Abstract
IgG4-related disease (IgG4-RD) represents an immune-mediated fibroinflammatory condition with peculiar histopathologic changes that can affect various organs. In 2012 its unified nomenclature was published, which allows to abandon other synonymous names. Up to now, only little is known about its epidemiology around the world. However, although it is generally considered a rare condition, the number of patients with IgG4-RD is increasing enormously. Likewise, the annual number of publications on this subject has increased progressively. The spectrum of clinical manifestations in IgG4-RD is highly variable, depending on the severity of the disease as well as the presence of organ(s) involvement. This review gives an overview on changing epidemiology of IgG4-RD focusing the attention on the large cohorts of patients published in the literature.
Keywords: IgG4, IgG4-related disease, Epidemiology, Genetic factors
Highlights
-
•
IgG4-related disease represents a multi-systemic immune-mediated fibroinflammatory condition.
-
•
The peculiar histopathology features include lymphoplasmacytic infiltration, storiform fibrosis, and obliterative phlebitis.
-
•
From the epidemiological point of view, Asian patients seem more susceptible to IgG4-related disease than others.
-
•
The disease affects individuals of middle to upper age, but is increasingly recognized to be diagnosed in children.
1. Introduction
IgG4-related disease (IgG4-RD) is a rare multiorgan condition characterized by three pathology features: lymphoplasmacytic infiltration, storiform fibrosis, and obliterative phlebitis [1]. The hallmark of this condition is the IgG4-positive plasma cell infiltration in affected organs. Various organs may be either simultaneously or consecutively be involved (Table 1 [2]), of whom the most commonly affected are pancreas, biliary tree, salivary and lacrimal glands, retroperitoneum and lymph nodes. The disease has been underrecognized for many years and was firstly described in 2003 in 7 patients with an initial diagnosis of autoimmune pancreatitis (AIP) in whom an extensive organ involvement with IgG4-positive plasma cell infiltration was described [3].
Table 1.
Organ involvement in IgG4-RD (from Della-Torre et al., modified [2].
| Head and neck | |
|---|---|
| Orbits and periorbital tissue | IgG4-related orbital disease |
| Salivary and lacrimal glands | IgG4-related sialadenitis |
| Thyroid | IgG4-related thyroiditis |
| Ear, nose, throat | IgG4-related sinusitis |
| Thorax | |
| Lungs | IgG4-related lung disease |
| Pleura | IgG4-related pleural disease |
| Mediastinum | IgG4-related mediastinitis |
| Breast | IgG4-related mastitis |
| Abdomen and pelvis | |
| Retroperitoneum | IgG4-related retroperitoneal fibrosis |
| Pancreas | IgG4-related pancreatitis |
| Biliary tree | IgG4-related sclerosing cholangitis |
| Liver | IgG4-related autoimmune hepatitis |
| Kidney | IgG4-related tubule-interstitial nephritis/glomerulonephritis |
| Gastrointestinal tract | IgG4-related gastrointestinal disease |
| Mesentery | IgG4-related sclerosing mesenteritis |
| Prostate | IgG4-related prostatitis |
| Testis | IgG4-related epididymitis-orchitis |
| Nervous system | |
| Pituitary gland | IgG4-related hypophysitis |
| Peripheral nerves | IgG4-related neuropathy |
| Meninges | IgG4-related pachymeningitis |
| Cardiovascular system | |
| Heart and pericardium | IgG4-related cardiac disease |
| Aorta | IgG4-related periaortitis |
| Lymph nodes | IgG4-related lymph adenopathy |
| Skin | IgG4-related skin disease |
| Bone | IgG4-related disease of the bone |
The first International Symposium on IgG4-RD was held in Boston in 2011, when the name and definition of these conditions were recognized internationally. Subsequently, the 2nd and the 3rd International Symposia were organized in 2014 and 2017 respectively, in order to establish the diagnostic standard and guidelines. Finally, the 4th Symposium on IgG4-RD was planned in November 2020 in Japan, but it is postponed in December 2021 because of COVID-19 pandemic expansion. This review gives an overview on changing epidemiology of IgG4-RD focusing the attention on the cohorts published in the literature and on IgG4-related pancreatitis and IgG4-sclerosing cholangitis.
1.1. Diagnosis
The diagnosis of IgG4-RD is based on a combination of clinical, histopathologic, and serologic grounds. In 2014 the Japanese team published comprehensive diagnostic criteria for IgG4-RD [4]. Diagnostic criteria are based on: 1) organ enlargement, mass or nodular lesions, or organ dysfunction; 2) increased serum IgG4 concentration >135 mg/dl, and 3) infiltration of IgG4+ cells (>10 cells/high power field (HPF) and IgG4+/IgG+ cell ratio >40%). Patients who fulfil the organ-specific criteria (1,2,3) have a definite diagnosis. A diagnosis of IgG4-RD is possible in patients who fulfil criteria (1) and (2), but with negative results on histopathology or without histopathologic examination, whereas a diagnosis of IgG4-RD is probable in in patients with organ involvement (1), but without increased IgG4 concentration. As observed by Fox et al. [5], IgG4-RD tends to be both underdiagnosed and overdiagnosed. Underdiagnosis is due to a lack of recognition of this condition, mainly for the lack of utilizing a specific immunohistochemical approach. Overdiagnosis can result from a non-correct approach or interpretation of a slight increase in serum IgG4, or nonspecific infiltration of IgG4-positive cells in other mimickers. Useful methods of diagnosing IgG4-RD have been suggested by Umehara et al. [6] with combination of comprehensive diagnostic and organ-specific criteria, or the 2019 American College of Rheumatology (ACR)/European Legue Against Rheumatism (EULAR) classification criteria for IgG4-RD [7].
1.2. Pathogenesis
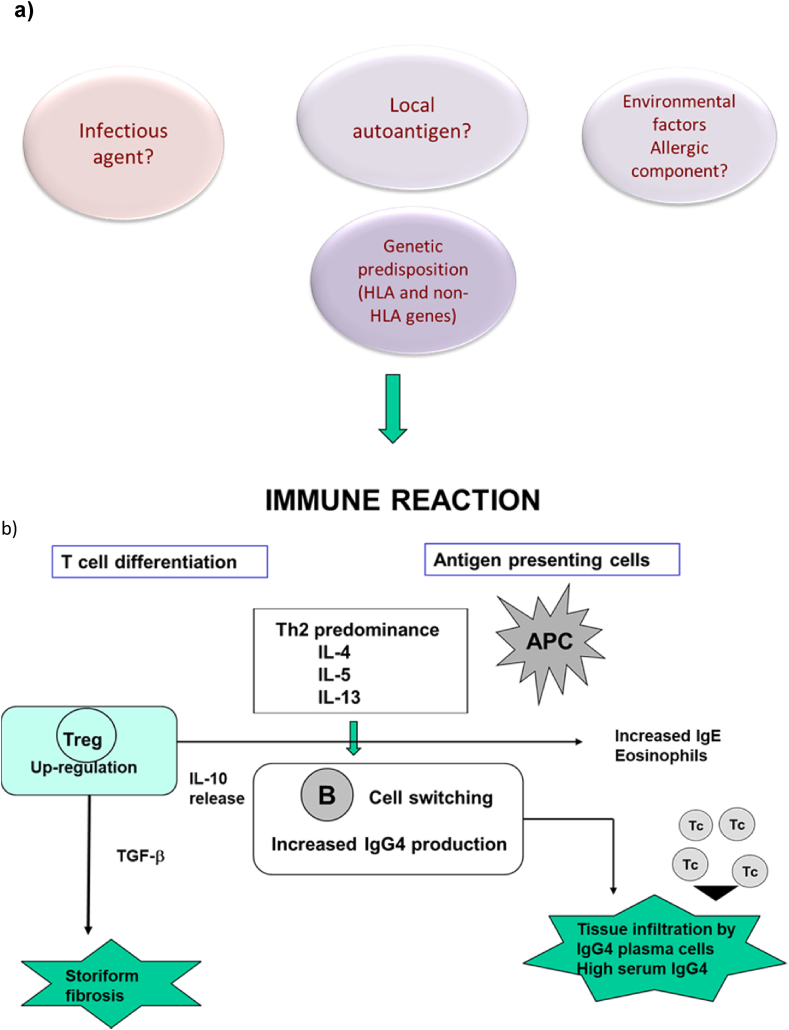
The pathogenic mechanisms underlying IgG4-RD are poorly understood. Indeed, it is not clear whether IgG4-RD is caused by an abnormal acquired immunity, like autoimmune disease, and whether the increased IgG4 production acts through a direct pathogenic mechanism triggering the disease (Fig. 1). It has been suggested that IgG4 have an anti-inflammatory effect, rather than a pro-inflammatory role [8]. The crosstalk between innate and acquired immunity has been hypothesized to have a key role [9]. An increased production of Th2 cytokines (IL-4, IL-5, IL-10. IL-13) has been found to be enhanced in IgG4-related pancreatitis and cholangitis [10]. These cytokines are potent activators of B-cell IgE production and eosinophil recruitment. Moreover, an increased infiltration of inducible-memory T regulatory cells (Tregs) in affected tissue and blood, with upregulation of IL-10 and tumour growth factor-β (TGF-β) has been observed in patients with IgG4-RD [11,12]. TGF-β production from Tregs may stimulate fibroblasts causing fibrogenesis, a prominent feature of advanced-stage disease. Moreover, Tregs can exert a regulatory function through IL-10 secretion, inducing IgG4-secreting plasma cells. Other potential sources of IL-10 include macrophage, dendritic cells and basophils. One study reported the involvement of IL-10 and TGF-β−secreting basophils in tissue of patients with IgG4-related submandibular gland disease [13]. BAFF and APRIL, in concert with IL-21 enhance IgG4 production via promoting the expansion of IgG4-committed B cells [14,15].
Fig. 1.
Pathogenic mechanisms of IgG4-RD: a) Eventual initial mechanisms include local autoantigens, infectious agents, allergic components in a genetically predisposed individual; b) These factors activate immune reaction in the target tissue (organ/organs) which is characterized by Th2 predominance with an increased production of cytokines (IL-4, IL-5, IL-13) and tumour growth factor -b (TGF-b). Moreover, an activation of Treg, a release of IL-10 and elevated serum IgE occur. The immune reaction also causes a B cell class switching with an increased production of IgG4 producing plasma cells. APC = antigen presenting cell; Tc = cytotoxic T lymphocyte; Treg = regulatory T cell.
1.3. Histopathology
The HISORt acronymous has been proposed for remember the clinical features of IgG4-RD: histopathology and immunostaining, imaging, serology, organ involvement, and response to therapy [16]. These criteria, originally developed for AIP, were adapted for IgG4-RD. First of all, histopathology and immunostaining must rule out malignant disease, which in practice will often require fine-needle aspiration, biopsy, or surgical samples. The major histological features include: i) a dense lymphoplasmacytic inflammatory infiltrate with increased numbers of IgG4+ plasma cells and often increased eosinophils; ii) a storiform pattern of fibrosis; iii) obliterative phlebitis [17]. IgG4+ plasma cells may be diffusely increased or distributed in variable dense patches. A Consensus Statement on the pathology of IgG4-RD was held in Boston in October 2011, with 35 International experts [18]. The quantitative assessment of the IgG4 staining was also recommended. In IgG4-related pancreatitis the finding of >30 IgG4+ plasma cells per high power field (hpf) has been reported to have acceptable specificity, otherwise a number >50/hpf is reported as highly specific [18]. The storiform fibrosis is a swirling pattern of fibrosis which may have a patchy distribution, and therefore be missed with small biopsies. It is constituted by fibrotic collagen deposition containing fibroblasts and myofibroblasts, in a similar way as fibro-histiocytic malignancies. Obliterative phlebitis can be identified in veins with inflammatory infiltrate filling both the walls and lumina [18].
1.4. Role of the immunoglobulin G4 molecule
The role of serum IgG4 concentration in the diagnosis of IgG4-RD is still debated. Based on the diagnostic criteria established by the Consensus Conference on IgG4 [7], a serum concentration of IgG4 >135 mg/dl has been included in the comprehensive criteria together with clinical symptoms (swelling or masses of single or multiple organs, histopathological findings (extensive infiltration of IgG4+ plasma cells and fibrosis), and imaging changes (depending on single or multiple organ involvement). A meta-analysis including 23 studies comprising 6.048 patients with IgG4-RD the pooled sensitivity was 85% (CI 78–90%), the pooled specificity was 93 (CI 90–95%), and area under curve was 0.95, which suggested a high diagnostic accuracy of serum IgG4 for the entity of IgG4-RD [19]. Limitation of this study include the variability of design in the studies and the poor quality of reporting in the primary studies. However, it should be noticed that in situations of chronic antigen exposure IgG4 result increased in 80% of cases, thus reflecting the development of humoral tolerance. In fact, IgG4 has been defined as “odd antibody” [20]. IgG4 is involved in attachment of heavy or light chain way in two different antigen-combining sites (this process has been defined as “Fab-arm exchange”). Another aspect is its tendency to mimic IgG rheumatoid factor activity by interacting with IgG on a solid support [21]. In many situations IgG4 are linked to allergies. In fact, both IgE and Ig4 require help by Th2 cells. Moreover, in clinical situations, such as pemphigoid disease, IgG4 is associated to a pathogenic effect [21]. A critical question is whether tissue infiltration with IgG4-positive plasma cells and increased serum IgG4 concentration contribute directly to disease pathogenesis. In patients with type 1AIP serum IgG4 was able to bind with normal human bile and pancreatic duct epithelia, a reaction that was abolished by steroids [22]. Evidence also exists on the correlation of serum IgG4 and severity of disease. In grafts from patients with primary sclerosing cholangitis (PSC) marked hilar IgG4-positive lymphoplasmacytic infiltration was found to be associated with the presence of dominant biliary strictures [23]. In IgG4-SC the presence of a subset of clonally expanded IgG4+ B cells in blood and affected tissue was demonstrated, suggesting that an antigen-mediated immune-response is pivotal in the pathogenesis of IgG4-SC [24]. Alternatively, this B cell expansion may reflect an amplified humoral response to an inflammatory environment with polyclonal expansion of pre-existing IgG4-switched B cells of multiple specificities [25].
1.5. Genetic factors
The majority studies on the role of genetic factors in the pathogenesis of IgG4-RD regard the autoimmune pancreatitis (AIP). An association between susceptibility for AIP and HLA genes has been studied in the Japanese population [26,27]. In the former study [26] 40 patients with AIP, 43 with chronic calcifying pancreatitis, and 201 healthy Japanese subjects were enrolled. An association between DRB1∗0405-DQB1∗0401 haplotype and susceptibility for AIP was observed [26]. In the latter study, further genetic analyses were addressed in the same population of the former study. The HLA-linked susceptibility region was confirmed for the DQB1∗0405-DQB1∗0401 haplotypes and was also observed an association between ABCF1 in the HLA I and AIP. Relapse in AIP was observed with HLA-A, HLA-C, HLA-DQB1 [27] and with the non-aspartic acid at DQB157 [27]. The newest genome-wide association study on IgG4-RD has been performed in Japan enrolling 857 patients with IgG4-RD and 2082 healthy controls [28]. Two susceptibility loci were identified: HLA-DRB1 and FCGR2B which may have important roles in IgG4-RD development. The strong genetic association between HLA DRB1 and IgG4-RD highlights the fundamental role of the HLA locus in the onset of immune-related disease, as in asthma [29]. On the other hand, FCGR2B is known to be a major genetic factor of systemic lupus erythematosus (SLE); The additional association in the variant rs1050501 found by the Japanese authors [28] has been found to destabilise the anchoring of FCGR2B in the cell membrane, resulting in a reduced suppressive effect on the immune response. These opposing genetic associations of HLA and FCGR2B between IgG4-RD and SLE are interesting and might in part explain the phenotypic differences between the two diseases.
Interestingly, a newly described gene variant in fibroblast growth factor binding protein 2 (FGFBP2) has been found in a heterozygous form in a father with IgG4-RD and in two sons with elevated plasma IgG4 concentrations and high levels of circulating IgG4 plasmablasts and CD4+ cytotoxic T cells [30]. This finding rises the possibility of familial clusters of IgG4-RD. Moreover, as FGFBP2 protein is secreted by cytotoxic T cells and binds fibroblast growth factor, an alteration in this protein might enhance the CD4+ T cell function, and consequently might play a role in the pathogenesis of IgG4-RD.
In addition to HLA loci, several non-HLA gene have been identified as risk genes in IgG4-RD [31,32] (Table 2). A high-throughput sequence analysis was performed in 27 Japanese patients with AIP with the aim to identify possible gene candidates for this disease [31]. The authors identified nine, eight and two candidate variants associated with the development of type 1 AIP, AIP relapse, and extra-pancreatic lesions. Among the 9 candidate genes associated to AIP susceptibility, P2RX3 (c.195delG) and TOP1 (c.2007delG) showed a high significant association. P2RX3 constitutes a positive autocrine signal for release of insulin from pancreatic β cells and contributes to the pancreatic pain in chronic pancreatitis. TOP1 which encodes for DNA topoisomerase, is associated with various autoimmune diseases. Moreover, a deletion in SMAD7 leading to a loss of TGF-β signalling, has been identified in the same study [31], as candidate genetic factor associated with the pathogenesis of AIP. A Chinese group in Taiwan firstly reported that the cytotoxic T antigen 4 (CTLA-4) gene polymorphism is associated with risk of IgG4-related pancreatitis [33]. More recently the same group reported an association study including 118 patients with IgG4-related pancreatitis vs 200 healthy controls [34]. They performed a genetic analysis for the cationic trypsinogen gene (protease, serine,1 [PRSS1] and the pancreatic secretory trypsin inhibitor gene (serine peptidase inhibitor, Kazal type 1 [SPINK1] both of which are involved in pancreatic acinar cell injury. They found a total of 19 PRSS1 variants and one SPINK1 variant in case subjects, but none in controls (p = 0.003). In the multivariate analysis PRSS1 was negatively associated with relapse of IgG4-related pancreatitis. These findings indicate that the reaction to PRSS1 or SPINK1 might stimulate the immune reaction that induces IgG4-related pancreatitis at very early stage. Indeed, the PRSS1 variants might be associated with endoplasmic reticulum stress and protein misfolding, with a consequent generation of a neoantigen and induction of autoantibodies.
Table 2.
Non-HLA genetic susceptibility for patients with IgG4-RD.
| Gene | SNP | Association | Country | Reference |
|---|---|---|---|---|
| CTLA4 | 49 A/G -318C/+49A/CT60G +6230 3’, untranslated region +6230G/G +6230A 49 A/A and +6230A/A genotypes |
IgG4-related pancreatitis IgG4-related pancreatitis IgG4-related pancreatitis IgG4-related pancreatitis IgG4-related pancreatitis IgG4-related pancreatitis |
China China Japan Japan Japan Japan |
Chang33 Umemura35 |
| TNFα | -863 A/G | Extra-pancreatic lesions | China | Chang34 |
| KCNA3 | rs2840381G>A | IgG4-related pancreatitis | Japan | Ota37 |
| FCGR | rs1058184A>C rs2640480C>A rs131978C>T rs1801274 |
Chronic periaortitis | Italy | Alberici39 |
| FCRL3 | -110A/A genotype | IgG4-related pancreatitis | Japan | Umemura36 |
| PRSS1 | c.2766G>T c346C>T c137C>T c415T>A c.416G>T |
IgG4-related pancreatitis | China | Chang32 |
| SPINK1 | 194 + 2(IVS3+2)TC | IgG4-related pancreatitis | China | Chang34 |
| P2RX3 | c.195delG | IgG4-related pancreatitis | China | Chang34 |
| TOP1 | c.2007delG | IgG4-related pancreatitis | China | Chang34 |
| CACNA1C | c.5996delC | IgG4-related pancreatitis | China | Chang34 |
| CXCR3 | c.630_631delGC | IgG4-related pancreatitis | China | Chang34 |
| MLL3 | rs111493987C>A | IgG4-related pancreatitis | China | Chang34 |
| CFTR | 26 variants | IgG4-related pancreatitis | China | Chang34 |
Umemura et al. [35] in Japan recruited 59 patients with IgG4-related pancreatitis, 62 patients with chronic calcifying pancreatitis and 97 healthy controls. They genotyped 4 SNPs in Fcγ receptor-like gene 3 (FCRL3) and found a significant association between-110 SNP and AIP. Interestingly, serum IgG4 concentrations were found to be significantly correlated with the number of susceptible alleles. The same group [36] studied 5 CTLA-4 polymorphisms in 59 patients with AIP and 102 healthy subjects. The +6230 G/G genotype was significantly increased in patients compared to controls, whereas haplotype 2 (+6230 A) was significantly associated with AIP resistance.
The same Japanese group [37] performed an association analysis using 400 microsatellite markers with an average spacing of 10.8 cM in the genome; they also evaluated 7 SNPs within member 3 gene (KCNA3) in 64 patients with AIP and 104 healthy controls. Six statistically significant markers for susceptibility were identified (D152726, D58410, D6S460, D105548, D15S128, D20S186). Further analysis by SNP genotyping in KCNA3 gene revealed that 4 SNPs were significantly associated with the AIP susceptibility. KCNA3 encodes the voltage-gated potassium channel Kv1.3 which regulates membrane potential ana Ca++-signalling in human T cells and plays an essential role in T-cell proliferation and activation [38].
An Italian group [39] genotyped 4 candidate SNPs of the Fc GR region in 183 cases with chronic peri-aortitis and 181 controls. They found an association between the SNP rs1801274 of Fc fragment of IgG receptor 11A restricted to cases of idiopathic retroperitoneal fibrosis with normal IgG4 levels.
1.6. Environmental factors and infections
Regarding IgG4-RSC and AIP a questionnaire-survey reported in two independent cohorts that 88% of patients had a career in “blue-collar” occupations with long-term exposure (for at least one year, but often for a whole career) to solvents, industrial gases, and other environmental agents, indicating a possible role of environmental factors in the development of IgG4-SC and type AIP [40]. The same questionnaire was also administered among a cohort of 44 patients in Oxford, and it was found that 61% of the patients had blue-collar profession with an exposure to potentially harmful products [41].
Gram-positive bacteria and Mycobacterium tuberculosis have been reported in IgG4-RD. In particular, two cases of IgG4 plasma cell infiltrates in association with infectious aortitis due to Gram-positive bacteria including Staphylococcus aureus have been described [42]. It has been suggested that Staphylococcus aureus may stimulate a Th2-mediated elevation in IgG4 [42]. Mycobacterium tuberculosis has been observed to be associated to a case of IgG4-related chronic sclerosing sialadenitis and dacryoadenitis [42]. A novel antibody, peptide AIP1-7 was found in 18 out of 20 patients with AIP [43]. This peptide shows homology with an amino-acid sequence of plasminogen-binding protein (PBP) of Helicobacter pylori, thus it was hypothesized that Helicobacter pylori may trigger disease in a genetically predisposed host via molecular mimicry [44]. However, a recent study investigated the exposure to Helicobacter pylori infection, cytokine response and immunological memory to Helicobacter pylori plasminogen binding protein (PBP) in a prospective IgG4-RD cohort in UK of 55 IgG4-RD and 52 controls [45]. In IgG4-RD gastric tissue contained a chronic inflammatory infiltrate with a low IgG4+ plasma cell count (<10/hpf; range 1–4/hpf) and duodenal specimens had an increased IgG4 count (>10/hpf, range 7–54) compared with controls (p < 0.01). Th1 and Th2 cytokine respond and immunological B-cell memory to Helicobacter pylori PBP did not differ between patients and controls. These results do not support a role for Helicobacter pylori as microbial agent in IgG4-RD patients [45].
1.7. Epidemiology
From the epidemiology point of view there are insufficient data on incidence/prevalence of IgG4-RD. The majority data come from Japan, but it is not known whether Asian patients are more susceptible to IgG4-RD than others; hence, the available epidemiologic data should be interpreted carefully and according to organ involvement [46]. In a Nationwide survey conducted in 2009 in the Ishikawa Prefecture in Japan, the incidence of IgG4-RD was 0.28–1.08/100.000 population with 336-1.300 patients newly diagnosed per year from 2003 to 2009; the estimated prevalence was 62 per million subjects [47]. IgG4-RD affects patients of middle to upper age, with an onset at 50–70 years [48].
1.8. Lessons from clinical studies including cohorts with IgG4-RD
Eleven studies have been published in the literature including 1498 patients diagnosed with IgG4-RD. The main clinical characteristics of these cohorts are summarized in Table 3 [[49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59]]. Four studies have been performed in Europe, one in USA, and six in Asia. Two studies utilized the data base from the France Registry and the Spanish Registry respectively [49,50]. Only two are prospective [51,58]. Among the 7 retrospective studies, only one is multicentre [57]. The mean age at inclusion ranged between 50.3 and 67 years, the M:F ratio was in favour of male gender, and mostly had IgG4 concentration >135 mg/dl. The first European multicentre study included 25 patients from the French Registry of the Internal Medicine Society [49]. The inclusion criteria were elevated serum IgG4 (>135 mg/dl), sclerosing and inflammatory involvement of one or more organs, and histologic features of fibrosis and/or lymphocytic and polyclonal plasma cell infiltration.
Table 3.
IgG4-RD cohorts published in the literature.
| Author | Year | Country | N. pts | M:F ratio | Age at onset | Organ involvement | Serum IgG4 >135 mg/dl | |
|---|---|---|---|---|---|---|---|---|
| Ebbo49 | 2012 | France | 25 | 2.6:1 | 58 | Lymph nodes, pancreas | 100% | French registry |
| Fernandez-Codina50 | 2015 | Spain | 55 | 3:1 | 56 | Retroperitoneum, orbital pseudotumor | – | Spanish registry |
| Lin51 | 2015 | China | 118 | 2.3:1 | 53.1 | Pancreas, lymph nodes, salivary glans | 97.5% | Prospective, multicentre |
| Wallace52 | 2015 | USA | 125 | 1.6:1 | 50.3 | Pancreas, lymph nodes, submandibular glands | 51.4% | Retrospective, single centre |
| Quero53 | 2019 | Spain | 15 | 4:1 | 60.7 | Pancreas, lymph nodes, Kidney, salivary glands, lungs, vascular system | – | Retrospective, single centre |
| Campochiaro54 | 2016 | Italy | 41 | 1.9:1 | 62 | Pancreas, retroperitoneum | 73% | Retrospective, single centre |
| Li55 | 2017 | Hong Kong | 104 | 3:1 | 62 | Hepatobiliary system, lungs, retroperitoneum, CNS, eye, skin | 95% | Retrospective, single centre |
| Fong56 | 2018 | Singapore | 42 (35 definite) | 2.9:1 | 66.3 | Pancreas, lymph nodes, bile ducts | 77.5% | Retrospective, single centre |
| Inoue57 | 2015 | Japan | 235 | 4:1 | 67 | Pancreas, salivary glands, kidney | 88% | Retrospective multicentre |
| Wang58 | 2019 | China | 403 | 1.69:1 | 55 | Salivary glands, pancreas, Lung, retroperitoneum, kidney, lymph nodes, liver, gastrointestinal tract, skin, prostate, sinus, orbits, thyroid | – | Prospective |
| Jamada59 | 2017 | Japan | 334 | 1.5:1 | 63.8 | Salivary and lacrimal glands, lymph nodes, retroperitoneum/periaorta, lung, prostate, biliary tree, skin, thyroid | 95% | Retrospective multicentre |
The largest series reported in Europe derived from a retrospective multicentre study including twelve hospitals in Spain [50]. The inclusion criteria fulfilled the recommendation of the Consensus Conference held in 2011 in Boston [15]. Fifty-five patients were included, 38 of whom were males; the most frequent affected organs were: retroperitoneum, orbital pseudo-tumours, pancreas, salivary and lacrimal glands, and maxillary sinuses.
In China, a prospective cohort of 118 patients with patients with IgG4-RD was enrolled between 2011 and 2013 [51]. Including criteria were divided as: “definite disease” in the presence of: i) organ involvement (enlargement, nodules or dysfunction); ii) serum IgG4 concentration >135 mg/dl; iii) histopathological findings of >10 IgG4+ cells per high power field, and IgG4+/IgG+ cell ratio >40%. “Possible disease” was defined in case of presence of i) and ii) but negative of non-available iii). “Probable disease” required i) and iii). Sixty-eight patients only (57.6%) underwent organ biopsy, 43.2% were diagnosed with “possible disease”, 53.2% had “definite disease”, and 2.5% had “probable disease”.
In USA medical records from 125 patients with biopsy-proven IgG4-RD were reviewed [52]. This is the largest study of IgG4-RD affecting a broad array of organs reported. The authors showed that IgG4-RD patients had elevated levels of circulating plasmablasts that were independent of serum IgG4 concentrations. Moreover, the levels of circulating plasmablasts correlated with the extent of disease activity assessed by the IgG4-RD reactivity index.
More recently, a retrospective study in Spain included 15 patients enrolled from 2008 to 2015 in a single centre in Barcelona [53]. All patients underwent organ biopsy and disease inclusion criteria included both serum IgG4 >135 mg/dl and histopathologic criteria established by Umehara et al. [6].
In Italy a large cohort of 41 patients was included [54]; 26 with “definite” and 15 with “possible” IgG4-RD. The mean age at diagnosis was 62 years. Pancreas, retroperitoneum and major salivary glands were the most frequently involved organs.
A total of 55 patients with IgG4-RD identified between January 2003 and December 2015 were studied in Hong Kong [55]. Forty-two (76.4%) were males and the mean age was 62 ± 12 years. Over 95% of patients had IgG4 level >135 mg/dl and a IgG4: IgG ratio of >8%. Serum IgG4 levels correlated with salivary gland involvement, but not with individual organ system.
Another retrospective study was performed in Singapore in 42 patients with IgG4-RD [56]; 76% were males and the median age was 59 years (range 18–79). Patients were identified from prospective database in four Departments (Gastroenterology, Rheumatology, Radiology and Pathology) in a tertiary referral centre. Seventy-nine percent of patients fulfilled the 2011 comprehensive diagnostic criteria for “definite” IgG4-RD, and 12.5% for “probable” IgG4-RD [5]; 83% of cases had ≥1 organ involvement.
A large cohort included 235 consecutive patients with histology-proven IgG4-RD diagnosed in 8 general hospitals in Japan [57]; 143 patients (60%) presented the clinical features of autoimmune pancreatitis. Of note, all patients were adults, 91% of whom had 50–80 years of age with a M/F ratio of 4:1. Interestingly, male and female patients differed in their organ manifestations. Males had a preponderance of all organs except for sialadenitis and dacryoadenitis which were much more frequent in females than in males.
The largest prospective cohort published so far have been enrolled ai Peking Union Medical College in China [58]. Interestingly, the authors analysed the clinical characteristics of patients with IgG4-RD in female and males. Male predominance was more significant in older patients, with a M:F ratio ranging between 1.12 < 44 years, 1.53 between 45 and 59 years, and 3.15 > 60 years of age. Females more than males had a history of allergy, and at disease onset more often presented with swelling of the lacrimal and submandibular glands. IgG4-related pancreatitis, IgG4-sclerosing cholangitis and retroperitoneal fibrosis were more common in male patients. Finally, males had a greater chance to respond to steroids than females and a greater risk of relapse during follow-up (hazard ratio 3.14, P = 0,003).
Finally, a more recent study has been conducted in Japan with rheumatologists, gastroenterologists, pulmonologists, and nephrologists [59]. In this study 334 patients with IgG4-RD enrolled from 5 University Hospitals in Japan have been retrospectively evaluated. The diagnosis was made according to the conventional criteria [7] in 304 patients, whereas in those diagnosed after 2005 (30 patients) a re-evaluation of histological samples was performed. The numbers of patients with definite, probable, and possible IgG4-RD according to the comprehensive diagnostic criteria were 280 (83.8%), 49 (14.7%) and 4 (1.2%) respectively. All patients were Japanese and it is noteworthy that the overall prevalence of diabetes was 34.4% and was significantly higher in those with AIP than without AIP (46.2% vs 30%, P = 0.005). The most impressive finding is that 67 malignancies appeared in 57 of the 334 patients (17.1%), lung cancer being the most frequent tumour followed by gastric cancer, colon cancer, and prostate cancer. Low serum levels of C3 were observed in 34.7% of patients in whom the affected organs were kidney, lung and pancreas.
The disease is also increasingly recognized to be diagnosed in children, and an exhaustive review has been recently published on paediatric cases [60]. In general, disease characteristics appear to be similar to those observed in the IgG4-RD of the adults. The median age of children was 13 years, and there was a predominance of girls (64% in the 22 reported cases. The most important conditions were IgG4-related orbital disease and IgG4-related pancreatitis [60]. A more recent review of the literature [61] reported that eye manifestations, expressed mainly as orbital mass and, to less extent, as dacryoadenitis are thought to be common in the paediatric population.
In general, these studies document that more cases have been described worldwide. Although an increasing incidence of IgG4-RD is recognized, there is a need to concentrate the efforts on multicentric studies. Several bias offer true limitations of these studies: i) the lack of a proper epidemiological design; ii) most studies have been performed in a single centre, with a consequence to describe a single-organ involvement; iii) Being rare disease in Europe a Registry for rare disease is ongoing but diagnosis of IgG4-RD is actually performed only in tertiary centres. Apparently, there are more cases diagnosed in Asia than in Europe or North America and this geographical difference has not been clarified.
Lessons from studies including IgG4-RD with involvement of a single organ or a group of organs.
1.9. IgG4-related kidney (IgG4-RKD) and retroperitoneum disease
Table 4 [[62], [63], [64], [65]] summarizes the most important published studies. Due to the increased incidence of IgG4-RKD the Japanese Society of Nephrology has established a working group to prepare diagnostic criteria for IgG4-RKD. Briefly, the diagnostic algorithm is based on: i) the presence of kidney damage manifested by abnormal urinalysis markers and/or decreased renal function; ii) serum IgG4 above 135 mg/dl; iii) renal imaging showing diffuse kidney enlargement, hypo-vascular solitary mass in the kidney, and hypertrophic lesion of the renal pelvic wall in the absence of irregularity in the renal pelvic surface; iv) renal histology showing the characteristic changes of IgG4-RD (i.e. storiform fibrosis and lymphoplasmacytic infiltrate). In contrast to the common IgG4-RD, obliterative phlebitis is not a peculiar histological sign of IgG4-RKD. Patients with IgG4-RKD present an extra-organ involvement in more than 75% of cases [62,63]. The authors recommend a careful differential diagnosis to rule out malignant disease, lupus erythematosus, Wegner’s granulomatosis, sarcoidosis, and metastatic carcinoma [63,64]. Finally, patients have an optimal response to steroid treatment. The mean starting dose suggested by Evans et al. is 40 mg/day with tapering dosage and a maintenance dosage [65]. It is not clear whether a steroid-sparing agent might be useful. However, IgG4-RKD is often a relapsing disease and may lead to progressive and permanent damage in 11% of cases [65]. Combined therapy with cyclophosphamide and rituximab with good efficacy has also been reported [66].
Table 4.
IgG4-related kidney disease and retroperitoneum cohorts.
| Author | Country | N. pts | % of males | Age at onset | Organ involvement | Serum IgG4 >135 mg/dl |
|---|---|---|---|---|---|---|
| Raissian62 | USA | 35 (kidney) | 86 | 65 | Lung, pancreas, bile ducts, liver thyroid | – |
| Kawano63 | Japan | 41 (kidney) | 73.2 | 63.7 | Retroperitoneum, pancreas, salivary glands, lymph nodes, prostate, periaortic area | 100% |
| Khosroshahi64 | USA | 13 (retroperitoneum) | 79 | 57 | Orbits, lymph nodes, salivary glands, kidney | – |
| Evans65 | UK | 28 | 92.9 | 58.2 | Pancreas, lymph nodes, lungs, liver, biliary tree | – |
The new discovering cases of IgG4-retroperitoneal fibrosis (IgG4-RPF) have contributed to changing the scenario of this rare condition formerly known as Ormond’s disease. In fact, before the description of IgG4-RPF, peritoneal fibrosis was divided into “idiopathic” and secondary type. The diagnosis of idiopathic cases fulfilled the criteria of exclusion of secondary cases, namely drug exposure, infection and malignancy [67]. Histopathology and immunostaining of tissue sampling obtained from needle or laparoscopic biopsies are the gold-standard for the diagnosis of IgG4-RPF. It should be noted that biopsies from IgG4-RPF patients are likely to be less cellular and more fibrotic than biopsies from other organs, and this can delay the diagnosis. The characteristics of IgG4-RPF include lymphoplasmacytic infiltrate, storiform fibrosis, and eosinophilia which strongly correlates with IgG4/IgG ratio [68].
1.10. IgG4-related pancreatitis
Limited data exist on incidence and prevalence of IgG4-related pancreatitis. In the newest nationwide survey of AIP in Japan, the overall prevalence rate was estimated on 10.1 per 100.000 inhabitants, with an annual incidence rate of 3.1 per 100.000 inhabitants [69]. Compared to the 2011 survey, both numbers are more than doubled [70]. IgG4-related pancreatitis, termed also as type1 AIP is the typical and the most studied manifestation of IgG4-RD on the basis of pancreatic specimens [[71], [72], [73], [74]]. The Honolulu Conference in 2009 established two forms of AIP on the basis of pancreatic specimens after surgery [75]. The presence of granulocyte epithelial lesions (GEL+) or IgG4+ plasma cells at immunohistochemistry are the main findings that allow the diagnosis of type 1 (IgG4+/GEL−) or type 2 (IgG4-/GEL+). However, since these findings are difficult to evaluate on the biopsies, the diagnosis of subtypes of IgG4-related pancreatitis is difficult in the clinical practice [76].
Patients with type I AIP usually present with abdominal pain, weight loss, anorexia and jaundice; usually have raised levels of IgG4 [77]. Patients can also present symptoms of pancreatic exocrine and endocrine insufficiency that include anorexia, weight loss and new-onset diabetes. A retrospective study performed in Toronto enrolled 57 patients with IgG4-related pancreatitis from 2000 to 2009, 40 of whom were males; the mean age was 59 years (range 18–85) [78]. More recently, a retrospective study carried out in Germany collected the clinical characteristics of 40 patients with type 1 and 32 patients with type 2 [79]. Patients with type 1 pancreatitis had a diffuse pancreatic swelling in 26/40 cases, compared to 1/30 of type 2 (P = 0.001), whereas type 2 patients presented with tumour-like pancreatic mass (suspicious for malignancy in 30/32 cases), compared to 16/40 cases of type 1 (P = 0.0001). Moreover, patients with type 1 AIP had a dramatic response to corticosteroids (whereas patients with type 2 underwent surgical resection).
Type 1 AIP is the most common form worldwide, accounting for almost all cases in Japan and Korea, and more than 80% of cases in Europe and US [80,81].
Diagnostic criteria proposed for IgG4-related pancreatitis by various research groups include imaging, histologic and serological findings, extra-pancreatic involvement and response to corticosteroid therapy. According to the Japanese Pancreas Society [82] 3 groups of parameters were considered: 1) diffuse or segmental narrowing of main pancreatic duct with irregular wall and diffuse or localized enlargement of the pancreas by imaging, such as ultrasound, computed tomography (CT) or magnetic resonance; 2) high serum gamma-globulin (IgG or IgG4) or the presence of autoantibodies. Such as anti-nuclear antibodies and rheumatoid factor; 3) marked interlobular fibrosis and prominent infiltration of lymphocytes and plasma cells in the peri-ductal area, occasionally with lymphoid follicles in the pancreas. In 2010 the International Consensus Diagnostic Criteria (ICDC) of the International Society of Pancreatology were proposed with the attempt to unify the AIP criteria. ICDC criteria used the previous proposed criteria from the Mayo Clinic utilizing the HISORt approach [83]. In late 2011 the Japanese Pancreas Society released the revised criteria which were more similar to ICDC with an important goal for each of diagnostic criteria, that is to avoid misdiagnosing pancreatic cancer.
Steroid treatment is recommended [84]. A large multicentre International study including 23 institutions from 10 different countries analysed the data from 1064 patients of whom 978 fulfilled the criteria for type 1 AIP and 86 for type 2 [81]. Steroid remission was obtained in 99% of patients with type 1 and 92% of patients with type 2. Most patients with jaundice required biliary stent placement (71% with type 1 and 77% with type 2). Relapses were recorded in 31% and 9% respectively, but retreatment was effective for induce remission (with steroids or azathioprine). Pancreatic cancer was diagnosed in 5 cases with type 1 AIP more than three years after the initial diagnosis [81].
1.11. IgG4-related sclerosing cholangitis (IgG4-SC)
IgG4-SC is a distinct form of chronic cholangitis characterized by infiltration of lymphocytes and abundant IgG4-positive plasma cells in the bile duct wall, elevated IgG4 serum levels in the majority, and strong response to corticosteroid therapy [85]. From the epidemiological point of view, a recent epidemiological survey conducted in 1180 departments from health centres across Japan on the basis of a questionnaire, estimated a 2.18 prevalence of IgG4-SC per 100,000 population [86]. This condition can be divided into two types: the major type is IgG4-SC associated with AIP, whereas the minor type is isolated IgG4-SC [85]. Patients with IgG4-SC and with associated AIP are generally older (mean age 62 years), men (85%) presenting with obstructive jaundice [86]. Indeed, IgG4-SC produces stenosis in the lower bile duct, and thus should be differentiated from pancreatic cancer and cholangiocarcinoma. Type 2 IgG4-SC in which the stenoses are diffusely distributed throughout the biliary tree the differential diagnosis should include primary sclerosing cholangitis (PSC) [87,88]. The clinic-pathologic features, imaging spectrum, and systemic approach to differential diagnosis of sclerosing cholangitis have been extensively reviewed [[87], [88], [89]]. The most important characteristics in the differential diagnosis of IgG4-SC and PSC, or secondary sclerosing cholangitis evaluated with radiological imaging are listed in Table 5 [[90], [91], [92]]. According to the Nakazawa classification [93] four types of cholangiographic findings for IgG4-RSC can be distinguished: 1) type 1 in which stenosis is located only in the lower part of the common bile duct; 2) type 2 in which stenosis is diffusely distributed throughout the intra- and extra-hepatic bile ducts; 3) type 3 characterized by the involvement of distal bile ducts and hepatic hilum; 4) type 4 characterized by involvement of ducts at hepatic hilum only. Magnetic resonance (MR) imaging shows long-segmental, symmetrical, circumferential wall thickening and delayed contrast enhancement. Long and continuous strictures of the bile ducts with the pre-stenotic dilation are typical finding of the IgG4-RSC. Furthermore, extra-biliary findings associated with IgG4-RD involvement strongly suggest IgG4-RSC. Typical cholangiographic features of primary sclerosing cholangitis include ductal irregularities of the intra- and extra-hepatic biliary tree, stenosis and saccular dilations which often produce “beaded” appearance. When fibrosis progresses “pruned tree” appearance becomes evident. Differential diagnosis includes also recurrent pyogenic cholangitis in which MR cholangiographic findings show dilation of the central bile ducts and rapid tapering in the peripheral bile ducts, showing multiple filling defects. This aspect, known as “arrowhead” appearance, suggests the presence of radiolucent stones; on the other hand, filling defects can also be represented by Clonorchis sinensis worms and should be differentiated by pneumobilia. Cholangiocarcinoma must be ruled out in case of dominant strictures with brush sampling obtained by endoscopic retrograde cholangiopancreatography (ERCP).
Table 5.
Magnetic resonance imaging in type I IgG4-sclerosing cholangitis (Type I IgG4-SC), isolated IgG4-sclerosing cholangitis, primary sclerosing cholangitis (PSC) and recurrent pyogenic cholangitis (isolated IgG4-SC).
| Type IgG4-SCa | Isolated IgG4-SC | PSC [93] | Recurrent pyogenic cholangitis |
|---|---|---|---|
| Type1: involvement of lower part of bile duct only Type 2a: involvement of distal bile duct and intrahepatic bile duct with proximal dilation Type 2b: involvement of distal bile duct and diffuse intrahepatic bile duct without proximal dilation Type 3: involvement of distal bile duct and ducts at hepatic hilum Type 4: involvement of ducts at hepatic hilum only |
Absence of intra-hepatic bile ducts, bile duct lesions. Cholangiographic findings based on Nakazawa classificationa | Ductal irregularities of the intra- and extra hepatic biliary tree. Stenosis and saccular dilations (beaded appearance). Pruned-tree appearance at chronic stage |
Dilated intrahepatic bile ducts, bile duct strictures, calculi within dilated bile ducts and morphologic changes like atrophy |
Nakazawa classification [92].
The most important clinical and demographic characteristics of IgG4-RD derive from a large-scale survey conducted in Japan [94]. The authors analysed a retrospective cohort of 527 patients with IgG4-RD enrolled from 2000 to 2015; 83% were males, and the median age at presentation was 66.2 years, range 23–89 years. Symptoms at presentation included jaundice (35% of cases) and itching (13%), whereas 28% of cases were asymptomatic. Serum IgG4 were increased (>135 mg/dl) in 85.4% of patients. Prednisolone was administered in 458 patients (88%) and was continued in 62%. A maintenance dose ≤5 mg was given to 220 patients. The treatment responses were excellent. It is noteworthy that no patients received liver transplant, and cholangiocarcinoma was found in only 4 patients.
1.12. IgG4-autoimmune hepatitis (AIH)
A subgroup of AIH called IgG4-AIH has been recently identified in patients who met the criteria for AIH. A review on this issue has been recently also published [95]. Indeed, the concept of IgG4-AIH was first published by Umemura et al., in 2007 (96); the authors reported a clinical case in whom the clinical and histological criteria for definite AIH were met and additionally two liver biopsies (the first during cholecystectomy and the second 6 months thereafter) showed hepatitis with abundant IgG4-bearing plasma cells in the liver and gallbladder. Table 6 summarizes the five studies reported in the literature [[96], [97], [98], [99], [100]]: four studies report adult cases and one [101] paediatric cases. The hallmark of this condition is the presence of abundant infiltration of IgG4-expressing plasma cells in the portal tracts together with serum concentration of IgG4 >135 mg/dl. According to the diagnostic criteria proposed by Nakanuma [102], IgG4-AIH can be defined in the presence of elevated concentration of serum IgG4 and infiltration of IgG4-expressing plasma cells in the liver ≥10/HPF. Applying the Nakanuma criteria, only 3 studies met the diagnosis of IgG4-AIH. However, several points should be clarified: i) whether IgG4-AIH is a hepatic involvement of IgG4-RD; ii) whether IgG4-AIH is a subtype of classical AIH. Moreover, in the Umemura’s study [98] the infiltration of IgG4-positive cells in the gallbladder and common bile duct was observed in one of the IgG4-AIH cases, indicating the presence of IgG4-RSC rather than IgG4-AIH.
Table 6.
IgG4-related AIH.
| Author |
Umemura97 |
Chung98 |
Amarapurkar99 |
Canivetet100 |
Aydamir101 |
|---|---|---|---|---|---|
| Country | Japan | Japan | India | France | Turkey |
| N. of pts with AIH | 60 | 26 | 40 | 28 | 40 (children) |
| IgG4-AIH | 2 (3.3%) | 9 (34.6%) | 10 (25%) | 7 (25%9 | 6 (15% |
| Immunostaining for IgG4+ cells/HPF | >10 | >5 | >5 | >10 | >10 |
1.13. IgGg4-related periaortitis/periarteritis
A complete review on this topic has been recently published [103]. The involvement of IgG4 in this district is very impressive. Briefly, periaortitis/periarteritis may be present in 10–30% of overall IgG4-RD and they may also appear as an isolated lesion of IgG4-RD. The majority of studies have been conducted in Japan and China. However, an important study from US has been reported in 2016 [104]. The authors defined two types of vasculitis, primary IgG4-related disease and secondary IgG4-related vascular involvement. The former was characterized by the presence of one or more of the followings: vessel wall thickening, vessel wall enhancement on contrast study with 18F-Fluoro-deoxyglucose (FDG) uptake and FDG-PET technology. Primary IgG4-vasculitis is characterized by a periaortic and concentric involvement. The latter is characterized by abnormal perivascular soft tissue with minimal vascular wall findings. Secondary IgG4-related vascular involvement is generally associated with retroperitoneal fibrosis. Since serum IgG4 are not always elevated in cases with IgG4-related aortitis or retroperitoneal fibrosis, the Japanese Circulation Society has established specific diagnostic criteria for IgG4-related periaortitis/periarteritis and retroperitoneal fibrosis, taking into account diagnostic factors (abnormal radiological findings, elevated serum IgG4, histological findings, and other organ involvement [105].
1.14. IgG4-RD of the head and neck
Submandibular, parotid, sublingual glands, and lacrimal glands are often involved, usually bilaterally. IgG4-related sclerosing sialadenitis of the submandibular gland is also known as Kuttner’s disease. According to the Sapporo Medical University and Related Institutes Database for Investigation and Best Treatment for IgG4-RD (SMART) database the sex ratio in IgG4-related dacryoadenitis and/or sialadenitis was roughly equal [106]. More recently this condition has been associated to IgG4-RD [107,108]. Patients complain of pain and swelling frequently associated with eating. At physical examination sialadenitis may present as a painless firm mass which mimics a tumour. Indeed, the differential diagnosis includes malignancy and Sjogren’s syndrome. Ultrasound reveals hypoechoic zones with duct dilations. CT imaging shows homogeneous attenuation and enhancement. At MR imaging the lesions demonstrate homogeneous low to intermediate signal intensity on T1-weighted images with homogeneous enhancement [109]. Mikulicz’s disease has been recently included in the IgG4-RD [110]. It is characterized by a bilateral painless and asymmetrical swelling of the lacrimal, parotid, and submandibular glands. The differential diagnosis includes Sjogren’s syndrome, but Mikulicz’s disease in contrast with Sjogren’s syndrome lacks anti-SSA and anti–SS–B antibodies. Thus, the diagnostic criteria for Mikulicz’s disease include: i) symmetrical and persistent swelling in more than two lacrimal glands and major salivary glands; ii) IgG4 serum concentration >135 mg/dl; iii) IgG4+ plasma cell infiltration.
IgG4-RD disease of the thyroid gland is a similarly newly recognized entity with four postulated subcategories: i) IgG4-related Hashimoto’s (HT); ii) fibrosing variant of HT; iii) Riedel’s thyroiditis (RT); iv) Graves disease with elevated IgG4 levels [111]. Histology is essential for the diagnosis with the typical changes: lymphoplasmacytic infiltrate rich in plasma cells, storiform fibrosis, obliterative phlebitis. Phlebitis without obliteration of the lumen and increased number of eosinophils are considered unspecific.
Studies including IgG4-RD with involvement of a single organ or a group of organs deserve some point of discussion. Firstly, several conditions have been recently revisited bearing in mind the IgG4 context (i.e. Mikulicz’s disease, Ormond’s disease, Riedel’s disease, Kuttner’s disease). Secondly, many conditions with clinical aspects resembling malignancy in whom tissue specimens are characterized by fibrosis should complete their histopathological diagnosis with immunohistochemistry for IgG4. Finally, the diagnosis of IgG4-RD requires comprehensive criteria (clinical, serological, histological) according to the International guidelines. These conditions should be managed in a multi-specialistic way, and Scientific Societies should deserve part of their programmes to discuss this emerging group of diseases.
1.15. Treatment
According to the International Consensus Guidance published in 2015 [112], all patients with symptomatic, active IgG4-RD require treatment, some urgently. Glucocorticoids at a dosage of 30–40 mg per day are the first-line agents; there is no significative difference between prednisolone at 30 mg and prednisolone at 40 mg. Most experts agree that initial dosage should be maintained for 2–4 weeks and tapered gradually thereafter. The dosage is different in various centres. The goal of induction therapy is to discontinue steroids 3–6 months after starting treatment; many Japanese clinicians recommend the low-dose maintenance therapy for up to 3 years [113]. In type1 AIP (IgG4-related pancreatitis), a Japanese RCT study for usefulness of low-dose maintenance Tx was recently published [114]. Based on expert opinion, international guidelines for Tx of AIP were proposed thereafter [115]. At least one third of patients experience relapse after discontinuation of steroids, and in this case, retreatment with steroids is indicated. Following relapse, the introduction of steroid-sparing agent should be considered.
Rituximab (RTX), an anti-CD20 monoclonal antibody represent a promising strategy. In a pilot study, 14 patients with IgG4-RD (7 retreated only in case of disease relapse [group 1], and 7 regularly retreated every 6 months [group 2]) were administered with RTX and retrospectively analysed [116]. The free relapse rate 18 months after induction of remission treatment was significantly lower in group 1 (29%) than in group 2 (100%, p = 0.006). However, it should be taken into account the development of infectious complications (6/14 patients). However, treatment is influenced by geographical, epidemiological, and clinical factors especially with respect to organ involvement. The results of a systematic search strategy in 62 studies that included a total of 3.034 patients has been recently published [117]. The following regimens have been considered: first-line regimens of whom glucocorticoids were given in 1.437 patients (74%), drug-free regimens in 11% and other therapies in 2%. No therapy (wait and see management) was reported in 264 cases (13%). Therapeutic efficacy on steroids was reported in 97% of cases (but with 95% of relapses). Relapses occurred also in 56/69 (81%) patients treated with azathioprine, 16/22 (72%) in those treated with immunosuppressive agents, and in 9 cases (100%) treated with rituximab. Among patients without therapy improvement occurred in 43% of cases.
2. Conclusion
The available epidemiological data on IgG-4-RD derived from cohort of patients with different organ involvement. It is still unknown whether Asian patients are more susceptible to IgG4-RD than others. IgG4-RD affects individuals of middle to upper age with onset at 50–70 years. Only rarely it can be seen in children. Most studies report an overall predilection for male sex. However, IgG4-related sialadenitis and dacryoadenitis may occur more frequently among females. However, its epidemiology is still widely unknown. The spectrum of clinical manifestations in IgG4-RD is highly variable, depending on the severity of the disease as well as the presence of organ(s) involvement. Correctly differentiating IgG4-RD from disease mimics is crucial and a multi-specialist team is often necessary.
Pathogenic mechanisms of IgG4-RD: a) Eventual initial mechanisms include local autoantigens, infectious agents, allergic components in a genetically predisposed individual; b) These factors activate immune reaction in the target tissue (organ/organs) which is characterized by Th2 predominance with an increased production of cytokines (IL-4, IL-5, IL-13) and tumour growth factor -β (TGF-β). Moreover, an activation of Treg, a release of IL-10 and elevated serum IgE occur. The immune reaction also causes a B cell class switching with an increased production of IgG4 producing plasma cells. APC = antigen presenting cell; Tc = cytotoxic T lymphocyte; Treg = regulatory T cell.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Kamisawa T., Zen Y., Pillai S., Stone J.H. IgG4-related disease. Lancet. 2015;385:1460–1471. doi: 10.1016/S0140-6736(14)60720-0. [DOI] [PubMed] [Google Scholar]
- 2.Della-Torre E., Lanzillotta M., Doglioni C. Immunology of IgG4-related disease. Clin. Exp. Immunol. 2015;181:191–206. doi: 10.1111/cei.12641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kamisawa T., Funata N., Hayashi Y., Eishi Y., Koike M., Tsuruta K. A new clinicopathological entity of IgG4-related autoimmune disease. J. Gastroenterol. 2003;38:982–984. doi: 10.1007/s00535-003-1175-y. [DOI] [PubMed] [Google Scholar]
- 4.Umehara H., Okazaki K., Masaki Y. Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD) Mod. Rheumathol. 2011;22(2012):21–30. doi: 10.1007/s10165-011-0571-z. [DOI] [PubMed] [Google Scholar]
- 5.Fox R.I., Fox C.M. IgG4 levels and plasmablasts as a marker for IgG4-related disease (IgG4-RD) Ann. Rheum. Dis. 2015;74:1–3. doi: 10.1136/annrheumdis-2014-205476. [DOI] [PubMed] [Google Scholar]
- 6.Umehara H., Okazaki K., Nakamura T., Satoh-Nakamura T., Nakajima A., Kawano M. Current approach to the diagnosis of IgG4-related disease – combination of comprehensive diagnostic and organ-specific criteria. Mod. Rheumatol. 2017;27:381–391. doi: 10.1080/14397595.2017.1290911. [DOI] [PubMed] [Google Scholar]
- 7.Wallace Z.S., Naden R.P., Chari S., Choi H., Della-Torre E., Dicaire J.-F. The 2019 American College of Rheumatology/European league against rheumatism classification criteria for IgG4-related disease. Arthritis & Rheumatology. 2020;72:7–19. doi: 10.1002/art.41120. [DOI] [PubMed] [Google Scholar]
- 8.Aalberse R.C., Schurman J. IgG4 breaking the rules. Immunology. 2002;105:9–19. doi: 10.1046/j.0019-2805.2001.01341.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Umehara I., Nakajima A., Nakamura T., Kawanami T., Tanaka M., Dong L., Kawano M. IgG4-related disease and its pathogenesis – cross-talk between innate and acquired immunity. Int. Immunol. 2014;26:585–595. doi: 10.1093/intimm/dxu074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zen Y., Fuji T., Harada K. Th2 and regulatory immune reactions are increased in immunoglobulin G-4 related sclerosing pancreatitis and cholangitis. Hepatology. 2007;45:1538–1546. doi: 10.1002/hep.21697. [DOI] [PubMed] [Google Scholar]
- 11.Kusuda T., Uchida K., Miyoshi H., Koyabu M., Sato S., Takaoka M. Involvement of inducible costimulatory- and interleukin 10-positive regulatory T cells in the development of IgG4-related autoimmune pancreatitis. Pancreas. 2011;40:1120–1130. doi: 10.1097/MPA.0b013e31821fc796. [DOI] [PubMed] [Google Scholar]
- 12.Tsuboi H., Matsuo N., Iizuka M. Analysis of IgG4 class switch-related molecules in IgG4-related disease. Arthritis Res. Ther. 2012;14:R171–R177. doi: 10.1186/ar3924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tacheuchi M., Sato Y., Ohno K. T helper 2 and regulatory T-cell cytokine production by mast cells: a key factor in the pathogenesis of IgG4-related disease. Mod. Pathol. 2014;27:1126–1136. doi: 10.1038/modpathol.2013.236. [DOI] [PubMed] [Google Scholar]
- 14.Watanabe T., Yamashita K., Sakurai T., Kudo M., Shiokawa M., Uza N. Toll-like receptor activation in basophils contributes to the development of IgG4-related disease. J. Gastroenterol. 2013;48:247–253. doi: 10.1007/s00535-012-0626-8. [DOI] [PubMed] [Google Scholar]
- 15.Watanabe T T., Yamashita K., Fujikawa S., Sakurai T., Kudo M., Shiokawa M. Involvement of activation of toll-like receptors and nucleotide-binding oligomerization domain-like receptors in enhanced IgG4 responses in autoimmune pancreatitis. Arthritis Rheum. 2012;64:914–924. doi: 10.1002/art.33386. [DOI] [PubMed] [Google Scholar]
- 16.Chiari S.T., Takahashi N., Levi M.J. A diagnostic strategy to distinguish autoimmune pancreatitis from pancreatic cancer. Clin. Gastroenterol. Hepatol. 2009;7:1097–1103. doi: 10.1016/j.cgh.2009.04.020. [DOI] [PubMed] [Google Scholar]
- 17.Weindorf S., Frederiksen J.K. IgG4-related disease. A reminder for practicing pathologists. Arch. Pathol. Lab Med. 2017;141:1476–1483. doi: 10.5858/arpa.2017-0257-RA. [DOI] [PubMed] [Google Scholar]
- 18.Deshpande V., Zen Y., Chan J.K.C., Yi E.E., Sato Y., Yoshino T. Consensus statement on the pathology of IgG4-related disease. Mod. Pathol. 2012;25:1181–1192. doi: 10.1038/modpathol.2012.72. [DOI] [PubMed] [Google Scholar]
- 19.Xu W.L., Ling Y.C., Wang Z.K., Deng F. Diagnostic performance of serum IgG4 level for IgG4-related disease: a meta-analysis. Sci. Rep. 2016;6:32035. doi: 10.1038/srep32035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Aalberse R.C., Stapel S.O., Schurman J., Rispens T. Immunoglobulin G4: an odd antibody. Clin. Exp. Allergy. 2009;39:469–477. doi: 10.1111/j.1365-2222.2009.03207.x. [DOI] [PubMed] [Google Scholar]
- 21.Rock B., Martins C.R., Theofilopulos A.N. The pathogenic effect of IgG4 autoantibodies in endemic pemphigus foliaceous (Fogo selvagens) N. Engl. J. Med. 1989;320:1463–1469. doi: 10.1056/NEJM198906013202206. [DOI] [PubMed] [Google Scholar]
- 22.Aoki S., Nakazawa T., Ohara H. Immunohistochemical study of autoimmune pancreatitis using anti-IgG4 antibody and patients’ sera. Histopathology. 2005;47:147–158. doi: 10.1111/j.1365-2559.2005.02204.x. [DOI] [PubMed] [Google Scholar]
- 23.Fischer S., Trivedi P.J., Ward S., Greig P.D., Therapondos G., Hirschfield G.M. Frequency and significance of IgG4 immunohistochemical staining in liver explants from patients with primary sclerosing cholangitis. Int. J. Exp. Pathol. 2014;95:209–2015. doi: 10.1111/iep.12076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.De Buy Wenninger L.J.M., Doorenspleet M.E., Klarenbeek P.L. IgG4+ clones identified by next-generation sequencing dominate the b-cell receptor repertoire in IgG4-associated cholangitis. Hepatology. 2013;57:2390–2398. doi: 10.1002/hep.26232. [DOI] [PubMed] [Google Scholar]
- 25.Culver E.L., Vermeulen E., Makuch M. Increased IgG4 responses to multiple food on animal antigens indicate a polyclonal expansion and differentiation of pre-existing B cells in IgG4-related disease. Ann. Rheum. Dis. 2015;74:994–997. doi: 10.1136/annrheumdis-2014-206405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kawa S., Ota M., Yoshizawa K., Horiuchi A., Hamano H., Ochi Y. HLA BRB10405-DQB10401 haplotype is assiciated with autoimmune pancreatitis in the Japanese population. Gastroenterology. 2002;122:1264–1269. doi: 10.1053/gast.2002.33022. [DOI] [PubMed] [Google Scholar]
- 27.Ota M., Katsuyama Y., Hamano H., Unemura T., Kimura A., Yoshizawa K. Two critical genes (HLA-DRB1 and ABCF1) in the HLA region are associated with the susceptibility to autoimmune pancreatitis. Immunogenetics. 2007;59:45–52. doi: 10.1007/s00251-006-0178-2. [DOI] [PubMed] [Google Scholar]
- 28.Terao C., Ota M., Iwasaki T., Shiokawa M., Kawagushi S., Kuriyama K. IgG4-related disease in the Japanese population: a genome-wide association study. Lancet Rheumatol. 2019;1:e14–e22. doi: 10.1016/S2665-9913(19)30006-2. [DOI] [PubMed] [Google Scholar]
- 29.X. Li, T.D. Howard, S.L. Zheng, T. Haselkorn, S.P. Peters, D.A. Meyers, et al. Genome-wide association study of asthma identifies RAD50-IL13 and HLA-DR/DQ regions, J. Allergy Clin. Immunol. 125 (210) 328-335. [DOI] [PMC free article] [PubMed]
- 30.Newman J.H., Shaver A., Sheehan J.H., Mallal S., Stone J.H., Pillai S. IgG4-related disease: association with a rare gene variant expressed in cytotoxic T cells, Mol. Genet. Genomic Med. 2019;7:e686–e693. doi: 10.1002/mgg3.686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fujbayashi S., Sasajima J., Goto T., Tanaka H., Kawabata H., Fujii T. A high throughput sequence analysis of Japanese patients revealed 11 candidate genes associated with type 1 autoimmune pancreatitis susceptibility. Biochem. Biophys. Rep. 2016;6:76–81. doi: 10.1016/j.bbrep.2016.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Park D.K., Kim M.-H., Oh H.B. Substitution of aspartic acid at position 57 of the DQbeta 1 affects relapse of autoimmune pancreatitis. Gastroenterology. 2008;134:440–446. doi: 10.1053/j.gastro.2007.11.023. [DOI] [PubMed] [Google Scholar]
- 33.Chang M.C., Chang Y.T., Tien Y.W., Liang P.C., Jan I.S., Wei S.C. T-cell regulatory gene CTLA-4 polymorphism/haplotype association with autoimmune pancreatitis. Clin. Chem. 2007;53:1700–1705. doi: 10.1373/clinchem.2007.085951. [DOI] [PubMed] [Google Scholar]
- 34.Chang M.C., Jan I.S., Liang P.C., Jeng Y.M., Yang C.Y., Tien Y.W. Hyman cationic trypsinogen but not serine peptidase inhibitor, Kaza 1 type 1 variants increase the risk of type 1 autoimmune pancreatitis. J. Gastroenterol. Hepatol. 2014;29:2038–2042. doi: 10.1111/jgh.12649. [DOI] [PubMed] [Google Scholar]
- 35.Umemura T., Ota M., Yoshizawa K. Genetic association of Fc receptor-like 3 polymorphisms with autoimmune pancreatitis in Japanese patients. Gut. 2006;55:1367–1368. doi: 10.1136/gut.2006.095059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Umemura T., Ota M., Hamano H., Katsuyama Y., Muraki T., Arakura N. Association of autoimmune pancreatitis with cytotoxic T-lymphocyte antigen 4 gene polymorphisms in Japanese patients. Am. J. Gastroenterol. 2008;103:588–594. doi: 10.1111/j.1572-0241.2007.01750.x. [DOI] [PubMed] [Google Scholar]
- 37.Ota M., Ito T., Unemura T., Katsuyama Y., Yoshizawa K., Hamano H., Kawa S. Polymorphisms in the KCNA3 gene is associated with susceptibility to autoimmune pancreatitis in the Japanese population. Dis. Markers. 2011;31:223–229. doi: 10.3233/DMA-2011-0820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fanger C.M., Rauer H., Neben A.I. Calcium-activated potassium channels sustain calcium signaling in T lymphocyte. Selective blockers and manipulated channel expression levels. J. Biol. Chem. 2001;276:12249–12256. doi: 10.1074/jbc.M011342200. [DOI] [PubMed] [Google Scholar]
- 39.Alberici F., Bonatti F., Maritati F., Urban M.L., Moroni G., Emmi G. Association of a polymorphism of the Fcγ-receptor “A (FCGR2A) gene with chronic periaortitis. Clin. Exp. Rheumatol. 2018;37:222–226. [PubMed] [Google Scholar]
- 40.de Buy Wellinger L.J.M., Culver E.L., Beuers U. Exposure to occupational agents might predispose to IgG4-related disease. Hepatology. 2014;60:1453–1454. doi: 10.1002/hep.26999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Siddiquee Z., Zane N.A., Smith R.N., Stone J.R. Dense IgG4 plasma cell infiltrates associated with chronic infections aortitis: implications for the diagnosis of IgG4-related disease. Cardiovasc. Pathol. 2012;21:470–475. doi: 10.1016/j.carpath.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 42.Kawano M., Yamada K., Kakuchi Y., Ito K., Hamano R., Fujii H. A case of immunoglobulin G4-related chronic sclerosing sialadenitis and dacryoadenitis associated with tuberculosis. Mod. Rheumatol. 2009;19:87–90. doi: 10.1007/s10165-008-0127-z. [DOI] [PubMed] [Google Scholar]
- 43.Frulloni L., Lunardi C., Simone R., Dolcino M., Scattolini C., Falconi M. Identification of a novel antibody associated with autoimmune pancreatitis. N. Engl. J. Med. 2009;361:2135–2142. doi: 10.1056/NEJMoa0903068. [DOI] [PubMed] [Google Scholar]
- 44.Kountouras J., Zavos C., Chatzopoulos D. A concept of the role of Helicobacter pylori infection in autoimmune pancreatitis. J. Cell Mol. Med. 2005;9:196–207. doi: 10.1111/j.1582-4934.2005.tb00349.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Culver E.L., Smit W.L., Evans C., Sadler R., Cargill T., Mazuk M. No evidence to support the role for Helicobacter pylori infection and plasminogen binding protein in autoimmune pancreatitis and IgG4-related disease in a UK cohort. Pancreatology. 2017;17:395–402. doi: 10.1016/j.pan.2017.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Brito-Zeron P., Ramos-Casals M., Bosch X., Stone J.H. The clinical spectrum of IgG4-related disease. Autoimmun. Rev. 2014;13:1203–1210. doi: 10.1016/j.autrev.2014.08.013. [DOI] [PubMed] [Google Scholar]
- 47.Uchida K., Masamune A., Shimosegawa T., Okazaki K. Prevalence of IgG4-related disease in Japan based on nationwide survey in 2009. Internet J. Rheumatol. 2012;358371:1–5. doi: 10.1155/2012/358371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lang D., Zwerina J., Pieringer H. IgG4-related disease: current challenges and future prospects. Therapeut. Clin. Risk Manag. 2016;12:189–199. doi: 10.2147/TCRM.S99985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ebbo M., Daniel L., Pavic M. IgG4-related systemic disease; features and treatment response in a French cohort: results of a multicenter registry. Medicine (Baltimore) 2012;91:49–56. doi: 10.1097/MD.0b013e3182433d77. [DOI] [PubMed] [Google Scholar]
- 50.Fernandez-Codina A., Martinez-Valle F., Pinilla B., Lopez C., De Torres I., Solans-Laquè R. IgG4-related disease: results from a multicenter Spanish registry. Medicine. 2015;94:e1275–e1279. doi: 10.1097/MD.0000000000001275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lin W., Lu S., Chen H., Wu Q., Fei Y., Li M. Clinical characteristics of immunoglobulin G4-related disease: a prospective study of 118 Chinese patients. Rheumatology. 2015;54:1982–1990. doi: 10.1093/rheumatology/kev203. [DOI] [PubMed] [Google Scholar]
- 52.Wallace Z., Deshpande V., Mattoo H., Mahajan V.S., Kukikova M., Pillai S., Stone J.H. IgG4-related disease. Clinical and laboratory features in one hundred twenty-five patients. Arch. Rheumatol. 2015;67:2466–2475. doi: 10.1002/art.39205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Quero M., Draibe J., Solanich X., Rama I., Gomà M., Martinez-Valenzuela L. Clinical features and outcomes in a cohort of patients with immunoglobulin G4-related disease at an university hospital in Spain. Clin. Kidney J. 2019;12:829–835. doi: 10.1093/ckj/sfz031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Campochiaro C., Ramirez G.A., Bozzolo E.P., Lanzillotta M., Berti A., Baldissera E. IgG4-related disease in Italy: clinical features and outcomes of a large cohort of patients. Scand. J. Rheumatol. 2016;45:135–145. doi: 10.3109/03009742.2015.1055796. [DOI] [PubMed] [Google Scholar]
- 55.Li P.H., Ho C.T.K., Lau L.L., Tsang R.K.J., Cheung T.T., Leung W.K., Lau C.S. Immunoglobulin G4-related disease in Hong Kong: clinical features, treatment practices, and its association with multisystem disease. Hong Kong Med. J. 2017;23:446–453. doi: 10.12809/hkmj176229. [DOI] [PubMed] [Google Scholar]
- 56.Fong W., Liaw I., Tan D., Lim K.H., How A., Leung Y.Y. IgG4-related disease: features and treatment response in a multi-ethnic cohort in Singapore. Clin. Exp. Rheumatol. 2018;36(suppl. 112):S89–S93. [PubMed] [Google Scholar]
- 57.Inoue D., Yoshida K., Yoneda N., Ozaki K., Matsubara T., Nagai K. IgG4-related disease: dataset of 235 consecutive patients. Medicine. 2015;94:e680–2687. doi: 10.1097/MD.0000000000000680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wang L., Zhang P., Zhang X., Wei L., Tang H., Jieqiong L. Sex disparities in clinical characteristics and prognosis in immunoglobulin G4-related disease: a prospective study of 403 patients. Rheumatology. 2019;58:820–830. doi: 10.1093/rheumatology/key397. [DOI] [PubMed] [Google Scholar]
- 59.Yamada K., Yamamoto M., Saeki T., Mizushima I., Matsui S., Fujisawa Y. New clues to the nature of immunoglobulin G4-related disease: a retrospective Japanese multicenter study of baseline clinical features of 334 cases. Arthritis Res. Ther. 2017;19:262–272. doi: 10.1186/s13075-017-1467-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Karim F., Loeffen J., Bramer W., Westenberg L., Verdijk R., van Hagen M., van Laar J. IgG4-related disease: a systematic review of this unrecognized disease in pediatrics. Pediatr. Rheumatol. 2016;14:18–27. doi: 10.1186/s12969-016-0079-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Smerla R.G., Rontogianni D., Fragoulis G.E. Ocular manifestations of IgG4-related disease in children. More common than anticipated? Review of the literature and case report. Clin. Rheumatol. 2018;37:1721–1727. doi: 10.1007/s10067-017-3934-9. [DOI] [PubMed] [Google Scholar]
- 62.Raissian Y., Hars S.H., Larsen C.P., Colvin R.B., Smyrk T.C., Nakanashi N. Diagnosis of IgG4-related tubulointerstitial nephritis. J. Am. Soc. Nephrol. 2011;22:1343–1352. doi: 10.1681/ASN.2011010062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kawano M., Saeki T., Nakashima H., Nishi S., Yamaguchi Y., Hisano S. Proposal for diagnostic criteria for IgG4-related kidney disease. Clin. Exp. Nephrol. 2011;15:615–626. doi: 10.1007/s10157-011-0521-2. [DOI] [PubMed] [Google Scholar]
- 64.Khosroshahi A., Carruthers M.N., Stone J.H., Shinagare S., Sainari N., Hasserjian R.P. Rethinking Ormond’s disease. Medicine. 2013;92:82–91. doi: 10.1097/MD.0b013e318289610f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Evans R.D.R., Cargill T., Goodchild G., Oliveira B., Rodriguez-Justo M., Pepper R. Clinical manifestations and long-term outcomes of IgG4-related kidney and retroperitoneal involvement in a United Kingdom IgG4-related disease cohort. Kidney Int Rep. 2019;4:48–58. doi: 10.1016/j.ekir.2018.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Quattrocchio G., Barreca A., De Marchi A. IgG4-related kidney disease. The effect of rituximab-based immunosuppressive therapy. Oncotarget. 2018;9:21337–21347. doi: 10.18632/oncotarget.25095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Vaglio A., Salvarani C., Buzio C. Retroperitoneal fibrosis. Lancet. 2006;367:241–251. doi: 10.1016/S0140-6736(06)68035-5. [DOI] [PubMed] [Google Scholar]
- 68.Khosroshah A., Carruthers M.N., Stone J.H., Shinagare S., Sainani N., Hasserjian R.P. Rethinking Ormond’s disease: “idiopatic” retroperitoneal fibrosis in the era of IgG4-related disease. Medicine. 2013;92:82–91. doi: 10.1097/MD.0b013e318289610f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Masamune A., Kikuta K., Hamada S., Tsuji I., Takeyama Y., Shimosegawa T. Nationwide epidemiological survey of autoimmune pancreatitis in Japan in 2016. J. Gastroenterol. 2020;55:462–470. doi: 10.1007/s00535-019-01658-7. [DOI] [PubMed] [Google Scholar]
- 70.Kanno A., Musamune A., Okazaki K., Kamisawa T., Kawa S., Nishimori I. Nationwide epidemiological survey of autoimmune pancreatitis in Japan 2011. Pancreas. 2015;44:535–539. doi: 10.1097/MPA.0000000000000325. [DOI] [PubMed] [Google Scholar]
- 71.Hart P.A., Zen Y., Chari S.T. Recent advances in autoimmune pancreatitis. Gastroenterology. 2015;149:39–51. doi: 10.1053/j.gastro.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 72.Miyabe K., Zen Y., Cornell L.D., Rajagopalan G., Chowdhary V.R., Roberts L.R., Chari S.T. Gastrointestinal and extra-intestinal manifestations of IgG4-related disease. Gastroenterology. 2018;155:990–1003. doi: 10.1053/j.gastro.2018.06.082. [DOI] [PubMed] [Google Scholar]
- 73.Uchida K., Okazaki K. Clinical and pathological aspects of type 1 autoimmune pancreatitis. J. Gastroenterol. 2018;53:475–483. doi: 10.1007/s00535-018-1440-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Okazaki K., Uchida K. Current concept of autoimmune pancreatitis and IgG4-related disease. Am. J. Gastroenterol. 2018;113:1412–1416. doi: 10.1038/s41395-018-0184-7. [DOI] [PubMed] [Google Scholar]
- 75.Chari S.T., Kloeppel G., Zhang L., Notohara K., Lerch M.M., Shimosegawa T. Histopathologic and clinical sybtypes of autoimmune pancreatitis. The Honolulu consensus document. Pancreas. 2010;39:549–554. doi: 10.1097/MPA.0b013e3181e4d9e5. [DOI] [PubMed] [Google Scholar]
- 76.de Pretis N., Vicceli F., Brandolese A., Brozzi L., Amodio A., Frulloni L. Autoimmune pancreatitis not otherwise specified (NOS): clinical features and outcomes of the forgotten type. Hepato. Pancreatic Dis. 2019;18:576–579. doi: 10.1016/j.hbpd.2019.05.010. [DOI] [PubMed] [Google Scholar]
- 77.Morselli-Labate A.M., Pezzilli R. Usefulness of serum IgG4 in the diagnosis and follow-up of autoimmune pancreatitis: a systematic literature review and meta-analysis. J. Gastroenterol. Hepatol. 2009;24:15–36. doi: 10.1111/j.1440-1746.2008.05676.x. [DOI] [PubMed] [Google Scholar]
- 78.Vlachou P.A., Khalili K., Jang H.-J., Fischer S., Hirschfield G.M., Kim T.K. IgG4-related sclerosing disease: autoimmune pancreatitis and extrahepatic manifestations. Radiographics. 2011;31:1379–1402. doi: 10.1148/rg.315105735. [DOI] [PubMed] [Google Scholar]
- 79.Fritz S., Bergmann F., Grenacher L., Sgroi M., Hinz U., Tackert T. Diagnosis and treatment of autoimmune pancreatitis type 1 and 2. BJS. 2014;101:1257–1265. doi: 10.1002/bjs.9574. [DOI] [PubMed] [Google Scholar]
- 80.Sah R.P., Chiari S.T. Autoimmune pancreatitis: an update on classification, diagnosis, natural history and management. Curr. Gastroenterol. Rep. 2012;14:95–105. doi: 10.1007/s11894-012-0246-8. [DOI] [PubMed] [Google Scholar]
- 81.Hart P.A., Kamisawa T., Brugge W.R. Long-term outcomes of autoimmune pancreatitis: a multicentre, international analysis. Gut. 2013;11:1771–1776. doi: 10.1136/gutjnl-2012-303617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Okazaki K., Kawa S., Kamisawa T. Clinical diagnostic criteria of autoimmune pancreatitis: revised proposal. J. Gastroenterol. 2006;41:626–631. doi: 10.1007/s00535-006-1868-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Chiari S.T., Smyrk T.C., Levy M.J., Topazian M.D., Takahashi N., Zhang L. Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin. Gastroenterol. Hepatol. 2006;4:1010–1016. doi: 10.1016/j.cgh.2006.05.017. [DOI] [PubMed] [Google Scholar]
- 84.Hivano K., Tada M., Isayama H., Yagioka H., Sasaki T., Kogure H. Long-term prognosis of autoimmune pancreatitis with and without corticosteroid treatment. Gut. 2007;56:1719–1724. doi: 10.1136/gut.2006.115246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Smit W.L., Culver E.L., Chapman R.W. New thoughts on immunoglobulin G4-related sclerosing cholangitis. Clin. Liver Dis. 2016;20:47–65. doi: 10.1016/j.cld.2015.08.004. [DOI] [PubMed] [Google Scholar]
- 86.Tanaka A., Mori M., Kubota K., Naitoh I., Nakazawa T., Takikawa H. Epidemiological features of immunoglobulin G4-related sclerosing cholangitis in Japan. Hepatobiliary Pancreat Sci. 2020;27:598–603. doi: 10.1002/jhbp.793. [DOI] [PubMed] [Google Scholar]
- 87.Ghazale A., Chari S.T., Zhang L., Smyrk T.C., Takahashi N., Levy M.J. Immunoglobulin G4-associate cholangitis: clinical profile and response to therapy. Gastroenterology. 2008;134:706–715. doi: 10.1053/j.gastro.2007.12.009. [DOI] [PubMed] [Google Scholar]
- 88.Kamisawa T., Nakazawa T., Tazuma S., Zen Y Y., Tanaka A., Ohara H. Clinical practice guidelines for IgG4-related sclerosing cholangitis. J Hepatobiliary Pancreat Sci. 2019;26:9–42. doi: 10.1002/jhbp.596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kamisawa T., Nakazawa Y.Z., Okazaki K. Advances in IgG4-related pancreatobiliary diseases. Lancet Gastroenterol Hepatol. 2018;3:575–585. doi: 10.1016/S2468-1253(18)30121-3. [DOI] [PubMed] [Google Scholar]
- 90.Takagi Y., Kubota K., Takayanagi T., Kurita Y., Ishii K., Hasegawa S. Clinical features of isolated proximal-type immunoglobulin G4-related sclerosing cholangitis. Dig. Endosc. 2019;31:422–430. doi: 10.1111/den.13320. [DOI] [PubMed] [Google Scholar]
- 91.Seo N., Kim S.Y., Lee S.S., Byun J.H., Kim J.H., Kim H.J. Sclerosing cholangitis: clinicopathologic features, imaging spectrum, and systemic approach to differential diagnosis. Korean J. Radiol. 2016;17:25–38. doi: 10.3348/kjr.2016.17.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Nakazawa T., Ohara H., Sano H., Ando T., Takashi T.J. Schematic classification of sclerosing cholangitis with autoimmune pancreatitis by cholangiography. Pancreas. 2006;32:229. doi: 10.1097/01.mpa.0000202941.85955.07. (letter) [DOI] [PubMed] [Google Scholar]
- 93.Kohosnpouri P., Habibabadi R.R., Kazhirkarzar B., Amelis S., Ghadimi M., Ghasabeh M. Imaging features of primary sclerosing cholangitis: from diagnosis to liver transplant follow-up. Radiographics. 2019;39:1939–1964. doi: 10.1148/rg.2019180213. [DOI] [PubMed] [Google Scholar]
- 94.Tanaka A., Tazuma S., Ozaki K., Nakazawa T., Inui K., Chiba T., Takikawa H. Clinical features, response to treatment, and outcomes of IgG4-related sclerosing cholangitis. Clin. Gastroenterol. Hepatol. 2017;15:920–926. doi: 10.1016/j.cgh.2016.12.038. [DOI] [PubMed] [Google Scholar]
- 95.Minaga K., Watanabe T., Chung H., Kudo M. Autoimmune hepatitis and IgG4-related disease. World J. Gastroenterol. 2019;25:2308–2314. doi: 10.3748/wjg.v25.i19.2308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Umemura T., Zen Y., Hamano H., Ichijo T., Kawa S., Nakanuma Y. IgG4 associated autoimmune hepatitis: a differential diagnosis for classical autoimmune hepatitis. Gut. 2007;56:1471–1472. doi: 10.1136/gut.2007.122283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Umemura T., Zen Y., Hamano H., Joshita S., Ichijo T., Kiyosawa K., Ota M. Clinical significance of immunoglobulin G4-associated autoimmune hepatitis. J. Gastroenterol. 2011;44(suppl. 1):48–55. doi: 10.1007/s00535-010-0323-4. [DOI] [PubMed] [Google Scholar]
- 98.Chung H., Watanabe T., Kudo M., Maenishi O., Wakatsuki Y., Chiba T. Identification and characterization of IgG4-associated autoimmune hepatitis. Liver Int. 2010;30:222–231. doi: 10.1111/j.1478-3231.2009.02092.x. [DOI] [PubMed] [Google Scholar]
- 99.Amarapurkar A.D., Amarapurkar D.N. Immunoglobulin IgG4 and autoimmune hepatitis. Trop. Gastroenterol. 2015;36:112–117. doi: 10.7869/tg.263. [DOI] [PubMed] [Google Scholar]
- 100.Canivet C.M., Anty R., Patouraux S., Saint-Paul M.C., Lebeaupin C. Immunoglobulin G4-associated autoimmune hepatitis may be found in Western countries. Dig. Liver Dis. 2016;48:302–308. doi: 10.1016/j.dld.2015.10.005. [DOI] [PubMed] [Google Scholar]
- 101.Ayademiz Y., Akcoren Z., Demiz H., Saltik Temizel I.N., Ozen H., Yuce A. Clinical and histopathological features of immunoglobulin G4-associated autoimmune hepatitis in children. J. Gastroenterol. Hepatol. 2019;34:742–746. doi: 10.1111/jgh.14525. [DOI] [PubMed] [Google Scholar]
- 102.Nakanuma Y., Ishizu Y., Zen Y., Harada K., Umemura T. Histopathology of IgG4-related autoimmune hepatitis and IgG4-related hepathopathy in IgG4-related disease. Semin. Liver Dis. 2016;36:229–241. doi: 10.1055/s-0036-1584320. [DOI] [PubMed] [Google Scholar]
- 103.Mizushima I., Kasashima S., Fujinaga Y., Kamano M., Ishizawa N. IgG4-related periaortitis/periarteritis: an under-recognized condition that is potentially life-threatening. Mod. Rheumatol. 2019;29:240–250. doi: 10.1080/14397595.2018.1546367. [DOI] [PubMed] [Google Scholar]
- 104.Perugino C.A., Wallace Z.S., Meyersohn N., Oliveira G., Stone J.R. Large vessel involvement by IgG4-related disease. Medicine (Baltimore) 2016;95 doi: 10.1097/MD.0000000000003344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Mizushima I., Kasashima S., Notohara K., Fujinaga Y., Saeki T., Zen Y. Clinical and diagnosed based on experts’ diagnosis. J.Jpn. Coll. Angiol. 2018;58:117–129. [Google Scholar]
- 106.Yamamoto M., Yajima H., Takahashi H., Yokoyama Y., Ishigami K. Everyday clinical practice in IgG4-related dacryoadenitis and/or sialadenitis: results from the SMART database. Mod Rheumatol; Early Online. 2014:1–6. doi: 10.3109/14397595.2014.950036. [DOI] [PubMed] [Google Scholar]
- 107.Kitigawa S., Zen Y., Harada K. IgG4-positive plasma cell infiltration characterizes chronic sclerosing sialadenitis (Kuttner’s tumor) Am. J. Surg. Pathol. 2005;29:783–791. doi: 10.1097/01.pas.0000164031.59940.fc. [DOI] [PubMed] [Google Scholar]
- 108.Wei T.W., Lien C.F., Hsu T.Y., He H.L. Chronic sclerosing sialadenitis of the submandibular gland: an entity of IgG4-related sclerosing disease. Int. J. Clin. Exp. Pathol. 2015;8:8628–8631. [PMC free article] [PubMed] [Google Scholar]
- 109.Razek A.A.K.A., Mukherji S. Imaging of sialadenitis. NeuroRadiol. J. 2017;30:205–215. doi: 10.1177/1971400916682752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Yamamoto M., Takahashi H., Ohara M., Suzuki C., Naishiro Y., Yamamoto H. A new conceptualization for Mikulicz’s disease as an IgG4-related plasmocytic disease. Mod. Rheumatol. 2006;16:335–340. doi: 10.1007/s10165-006-0518-Y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kottahachchi D., Topliss D.J. Immunoglobulin G4-related thyroid disease. Eur. Thyroid. 2016;5:231–239. doi: 10.1159/000452623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Khosroshahi A., Wallace Z.S., Crowe J.L., Akamiau T., Azumi A., Carruthers M.N. International consensus guidance statement on the management and treatment of IgG4-related disease. Arthritis Rheum. 2015;67:1688–1699. doi: 10.1002/art.39132. [DOI] [PubMed] [Google Scholar]
- 113.Kamisawa T., Okazawi K., Kawa S., Ito T., Inui K., Irie H. Amendment of the Japanese Consensus Guidelines for autoimmune pancreatitis, 2013 III. Treatment and prognosis of autoimmune pancreatitis. J. Gastroenterol. 2014;49:961–970. doi: 10.1007/s00535-014-0945-z. [DOI] [PubMed] [Google Scholar]
- 114.Masamune A., Nishimori I., Kikuta K., Tsuji I., Mizuno N., Iiyama T. Randomised controlled trial of long-term maintenance corticosteroid therapy in patients with autoimmune pancreatitis. Gut. 2017;66:487–494. doi: 10.1136/gutjnl-2016-312049. [DOI] [PubMed] [Google Scholar]
- 115.Okazaki K., Chari S.T., Frulloni L., Lerch M.M., Kamisawa T., Kawa S. International consensus for the treatment of autoimmune pancreatitis. Pancreatology. 2017;17:1–6. doi: 10.1016/j.pan.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 116.Campochiaro C., Della-Torre E., Lanzillotta M., Bozzolo E., Baldissera E., Milani R. Long-term efficacy of maintenance therapy with rituximab for IgG4-related disease. Eur. J. Intern. Med. 2020;74:92–98. doi: 10.1016/j.ejim.2019.12.029. [DOI] [PubMed] [Google Scholar]
- 117.Brito-Zeron P., Kostov B., Bosch X., Acar-Denizli N., Ramos-Casals M., Stone J.H. Therapeutic approach to IgG4-related disease. A systematic review. Medicine. 2016;95 doi: 10.1097/MD.0000000000004002. [DOI] [PMC free article] [PubMed] [Google Scholar]