Abstract
Long-term culture of canine artificial hepatocytes has not been established. We hypothesized that human placental hydrolysate (hPH) may support the long-term culture of differentiated hepatocyte-like cells. Canine bone marrow cells were cultured using modified hepatocyte growth medium supplemented with hPH. Quantitative reverse transcription polymerase chain reaction (RT-PCR) and immunocytochemical analysis for albumin, qualitative RT-PCR for cytochrome P450 1A1 (CYP1A1), hepatocyte growth factor (HGF), Cytokeratin 7 (CK7), CD90, CD44, and CD34, and functional analyses of CYP450 activity and low-density lipoprotein (LDL) uptake were performed. Cultured hepatocyte-like cells were able to maintain hepatocyte characteristics, including morphology, albumin synthesis, CYP450 activity, and LDL uptake for 80 days. Thus, hPH may be a potential facilitator for the long-term culture of hepatocyte-like cells. Clinicopathologically, this culture protocol of artificial hepatocytes will contribute to liver function evaluation.
Keywords: bone marrow, dog, hepatocyte, long-term culture, placenta
The loss of liver function due to drug toxicity or viral infection related to acute or chronic hepatitis or cirrhosis is a major cause of death in humans and dogs [1, 29]. Canine liver failure is common and is caused by hepatitis, portosystemic shunts, and hepatic cirrhosis, whose mechanisms are similar to those in humans, at a molecular and cellular level [6, 9, 17, 29]. Liver transplant is the gold standard therapy for liver failure in humans; however, the shortage of liver organ donors and the adverse effects increase the need for alternatives. Hepatocyte transplantation using differentiated hepatocytes from induced pluripotent stem cells (iPSCs), embryonic stem cells (ESCs), or mesenchymal stem cells (MSCs), as well as liver bud transplantation have been proposed for humans and dogs [10, 21, 27]. However, an efficient and long-term cell culture of primary and artificial hepatocytes for practical use and transplantation has not been established in humans or dogs.
Long-term hepatocyte culture techniques are also required to establish artificial hepatocytes for pharmaceutical toxicity screening [25]. Primary hepatocyte cultures use is generally considered the gold standard for liver-based in vitro toxicity testing [30]. However, the long-term culture of primary hepatocytes is largely impeded by the progressive loss of the hepatocyte-specific morphological and functional phenotype [3, 18, 22]. Multiple culture conditions have been investigated to improve the long-term survival of hepatocytes or artificial hepatocytes, maintaining an appropriate liver function. Zhang et al. developed a defined medium using human liver isolation medium with no Rspo1, Noggin, and forskolin, to expand a large quantity of functional primary human hepatocytes for more than a few months [31]. Mouse ESCs and iPSCs were cultured with supplements, including cytokines and sodium butyrate, and efficiently differentiated into functional hepatocytes in vitro, after 20 days [32]. However, the long-term hepatocyte culture methods are limited and far from practical use in transplantation and pharmaceutical toxicity studies.
Placenta is a rich source of many biological components, including hormones, cytokines, chemokines, and growth factors required for liver regeneration [11, 12]. Previously, we found that human placental hydrolysate (hPH) may be an effective inducer of hepatic differentiation of canine bone marrow cells (cBMCs) [19]. The complex mixture of amino acids, peptides, DNA bases, carbohydrates, and cytokines in hPH may contain potential cofactors to induce the differentiation of cBMCs into hepatocytes. In fact, placental growth factor upregulation, which is observed in regenerating livers, is correlated to liver growth [28]. Furthermore, hPH contains HGF, epidermal growth factor (EGF), and fibroblast growth factor, and has been shown to induce liver regeneration in rats, HepG2 cells, and human primary hepatocytes [15].
Clinicopathologically, the evaluation of the toxic effect of liver function is highly dependent on serum hepatic enzymes, such as alanine aminotransferase (ALT), or serum bile acid test, but these are not specific markers [7]. Therefore, new parameters to evaluate the toxic damage or function of hepatocytes need to be established. Long-term survival of functional cBMCs-derived hepatocyte-like cells using hPH may be able to support in vitro pharmacologic testing of new drugs, as well as the detection of novel biomarkers for canine liver disease.
We hypothesized that hPH may support the long-term culture of differentiated hepatocytes. To confirm our hypothesis, we cultured cBMC-derived hepatocyte-like cells for 80 days in hepatocyte growth medium (HGM) containing hPH and assessed their morphology, gene and protein expressions, and hepatic function.
cBMCs were collected from clinically healthy dogs (2–5-year-old beagles, n=4) under sedation with butorphanol tartrate (0.2 mg/kg IM) and medetomidine hydrochloride (0.04 mg/kg IM). Animal care and handling were in accordance with the Azabu University Animal Experiment Guidelines. All experiments were reviewed and approved by the Ethics Committee of Azabu University (approval number: 180220-1). The marrow cells were separated using Lymphoprep (Fresenius Kabi Norge AS Axis-Shield proCAS, Oslo, Norway) and inoculated at a density of 1 × 106/cm2 in 25-cm collagen-coated plastic dishes. The cells were cultured at 37°C and 5% CO2 using modified HGM medium supplemented with 10% fetal bovine serum, 20 ng/ml of EGF (Sigma Aldrich, St. Louis, MO, USA), and 0.2 ml/ml of hPH (Laennec; kindly provided by Japan Bio Products Co., Ltd., Tokyo, Japan) [19]. The cells were cultured for 80 days and assessed on days 7, 21, and 80. The medium was changed every 3 days. Fresh hepatocytes were also obtained from healthy dogs via true-cut biopsy using the Super Core Biopsy Instrument 14 ga × 9 cm (Angiotech, Vancouver, BC, Canada). Filamentous actin (F-actin) was detected using confocal microscopy by using Alexa Fluor 488 phalloidin 3000 (Invitrogen, Carlsbad, CA, USA) to stain cultured cBMCs on day 21, after fixation in 3.7% paraformaldehyde, permeabilization in 0.1% Triton X-100 (Sigma Aldrich), and blocking with Block Ace (DS Pharma Biomedical Co., Ltd., Tokyo, Japan).
For the qualitative and quantitative RT-PCR, total RNA was extracted from cultured cBMCs and hepatocytes using the RiboPure kit (Applied Biosystems, Foster City, CA, USA) and 100 ng of total RNA was converted to cDNA using the High Capacity RNA-to-cDNA kit (Applied Biosystems). Synthesized cDNA was amplified using RT-PCR. The primer sequences and amplification conditions used are listed in Table 1. PCR products were visualized after electrophoresis on a 3% (w⁄v) agarose gel containing 0.1 µl/ml of UltraPower DNA Safedye (Gellex International Co., Ltd., Tokyo, Japan). Canine β-actin was used as an internal control. Direct DNA sequencing of the amplified PCR products was performed using an Applied Biosystem Model 310 sequencer (Applied Biosystems) and the dideoxy-mediated chain-termination method. Quantitative RT-PCR of albumin mRNA was performed using the Thermal Cycler Dice Real Time System II (TaKaRa Bio Inc., Kusatsu, Japan). Primers and probes were provided with the TaqMan Gene Expression Assay (Applied Biosystems), and eukaryotic 18S rRNA (Hs99999901_s1) and albumin (Cf02622284_m1) were included as internal controls. Data were analyzed using the 2-ΔΔCT method. Statistical analysis was performed using the Mann-Whitney test. P values <0.05 were considered statistically significant and all calculations were performed using GraphPad Prism 5 (GraphPad Software Inc., San Diego, CA, USA).
Table 1. Primer list used for qualitative RT-PCR and amplification conditions.
| Accession number | Sequence (5′→3′) | Product size (bp) | Amplification conditions of qualitative RT-PCR | |
|---|---|---|---|---|
| Alb | NM_001003026 | F:gtt cct ggg cac gtt ttt gta tga | 278 | 3 min at 95°C, followed by 36 cycles at 95°C for 30 sec, |
| R:ctt ggg gtg ctt tct tgg tgt aac | 55°C for 30 sec, 72°C for 30 sec, and 5 min at 72°C | |||
| HGF | NM_001002964 | F:ggt gct ata ctc ttg acc ctg c | 286 | 3 min at 95°C, followed by 36 cycles at 95°C for 30 sec, |
| R:cac cag ggt gac tca gcc cca tct g | 55°C for 30 sec, 72°C for 30 sec, and 5 min at 72°C | |||
| CYP1A1 | AACN010067442 | F:ccc tct ggc aca cta aat tcc a | 90 | 5 min at 95°C, followed by 36 cycles at 95°C for 20 sec, |
| R:gaa gtg cat cgg tga gac cat | 60°C for 30 sec, and 30 sec at 60°C | |||
| CK7 | XM_534795 | F:gcg tgg gag ccg tga aca tc | 109 | 5 min at 95°C, followed by 36 cycles at 95°C for 20 sec, |
| R:ccg ccg ccg ctg gag aa | 55°C for 30 sec, and 30 sec at 60°C | |||
| CD90 | XM_844606 | F:cag cat gac ccg gga gaa aaa g | 134 | 3 min at 95°C, followed by 36 cycles at 95°C for 30 sec, |
| R:tgg tgg tga agc cgg ata agt aga | 55°C for 30 sec, 72°C for 30 sec, and 5 min at 72°C | |||
| CD44 | Z27115 | F:tcc tca tat cca aca cct cc | 177 | 5 min at 95°C, followed by 40 cycles at 94°C for 1 min, |
| R:tgg aag atg ttg tag cct gc | 59.5°C for 2 min, 72°C for 1 min, and 5 min at 72°C | |||
| CD34 | NM_001003341 | F:tga gac ctc cag ctg tga | 550 | 3 min at 95°C, followed by 36 cycles at 95°C for 30 sec, |
| R:cag gtg ttg tct tgc tga atg g | 55°C for 30 sec, 72°C for 30 sec, and 5 min at 72°C | |||
| β-actin | AF021873 | F:ttc gcc atg gat gac gat atc g | 143 | 3 min at 95°C, followed by 36 cycles at 95°C for 30 sec, |
| R:ccc acc atc acg ccc tgg | 55°C for 30 sec, 72°C for 30 sec, and 5 min at 72°C | |||
F, forward primer; R, reverse primer.
Immunocytochemical staining was performed on cBMCs cultured for 80 days. Cells were fixed for 30 min with 4% paraformaldehyde in phosphate-buffered saline (PBS) at 4°C. Next, antigen activation with PBS containing 0.1% Triton X-100 was performed for 4 min at room temperature. To block internal peroxidase, slides were incubated in methanol with 3% H2O2 for 15 min at room temperature. After blocking with 10% skim milk containing PBS at room temperature for 1 hr, the slides were incubated at 4°C overnight with goat anti-dog albumin polyclonal antibody (Bethyl Laboratory Inc., Montgomery, TX, USA) as the primary antibody [dilution of 1:200 with Dako REAL antibody diluent (Dako, Glostrup, Denmark)]. Then, slides were incubated in 0.2 M glycine-containing PBS at room temperature for 30 min before incubation with the secondary antibody, goat MAX PO (Histofine; Nichirei, Tokyo, Japan), at room temperature for 30 min. Between each step, the slides were washed with PBS. The slides were visualized with 3,3′-diaminobenzidine (DAB; Nichirei) and counterstained with Dako REAL hematoxylin (Dako).
To test hepatocyte function, pentoxyresorufin was used to determine cytochrome P450 (CYP450) activity and the low-density lipoprotein (LDL) uptake was measured as previously described [19] in hepatocyte-like cells cultured for 80 days. The number of albumin- and F-actin-positive cells per field of cultured cBMCs was counted at × 400 magnification, and the positive cells: total cells ratio was averaged from 10 different fields.
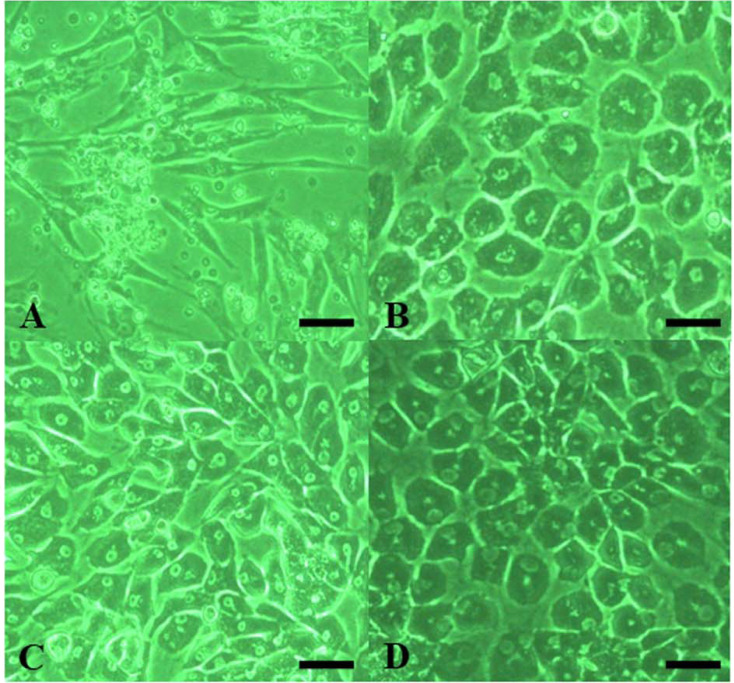
Here, cBMCs from a total of 4 dogs cultured in hPH for over 80 days were able to maintain hepatocyte characteristics, including morphology (Fig. 1), whereas without hPH in the culture medium, cells had a fibroblast-like morphology, which did not change, and they did not express albumin mRNA during culture (data not shown). Cultured cBMCs attached to the bottom of the flask had a spindle-like shape on day 7 and had become polygonal (pentagonal)-shaped, resembling mature hepatocytes, on day 21. They retained their polygonal shape until day 80 (Fig. 1). F-actin was detected cortically within the cell membrane (approximately 85% of cells) in hepatocyte-like cells on day 21 (Fig. 2). Immunocytochemical staining of albumin revealed the presence of albumin within the cytoplasm of 10–15% of the hepatocyte-like cells on day 80 (Fig. 2).
Fig. 1.
The morphology of cultured canine bone marrow cells (cBMCs) on days 7 (A), 21 (B), 60 (C), and 80 (D). cBMCs attached to the bottom of the flask were spindle-like shaped on day 7, and became polygonal (pentagonal), adult hepatocyte-like cells, on days 21, 60, and 80. Scale bars represent 20 µm.
Fig. 2.
F-actin staining and immunocytochemical staining of albumin in cultured bone marrow (BM) cells. (A) F-actin staining of cultured BM on day 21, viewed using a confocal microscope. Strong fluorescence was observed within the cell membrane, showing epithelial cell characteristics. Scale bar represents 20 µm. (B) Immunofluorescent staining of BM cells with anti-human albumin polyclonal antibody. Positively stained cells on day 80 (C), which were more strongly stained compared to cells in the negative control (B), were counted within ten randomly selected areas; 10–15% of hepatocyte-like cells were positive for albumin. Scale bars represent 50 µm.
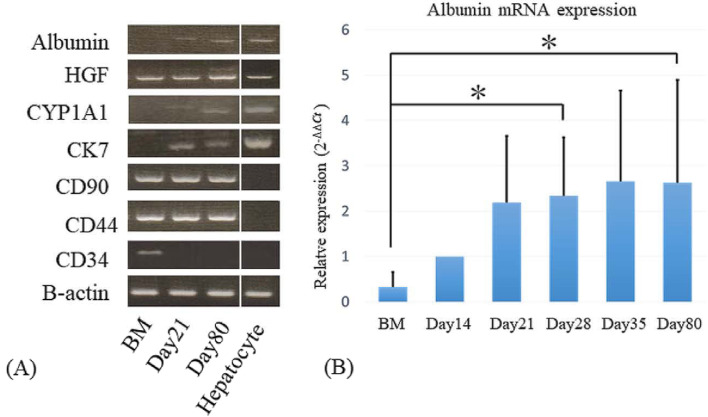
Gene expression analysis of the cultured cBMCs using qualitative RT-PCR analysis revealed the expression of albumin, CYP1A1, and CK7 in cultured hepatocyte-like cells on days 21 and 80. Albumin expression on day 21 showed slight variation, for example, 3 of 4 samples were positive and only one sample was negative. HGF, CD90, and CD44 were detected in cBMCs, as well as in hepatocyte-like cells on days 21 and 80. CD34 was only detected in cBMCs. Hepatocytes from adult canine liver expressed albumin, HGF, CYP1A1, and CK7 (Fig. 3A).
Fig. 3.
Gene expression analysis of cultured canine bone marrow cells using qualitative RT-PCR. (A) Qualitative RT-PCR analysis revealed that albumin (ALB) and cytochrome P450 (CYP1A1) were expressed in cultured bone marrow cells on days 21 and 80. Hepatocyte growth factor (HGF), CD90, and CD44 were detected in bone marrow cells and in cultured bone marrow cells on days 21 and 80. CD34 was only detected in bone marrow cells. Hepatocytes from adult canine liver expressed ALB, HGF, CYP1A1, and CK7. (B) Quantitative RT-PCR analysis of albumin mRNA. Using the 2-ΔΔCt method, the mRNA level of hepatocyte-like cells on day 14 was set to 1 and the relative mRNA levels on days 21, 28, 35, and 80 were expressed as mean ± standard error of the mean. * (P<0.05) indicates a significant difference from the bone marrow cells from the same dogs.
Quantitative RT-PCR revealed that the mRNA expression of albumin was increased in hepatocyte-like cells on days 14, 21, 28, and 80, compared to cBMCs; statistically significant differences were observed between cBMCs and day 28 hepatocyte-like cells, and cBMCs and day 80 hepatocyte-like cells, with approximately 3 times the albumin mRNA level, compared to that of day 14 cultured cBMCs (P=0.02; Fig. 3B).
Pentoxyresorufin is a nonfluorescent compound that is O-dealkylated by CYP450 into resorufin and emits red fluorescence. Hepatocyte-like cells showed a markedly increased fluorescence within the cytoplasm (69% of cells) on day 80 (Fig. 4B). LDL uptake is represented by the red fluorescent dye, indicating Dil, in which acetylated low density lipoprotein, labeled with 1,1-dioctadecyl-3,3,3′,3′-tetramethyl-indocarbocyanine perchlorate (Dil-Ac-LDL) is taken into the cell and metabolized, and was detected as a moderate red fluorescence within the cytoplasm of hepatocyte-like cells (20% of cells) on day 80 (Fig. 4D). No fluorescence was detected in cultured cBMCs on day 80 (Fig. 4A, 4C).
Fig. 4.
In vitro functional characterization of hepatocyte-like cells differentiated from bone marrow cells. Nuclei were stained using 4′,6-diamidino-2-phenylindole (blue), and the photomicrographs were overlaid with those of cytochrome P450 (CYP450) and the low-density lipoprotein (LDL) fluorescence. CYP450 and LDL were detected in differentiated cells at day 80 (B, D). N.C. indicates negative control, differentiated cells without CYP450 and LDL fluorescence (A, C). Scale bars represent 50 µm.
In the present study, hepatocyte-like cells differentiated from cBMCs were cultured for a long period of 80 days using hPH. On day 21, cultured cBMCs were polygonal-shaped, resembled hepatocytes, and maintained morphology throughout the 80 days of culture. F-actin was detected within the cellular membrane on day 21, which further confirmed that the hepatocyte-like cells had epithelial characteristics.
Interestingly, the albumin mRNA and protein were consistently expressed in hepatocyte-like cells until day 80, and hepatocyte function, including LDL synthesis and CYP function, were detected on day 80. Albumin is a protein synthesized by hepatocytes and is a well-known hepatocyte marker [25]. Hepatocyte markers that reflect hepatic function include those involved in drug metabolism, such as CYP450 and LDL uptake [8, 19]. The presence of these markers indicates that hepatocyte-like cells have some characteristics of canine hepatocytes and can maintain their features in the long term. Albumin mRNA expression was significantly increased in hepatocyte-like cells compared to cBMCs and did not change significantly from day 21 to day 80. The main challenges regarding culture of primary hepatocytes or artificial hepatocytes are the short survival and the loss of an appropriate hepatic function. A recent study showed that mouse primary hepatocytes lost the ability to synthesize albumin after 7 days of culture [14]. The activity of CYPs (CYP1A2 and CYP3A4) expressed by ESC-derived hepatocytes was significantly lower than that of primary hepatocytes [33] and a number of liver-essential functions were progressively lost with time, in cultured primary hepatocytes [4]. Additionally, directed differentiation of mouse bone marrow cells into hepatocyte-like cells was reported; however, the culture time was 21 days [23]. CYP activity in human hepatocyte-like cells derived from bone marrow cells was observed on days 21 and 28; however, evaluation of CYP activity has not been performed in a long-term culture [24]. Here, we proposed that hPH culture conditions would allow differentiated hepatocyte-like cells to survive longer, while maintaining hepatic function.
HGF, CK7, CD44, and CD90 are highly expressed in hepatic progenitor cells and liver tissues and, here, they were expressed in differentiated hepatocyte-like cells on day 80 in a long-term culture. HGF is essential growth factor for liver regeneration and is synthesized by hepatocytes and mesenchymal cells. Here, HGF was not supplemented, and hPH contains only a low amount of HGF, which may not be enough for hepatocyte-like cells to regenerate. HGF is synthesized by bone marrow stromal cells and stimulates liver regeneration in an autocrine and paracrine manner [10, 20, 26]. Here, HGF produced by cBMCs may have contributed to the regeneration and maintenance of hepatocyte characteristics for a long period of 80 days. CK7 mRNA was not expressed in cBMCs and was only detected in differentiated hepatocyte-like cells from day 21 to day 80. CK7 expression is observed in bile duct epithelial cells in the liver and in other epithelial cells, such as the acini and ductal epithelial cells of the salivary gland and anal sac gland, and the transitional cells of the urinary bladder; however, it is not expressed in hepatocytes [5]. These findings indicate that cBMCs can differentiate not only into hepatocyte-like cells, but also other epithelial cells. CD44 and CD90 are surface markers expressed in mesenchymal stroma cells derived from cBMCs [16] and are also liver progenitor markers [13]. Here, CD44 and CD90 were observed in cBMCs and in differentiated hepatocyte-like cells until day 80. Persistent expression of CD44 and CD90, which are stem cell markers, may contribute to the long-term survival of hepatocyte-like cells. The progenitor cell population expressing CD44 and CD90 might be the source for the production of mature hepatocytes.
Although it was not clear in the present study, there are a few possible reasons why hPH may contribute to the prolonged period of hepatocyte-like cell culture. hPH contains HGF, EGF, stem cell factor, amino acids, and interleukins (IL1 and IL6) [15]. hPH promotes liver regeneration by activating cytokines and growth factors associated with liver regeneration and eliminating oxidative stress [15]. Additionally, hPH exhibits a protective role in hepatocyte apoptosis by inhibiting oxidative stress and maintaining cell homeostasis [2]. Collectively, these factors may affect the long-term culture of hepatocyte-like cells. However, the precise level of each component in the hPH used here is unknown, since some of the factors in hPH were not measured correctly because of the low concentration of these components.
There are some limitations of this study: the albumin expression level in our cultured hepatocyte-like cells was only 1/105–1/107, much lower than in hepatocytes (data not shown), which emphasized the necessity for further improvement of the culture conditions for the hepatocyte-like cells to have an appropriate hepatic function. Moreover, additional effective components necessary for the long-term culture of hepatocyte-like cells in hPH should be elucidated. The next step for the effective long-term culture of hepatocytes or hepatocyte-like cells would be to use 3D cultures and iPS or directly reprogrammed cells as the cell source.
Here, cBMC-derived hepatocyte-like cells were able to maintain hepatocyte characteristics for a long period, and the potential contribution of hPH was indicated. Clinicopathologically, the establishment of this long-term culture protocol for cBMC-derived hepatocyte-like cells will significantly contribute to establish new parameters to evaluate liver function. Further studies are warranted to investigate the individual hPH components and mechanisms of action to establish the optimal culture conditions for hepatocytes or hepatocyte-like cells.
POTENTIAL CONFLICTS OF INTEREST
The authors have nothing to disclose.
Acknowledgments
We appreciate Dr. Takefumi Ishikawa for the valuable assistance. We also thank Japan Bio Products Co., Ltd., Tokyo, Japan for kindly providing the hPH. This work was supported by JSPS KAKENHI Grant Number 25850214, 17K08116, and 16K15050.
REFERENCES
- 1.Asrani S. K., Devarbhavi H., Eaton J., Kamath P. S.2019. Burden of liver diseases in the world. J. Hepatol. 70: 151–171. doi: 10.1016/j.jhep.2018.09.014 [DOI] [PubMed] [Google Scholar]
- 2.Bak D. H., Na J., Choi M. J., Lee B. C., Oh C. T., Kim J. Y., Han H. J., Kim M. J., Kim T. H., Kim B. J.2018. Anti‑apoptotic effects of human placental hydrolysate against hepatocyte toxicity in vivo and in vitro. Int. J. Mol. Med. 42: 2569–2583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cohen M., Levy G., Nahmias Y.2015. Coculture and long-term maintenance of hepatocytes. Methods Mol. Biol. 1250: 161–173. doi: 10.1007/978-1-4939-2074-7_11 [DOI] [PubMed] [Google Scholar]
- 4.Elaut G., Henkens T., Papeleu P., Snykers S., Vinken M., Vanhaecke T., Rogiers V.2006. Molecular mechanisms underlying the dedifferentiation process of isolated hepatocytes and their cultures. Curr. Drug Metab. 7: 629–660. doi: 10.2174/138920006778017759 [DOI] [PubMed] [Google Scholar]
- 5.Espinosa de los Monteros A., Fernández A., Millán M. Y., Rodríguez F., Herráez P., Martín de las Mulas J.1999. Coordinate expression of cytokeratins 7 and 20 in feline and canine carcinomas. Vet. Pathol. 36: 179–190. doi: 10.1354/vp.36-3-179 [DOI] [PubMed] [Google Scholar]
- 6.Eulenberg V. M., Lidbury J. A.2018. Hepatic fibrosis in dogs. J. Vet. Intern. Med. 32: 26–41. doi: 10.1111/jvim.14891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Harrill A. H., Eaddy J. S., Rose K., Cullen J. M., Ramanathan L., Wanaski S., Collins S., Ho Y., Watkins P. B., Lecluyse E. L.2014. Liver biomarker and in vitro assessment confirm the hepatic origin of aminotransferase elevations lacking histopathological correlate in beagle dogs treated with GABAA receptor antagonist NP260. Toxicol. Appl. Pharmacol. 277: 131–137. doi: 10.1016/j.taap.2014.03.015 [DOI] [PubMed] [Google Scholar]
- 8.Hay D. C., Zhao D., Fletcher J., Hewitt Z. A., McLean D., Urruticoechea-Uriguen A., Black J. R., Elcombe C., Ross J. A., Wolf R., Cui W.2008. Efficient differentiation of hepatocytes from human embryonic stem cells exhibiting markers recapitulating liver development in vivo. Stem Cells 26: 894–902. doi: 10.1634/stemcells.2007-0718 [DOI] [PubMed] [Google Scholar]
- 9.Ijzer J., Schotanus B. A., Vander Borght S., Roskams T. A., Kisjes R., Penning L. C., Rothuizen J., van den Ingh T. S.2010. Characterisation of the hepatic progenitor cell compartment in normal liver and in hepatitis: an immunohistochemical comparison between dog and man. Vet. J. 184: 308–314. doi: 10.1016/j.tvjl.2009.02.016 [DOI] [PubMed] [Google Scholar]
- 10.Jiang Y., Jahagirdar B. N., Reinhardt R. L., Schwartz R. E., Keene C. D., Ortiz-Gonzalez X. R., Reyes M., Lenvik T., Lund T., Blackstad M., Du J., Aldrich S., Lisberg A., Low W. C., Largaespada D. A., Verfaillie C. M.2002. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418: 41–49. doi: 10.1038/nature00870 [DOI] [PubMed] [Google Scholar]
- 11.Jung J., Lee H. J., Lee J. M., Na K. H., Hwang S. G., Kim G. J.2011. Placenta extract promote liver regeneration in CCl4-injured liver rat model. Int. Immunopharmacol. 11: 976–984. doi: 10.1016/j.intimp.2011.02.012 [DOI] [PubMed] [Google Scholar]
- 12.Jung J., Moon J. W., Choi J. H., Lee Y. W., Park S. H., Kim G. J.2015. Epigenetic alterations of IL-6/STAT3 signaling by placental stem cells promote hepatic regeneration in a rat model with CCl4-induced liver injury. Int. J. Stem Cells 8: 79–89. doi: 10.15283/ijsc.2015.8.1.79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Khuu D. N., Nyabi O., Maerckx C., Sokal E., Najimi M.2013. Adult human liver mesenchymal stem/progenitor cells participate in mouse liver regeneration after hepatectomy. Cell Transplant. 22: 1369–1380. doi: 10.3727/096368912X659853 [DOI] [PubMed] [Google Scholar]
- 14.Kim Y., Kang K., Yoon S., Kim J. S., Park S. A., Kim W. D., Lee S. B., Ryu K. Y., Jeong J., Choi D.2018. Prolongation of liver-specific function for primary hepatocytes maintenance in 3D printed architectures. Organogenesis 14: 1–12. doi: 10.1080/15476278.2018.1423931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee T. H., Park D. S., Jang J. Y., Lee I., Kim J. M., Choi G. S., Oh C. T., Kim J. Y., Han H. J., Han B. S., Joh J. W.2019. Human placenta hydrolysate promotes liver regeneration via activation of the cytokine/growth factor-mediated pathway and anti-oxidative effect. Biol. Pharm. Bull. 42: 607–616. doi: 10.1248/bpb.b18-00712 [DOI] [PubMed] [Google Scholar]
- 16.Lee W. S., Suzuki Y., Graves S. S., Iwata M., Venkataraman G. M., Mielcarek M., Peterson L. J., Ikehara S., Torok-Storb B., Storb R.2011. Canine bone marrow-derived mesenchymal stromal cells suppress alloreactive lymphocyte proliferation in vitro but fail to enhance engraftment in canine bone marrow transplantation. Biol. Blood Marrow Transplant. 17: 465–475. doi: 10.1016/j.bbmt.2010.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mankin K. M.2015. Current concepts in congenital portosystemic shunts. Vet. Clin. North Am. Small Anim. Pract. 45: 477–487. doi: 10.1016/j.cvsm.2015.01.008 [DOI] [PubMed] [Google Scholar]
- 18.Miyamoto Y., Ikeuchi M., Noguchi H., Yagi T., Hayashi S.2014. Three-dimensional in vitro hepatic constructs formed using combinatorial tapered stencil for cluster culture (TASCL) device. Cell Med. 7: 67–74. doi: 10.3727/215517914X685187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Neo S., Ishikawa T., Ogiwara K., Kansaku N., Nakamura M., Watanabe M., Hisasue M., Tsuchiya R., Yamada T.2009. Canine bone marrow cells differentiate into hepatocyte-like cells and placental hydrolysate is a potential inducer. Res. Vet. Sci. 87: 1–6. doi: 10.1016/j.rvsc.2008.11.008 [DOI] [PubMed] [Google Scholar]
- 20.Neuss S., Becher E., Wöltje M., Tietze L., Jahnen-Dechent W.2004. Functional expression of HGF and HGF receptor/c-met in adult human mesenchymal stem cells suggests a role in cell mobilization, tissue repair, and wound healing. Stem Cells 22: 405–414. doi: 10.1634/stemcells.22-3-405 [DOI] [PubMed] [Google Scholar]
- 21.Nitta S., Kusakari Y., Yamada Y., Kubo T., Neo S., Igarashi H., Hisasue M.2020. Conversion of mesenchymal stem cells into a canine hepatocyte-like cells by Foxa1 and Hnf4a. Regen Ther 14: 165–176. doi: 10.1016/j.reth.2020.01.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Richert L., Liguori M. J., Abadie C., Heyd B., Mantion G., Halkic N., Waring J. F.2006. Gene expression in human hepatocytes in suspension after isolation is similar to the liver of origin, is not affected by hepatocyte cold storage and cryopreservation, but is strongly changed after hepatocyte plating. Drug Metab. Dispos. 34: 870–879. doi: 10.1124/dmd.105.007708 [DOI] [PubMed] [Google Scholar]
- 23.Shi X. L., Qiu Y. D., Li Q., Xie T., Zhu Z. H., Chen L. L., Li L., Ding Y. T.2005. Hepatocyte-like cells from directed differentiation of mouse bone marrow cells in vitro. Acta Pharmacol. Sin. 26: 469–476. doi: 10.1111/j.1745-7254.2005.00093.x [DOI] [PubMed] [Google Scholar]
- 24.Schwartz R. E., Reyes M., Koodie L., Jiang Y., Blackstad M., Lund T., Lenvik T., Johnson S., Hu W. S., Verfaillie C. M.2002. Multipotent adult progenitor cells from bone marrow differentiate into functional hepatocyte-like cells. J. Clin. Invest. 109: 1291–1302. doi: 10.1172/JCI0215182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shulman M., Nahmias Y.2013. Long-term culture and coculture of primary rat and human hepatocytes. Methods Mol. Biol. 945: 287–302. doi: 10.1007/978-1-62703-125-7_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stuart K. A., Riordan S. M., Lidder S., Crostella L., Williams R., Skouteris G. G.2000. Hepatocyte growth factor/scatter factor-induced intracellular signalling. Int. J. Exp. Pathol. 81: 17–30. doi: 10.1046/j.1365-2613.2000.00138.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Takebe T., Zhang R. R., Koike H., Kimura M., Yoshizawa E., Enomura M., Koike N., Sekine K., Taniguchi H.2014. Generation of a vascularized and functional human liver from an iPSC-derived organ bud transplant. Nat. Protoc. 9: 396–409. doi: 10.1038/nprot.2014.020 [DOI] [PubMed] [Google Scholar]
- 28.Vanheule E., Fan Y. D., Van Huysse J., Meester D., Olievier K., Praet M., de Hemptinne B., Colle I.2011. Expression of placental growth factor in regenerating livers after partial hepatectomy in the rat. Eur. J. Gastroenterol. Hepatol. 23: 66–75. doi: 10.1097/MEG.0b013e328341ef35 [DOI] [PubMed] [Google Scholar]
- 29.Watson P. J.2004. Chronic hepatitis in dogs: a review of current understanding of the aetiology, progression, and treatment. Vet. J. 167: 228–241. doi: 10.1016/S1090-0233(03)00118-7 [DOI] [PubMed] [Google Scholar]
- 30.Zeilinger K., Freyer N., Damm G., Seehofer D., Knöspel F.2016. Cell sources for in vitro human liver cell culture models. Exp. Biol. Med. (Maywood) 241: 1684–1698. doi: 10.1177/1535370216657448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhang K., Zhang L., Liu W., Ma X., Cen J., Sun Z., Wang C., Feng S., Zhang Z., Yue L., Sun L., Zhu Z., Chen X., Feng A., Wu J., Jiang Z., Li P., Cheng X., Gao D., Peng L., Hui L.2018. In vitro expansion of primary human hepatocytes with efficient liver repopulation capacity. Cell Stem Cell 23: 806–819.e4. doi: 10.1016/j.stem.2018.10.018 [DOI] [PubMed] [Google Scholar]
- 32.Zhang Q., Yang Y., Zhang J., Wang G. Y., Liu W., Qiu D. B., Hei Z. Q., Ying Q. L., Chen G. H.2011. Efficient derivation of functional hepatocytes from mouse induced pluripotent stem cells by a combination of cytokines and sodium butyrate. Chin. Med. J. (Engl.) 124: 3786–3793. [PubMed] [Google Scholar]
- 33.Zhao D., Chen S., Duo S., Xiang C., Jia J., Guo M., Lai W., Lu S., Deng H.2013. Promotion of the efficient metabolic maturation of human pluripotent stem cell-derived hepatocytes by correcting specification defects. Cell Res. 23: 157–161. doi: 10.1038/cr.2012.144 [DOI] [PMC free article] [PubMed] [Google Scholar]