Abstract
There is strong evidence that the seafloor constitutes a final sink for plastics from land sources. There is also evidence that part of the plastics lying on the shallow seafloor are washed up back to the shoreline. However, little is known on the natural trapping processes leading to such landwards return. Here we investigate microplastics and larger plastic debris within beached seagrass remains including balls (aegagropilae) made of natural aggregates of vegetal fibers intertwined by seawater motion. We found up to 1470 plastic items per kg of plant material, which were mainly composed of negatively buoyant polymer filaments and fibers. Our findings show that seagrass meadows promote plastic debris trapping and aggregation with natural lignocellulosic fibers, which are then ejected and escape the coastal ocean. Our results show how seagrasses, one of the key ecosystems on Earth in terms of provision of goods and services, also counteract marine plastic pollution. In view of our findings, the regression of seagrass meadows in some marine regions acquires a new dimension.
Subject terms: Environmental impact, Ocean sciences
Introduction
Microplastics -plastic particles smaller than 5 mm in size1-derive from fragmentation and degradation of large plastic items2,3, and also from direct manufacturing of microscopic particles such as virgin plastic pellets, cosmetic microbeads and clothing microfibres4,5. Research on microplastic pollution has long focused on sea surface accumulations6–8. However, there is a growing body of evidence that floating plastic debris account for less than 1% of the global ocean plastic inventory9, whereas the vast majority sinks to the seafloor1,10,11. Microplastics have indeed been found in all marine environments, shallow and deep, close to shore and amidst ocean basins11–15. Further, recent studies have shown that bottom currents control the distribution of microplastics on the seafloor, transporting them from shallow to deep waters where they accumulate14,16. In this study, we provide evidence of the entrapment of plastic debris from the shallow marine environment by seagrasses. This represents a continuous purge of plastic debris out of the sea that has been omitted in surface (nearshore to offshore) and bottom (shallow to deep) simulations of microplastics transport3,8,17,18.
Seagrass meadows are widespread in shallow coastal waters19 and provide important ecosystem services and benefits, such as water quality improvement20, CO2 absorption21, climate change mitigation22, sediment production for seafloor and beach stabilization23, coastal protection24, nursery and refuge areas for many species25, and support in fisheries production26. We have investigated microplastics and larger plastic debris washed ashore together with natural debris of the seagrass Posidonia oceanica, a Mediterranean endemic seagrass forming lush, extensive meadows from 0.5 to 40 m of water depth. According to the latest and more accurate estimate the total area covered by P. oceanica is 1.2 M Ha27. P. oceanica has long, ribbon-like leaves, with a clear differentiation in leaf blade (photosynthetic) and leaf base or leaf sheath (non pigmented and fibrous) that attaches the leaf to the stem, called rhizome28.
As a temperate species, P. oceanica loses leaves in autumn, which are washed by waves and currents and accumulate on adjacent beaches as wrack beds. These vegetal deposits, besides attenuating wave energy, protecting the shoreline and preventing coastal erosion, influence also dune vegetation not only by providing it with nutrients but also by preventing substrate aridity29. In addition, in this species (as in other congenerics in the southern hemisphere) leaf sheaths remain attached to the rhizome when leaves shed, and are slowly buried by sedimentation in the so called “matte”, an accumulation of dead rhizomes and roots that can persist for millennia30. During the burial process, leaf sheaths, which are rich in lignocellulose, suffer mechanical erosion, releasing part of the constituent fibers that intertwine to form ball-shaped agglomerates known as seaballs, Neptune balls or aegagropilae (EG)31. These balls are also washed ashore. While leaf sheaths are present in almost all seagrass genera, only sheath cells in Posidonia have thin and lignified walls28, and thus fibers provide the needed stiffness to form EG32. The genus Posidonia has a unique fragmented distribution in the temperate waters of the Mediterranean Sea and southern Australia. Regrettably, it is estimated that between 13 and 50% of potential initial P. oceanica area may have been lost since 196027,33.
To examine the role played by these piles of vegetal remains in trapping and extracting plastic debris from sea and carrying them to shore we have examined both EG and beach wracks accumulated on different beaches of Mallorca Island, in the Western Mediterranean Sea. This island, 3640 km2 in area and ca. 560 km in perimeter, is an optimal site to address these issues, because of both the extensive meadows of P. oceanica in its waters34 and the fact that the highest accumulations of floating plastic debris in the Mediterranean Sea occur in its nearshore35.
Results and discussion
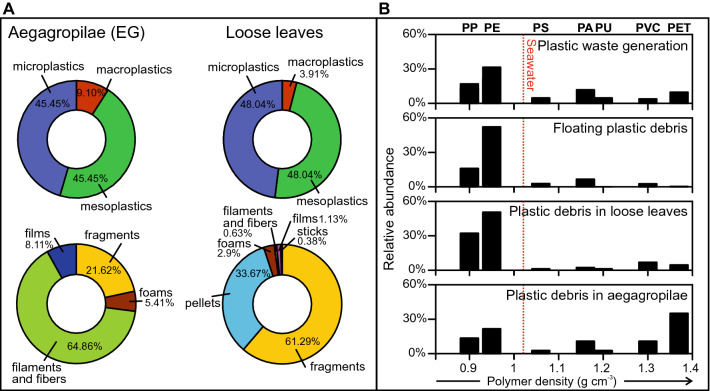
Plastic debris in loose leaves (wracks) were found in 50% of the samples, with up to 613 plastic items per kg of dead leaves. Plastic items consisted mostly of fragments (61.29%) followed by pellets (33.67%) and foams (2.90%). The polymers were identified by spectrometry and included polyethylene (PE) (50.57%) followed by polypropylene (PP) (32.18%) and polyvinyl chloride (PVC) (6.90%). Plastic sizes ranged from 0.55 to 287 mm and averaged 9.08 mm.
Plastics of different sizes were found intertwined in 17% of the inspected EG (Fig. 1A, Table S2), with up to 1,470 plastic items per kg of dead seagrass remains. The plastic debris found were mostly filaments and fibers (64.86%), fragments (21.62%), films (8.11%) and foams (5.41%) (Fig. 1A). The polymers identified by spectrometry (n = 124) included polyethylene terephthalate (PET) (35.14%), PE (21.62%), PP (13.51%), polyamide (PA) (10.81%) and PVC (10.81%) (Fig. 1B). Plastic sizes ranged from 1.05 to 59.02 mm and averaged 9.48 mm.
Figure 1.
Plastic debris in seagrass remains. (A) Pie chart illustrating size of plastic debris (microplastics < 5 mm, mesoplastics 5–25 mm, and macroplastics > 25 mm), and type of plastic (filaments and fibers, fragments, films, foams, pellets and sticks) found in beached aegagropilae (EG) and loose leaves (wracks). (B) Relative abundance of each plastic polymer in plastic waste generation, floating plastic debris, plastic debris in loose leaves and aegagropilae (EG) vs. polymer density. Plastic waste generation is from Geyer et al.64, and plastic polymers floating at the sea surface are from Suaria et al.43. Only plastic polymers with percentages > 1% in at least two matrices (floating, leaves, EG) are taken into account. PA polyamide, PE polyethylene, PET polyethylene terephthalate, PP polypropylene, PS polyestyrene, PU polyurethane, PVC polyvinyl chloride.
Plastic debris weight represented < 1.6% and < 0.15% of the mass of dead leaves and EG, respectively. Plastic weight ranged 0–13.3 mg per EG unit.
Our findings demonstrate that high density plastic items in shallow meadows are intertwined, and subsequently trapped, with the lignocellulosic debris of the seagrass P. oceanica to form EG, which are washed ashore by sea waves mainly during stormy conditions (Fig. 2). Indeed, seagrass beds are known to promote deposition and reduce resuspension of sedimentary particles as a result of the reduction of water flow, turbulence and wave action by the plant canopies36. In addition, particle collision with dense seagrass canopies plays a role in sediment transport to the seabed and extraction from the water column37. Sediment trapping by seagrass meadows may also include plastic debris. Huang et al.38 found that microplastics in seagrass meadows were enriched by a factor up to 2.9 compared to non-vegetated areas. The reduction of the flow and the trapping effect of canopies may create soft bottom accumulations of plastic debris, deposited and stranded on the seabed, from where they can hardly escape. Accordingly, a statistically significant higher loading of polymers denser than seawater such as PET, PA, and PVC (Chi-square test = 22.6, df = 1, p < 0.001) were found in EG (Fig. 1B). Their negative buoyancy may have favoured their accumulation and aggregation with natural fibres in the meadow. Low density PE and PPs, which float in sea water, still represented a significant proportion among EG trapped plastic debris. Positively buoyant plastic debris may have reached the seabed due to collision with seagrass leaves, momentum loss and particle deposition37, or due to buoyancy loss because of biofouling39 or aggregation40.
Figure 2.
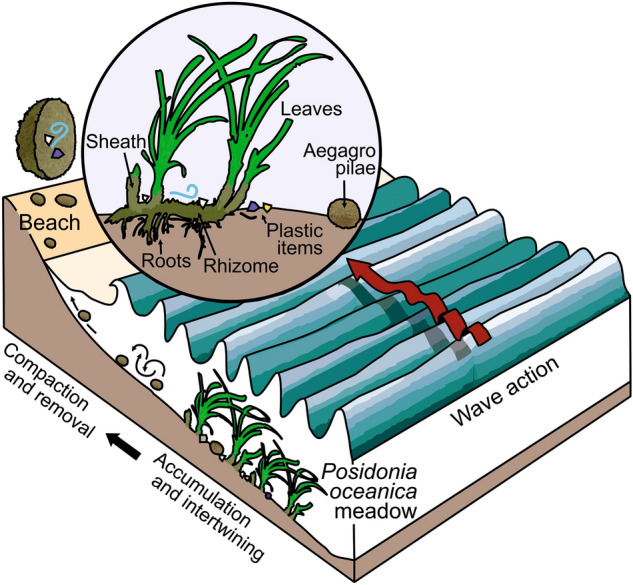
Trapping of plastic debris by seagrasses. Representation of the processes involved in the accumulation and intertwining of plastic items and sheath fibers to form plastic-rich aegagropilae (EG) found stranded in beaches.
In the seabed, it is likely that agitation caused by sea motion aggregates and holds together sheath fibers and plastic debris forming EG (Fig. 2). It has been suggested that EG formation is initiated at low abundances of vegetal fibers41, and that they grow by random aggregation of smooth fibers under relatively calmed conditions and for a long period of time32. Then, on a shorter time, sudden change in sea motion may cause repeated collisions of the EG with the seabed subsequently inducing the formation of an outward dense shell (hardening), which in turn, inhibits addition of new material32. EG size was observed to be inversely correlated with plastic abundance (r(27) = − 0.30, p < 0.01; including only EG with plastic presence). This suggests that the relatively rigidity of plastic debris, and specially polyamide filaments from fishing lines (Fig. 3), may decrease the natural tendency of the lignocellulosic fibers to agglomerate to form balls stiff when agitated by sea motion.
Figure 3.
Plastic debris in aegagropilae (EG) and loose leaves found in beaches. (A) Polyamide filaments trapped in aegagropilae (EG), and (B) beached EG and loose leaves along with plastic debris.
Plastic debris abundance per mass unit in loose dead leaves was significantly lower than those in EG (p < 0.001, Mann–Whitney U test) and mostly composed of low-density polymers (PE and PP) fragments and pellets (Fig. 1, Table S1). This suggests that plastic debris found stranded in wracks are, for the most, those drifting at the sea surface, which are transported close to shore by surface currents and wind waves, and are washed ashore at the same time as leaf litter. Indeed, similar low density polymer contributions have been found floating off the investigated beaches35 and drifting in open waters of the Mediterranean Sea43 (Fig. 1B). The occurrence of an extraordinary convective rainfall event in Mallorca Island in autumn 201744 may had loaded coastal waters with an extra amount of PP pellets (Supplementary Table 2). Drifting floating plastic items were flushed ashore during autumn storms, alongside with seagrass remains from natural massive seasonal leaf loss45. Interestingly, plastic debris in wracks along the shoreline undergo photo-oxidative degradation and gradual fragmentation46, and may be eventually backwashed to the coastal sea and transported seaward by swash waves47. Accordingly, low-density plastic debris found in wracks show smaller size (Fig. 1) and thus higher deterioration than those in EG. In contrast, enclosure of plastic debris in EG protects them from exposure to UV radiation and mechanical abrasion and thus from breakdown into smaller sized pieces.
The fate of the EG is an open question. On the one hand, and in the context of beach residues management, one possibility would be to remove the balls to eliminate their associated plastic debris. However, it would be difficult to remove the balls without removing the stranded leaf litter, which is known to protect beaches against erosion48, provide nutrients for dune plants29 and feed beach arthropod communities49. On the other hand, the dense outward shell of the EG32,42, the refractory character of their lignocellulosic fibers50 and their low N content51 provide them with a high stiffness and resistance to degradation. Thus, the probability of a plastic debris being disentangled and degraded and/or backwashed may be relatively low. In any case, what happens to the plastic debris in the EG once ashore deserves further investigation.
Our understanding of plastic fluxes and pathways is incomplete. There appears to be a considerable proportion of all plastic dumped into the ocean that is missing as it has not been found in surveys tracking floating plastic debris3,6–9. Where this “missing” plastic is has been a longstanding scientific question after being posed by Thompson et al.1. During more than 15 years, an increasing number of studies have supported the idea that most plastic ends up in the seafloor11,13,15, acting as the ultimate reservoir of our throw-away society. However, other processes may also account for some of this discrepancy between estimations of plastics dumped and plastics floating at the sea surface. There are evidences that a natural sorting for plastic debris is occurring in coastal environments, where a major part of entering plastics are stranded and captured, and only a small fraction escapes offshore52. Here we show that plastic debris in the shallow seafloor could be trapped in seagrass remains, eventually leaving the marine environment through beaching. Furthermore, taking an average plastic debris abundance in EG of 57.8 items kg−1, and a Mediterranean basin sheath fiber production of 6000–15,000 tonnes y−1 (estimate obtained combining data from Romero et al.51, Mateo and Romero53, Khiari et al.54, and Telesca et al.27) our results give a potential entrapment of 867 million plastic debris in EG each year. How many of these plastic-rich EG are annually flushed ashore is unknown. Given the ever-increasing plastic load reaching our oceans3, seagrass ecosystems such as P. oceanica meadows will play a crucial role. Therefore, in addition to the key and extensively documented ecosystem services provided by seagrass beds22,24,55, P. oceanica may provide a valuable added plastic buffering and trapping service. This may be particularly important in the Mediterranean Sea, where this species is endemic, where high microplastics loadings have been found at surface waters and on the seafloor14,40,43,56. The declining trend of the areal extent (13–50%), cover (− 1.22% yr−1 in average) and shoot density (50% thinning) of P. oceanica meadows during the last few decades27,33, though de los Santos et al.,57 report deceleration trends, points to a severe reduction of the marine plastic trapping role by this seagrass species right when we are starting to realise it.
In addition, besides climate change, spreading of invasive species, excess nutrient inputs, coastal erosion and mechanical impacts33,58, plastic pollution may also pose a significant threat to seagrasses around the world. There are some evidence of alteration of seagrass competitive intensity59, adherence to seagrass tissues60, and consumption by herbivores61,62, even though current studies are not sufficient to provide a clear picture of the consequences of plastics in seagrass ecosystems63. What is clear is that the deterioration of seagrass meadows may compromise the services they provide, so it is crucial to undertake specific actions to mitigate threats causing regression and ensure conservation.
Methods
EG and loose leaves were collected in four beaches of Mallorca island in the western Mediterranean Sea: Sa Marina, Son Serra de Marina, Costa dels Pins, in the north-east of the island, and Es Peregons Petits, in the south-west. Beaches were microtidal, gently sloping and composed by medium to fine carbonate-rich sand, and were adjacent to extensive Posidonia oceanica meadows. EG and quadrats of loose leaves (500–1000 g) were collected in July–August 2018 and December 2018–January 2019 along 50 m long transects at 0 m, 2.5 m and 7 m of the shoreline.
Once in the laboratory, samples were dried at room temperature (25 °C) and low humidity for several days. We assume low moisture content and comparability of samples (EG and loose leaves) in identical operative conditions. EGs were weighted with an analytical balance with a sensitivity of 0.01 mgr, and the length of their principal axes was measured. EG were then carefully disentangled and fibers sieved at 8 mm, 5 mm, 1 mm and 0.63 mm using stainless steel sieves. Accordingly, the smallest plastic particle size detected was 0.54 mm. The content of the sieves was transferred to petri dishes, and H2O2 30% and HCl 10% were added to remove most of the organic matter and calcium carbonate. Samples were oven-dried at 50 °C for > 24 h. The Petri dishes were inspected for plastic debris under a Nikon SMZ1000 stereo-microscope (10 ×–40 ×) coupled with a DS-Fi2 camera in a clean laboratory. All extracted plastic particles were weighted and transferred to a 90 mm Petri dish containing a black and white background that enabled high contrast with plastic colors. The same procedure was applied to loose leaves.
Each Petri dish containing plastic particles was photographed with a Jai 3CCD High Speed Color Line Scan Camera with a 50 mm F2.8–22 lens and a resolution of 150 pixel cm−1 at the CORELAB Laboratory of the University of Barcelona using a fixed light temperature (4000 K) and lens aperture (f/11). High-resolution images were processed using the image-processing ImageJ software v1.50i (https://imagej.net/), which allowed gathering information on size (i.e. the maximum distance between any two points calculated at various angles)40. Then plastics were classified into four size categories: microplastics (< 5 mm), mesoplastics (5–25 mm) and macroplastics (> 25 mm). Plastic items were also classified according to their nature and shape in fragments, filaments and fibers, films, pellets, sticks and foams. Finally, a subset of 124 particles were randomly selected and chemically identified using a Perkin Elmer Frontier FT-IR Spectrometer with a diamond crystal ATR accessory at the Scientific and Technological Centres of the University of Barcelona (CCiTUB). FT-IR spectroscopy allowed the identification of the polymer composition of each item based on IR absorption bands that represent the presence or absence of specific functional groups in the material. The spectral range analysed was between 4000 and 220 cm−1 with a 4 cm−1 resolution and 16 accumulations. Each spectrum was compared with known spectrums of Bio-Rad Sadtler Raman Spectra databases (BioRad-KnowItAll Informatics System 2015, Raman ID Expert Inc.).
Samples, sieves and petri dishes were covered wherever possible to minimize periods of exposure. A clean workspace was maintained by keeping all surfaces and equipment clean using ethanol wipes, and cotton clothing was worn.
Pairwise comparisons were performed using the Mann–Whitney U test between unpaired groups. The chi-square test was used to determine the significance between expected and observed frequencies. Furthermore, t tests were performed on the regression coefficients to establish statistical significance. All statistical tests was conducted in R (version 3.6.3) using packages dplyr, car, corrplot, qqplotr, gmodels and ggplot2. All values were considered significant when p ≤ 0.05.
Supplementary Information
Acknowledgements
We would like to thank M. Guart from the Faculty of Earth Sciences for her precious help during laboratory work, and N. Ferrer from the Scientific and Technical Centres of the University of Barcelona for technical assistance with the FT-IR spectrometer. We would like to thank Paul Lavery from Edith Cowan University for fruitful discussion on EG formation. This research has been supported by a Catalan Government Grups de Recerca Consolidats grant to GRC Geociències Marines (ref. 2017 SGR 315). Earlier suport by BAMAR thematic network (Red Española sobre BAsuras MARinas, ref.: CGL2016-81854-REDT) is equally acknowledged.
Author contributions
A.S-V. and M.C. designed the study. M.V. and W.P.d.H. undertook data acquisition and analysis. All authors contributed to interpretation of data and have drafted and revised the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-020-79370-3.
References
- 1.Thompson RC, Olsen Y, Mitchell RP, Davis A, Rowland SJ, John AWG, McGonigle A, Russell AE. Lost at sea: where is all the plastic? Science. 2004;304:838. doi: 10.1126/science.1094559. [DOI] [PubMed] [Google Scholar]
- 2.Barnes DKA, Galgani F, Thompson RC, Barlaz M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B. 2009;364:1985–1998. doi: 10.1098/rstb.2008.0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, Narayan A, Law KL. Plastic waste inputs from land into the ocean. Science. 2015;347:768–771. doi: 10.1126/science.1260352. [DOI] [PubMed] [Google Scholar]
- 4.Browne MA, Crump P, Niven SJ, Teuten E, Tonkin A, Galloway T, Thompson RC. Accumulation of microplastic on shorelines worldwide: sources and sinks. Environ. Sci. Technol. 2011;45:9175–9179. doi: 10.1021/es201811s. [DOI] [PubMed] [Google Scholar]
- 5.Napper IE, Thompson RC. Release of synthetic microplastic plastic fibres from domestic washing machines: Effects of fabric type and washing conditions. Mar. Pollut. Bull. 2016;112(1–2):39–45. doi: 10.1016/j.marpolbul.2016.09.025. [DOI] [PubMed] [Google Scholar]
- 6.Law KL, Moret-Ferguson S, Maximenko N, Proskurowski G, Peacock EE, Hafner J, Reddy CM. Plastic accumulation in the North Atlantic subtropical gyre. Science. 2010;329(5996):1185–1188. doi: 10.1126/science.1192321. [DOI] [PubMed] [Google Scholar]
- 7.Cózar A, Echevarría F, González-Gordillo JI, Irigoien X, Úbeda B, Hernández-León S, Palma AT, Navarro S, García-de-Lomas J, Ruiz A, Fernández-de-Puelles ML, Duarte CM. Plastic debris in the open ocean. PNAS. 2014;111(28):10239–10244. doi: 10.1073/pnas.1314705111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eriksen M, Lebreton LCM, Carson HS, Thiel M, Moore CJ, Borerro JC, Galgani F, Ryan PG, Reisser J. Plastic pollution in the world’s oceans: More than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS ONE. 2014;9:e111913. doi: 10.1371/journal.pone.0111913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Koelmans AA, Kooi M, Law KL, van Sebille E. All is not lost: deriving a top-down mass budget of plastic at sea. Environ. Res. Lett. 2017;12:114028. doi: 10.1088/1748-9326/aa9500. [DOI] [Google Scholar]
- 10.Claessens M, Meester SD, Landuyt LV, Clerck KD, Janssen CR. Occurrence and distribution of microplastics in marine sediments along the Belgian coast. Mar. Pollut. Bull. 2011;62:2199–2204. doi: 10.1016/j.marpolbul.2011.06.030. [DOI] [PubMed] [Google Scholar]
- 11.Woodall L, Sanchez-Vidal A, Canals M, Paterson GLJ, Coppock R, Sleight V, Calafat A, Rogers AD, Narayanaswamy BE, Thompson RC. The deep sea is a major sink for microplastic debris. R. Soc. Open Sci. 2014;1:140317. doi: 10.1098/rsos.140317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Browne M, Galloway T, Thompson RC. Spatial patterns of plastic debris along Estuarine shorelines. Environ. Sci. Technol. 2010;44:3404–3409. doi: 10.1021/es903784e. [DOI] [PubMed] [Google Scholar]
- 13.Bergmann M, Wirzberger V, Krumpen T, Lorenz C, Primpke S, Tekman MB, Gerdts G. High quantities of microplastic in arctic deep-sea sediments from the HAUSGARTEN observatory. Environ. Sci. Technol. 2017;51:11000–11010. doi: 10.1021/acs.est.7b03331. [DOI] [PubMed] [Google Scholar]
- 14.Sanchez-Vidal A, Thompson RC, Canals M, de Haan WP. The imprint of microfibres in southern European deep seas. PLoS ONE. 2018;13(11):e0207033. doi: 10.1371/journal.pone.0207033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Courtene-Jones W, Quinn B, Ewins E, Gary SF, Narayanaswamy BE. Microplastic accumulation in deep-sea sediments from the Rockall Trough. Mar. Pollut. Bull. 2020;154:111092. doi: 10.1016/j.marpolbul.2020.111092. [DOI] [PubMed] [Google Scholar]
- 16.Kane IA, Clare MA, Miramontes E, Wogelius R, Rothwell JJ, Garreau P, Pohl F. Seafloor microplastic hotspots controlled by deep-sea circulation. Science. 2020;30:eaba5899. doi: 10.1126/science.aba5899. [DOI] [PubMed] [Google Scholar]
- 17.van Sebille E, Wilcox C, Lebreton L, Maximenko M, Hardesty BD, van Franeker JA, Eriksen M, Siegel S, Galgani F, Law KL. A global inventory of small floating plastic debris. Environ. Res. Lett. 2015;10:124006. doi: 10.1088/1748-9326/10/12/124006. [DOI] [Google Scholar]
- 18.Pohl F, Eggenhuisen JT, Kane IA, Clare MA. Transport and burial of microplastics in deep-marine sediments by turbidity currents. Environ. Sci. Technol. 2020;54:4180–4189. doi: 10.1021/acs.est.9b07527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cullen-Unsworth L, Unsworth R. Seagrass meadows, ecosystem services, and sustainability. Environment. 2013;55:14–28. [Google Scholar]
- 20.de los Santos, C.M., Olivé, I., Moreira, M., Silva, A., Freitas, C., Araújo Luna, R., Quental-Ferreira, H., Martins, M., Costa, M.M., Silva, J., Cunha, M.E., Soares, F., Pousão-Ferreira, P., Santos, R. Seagrass meadows improve inflowing water quality in aquaculture ponds. Aquaculture528, 735502 (2020).
- 21.Deyanova D, Gullström M, Lyimo LD, Dahl M, Hamisi MI, Mtolera MSP, Björk M. Contribution of seagrass plants to CO2 capture in a tropical seagrass meadow under experimental disturbance. PLoS ONE. 2017;12:e0181386. doi: 10.1371/journal.pone.0181386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Duarte CM, Losada IJ, Hendriks IE, Mazarrasa I, Marba N. The role of coastal plant communities for climate change mitigation and adaptation. Nat. Clim. Change. 2013;3:961–968. doi: 10.1038/nclimate1970. [DOI] [Google Scholar]
- 23.Gacia E, Duarte CM, Marbà N, Terrados J, Kennedy H, Fortes MD, Tri NH. Sediment deposition and production in SE-Asia seagrass meadows. Estuar. Coast. Shelf Sci. 2003;56:909–919. doi: 10.1016/S0272-7714(02)00286-X. [DOI] [Google Scholar]
- 24.Ondiviela B, Losada I, Lara JL, Maza M, Galván C, Bouma J, van Belzen J. The role of seagrasses in coastal protection in a changing climate. Coast. Eng. 2014;87:158–168. doi: 10.1016/j.coastaleng.2013.11.005. [DOI] [Google Scholar]
- 25.Whitfield AK. The role of seagrass meadows, mangrove forests, salt marshes and reed beds as nursery areas and food sources for fishes in estuaries. Rev. Fish Biol. Fish. 2017;27:75–110. doi: 10.1007/s11160-016-9454-x. [DOI] [Google Scholar]
- 26.Unsworth RKF, Nordlund LM, Cullen-Unsworth LC. Seagrass meadows support global fisheries production. Conserv. Lett. 2019;12:e12566. doi: 10.1111/conl.12566. [DOI] [Google Scholar]
- 27.Telesca L, Belluscio A, Criscoli A, et al. Seagrass meadows (Posidonia oceanica) distribution and trajectories of change. Sci. Rep. 2015;5:12505. doi: 10.1038/srep12505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hartog C, Kuo J. Taxonomy and Biogeography of Seagrasses. Seagrasses: Biology, Ecology and Conservation. Amsterdam: Springer; 2006. pp. 1–24. [Google Scholar]
- 29.Del Vecchio S, Marbà N, Acosta A, Vignolo C, Traveset A. Effects of Posidonia oceanica beach-cast on germination, growth and nutrient uptake of coastal dune plants. PLoS ONE. 2013;8(7):1–8. doi: 10.1371/journal.pone.0070607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mateo MA, Romero J, Pérez M, Littler MM, Littler DS. Dynamics of millenary organic deposits resulting from the growth of the Mediterranean Seagrass Posidonia oceanica. Estuar. Coast. Shelf Sci. 1997;44:103–110. doi: 10.1006/ecss.1996.0116. [DOI] [Google Scholar]
- 31.Pietrelli L, Di Gennaro A, Menegoni P, Lecce F, Poeta G, Acosta ATR, Battisti C, Iannilli V. Pervasive plastisphere: first record of plastics in egagropiles (Posidonia spheroids) Environ. Pollut. 2017;229:1032–1036. doi: 10.1016/j.envpol.2017.07.098. [DOI] [PubMed] [Google Scholar]
- 32.Verhille G, Moulinet S, Vandenberghe N, Adda-Bedia M, Le Gal P. Structure and mechanics of aegagropilae fiber network. Proc. Natl. Acad. Sci. 2017;114:4607–4612. doi: 10.1073/pnas.1620688114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Marbà N, Díaz-Almela E, Duarte CM. Mediterranean seagrass (Posidonia oceanica) loss between 1842 and 2009. Biol. Cons. 2014;176:183–190. doi: 10.1016/j.biocon.2014.05.024. [DOI] [Google Scholar]
- 34.Ruiz, J.M., Guillén, J.E., Ramos Segura, A., Otero, M.M. Atlas de las praderas marinas de España. IEO/IEL/UICN, Murcia-Alicante-Málaga, 681 pp (2015).
- 35.Compa M, Alomar C, Mourre B, March D, Tintoré J, Deudero S. Nearshore spatio-temporal sea surface trawls of plastic debris in the Balearic Islands. Mar. Environ. Res. 2020;158:104945. doi: 10.1016/j.marenvres.2020.104945. [DOI] [PubMed] [Google Scholar]
- 36.Gacia E, Granata TC, Duarte CM. An approach to measurement of particle flux and sediment retention within seagrass (Posidonia oceanica) meadows. Aquat. Bot. 1999;65(1–4):255–268. doi: 10.1016/S0304-3770(99)00044-3. [DOI] [Google Scholar]
- 37.Hendriks IE, Sintes T, Bouma TJ, Duarte CM. Experimental assessment and modeling evaluation of the effects of the seagrass Posidonia oceanica on flow and particle trapping. Mar. Ecol. Prog. Ser. 2008;356:163–173. doi: 10.3354/meps07316. [DOI] [Google Scholar]
- 38.Huang Y, Xiao X, Xu C, Perianen YD, Hu J, Holmer M. Seagrass beds acting as a trap of microplastics—emerging hotspot in the coastal region? Environ. Pollut. 2020;257:113450. doi: 10.1016/j.envpol.2019.113450. [DOI] [PubMed] [Google Scholar]
- 39.Ryan PG. Does size and buoyancy affect the long-distance transport of floating debris? Environ. Res. Lett. 2015;10:084019. doi: 10.1088/1748-9326/10/8/084019. [DOI] [Google Scholar]
- 40.de Haan WP, Sanchez-Vidal A, Canals M. Floating microplastics and aggregate formation in the Western Mediterranean Sea. Mar. Pollut. Bull. 2019;140:523–535. doi: 10.1016/j.marpolbul.2019.01.053. [DOI] [PubMed] [Google Scholar]
- 41.Cannon J. An experimental investigations of Posidonia balls. Aquat. Bot. 1979;6:407–410. doi: 10.1016/0304-3770(79)90080-9. [DOI] [Google Scholar]
- 42.Verhille G, Le Gal P. Aggregation of fibers by waves. In: Abcha N, Pelinovsky E, Mutabazi I, editors. Nonlinear Waves and Pattern Dynamics. Cham: Springer; 2018. [Google Scholar]
- 43.Suaria G, Avio C, Mineo A, et al. The Mediterranean plastic soup: synthetic polymers in Mediterranean surface waters. Sci. Rep. 2016;6:37551. doi: 10.1038/srep37551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lorenzo-Lacruz J, Amengual A, Garcia C, Morán-Tejeda E, Homar V, Maimó-Far A, Hermoso A, Ramis C, Romero R. Hydro-meteorological reconstruction and geomorphological impact assessment of the October 2018 catastrophic flash flood at Sant Llorenç, Mallorca (Spain) Nat. Hazards Earth Syst. Sci. 2019;19:2597–2617. doi: 10.5194/nhess-19-2597-2019. [DOI] [Google Scholar]
- 45.Jiménez MA, Beltran R, Traveset A, Calleja Cortes ML, Delgado-Huertas A, Marbà N. Aeolian transport of seagrass (Posidonia oceanica) beach-cast to terrestrial systems. Estuarine Coast. Shelf Sci. 2017;196:31–44. doi: 10.1016/j.ecss.2017.06.035. [DOI] [Google Scholar]
- 46.Rånby B. Photodegradation and photo-oxidation of synthetic polymers. J. Anal. Appl. Pyrol. 1989;15:237–247. doi: 10.1016/0165-2370(89)85037-5. [DOI] [Google Scholar]
- 47.Hinata H, Mori K, Ohno K, Miyao Y, Kataoka T. An estimation of the average residence times and onshore-offshore diffusivities of beached microplastics based on the population decay of tagged meso- and macrolitter. Mar. Pollut. Bull. 2017;122(1–2):17–26. doi: 10.1016/j.marpolbul.2017.05.012. [DOI] [PubMed] [Google Scholar]
- 48.Boudouresque CF, Pergent G, Pergent-Martini C, Ruitton S, Thibault T, Verlaque M. The necromass of the Posidonia oceanica seagrass meadow: fate, role, ecosystem services and vulnerability. Hydrobiologia. 2016;781:25–42. doi: 10.1007/s10750-015-2333-y. [DOI] [Google Scholar]
- 49.Colombini I, Mateo MA, Serrano O, Fallaci M, Gagnarli E, Serrano L, Chelazzi L. On the role of Posidonia oceanica beach wrack for macroinvertebrates of a Tyrrhenian sandy shore. Acta Oecol. 2009;35:32–44. doi: 10.1016/j.actao.2008.07.005. [DOI] [Google Scholar]
- 50.Trevathan-Tackett SM, Macreadie PI, Sanderman J, Baldock J, Howes JM, Ralph PJ. A global assessment of the chemical recalcitrance of seagrass tissues: implications for long-term carbon sequestration. Front. Plant Sci. 2017;8:925. doi: 10.3389/fpls.2017.00925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Romero J, Pergent G, Pergent-Martin C, Mateo MA, Regnier C. The detritic compartment in a Posidonia oceanica meadow: litter features, decomposition rates, and mineral stocks. Mar. Ecol. 1992;13(1):69–83. doi: 10.1111/j.1439-0485.1992.tb00341.x. [DOI] [Google Scholar]
- 52.Lebreton L, Egger M, Slat B. A global mass budget for positively buoyant macroplastic debris in the ocean. Sci. Rep. 2019;9:12922. doi: 10.1038/s41598-019-49413-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mateo MA, Romero J. Detritus dynamics in the seagrass Posidonia oceanica: elements for an ecosystem carbon and nutrient budget. Mar. Ecol. Prog. Ser. 1997;151:43–53. doi: 10.3354/meps151043. [DOI] [Google Scholar]
- 54.Khiari R, Mhenni MF, Belgacem MN, Mauret E. Chemical composition and pulping of date palm rachis and Posidonia oceanica—a comparison with other wood and non-wood fibre sources. Biores. Technol. 2010;101:775–780. doi: 10.1016/j.biortech.2009.08.079. [DOI] [PubMed] [Google Scholar]
- 55.Hemminga MA, Duarte CM. Seagrass Ecology. Cambridge: Cambridge University Press; 2000. [Google Scholar]
- 56.Pedrotti ML, Petit S, Elineau A, Bruzaud S, Crebassa JC, Dumontet B, et al. Changes in the Floating Plastic Pollution of the Mediterranean Sea in relation to the distance to land. PLoS ONE. 2016;11(8):e0161581. doi: 10.1371/journal.pone.0161581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.de los Santos CB, Krause-Jensen D, Alcoverro T, et al. Recent trend reversal for declining European seagrass meadows. Nat. Commun. 2019;10:3356. doi: 10.1038/s41467-019-11340-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Orth RJ, Carruthers TJB, Dennison WC, Duarte C, Fourquerean JW, Heck KL, Hughes RA, Kendrick G, Kenworthy JW, Olyarnik S, Short FT, Waycott M, Williams SL. A global crisis for Seagrass ecosystems. Bioscience. 2006;56:987–996. doi: 10.1641/0006-3568(2006)56[987:AGCFSE]2.0.CO;2. [DOI] [Google Scholar]
- 59.Balestri E, Menicagli V, Vallerini F, Lardicci C. Biodegradable plastic bags on the seafloor: a future threat for seagrass meadows? Sci. Total Environ. 2017;605–606:755–763. doi: 10.1016/j.scitotenv.2017.06.249. [DOI] [PubMed] [Google Scholar]
- 60.Jones KL, Hartl MGJ, Bell MC, Capper A. Microplastic accumulation in a Zostera marina L. bed at Deerness sound, Orkney, Scotland. Mar. Pollut. Bull. 2020;152:11083. doi: 10.1016/j.marpolbul.2020.110883. [DOI] [PubMed] [Google Scholar]
- 61.Remy F, Collard F, Gilbert B, Compère P, Eppe G, Lepoint G. When microplastic is not plastic: the ingestion of artificial cellulose fibers by macrofauna living in seagrass macrophytodetritus. Environ. Sci. Technol. 2015;49:11158–11166. doi: 10.1021/acs.est.5b02005. [DOI] [PubMed] [Google Scholar]
- 62.Yokota K, Waterfield H, Hastings C, Davidson E, Kwietniewski E, Wells B. Finding the missing piece of the aquatic plastic pollution puzzle: interaction between primary producers and microplastics. Limnol. Oceanogr. Lett. 2017;2:91–104. doi: 10.1002/lol2.10040. [DOI] [Google Scholar]
- 63.Bonnano G, Orlando-Bonaca M. Marine plastics: what risks and policies exist for seagrass ecosystems in the Plasticene? Mar. Pollut. Bull. 2020;158:111425. doi: 10.1016/j.marpolbul.2020.111425. [DOI] [PubMed] [Google Scholar]
- 64.Geyer R, Jambeck J, Law KL. Production, use, and fate of all plastics ever made. Sci. Adv. 2017;3(7):e1700782. doi: 10.1126/sciadv.1700782. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.