Abstract
Background: The present study aimed to investigate and compare mental health, health-related quality of life, and sleep levels of patients with various stages of chronic kidney disease (CKD) and undergoing different renal replacement therapies and analyze the factors affecting these parameters.
Methods: Overall, 140 patients with a mean age of 43 ± 14 years were recruited into this study. Study groups [controls and patients with CKD undergoing predialysis, hemodialysis (HD), peritoneal dialysis, kidney transplantation (KT)] were evaluated using Short Form Health Survey-36 (SF-36), Kidney Disease Quality of Life-36 (KDQoL-36), Pittsburgh Sleep Quality Index (PSQI), and General Health Questionnaire-12 (GHQ-12).
Results: The KT group had the highest scores in physical and mental components of the subscales of SF-36 and KDQoL-36 but the lowest scores in PSQI and GHQ-12, indicating the best results in terms of mental health and quality of life, and sleep. Serum albumin and hemoglobin levels were positively correlated with several subscales of quality of life. Significant negative correlations were observed among PSQI, GHQ-12, and subscale scores of SF-36 and KDQoL-36. The HD group showed significantly lower scores in the subscales of symptoms and burden of kidney disease of KDQoL-36.
Conclusion: KDQoL was worse in the HD group and better in the KT group than in other groups. Serum albumin and hemoglobin levels, and Kt/V (dialyzer clearance of urea multiplied by dialysis time and normalized for urea distribution volume ) values of patients with CKD exerted a linear and significant effect on the quality of life, which showed a significant positive correlation with the quality of sleep and mental health. In contrast, serum calcium x phosphorus levels showed an inverse correlation with the subscale scores of KDQoL. HIPPOKRATIA 2020, 24(2): 51-58.
Keywords: Hemodialysis, mental health, peritoneal dialysis, predialysis, kidney transplantation, quality of life, quality of sleep
Introduction
Health-related quality of life (HRQoL) is defined as the extent to which an individual’s routine or expected physical, emotional, and social well-being are affected by a medical condition or its treatment. Thus, HRQoL is an important factor for all types of treatment outcomes and can differ according to illnesses and cultures1. Chronic kidney disease (CKD), which is defined as an irreversible loss of kidney function, affects mental health and quality of sleep, and patients’ lives worldwide. The underlying etiologies for the negative impact of CKD on the HRQoL of these patients include malnutrition, deteriorated cognitive function, sleep disorders, and depressive tendency2. In addition to CKD itself, the treatment modalities of CKD, including hemodialysis (HD), peritoneal dialysis (PD), and kidney transplantation (KT), can have different effects on the daily life of patients3.
There is extensive research from various countries investigating the HRQoL of patients with CKD in the literature. These studies have reported decreases in HRQoL with decreases in glomerular filtration rate (GFR) and CKD progression. However, to the best of our knowledge, the present study is the first to evaluate all parameters related to HRQoL, including mental health, quality of sleep, and laboratory parameters in patients undergoing predialysis, HD, PD, and KT together and with comparison to healthy controls. Therefore, our aim in this study was to investigate both the factors affecting mental health and quality of life and sleep of patients with CKD as well as the differences in renal replacement therapies concerning these parameters.
Material and methods
The present study was conducted with the cooperation of the Psychiatry and Nephrology Departments from October 2016 to June 2017. Our clinic’s regular follow-up patients were included based on the nephrologist’s and internist’s general medical examinations. Pediatric patients and those with acute kidney disease, history of mental illness, active infection, advanced heart failure, history of hospitalization in the past month, and failure to obtain informed consent were excluded from the study. All participants were aware of the study design and signed an informed consent form after reading the document. The study protocol was approved by the Ethics Committee of the Faculty of Medicine, Karadeniz Technical University (24237859-342; 2016/61). The study was conducted in accordance with the declaration of Helsinki.
Study Groups
Overall, 140 patients (48.6 % females) with a mean age of 43 ± 14 years were recruited. The patients were categorized into the five groups4. Group 1, defined as the control group, consisted of 30 healthy individuals who were healthcare workers in our hospital, with a mean serum creatinine level of 0.73 ± 0.18 mg/dL. Group 2 (CKD group) consisted of 30 patients with stage 4 CKD with a mean serum creatinine level of 3.66 ± 1.49 mg/dL and a mean GFR level of 18.07 ± 6.61 mL/min/1.73m2). Group 3 (HD group) consisted of 30 patients undergoing chronic HD program for 32 ± 31 months). Group 4 (PD group) consisted of 20 patients on chronic PD program for 57 ± 38 months (by that time, the number of PD patients with regular follow-up by our clinic had decreased), and Group 5 (KT group) consisted of 30 patients with functioning kidney allografts, whose mean lifespan was 60 ± 71 months, with a mean serum creatinine level of 1.19 ± 0.46 mg/dL.
Questionnaires
1. Short Form Health Survey-36 (SF-36) is a self-report questionnaire that evaluates eight aspects of quality of life [physical functioning (PF), physical role functioning (RP), bodily pain (BP), general health (GH), vitality (VT), social functioning (SF), emotional role functioning (RE), and mental health (MH)] with 36 items5. Scores of the groups were rated using the Likert method, with 0 and 100 being assigned to the lowest and highest values, respectively; higher scores indicated a better outcome. The reliability and validity of SF-36 for the Turkish population have been demonstrated by Koçyiğit et al6. This survey was applied to all study groups.
2. Kidney Disease Quality of Life-36 (KDQoL-36) is a self-report questionnaire that has two primary components: a generic core [which has two domains: physical (PCS) and mental (MCS) components] and a disease-specific core (which has three domains: the burden of kidney disease, symptoms, and effects of kidney disease), with scores ranging from 0 to 100 according to the Likert method7. Higher scores indicate better states of HRQoL and vice versa. The validity and reliability of KDQoL-36 for the Turkish population have been confirmed by Yıldırım et al1. This survey was applied only to CKD patients (Groups 2-5, except the control group).
3. The Pittsburgh Sleep Quality Index (PSQI) is another self-rated questionnaire designed to measure sleep quality in clinical conditions during the preceding one month8. A total of 19 items for assessing individual subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, sleeping medication use, and daytime dysfunction are scored. A sum of the scores ≥5 indicates poor sleepers. The reliability and validity of PSQI for the Turkish population have been demonstrated by Ağargün et al9. This survey was applied to all study groups.
4. The General Health Questionnaire-12 (GHQ-12), a self-replied questionnaire comprising 12 items evaluating anxiety and depression, was applied to all study groups. A sum of scores ≥2 indicates a high risk for mental disorder10. The reliability and validity of GHQ-12 for the Turkish population have been shown by Kılıç et al11.
The scores obtained in all these surveys for all the study groups were evaluated by a clinical psychiatrist.
Laboratory parameters
The mean of at least three values of blood urea nitrogen (BUN), hemoglobin (Hgb), hematocrit (Hct), calcium (Ca), phosphorus (P), C-reactive protein (CRP), albumin (Alb), creatinine (Cre), and low-density lipoprotein-cholesterol (LDL-C) levels (measured by Beckman Coulter analyzer; Beckman Coulter Inc., Brea, CA, USA) of the study groups was recorded from the computer data.
Statistical analysis
Statistical analyses were conducted using the IBM SPSS Statistics for Windows, Version 23.0. (IBM Corp., Armonk, NY, USA). Variables were checked by the Kolmogorov-Smirnov analytic method to determine whether data were normally distributed. Descriptive results are presented using means and standard deviation for the non-normally distributed and ordinal variables. When normal distribution was not observed, the Mann-Whitney U, Kruskal-Wallis, and chi-square tests were conducted to assess the significance of pairwise differences using the Bonferroni correction to adjust for multiple comparisons. Correlation coefficients and their significance were calculated using Spearman’s rank correlation coefficient. A p-value of <0.05 was considered to reveal significance.
Results
Baseline characteristics of participants
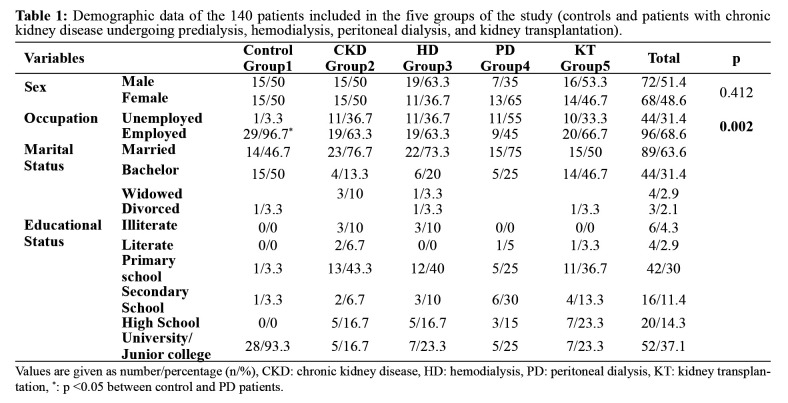
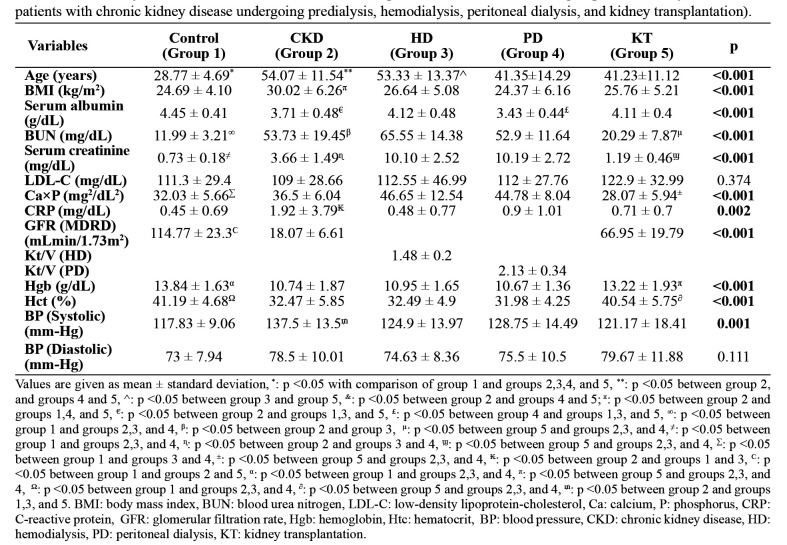
Of the 140 participants, 51.4 % were males, and there was no difference between the groups in terms of sex and marital status (Table 1). The educational status of the study groups is also shown in Table 1. Hypertension was the leading cause of CKD in all the patient groups, with frequencies of 36.7 %, 33.3 %, 25 %, and 43.3 % in groups 2, 3, 4, and 5, respectively. The frequencies of smoking history were 6.7 %, 10 %, 6.7 %, 20 %, and 3.3 % in groups 1, 2, 3, 4, and 5, respectively and those of social alcohol drinking were 10 %, 0 %, 6.7 %, 0 %, and 3.3 %, respectively. The frequencies of diabetes mellitus in groups 2, 3, 4, and 5 were 33.3 %, 26.7 %, 10 %, and 6.7 %, respectively. Serum albumin, BUN, creatinine, calcium x phosphorus, CRP, Hgb, and Hct levels, and blood pressure values showed significant differences among the study groups (Table 2).
Table 1. Demographic data of the 140 patients included in the five groups of the study (controls and patients with chronic kidney disease undergoing predialysis, hemodialysis, peritoneal dialysis, and kidney transplantation).
Values are given as number/percentage (n/%), CKD: chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplantation, *: p <0.05 between control and PD patients.
Table 2. Laboratory values and clinical features of the 140 patients included in the five groups of the study (controls and patients with chronic kidney disease undergoing predialysis, hemodialysis, peritoneal dialysis, and kidney transplantation).
Values are given as mean ± standard deviation, *: p <0.05 with comparison of group 1 and groups 2,3,4, and 5, **: p <0.05 between group 2, and groups 4 and 5, ^: p <0.05 between group 3 and group 5, &: p <0.05 between group 2 and groups 4 and 5; π: p <0.05 between group 2 and groups 1,4, and 5, €: p <0.05 between group 2 and groups 1,3, and 5, £: p <0.05 between group 4 and groups 1,3, and 5, ∞: p <0.05 between group 1 and groups 2,3, and 4, β: p <0.05 between group 2 and group 3, μ: p <0.05 between group 5 and groups 2,3, and 4, ≠: p <0.05 between group 1 and groups 2,3, and 4, ɳ: p <0.05 between group 2 and groups 3 and 4, ϣ: p <0.05 between group 5 and groups 2,3, and 4, Σ: p <0.05 between group 1 and groups 3 and 4, ±: p <0.05 between group 5 and groups 2,3, and 4, ₭: p <0.05 between group 2 and groups 1 and 3, Ϲ: p <0.05 between group 1 and groups 2 and 5, α: p <0.05 between group 1 and groups 2,3, and 4, π: p <0.05 between group 5 and groups 2,3, and 4, Ω: p <0.05 between group 1 and groups 2,3, and 4, ∂: p <0.05 between group 5 and groups 2,3, and 4, ₥: p <0.05 between group 2 and groups 1,3, and 5. BMI: body mass index, BUN: blood urea nitrogen, LDL-C: low-density lipoprotein-cholesterol, Ca: calcium, P: phosphorus, CRP: C-reactive protein, GFR: glomerular filtration rate, Hgb: hemoglobin, Htc: hematocrit, BP: blood pressure, CKD: chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplantation.
SF-36 scores of the participants
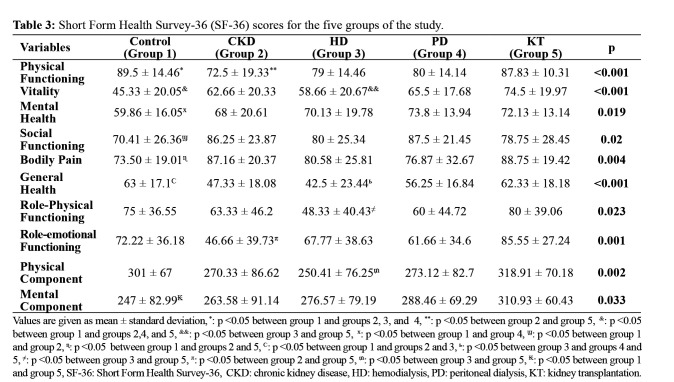
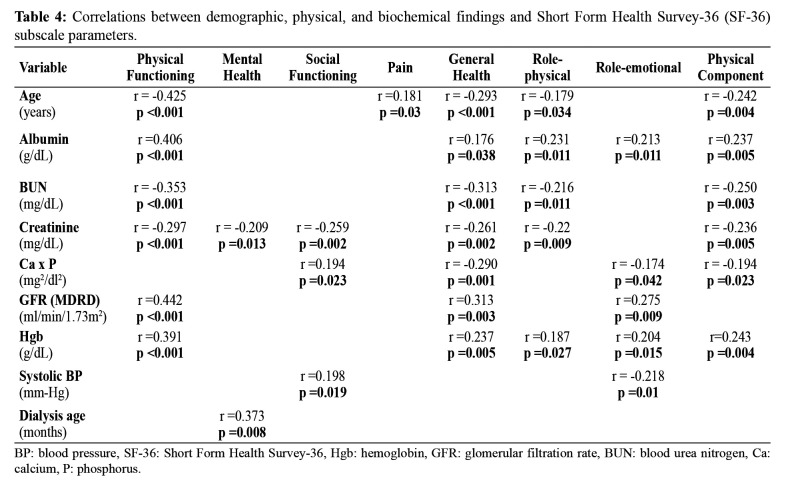
The CKD groups’ PF scores were significantly lower than those of the KT group, and the HD group’s VT scores were significantly lower than those of the KT group. GH scores of the HD group were significantly lower than those of the PD and KT groups, whereas the control group showed significantly higher GH scores than the CKD and HD groups. There was no difference in GH scores between the control and KT groups. The RP and RE scores were significantly higher in the KT group than in the HD and CKD groups (Table 3). Furthermore, among the study groups, the KT group showed the highest scores in the physical and mental components of the subscales of SF-36. Serum albumin and hemoglobin levels showed a positive correlation with several subscales of the quality of life (Table 4). Age showed a significant negative correlation with PF, GH, RP, and RE subscales. Age and pain scores showed a linear correlation. Significant negative correlations were observed between PSQI and GHQ-12 and the subscale scores of SF-36 and KDQoL-36. A significant negative correlation was observed between serum creatinine and BUN levels and SF, MH, GH, and RP subscales (Table 4). Serum calcium x phosphorus levels were negatively correlated with GH, RE, and RP scores but were positively correlated with SF. With a decrease in GFR level, the PF, GH, and RE scores also decreased.
Table 3. Short Form Health Survey-36 (SF-36) scores for the five groups of the study.
Values are given as mean ± standard deviation, *: p <0.05 between group 1 and groups 2, 3, and 4, **: p <0.05 between group 2 and group 5, &: p <0.05 between group 1 and groups 2,4, and 5, &&: p <0.05 between group 3 and group 5, x: p <0.05 between group 1 and group 4, ϣ: p <0.05 between group 1 and group 2, ɳ: p <0.05 between group 1 and groups 2 and 5, Ϲv: p <0.05 between group 1 and groups 2 and 3, ь: p <0.05 between group 3 and groups 4 and 5, ≠: p <0.05 between group 3 and group 5, π: p <0.05 between group 2 and group 5, ₥: p <0.05 between group 3 and group 5, ₭: p <0.05 between group 1 and group 5, SF-36: Short Form Health Survey-36, CKD: chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplantation.
Table 4. Correlations between demographic, physical, and biochemical findings and Short Form Health Survey-36 (SF-36) subscale parameters.
BP: blood pressure, SF-36: Short Form Health Survey-36, Hgb: hemoglobin, GFR: glomerular filtration rate, BUN: blood urea nitrogen, Ca: calcium, P: phosphorus.
KDQoL scores of the participants
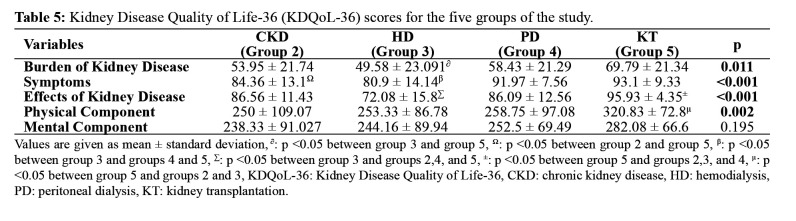
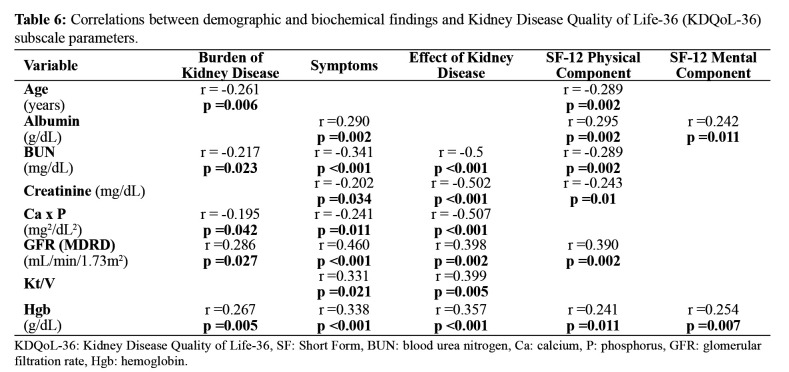
KDQoL-36 showed significantly lower scores in the subscales of symptoms and burden of kidney disease in the HD group than in other groups (Table 5). In other words, the HD group had the lowest perception of the quality of life. KDQoL scores disclosed that age had a significant negative impact on the burden of kidney disease and physical component (Table 6). Serum albumin level positively correlated with symptoms, physical component, and mental component scores, whereas GFR positively correlated with the burden of kidney disease, symptoms, effects of kidney disease, and physical component scores, and Hgb level positively correlated with all the subscales of KDQoL scores (Table 6). Serum creatinine, BUN, and calcium x phosphorus levels were negatively correlated with several subscales of KDQoL.
Table 5. Kidney Disease Quality of Life-36 (KDQoL-36) scores for the five groups of the study.
Values are given as mean ± standard deviation, ∂: p <0.05 between group 3 and group 5, Ω: p <0.05 between group 2 and group 5, β: p <0.05 between group 3 and groups 4 and 5, Σ: p <0.05 between group 3 and groups 2,4, and 5, ±: p <0.05 between group 5 and groups 2,3, and 4, μ: p <0.05 between group 5 and groups 2 and 3, KDQoL-36: Kidney Disease Quality of Life-36, CKD: chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplantation.
Table 6. Correlations between demographic and biochemical findings and Kidney Disease Quality of Life-36 (KDQoL-36) subscale parameters.
KDQoL-36: Kidney Disease Quality of Life-36, SF: Short Form, BUN: blood urea nitrogen, Ca: calcium, P: phosphorus, GFR: glomerular filtration rate, Hgb: hemoglobin.
PSQI scores of the participants
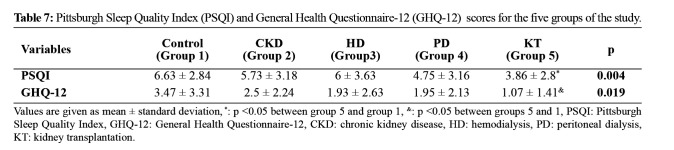
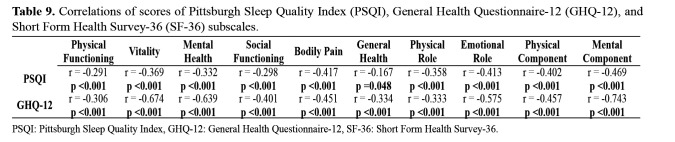
The KT group showed the lowest PSQI and GHQ-12 scores, indicating the best results among all study groups regarding mental health and quality of life, and sleep. Based on the cut-off point (5 points), the PSQI score percentages in the control, CKD, HD, PD, and KT groups were 80 %, 56.7 %, 60 %, 40 %, and 33.3 %, respectively (Table 7 and Table 8). There were significant differences between the control and KT groups. PSQI scores of the KT group were the lowest indicating the best quality of sleep among the study groups. Significant negative correlations were detected between PSQI and the subscale scores of SF-36 and KDQoL-36 (Table 9).
Table 7. Pittsburgh Sleep Quality Index (PSQI) and General Health Questionnaire-12 (GHQ-12) scores for the five groups of the study.
Values are given as mean ± standard deviation, *: p <0.05 between group 5 and group 1, &: p <0.05 between groups 5 and 1, PSQI: Pittsburgh Sleep Quality Index, GHQ-12: General Health Questionnaire-12, CKD: chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplantation.
Table 8. Pittsburgh Sleep Quality Index (PSQI) and General Health Questionnaire-12 (GHQ-12) score distribution according to cut-off points.
Values are given as number/percentage (n/%), *: p <0.05 between groups 1 and 5, **: p <0.05 between groups 1 and 5, PSQI: Pittsburgh Sleep Quality Index, GHQ-12: General Health Questionnaire-12, CKD: chronic kidney disease, HD: hemodialysis, PD: peritoneal dialysis, KT: kidney transplantation.
Table 9. Correlations of scores of Pittsburgh Sleep Quality Index (PSQI), General Health Questionnaire-12 (GHQ-12), and Short Form Health Survey-36 (SF-36) subscales.
PSQI: Pittsburgh Sleep Quality Index, GHQ-12: General Health Questionnaire-12, SF-36: Short Form Health Survey-36.
GHQ-12 scores of the participants
Scores >2 for GHQ-12 (cut-off point) were recorded in 63.3 %, 50 %, 36.7 %, 45 %, and 20 % of participants in the control, CKD, HD, PD, and KT groups, respectively. The KT group showed better scores than the other groups (Table 7 and Table 8). No significant difference was observed with respect to psychiatric disease among the groups. Significant negative correlations were observed between GHQ-12 and the subscale scores of SF-36 and KDQoL-36, whereas significant positive correlations were observed between GHQ-12 and PSQI.
Discussion
Worldwide, CKD affects patients’ morbidity and mortality, including their quality of life, by exerting a negative impact on their social and psychological wellness2,3,12,13. We aimed to investigate mental health and quality of sleep and life among patients with CKD followed-up at our center in the present study. First, our study groups were not homogeneous in terms of age and occupational status, which might have affected the quality of life in several aspects. Under this limitation, our study highlighted that the HD group had the lowest (worse) KDQoL-36 score, and the KT group had the highest (best) score (p =0.011). This finding is consistent with that reported by Cryzevski et al14, who also found the highest values among post-KT patients and lowest values among HD patients. In our study, the healthy control group had the highest scores for PF, but the lowest scores for VT, MH, SF, BP scales compared with those of patients with CKD (both dialysis and KT patients). This result differs from those of other studies. For instance, Reimer et al reported that KT patients and healthy controls had a similar quality of life in terms of psychological and physical aspects15. In our study, the healthy control group primarily consisted of assistants in our Internal Medicine Department. Their busy working schedules might have contributed to their low scores of mental component scales of the quality of life (SF-36).
Sleep is one of the primary requirements and has a significant impact on the quality of life. In our study, 60 % of HD patients had sleep disturbances (high scores on the PSQI scale), whereas 33 % of KT patients had low sleep quality. Brekke et al found that KT led to an improvement in the sleep quality of their study patients16. Eryılmaz et al reported a similar proportion of poor sleepers (30 %) among KT patients17. Also, Sung et al reported a relationship between sleep duration and HRQoL (7-h sleepers had the highest HRQoL scores) among 1,910 predialysis patients with CKD18. Zhenc et al revealed a prevalence of 67 % of sleep disorders among dialysis patients, which is similar to our result, and sleep disorders were closely associated with deterioration of quality of life19. Similarly, in the present study, we detected significant negative correlations between PSQI and GHQ-12 and the subscale scores of SF-36 and KDQoL-36. This result implies that a significant linear relationship exists between mental health and quality of sleep and life. Our study’s correlations revealed that better mental health scores are associated with better quality of life and quality of sleep scores.
Our study findings further demonstrated that as the kidney functions decrease and the need for dialysis emerges, both the physical and mental burden of kidney disease become worse and improve after successful KT. As a possible explanation for this outcome, we observed that participants’ serum albumin levels were positively correlated with PF, GH, RP, and RE subscales of SF-36. A similar association between high serum albumin levels and HRQoL has been reported in the literature20. Moreover, our study participants’ serum hemoglobin levels showed a significant and positive correlation with PF, GH, RP, RE, and physical component subscales of SF-36. Similarly, Chen et al reported that patients who did not achieve target serum albumin and hemoglobin levels showed poor HRQoL outcomes21. In contrast, age, serum calcium x phosphorus levels, and serum creatinine levels were negatively correlated with several subscales of SF-36. In the KDQoL-36 scale, we detected negative correlations of age, serum calcium x phosphorus levels, and serum creatinine levels and positive correlations of serum albumin, hemoglobin, and Kt/V values with symptoms, burden, and effects of kidney disease subscale scores. Age at dialysis showed a positive correlation with MH subscale scores of KDQoL-36 in our study. Based on these findings, we believe that the perception of sickness among dialysis patients changes favorably over time.
It has been well established that worsening of life quality increases the diagnostic and therapeutic costs, particularly among patients with CKD22. Moreover, these patients have a higher frequency of anxiety and depressive episodes leading to the initiation of early dialysis23. In our study, patients with CKD had lower RE scores than dialysis patients. We suggest that more attention focused on predialysis patients in terms of role functioning, as suggested earlier22,23. We observed that the subscales of quality of life directly impact the quality of sleep and mental health status. KT patients had the best quality of life subscale scores among all study groups. Recent research has reported that KT patients have less anxiety and depression, and dialysis patients have a higher frequency of anxiety and depression24. Nevertheless, we found no significant difference in terms of these parameters between the dialysis and KT groups. Biochemically, linear correlations were found among serum hemoglobin, albumin levels, and Kt/V values, whereas serum calcium x phosphorus showed an inverse correlation with KDQoL subscale scores. The ability to function and maintain well-being and psychological health is one of the goals recommended by the World Health Organization in several diseases. To achieve these goals among dialysis patients, we suggest optimum nutrition and dialysis dose, anemia management, and successful KT opportunity because KT is the best method in medical and economic terms and terms of mental health and quality of life and sleep. It is necessary to focus on this aspect for better management of patients with CKD.
A limitation of the current study is that the study groups were not homogeneous in terms of age and educational status. The healthy control group consisted of our hospital assistants and healthcare workers who were found to have more depressive disorders than patient groups. In particular, our study’s healthy control group consisting of our hospital healthcare workers, primarily resident doctors, and had lower VT, MH, and SF scores than the patient groups. This could be due to their stressful working conditions, as reported in the majority of countries worldwide25-29. Therefore, we believe that merely having a normal kidney function is not sufficient to have a satisfactory quality of life and sleep required for good mental health.
Conflict of interest
The authors declared no conflicts of interest with respect to the research, authorship, and publication of this article.
Acknowledgement
This study was reported in the abstract form at 35thNephrology, Hypertension, Dialysis and Transplantation Congress of the Turkish Society of Nephrology.
References
- 1.Yildirim A, Ogutmen B, Bektas G, Isci E, Mete M, Tolgay HI. Translation, cultural adaptation, initial reliability, and validation of the Kidney Disease and Quality of Life-Short Form (KDQOL-SF 1.3) in Turkey. Transplant Proc. 2007;39:51–54. doi: 10.1016/j.transproceed.2006.10.196. [DOI] [PubMed] [Google Scholar]
- 2.Afsar B, Siriopol D, Aslan G, Eren OC, Dagel T, Kilic U, et al. The impact of exercise on physical function, cardiovascular outcomes and quality of life in chronic kidney disease patients: a systematic review. Int Urol Nephrol. 2018;50:885–904. doi: 10.1007/s11255-018-1790-4. [DOI] [PubMed] [Google Scholar]
- 3.Barotfi S, Molnar MZ, Almasi C, Kovacs AZ, Remport A, Szeifert L, et al. Validation of the Kidney Disease Quality of Life-Short Form questionnaire in kidney transplant patients. J Psychosom Res. 2006;60:495–504. doi: 10.1016/j.jpsychores.2005.09.009. [DOI] [PubMed] [Google Scholar]
- 4.Günalay S, Öztürk YK, Akar H, Mergan H. The relationship between malnutrition and quality of life in hemodialysis and peritoneal dialysis patients. Rev Assoc Med Bras (1992) 2018;64:845–852. doi: 10.1590/1806-9282.64.09.845. [DOI] [PubMed] [Google Scholar]
- 5.Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30:473–483. [PubMed] [Google Scholar]
- 6.Koçyiğit H, Aydemir Ö, Fişek G, Memiş A. Kısa form-36 (KF-36)’nın Türkçe versiyonunun güvenilirliği ve geçerliliği: romatizmal hastalığı olan bir grup ile çalışma. İlaç ve Tedavi Dergisi. 1999;12:102–106. [Google Scholar]
- 7.Hays RD, Kallich JD, Mapes DL, Coons SJ, Carter WB. Development of the kidney disease quality of life (KDQOL) instrument. Qual Life Res. 1994;3:329–338. doi: 10.1007/BF00451725. [DOI] [PubMed] [Google Scholar]
- 8.Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 9.Ağargün MY, Kara H, Anlar Ö. Pittsburgh Uyku Kalitesi İndeksinin Geçerliği ve Güvenirliği. Türk Psikiyatri Dergisi. 1996;7:107–115. [Google Scholar]
- 10.Goldberg DP, Gater R, Sartorius N, Ustun TB, Piccinelli M, Gureje O, et al. The validity of two version of the GHQ in the WHO study of mental illness in general health care. Psychol Med. 1997;27:191–197. doi: 10.1017/s0033291796004242. [DOI] [PubMed] [Google Scholar]
- 11.Kılıç C. Genel Sağlık Anketi: Güvenilirlik ve Geçerlilik Çalışması. Türk Psikiyatri Dergisi. 1996;7:3–10. [Google Scholar]
- 12.Ng JK, Li PK. Chronic kidney disease epidemic: How do we deal with it? Nephrology (Carlton) 2018;23 Suppl 4:116–120. doi: 10.1111/nep.13464. [DOI] [PubMed] [Google Scholar]
- 13.Ghiasi B, Sarokhani D, Dehkordi AH, Sayehmiri K, Heidari MH. Quality of Life of patients with chronic kidney disease in Iran: Systemic Review and Meta-analysis. Indian J Palliat Care. 2018;24:104–111. doi: 10.4103/IJPC.IJPC_146_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Czyżewski L, Sańko-Resmer J, Wyzgał J, Kurowski A. Assessment of health-related quality of life of patients after kidney transplantation in comparison with hemodialysis and peritoneal dialysis. Ann Transplant. 2014;19:576–585. doi: 10.12659/AOT.891265. [DOI] [PubMed] [Google Scholar]
- 15.Reimer J, Franke GH, Lütkes P, Kohnle M, Gerken G, Philipp T, et al. [Quality of life in patients before and after kidney transplantation] Psychother Psychosom Med Psychol. 2002;52:16–23. doi: 10.1055/s-2002-19662. [DOI] [PubMed] [Google Scholar]
- 16.Brekke FB, Waldum-Grevbo B, von der Lippe N, Os I. The effect of renal transplantation on quality of sleep in former dialysis patients. Transpl Int. 2017;30:49–56. doi: 10.1111/tri.12866. [DOI] [PubMed] [Google Scholar]
- 17.Eryılmaz MM, Ozdemir C, Yurtman F, Cilli A, Karaman T. Quality of sleep and quality of life in renal transplantation patients. Transplant Proc. 2005;37:2072–2076. doi: 10.1016/j.transproceed.2005.03.084. [DOI] [PubMed] [Google Scholar]
- 18.Sung SA, Hyun YY, Lee KB, Park HC, Chung W, Kim YH, et al. Sleep Duration and Health-Related Quality of Life in Predialysis CKD. Clin J Am Soc Nephrol. 2018;13:858–865. doi: 10.2215/CJN.11351017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zheng C, Xu J, Chen C, Lin F, Shao R, Lin Z, et al. Effects of sleep disorders and sedative-hypnotic medications on health-related quality of life in dialysis patients. Int Urol Nephrol. 2019;51:163–174. doi: 10.1007/s11255-018-2018-3. [DOI] [PubMed] [Google Scholar]
- 20.Romano-Zelekha O, Golan E, Ifrah A, Weinstein T, Shohat T. Differences in quality of life between Jewish and Arab patients on hemodialysis. Qual Life Res. 2017;26:3343–3352. doi: 10.1007/s11136-017-1661-y. [DOI] [PubMed] [Google Scholar]
- 21.Chen JY, Wan EYF, Choi EPH, Chan AKC, Chan KHY, Tsang JPY, et al. The Health-Related Quality of Life of Chinese Patients on Hemodialysis and Peritoneal Dialysis. Patient. 2017;10:799–808. doi: 10.1007/s40271-017-0256-6. [DOI] [PubMed] [Google Scholar]
- 22.Karatas A, Canakci E, Turkmen E. Comparison of sleep quality and quality of life indexes with sociodemographic characteristics in patients with chronic kidney disease. Niger J Clin Pract. 2018;21:1461–1467. doi: 10.4103/njcp.njcp_146_18. [DOI] [PubMed] [Google Scholar]
- 23.Mosleh H, Alenezi M, Al Johani S, Alsani A, Fairaq G, Bedeiwi R. Prevalence and Factors of Anxiety and Depression in Chronic Kidney Disease Patients Undergoing Hemodialysis: A Cross-sectional Single-Center Study in Saudi Arabia. Cureus. 2020;12:e6668. doi: 10.7759/cureus.6668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brito DCS, Machado EL, Reis IA, Carmo LPFD, Cherchiglia ML. Depression and anxiety among patients undergoing dialysis and kidney transplantation: a cross-sectional study. Sao Paulo Med J. 2019;137:137–147. doi: 10.1590/1516-3180.2018.0272280119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Asante JO, Li MJ, Liao J, Huang YX, Hao YT. The relationship between psychosocial risk factors, bunout and quality of life among primary healthcare workers in rural Guangdong province: a cross-sectional study. BMC Health Serv Res. 2019;19:447. doi: 10.1186/s12913-019-4278-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Austin CL, Saylor R, Finley PJ. Moral distress in physicians and nurses: Impact on professional quality of life and turnover. Psychol Trauma. 2017;9:399–406. doi: 10.1037/tra0000201. [DOI] [PubMed] [Google Scholar]
- 27.Saitoh K, Furihata R, Kaneko Y, Suzuki M, Takahashi S, Uchiyama M. Association of serum BDNF levels and the BDNF Val66Met polymorphism with the sleep pattern in healthy young adults. PLoS One. 2018;13:e0199765. doi: 10.1371/journal.pone.0199765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yamuchi T, Sasaki T, Takahashi K, Umezaki S, Takahashi M, Yoshikawa T, et al. Long working hours, sleep-related problems, and near-misses/injuries in industrial settings using a nationally representative sample of workers in Japan. PLoS One. 2019;14:e0219657. doi: 10.1371/journal.pone.0219657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.İlçin N, Tomruk M, Yeşilyaprak SS, Karadibak D, Savcı S. The relationship between learning styles and academic performance in TURKISH physiotherapy students. BMC Med Educ. 2018;18:291. doi: 10.1186/s12909-018-1400-2. [DOI] [PMC free article] [PubMed] [Google Scholar]