To the Editor:
The COVID-19 pandemic is a worldwide health and socioeconomic burden requiring optimization of disease prevention, treatment, and management strategies. Here, this review of physiologic rationales, basic science and translational data, and preliminary clinical results suggests the involvement of C1 esterase inhibitor-related processes in COVID-19 pathology and specifies opportunities for further investigation.
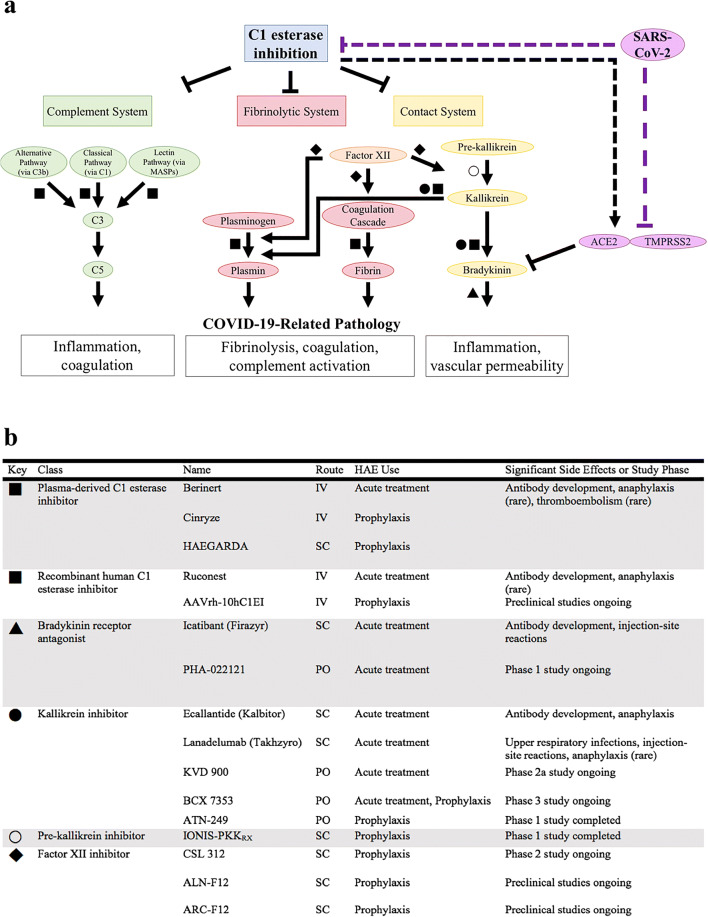
C1 esterase inhibition regulates the complement, fibrinolytic, and contact systems (Fig. 1a). These cascades stimulate a range of immunologic and hematologic processes including inflammation, coagulation, and fibrinolysis, all of which are activated in COVID-19 [1–3]. In the complement system, C1 esterase inhibitor (C1-INH) protein binds to and inactivates C1r and C1s of the classical pathway, C3b of the alternative pathway, and mannose-binding lectin-associated serine proteases (MASPs) of the lectin pathway [4, 5]. This inhibition reduces complement-driven inflammation and coagulation. In the fibrinolytic and contact systems, C1-INH inhibits components of the coagulation cascade, plasmin, and kallikrein to affect both coagulation and fibrinolysis. Additionally, bradykinin, downstream of kallikrein, is an integral mediator of the contact system that increases vascular permeability.
Fig. 1.
C1 esterase inhibitor-related cellular processes and therapeutic targets. a C1 esterase inhibition physiologically regulates the complement, fibrinolytic, and contact systems, all of which may be involved in COVID-19 pathology. SARS-CoV-2 could dysregulate these systems by direct interaction with C1 esterase inhibitor or ACE2 and TMPRSS2. b Multiple medications target C1 esterase inhibitor-related pathways for management of hereditary angioedema, which is caused by C1 esterase inhibitor deficiency or dysfunction. Dash: hypothesized mechanism; HAE, hereditary angioedema; IV, intravenous; SC, subcutaneous; PO, oral
Numerous medications involved in C1-INH pathways are clinically available for the treatment of hereditary angioedema (HAE), which is caused by deficient or dysfunctional C1-INH (Fig. 1b) [6, 7]. COVID-19 and HAE share clinical overlap with symptoms such as shortness of breath, diarrhea, abdominal pain, and facial swelling [8]. In HAE, increased vascular permeability results in angioedema that can manifest as facial and extremity swelling or mucosal edema of the respiratory and gastrointestinal tracts. It has been hypothesized that the dry cough, ground-glass lung opacities, and sensitivity to fluid overload in COVID-19 may be related to pulmonary angioedema [9–11]. Mechanistically, COVID-19 and HAE share innate immune activation that results in inflammation, endothelial dysfunction, and fibrinolysis. In the terminal complement cascade, dysregulation can result in thrombotic microangiopathy characterized by thrombosis and organ injury, a common finding in COVID-19 [1]. The terminal complement cascade can be initiated by the classical pathway via the C1 complex, the alternative pathway, and the lectin pathway.
Early use of C1 esterase inhibition for the treatment of COVID-19 has been promising. In Switzerland, administration of recombinant C1-INH reduced fever and inflammatory markers in patients with COVID-19, who had not improved despite hydroxychloroquine and antiviral therapies [12]. In the Netherlands, inhibition of the contact system via icatibant, a bradykinin receptor antagonist, was associated with decreased oxygen requirements in patients with COVID-19 [13]. Applicability of these results is limited by their observational nature and the small number and male predominance of enrolled patients. Expanded trials with recombinant C1-INH, icatibant, and lanadelumab—a kallikrein inhibitor—are currently under investigation and will assess clinical outcomes in larger, randomized studies [14–16]. Although preliminary results are promising, C1 esterase inhibition may not be sufficient to significantly modulate all potential downstream effectors within the complement cascade, which can have multiple means of activation. Clinical trials of these downstream targets such as C3, C5, and C5a are currently under investigation [17]. Randomized controlled clinical trials are needed.
In vitro and bioinformatic studies suggest that SARS-CoV can interact with C1-INH and dysregulate its anti-inflammatory cellular processes. High-throughput yeast two-hybrid screening demonstrates interactions between human C1-INH and seven different SARS-CoV proteins [18, 19]. C1-INH is predicted to interact directly with ACE (61% similar to ACE2) and TMPRSS1 (50% similar to TMPRSS2) [20]. Interestingly, bradykinin, a downstream effector of the contact system, is a substrate of the angiotensin-converting enzyme 2 (ACE2) protein that is required along with transmembrane protease, serine 2 (TMPRSS2) for SARS-CoV-2 cell entry [21]. In addition, C1-INH was identified amongst ten serum proteins that most accurately predict progression to respiratory distress in COVID-19 and amongst the six proteins predicted to interact with SARS-CoV-2 that are most broadly connected to expression of other genes [22, 23]. A genetic association study identified a SERPING1 single-nucleotide polymorphism (SNP) as one of seven complement-related SNPs most associated with adverse clinical outcomes in COVID-19 infection [24].
C1-INH may be suppressed in SARS-CoV infection, leading to unchecked inflammation. Antiviral interferon cytokines can induce C1-INH expression, and preclinical studies have shown that C1 esterase inhibition reduces inflammation [25, 26]. However, in vitro SARS-CoV infection increases interferon-stimulated gene expression—with the notable exceptions of ACE2 and SERPING1, the gene that encodes the serine protease inhibitor: C1-INH [27]. As a whole, the SERPING1-related gene networks are amongst the most upregulated in SARS-CoV infection [28]. In two genomic analyses of patients with COVID-19 infection, SERPING1 expression in bronchoalveolar lavage fluid (BALF) was decreased [29–31]. These BALF decreases are coupled with increases in SERPING1 blood expression in COVID-19 [23, 30, 32]. Another study showed that SERPING1 BALF expression was generally decreased in patients with non-SARS-CoV-2 community-acquired pneumonias, suggesting a broader role for C1-INH involvement in pneumonia [31]. Decreases in SERPING1 BALF expression are likely cell-specific processes, secondary to secretion from or consumption in resident lung cells. Serologic increases in SERPING1 expression likely represent a physiologic response rather than a pathologic propagation.
Administration of medications that target C1-INH pathways may supplement endogenous anti-inflammatory and antiviral efforts to improve COVID-19 disease courses. Medication repurposing utilizes the benefits of already-established pharmaceutical production protocols and awareness of medication pharmacokinetics and pharmacodynamics. It will be particularly important to carefully assess the risk-benefit profiles of such strategies in COVID-19. Lanadelumab, a kallikrein inhibitor, is associated with increased risk of upper respiratory tract infections in clinical trials [33]. There are conflicting preclinical reports about increased thrombogenesis with C1 esterase inhibitors [34].
Thrombosis also occurs in several complementopathies including paroxysmal nocturnal hemoglobinuria, atypical hemolytic uremic syndrome, and complement hyperactivation, angiopathic thrombosis, and protein-losing enteropathy (CHAPLE) syndrome [35]. All of these diseases respond to complement inhibition. These clinical observations and the association of SNPs in complementopathy-related genes with adverse COVID-19 outcomes may reflect complement-driven hypercoagulability and endothelial dysfunction as shared processes in COVID-19 and complement disorders [24]. Trials of various complement inhibition strategies in COVID-19 are ongoing. Targeting these pathways at a proximal step via C1-INH may be a promising approach.
Involvement of C1-INH-related pathways in COVID-19 pathology collectively appears rooted in basic science rationale, supporting translational data, and promising preliminary clinical results. The results from the aforementioned trials should prompt collaborative research into C1-INH-related mechanisms of disease etiology and therapy. Specifically, investigations could explore the biochemical confirmation of host and host-viral protein interactions, analysis of host genetics and protein expression to determine predictors of morbidity, clinical review of patients diagnosed with both COVID-19 and complement disorders, and expansion of randomized controlled studies. While social distancing, personal protective equipment, disease screening, and vaccine development are cornerstones of COVID-19 prevention, studies of therapeutic intervention should be concomitantly advanced. Such efforts will be critical to efficiently and effectively reduce the health and socioeconomic burdens of the COVID-19 pandemic.
Authors’ Contributions
TA wrote and prepared the manuscript. CC, LC, PO, and RK edited and reviewed the manuscript.
Data Availability
Not applicable
Compliance with Ethical Standards
Conflict of Interests
The authors declare that they have no conflict of interest.
Ethical Approval and Consent to Participate
Not applicable
Consent for Publication
Not applicable
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Campbell CM, Kahwash R. Will complement inhibition be the new target in treating COVID-19 related systemic thrombosis? Circulation. 2020;141:1739–1741. doi: 10.1161/CIRCULATIONAHA.120.047419. [DOI] [PubMed] [Google Scholar]
- 2.Risitano AM, Mastellos DC, Huber-Lang M, Yancopoulou D, Garlanda C, Ciceri F, Lambris JD. Complement as a target in COVID-19? Nat Rev Immunol. 2020;20:343–344. doi: 10.1038/s41577-020-0320-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tay MZ, Poh CM, Renia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jiang H, Wagner E, Zhang H, Frank MM. Complement 1 inhibitor is a regulator of the alternative complement pathway. J Exp Med. 2001;194(11):1609–1616. doi: 10.1084/jem.194.11.1609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Panagiotou A, Trendelenburg M, Osthoff M. The lectin pathway of complement in myocardial ischemia/reperfusion injury-review of its significance and the potential impact of therapeutic interference by C1 esterase inhibitor. Front Immunol. 2018;9:1151. doi: 10.3389/fimmu.2018.01151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Busse PJ, Christiansen SC. Hereditary angioedema. N Engl J Med. 2020;382(12):1136–1148. doi: 10.1056/NEJMra1808012. [DOI] [PubMed] [Google Scholar]
- 7.Cotiguala L, Coover K, Bewtra A, Malesker M. Advances in the pharmacotherapy of hereditary angioedema. US Pharmacist. 2015;40(10):HS17–HS21. [Google Scholar]
- 8.Batarseh E, Kersten BP, Pinelo AC, Nadler JN, Schwartz SA. Angioedema in African Americans patients hospitalized for COVID-19. Am J Respir Crit Care Med. 2020;202:1581–1584. doi: 10.1164/rccm.202006-2223LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Roche J, Roche R. An open letter to the scientific community on the possible role of dysregulated bradykinin signaling in COVID-19 respiratory complications. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tolouian R, Vahed S, Ghiyasvand S, Tolouian A, Ardalan M. COVID-19 interactions with angiotensin-converting enzyme 2 (ACE2) and the kinin system; looking at a potential treatment. J Renal Inj Prev. 2020;9(2):e19. doi: 10.34172/jrip.2020.19. [DOI] [Google Scholar]
- 11.van de Veerdonk FL, Netea MG, van Deuren M, van der Meer JWM, de Mast Q, Brüggemann RJ, van der Hoeven H. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. eLife. 2020;9:e57555. doi: 10.7554/eLife.57555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Urwyler P, Moser S, Charitos P, Heijnen I, Rudin M, Sommer G, et al. Treatment of COVID-19 with Conestat Alfa, a regulator of the complement, contact activation and kallikrein-kinin system. Front Immunol. 2020;11:2072. doi: 10.3389/fimmu.2020.02072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van de Veerdonk FL, Kouijzer IJE, de Nooijer AH, van der Hoeven HG, Maas C, Netea MG, Brüggemann RJM. Outcomes associated with use of a kinin B2 receptor antagonist among patients with COVID-19. JAMA Netw Open. 2020;3(8):e2017708. doi: 10.1001/jamanetworkopen.2020.17708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.I-SPY COVID-19 TRIAL: an adaptive platform trial for critically Ill patients. https://clinicaltrials.gov/show/NCT04488081.
- 15.Lanadelumab for treatment of COVID-19 disease. https://clinicaltrials.gov/show/NCT04422509.
- 16.Prevention of severe SARS-CoV-2 infection in hospitalized patients with COVID-19. https://clinicaltrials.gov/show/NCT04530136.
- 17.Campbell CM. The opening salvo of anti-complement therapy against COVID-19. Lancet Rheumatol. 2020;2:e729–e730. doi: 10.1016/S2665-9913(20)30353-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pfefferle S, Schopf J, Kogl M, Friedel CC, Muller MA, Carbajo-Lozoya J, et al. The SARS-coronavirus-host interactome: identification of cyclophilins as target for pan-coronavirus inhibitors. PLoS Pathog. 2011;7(10):e1002331. doi: 10.1371/journal.ppat.1002331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Srinivasan S, Cui H, Gao Z, Liu M, Lu S, Mkandawire W, et al. Structural genomics of SARS-CoV-2 indicates evolutionary conserved functional regions of viral proteins. Viruses. 2020;12:4. doi: 10.3390/v12040360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McDowall MD, Scott MS, Barton GJ. PIPs: human protein-protein interaction prediction database. Nucleic Acids Res. 2009;37(Database issue):D651–D656. doi: 10.1093/nar/gkn870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hoffmann M, Kleine-Weber H, Schroeder S, Kruger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Carnivali G. Analyzing characteristics of the genetic network generated by genes linked to COVID-19. InterAm J Med Health. 2020;3.
- 23.Shen B, Yi X, Sun Y, Bi X, Du J, Zhang C, et al. Proteomic and metabolomic characterization of COVID-19 patient sera. Cell. 2020;182:59–72.e15. doi: 10.1016/j.cell.2020.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ramlall V, Thangaraj PM, Meydan C, Foox J, Butler D, Kim J, May B, de Freitas JK, Glicksberg BS, Mason CE, Tatonetti NP, Shapira SD. Immune complement and coagulation dysfunction in adverse outcomes of SARS-CoV-2 infection. Nat Med. 2020;26(10):1609–1615. doi: 10.1038/s41591-020-1021-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Caccia S, Suffritti C, Carzaniga T, Berardelli R, Berra S, Martorana V, Fra A, Drouet C, Cicardi M. Intermittent C1-inhibitor deficiency associated with recessive inheritance: functional and structural insight. Sci Rep. 2018;8(1):977. doi: 10.1038/s41598-017-16667-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caliezi C, Wuillemin WA, Zeerleder S, Redondo M, Eisele B, Hack CE. C1-esterase inhibitor: an anti-inflammatory agent and its potential use in the treatment of diseases other than hereditary angioedema. Pharmacol Rev. 2000;52(1):91–112. [PubMed] [Google Scholar]
- 27.Gralinski LE, Baric RS. Molecular pathology of emerging coronavirus infections. J Pathol. 2015;235(2):185–195. doi: 10.1002/path.4454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guzzi PH, Mercatelli D, Ceraolo C, Giorgi FM. Master regulator analysis of the SARS-CoV-2/human interactome. J Clin Med. 2020;9:4. doi: 10.3390/jcm9040982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liao M, Liu Y, Yuan J, Wen Y, Xu G, Zhao J, Cheng L, Li J, Wang X, Wang F, Liu L, Amit I, Zhang S, Zhang Z. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020;26:842–844. doi: 10.1038/s41591-020-0901-9. [DOI] [PubMed] [Google Scholar]
- 30.Xiong Y, Liu Y, Cao L, Wang D, Guo M, Jiang A, Guo D, Hu W, Yang J, Tang Z, Wu H, Lin Y, Zhang M, Zhang Q, Shi M, Liu Y, Zhou Y, Lan K, Chen Y. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients. Emerg Microbes Infect. 2020;9(1):761–770. doi: 10.1080/22221751.2020.1747363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhou Z, Ren L, Zhang L, Zhong J, Xiao Y, Jia Z, Guo L, Yang J, Wang C, Jiang S, Yang D, Zhang G, Li H, Chen F, Xu Y, Chen M, Gao Z, Yang J, Dong J, Liu B, Zhang X, Wang W, He K, Jin Q, Li M, Wang J. Heightened innate immune responses in the respiratory tract of COVID-19 patients. Cell Host Microbe. 2020;27:883–890.e2. doi: 10.1016/j.chom.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yan Q, Li P, Ye X, Huang X, Mo X, Wang Q et al. Longitudinal peripheral blood transcriptional analysis of COVID-19 patients captures disease progression and reveals potential biomarkers. medRxiv. 2020:2020.05.05.20091355. doi:10.1101/2020.05.05.20091355.
- 33.Banerji A, Riedl MA, Bernstein JA, Cicardi M, Longhurst HJ, Zuraw BL, Busse PJ, Anderson J, Magerl M, Martinez-Saguer I, Davis-Lorton M, Zanichelli A, Li HH, Craig T, Jacobs J, Johnston DT, Shapiro R, Yang WH, Lumry WR, Manning ME, Schwartz LB, Shennak M, Soteres D, Zaragoza-Urdaz RH, Gierer S, Smith AM, Tachdjian R, Wedner HJ, Hebert J, Rehman SM, Staubach P, Schranz J, Baptista J, Nothaft W, Maurer M, for the HELP Investigators Effect of lanadelumab compared with placebo on prevention of hereditary angioedema attacks: a randomized clinical trial. JAMA. 2018;320(20):2108–2121. doi: 10.1001/jama.2018.16773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Crowther M, Bauer KA, Kaplan AP. The thrombogenicity of C1 esterase inhibitor (human): review of the evidence. Allergy Asthma Proc. 2014;35(6):444–453. doi: 10.2500/aap.2014.35.3799. [DOI] [PubMed] [Google Scholar]
- 35.Ozen A, Comrie WA, Ardy RC, Dominguez Conde C, Dalgic B, Beser OF et al. CD55 deficiency, early-onset protein-losing enteropathy, and thrombosis. N Engl J Med. 2017;377(1):52–61. doi: 10.1056/NEJMoa1615887. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable