Abstract
It remains controversial whether entecavir (ETV) and tenofovir disoproxil fumarate (TDF) is associated with different clinical outcomes for chronic hepatitis B (CHB). This study aimed to compare the long-term risk of ETV versus TDF on hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (ICC) in CHB patients from a large multi-institutional database in Taiwan. From 2011 to 2018, a total of 21,222 CHB patients receiving ETV or TDF were screened for eligibility. Patients with coinfection, preexisting cancer and less than 6 months of follow-up were excluded. Finally, 7248 patients (5348 and 1900 in the ETV and TDF groups, respectively) were linked to the National Cancer Registry database for the development of HCC or ICC. Propensity score matching (PSM) (2:1) analysis was used to adjust for baseline differences. The HCC incidence between two groups was not different in the entire population (hazard ratio [HR] 0.82; 95% confidence interval [CI] 0.66–1.02, p = 0.078) and in the PSM population (HR 0.83; 95% CI 0.65–1.06, p = 0.129). Among decompensated cirrhotic patients, a lower risk of HCC was observed in TDF group than in ETV group (HR 0.54; 95% CI 0.30–0.98, p = 0.043, PSM model). There were no differences between ETV and TDF groups in the ICC incidence (HR 1.84; 95% CI 0.54–6.29, p = 0.330 in the entire population and HR 1.04; 95% CI 0.31–3.52, p = 0.954 in the PSM population, respectively). In conclusion, treatment with ETV and TDF showed a comparable long-term risk of HCC and ICC in CHB patients.
Subject terms: Gastroenterology, Oncology
Introduction
Chronic hepatitis B (CHB) is one of the most common chronic viral infections worldwide, affecting approximately 350 million people1. It may result in serious complications, such as liver failure, advanced cirrhosis, and/or hepatocellular carcinoma (HCC) among 15–40% of infected patients1–3. The risk of cirrhosis and/or HCC in CHB increases proportionally as serum HBV DNA levels increase3. Previous studies have demonstrated that antiviral treatment with nucleos(t)ide analogues (NA) treatment, either by lamivudine, entecavir (ETV) or tenofovir disoproxil fumarate (TDF), can reduce the risk of HCC development, cirrhotic events and mortality4–8.
Currently, both ETV and TDF are equally recommended as first-line NAs treatment for CHB patients. ETV and TDF are high potent and high genetic barrier NAs, with excellent antiviral activity in both hepatitis B e antigen (HBeAg)-positive and HBeAg-negative CHB. Nevertheless, a recent study from Choi et al. showed that TDF treatment was more effective in lowering risk of HCC compared with ETV treatment in a Korean nationwide cohort of CHB patients9. This result was also successfully validated from a hospital-based cohort9. However, subsequent studies did not show the consistent results even though propensity score (PS) matching and inverse probability of treatment weighting analysis was used to minimize selection bias9–13. A multi-center cohort study in South Korea demonstrated that the overall HCC incidence was not statistically different between ETV and TDF treatment10. More recently, TDF treatment was also not associated with a lower risk of HCC than ETV treatment among Asian and non-Asian patients in a multi-center study12.
Intrahepatic cholangiocarcinoma (ICC) is the second most common type of primary liver cancer behind HCC, accounting for 10–15% of primary liver cancers14. The incidence and mortality of ICC is markedly increasing over the past two decades worldwide15–17. In recent years, several studies have indicated that chronic HBV infection is a significant risk factor for ICC17–19. HBV-associated ICC patients displays significantly different clinicopathological characteristics as well as survival outcomes17–19. A recent study has demonstrated that preoperative NAs therapy could decrease HBV reactivation and prolong long-term survival after liver resection for ICC patients with a high HBV DNA level20. To our knowledge, there were no studies to evaluated the ICC incidence in patients treated with ETV vs. TDF.
We therefore conducted a retrospective cohort study using the Chang Gung Research Database (CGRD) for CHB patients undergoing ETV or TDF treatment since 2011. CGRD is a de-identified database derived from medical records of Chang Gung Memorial Hospital (CGMH), which was the largest multi-institutional electronic medical records collection in Taiwan21. CGMH is currently the largest Taiwanese medical care system, comprising 4 tertiary‐care medical centers and 3 major teaching hospitals. This medical care system, with more than 10,000 beds and over 280,000 inpatients per year, provides about 10% of all medical service used by the Taiwanese people annually21.
Materials and methods
Study cohorts
Between January 2011 and October 2018, a total of 21,222 CHB patients who were initially treated with ETV or TDF were screened for eligibility. Patients who had co-infection with human immunodeficiency virus or hepatitis C virus by serological assays or preexisting cancer were excluded. Also, patients with follow-up duration and exposure to ETV or TDF of less than 6 months, HCC or ICC development during the first 6 months, and missing data of serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), platelet counts, alpha-fetoprotein protein (AFP), albumin, bilirubin, international normalized ratio (INR) or exposure to both ETV and TDF during the follow-up period were excluded. Finally, 7248 patients (ETV: 5348, TDF: 1900) were included in the analyses. We collected the date of NAs prescription and the number of days supplied. The defined daily doses (DDDs) recommended by the WHO (World Health Organization)22 were used to quantify the usage of ETV or TDF. Cumulative DDD was estimated as the sum of dispensed DDD of ETV or TDF from the starting date.
Liver cirrhosis was either histopathologically (n = 65) or clinically diagnosed (n = 2115). Clinical diagnosis was based on the ultrasound findings as coarse liver parenchyma with nodular contour and small liver size and the presence of features of portal hypertension23,24. The Child–Turcotte–Pugh (CTP) score was calculated to classify cirrhosis, and those with a CTP score above 6 (class B or C) were defined as having hepatic decompensation. The endpoints of this study were HCC or ICC, which were identified based on the diagnosis codes retrieved from Cancer Registry Database and medical records in CGRD. The diagnosis of HCC or ICC was ascertained by histology or imaging criteria compatible with the guidelines of the American Association for the Study of Liver Disease25. In clinical principle, patients with combined HCC and ICC were classified as ICC. Informed consent was obtained from all participants. This study was approved by the Research Ethics Committee of Chang Gung Memorial Hospital (IRB no. 201901734B0C501) and was conducted in accordance with the principles of Declaration of Helsinki and the International Conference on Harmonization for Good Clinical Practice.
Matched cohort
To further examine the effect of NAs use, we used PS to estimate the probabilities of assigning a patient to use ETV or TDF, given background variables including age, sex, ALT, AST, platelet counts, AFP, albumin, bilirubin, INR, AST to platelet ratio index (APRI), fibrosis index based on 4 factors (FIB-4), cirrhosis, diabetes, CTP score, and Charlson Comorbidity Index (CCI). ETV and TDF patients were matched by using PS at a ratio of 2:1. Overall, 4956 patients (1652 matched sets) were included in the matched cohort. Serum concentrations of albumin, INR of prothrombin time, AFP, platelet count, liver cirrhosis, diabetes, FIB-4, APRI, CTP score and CCI were regarded as potential confounders and identified form medical records. Besides, previous exposure to lamivudine, telbivudine or adefovir was identified as a confounder.
Statistical analyses
The distribution of demographic factors, laboratory data and the proportions of co-morbidities between the ETV and TDF patients in the study cohort and matched cohort were compared. We used the Kaplan–Meier method to estimate the cumulative incidences of HCC and ICC. The log-rank test was performed to examine differences in the risk for HCC and ICC in the cohort. Finally, Cox proportional hazards models were used to compute the hazard ratios (HRs) accompanying 95% confidence interval (CI) after adjustment for potential confounders. Two-tailed p < 0.05 was considered to be significant.
Patients with a death date in the admission file and those from the beneficiaries register who were lost to follow-up were censored. To examine potential effect modifiers, we conducted sensitivity analyses in main model with additional covariates and analyses stratified by groups according to sex and age and with or without cirrhosis and diabetes. We also examined the outcomes stratified by different CTP score and previous exposure to lamivudine, telbivudine, or adefovir. These analyses were applied to evaluate the difference and consistency between ETV or TDF use and the risk of HCC and ICC. All of these analyses were carried out using SAS statistical software (Version 9.4; SAS Institute, Cary, NC, USA).
Results
Baseline characteristics
The baseline characteristics of the study population are shown in Table 1. Patients treated with TDF were younger, less likely to be diabetic, and more likely to be compensated, and have exposure to NAs use and positive hepatitis B e antigen (HBeAg) as compared to those receiving ETV therapy. The mean HBV DNA and duration of follow-up were comparable between these two groups.
Table 1.
Baseline characteristics.
| Cohort | PS Matched cohort | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Entecavir | Tenofovir | p-value | Entecavir | Tenofovir | p-value | |||||
| N | 5348 | 1900 | 3304 | 1652 | ||||||
| Age, year (SD) | 54 | 11.92 | 51 | 12.19 | < 0.001 | 52 | 11 | 52 | 12 | 0.989 |
| Gender | 0.072 | 0.966 | ||||||||
| Male | 3544 | 66% | 1302 | 69% | 2210 | 67% | 1106 | 67% | ||
| Female | 1804 | 34% | 598 | 31% | 1094 | 33% | 546 | 33% | ||
| BMI, mean (SD) | 24.65 | 4.33 | 24.85 | 4.38 | 0.143 | 24.7 | 4.1 | 24.8 | 4.4 | 0.644 |
| Comorbidity, N (%) | ||||||||||
| Liver cirrhosis | 1590 | 30% | 590 | 31% | 0.347 | 930 | 28% | 509 | 31% | 0.732 |
| DM | 1165 | 22% | 336 | 18% | < 0.001 | 632 | 19% | 310 | 19% | 0.759 |
| Prior NA use* | 460 | 9% | 440 | 23% | < 0.001 | 268 | 8% | 383 | 23% | < 0.001 |
| cDDDs, mean (SD) | 647.4 | 576.8 | 907.4 | 695.6 | < 0.001 | 658.4 | 573.3 | 913.7 | 699.9 | < 0.001 |
| Stratified cDDD, N (%) | < 0.001 | < 0.001 | ||||||||
| cDDDs ≤ 1095 | 4377 | 82% | 1266 | 67% | 2682 | 81% | 1094 | 66% | ||
| cDDDs > 1095 | 971 | 18% | 634 | 33% | 622 | 19% | 558 | 34% | ||
| Follow-up (years) | 0.050 | 0.025 | ||||||||
| Mean (SD) | 3.30 | 2.05 | 3.34 | 1.84 | 3.34 | 2.03 | 3.42 | 1.85 | ||
| Median (Q1–Q3) | 2.95 | 1.47–4.83 | 3.16 | 1.72–4.94 | 3.01 | 1.53–4.88 | 3.26 | 1.78–5.01 | ||
| Lab data, mean (SD) | ||||||||||
| Creatinine (mg/dL) | 1.05 | 1.33 | 0.90 | 0.89 | 0.755 | 0.98 | 1.18 | 0.9 | 1.0 | 0.468 |
| AST (U/L) | 165 | 397 | 165 | 349 | 0.002 | 166 | 405 | 166 | 355 | 0.062 |
| ALT (U/L) | 213 | 469 | 233 | 459 | < 0.001 | 227 | 496 | 228 | 458 | 0.002 |
| Platelet (103/μL) | 181 | 87 | 18 | 72 | 0.206 | 182 | 82 | 179 | 74 | 0.940 |
| AFP (ng/mL) | 169 | 6669 | 26 | 115 | 0.003 | 30 | 444 | 27 | 122 | 0.003 |
| Albumin (g/dL) | 3.96 | 0.69 | 4.15 | 0.61 | < 0.001 | 4.08 | 0.62 | 4.1 | 0.6 | 0.734 |
| Bilirubin (mg/dL) | 2.07 | 4.14 | 1.87 | 3.56 | 0.002 | 1.95 | 3.94 | 1.9 | 3.7 | 0.001 |
| INR | 1.16 | 0.28 | 1.14 | 0.27 | 0.006 | 1.15 | 0.26 | 1.1 | 0.3 | 0.928 |
| HBV DNA (log IU/mL) | 3.5 | 2.32 | 3.18 | 2.29 | 0.150 | 3.38 | 2.29 | 3.1 | 2.3 | 0.171 |
| HBeAg positive, N (%) | 849 | 16% | 544 | 29% | < 0.001 | 586 | 18% | 435 | 26% | < 0.001 |
| FIB-4 | < 0.001 | 0.807 | ||||||||
| Mean (SD) | 4.28 | 6.54 | 3.53 | 4.66 | 3.73 | 4.79 | 3.76 | 4.91 | ||
| Median (Q1-Q3) | 2.32 | 1.35–4.71 | 2.11 | 1.25–3.80 | 2.24 | 1.32–4.31 | 2.24 | 1.33–4.14 | ||
| APRI | 0.126 | 0.200 | ||||||||
| Mean (SD) | 3.75 | 11.00 | 3.41 | 8.19 | 3.53 | 9.87 | 3.52 | 8.52 | ||
| Median (Q1–Q3) | 1.08 | 0.48–2.77 | 1.10 | 0.55–2.57 | 1.09 | 0.50–2.61 | 1.11 | 0.54–2.63 | ||
| CTP class | < 0.001 | 0.469 | ||||||||
| A | 4214 | 79% | 1584 | 83% | 2741 | 83% | 1355 | 82% | ||
| B | 877 | 16% | 268 | 14% | 458 | 14% | 249 | 15% | ||
| C | 257 | 5% | 48 | 3% | 105 | 3% | 48 | 3% | ||
| CCI, N (%) | < 0.001 | 0.003 | ||||||||
| 0 | 134 | 3% | 52 | 3% | 90 | 3% | 30 | 2% | ||
| 1–2 | 2188 | 41% | 1087 | 57% | 1606 | 49% | 881 | 53% | ||
| ≥ 3 | 3026 | 57% | 761 | 40% | 1608 | 49% | 741 | 45% | ||
| Mean (SD) | 3.32 | 2.19 | 2.66 | 1.90 | < 0.001 | 2.86 | 1.90 | 2.85 | 1.94 | 0.520 |
| Median (Q1–Q3) | 3 | 2–5 | 2 | 1–4 | 2 | 1–4 | 2 | 1–4 | ||
PS propensity score, SD standard deviation, BMI body mass index, DM diabetes mellitus, NA nucleos(t)ide analogue, cDDD cumulative defined daily doses (DDDs), AST aspartate aminotransferase, ALT alanine aminotransferase, AFP alpha-fetoprotein, INR international normalized ratio, HBV hepatitis B virus, HBeAg hepatitis B e antigen, FIB-4 fibrosis index based on 4 factors, APRI AST to platelet ratio index, CTP Child–Turcotte–Pugh, CCI Charlson comorbidity index.
*Lamivudine, telbivudine, adefovir.
HCC occurrence
During the study period, 375 and 100 patients in the ETV and TDF groups developed HCC, with the annual incidence rate of 2.13 (95% CI 1.92–2.35) per 100 person-years (PY) and 1.58 (95% CI 1.30–1.92) per 100 PY, respectively (p = 0.007) (Fig. 1A). However, the incidence of HCC was not significantly different between these two groups by multivariable-adjusted analysis with full model (adjusted for age, sex, liver cirrhosis, diabetes, APRI, FIB4, CCI, CTP, AST, ALT, platelet, AFP, albumin, bilirubin and INR) or with main model (adjusted for age, sex, liver cirrhosis and diabetes) (Table 2).
Figure 1.
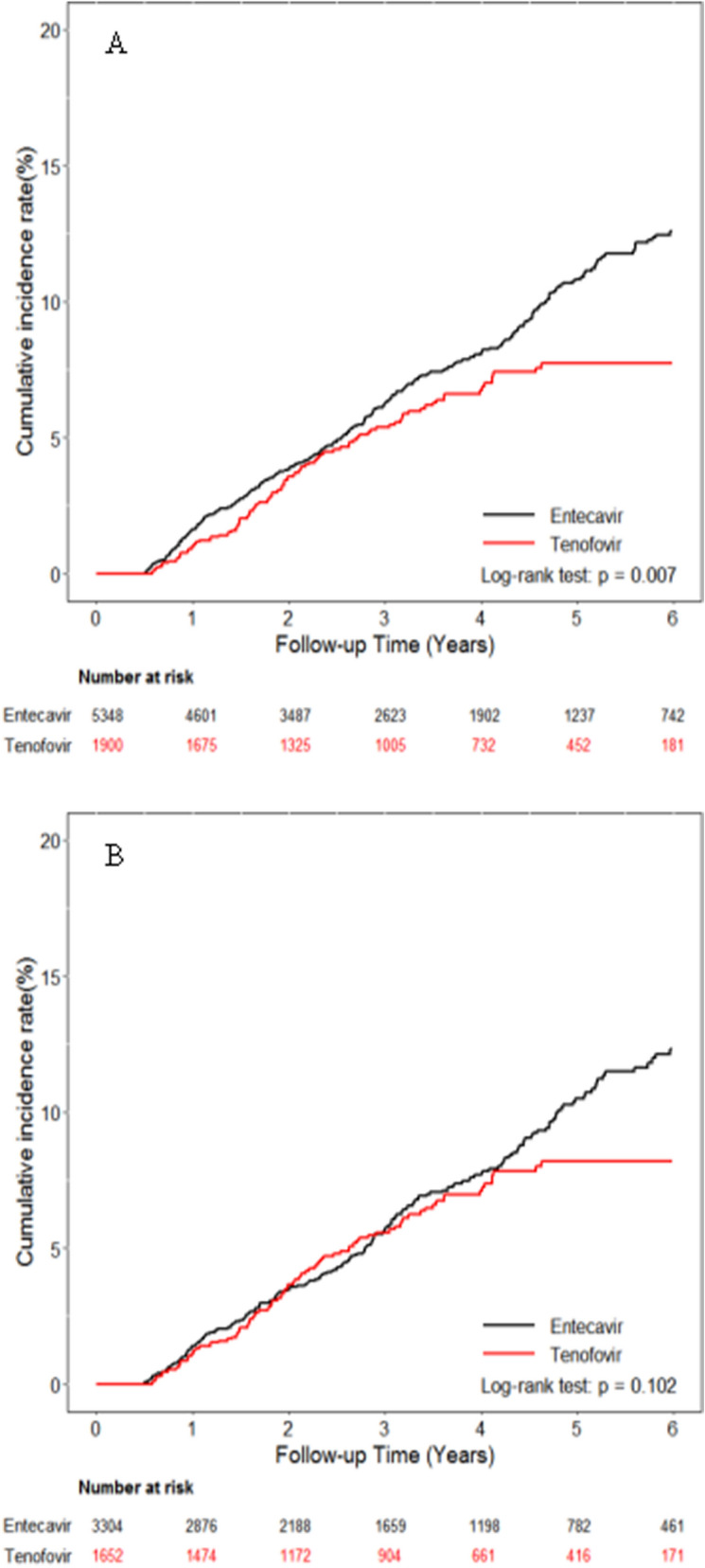
Cumulative incidences of HCC. (A) Entire cohort (B) Propensity score matching cohort.
Table 2.
HR for HCC.
| Cohort | PS matched cohort | |||||||
|---|---|---|---|---|---|---|---|---|
| Tenofovir (ref: Entecavir) | Tenofovir (ref: Entecavir) | |||||||
| HR | 95% CI | P-value | HR | 95% CI | P-value | |||
| Crude model | 0.74 | 0.59 | 0.92 | 0.007 | 0.82 | 0.64 | 1.04 | 0.102 |
| Full model* | 0.89 | 0.71 | 1.12 | 0.315 | 0.87 | 0.69 | 1.11 | 0.274 |
| Main model** | 0.82 | 0.66 | 1.02 | 0.078 | 0.83 | 0.65 | 1.06 | 0.129 |
| IPTW | 0.86 | 0.69 | 1.06 | 0.149 | ||||
| Additional covariates | ||||||||
| Main model + APRI | 0.82 | 0.66 | 1.02 | 0.076 | 0.83 | 0.65 | 1.06 | 0.133 |
| Main model + FIB4 | 0.83 | 0.67 | 1.04 | 0.106 | 0.83 | 0.65 | 1.05 | 0.123 |
| Main model + CTP | 0.83 | 0.66 | 1.03 | 0.091 | 0.83 | 0.65 | 1.06 | 0.130 |
| Main model + AFP | 0.82 | 0.66 | 1.03 | 0.083 | 0.83 | 0.65 | 1.06 | 0.137 |
| Main model + CCI | 0.81 | 0.65 | 1.01 | 0.065 | 0.84 | 0.66 | 1.06 | 0.145 |
| Main model + Prior NA+ use | 0.86 | 0.69 | 1.08 | 0.189 | 0.85 | 0.66 | 1.09 | 0.193 |
| Subgroup analysis*** | ||||||||
| Sex | ||||||||
| Female | 0.68 | 0.40 | 1.14 | 0.145 | 0.72 | 0.41 | 1.26 | 0.246 |
| Male | 0.86 | 0.67 | 1.10 | 0.216 | 0.85 | 0.65 | 1.12 | 0.248 |
| Age, y/o | ||||||||
| ≤ 50 | 0.74 | 0.48 | 1.14 | 0.167 | 0.79 | 0.48 | 1.30 | 0.358 |
| > 50 | 0.84 | 0.65 | 1.09 | 0.187 | 0.86 | 0.65 | 1.13 | 0.271 |
| DM | ||||||||
| No | 0.83 | 0.64 | 1.07 | 0.141 | 0.84 | 0.64 | 1.11 | 0.218 |
| Yes | 0.80 | 0.51 | 1.28 | 0.352 | 0.78 | 0.48 | 1.28 | 0.328 |
| Liver cirrhosis | ||||||||
| No | 0.95 | 0.60 | 1.48 | 0.811 | 1.08 | 0.66 | 1.76 | 0.775 |
| Yes | 0.78 | 0.61 | 1.01 | 0.061 | 0.77 | 0.58 | 1.01 | 0.060 |
| Prior NA | ||||||||
| No | 0.78 | 0.60 | 1.01 | 0.054 | 0.79 | 0.60 | 1.04 | 0.095 |
| Yes | 1.25 | 0.74 | 2.11 | 0.406 | 1.10 | 0.61 | 2.01 | 0.748 |
| Stratified cDDD | ||||||||
| cDDDs ≤ 1095 | 1.03 | 0.82 | 1.31 | 0.791 | 1.07 | 0.83 | 1.39 | 0.588 |
| cDDDs > 1095 | 0.72 | 0.36 | 1.43 | 0.342 | 0.59 | 0.28 | 1.25 | 0.170 |
| CTP score | ||||||||
| A | 0.92 | 0.72 | 1.18 | 0.530 | 0.91 | 0.70 | 1.19 | 0.498 |
| B/C | 0.51 | 0.29 | 0.89 | 0.018 | 0.54 | 0.30 | 0.98 | 0.043 |
PS propensity score, HR hazard ratio, CT confidence interval, IPTW inverse probability treatment weighting, APRI AST to platelet ratio index, FIB-4 fibrosis index based on 4 factors, CTP Child–Turcotte–Pugh, AFP alpha-fetoprotein, CCI Charlson comorbidity index, NA nucleos(t)ide analogue, DM diabetes mellitus.
*Full model is adjusted for age, sex, liver cirrhosis, DM, APRI, FIB4, CCI, CTP, AST, ALT, platelet, AFP, albumin, bilirubin and INR. **Main model is adjusted for age, sex, liver cirrhosis, DM. ***HRs were estimated by fitting the main model.
+lamivudine, telbivudine, adefovir.
In the PS-matched analysis, the risk of HCC was comparable between ETV and TDF groups, with the annual incidence of 2.03 (95% CI 1.78–2.31) per 100 PY and 1.67 (95% CI 1.36–2.04) per 100 PY, respectively (p = 0.102) (Fig. 1B). There was no significant difference in HCC occurrence between these two groups by multivariable-adjusted analysis with full model or with main model (Table 2).
In subgroup analyses, Kaplan–Meier curves showed that a lower risk of HCC was found in TDF group than in ETV group among decompensate patients in either the entire cohort (p = 0.003) (Fig. 2A) or PS-matched cohort (p = 0.042) (Fig. 2B). In contrast, there were no differences in HCC incidence between ETV and TDF groups in subgroup analyses stratified by age, sex, liver cirrhosis, diabetes and exposure to NUCs use (Table 2).
Figure 2.
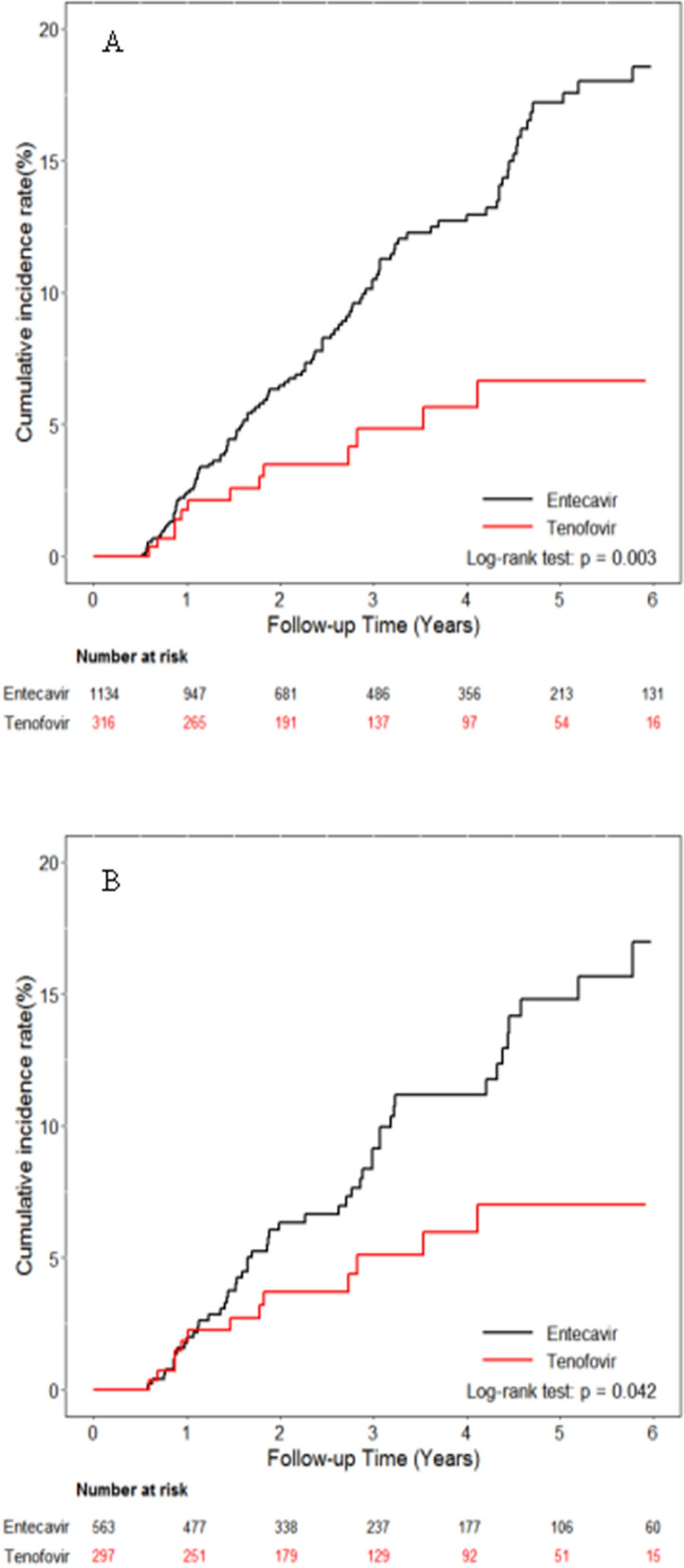
Cumulative incidences of HCC in patients with decompensated liver cirrhosis. (A) Entire cohort (B) Propensity score matching cohort.
Based on Cox proportional hazards analysis, old age (HR, 1.04; 95% CI 1.03–1.05; p < 0.001), male gender (HR, 1.90; 95% CI 1.43–2.51; p < 0.001), liver cirrhosis (HR, 3.20; 95% CI 2.43–4.20; p < 0.001), low platelet count (HR, 0.994; 95% CI 0.992–0.996; p < 0.001), low albumin level (HR, 0.77; 95% CI 0.65–0.91; p = 0.002) and high FIB-4 (HR, 1.02; 95% CI 1.01–1.04; p = 0.012) were independent risk factors for HCC development (Table 3).
Table 3.
Risk factors for HCC in PS matched cohort.
| Univariate analysis | Multivariate analysis | |||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | |||
| Main model | ||||||||
| Tenofovir (ref: Entecavir) | 0.82 | 0.64 | 1.04 | 0.102 | 0.83 | 0.65 | 1.06 | 0.129 |
| Age, y/o | 1.05 | 1.04 | 1.06 | < 0.001 | 1.04 | 1.03 | 1.05 | < 0.001 |
| Male (ref: female) | 1.95 | 1.48 | 2.58 | < 0.001 | 1.90 | 1.43 | 2.51 | < 0.001 |
| Liver cirrhosis (ref: without) | 4.07 | 3.11 | 5.31 | < 0.001 | 3.20 | 2.43 | 4.20 | < 0.001 |
| DM (ref: without) | 1.63 | 1.26 | 2.10 | < 0.001 | 1.19 | 0.92 | 1.54 | 0.192 |
| Covariates | ||||||||
| Plateleta | 0.990 | 0.988 | 0.992 | < 0.001 | 0.994 | 0.992 | 0.996 | < 0.001 |
| AFPa | 0.999 | 0.998 | 1.001 | 0.448 | 0.999 | 0.997 | 1.001 | 0.222 |
| Albumina | 0.60 | 0.51 | 0.70 | < 0.001 | 0.77 | 0.65 | 0.91 | 0.002 |
| Bilirubina | 0.99 | 0.96 | 1.02 | 0.488 | 0.96 | 0.92 | 0.99 | 0.488 |
| INRa | 1.57 | 1.16 | 2.14 | 0.004 | 1.04 | 0.68 | 1.59 | 0.854 |
| FIB-4a | 1.04 | 1.03 | 1.05 | < 0.001 | 1.02 | 1.01 | 1.04 | 0.012 |
| APRIa | 1.01 | 1.00 | 1.02 | 0.204 | 0.99 | 0.98 | 1.01 | 0.407 |
| Prior NAa (ref: no use) | 0.96 | 0.70 | 1.31 | 0.782 | 0.87 | 0.63 | 1.19 | 0.388 |
PS propensity score, HR hazard ratio, CT confidence interval, DM diabetes mellitus, AFP alpha-fetoprotein, INR international normalized ratio, FIB-4 fibrosis index based on 4 factors, APRI AST to platelet ratio index, NA nucleos(t)ide analogue.
aAdjusted for tenofovir, sex, age, liver cirrhosis, DM.
As shown in Fig. 3, patients treated with TDF had significantly better overall survival compared to those receiving ETV in the entire cohort (p < 0.001) (Fig. 3A) and in the PS-matched cohort (p < 0.001) (Fig. 3B). While as regards to liver-related death, there was no significant difference between these two groups in the PS-matched cohort (p = 0.99) (Fig. 4B), although patients treated with TDF had significantly lower liver-related death compared to those receiving ETV in the entire cohort (p = 0.009) (Fig. 4A).
Figure 3.
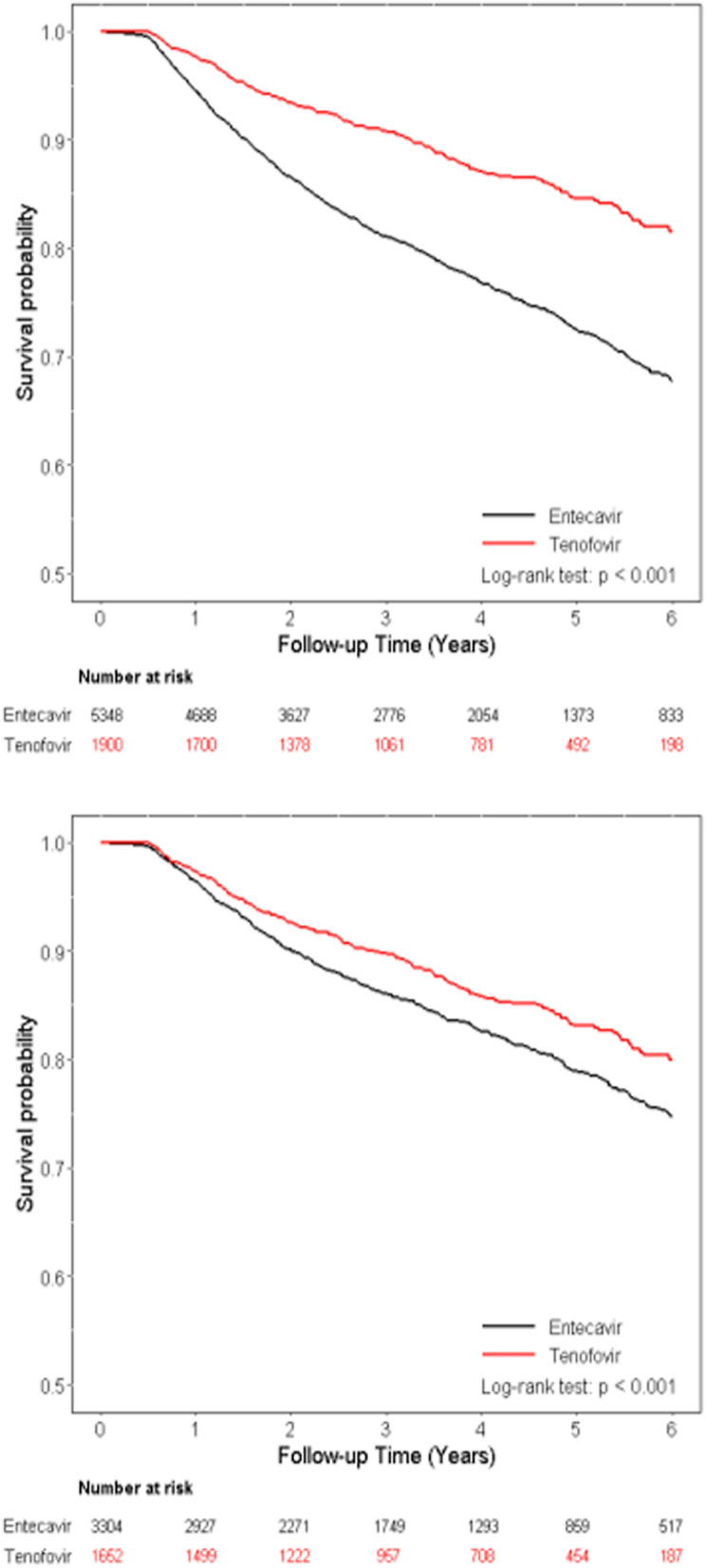
Cumulative incidences of overall survival. (A) Entire cohort (B) Propensity score matching cohort.
Figure 4.
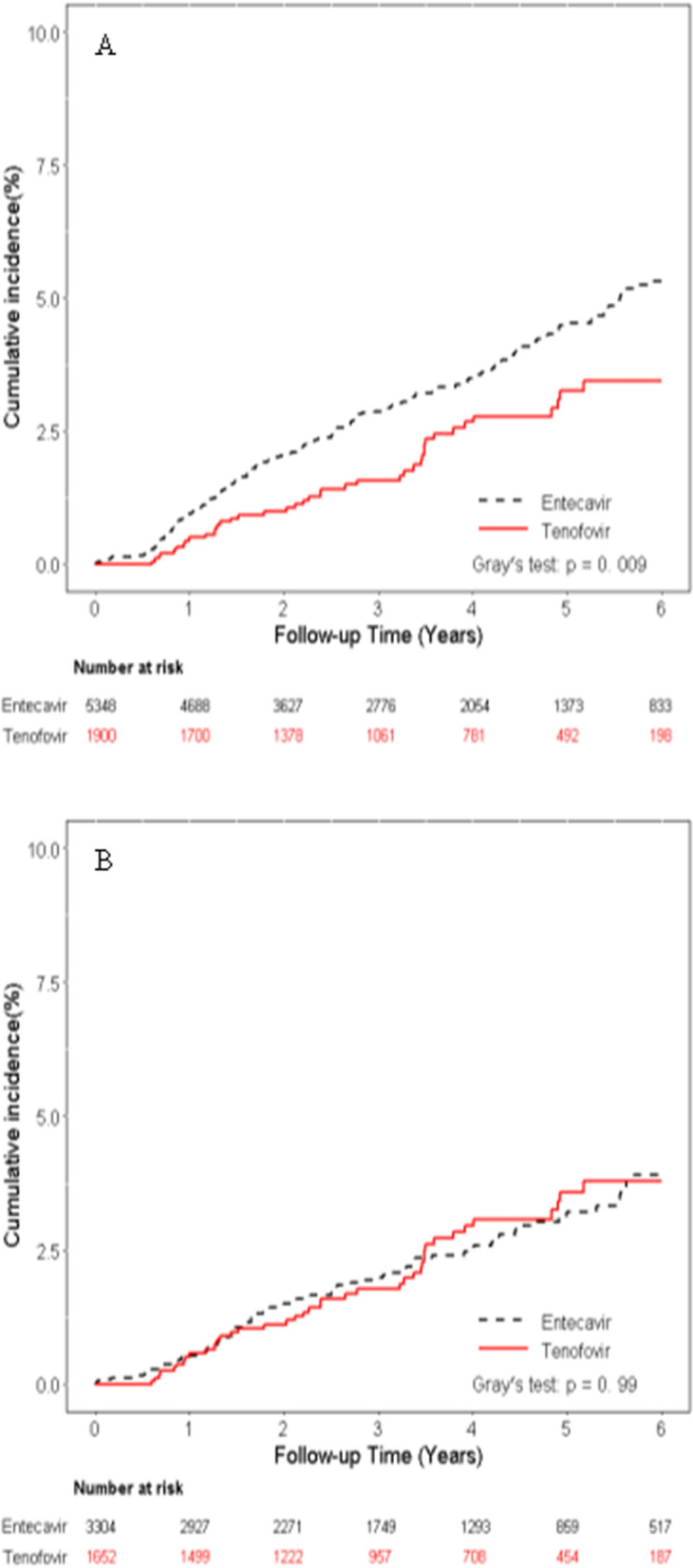
Cumulative incidences of liver-related death. (A) Entire cohort (B) Propensity score matching cohort.
ICC occurrence
During follow-up, 10 and 4 patients in the ETV and TDF groups developed ICC, with the annual incidence rate of 0.05 (95% CI 0.03–0.10) per 100 PY and 0.06 (95% CI 0.02–0.16) per 100 PY, respectively (p = 0.642) (Fig. 5A). In the PS-matched ETV and TDF cohorts, the incidence of ICC was also similar, with the annual incidence of 0.09 (95% CI 0.05–0.16) per 100 PY and 0.07 (95% CI 0.03–0.18) per 100 PY, respectively (p = 0.89) (Fig. 5B). No difference was found between ETV and TDF groups by multivariable-adjusted analysis with full model or with main model (Table 4).
Figure 5.
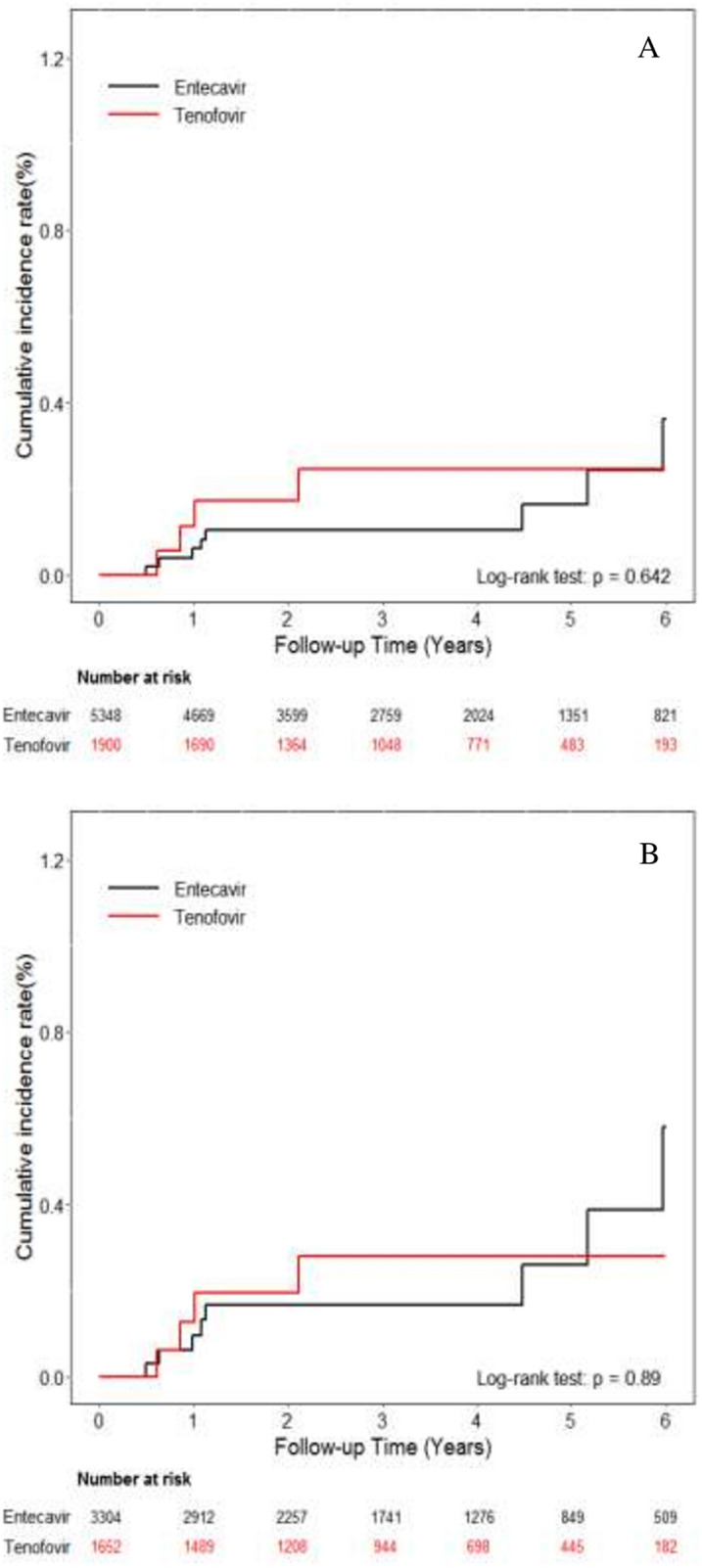
Cumulative incidences of ICC. (A) Entire cohort (B) Propensity score matching cohort.
Table 4.
HR for ICC.
| Cohort | PS matched cohort | |||||||
|---|---|---|---|---|---|---|---|---|
| Tenofovir (ref: Entecavir) | Tenofovir (ref: Entecavir) | |||||||
| HR | 95% CI | P-value | HR | 95% CI | P-value | |||
| Crude model | 1.32 | 0.41 | 4.30 | 0.643 | 0.92 | 0.28 | 2.99 | 0.890 |
| Full model* | 1.84 | 0.54 | 6.29 | 0.330 | 1.04 | 0.31 | 3.52 | 0.954 |
| Main model** | 1.67 | 0.51 | 5.51 | 0.397 | 0.94 | 0.29 | 3.07 | 0.914 |
| IPTW | 1.70 | 0.57 | 5.02 | 0.341 | ||||
| Additional covariates | ||||||||
| Main model + APRI | 1.86 | 0.56 | 6.21 | 0.313 | 1.03 | 0.31 | 3.42 | 0.961 |
| Main model + FIB4 | 1.72 | 0.52 | 5.69 | 0.371 | 0.93 | 0.29 | 3.06 | 0.909 |
| Main model + CTP | 1.65 | 0.50 | 5.45 | 0.413 | 0.94 | 0.29 | 3.06 | 0.912 |
| Main model + AFP | 1.67 | 0.51 | 5.49 | 0.401 | 0.94 | 0.29 | 3.07 | 0.915 |
| Main model + CCI | 1.66 | 0.50 | 5.48 | 0.404 | 0.93 | 0.28 | 3.06 | 0.904 |
| Subgroup analysis*** | ||||||||
| Sex | ||||||||
| Female | 3.36 | 0.21 | 53.80 | 0.392 | 1.47 | 0.09 | 24.51 | 0.787 |
| Male | 1.40 | 0.37 | 5.35 | 0.624 | 0.81 | 0.21 | 3.09 | 0.762 |
| Age, y/o | ||||||||
| ≤ 50 | 0.00 | 0.00 | 0.997 | 0.00 | 0.00 | 0.997 | ||
| > 50 | 2.12 | 0.61 | 7.30 | 0.235 | 1.24 | 0.36 | 4.26 | 0.733 |
| DM | ||||||||
| No | 2.08 | 0.49 | 8.80 | 0.319 | 1.27 | 0.30 | 5.35 | 0.745 |
| Yes | 1.03 | 0.12 | 9.23 | 0.980 | 0.50 | 0.06 | 4.52 | 0.537 |
| Liver cirrhosis | ||||||||
| No | 0.00 | 0.00 | 0.998 | 0.00 | 0.00 | 0.998 | ||
| Yes | 2.00 | 0.58 | 6.87 | 0.271 | 1.09 | 0.32 | 3.74 | 0.887 |
| Prior NA | ||||||||
| No | 0.78 | 0.60 | 1.01 | 0.054 | 0.79 | 0.60 | 1.04 | 0.095 |
| Yes | 1.25 | 0.74 | 2.11 | 0.406 | 1.10 | 0.61 | 2.01 | 0.748 |
| Stratified cDDD | ||||||||
| cDDDs ≤ 1095 | 2.16 | 0.65 | 7.19 | 0.211 | 1.19 | 0.36 | 3.95 | 0.780 |
| CDDDs > 1095 | – | – | – | – | – | – | – | – |
| CTP score | ||||||||
| A | 1.71 | 0.42 | 6.97 | 0.455 | 1.11 | 0.28 | 4.48 | 0.882 |
| B/C | 1.60 | 0.16 | 15.89 | 0.688 | 0.56 | 0.06 | 5.49 | 0.615 |
PS propensity score, HR hazard ratio, CT confidence interval, IPTW inverse probability treatment weighting, APRI AST to platelet ratio index, FIB-4 fibrosis index based on 4 factors, CTP Child–Turcotte–Pugh, AFP alpha-fetoprotein, CCI Charlson comorbidity index, DM diabetes mellitus.
*Full model is adjusted for age, sex, liver cirrhosis, DM, APRI, FIB4, CCI, CTP, AST, ALT, platelet, AFP, albumin, bilirubin and INR. **Main model is adjusted for age, sex, liver cirrhosis, DM. ***HRs were estimated by fitting the main model.
The univariate analysis found that old age (HR, 1.10; 95% CI 1.05–1.16; p < 0.001), liver cirrhosis (HR, 6.27; 95% CI 1.40–28.01; p = 0.016) and high APRI (HR, 1.10; 95% CI 1.05–1.16; p < 0.001) were significant risk factors of ICC development. Further multivariate analyses showed that old age (HR, 1.09; 95% CI 1.04–1.15; p = 0.001) and high APRI (HR, 1.04; 95% CI 1.00–1.09; p = 0.044) were independent variables (Table 5).
Table 5.
Risk factors for ICC in PS matched cohort.
| Univariate analysis | Multivariate analysis | |||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | |||
| Main model | ||||||||
| Tenofovir (ref: Entecavir) | 0.92 | 0.28 | 2.99 | 0.890 | 0.94 | 0.29 | 3.07 | 0.914 |
| Age, y/o | 1.10 | 1.05 | 1.16 | < 0.001 | 1.09 | 1.04 | 1.15 | 0.001 |
| Male (ref: Female) | 2.70 | 0.60 | 12.08 | 0.194 | 2.94 | 0.65 | 13.23 | 0.160 |
| Liver Cirrhosis (ref: without) | 6.27 | 1.40 | 28.01 | 0.016 | 3.59 | 0.78 | 16.42 | 0.100 |
| DM (ref: without) | 2.69 | 0.90 | 8.04 | 0.077 | 1.54 | 0.50 | 4.72 | 0.449 |
| Covariates | ||||||||
| Plateleta | 0.996 | 0.988 | 1.004 | 0.297 | 1.004 | 0.996 | 1.012 | 0.365 |
| AFPa | 0.999 | 0.989 | 1.008 | 0.790 | 0.998 | 0.986 | 1.010 | 0.763 |
| Albumina | 0.60 | 0.29 | 1.24 | 0.167 | 0.80 | 0.36 | 1.77 | 0.585 |
| Bilirubina | 0.95 | 0.75 | 1.19 | 0.643 | 0.91 | 0.67 | 1.23 | 0.530 |
| INRa | 2.00 | 0.63 | 6.30 | 0.237 | 1.73 | 0.43 | 6.93 | 0.436 |
| FIB-4a | 1.14 | 0.93 | 1.39 | 0.223 | 1.12 | 0.88 | 1.42 | 0.353 |
| APRIa | 1.05 | 1.02 | 1.08 | < 0.001 | 1.04 | 1.00 | 1.09 | 0.044 |
PS propensity score, HR hazard ratio, CT confidence interval, DM diabetes mellitus, AFP alpha-fetoprotein, INR international normalized ratio, FIB-4 fibrosis index based on 4 factors, APRI AST to platelet ratio index.
aAdjusted for tenofovir, sex, age, liver cirrhosis, DM.
Discussion
This is one of the largest PS-matched cohort studies comparing the long-term effect of ETV and TDF on incidence of HCC in CHB patients. In this multi-institutional study, we did not find the difference between ETV and TDF in the cumulative risk of HCC development despite a lower incidence of TDF-treated patients in the unadjusted analysis. However, a lower risk of HCC was observed in TDF group compared to ETV group among decompensated cirrhotic patients after adjustment for confounding factors and with PS matching analysis. Meanwhile, we provided the first evidence that there was no difference in the ICC incidence between CHB patients treated with ETV and TDF.
Our study consisted of a large database of patients enrolled since 2011 when TDF was available and reimbursed in Taiwan, thus both ETV and TDF groups of patients had comparable treatment duration. This is considerably different from most studies in which patients treated with ETV had more than 3 years of follow-up period compared to TDF group9–13. Although patients receiving ETV appeared to be older and have more advanced disease than TDF group, these could be explained that TDF might be less prescribed in the elderly for the concerns about osteoporosis and renal toxicity26,27. Furthermore, our patients were collected from CGMH care system, in which physicians used the same ultrasonographic scoring system to evaluate cirrhosis23,24 and similar surveillance protocol for ETV or TDF-treated patients using serum alpha-fetoprotein and ultrasonography28–30. Taken together, we could speculate that the bias in our study might be relatively smaller than those reported previously9–13.
In analysis for HCC development, our findings confirmed that age, male gender, cirrhosis, platelet count, FIB-4 and albumin level were associated with HCC development in CHB patients treated with ETV or TDF. On the contrary, there were no associations of pretreatment HBV DNA, serum AST and ALT levels with HCC incidence in CHB patients continuously receiving NUCs therapy. These results were in accordance with those in previous cohort studies9–13. Although the risk of HCC was comparable between the two groups, subgroup analysis showed that a lower risk of HCC was found among decompensated cirrhotic patients treated with TDF than ETV. The mechanisms for this significant difference remained unclear. One of the possible explanations was that ETV has been shown to be carcinogenic in mice and rats when administered at higher doses those used in humans31. ETV is recommended as 0.5 mg daily for NAs-naïve and 1.0 mg daily for NAs-resistant CHB and decompensated liver diseases25,26. Whether higher dose of ETV during long-term treatment was associated with carcinogenic potential especially in patients with decompensated liver cirrhosis who have increased chromosomal instability of hepatocytes should be further studied32,33. The other possible explanation was that TDF as a nucleotide analogue might have an additional pharmacological effect by inducing a rise in the serum levels of interferon-lambda 3, a potent antitumor activity in murine models of cancer including HCC34,35.
A number of risk factors have been shown to be associated with the occurrence of ICC. In particular, persons with HBV infection had an increased risk of ICC (rate ratio 3.17–3.42) than those without HBV infection in meta-analyses36,37. Previous epidemiological studies have shown that the incidence rates of ICC were 0.43–9.08 per 100,000 PY among patients who were hepatitis B surface antigen seropositive38,39. Compared to these data, the incidence rate of ICC in our patients appeared to be higher (0.09 and 0.07 per 100 PY in the PS-matched ETV and TDF cohorts, respectively). This should be attributed to the more advanced disease as well as high HBV load in our study population. Of note, we first found that old age and high APRI were independent risk factors of ICC development in CHB patients treated with NUCs. Moreover, there was no difference in the ICC incidence between CHB patients receiving ETV and TDF.
The strength of our study was its large sample size of patients and detailed subgroup analyses which made our data more reliable. Nevertheless, this study has the usual limitations related to its retrospective and observational design and to electronic data collection, including incomplete patient records and potential selection bias. However, we adjusted this shortcoming by using PS matching and multivariable adjustment to minimize the influence of the baseline characteristics. Second, our study did not analyze the on-treatment parameters including virological and biochemical responses. Although previous studies showed that a higher virological response by TDF than ETV treatment might explain in part for the difference of HCC incidence between TDF and ETV cohorts, on-treatment virologic and biochemical response such as at 1 year of treatment was not independently associated with HCC9,11.
In conclusion, this study showed that treatment with ETV and TDF in CHB patients did not differ in the long-term incidence of HCC and ICC. While in patients with decompensated cirrhosis, a lower risk of HCC was found in TDF group than in ETV group. Further prospective studies are needed to validate our findings.
Supplementary Information
Acknowledgements
This study was supported by contract grants CGRPG6K0011 from Chang Gung Memorial Hospital, Taiwan. We thank Health Information and Epidemiology Laboratory at Chiayi Chang Gung Memorial Hospital for the statistical work.
Author contributions
Y.H.Y. and C.H.H. were involved in study design and interpretation of data. T.S.C., W.M.C., C.H.S., S.Y.T., C.W.Y., Y.Y.H. and S.N.L. were in charge of the patients and participated in interpretation of data. C.P.L. and M.L.T. performed the statistical analysis and participated in interpretation of data. All authors reviewed and approved the final version of the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Te-Sheng Chang and Yao-Hsu Yang.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-020-80523-7.
References
- 1.Kim BK, Revill PA, Ahn SH. HBV genotypes: Relevance to natural history, pathogenesis and treatment of chronic hepatitis B. Antivir. Ther. 2011;16:1169–1186. doi: 10.3851/IMP1982. [DOI] [PubMed] [Google Scholar]
- 2.Iloeje UH, Yang HY, Su J, Jen CL, You SL, Chen CJ. Predicting cirrhosis risk based on the level of circulating hepatitis B viral load. Gastroenterology. 2006;130:678–686. doi: 10.1053/j.gastro.2005.11.016. [DOI] [PubMed] [Google Scholar]
- 3.Chen CJ, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA. 2006;295:65–73. doi: 10.1001/jama.295.1.65. [DOI] [PubMed] [Google Scholar]
- 4.Liaw YF, et al. Lamivudine for patients with chronic hepatitis B and advanced liver disease. N. Engl. J. Med. 2004;351:1521–1531. doi: 10.1056/NEJMoa033364. [DOI] [PubMed] [Google Scholar]
- 5.Wong GLH, et al. Entecavir treatment reduces hepatic events and deaths in chronic hepatitis B patients with liver cirrhosis. Hepatology. 2013;58:1537–1547. doi: 10.1002/hep.26301. [DOI] [PubMed] [Google Scholar]
- 6.Hosaka T, et al. Long-term entecavir treatment reduces hepatocellular carcinoma incidence in patients with hepatitis B virus infection. Hepatology. 2013;58:98–107. doi: 10.1002/hep.26180. [DOI] [PubMed] [Google Scholar]
- 7.Su TH, et al. Four-year entecavir therapy reduces hepatocellular carcinoma, cirrhotic events and mortality in chronic hepatitis B patients. Liver Int. 2016;36:1755–1764. doi: 10.1111/liv.13253. [DOI] [PubMed] [Google Scholar]
- 8.Nguyen MH, et al. Reduced incidence of hepatocellular carcinoma in cirrhotic and noncirrhotic patients with chronic hepatitis B treated with tenofovir-A propensity score-matched study. J. Infect. Dis. 2019;219:10–18. doi: 10.1093/infdis/jiy391. [DOI] [PubMed] [Google Scholar]
- 9.Choi J, et al. Risk of hepatocellular carcinoma in patients treated with entecavir vs tenofovir for chronic hepatitis B: A Korean Nationwide Cohort Study. JAMA Oncol. 2019;5:30–36. doi: 10.1001/jamaoncol.2018.4070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim SU, et al. A multicenter study of entecavir vs. tenofovir on prognosis of treatment-naive chronic hepatitis B in South Korea. J. Hepatol. 2019;71:456–464. doi: 10.1016/j.jhep.2019.03.028. [DOI] [PubMed] [Google Scholar]
- 11.Yip TCF, et al. Tenofovir is associated with lower risk of hepatocellular carcinoma than entecavir in patients with chronic HBV Infection in China. Gastroenterology. 2020;158:215–225. doi: 10.1053/j.gastro.2019.09.025. [DOI] [PubMed] [Google Scholar]
- 12.Hsu YC, et al. Tenofovir versus entecavir for hepatocellular carcinoma prevention in an international consortium of chronic hepatitis B. Am. J. Gastroenterol. 2020;115:271–280. doi: 10.14309/ajg.0000000000000428. [DOI] [PubMed] [Google Scholar]
- 13.Lee SW, et al. Comparison of tenofovir and entecavir on the risk of hepatocellular carcinoma and mortality in treatment-naïve patients with chronic hepatitis B in Korea: A large-scale, propensity score analysis. Gut. 2020;69:1301–1308. doi: 10.1136/gutjnl-2019-318947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bridgewater J, et al. Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J. Hepatol. 2014;60:1268–1289. doi: 10.1016/j.jhep.2014.01.021. [DOI] [PubMed] [Google Scholar]
- 15.Chang KY, Chang JY, Yen Y. Increasing incidence of intrahepatic cholangiocarcinoma and its relationship to chronic viral hepatitis. J. Natl. Compr. Cancer Netw. 2009;7:423–427. doi: 10.6004/jnccn.2009.0030. [DOI] [PubMed] [Google Scholar]
- 16.Lee TY, et al. Hepatitis B virus infection and intrahepatic cholangiocarcinoma in Korea: A case–control study. Am. J. Gastroenterol. 2008;103:1716–1720. doi: 10.1111/j.1572-0241.2008.01796.x. [DOI] [PubMed] [Google Scholar]
- 17.Fujita T. An unusual risk factor for intrahepatic cholangiocarcinoma. J. Hepatol. 2012;57:1396–1397. doi: 10.1016/j.jhep.2012.07.021. [DOI] [PubMed] [Google Scholar]
- 18.Palmer WC, Patel T. Are common factors involved in the pathogenesis of primary liver cancers? A meta-analysis of risk factors for intrahepatic cholangiocarcinoma. J. Hepatol. 2012;57:69–76. doi: 10.1016/j.jhep.2012.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ahn CS, et al. Prognostic impact of hepatitis B virus infection in patients with intrahepatic cholangiocarcinoma. ANZ J. Surg. 2018;88:212–217. doi: 10.1111/ans.13753. [DOI] [PubMed] [Google Scholar]
- 20.Lei Z, et al. Antiviral therapy improves survival in patients with HBV infection and intrahepatic cholangiocarcinoma undergoing liver resection. J. Hepatol. 2018;68:655–662. doi: 10.1016/j.jhep.2017.11.015. [DOI] [PubMed] [Google Scholar]
- 21.Tsai MS, et al. Chang Gung Research Database: A multi-institutional database consisting of original medical records. Biomed. J. 2017;40:263–269. doi: 10.1016/j.bj.2017.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.WHO Collaborating Center for Drugs Statistics Methodology. ATC Index with DDDs 2003. (WHO, Oslo, 2003).
- 23.Lin DY, et al. Ultrasonographic changes of early liver cirrhosis in chronic hepatitis B: A longitudinal study. J. Clin. Ultrasound. 1993;21:303–308. doi: 10.1002/jcu.1870210502. [DOI] [PubMed] [Google Scholar]
- 24.Hung CH, et al. Correlation between ultrasonographic and pathologic diagnoses of hepatitis B and C virus-related cirrhosis. J. Gastroenterol. 2003;38:153–157. doi: 10.1007/s005350300025. [DOI] [PubMed] [Google Scholar]
- 25.Bruix J, Sherman M. Practice Guidelines Committee, American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236. doi: 10.1002/hep.20933. [DOI] [PubMed] [Google Scholar]
- 26.Terrault NA, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology. 2018;67:1560–1599. doi: 10.1002/hep.29800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.European Association for the Study of the Liver EASL 2017 clinical practice guidelines on the management of hepatitis B virus infection. J. Hepatol. 2017;67:370–398. doi: 10.1016/j.jhep.2017.03.021. [DOI] [PubMed] [Google Scholar]
- 28.Chen CH, et al. Entecavir vs. lamivudine in chronic hepatitis B patients with severe acute exacerbation and hepatic decompensation. J. Hepatol. 2014;60:127–134. doi: 10.1016/j.jhep.2013.08.024. [DOI] [PubMed] [Google Scholar]
- 29.Chen YC, Peng CY, Jeng WJ, Chien RN, Liaw YF. Clinical outcomes after interruption of entecavir therapy in HBeAg-negative chronic hepatitis B patients with compensated cirrhosis. Aliment Pharmacol. Ther. 2015;42:1182–1191. doi: 10.1111/apt.13409. [DOI] [PubMed] [Google Scholar]
- 30.Tsai MC, et al. Long-term outcomes of hepatitis B virus-related cirrhosis treated with nucleos(t)ide analogs. J. Formos Med. Assoc. 2017;116:512–521. doi: 10.1016/j.jfma.2016.08.006. [DOI] [PubMed] [Google Scholar]
- 31.US Food and Drug Administration. NDA Review pharmacology/toxicology review and evaluation: NDA No. 21-797: Submitted September 30, 2004. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2005/21797_BARACLUDE_pharmr.PDF. Accessed August 21 (2018).
- 32.Wilkens L, et al. Induction of aneuploidy by increasing chromosomal instability during dedifferentiation of hepatocellular carcinoma. Proc. Natl. Acad. Sci. U.S.A. 2004;101:1309–1314. doi: 10.1073/pnas.0305817101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wiemann SU, et al. Hepatocyte telomere shortening and senescence are general markers of human liver cirrhosis. FASEB J. 2002;16:935–942. doi: 10.1096/fj.01-0977com. [DOI] [PubMed] [Google Scholar]
- 34.Murata K, et al. Induction of IFN-lambda3 as an additional effect of nucleotide, not nucleoside, analogues: A new potential target for HBV infection. Gut. 2018;67:362–371. doi: 10.1136/gutjnl-2016-312653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Abushahba W, et al. Antitumor activity of type I and type III interferons in BNL hepatoma model. Cancer Immunol. Immunother. 2010;59:1059–1071. doi: 10.1007/s00262-010-0831-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li M, et al. Hepatitis B virus infection increases the risk of cholangiocarcinoma: A meta-analysis and systematic review. J. Gastroenterol. Hepatol. 2012;27:1561–1568. doi: 10.1111/j.1440-1746.2012.07207.x. [DOI] [PubMed] [Google Scholar]
- 37.Zhou Y, et al. Hepatitis viruses infection and risk of intrahepatic cholangiocarcinoma: Evidence from a meta-analysis. BMC Cancer. 2012;12:289. doi: 10.1186/1471-2407-12-289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tanaka M, et al. Risk factors for intrahepatic cholangiocarcinoma: A possible role of hepatitis B virus. J. Viral Hepat. 2010;17:742–748. doi: 10.1111/j.1365-2893.2009.01243.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fwu CW, et al. Hepatitis B virus infection and risk of intrahepatic cholangiocarcinoma and non-Hodgkin lymphoma: A cohort study of parous women in Taiwan. Hepatology. 2011;53:1217–1225. doi: 10.1002/hep.24150. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


