Abstract
Background/Aim: The visualization of hepatic segments with indocyanine green (ICG) fluorescence can aid in anatomic liver resection. The present study aimed to develop a method to specifically label an hepatic segment in a nude mouse model with liver metastasis. Materials and Methods: An orthotopic mouse model was established by surgical orthotopic implantation (SOI) of a patient-derived colon-cancer liver metastasis in the left lobe of the liver. Three weeks after SOI, the left Glissonean pedicle was ligated and 10 μg ICG was administrated intravenously. Images were obtained with the Pearl Trilogy Imaging System. Results: All mice expressed an 800 nm signal from ICG on the right lobe of the liver. The left lobe of the liver, in which the tumor was located, showed no fluorescence and had ischemia due to successful ligation of the Glissonean pedicle. Conclusion: The ligation of the Glissonean pedicle enables specific liver-segment labeling with ICG, which has potential clinical application for liver metastasectomy.
Keywords: Indocyanine green, liver, hepatic segment, labeling, liver metastasis, patient-derived orthotopic xenographt, PDOX, Glissonean pedicle, ligation, imaging, nude mouse
Liver resection is the mainstay curative treatment of liver tumors (1,2). Anatomic liver resection is recommended for the treatment of hepatocellular carcinoma (3). Resectability is determined by the number and location of tumors in the liver. Accurate visualization of the liver segmental anatomy at the time of tumor resection is critical for a successful procedure (4).
Indocyanine green (ICG) and a near-infrared camera system are effective for tumor visualization in the clinical setting during resection of liver tumors and metastases (5). ICG enters hepatocytes through a transporter expressed on the sinusoidal membrane of the hepatocytes. ICG accumulates before excretion into bile canaliculi (6).
Hepatic segments can also be specifically labelled by intravenous ICG injection after blocking the blood flow of the cancer-bearing segment (7,8) by ligation of the Glissonean pedicle (9). Surgeons can then perform anatomic liver resection of the non-fluorescent area containing the tumor.
The present study applies the ligation technique to specifically label a liver segment in a patient-derived orthotopic xenograft (PDOX) nude-mouse liver-metatasis model, for anatomic liver resection.
Materials and Methods
Animals. Nude (nu/nu) mice, age 4-6 weeks purchased from Jackson Lab (Bar Harbor, ME, USA), were used for this study. The animals were fed an autoclaved laboratory diet. All surgical procedures were performed with anesthesia by intra-muscular injection of ketamine, xylazine and acepromazine reconstituted in phosphate-buffered saline (PBS). Mice were treated with buprenorphine for pain control after surgical procedures. At the conclusion of the study, mice were euthanized with CO2 inhalation, which was confirmed with cervical dislocation. All studies were approved by the San Diego Veterans Administration Medical Center Institutional Animal Care and Use Committee (IACUC), animal -use protocol A17-020 (10).
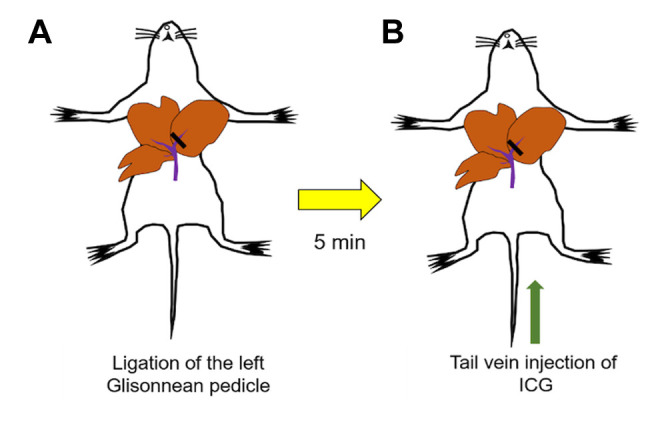
Ligation of the left Glissonean pedicle of nude mice. A 15-mm incision was made vertically in the midline of the upper abdomen through the skin and peritoneum. The whole liver was exposed and set in the cranial-inversion position. The left Glissonean pedicle was isolated and ligated with 6-0 surgical sutures under magnification using an MVX10 microscope (Olympus, Tokyo, Japan). The liver was gently returned to the peritoneal cavity and the abdominal wall and skin were closed with 6-0 surgical sutures (Ethicon Inc., NJ, USA) (Figure 1A).
Figure 1. Experimental schema. (A) Isolation and ligation of the left Glissonean pedicle. (B) ICG (10 μg) was injected via the tail vein, resulting in labeling of the area with preserved blood flow in the liver.
Tumor establishment. A colon-cancer liver metastasis was obtained from UCSD Thornton Hospital under UCSD IRB protocol 140046 with informed patient consent. A portion of the tumor was placed in phosphate-buffered saline (PBS) on ice and transferred to the laboratory. Fresh tumor fragments were subcutaneously implanted in nude mice. Grown tumors were harvested and cut into small fragments.
Nude mice (n=4) were anesthetized by intra-muscular injection of 0.02 ml of the ketamine solution. The abdomen was prepped with 70% ethanol solution. A 10-mm incision was made vertically in the midline of the upper abdomen through the skin and peritoneum. The left lobe of the liver was exposed and a 1 mm incision was made through the liver parenchyma. Surgical orthotopic implantation (SOI) was performed by implanting a single 1 mm3 tumor fragment in the left lobe of the liver parenchyma. After hemostasis was achieved, the liver was gently returned to the peritoneal cavity, and the abdominal wall and skin were closed with 6-0 surgical sutures.
Administration of ICG. Three weeks after SOI, a 15 mm incision was made vertically in the midline of the upper abdomen through the skin and peritoneum. The whole liver was exposed to ensure adequate establishment of the tumor in the liver parenchyma. The left Glissonean pedicle was isolated and ligated with 6-0 surgical sutures as described above (Figure 1A). Five minutes after isolation and ligation of the Glissonean pedicle, 10 μg of ICG was administered via tail vein injection (Figure 1B).
In vivo imaging. The Pearl Trilogy Small Animal Fluorescence Imaging System (LI-COR, Lincoln. NE, USA) was used. The Pearl Trilogy is equipped for sensitive imaging of 700- and 800- nanometer near-infrared fluorophores. Mice were euthanized prior to imaging. Laparotomy was then performed to expose the liver for imaging.
Results
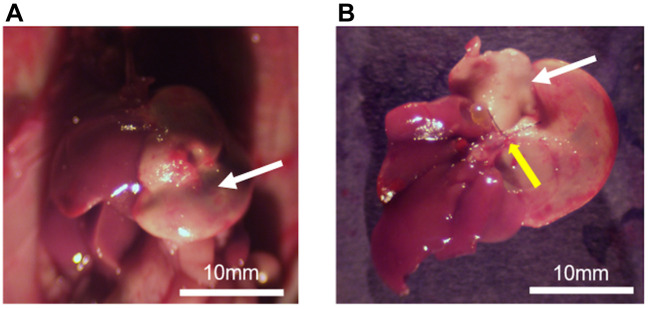
Liver ischemia induced by ligation of the Glissonean pedicle. Three days after left-Glissonean-pedicle ligation in the liver of the nude mice, laparotomy was performed to observe the liver. White areas in the left lobe of the liver indicated necrosis due to ischemia, (Figure 2A). Figure 2B shows the resected whole-liver specimen with the left Glissonean-pedicle ligated, indicated by the yellow arrow.
Figure 2. Liver ischemia caused by ligation of the Glissonean pedicle. (A) Three days after left-Glissonean-pedicle ligation, necrosis occurred due to ischemia (white arrow). (B) Entire liver after removal. Left Glissonean pedicle ligation is depicted by the yellow arrow. Necrosis is indicated by the white arrow.
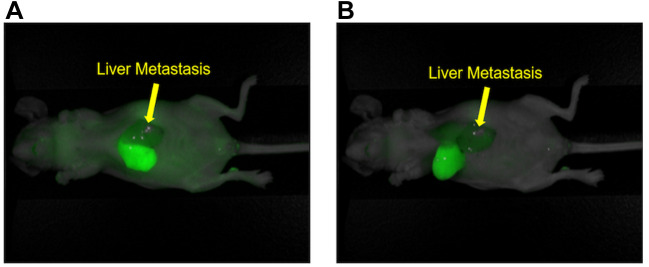
ICG specifically labels the right lobe of the liver and not the metastasis-bearing left lobe due to Glissonean-pedicle ligation. Images were then acquired under anesthesia with the Pearl Trilogy Small Animal Imaging System five minutes after ICG administration via the tail vein. All mice (n=4) showed an 800 nm signal from ICG on the right lobe of the liver. The left lobe of the liver, which contained the colon-cancer liver metastasis, showed no fluorescence (Figure 3A). Figure 3B shows cranial inversion of the liver, which also demonstrated distinct demarcation of the liver segment with preserved blood flow.
Figure 3. Fluorescence image of the ICG-labeled tumor segment. (A) Five minutes after indocyanine green injection (10 μg) via the tail vein, the 800- nm signal was observed on the right lobe of the liver due to blockage of blood flow into the left lobe of the liver due to Glissonean pedicle-ligation. Liver metastasis is indicated by the arrow. (B) Cranial inversion of the liver in the same mouse as Figure 3A. Arrow: liver-metastatic tumor.
Discussion
This is the first study describing specific hepatic-segment labeling using ICG in a mouse model of colon-cancer liver metastases. In the present study, we demonstrated that fluorescence labeling using ICG clearly visualized the liver segment in which the tumor is not located. Necrosis was observed in the ligated segments of the liver, indicating successful ligation of the Glissonean pedicle. A left lobectomy for tumor resection on the basis of the fluorescence boundary will be performed in future experiments. The major importance of left lobectomy is for metastasectomy, which will be more accurate due to specific ICG labelling, as described in the present study.
In clinical practice, there are two main means of administering ICG: positive and negative staining-methods (7). In the present study, we selected the negative-staining method for hepatic segmentation with ICG. We used Glissonean-pedicle ligation for the negative-staining method.
Primary and metastatic hepatic tumors can be intra-operatively detected by ICG fluorescence imaging after pre-operative ICG injection (11). Recently, a small colon-cancer liver metastasis was identified using ICG fluorescence in an orthotopic nude-mouse model (12). However, ICG is not tumor specific (13). The potential disadvantage is non-specific labelling of adjacent tissue, which can be reduced following the technique of the present study. Differentiation between the tumor and liver segments using ICG in combination with other fluorescent agents, such as tumor-specific antibodies, may improve differentiation of tumor from the surrounding liver segment and will be tested in future studies. The major advantage of ICG accumulation in hepatic tumors such as liver metastasis and hepatocellular carcinoma is to secure surgical margins of the liver tumors (14).
Hepatic-segment labeling using ICG in mouse models is a promising method since this method can also similarly visualize liver segments in the clinical setting. In conclusion, the visualization of an hepatic segment using ICG fluorescence in orthotopic mouse models after Glissonean-pedicle ligation has potential for more accurate demarcation of tumor margins in the liver.
Conflicts of Interest
There are no financial or other interests regarding the submitted manuscript that might be construed as a conflict of interest.
Authors’ Contributions
H.N., Y.T., J.Y. and M.B. designed the experiments. H.N., H.M.H., S.A., and F.F. were involved in the acquisition of the data. N.S. and R.M.H. gave technical support and conceptual advice. H.N. and H.M.H. were involved in drafting of the manuscript. R.M.H. revised the manuscript. All Authors were involved in editing and approval of the final manuscript.
Acknowledgements
This study was funded by VA Merit Review grant numbers 1 I01 BX003856-01A1 and 1 I01 BX004494-01 (MB) and NIH/NCI T32CA121938 (HH).
References
- 1.Benson AB 3rd, Abrams TA, Ben-Josef E, Bloomston PM, Botha JF, Clary BM, Covey A, Curley SA, D'Angelica MI, Davila R, Ensminger WD, Gibbs JF, Laheru D, Malafa MP, Marrero J, Meranze SG, Mulvihill SJ, Park JO, Posey JA, Sachdev J, Salem R, Sigurdson ER, Sofocleous C, Vauthey JN, Venook AP, Goff LW, Yen Y, Zhu AX. NCCN clinical practice guidelines in oncology: hepatobiliary cancers. J Natl Compr Canc Netw. 2009;7(4):350–391. doi: 10.6004/jnccn.2009.0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Benson AB 3rd, Bekaii-Saab T, Chan E, Chen YJ, Choti MA, Cooper HS, Engstrom PF, Enzinger PC, Fakih MG, Fenton MJ, Fuchs CS, Grem JL, Hunt S, Kamel A, Leong LA, Lin E, May KS, Mulcahy MF, Murphy K, Rohren E, Ryan DP, Saltz L, Sharma S, Shibata D, Skibber JM, Small W Jr., Sofocleous CT, Venook AP, Willett CG, Gregory KM, Freedman-Cass DA, National Comprehensive Cancer Network Metastatic colon cancer, version 3.2013: featured updates to the NCCN guidelines. J Natl Compr Canc Netw. 2013;11(2):141–152. doi: 10.6004/jnccn.2013.0022. [DOI] [PubMed] [Google Scholar]
- 3.Hasegawa K, Kokudo N, Imamura H, Matsuyama Y, Aoki T, Minagawa M, Sano K, Sugawara Y, Takayama T, Makuuchi M. Prognostic Impact of anatomic resection for hepatocellular carcinoma. Ann Surg. 2005;242(2):252–259. doi: 10.1097/01.sla.0000171307.37401.db. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Majno P, Mentha G, Toso C, Morel P, Peitgen HO, Fasel JH. Anatomy of the liver: an outline with three levels of complexity – a further step towards tailored territorial liver resections. J Hepatol. 2014;60(3):654–662. doi: 10.1016/j.jhep.2013.10.026. [DOI] [PubMed] [Google Scholar]
- 5.Aoki T, Yasuda D, Shimizu Y, Odaira M, Niiya T, Kusano T, Mitamura K, Hayashi K, Murai N, Koizumi T, Kato H, Enami Y, Miwa M, Kusano M. Image-guided liver mapping using fluorescence navigation system with indocyanine green for anatomical hepatic resection. World J Surg. 2008;32(8):1763–1767. doi: 10.1007/s00268-008-9620-y. [DOI] [PubMed] [Google Scholar]
- 6.Cusin F, Fernandes Azevedo L, Bonnaventure P, Desmeules J, Daali Y, Pastor CM. Hepatocyte concentrations of indocyanine green reflect transfer rates across membrane transporters. Basic Clin Pharmacol Toxicol. 2017;120(2):171–178. doi: 10.1111/bcpt.12671. [DOI] [PubMed] [Google Scholar]
- 7.Ishizawa T, Zuker NB, Kokudo N, Gayet B. Positive and negative staining of hepatic segments by use of fluorescent imaging techniques during laparoscopic hepatectomy. Arch Surg. 2012;147(4):393–394. doi: 10.1001/archsurg.2012.59.. [DOI] [PubMed] [Google Scholar]
- 8.Inoue Y, Arita J, Sakamoto T, Ono Y, Takahashi M, Takahashi Y, Kokudo N, Saiura A. Anatomical liver resections guided by 3-dimensional parenchymal staining using fusion indocyanine green fluorescence imaging. Ann Surg. 2015;262(1):105–111. doi: 10.1097/SLA.0000000000000775. [DOI] [PubMed] [Google Scholar]
- 9.Takasaki K. Glissonean pedicle transection method for hepatic resection: a new concept of liver segmentation. J Hepatobiliary Pancreat Surg. 1998;5(3):286–291. doi: 10.1007/s005340050047. [DOI] [PubMed] [Google Scholar]
- 10.Yang M, Reynoso J, Bouvet M, Hoffman RM. A transgenic red fluorescent protein-expressing nude mouse for color-coded imaging of the tumor microenvironment. J Cell Biochem. 2009;106(2):279–284. doi: 10.1002/jcb.21999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ishizawa T, Fukushima N, Shibahara J, Masuda K, Tamura S, Aoki T, Hasegawa K, Beck Y, Fukayama M, Kokudo N. Real-time identification of liver cancers by using indocyanine green fluorescent imaging. Cancer. 2009;115(11):2491–2504. doi: 10.1002/cncr.24291. [DOI] [PubMed] [Google Scholar]
- 12.Tashiro Y, Hollandsworth HM, Nishino H, Yamamoto J, Amirfakhri S, Filemoni F, Sugisawa N, Aoki T, Murakami M, Hoffman RM, Bouvet M. Indocyanine green labels orthotopic nude mouse models of very early colon cancer liver metastases. In Vivo. 2020;34(5):2277–2280. doi: 10.21873/invivo.12038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miyata Y, Ishizawa T, Kamiya M, Yamashita S, Hasegawa K, Ushiku A, Shibahara J, Fukayama M, Urano Y, Kokudo N. Intraoperative imaging of hepatic cancers using γ-glutamyl-transpeptidase-specific fluorophore enabling real-time identification and estimation of recurrence. Sci Rep. 2017;7(1):3542. doi: 10.1038/s41598-017-03760-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tashiro Y, Aoki T, Hirai T, Koizumi T, Mansou DA, Kusano T, Matsuda K, Yamada K, Nogaki K, Hakozaki T, Wada Y, Shibata H, Kodai T, Yamazaki T, Saito K, Fujimori A, Enami Y, Hoffman RM, Murakami M. Pathological validity of using near-infrared fluorescence imaging for securing surgical margins during liver resection. Anticancer Res. 2020;40(7):3873–3882. doi: 10.21873/anticanres.14377. [DOI] [PubMed] [Google Scholar]