Abstract
The COVID-19 pandemic has severely impacted public health worldwide. Evidence of SARS-CoV-2 transmission via aerosols and surfaces has highlighted the need for efficient indoor disinfection methods. For instance, the use of ozone gas as a safe and potent disinfectant against SARS-CoV-2 virus is of particular interest. Here we tested the use of pseudoviruses as a model for evaluating ozone disinfection of the coronavirus at ozone concentrations of 30, 100, and 1000 ppmv. Results show that ozone disinfection rate of pseudoviruses was similar to that of coronavirus 229E (HuCoV-229E) at short contact times, below 30 min. Viral infection decreased by 95% following ozone exposure for 20 min at 1000 ppmv, 30 min at 100 ppmv and about 40 min at 30 ppmv. This findings mean that ozone is a powerful disinfectant toward the enveloped pseudovirus even at low ozone exposure. We also showed that viral disinfection occurs on various contaminated surfaces, with a positive association between disinfection and surface hydrophilicity. Infected surfaces made of aluminum alloy, for example, were better disinfected with ozone as compared to brass, copper, and nickel surfaces. Lastly, we demonstrate the advantage of ozone over liquid disinfectants by showing similar viral disinfection on top, side, bottom, and interior surfaces. Overall, our study demonstrates the potential use of ozone gas disinfection to combat the COVID-19 outbreak.
Electronic supplementary material
The online version of this article (10.1007/s10311-020-01160-0) contains supplementary material, which is available to authorized users.
Introduction
The COVID-19 pandemic has severely impacted public health, with almost 63 M total cases and 1.5 M deaths worldwide as of November 2020 (WHO 2020). The ongoing pandemic is caused by a new coronavirus (SARS-CoV-2), causing severe respiratory tract infection in humans. SARS-CoV-2 is highly contagious and spreads by close personal contact through respiratory droplets, or indirectly through either infected surfaces or aerosols (Ashour et al. 2020; Kampf et al. 2020). Liquid disinfectants—such as ethanol, hypochlorite, and quaternary ammonium—are often applied to reduce dissemination through indirect pathways. However, these liquid-based disinfectants suffer from technical and economical limitations, including transportation requirements, cost, difficulty in application on hard-to-reach locations, and a large environmental footprint. An attractive, less employed, alternative disinfectant is gaseous ozone.
Ozone is a powerful oxidant frequently used for inactivating pathogenic microorganisms in water and wastewater (Crini and Lichtfouse 2019; Mecha and Chollom 2020; von Gunten 2003). In addition to its qualities as a water disinfectant, ozone gas has been successfully applied for the disinfection of viruses on surfaces and in aerosols (Cannon et al. 2013; Tseng and Li 2006). Tseng and Li (2008) have found that ozone exposure—a product of exposure time and ozone concentration—of 9–52 ppm·min results in 90% inactivation (1-log10) of four different bacteriophages placed on a gelatin-based surface. Hudson et al. (2009) have suggested the use of a mobile ozone generator to disinfect rooms in health care facilities and hotels. Over 3-log10 inactivation of 12 different viruses placed on porous surfaces was achieved by applying ozone at peak gas concentration of 20–25 ppm with an exposure time of 45 min (Hudson et al. 2009). Recently, Yeargin et al. (2016) concluded that gaseous ozone is one of the most efficient disinfectants to treat enteric viruses on soft surfaces (e.g., carpets). However, studies evaluating ozone disinfection of coronaviruses at specific local conditions are scarce (Hudson et al. 2009).
A key challenge in the development and optimization of new technologies to fight COVID-19 is the difficulty to work directly with SARS-CoV-2, which requires biosafety level 3 (BSL3) or higher. In light of these severe restrictions, less-pathogenic viruses have been suggested as representative models to develop methods preventing SARS-CoV-2 infection (Aquino De Carvalho et al. 2017; Buonanno et al. 2020; Geller et al. 2012; Kampf et al. 2020; Liu et al. 2020). For example, HuCoV-229E is a human coronavirus which originated from bats, displays structural and genomic similarity to SARS-CoV-2 (Li et al. 2020) and is currently used as a model system to demonstrate disinfection technologies (Buonanno et al. 2020; Lee et al. 2020). An alternative approach to overcome biosafety limitations of infectious coronaviruses (CDC 2020) is the use of pseudotyped, non-replicative viral particles (Letko et al. 2020). Today, there are several technologies based on either Retrovirus, Lentivirus, or Vesicular Stomatitis Virus (VSV) to generate single cycle infecting particles (Steffen and Simmons 2016). In principle, the genes required for particle assembly—except the envelope protein responsible for cell recognition and entry—are co-transfected to susceptible cells together with a vector encoding an envelope protein from a different virus to create virus particles that present the target envelop protein on their membrane. Such pseudoviruses may offer simplicity and high throughput, while still demonstrating high similarity to the infective virus envelope and internalization mechanism (i.e., spike-dependent entry of these particles into cells) (Crawford et al. 2020; Walls et al. 2020). Pseudoviruses are frequently employed for fundamental mechanistic immunity and neutralization assays; however, their use as a model in disinfection studies has never been attempted.
The main goals of this study are to (1) demonstrate the use of pseudoviruses as a model for SARS-CoV-2 in ozone disinfection and (2) use the pseudoviruses to determine the feasibility of ozone for indoor inactivation of SARS-CoV-2. For this purpose, we first compared the ozone disinfection rate of the pseudoviruses to coronavirus 229E (HuCoV-229E). Subsequently, we used the pseudoviruses to determine the efficacy of ozone in minimizing coronaviruses infectivity on different surfaces and at various ozone exposures. Finally, we applied gaseous ozone for inactivating viruses placed at different, hard-to-reach, locations (e.g., inside a closet), and validated its advantage over liquid chemicals for indoor SARS-CoV-2 disinfection. Our work shows the potential use of ozone gas disinfection to combat COVID-19 outbreak and propose a simple pseudoviral model to enable the design and development of ozone-based systems for the disinfection of infected surfaces and aerosols.
Experimental
Viral infection tests
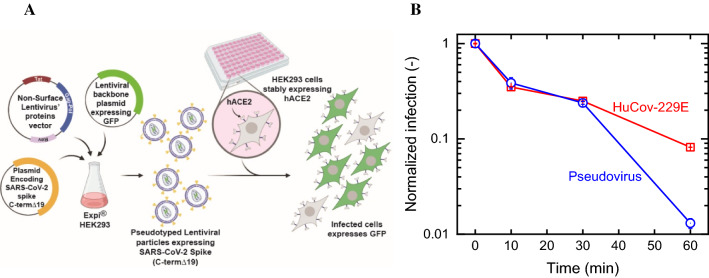
SARS-CoV-2-Spike pseudoviruses were synthesized by co-transfection of Expi293F™ cells with three plasmids encoding SARS-CoV-2-Spike (pCMV delta R8.2, pLenti-GFP, and pCMV3-ORF SΔC19 at a ratio of 1:2:1, respectively). Post transfection, cell debris were removed, and the pseudoviruses-containing supernatant was concentrated to yield 1 × 106 particles per mL. To evaluate the pseudovirus infectivity following ozone treatment, reconstituted pseudoviruses were added to HEK-293 cells stably expressing hACE2 (Fig. 1a). After 48 h, cells media was replaced with fresh DMEM (Sigma Aldrich, Israel) media excluding Phenol Red. Cells were imaged using IncuCyte ZOOM system (488 nm-channel, Essen BioScience) after an additional 24 h to calculate the number of GFP-positive cells from four images in each well. Coronavirus 229E (HuCoV-229E) was analyzed at the laboratories of Smart Assays (Nes Tsiona, Israel), using MRC-5 cells (ATCC, VA, USA). Following exposure to ozone, HuCoV-229E viruses were mixed MRC-5 cells and incubated for 120 h at 35 °C in CO2 incubator. Cells’ viability was determined using Cell titer glo reagent and viral infectivity, and the corresponding tissue culture infective dose (TCID50) was calculated. Further details of the synthesis method of SARS-CoV-2-Spike pseudoviruses and infection tests of both viruses can be found in the Supporting Information (SI).
Fig. 1.
A Pseudovirus production, left, and usage, right. B Kinetics of viral infection following exposure to ozone (100 ppmv) at different exposure times of HuCoV-229E and SARS-CoV-2-Spike pseudoviral model. Relative humidity and temperature were 60% and 25 °C. Viral infection values were normalized to those obtained in control experiments to assess any changes arising from the reconstitution procedure and drying effects. Each data point represents the average and standard error of three repetitions
Ozone disinfection setup
Ozone gas was produced by either an air-fed UV light ozone generator (APS 450, Pool-Purity, Israel) for low- and medium-ozone concentrations (30 and 100 ppmv, respectively), or a corona-discharge generator for high ozone levels (BMT 802 N, Germany). Ozone concentration was measured using low-range (106 M, 2B-Technologies, CO, USA) or high-range (BMT 963, Germany) ozone analyzers. Ozone generators and analyzers were connected to an 18 L sealed reaction chamber. In the chamber, relative humidity and temperature were set and maintained at 60% (± 3%) and 25 °C (± 1 °C) using humidity and temperature controllers (INKBIRD, China). Ozone off-gas was decomposed prior to release using a catalytic scrubber (Bircon, Israel).
Experimental procedure
Inactivation experiments took place in the reaction chamber connected to the ozone generators and analyzers (Fig. S1). All ozone experiments were conducted on virally contaminated surfaces. In a typical experiment, drops of concentrated viruses were placed on sterile surfaces (13 µL), and the surfaces were immediately inserted into the reaction chamber and exposed to ozone. Surfaces were removed from the chamber at different time intervals (different exposure times), reconstituted using DMEM media, and analyzed for the virus’s infectivity. All experiments were conducted in duplicates or triplicates.
Surface characterization
Six types of commonly-used surface materials were studied: glass (microscope slides), stainless steel (316), copper (C110), brass (C260), aluminum alloy (6061), and nickel (foil, GoodFellow). Hydrophilicity and surface functionality of the inanimate surfaces used in this study were determined using static contact angle measurements and Fourier-transform infrared spectroscopy—attenuated total reflection (FTIR-ATR), respectively. Contact angle was measured by placing a 13 μL water droplet (γ = 72.8 mN m−1) on each surface and analyzing left and right contact angles from digital images. FTIR-ATR (Tensor 27, Bruker, USA) analysis was conducted between 400 and 4000 cm−1 at spectral resolution of 4 cm−1 with 16 scans. The spectrum obtained was normalized to the background spectrum.
Results and discussion
Corona pseudoviruses can be used to assess ozone disinfection
Viral infection following ozone treatment was evaluated for the SARS-CoV-2-Spike pseudoviruses and compared to ozone inactivation of coronavirus 229E (HuCoV-229E). As mentioned above, HuCoV-229E coronavirus displays structural and genomic resemblance to SARS-CoV-2 (Li et al. 2020); because morphological and genomic structure are major contributors to viral inactivation kinetics, we hypothesize that HuCoV-229E’s reaction rate with ozone is approximately the same as SARS-CoV-2. Virus-contaminated plates (tissue culture treated 6-well polystyrene plates, Corning, USA) were exposed to ozone gas in the reaction chamber and to air (i.e., controls to account for all non-ozone inactivation paths). Figure 1b show the relative infectivity of the virus at different exposure times with 100 ppmv ozone (normalized to controls). HuCoV-229E and the pseudovirus showed similar reduction in infectivity after exposure times of 10 (~ 65%) and 20 min (7%); however, longer exposure (60 min) resulted in different reductions: ~ 99% for the pseudovirus and 92% for HuCoV-229E. The difference in the viruses’ behavior at long contact times may arise from a drying effect of the virus suspension. Specifically, following 60 min, the drop was completely dry for both viruses; preliminary tests showed that the pseudovirus reconstitution and infectivity are more sensitive to drying than HuCoV-229E.
Coronaviruses are enveloped, single-stranded, positive-sense RNA viruses. Ozone inactivation of enveloped viruses most often takes place through oxidation and disruption of the bilayer lipid structure (i.e., envelope) or the attached glycoproteins, which leads to loss of the virus’s infectivity (Bogler et al. 2020; Sunnen 2005, 1988). The pseudovirus employed herein accurately mimics the outer layer of SARS-CoV-2, including its enveloped surface and spike proteins, which may explain its similar disinfection rates with HuCoV-229E. Therefore, pseudoviruses may act as a surrogate to assess the effectivity of ozone treatment (within short contact times). Moving forward, we used the pseudoviral model to evaluate the applicability of ozone as an indoor disinfectant for coronavirus.
Ozone decreases virus infectivity in a wide range of concentrations
In this section, ozone inactivation of the SARS-CoV-2-Spike pseudovirus on polystyrene plates was measured for incubation at three ozone concentrations: 30 ppmv (low), 100 ppmv (medium), and 1000 ppmv (high). As expected, increasing ozone concentration reduced the pseudovirus’ infectivity (Fig. 2). For example, at 30 min exposure time, inactivation reached 90% (1-log10), 94% (1.2-log10), and 99% (2-log10) for the low, medium, and high ozone concentrations (respectively).
Fig. 2.
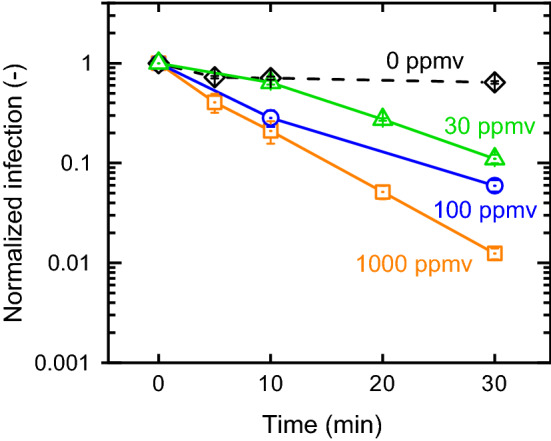
Infection of the SARS-CoV-2-Spike pseudoviruses following exposure to three ozone concentrations in different time intervals. All data were normalized to viral infection of freshly reconstituted drop. Relative humidity and temperature were 62% and 26 °C. Each data point represents the average and standard error of three repetitions
Quantitative comparison to other studies and viruses is not straight forward, due to variations in key experimental conditions such as size and dryness of the virus sample (for example, in most studies viruses were dried prior to ozone exposure). Experimental conditions in tests conducted in a large 65 m3 room, applying ozone at peak concentrations of 28 ppm and relative humidity of 40%, are comparable with the conditions in our low ozone tests (Hudson et al. 2009). These studies showed that total exposure time of 60 min resulted in at least 2-log10 inactivation of various viruses including herpes simplex virus 1, rhinovirus, and poliovirus. In our case, a 1-log10 inactivation was achieved with 30 ppmv ozone and exposure time of 30 min. Hence, we estimate that ozone inactivation rate of the pseudoviruses (simulating coronavirus) is in the same range of other previously tested viruses.
The inactivation effectivity at low ozone concentration and short exposure times strengths a newly emerged hypothesis, which links local increases in concentration of ambient (tropospheric) ozone (formed naturally by reaction of sunlight with hydrocarbons and nitrogen oxides) to the spread of the pandemic (Wang et al. 2020; Yao et al. 2020; Yu 2019). For example, a recent study published by Yao et al. (2020) found a negative correlation between ambient ozone levels and COVID-19 cases in major Chinese cities during Jan–March, 2020. A different study by Wang et al. (2020) showed that ambient ozone concentration largely increases in urban and rural areas during lockdown, which might contribute to the success of the lockdown in controlling COVID-19. Further studies, integrating statistical and engineering tools, should be performed to effectively determine the impact of ambient ozone on air-borne viruses in general and SARS-CoV-2 in particular.
Surface material affects viral infection
An important parameter affecting indoor disinfection is the surface material to be treated (Kampf et al. 2020; Tseng and Li 2008). The tested surfaces were selected based on their abundance in commonly-used indoor surfaces; nickel and copper, for example, are used in electrical devices through coating and wiring. Brass is a popular material used for decoration, musical instruments, and jewelry. Stainless steel is frequently used in kitchen appliances, tableware, and elevators including doors, walls, and bottom panels. Glass and aluminum alloys are also common materials for general-purpose use.
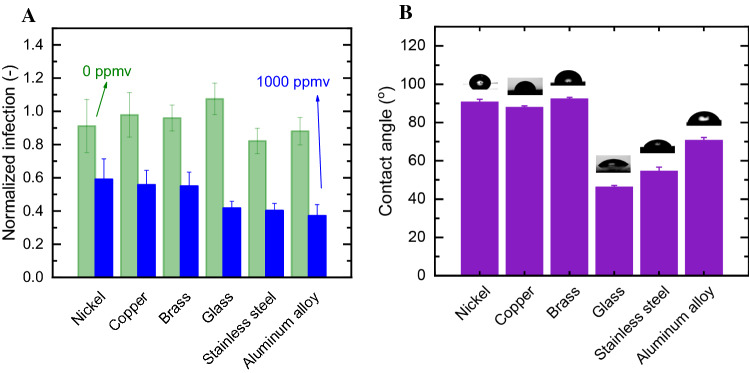
We evaluated the effect of inanimate surfaces on the disinfection rate by exposing pseudovirus-contaminated surfaces to a concentration of 1000 ppmv ozone for 5 min. This moderate ozone exposure assured that differences in performance can be monitored. Figure 3a shows the normalized infection arising from control experiments (green bars) and ozone treatment (blue bars). Controls showed minimal effect on infection due to irreversible adsorption, drying, or other physical interface phenomena. In contrast, ozone treatment strongly inhibited the pseudovirus’ infectivity on the tested surfaces, which ranged from 60% (nickel) to 37% (aluminum alloy). Analysis of variance (ANOVA) and Tukey’s post hoc test was used to evaluate differences between pseudovirus disinfection on different surfaces. The surface material had noticeable effect on inactivation; glass, stainless steel, and aluminum alloy surfaces statistically significantly (p < 0.05) enhanced ozone disinfection, compared to the other tested surfaces (nickel, copper, and brass). Our results agree with previous data published by Hudson et al. (2009), which found a similar ozone inactivation rate of viruses (including mouse coronavirus) placed on glass and stainless steel. The low inactivation rate for brass and copper was unexpected since these materials are known to generate reactive species under ambient environmental conditions (i.e., without ozone), which attack and inactivate attached viruses (Warnes et al. 2015).
Fig. 3.
A Material-dependent viral disinfection. Surfaced were contaminated with SARS-CoV-2-Spike pseudoviruses (1 × 106 particles per mL), exposed to 1000 ppmv ozone (blue) and 0 ppmv (green) for 5 min. Viral infection following exposure is presented as normalized infection to the reconstitution value of each surface examined. B Contact angle of the surfaces used in the study. Statistically significant differences (p < 0.05) were found between inactivation on hydrophobic surfaces compared to hydrophilic ones under ozone conditions, but not in air
To determine the cause for enhanced inactivation on aluminum, glass, and stainless steel, we further analyzed properties potentially contributing to this activity, such as surface hydroxyl groups (Ernst et al. 2004) and hydrophilicity. FTIR-ATR spectra (Figure S2) highlighted the OH stretching (3000–2900 cm−1) and bending (1400 cm−1) wavenumbers. While hydroxyl functional groups were observed for most surfaces, no clear association was found between surface functionality and infectivity following ozonation. However, contact angle—an indication of surface energy and hydrophilicity—could be associated with the normalized infection values (Fig. 3b), suggesting infectivity rates may increase with hydrophilicity. While nickel, copper, and brass are hydrophobic surfaces (i.e., contact angle larger than 90°), surfaces made of glass, stainless steel, and aluminum alloy are of hydrophilic nature. Therefore, we can hypothesize that surface hydrophilicity, which is associated with surface-functional groups, increases the overall ozone inactivation of the viruses. The reasons for this phenomenon might be enhanced ozone-surface interactions or shape variations of the liquid drop, impacting the mass transfer of ozone inside.
Ozone can effectively penetrate every part of a room
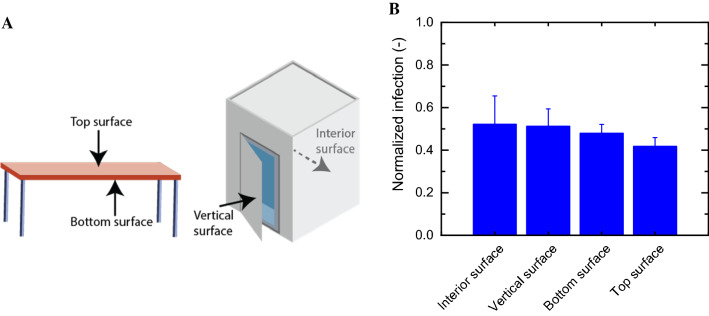
To demonstrate the advantage of gaseous disinfectants over liquid chemicals, we tested ozone inactivation of SARS-CoV-2-Spike pseudoviruses positioned in hard-to-reach locations. To do so, we equipped the reaction chamber with a miniature table and cabinet, and placed the contaminated surfaces below the table and inside the cabinet, simulating “real” indoor contamination events (Fig. 4a and Fig. S3). We used these contamination events in three-dimensional space to demonstrate the ability to treat entire rooms with gaseous ozone and the potential to effectively disinfect surfaces not typically disinfected with manually-applied liquid disinfectants, like the back of plane seats. Experiments were conducted with ozone concentration of 1000 ppmv and exposure time of 5 min. As can be seen in Fig. 4b, similar viral infection values were obtained for all surfaces (top, bottom, interior, and vertical). This suggests that applying ozone gas for disinfecting SARS-CoV-2 may overcome the low accessibility of commonly applied liquid disinfectants and enables a complete and effective disinfection.
Fig. 4.
A Positions by which a contaminated glass surface was placed and ozonated. B Ozone disinfection of several surface positions. Surfaced were contaminated with SARS-CoV-2-Spike pseudoviruses (1 × 106 particles per mL) and exposed to 1000 ppmv and 0 ppmv ozone (air) for 5 min. Viral infection following exposure is presented as normalized infection of 1000 ppmv and 0 ppmv
Other studies have proven ozone as an effective disinfectant and, if applied properly, it can be a powerful tool to minimize viral transmission indoors (Cristiano 2020; Yao et al. 2020; Zhou 2020). The gas is easy and economical to produce on-site, and it quickly reverts back to oxygen (half-life of about 20 min), leaving the treated area with no disinfection residues. On the other hand, the ozone processes must be optimized to prevent undesired phenomena such as corrosion and toxic effect on humans. Additionally, ozonation can be optimized and applied as a tertiary treatment for secondary effluents to ensure SARS-CoV disinfection and avoid transmission via wastewater (Medema et al. 2020; Randazzo et al. 2020), as an additional component of overall COVID-19 pandemic containment. Overall, this work suggests that development of intensive ozone-based disinfection systems could prove highly effective toward viruses similar to SARS-CoV-2, and proposes a simple model system for the design and optimization of such systems.
Conclusion
Our study suggests that corona pseudoviruses can be used as a viral model to assess ozone disinfection, owing to its similar ozone inactivation rate compared to coronavirus. We demonstrated that ozone decreases virus infectivity in a wide range of concentrations and exposure times, with 99% decrease recorded following 30 min of exposure to 1000 ppmv ozone. Surface material affected viral infection, with a positive association between disinfection and surface hydrophilicity. We eventually showed that ozone can penetrate every part of a room, suggesting ozone gas as an effective disinfectant for SARS-CoV-2 with high potential to overcome the low accessibility of commonly applied liquid disinfectants.
Supplementary information
Below is the link to the electronic supplementary material.
Supplementary material 1 (DOCX 15715 kb)
Acknowledgements
The authors would like to thank Dr. Bat-El Pinchasik and Hagit Gilron for their assistance with contact angle measurements, and Dr. Amit Kumar Sarkar for his assistance with FTIR analysis. We would like also to thank Dr. Alex Rouvinsky from the Hebrew University in Jerusalem for kindly providing the vector encoding the SARS-CoV-2-Spike truncated in its C-terminus (S∆C19).
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ines Zucker, Email: ineszucker@tauex.tau.ac.il.
Moshe Dessau, Email: moshe.dessau@biu.ac.il.
References
- Aquino De Carvalho N, Stachler EN, Cimabue N, Bibby K. Evaluation of Phi6 persistence and suitability as an enveloped virus surrogate. Environ Sci Technol. 2017;51:8692–8700. doi: 10.1021/acs.est.7b01296. [DOI] [PubMed] [Google Scholar]
- Ashour HM, Elkhatib WF, Rahman MM, Elshabrawy HA. Insights into the recent 2019 novel coronavirus (SARS-CoV-2) in light of past human coronavirus outbreaks. Pathogens. 2020;9:1–15. doi: 10.3390/pathogens9030186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bogler A, Packman A, Furman A, Gross A, Kushmaro A, Ronen A, Dagot C, Hill C, Vaizel-Ohayon D, Morgenroth E, Bertuzzo E, Wells G, Kiperwas HR, Horn H, Negev I, Zucker I, Bar-Or I, Moran-Gilad J, Balcazar JL, Bibby K, Elimelech M, Weisbrod N, Nir O, Sued O, Gillor O, Alvarez PJ, Crameri S, Arnon S, Walker S, Yaron S, Nguyen TH, Berchenko Y, Hu Y, Ronen Z, Bar-Zeev E. Rethinking wastewater risks and monitoring in light of the COVID-19 pandemic. Nat Sustain. 2020 doi: 10.1038/s41893-020-00605-2. [DOI] [Google Scholar]
- Buonanno M, Welch D, Shuryak I, Brenner DJ. Far-UVC light (222 nm) efficiently and safely inactivates airborne human coronaviruses. Sci Rep. 2020;10:10285. doi: 10.1038/s41598-020-67211-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cannon JL, Kotwal G, Wang Q. Inactivation of norovirus surrogates after exposure to atmospheric ozone. Ozone Sci Eng. 2013;35:217–219. doi: 10.1080/01919512.2013.771953. [DOI] [Google Scholar]
- CDC (2020) Interim laboratory biosafety guidelines for handling and processing specimens associated with coronavirus disease 2019 (COVID-19) [WWW Document]. Centers Dis Control Prev. https://www.cdc.gov/coronavirus/2019-ncov/lab/lab-biosafety-guidelines.html
- Crawford KHD, Eguia R, Dingens AS, Loes AN, Malone KD, Wolf CR, Chu HY, Tortorici MA, Veesler D, Murphy M, Pettie D, King NP, Balazs AB, Bloom JD. Protocol and reagents for pseudotyping lentiviral particles with SARS-CoV-2 spike protein for neutralization assays. Viruses. 2020;12:513. doi: 10.3390/v12050513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crini G, Lichtfouse E. Advantages and disadvantages of techniques used for wastewater treatment. Environ Chem Lett. 2019;17:145–155. doi: 10.1007/s10311-018-0785-9. [DOI] [Google Scholar]
- Cristiano L. Could ozone be an effective disinfection measure against the novel coronavirus (SARS-CoV-2) J Prev Med Hyg. 2020;61:301–303. doi: 10.15167/2421-4248/jpmh2020.61.3.1596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ernst M, Lurot F, Schrotter J-C. Catalytic ozonation of refractory organic model compounds in aqueous solution by aluminum oxide. Appl Catal B Environ. 2004;47:15–25. doi: 10.1016/S0926-3373(03)00290-X. [DOI] [Google Scholar]
- Geller C, Varbanov M, Duval RE. Human coronaviruses: insights into environmental resistance and its influence on the development of new antiseptic strategies. Viruses. 2012;4:3044–3068. doi: 10.3390/v4113044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson JB, Sharma M, Vimalanathan S. Development of a practical method for using ozone gas as a virus decontaminating agent. Ozone Sci Eng. 2009;31:216–223. doi: 10.1080/01919510902747969. [DOI] [Google Scholar]
- Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J Hosp Infect. 2020;104:246–251. doi: 10.1016/j.jhin.2020.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J, Bong C, Bae PK, Abafog AT, Baek SH, Shin Y-B, Park MS, Park S. Fast and easy disinfection of coronavirus-contaminated face masks using ozone gas produced by a dielectric barrier discharge plasma generator. medRxiv. 2020 doi: 10.1101/2020.04.26.20080317. [DOI] [PubMed] [Google Scholar]
- Letko M, Marzi A, Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat Microbiol. 2020;5:562–569. doi: 10.1038/s41564-020-0688-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Liu B, Cui J, Wang Z, Shen Y, Xu Y, Yao K, Guan Y (2020) Similarities and evolutionary relationships of COVID-19 and related viruses. Preprints 10.20944/preprints202003.0316.v1
- Liu DX, Liang JQ, Fung TS. Human coronavirus-229E, -OC43, -NL63, and -HKU1. Ref Modul Life Sci. 2020 doi: 10.1016/B978-0-12-809633-8.21501-X. [DOI] [Google Scholar]
- Mecha AC, Chollom MN. Photocatalytic ozonation of wastewater: a review. Environ Chem Lett. 2020;18:1491–1507. doi: 10.1007/s10311-020-01020-x. [DOI] [Google Scholar]
- Medema G, Heijnen L, Elsinga G, Italiaander R, Brouwer A. Presence of SARS-coronavirus-2 in sewage. medRxiv. 2020 doi: 10.1101/2020.03.29.20045880. [DOI] [PubMed] [Google Scholar]
- Randazzo W, Truchado P, Cuevas-Ferrando E, Simón P, Allende A, Sánchez G. SARS-CoV-2 RNA in wastewater anticipated COVID-19 occurrence in a low prevalence area. Water Res. 2020;181:115942. doi: 10.1016/j.watres.2020.115942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steffen I, Simmons G. Pseudotyping viral vectors with emerging virus envelope proteins. Curr Gene Ther. 2016;16:47–55. doi: 10.2174/1566523216666160119093948. [DOI] [PubMed] [Google Scholar]
- Sunnen GV. Ozone in medicine: overview and future directions. J Adv Med Fall. 1988;1:159–174. [Google Scholar]
- Sunnen G (2005) A virology primer: with special reference to ozone. Ozonics Int. report. http://ozoneinmedicine.com/virologyprimer.pdf
- Tseng CC, Li CS. Ozone for inactivation of aerosolized bacteriophages. Aerosol Sci Technol. 2006;40:683–689. doi: 10.1080/02786820600796590. [DOI] [Google Scholar]
- Tseng C, Li C. Inactivation of surface virases by gaseous ozone. Int Perspect. 2008;70:56–62. [PubMed] [Google Scholar]
- von Gunten U. Ozonation of drinking water: part II. Disinfection and by-product formation in presence of bromide, iodide or chlorine. Water Res. 2003;37:1469–1487. doi: 10.1016/S0043-1354(02)00458-X. [DOI] [PubMed] [Google Scholar]
- Walls AC, Park Y-J, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:281–292.e6. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Li M, Yu S, Chen X, Li Z, Zhang Y, Jiang L, Xia Y, Li J, Liu W, Li P, Lichtfouse E, Rosenfeld D, Seinfeld JH. Unexpected rise of ozone in urban and rural areas, and sulfur dioxide in rural areas during the coronavirus city lockdown in Hangzhou, China: implications for air quality. Environ Chem Lett. 2020;18:1713–1723. doi: 10.1007/s10311-020-01028-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warnes SL, Little ZR, Keevil CW. Human coronavirus 229E remains infectious on common touch surface materials. MBio. 2015;6:1–10. doi: 10.1128/mBio.01697-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO (2020) World Health Organization [WWW Document]. Situat. reports, Coronavirus Dis. 10.1515/9783110491326-003
- Yao M, Zhang L, Ma J, Zhou L. On airborne transmission and control of SARS-Cov-2. Sci Total Environ. 2020;731:139178. doi: 10.1016/j.scitotenv.2020.139178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeargin T, Buckley D, Fraser A, Jiang X. The survival and inactivation of enteric viruses on soft surfaces: a systematic review of the literature. Am J Infect Control. 2016;44:1365–1373. doi: 10.1016/j.ajic.2016.03.018. [DOI] [PubMed] [Google Scholar]
- Yu S. Fog geoengineering to abate local ozone pollution at ground level by enhancing air moisture. Environ Chem Lett. 2019;17:565–580. doi: 10.1007/s10311-018-0809-5. [DOI] [Google Scholar]
- Zhou M (2020) Ozone: a powerful weapon to combat COVID-19 outbreak [WWW Document]. http://www.china.org.cn/opinion/2020-02/26/content_75747237_4.htm
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material 1 (DOCX 15715 kb)