Abstract
Purpose
Reirradiation poses a distinct therapeutic challenge owing to risks associated with exceeding normal tissue tolerances and possibly more therapeutically resistant disease biology. We report our experience with reirradiation for locoregional recurrent or second primary breast cancer.
Methods and Materials
Between 1999 and 2019, all patients with breast cancer treated with repeat breast/chest wall radiation therapy (RT) at our institution were identified. Adverse events were assessed using the Common Terminology Criteria for Adverse Events v5.0. Fisher exact, Mann-Whitney rank-sum, and unpaired t tests were used for statistical analysis. Freedom from locoregional recurrence and distant metastasis as well as overall survival were calculated using the Kaplan-Meier method.
Results
Seventy-two patients underwent reirradiation. Median prior RT dose, reirradiation dose, and cumulative dose were 60 Gy (interquartile range [IQR], 50-60.4 Gy), 45 Gy (IQR, 40-50 Gy), and 103.54 Gy2 (IQR, 95.04-109.62 Gy2), respectively. Median time between RT courses was 73 months (IQR, 29-129 months). Thirty-four patients (47%) had gross residual disease at time of reirradiation. Course intent was described as curative in 44 patients (61%) and palliative in 28 (39%). Fifty-two patients (72%) were treated with photons ± electrons and 20 (28%) with protons. With a median follow-up of 22 months (IQR, 10-43 months), grade 3 adverse events were experienced by 13% of patients (10% acute skin toxicity and 3% late skin necrosis). Time between RT courses and reirradiation fields was significantly associated with the development of grade 3 toxicity at any point. Proton therapy conferred a dosimetric advantage without difference in toxicity. At 2 years, locoregional recurrence-free survival was 74.6% and overall survival was 65.5% among all patients, and 93.1% and 76.8%, respectively, among curative intent patients treated without gross disease. Distant metastasis-free survival was 59.0% among all curative intent patients.
Conclusions
Reirradiation for locoregional recurrent breast cancer is feasible with acceptable rates of toxicity. Disease control and survival are promising among curative intent reirradiation patients without gross disease.
Introduction
Breast-conserving therapy with radiation and mastectomy with or without radiation are part of standard local therapy for breast cancer.1, 2, 3 Although overall outcomes for breast cancer have improved, even with modern systemic therapy and optimal treatment, locoregional recurrence (LRR) occurs in a portion of patients.1, 2, 3, 4, 5 Treatment of LRR or second primary breast cancer in the setting of prior radiation therapy (RT) remains a therapeutic challenge, with primary consideration for further resection, systemic therapy, RT, and possibly reirradiation. Reirradiation may be a component of definitive locoregional therapy for isolated LRR, or may be employed palliatively for symptoms, such as bleeding or pain. The best reported outcomes for patients with local recurrence appear to be achieved with gross total resection, systemic therapy, and RT.6 However, selection of patients for reirradiation is complex, as risk for treatment-related toxicity increases with repeat radiation.7 Further, recurrent breast cancer may be more therapeutically resistant.8 More data are necessary to characterize outcomes with reirradiation in the modern era of systemic therapy and advanced RT techniques to guide the management of recurrent breast cancer.
Previous studies have reported a variety of techniques for reirradiation to the breast or chest wall including limited radiation fields, electrons, twice-daily radiation, superficial chest wall brachytherapy, concurrent systemic therapy, and hyperthermia.7,9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24 Whether improvements in radiation treatment delivery, such as optimized 3-dimensional conformal RT, intensity modulated RT, deep inspiratory breath hold, and particle therapy, increase the therapeutic ratio of reirradiation is not well established. Herein, we report toxicities and outcomes for a cohort of patients with breast cancer treated at a single institution with a second course of repeat breast/chest wall/nodal RT.
Methods and Materials
This study was institutional review board approved. We identified 74 consecutive patients with breast cancer who were treated with repeat breast/chest wall/nodal RT between 1999 and 2019. Two identified patients received only a third overlapping course at our institution and were excluded due to inability to fully characterize prior courses. All others received a second course of RT to previously treated breast, chest wall, and/or nodal regions for recurrent or second primary breast cancer. Sixty-eight patients had pathologic confirmation of disease before reirradiation, and the remaining 4 patients were diagnosed clinically. The courses of RT were evaluated for overlap and characterized as fully or partially overlapping. Plans were considered fully overlapping if the entire volume of the first or second treatment was encompassed by the other. The first course was photon or photon and electron based in all but 3 cases, 1 proton based and 2 brachytherapy based. The second course treatment technique was either photon +/- electrons, or protons. Hyperthermia was used in select cases. The intensity modulated proton therapy technique has been previously described.25 Treatment plans were evaluated for doses to the heart, lung, and coronary arteries, when contoured, for the second course of RT. The equivalent dose in 2 Gy for each course was calculated using an α/β ratio of 3. Each patient was classified as curative or palliative at reirradiation based on the presence of distant metastasis and reported physician intent.
Patients were followed for any locoregional or distant recurrence until date of last follow-up or date of death. LRR was defined as recurrence within the ipsilateral chest/breast or regional lymphatics. Adverse events (AEs) were collected prospectively at the point of care and assessed using the Common Terminology Criteria for Adverse Events, version 5.0.26 AEs were considered acute if they occurred within 90 days from the first day of reirradiation, late if they occurred after 90 days, and persistent if they developed before reirradiation and persisted beyond the last day of reirradiation.
Locoregional recurrence-free survival (LRFS), distant metastasis-free survival, and overall survival (OS) were estimated using the Kaplan-Meier method. Survival, recurrences, follow-up, and AEs were measured from day of reirradiation completion. Patients without LRR were considered censored for LRR, distant metastasis, or OS at the last date of follow-up free from the event. Log-rank (Mantel-Cox) test P value was used to determine statistical significance among Kaplan-Meier estimates. Fisher’s exact test was used for categorical variables, Mann-Whitney rank-sum test was used for continuous variables, and unpaired t test was used for independent means. P values less than .05 were considered to be statistically significant.
Results
Between 1999 and 2019, 72 women were treated with repeat breast, chest wall, and nodal radiation, as shown in Tables 1 and 2. Before the first course of RT, 51 patients (71%) underwent wide local excision, 14 (19%) underwent mastectomy, and 1 underwent axillary lymph node dissection only for occult primary presentation. Five (7%) underwent biopsy only, as the first course of radiation was given for postmastectomy recurrence, and 1 underwent biopsy only because the initial intent was palliative. Overall, 8 patients (11%) were classified as recurrent before the first course of radiation. For the first course of RT, median dose including boost was 60 Gy (interquartile range [IQR], 50.4-60.4 Gy) in patients receiving 1.8-2 Gy/d (n = 61), with 39 (54%) receiving median sequential boost of 10 Gy (IQR, 10-12.6) and 2 (3%) having integrated boost of 6.25 Gy and 8.75 Gy. The median dose including boost was 43.78 (IQR, 40.05-52.56 Gy) in patients receiving 2.25-3 Gy/d (n = 8), with 3 (4%) receiving sequential boost of 10 Gy (2) and 20 Gy (1). Three patients received 32 to 38.5 Gy in 10 fractions bid.
Table 1.
First radiation therapy tumor and treatment characteristics
| N = 72 (%) | |
|---|---|
| Age (years), median (range) | 51 (28-85) |
| Recurrent | 8 (11%) |
| Primary surgery | |
| Mastectomy | 14 (19%) |
| Wide local excision | 51 (71%) |
| Axillary lymph node dissection only∗ | 1 (1%) |
| Biopsy only† | 6 (8%) |
| NR/NA | - |
| Axillary surgery | |
| Axillary lymph node dissection | 40 (56%) |
| Sentinel lymph node biopsy | 16 (22%) |
| No axillary surgery | 11 (15%) |
| NR/NA | 5 (7%) |
| Pathologic T stage | |
| Tx | 1 (1%) |
| T0 | 2 (3%) |
| Tis | 6 (8%) |
| T1 | 23 (32%) |
| T2 | 15 (21%) |
| T3 | 6 (8%) |
| T4 | 2 (3%) |
| Recurrent | 8 (11%) |
| NR/NA | 9 (13%) |
| Pathologic N stage | |
| Nx | 2 (3%) |
| N0 | 25 (35%) |
| N1 | 17 (24%) |
| N2 | 7 (10%) |
| N3 | 4 (6%) |
| Recurrent | 8 (11%) |
| NR/NA | 9 (12%) |
| Complete pathologic response | 7 (10%) |
| AJCC 7th edition stage | |
| Stage 0 | 8 (11%) |
| Stage I | 17 (24%) |
| Stage II | 17 (24%) |
| Stage III | 13 (18%) |
| Stage IV | - |
| Recurrent | 8 (11%) |
| NR/NA | 9 (12%) |
| Histology | |
| IDC | 42 (58%) |
| ILC | 5 (7%) |
| Invasive carcinoma NOS | 8 (11%) |
| DCIS | 10 (14%) |
| LCIS | - |
| Phyllodes tumor | 1 (1%) |
| NR/NA | 6 (8%) |
| Receptor status | |
| ERPR+HER2- | 40 (56%) |
| ERPR+HER2+ | 1 (1%) |
| ERPR-HER2+ | 5 (7%) |
| TN | 14 (19%) |
| NR/NA | 12 (17%) |
| Modality | |
| Photons +/- electrons | 69 (96%) |
| Protons | 1 (1%) |
| Brachytherapy | 2 (3%) |
| RT fields | |
| Unilateral chest wall +/- nodes | 18 (25%) |
| Bilateral chest wall +/- nodes | - |
| Unilateral whole breast +/- nodes | 51 (71%) |
| Bilateral whole breast +/- nodes | - |
| Partial breast | 1 (1%) |
| Nodes only | 2 (3%) |
| NR/NA | - |
| Chemotherapy‡ | |
| Neoadjuvant | 16 (22%) |
| Adjuvant | 30 (42%) |
| Concurrent | 1 (1%) |
| NR/NA | 4 (6%) |
| Hormone therapy‡ | |
| Neoadjuvant | 1 (3%) |
| Adjuvant | 30 (42%) |
| Concurrent | - |
| NR/NA | 4 (6%) |
| Concurrent hyperthermia | |
| Yes | - |
| No | 68 (94%) |
| NR/NA | 4 (6%) |
| bid treatment | |
| Yes | 3 (4%) |
| No | 67 (93%) |
| NR/NA | 2 (3%) |
Abbreviations: AJCC = American Joint Committee on Cancer; DCIS = ductal carcinoma in situ; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; IDC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; LCIS = lobular carcinoma in situ; NA = not applicable; NOS = not otherwise specified; NR = not reported; PR = progesterone receptor; RT = radiation therapy; TN = triple negative.
Occult primary presentation
Five were recurrent with prior mastectomy and 1 was treated palliatively.
Categories are not mutually exclusive.
Table 2.
Second radiation therapy treatment characteristics
| N = 72 (%) | |
|---|---|
| Age (years), median (range) | 62 (31-91) |
| Multiply recurrent | 19 (26%) |
| Primary surgery | |
| Mastectomy | 23 (32%) |
| Wide local excision | 11 (15%) |
| Axillary mass excision | 3 (4%) |
| Axillary lymph node dissection only | 4 (6%) |
| Biopsy only∗ | 27 (38%) |
| No biopsy | 4 (6%) |
| NR/NA | - |
| Axillary surgery | |
| Axillary lymph node dissection | 30 (42%) |
| Sentinel lymph node biopsy | 3 (4%) |
| No axillary surgery | 38 (53%) |
| NR/NA | 1 (1%) |
| Gross residual disease at reirradiation | 34 (47%) |
| Modality | |
| Photons +/- electrons | 52 (72%) |
| Protons | 20 (28%) |
| Intent | |
| Curative | 44 (61%) |
| Palliative | 28 (39%) |
| RT fields | |
| Unilateral breast/chest wall +/- nodes | 43 (60%) |
| Bilateral breast/chest wall +/- nodes | 9 (13%) |
| Partial breast | 1 (1%) |
| Nodes only | 19 (26%) |
| NR/NA | - |
| Chemotherapy† | |
| Neoadjuvant | 27 (38%) |
| Adjuvant | 28 (39%) |
| Concurrent | 17 (24%) |
| NR/NA | 1 (1%) |
| Hormone therapy† | |
| Neoadjuvant | 5 (6%) |
| Adjuvant | 25 (35%) |
| Concurrent | 4 (6%) |
| NR/NA | 1 (1%) |
| Concurrent hyperthermia | |
| Yes | 15 (21%) |
| No | 57 (79%) |
| NR/NA | - |
| bid treatment | |
| Yes | 4 (5%) |
| No | 68 (95%) |
| NR/NA | - |
Abbreviations: NA = not applicable; NR = not reported; RT = radiation therapy.
Patients underwent mastectomy previously, were already metastatic at presentation, and/or were being treated palliatively.
Categories are not mutually exclusive.
The median interval between radiation courses was 73 months (IQR, 29-129 months). Salvage surgery for recurrence included biopsy only in 27 (38%), mastectomy in 23 patients (32%), wide local excision in 11 (15%), axillary lymph node biopsy only in 4 (6%), and axillary mass excision in 3 (4%). The remaining 4 patients (6%) received diagnoses clinically. Those who underwent biopsy only had prior mastectomy (n = 21), were already metastatic at second course (n = 9), and/or were being treated palliatively (n = 20). Overall, 19 patients (26%) were multiply recurrent before the second course of radiation. Thirty-three women (46%) had axillary surgery, consisting of axillary node dissection in 30 (42%) and sentinel node biopsy in 3 (4%). Thirty-four patients (47%) had gross residual disease at time of reirradiation.
The reirradiation course intent was described as curative in 44 patients (61%) and palliative in 28 (39%). Fifty-two patients (72%) were treated with photons ± electrons and 20 (28%) with protons. Clinical target volume included unilateral breast/chest wall ± nodes in 43 patients (60%), bilateral breast/chest wall ± nodes in 9 patients (13%), partial breast in 1 patient, and regional nodes only in 19 patients (26%). Reirradiation was conventionally fractionated (1.8-2 Gy/d) to a median dose of 50 Gy (IQR, 40-50.4 Gy) in 47 patients (65%)—with 5 patients receiving an additional boost of 10 Gy (IQR, 9.5-12 Gy) and 2 having an integrated boost of 5.6 Gy and 6.25 Gy—and hypofractionated (2.25-7.3 Gy/d) to a median dose of 40.05 Gy (IQR, 40.03-43.78 Gy) in 21 (29%), with 1 patient receiving a sequential boost of 10 Gy. Four patients (6%) received 39.6-60 Gy in 1.2-3.7 Gy/d bid. Median cumulative dose in 2-Gy equivalents for the 2 courses was calculated as 103.54 Gy2 (IQR, 95.04-109.62 Gy2), including boosts. Seventeen patients (24%) received concurrent chemotherapy with capecitabine, 4 (6%) received concurrent hormone therapy, and 15 (21%) were treated with concurrent hyperthermia.
Table 3 lists available doses to organs at risk (OAR) with the second course of radiation. Compared with photons, proton beam therapy (PBT) resulted in lower mean doses to the heart, lungs, and coronary arteries. Significance was seen for all parameters between left-sided proton and photon patients. All parameters were numerically reduced with PBT for right-sided patients, with statistical significance reached for the heart mean dose, right coronary, lung mean, and lung V5.
Table 3.
Second course radiation dose to organs at risk
| Structure | Parameter | Proton left-sided (n = 12) mean (n; range) | Photon left-sided (n = 23) mean (n; range) | P value | Proton right-sided (n = 8) mean (n; range) | Photon right-sided (n = 29) mean (n; range) | P value |
|---|---|---|---|---|---|---|---|
| Heart | |||||||
| Mean (Gy) | 0.8 (11; 0.0-3.1) | 5.6 (11; 0.5-22.5) | .02 | 0.4 (7; 0.1-1.5) | 4.5 (12; 0.8-17.1) | .04 | |
| V5Gy (%) | 4.4 (11; 0.0-20.8) | 20.2 (11; 0.0-100.0) | .02 | 2.2 (7; 0.0-8.0) | 30.9 (12; 0.0-100.0) | .06 | |
| Coronary vessels | |||||||
| Left anterior descending artery | Mean (Gy) | 1.2 (8; 0.0-2.6) | 3.4 (2; 3.0-3.9) | .02 | 1.3 (3; 0.0-2.4) | - | - |
| Right coronary artery | Mean (Gy) | 0.5 (3; 0.0-1.0) | - | - | 1.9 (4; 1.6-2.5) | 6.8 (1) | .002 |
| Ipsilateral lung | |||||||
| Mean (Gy) | 4.7 (12; 0.0-9.0) | 10.9 (15; 2.6-23.9) | .002 | 6.8 (8; 2.7-10.3) | 11.3 (22; 0.8-18.8) | .02 | |
| V5Gy (%) | 23.9 (12; 0.0-47.6) | 54.4 (15; 10.1-100.0) | .004 | 36.4 (8; 16.2-50.6) | 57.3 (22; 1.9-100.0) | .04 | |
| V10Gy (%) | 17.1 (12; 0.0-33.4) | 36.4 (15; 4.7-92.8) | .01 | 24.9 (8; 7.0-36.3) | 38.2 (22; 1.2-100.0) | .10 | |
| V20Gy (%) | 8.8 (12; 0.0-16.8) | 19.5 (15; 2.8-58.1) | .02 | 12.0 (8; 0.9-19.3) | 18.5 (22; 0.8-32.1) | .08 |
P values less than .05 have been bolded to highlight significance.
Median follow-up time from completion of reirradiation was 22 months (IQR, 11-43 months). The highest grade toxicity experienced at any point was grade 1 by 30 patients (42%), grade 2 by 33 (46%), and grade 3 by 9 (13%). There were no reported grade 4/5 AEs.
The most common acute toxicity was radiation dermatitis grade 1 in 43 patients (60%), grade 2 in 22 (31%), and grade 3 in 6 (8%). One additional patient experienced grade 3 skin necrosis during treatment. This patient had diffuse dermal lymphovascular invasion, which was identified as a probable contributor. Table 4 displays a univariate analysis of selected patient and treatment variables by incidence of acute grade 1 to 2 versus grade 3 AEs. Reirradiation fields (P = .05) and concurrent capecitabine at reirradiation (P = .05) were the only variables significantly associated with the development of acute grade 3 AEs.
Table 4.
Patient and treatment characteristics by AE grade for acute toxicities
| Grade 1-2 n = 65 | Grade 3 n = 7 | P value | |
|---|---|---|---|
| Age at reirradiation (years), median (range) | 63 (31-91) | 62 (39-80) | .61 |
| BMI (kg/m2) at reirradiation | |||
| <30 | 43 | 5 (10.4%) | 1.00 |
| >30 | 20 | 2 (22.2%) | |
| NR/NA | 2 | 0 (0%) | |
| Smoking history | |||
| Yes | 26 | 3 (10.3%) | 1.00 |
| No | 34 | 4 (10.5%) | |
| NR/NA | 5 | 0 (0%) | |
| Treatment modality at reirradiation | |||
| Photons | 48 | 4 (7.7%) | .39 |
| Protons | 17 | 3 (15.0%) | |
| Reirradiation fields | |||
| Unilateral breast/chest wall +/- nodes | 39 | 4 (9.3%) | .05 |
| Bilateral breast/chest wall +/- nodes | 6 | 3 (33.3%) | |
| Partial breast | 1 | 0 (0%) | |
| Regional nodes only | 19 | 0 (0%) | |
| Reirradiation to regional nodes | |||
| Yes | 47 | 5 (9.6%) | 1.00 |
| No | 18 | 2 (10.0%) | |
| Gross disease present at the time of reirradiation | |||
| Yes | 29 | 5 (14.7%) | .24 |
| No | 36 | 2 (5.3%) | |
| Concurrent capecitabine at reirradiation | |||
| Yes | 13 | 4 (23.5%) | .05 |
| No | 52 | 3 (5.5%) | |
| Concurrent hyperthermia at reirradiation | |||
| Yes | 13 | 2 (13.3%) | .63 |
| No | 52 | 5 (8.8%) | |
| bid treatment at reirradiation | |||
| Yes | 3 | 1 (25.0%) | .34 |
| No | 62 | 6 (8.8%) | |
| Prior RT dose including boost (Gy), median (IQR) | 60 (50-60.4) | 50 (38.5-60.0) | .14 |
| Reirradiation dose including boost (Gy), median (IQR) | 45 (40-50) | 50 (45-50.4) | .09 |
| Use of boost at reirradiation | |||
| Yes | 7 | 1 (12.5%) | .58 |
| No | 58 | 6 (9.4%) | |
| Cumulative RT dose including boost (Gy2), median (IQR) | 103.4 (94.79-109.1) | 110 (100-114) | .21 |
| Time between RT courses (months), median (IQR) | 76 (30-141) | 51 (11-61) | .11 |
| Degree of RT overlap | |||
| Full overlap | 56 | 7 (11.1%) | .58 |
| Partial overlap | 9 | 0 (0%) |
Abbreviations: AE = adverse events; BMI = body mass index; IQR = interquartile range; NA = not applicable; NR = not reported; RT = radiation therapy.
P values less than .05 have been bolded to highlight significance.
Late grade 1 AEs included brachial plexopathy in 1 patient, osteonecrosis in 1, soft tissue necrosis in 2 (3%), decreased range of motion in 7 (10%), wound complication in 7 (10%), lymphedema in 8 (11%), new chest wall and soft tissue fibrosis in 11 (15%), and lung fibrosis in 13 (18%). Osteonecrosis of the anterior second rib was experienced by a patient treated with curative intent using photons initially and at reirradiation. Field overlap was complete and courses were 46 months apart with cumulative dose 100.4 Gy (50 Gy to the breast followed by 50.4 Gy to the chest wall and nodes at reirradiation). Late grade 2 AEs included brachial plexopathy in 2 (3%), lymphedema in 2 (3%), soft tissue necrosis in 2 (3%), wound complication in 3 (4%), decreased range of motion in 4 (6%), skin infection in 6 (8%), new chest wall and soft tissue fibrosis in 9 (13%), and telangiectasia in 9 (13%). Both patients with brachial plexopathy were treated using photons initially and at reirradiation. In both cases, the axilla was not retreated, and the plexopathy was noted with subsequent axillary recurrence with plexus involvement. The first was treated initially with whole breast RT, followed by a second course to the axilla, 176 months apart, for a total of 100 Gy, with little expected overlap at the plexus. Brachial plexopathy was diagnosed 63 months later due to metastatic tumor involvement. The second was treated with palliative intent to a chest wall target twice with complete field overlap, 29 months apart, for a total of 80.6 Gy. Little cumulative plexus dose was expected, and brachial plexopathy was diagnosed 22 months after reirradiation owing to metastatic tumor involvement. One patient had a chronic nonhealing wound before reirradiation, which worsened after treatment but was managed conservatively. Chest wall fibrosis before second RT course was grade 1 in 25 patients (35%) and grade 2 in 3 (4%), which persisted along with the additional 28% noted previously for a total reported rate of 67% in the cohort. After reirradiation, 2 patients (3%) developed late grade 3 skin necrosis, 1 of whom required a flap-based surgical closure 2 years after reirradiation. Both were treated with palliative intent, with photon-based planning, and had gross disease at time of reirradiation. Fourteen patients (19%) developed rib fracture at a median time of 11 months (IQR, 7-31). Ten were asymptomatic and identified by imaging. We did not observe a significant association between any of the selected patient and treatment variables with grade ≥2 late AEs (not shown). Times between RT courses (P = .04) and reirradiation fields (P = .01) were significant for the development of any grade 3 (acute or late) toxicity (Table E1), with 4 of 9 patients treated bilaterally having any grade 3 toxicity. Although it was not significant, all the grade 3 toxicity occurred in patients characterized as having completely overlapping fields.
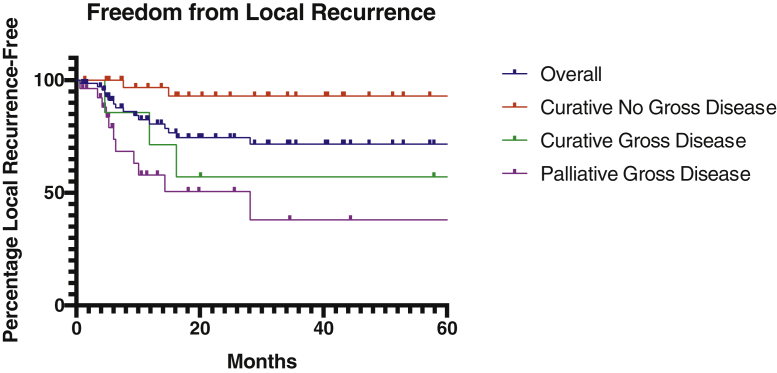
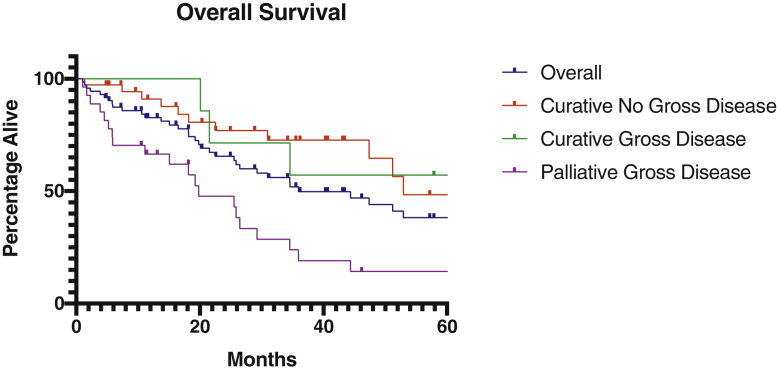
The 2-year LRFS and OS estimates for the overall cohort were 74.6% and 65.5%, respectively (Figs 1 and 2). We also evaluated LRFS and OS at 2 years among 3 different subgroups: 37 curative intent patients without gross disease (51%), 7 curative intent patients with gross disease (10%), and 27 palliative intent patients with gross disease (38%). One additional palliative intent patient underwent reirradiation without gross disease and was alive at last follow-up at 96 months without LRR or distant metastasis. Two-year LRFS was 93.1% in curative intent patients without gross disease, 57.1% in curative intent patients with gross disease, and 50.7% in palliative intent patients with gross disease (P = .005, Fig 1). The cumulative median biologically effective dose for curative patients without gross disease was 105.42 Gy2 (IQR, 99.19-109.83 Gy2), compared with 114 Gy2 (IQR, 101.18-115.78 Gy2) for curative patients with gross disease (P = .08). Two-year OS was 76.8%, 71.4%, and 47.7%, respectively (P < .001, Fig 2). Distant metastasis-free survival was 59.0% among all curative intent patients.
Fig. 1.
Overall survival Kaplan-Meier estimates for the entire cohort, as well as 3 subgroups: curative intent patients without gross disease, curative intent patients with gross disease, and palliative intent patients with gross disease.
Fig. 2.
Locoregional recurrence-free survival Kaplan-Meier estimates for the entire cohort, as well as 3 subgroups: curative intent patients without gross disease, curative intent patients with gross disease, and palliative intent patients without gross disease.
Discussion
In the present study, we report favorable LRFS and OS after reirradiation for LRR breast cancer, similar to other cohorts.7,14,15,17,19,20,27 Particularly, high rates of disease control were seen among curative intent patients undergoing complete surgical excision followed by reirradiation. Although many patients experienced late grade 1 or 2 soft tissue toxicity, high-grade toxicity was limited, with 13% of patients experiencing grade 3 toxicity at any point (10% acute skin toxicity and 3% late skin necrosis), and no patients experiencing grade 4 or 5 toxicities. The most common delayed toxicity, aside from soft tissue fibrosis and telangiectasia, was rib fracture, many of which were asymptomatic. Although the absence of high-grade fibrosis is reassuring, based on our clinical experience, at least low-grade soft tissue fibrosis is likely after reirradiation. Even with 67% reported to have grade 1 to 2 fibrosis, low-grade soft tissue fibrosis may be underreported, and patient-reported outcomes could be helpful to fully characterize normal tissue effects and function following reirradiation.
Proton therapy for breast cancer offers enhanced ability to spare nontarget tissue, including the heart and lungs,24,28, 29, 30, 31, 32, 33 which is of particular importance and interest for reirradiation. Reirradiation with PBT has been reported in multiple disease sites, including in central nervous system, head and neck, gynecologic, gastrointestinal, and lung tumors.34, 35, 36, 37, 38, 39, 40, 41 There are limited data in the setting of breast reirradiation. The Proton Collaborative Group reported favorable local control in 50 proton patients at a median follow-up of 12.7 months.28 LRFS was 93% and OS was 97% at 1 year, with only 16% of patients experiencing grade 3 AEs at any point. We observed similar low rates of grade 3 toxicity at any point (13%), all of which was observed in patients with fully overlapping fields. In our study, PBT was significantly associated with lower doses to the heart, coronaries, and lung, compared with photon treatment in both left-and right-sided patients. Despite the dosimetric advantage, there was no association with late toxicity, possibly because of few events and limited follow-up. In addition, there may be selection bias, with proton therapy considered for the most complex and difficult-to-treat tumors and target volumes. Nevertheless, sparing of OAR is of heightened importance with reirradiation, given the possible toxicity from both systemic therapy and prior RT. Long-term follow-up may be necessary to observe the benefits of reduced OAR doses. Our study suggests that patients with complete field overlap, shorter intervals to retreatment, and larger reirradiation fields may have a heightened risk for toxicity, and could have the greatest potential benefit from proton therapy. Although proton therapy would not be expected to reduce toxicity within overlapping targets, it could reduce the degree of overlap, particularly adjacent and deep to the targets. Additionally, patients treated with curative intent without gross disease had the best oncologic outcomes, suggesting a greater potential for long-term benefit for OAR sparing.
Toxicity rates have varied significantly in studies evaluating reirradiation.12,23,30, 31, 32, 33 Our rate of new onset grade 3 toxicities at any point (13%) appears favorable. Previous studies have looked at factors contributing to increased rates and higher grades of toxicity, including higher radiation dose, presence of gross disease, and concurrent systemic therapy.28,42,43 However, to our knowledge, no other studies have evaluated rates and grades of toxicity with regards to degree of field overlap at time of reirradiation. As demonstrated in Table 4, reirradiation fields and concurrent capecitabine were significant for the development of acute grade 3 toxicity. No characteristics were significant for the development of late grade 2+ toxicity, and the low number of late grade 3 AEs limited further analysis. However, given short follow-up and small number of events, further follow-up of this cohort over a longer period of time is planned to better understand the effect of treatment characteristics on rates and grades of toxicities.
As noted, curative intent patients without gross disease had the best oncological outcomes compared with the overall cohort, whereas palliative intent patients with gross disease had the worst. Our curative intent patients without gross disease compare favorably relative to other similar studies with an LRFS of 93.1% and OS of 76.8% at 2 years. LRFS was significantly worse for curative intent patients treated with gross disease (57.1%), despite the trend to higher equivalent dose in 2 Gy, emphasizing the importance of excision where feasible and consideration for alternative escalation strategies where not. Our palliative intent patients with gross disease had less favorable outcomes, but were similar to previous studies with LRFS of 50.7% and OS of 47.7% at 2 years.7,14,15,17,19,20,27 The development of distant metastasis remained a substantial concern for patients treated with curative intent, suggesting that continued study into optimization of systemic therapy for LRR is warranted.8
There are several limitations to our study. First, our cohort is small and heterogeneous with regards to both initial and subsequent treatment and follow-up times. Despite this, the heterogeneity allows for comparison across differing treatment modalities. Although small, the cohort adds to the growing body of data supporting that reirradiation may be an appropriate consideration for select patients with recurrent or second primary breast cancer. The small event numbers highlight the need for ongoing follow-up for full characterization of toxicity. Future studies that incorporate patient-reported outcomes would be valuable.
Conclusions
Our study shows that reirradiation for breast cancer is feasible with acceptable toxicity. In particular, local disease control outcomes appear promising, especially among resected patients treated with curative intent. Early follow-up shows acceptable acute and late toxicity, with higher rates of grade 3 toxicity when treatment fields are fully overlapping, intervals between radiation are shorter, or larger volumes are treated. Proton therapy has dosimetric advantages for OAR sparing, though additional follow-up is needed to evaluate whether this translates into reduction of toxicity.
Footnotes
Sources of support: This work had no specific funding.
Disclosures: none.
All data are housed on an institutional server accessible by team members only.
Supplementary material for this article can be found at https://doi.org/10.1016/j.adro.2020.100640.
Supplementary Materials
References
- 1.Early Breast Cancer Trialists’ Collaborative Group Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: Meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet. 2014;383:2127–2135. doi: 10.1016/S0140-6736(14)60488-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fisher B., Anderson S., Bryant J. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002;347:1233–1241. doi: 10.1056/NEJMoa022152. [DOI] [PubMed] [Google Scholar]
- 3.Veronesi U., Cascinelli N., Mariani L. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med. 2002;347:1227–1232. doi: 10.1056/NEJMoa020989. [DOI] [PubMed] [Google Scholar]
- 4.Whelan T.J., Olivotto I.A., Parulekar W.R. Regional nodal irradiation in early-stage breast cancer. N Engl J Med. 2015;373:307–316. doi: 10.1056/NEJMoa1415340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haviland J.S., Owen J.R., Dewar J.A. The UK Standardisation of Breast Radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early breast cancer: 10-year follow-up results of two randomised controlled trials. Lancet Oncol. 2013;14:1086–1094. doi: 10.1016/S1470-2045(13)70386-3. [DOI] [PubMed] [Google Scholar]
- 6.Hanrahan E.O., Broglio K.R., Buzdar A.U. Combined-modality treatment for isolated recurrences of breast carcinoma: Update on 30 years of experience at the University of Texas M.D. Anderson Cancer Center and assessment of prognostic factors. Cancer. 2005;104:1158–1171. doi: 10.1002/cncr.21305. [DOI] [PubMed] [Google Scholar]
- 7.Oldenborg S., Griesdoorn V., van Os R. Reirradiation and hyperthermia for irresectable locoregional recurrent breast cancer in previously irradiated area: Size matters. Radiother Oncol. 2015;117:223–228. doi: 10.1016/j.radonc.2015.10.017. [DOI] [PubMed] [Google Scholar]
- 8.Wapnir I.L., Price K.N., Anderson S.J. Efficacy of chemotherapy for ER-negative and ER-positive isolated locoregional recurrence of breast cancer: Final analysis of the CALOR trial. J Clin Oncol. 2018;36:1073–1079. doi: 10.1200/JCO.2017.76.5719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Harkenrider M.M., Wilson M.R., Dragun A.E. Reirradiation as a component of the multidisciplinary management of locally recurrent breast cancer. Clin Breast Cancer. 2011;11:171–176. doi: 10.1016/j.clbc.2011.03.014. [DOI] [PubMed] [Google Scholar]
- 10.Kouloulias V.E., Pataniotis G.A., Kouvaris J.R. Re-irradiation in conjunction with liposomal doxorubicin for the treatment of skin metastases of recurrent breast cancer: A radiobiological approach and 2 year of follow-up. Cancer Lett. 2003;193:33–40. doi: 10.1016/s0304-3835(02)00742-5. [DOI] [PubMed] [Google Scholar]
- 11.Deutsch M. Repeat high-dose external beam irradiation for in-breast tumor recurrence after previous lumpectomy and whole breast irradiation. Int J Radiat Oncol Biol Phys. 2002;53:687–691. doi: 10.1016/s0360-3016(02)02785-2. [DOI] [PubMed] [Google Scholar]
- 12.Harms W., Krempien R., Hensley F.W., Berns C., Wannenmacher M., Fritz P. Results of chest wall reirradiation using pulsed-dose-rate (PDR) brachytherapy molds for breast cancer local recurrences. Int J Radiat Oncol Biol Phys. 2001;49:205–210. doi: 10.1016/s0360-3016(00)01360-2. [DOI] [PubMed] [Google Scholar]
- 13.Kauer-Dorner D., Potter R., Resch A. Partial breast irradiation for locally recurrent breast cancer within a second breast conserving treatment: alternative to mastectomy? Results from a prospective trial. Radiother Oncol. 2012;102:96–101. doi: 10.1016/j.radonc.2011.07.020. [DOI] [PubMed] [Google Scholar]
- 14.Muller A.C., Eckert F., Heinrich V., Bamberg M., Brucker S., Hehr T. Re-surgery and chest wall re-irradiation for recurrent breast cancer: A second curative approach. BMC Cancer. 2011;11:197. doi: 10.1186/1471-2407-11-197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wahl A.O., Rademaker A., Kiel K.D. Multi-institutional review of repeat irradiation of chest wall and breast for recurrent breast cancer. Int J Radiat Oncol Biol Phys. 2008;70:477–484. doi: 10.1016/j.ijrobp.2007.06.035. [DOI] [PubMed] [Google Scholar]
- 16.Resch A., Fellner C., Mock U. Locally recurrent breast cancer: Pulse dose rate brachytherapy for repeat irradiation following lumpectomy— a second chance to preserve the breast. Radiology. 2002;225:713–718. doi: 10.1148/radiol.2253011913. [DOI] [PubMed] [Google Scholar]
- 17.Merino T., Tran W.T., Czarnota G.J. Re-irradiation for locally recurrent refractory breast cancer. Oncotarget. 2015;6:35051–35062. doi: 10.18632/oncotarget.6036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wurschmidt F., Dahle J., Petersen C., Wenzel C., Kretschmer M., Bastian C. Reirradiation of recurrent breast cancer with and without concurrent chemotherapy. Radiat Oncol. 2008;3:28. doi: 10.1186/1748-717X-3-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Linthorst M., van Geel A.N., Baaijens M., Ameziane A., Ghidey W., van Rhoon G.C., vander Zee J. Re-irradiation and hyperthermia after surgery for recurrent breast cancer. Radiother Oncol. 2013;109:188–193. doi: 10.1016/j.radonc.2013.05.010. [DOI] [PubMed] [Google Scholar]
- 20.Linthorst M., Baaijens M., Wiggenraad R. Local control rate after the combination of re-irradiation and hyperthermia for irresectable recurrent breast cancer: Results in 248 patients. Radiother Oncol. 2015;117:217–222. doi: 10.1016/j.radonc.2015.04.019. [DOI] [PubMed] [Google Scholar]
- 21.Notter M., Thomsen A.R., Nitsche M. Combined wIRA-hyperthermia and hypofractionated re-irradiation in the treatment of locally recurrent breast cancer: Evaluation of therapeutic outcome based on a novel size classification. Cancers (Basel) 2020;12:606. doi: 10.3390/cancers12030606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Montagne L., Hannoun A., Hannoun-Levi J.M. Second conservative treatment for second ipsilateral breast tumor event: A systematic review of the different re-irradiation techniques. Breast. 2020;49:274–280. doi: 10.1016/j.breast.2020.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Arthur D.W., Winter K.A., Kuerer H.M. Effectiveness of breast-conserving surgery and 3-dimensional conformal partial breast reirradiation for recurrence of breast cancer in the ipsilateral breast: The NRG Oncology/RTOG 1014 Phase 2 Clinical Trial. JAMA Oncol. 2019 doi: 10.1001/jamaoncol.2019.4320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gabani P., Patel H., Thomas M.A. Clinical outcomes and toxicity of proton beam radiation therapy for re-irradiation of locally recurrent breast cancer. Clin Transl Radiat Oncol. 2019;19:116–122. doi: 10.1016/j.ctro.2019.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mutter R.W., Remmes N.B., Mh Kahila M. Initial clinical experience of postmastectomy intensity modulated proton therapy in patients with breast expanders with metallic ports. Pract Radiat Oncol. 2017;7:e243–e252. doi: 10.1016/j.prro.2016.12.002. [DOI] [PubMed] [Google Scholar]
- 26.National Cancer Institute . Common Terminology Criteria for Adverse Events version 5.0. U.S. Department of Health and Human Services; 2017. [Google Scholar]
- 27.Datta N.R., Puric E.M., Klingbiel D., Gomez S., Bodis S. Hyperthermia and radiation therapy in locoregional recurrent breast cancers: A systematic review and meta-analysis. Int J Radiat Oncol Biol Phys. 2016;94:1073–1087. doi: 10.1016/j.ijrobp.2015.12.361. [DOI] [PubMed] [Google Scholar]
- 28.Thorpe C.S., Niska J.R., Girardo M.E. Proton beam therapy reirradiation for breast cancer: Multi-institutional prospective PCG registry analysis. Breast J. 2019;25:1160–1170. doi: 10.1111/tbj.13423. [DOI] [PubMed] [Google Scholar]
- 29.Thorpe C.S., Niska J.R., Anderson J.D. Acute toxicities after proton beam therapy following breast-conserving surgery for breast cancer: Multi-institutional prospective PCG registry analysis. Breast J. 2020;26:1760–1764. doi: 10.1111/tbj.13812. [DOI] [PubMed] [Google Scholar]
- 30.Verma V., Rwigema J.-C.M., Malyapa R.S., Regine W.F., Simone C.B. Systematic assessment of clinical outcomes and toxicities of proton radiotherapy for reirradiation. Radiother Oncol. 2017;125:21–30. doi: 10.1016/j.radonc.2017.08.005. [DOI] [PubMed] [Google Scholar]
- 31.Moon S.H., Shin K.H., Kim T.H. Dosimetric comparison of four different external beam partial breast irradiation techniques: Three-dimensional conformal radiotherapy, intensity-modulated radiotherapy, helical tomotherapy, and proton beam therapy. Radiother Oncol. 2009;90:66–73. doi: 10.1016/j.radonc.2008.09.027. [DOI] [PubMed] [Google Scholar]
- 32.Kozak K.R., Katz A., Adams J. Dosimetric comparison of proton and photon three-dimensional, conformal, external beam accelerated partial breast irradiation techniques. Int J Radiat Oncol Biol Phys. 2006;65:1572–1578. doi: 10.1016/j.ijrobp.2006.04.025. [DOI] [PubMed] [Google Scholar]
- 33.Cuaron J.J., Chon B., Tsai H. Early toxicity in patients treated with postoperative proton therapy for locally advanced breast cancer. Int J Radiat Oncol Biol Phys. 2015;92:284–291. doi: 10.1016/j.ijrobp.2015.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.McDonald M.W., Linton O.R., Shah M.V. Proton therapy for reirradiation of progressive or recurrent chordoma. Int J Radiat Oncol Biol Phys. 2013;87:1107–1114. doi: 10.1016/j.ijrobp.2013.09.038. [DOI] [PubMed] [Google Scholar]
- 35.McDonald M.W., Zolali-Meybodi O., Lehnert S.J. Reirradiation of recurrent and second primary head and neck cancer with proton therapy. Int J Radiat Oncol Biol Phys. 2016;96:808–819. doi: 10.1016/j.ijrobp.2016.07.037. [DOI] [PubMed] [Google Scholar]
- 36.Romesser P.B., Cahlon O., Scher E.D. Proton beam reirradiation for recurrent head and neck cancer: Multi-institutional report on feasibility and early outcomes. Int J Radiat Oncol Biol Phys. 2016;95:386–395. doi: 10.1016/j.ijrobp.2016.02.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Oshiro Y., Mizumoto M., Okumura T. Analysis of repeated proton beam therapy for patients with hepatocellular carcinoma. Radiother Oncol. 2017;123:240–245. doi: 10.1016/j.radonc.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 38.Eaton B.R., Chowdhry V., Weaver K. Use of proton therapy for re-irradiation in pediatric intracranial ependymoma. Radiother Oncol. 2015;116:301–308. doi: 10.1016/j.radonc.2015.07.023. [DOI] [PubMed] [Google Scholar]
- 39.Chao H.H., Berman A.T., Simone C.B. Multi-institutional prospective study of reirradiation with proton beam radiotherapy for locoregionally recurrent non-small cell lung cancer. J Thorac Oncol. 2017;2:281–292. doi: 10.1016/j.jtho.2016.10.018. [DOI] [PubMed] [Google Scholar]
- 40.Li Y.R., Kirk M., Lin L. Proton therapy for vaginal reirradiation. Int J Part Ther. 2016;3:320–326. doi: 10.14338/IJPT-16-00013.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Boimel P.J., Berman A.T., Li J. Proton beam reirradiation for locally recurrent pancreatic adenocarcinoma. J Gastrointest Oncol. 2017;8:665–674. doi: 10.21037/jgo.2017.03.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Parekh A., Dholakia A.D., Zabransky D.J. Predictors of radiation-induced acute skin toxicity in breast cancer at a single institution: Role of fractionation and treatment volume. Adv Radiat Oncol. 2018;3:8–15. doi: 10.1016/j.adro.2017.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Woodward W.A., Fang P., Arriaga L. A phase 2 study of capecitabine and concomitant radiation in women with advanced breast cancer. Int J Radiat Oncol Biol Phys. 2017;99:777–783. doi: 10.1016/j.ijrobp.2017.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.