] [PubMed] [Google Scholar]
- 8.Pivatto Júnior F, Bellagamba CCA, Pianca EG, Fernandes FS, Butzke M, Busato SB, et al. Analysis of risk scores to predict mortality in patients undergoing cardiac surgery for endocarditis. Arq Bras Cardiol. 2020;114(3):518–524. doi: 10.36660/abc.20190050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Thuny F, Grisoli D, Collart F, Habib G, Raoult D. Management of infective endocarditis: challenges and perspectives. Lancet. 2012;379:965–975. doi: 10.1016/S0140-6736(11)60755-1. [DOI] [PubMed] [Google Scholar]
- 10.Cahill TJ, Baddour LM, Habib G, Hoen B, Salaun E, Pettersson GB. Challenges in infective endocarditis. JACC. 2017;69:325–344. doi: 10.1016/j.jacc.2016.10.066. [DOI] [PubMed] [Google Scholar]
- 11.Delamaire M, Maugendre D, Moreno M, Le Goff MC, Allannic H, Genetet B. Impaired leucocyte functions in diabetic patients. Diabet Med. 1997;14(1):29–34. doi: 10.1002/(SICI)1096-9136(199701)14:1<29::AID-DIA300>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 12.Benvenga RM, De Rosa R, Silverio A, Matturro R, Zambrano C, Masullo A. Infective endocarditis and diabetes mellitus: results from a single-center study from 1994 to 2017. PLoS ONE. 2019;14(11):e0223710. doi: 10.1371/journal.pone.0223710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cornelissen CG, Frechen DA, Schreiner K, Marx N, Krüger S. Inflammatory parameters and prediction of prognosis in infective endocarditis. BMC Infect Dis. 2013;13:272. doi: 10.1186/1471-2334-13-272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Agrawal A, Virk HUH, Riaz I, Jain D, Tripathi B, Krittanawong C, et al. Predictors of 30-day re-admissions in patients with infective endocarditis: a national population based cohort study. Rev Cardiovasc Med. 2020;21(1):123–127. doi: 10.31083/j.rcm.2020.01.552. [DOI] [PubMed] [Google Scholar]
- 15.Emrecan B, Taştan H, Şimşek Ş, Çekirdekoğlu K. Prosthetic valve endocarditis: a challenging complication of prosthetic valves. Turk Gogus Kalp Damar Cerrahisi Derg. 2019;27(2):159–164. doi: 10.5606/tgkdc.dergisi.2019.16796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ilhão Moreira R, Coutinho Cruz M, Moura Branco L, Galrinho A, Coutinho Miranda L, Fragata J, et al. Infective endocarditis: surgical management and prognostic predictors. Rev Port Cardiol. 2018;37(5):387–394. doi: 10.1016/j.repc.2017.08.007. [DOI] [PubMed] [Google Scholar]
- 17.Cahill TJ, Prendergast BD. Infective endocarditis. Lancet. 2016;387(10021):882–893. doi: 10.1016/S0140-6736(15)00067-7. [DOI] [PubMed] [Google Scholar]
- 18.Reference obscured for peer review process.
- 19.Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta J-P, Del Zotti F, et al. ESC Scientific Document Group, 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC) Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM) Eur Heart J. 2015;36(44):100–101. doi: 10.1093/eurheartj/ehv319. [DOI] [PubMed] [Google Scholar]
- 20.Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas and Bennett’s principles and practice of infectious diseases. 5. Philadelphia: Churchill Livingstone; 2000. [Google Scholar]
- 21.De Rosa FG, Cicalini S, Canta F, Audagnotto S, Cecchi E, Di Perri G. Infective endocarditis in intravenous drug users from Italy: the increasing importance in HIV-infected patients. Infection. 2007;35(3):154–160. doi: 10.1007/s15010-007-5125-0. [DOI] [PubMed] [Google Scholar]
- 22.Mathew J, Addai T, Anand A, Morrobel A, Maheshwari P, Freels S. Clinical features, site of involvement, bacteriologic findings, and outcome of infective endocarditis in intravenous drug users. Arch Intern Med. 1995;155(15):1571–1572. [PubMed] [Google Scholar]
- 23.Rudasill SE, Sanaiha Y, Mardock AL, Khoury H, Xing H, Antonios JW, et al. Clinical outcomes of infective endocarditis in injection drug users. J Am Coll Cardiol. 2019;73(5):571–572. doi: 10.1016/j.jacc.2018.10.082. [DOI] [PubMed] [Google Scholar]
- 24.Mathew J, Anand A, Addai T, Freels S. Value of echocardiographic findings in predicting cardiovascular complications in infective endocarditis. London: Westminster Publications; 2001. p. 52. [DOI] [PubMed] [Google Scholar]
- 25.Schmid E, Hilberath JN, Blumenstock G, Shekar PS, Kling S, Shernan SK, et al. Tricuspid annular plane systolic excursion (TAPSE) predicts poor outcome in patients undergoing acute pulmonary embolectomy. Heart Lung Vessel. 2015;7(2):151–158. [PMC free article] [PubMed] [Google Scholar]
- 26.Lee S, Kamdar F, Madlon-Kay R, Boyle A, Colvin-Adams M, Pritzker M. Effects of the Heart Mate II continuous-flow left ventricular assist device on right ventricular function. J Heart Lung Transplant. 2010;29:209–215. doi: 10.1016/j.healun.2009.11.599. [DOI] [PubMed] [Google Scholar]
- 27.David TE, Feindel CM, Armstrong S, Sun Z. Reconstruction of the mitral annulus. A ten-year experience. J Thorac Cardiovasc Surg. 1995;110(5):1323–1332. doi: 10.1016/S0022-5223(95)70055-2. [DOI] [PubMed] [Google Scholar]
- 28.Gillinov AM, Diaz R, Blackstone EH, Pettersson GB, Sabik JF, Lytle BW, Cosgrove DM., 3rd Double valve endocarditis. Ann Thorac Surg. 2001;71(6):1874–1879. doi: 10.1016/s0003-4975(01)02603-0. [DOI] [PubMed] [Google Scholar]
- 29.Selton-Suty C, Doco-Lecompte T, Bernard Y, Duval X, Letranchant L, Delahaye F, the AEPEI Study Group et al. Clinical and microbiologic features of multivalvular endocarditis. Curr Infect Dis Rep. 2010;12:237–243. doi: 10.1007/s11908-010-0112-5. [DOI] [PubMed] [Google Scholar]
- 30.Chirouze C, Athan E, Alla F, Chu VH, Ralph Corey G, Selton-Suty C, et al. International Collaboration on Endocarditis Study Group. Enterococcal endocarditis in the beginning of the 21st century: analysis from the International Collaboration on Endocarditis-Prospective Cohort Study. Clin Microbiol Infect. 2013;19(12):1140–1147. doi: 10.1111/1469-0691.12166. [DOI] [PubMed] [Google Scholar]
- 31.Abdallah L, Habib G, Remadi JP, Salaun E, Casalta JP, Tribouilloy C. Comparison of prognoses of Staphylococcus aureus left-sided prosthetic endocarditis and prosthetic endocarditis caused by other pathogens. Arch Cardiovasc Dis. 2016;109(10):542–549. doi: 10.1016/j.acvd.2016.02.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.