Abstract
Background
Babies differ from older children with regard to their exposure to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). However, data describing the effect of SARS-CoV-2 in this group are scarce, and guidance is variable. We aimed to describe the incidence, characteristics, transmission, and outcomes of SARS-CoV-2 infection in neonates who received inpatient hospital care in the UK.
Methods
We carried out a prospective UK population-based cohort study of babies with confirmed SARS-CoV-2 infection in the first 28 days of life who received inpatient care between March 1 and April 30, 2020. Infected babies were identified through active national surveillance via the British Paediatric Surveillance Unit, with linkage to national testing, paediatric intensive care audit, and obstetric surveillance data. Outcomes included incidence (per 10 000 livebirths) of confirmed SARS-CoV-2 infection and severe disease, proportions of babies with suspected vertically and nosocomially acquired infection, and clinical outcomes.
Findings
We identified 66 babies with confirmed SARS-CoV-2 infection (incidence 5·6 [95% CI 4·3–7·1] per 10 000 livebirths), of whom 28 (42%) had severe neonatal SARS-CoV-2 infection (incidence 2·4 [1·6–3·4] per 10 000 livebirths). 16 (24%) of these babies were born preterm. 36 (55%) babies were from white ethnic groups (SARS-CoV-2 infection incidence 4·6 [3·2–6·4] per 10 000 livebirths), 14 (21%) were from Asian ethnic groups (15·2 [8·3–25·5] per 10 000 livebirths), eight (12%) were from Black ethnic groups (18·0 [7·8–35·5] per 10 000 livebirths), and seven (11%) were from mixed or other ethnic groups (5·6 [2·2–11·5] per 10 000 livebirths). 17 (26%) babies with confirmed infection were born to mothers with known perinatal SARS-CoV-2 infection, two (3%) were considered to have possible vertically acquired infection (SARS-CoV-2-positive sample within 12 h of birth where the mother was also positive). Eight (12%) babies had suspected nosocomially acquired infection. As of July 28, 2020, 58 (88%) babies had been discharged home, seven (11%) were still admitted, and one (2%) had died of a cause unrelated to SARS-CoV-2 infection.
Interpretation
Neonatal SARS-CoV-2 infection is uncommon in babies admitted to hospital. Infection with neonatal admission following birth to a mother with perinatal SARS-CoV-2 infection was unlikely, and possible vertical transmission rare, supporting international guidance to avoid separation of mother and baby. The high proportion of babies from Black, Asian, or minority ethnic groups requires investigation.
Funding
UK National Institute for Health Research Policy Research Programme.
Introduction
Children appear to be less severely affected by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) than adults.1 However, there is a paucity of data describing the effect of the virus on babies in the first 28 days after birth. Neonates are likely to differ from older groups in their exposure to the virus: although they can contract SARS-CoV-2 through close personal contact in much the same way as other groups, they might also contract the virus vertically before or at birth.2 The neonatal response to infection differs from that of older children and adults,3 and hence neonates might be more susceptible to infection or to severe disease.
The scarce data available suggest that severe disease might be more common in infants4 and neonates5 than in older children. However, these data are largely from case reports and series and, to the best of our knowledge, are not population-based. Consequently, the incidence of symptomatic neonatal SARS-CoV-2 infection and vertical transmission, features of presentation, severity, and outcomes at the population level are unknown.
The paucity of information about neonatal disease and concerns about SARS-CoV-2 transmission from mother to newborn6 have resulted in wide variation in guidance for the management of neonates at risk of SARS-CoV-2.7 In some countries, such as China, the immediate, routine separation of newborn infants from SARS-CoV-2-infected mothers is recommended, with no breastfeeding.7 By contrast, WHO recommendations8 and UK guidance9 support keeping mother and baby together and encouraging breastfeeding with hygiene precautions. Separating a mother and her newborn baby is likely to have deleterious consequences for mother–infant bonding, perinatal mental health,10 and breastfeeding.11 It is therefore crucial that, with the emergence of new diseases, policy is guided by robust and unbiased data.
Research in context.
Evidence before this study
This study was developed and initiated at the outset of the COVID-19 pandemic in the UK in March, 2020. At inception we searched PubMed and the preprint server MedRxiv using the terms “neonate”, “neonatal”, “SARS-CoV-2”, and “COVID-19”, from Jan 1, 2020, and without language limitations, for papers describing the incidence, presentation, and outcomes of neonatal severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. This search identified a single national population-based study of 2143 children with confirmed or suspected SARS-CoV-2 infection in China, in which the authors had developed a disease severity grading for children, and found that infants in the first year of life might have more severe disease compared with older children. Case reports and series were identified from China indicating possible vertical transmission of SARS-CoV-2 from mother to newborn baby.
A search was repeated using the same search terms and databases on July 28, 2020, with an additional search of the relevant Johns Hopkins Center for Humanitarian Health repository (COVID-19, Maternal and Child Health, Nutrition) using the search terms “neonate” and “neonatal”. Proposed criteria for vertical transmission of SARS-CoV-2 had been published, as well as further detailed case reports describing babies in whom vertical transmission was highly suggested. We found one systematic review of births following maternal SARS-CoV-2 infection, which included numerous case reports, single-centre case series, voluntary registries (at high risk of bias), and a single population-level study from the UK Obstetric Surveillance System that used active national surveillance. This UK-based study described 12 babies with SARS-CoV-2 following birth to mothers with confirmed infection, but did not describe clinical presentation or detailed information about diagnosis. A European multicentre voluntary registry study of paediatric SARS-CoV-2 infection reported 40 babies across 84 institutions, and a national cohort study of children with SARS-CoV-2 infection in the UK reported 53 babies. These studies described presentation, clinical course, and outcome across all paediatric groups, with little neonatal data, and were not sufficient to inform population-level incidence because of uncertainty about the completeness of case ascertainment and population coverage. We also identified guideline reviews showing that guidance for the management of babies born to women with confirmed or suspected SARS-CoV-2 infection varied between countries, with routine separation of mother and baby advised in many countries.
Added value of this study
Our study is the first national, active surveillance study of neonatal SARS-CoV-2 infection. We found that SARS-CoV-2 infection was uncommon among neonates receiving inpatient care, with only 66 cases identified in the UK between March and April, 2020. Most babies were mildly affected, with cases of severe disease being very rare. Infection requiring admission to a neonatal care unit following birth to a mother with perinatal SARS-CoV-2 infection was uncommon, with only 17 cases identified across the UK during the study period, and only two babies with possible vertical transmission were identified in the UK during the first peak of SARS-CoV-2 transmission.
Implications of all the available evidence
This study supports international guidance to avoid separation of mother and baby after birth in situations in which the mother has suspected or confirmed SARS-CoV-2 infection.
The aim of this study was to describe, on a population basis, the incidence, characteristics, transmission, and outcomes of SARS-CoV-2 infection in babies who received inpatient hospital care in the UK in the first 28 days after birth, in order to inform policy, ongoing management, and guidance for health-care professionals, pregnant women, and new parents.
Methods
Study design and procedures
A national prospective cohort study was carried out using the British Paediatric Surveillance Unit (BPSU).12 Established in 1985, the BPSU is a surveillance platform designed to carry out national studies of rare neonatal and paediatric diseases. From April 1, 2020, all consultant paediatricians in all 155 hospital trusts and health boards with their associated 190 neonatal units in the UK received a weekly electronic BPSU reporting card asking them to notify any eligible cases of neonatal SARS-CoV-2 infection diagnosed on or after March 1, 2020. Babies were eligible for inclusion if they had a diagnosis of SARS-CoV-2 infection made on a sample taken in the first 28 days after birth and received inpatient care on a postnatal ward, neonatal unit, paediatric inpatient ward, or paediatric intensive care unit (PICU). At the end of each month, an additional BPSU reporting card asked for confirmation that all eligible babies had been reported or to confirm zero reports for the month (active negative surveillance). During the study period, national policy did not recommend routine testing of neonates for SARS-CoV-2 in any clinical setting.
To maximise case ascertainment, data from Public Health England, Health Protection Scotland, the Paediatric Intensive Care Audit Network (PICANet),13 and the UK Obstetric Surveillance System (UKOSS)14 were used to identify any baby with a positive SARS-CoV-2 test taken in the first 28 days not reported through the BPSU. Following linkage, newly identified cases from these sources were followed up through local BPSU reporters and research nurses. Where cases identified through national testing data were unable to be matched to hospital records at the site of the test, they were categorised as not admitted for inpatient care and therefore excluded from the study. Deaths were verified with data from the MBRRACE-UK national surveillance of perinatal deaths.15
Following receipt of a report, notifying clinicians were asked to complete a data collection form (appendix 1) with details of the pregnancy, baby characteristics, neonatal management, and outcomes. Reporters who had not returned the form were contacted by email at 1, 2, and 4 weeks after notification. Data collection was supported by hospital-based research nurses from the UK's National Institute of Health Research (NIHR) Clinical Research Network following the study's adoption as an urgent public health priority study.
Parents, patients, and the public were consulted during the design of the study through the MBRRACE-UK third-sector stakeholder group, which comprises representatives from all the major mother and baby charities in the UK. The study protocol is available online.
The study was approved by the North East—Newcastle & North Tyneside 2 Research Ethics Committee (IRAS ID 282127; REC 20/NE/0107). Data were collected in England and Wales without parental consent following Section 251 advice from the Confidentiality Advisory Group of the Health Research Authority (20/CAG/0058) and under the COVID-19 notice issued by the Secretary of State for Health and Social Care under Regulation 3(4) of the Health Service Control of Patient Information Regulations 2002. Data were collected in Scotland without parental consent following COVID-19 rapid review and advice from the Public Benefit and Privacy Panel for Health and Social Care (1920-0288), and with maternal consent following advice from the Privacy Commissioner in Northern Ireland.
Outcomes and definitions
This analysis presents characteristics and outcomes for babies reported as having confirmed SARS-CoV-2 infection before 29 days of age between March 1 and April 30, 2020, and for whom complete data had been received by July 28, 2020.
For the purpose of this study, severe disease included both severe and critical disease, defined according to Dong and colleagues' criteria,4 which required that cases meet at least two of the following: (1) any of hyperthermia (>37·5°C), apnoea, cough, tachypnoea, respiratory distress or recession, supplemental oxygen requirement, poor feeding or vomiting, or diarrhoea; (2) any of low white blood cell count (<5 × 109/L), low lymphocyte count (<1 × 109/L), or raised C-reactive protein concentration (>5 mg/L); and (3) abnormal chest x-ray.
Possible vertically acquired infection was defined as a positive neonatal sample taken within the first 12 h following birth to a mother with confirmed SARS-CoV-2 infection; this definition is consistent with UKOSS criteria14 and with the criteria for probable or possible neonatal infection acquired intrapartum as defined by Shah and colleagues.16
Statistical analysis
Descriptive statistics are presented as frequencies, proportions, means, and standard deviations as appropriate. The incidence and 95% CIs of babies in hospital with confirmed SARS-CoV-2 infection were estimated using denominator estimates of total UK livebirths based on the most recently available (2018) complete national birth data for the four countries of the UK. Incidence according to gestational age at birth and ethnicity was also estimated using denominator estimates of livebirths in England and Wales in 2018.
The study is registered with ISRCTN (ISRCTN60033461).
Role of the funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
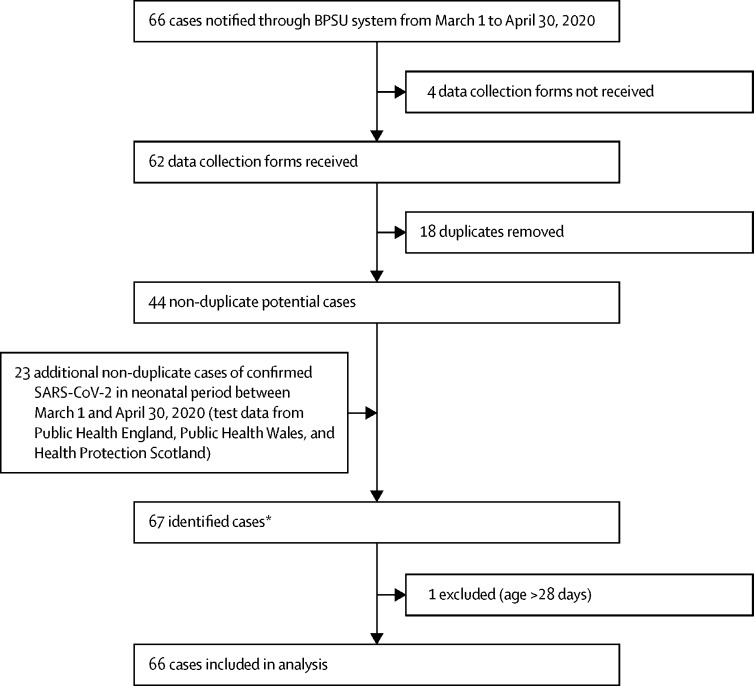
Monthly BPSU card returns were received from 3530 (87%) of 4063 UK paediatricians in March and 3480 (85%) of 4084 in April, 2020. 66 potentially eligible neonates were reported to the BPSU system over the study period and 23 additional babies were identified from other sources (figure 1 ).
Figure 1.
SARS-CoV-2 infection reporting profile
BPSU=British Paediatric Surveillance Unit. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2. *There were three more cases notified that were not confirmed as SARS-CoV-2 infection in the first 28 days because the date of birth or date of diagnosis was missing.
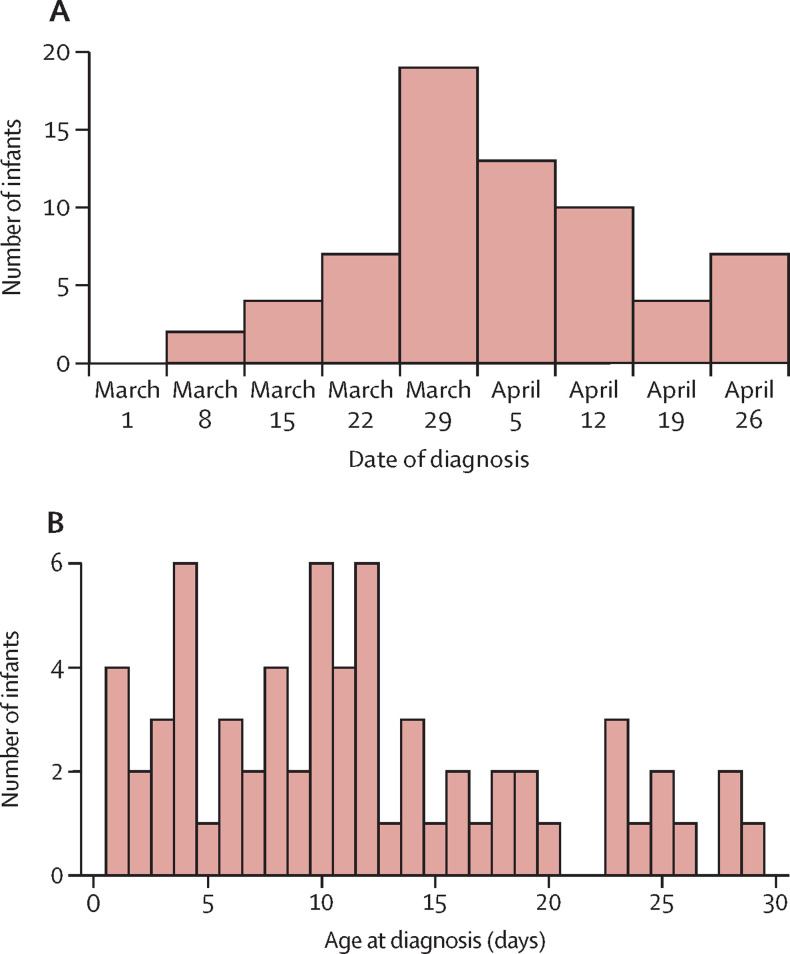
66 neonates with SARS-CoV-2 infection diagnosed between March 1 and April 30, 2020, received inpatient care in the UK. Over the same period, there were an estimated 118 347 livebirths, giving a neonatal incidence of SARS-CoV-2 infection of 5·6 (95% CI 4·3–7·1) per 10 000 livebirths. The England-specific incidence was 6·4 (4·9–8·2) per 10 000 livebirths. 28 babies in the UK met the criteria for severe paediatric SARS-CoV-2 infection4 (incidence 2·4 [1·6–3·4] per 10 000 births; table 1 ). Neonatal diagnoses peaked in early April (figure 2 ). The median age of diagnosis was 9·5 days (IQR 7·5–11·0) and 45 (68%) babies were diagnosed more than 7 days after birth (figure 2).
Table 1.
Incidence of SARS-CoV-2 infection in March and April, 2020
| Cases | Incidence of SARS-CoV-2 infection per 10 000 livebirths | ||
|---|---|---|---|
| Total | 66 | 5·6 (4·3–7·1) | |
| Gestational age at birth, weeks | |||
| ≥37 | 48 (73%) | 4·9 (3·6–6·5) | |
| 32 to <37 | 14 (21%) | 18·4 (9·8–31·4) | |
| 28 to <32 | 1 (2%) | 12·1 (0·3–67·0) | |
| <28 | 1 (2%) | 20·2 (0·5–112·5) | |
| Missing data | 2 (3%) | .. | |
| Ethnicity | |||
| White | 36 (55%) | 4·6 (3·2–6·4) | |
| Asian or Asian British | 14 (21%) | 15·2 (8·3–25·5) | |
| Black, African, Caribbean, or Black British | 8 (12%) | 18·0 (7·8–35·5) | |
| Mixed or other | 7 (11%) | 5·6 (2·2–11·5) | |
| Missing data | 1 (2%) | .. | |
| Severe disease* | 28 (42%) | 2·4 (1·6–3·4)† | |
Data are n (%) or incidence (95% CI). SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.
Cases meeting at least two of following: (1) any of hyperthermia (>37·5°C), apnoea, cough, tachypnoea, respiratory distress or recession, supplemental oxygen requirement, poor feeding or vomiting, or diarrhoea; (2) any of low white blood cell count, low lymphocyte count, or raised C-reactive protein concentration; and (3) abnormal chest x-ray.4
Incidence of severe disease per 10 000 livebirths in the population.
Figure 2.
Date of diagnosis (A) and age at diagnosis (B) of babies diagnosed with severe acute respiratory syndrome coronavirus 2 infection from March to April, 2020 (n=66)
17 (26%) babies with SARS-CoV-2 infection were born to a mother who had confirmed infection within 7 days before or after birth (table 2 ). Two of these babies (including one identified by UKOSS) had SARS-CoV-2 detected on a nasopharyngeal swab taken within 12 h of birth and were therefore deemed to have a possible vertically acquired infection; both had no further SARS-CoV-2 testing. UKOSS14 previously identified six babies as having possible vertically acquired infection using the same criterion (positive test taken within 12 h of birth to a mother with SARS-CoV-2 infection). These six cases were included in our study via database linkage, but further follow-up found that five of these cases were false positives; therefore, our final cohort only included one of these six babies, who had confirmed SARS-CoV-2 infection identified from a sample taken in the first 12 h after birth. Eight (47%) of the 17 cases in which both baby and mother had confirmed SARS-CoV-2 infection (including one baby with possible vertically acquired infection) had remained with their mother after birth. Seven (41%) had been separated immediately after birth, of whom three (including one preterm baby with possible vertically acquired infection) were admitted to a neonatal care unit. Where babies remained with their mother, the mother was advised regarding hand washing (to wash hands before touching the baby, breast pump, or bottles) and to wear a fluid-resistant surgical mask while handling the baby, and breastfeeding was recommended.9
Table 2.
Transmission of SARS-CoV-2 to neonates
| Cases (n=66) | |||
|---|---|---|---|
| Immediate family or close contacts with signs or symptoms of COVID-19 | |||
| Yes | 34 (52%) | ||
| No | 20 (30%) | ||
| Unsure | 12 (18%) | ||
| Mother confirmed to have SARS-CoV-2 infection at birth | 17 (26%) | ||
| Baby separated from mother immediately after birth | 7 (11%) | ||
| Not admitted to neonatal care unit | 4 (6%) | ||
| Admitted to neonatal care unit | 3 (5%) | ||
| Baby not separated from mother | 8 (12%) | ||
| Separation status not known | 2 (3%) | ||
| Possible vertically acquired infection* | 2 (3%) | ||
| Suspected nosocomially acquired infection† | 8 (12%) | ||
Data are n (%). Categories overlap and the potential source of transmission was not known or reported in some cases. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.
SARS-CoV-2 isolated from a sample taken from infant within 12 h of birth where mother was positive.
Not born to a mother with confirmed SARS-CoV-2 infection.
21 (32%) babies were diagnosed with SARS-CoV-2 infection in the first 7 days after birth, including 14 babies born to a mother with confirmed infection. Overall, 34 (52%) babies had immediate family or close contacts with signs or symptoms consistent with SARS-CoV-2 infection. Eight (12%) babies were suspected to have SARS-CoV-2 infection from nosocomial transmission and were not born to a mother with confirmed SARS-CoV-2 infection. Six (75%) instances of presumed nosocomial transmission, all in preterm babies, occurred more than 7 days after birth in babies hospitalised in neonatal units (six babies) or PICUs (one baby, who was also cared for in a neonatal unit) following preterm birth or a congenital condition. Further details of SARS-CoV-2 testing are presented in appendix 2 (p 5).
Of the 62 babies for whom sex was reported, 27 (44%) were male and 35 (56%) were female. 29 (45%) were from Black, Asian, or other minority ethnic (BAME) groups, an incidence of 11·1 (95% CI 7·4–15·9) compared with 4·6 (3·2–6·4) per 10 000 livebirths in babies from white ethnic groups (table 1). Although the majority of affected babies were born at term, incidence was highest in preterm infants (table 1).
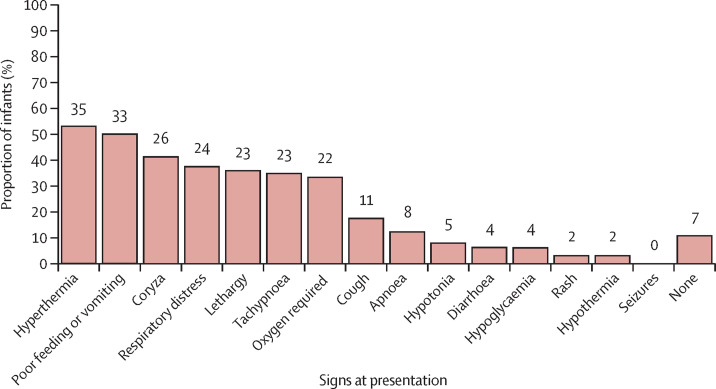
The most common signs at presentation were hyperthermia and poor feeding or vomiting. Coryza, respiratory signs, and lethargy were also commonly reported, and seven (11%) infants (all diagnosed in the first 7 days after birth) had no reported signs of infection, having only been tested following suspected maternal infection (figure 3 ). Hyperthermia, coryzal signs, and poor feeding were more commonly reported in babies diagnosed with SARS-CoV-2 later than 7 days after birth. Of the 25 babies who had a chest x-ray, 14 (56%) had abnormal findings, with ground-glass changes reported in seven (28%) babies (of whom four were preterm). The most common biochemical and haematological abnormalities were raised lactate (>2 mmol/L; 17 (55%) of 31 babies tested) and raised C-reactive protein (>5 mg/L; 14 (29%) of 49 babies). Five (9%) of 54 babies tested had a low lymphocyte count (<1 × 109/L). Further details are provided in appendix 2 (p 5). Eight babies underwent cranial ultrasound, and no abnormalities were detected.
Figure 3.
Clinical signs at presentation (n=66)
The number of patients in each category is shown above each bar. Percentages are calculated on non-missing data. Missing data: n=1 for apnoea, hypoglycaemia, rash, seizures, and none; n=2 for hypothermia, lethargy, diarrhoea, and respiratory distress; and n=3 for coryza, cough, and hypotonia.
42 (64%) babies were looked after on a paediatric or postnatal ward, 20 (30%) received care in a neonatal unit, and four (6%) in a PICU. In total, 22 (33%) babies received one or more types of respiratory support: three (one born preterm at 36 weeks' gestation) received invasive ventilation, ten (four born preterm at 30, 32, 34, and 35 weeks' gestation) received non-invasive ventilatory support, and 22 (five born preterm at 30, 32, 34, 35, and 36 weeks' gestation) received supplemental oxygen.
Of the 21 babies who were diagnosed with SARS-CoV-2 infection within 7 days of birth, 14 (67%) were born at term, one (5%) baby (born preterm) was ventilated, eight (38%) babies (three born at term) received supplemental oxygen, and five (24%) babies (one born at term) received non-invasive respiratory support. Overall, two (3%) of 66 babies were treated with antiviral agents, two (3%) others were treated with corticosteroids, and one (2%) other received pooled immunoglobulin (table 3 ).
Table 3.
Treatment and outcomes
| Cases (n=66) | ||
|---|---|---|
| Highest location of care | ||
| Neonatal unit | 20 (30%) | |
| Paediatric intensive care unit | 4 (6%) | |
| Paediatric inpatient ward | 30 (45%) | |
| Postnatal ward | 12 (18%) | |
| Clinical care received for COVID-19 | ||
| Invasive ventilation (endotracheal intubation)* | 3 (5%) | |
| Non-invasive ventilation* | 10 (15%) | |
| Supplemental oxygen* | 22 (33%) | |
| High-frequency oscillatory ventilation | 0 | |
| Nitric oxide | 0 | |
| Extracorporeal membrane oxygenation | 0 | |
| No respiratory support | 42 (64%) | |
| Therapeutic hypothermia | 0 | |
| Missing data | 2 (3%) | |
| Treatments received at time of COVID-19 | ||
| Antibiotics | 46 (70%) | |
| Antivirals | 2 (3%) | |
| Corticosteroids | 2 (3%) | |
| Anti-arrhythmic treatment | 1 (2%)† | |
| Immunoglobulin | 1 (2%) | |
| Outcome | ||
| Still admitted | 3 (5%) | |
| Discharged home | 58 (88%) | |
| With home oxygen | 0 | |
| With home respiratory support | 0 | |
| For palliative care | 0 | |
| With community nursing | 2 (3%) | |
| No additional support | 56 (85%) | |
| Transferred to another hospital | 4 (6%) | |
| Died | 1 (2%) | |
| Missing data | 0 | |
Data are n (%).
Some babies received multiple forms of respiratory support.
Administered for condition considered unrelated to COVID-19.
Median length of stay for babies with SARS-CoV-2 infection was 2 days (IQR 1–4); those admitted to a neonatal unit had a longer median length of stay (7 days [2–55]) than those admitted to a PICU or a paediatric ward (2 days [0–4]). 58 (88%) babies were discharged home, seven (11%) were still inpatients at the time of last data collection form submission, and one (2%) died from a cause unrelated to SARS-CoV-2.
Discussion
Using population-level active surveillance data, we confirmed that inpatient care for neonates with confirmed SARS-CoV-2 is rare, with 5·6 cases per 10 000 livebirths (one in 1785) at the UK peak in March and April, 2020. Infection in the first 7 days after birth to a mother with perinatal SARS-CoV-2 infection was uncommon and generally mild or asymptomatic, despite a national policy that promoted keeping mother and neonate together. We identified only two babies with possible vertically acquired infection and six who were suspected of contracting SARS-CoV-2 within a neonatal unit or PICU. Neonatal SARS-CoV-2 infection led to severe disease in 42% of cases, and 36% of the babies in this study received care in a neonatal unit or PICU. 33% of babies required some form of respiratory support. However, this requirement might have been related to other conditions, such as prematurity, rather than SARS-CoV-2 infection. Neonates from Black, Asian, and mixed or other ethnic groups accounted for almost half of neonates admitted with SARS-CoV-2 infection.
Newborn babies can be affected by SARS-CoV-2 in a number of different ways: through infection, following vertical or horizontal transmission, and indirectly, through maternal COVID-19 (eg, in cases of preterm birth caused by maternal disease). The data presented here provide evidence on the incidence and outcomes of horizontal and possible vertical transmission among neonates who received inpatient hospital care. Previous systematic review data describing 666 births following maternal SARS-CoV-2 infection identified 28 cases of early neonatal infection,17 mainly from case reports, single-centre case series, and voluntary registries, with a lack of clarity about the proportion of ascertained, duplicate, and overlapping cases. Representative population-based data have so far been limited to that reported through UKOSS,14 which described 12 infants with confirmed SARS-CoV-2 infection. In this study, we report in greater detail the samples, clinical presentation, course, and outcomes for 17 babies in whom neonatal infection followed maternal perinatal infection. The data presented include the SARS-CoV-2-positive babies reported by UKOSS. We confirmed that early neonatal infection is rare and generally mild: only four babies born at term with early neonatal SARS-CoV-2 infection received any respiratory support, and none were ventilated. This study took place in the UK, where guidance was, and remains, to keep mother and baby together when the mother has confirmed perinatal SARS-CoV-2 infection.9 Separation of mother and baby has multiple detrimental consequences for both mother and baby,10, 11 and is not recommended by WHO guidance.8 Over the study period, more than 300 mothers with confirmed SARS-CoV-2 infection gave birth,14 and the low number of early neonatal SARS-CoV-2 infections and the mild disease course we documented support the approach taken in the UK. Furthermore, seven babies who were separated from their mothers immediately after birth still contracted SARS-CoV-2, suggesting that such action might not be effective at preventing early neonatal transmission. It is unclear why four of these seven babies were separated from their mothers, but it might reflect evolving local guidance early in the UK pandemic.
There remains considerable uncertainty about vertical transmission of SARS-CoV-2. Raised IgM titres in neonatal umbilical blood samples following maternal perinatal SARS-CoV-2 infection18, 19 and detailed case reports2, 20 support possible vertical transmission, although no reports to date meet the criteria for confirmed vertical transmission proposed by Shah and colleagues.16 Data from UKOSS provide the only available population-level data to inform the possible incidence of vertical transmission: six babies with SARS-CoV-2 isolated within the first 12 h. More focused neonatal follow-up through the BPSU surveillance system showed that five of these six babies were later confirmed to be negative for SARS-CoV-2, consistent with data from a single-centre study indicating that SARS-CoV-2 tests taken immediately after birth can have a considerable false positive rate.21 Thus, linked population-level obstetric and neonatal active surveillance data suggest that vertically transmitted neonatal SARS-CoV-2 infection is very rare, even at the height of a pandemic. However, this finding must be interpreted with caution, for two main reasons. First, our pre-specified definition of possible vertically acquired infection might have underestimated the true frequency because testing of newborn babies within 48 h after birth was not recommended in the UK, and thus few babies were tested within the first 12 h. Second, the proportion of asymptomatic infections is unknown, so it is not possible to accurately determine the population of SARS-CoV-2-positive mothers necessary to calculate incidence. Pregnant women in the UK were asked to shield from March, 2020, so their incidence of infection was likely to have been lower than that of the general population. For these reasons we have not attempted to estimate the incidence of possible vertically acquired infection. The rarity of such cases is consistent with data from three hospitals in New York City (NY, USA), in which routine neonatal testing of babies from 116 mothers with confirmed perinatal SARS-CoV-2 infection identified no neonatal cases.22
Neonates can also be infected with SARS-CoV-2 outside the immediate perinatal period. There are, however, no other population-level data describing later-onset neonatal infection up to 28 days after birth. The largest descriptive cohorts to date were drawn from a multinational study that included data from 84 institutions in Europe (including 15 in the UK)5 and the UK-based ISARIC4C prospective observational cohort study, which reported data from Jan 17 to July 3, 2020.23 The European registry cohort describes 40 and the ISARIC4C cohort 53 babies with SARS-CoV-2 infection in the first month after birth (some of whom are likely to be a subset of our cohort).
Our population-level data confirm that neonates have more severe disease than older children: 36% received critical care or respiratory support, in contrast to 13% in older children;5 42% of neonates met the criteria for severe disease, in contrast to 6% of children up to 18 years of age.4 Although viral respiratory infections in neonates24 are associated with an increased requirement for respiratory support, critical care or respiratory support in some cases might have been related to other newborn conditions, such as preterm birth, which was also more common among babies with SARS-CoV-2 infection than among the general population. Crucially, although the long-term consequences of early-life SARS-CoV-2 exposure or infection are as yet unknown, short-term outcomes were excellent in this cohort: 60% of babies were discharged home without the requirement for additional support, and there were no deaths attributable to SARS-CoV-2 infection. By contrast, poor perinatal outcomes were observed during the 2009–10 H1N1 influenza pandemic.25 We also found that babies were more likely to present with poor feeding or vomiting or with coryzal signs than were older children23 or adults.1
9% of neonatal SARS-CoV-2 cases followed suspected nosocomial transmission within neonatal or PICU settings, indicating that such units might need to review facilities and procedures for isolation of highly infectious cases during this and future pandemics. Further research is required to understand the effects of hospital visiting restrictions on the nosocomial spread of this and other highly transmissible viruses.26
The over-representation of BAME groups in neonatal SARS-CoV-2 infection is consistent with patterns described in UK maternity,14 paediatric,23 and adult data. This finding could be explained by the increased incidence of SARS-CoV-2 infections in March and April in areas with higher proportions of BAME groups relative to the whole UK, or might be due to other predisposing factors in the BAME population, and highlights the need for further urgent research in this area. We also found that babies born preterm were over-represented (24% of babies in this cohort compared with 8% of livebirths in England and Wales in 2018). Immune function3 differs and susceptibility to a range of pathogens is greater in preterm than in term babies;27 however, the finding that six (38%) of the 16 preterm babies had suspected nosocomial infection suggests that viral exposure within health-care settings could also contribute to the over-representation of preterm babies in the SARS-CoV-2 cohort.
The major strength of this study is that it reports UK-wide, population-level incidence data from a single national health service through the use of an established active surveillance system,28 with high reporting levels by UK paediatricians during the study period. The long-standing monthly BPSU reporting cards were augmented with additional weekly reporting cards for this study, and surveillance was further supplemented by national virology testing, PICU, and mortality data to maximise case ascertainment. The number of babies identified through these other sources highlights the importance of multiple-source notification and linkage during a health crisis. This study was prioritised as an urgent public health COVID-19 study in the UK, which ensured involvement of NIHR Clinical Research Network staff across paediatric and neonatal units. This study was done in conjunction with the national surveillance of maternal COVID-19 through the UKOSS system in order to facilitate the ascertainment of cases of potential vertical transmission and more detailed neonatal data describing cases reported by UKOSS.14 This linked surveillance will also be used to describe the indirect effects of maternal SARS-CoV-2 infection on neonates, including babies without confirmed SARS-CoV-2 infection. More detailed data will be available through future linkage with data held by PICANet13 and the National Neonatal Research Database.29 It is important to note that BPSU is the only national active surveillance platform in the UK for neonatal SARS-CoV-2 infection, and that all other reports of babies admitted to hospital with SARS-CoV-2 infection in the first 28 days in the UK will be subsets of these data.
Limitations of this study include a focus on essential data to limit the reporting burden on paediatricians in the setting of a coordinated national health service response to the COVID-19 pandemic; therefore, we did not collect detailed longitudinal data on physiological status or biochemical or haematological test results. Consequently, we operationalised the definition of critical or severe disease4 as described in table 1. We also did not specify the testing to be done and national policy varied over the study period, and we were therefore unable to confirm any baby infected as a consequence of vertical transmission in line with the classification proposed by Shah and colleagues.16 Additionally, we did not collect longitudinal data describing infant feeding and are thus unable to examine the relationship between breastfeeding and neonatal SARS-CoV-2 transmission. Finally, because our aim was to collect population-based information about neonatal SARS-CoV-2 infection in babies receiving inpatient hospital care, this study does not provide any data about overall infection rates or asymptomatic infection.
Inpatient admission in neonates with SARS-CoV-2 infection is rare and most babies are only mildly affected in the neonatal period. The long-term effects of early-life exposure to SARS-CoV-2 are unknown, and ongoing data collection, linkage, and follow-up are crucial. Furthermore, inpatient admission for neonatal infection following birth to a mother with perinatal SARS-CoV-2 infection was especially uncommon, with only 17 cases found across the UK during the study period. Although social, economic, and other factors must be incorporated in policy decisions, these data support current UK9 and international guidance8 to avoid routine separation of mother and baby and ensure that new parents can make informed decisions about neonatal care.
Data sharing
Data from this study will be shared according to the National Perinatal Epidemiology Unit's data sharing policy, which is available online.
Acknowledgments
Acknowledgments
This research is funded by the UK NIHR Policy Research Programme, conducted through the Policy Research Unit in Maternal and Neonatal Health and Care (PR-PRU-1217-21202). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. We acknowledge the assistance of Jacob Avis and Richard Lynn at the BPSU, the BPSU reporting clinicians, the NIHR Clinical Research Networks, and other people without whose support this research would not have been possible in such a timely manner. Full details of all collaborators and acknowledgments can be found in appendix 2 (pp 2–4).
Contributors
CG wrote the first draft of the article with contributions from MAQ and JJK. MAQ, CG, JJK, and AP carried out the analyses. All authors edited and approved the final version of the Article. CG, MK, SL, ESD, DS, CD, HM, and JJK contributed to the development and conduct of the study. CG as guarantor accepts full responsibility for the conduct of the study, had access to the data, and controlled the decision to publish. CG, MAQ, and JJK have accessed and verified the data underlying the study.
Declaration of interests
MK, MAQ, and JJK received grants from the UK NIHR Policy Research Programme in relation to the submitted work. AP, SL, ESD, DS, CD, HM, and CG declare no competing interests.
Supplementary Materials
References
- 1.Docherty AB, Harrison EM, Green CA, et al. Features of 20 133 UK patients in hospital with COVID-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ. 2020;369 doi: 10.1136/bmj.m1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kirtsman M, Diambomba Y, Poutanen SM, et al. Probable congenital SARS-CoV-2 infection in a neonate born to a woman with active SARS-CoV-2 infection. CMAJ. 2020;192:e647–e650. doi: 10.1503/cmaj.200821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kamdar S, Hutchinson R, Laing A, et al. Perinatal inflammation influences but does not arrest rapid immune development in preterm babies. Nat Commun. 2020;11 doi: 10.1038/s41467-020-14923-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dong Y, Mo X, Hu Y, et al. Epidemiology of COVID-19 among children in China. Pediatrics. 2020;145 doi: 10.1542/peds.2020-0702. [DOI] [PubMed] [Google Scholar]
- 5.Götzinger F, Santiago-García B, Noguera-Julián A, et al. COVID-19 in children and adolescents in Europe: a multinational, multicentre cohort study. Lancet Child Adolesc Health. 2020;4:653–661. doi: 10.1016/S2352-4642(20)30177-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zeng L, Xiao T, Zhou W. Vertical transmission of severe acute respiratory syndrome coronavirus 2 from the mother to the infant—reply. JAMA Pediatr. 2020;174 doi: 10.1001/jamapediatrics.2020.2156. [DOI] [PubMed] [Google Scholar]
- 7.Yeo KT, Oei JL, De Luca D, et al. Review of guidelines and recommendations from 17 countries highlights the challenges that clinicians face caring for neonates born to mothers with COVID-19. Acta Paediatr. 2020 doi: 10.1111/apa.15495. published online July 27. [DOI] [PubMed] [Google Scholar]
- 8.WHO Clinical management of COVID-19—interim guidance. May 27, 2020. https://www.who.int/publications/i/item/clinical-management-of-covid-19
- 9.Royal College of Obstetricians and Gynecologists Coronavirus (COVID-19) infection in pregnancy. July 24, 2020. https://www.rcog.org.uk/coronavirus-pregnancy
- 10.Bonacquisti A, Geller PA, Patterson CA. Maternal depression, anxiety, stress, and maternal-infant attachment in the neonatal intensive care unit. J Reprod Infant Psychol. 2020;38:297–310. doi: 10.1080/02646838.2019.1695041. [DOI] [PubMed] [Google Scholar]
- 11.Crenshaw JT. Healthy birth practice #6: keep mother and baby together—it's best for mother, baby, and breastfeeding. J Perinat Educ. 2014;23:211–217. doi: 10.1891/1058-1243.23.4.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Verity C, Preece M. Surveillance for rare disorders by the BPSU. Arch Dis Child. 2002;87:269–271. doi: 10.1136/adc.87.4.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Paediatric Intensive Care Audit Network Annual report 2019: summary report—data collection period January 2016–December 2018. https://www.picanet.org.uk/wp-content/uploads/sites/25/2019/12/PICANet-2019-Annual-Report-Summary_v1.0.pdf
- 14.Knight M, Bunch K, Vousden N, et al. Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369 doi: 10.1136/bmj.m2107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Draper ES, Gallimore ID, Kurinczuk JJ, et al. MBRRACE-UK perinatal mortality surveillance report: UK perinatal deaths for births from January to December 2017—summary report. October, 2019. https://www.npeu.ox.ac.uk/assets/downloads/mbrrace-uk/reports/MBRRACE-UK%20Perinatal%20Mortality%20Surveillance%20Summary%20Report%20for%202017%20-%20FINAL.pdf
- 16.Shah PS, Diambomba Y, Acharya G, Morris SK, Bitnun A. Classification system and case definition for SARS-CoV-2 infection in pregnant women, fetuses, and neonates. Acta Obstet Gynecol Scand. 2020;99:565–568. doi: 10.1111/aogs.13870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Walker KF, O'Donoghue K, Grace N, et al. Maternal transmission of SARS-COV-2 to the neonate, and possible routes for such transmission: a systematic review and critical analysis. BJOG. 2020;127:1324–1336. doi: 10.1111/1471-0528.16362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dong L, Tian J, He S, et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn. JAMA. 2020;323:1846–1848. doi: 10.1001/jama.2020.4621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zeng H, Xu C, Fan J, et al. Antibodies in infants born to mothers with COVID-19 pneumonia. JAMA. 2020;323:1848–1849. doi: 10.1001/jama.2020.4861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vivanti AJ, Vauloup-Fellous C, Prevot S, et al. Transplacental transmission of SARS-CoV-2 infection. Nat Commun. 2020;11 doi: 10.1038/s41467-020-17436-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martínez-Perez O, Vouga M, Cruz Melguizo S, et al. Association between mode of delivery among pregnant women with COVID-19 and maternal and neonatal outcomes in Spain. JAMA. 2020;324:296–299. doi: 10.1001/jama.2020.10125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Salvatore CM, Han JY, Acker KP, et al. Neonatal management and outcomes during the COVID-19 pandemic: an observation cohort study. Lancet Child Adolesc Health. 2020;4:721–727. doi: 10.1016/S2352-4642(20)30235-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Swann OV, Holden KA, Turtle L, et al. Clinical characteristics of children and young people admitted to hospital with COVID-19 in United Kingdom: prospective multicentre observational cohort study. BMJ. 2020;370 doi: 10.1136/bmj.m3249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zinna S, Lakshmanan A, Tan S, et al. Outcomes of nosocomial viral respiratory infections in high-risk neonates. Pediatrics. 2016;138 doi: 10.1542/peds.2016-1675. [DOI] [PubMed] [Google Scholar]
- 25.Pierce M, Kurinczuk JJ, Spark P, Brocklehurst P, Knight M. Perinatal outcomes after maternal 2009/H1N1 infection: national cohort study. BMJ. 2011;342 doi: 10.1136/bmj.d3214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Szatkowski L, McClaughry R, Clarkson M, Sharkey D. Restricted visiting reduces nosocomial viral respiratory tract infections in high-risk neonates. Eur Respir J. 2019;53 doi: 10.1183/13993003.01874-2018. [DOI] [PubMed] [Google Scholar]
- 27.Afonso EDP, Blot S. Effect of gestational age on the epidemiology of late-onset sepsis in neonatal intensive care units—a review. Expert Rev Anti Infect Ther. 2017;15:917–924. doi: 10.1080/14787210.2017.1379394. [DOI] [PubMed] [Google Scholar]
- 28.Gale C, Knight M, Ladhani S, et al. National active surveillance to understand and inform neonatal care in COVID-19. Arch Dis Child Fetal Neonatal Ed. 2020;105:346–347. doi: 10.1136/archdischild-2020-319372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gale C, Morris I. The UK National Neonatal Research Database: using neonatal data for research, quality improvement and more. Arch Dis Child Educ Pract Ed. 2016;101:216–218. doi: 10.1136/archdischild-2015-309928. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data from this study will be shared according to the National Perinatal Epidemiology Unit's data sharing policy, which is available online.