Abstract
Background and purpose
Due to a smaller target volume when delineating prostate on magnetic resonance imaging (MRI), margins may be too tight as compared to computed tomography (CT) delineation, potentially reducing tumor control probability (TCP) in prostate radiotherapy. This study evaluated a clinically implemented MRI-based target expansion method to provide adequate margins yet limit organ-at-risk (OAR) dose as compared to CT-based delineation.
Methods and materials
Patients in this study were treated to 79.2 Gy in 44 fractions via intensity modulated radiotherapy using an MRI-based expansion method, which excluded OARs when performing a 5 mm isotropic (except 4 mm posterior) expansion from gross tumor volume to clinical target volume (CTV), followed by an isotropic 5 mm expansion to generate the planning target volume (PTV). Ten cases were re-planned using CT-delineated prostate with CTV-to-PTV expansion of isotropic 8 mm, except for a 5 mm posterior expansion, with comparison of PTV volumes, TCP and normal tissue complication probability (NTCP) to the MRI-based method. Under IRB approved protocol, we retrospectively evaluated 51 patients treated with the MRI-based method for acute bladder and rectal toxicity with CTC-AE version 4.0 used for scoring.
Results
MRI-based PTV volume differed by 4% compared to CT-based PTV volume. Radiobiological calculated TCP of the MRI-based method was found comparable to CT-based methods with an average equivalent uniform dose of 80.5 Gy and 80.1 Gy respectively. Statistically significant decrease in bladder NTCP (toxicity Grade 2 and above for 5% complications within 5 years post radiotherapy) was observed in the MRI-based method. Outcomes data collected showed 65% and 100% of patients studied experienced Grade 0/1 bladder and rectal acute toxicity respectively. Grade 2 bladder toxicity was indicated in the remaining 35% of patients studied with no Grade 3 toxicity reported.
Conclusions
Results showed comparable PTV volume with MRI-based method, and NTCP was reduced while maintaining TCP. Clinically, bladder and rectal toxicities were observed to be minimal.
1. Introduction
Radiation therapy plays an important role in the current treatment of prostate cancer. There have been several published randomized trials that have reported dose escalation as improving biochemical control, but it is also followed by an increase in toxicity [1], [2], [3]. In order for radiotherapy to be effective in the treatment of prostate cancer, and potentially limit genitourinary (GU) and gastrointestinal (GI) toxicities, high sensitivity and specificity imaging techniques, such as magnetic resonance imaging (MRI), are needed to accurately visualize the prostate for gross tumor volume (GTV) delineation. Currently, the most commonly used method for prostate target delineation is via treatment planning on computed tomography (CT) [4]. However, this method has been shown to overestimate the true volume of the prostate by up to 30% when compared to target delineation on MRI [5], [6], [7]. Moreover, it has been documented that treatment planning with an MRI delineated prostate registered to CT may reduce both bladder and rectal toxicities due to a smaller target volume [8]. Despite this reduction in target volume, MRI-based planning has not replaced conventional CT-based planning [9]. A delicate balance exists between the aim of maximum accuracy and margin reduction to avoid normal tissue toxicity, and the risk of missing microscopic extra prostatic tumor extension as a consequence of irradiated target volume reduction [4].
Target expansions beyond the delineated prostate GTV are largely motivated by anatomical, clinical, and practical implications, with many guidelines available in literature. The International Commission on Radiation Units and Measurements (ICRU) has provided recommendations for consistent volume specifications with reports 29, 50, and 62 [10], [11], [12]. In most cases, a GTV to clinical target volume (CTV) is performed to account for subclinical cancer involvement, which for prostate radiotherapy could include extraprostatic extension, seminal vesicles, or pelvic lymph nodes [13]. Further expansion to generate the planning target volume (PTV) is employed to account for patient movement, inaccurate patient positioning, and organ motion, with the PTV being a significant factor in dose escalated prostate radiotherapy [14]. These guidelines are widely varying however, with a lack of uniformity in margin selection among institutions, and a lack of definitive experimental/clinical studies providing robust information on selection of exact PTV values [15].
It has been reported that continuous prostate motion was unpredictable, and prostate displacements on the order of ≥5 mm for cumulative durations of at least 30 s were observed, thus demonstrating that prostate motion during treatment could be significant [16]. With the introduction of image guidance via fiducial tracking, it is thought that PTV margin reduction could be achieved [17]. Zhang et al reported reduced PTV margins that may allow for dose escalation by comparing tumor control probability (TCP) of two different treatment schemes: 70 Gy/35 fractions with a 10 mm PTV and no patient realignment, and dose escalated 79 Gy/39 fractions with a 5 mm PTV with patient realignment [18]. Their results concluded more favorable TCP for the dose escalated plan with 5 mm PTV margin and daily image guidance (assuming geometric uncertainties of <5 mm). However, their target delineation was performed on CT alone, and they considered intrafraction motion to be insignificant compared to interfraction motion. The results of the normal tissue complication probability (NTCP) modeling by Gauthier et al suggested that the reduction of PTV margin afforded by fiducial marker localization should decrease the rate of late rectal toxicities [19]. Again, their planning techniques were performed on CT alone, highlighting the possibility of further benefit with MRI-based delineation. Tzikas et al investigated the clinical aspects of using CT vs. MRI-based treatment planning in prostate radiotherapy, and their results showed that the use of fused CT-MRI images produce dose distributions that lead on average to better expected treatment outcome compared to the use of CT images alone [20]. Their PTV expansion methods employed an isotropic 10 mm expansion from the CTV, except 6 mm posteriorly, of which they acknowledged the CTV expansion did not allow for much organ sparing when the target is adjacent to highly radiosensitive healthy organs. Several others have developed expansion techniques based on their own clinical experience [21], [22], [23], [24]. Memorial Sloan‐Kettering Cancer Center reported their toxicity results employing a 10 mm PTV margin, except 6 mm posteriorly towards the prostate-rectum interface, with rectal grade 2 or higher (RTOG scale) complication rate of 16.5% when delivering a prescription dose of 81 Gy [25]. M.D. Anderson reported similar toxicity rates when employing their own target expansions [26]. Both of these studies were performed with prostate delineated on CT alone. Sander et al compared long-term toxicity (36 months) using CT or MRI delineated prostate with 78 Gy in 39 fractions and employed a 5 mm CTV to PTV margin to both methods [4]. Their results showed significantly lower urinary frequency and urinary retention toxicity scores, but did not find significant differences in overall urinary or rectal toxicity between the two groups. They acknowledged that longer follow up times may show a clinical relevance in terms of possible toxicity reduction, however, no acute toxicity data was collected from this study. Given that literature suggests favorable TCP and treatment outcomes with CT-based delineation methods, further investigation on the role of MRI-based target expansions, their associated clinical outcomes and any potential benefits, is warranted.
This study aimed to determine an optimum MRI-based expansion technique that can maintain target coverage as compared to CT-based methods, yet limit bladder and rectal toxicity. We hypothesized our MRI-based delineation and expansion method dependent on exclusion of organs-at-risk (OAR) might achieve similar CT-based PTV volumes. Radiobiological model (TCP and NTCP) predictions were compared between treatment plans generated using the MRI-based OAR exclusion method and conventional CT-based methods. A retrospective study was then performed under approved IRB-protocol on the evaluation of acute toxicities among 51 men diagnosed with prostate cancer who received treatment with radiation therapy utilizing the MRI-based prostate delineation and expansion technique.
2. Methods and materials
2.1. Treatment planning methods and initial PTV coverage plan comparison
Retrospective treatment plan data of ten prostate cancer patients (T1-T3N0M0) treated with radiotherapy was used for comparison study. CT scans obtained were performed on a Toshiba Aquilion (Toshiba Medical System, Otawara, Japan) using 2.5 mm slice spacing. MRI scans of 1.5 Tesla strength were obtained with T2-weighted axial scans. All images were imported into MIM 6.4.3 (MIM Software, Cleveland, OH, USA) for image fusion. Treatment plans were generated uniformly using Philips Pinnacle3 version 9.2 treatment planning system on a Varian linear accelerator (Varian Medical Systems, Palo Alto, CA, USA) with Millennium 120 leaf MLC and 0.5 cm MLC leaf width within the modulation field. Prescription used was 1.8 Gy for 44 fractions for a total dose of 79.2 Gy with plans designed to deliver 95% of the prescription dose to at least 95% of the PTV. Planning techniques were performed using a 9 field beam arrangement and uniform intensity modulation radiation therapy (IMRT) optimization.
To facilitate determination of optimum margins, plans were first contoured on MRI by radiation oncologist with target considered the CTV, then expanded isotropically by 8 mm, except 5 mm in the posterior dimension, to generate the PTV. The prostate was then delineated on CT by an alternate clinician (observer blinded) and expanded in the same manner. Treatment plans were optimized to the MRI-based PTV and the CT-based PTV volume coverage was compared on dose volume histogram (DVH).
2.2. Expansion method for MRI-based target delineation with PTV volume comparison
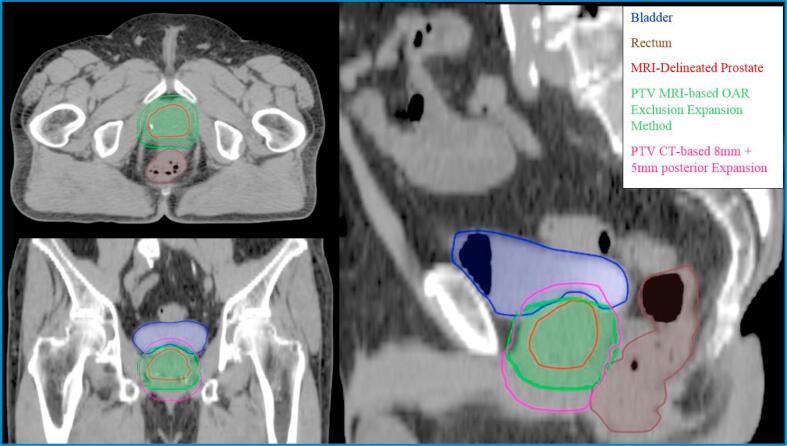
In developing the MRI-based delineation and expansion method, ICRU 50 and 62 were utilized and strictly interpreted for PTV generation. The ICRU reports call for a GTV-to-CTV expansion to account for microscopic disease extension. It is known that the peripheral zone extends postero-laterally around the gland from the apex to the base and represents the most common site in the prostate for developing prostate carcinomas [27]. To account for any microscopic extension outside the periphery, the delineated GTV on MRI was first expanded by 5 mm isotropically except for 4 mm posteriorly. Furthermore, any expansion into the bladder and rectum was carved out in order to protect both the bladder and rectum (Fig. 1). This method allowed for the CTV to curve around the critical structures and provided an adequate margin around the prostate for tumor control. The CTV was further expanded uniformly by 5 mm to create the PTV. Plans created using this method were compared to CT-delineated prostate with conventional expansions in terms of PTV volume.
Fig. 1.
Axial, coronal, and sagittal view comparing conventional expansions (isotropic 8 mm, except 5 mm posterior) to the MRI-based delineation and expansion method. The extent of expansion into the bladder is less pronounced and expansion into the rectum is comparable to conventional expansions for this one representative case. PTV expansion into the rectum is variable depending on patient anatomy.
2.3. TCP and NTCP calculations
In order to provide insight into the predictive outcomes of our proposed method, radiobiological evaluation of Equivalent Uniform Dose (EUD)-based TCP and NTCP was undertaken using an open source MATLAB code following the methods and workflow (Fig. S1 in supplementary material) of Gay and Niemierko’s published work [28]. This EUD-based model is simple and versatile as the model may be used for both TCP and NTCP calculations. This model takes the form of
| (1) |
where “a” is a dimensionless model parameter that is specific to the normal structure or tumor of interest, and Vi is dimensionless and represents the i’th partial volume receiving dose Di in Gy. To calculate the EUD-based NTCP, the following logistic function is used.
| (2) |
where the TD5/5 term is the tolerance dose for a 5% complication rate within 5 years post radiotherapy, and the γ50 is a dimensionless model parameter that is specific to the normal structure or tumor of interest and describes the slope of the dose response curve. NTCP parameters used for “a” and γ50 were 6 and 4 respectively, and a TD5/5 of 65 Gy and 60 Gy were used for the bladder and rectum respectively. To calculate TCP, the EUD is substituted into the following formula.
| (3) |
where the tumor control dose (TCD50) is the tumor dose to control 50% of the tumors when the tumor is homogeneously irradiated. TCP parameters of 70.5 Gy and 2.9 were used for TCD50 and γ50 respectively [29]. α/β ratios (tissue specific Linear Quadratic parameter of the organ being exposed) of 3 Gy were used for both bladder and rectum in the NTCP calculations [30]. In addition to TCP/NTCP calculations, mean dose to the bladder and rectum was collected and averaged across all trials for both MRI-based and CT-based methods. Statistical analysis of results was performed using the paired Student’s t-test technique.
2.4. Clinical evaluation/outcomes of the MRI-based delineation and expansion technique
Medical records, including consult notes and on treatment weekly visit notes, of 51 patients treated for prostate cancer between 2011 and 2015 were retrospectively reviewed under IRB-protocol. Patients were of average age 66.5 years with the most prominent Gleason Score being 3 + 4 = 7 and were monitored weekly for clinical progress during their treatment. Notes of adverse effects and the dose delivered at time of onset were documented. Any toxicity noted is to be assessed using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTC-AE) version 4.0. Rectal symptoms to be assessed include stool frequency, stool incontinence, rectal pain, proctitis, and rectal bleeding. Urinary symptoms to be assessed include frequency, urgency, incontinence, dysuria, urinary retention, nocturia and hematuria. Grades 0/1, 2, 3 and above were assessed for both genitourinary and gastrointestinal effects during treatment. Per the CTC-AE guidelines, in general we considered the distinguishing factor between Grade 0/1 and Grade 2 to be an intervention indicated by physician but not beyond medication prescribed. Grade 3 and above would be reserved for effects exhibiting severe pain and/or requiring further medical intervention beyond prescribed medication.
3. Results
3.1. PTV volume comparisons
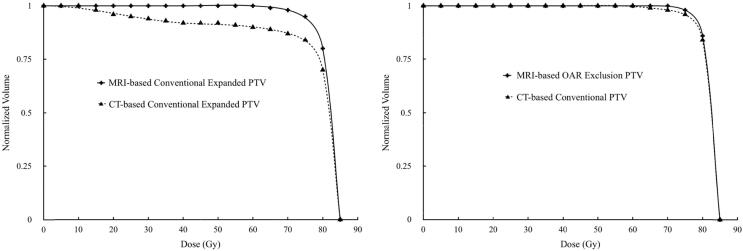
Comparisons between CT-based PTV coverage to MRI-based PTV coverage on plans optimized using MRI-based PTV with conventional expansions showed under-dosing of the CT-based PTV (Fig. 2). PTV volume of CT-delineated prostate with conventional expansions was on average 28% larger than the MRI-delineated PTV volume with conventional expansions. MRI-based delineation and expansion method dependent on OAR exclusions had an average PTV volume comparable to conventional CT-based PTV at 4% difference, and similar PTV coverage was observed on DVH for plans optimized on MRI-based delineation and expansion method (Fig. 2).
Fig. 2.
(Left) DVH of PTV comparison for plan optimized with MRI-delineated prostate and conventional expansion methods (isotropic 8 mm, except 5 mm posterior) showing under-dosing of the conventional CT-expanded PTV. (Right) DVH of PTV comparison for plan optimized with MRI-based delineation and expansion method showing comparable coverage to conventional CT-expanded PTV.
3.2. TCP and NTCP calculations with comparisons
With respect to prostate TCP calculations, results showed that the MRI-based delineation and expansion method had an EUD of 80.5 Gy and a TCP of 82%, compared to an EUD of 80.1 Gy and a TCP of 81% for the CT-based conventional expansion method, thus showing similarity between the two techniques (Table 1). Reduced bladder EUD and NTCP was observed with a minimum and maximum decrease in NTCP of <1% and 6% respectively. These results were deemed statistically significant. All but two cases exhibited a reduction in NTCP for the MRI-based expansion method, with those cases only exhibiting a <1% increase in NTCP. Rectum EUD and NTCP calculations were relatively similar with a difference of −0.3 Gy EUD and −1% NTCP between MRI-based and CT-based expansion methods. These results were not considered statistically significant. Differences in rectal NTCP were variable with some cases exhibiting an increase in NTCP and others decreasing. Minimum and maximum reduction of NTCP ranged from <1% to 9%, while minimum and maximum increase in NTCP ranged from 2% to 3%. MRI-based delineation and expansion method showed a lower average mean dose of 28.4 Gy as compared to the 36.3 Gy average mean dose for the CT-based target expansion methods. Average mean dose for the rectum were 44.7 Gy and 46.0 Gy for the MRI-based and CT-based expansion methods respectively, showing similarity across all plan trials.
Table 1.
EUD, TCP, and NTCP calculation results averaged across trials compared.
| MRI-based delineation and expansion | CT-based conventional expansion | p-value | ||
|---|---|---|---|---|
| Prostate | EUD (Gy) | 80.5 | 80.1 | 0.06 |
| TCP (%) | 82 | 81 | 0.06 | |
| Bladder | EUD (Gy) | 72.9 | 73.4 | 0.05 |
| NTCP (%) | 19 | 20 | 0.05 | |
| DMean (Gy) ± standard dev. | 28.4 ± 12.3 | 36.3 ± 4.8 | 0.01 | |
| Rectum | EUD (Gy) | 53.3 | 53.6 | 0.4 |
| NTCP (%) | 15 | 16 | 0.4 | |
| DMean (Gy) ± standard dev. | 44.7 ± 6.3 | 46.0 ± 7.5 | 0.7 | |
3.3. Toxicity scoring
For toxicity scored, all GU and GI symptoms were grouped together by grade: 33 out of the 51 patient cohort (65%) experienced Grade 0/1 acute urinary toxicities, while the remaining 18 (35%) experienced Grade 2 acute urinary toxicity. The most common Grade 2 symptoms were urinary frequency requiring prescribed medication (e.g. Tamsulosin HCL) followed by dysuria also requiring prescription medication (e.g. Phenazopyridine). All 51 patients scored for GI symptoms exhibited only Grade 0/1 toxicities. The most common side effect scored for GI toxicity was increased stool frequency with no medical intervention necessary. Acute toxicity data collected showed no Grade 3 and above GU toxicity, as well as no Grade 2 and above GI toxicity.
4. Discussion
Several guidelines exist in literature discussing prostate volume delineation and expansion in radiotherapy, as summarized by Yartsev et al [15]. However, these expansion guidelines are largely based on CT delineated prostate with minimal literature on detailed target expansions referencing exclusion of OAR’s within generated PTV’s. It was shown that CT-based PTV coverage was under-dosed when compared to MRI-based PTV coverage on plans optimized using MRI-based PTV with conventional expansions, potentially compromising TCP. Furthermore, given the possibility of prostate intrafaction motion to be significant (ranging from 2 to 7 mm), larger treatment margins may be used as an option to minimize loss of TCP, which may also compromise bladder/rectal sparing [31]. Therefore, in this present study, we provided a “recipe” to perform an MRI-based delineation and expansion method based on exclusion of OAR’s for prostate radiotherapy by use of MRI-registered to CT in order to provide adequate target coverage and limit OAR complications.
In order to further evaluate the efficacy of our expansion technique in prostate radiotherapy, we employed biological modeling in terms of both TCP and NTCP. It was shown that the EUD and TCP were slightly higher for plans employing the MRI-based delineation and expansion technique as compared to CT delineated plans with conventional expansions. Although the purpose of this comparison was mainly to show that the outcomes would be near identical to the CT-delineation with conventional expansions, it is possible that there exists a further radiobiological advantage. The max dose to the prostate was slightly higher and more conformal within the GTV for the MRI-based method than plans using the CT-delineation with conventional expansions. Due to the exclusion of portions of OAR’s within the PTV, minimizing the objective cost function of OAR’s may be more easily achieved, giving way to superior constraint of high isodose lines to within the GTV. These results are in line with prior literature, as Tzikas et al had shown that their contoured CTV target was irradiated more effectively on MRI-based treatment plans [20].
Bladder NTCP results were shown to improve as compared to CT-based expansion method, most likely as a result of the exclusion of portions of the CTV and subsequent PTV from the bladder itself, leading to a superior treatment plan. Results trended more consistently with improved calculated complication rates as compared to rectal NTCP. Rectal filling may be a significant contributing factor for the lack of statistical significance in a reduction of rectal NTCP, as it can vary greatly per patient, and be less uniform as compared to the bladder. This may lead to regions of dose that traverse more volume of rectum, leading to an increase in NTCP. This effect was evident within our results with wider variation in NTCP calculated per patient observed. However, our results on average showed a lower NTCP for rectum as compared to bladder, which is consistent for IMRT-based prostate radiotherapy treatment planning as was also noted by Tzikas et al [20].
We acknowledge the likelihood of alternative expansion methods (both theorized and in literature) that may further decrease NTCP and increase TCP. For example, a target expansion based on MRI with only a 5 mm margin that excludes expansion into the bladder and rectum. However, analysis of shift data in literature and our own institution has shown the possibility of the prostate to shift ≥5 mm in the transverse and longitudinal dimensions. This could lead to margins being too tight, compromising both TCP and NTCP, as is also noted in literature [32]. Hence, our proposed method attempts to reduce the likely-hood of the prostate shifting outside of margins to maintain TCP during treatment by performing a tiered expansion (GTV-CTV-PTV with varying expansion dimensions), but also reduce NTCP by shaping the expansion around both the bladder and the rectum.
Clinical data collected resulted in minimal bladder/rectum toxicities that were reported in patient charts. Gill, et al, published their own clinical results on GI and GU toxicities for patients treated with image guidance radiotherapy (IGRT) for prostate cancer, along with summarized acute GU and GI toxicities of other published studies with large cohorts [33]. Lips et al. reported acute Grade 2 GU toxicity in 47% and Grade 2 GI toxicity in 30% of patients within their study (IGRT-based prostate radiotherapy) [34]. In another cohort of 238 patients treated with IGRT, Soete et al. reported a higher rate of 16% with Grade 3 GU and 6% Grade 3 GI toxicity [35]. It is important to note that data collected from these studies vary widely in methodology. As such, the authors acknowledge that there are several compounding reasons for the clinical results obtained. Different target expansions, treatment planning techniques, treatment assessment tools, and patient population may each have an effect on toxicity data collected. Further investigation is warranted in order to determine the overall effect that IMRT optimization and image guidance during treatment has on the overall rectal NTCP in conjunction with our MRI-based delineation and expansion methods.
To bring light to a few limitations of this study, the authors acknowledge the limited sample size present within the data, the retrospective nature of the toxicity scoring, and a lack of follow-up data extending beyond 4 years with some data only available for approximately 2 years. Hence long term survival was not assessed in this study. Moreover, with respect to recorded prostate specific antigen (PSA) in patient charts, due to the addition of hormone therapy and lack of long term follow-up data, the PSA data reviewed only demonstrated that PSA levels were either decreasing or stable at the conclusion of follow-up by clinician.
It should be stated that successful implementation of this method will require some changes to a clinic’s workflow in prostate radiotherapy. The adoption of two imaging modalities per patient will be necessary, with staff ideally trained in appropriate patient set-up and immobilization during the MRI acquisition. Image fusion software, if not already present in the department’s treatment planning system, in addition to a minimal amount of extra time to perform requisite image fusion will be necessary. Most commercially available image fusion and contouring tools available today include workflows to better streamline and reduce the additional time to implement this MRI-based technique, and is therefore encouraged when employing for clinical use.
In this study, we presented our experience and evaluation of a MRI-based delineation and expansion method with PTV generation dependent on OAR exclusions. With our MRI-based methods we observed a reduction in NTCP while maintaining TCP as compared to conventional CT-based target delineation and expansion. Our findings also showed minimal acute toxicities in our clinical assessment. However, investigation with a larger cohort and longer follow up data is warranted to further validate our findings.
Conflict of interest
The authors declared that there is no conflict of interest.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.phro.2018.11.007.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
Schematic diagram of TCP/NTCP calculations.
References
- 1.Al-Mamgani A., van Putten W.L., Heemsbergen W.D., van Leenders G.J., Slot A., Dielwart M.F. Update of Dutch multicenter dose-escalation trial of radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys. 2008;72:980–988. doi: 10.1016/j.ijrobp.2008.02.073. [DOI] [PubMed] [Google Scholar]
- 2.Dearnaley D.P., Sydes M.R., Graham J.D., Aird E.G., Bottomley D., Cowan R.A. Escalated-dose versus standard-dose conformal radiotherapy in prostate cancer: first results from the MRC RT01 randomised controlled trial. Lancet Oncol. 2007;8:475–487. doi: 10.1016/S1470-2045(07)70143-2. [DOI] [PubMed] [Google Scholar]
- 3.Kuban D.A., Tucker S.L., Dong L., Starkschall G., Huang E.H., Cheung M.R. Long-term results of the M. D. Anderson randomized dose-escalation trial for prostate cancer. Int J Radiat Oncol Biol Phys. 2008;70:67–74. doi: 10.1016/j.ijrobp.2007.06.054. [DOI] [PubMed] [Google Scholar]
- 4.Sander L., Langkilde N.C., Holmberg M., Carl J. MRI target delineation may reduce long-term toxicity after prostate radiotherapy. Acta Oncol. 2014;53:809–814. doi: 10.3109/0284186X.2013.865077. [DOI] [PubMed] [Google Scholar]
- 5.Gao Z., Wilkins D., Eapen L., Morash C., Wassef Y., Gerig L. A study of prostate delineation referenced against a gold standard created from the visible human data. Radiother Oncol. 2007;85:239–246. doi: 10.1016/j.radonc.2007.08.001. [DOI] [PubMed] [Google Scholar]
- 6.McLaughlin P.W., Evans C., Feng M., Narayana V. Radiographic and anatomic basis for prostate contouring errors and methods to improve prostate contouring accuracy. Int J RadiatOncol Biol Phys. 2010;76:369–378. doi: 10.1016/j.ijrobp.2009.02.019. [DOI] [PubMed] [Google Scholar]
- 7.Rasch C., Barillot I., Remeijer P., Touw A., van H.M., Lebesque J.V. Definition of the prostate in CT and MRI: a multi-observer study. Int J Radiat Oncol Biol Phys. 1999;43:57–66. doi: 10.1016/s0360-3016(98)00351-4. [DOI] [PubMed] [Google Scholar]
- 8.Ali A.N., Rossi P.J., Godette K.D., Martin D., Liauw S., Vijayakumar S. Impact of magnetic resonance imaging on computed tomography-based treatment planning and acute toxicity for prostate cancer patients treated with intensity modulated radiation therapy. Pract Radiat Oncol. 2013;3:e1–e9. doi: 10.1016/j.prro.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 9.Korhonen J., Kapanen M., Keyriläinen J., Seppälä T., Tenhunen M. A dual model HU conversion from MRI intensity values within and outside of bone segment for MRI-based radiotherapy treatment planning of prostate cancer. Med. Phys. 2014;41:011704. doi: 10.1118/1.4842575. [DOI] [PubMed] [Google Scholar]
- 10.International Commission on Radiation Units and Measurements . ICRU; Washington, DC: 1978. ICRU report 29: dose specification for reporting external beam therapy with photons and electrons. [Google Scholar]
- 11.International Commission on Radiation Units and Measurements . ICRU; Bethesda, MD: 1993. ICRU report 50: prescribing, recording, and reporting photon beam therapy. [Google Scholar]
- 12.ICRU Report 62: prescribing, recording, and reporting photon beam therapy (Supplement to ICRU Report 50). Bethesda, MD: ICRU; 1999.
- 13.Zaorsky N.G., Showalter T.N., Ezzell G.A. ACR Appropriateness Criteria® external beam radiation therapy treatment planning for clinically localized prostate cancer, part I of II. Adv Radiat Oncol. 2017;2:62–84. doi: 10.1016/j.adro.2016.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Langen K.M., Jones D.T. Organ motion and its management. Int J Radiat Oncol Biol Phys. 2001;50:265–278. doi: 10.1016/s0360-3016(01)01453-5. [DOI] [PubMed] [Google Scholar]
- 15.Yartsev S., Bauman G. Target margins in radiotherapy of prostate cancer. Br J Radiol. 2016;89:1–12. doi: 10.1259/bjr.20160312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kupelian P., Willoughby T., Mahadevan A., Djemil T., Weinstein G., Jani S. Multi-institutional clinical experience with the Calypso System in localization and continuous, real-time monitoring of the prostate gland during external radiotherapy. Int J Radiat Oncol Biol Phys. 2007;67:1088–1098. doi: 10.1016/j.ijrobp.2006.10.026. [DOI] [PubMed] [Google Scholar]
- 17.Tudor G.S., Rimmer Y.L., Nguyen T.B., Cowen M.A., Thomas S.J. Consideration of the likely benefit from implementation of prostate image-guided radiotherapy using current margin sizes: a radiobiological analysis. Br J Radiol. 2012;85:1263–1271. doi: 10.1259/bjr/27924223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang M., Moiseenko V., Liu M. PTV margin for dose escalated radiation therapy of prostate cancer with daily on-line realignment using internal fiducial markers: Monte Carlo approach and dose population histogram (DPH) analysis. J Appl Clin Med Phys. 2006;7:38–49. doi: 10.1120/jacmp.v7i2.2210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gauthier I., Carrier J.F., Béliveau-Nadeau D., Fortin B., Taussky D. Dosimetric impact and theoretical clinical benefits of fiducial markers for dose escalated prostate cancer radiation treatment. Int J Radiat Oncol Biol Phys. 2009;74:1128–1133. doi: 10.1016/j.ijrobp.2008.09.043. [DOI] [PubMed] [Google Scholar]
- 20.Tzikas A., Karaiskos P., Papanikolaou N., Sandilos P., Koutsouveli E., Lavdas E. Investigating the clinical aspects of using CT vs. CT-MRI images during organ delineation and treatment planning in prostate cancer radiotherapy. Technol Cancer Res Treat. 2011;10:231–242. doi: 10.7785/tcrt.2012.500198. [DOI] [PubMed] [Google Scholar]
- 21.Zelefsky M.J., Fuks Z., Happersett L., Lee H.J., Ling C.C., Burman C.M. Clinical experience with intensity modulated radiation therapy (IMRT) in prostate cancer. Radiother Oncol. 2000;55:241–249. doi: 10.1016/s0167-8140(99)00100-0. [DOI] [PubMed] [Google Scholar]
- 22.Schultheiss T.E., Hanks G.E., Hunt M.A., Lee W.R. Incidence of and factors related to late complications in conformal and conventional radiation treatment of cancer of the prostate. Int J Radiat Oncol Biol Phys. 1995;32:643–649. doi: 10.1016/0360-3016(95)00149-s. [DOI] [PubMed] [Google Scholar]
- 23.Pollack A., Zagars G.K., Starkschall G., Childress C.H., Kopplin S., Boyer A.L. Conventional vs. conformal radiotherapy for prostate cancer: preliminary results of dosimetry and acute toxicity. IntJ Radiat Oncol Biol Phys. 1996;34:555–564. doi: 10.1016/0360-3016(95)02103-5. [DOI] [PubMed] [Google Scholar]
- 24.Pickett B., Roach M., III, Verhey L., Horine P., Malfatti C., Akazawa C. The value of nonuniform margins for six-field conformal irradiation of localized prostate cancer. IntJ Radiat Oncol Biol Phys. 1995;32:211–218. doi: 10.1016/0360-3016(95)00512-W. [DOI] [PubMed] [Google Scholar]
- 25.Skwarchuk M.W., Jackson A., Zelefsky M.J., Venkatraman E.S., Cowen D.M., Levegrün S. Late rectal toxicity after conformal radiotherapy of prostate cancer (I): multivariate analysis and dose-response. Int J Radiat Oncol Biol Phys. 2000;47:103–113. doi: 10.1016/s0360-3016(99)00560-x. [DOI] [PubMed] [Google Scholar]
- 26.Storey M.R., Pollack A., Zagars G., Smith L., Antolak J., Rosen I. Complications from radiotherapy dose escalation in prostate cancer: preliminary results of a randomized trial. Int J Radiat Oncol Biol Phys. 2000;48:635–642. doi: 10.1016/s0360-3016(00)00700-8. [DOI] [PubMed] [Google Scholar]
- 27.Oh WK, Hurwitz M, D'Amico AV, Richie JP, Kantoff PW. Biology of prostate cancer. In: Kufe DW, Pollock RE, Weichselbaum RR, Bast RC, Gansler TS, Holland JF, et al., editors. Cancer medicine 6. 6th ed. Hamilton (ON): BC Decker Inc; 2003.
- 28.Gay H.A., Niemierko A. A free program for calculating EUD-based NTCP and TCP in external beam radiotherapy. Phys Med. 2007;23:115–125. doi: 10.1016/j.ejmp.2007.07.001. [DOI] [PubMed] [Google Scholar]
- 29.Levegrün S., Jackson A., Zelefsky M.J., Venkatraman E.S., Skwarchuk M.W., Schlegel W. Risk group dependence of dose-response for biopsy outcome after three-dimensional conformal radiation therapy of prostate cancer. Radiother Oncol. 2002;63:11–26. doi: 10.1016/s0167-8140(02)00062-2. [DOI] [PubMed] [Google Scholar]
- 30.Oliveira S.M., Teixeira N.J., Fernandes L. What do we know about the α/β for prostate cancer? Med Phys. 2012;39:3189. doi: 10.1118/1.4712224. [DOI] [PubMed] [Google Scholar]
- 31.Tong X., Chen X., Li J., Xu Q., Lin M.H., Chen L. Intrafractional prostate motion during external beam radiotherapy monitored by a real-time target localization system. J Appl Clin Med Phys. 2015;16:5013. doi: 10.1120/jacmp.v16i2.5013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zarkar A., Southgate E., Karania A. Prostate radiotherapy: have our margins become too tight? Clin Oncol (R Coll Radiol) 2011;23:58. [Google Scholar]
- 33.Gill S., Thomas J., Fox C., Kron T., Rolfo A., Leahy M. Acute toxicity in prostate cancer patients treated with and without image-guided radiotherapy. Radiat Oncol. 2011;6:145. doi: 10.1186/1748-717X-6-145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lips I.M., Dehnad H., van Gils C.H., Boeken Kruger A.E., van der Heide U.A., van Vulpen M. High-dose intensity-modulated radiotherapy for prostate cancer using daily fiducial marker-based position verification: acute and late toxicity in 331 patients. Radiat Oncol. 2008;3:15. doi: 10.1186/1748-717X-3-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Soete G., Verellen D., Michielsen D., Rappe B., Keuppen F., Storme G. Image-guided conformation arc therapy for prostate cancer: early side effects. Int J Radiat Oncol Biol Phys. 2006;66:141–144. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Schematic diagram of TCP/NTCP calculations.