Key Points
Question
Does the use of sodium nitrite during resuscitation from out-of-hospital cardiac arrest improve survival to hospital admission?
Finding
In this phase 2 randomized clinical trial that included 1502 patients, out-of-hospital administration with 45 mg or 60 mg of sodium nitrite during active resuscitation by paramedics, compared with placebo, did not significantly improve survival to hospital admission (41% for the 45-mg dose and 43% for the 60-mg dose vs 44% for placebo).
Meaning
These findings do not support the use of sodium nitrite during resuscitation from out-of-hospital cardiac arrest.
Abstract
Importance
Therapeutic delivery of sodium nitrite during resuscitation improved survival in animal models of cardiac arrest, but efficacy has not been evaluated in clinical trials in humans.
Objective
To determine whether parenteral administration of sodium nitrite given by paramedics during resuscitation for out-of-hospital cardiac arrest improved survival to hospital admission.
Design, Setting, and Participants
Double-blind, placebo-controlled, phase 2 randomized clinical trial including 1502 adults in King County, Washington, with out-of-hospital cardiac arrest from ventricular fibrillation or nonventricular fibrillation. Patients underwent resuscitation by paramedics and were enrolled between February 8, 2018, and August 19, 2019; follow-up and data abstraction were completed by December 31, 2019.
Interventions
Eligible patients with out-of-hospital cardiac arrest were randomized (1:1:1) to receive 45 mg of sodium nitrite (n = 500), 60 mg of sodium nitrite (n = 498), or placebo (n = 499), which was given via bolus injection by the paramedics as soon as possible during active resuscitation.
Main Outcomes and Measures
The primary outcome was survival to hospital admission and was evaluated with 1-sided hypothesis testing. The secondary outcomes included out-of-hospital variables (rate of return of spontaneous circulation, rate of rearrest, and use of norepinephrine to support blood pressure) and in-hospital variables (survival to hospital discharge; neurological outcomes at hospital discharge; cumulative survival to 24 hours, 48 hours, and 72 hours; and number of days in the intensive care unit).
Results
Among 1502 patients with out-of-hospital cardiac arrest who were randomized (mean age, 64 years [SD, 17 years]; 34% were women), 99% completed the trial. Overall, 205 patients (41%) in the 45 mg of sodium nitrite group and 212 patients (43%) in the 60 mg of sodium nitrite group compared with 218 patients (44%) in the placebo group survived to hospital admission; the mean difference for the 45-mg dose vs placebo was −2.9% (1-sided 95% CI, −8.0% to ∞; P = .82) and the mean difference for the 60-mg dose vs placebo was −1.3% (1-sided 95% CI, −6.5% to ∞; P = .66). None of the 7 prespecified secondary outcomes were significantly different, including survival to hospital discharge for 66 patients (13.2%) in the 45 mg of sodium nitrite group and 72 patients (14.5%) in the 60 mg of sodium nitrite group compared with 74 patients (14.9%) in the placebo group; the mean difference for the 45-mg dose vs placebo was −1.7% (2-sided 95% CI, −6.0% to 2.6%; P = .44) and the mean difference for the 60-mg dose vs placebo was −0.4% (2-sided 95% CI, −4.9% to 4.0%; P = .85).
Conclusions and Relevance
Among patients with out-of-hospital cardiac arrest, administration of sodium nitrite, compared with placebo, did not significantly improve survival to hospital admission. These findings do not support the use of sodium nitrite during resuscitation from out-of-hospital cardiac arrest.
Trial Registration
ClinicalTrials.gov Identifier: NCT03452917
This randomized trial compares the effects of paramedic-administered parenteral sodium nitrite (45 mg vs 60 mg) vs placebo on survival to hospital admission among adult patients with out-of-hospital pulseless collapse and cardiac arrest.
Introduction
Out-of-hospital cardiac arrest is a common cause of premature morbidity and mortality and hence a major public health challenge.1 Existing treatments for out-of-hospital cardiac arrest combine cardiopulmonary resuscitation (CPR) and early defibrillation by bystanders or first responders with advanced cardiac life support by emergency medical services (EMS) personnel. The advanced life support treatments include CPR, defibrillation, intravenous drugs, and postresuscitation care.2,3,4 Neurological injury is the major cause of morbidity and mortality in these patients; most resuscitated patients never regain consciousness.5,6,7,8 Despite advances in resuscitation, more than 80% of those in whom circulation was restored after out-of-hospital cardiac arrest died before hospital discharge in studies with data collected between 2005 and 2015.9,10
Nitrite therapy limits cellular injury and apoptosis after ischemia and reperfusion and nitrite therapy is cytoprotective in numerous animal models of focal ischemia.11 In a rodent model of cardiac arrest, giving a single, low intravenous dose of nitrite during resuscitation significantly improved relative survival by 48% compared with placebo.12 Other animal models suggest that serum nitrite levels between 10 μM and 20 μM during the early reperfusion period following cardiac arrest are associated with improved survival.13 Based on results from a phase 1, open-label study in 125 patients with out-of-hospital cardiac arrest, a sodium nitrite dose of 45 mg or 60 mg given during resuscitation was sufficient to achieve serum nitrite levels between 10 μM and 20 μM within 10 to 15 minutes after administration.14
The aim of this randomized trial was to determine whether sodium nitrite given during active resuscitation of patients with out-of-hospital cardiac arrest improved survival to hospital admission.
Methods
Ethical Oversight
The trial was conducted with exception from informed consent for emergency research conditions in accordance with all applicable federal regulations (21 CFR §50.24) and investigational new drug provisions by the US Food and Drug Administration. The institutional review boards at the University of Washington and at all the acute care hospitals in Seattle and King County, Washington, provided approval. Oversight was performed by an independent data and safety monitoring board. Study personnel contacted the patient’s family as soon as feasible after enrollment to explain the study and seek written informed consent to review the patient’s medical records. Families of deceased patients were notified of participation by mail.
Study Setting and Patients
This double-blind, randomized clinical trial assigned adults with out-of-hospital cardiac arrest to 45 mg of sodium nitrite, 60 mg of sodium nitrite, or normal saline (placebo) during active resuscitation by paramedics. Seattle and King County, Washington, EMS collectively serve a population of 2.3 million residents and respond to more than 1100 nontraumatic cardiac arrests annually using a 2-tiered response. First-tier responders are trained in high-performance CPR and are equipped with automated external defibrillators. Second-tier responders are paramedics who provide advanced cardiac life support including defibrillation, intubation, and administration of resuscitation drugs. Patients with return of pulses are transported to 1 of 10 hospitals, each with intensive care units (ICUs) and 24-hour coronary catherization laboratories.
Cardiac arrest was defined as a sudden pulseless collapse requiring CPR by EMS personnel. Except for trauma, all causes of cardiac arrest were considered, including those with ventricular fibrillation (VF), pulseless ventricular tachycardia, or non-VF (defined as nonshockable rhythms) as the first recorded rhythm. Eligible patients received advanced life support by paramedics, had intravenous or intraosseous access, were older than aged 18 years, and were comatose at enrollment.
The study began on February 8, 2018, and the last patient was enrolled on August 19, 2019, with follow-up and data abstraction completed by December 31, 2019. The trial protocol appears in Supplement 1. All investigators, key personnel, and paramedics in Seattle and King County, Washington, were trained on procedures prior to study commencement.
Study Intervention
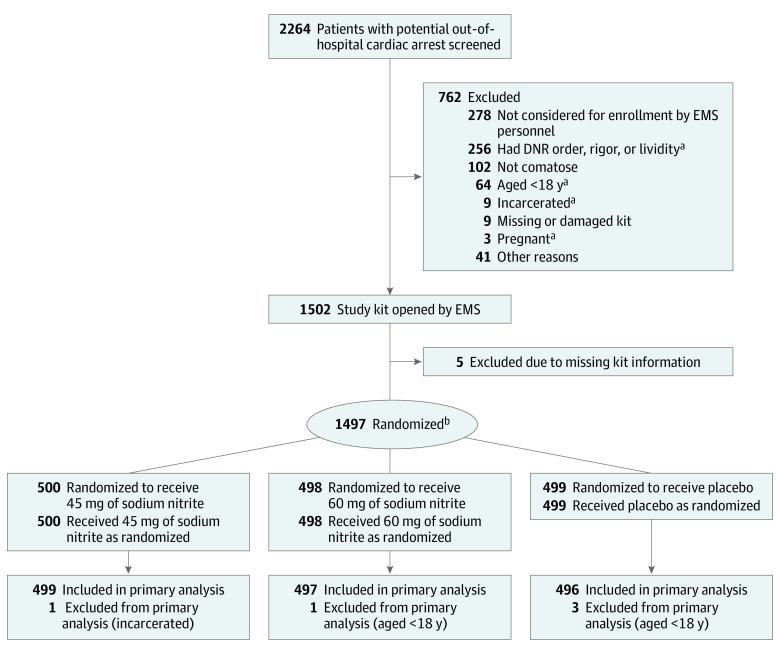
Patients were randomized 1:1:1 to 45 mg of sodium nitrite, 60 mg of sodium nitrite, or placebo based on the contents of a blinded study drug kit identifiable with a study number (Figure 1). Randomization was performed using computer-generated random samples and permuted block sizes of 3, 6, and 9 and was stratified by 4 regions in Washington State (South County, Redmond/Bellevue, Shoreline, and Seattle). A study kit contained a 3-mL glass syringe filled with 2 mL of sodium nitrite (22.5 mg/mL), 2 mL of sodium nitrite (30 mg/mL), or 2 mL of normal saline (Exela Pharma Sciences) that were indistinguishable in appearance. Stability testing of each product was performed throughout the course of the trial period. In addition, assays for sodium nitrite levels were performed on syringe contents from predesignated study kits that were periodically removed from paramedic units during the trial.
Figure 1. Study Enrollment and Analysis.
The study inclusion criteria were patients with out-of-hospital cardiac arrest who received advanced life support by paramedics, those having intravenous or intraosseous access, being aged 18 years or older, and being comatose. DNR indicates do not resuscitate; EMS, emergency medical services.
aIndicates an exclusion criterion.
bRandomization was stratified by 4 geographic regions in Washington State (South County, Redmond/Bellevue, Seattle, and Shoreline).
Eligible patients received intravenous or intraosseous sodium nitrite or placebo as soon as possible. The content of the study syringe was administered over 1 to 2 minutes by the paramedics. Once a study kit had been opened, the patient was considered randomized to that treatment group whether or not any study drug was administered.
The intervention and control groups were otherwise treated the same according to standard out-of-hospital resuscitation protocols. Paramedics transported patients to all acute care hospitals in King County, Washington, and provided information sheets describing the study to hospital physicians. Paramedics, emergency department staff, inpatient physicians, and nursing staff at receiving hospitals were blinded to treatment assignment. Study personnel who abstracted medical records for the outcomes were unaware of study allocation.
Outcomes
The primary outcome was survival to hospital admission, which was defined as formal inpatient assignment and arrival to a hospital ICU bed. The secondary outcomes included both out-of-hospital and in-hospital variables and included rate of rearrest (defined as loss of pulse); use of norepinephrine by paramedics; rate of return of spontaneous circulation; number of days in the ICU; cumulative survival to 24 hours, 48 hours, and 72 hours; survival to hospital discharge; and neurological status at hospital discharge. Data from standard reports completed by paramedics and from hospital records were collected as previously described.15,16 The time intervals included (1) from receipt of the 9-1-1 call to the arrival of EMS personnel and (2) from receipt of the 9-1-1 call to randomization (ie, study kit was opened) and were determined from review of the EMS records and from review of downloaded defibrillator data.
Neurological status at the time of hospital discharge was assessed by review of the daily progress notes and was assigned a Cerebral Performance Category (CPC) score as follows: a score of 1 indicates good cerebral performance (conscious, alert, and able to work); 2, moderate cerebral disability (conscious, sufficient cerebral function for independent activity); 3, severe cerebral disability (conscious, dependent on others for daily support due to impaired brain function); 4, comatose or in a vegetative state; and 5, dead.16,17 The CPC scores of 1 and 2 were grouped as mild to moderate impairment and the CPC scores of 3 and 4 were grouped as severe impairment. The initial plan was to use the modified Rankin Scale; however, the CPC scoring system was used instead because the medical record abstracters did not have experience using the modified Rankin Scale. This change was made before the first patient was enrolled.
To assess for potential adverse effects of the intervention, several prespecified and post hoc variables and outcomes were evaluated. The half-life of sodium nitrite is 40 to 50 minutes; sodium nitrite infusion is associated with a reduction in mean blood pressure of 7 mm Hg in healthy individuals.18 Therefore, we examined the first recorded blood pressure level at hospital arrival and any evidence of sustained hypotension in the hospital, which was defined as a sustained systolic blood pressure level of less than 90 mm Hg. We examined the medical record for any withdrawal of life-sustaining support as a measure of the level of intensity of patient care.
In addition to these prespecified outcomes, both post hoc out-of-hospital and post hoc in-hospital outcomes also were examined to further assess the treatment effects on hemodynamics and the intensity of hospital treatment. For out-of-hospital events, we assessed the first blood pressure level obtained by paramedics in the field after randomization. In the hospital setting, evidence for rearrest during the first 24 hours of hospitalization, use of vasopressors, pH determined by the first arterial blood gas measurement, and a positive troponin level (defined as >0.02-0.08 ng/mL at any time during the hospitalization) were used as measures of possible adverse effects of the treatment. The use of targeted temperature management and cardiac catherization during hospitalization were used as measures of the intensity of in-hospital treatment. Survival outside the ICU (defined as the number of days alive but not in the ICU during first 30 days after index cardiac arrest) was used as a composite measure of morbidity and mortality.
Sample Size
We estimated a priori that a sample size of 1500 (500 per group) would provide 80% power to detect an absolute increase of 8% for the hospital admission rate between each of the treatment groups and the placebo group using a 1-sided test with an α level of .05 for each comparison and a hospital admission rate of 40% in the placebo group. No human studies were available. The animal study demonstrating an absolute increase in survival of 28%12 seemed overly optimistic, therefore, we chose an increase of 8% for the hospital admission rate to be a conservative, yet clinically meaningful, estimate. This 1-sided test was used because only an increase in the hospital admission rate would support progression to a multicenter, phase 3 clinical trial.19,20
Statistical Methods
Categorical variables are presented as number (percentage) and continuous variables are presented as median (interquartile range). One-sided tests of proportions were used for the primary outcome with an analysis of patients by the group to which they were randomized, complete case analysis, and an α level of .05 for each of the 2 comparisons (45 mg of sodium nitrite vs placebo and 60 mg of sodium nitrite vs placebo). For the secondary and other analyses, the mean differences (2-sided 95% CIs) for the 2 comparisons are reported.
Furthermore, for each of the 2 comparisons, 2-sided statistical tests were used. The χ2 test was used for categorical variables and the Wilcoxon rank sum test was used for continuous variables. Because of the potential for type I error due to multiple comparisons, the findings from these secondary end points should be interpreted as exploratory.
Additional secondary analyses of the primary end point used generalized estimating equations and 2-sided 95% CIs for binomial outcomes to estimate differences and to adjust for age, witnessed cardiac arrest status, initial rhythm (VF vs non-VF), location (public vs private), bystander CPR, and response time (from 9-1-1 call to arrival of EMS personnel at the scene), allowing different correlation for patients treated within the same geographic region in Washington State (South County, Seattle, Redmond/Bellevue, Shoreline).
No adjustments were made for multiple comparisons. A priori subgroup analysis included bystander CPR (yes vs no), initial rhythm (VF vs pulseless electrical activity and asystole), observational status of cardiac arrest (witnessed by EMS personnel vs witnessed by bystander vs unwitnessed), response time interval from 9-1-1 call to arrival at scene by EMS personnel among witnessed cardiac arrests (<10 minutes vs ≥10 minutes), and sex (male vs female). The potential for interaction effects between the treatment groups and these factors were assessed using generalized estimating equations for binomial outcomes, allowing different correlation for patients treated within the same geographic region (including main effect and interaction effect terms) and testing (using the Wald test) whether all interaction terms are simultaneously 0 with 2 degrees of freedom (bystander CPR, initial rhythm, response time interval from 9-1-1 call to arrival of EMS personnel, sex) or with 4 degrees of freedom (observational status of cardiac arrest). One formal interim analysis was reviewed by the independent data and safety monitoring board using only a futility boundary.
P < .05 was considered statistically significant. For the statistical analysis, SPSS version 19.0 (SPSS Inc) and Stata version 16.1 (StataCorp) were used.
Post Hoc Analyses
Time to death was graphically represented using Kaplan-Meier plots and statistically tested using the log-rank statistic. Patients who were discharged alive were censored at the time of hospital discharge. The test by Grambsch and Therneau was used as a global test for the proportionality of hazards.
Results
Enrollment and Randomization
During the enrollment period, participating paramedics attended to 2264 patients with cardiac arrest, of whom 1502 (66%) were enrolled (mean age, 64 years [SD, 17 years]; 34% were women) (Figure 1). A total of 762 patients were not enrolled. Of these 762 patients, 278 (36%) were not considered for enrollment by the EMS personnel; 102 (13%) were awake (not comatose); and there were 256 (34%) for whom advanced cardiac life support was not started because the paramedics were aware of a do-not-resuscitate order or because of evidence of lividity or rigor. Characteristics of the randomized and excluded patients appear in eTable 1 in Supplement 2.
The study kit number was missing for 5 patients, so they were not included in the analysis. Five additional patients were later withdrawn from the study after having been identified as being in a protected population (4 were aged <18 years and 1 was incarcerated). Thus, 1492 patients were included in the primary analysis and out-of-hospital and in-hospital variables were complete in more than 99% of these patients. There were 5 patients for whom consent for hospital medical record review could not be obtained. Only out-of-hospital, emergency department, and hospital discharge data were available for these 5 patients, but not neurological outcomes or data on in-hospital variables. Baseline out-of-hospital patient characteristics appear in Table 1; an intraosseous line was used in 27.8%.
Table 1. Baseline Out-of-Hospital Patient Characteristicsa.
| Characteristic | Sodium nitrite | Placebo (n = 496) | |
|---|---|---|---|
| 45 mg (n = 499) | 60 mg (n = 497) | ||
| Age, median (IQR), y | 65 (56-76) | 64 (52-76) | 67 (55-76) |
| Sex, No. (%) | |||
| Male | 329 (66) | 330 (66) | 331 (67) |
| Female | 170 (34) | 167 (34) | 165 (33) |
| Witnessed cardiac arrest, No. (%) | 251 (50) | 251 (50) | 249 (50) |
| Bystander CPR, No. (%) | 269 (54) | 268 (54) | 279 (56)b |
| Cardiac arrest before arrival of EMS personnel, No. (%) | 435 (87) | 431 (87) | 444 (90) |
| Time from 9-1-1 call, median (IQR), min | |||
| To arrival of EMS personnel | 5.6 (4.4-6.9)b | 5.4 (4.4-6.7) | 5.3 (4.3-6.9) |
| To randomization | 19.2 (15.8-24.4)c | 20.0 (16.0-25.4)d | 19.1 (15.9-24.3)e |
| Initial rhythm, No. (%) | |||
| Asystole | 229 (45.9) | 219 (44.1) | 205 (41.3) |
| Pulseless electrical activity | 154 (30.9) | 136 (27.4) | 147 (29.6) |
| Ventricular fibrillation or ventricular tachycardia | 96 (19.2) | 126 (25.3) | 119 (24.0) |
| Otherf | 20 (4.0) | 16 (3.2) | 25 (5.0) |
| Treatment details | |||
| Intubation, No. (%) | 483 (97) | 475 (96) | 472 (95) |
| Intraosseous delivery, No. (%) | 128 (26)g | 172 (35)c | 113 (23)c |
| Total No. of shocks, median (IQR) | 0 (0-1) | 0 (0-2) | 0 (0-2) |
Abbreviations: CPR, cardiopulmonary resuscitation; EMS, emergency medical services; IQR, interquartile range.
The patients who were incarcerated (1 in the 45-mg dose group) or younger than 18 years (1 in the 60-mg dose group and 3 in the placebo group) were excluded from all analyses.
Data were missing for 1 patient.
Data were missing for 2 patients.
Data were missing for 4 patients.
Data were missing for 5 patients.
Includes pulses on first rhythm check after CPR by EMS personnel, automatic external defibrillator without shock by EMS personnel, bystander automatic external defibrillator without shock, or unknown.
Data were missing for 3 patients.
Primary Outcome
Among randomized patients with out-of-hospital cardiac arrest, 205 (41%) in the 45 mg of sodium nitrite group and 212 (43%) in the 60 mg of sodium nitrite group compared with 218 (44%) in the placebo group survived to hospital admission; the mean difference for the 45-mg dose vs placebo was −2.9% (1-sided 95% CI, −8.0 to ∞; P = .82) and the mean difference for the 60-mg dose vs placebo was −1.3% (1-sided 95% CI, −6.5% to ∞; P = .66) (Table 2).
Table 2. Primary and Secondary Outcomes.
| Sodium nitrite | Placebo (n = 496) |
45-mg dose vs placebo | 60-mg dose vs placebo | ||||
|---|---|---|---|---|---|---|---|
| 45 mg (n = 499) |
60 mg (n = 497) |
Mean difference (95% CI) | P value | Mean difference (95% CI) | P value | ||
| Primary outcome | |||||||
| Survived to hospital admission, No. (%) | 205 (41) | 212 (43) | 218 (44) | −2.9 (−8.0 to ∞) | .82 | −1.3 (−6.5 to ∞) | .66 |
| Secondary outcomes | |||||||
| Out-of-hospital return of spontaneous circulation, No. (%) | 270 (54) | 294 (59) | 291 (59) | −4.6 (−10.7 to 1.6) | .15 | 0.5 (−5.6 to 6.6) | .88 |
| Cardiac rearrest, No./total (%) | 130/270 (48) | 156/294 (53) | 139/291 (48) | 0.4 (−7.8 to 8.6) | .93 | 5.3 (−2.8 to 13.3) | .20 |
| Out-of-hospital use of norepinephrine after randomization, No. (%) | 79 (16) | 77 (16) | 75 (15) | 0.7 (−3.8 to 5.2) | .76 | 0.4 (−4.1 to 4.9) | .87 |
| Time in ICU, mean (SD), d | 8.9 (10.4) | 7.3 (7.2) | 6.7 (8.1) | 2.2 (−0.9 to 5.4) | .12 | 0.5 (−2.5 to 3.6) | .41 |
| Cumulative survival during hospital stay, No. (%) | |||||||
| 24 h | 170 (34.1) | 171 (34.4) | 181 (36.5) | −2.4 (−8.3 to 3.5) | .42 | −2.1 (−8.0 to 3.9) | .49 |
| 48 h | 152 (30.5) | 151 (30.4) | 161 (32.5) | −2.0 (−7.7 to 3.8) | .50 | −2.1 (−7.8 to 3.7) | .48 |
| 72 h | 133 (26.7) | 131 (26.4) | 142 (28.6) | −2.0 (−7.7 to 3.6) | .49 | −2.3 (−7.8 to 3.3) | .42 |
| Discharge from hospital | 66 (13.2) | 72 (14.5) | 74 (14.9) | −1.7 (−6.0 to 2.6) | .44 | −0.4 (−4.9 to 4.0) | .85 |
| Neurological status at hospital discharge, No. (%) | (n = 498) | (n = 496) | (n = 495) | .69a | .84a | ||
| Moderate cerebral disability or better (CPC score of 1 or 2)b | 60 (12.0) | 66 (13.3) | 66 (13.3) | −1.3 (−5.4 to 2.9) | 0 (−4.3 to 4.2) | ||
| Severe cerebral disability or coma (CPC score of 3 or 4)b | 5 (1.0) | 5 (1.0) | 7 (1.4) | −0.4 (−2.0 to 1.1) | −0.4 (−2.0 to 1.1) | ||
Abbreviation: ICU, intensive care unit.
Overall P values are reported. For each 2-way comparison (45-mg dose vs placebo and 60-mg dose vs placebo), the χ2 statistic was used to assess statistical significance. There were 3 categories: moderate, severe, and dead.
The Cerebral Performance Category (CPC) scores range from 1 (good cerebral performance [conscious, alert, and able to work]), 2 (moderate cerebral disability [conscious, sufficient cerebral function for independent activity]), 3 (severe cerebral disability [conscious, dependent on others for daily support due to impaired brain function]), 4 (comatose or in a vegetative state), and 5 (dead). The CPC score at hospital discharge was determined by investigator review of the patient’s chart.
Secondary Outcomes
None of the 7 prespecified secondary outcomes were significantly different. There were 270 patients (54%) in the 45 mg of sodium nitrite group and 294 patients (59%) in the 60 mg of sodium nitrite group who achieved out-of-hospital return of spontaneous circulation compared with 291 patients (59%) in the placebo group; the mean difference for the 45-mg dose vs placebo was −4.6% (95% CI, −10.7% to 1.6%; P = .15) and the mean difference for the 60-mg dose vs placebo was 0.5% (95% CI, −5.6% to 6.6%; P = .88). There were 79 patients (16%) in the 45 mg of sodium nitrite group and 77 patients (16%) in the 60 mg of sodium nitrite group who received out-of-hospital norepinephrine compared with 75 patients (15%) in the placebo group. The rate of out-of-hospital rearrest (defined by loss of pulse following return of spontaneous circulation) was 48% in the 45-mg dose group, 53% in the 60-mg dose group, and 48% in the placebo group (Table 2).
Among patients who survived to hospital admission, there were no significant differences in the number of ICU days between the sodium nitrite groups vs placebo; the mean difference for 45-mg dose vs placebo was 2.2 days (95% CI, −0.9 to 5.4 days; P = .12) and the mean difference for the 60-mg dose vs placebo was 0.5 days (95% CI, −2.5 to 3.6 days; P = .41) (Table 2). There were 66 patients (13.2%) in 45-mg dose group and 72 patients (14.5%) in the 60-mg dose group who survived to hospital discharge compared with 74 patients (14.9%) in the placebo group; the mean difference for the 45-mg dose vs placebo was −1.7% (95% CI, −6.0% to 2.6%; P = .44) and the mean difference for the 60-mg dose vs placebo was −0.4% (95% CI, −4.9% to 4.0%; P = .85) (Table 2). Survival at 24 hours, 48 hours, and 72 hours did not differ significantly among the groups (Table 2).
The generalized estimating equations used to adjust for age, witnessed cardiac arrest status, initial rhythm (VF vs non-VF), location, bystander CPR, and response time did not demonstrate a significant difference between the 45-mg dose and placebo (odds ratio, 0.94; 95% CI, 0.72-1.24) or between the 60-mg dose and placebo (odds ratio, 0.92; 95% CI, 0.70-1.20) (eTable 2 in Supplement 2).
Additional Prespecified and Post Hoc Analyses
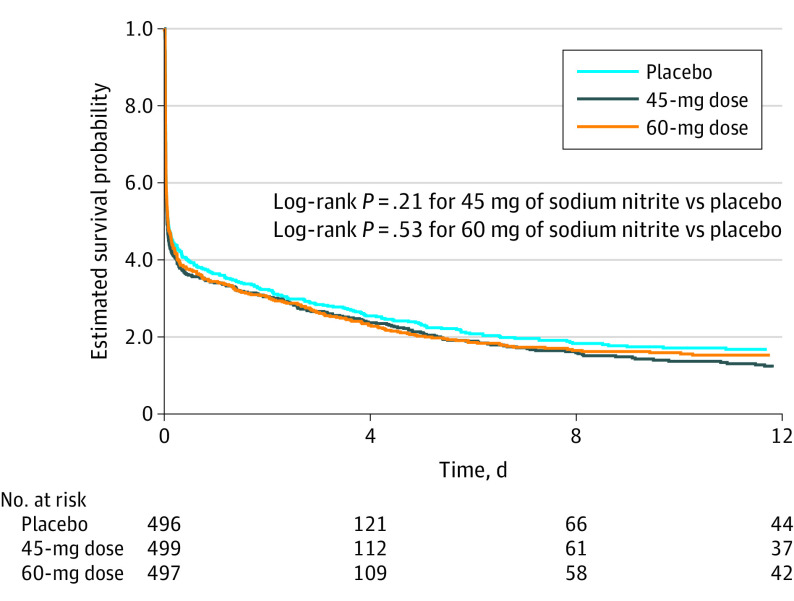
Neither dose of sodium nitrite was associated with a significant difference for time to death (P = .37 using a proportional hazards test; Figure 2). Sodium nitrite administration was not associated with a significant reduction in the first recorded blood pressure level at hospital arrival, sustained hypotension, or the incidence of withdrawal of life-sustaining support (eTable 3 in Supplement 2).
Figure 2. Time to Death by Treatment Group.
Observations were censored at hospital discharge. The median observation time was 0.07 days (interquartile range, 0.03-3.81 days).
For the post hoc analyses of the in-hospital outcomes, no significant differences were evident in the proportion of patients who underwent cardiac catheterization at any time during their hospitalization (26% in 45-mg dose group and 26% in 60-mg dose group compared with 24% in the placebo group), and use of targeted temperature management (51% in 45-mg dose group and 51% in 60-mg dose group compared with 49% in the placebo group) (eTable 3 in Supplement 2).
A Priori Subgroups
Among patients who had initial rhythm of VF, no significant differences were noted in survival to hospital admission (eFigure in Supplement 2). Hospital outcomes in patients with the lowest 25th percentile time from 9-1-1 call to randomization (<14.6 minutes) and the highest 25th percentile time from 9-1-1 call to randomization (>21.6 minutes) appear in eTables 4 and 5, respectively, in Supplement 2.
Discussion
In this phase 2, double-blind, randomized clinical trial of patients undergoing resuscitation from out-of-hospital cardiac arrest, administration of sodium nitrite, compared with placebo, did not significantly improve survival to hospital admission or survival to hospital discharge. The intervention was not associated with a significant effect on return of spontaneous circulation, rate of rearrest, or requirement for vasopressor support prior to hospital arrival but the 95% CIs did not exclude potentially clinically meaningful differences. Although the intervention was based on promising studies using animal models of cardiac arrest and the results from a phase 1 open-labeled study,13,14,21 the results of this study do not support the use of sodium nitrite for out-of-hospital cardiac arrest.
Experimental animal evidence suggests that increasing nitric oxide bioavailability using pharmacological or genetic mechanisms protects organs during ischemia-reperfusion injury.22 Nitric oxide reduces inflammation, thrombosis, and production of reactive oxygen species, which are all biochemical processes involved in neuronal cell injury. Importantly, nitric oxide production is inhibited during hypoxia23 and ischemia,24,25,26 which limits the body’s ability to protect organs from tissue damage during reperfusion. The nitrite anion (NO2−) is converted to nitric oxide during hypoxia or low pH, independent of nitric oxide synthase activity, making NO2− an ideal drug to increase nitric oxide bioavailability during ischemia and in reperfusion.27 Nitrite offers an additional benefit of S-nitrosating mitochondrial complex I,28 permitting protective signaling through an additional mechanism.29
Previous studies suggested that during CPR, nitrite offered promise for global ischemia and reperfusion because of its simple dosing at the time of reperfusion, efficacy in multiple organs (heart, liver, brain, lung, and kidney),11 and lack of adverse events at the low doses used to attenuate ischemia-reperfusion injury. Relevant animal studies provided a rationale to test this therapy in patients with out-of-hospital cardiac arrest.
The investigation evaluated a generalizable, low-cost intervention for a condition that accounts for substantial public morbidity and mortality. The study was conducted in an EMS system with an established record of research and effective out-of-hospital resuscitation, which are characteristics essential for the internal and external validity of the study results. The investigation had adequate power to detect clinically important differences in survival to hospital admission for this phase 2 study. The effect of sodium nitrite in this trial was unlikely to be modified or confounded by the quality of out-of-hospital emergency care because of the randomized, blinded treatment and the fact that the baseline outcomes achieved by the EMS agencies that participated in this study were good. The effect of sodium nitrite was unlikely to have been modified by the quality of hospital-based care because prespecified and post hoc secondary analyses did not demonstrate a relationship between any outcomes and the use of early angiography, use of targeted temperature management, or withdrawal of life-sustaining support.
In prior animal studies, delivery of nitrite occurred within minutes following 12 minutes of asystole.12 In the present clinical study, delivery of sodium nitrite or placebo occurred within 25 to 30 minutes from time of 9-1-1 call (approximate time of loss of pulse) to nitrite administration, which is longer than the time in prior animal experiments. However, this time frame of drug delivery is consistent with that of other drugs in similar trials,30,31,32 which ultimately limits the applicability of intravenous sodium nitrite.
It is possible that the dose of sodium nitrite was not sufficient or there was a loss of drug potency. Two doses of sodium nitrite were used (45 mg and 60 mg), which were chosen based on phase 1 study data, and were sufficient to achieve serum nitrite levels between 10 μM and 20 μM within 10 to 15 minutes.14 Higher doses of sodium nitrite could lead to hypotension and serum nitrite levels greater than 100 μM are associated with neurological injury,12 therefore, higher doses of sodium nitrite would not likely be safe. However, the 45-mg dose did not achieve better outcomes than the 60-mg dose, suggesting that lower doses would not have proved more beneficial.
It is possible that certain subgroups of patients may have a greater benefit from sodium nitrite. Age, witnessed cardiac arrest, and paramedic response time are important predictors of outcome. However, exploratory and subgroup analyses did not demonstrate a significant effect for out-of-hospital use of sodium nitrite on hospital admission, even after accounting for these differences in the cardiac arrest population.
Limitations
The study has several limitations. First, the sample size calculations of the present study were based on assumptions from animal studies because human studies were limited. Therefore, the survival to hospital admission effect size may have been overestimated. This study may be underpowered to detect smaller differences.
Second, 278 patients (36% of unenrolled patients) were not considered for enrollment by paramedics. This might suggest enrollment bias in that patients who were older or who had more severe underlying illness were not enrolled. However, the patients not considered for enrollment were generally younger and there were higher proportions who were admitted to the hospital and who survived to hospital discharge (eTable 1 in Supplement 2).
Third, this study was not powered or designed specifically to examine the VF population, however, the absence of an observed significant difference in the primary or secondary outcomes or in the planned subgroup analyses suggests that a larger trial would be unlikely to detect such a difference in favor of sodium nitrite therapy.
Conclusions
Among patients with out-of-hospital cardiac arrest, administration of sodium nitrite, compared with placebo, did not significantly improve survival to hospital admission. These findings do not support the use of sodium nitrite during resuscitation from out-of-hospital cardiac arrest.
Trial protocol
eTable 1. Characteristics of randomized and missed subjects
eTable 2. Factors associated with admission to the hospital (n=1309)
eTable 3. Pre-specified and post-hoc out-of-hospital and hospital variables
eTable 4. Hospital outcomes (lowest 25th (<14.6 minutes) percentile time from call to randomization, VF after EMS arrival)
eTable 5. Hospital outcomes (highest 25th (>21.6 minutes) percentile time from call to randomization, VF after EMS arrival)
eFigure. Forest plot of absolute differences in percent admitted to hospital for prespecified subgroups
Data sharing statement
References
- 1.Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics—2014 update. Circulation. 2014;129(3):e28-e292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Panchal AR, Berg KM, Cabañas JG, et al. 2019 American Heart Association focused update on systems of care. Circulation. 2019;140(24):e895-e903. [DOI] [PubMed] [Google Scholar]
- 3.Neumar RW, Nolan JP, Adrie C, et al. Post-cardiac arrest syndrome: epidemiology, pathophysiology, treatment, and prognostication. Circulation. 2008;118(23):2452-2483. [DOI] [PubMed] [Google Scholar]
- 4.Callaway CW, Soar J, Aibiki M, et al. Part 4: advanced life support: 2015 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Circulation. 2015;132(16)(suppl 1):S84-S145. [DOI] [PubMed] [Google Scholar]
- 5.Longstreth WT., Jr Brain resuscitation after cardiopulmonary arrest. Acta Anaesthesiol Belg. 1988;39(3)(suppl 2):115-119. [PubMed] [Google Scholar]
- 6.Longstreth WT Jr, Copass MK, Dennis LK, et al. Intravenous glucose after out-of-hospital cardiopulmonary arrest: a community-based randomized trial. Neurology. 1993;43(12):2534-2541. [DOI] [PubMed] [Google Scholar]
- 7.Raichle ME. The pathophysiology of brain ischemia. Ann Neurol. 1983;13(1):2-10. [DOI] [PubMed] [Google Scholar]
- 8.Laver S, Farrow C, Turner D, Nolan J. Mode of death after admission to an intensive care unit following cardiac arrest. Intensive Care Med. 2004;30(11):2126-2128. [DOI] [PubMed] [Google Scholar]
- 9.Peberdy MA, Callaway CW, Neumar RW, et al. Part 9: post-cardiac arrest care: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2010;122(18)(suppl 3):S768-S786. [DOI] [PubMed] [Google Scholar]
- 10.Girotra S, van Diepen S, Nallamothu BK, et al. Regional variation in out-of-hospital cardiac arrest survival in the United States. Circulation. 2016;133(22):2159-2168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dezfulian C, Raat N, Shiva S, Gladwin MT. Role of the anion nitrite in ischemia-reperfusion cytoprotection and therapeutics. Cardiovasc Res. 2007;75(2):327-338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dezfulian C, Shiva S, Alekseyenko A, et al. Nitrite therapy after cardiac arrest reduces reactive oxygen species generation, improves cardiac and neurological function, and enhances survival via reversible inhibition of mitochondrial complex I. Circulation. 2009;120(10):897-905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dezfulian C, Alekseyenko A, Dave KR, et al. Nitrite therapy is neuroprotective and safe in cardiac arrest survivors. Nitric Oxide. 2012;26(4):241-250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim F, Dezfulian C, Empey PE, et al. Usefulness of intravenous sodium nitrite during resuscitation for the treatment of out-of-hospital cardiac arrest. Am J Cardiol. 2018;122(4):554-559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim F, Olsufka M, Longstreth WT Jr, et al. Pilot randomized clinical trial of prehospital induction of mild hypothermia in out-of-hospital cardiac arrest patients with a rapid infusion of 4 degrees C normal saline. Circulation. 2007;115(24):3064-3070. [DOI] [PubMed] [Google Scholar]
- 16.Kim F, Nichol G, Maynard C, et al. Effect of prehospital induction of mild hypothermia on survival and neurological status among adults with cardiac arrest: a randomized clinical trial. JAMA. 2014;311(1):45-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jennett B, Bond M. Assessment of outcome after severe brain damage. Lancet. 1975;1(7905):480-484. [DOI] [PubMed] [Google Scholar]
- 18.Cosby K, Partovi KS, Crawford JH, et al. Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat Med. 2003;9(12):1498-1505. [DOI] [PubMed] [Google Scholar]
- 19.Fisher LD. The use of one-sided tests in drug trials. J Biopharm Stat. 1991;1(1):151-156. [DOI] [PubMed] [Google Scholar]
- 20.Freedman LS. An analysis of the controversy over classical one-sided tests. Clin Trials. 2008;5(6):635-640. [DOI] [PubMed] [Google Scholar]
- 21.Dezfulian C, Olsufka M, Fly D, et al. Hemodynamic effects of IV sodium nitrite in hospitalized comatose survivors of out of hospital cardiac arrest. Resuscitation. 2018;122:106-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jones SP, Bolli R. The ubiquitous role of nitric oxide in cardioprotection. J Mol Cell Cardiol. 2006;40(1):16-23. [DOI] [PubMed] [Google Scholar]
- 23.Muramatsu K, Sheldon RA, Black SM, et al. Nitric oxide synthase activity and inhibition after neonatal hypoxia ischemia in the mouse brain. Brain Res Dev Brain Res. 2000;123(2):119-127. [DOI] [PubMed] [Google Scholar]
- 24.Iadecola C, Li J, Ebner TJ, Xu X. Nitric oxide contributes to functional hyperemia in cerebellar cortex. Am J Physiol. 1995;268(5 pt 2):R1153-R1162. [DOI] [PubMed] [Google Scholar]
- 25.Elrod JW, Calvert JW, Gundewar S, et al. Nitric oxide promotes distant organ protection. Proc Natl Acad Sci U S A. 2008;105(32):11430-11435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xia Z, Vanhoutte PM. Nitric oxide and protection against cardiac ischemia. Curr Pharm Des. 2011;17(18):1774-1782. [DOI] [PubMed] [Google Scholar]
- 27.Lundberg JO, Weitzberg E, Gladwin MT. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat Rev Drug Discov. 2008;7(2):156-167. [DOI] [PubMed] [Google Scholar]
- 28.Chouchani ET, Methner C, Nadtochiy SM, et al. Cardioprotection by S-nitrosation of a cysteine switch on mitochondrial complex I. Nat Med. 2013;19(6):753-759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Alencar JL, Lobysheva I, Chalupsky K, et al. S-nitrosating nitric oxide donors induce long-lasting inhibition of contraction in isolated arteries. J Pharmacol Exp Ther. 2003;307(1):152-159. [DOI] [PubMed] [Google Scholar]
- 30.Perkins GD, Ji C, Deakin CD, et al. A randomized trial of epinephrine in out-of-hospital cardiac arrest. N Engl J Med. 2018;379(8):711-721. [DOI] [PubMed] [Google Scholar]
- 31.Hansen M, Schmicker RH, Newgard CD, et al. Time to epinephrine administration and survival from nonshockable out-of-hospital cardiac arrest among children and adults. Circulation. 2018;137(19):2032-2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nakahara S, Tomio J, Nishida M, et al. Association between timing of epinephrine administration and intact neurologic survival following out-of-hospital cardiac arrest in Japan. Acad Emerg Med. 2012;19(7):782-792. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Trial protocol
eTable 1. Characteristics of randomized and missed subjects
eTable 2. Factors associated with admission to the hospital (n=1309)
eTable 3. Pre-specified and post-hoc out-of-hospital and hospital variables
eTable 4. Hospital outcomes (lowest 25th (<14.6 minutes) percentile time from call to randomization, VF after EMS arrival)
eTable 5. Hospital outcomes (highest 25th (>21.6 minutes) percentile time from call to randomization, VF after EMS arrival)
eFigure. Forest plot of absolute differences in percent admitted to hospital for prespecified subgroups
Data sharing statement