Abstract
Background
Patients with severe COVID-19 disease frequently develop anaemia as the result of multiple mechanisms and often receive transfusions. The aims of this study were to assess the impact of repeated blood samplings on patients’ anaemic state using standard-volume tubes, in comparison with the hypothetical use of low-volume tubes and to evaluate the transfusion policy adopted.
Study design and methods
Transfusion data of mechanically ventilated non-bleeding patients with COVID-19 disease hospitalized in ICU for a minimum of 20 days were recorded. The total volume of blood drawn for samplings with standard-volume tubes and the corresponding red blood cell mass (RBCM) removed during hospitalization for each patient were calculated and compared with the hypothetical use of low-volume tubes.
Results
Twenty-four patients fulfilled the inclusion criteria. Ten patients were anaemic at ICU admission (41.7 %). Overall, 6658 sampling tubes were employed, for a total of 16,786 mL of blood. The median RBCM subtracted by blood samplings per patient accounted for about one third of the total patients’ RBCM decrease until discharge.
The use of low-volume tubes would have led to a median saving of about one third of the drawn RBCM.
Eleven patients were transfused (45.8 %) at a mean Hb value of 7.7 (± 0.5) g/dL.
Conclusion
The amount of blood drawn for sampling has a significant role in the development of anaemia and the use of low-volume tubes could minimize the problem.
Large high-powered studies are warranted to assess the more appropriate transfusion thresholds in non-bleeding critically ill patients with COVID-19 disease.
Keywords: Iatrogenic anaemia, COVID-19, Patient blood management
1. Introduction
The development of anaemia is common in patients during hospitalization in Intensive Care Unit (ICU) [1]. Several causes can lead to a reduction of the Red Blood Cell Mass (RBCM) in this setting. Bleeding caused by the underlying disease or induced by invasive procedures, impaired iron-restricted hematopoiesis secondary to inflammation [2] and reduced red blood cell lifespan are known as the most frequent causes [3]. Moreover, the daily amount of blood drawn for the frequent blood testing can represent a worsening factor.
With the aim to reduce, inter alia, the impact of iatrogenic anaemia, the Society of Critical Care Medicine and the American Association of Blood Banks support the Choosing Wisely campaign: “Do not order diagnostic test at regular intervals (such as every day), but rather in response to specific questions” [4] and “Don’t perform serial blood counts on clinically stable patients” [5], respectively.
After January 30th 2020, when the first two imported cases of SARS-CoV-2 infection in Italy were documented, a rapid increase in admissions of patients with severe acute respiratory syndrome to ICU has been observed during the following weeks. Overall, from March 8th until May 20th 2020 eighty-four SARS-CoV-2 patients were treated in ICU at the tertiary care Legnano General Hospital.
Our first aim was to retrospectively evaluate the amount of RBCM drawn for blood testing during the ICU stay as a causative factor of anaemia, and the consequent transfusion approach. Since standard-volume sampling tubes were currently in use at our hospital, the second aim of this study was to estimate the theoretical RBCM that would have been spared with the use of reduced-volume sampling devices and the potential changes in the consequent transfusion policy.
2. Materials and methods
2.1. Patients and data collection
After exclusion of patients hospitalized in ICU for less than twenty days, with a non-invasive respiratory support only, those with overt bleeding and patients who died, twenty-four patient fulfilled the inclusion criteria and represented our study cohort.
The baseline and transfusion data of patients admitted to ICU with SARS-CoV-2 infection from March 8th to May 20th 2020 were retrospectively collected. The analysis was restricted to patients undergoing orotracheal intubation and discharged alive from ICU after at least 20 days.
In ICU routine blood testing were usually drawn with a closed blood conservation device (VAMP adult system, Edwards Lifescience, Irvine, CA, USA), with the aim to reduce the waste of patients' blood. Anthropometric, hematological and transfusion data were recorded, along with the number and type of tubes used for tests throughout the ICU stay. The total volume of blood drawn with standard Vacutainer™ tubes, Bactec™culture vials (Becton Dickinson, Milan, Italy), SafePICO™ arterial blood gas syringes (Radiometer, Brønshøj, Denmark) and the corresponding RBCM removed during hospitalization for each patient were calculated, based on the daily record of ordered testing.
The resulting parameters were then recalculated in a hypothetical scenario in which commercially available low-volume sampling tubes intended for use in adults were employed (S-Monovette®, Sarstedt AG & Co., Nümbrecht, Germany). Both the actual and the simulated RBCM losses were evaluated as potential triggers for transfusion, taking into account that one unit of Packed Red Blood Cells (PRBC) for transfusion contains an average of 170 g of RBCM.
The Nadler’s formula was used to calculate the patients' total blood volume. Baseline patient RBCM was calculated multiplying the total blood volume by the baseline hematocrit. The drawn RBCM was calculated by multiplying the tube numbers and volumes by the corresponding daily hematocrit. The RBCM at discharge from ICU was calculated for each patient. In case of transfusion, the corresponding administered RBCM was subtracted from the final calculated value.
2.2. Statistical analysis
Normality was checked using Shapiro Wilk test. Data are presented as percentage, mean ± Standard Deviation, where applicable, or as median ± Interquartile Range [IQR]. Pearson’s Chi-Square test with Yates’ correction or Fisher exact test were performed to assess the differences among categorical variables. Mann-Whitney U test was used in case of non-normally distributed continuous variables. Data were analysed using IBM SPSS software (version 20). A value of p < 0.05 was considered as statistically significant.
3. Results
3.1. Measured parameters
Data from the 24 eligible patients were summarized in Table 1 . Median length of stay was 29 days (min 20 – max 43), male/female ratio was 1.6 (15/9) and median age 65. Nineteen patients (79 %) were overweight, with BMI > 25 Kg/m2 and 41.7 % (10/24) were anaemic at ICU admission, as defined by World Health Organization criteria in adults and non-pregnant women [6] (i.e. Haemoglobin < 13.0 g/dL for men and < 12.0 g/dL for women). One patient had a previous episode of myocardial infarction.
Table 1.
Patients baseline and transfusion data.
| Gender (M/F) | 19/5 |
| Age, years - mean (min – max) | 65 (44–77) |
| Weight, Kg - mean (± 1 SD) | 89 (17) |
| Height, cm - mean (± 1 SD) | 172 (1.0) |
| BMI, Kg/m2 - mean (± 1 SD) | 30.5 (5.8) |
| Baseline hemoglobin, g/dL - mean (± 1 SD) | 13.0 (1.4) |
| Transfuseda | 13.2 (1.4) |
| Non-transfuseda | 12.8 (1.5) |
| Baseline RBCM, L - mean (± 1 SD) | 1.984 (0.402) |
| Anemic pts at ICU admission, n. (%) | 10/24 (41.7) |
| Days in ICU, median (min – max) | 29 (20–43) |
| Total iatrogenic blood loss, mL – median (min – max) | 719 (424–1342) |
| Iatrogenic blood loss per day, mL – median [IQR] | 21.7 [18.7–26.7] |
| Transfusedb | 23.4 [19.7–28.4] |
| Non-transfusedb | 20.7 [17.0–25.4] |
| Patients transfusedc | 11/24 (45.8) |
| Hemoglobin at transfusion, g/dL - mean (± 1 SD) | 7.7 (0.5) |
| PRBC units transfused, median [IQR] | 2 [1–4] |
IQR: Interquartile range ICU: Intensive Care Unit RBCM: Red Blood Cell Mass PRBC: Packed Red Blood Cells.
p = 0.160.
p = 0.007.
Number of patients/all patients (percentage).
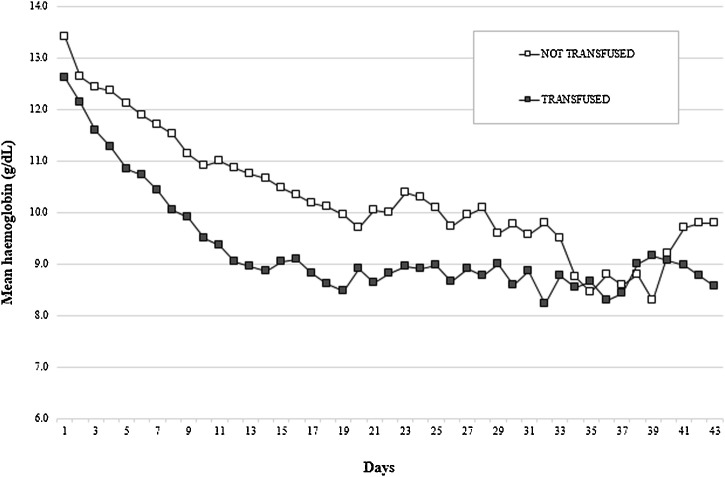
The mean Hb level decreased progressively throughout the ICU stay (Fig. 1 ). Among non-anaemic patients, the development of anaemia started at a median of three days (min 2 – max 9).
Fig. 1.
Median daily haemoglobin in non-transfused and transfused patients.
Overall, 6658 sampling tubes were employed, for a total 16,786 mL of blood drawn. The distribution of tubes used for blood testing is reported in Table 2 . Median blood drawn volume per patient per day was 21.7 mL, with a total median 719 mL loss throughout the ICU stay.
Table 2.
Number of tubes and drawn volumes (Real Standard Tubes vs Simulated Low-Volume Tubes).
|
Tubes |
Standard Tubes |
Low Volume Tubes |
|||
|---|---|---|---|---|---|
| n. (%) | (mL/tube) | total mL (%) | (mL/tube) | total mL (%) | |
| Full Blood Count | 711 (10.7) | 3.0 |
2133 (12.7) | 1.2 | 853.2 (7.6) |
| Coagulation | 908 (13.6) | 2.7 |
2451.6 (14.6) | 1.4 | 1271.2 (11.4) |
| Clinical chemistry | 1294 (19.4) | 5.0 |
6470 (38.5) | 2.7 | 3493.8 (31.2) |
| Blood gases | 3431 (51.5) | 1.0 | 3431 (20.4) | 1.0 | 3431 (30.6) |
| Othersa | 314 (4.8) | -- | 2300.4 (13.8) | -- | 2153.8 (19.2) |
| Total | 6658 (100) | -- | 16786 (100) | -- | 11203 (100) |
i.e. Blood cultures, infectious tests and other clinical chemistry tests performed with additional tubes.
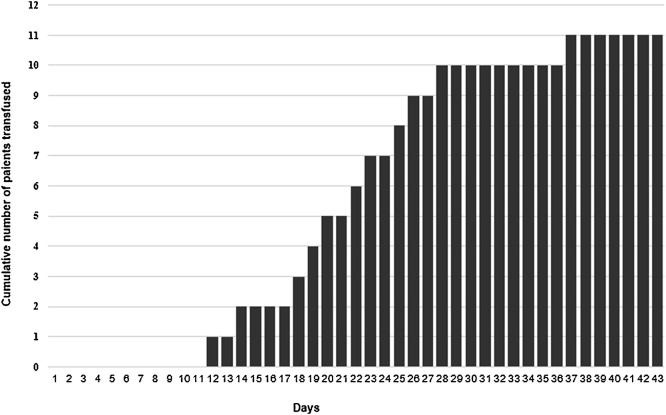
Eleven out of 24 patients (45.8 %) were transfused, starting from the 12th day in ICU (Fig. 2 ), with a mean of 7.7 (± SD 0.5) g/dL haemoglobn at transfusion and a median number of 2 RBC [IQR 1–4] units transfused. The differences in the mean haemoglobn value at day zero between subsequently transfused and non-transfused patients did not reach the statistical significance (p = 0.160).
Fig. 2.
Cumulative number of patients transfused by day of ICU admission.
Pre and post-transfusion PaO2/FiO2 ratio, Blood Lactate (LCT) and Base excess (BE) were 210 (± SD 59) vs 217 mmHg (± SD 59), 0.9 (± SD 0.3) vs 1.0 (± SD 0.3) mmol/L and 8.7 (± SD 5.4) vs 7.8 (± SD 5.7) mmol/L, respectively (p = n.s. for all three comparisons).
Median daily blood sampling volume was significantly higher in patients subsequently receiving transfusions: 23.4 mL [19.7–28.4] vs 20.7 [17.0–25.4] mL in non-transfused patients (p = 0.007). Overall, the median RBCM subtracted by blood samplings accounted for about one third of the total patients’ RBCM decrease until discharge: -31.1 % [21.7–49.5], with a nadir of -75.5 % in one case.
3.2. Recalculated simulated parameters
A simulation was run with the hypothetical usage of commercially available low-volume sampling tubes, as detailed in Table 2. Blood gas analysis requires an irreducible sampling of 1 mL, as well as blood cultures with Bactec™ vials cannot be performed in lower-volume containers.
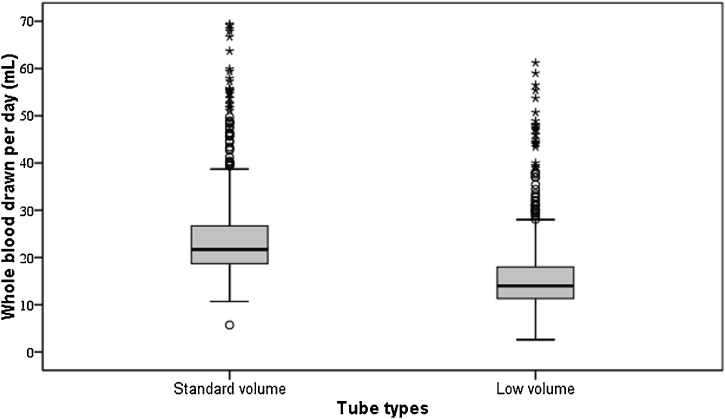
Overall, median differences between standard and low-volume tubes in terms of mL/day were 21.7 [18.7–26.7] versus 14.0 [11.3–18], respectively, p < 0.001, corresponding to a median decrease in blood drawn per day of 35.5 % (Fig. 3 ). The differences in RBCM decrease per patient using the low-volume tubes ranged from 28.5%–45.9% with a median value of 33.3 %.
Fig. 3.
Whole blood drawn by patient and by day in real cases and simulating the usage of reduced-volume sampling tubes.
Note to Fig. 3: Within each box, horizontal black lines denote median values; the boxes represent the 25th and 75th percentiles and the whiskers are the upper and lower values. Open circles denote cases between 75th and 95th percentiles. Asterisks denote outlier values.
Focusing on tubes for blood cell count, coagulation and clinical chemistry, that have their respective low-volume counterparts, the decrease in drawn blood volume would have been 49.2 % (overall 5,618.2 mL versus 11,054 mL).
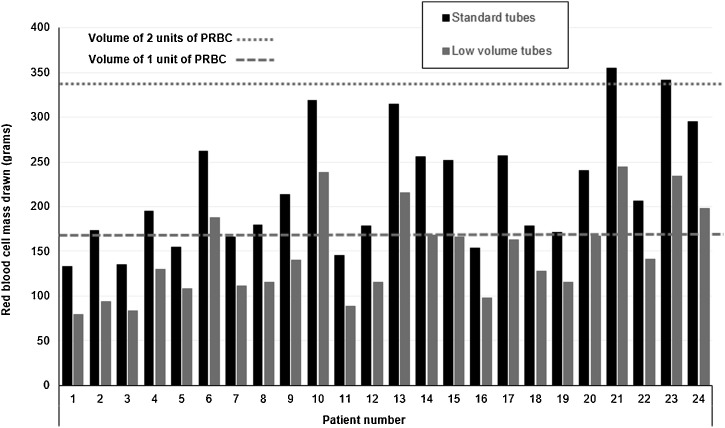
The comparison between the actual and the simulated RBCM drawn for each patient is shown in Fig. 4 . Using standard tubes, in 19/24 patients (79.2 %) the total blood loss by sampling over a mean of 27 days (min 21 – max 42) approached the RBCM contained in 1 unit of PRBC, whereas in 5 patients (20.8 %) the lost volume was near to two PRBC units over a mean of 42 days (min 39 – max 43). With the hypothetical usage of low-volume tubes, in 14 out of 24 patients (58.3 %) the blood loss would have been below the corresponding RBCM of 1 unit of packed red cells, instead of 6/24 (25.0 %, p = 0.040) with standard tubes.
Fig. 4.
Red blood cell volume drawn by blood sampling (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article).
Note to Fig. 4: dashed line and dotted line denote the corresponding red blood cell volume of one and two standard units of packed red blood cell, respectively.
4. Discussion
4.1. General considerations
In this study we analysed data of 24 patients admitted to ICU and undergoing orotracheal intubation due to Acute Respiratory Distress Syndrome (ARDS) induced by SARS-CoV-2 infection. The reasons for restricting our evaluation to surviving and non-acutely bleeding patients were the gathering of a homogeneous cohort of subjects with an ICU stay of at least twenty days, excluding the confounding effect of ongoing hemorrhages on RBCM loss calculations.
The COVID-19 disease outbreak had a tremendous impact on the Italian health system in general, especially putting in strain the ICUs’ capacity of many hospitals. Moreover, in the first week of March 2020 the epidemic caused the crisis of donated blood inventory, although mitigated by a reduction in blood product consumption (e.g. breaking non urgent admissions and postponing programmed surgery) [7].
Hospital-acquired anaemia is a common condition in critical patients admitted to ICU and is associated with worse clinical outcomes [8].
Anaemia in SARS-CoV-2 infection seems to be correlated with illness severity and [9] is the results of a multiple pathogenetic mechanisms [10]. The cytokine storm, induced by the virus in the more severe cases, leads to an impaired iron utilization through the interference on the hepcidin/ferroportin axis, that ultimately causes the sequestration of iron into storage cells [11]. In our cohort, 79 % of patients were overweight (i.e. BMI > 25 Kg/sqm) and obesity is also a recognized cause of impaired iron absorption [12]. Moreover, data are accumulating about the development of hemolytic autoimmune anaemia with a positive direct anti-globulin test in some infected patients [[13], [14], [15]]. Inflammation can also lead to a reduced RBC lifespan, through a membrane alteration [16,17]. Finally, a direct infection of precursor cells by the virus itself could be a further mechanism inducing anaemia [18].
4.2. Iatrogenic anaemia evaluation
Iatrogenic anaemia is a well-recognized cause of anaemia in patients in ICU [1,19] and the minimization of clinically unnecessary blood sampling is a recommended practice through all the three pillars of the patient blood management approach [[20], [21], [22]]. However, in the context of acute COVID-19 disease the frequency and the amount of blood samplings have been overlooked as an important factor causing the continuous drop in patients' haemoglobin and the need of blood transfusion throughout the ICU stay.
In our studied cohort a total of more than sixteen liters of blood was subtracted from 24 patients over about 20 days and the median value of RBCM drawn by blood sampling accounted for about a third of patients’ total RBCM decrease until discharge, with a nadir of -75 % in one patient. In five cases the amount of RBCM subtracted by phlebotomy approached or exceeded the corresponding volume of two RBC units.
In a recent study performed in a mixed ICU population of 7273 patients [23], the authors found a median daily blood loss due to laboratory testing of 25 mL [IQR 14–43]. Interestingly, in the same cohort the cumulative blood loss from day 2–7 was independently associated with the risk of transfusion.
The daily median volume of blood drawn in our study population approached that of the above-mentioned study, i.e. 21.7 mL [IQR 18.7–26.7], with a difference between transfused and non-transfused patients (2.7 mL) that reached the statistical significance. Although this difference may appear small, nevertheless similar values were associated with a doubling of the odds of transfusion in a previous study [24].
Various approaches have been recommended and put into practice with the aim to prevent and minimize iatrogenic anaemia in ICU [21,[25], [26], [27], [28]]. In accord with our simulation, the use of low-volume tubes would have reduced the RBCM drawn of about one third. However, it is not possible to predict accurately the efficacy of this intervention on transfusion rate, and studies evaluating the impact of reducing blood samplings on transfusion requirements have given conflicting results [26,29]. Nevertheless, in a recent systematic review based on twenty-one studies performed in mixed settings, the Authors concluded that the routine use of low-volume tubes as blood-sparing devices can be recommended [30].
The mean amount of blood needed for tests, blood culture excluded, by modern analyzers range between 100–200 μL, about twenty-fold more than blood collected by standard tubes (i.e. 2–5 mL). This leads to a more than 90 % of blood discarded [31]. In the face of this evidence, the extended use of low-volume tubes should be advisable.
4.3. Transfusion thresholds
Despite we have selected a group of non-bleeding patients, the overall transfusion rate was rather high (48.5 %). In a study by Chant et al., the authors reported a transfusion rate of 62 % in a mixed ICU population. However, the reason for transfusion was active bleeding in 17 % of cases, whereas in 26 % of cases the reason of the transfusion was not identifiable [24].
In absence of specific haemoglobin thresholds tailored for patients with COVID-19 disease, transfusion therapy with RBC can be safely performed according to current guidelines recommending a restrictive approach in critically ill patients [32].
However, as recently reaffirmed [33,34], transfusion is a complex decision process. A transfusion policy guided only by strict transfusion thresholds could be harmful for the frail ICU population affected by a possible impairment of peripheral perfusion and limited oxygen delivery and/or extraction. Nevertheless, data based on large observational studies can be unaffected by the biases of randomized clinical trials, thus possibly giving more detailed answers on patients’ outcomes depending on transfusion policy. Moreover, despite current guidelines, as shown by a recent worldwide audit, the transfusion thresholds applied in “real-life” seems slightly higher than recommended (Hb 8.3 ± 1.7 g/dL) [35].
In our cohort, the mean haemoglobn at transfusion was 7.7 g/dL (SD ± 0.5). Nine out eleven patients transfused (81.8 %) were older than 60 years, and possibly with age-dependent comorbidities (i.e. cardiorespiratory insufficiency), conditioning less restrictive transfusion thresholds. [36]. In absence of sound evidence, more recent guidelines in critically ill patients [37] give no or weak transfusion recommendations in a number of acute clinical conditions that are represented in our series. Moreover, we can justify the slightly liberal approach that was applied as a consequence of facing a new, clinically unknown and lethal disease.
Actually, a just published retrospective study [38] comparing patients with ARDS transfused at low (< 8.0 g/dL) versus high haemoglobin threshold (< 10.0 g/dL), although did not found differences in 28-days mortality and within 28-days ECMO-free, sedation-free and organ dysfunction-free composites, demonstrated a delayed weaning from mechanical ventilation in the low threshold group (p = 0.02).
Our study has some limitations. First of all, the number of cases is low to draw definitive conclusions. The reasons for repetitive withdrawals by ICU’s attending physician was not strictly protocol-based, leading to a possible over-sampling respect to a real clinical usefulness. Furthermore, we could not verify that the blood conservation device was always used, with a possible underestimation of iatrogenic blood loss in some cases. In this regard, our decision to focus on RBCM was aimed at obtaining an haemodilution-independent parameter, and this could be a point of strength of the present study.
Lastly, any theoretical simulation based on calculations is intrinsically limited.
5. Conclusions
The amount and frequency of blood sampling considerably account for the development of anaemia in COVID-19 patients in ICU and the use of low-volume sampling tubes could minimize the progressive fall of haemoglobin and possibly the risk of transfusion.
Larger high-powered studies are warranted to assess the more appropriate transfusion thresholds in non-bleeding critically ill patients with COVID-19 disease.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authorship contribution
IB: study design, data collection, analysis and interpretation; manuscript writing; CN data collection, manuscript comment and review; EB, DR, BB: manuscript comment and review.
All the authors have read and approved the final manuscript.
Declaration of Competing Interest
All the Authors declare no conflicts of interest.
References
- 1.Vincent J.L., Baron J.-F., Reinhart K., Gattinoni L., Thijs L., Webb A., et al. Anemia and blood transfusion in critically ill patients. JAMA. 2002;288:1499–1507. doi: 10.1001/jama.288.12.1499. [DOI] [PubMed] [Google Scholar]
- 2.Ganz T. Anemia of inflammation. N Engl J Med. 2019;381:1148–1157. doi: 10.1056/NEJMra1804281. [DOI] [PubMed] [Google Scholar]
- 3.Hayden S.J., Albert T.J., Watkins T.R., Swenson E.R. Anemia in critical illness: insights into etiology, consequences, and management. Am J Respir Crit Care Med. 2012;185:1049–1057. doi: 10.1164/rccm.201110-1915CI. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Halpern S.D., Becker D., Curtis J.R., Fowler R., Hyzy R., Kaplan L.J., et al. An official American thoracic society/American association of critical-care nurses/American college of chest physicians/society of critical care medicine policy statement: the Choosing Wisely® top 5 list in critical care medicine. Am J Respir Crit Care Med. 2014;190:818–826. doi: 10.1164/rccm.201407-1317ST. [DOI] [PubMed] [Google Scholar]
- 5.Callum J.L., Waters J.H., Shaz B.H., Sloan S.R., Murphy M.F. The AABB recommendations for the Choosing Wisely campaign of the american board of internal medicine: AABB Choosing Wisely campaign. Transfusion. 2014;54:2344–2352. doi: 10.1111/trf.12802. [DOI] [PubMed] [Google Scholar]
- 6.World Health Organization . WHO; 2011. Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity.http://www.who.int/vmnis/indicators/haemoglobin/en/ (accessed March 12, 2018) [Google Scholar]
- 7.Mascaretti L.G., De Angelis V., Berti P. The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic and transfusion medicine: reflections from Italy. Blood Transfus. 2020;18:77–78. doi: 10.2450/2020.0071-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Corwin H.L., Gettinger A., Pearl R.G., Fink M.P., Levy M.M., Abraham E., et al. The CRIT Study: anemia and blood transfusion in the critically ill--current clinical practice in the United States. Crit Care Med. 2004;32:39–52. doi: 10.1097/01.CCM.0000104112.34142.79. [DOI] [PubMed] [Google Scholar]
- 9.Tao Z., Xu J., Chen W., Yang Z., Xu X., Liu L., et al. Anaemia is associated with severe illness in COVID‐19: a retrospective cohort study. J Med Virol. 2020 doi: 10.1002/jmv.26444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bellmann-Weiler R., Lanser L., Barket R., Rangger L., Schapfl A., Schaber M., et al. Prevalence and predictive value of anemia and dysregulated Iron homeostasis in patients with COVID-19 infection. J Clin Med. 2020;9:2429. doi: 10.3390/jcm9082429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Taneri P.E., Gomez-Ochoa S.A., Llanaj E., Raguindin P.F., Rojas L.Z., Wyssmann B.M., et al. Anemia and iron metabolism in COVID-19: a systematic review and meta-analysis. Eur J Epidemiol. 2020;35:763–773. doi: 10.1101/2020.06.04.20122267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stoffel N., Mallah C.E., Obeid O., Zeder C., Herter-Aeberli I., Zimmermann M. In women, central obesity predicts higher inflammation, higher serum hepcidin, lower absorption and hypoferremia. Proc Nutr Soc. 2020;79(OCE2):E131. doi: 10.1017/S0029665120000798. [DOI] [Google Scholar]
- 13.Lazarian G., Quinquenel A., Bellal M., Siavellis J., Jacquy C., Re D., et al. Autoimmune haemolytic anaemia associated with COVID‐19 infection. Br J Haematol. 2020;190:29–31. doi: 10.1111/bjh.16794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Berzuini A., Bianco C., Paccapelo C., Bertolini F., Gregato G., Cattaneo A., et al. Red cell bound antibodies and transfusion requirements in hospitalized patients with COVID-19. Blood. 2020;136:766–768. doi: 10.1182/blood.2020006695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wahlster L., Weichert‐Leahey N., Trissal M., Grace R.F., Sankaran V.G. COVID‐19 presenting with autoimmune hemolytic anemia in the setting of underlying immune dysregulation. Pediatr Blood Cancer. 2020;67 doi: 10.1002/pbc.28382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Piagnerelli M., Boudjeltia K.Z., Brohee D., Piro P., Carlier E., Vincent J.-L., et al. Alterations of red blood cell shape and sialic acid membrane content in septic patients. Crit Care Med. 2003;31:2156–2162. doi: 10.1097/01.CCM.0000079608.00875.14. [DOI] [PubMed] [Google Scholar]
- 17.Piagnerelli M., Boudjeltia K.Z., Rapotec A., Richard T., Brohée D., Babar S., et al. Neuraminidase alters red blood cells in sepsis. Crit Care Med. 2009;37:1244–1250. doi: 10.1097/CCM.0b013e31819cebbe. [DOI] [PubMed] [Google Scholar]
- 18.Yang M., Li C., Li K., Hon K., Ng M., Chan P., et al. Hematological findings in SARS patients and possible mechanisms (Review) Int J Mol Med. 2004;14:311–315. doi: 10.3892/ijmm.14.2.311. [DOI] [PubMed] [Google Scholar]
- 19.Shander A., Corwin H.L. A narrative review on hospital-acquired anemia: keeping blood where it belongs. Transfus Med Rev. 2020;34:195–199. doi: 10.1016/j.tmrv.2020.03.003. [DOI] [PubMed] [Google Scholar]
- 20.Shander A., Van Aken H., Colomina Mj, Gombotz H., Hofmann A., Krauspe R., et al. Patient blood management in Europe. Br J Anaesth. 2012;109:55–68. doi: 10.1093/bja/aes139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Burns C.D., Brown J.P., Corwin H.L., Gross I., Ozawa S.J., Shander A. Special report from the society for the advancement of blood management: the Choosing Wisely campaign. Anesth Analg. 2019;129:1381–1386. doi: 10.1213/ANE.0000000000004415. [DOI] [PubMed] [Google Scholar]
- 22.Baron Dm, Franchini M., Goobie Sm, Javidroozi M., Klein Aa, Lasocki S., et al. Patient blood management during the COVID–19 pandemic: a narrative review. Anaesthesia. 2020;75:1105–1113. doi: 10.1111/anae.15095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jackson Chornenki N.L., James T.E., Barty R., Liu Y., Rochwerg B., Heddle N.M., et al. Blood loss from laboratory testing, anemia, and red blood cell transfusion in the intensive care unit: a retrospective study. Transfusion. 2020;60:256–261. doi: 10.1111/trf.15649. [DOI] [PubMed] [Google Scholar]
- 24.Chant C., Wilson G., Friedrich J.O. Anemia, transfusion, and phlebotomy practices in critically ill patients with prolonged ICU length of stay: a cohort study. Crit Care. 2006;10:R140. doi: 10.1186/cc5054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lasocki S., Pène F., Ait-Oufella H., Aubron C., Ausset S., Buffet P., et al. Management and prevention of anemia (acute bleeding excluded) in adult critical care patients. Ann Intensive Care. 2020;10:97. doi: 10.1186/s13613-020-00711-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Riessen R., Behmenburg M., Blumenstock G., Guenon D., Enkel S., Schäfer R., et al. A simple “blood-saving bundle” reduces diagnostic blood loss and the transfusion rate in mechanically ventilated patients. PLoS One. 2015;10 doi: 10.1371/journal.pone.0138879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cismondi F., Celi L.A., Fialho A.S., Vieira S.M., Reti S.R., Sousa J.M.C., et al. Reducing unnecessary lab testing in the ICU with artificial intelligence. Int J Media Inf Lit. 2013;82:345–358. doi: 10.1016/j.ijmedinf.2012.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lee J., Maslove D.M. Using information theory to identify redundancy in common laboratory tests in the intensive care unit. BMC Med Inform Decis Mak. 2015:59. doi: 10.1186/s12911-015-0187-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Macisaac C.M., Presneill J.J., Boyce C.A., Byron K.L., Cade J.F. The influence of a blood conserving device on anaemia in intensive care patients. Anaesth Intensive Care. 2003;31:653–657. doi: 10.1177/0310057X0303100607. [DOI] [PubMed] [Google Scholar]
- 30.Whitehead N.S., Williams L.O., Meleth S., Kennedy S.M., Ubaka-Blackmoore N., Geaghan S.M., et al. Interventions to prevent iatrogenic anemia: a laboratory medicine best practices systematic review. Crit Care. 2019:23. doi: 10.1186/s13054-019-2511-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sanchez-Giron F., Alvarez-Mora F. Reduction of blood loss from laboratory testing in hospitalized adult patients using small-volume (pediatric) tubes. Arch Pathol Lab Med. 2008;132:1916–1919. doi: 10.1043/1543-2165-132.12.1916. [DOI] [PubMed] [Google Scholar]
- 32.Carson J.L., Guyatt G., Heddle N.M., Grossman B.J., Cohn C.S., Fung M.K., et al. Clinical practice guidelines from the AABB: red blood cell transfusion thresholds and storage. JAMA. 2016;316:2025–2035. doi: 10.1001/jama.2016.9185. [DOI] [PubMed] [Google Scholar]
- 33.Vincent J.-L. Transfusion thresholds: the dangers of guidelines based on randomized controlled trials. Intensive Care Med. 2020;46:714–716. doi: 10.1007/s00134-019-05889-3. [DOI] [PubMed] [Google Scholar]
- 34.Hare G.M.T., Cazorla-Bak M.P., Ku S.F.M., Chin K., Mistry N., Sklar M.C., et al. When to transfuse your acute care patient? A narrative review of the risk of anemia and red blood cell transfusion based on clinical trial outcomes. Can J Anesth Can Anesth. 2020 doi: 10.1007/s12630-020-01763-9. [DOI] [PubMed] [Google Scholar]
- 35.Vincent J.-L., Wittebole X., Lefrant J.-Y., Jakob S.M., Almekhlafi G.A., Pellis T., et al. Worldwide audit of blood transfusion practice in critically ill patients. Crit Care. 2018;22:102. doi: 10.1186/s13054-018-2018-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Simon Gi, Craswell A., Thom O., Chew Ms, Anstey Cm, Fung Yl. Impacts of aging on anemia tolerance, transfusion thresholds and patient blood management. Transfus Med Rev. 2019;33:154–161. doi: 10.1016/j.tmrv.2019.03.001. [DOI] [PubMed] [Google Scholar]
- 37.Vlaar A.P., Oczkowski S., de Bruin S., Wijnberge M., Antonelli M., Aubron C., et al. Transfusion strategies in non-bleeding critically ill adults: a clinical practice guideline from the European Society of Intensive Care Medicine. Intensive Care Med. 2020;46:673–696. doi: 10.1007/s00134-019-05884-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hunsicker O., Materne L., Bünger V., Krannich A., Balzer F., Spies C., et al. Lower versus higher hemoglobin threshold for transfusion in ARDS patients with and without ECMO. Crit Care. 2020:24. doi: 10.1186/s13054-020-03405-4. [DOI] [PMC free article] [PubMed] [Google Scholar]