Abstract
We describe a case of delayed onset, acute demyelinating neuropathy secondary to novel SARS-CoV-2 infection. A previously healthy 46-year-old man presented with bilateral leg pain and loss of sensation in his feet 53 days after having COVID-19 pneumonitis. He developed painful sensory symptoms followed by a rapidly progressive lower motor neuron weakness involving all limbs, face and respiratory muscles, needing ventilatory support. In keeping with a diagnosis of Guillain-Barré syndrome, cerebrospinal fluid examination showed albuminocytologic dissociation and nerve conduction studies supported the diagnosis of an acute inflammatory demyelinating polyradiculoneuropathy. The delayed neurological dysfunction seen in our patient following SARS-CoV-2 infection may indicate a novel mechanism of disease that is part of the emerging ‘long COVID-19 syndrome’.
Keywords: infectious diseases, neurology, immunology, COVID-19
Background
Throughout the COVID-19 pandemic, neurological complications have been reported, including cerebrovascular disease, seizures, meningitis, encephalitis, loss of smell (anosmia), taste (ageusia) and myositis.1 Peripheral nerve damage has been reported as mononeuropathy and a more generalised acute polyneuropathy, most often reported as Guillain-Barré syndrome (GBS).1–3 More recently, longer term complications ‘long COVID-19’ have been frequently described, including fatigue, cognitive problems (especially memory loss), persistent fevers, myalgia, muscle weakness and breathlessness (particularly on exertion).4–6 The pathogenesis of these ongoing problems has yet to be established.
GBS refers to a group of associated acute inflammatory neuropathies, most commonly caused by an immune reaction following exposure to certain viral or bacterial antigens. Two-thirds of GBS cases report a history of infection prior to symptom onset. They can be triggered by bacterial (Campylobacter jejuni, Mycoplasma sp) and viral infections (Cytomegalovirus, Ebstein-Barr virus, HIV, hepatitis B and C, Zika virus, influenza etc.).7 The most common form in the UK is acute inflammatory demyelinating polyradiculopathy (AIDP), often seen after gastrointestinal or respiratory infections.8
Case presentation
A previously healthy 46-year-old Caucasian man presented with a week’s history of lethargy and progressive neurological symptoms. Initially he experienced sensory loss in his feet, progressing to gait unsteadiness and distal lower limb weakness. He later developed severe bilateral leg pain (shooting and burning) alongside paraesthesia and clumsiness in his hands. There was no history of recent fever, night sweats or weight loss. There was no reported visual, swallowing, bowel or bladder dysfunction and no back or neck pain. He had a previous medical history of borderline hypertension (on no regular medication; BMI 29.5). There was no history of alcohol intake, cigarette smoking or recreational drug use. He worked as a project manager and lived with his wife and children who had all been well, with no history of viral illness or diarrhoea.
Fifty-three days earlier he had a 7-day admission with COVID-19 pneumonitis (confirmed by nasopharyngeal swab PCR) complicated by type 1 respiratory failure requiring non-invasive ventilation. He was enrolled into the RECOVERY Trial and received standard supportive care.9 Following discharge, he made a full recovery.
At the second presentation, cardiorespiratory and abdominal examination were normal. Neurological examination revealed normal speech and intact cranial nerves, in particular, no ophthalmoplegia or nystagmus and no facial nerve palsy. Lower limb examination revealed normal tone and muscle mass, bilateral distal weakness (3+/5 power in dorsi and plantar flexion of his feet, 3/5 in hallux flexion) with areflexia and an ataxic gait. There was bilateral loss of light touch and pinprick sensation in a stocking distribution. Proprioception was reduced in both legs with complete loss of joint position of both hallux and partial loss in both ankle joints. In his upper limbs, power was preserved but reflexes were symmetrically diminished. There was reduced light touch and pinprick sensation over the lateral aspect of the left upper limb and hand (C5/7 dermatomes).
Investigations
Cerebrospinal fluid (CSF) analysis (day 1 of admission) showed raised CSF total protein (table 1).
Table 1.
CSF results
| Results | Reference values | |
| CSF total protein | 1.27 | 0.15–0.45 g/L |
| CSF glucose | 4.0 | mmol/L |
| Cell count CSF | <2 WBC /μL | Bottle 1 and 3 |
| 189 RBC /μL | Bottle 1 | |
| 9 RBC /μL | Bottle 3 | |
| Microscopy | Organisms not seen | |
| CSF culture | Sterile after 48 hours incubation |
There is a clear albumino-cytologic dissociation.
CSF, cerebrospinal fluid; RBC, Red blood cells; WBC, White blood cells.
ECG showed sinus rhythm. Initial basic blood tests (full blood count, urea and electrolytes, liver, bone profile, C-reactive protein, B12, folate), CT brain and MRI cervical spine were unremarkable. Admission bedside spirometry showed a forced vital capacity (FVC) of 2.8 L (normal value for weight >1.9 L). Infection and vasculitis screens were negative (HIV, syphilis and lyme disease).
Peripheral nerve conduction studies were completed on day 1 of admission and supported a diagnosis of acute inflammatory demyelinating polyneuropathy (table 2).
Table 2.
Nerve conduction study results show a demyelinating polyneuropathy in keeping with AIDP.
| Nerve conduction study results | ||||
| Nerve | Latency (ms) | Amplitude (mV) | Conduction velocities (m/s) | F latency (ms) |
| Motor | ||||
| Medianus motor right | ||||
| Wrist—APB | 3.95 | 8.9 | 50 | 32 |
| Elbow—wrist | 8.9 | 8.5 | ||
| Ulnaris motor right | ||||
| Wrist—ADM | 2.9 | 7.0 | ||
| Below elbow—wrist | 14.0 | 6.3 | 52 | 29 |
| Tibialis motor right | ||||
| Ankle—abductor hallucis | 4.1 | 2 D* | Absent | |
| Tibialis motor left | ||||
| Ankle—abductor hallucis | 4.25 | 1.6 D* | ||
| Peroneus motor right | ||||
| Ankle—EDB | 13.3 | 2.7 | ||
| Fibular head—ankle | 21.7 | 2.6 | 46 | 78 |
| Peroneus motor left | ||||
| Ankle—EDB | 10.9 | 2.2 | ||
| Fibular head—ankle | 21.6 | 2.2 | 38 | |
| Sensory | ||||
| Medianus sensory right | ||||
| Dig II—wrist | 2.5 | 4.1 | 51 | |
| Dig III—wrist | 2.6 | 3.1 | 50 | |
| Ulnaris sensory right | ||||
| Dig V—wrist | 2.1 | 3.4 | 51 | |
| Radialis sensory right | ||||
| Forearm—first web space | 1.4 | 24 | 59 | |
| Peroneus superficialis sensory right | ||||
| Mid calf—anterior ankle | 2 | 6 | 44 | |
| Suralis sensory right | ||||
| Mid calf—anterior ankle | 1.9 | 5 | 51 | |
Abnormal responses shown in bold type.
ADM, abductor digiti minimi; APB, abductor pollicis brevis; D, dispersed; Dig, digit; EDB, extensor digitorum brevis.
Treatment and progress
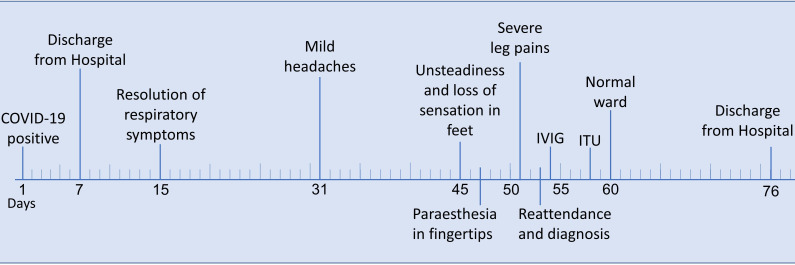
He was readmitted and treated with analgesia and intravenous immunoglobulins (IVIG) 2 g/kg divided over 5 days. His vital capacity was closely monitored. He developed progressive ascending weakness, including a facial droop, dysphagia and breathing difficulties (FVC of 1.2 L at day 5). He was admitted to the intensive care unit where he received non-invasive ventilation for type 1 respiratory failure for approximately 36 hours. His neuropathic pain remained difficult to control. As his condition improved, he was moved to a rehabilitation ward and managed by a multidisciplinary team. He recovered gradually and was discharged home, with ongoing community physiotherapy, after 23 days. The timeline of events is illustrated in figure 1.
Figure 1.
Timeline of events.
Outcome and follow-up
He has made a good functional recovery. Three months after his hospital discharge, he is able to walk independently, occasionally using a stick for longer distances. His main residual symptoms are intermittent neuropathic pain and paraesthesia in his distal limbs. He has stopped analgesia and continues to improve.
Follow-up nerve conduction studies at 5 months show improvement in nerve function, with normalising distal motor latencies. This indicates a return of distal myelin function.
Discussion
AIDP is the most common form of GBS. It classically presents as a rapidly evolving, ascending, symmetrical, flaccid paralysis with loss or diminished tendon reflexes. Peripheral sensory symptoms, including severe back pain, are common (80% of cases) and often precede muscle weakness. Diagnostic criteria to facilitate the largely clinical diagnosis of GBS are summarised in table 3.10 11
Table 3.
Modified Asbury Cornblath diagnostic criteria for GBS10 11
| Required features |
| Progressive motor weakness of more than one limb. |
| Areflexia or decreased reflexes in weak limbs. |
| Supportive features |
| Progressive phase lasts up to 4 weeks. |
| Recovery beginning 2–4 weeks after progression ceases. |
| Autonomic dysfunction. |
| Relative symmetry. |
| Cranial nerve involvement, especially bilateral facial nerve weakness. |
| Mild sensory symptoms or signs. |
| Pain. |
| No fever at the onset. |
| Typical features of cerebrospinal fluid and nerve conduction studies. |
| The following features make the diagnosis of GBS doubtful | Possible alternative diagnosis |
| Sensory level. | Cord compression/demyelination |
| Bowel and bladder dysfunction at onset. | Cord compression |
| Severe and persistent bowel and bladder dysfunction. | Cauda equina syndrome |
| Severe pulmonary dysfunction with little or no limb weakness at onset. | Myasthenia gravis |
| Severe sensory signs with little or no weakness at onset. | |
| Fever at onset. | |
| CSF pleocytosis with a white cell count >50/µL. | Meningitis |
CSF, cerebrospinal fluid; GBS, Guillain-Barré syndrome.
GBS is potentially life threatening and should therefore be monitored meticulously. Complications include respiratory failure, requiring intubation and ventilation (20%–30% of patients), bulbar dysfunction and aspiration and autonomic dysfunction leading to haemodynamic instability.11 Disease-modifying therapy comprises of high-dose IVIG or plasmapheresis.6 12 Eighty-five per cent of patients make a good functional recovery. Twenty per cent of patients experience long-term complications including severe disability, pain and fatigue; mortality is 5%.11 13
The aetiology of GBS is complex and most likely caused by an immune-mediated reaction secondary to a preceding infection (likely due to molecular mimicry).14 GBS has previously been reported following corona virus infections, including MERS-CoV15 and more recently SARS-CoV-2.16 17 COVID-19 causes an exaggerated immune response with persistent fevers, elevated inflammatory markers and elevated proinflammatory cytokines. It is likely that COVID-19-associated immune dysregulation increases the risk of immune-mediated conditions such as GBS.18
A predominant feature of our patient’s presentation was muscle and radicular pain, which is present in two-thirds of GBS cases but has been less frequently reported in case reports of GBS following COVID-19 (14.8% of cases).11 12 He also reported lethargy at the time of onset of paraesthesia, this may be attributed to recent COVID-19 pneumonitis, as it is not usually described as an early feature of GBS and could represent part of the ‘long COVID syndrome’. The persistence of sensory deficits may also represent a feature of this.
Of particular interest, here is the time between the COVID-19 pneumonitis and the onset of the acute neuropathy. The 53 days between the index illness and GBS symptoms reported here is significantly longer than expected. The median time of onset from index SARS-CoV-2 infection reported in recent systematic reviews was 14 days (IQR 7–20 and 9–20 days) and 11.5 days (IQR 7.7–16 days).12 16 17 Newer treatments of COVID-19 pneumonitis with dexamethasone and other immunomodulatory medications may affect the prevalence and time of onset of GBS following SARS-CoV-2 infection.9
Men have a higher risk of all cause death and severe disease in COVID-19,19 which is mirrored by reports of male predominance (68.5%) in GBS following COVID-19.13 This gender difference is not seen in classical GBS and may add to speculation that gender-specific immune responses account for the higher male mortality with COVID-19.16 17 20
An important question regarding the pathophysiology of GBS following COVID-19 is whether it reflects a para-infectious response related to the acute inflammation or a true postinfectious immune-mediated response.21 22 The case we have presented would favour the latter and would give credence to the hypothesis that it is the immune response to COVID-19 and not the virus itself or the acute vascular changes that underly the pathophysiology of ‘long COVID-19 syndrome’.
Learning points.
Guillain-Barré syndrome (GBS) is a rare but recognised late complication of acute infection including following SARS-CoV-2. In our case, onset was relatively late compared with other reports.
In acute presentations of GBS with recent influenza-like or respiratory symptoms, COVID-19 serology could be considered to investigate the causative aetiology.
Long COVID-19 may share an immune-related pathology similar to the pathophysiology in GBS.
Patients need vigilant monitoring for disease progression: particularly respiratory function (frequent forced vital capacity monitoring).
Pain and paraesthesia may precede muscle weakness in GBS.
Acknowledgments
Many thanks to Dr Andrew Flatt (consultant Microbiologist) for his support and helpful ideas.
Footnotes
Contributors: MMR—first author wrote this case report in collaboration with AK (second). The timeline and most tables were edited, if not designed by MMR. CM helped to rewrite parts of the discussion and abstract. He also assisted AWA in the development of table 2 and modified the table of the Asbury Cornblath criteria. CM and AK ensured the text reads well. AWA wrote parts of the investigations and created table 2.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Pezzini A, Padovani A. Lifting the mask on neurological manifestations of COVID-19. Nat Rev Neurol 2020;16:636–44. 10.1038/s41582-020-0398-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Othenin-Girard A, Regamey J, Lamoth F, et al. Multisystem inflammatory syndrome with refractory cardiogenic shock due to acute myocarditis and mononeuritis multiplex after SARS-CoV-2 infection in an adult. Swiss Med Wkly 2020;150:w20387. 10.4414/smw.2020.20387 [DOI] [PubMed] [Google Scholar]
- 3.Needham E, Newcombe V, Michell A. Mononeuritis multiplex: an unexpectedly common feature of severe COVID-19. medRxiv 2020. 10.1101/2020.07.19.20149898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garrigues E, Janvier P, Kherabi Y, et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J Infect 2020;81:e4–6. 10.1016/j.jinf.2020.08.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Carfì A, Bernabei R, Landi F, et al. Persistent symptoms in patients after acute COVID-19. JAMA 2020;324:603–5. 10.1001/jama.2020.12603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tenforde MW, Kim SS, Lindsell CJ, et al. Symptom Duration and Risk Factors for Delayed Return to Usual Health Among Outpatients with COVID-19 in a Multistate Health Care Systems Network - United States, March-June 2020. MMWR Morb Mortal Wkly Rep 2020;69:993–8. 10.15585/mmwr.mm6930e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hahn AF. Guillain-Barré syndrome. Lancet 1998;352:635–41. 10.1016/S0140-6736(97)12308-X [DOI] [PubMed] [Google Scholar]
- 8.Ropper AH. The Guillain-Barré syndrome. N Engl J Med 1992;326:1130–6. 10.1056/NEJM199204233261706 [DOI] [PubMed] [Google Scholar]
- 9.Horby P, Landary M. Recovery trial, 2020. Available: https://www.recoverytrial.net/
- 10.Asbury AK, Cornblath DR. Assessment of current diagnostic criteria for Guillain-Barré syndrome. Ann Neurol 1990;27 Suppl:S21–4. 10.1002/ana.410270707 [DOI] [PubMed] [Google Scholar]
- 11.Willison HJ, Jacobs BC, van Doorn PA. Guillain-Barré syndrome. Lancet 2016;388:717–27. 10.1016/S0140-6736(16)00339-1 [DOI] [PubMed] [Google Scholar]
- 12.Hasan I, Saif-Ur-Rahman KM, Hayat S, et al. Guillain-Barré syndrome associated with SARS-CoV-2 infection: a systematic review and individual participant data meta-analysis. J Peripher Nerv Syst 2020;25:335–43. 10.1111/jns.12419 [DOI] [PubMed] [Google Scholar]
- 13.Hughes RAC, Swan AV, Raphaël J-C, et al. Immunotherapy for Guillain-Barré syndrome: a systematic review. Brain 2007;130): :2245–57. 10.1093/brain/awm004 [DOI] [PubMed] [Google Scholar]
- 14.van den Berg B, Walgaard C, Drenthen J, et al. Guillain-Barré syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol 2014;10:469–82. 10.1038/nrneurol.2014.121 [DOI] [PubMed] [Google Scholar]
- 15.Kim JE, Heo JH, Kim HO, et al. Neurological complications during treatment of middle East respiratory syndrome. J Clin Neurol 2017;13:227–33. 10.3988/jcn.2017.13.3.227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Uncini A, Vallat J-M, Jacobs BC. Guillain-Barré syndrome in SARS-CoV-2 infection: an instant systematic review of the first six months of pandemic. J Neurol Neurosurg Psychiatry 2020;91:1105–10. 10.1136/jnnp-2020-324491 [DOI] [PubMed] [Google Scholar]
- 17.Abu-Rumeileh S, Abdelhak A, Foschi M. Guillain-Barré syndrome spectrum associated with COVID-19: an up-to-date systematic review of 73 cases. J Neurol 2020:1–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020;395:1033–4. 10.1016/S0140-6736(20)30628-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kragholm K, Andersen MP, Gerds TA, et al. Association between male sex and outcomes of Coronavirus Disease 2019 (Covid-19) - a Danish nationwide, register-based study. Clin Infect Dis 2020. 10.1093/cid/ciaa924. [Epub ahead of print: 08 Jul 2020]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Carrillo-Larco RM, Altez-Fernandez C, Ravaglia S, et al. COVID-19 and Guillain-Barre syndrome: a systematic review of case reports. Wellcome Open Res 2020;5:107. 10.12688/wellcomeopenres.15987.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhao H, Shen D, Zhou H, et al. Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol 2020;19:383–4. 10.1016/S1474-4422(20)30109-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Paterson RW, Brown RL, Benjamin L, et al. The emerging spectrum of COVID-19 neurology: clinical, radiological and laboratory findings. Brain 2020;143:3104–20. 10.1093/brain/awaa240 [DOI] [PMC free article] [PubMed] [Google Scholar]