Abstract
25-Hydroxyvitamin D (25OHD) and 1,25-dihydroxyvitamin D (1,25(OH)2D) need to be bound to carrier proteins to be transported to their target cells. The majority of either 25OHD or 1,25(OH)2D is bound to vitamin D-binding protein (DBP), a smaller fraction is bound to albumin and only very small amounts of 25OHD or 1,25(OH)2D are free. Albumin-bound 25OHD or 1,25(OH)2D is relatively easily available after dissociation from albumin. Thus, the sum of free and albumin-bound forms is called bioavailable 25OHD and bioavailable 1,25(OH)2D. Total 25OHD and 1,25(OH)2D are defined as the sum of free, albumin-bound and DBP-bound 25OHD and 1,25(OH)2D, respectively. This cross-sectional study in 427 pregnant women compared the correlation of the six vitamin D compounds with biomarkers of bone health, lipid metabolism, kidney function, endocrine parameters, and group B water-soluble vitamins. Among the 25OHD metabolites analysed, total 1,25(OH)2D showed clearly the best correlation with calcium, bone-specific alkaline phosphatase, adiponectin, LDL, HDL, urea, thyroxine, and group B water-soluble vitamins. When comparing the three 25OHD metabolites, both free 25OHD and bioavailable 25OHD showed overall good correlations with calcium, bone-specific alkaline phosphatase, adiponectin, LDL, HDL, urea, thyroxine, triiodothyronine, and group B water-soluble vitamins, The correlations of 1,25(OH)2D and 25OHD metabolites went always in opposite directions. Only PTH correlates always inversely with all six vitamin D compounds. In conclusion, free 25(OH)D and bioavailable 25(OH)D are more precise determinants of the vitamin D status than total 25(OH)D in normal pregnancy, whereas total 1,25(OH)2D is superior to free and bioavailable 1,25(OH)2D. Except for PTH, correlations of 25(OH)D and 1,25(OH)2D metabolites with typical clinical chemistry readouts go in opposite directions.
Subject terms: Biochemistry, Hormones, Steroid hormones
Introduction
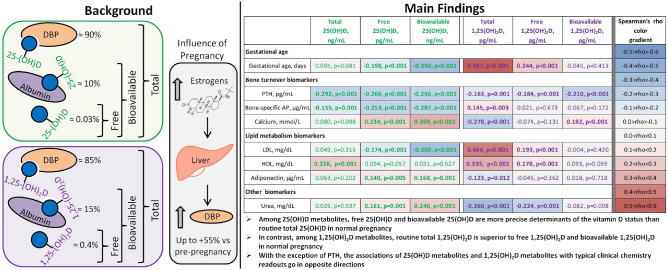
Both 25OHD and 1,25(OH)2D, being lipophilic molecules, have a special carrying protein, enabling their transport in the circulation, protection from hepatic degradation and accumulation of circulating reserves, called vitamin D-binding protein (DBP). In the serum, 85–90% of the whole 25OHD/1,25(OH)2D pool is present in the DBP-bound condition. The remaining 10–15% are loosely bound to serum albumin and lipoproteins (10–15%), and just a tiny fraction of 25OHD/1,25(OH)2D (less than 0.1% of 25OHD and about 0.4% of 1,25(OH)2D1,2) is available in the free, unbound condition. Thus, from this point of view, the terminology “total 25OHD” “total 1,25(OH)2D”, which will be used throughout this manuscript, refers to a sum of three 25OHD/1,25(OH)2D fractions: DBP-bound, albumin-bound (the latter fraction is also called bioavailable, since this binding is very weak and spontaneous dissociation of vitamin D metabolites from albumin is very easy to occur), and free compounds. Since serum levels of liver-produced DBP is subject to substantiate changes under numerous physiologic and pathologic conditions (genetic background, endocrine status in particular with regards to estrogens, liver diseases and kidney diseases), the values of total 25OHD—the current gold standard vitamin D status marker—might not precisely clarify the status of vitamin D, particularly in such conditions as normal pregnancy, estrogen-containing oral contraceptive intake, as well as liver and renal pathologies.
Since the mid-1980s it is known that free 25OHD/1,25(OH)2D determination is possible based on the laws of protein–ligand binding kinetics using a formula employing serum concentrations of total 25OHD, DBP, albumin, as well as 25OHD/1,25(OH)2D-DBP and 25OHD/1,25(OH)2D-albumin binding affinity constants, which was originally suggested by Bikle et al.3.
The present study was designed to perform for the first time a systematic head-to-head comparison of three ways of measuring 25-hydroxyvitamin D (free, bioactive, ant total 25OHD) with three ways to measure 1,25-dihydroxyvitamin D (free, bioactive, ant total 1,25(OH)2D) using pregnant women as a test system, because vitamin D binding protein is incresed in this population.
Materials and methods
Study population
The study was a part of the Berlin Birth Cohort (BBC) study4,5. This study was approved by the local ethical committee of the Charité—Universitätsmedizin Berlin, Campus Mitte, Berlin, Germany and written informed consent was obtained from all participants. The study cohort consisted of 427 Caucasian pregnant healthy women aged > 18 years. All methods were carried out in accordance with relevant German guidelines and regulations.
The study participants were categorized into three trimester groups according to their gestational age: the 1st trimester (≤ 12 weeks + 6 days), the 2nd trimester (13 weeks–27 weeks + 6 days), and the 3rd trimester (≥ 28 weeks until delivery). The mean gestational age of the entire study cohort was 18.49 weeks (n = 427). The group sizes were 211, 78, and 135 participants in 1st, 2nd, and 3rd pregnancy trimesters, respectively.
The study was carried out at clinical laboratory medical institution in Berlin, Germany. Venous blood samples were collected in the period from May till June 2016. The study was approved by the local ethics committee.
Measurement of 25OHD and 1,25(OH)2D metabolites, DBP, and albumin
All analytes were measured in serum samples. Total 25OHD (hereafter “25OHD” is referred to as a sum of 25OHD2 and 25OHD3 metabolites) was measured by Abbott Architect i2000 (Abbott Laboratories, Wiesbaden, Germany) using Abbott Architect 25OHD automated chemiluminescent microparticle immunoassay (Abbott Laboratories, Wiesbaden, Germany). As declared by the assay manufacturer, the kit has the following cross-reactivities with vitamin D metabolites: 98.6–101.1% with 25OHD3, 54.0% with 25OHD2, 101.9–189.2% with 24,25OH2D3, 71.4–114.2% with 24,25OH2D2, and < 1% with other vitamin D forms (vitamin D3, vitamin D2, 1,25(OH)2D3, and 1,25(OH)2D2). Total 1,25(OH)2D (hereafter “1,25(OH)2D” is referred to as a sum of 1,25(OH)2D2 and 1,25(OH)2D3 metabolites) was determined using IDS-iSYS 1,25 VitDXP chemiluminescence-immunoassay on the IDS-iSYS Multi-Discipline Automated system (both by IDS Immunodiagnostic Systems GmbH, Frankfurt am Main, Germany). According to the manufacturer’s data sheet, this assay has the following cross-reactivities with 1,25(OH)2D metabolites: 97% with 1,25(OH)2D3, 72% with 1,25(OH)2D2, 106.7% with 1,24,25OH3D3, and < 1% with other vitamin D metabolites. Serum DBP was determined by a polyclonal antibody immunoassay (Immundiagnostik AG, Bensheim, Germany). Serum albumin was assessed using an automated analyzer (Beckman Coulter Inc., Krefeld, Germany). Regarding the quality control in total 25OHD, total 1,25(OH)2D and albumin measurement, our lab constantly fulfill the requirements of the round robin tests organized by the Reference Institute for Bioanalytics (RfB)—one of the two German proficiency testing organizations which have been officially charged by the German Medical Association, in which our lab participates regularly. Regarding DBP, this analyte does not belong to the list of routine clinical biomarkers and was measured exclusively for the present study. However, this kit is IVD- (“In-Vitro-Diagnostic Medical Device”) and CE- (“Conformité Européene”) certified, which means that it can be used in-vitro for the examination of specimens, including blood- and tissue donations, derived from the human body within the European Economic Area.
Free 25OHD was measured based on the laws of protein–ligand binding kinetics by calculation method using a formula employing serum concentrations of total 25OHD, DBP, albumin, as well as 25OHD-DBP and 25OHD-albumin binding affinity constants, which was originaly suggested by Bikle et al.6:
where Free 25OHD = concentration of free 25OHD in mol/L; Kalb = affinity constant between 25OHD and albumin equal to 6 × 105 M−1; KDBP = affinity constant between 25OHD and DBP, equal to 7 × 108 M−16,7; albumin = concentration of total serum albumin in mol/L; DBP = concentration of total vitamin D-binding protein in mol/L; Total 25OHD = concentration of total 25OHD in mol/L.
Utilizing exactly the same principle, but using 1,25(OH)2D-specific binding affinity constants—KDBP = 4.0 × 107 M−1 and Kalb = 5.4 × 104 M−1—we calculated free 1,25(OH)2D6,7:
Bioavailable 25OHD and bioavailable 1,25(OH)2D were calculated using the following formula based on precalculated free 25OHD or free 1,25(OH)2D levels, respectively8,9:
where D = concentration of vitamin D metabolite (25OHD or 1,25(OH)2D) in mol/L; Kalb = affinity constant between a respective vitamin D metabolite (6 × 105 M−1 for 25OHD and 5.4 × 104 M−1 for 1,25(OH)2D) and albumin in mol−1; albumin = concentration of serum albumin in mol/L.
Measurement of other serum analytes
The detailed data on diagnostic assays and instrumental devices used for measurement of other serum parameters (calcium, PTH, bone-specific alkaline phosphatase, LDL, HDL, hemoglobin, etc.) are provided in Supplementary Table 1.
Statistical analyses
Statistical analyses were performed using SPSS 20.0 (IBM, New York, NY, USA) and GraphPad Prism version 5 software (GraphPad Software, San Diego, CA, USA) as recently described10,11. All relevant parameters were tested for normality using a D’Agostino and Pearson omnibus normality test and were found to be not normally distributed (p < 0.05) in the majority of the cases. For this reason, we used Spearman’s rank correlation analysis to assess bivariate associations between vitamin D metabolites with each other and with other analytes. The correlation strength (measured as Spearman’s rho values) was classified as follows: ≤ 0.1 no correlation; 0.1–0.3 weak to modest; 0.3–0.49 moderate; > 0.5 strong correlation. To assess statistical differences among three trimester groups we used either one-way ANOVA followed by Tukey post hoc test (in cases when all three trimester groups were normally distributed, as determined by D’Agostino and Pearson omnibus normality text), or Kruskal–Wallis analysis with Dunn’s multiple comparison test (in cases when at least one group out of three analyzed groups was found to be not normally distributed). In both correlation and group comparison analyses, correlations/group differences were considered significant if p values were < 0.05.
Results
Study participant characteristics
The mean values of participant’s age and gestational age, as well as selected serum biomarkers for the pregnancy overall and for early, mid-, and late pregnancy are presented in Table 1.
Table 1.
Biochemical characteristics of the study population. Mean values ± standard deviation of age and gestational age, vitamin D metabolites and biochemical, hematological, and endocrinological serum parameters in 1st, 2nd, and 3rd pregnancy trimesters.
| Variable, units | 1st trimester of pregnancy, mean ± SD | 2nd trimester of pregnancy, mean ± SD | 3rd trimester of pregnancy, mean ± SD | Changes throughout pregnancy |
|---|---|---|---|---|
| T25OHD, ng/mL | 18.80 ± 9.96 | 20.26 ± 11.09 | 20.55 ± 11.56 | ↔ |
| F25OHD, pg/mL | 2.84 ± 1.54 | 2.41 ± 1.41 | 2.31 ± 1.48*** | ↓ |
| B25OHD, ng/mL | 1.05 ± 0.57 | 0.76 ± 0.47*** | 0.69 ± 0.44*** | ↓ |
| T1,25(OH)2D, pg/mL | 105.80 ± 42.59 | 150.95 ± 47.28*** | 163.28 ± 43.68*** | ↑ |
| F1,25(OH)2D, pg/mL | 0.27 ± 0.10 | 0.30 ± 0.10** | 0.31 ± 0.10*** | ↑ |
| B1,25(OH)2D, pg/mL | 9.05 ± 3.47 | 8.84 ± 2.93 | 8.58 ± 2.68 | ↔ |
| Age, years | 30.63 ± 6.36 | 29.54 ± 5.28 | 29.87 ± 5.86 | ↔ |
| Gestational age, weeks | 7.98 ± 1.84 | 21.29 ± 4.06*** | 33.25 ± 2.19§§§, *** | ↑ |
| PTH, pg/mL | 15.37 ± 10.76 | 14.60 ± 13.48 | 17.93 ± 13.98 | ↔ |
| BSAP, µg/mL | 12.93 ± 5.12 | 13.97 ± 5.86 | 18.01 ± 8.21§§§,*** | ↑ |
| Calcium, mmol/L | 2.38 ± 0.12 | 2.25 ± 0.13*** | 2.24 ± 0.10*** | ↓ |
| Phosphate, U/L | 4.39 ± 1.65 | 4.07 ± 1.24 | 4.33 ± 2.23 | ↔ |
| DBP, mg/L | 532.56 ± 138.73 | 690.00 ± 171.15*** | 762.70 ± 254.08*** | ↑ |
| LDL, mg/dL | 118.04 ± 34.00 | 158.52 ± 39.12*** | 184.16 ± 40.60§§,*** | ↑ |
| HDL, mg/dL | 59.36 ± 13.25 | 65.10 ± 12.40** | 67.48 ± 12.64*** | ↑ |
| LDL/HDL ratio | 2.05 ± 0.61 | 2.49 ± 0.63*** | 2.83 ± 0.82*** | ↑ |
| Adiponectin, µg/mL | 9.28 ± 4.15 | 8.06 ± 3.73* | 8.41 ± 3.97 | ↓ |
| Free T3, pg/mL | 3.10 ± 0.38 | 3.15 ± 0.38 | 3.03 ± 0.38§ | ↓ |
| Free T4, ng/dL | 1.02 ± 0.14 | 0.87 ± 0.12*** | 0.85 ± 0.11*** | ↓ |
| TSH, uU/mL | 1.48 ± 0.97 | 1.54 ± 0.79 | 1.45 ± 0.66 | ↔ |
| Hemoglobin, g/dL | 12.55 ± 0.94 | 11.53 ± 0.83*** | 11.66 ± 0.93*** | ↓ |
| RBC count, 106/µL | 4.34 ± 0.39 | 3.90 ± 0.35*** | 4.00 ± 0.34*** | ↓ |
| WBC count, 103/µL | 8.54 ± 2.14 | 9.55 ± 2.83* | 9.75 ± 2.33*** | ↑ |
| Platelet count, 103/µL | 232.55 ± 50.51 | 214.12 ± 42.94 | 220.68 ± 54.10 | ↔ |
| MCV, fL | 89.13 ± 5.42 | 89.77 ± 5.67 | 89.34 ± 5.39 | ↔ |
| MCH, pg | 29.18 ± 1.96 | 29.73 ± 2.12 | 29.32 ± 1.95 | ↔ |
| MCHC, g/dL | 32.75 ± 0.97 | 33.15 ± 0.96* | 32.93 ± 1.11 | ↔ |
| RDW, % | 13.97 ± 1.23 | 13.99 ± 1.11 | 13.87 ± 1.16 | ↔ |
| Hematocrit, % | 38.57 ± 2.85 | 34.86 ± 2.39*** | 35.51 ± 2.96*** | ↓ |
| Urea, mg/dL | 23.07 ± 5.85 | 17.97 ± 4.89*** | 17.93 ± 4.58*** | ↓ |
| Albumin, g/dL | 4.09 ± 0.38 | 3.45 ± 0.30*** | 3.30 ± 0.24*** | ↓ |
| Vitamin B6, ng/mL | 14.43 ± 18.70 | 7.34 ± 7.78*** | 5.55 ± 13.11§,*** | ↓ |
| Vitamin B12, pg/mL | 368.63 ± 157.81 | 279.23 ± 123.68*** | 275.75 ± 121.83*** | ↓ |
| Sodium, mmol/L | 137.80 ± 4.46 | 138.03 ± 2.80 | 137.77 ± 3.43 | ↔ |
| Zinc, µmol/L | 12.50 ± 2.83 | 10.13 ± 2.21** | 9.86 ± 2.13*** | ↓ |
B25OHD, bioavailable 25OHD; B1,25(OH)2D, bioavaialble 1,25(OH)2D; BSAP, bone-specific alkaline phosphatase; F25OHD, free 25OHD; F1,25(OH)2D, free 1,25(OH)2D; fT3, free triiodothyronine; fT4, free thyroxine; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume of red blood cells; PTH, parathyroid hormone; RBC, red blood cell; RDW, red blood cell distribution width; T25OHD, total 25OHD; T1,25(OH)2D, total 1,25(OH)2D; WBC, white blood cell. *p < 0.05 versus 1st trimester; **p < 0.01 versus 1st trimester; ***p < 0.001 versus 1st trimester; §§p < 0.01 versus 2nd trimester; §§§p < 0.001 versus 2nd trimester.
Changes in serum parameters throughout pregnancy
The data on changes of biochemical and biochemical parameters throughout pregnancy (split by three trimesters) are presented in Table 1. Importantly, a progressive increase in DBP during pregnancy was observed (43% increase in 3rd trimester compared to the 1st trimester, p < 0.001, Table 1).
Regarding 25(OH)D metabolites, we found no significant changes in total 25OHD throughout pregnancy. In contrast, free 25OHD and bioavailable 25OHD were both significantly decreased with gestational age (Table 1). The dynamics in changes of 1,25(OH)2D isoform levels was totally different compared to the behavior of 25OHD compounds. Indeed, total 1,25(OH)2D showed a progressive increase throughout pregnancy. The same trend revealed free 1,25(OH)2D with however weaker numerical rise. In contrast, bioavailable 1,25(OH)2D showed no significant differences between early and late gestation groups (Table 1).
Associations between 25OHD and 1,25(OH)2D metabolites
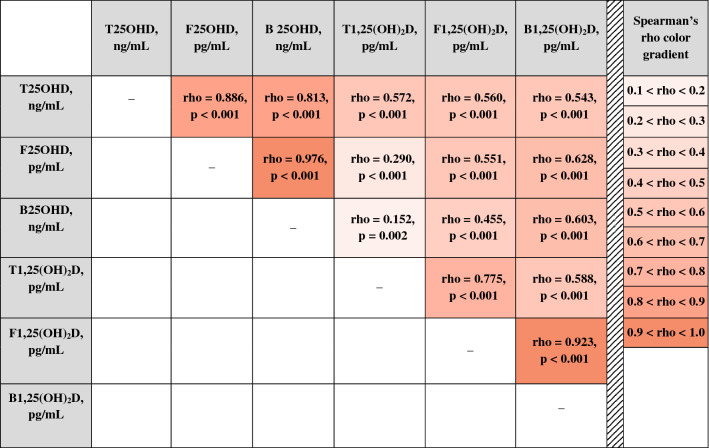
In general, absolutely all analyzed compounds of 25OHD and 1,25(OH)2D showed a significant positive correlation with each other, however, the degree of such association (characterized by Spearman’s rho) varied from 0.15 until 0.98 (p < 0.001 for all pairs of comparison, except of the pair total 1,25(OH)2D versus bioavailable 25OHD with p = 0.002, Table 2). Regarding 25OHD isoforms, total 25OHD, free 25OHD, and bioavailable 25OHD revealed very strong correlation between each other (0.813 < rho < 0.976, Table 2). Regarding interrelations between 1,25(OH)2D compounds, the correlation was substantially weaker (0.588 < rho < 0.923, p < 0.001 for all pairs of comparison) compared to the ones between 25OHD isoforms and more variable depending on certain pair of comparison. In addition, total 25OHD showed a medium correlation with all three 1,25(OH)2D compounds of nearly equal strength (rho around 0.55). In contrast, free 25OHD and bioavailable 25OHD showed much weaked correlation with total 1,25(OH)2D than with free 1,25(OH)2D and bioavailable 1,25(OH)2D (rho = 0.29, 0.55, and 0.63, respectively, see Table 3). The relationship between bioavailable 25OHD and 1,25(OH)2D compounds was very similar to interrelations between free 25OHD and 1,25(OH)2D.
Table 2.
Interrelations between 25OHD and 1,25(OH)2D isoforms.
The correlation strength is defined by Spearman's rank correlation coefficient rho. Abbreviations: B25OHD, bioavailable 25OHD; B1,25(OH)2D, bioavaialble 1,25(OH)2D; F25OHD, free 25OHD; F1,25(OH)2D, free 1,25(OH)2D; T25OHD, total 25OHD; T1,25(OH)2D, total 1,25(OH)2D.
Table 3.
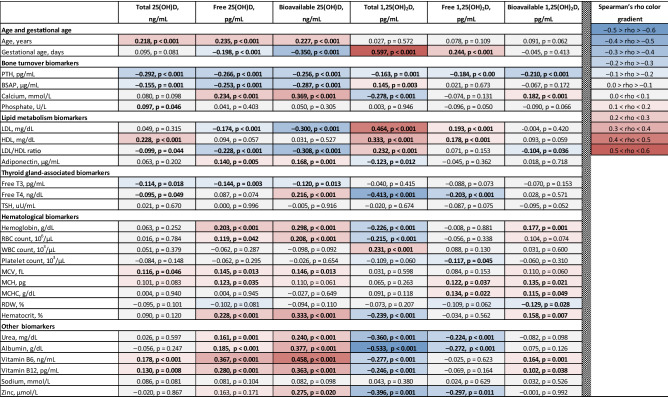
Interrelations between 25OHD and 1,25(OH)and 1,25(OH)2D isoforms with age, gestational age, biochemical, hematological, and endocrinological serum parameters.
The correlation strength is defined by Spearman's rank correlation coefficient rho (appears first in each cell). Abbreviations: B25OHD, bioavailable 25OHD; B1,25(OH)2D, bioavaialble 1,25(OH)2D; BSAP, bone-specific alkaline phosphatase; F25OHD, free 25OHD; F1,25(OH)2D, free 1,25(OH)2D; fT3, free triiodothyronine; fT4, free thyroxine; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume of red blood cells; PTH, parathyroid hormone; RBC, red blood cell; RDW, red blood cell distribution width; T25OHD, total 25OHD; T1,25(OH)2D, total 1,25(OH)2D; WBC, white blood cell.
Correlations of free and total 25(OH)D with serum biochemical and hematological parameters
Overall, free 25OHD and, particularly bioavailable 25OHD, showed stronger associations with distinct parameters than total 25OHD (Table 3).
Total 25OHD showed no correlation with serum calcium—a key parameter of bone health (rho = 0.080, p = 0.098), whereas free 25OHD revealed weak but significant positive correlation with calcium (rho = 0.234, p < 0.001, Table 3). Bioavailable 25OHD showed even stronger association with calcium (rho = 0.369, p < 0.001, Table 3).
Total 25(OH)D showed no correlation with serum albumin, free thyroxin, urea, adiponectin, LDL, LDL/HDL ratio, vitamin B12, hemoglobin, white blood cell count, and red blood cell distribution width, in contrast to free 25(OH)D, which did show weak but significant correlations with these analytes (Table 3). In addition, despite the presence of the correlation between total 25(OH)D and bone-specific alkaline phosphatase and vitamin B6, it was borderline significant, in contrast to free 25(OH)D, which was correlated with these parameters with p < 0.001. The associations of total and free 25(OH)D with other biochemical and hematological parameters are presented in Table 3.
Discussion
Summary of the key findings
To the best of our knowledge, the present cross-sectional study is the first report comparing the behavior of the six vitamin D compounds—free, bioavailable, and total 25-hydroxyvitamin D as well as free, bioavailable, and total 1,25-dihydroxyvitamin D—in a relatively large population of healthy pregnant women (n = 427). Among the 1,25-dihydroxyvitamin D metabolites analyzed, total 1,25(OH)2D showed clearly the best correlation with components of the endocrine vitamin D system and/or potential cellular targets of activation of the vitamin D receptor (see Table 3). When comparing the three 25OHD metabolites, both free 25OHD and bioavailable 25OHD showed overall good correlations with the analyzed clinical laboratory parameters, bioavailable 25OHD might perform even better then free 25OHD. The only exception from the above described overall findings is PTH. It correlates well with total 25OHD most likely because the parathyroid cells have a highly specific uptake mechanism for bound 25OHD—megalin/cubilin-uptake system, see below. With the exception of PTH, associations of 25(OH)D metabolites and 1,25(OH)2D metabolites with typical clinical chemistry readouts goes in opposite directions.
Moreover, our study confirmed the finding of the study by Bouillon et al.12 that serum total 25OHD doesn’t markedly change during pregnancy, whereas serum total 1,25(OH)2D increased significantly along with serum DBP, which in turn resulted in the increase in free vitamin D metabolites: free 25OHD was declined and free 1,25(OH)2D was slightly elevated as total 1,25(OH)2D was increased slighthy more than DBP.
Free 25OHD and bioavailable 25OHD were found to be superior to traditional total 25OHD
Our data suggest that in normal human pregnancy both free 25OHD and bioavailable 25OHD (the latter is tendentially slightly superior to free 25OHD) appear to be more precise indicators of vitamin D status during pregnancy than routine total 25OHD in terms of overall better correlations with gestational age, calcium, bone-specific alkaline phosphatase, adiponectin, hemoglobin and red blood cell count, vitamins B6 and B12 and other analytes (Table 3). In other words, our data are in favor of clinical relevance of free hormone hypothesis for 25OHD.
To date, the data on changes in free/bioavailable 25OHD with total 25OHD throughout pregnancy were analyzed in nine studies9–11,13–18, including our own10,11. Overall, the data regarding the comparison of the diagnostic potential of free/bioavailable 25OHD versus total 25OHD, reported by these authors, are conflicting due to low sample sizes (varying between 10 and 60 participants), with the exception of two studies by Powe et al.18 and Gustafsson et al.17 , which was based on 170 and 855 pregnant women, respectively. This discrepancy was discussed in details in our recent publications11,19. This study by Powe et al.18 differs from ours: just three-fourth of the participating women were healthy and only around 60% were of White ethnicity. Although F25OHD was assessed by calculation method using vitamin D-binding protein (DBP) levels like in our study, DBP in this study was measured using a monoclonal antibody-based ELISA. This DBP measurement technique is considered unreliable, especially when having populations with different ethnic backgrounds due to DBP polymorphism20–23. Our study is larger, conducted in an ethnically homogenous cohort and by using the state-of-the-art way to measure DBP. We thus believe that our data are more relevant. Moreover, these results are in good agreement with the findings of our previous study11, where we compared total 25OHD with directly measured free 25OHD. Regarding the large longitudinal study by Gustafsson et al.17, since the authors did not present the data of bivariate correlation analysis (free 25OHD/total 25OHD versus PTH), a one-to-one comparison of our results with their results is impossible.
Neither free 1,25(OH)2D nor bioavailable 1,25(OH)2D showed superiority to total 1,25(OH)2D
In contrast to 25OHD—vitamin D’s precursor and reservoir—our data do not support the free hormone hypothesis for 1,25(OH)2D—vitamin D’s biologically active metabolite. Indeed, with the exception of PTH, total 1,25(OH)2D showed constantly stronger correlations with all analyzed serum biomarkers than free 1,25(OH)2D and bioavailable 1,25(OH)2D (Table 3).
According to the data of numerous cross-sectional and longitudinal studies, total 1,25(OH)2D is known to rise up to threefold versus non-pregnant state24. This is the result of the increased production of 1,25(OH)2D by 25-hydroxyvitamin D-1α-hydroxylase (CYP27B1) by not only maternal kidneys, but also, although to a lesser extent, by the fetoplacental unit (maternal decidual and fetal trophoblast tissues of the placenta and fetal kidney)24–26. We likewise detected total 1,25(OH)2D increase throughout gestation. Notably, free 1,25(OH)2D but not bioavailable 1,25(OH)2D was found to rise throughout pregnancy course. Three other studies3,17,27 likewise reported an increase in free 1,25(OH)2D with pregnancy course.
Correlation with PTH stays apart: all six vitamin D isoforms showed nearly equal associations with PTH
PTH—a major secretory product of parathyroid gland and a second, along with 1,25(OH)2D, hormonal regulator of calcium homeostasis—was found to be the only biomarker which showed the same direction of correlation (negative correlation) and strength (weak but still significant negative correlation) with all six analysed vitamin D metabolites. The most likely explanation of this finding could be that parathyroid gland, along with kidney and placenta, are exceptional organs, which have megalin/cubilin-mediated system of 25OHD/1,25(OH)2D uptake from 25OHD-DBP and 1,25(OH)2D-DBP complexes28,29 and thus the cells of parathyroid gland are influenced by all circulating isoforms of 25OHD/1,25(OH)2D at nearly the equal extent. This mechanism is absent in most of the other cell types and organs, leaving for those cells only the option of the freely circulating 25OHD/1,25(OH)2D uptake via passive diffusion through plasma membranes.
25OHD and 1,25(OH)2D correlate in opposite directions with biochemical and endocrinological biomarkers
The direction of correlation (i.e., positive vs negative) between 25OHD metabolites on one hand, and 1,25(OH)2D metabolites on the other hand, with other serum biomarkers was found to be opposite. The most prominent examples are the differences in the correlation directions with such biomarkers as LDL, free thyroxin, urea, vitamins B6, B12, and zinc (Table 3). This emphasizes the different biological roles of total 1,25(OH)2D—vitamin D’s fully active isoform, which (total 1,25(OH)2D)—is present in the circulation in a picomolar range and have a half-life of 5–80 h30 and total 25OHD—vitamin D’s precursor, which is present in the circulation in nearly thousand-fold higher concentration range and have a half-live of 10–40 days30 and which is well known to serve as a reservoir/storage fraction of vitamin D, responsible for balancing/minimization of short-time (intraday) vitamin D isoform fluctuations. The 10–100-fold lower affinity between DBP and free 1,25(OH)2D compared to the affinity between DBP and free 25OHD (to the lesser extent this is valid likewise for albumin) as well as much higher (up to 100-fold) affinity of nuclear VDR for free 1,25(OH)2D than for free 25OHD are factors influencing concentrations of the vitamin D metabolites at the nuclear receptor. Whether or not the strength of the binding of either free 1,25(OH)2D or free 25OHD to the nuclear receptor is affecting gene expression in an opposite manner is unknown so far. It is conceivable that hormone receptor ligands with low binding affinity for the receptor may act as partial receptor antagonists, whereas ligands with high affinity fully stimulate the receptor. However, this needs to be proven in adequately designed basic science experiments. In any case, this is the first study reporting differences in correlation strength between 25OHD/1,25(OH)2D isoforms versus key clinical parameters. This for sure needs to be repeated in independent cohorts and the underlying molecular mechanisms (see our hypothesis above) need to be explored in future studies.
Study limitations
The data on free and bioavailable 25OHD and 1,25(OH)2D were based on calculations using the affinity constants between 25OHD/1,25(OH)2D and DBP and 25OHD/1,25(OH)2D and albumin. These affinity constants were measured experimentally in serum samples of non-pregnant individuals and has not been verified in pregnant women so far. However, the possible inaccuracies (a systematic shift in calculated free/bioavailable 25OHD/1,25(OH)2D absolute levels versus their real values) are not influencing the key findings of our study, because our conclusions depend on the relative changes of free, bioavailable and total 25OHD and 1,25(OH)2D, respectively rather than absolute changes of these parameters.
It is very well known that during pregnancy, especially in the 3rd trimester, the blood volume of the pregnant women increases and thus the blood is diluted to a certain extent31. However, the influence of hemodilution as a confounding factor in bivariate associations presented in this study will not change the correlation direction. Hemodilution might have just flatten the correlation strength (as presented by Spearman’s rho values), but it will not reverse the direction of correlation.
The accuracy of total 25OHD levels produced by fully-automated commercial immunoassays, especially in the conditions of pregnancy-induced increased DBP levels, is a well-known issue32. However, we used the total 25OHD kit with the best diagnostic precision characteristics in pregnancy among the other tested commercial kits when comparing to total 25OHD concentrations measured by mass-spectrometry—a technique considered the gold standard for the measurement of 25OHD32. Undoubtedly, the usage of LC–MS/MS might have further improved the data quality.
DBP, being a highly polymorphic protein, was reported to have genotype differences (up to threefold) in affinity constants with 25OHD33. Although these data were not confirmed by three other studies34–36, we are on the safe side with our key conclusions, because we used quite a monogenic (Caucasian) cohort and we measured DBP with a polyclonal-antibody-based ELISA kit. Finally, we conducted a single-centered study and thus single-centered effects cannot be excluded.
Conclusions
The present study compared the performance of the six vitamin D compounds—free, bioavailable, and total 25-hydroxyvitamin D (free 25OHD, bioavailable 25OHD, total 25OHD) as well as free, bioavailable, and total 1,25-dihydroxyvitamin D (free 1,25(OH)2D, bioavailable 1,25(OH)2D, total 1,25(OH)2D). Among the 1,25-dihydroxyvitamin D metabolites, total 1,25(OH)2D clearly showed the best correlation with components of the endocrine vitamin D system and/or potential cellular targets of activation of the vitamin D receptor. When comparing the three 25OHD metabolites, both free 25OHD and bioavailable 25OHD showed an overall good correlation with the analyzed clinical laboratory parameters, bioavailable 25OHD might perform even better then free 25OHD. The only exception is PTH. It correlates well with total 25OHD most likely because the parathyroid cells have a highly specific uptake mechanism for total 25OHD—the megalin/cubilin-uptake system. This specific system acts much faster as compared to the uptake via the cell membrane. With the exception of PTH, correlations of 25(OH)D and 1,25(OH)2D metabolites with typical clinical chemistry readouts go in opposite directions suggesting potentially opposite effects of either 25(OH)D or 1,25(OH)2D on the nuclear vitamin D receptor in some target cells of vitamin D (see also Fig. 1 summarizing the key findings of our study). This hypothesis based on clinical data needs to be confirmed in adequately designed basic science studies.
Figure 1.
Key findings of our study are: (i) Both free 25(OH)D and bioavailable 25(OH)D showed better correlations with gestational age, bone and lipid metabolism biomarkers than total 25(OH)D. (ii) Total 1,25(OH)2D is superior to free 1,25(OH)2D and bioavailable 1,25(OH)2D. (iii) 25(OH)D metabolites and 1,25(OH)2D metabolites correlated with gestational age, bone and lipid biomarkers in opposite directions.
Supplementary Information
Abbreviations
- 125(OH)2D
1,25-Dihydroxyvitamin D
- 25OHD
25-Hydroxyvitamin D
- ANOVA
Analysis of variance
- B25OHD
Bioavailable 25-hydroxyvitamin D
- B125(OH)2D
Bioavailable 1,25-dihydroxyvitamin D
- BSAP
Bone-specific alkaline phosphatase
- CE
Conformité Européene
- DBP
Vitamin D-binding protein
- ELISA
Enzyme-linked immunosorbent assay
- F25OHD
Free 25-hydroxyvitamin D
- F125(OH)2D
Free 1,25-dihydroxyvitamin D
- fT3
Free triiodothyronine
- fT4
Free thyroxine
- HDL
High-density lipoprotein cholesterol
- IVD
In-Vitro-Diagnostic Medical Device
- LC–MS/MS
Liquid chromatography–tandem mass spectrometry
- LDL
Low-density lipoprotein cholesterol
- MCH
Mean corpuscular hemoglobin
- MCHC
Mean corpuscular hemoglobin concentration
- MCV
Mean corpuscular volume of red blood cells
- PTH
Parathyroid hormone
- RBC
Red blood cell
- RDW
Red blood cell distribution width
- SD
Standard deviation
- T25OHD
Total 25-hydroxyvitamin D
- T125(OH)2D
Total 1,25-dihydroxyvitamin D
- VDR
Vitamin D receptor
- WBC
White blood cell
Author contributions
All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission. BH designed the study. O.T., B.K.K. and B.H. wrote the manuscript. B.H. and O.T. did statistics, O.T., C.C., C.B. and S.E. collected the data.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-81452-9.
References
- 1.Bikle D, Bouillon R, Thadhani R, Schoenmakers I. Vitamin D metabolites in captivity? Should we measure free or total 25(OH)D to assess vitamin D status? J. Steroid Biochem. Mol. Biol. 2017;173:105–116. doi: 10.1016/j.jsbmb.2017.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bikle DD, Gee E. Free, and not total, 1,25-dihydroxyvitamin D regulates 25-hydroxyvitamin D metabolism by keratinocytes. Endocrinology. 1989;124:649–654. doi: 10.1210/endo-124-2-649. [DOI] [PubMed] [Google Scholar]
- 3.Bikle DD, Gee E, Halloran B, Haddad JG. Free 1,25-dihydroxyvitamin D levels in serum from normal subjects, pregnant subjects, and subjects with liver disease. J. Clin. Invest. 1984;74:1966–1971. doi: 10.1172/JCI111617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dwi Putra SE, et al. Being born large for gestational age is associated with increased global placental DNA methylation. Sci. Rep. 2020;10:927. doi: 10.1038/s41598-020-57725-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yong-Ping L, et al. Impact of maternal smoking associated lyso-phosphatidylcholine 20:3 on offspring brain development. J. Steroid Biochem. Mol. Biol. 2020;199:105591. doi: 10.1016/j.jsbmb.2020.105591. [DOI] [PubMed] [Google Scholar]
- 6.Bikle DD, et al. Assessment of the free fraction of 25-hydroxyvitamin D in serum and its regulation by albumin and the vitamin D-binding protein. J. Clin. Endocrinol. Metab. 1986;63:954–959. doi: 10.1210/jcem-63-4-954. [DOI] [PubMed] [Google Scholar]
- 7.Bikle DD, Siiteri PK, Ryzen E, Haddad JG. Serum protein binding of 1,25-dihydroxyvitamin D: A reevaluation by direct measurement of free metabolite levels. J. Clin. Endocrinol. Metab. 1985;61:969–975. doi: 10.1210/jcem-61-5-969. [DOI] [PubMed] [Google Scholar]
- 8.Powe CE, et al. Vitamin D-binding protein modifies the vitamin D-bone mineral density relationship. J. Bone Miner. Res Off. J. Am. Soc. Bone Miner. Res. 2011;26:1609–1616. doi: 10.1002/jbmr.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim HJ, et al. Clinical utility of measurement of vitamin D-binding protein and calculation of bioavailable vitamin D in assessment of vitamin D status. Ann. Lab. Med. 2017;37:34–38. doi: 10.3343/alm.2017.37.1.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tsuprykov O, Buse C, Skoblo R, Haq A, Hocher B. Reference intervals for measured and calculated free 25-hydroxyvitamin D in normal pregnancy. J. Steroid Biochem. Mol. Biol. 2018;181:80–87. doi: 10.1016/j.jsbmb.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 11.Tsuprykov O, Buse C, Skoblo R, Hocher B. Comparison of free and total 25-hydroxyvitamin D in normal human pregnancy. J. Steroid Biochem. Mol. Biol. 2019;190:29–36. doi: 10.1016/j.jsbmb.2019.03.008. [DOI] [PubMed] [Google Scholar]
- 12.Bouillon R, Van Assche FA, Van Baelen H, Heyns W, De Moor P. Influence of the vitamin D-binding protein on the serum concentration of 1,25-dihydroxyvitamin D3. Significance of the free 1,25-dihydroxyvitamin D3 concentration. J. Clin. Invest. 1981;67:589–596. doi: 10.1172/JCI110072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schwartz JB, et al. A comparison of measured and calculated free 25(OH) vitamin D levels in clinical populations. J. Clin. Endocrinol. Metab. 2014;99:1631–1637. doi: 10.1210/jc.2013-3874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schwartz JB, et al. Variability in Free 25(OH) Vitamin D Levels in Clinical Populations. J. Steroid Biochem. Mol. Biol. 2014;144PA:156–158. doi: 10.1016/j.jsbmb.2013.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jones KS, Assar S, Prentice A, Schoenmakers I. Vitamin D expenditure is not altered in pregnancy and lactation despite changes in vitamin D metabolite concentrations. Sci. Rep. 2016;6:26795. doi: 10.1038/srep26795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Best CM, Pressman EK, Queenan RA, Cooper E, O’Brien KO. Longitudinal changes in serum vitamin D binding protein and free 25-hydroxyvitamin D in a multiracial cohort of pregnant adolescents. J. Steroid Biochem. Mol. Biol. 2019;186:79–88. doi: 10.1016/j.jsbmb.2018.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gustafsson MK, et al. Alterations in the vitamin D endocrine system during pregnancy: A longitudinal study of 855 healthy Norwegian women. PLoS ONE. 2018;13:e0195041. doi: 10.1371/journal.pone.0195041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Powe Camille E, et al. First trimester vitamin D, vitamin D binding protein, and subsequent preeclampsia. Hypertension. 2010;56:758–763. doi: 10.1161/HYPERTENSIONAHA.110.158238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tsuprykov O, et al. Why should we measure free 25(OH) vitamin D? J. Steroid Biochem. Mol. Biol. 2018;180:87–104. doi: 10.1016/j.jsbmb.2017.11.014. [DOI] [PubMed] [Google Scholar]
- 20.Nielson CM, et al. Role of assay type in determining free 25-hydroxyvitamin D levels in diverse populations. N. Engl. J. Med. 2016;374:1695–1696. doi: 10.1056/NEJMc1513502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nielson CM, et al. Free 25-hydroxyvitamin D: Impact of vitamin D binding protein assays on racial-genotypic associations. J. Clin. Endocrinol. Metab. 2016;101:2226–2234. doi: 10.1210/jc.2016-1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hoofnagle AN, Eckfeldt JH, Lutsey PL. Vitamin D-binding protein concentrations quantified by mass spectrometry. N. Engl. J. Med. 2015;373:1480–1482. doi: 10.1056/NEJMc1502602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bouillon R, Schuit F, Antonio L, Rastinejad F. Vitamin D binding protein: A historic overview. Front. Endocrinol. (Lausanne) 2020 doi: 10.3389/fendo.2019.00910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kovacs CS. Chapter 42—Pregnancy, Lactation, and Postweaning Recovery. In: Feldman D, editor. Vitamin D. 4. Cambridge: Academic Press; 2018. pp. 755–782. [Google Scholar]
- 25.Olmos-Ortiz A, Avila E, Durand-Carbajal M, Díaz L. Regulation of calcitriol biosynthesis and activity: Focus on gestational vitamin D deficiency and adverse pregnancy outcomes. Nutrients. 2015;7:443–480. doi: 10.3390/nu7010443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Karras SN, Wagner CL, Castracane VD. Understanding vitamin D metabolism in pregnancy: From physiology to pathophysiology and clinical outcomes. Metab. Clin. Exp. 2018;86:112–123. doi: 10.1016/j.metabol.2017.10.001. [DOI] [PubMed] [Google Scholar]
- 27.Wilson SG, Retallack RW, Kent JC, Worth GK, Gutteridge DH. Serum free 1,25-dihydroxyvitamin D and the free 1,25-dihydroxyvitamin D index during a longitudinal study of human pregnancy and lactation. Clin. Endocrinol. (Oxf.) 1990;32:613–622. doi: 10.1111/j.1365-2265.1990.tb00905.x. [DOI] [PubMed] [Google Scholar]
- 28.Lundgren S, et al. Tissue distribution of human gp330/megalin: A putative Ca2+-sensing protein. J. Histochem. Cytochem. 1997;45:383–392. doi: 10.1177/002215549704500306. [DOI] [PubMed] [Google Scholar]
- 29.Bikle DD, Malmstroem S, Schwartz J. Current controversies: Are free vitamin metabolite levels a more accurate assessment of vitamin D status than total levels? Endocrinol. Metab. Clin. North Am. 2017;46:901–918. doi: 10.1016/j.ecl.2017.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schoenmakers I, Jones KS. Chapter 37—Pharmacology and Pharmacokinetics. In: Feldman D, editor. Vitamin D. 4. Cambridge: Academic Press; 2018. pp. 635–661. [Google Scholar]
- 31.Soma-Pillay P, et al. Physiological changes in pregnancy. Cardiovasc. J. Afr. 2016;27:89–94. doi: 10.5830/CVJA-2016-021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Heijboer AC, Blankenstein MA, Kema IP, Buijs MM. Accuracy of 6 routine 25-hydroxyvitamin D assays: Influence of vitamin D binding protein concentration. Clin. Chem. 2012;58:543–548. doi: 10.1373/clinchem.2011.176545. [DOI] [PubMed] [Google Scholar]
- 33.Arnaud J, Constans J. Affinity differences for vitamin D metabolites associated with the genetic isoforms of the human serum carrier protein (DBP) Hum. Genet. 1993;92:183–188. doi: 10.1007/BF00219689. [DOI] [PubMed] [Google Scholar]
- 34.Bouillon R, Van Baelen H, De Moor P. Comparative study of the affinity of the serum vitamin d-binding protein. J. Steroid Biochem. 1980;13:1029–1034. doi: 10.1016/0022-4731(80)90133-8. [DOI] [PubMed] [Google Scholar]
- 35.Kawakami M, Imawari M, Goodman DS. Quantitative studies of the interaction of cholecalciferol (vitamin D3) and its metabolites with different genetic variants of the serum binding protein for these sterols. Biochem. J. 1979;179:413–423. doi: 10.1042/bj1790413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Boutin B, Galbraith RM, Arnaud P. Comparative affinity of the major genetic variants of human group-specific component (vitamin D-binding protein) for 25-(OH) vitamin D3. J. Steroid Biochem. 1989;32:59–63. doi: 10.1016/0022-4731(89)90014-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.