Abstract
With the global spread of SARS-CoV-2, millions of people have been affected leading to the declaration of coronavirus disease 2019 (COVID-19) as a pandemic by the WHO. Several studies have linked the severity of COVID-19 cases and increased fatality in patients with obesity and other comorbid conditions such as diabetes, cardiovascular diseases, hypertension, and kidney disease. Obesity, a metabolically deranged condition, establishes a low-grade chronic inflammation in the body, which affects different organs and promotes the development of several other diseases. The ways in which SARS-CoV-2 infection aggravates the already overloaded body organs with inflammation or vice versa has perplexed the researchers. As a result, there is an intensified search for the clear-cut mechanism to understand the link of obesity with the increased severity of COVID-19 in obese patients. In this article we have discussed various mechanisms linking obesity, inflammation, and COVID-19 to enhance the understanding of the disease process and help the clinicians and scientists develop potential cellular, molecular and metabolic targets for clinical intervention and management of COVID-19 severity in obese patients.
Keywords: SARS-CoV-2, COVID-19, Obesity, Inflammation, Coronavirus, Metabolic syndrome
Introduction
Coronavirus disease 2019 (COVID-19), declared as a pandemic by World Health Organization (WHO), is the result of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection [1]. The disease has affected more than 75 million people globally; more than 1.6 million people have succumb to the COVID-19 disease and died, and the number of infected is increasing consistently [2]. Among the dead, a majority of the patients were reportedly obese and/or had other comorbid conditions. The obese people with >40 body mass index (BMI) have been reported to be at the increased risk of showing severity from COVID-19 [3]. However, this intricate relationship surfaced pretty late in the course of pandemic with a scarce reporting on the metabolic parameters such as BMI in the preliminary data from the hospitalized COVID-19 patients [4].
Obesity as a medical condition has itself reached a pandemic stage and globally there are 650 million (13%) obese (BMI ≥ 30 kg/m²) and over 2 billion (39%) are overweight [5]. Obesity predisposes an individual to develop many other comorbid conditions including type 2 diabetes, cardiovascular disease, hypertension, dyslipidemia, and several types of cancers. However, a miniscule subpopulation of metabolically healthy obese seems to be at low risk of developing obesity-associated comorbidities and mortality [6]. An increase in the visceral fat depots and the overall fat mass is believed to be orchestrating metabolic syndrome (MS) in the obese and overweight. It is thus recommended by clinicians that people with obesity follow a healthy diet and lifestyle in order to have a reduced overall fat mass and thus stay metabolically healthy [7]. Furthermore, the presence of low-grade chronic inflammatory condition in people with obesity is believed to facilitate MS [8]. The level of several key cytokines and chemokines such as interleukin (IL)-6, IL-1β, IL-8, and tumor necrosis factor-alpha (TNF-α) among others are elevated in the people with obesity as compared to their lean counterparts [9]. The consistently elevated levels of these inflammatory molecules weaken the response of innate immune cells and the response of the first line of defense including the mucosal lining of the airway is compromised. Additionally, elevated levels of lipid-derived inflammatory mediators also affect the airway epithelial cells (AEC) functions [10]. Since low-grade chronic inflammation and its associated metabolic illness also corroborate with both inherent [11] and acquired lipodystrophy [12], the potential cellular & molecular targets for clinical intervention in the obese COVID-19 patients discussed in this article can also be exploited for metabolically unhealthy lean COVID-19 patients.
Obesity is known as a predisposing factor for developing asthma because of the presence of elevated levels of cytokines, adipokines, and lipid-derived inflammatory mediators [13,14]. Activation of several inflammatory genes by nuclear factor kappa B (NF-κB) is also implicated in the obese asthmatic patients [15]. Besides, the SARS-CoV-2 infection of obese could further aggravate the condition by inducing altered innate immune response involving inflammasome activation. Studies have shown that RNA viruses could activate the nucleotide-binding domain, leucine-rich-containing family, pyrin domain-containing-3 (NLRP3) inflammasome through increasing potassium efflux [16]. Although not many studies have shown the same in the case of SARS-CoV-2, a recent study has implicated a likely mechanism involved in promoting NLRP3 based excessive innate immune response in SARS-CoV-2 patients [17]. In this article, we have discussed these and other mechanisms with an aim to enhance the understanding and develop an insight on the potential molecular targets for clinical intervention and management of COVID-19 severity in obese patients.
Pathways of inflammation
Inflammation is body’s biological response to defend itself from invading foreign entities such as pathogens and/or their toxins. It involves an organized response from the host’s structural and immune cells and secreted molecular signals. The invading foreign bodies act as stimulants that prime and activate immune response pathways [18,19]. Largely, the inflammatory pathways involve conventional transcription factors including signal transducer and activator of transcription (STAT), NF-κB, and hypoxia inducing factor-1α (HIF-1α) that are turned on by different stimuli and further generate effector response by these immune cells. NF-κB is expressed in various cell types where it can be activated in the cytoplasm by canonical and non-canonical signaling pathways. The end products of NF-κB inflammatory response are pro-inflammatory genes expression such as that of IL-1, IL-6, IL-8, TNF-α, monocytes chemoattractant protein-1 (MCP-1), CXCL-10, CXCL1 etc. The activation of canonical NF-κB inflammatory response is mediated through the ligand and receptor interaction of the pathogen-associated molecular patterns/pattern recognition receptors (PAMP/PRR). The non-canonical NF-κB signaling pathway activation is mediated via ligand-receptor interaction of TNF-α/tumor necrosis factor receptor (TNFR). It may be noted here that NF-κB mediated inflammatory pathway response is linked to the acute phase of the inflammation where the homeostasis is reached quickly.
The myeloid cells, a part of innate immune cells, after infiltrating the site of injury or inflammation, may encounter a low oxygen condition. In order to better survive and perform their functions, these cells rely upon induction of HIF-1α, a fundamental regulator of hypoxia that also regulates pro-inflammatory cytokine genes. Reports show that both TNFα and IL-1β, through the downstream signaling, can induce the expression of HIF-1α via NF-κB activation [20].
STAT-based inflammatory immune response is widely mediated by the interferons (IFN). IFNs are pro-inflammatory cytokines that render host protection by inducing the expression of antiviral proteins. Viral-PAMPs such as the DNA or RNA can trigger IFN production, which activate signaling cascade for lodging antiviral protein response in an autocrine or paracrine manner [21]. Type I, II, and III interferons differentially regulate the immune response through Janus associated kinases (JAK) and STAT pathway eventually stimulating the interferon stimulated genes (ISGs) [21], concerned with antiviral response protein such as 2′5′ oligoadenylate synthetase [22].
The purview of this article is to understand the mechanisms of acute phase of inflammation lodged by the immune cells and their mediators in the disease states such as obesity and COVID-19. Both COVID-19 and obesity could be viewed as inflammatory pathologies with different etiologies, making it essential to highlight the pathways and inflammatory mediators involved [8,23]. This would help devise effective strategies for intervention in obese as well as metabolically ill lean COVID-19 patients in determining the better clinical outcome.
Mediators of inflammation
The key inflammatory signaling pathways through which immune response is generated were discussed in the previous section. These signaling cascades culminate in the synthesis and release of inflammatory mediators. Out of the several mediators that are found elevated in the obese, the significant ones such as TNF-α, IL-6, and IL-1β could be implicated in the severity of COVID-19 in obese patients.
TNF-α
In the acute inflammatory response TNF-α is believed to play an important pro-inflammatory role [24]. Activated macrophages and T cells are the prominent producers of TNF-α which acts through two different receptors, TNFR-1 and TNFR-2 expressed on the target cells [25]. TNF-α signaling cascade activates the transcription factors such as NF-κB and activated protein (AP)-1 which can further induce the expression of other pro-inflammatory gene targets [26]. Recently, it was found that TNF-α can activate the coagulation of platelets in whole blood samples from humans, potentiating its role in thrombosis [27]. Interestingly, obesity-associated prothrombic state has been reported particularly in visceral adiposity that mediates obesity-associated thrombosis [28,29]. Further, the clinical cases of COVID-19 patients have also depicted the characteristics of thrombosis [30]. Since serum concentration of TNF-α is elevated in COVID-19 patients [31], and also during low-grade chronic inflammation in the obese, inflammation and thrombosis could both be attributed to the increased TNF-α activity. Therefore, serum TNF-α could be considered as potential biomarker in acute respiratory distress syndrome (ARDS) in obese COVID-19 patients [32], and thus can be targeted to control the severity of the disease.
IL-6
One of the important functions of IL-6 as a mediator of inflammation is the induction & secretion of acute phase proteins (APP) from hepatocytes. APPs such as c-reactive proteins, serum amyloid A, haptoglobin act to either inhibit or destroy the invading pathogens and have a systemic response. IL-6, a pro-inflammatory cytokine with pleiotropic roles, can be secreted at the site of injury or infection by the activated monocytes/macrophages that have encountered the PAMP and triggered PRR-mediated immune response and activation of downstream mediators such as NF-κB [33]. Acting via JAK/STAT signal transduction pathway IL-6 can activate STAT-3, which causes attenuation of adhesion and chemoattractant protein-coding genes such as intercellular adhesion molecule-1 (ICAM-1) and MCP-1, respectively [34]. In this way, IL-6 acts as a pro-inflammatory mediator of acute inflammation. Visceral fat in the obese was characterized for the secretion of IL-6 and found to be associated with the co-morbidities such as diabetes & MS [35]. Also, in the severe cases of COVID-19, serum concentration of IL-6 was found elevated [36] and was reportedly a good prognostic indicator of mortality associated with COVID-19 [37]. Thus, IL-6 can be targeted for better clinical outcome of the disease [38].
IL-1β
Studies have shown that priming and activation of the inflammasome complex and subsequent activation of caspase-1 contribute towards the activation and secretion of IL-1β [39], a pleiotropic pro-inflammatory cytokine. In obese, the level of IL-1β is elevated and implicated in the development of systemic chronic inflammation and other comorbidities. Once secreted in the extracellular milieu, IL-1β binds to its receptor, IL-1R1 & activates NF-κB transcription factor to mediate an inflammatory response [40]. IL-1β, after getting secreted from the innate immune cells including the AECs upon virus infection can act as chemoattractant & aid in the clearance of pathogen-infected host cells. This response, depending on the viral load of SARS-CoV-2 and activities of infiltrated innate immune cells including macrophages, neutrophils, B cells, and T cells etc. can be severe in obese patients leading to cytokine storm, which if not controlled, can be fatal [41].
Inflammation as a part of obesity
Before the COVID-19 pandemic, there was another silent pandemic of obesity which has affected millions across the globe. According to the CDC, people with BMI between 25–30 kg/m2 are considered overweight and those with >30 kg/m2 are obese [42]. Obesity is driven by excessive intake of calorie-rich diet, sedentary lifestyle and in some cases genetic predisposition which promotes positive energy balance and subsequently dis-balanced adipocyte homeostasis [43,44]. Low-grade systemic chronic inflammation during obesity has been identified as an underlying feature of the MS and can influence the clinical outcome of other pathogenetic conditions in the patients including COVID-19 [45]. Increased visceral adipose tissue mass affects the resident adipocytes, pre-adipocytes, residential immune cells, and endothelial cells [46]. Interaction among these cellular players gives way to the establishment of a chronic pro-inflammatory state, which is discussed below briefly.
Adipocytes
The adipocytes may contribute towards the development of a pro-inflammatory state of the adipose tissues in two ways. Firstly, owing to their endocrine nature, adipocytes are known to secrete adipokines that have pleiotropic effects. Leptin and resistin are pro-inflammatory adipokines secreted by the adipocytes in the white adipose tissue (WAT) of obese individuals. Leptin signaling can activate effector response in macrophages to secrete IL-6, IL-18, IL-12, and TNF-α via transcription factor STAT-3 activation [47]. Secondly, hypoxia may prevail in the adipose tissues due to adipocytes hypertrophy and hyperplasia, which may further enhance inflammation. The hypoxic adipocytes die through necrosis and induce infiltration of the leukocytes and further augmentation of inflammation [48]. These conditions weaken the innate immune response and may not display appropriate defense against the pathogens like Coronavirus and result in severe consequences for the host.
Resident and recruited macrophages
As specified earlier, prevalent hypoxia in the adipose tissues promotes the infiltration of circulatory macrophages. Upon infiltration these macrophages convert into M1 type of inflammatory macrophages and release pro-inflammatory cytokines and chemokines such as IL-6, TNF-α, and IL-1β [49]. The presence of preexisting pro-inflammatory M1 polarization in obese may further aggravate the macrophages response to the invading virus leading to increased severity of COVID-19.
Other myeloid cells
Mast cells infiltrate the adipose tissue in the early stage of obesity and influence adipocytes behavior and function by secreting IFN-ϒ and IL-6 [50,51]. The dendritic cells, which are professional antigen-presenting cells, may stimulate Th17 response by aiding in the chemoattraction of macrophages and neutrophils in the obese adipose tissues [52]. Thus, they may drive the vicious cycle of immune cell infiltration in the adipose tissues and promotion of pro-inflammatory milieu in obese patients.
Lymphoid cells
The B cells are also reported to infiltrate the adipose tissues at the early onset of obesity [53]. They can regulate the function of macrophages and T cells that are already present in the adipose tissues. The data suggest that activated B cells in the obese adipocytes can produce auto-antibodies that can induce CD8+ & CD4+ T cells to produce IFN-ϒ [54]. It is already known that IFN-ϒ can induce a positive feedback loop in the macrophages to produce TNF-α and other pro-inflammatory cytokines in the adipose tissues [55].
Thus, with the prevalence of an obese state, pro-inflammatory mediators such as cytokines, adipokines, chemokines, and pro-inflammatory immune cells can escalate to systemic inflammation. A resultant systemic chronic inflammation may affect multiple organ systems of the body leading to poor clinical outcomes in obese COVID-19 patients.
Obesity and metabolic syndrome
MS can be defined as a combination of clinical features such as insulin resistance and elevated levels of blood glucose, cholesterol or triglycerides, and blood pressure that converge to a poor clinical condition in obese [56]. Obesity is now considered as a defining factor for the progression of MS where the underlying low-grade chronic inflammation predisposes the obese to develop MS-associated comorbidities [45]. In obese patients, the pro-inflammatory cytokines may destabilize insulin receptor substrate (IRS) signaling leading to the development of insulin resistance. TNF-α is known to promote insulin resistance in the obese adipocytes through several inhibitory serine phosphorylation events. After binding to its receptor TNFR-1, it can activate a downstream enzyme called sphingomyelinase which may give rise to ceramides & phosphocholine [57]. The ceramides can induce the ceramide activated kinases, which lead to serine phosphorylation of the Insulin receptor [58]. Further, TNF-α can also activate JNK that may lead to serine phosphorylation of IRS-1, a crucial downstream protein of the insulin receptor [59].
IL-6 is also implicated in the induction of insulin resistance in insulin-sensitive tissue microenvironment via activation of STAT-3. Once activated, STAT-3 transcribes the gene coding for suppressor of cytokine signaling-3 (SOCS-3) protein. In the cytoplasm, SOCS-3 protein interacts with the phosphotyrosine binding domain of IRS-1 or it may directly phosphorylate the insulin receptor and inhibit it [60]. Insulin resistance and/or metabolic syndrome manifested in obese individuals may also increase their risk for the COVID-19 [61]. Thus, it becomes pertinent for the obese patients to shed the body weight by following a healthy diet and lifestyle and secure a better metabolic health.
SARS-CoV-2 and COVID-19
SARS-CoV-2 is the new variant of the erstwhile SARS-CoV virus that caused the SARS pandemic in 2003 and has been identified as a causative agent for COVID-19 in humans [62]. When the infectious nature and mortality associated with SARS-CoV are contrasted with SARS-CoV-2, it turns out that SARS-CoV-2 is more contagious in causing infection and fatality [63,64]. Further, unlike SARS-CoV that used to colonize lungs, SARS-CoV-2 is perceived to colonize the upper respiratory system with the capability of steering down towards the lungs [65].
One of the pathological features of COVID-19 is the virus-induced cytopathy as evident from the biopsies of the SARS-CoV-2 infected lung tissues [66]. Apart from that, pyroptosis, a caspase-1 mediated inflammatory cell death in the virus-infected epithelial as well as infiltrating myeloid cells, has also been reported [67]. Further, this pathological event can cause the secretion of several chemokines and cytokines such as macrophage inflammatory protein-1α (MIP-1α), MIP-1β, IL-6, MCP-1 and interferon-gamma induced-protein-10 (IP-10) [68]. The released chemokines and cytokines can drive the vicious cycle of immune cell infiltration leading to cytokine storm and further organ damage. Usually, this is the late stage of COVID-19, which due to an exaggerated host response, may lead to the development of ARDS and pneumonia [69].
Since obese and overweight already possess systemic low-grade inflammation therefore, it is clinically important to understand the impact of COVID-19 in these patients and vice versa for better clinical management of patients.
Obesity and COVID-19
The prevalence of obesity has been on a continuous rise globally, and given its impact on respiratory ailments, it becomes critical to underline the effects it may have on COVID-19. In the past as well, the prevalence of obesity amongst hospitalized cases of H1N1 has been shown to correlate with the poor prognosis and fatality [70]. Unfortunately, in the initial Chinese reports, the BMI of the hospitalized COVID-19 patients was not considered sufficiently. However, later on a BMI of >40 kg/m2 emerged as an important factor that needed to be taken into consideration for the early hospitalization of the COVID-19 patients [71]. Emerging data on the increase in the prevalence of obesity amongst hospitalized COVID-19 patients has also attracted the attention of clinicians. Simonnet et al., (2020) conducted a retrospective cohort study to determine the requirement of mechanical ventilator support by the inpatients with COVID-19 based on their BMI. It was reported that the requirement of mechanical ventilator support increased parallelly with the increase in BMI of COVID-19 patients, thus indicating a strong correlation of the disease severity with obesity [72]. The prevalence of obesity amongst severe and critical patients with or without COVID-19 in an ICU setup was also analyzed by Caussy et al., (2020) who reported a 2.8% increase in the prevalence of obesity in those with COVID-19 [73]. Since obesity is known to alter the homeostasis of the lungs and affect respiratory health via different mechanisms, the emerging data of its prevalence as comorbidity in hospitalized cases of COVID-19 highlight its importance as a factor worth considering for early hospitalization and care of obese COVID-19 patients. The odds of obese patients developing a severe form of COVID-19 were 3.4 folds high when compared to the non-obese counterparts admitted at a designated hospital in China [74]. Also, when the clinical features of the COVID-19 patients hospitalized at a University Hospital Aachen were analyzed, apart from the preexisting respiratory ailments, obesity was found to increase the chances of developing the ARDS and prolong the hospitalization [75]. Several of the adipose tissue residential cells such as adipocytes, stromal cells, endothelial cells and macrophages are reported to be a target of different viruses such as the H1N1, the adenoviruses or the Type A Influenza [76]. Further, since these cells also express angiotensin converting enzyme 2 (ACE-2) receptors [77], it could be speculated that obese patients may have a higher viral load for SARS-CoV-2 and thus an increased virus shedding. It is reported that several RNA viruses such as vesicular stomatitis virus or encephalomyocarditis can activate NLRP3 inflammasome via potassium ion efflux and promote the secretion of IL-1β [16]. Other RNA viruses have also been shown to mediate NLRP3 inflammasome activation by the viral RNA and subsequent IL-1β release [78]. Since SARS-CoV-2 is an RNA virus, it would be rational to contemplate that after infecting the host cells its RNA may interact with the toll-like receptor 7/8. Following this, there might be a subsequent NLRP3 inflammasome priming and activation that can ultimately lead to IL-1β secretion. Therefore, the SARS-CoV-2 infection of obese could further aggravate the condition by inducing altered innate immune response involving inflammasome activation & contemplating the role of molecular players like NLRP3 inflammasome can bring more clarity to understand the less studied inflammatory pathologies in many COVID-19 patients. Indeed, a recent study has implicated the involvement of this pathway in COVID-19 patients and shown primary evidence of NLRP3 activation [17]. With obese adipocytes already infiltrated with the macrophages and owing to the evidence of SARS-CoV-2 tropism towards adipose tissue [79], activation of NLRP3 inflammasome and successive IL-1β release may aid to the cytokine storm. The low-grade systemic chronic inflammation in the obese is implicated in endothelial dysfunction, which is considered a common feature of the associated comorbidities such as hypertension, type 2 diabetes, and dyslipidemia [80]. Since endothelial cells express ACE-2 receptors, a site for SARS-CoV-2 entry and infection, they demonstrate endothelial inflammation in the laboratory confirmed cases of COVID-19 [81]. Moreover, the low-grade chronic inflammation is also known to activate prothrombotic signaling in the vascular endothelial cells in the obese. Apart from that, the adipocytes of the obese individuals may get infected with SARS-CoV-2 and undergo necrotic cell death liberating free fatty acids in the adipose interstitium which may further predispose the obese COVID-19 patients to fat embolism syndrome [82]. Hence, the direct recruitment of SARS-CoV-2 to endothelium or the adipocytes as well as preexisting systemic chronic inflammation in the obese may lead to severe form of the COVID-19.
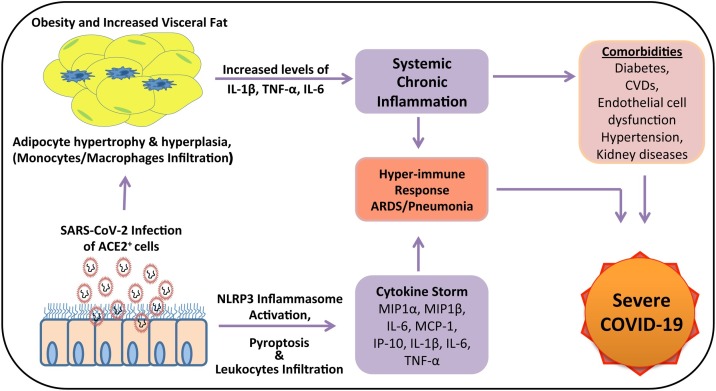
Although many of the molecular and clinical aspects of COVID-19 are either under investigation or yet to be studied, the potential interrelationship between COVID-19 and obesity and disease mechanism in obese patients has been summarized in Fig. 1 , which will be helpful in developing potential cellular and molecular therapeutic targets for clinically beneficial outcome in severe to critical obese COVID-19 patients.
Fig. 1.
A schematic representation of the interrelationship between COVID-19 and obesity. SARS-CoV-2 utilizes ACE-2 receptors to infect a wide variety of cells. The cytopathic effect of the infection on the AECs leads to the activation of inflammasome complex and the secretion of the pro-inflammatory mediators culminating into the cytokine storm. The obese or metabolically ill lean patients with preexisting systemic chronic inflammation and comorbidities like diabetes, CVDs, endothelial dysfunction, hypertension, chronic kidney diseases when infected with the virus, may develop severe COVID-19 symptoms due to the release of more chemokines and cytokines by accentuating the immune response. (IP-10, Interferon gamma-induced protein 10; MCP-1, Monocyte chemoattractant protein-1; IL-1β, Interleukin-1beta; TNF-α, Tumor necrosis factor-alfa; IL-6, Interleukin-6; ACE-2, Angiotensin converting enzyme-2. CVDs, Cardiovascular diseases; SARS-CoV-2, Severe acquired respiratory syndrome-coronavirus-2; ARDS, Acute respiratory distress syndrome; COVID-19, Coronavirus disease-2019).
Potential cellular and molecular targets to decrease COVID-19 severity in obese patients
As the cases of infection and mortality are rising unabated on a daily basis, understanding the potential cellular and molecular targets for the development of drugs or therapeutic strategies to manage COVID-19 is a race against the time. As discussed below some important molecular targets and strategies are already under investigation for developing potential therapeutics, while some additional cellular and molecular targets can also be explored as new potential strategies for clinical intervention and management of COVID-19 severity in obese patients.
Viral components and host response elements
Since we know that SARS-Cov-2 is efficient in utilizing its spike protein’s receptor binding domain to interact with the host ACE-2 receptor, this interaction could be molecularly targeted. Arbidol, a drug that can target this interaction, has entered a clinical trial in China (Clinical Trial Identifier: NCT04273763). It is reported to block the fusion of viral envelope with host cell membrane by targeting the viral glycoproteins [83]. Arbidol is an approved drug for Influenza where it is illustrated to bind with haemagglutinin glycoprotein of influenza virus. It can thus stop the fusion of virus with the host cells preventing an infection [84]. Further, a membrane-based protease, transmembrane protease serine 2 (TMPRSS2), that helps SARS-CoV-2 fuse with the host cell membrane, can also be a crucial target. The action of this serine protease could be blocked by utilizing its inhibitor to prevent SARS-CoV-2 from entering the host cells. A clinically proven protease inhibitor camostat mesylate has been recently shown to effectively engage with the TMPRSS2 on the AECs. Camostat mesylate treatment to the AECs showed a significantly reduced entry of SARS-CoV-2 inside the cells and its efficacy in blocking the TMPRSS2 did not cause any cytotoxic effects [85].
Once inside the host cells, SARS-CoV-2 exploits the host machinery for completion of the infection cycle through its constituent factors such as viral RNA and other proteins. One such viral factor is the enzyme RNA dependent RNA polymerase (RdRP) which catalyzes the synthesis of viral RNA strands from the template viral-genomic RNA. Since RdRP uses the host nucleotides to synthesize the new viral genomic-RNA strands, it could be targeted via nucleotide analog molecules. Nucleoside and nucleotide analogs such as Ribavirin, Remdesivir, and Favipiravir are found to be effective towards blocking the function of RdRP and can thus be exploited to target the SARS-CoV-2 replication [86]. Favipiravir in the combination of other drugs (Clinical Trial Identifier: NCT04303299) and Remdesivir (Clinical Trial Identifier: NCT04257656) are being investigated for their safety and wide-scale applicability in different clinical trials. Form these, Remdesivir showed a reduction in the recovery time in COVID-19 patients [87] and thus has been issued an emergency use authorization by the FDA as a potential treatment option for COVID-19 [88].
Other SARS-CoV-2 factors such as replicase polyproteins, which are crucial for virus replication and transcription, are subjected to proteolytic processing and maturation by the viral proteases. The main protease (Mpro) of SARS-CoV-2 seems to be involved in this proteolytic processing of the crucial replicase polyproteins and is highlighted as a potential target for therapeutic development. Following the molecular modeling approach Kumar et al., (2020) have recently demonstrated an efficient binding efficiency of two phytochemicals, withanone and caffeic acid phenethyl ester with the Mpro of SARS-CoV-2. Their studies concluded that these two phytochemicals were significantly similar to a known protease inhibitor of Mpro and may bind and inhibit the Mpro activity leaving it functionless. Thus, withanone and caffeic acid phenethyl ester may be utilized as potential therapeutic drugs in COVID-19 [89].
Many of the pro-inflammatory cytokines are enhanced in the hyper-inflammatory response of the host during COVID-19 where IL-6 has been highlighted to be a central player [90]. Tocilizumab and Sarilumab are the monoclonal antibodies that are both IL-6 receptor antagonists and FDA approved agents to be utilized for treatment against rheumatoid arthritis [91]. These monoclonal antibodies work by blocking IL-6 signaling cascade and preventing the pro-inflammatory response of these cytokines. Since COVID-19 patients deal with an enhanced IL-6 plasma level, such antibodies can help dampen the heightened immune response and manage the symptoms. To test the efficacy and assess the side effects, Sarilumab (Clinical Trial Identifier: NCT04315298) and Tocilizumab (Clinical Trial Identifier: ChiCTR2000030196) have thus been into clinical trials and found helpful in decreasing the severity of COVID-19 in obese patients. Since the host response to SARS-CoV-2 may lead to an inflammatory damage to the body organs, although contraindicated, preliminary results of dexamethasone use for COVID-19 have shown significant efficacy. At randomization, dexamethasone was found to reduce one third of the mortality amongst the severe and critical cases of COVID-19 that needed mechanical ventilator support [92].
Targeting acute inflammatory response
In the following sections, we have highlighted several other host response factors that are known to be involved in the attenuation of pro-inflammatory state, and thus can be the potential therapeutic targets in COVID-19 patients.
Receptors mediating innate immune response
The receptors responsible for innate immune responses act via recognizing the pathogens or relay a signal downstream for an effector immune response. These receptors, as discussed earlier, are called PRRs and can recognize the PAMPs leading to the assembly and activation of inflammasome. We have further implicated the possible involvement of NLRP3 class of inflammasome activation and secretion of IL-1β in COVID-19. This implication is in line with the increased concentration of pro-inflammatory cytokine IL-1β in COVID-19 patients. Since aberrant NLRP3 activation in obese patients may lead to excessive secretion of IL-1β and cause sterile inflammation that may aggravate COVID-19 inflammatory pathology, it is rational to target IL-1β and improve the prognosis of COVID-19. Anakinra, an IL-1β receptor antagonist, and approved agent for the treatment of rheumatoid arthritis [93], could be repurposed for targeting the inflammation caused by IL-1β in critically ill obese COVID-19 patients. Targeting these receptors in the early stage of COVID-19 illness may turn out to be beneficial in case of obese patients as this can stop the cytokines storm in the bud.
Inflammatory cell death and immune responses
SARS-CoV-2 is a cytopathic virus and may thus lead to pyroptosis of the infected host cells leading to extensive tissue damage and aggravated inflammation. One of the characteristic features of pyroptosis is the activation of inflammasome complex and induction of gasdermin D (GSDMD) protein. The activated GSDMD after processing from inflammasome can form pores in the host cells membrane leading to the leakage of pro-inflammatory cytokines [94]. If this particular protein can be exploited as a molecular target in COVID-19, the spillage of pro-inflammatory cytokines and chemokines could be blocked at the source. By doing so, the pro-inflammatory response could be dampened. Disulfiram, an approved drug used to manage alcohol addiction, is reported to deactivate GSDMD pore-forming protein in pyroptotic cells. It causes the modification of cysteine residue on GSDMD, ultimately blocking the pore formation and further cytokine release [95]. Hence, targeting activated GSDMD in SARS-CoV-2 infected cells undergoing pyroptosis can be another potential therapeutic strategy.
Targeting augmentation of the anti-inflammatory immune response
Resolution of inflammation
An aberrant inflammatory response can lead to several forms of devastating effects on the tissues and organs. Pathological features of aberrant and dysregulated inflammation have been depicted in patients with COVID-19. Undertaking a different strategy to tackle the inflammatory pathogenesis, targeting the resolution of inflammation (RoI) seems quite viable option. RoI is mediated by anti-inflammatory lipid mediators that prevent the infiltration of immune cells at the site of inflammation, promotes the clearance of apoptotic cells, and ultimately reduce the availability of pro-inflammatory cytokines. RoI mediating lipid mediators are the metabolites derived from omega-3 fatty acid such eicosapentaenoic acid and docosahexaenoic acid e.g. resolvin, protectin and maresin. Levels of these anti-inflammatory mediators are reportedly low in obese people that give way to pro-inflammatory stimuli an upper hand leading to delay in RoI. These metabolites of omega-3 fatty acids act on the respective immune cells via G-protein coupled receptors (GPCRs) expressed on them such as ChemR23 (expressed on dendritic cells and monocytes), GPR37 and LGR-6 [[96], [97], [98]]. Targeting these receptors by developing their agonists and timely induction of RoI in an aberrant inflammatory response such as COVID-19 may also seem a promising approach to tackle COVID-19 severity in obese patients.
Metabolic targets in pro-inflammatory immune response
Immune cell metabolism is an important player in driving immune cells mediated inflammatory response [99]. The pro-inflammatory mediators are believed to activate the metabolic switch leading to a dependence on the glycolysis for these cells just like in the case of the Warburg effect observed in the cancer cells [99]. Ultimately, a dependence on glycolysis leads to the activation of pro-inflammatory phenotypes in these cells [100]. Such a phenomenon of metabolic reprogramming could thus be targeted to develop therapeutics that can drive the pro-inflammatory cells towards a more anti-inflammatory state and regulate the hyper immune response such as seen in the critical cases of COVID-19. Dimethyl fumarate (DMF), an approved drug candidate for the treatment of Multiple sclerosis, is effective in reprogramming the pro-inflammatory response of the activated immune cells towards an anti-inflammatory state [101]. DMF is reported to reprogramme the classically activated, pro-inflammatory M1 macrophage towards the anti-inflammatory M2 macrophage. Further, DMF has been shown to achieve the switching of M1 to M2 macrophages and Th1 to Treg phenotype, respectively by targeting a key enzyme called glyceraldehyde 3-phosphate dehydrogenase (GAPDH), active in the glycolysis. Furthermore, DMF is reported to cause the succinylation of cysteine residues at the active site of GAPDH ultimately inhibiting its function [102]. Since the activation of the macrophages and Th1 cells is dependent on glycolysis, inhibition of GAPDH, a rate limiting enzyme, may alter their fate and push them towards an anti-inflammatory M2 and Treg types. Since the active pro-inflammatory macrophages have been implicated in COVID-19 related cytokine storm [103], targeting metabolic reprogramming of these cells to achieve an anti-inflammatory state warrants further exploration. Another advantage of this cellular target could be the selectivity in blocking certain activated immune cells only as compared to the broad-spectrum inhibition of inflammatory response as seen with steroid based drugs.
Finally, it is being emphasized that the selection of the targets to devise therapeutic strategies should be based on the stages of the COVID-19 illness. For example, in the early stage of SARS-CoV-2 infection, halting the replication of the virus can be effective in activating a judicious host immune response. In the late stage of the illness that is driven by the host-mediated pro-inflammatory response, its effective suppression can be beneficial. Thus, novel therapeutics could be designed keeping into consideration this fine balance and the associated side effects of the treatment. It can be further speculated that the obese COVID-19 patients might respond much better to the therapies designed keeping into consideration the late stage of the illness. This is owing to the possibility of an involvement of obesity-induced chronic systemic inflammation in aggravating the host response in the late stage of COVID-19. Some molecular players discussed above, such as NLRP3 inflammasome activation, pyroptosis, and the augmentation of pro- and anti-inflammatory cytokines could be exploited as possible cellular and molecular targets to devise therapeutics for COVID-19, especially for the obese as well as metabolically ill lean COVID-19 patients. A few of these prominent and potential molecular targets have been summarized below in Table 1 .
Table 1.
Summary of plausible pharmacological intervention in COVID-19 patients with obesity.
| S. No. | Target | Therapy | Effects | Ref. |
|---|---|---|---|---|
| 1. | TMPRSS | Camostat Mesylate (FOY-305) | Blocks TMPRSS activity, reducing entry of SARS-CoV-2 inside host cells | [84] |
| 2. | ACE-2 | Umifenovir (Arbidol) | Blocks the fusion of viral envelope with host cell membrane | [82] [83] |
| 3. | RdRPs | Ribavirin, Remdesivir, and Favipiravir | Blocks the function of RdRPs and thus checks the incorporation of nucleotides in newly synthesized viral RNA | [85] |
| 4. | Main protease (Mpro) | Withanone and Caffeic Acid-Phenethyl Ester | Demonstrated to prevent the proteolytic processing of the crucial polyproteins involved with viral replication & transcription | [88] |
| 5. | IL-6 | Tocilizumab and Sarilumab | IL-6 receptor agonist may prevent the IL-6 mediated signaling cascade & proinflammatory response | [90] |
| 6. | IL-1β | Anakinra | IL-1β agonist may prevent the proinflammatory activity of heightened IL-1β in the organs and tissues during SARS-CoV-2 infection | [93] |
| 7. | Gasdermin- D | Disulfiram | Inactivate gasdermin- D activity and thus prevent pro-inflammatory cytokine release from the inflammatory dead cells | [95] |
| 8. | Glyceraldehyde 3-phosphate dehydrogenase | Dimethyl fumarate | Reprograms the metabolism in the pro-inflammatory macrophages and Tells so as to push them towards an anti-inflammatory state | [101] |
Conclusion
The pandemic of COVID-19 involves a novel Coronavirus-2 induced respiratory ailment that has shown serious repercussions on the people suffering from comorbidities such as obesity and associated metabolic illnesses. Thus, we have reviewed the available literature and clinical studies and highlighted some of the potential and novel cellular and molecular targets that can be explored, understood, and exploited to develop new therapeutic strategies for better clinical outcome in obese and metabolically ill patients with SARS-CoV-2 infection.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Authors statement
Umesh C.S. Yadav: Conceptualized, edited, supervised and finalised the manuscript.
Jiten R. Sharma: Performed literature search, drafted and revised the manuscript.
Acknowledgment
JS acknowledges junior research fellowship (JRF) from UGC, New Delhi, India.
References
- 1.World Health Organisation . 2020. WHO announces COVID-19 outbreak a pandemic. [Available from: http://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/news/news/2020/3/who-announces-covid-19-outbreak-a-pandemic. [Accessed 20 May 2020] [Google Scholar]
- 2.World Health Organisation . 2020. Weekly epidemiological update - 15 December 2020. [Available from: https://www.who.int/publications/m/item/weekly-epidemiological-update---15-december-2020. [Accessed 18 Dec 2020] [Google Scholar]
- 3.Center for Disease Control . 2020. Coronavirus disease 2019 (COVID-19) [Available from: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/groups-at-higher-risk.html#severe-obesity. [Accessed 20 May 2020] [Google Scholar]
- 4.Stefan N., Birkenfeld A.L., Schulze M.B., Ludwig D.S. Obesity and impaired metabolic health in patients with COVID-19. Nat Rev Endocrinol. 2020;16(7):341–342. doi: 10.1038/s41574-020-0364-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.World obesity Federation . 2019. Prevelance of obesity. [Available from: https://www.worldobesity.org/about/about-obesity/prevalence-of-obesity. [Accessed 20 May 2020] [Google Scholar]
- 6.Stefan N., Häring H.-U., Schulze M.B. Metabolically healthy obesity: the low-hanging fruit in obesity treatment? Lancet Diabetes Endocrinol. 2018;6(3):249–258. doi: 10.1016/S2213-8587(17)30292-9. [DOI] [PubMed] [Google Scholar]
- 7.Stefan N. Causes, consequences, and treatment of metabolically unhealthy fat distribution. Lancet Diabetes Endocrinol. 2020;8(7):616–627. doi: 10.1016/S2213-8587(20)30110-8. [DOI] [PubMed] [Google Scholar]
- 8.Saltiel A.R., Olefsky J.M. Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest. 2017;127(1):1–4. doi: 10.1172/JCI92035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ashraf H., Laway B.A., Afroze D., Wani A.I. Evaluation of proinflammatory cytokines in obese vs non-obese patients with metabolic syndrome. Indian J Endocrinol Metab. 2018;22(6):751–756. doi: 10.4103/ijem.IJEM_206_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dholia N., Yadav U.C.S. Lipid mediator Leukotriene D4-induces airway epithelial cells proliferation through EGFR/ERK1/2 pathway. Prostaglandins Other Lipid Mediat. 2018;136:55–63. doi: 10.1016/j.prostaglandins.2018.05.003. [DOI] [PubMed] [Google Scholar]
- 11.Mann J.P., Savage D.B. What lipodystrophies teach us about the metabolic syndrome. J Clin Invest. 2019;129(10):4009–4021. doi: 10.1172/JCI129190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eigentler T., Lomberg D., Machann J., Stefan N. Lipodystrophic Nonalcoholic Fatty Liver Disease Induced by Immune Checkpoint Blockade. Ann Intern Med. 2020;172(12):836–837. doi: 10.7326/L19-0635. [DOI] [PubMed] [Google Scholar]
- 13.Dholia N., Sethi G.S., Naura A.S., Yadav U.C. Cysteinyl leukotriene D 4 (LTD 4) promotes airway epithelial cell inflammation and remodelling. Inflamm Res. 2020:1–18. doi: 10.1007/s00011-020-01416-z. [DOI] [PubMed] [Google Scholar]
- 14.Yadav U., Srivastava S. Cysteinyl leukotrienes (CysLTs): role in obesity-induced asthma. Curr Mol Med. 2015;15(7):598–605. doi: 10.2174/1566524015666150831130954. [DOI] [PubMed] [Google Scholar]
- 15.Wood L.G., Garg M.L., Gibson P.G. A high-fat challenge increases airway inflammation and impairs bronchodilator recovery in asthma. J Allergy Clin Immunol. 2011;127(5):1133–1140. doi: 10.1016/j.jaci.2011.01.036. [DOI] [PubMed] [Google Scholar]
- 16.da Costa L.S., Outlioua A., Anginot A., Akarid K., Arnoult D. RNA viruses promote activation of the NLRP3 inflammasome through cytopathogenic effect-induced potassium efflux. Cell Death Dis. 2019;10(5):346. doi: 10.1038/s41419-019-1579-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roschewski M., Lionakis M.S., Sharman J.P., Roswarski J., Goy A., Monticelli M.A. Inhibition of Bruton tyrosine kinase in patients with severe COVID-19. Sci Immunol. 2020;5(48):eabd0110. doi: 10.1126/sciimmunol.abd0110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rivera A., Siracusa M.C., Yap G.S., Gause W.C. Innate cell communication kick-starts pathogen-specific immunity. Nat Immunol. 2016;17(4):356–363. doi: 10.1038/ni.3375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tartey S., Takeuchi O. Pathogen recognition and Toll-like receptor targeted therapeutics in innate immune cells. Int Rev Immunol. 2017;36(2):57–73. doi: 10.1080/08830185.2016.1261318. [DOI] [PubMed] [Google Scholar]
- 20.Rius J., Guma M., Schachtrup C., Akassoglou K., Zinkernagel A.S., Nizet V. NF-kappaB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1alpha. Nature. 2008;453(7196):807–811. doi: 10.1038/nature06905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mesev E.V., LeDesma R.A., Ploss A. Decoding type I and III interferon signalling during viral infection. Nat Microbiol. 2019;4(6):914–924. doi: 10.1038/s41564-019-0421-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liao X., Xie H., Li S., Ye H., Li S., Ren K. 2’, 5’-Oligoadenylate synthetase 2 (OAS2) inhibits zika virus replication through activation of type I IFN signaling pathway. Viruses. 2020;12(4):418. doi: 10.3390/v12040418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hirano T., Murakami M. COVID-19: a new virus, but a familiar receptor and cytokine release syndrome. Immunity. 2020;52(5):731–733. doi: 10.1016/j.immuni.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Akdis M., Aab A., Altunbulakli C., Azkur K., Costa R.A., Crameri R. Interleukins (from IL-1 to IL-38), interferons, transforming growth factor β, and TNF-α: receptors, functions, and roles in diseases. J Allergy Clin Immunol. 2016;138(4):984–1010. doi: 10.1016/j.jaci.2016.06.033. [DOI] [PubMed] [Google Scholar]
- 25.Mitoma H., Horiuchi T., Tsukamoto H., Ueda N. Molecular mechanisms of action of anti-TNF-α agents – comparison among therapeutic TNF-α antagonists. Cytokine. 2018;101:56–63. doi: 10.1016/j.cyto.2016.08.014. [DOI] [PubMed] [Google Scholar]
- 26.Bouwmeester T., Bauch A., Ruffner H., Angrand P.-O., Bergamini G., Croughton K. A physical and functional map of the human TNF-α/NF-κB signal transduction pathway. Nat Cell Biol. 2004;6(2):97–105. doi: 10.1038/ncb1086. [DOI] [PubMed] [Google Scholar]
- 27.Page M.J., Bester J., Pretorius E. The inflammatory effects of TNF-α and complement component 3 on coagulation. Sci Rep. 2018;8(1):1812. doi: 10.1038/s41598-018-20220-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Blokhin I.O., Lentz S.R. Mechanisms of thrombosis in obesity. Curr Opin Hematol. 2013;20(5):437–444. doi: 10.1097/MOH.0b013e3283634443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chitongo P.B., Roberts L.N., Yang L., Patel R.K., Lyall R., Luxton R. Visceral Adiposity Is an Independent Determinant of Hypercoagulability as Measured by Thrombin Generation in Morbid Obesity. TH Open: Companion Journal Thrombosis Haemostasis. 2017;1(2) doi: 10.1055/s-0037-1608942. e146-e54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Klok F.A., Kruip M., Van der Meer N., Arbous M., Gommers D., Kant K. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis. Thromb Res. 2020 doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Leija-Martínez J.J., Huang F., Del-Río-Navarro B.E., Sanchéz-Muñoz F., Muñoz-Hernández O., Giacoman-Martínez A. IL-17A and TNF-α as potential biomarkers for acute respiratory distress syndrome and mortality in patients with obesity and COVID-19. Med Hypotheses. 2020;144 doi: 10.1016/j.mehy.2020.109935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Slaats J., ten Oever J., van de Veerdonk F.L., Netea M.G.J.Pp. IL-1β/IL-6/CRP and IL-18/ferritin: distinct inflammatory programs in infections. PLoS Pathog. 2016;12(12) doi: 10.1371/journal.ppat.1005973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ljungberg L.U., Zegeye M.M., Kardeby C., Fälker K., Repsilber D., Sirsjö A.J.Mo I. Global Transcriptional Profiling Reveals Novel Autocrine Functions of Interleukin 6 in Human Vascular Endothelial Cells. Mediators Inflamm. 2020;2020 doi: 10.1155/2020/4623107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.van Greevenbroek M.M., Schalkwijk C.G., Stehouwer C.D. Obesity-associated low-grade inflammation in type 2 diabetes mellitus: causes and consequences. Neth J Med. 2013;71(4):174–187. [PubMed] [Google Scholar]
- 36.Gao Y., Li T., Han M., Li X., Wu D., Xu Y. Diagnostic utiliT.y of clinical laboratory data determinations for patients with the severe COVID-19. J Med Virol. 2020;92(7):791–796. doi: 10.1002/jmv.25770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020 doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Xu X., Han M., Li T., Sun W., Wang D., Fu B. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci. 2020;117(20):10970. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Martín-Sánchez F., Diamond C., Zeitler M., Gomez A.I., Baroja-Mazo A., Bagnall J. Inflammasome-dependent IL-1β release depends upon membrane permeabilisation. Cell Death Differ. 2016;23(7):1219–1231. doi: 10.1038/cdd.2015.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Weber A., Wasiliew P., Kracht M. Interleukin-1 (IL-1) pathway. Sci Signal. 2010;3(105):cm1. doi: 10.1126/scisignal.3105cm1. [DOI] [PubMed] [Google Scholar]
- 41.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20(5):269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.2020. Defining adult overweight and obesity: division of nutrition, physical activity, and obesity, national center for chronic disease prevention and health promotion. [Available from: https://www.cdc.gov/obesity/adult/defining.html [Acessed 27 May 2020] [Google Scholar]
- 43.Trayhurn P.J.B. Origins and early development of the concept that brown adipose tissue thermogenesis is linked to energy balance and obesity. Biochimie. 2017;134:62–70. doi: 10.1016/j.biochi.2016.09.007. [DOI] [PubMed] [Google Scholar]
- 44.Konttinen H., Llewellyn C., Silventoinen K., Joensuu A., Männistö S., Salomaa V. Genetic predisposition to obesity, restrained eating and changes in body weight: a population-based prospective study. Int J Obes. 2018;42(4):858–865. doi: 10.1038/ijo.2017.278. [DOI] [PubMed] [Google Scholar]
- 45.Wiebe N., Stenvinkel P., Tonelli M.J.Jno. Associations of chronic inflammation, insulin resistance, and severe obesity with mortality, myocardial infarction, cancer, and chronic pulmonary disease. JAMA Network Open. 2019;2(8) doi: 10.1001/jamanetworkopen.2019.10456. e1910456-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Martyniak K., Masternak M.M. Changes in adipose tissue cellular composition during obesity and aging as a cause of metabolic dysregulation. Exp Gerontol. 2017;94:59–63. doi: 10.1016/j.exger.2016.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Desai H.R., Sivasubramaniyam T., Revelo X.S., Schroer S.A., Luk C.T., Rikkala P.R. Macrophage JAK2 deficiency protects against high-fat diet-induced inflammation. Sci Rep. 2017;7(1):7653. doi: 10.1038/s41598-017-07923-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lee C.H., Lam K.S.J.Jodi. Obesity‐induced insulin resistance and macrophage infiltration of the adipose tissue: a vicious cycle. J Diabetes Investig. 2019;10(1):29. doi: 10.1111/jdi.12918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Acedo S.C., Gambero S., Cunha F.G.P., Lorand-Metze I., Gambero A. Participation of leptin in the determination of the macrophage phenotype: an additional role in adipocyte and macrophage crosstalk. Vitr Cell Dev Biol - Anim. 2013;49(6):473–478. doi: 10.1007/s11626-013-9629-x. [DOI] [PubMed] [Google Scholar]
- 50.Zhou Y., Yu X., Chen H., Sjöberg S., Roux J., Zhang L. Leptin deficiency shifts mast cells toward anti-inflammatory actions and protects mice from obesity and diabetes by polarizing M2 macrophages. Cell Metab. 2015;22(6):1045–1058. doi: 10.1016/j.cmet.2015.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Elieh Ali Komi D., Shafaghat F., Christian M. Crosstalk between mast cells and adipocytes in physiologic and pathologic conditions. Clin Rev Allergy Immunol. 2020;58(3):388–400. doi: 10.1007/s12016-020-08785-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chen Y., Tian J., Tian X., Tang X., Rui K., Tong J. Adipose tissue dendritic cells enhances inflammation by prompting the generation of Th17 cells. PLoS One. 2014;9(3) doi: 10.1371/journal.pone.0092450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Duffaut C., Galitzky J., Lafontan M., Bouloumié A.J.B. Communications br. Unexpected trafficking of immune cells within the adipose tissue during the onset of obesity. Biochem Biophys Res Commun. 2009;384(4):482–485. doi: 10.1016/j.bbrc.2009.05.002. [DOI] [PubMed] [Google Scholar]
- 54.Winer D.A., Winer S., Shen L., Wadia P.P., Yantha J., Paltser G. B cells promote insulin resistance through modulation of T cells and production of pathogenic IgG antibodies. Nat Med. 2011;17(5):610. doi: 10.1038/nm.2353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Salim T., Sershen C.L., May E.E.J.Po. Investigating the role of TNF-α and IFN-γ activation on the dynamics of iNOS gene expression in LPS stimulated macrophages. PLoS One. 2016;11(6) doi: 10.1371/journal.pone.0153289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Saklayen M.G.J.Chr. The global epidemic of the metabolic syndrome. Curr Hypertens Rep. 2018;20(2):12. doi: 10.1007/s11906-018-0812-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vandenabeele P., Declercq W., Beyaert R., Fiers W. Two tumour necrosis factor receptors: structure and function. Trends Cell Biol. 1995;5(10):392–399. doi: 10.1016/s0962-8924(00)89088-1. [DOI] [PubMed] [Google Scholar]
- 58.Peraldi P., Hotamisligil G.S., Buurman W.A., White M.F., Spiegelman B.M.J.Jo B.C. Tumor necrosis factor (TNF)-α inhibits insulin signaling through stimulation of the p55 TNF receptor and activation of sphingomyelinase. J Biol Chem. 1996;271(22):13018–13022. doi: 10.1074/jbc.271.22.13018. [DOI] [PubMed] [Google Scholar]
- 59.Solinas G., Becattini B.J.Mm. JNK at the crossroad of obesity, insulin resistance, and cell stress response. Journal of Molecular Metabolism. 2017;6(2):174–184. doi: 10.1016/j.molmet.2016.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sarvas J.L., Khaper N., Lees S.J. The IL-6 paradox: context dependent interplay of SOCS3 and AMPK. J Diabetes Metab. 2013;(Suppl 13) doi: 10.4172/2155-6156.S13-003. 10.4172/2155-6156.S13-003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bansal R., Gubbi S., Muniyappa R. Metabolic syndrome and COVID 19: endocrine-immune-Vascular interactions shapes clinical course. Endocrinology. 2020;161(10) doi: 10.1210/endocr/bqaa112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lai C.-C., Shih T.-P., Ko W.-C., Tang H.-J., Hsueh P.-R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and corona virus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents. 2020 doi: 10.1016/j.ijantimicag.2020.105924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chu H., Chan J.F.-W., Yuen T.T.-T., Shuai H., Yuan S., Wang Y. Comparative tropism, replication kinetics, and cell damage profiling of SARS-CoV-2 and SARS-CoV with implications for clinical manifestations, transmissibility, and laboratory studies of COVID-19: an observational study. The Lancet Microbe. 2020 doi: 10.1016/S2666-5247(20)30004-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Xu J., Zhao S., Teng T., Abdalla A.E., Zhu W., Xie L. Systematic comparison of two animal-to-human transmitted human coronaviruses: SARS-CoV-2 and SARS-CoV. Viruses. 2020;12(2):244. doi: 10.3390/v12020244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zou L., Ruan F., Huang M., Liang L., Huang H., Hong Z. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med. 2020;382(12):1177–1179. doi: 10.1056/NEJMc2001737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Vabret N., Britton G.J., Gruber C., Hegde S., Kim J., Kuksin M. Immunology of COVID-19: current state of the science. Immunity. 2020;52(6):910–941. doi: 10.1016/j.immuni.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tay M.Z., Poh C.M., Rénia L., MacAry P.A., Ng L.F. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020:1–12. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Gattinoni L., Chiumello D., Rossi S. COVID-19 pneumonia: ARDS or not? Crit Care. 2020;24(1):154. doi: 10.1186/s13054-020-02880-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Poulakou G., Pérez M., Rello J. Severe acute respiratory infections in the postpandemic era of H1N1. Curr Opin Crit Care. 2012;18(5):441–450. doi: 10.1097/MCC.0b013e32835605f2. [DOI] [PubMed] [Google Scholar]
- 71.Petrilli C.M., Jones S.A., Yang J., Rajagopalan H., O’Donnell L., Chernyak Y. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. bmj. 2020;369:m1966. doi: 10.1136/bmj.m1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Simonnet A., Chetboun M., Poissy J., Raverdy V., Noulette J., Duhamel A. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity. 2020;28(7):1195–1199. doi: 10.1002/oby.22831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Caussy C., Pattou F., Wallet F., Simon C., Chalopin S., Telliam C. Prevalence of obesity among adult inpatients with COVID-19 in France. Lancet Diabetes Endocrinol. 2020 doi: 10.1016/S2213-8587(20)30160-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cai Q., Chen F., Wang T., Luo F., Liu X., Wu Q. Obesity and COVID-19 severity in a designated hospital in Shenzhen. China. Diabetes Care. 2020;43(7):1392–1398. doi: 10.2337/dc20-0576. [DOI] [PubMed] [Google Scholar]
- 75.Dreher M., Kersten A., Bickenbach J., Balfanz P., Hartmann B., Cornelissen C. The characteristics of 50 hospitalized COVID-19 patients with and without ARDS. Deutsches Ärzteblatt International. 2020;117(10):271. doi: 10.3238/arztebl.2020.0271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ryan P.M., Caplice N.M. Is adipose tissue a reservoir for viral spread, immune activation, and cytokine amplification in coronavirus disease 2019? Obesity. 2020;28(7):1191–1194. doi: 10.1002/oby.22843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Gupte M., Thatcher S.E., Boustany-Kari C.M., Shoemaker R., Yiannikouris F., Zhang X. Angiotensin converting enzyme 2 contributes to sex differences in the development of obesity hypertension in C57BL/6 mice. Arterioscler Thromb Vasc Biol. 2012;32(6):1392–1399. doi: 10.1161/ATVBAHA.112.248559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Allen I.C., Scull M.A., Moore C.B., Holl E.K., McElvania-TeKippe E., Taxman D.J. The NLRP3 inflammasome mediates in vivo innate immunity to influenza A virus through recognition of viral RNA. Immunity. 2009;30(4):556–565. doi: 10.1016/j.immuni.2009.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Pasquarelli-do-Nascimento G., Braz-de-Melo H.A., Faria S.S., Santos Id O., Kobinger G.P., Magalhães K.G. Hypercoagulopathy and adipose tissue exacerbated inflammation may explain higher mortality in COVID-19 patients with obesity. Front Endocrinol (Lausanne) 2020;11(530) doi: 10.3389/fendo.2020.00530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lobato N.S., Filgueira F.P., Akamine E.H., Tostes R.C., Carvalho M.H.C., Fortes Z.B. Mechanisms of endothelial dysfunction in obesity-associated hypertension. Braz J Med Biol Res. 2012;45(5):392–400. doi: 10.1590/S0100-879X2012007500058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cinti S., Graciotti L., Giordano A., Valerio A., Nisoli E. COVID-19 and fat embolism: a hypothesis to explain the severe clinical outcome in people with obesity. Int J Obes. 2020;44(8):1800–1802. doi: 10.1038/s41366-020-0624-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Vankadari N. Arbidol: a potential antiviral drug for the treatment of SARS-CoV-2 by blocking trimerization of the spike glycoprotein. Int J Antimicrob Agents. 2020;56(2) doi: 10.1016/j.ijantimicag.2020.105998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Pshenichnaya N.Y., Bulgakova V.A., Lvov N.I., Poromov A.A., Selkova E.P., Grekova A.I. Clinical efficacy of umifenovir in influenza and ARVI (study ARBITR) Therapeutic Archive. 2019;91(3):56–63. doi: 10.26442/00403660.2019.03.000127. [DOI] [PubMed] [Google Scholar]
- 85.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2) doi: 10.1016/j.cell.2020.02.052. 271-80.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Elfiky A.A. Ribavirin, Remdesivir, Sofosbuvir, Galidesivir, and Tenofovir against SARS-CoV-2 RNA dependent RNA polymerase (RdRp): A molecular docking study. Life Sci. 2020;253 doi: 10.1016/j.lfs.2020.117592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Beigel J.H., Tomashek K.M., Dodd L.E., Mehta A.K., Zingman B.S., Kalil A.C. Remdesivir for the treatment of Covid-19 — preliminary report. N Engl J Med. 2020 doi: 10.1056/NEJMc2022236. [DOI] [PubMed] [Google Scholar]
- 88.Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment. [press release]. FDA, 2020 May 01.
- 89.Kumar V., Dhanjal J.K., Kaul S.C., Wadhwa R., Sundar D. Withanone and caffeic acid phenethyl ester are predicted to interact with main protease (Mpro) of SARS-CoV-2 and inhibit its activity. J Biomol Struct Dyn. 2020:1–17. doi: 10.1080/07391102.2020.1772108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.McGonagle D., Sharif K., O’Regan A., Bridgewood C. The Role of Cytokines including Interleukin-6 in COVID-19 induced Pneumonia and Macrophage Activation Syndrome-Like Disease. Autoimmun Rev. 2020;19(6) doi: 10.1016/j.autrev.2020.102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Choy E., Caporali R., Xavier R., Fautrel B., Sanmarti R., Bao M. Subcutaneous tocilizumab in rheumatoid arthritis: findings from the common-framework phase 4 study programme TOZURA conducted in 22 countries. Rheumatology. 2018;57(3):499–507. doi: 10.1093/rheumatology/kex443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Group TRC Effect of dexamethasone in hospitalized patients with COVID-19: preliminary report. N Engl J Med. 2020 [Google Scholar]
- 93.Dinarello C.A., Simon A., van der Meer J.W.M. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov. 2012;11(8):633–652. doi: 10.1038/nrd3800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Shi J., Zhao Y., Wang K., Shi X., Wang Y., Huang H. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature. 2015;526(7575):660–665. doi: 10.1038/nature15514. [DOI] [PubMed] [Google Scholar]
- 95.Hu J.J., Liu X., Xia S., Zhang Z., Zhang Y., Zhao J. FDA-approved disulfiram inhibits pyroptosis by blocking gasdermin D pore formation. Nat Immunol. 2020;21(7):736–745. doi: 10.1038/s41590-020-0669-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Cash J.L., Norling L.V., Perretti M. Resolution of inflammation: targeting GPCRs that interact with lipids and peptides. Drug Discov Today. 2014;19(8):1186–1192. doi: 10.1016/j.drudis.2014.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hansen T.V., Vik A., Serhan C.N. The protectin family of specialized pro-resolving mediators: potent immunoresolvents enabling innovative approaches to target obesity and diabetes. Front Pharmacol. 2019;9(1582) doi: 10.3389/fphar.2018.01582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Chiang N., Libreros S., Norris P.C., de la Rosa X., Serhan C.N. Maresin 1 activates LGR6 receptor promoting phagocyte immunoresolvent functions. J Clin Invest. 2019;129(12):5294–5311. doi: 10.1172/JCI129448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Jha A.K., Huang S.C.-C., Sergushichev A., Lampropoulou V., Ivanova Y., Loginicheva E. Network integration of parallel metabolic and transcriptional data reveals metabolic modules that regulate macrophage polarization. Immunity. 2015;42(3):419–430. doi: 10.1016/j.immuni.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 100.Viola A., Munari F., Sánchez-Rodríguez R., Scolaro T., Castegna A. The metabolic signature of macrophage responses. Front Immunol. 2019;10:1462. doi: 10.3389/fimmu.2019.01462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bomprezzi R. Dimethyl fumarate in the treatment of relapsing-remitting multiple sclerosis: an overview. Ther Adv Neurol Disord. 2015;8(1):20–30. doi: 10.1177/1756285614564152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Angiari S., O’Neill L.A. Dimethyl fumarate: targeting glycolysis to treat MS. Cell Res. 2018;28(6):613–615. doi: 10.1038/s41422-018-0045-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Moore J.B., June C.H. Cytokine release syndrome in severe COVID-19. Science. 2020;368(6490):473–474. doi: 10.1126/science.abb8925. [DOI] [PubMed] [Google Scholar]