Abstract
Background
COVID-19 is a disease associated with an intense systemic inflammation that could induce severe acute respiratory distress syndrome (ARDS), with life-threatening hypoxia and hypercapnia. We present a case where mild therapeutic hypothermia was associated with improved gas exchange, facing other therapies' unavailability due to the pandemic.
Case report
A healthy 38-year-old male admitted for COVID-19 pneumonia developed extreme hypoxia (PaO2/FiO2 ratio 42 mmHg), respiratory acidosis, and hyperthermia, refractory to usual treatment (mechanical ventilation, neuromuscular blockade, and prone position), and advanced therapies were not available. Mild therapeutic hypothermia management (target 33–34 °C) was maintained for five days, with progressive gas exchange improvement, which allowed his recovery over the following weeks. He was discharged home after 68 days without significant ICU associated morbidity.
Conclusions
Mild hypothermia is a widely available therapy, that given some specific characteristics of COVID-19, may be explored as adjunctive therapy for life-threatening ARDS, especially during a shortage of other rescue therapies.
Keywords: COVID-19, Mild hypothermia, Acute respiratory distress syndrome, Hypoxemia
1. Background
Some patients with COVID-19 pneumonia develop severe acute respiratory distress syndrome (ARDS). In this setting, the profound alteration of gas exchange refractory to mechanical ventilation (MV) leads to multiorgan failure, ultimately responsible for the high mortality observed. Rescue therapies for severe hypoxia, like Extra Corporeal Membrane Oxygenation (ECMO), have been limited due to the surge of cases during the COVID-19 pandemic, that exceeded the response capacity of most health systems [1].
COVID-19 frequently triggers a hyperinflammatory state that has been associated with the escalation of care and mortality [2]. Systemic hyperinflammation in COVID-19 produces a severe derangement of metabolism, with high energy expenditure, oxygen consumption, and protein catabolism, among others [3]. In the respiratory system, this dramatic shift causes an increase in minute ventilation and work of breathing, being one of the mechanisms proposed to explain the progression of lung injury in this entity [4].
Induced hypothermia is the controlled lowering of body temperature for therapeutic purposes. Hypothermia has many effects on homeostasis, mainly due to a nonspecific hypometabolic state, although many other mechanisms have been described. On this basis, therapeutic hypothermia has been extended to several clinical situations where tissue dysoxia is an important issue, like management after cardiac arrest, during cardiopulmonary bypass, and preservation of organ grafts [[5], [6], [7], [8], [9], [10]].
We report the use of mild therapeutic hypothermia as rescue therapy in a patient with ARDS due to COVID-19, who developed catastrophic respiratory system dysfunction with severe hypoxemia and hypercapnia refractory to MV, prone position, and neuromuscular blockers, facing the unavailability of extracorporeal support therapies.
2. Case report
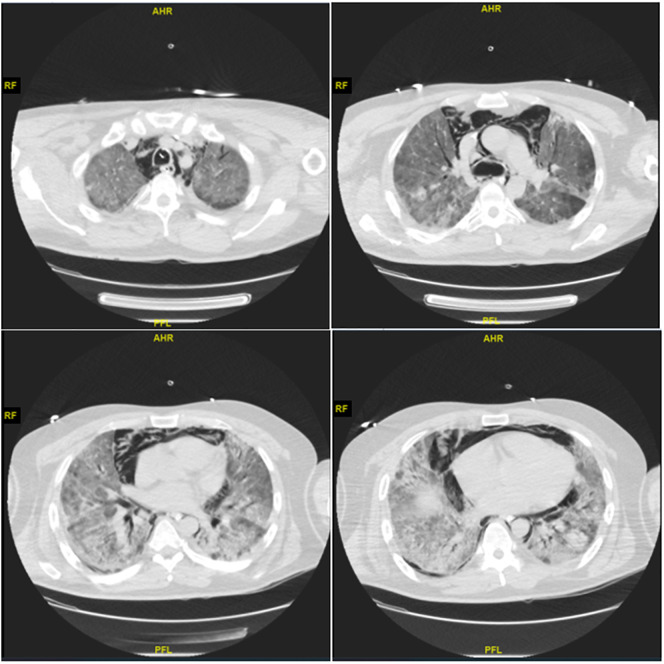
A previously healthy eutrophic (BMI 24 kg/m2), 38 years-old male of native south-american ethnicity (Mapuche) was admitted due to COVID-19 pneumonia. Initial respiratory support was a high-flow nasal cannula, but he developed severe hypoxemia and dyspnea ten days after admission. He required emergency intubation, and he was transferred to the pediatric intensive care unit due to the allocation of critical beds during the pandemic. A Chest CT scan ruled out pulmonary embolism and showed large bilateral consolidations and a moderate pneumomediastinum (Fig. 1 ).
Fig. 1.
Thoracic computed tomographic scan showing extensive bilateral pulmonary consolidation and pneumomediastinum in a patient with severe ARDS associated with SARS-CoV-2.
During the first week of MV, he had persistent hypoxemia, PaO2/FIO2 between 71 and 118 mmHg, and respiratory acidosis, PaCO2 between 65 and 84 mmHg. In this period, respiratory management included low tidal volume (VT) strategy, prone position, and neuromuscular blockade. Compliance of respiratory system (CRS) remained about 23 mL/cmH2O and MV parameters were: VT 350 mL (VT 4.7 mL/kg IBW), Plateau pressure (PPL) 26 cmH2O, total PEEP 11 cmH2O and driving pressure (DP) 15 cmH2O. He received empirical wide-spectrum antibiotics and steroids.
On the 17th day of MV, his condition progressively deteriorated, developing over 48 h profound hypoxemia and severe respiratory acidosis (PaO2/FIO2 42 mmHg, PaCO2 128 mmHg, and pH 7.0). His core body temperature was persistently higher than 39 °C, and he also had severe hemodynamic alterations, tachycardia between 150 and 175 bpm, profound hypotension that required norepinephrine up to 0.7 μg·kg−1·min−1. Remarkably, during that 48 h, his inflammatory markers spiked, D-Dimer 1093 mg/mL, ferritin 3652 ng/mL, and C-reactive protein 332 mg/L.
He was presented to the national program of extracorporeal therapies for ECMO or Interventional Lung Assist Device (i.e., Novalung®) for emergency cannulation due to progressive deterioration of gas exchange, and eventually, as a bridge to lung transplantation. Their final decision was that he was not suitable for extracorporeal therapies due to hemodynamic instability, MV greater than eight days, and poor prognosis. This decision was taken even considering our case's characteristics, like the patient's age, no previous comorbidities, no renal replacement therapy during ICU stay, and neurologic function assessment without signs of severe injury (normal cerebral CT scan and only sedation-induced changes in electroencephalography). Mild therapeutic hypothermia (33–34 °C) was proposed as a compassionate treatment given our patient's particular clinical characteristics, and the patient's spouse consented to its use. Mild therapeutic hypothermia was induced by Blanketrol® III (Cincinnati Sub-Zero) automated surface cooling device, aiming 33.5 °C servo-controlled core body temperature. The temperature was measured using an esophageal probe, and the gas exchange measurements were corrected to the patient's core body temperature at the time of blood sampling.
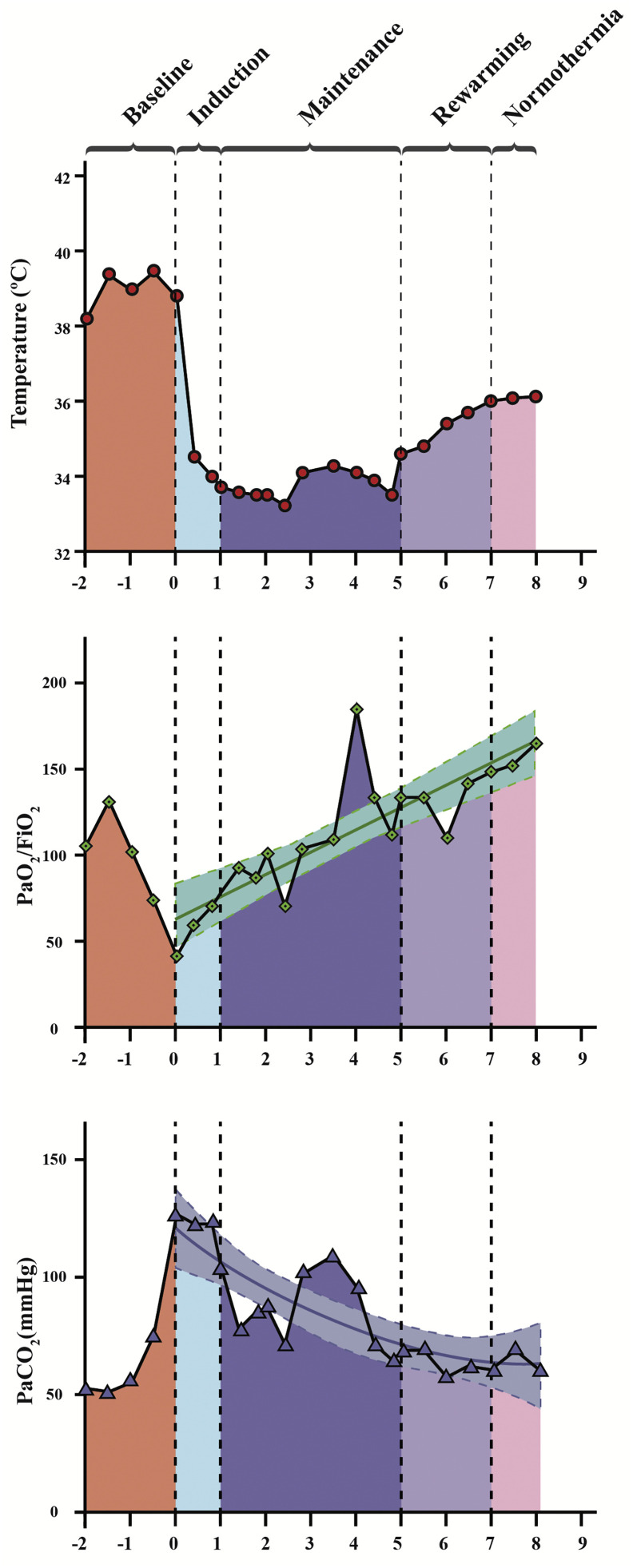
The patient was cooled to the target temperature in less than 1 h. Hemodynamics quickly improved, decreasing norepinephrine to 0.3 μg·kg−1·min−1 and heart rate to 120 bpm (Table 1 ). Gas exchange improved slowly and steadily over 72 h of therapeutic hypothermia (Fig. 2 ), maintaining FIO2 1, deep sedation, and neuromuscular blockers and MV within safety thresholds. We planned a rewarming of 1 °C over 24 h till normothermia (36 °C). Rewarming speed was reduced due to the deterioration of gas exchange during the third day. The patient completed five days of servo-controlled hypothermia between 33 and 34 °C. During the whole period of mild hypothermia, the patient maintained normal central venous saturation, normal arterial lactate, and venous-to-arterial CO2 gradient (Table 1). Oxygen extraction was 30% at baseline, decreasing to less than 20% during hypothermia, and then increasing to normal values. All improvements were sustained after full rewarming. CRS did not change during hypothermia and rewarming. No complications, including bleeding or alteration in coagulation tests, were identified. Pseudomona aeruginosa and Klebsiella pneumoniae were obtained in tracheal culture, and antibiotics were adjusted accordingly.
Table 1.
Selected hemodynamic and respiratory parameters of the patient with severe COVID-19 associated acute respiratory index during the phases of mild therapeutic hypothermia treatment. Blood gas analysis was corrected by core body temperature of the patient.
| Baseline | Induction | Maintenance | Rewarming | Normothermia | |
|---|---|---|---|---|---|
| Temperature (°C) | 38.8 | 34.0–34.5 | 33.2–34.3 | 34.6–35.7 | 36.1–36.3 |
| Time (days) | -1 | 0–1 | 2–4 | 5–6 | 7 |
| pH | 7.05 | 7.09–7.19 | 7.25–7.45 | 7.37–7.48 | 7.34–7.46 |
| ScvO2 (%) | 46 | 72–75 | 76–77 | 78 | 74 |
| Lactate (mmol/L) | 1.0 | 1.0–1.1 | 0.5–0.9 | 0.8–1.0 | 0.5–1.1 |
| Veno-arterial PCO2 gradient (mmHg) | 6 | 11 | 0 | 0 | 9 |
| Oxygen extraction ratio (%) | 30.3 | 15.7 | 17.2–18.8 | 19.1 | 22.1 |
| Heart rate (bpm) | 172 | 114–126 | 96–112 | 98–114 | 102–108 |
| Norepinephrine (mcg·kg−1·min−1) | 0.7 | 0.2–0.4 | 0–0.2 | 0 | 0 |
Abbreviations: ScvO2, central venous oxygen saturation; PCO2, carbon dioxide partial pressure.
Fig. 2.
Effects of mild therapeutic hypothermia on gas exchange in a patient with severe ARDS associated with SARS-CoV-2.
He required a tracheostomy due to prolonged MV, weaning, and rehabilitation. MV was discontinued at day 49 after admission, the artificial airway was removed at day 52 after admission, and supplementary oxygen was discontinued at day 61. He was discharged home for ambulatory rehabilitation on day 68 after admission. He did not require supplemental oxygen, feeding per mouth with normal swallowing, and without significant neurologic sequelae.
3. Discussion
We report a case of compassionate use of mild therapeutic hypothermia for extreme hypoxemia and hypercapnia in a patient with COVID-19 ARDS and persistent hyperthermia. No rescue therapies for severe ARDS, including extracorporeal therapies, were available because of the overwhelmed health system during the SARS-CoV-2 pandemic peak. Mild therapeutic hypothermia followed by servo-controlled normothermia resulted in sustained improvement of the patient's clinical condition and gas exchange. Despite the high predicted mortality, the patient survived hospital discharge, with no significant ICU- acquired morbidity.
The protectives effects of hypothermia have been largely studied in preclinical studies [[11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23]]. From the clinical standpoint, Villar J. and Slutsky A. conducted a small, non-randomized, controlled study in 19 moribund adults with severe ARDS almost 30 years ago [24]. They reported improved lung function and lower mortality in patients who received mild hypothermia (32–35 °C) for 70 ± 15 h compared to normothermia. It is important to recall that we cannot directly extrapolate the results of the study of Villar J. and Slutsky A. to current clinical practice because it was done before the contemporary use of protective MV.
Oxygen uptake (VO2) and CO2 production (VCO2) also decrease during hypothermia, which could explain the sustained improvement in gas exchange in this patient [3,23]. Hypothermia can also reduce pulmonary shunt, improve oxygen alveolar partial pressure and arterial–mixed venous oxygen content, improving arterial oxygen partial pressure, coupling DO2 to VO2, thus reducing oxygen extraction [[24], [25], [26]]. The reduction of veno-arterial PCO2 gradient in our patient reflects the same physiologic effect. These effects might be more pronounced in the described case since the temperature was reduced by 5 °C from hyperthermia to mild hypothermia.
Preliminary data shows that energy expenditure is still high after the second week of severe COVID-19 cases, and a hyperinflammatory profile is associated with worse outcomes [27,28]. Some authors define a hyperinflammatory state as a C-reactive protein concentration greater than 150 mg/L, ferritin concentration of greater than 1500 μg/L, or D-dimer concentration greater than 2500 mg/L [29,30]. Two of those criteria were present in our case, supporting a severe case of COVID-19 in the inflammatory phase [3,27,31,32]. In this stage of the disease, the physiologic effects of controlled hypothermia may play a role in improving gas exchange, being more intense in hyperthermic subjects [20]. More important, maintaining mechanical ventilation strategy within safety limits due to lower minute ventilation may lead to a decrease in the energy dissipated by the MV over the lung parenchyma [11], one of the leading causes of VILI and the decompartmentalization of lung injury and failure of distant organs [21,33,34].
Regarding hemodynamics, mild hypothermia reduces cardiac output without evidence of increasing anaerobic oxygen consumption due to the decrease in VO2 proportional to the core body temperature and the coupling of DO2 and VO2. Our patient had an increased oxygen extraction ratio, up to 40%, that rapidly decreased to less than 20% during hypothermia. Mechanism not related to the metabolic rate can explain this observation, for example, the shift to the left of the dissociation curve of hemoglobin, increasing the diffusion of oxygen to tissues, regulation of microcirculation shunt in end-organ parenchyma, and improving oxygen utilization by tissues [23,24].
Rescue therapies for severe ARDS during the COVID-19 pandemic might become a scarce resource. Most indications and protocols need to adapt to widely available, not expensive, and easily applied therapies, and more importantly, interventions with less human resources needs. In this way, mild therapeutic hypothermia is a therapy that does not require expensive technology nor staffing, being an interesting intervention to explore in COVID-19 catastrophic respiratory failure.
Finally, we believe that the adequate selection of our patient (healthy young adult, eutrophic, without metabolic syndrome or comorbidities) was also a determining factor in its good outcome.
In conclusion, our patient treated with mild therapeutic hypothermia survived COVID-19 severe acute respiratory failure when other rescue therapies were unavailable due to the pandemic's stressed health system. Given our current understanding of COVID-19 as a trigger of an intense inflammatory response, mild therapeutic hypothermia might be a promising therapy due to its anti-inflammatory and hypometabolic effects. Future studies should better elucidate its effectiveness, timing, optimal target, use times, and rewarming speed, such as the new proposed clinical trial, the CHILL study [35].
Declaration of Competing Interest
The authors declare that they have no competing interests.
References
- 1.Richardson S., Hirsch J.S., Narasimhan M., et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the new York City area. JAMA. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Manson J.J., Crooks C., Naja M., et al. COVID-19-associated hyperinflammation and escalation of patient care: a retrospective longitudinal cohort study. Lancet Rheumatol. 2020 doi: 10.1016/S2665-9913(20)30275-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yu P.J., Cassiere H., Bocchieri K., DeRosa S., Yar S., Hartman A. Hypermetabolism in critically ill patients with COVID-19 and the effects of hypothermia: a case series. Metabol Open. 2020;7:100046. doi: 10.1016/j.metop.2020.100046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cruces P., Retamal J., Hurtado D.E., et al. A physiological approach to understand the role of respiratory effort in the progression of lung injury in SARS-CoV-2 infection. Crit Care. 2020;24:494. doi: 10.1186/s13054-020-03197-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rittenhouse E.A., Mori H., Dillard D.H., Merendino K.A. Deep hypothermia in cardiovascular surgery. Ann Thorac Surg. 1974;17:63–98. doi: 10.1016/s0003-4975(10)65062-x. [DOI] [PubMed] [Google Scholar]
- 6.Belzer F.O., Southard J.H. Principles of solid-organ preservation by cold storage. Transplantation. 1988;45:673–676. doi: 10.1097/00007890-198804000-00001. [DOI] [PubMed] [Google Scholar]
- 7.Moshkovitz Y., David T.E., Caleb M., Feindel C.M., de Sa M.P. Circulatory arrest under moderate systemic hypothermia and cold retrograde cerebral perfusion. Ann Thorac Surg. 1998;66:1179–1184. doi: 10.1016/s0003-4975(98)00805-4. [DOI] [PubMed] [Google Scholar]
- 8.The Hypothermia after Cardiac Arrest Study Group Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346:549–556. doi: 10.1056/NEJMoa012689. [DOI] [PubMed] [Google Scholar]
- 9.Bernard S.A., Gray T.W., Buist M.D., et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346:557–563. doi: 10.1056/NEJMoa003289. [DOI] [PubMed] [Google Scholar]
- 10.McIntyre L.A., Fergusson D.A., Hébert P.C., Moher D., Hutchison J.S. Prolonged therapeutic hypothermia after traumatic brain injury in adults: a systematic review. JAMA. 2003;289:2992–2999. doi: 10.1001/jama.289.22.2992. [DOI] [PubMed] [Google Scholar]
- 11.Hong S.B., Koh Y., Lee I.C., et al. Induced hypothermia as a new approach to lung rest for the acutely injured lung. Crit Care Med. 2005;33:2049–2055. doi: 10.1097/01.ccm.0000178186.37167.53. [DOI] [PubMed] [Google Scholar]
- 12.Lim C.M., Hong S.B., Koh Y., et al. Hypothermia attenuates vascular manifestations of ventilator-induced lung injury in rats. Lung. 2003;181:23–34. doi: 10.1007/s00408-002-0111-x. [DOI] [PubMed] [Google Scholar]
- 13.Lim C.M., Kim E.K., Koh Y., Kim W.S., Kim D.S., Kim W.D. Hypothermia inhibits cytokine release of alveolar macrophage and activation of nuclear factor kb in endotoxemic lung. Intensive Care Med. 2004;30:1638–1644. doi: 10.1007/s00134-004-2336-z. [DOI] [PubMed] [Google Scholar]
- 14.Lim C.M., Kim M.S., Ahn J.J., et al. Hypothermia protects against endotoxin-induced acute lung injury in rats. Intensive Care Med. 2003;29:453–459. doi: 10.1007/s00134-002-1529-6. [DOI] [PubMed] [Google Scholar]
- 15.Huang P.S., Tang G.J., Chen C.H., Kou Y.R. Whole-body moderate hypothermia confers protection from wood smoke-induced acute lung injury in rats: the therapeutic window. Crit Care Med. 2006;34:1160–1166. doi: 10.1097/01.CCM.0000207342.50559.0F. [DOI] [PubMed] [Google Scholar]
- 16.Kim K., Kim W., Rhee J.E., et al. Induced hypothermia attenuates the acute lung injury in hemorrhagic shock. J Trauma. 2010;68:373–381. doi: 10.1097/TA.0b013e3181a73eea. [DOI] [PubMed] [Google Scholar]
- 17.Suzuki S., Hotchkiss J., Takahashi T., Olson D., Adams A.B., Marini J.J. Effect of core body temperature on ventilator-induced lung injury. Crit Care Med. 2004;32:144–149. doi: 10.1097/01.CCM.0000098857.14923.44. [DOI] [PubMed] [Google Scholar]
- 18.Kira S., Daa T., Kashima K., Mori M., Noguchi T., Yokohama S. Mild hypothermia reduced expression of intercellular adhesion molecule-1 (ICAM-1) and the accumulation of neutrophils after acid-induced lung injury in the rat. Acta Anaesthesiol Scand. 2005;49:351–359. doi: 10.1111/j.1399-6576.2005.00593.x. [DOI] [PubMed] [Google Scholar]
- 19.Chu S.J., Perng W.C., Hung C.M., Chang D.M., Lin S.H., Huang K.L. Effects of various body temperatures after lipopolysaccharide-induced lung injury in rats. Chest. 2005;128:327–336. doi: 10.1378/chest.128.1.327. [DOI] [PubMed] [Google Scholar]
- 20.Morita Y., Oda S., Sadahiro T., et al. The effects of body temperature control on cytokine production in a rat model of ventilator-induced lung injury. Cytokine. 2009;47:48–55. doi: 10.1016/j.cyto.2009.04.004. [DOI] [PubMed] [Google Scholar]
- 21.Hasday J., Shah N., Mackowiak P., Tulapurkar M., Nagarsekar A., Singh I. Fever, hyperthermia, and the lung: it's all about context and timing. Trans Am Clin Climatol Assoc. 2011;122:34–47. [PMC free article] [PubMed] [Google Scholar]
- 22.Cruces P., Ronco R., Erranz B., et al. Mild hypothermia attenuates mechanical ventilation-induced lung injury in a rat model. Exp Lung Res. 2011;37:549–554. doi: 10.3109/01902148.2011.616983. [DOI] [PubMed] [Google Scholar]
- 23.Cruces P., Erranz B., Donoso A., et al. Mild hypothermia increases pulmonary anti-inflammatory response during protective mechanical ventilation in a piglet model of acute lung injury. Paediatr Anaesth. 2013;23:1069–1077. doi: 10.1111/pan.12209. [DOI] [PubMed] [Google Scholar]
- 24.Villar J., Slutsky A.S. Effects of induced hypothermia in patients with septic adult respiratory distress syndrome. Resuscitation. 1993;26:183–192. doi: 10.1016/0300-9572(93)90178-s. [DOI] [PubMed] [Google Scholar]
- 25.Karnatovskaia L.V., Festic E., Freeman W.D., Lee A.S. Effect of therapeutic hypothermia on gas exchange and respiratory mechanics: a retrospective cohort study. Ther Hypothermia Temp Manag. 2014;4(2):88–95. doi: 10.1089/ther.2014.0004. [DOI] [PubMed] [Google Scholar]
- 26.Schumacker P.T., Rowland J., Saltz S., Nelson D.P., Wood L.D. Effects of hyperthermia and hypothermia on oxygen extraction by tissues during hypovolemia. J Appl Physiol. 1987;63(3):1246–1252. doi: 10.1152/jappl.1987.63.3.1246. [DOI] [PubMed] [Google Scholar]
- 27.Whittle J., Molinger J., MacLeod D., et al. Persistent hypermetabolism and longitudinal energy expenditure in critically ill patients with COVID-19. Crit Care. 2020;24:581. doi: 10.1186/s13054-020-03286-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dietz M., Chironi G., Claessens Y.E., et al. COVID-19 pneumonia: relationship between inflammation assessed by whole-body FDG PET/CT and short-term clinical outcome. Eur J Nucl Med Mol I. 2020:1–9. doi: 10.1007/s00259-020-04968-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reddy K., Rogers A.J., Mcauley D.F. Delving beneath the surface of hyperinflammation in COVID-19. Lancet Rheumatol. 2020;2:e578–e579. doi: 10.1016/S2665-9913(20)30304-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gustine J.N., Jones D. Immunopathology of hyperinflammation in COVID-19. Am J Pathol. 2021;19(1):4–17. doi: 10.1016/j.ajpath.2020.08.009. S0002-9440(20)30409-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ruiz-Irastorza G., Pijoan J.I., Bereciartua E., et al. Second week methyl-prednisolone pulses improve prognosis in patients with severe coronavirus disease 2019 pneumonia: an observational comparative study using routine care data. PLoS One. 2020;15:28–68. doi: 10.1371/journal.pone.0239401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.De Simone G., Mancusi C. Finding the right time for anti-inflammatory therapy in COVID-19 [published online ahead of print, 2020 Oct 1] Int J Infect Dis. 2020;S1201-9712(20):32170–32176. doi: 10.1016/j.ijid.2020.09.1454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gattinoni L., Tonetti T., Cressoni M., et al. Ventilator-related causes of lung injury: the mechanical power. Intensive Care Med. 2016;42:1567–1575. doi: 10.1007/s00134-016-4505-2. [DOI] [PubMed] [Google Scholar]
- 34.Polidoro R.B., Hagan R.S., De Santis Santiago R., Schmidt N.W. Overview: systemic inflammatory response derived from lung injury caused by SARS-CoV-2 infection explains severe outcomes in COVID-19. Front Immunol. 2020;11:1626. doi: 10.3389/fimmu.2020.01626. https://clinicaltrials.gov/ct2/show/NCT03376854?term=hypothermia&cond=COVID&draw=2&rank=1 [DOI] [PMC free article] [PubMed] [Google Scholar]