Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is responsible for the ongoing global Coronavirus 2019 (COVID-19) pandemic, resulting in thousands of deaths worldwide and representing a health challenge with few precedents in human history. Angiotensin-converting enzyme 2 (ACE-2) facilitates the access of SARS-CoV-2 to cells. Therapeutic agents acting on the renin–angiotensin system (RAS) might be able to modulate the concentration of ACE-2 and the various components of the system. Here, we discuss current pharmacological, molecular, and clinical evidence to investigate whether drugs acting on RAS with modulation of the ACE-2 concentration have added value in combating SARS-CoV-2 infection. We also highlight the possible deleterious action of the ACE/Ang-II/AT-1r axis and possible beneficial role of the ACE-2/Ang 1-7/MasR axis in acute respiratory distress syndrome (ARDS) caused by SARS-CoV-2, discussing the possibility of addressing the various RAS components with drug treatments to improve clinical outcomes.
SARS-CoV-2
SARS-CoV-2 is responsible for the current global COVID-19 pandemic, causing thousands of deaths and for a health challenge with few precedents in human history. SARS-CoV-2 is a family of RNA viruses capable of infecting humans and causing respiratory tract infections, respiratory distress syndrome, and serious lung injury in some cases [1]. SARS-Cov-2 has an ∼80% genome similarity to that of SARS-CoV responsible for a 2003 outbreak 2, 3. SARS-Cov-2 infection occurs in three stages: (i) asymptomatic or mildly symptomatic; (ii) moderately severe, characterized by a pulmonary inflammation; and (iii) very severe, characterized by a generalised inflammatory state affecting all tissues causing multiorgan dysfunction [4]. Biochemical interaction studies have shown that SARS-CoV-2 enters host cells mainly through the use of the spike protein (S) [5]. This protein contains two functional subunits S1 and S2: S1 binds the surface receptors of the host cells, whereas S2 is responsible for membrane fusion between the virus and the host cell [6]. SARS-CoV-2 penetrates cells using the S protein via the ACE-2 receptor on the cell surface, which is widely present in epithelial cells of the respiratory mucosa [6]. ACE-2 is also a converting enzyme that is part of RAS. It is unclear whether an increase in ACE-2 is responsible for a greater likelihood of COVID-19 infection. The level of expression of ACE-2 in the respiratory tract of smokers is higher than that of nonsmokers and SARS-CoV-2 is more likely to infect smokers [7]. Lung tissues are probably an easier entry route for SARS-CoV-2 because 83% of ACE-2 receptors are present in type-II pneumocytes that produce surfactants that prevent alveoli from collapsing [8]. In addition, the expression of ACE-2 changes during the various stages of the infection. To date, there is no conclusive evidence on the role of RAS and ACE-2 in the three stages of SARS-CoV-2 infection. Drugs acting on RAS are widely used in the treatment of cardiovascular diseases, but what is the correlation between these drugs, RAS, and COVID-19? Could they have a protective role by modulating the expression of RAS components or should they be considered risk factors for COVID-19?
RAS and SARS-CoV-2
RAS is a physiological system controlling blood pressure and electrolyte balance in the body. It also exerts numerous local effects aimed at regulating tissue physiology and homeostasis [9]. RAS can influence growth, proliferation, differentiation, and cell apoptosis, and also has effects on inflammation. RAS alterations are involved in many diseases, including atherosclerosis, diabetes mellitus, and viral infections, such as SARS-CoV-2 10, 11. Therefore, it is crucial to investigate how the expression of the various components of RAS vary in tissues during SARS-CoV-2 infection and whether a change in their expression caused by drugs acting on RAS could have repercussions and correlations with COVID-19 infection at various stages.
The classical route of RAS includes subsequent enzymatic reactions that lead to the conversion of the angiotensinogen substrate (AGT) into the active angiotensin-II (Ang-II) peptide, which binds to its specific membrane AT receptors to exert its effects. In the bloodstream, AGT is converted to angiotensin I (Ang-I) by renin; Ang-I is further processed by angiotensin-converting enzyme (ACE) to release vasoactive angiotensin-II (Ang-II). In addition to Ang-II production, ACE can degrade several vasodilator peptides, including bradykinin and kallikrein [12]. Bradykinin causes vasodilation through the production of prostaglandins and nitric oxide (NO) and inhibits the proliferation of smooth vascular muscle.
Ang-II acts on receptors to regulate blood pressure and electrolyte balance. At the cellular level, Ang-II modulates cell contraction, growth, differentiation, and apoptosis; it can also promote the production of other cytokines, the expression of adhesion molecules and subsequent recovery of inflammatory cells, chemotaxis, and macrophage activation. The increase in Ang-II formation induces inflammation and Ang-II is itself a powerful proinflammatory cytokine as well as a growth factor. It also activates the transcriptional factor nuclear factor (NF)-κb, a key factor of nuclear transcription in inflammatory and fibrotic diseases, and its activation allows the transcription of several inflammatory genes, including IL6 and IL1, which are responsible for the cytokinic cascade and the hyperactive inflammatory state that is generated mainly during the third stage of COVID infection 13, 14.
Nonclassical RAS pathways include angiotensin 1-7 (Ang 1–7) from Ang-II and angiotensin 1–9 (Ang 1–9) from Ang-I by ACE-2 conversion. Ang 1–7 oppose the actions of the AT1 receptor (AT-1 r) mediated by Ang-II [15] and this leads to vasodilator/antiproliferative effects. ACE-2 can generate Ang 1–7 directly from Ang-II, or indirectly from Ang-I via Ang 1–9 intermediates [16]. Ang 1–7 exert their effects via the Mas receptor (MasR). By binding to MasR, Ang 1–7 can induce vasodilation, cell growth inhibition, antithrombosis, antifibrosis and anti-inflammatory effects [17].
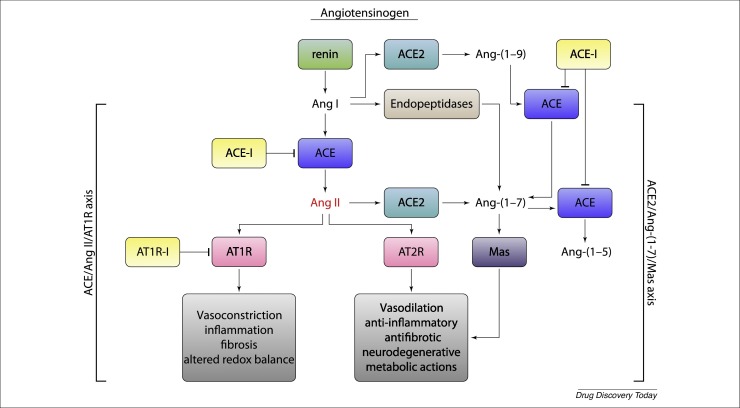
Ang 1–9 exert several eff ;ects in vivo and in vitro independently of Ang 1–7-mediated MasR activation, possibly through AT2 receptors (AT2-r). Ang 1–9 also have antifibrotic and antiproliferative effects [18] (Fig. 1 ).
Figure 1.
The renin–angiotensin system (RAS). Vasodilatory, anti-inflammatory, and antifibrotic effects mediated by stimulation of the AT2 (AT2R) and Mas receptors (MasR) could be of clinical benefit during the most severe stages of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, whereas the vasoconstrictory, proinflammatory, and profibrotic effects mediated by stimulation of AT1-r could increase lung damage.
Roles of ACE-2, Ang 1–7, and Ang 1–9
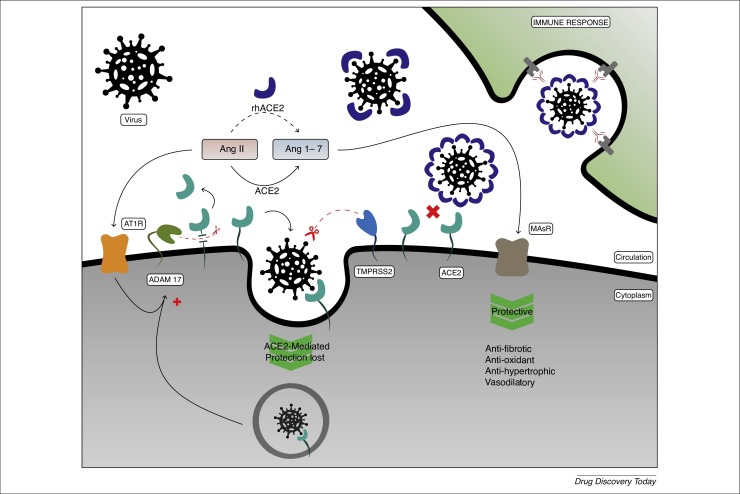
SARS-CoV-2 penetrates cells by binding S protein to ACE-2 (Fig. 2 ) [19]. In the current SARS-CoV-2 pandemic, evidence shows that ACE-2 receptors are the gateway to the virus. ACE-2 converts Ang-II to Ang 1–7, and converts Ang-I to Ang 1–9, which in turn is converted to Ang 1–7 by ACE and neprilysin. ACE-2 receptors are expressed in the heart, vessels, intestine and lungs 20, 21. ACE-2 occurs mainly on cell membranes and there is very little in circulation in soluble form.
Figure 2.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) penetrates cells by binding of the peak viral protein (spike; S) to angiotensin-converting enzyme 2 (ACE-2). ACE-2 converts angiotensin (Ang)-II to Ang 1–7. The latter have opposite biological actions to Ang-II (i.e., antifibrotic, antioxidant, and antihypertrophic effects) through stimulation of Mas receptor (MasR).
The role of ACE-2 in the pathophysiology of SARS-Cov-2 infection is not yet fully understood, because ACE-2 is not only the virus entry receptor, the modulation and variation of RAS and ACE-2 during the various stages of infection have been demonstrated, and the role of ACE-2 in infected lung tissue is unclear, especially during the more severe stages of infection, where it appears to have a protective role. These new findings could have a major impact on the development of new effective therapies to combat SARS-CoV-2 infection, or suggest additional therapeutic solutions for drugs modulating the RAS system.
To prevent viral replication, an interesting line of research could be that of the use of anti-ACE-2 antibodies to block the binding of SARS-CoV-2 to the receptor, whereas another option could be to block upstream the enzymatic cascade of RAS and decrease the expression of ACE-2 [8]. During the more severe stages of SARS-CoV-2 infection, where ACE-2 could have a protective role, an increase in ACE-2 expression could be beneficial. Other important aspects to consider are that the biological effects of Ang-II are vasoconstrictive and stimulate the release of aldosterone, which, in high concentrations, can also cause myocardial hypertrophy, interstitial fibrosis, endothelial dysfunction, and increased inflammation, oxidative stress, and coagulation, biological effects taht, if altered, can cause serious complications in patients with COVID-19 22, 23.
Finally, Ang-II can cause increased inflammation through production of IL-6, tumor necrosis factor (TNF)-α and other inflammatory cytokines 24, 25, 26. However, all these biological effects are mediated by AT-1 receptors. By contrast, ACE-2 can reduce the negative effects of Ang-II through several mechanisms, such as degradation of Ang-II and Ang 1–7 formation. As described earlier, recent evidence showed that Ang 1–7 can also be formed by the action of neprilysin. Ang 1–7 has opposite biological effects to Ang-II through MasR and AT2-r. MasRs are expressed on the surface of the smooth muscle cells of the bronchial muscle and alveolar epithelium 27, 28, where Ang 1–7 exerts anti-inflammatory and antifibrosis effects 29, 30. In addition, MasR are expressed on platelets, where Ang 1–7 mediates antiaggregant effects 31, 32, 33 by increasing prostacyclin and NO release [34]. Studies have demonstrated an antihypertrophic role for the Ang (1–9) receptors independent of Ang 1–7 MasR [35], proactively mediated by the AT2 receptor (AT2R) [8].
Agents acting on RAS and effects on ACE-2, Ang-II, Ang 1–7, and Ang 1–9
Based on the earlier considerations, the crucial role of RAS and ACE-2 in acute respiratory distress syndrome (ARDS) caused by SARS-CoV-2, and the correlation with the inflammatory status of lung tissues during the more severe stages of COIVD-19 infection, RAS-modulating therapeutic agents could indirectly have an important role in treating SARS-CoV-2 viral infection. Agents that are active against RAS interfere with various stages of the system, such as ACE inhibitors (ACEis), which block the AT-1 receptors of Ang-II (ARBs), or by directly inhibiting renin (renin inhibitors; DRis) [36]. These therapeutic agents are indicated for the treatment of cardiovascular diseases, such as hypertension and ischemic heart disease. Acting at different levels of the system, drugs acting on RAS can cause heterogeneous effects on the modulation of peptides and enzymes involved in the cascade. In vitro and animal studies have shown that the use of ACEis increases the levels of ACE-2, while the use of ARBs causes an increase in the expression and activity levels of ACE and ACE-2, 37, 38. Finally, administration of DRis is associated with a reduction in the expression of ACE and ACE-2 39, 40 (Table 1 ).
Table 1.
Agents active against RAS that modulate ACE-2 and their hypothetical use in the various stages of SARS-CoV-2 infectiona
| Phase of infection | ACE-2 (-) | ACE-2 (+) |
|---|---|---|
| 1 | DRis | |
| 2 | ACEis | |
| 3 | ARBs |
During the prevention and Phase 1 stages of viral infection, it might be useful to decrease ACE-2 concentrations with DRis, whereas, during the more severe stages, an increase in ACE-2 with ACEis and ARBs might have a protective role.
An increase in ACE-2 concentrations has already been documented in both mouse models and patients treated with ACEis and ARBs. An increase in the level of expression of these viral receptor suggest an increase in the infectious power SARS-CoV-2. During phase 1 of the infection, the virus penetrates the cell and replicates; thus, during this phase it might be useful to administer a DRi that, by acting upstream [41], lowers concentrations of ACE and ACE-2, decreasing the concentration of receptor proteins for the virus to attach to. Conversely, during phase 2 or 3 of the disease, where there is an overactive inflammatory state and where ACE-2 appears to have a protective role particularly in the respiratory tract [42], it might be appropriate to increase ACE-2 with ACEis or block the inflammatory effects of Ang-II using ARBs, which also increase the expression of ACE-2 itself. Other aspects to consider are that the use of ACEis could lead to an increase in bradykinin by decreasing ACE metabolization [43] and activating the inflammatory system.
Moreover, the use of ARBs could bring further benefits, in that blocking the AT-1 receptor would cause greater activation of AT2R by Ang-II, with resulting anti-inflammatory and antifibrosis effects. Thus, the use of ARBs might have added value compared with ACEis 44, 45. Finally, an increase in ACE or ACE-2 could lead to increased degradation of bradykinin, avoiding pro-inflammatory effects. In addition, for these three classes of drug acting on RAS, there is a potential indirect protection against SARS-CoV-2. Patients with cardiovascular disease are at high risk of pneumonia and studies have shown that the use of RAS-blocking drugs decreases this risk 46, 47.
Epidemiological evidence
As described earlier, in vitro and animal studies have shown that RAS-modulating agents can modify polypeptides and enzymes in the system; in particular, ACEis and ARBs can increase the expression of ACE-2. However, current epidemiological evidence is at odds with expected results based on in vitro and animal evidence. Mehra et al. [47] conducted a study with 8910 patients, and neither ACEis or ARBs were associated with an increased risk of infection or complications from COVID-19. Mancia et al. [48] conducted a case-control study of patients with COVID-19 in Italy and also showed no correlation between ACEi and ARB intake and increased risk of infection or COVID-19 complications. In addition, Reynolds et al. [49] conducted a study on 12 594 patients and showed no positive association between COVID-19 symptoms and ACEi and ARB use. Other epidemiological studies have reached similar conclusions 50, 51.
Despite each of these studies having its own weaknesses, they all suggest that the continued use of ACEis and ARBs is not harmful in patients with Covid-19, and does not increase the likelihood of infection. Ultimately, more studies will be needed to clarify whether ACEis or ARBs are harmful to patients with COVID-19.
Discussion
During the current COVID-19 pandemic, concerns have emerged over the use of different classes of drugs and treatments [52], particularly whether drugs that modulate RAS increase the risk of infection and complications associated with SARS-CoV-2, or whether, by modulating the components of RAS, they are indirectly useful? It is currently recommended not to discontinue treatment in patients with heart failure, chronic kidney disease, hypertension and associated diseases 53, 54. Therefore, there is currently no evidence to support the notion that the use of ACEis, ARBs, or DRis could increase SARS-CoV-2 infectivity. By discontinuing therapy with RAS blockers, patients will experience a significantly increased risk of cardiovascular morbidity and mortality considering the proven benefit of the therapy [55]. In addition, there is evidence showing that the activation of the Ang-II/AT1 r axis promotes lung injury and is harmful, whereas an increase in the ACE-2/Ang 1–7/MasR axis protects against lung injury. For the latter, there is the potential to protect lung injury by increasing ACE-2 using recombinant ACE-2 soluble in humans (rhACE-2) in patients with COVID-19 (Clinicaltrials.gov #NCT04287686).
The discrepancy between in vitro and animal results and clinical epidemiological data can be explained in terms of the selective variation in ACE and ACE-2 in tissues during treatment with RAS blockers, because no studies on the effects of RAS blockers on the activity of ACE-2 in tissues in the upper or lower respiratory tract have been reported [56]. However, we believe that reducing ACE2 expression with DRis either preventively or during phase 1 of viral infection could be an interesting option in the context of SARS-CoV-2 infection, and requires further investigation [57]. Although the clinical benefit of these pharmacological approaches has yet to be demonstrated, there is ample evidence to suggest that treatment with RAS blockers should not be discontinued in patients with COVID-19.
Declaration of interests
The authors declare no conflict of interest.
References
- 1.Baig A.M., et al. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chem. Neurosci. 2020;11:995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- 2.Liu Z., et al. Composition and divergence of coronavirus spike proteins and host ACE2 receptors predict potential intermediate hosts of SARS-CoV. Med. Virol. 2020;92:595–601. doi: 10.1002/jmv.25726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cascella M., et al. StatPearls; 2020. Features, Evaluation and Treatment Coronavirus (COVID-19) [PubMed] [Google Scholar]
- 4.Ashour H.M., et al. Insights into the recent 2019 novel coronavirus (SARS-CoV-2) in light of past human coronavirus outbreaks. Pathogens. 2020;4:9. doi: 10.3390/pathogens9030186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yan R., et al. Structural basis for the recognition of the SARS-CoV-2 by full-length human ACE2. Science. 2020;367:1444–1448. doi: 10.1126/science.abb2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Walls A.C., et al. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:894–904. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cai G., Bossé Y. Tobacco smoking increases the lung gene expression of ACE2, the receptor of SARS-CoV. Am. J. Respir. Crit. Care Med. 2020;201:1557–1559. doi: 10.1164/rccm.202003-0693LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang H., et al. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bader M. Tissue renin-angiotensin-aldosterone systems: targets for pharmacological therapy. Annu. Rev. Pharmacol. Toxicol. 2010;50:439–465. doi: 10.1146/annurev.pharmtox.010909.105610. [DOI] [PubMed] [Google Scholar]
- 10.Nehme A., Zibara K. Efficiency and specificity of RAAS inhibitors in cardiovascular diseases: How to achieve better end-organ protection? Hypertens. Res. 2017;40:903–909. doi: 10.1038/hr.2017.65. [DOI] [PubMed] [Google Scholar]
- 11.Meng J., et al. Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg. Microbes Infect. 2020;9:757–760. doi: 10.1080/22221751.2020.1746200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Atlas S.A. The renin-angiotensin aldosterone system: pathophysiological role and pharmacologic inhibition. J. Manag. Care Pharm. 2007;13:9–20. doi: 10.18553/jmcp.2007.13.s8-b.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nehme A., et al. An update on the tissue renin angiotensin system and its role in physiology and pathology. J. Cardiovasc. Dev. Dis. 2020;6:14. doi: 10.3390/jcdd6020014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Patel S., et al. Renin-angiotensin-aldosterone (RAAS): The ubiquitous system for homeostasis and pathologies. Biomed. Pharmacother. 2017;94:317–325. doi: 10.1016/j.biopha.2017.07.091. [DOI] [PubMed] [Google Scholar]
- 15.Ferrario C.M., et al. An evolving story of angiotensin-II-forming pathways in rodents and humans. Clin. Sci. 2014;126:461–469. doi: 10.1042/CS20130400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Donoghue M., et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ. Res. 2000;87:E1–E9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- 17.Simões Silva A.C., et al. ACE2, angiotensin-(1–7) and Mas receptor axis in inflammation and fibrosis. Br. J. Pharmacol. 2013;169:477–492. doi: 10.1111/bph.12159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hoffmann M., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kuba K., et al. Multiple functions of angiotensin-converting enzyme 2 and its relevance in cardiovascular diseases. Circ. J. 2013;77:301–308. doi: 10.1253/circj.cj-12-1544. [DOI] [PubMed] [Google Scholar]
- 20.Patel V.B., et al. Role of the ACE2/angiotensin 1–7 axis of the renin-angiotensin system in heart failure. Circ. Res. 2016;118:1313–1326. doi: 10.1161/CIRCRESAHA.116.307708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tan W.S.D., et al. Targeting the renin-angiotensin system as novel therapeutic strategy for pulmonary diseases. Curr. Opin. Pharmacol. 2018;40:9–17. doi: 10.1016/j.coph.2017.12.002. [DOI] [PubMed] [Google Scholar]
- 22.Recinos A., et al. Angiotensin-II induces IL-6 expression and the Jak-STAT3 pathway in aortic adventitia of LDL receptor-deficient mice. Atherosclerosis. 2007;194:125–133. doi: 10.1016/j.atherosclerosis.2006.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ferrara F., et al. The added value of Pirfenidone to fight inflammation and fibrotic state induced by SARS-CoV 2. Eur. J. Clin. Pharmacol. 2020;76:1615–1618. doi: 10.1007/s00228-020-02947-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yamamoto S., et al. Macrophage polarization by angiotensin-II-type 1 receptor aggravates renal injury-acceleration of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2011;31:2856–2864. doi: 10.1161/ATVBAHA.111.237198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee Y.B., et al. Cytokines, chemokines, and cytokine receptors in human microglia. J. Neurosci. Res. 2002;69:94–103. doi: 10.1002/jnr.10253. [DOI] [PubMed] [Google Scholar]
- 26.Magalhaes G.S., et al. Angiotensin-(1–7) attenuates airway remodelling and hyperresponsiveness in a model of chronic allergic lung inflammation. Br. J. Pharmacol. 2015;172 doi: 10.1111/bph.13057. 2330–2234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.El-Hashim A.Z., et al. Angiotensin-(1–7) inhibits allergic inflammation, via the MAS1 receptor, through suppression of ERK1/2- and NF-kappaB-dependent pathways. Br. J. Pharmacol. 2012;166:1964–1976. doi: 10.1111/j.1476-5381.2012.01905.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen Q., et al. Angiotensin-(1–7) attenuates lung fibrosis by way of Mas receptor in acute lung injury. J. Surg. Res. 2013;185:740–747. doi: 10.1016/j.jss.2013.06.052. [DOI] [PubMed] [Google Scholar]
- 29.Meng Y., et al. Angiotensin converting enzyme 2/angiotensin-(1–7)/Mas axis protects against lung fibrosis by inhibiting the MAPK/NF-kappaB pathway. Am. J. Respir. Cell Mol. Biol. 2014;50:723–736. doi: 10.1165/rcmb.2012-0451OC. [DOI] [PubMed] [Google Scholar]
- 30.Fang C., et al. Angiotensin 1–7 and Mas decrease thrombosis in Bdkrb2-/- mice by increasing NO and prostacyclin to reduce platelet spreading and glycoprotein VI activation. Blood. 2013;121:3023–3032. doi: 10.1182/blood-2012-09-459156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pai W.Y., et al. Angiotensin-(1–7) inhibits thrombin induced endothelial phenotypic changes and reactive oxygen species production via NADPH oxidase 5 downregulation. Front Physiol. 2017;8:994. doi: 10.3389/fphys.2017.00994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fraga-Silva R.A., et al. An orally active formulation of angiotensin-(1–7) produces an antithrombotic effect. Clinics (Sao Paulo) 2011;66:837–841. doi: 10.1590/S1807-59322011000500021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fraga-Silva R.A., et al. The antithrombotic effect of angiotensin-(1–7) involves mas-mediated NO release from platelets. Mol. Med. 2008;14:28–35. doi: 10.2119/2007-00073.Fraga-Silva. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ocaranza M.P., et al. Angiotensin-(1–9) regulates cardiac hypertrophy in vivo and in vitro. J. Hypertens. 2010;28:1054–1064. doi: 10.1097/hjh.0b013e328335d291. [DOI] [PubMed] [Google Scholar]
- 35.Flores-Munoz M., et al. Angiotensin-1–9 antagonises pro-hypertrophic signalling in cardiomyocytes via the angiotensin type2 receptor. J. Physiol. 2011;589:939–951. doi: 10.1113/jphysiol.2010.203075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Keidar S., et al. Mineralocorticoid receptor blocker increases angiotensin- converting enzyme 2 activity in congestive heart failure patients. Circ. Res. 2005;97:946–953. doi: 10.1161/01.RES.0000187500.24964.7A. [DOI] [PubMed] [Google Scholar]
- 37.Zhong J.C., et al. Telmisartan attenuates aortic hypertrophy in hypertensive rats by the modulation of ACE2 and profilin-1 expression. Regul. Pept. 2011;166:90–97. doi: 10.1016/j.regpep.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 38.Ferrario C.M., et al. Effect of angiotensin- converting enzyme inhibition and angiotensin-II receptor blockers on cardiac angiotensin- converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 39.Tipnis S.R., et al. A human homolog of angiotensin- converting enzyme: cloning and functional expression as a captopril- insensitive carboxypeptidase. J. Biol. Chem. 2000;275:33238–33243. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- 40.Mirabito Colafella K.M., et al. The renin-angiotensin-aldosterone system and its therapeutic targets. Exp. Eye Res. 2019;186:1076–1080. doi: 10.1016/j.exer.2019.05.020. [DOI] [PubMed] [Google Scholar]
- 41.Slabý A., Zák A. Renin, prorenin, and the direct renin inhibitor aliskiren. Cas. Lek. Cesk. 2010;149:55–60. [PubMed] [Google Scholar]
- 42.Aulakh G.K., et al. An update on non-peptide angiotensin receptor antagonists and related RAAS modulators. Life Sci. 2007;81:615–639. doi: 10.1016/j.lfs.2007.06.007. [DOI] [PubMed] [Google Scholar]
- 43.Ding W., et al. Aliskiren inhibits angiotensin-II/angiotensin 1–7(Ang-II/Ang1–7) signal pathway in rats with diabetic nephropathy. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2018;34:891–895. [PubMed] [Google Scholar]
- 44.Gurwitz D. Angiotensin receptor blockers as tentative SARS-CoV-2 therapeutics. Drug Dev. Res. 2020;81:537–540. doi: 10.1002/ddr.21656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Soto M., et al. Renin angiotensin system-modifying therapies are associated with improved pulmonary health. Clin. Diabetes Endocrinol. 2017;3:6. doi: 10.1186/s40842-017-0044-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Henry C., et al. Impact of angiotensin-converting enzyme inhibitors and statins on viral pneumonia. Proc. (Bayl. Univ. Med. Cent.) 2018;31:419–423. doi: 10.1080/08998280.2018.1499293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mehra M.R., et al. Cardiovascular disease, drug therapy, and mortality in Covid-19. N. Engl. J. Med. 2020;382:e102. doi: 10.1056/NEJMoa2007621. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 48.Mancia G., et al. Renin–angiotensin–aldosterone system blockers and the risk of Covid-19. N. Engl. J. Med. 2020;382:2431–2440. doi: 10.1056/NEJMoa2006923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Reynolds H.R., et al. Renin–angiotensin–aldosterone system inhibitors and risk of Covid-19. N. Engl. J. Med. 2020;382:2441–2448. doi: 10.1056/NEJMoa2008975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bean D., et al. Treatment with ACE-inhibitors is associated with less severe SARS-Covid-19 infection in a multi-site UK acute hospital trust. Eur. J. Heart Fail. 2020;22:967–974. doi: 10.1002/ejhf.1924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.De Abajo F., et al. Use of renin–angiotensin–aldosterone system inhibitors and risk of COVID-19 requiring admission to hospital: a case-population study. Lancet. 2020;395:1705–1714. doi: 10.1016/S0140-6736(20)31030-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ferrara F., et al. Worldwide recommendations and therapies for multiple sclerosis: are they safe in the COVID-19 pandemic period? SN Compr. Clin. Med. 2020 doi: 10.1007/s42399-020-00375-0. Published online June 26, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Williams B., et al. ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Cardiology and the European Society of Hypertension. J. Hypertens. 2018;36:1953–2041. doi: 10.1097/HJH.0000000000001940. [DOI] [PubMed] [Google Scholar]
- 54.Whelton P.K., et al. ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2018;71:e127–e248. doi: 10.1016/j.jacc.2017.11.006. [DOI] [PubMed] [Google Scholar]
- 55.Ponikowski P., et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016;37:2129–2200. doi: 10.1093/eurheartj/ehw128. [DOI] [PubMed] [Google Scholar]
- 56.Kreutz R., et al. Hypertension, the renin–angiotensin system, and the risk of lower respiratory tract infections and lung injury: implications for COVID-19. Cardiovasc. Res. 2020;116:1688–1699. doi: 10.1093/cvr/cvaa097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mourad J.J., Levy B.I. Interaction between RAAS inhibitors and ACE2 in the context of COVID-19. Nat. Rev. Cardiol. 2020;13:313. doi: 10.1038/s41569-020-0368-x. [DOI] [PMC free article] [PubMed] [Google Scholar]