Abstract
Currently, COVID-19 has created difficulties in understanding the pathological mechanisms and therapeutic options for treatment. COVID-19 patients have shown to be hypoxic, and hypoxia causes alteration of the cell calcium dynamics, which leads to alterations in many signal transduction pathways and gene expression. Also, both viruses and hypoxia directly alter many pathological and biochemical pathways, such as inflammation, cytokine signaling, glycolysis, and calcium signaling. Therefore, understanding of these cellular events would be useful in finding therapeutic options.
Keywords: SARS-CoV-2, COVID-19, hypoxia, calcium signaling, inflammation, therapeutics
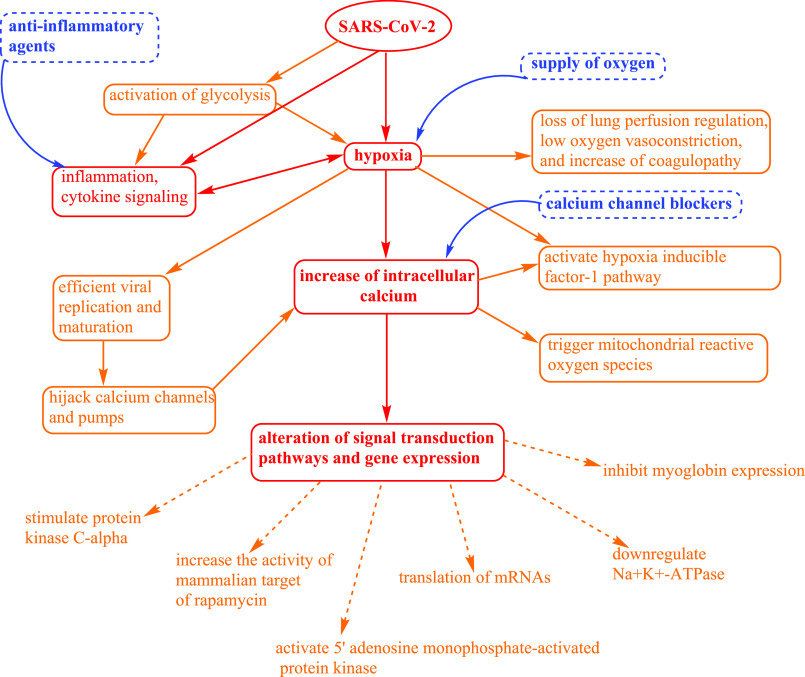
Human cells need a constant supply of oxygen for their survival, energy production, and normal function. However, hypoxia may lead to cell death. Therefore, many cellular mechanisms have been predicted of how cells adapt and behave in hypoxic conditions, which have been investigated through oxygen-sensing cells that how changes in the oxygen tension transduce signals into different organ systems and their functions. The hypoxic response is very complex as it regulates many signal transduction pathways and expression of genes. Recently, it has been reported that the novel SARS-CoV-2 causes hypoxia,1 and earlier investigation has supported that hypoxia leads to alteration in Ca2+ cell signaling.2 During hypoxia, an increase in intracellular Ca2+ concentration has been observed in many cells.3,4 Furthermore, the pathophysiological correlation between hypoxia signaling and inflammation are bidirectional. Hypoxia could activate the cytokine storm and inflammatory pathways and affect the fate of immune cells and functions.1 Recently, it has been reported that the severity and death of COVID-19 patients is due to the hyperinflammation and activation of cytokine signaling.5 Therefore, understanding these molecular mechanisms would be highly useful in prescribing or designing therapeutic approaches for the treatment of COVID-19 during the present global pandemic situation. The clinical symptoms of COVID-19 infected patients have been evidenced that the novel SARS-CoV-2 is responsible for causing hypoxia and respiratory distress. Earlier, it has been well established that a virus infection can trigger a metabolic shift and can activate the glycolytic process which leads to hypoxia and inflammation. Recently, it has been reported that the loss of lung perfusion regulation, low oxygen vasoconstriction, and increase of coagulopathy are the major mechanisms of hypoxia in COVID-19.6 The critical hypoxic state observed in COVID-19 patients is due to the small increase in partial arterial pressure of oxygen, even though the increase amount of inspired oxygen fraction delivered has been significantly noted during conventional oxygen supplementation.7 Additionally, many viruses replicate efficiently under hypoxia.8 Viruses also reprogram the host cellular function, which is an essential strategy for their replication, maturation, and survival, including hijacking the calcium channels and pumps and increasing the intracellular Ca2+ load.9
SARS-CoV-2 has been reported to activate the hypoxia-inducible factor (HIF-1) pathway.1 Therefore, it can induce many downstream processes such as alteration of host cellular metabolism, acceleration of inflammation, and facilitation of rapid viral replication. In a hypoxic state, cells generally regulate the expression of genes, mostly those involved in glycolysis and metabolism of iron and in the control of angiogenesis for adaptation and survival. HIF-1 is the transcriptional activator of several genes concerning cellular adaption in the hypoxic state.1 HIF-1α can increase the expression of glycolytic genes through binding hypoxia-responsive elements of glycolytic promoter genes.8
Furthermore, lactic acid is another viral-induced glycolytic product which reduces blood pH and causes respiratory acidosis and hypercapnia. Recently, an increase in lactate dehydrogenase (LDH) has been observed in severe COVID-19 patients.10 It is important to emphasize that increased LDH levels have been associated with severe disease conditions and an increased mortality rate of COVID-19 patients.10 Furthermore, LDH converts pyruvic acid to lactic acid during low oxygen environment. LDH has also been a marker of cardiac damage, therefore, its abnormal values may be due to multiple organ injury and may also be due to hypoxia and upregulation of the glycolytic pathway. The critical respiratory conditions in COVID-19 patients may be due to the increase of LDH levels.
Besides, hypoxia also leads to alteration of Ca2+ signaling in cells. The experimental investigation suggested that hypoxia could increase the intracellular Ca2+ concentration in pulmonary arterial myocytes through triggering mitochondrial reactive oxygen species (ROS).4 ROS are the high energetic free radical species which can cause damage to macro-biomolecules, such as nucleic acids, proteins, lipids, and carbohydrates, and ultimately lead to apoptotic cell death.11 Through an in vitro study, it has been analyzed that chronic hypoxia altered the Ca2+ signaling in airway smooth muscle cells, which led to an increase in airway responsiveness.12 Furthermore, it has been reported that hypoxia can stimulate translation of HIF-1α and HIF-2α through the distribution of HIF-α mRNAs to a larger fraction of polysomes. This process needs extracellular Ca2+ entry, stimulation of protein kinase Cα (cPKCα), and the activity of mammalian target of rapamycin (mTOR). This experiment has established an important physiological mechanism that hypoxia stimulates extracellular Ca2+ entry selectively, which induces the translation of mRNAs that required adaptation under hypoxia conditions.13 Working with rats’ hippocampal neurons, it was concluded that both types of high voltage-activated (HVA) Ca2+ channels such as N-type and L-type were sensitive in hypoxia conditions. Also, the L-type Ca2+ channel was found to be more sensitive to a low oxygen environment.14 Furthermore, experimental data suggested that in hypoxia, Ca2+ influx via Ca2+ release-activated Ca2+ (CRAC) channels led to Na+K+-ATPase downregulation, 5′ adenosine monophosphate-activated protein kinase (AMPK) activation, and alveolar epithelial dysfunction.2 To understand the role of Ca2+ in signal transduction pathways which regulate gene expression, dopaminergic pheochromocytoma (PC12) on the oxygen sensing cell line has been used in hypoxic conditions. The results showed an immediate response from PC12 cells causing membrane depolarization and an increase of the intracellular Ca2+ concentration in hypoxia. It was concluded that the increase of intracellular Ca2+ led to cell injury and death due to hypoxia.3 Hypoxia can also be responsible for the inhibition of myoglobin expression through the alteration of Ca2+ entry in response to cell depolarization or by the depletion of Ca2+ storage from the endoplasmic reticulum.15 The cellular events due to SARS-CoV-2, hypoxia, inflammation, and Ca2+ signaling along with the therapeutic options have been summarized in Figure 1.
Figure 1.
Cellular events due to SARS-CoV-2, hypoxia, inflammation, and Ca2+ signaling and the therapeutic options.
Therefore, clinical investigation on the application of oxygen, anti-inflammatory agents, and calcium channel blockers for the treatment of COVID-19 patients could be very useful.
Acknowledgments
The author is highly thankful to Dr. Maghsoud Alizad Farrokhi and Mr. Rahul Malhotra for proofreading.
The author declares no competing financial interest.
This article is made available via the ACS COVID-19 subset for unrestricted RESEARCH re-use and analyses in any form or by any means with acknowledgement of the original source. These permissions are granted for the duration of the World Health Organization (WHO) declaration of COVID-19 as a global pandemic.
References
- Serebrovska Z. O.; Chong E. Y.; Serebrovska T. V.; Tumanovska L. V.; Xi L. (2020) Hypoxia, HIF-1α, and COVID-19: From pathogenic factors to potential therapeutic targets. Acta Pharmacol. Sin. 41 (12), 1539–1546. 10.1038/s41401-020-00554-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gusarova G. A.; Trejo H. E.; Dada L. A.; Briva A.; Welch L. C.; Hamanaka R. B.; Mutlu G. M.; Chandel N. S.; Prakriya M.; Sznajder J. I. (2011) Hypoxia leads to Na,K-ATPase downregulation via Ca(2+) release-activated Ca(2+) channels and AMPK activation. Mol. Cell. Biol. 31 (17), 3546–3556. 10.1128/MCB.05114-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seta K. A.; Yuan Y.; Spicer Z.; Lu G.; Bedard J.; Ferguson T. K.; Pathrose P.; Cole-Strauss A.; Kaufhold A.; Millhorn D. E. (2004) The role of calcium in hypoxia-induced signal transduction and gene expression. Cell Calcium 36 (3–4), 331–340. 10.1016/j.ceca.2004.02.006. [DOI] [PubMed] [Google Scholar]
- Waypa G. B.; Marks J. D.; Mack M. M.; Boriboun C.; Mungai P. T.; Schumacker P. T. (2002) Mitochondrial reactive oxygen species trigger calcium increases during hypoxia in pulmonary arterial myocytes. Circ. Res. 91 (8), 719–26. 10.1161/01.RES.0000036751.04896.F1. [DOI] [PubMed] [Google Scholar]
- Cao X. (2020) COVID-19: Immunopathology and its implications for therapy. Nat. Rev. Immunol. 20 (5), 269–270. 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan C. W.; Low J. G. H.; Wong W. H.; Chua Y. Y.; Goh S. L.; Ng H. J. (2020) Critically ill COVID-19 infected patients exhibit increased clot waveform analysis parameters consistent with hypercoagulability. Am. J. Hematol. 95 (7), E156–E158. 10.1002/ajh.25822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gattinoni L.; Coppola S.; Cressoni M.; Busana M.; Rossi S.; Chiumello D. (2020) COVID-19 does not lead to a ″Typical″ acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 201 (10), 1299–1300. 10.1164/rccm.202003-0817LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ren L.; Zhang W.; Han P.; Zhang J.; Zhu Y.; Meng X.; Zhang J.; Hu Y.; Yi Z.; Wang R. (2019) Influenza A virus (H1N1) triggers a hypoxic response by stabilizing hypoxia-inducible factor-1α via inhibition of proteasome. Virology 530, 51–58. 10.1016/j.virol.2019.02.010. [DOI] [PubMed] [Google Scholar]
- Danta C. C. (2020) Calcium channel blockers: A possible potential therapeutic strategy for the treatment of Alzheimer’s dementia patients with SARS-CoV-2 infection. ACS Chem. Neurosci. 11 (15), 2145–2148. 10.1021/acschemneuro.0c00391. [DOI] [PubMed] [Google Scholar]
- Henry B. M.; Aggarwal G.; Wong J.; Benoit S.; Vikse J.; Plebani M.; Lippi G. (2020) Lactate dehydrogenase levels predict coronavirus disease 2019 (COVID-19) severity and mortality: A pooled analysis. Am. J. Emerg. Med. 38 (9), 1722–1726. 10.1016/j.ajem.2020.05.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danta C. C.; Piplani P. (2014) The discovery and development of new potential antioxidant agents for the treatment of neurodegenerative diseases. Expert Opin. Drug Discovery 9 (10), 1205–1222. 10.1517/17460441.2014.942218. [DOI] [PubMed] [Google Scholar]
- Belouchi N. E.; Roux E.; Savineau J. P.; Marthan R. (1999) Effect of chronic hypoxia on calcium signalling in airway smooth muscle cells. Eur. Respir. J. 14 (1), 74–79. 10.1034/j.1399-3003.1999.14a13.x. [DOI] [PubMed] [Google Scholar]
- Hui A. S.; Bauer A. L.; Striet J. B.; Schnell P. O.; Czyzyk-Krzeska M. F. (2006) Calcium signaling stimulates translation of HIF-alpha during hypoxia. FASEB J. 20 (3), 466–475. 10.1096/fj.05-5086com. [DOI] [PubMed] [Google Scholar]
- Lukyanetz E. A.; Shkryl V. M.; Kravchuk O. V.; Kostyuk P. G. (2003) Action of hypoxia on different types of calcium channels in hippocampal neurons. Biochim. Biophys. Acta, Biomembr. 1618 (1), 33–38. 10.1016/j.bbamem.2003.10.003. [DOI] [PubMed] [Google Scholar]
- Kanatous S. B.; Mammen P. P.; Rosenberg P. B.; Martin C. M.; White M. D.; Dimaio J. M.; Huang G.; Muallem S.; Garry D. J. (2009) Hypoxia reprograms calcium signaling and regulates myoglobin expression. Am. J. Physiol. Cell Physiol. 296 (3), C393–402. 10.1152/ajpcell.00428.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]